Abstract
Saquarema-Jaconé lagoonal system (SJLS) comprises a sequence of five interconnected shallow brackish lagoons with access to the sea by a single permanent tidal channel. It is a eutrophic system, receiving constant input of organic load from its urbanized catchments. The relationship between several environmental variables and the spatial-temporal distribution of the benthic macrofauna was assessed during four seasonal samplings (dry and wet periods of 2007-2009). Sediment replicates were sampled at seven sites for biological identification and analyzes of organic matter, carbonates, phytopigments, grain size and heavy metals. Salinity, dissolved oxygen and redox potential were measured in situ. SJLS was characterized by sandy bottoms with very reducing conditions. Redox potential significantly discriminated between the dry and wet periods and anoxic conditions were observed in the latter. No significant seasonal differences were observed in the macrofauna. A total of 37 taxa were identified, of which Capitella sp, oligochaetes and Laeonereis culveri (Webster, 1880) were the dominant, representing the early stage of community recovery following dystrophic crises. The faunistic pattern seems to be determined by complex combinations of silt+clay with salinity, organic matter and redox potential. On the other hand, the low concentrations of heavy metals found did not seem to influence the structure and distribution of the biota. SJLS is undergoing persistent environmental stress, dominated by first-order opportunistic species linked to organically enriched sediments.
Abiotic variables; disturbance; macrozoobenthos; seasonal and spatial scales; tropical coastal lagoons
ECOLOGY
Macrobenthic community structure in a Brazilian chocked lagoon system under environmental stress
Carla Lima Torres MendesI,II; Abilio Soares-GomesI
IDepartamento de Biologia Marinha, Laboratório de Ecologia de Sedimentos, Universidade Federal Fluminense, 24010-970 Niterói, RJ, Brazil
IICorresponding author, E-mail: cltmendes@gmail.com
ABSTRACT
Saquarema-Jaconé lagoonal system (SJLS) comprises a sequence of five interconnected shallow brackish lagoons with access to the sea by a single permanent tidal channel. It is a eutrophic system, receiving constant input of organic load from its urbanized catchments. The relationship between several environmental variables and the spatial-temporal distribution of the benthic macrofauna was assessed during four seasonal samplings (dry and wet periods of 2007-2009). Sediment replicates were sampled at seven sites for biological identification and analyzes of organic matter, carbonates, phytopigments, grain size and heavy metals. Salinity, dissolved oxygen and redox potential were measured in situ. SJLS was characterized by sandy bottoms with very reducing conditions. Redox potential significantly discriminated between the dry and wet periods and anoxic conditions were observed in the latter. No significant seasonal differences were observed in the macrofauna. A total of 37 taxa were identified, of which Capitella sp, oligochaetes and Laeonereis culveri (Webster, 1880) were the dominant, representing the early stage of community recovery following dystrophic crises. The faunistic pattern seems to be determined by complex combinations of silt+clay with salinity, organic matter and redox potential. On the other hand, the low concentrations of heavy metals found did not seem to influence the structure and distribution of the biota. SJLS is undergoing persistent environmental stress, dominated by first-order opportunistic species linked to organically enriched sediments.
Key words: Abiotic variables; disturbance; macrozoobenthos; seasonal and spatial scales; tropical coastal lagoons.
doi: 10.1590/S1984-46702011000300011
In the context of the European Water Framework Directive (WFD), coastal lagoons can be considered complex transitional water systems in the boundary between continental and marine domains, ranking among the most productive on Earth (Magni et al. 2008, Munari & Mistri 2010, Pérez-Ruzafa et al. 2010). According to Moreno et al. (2010), they occupy ca. 10% of the sea-land interface worldwide and provide feeding, reproduction and nursery areas for marine and terrestrial biota, fisheries, aesthetic values, tourism and other services to the human population, with a potential economical value estimated at over $22,000 ha-1y-1 (Costanza et al. 1997).
Choked coastal lagoons are most common on coasts with high energy and low tidal range (Kjerfve 1994). They are characterized by a single entrance channel, which allows a perennial exchange of water, sediment, dissolved materials and organisms between the lagoon and the open sea. Kjerfve & Magill (1989) suggested that this entrance channel can naturally serve as a dynamic filter that attenuates currents and tidal water-level fluctuations. The tidal gradient within coastal lagoons diminishes rapidly away from the entrance when subjected to microtidal conditions and large river flows. As a consequence, the more remote sections of these systems tend to be unaffected by marine tides and the dynamics are usually governed by the local wind action and freshwater discharge (Bird 1994).
Depending on their geomorphological and hydrological status, these shallow transitional water systems display gradual or sharp variations in physical-chemical parameters on a daily and seasonal basis, which cause changes in the structure and distribution pattern of organisms (Koutsoubas et al. 2000, Gamito 2006). In this sense coastal lagoons can be considered as harsh and naturally stressed habitats. In some cases, the change in the environmental variables in lagoons is severe and leads to dystrophic crisis (e.g. eutrophication, anoxia), generally resulting in the marked decrease in the number of individuals. According to Lardicci et al. (2001), the ecosystem recovers quickly once the crisis is over as species colonizing these environments are highly adapted to extreme conditions, and assemblages are characterized by high resilience. However, assemblages can be driven towards a different structure when environmental changes become permanent (Schirosi et al. 2010), mainly induced by anthropogenic disturbances, such as sewage discharge.
Transitional waters are sensitive aquatic system where benthic components and processes play an important regulatory function for the whole ecosystem (Magni et al. 2008). Because of the sedentary nature of most benthic organisms, these can integrate environmental conditions over time better than pelagic fauna (Gamito 2006). Therefore, surveys of macrozoobenthic communities are useful tools for assessing short and long-term changes in brackish lagoons, representing potential discriminators between natural and man-induced disturbances (Koutsoubas et al. 2000, Blanchet et al. 2005, Schirosi et al. 2010).
The eastern coastline of the state of Rio de Janeiro, southeast Brazil, harbors a series of chocked lagoons. The systems exhibit well-defined interconnected lagoon-cells, including one external cell with access to the sea by either a permanent or an ephemeral tidal channel and the remaining internal cells strongly influenced by fresh water discharge (Knoppers et al. 1991). The Saquarema-Jaconé lagoonal system (SJLS), located from 23°53'S, 42°39'W to 23°56'S, 42°28'W, comprises a sequence of five lagoons: Fora (7,4 km2), Boqueirão (0,6 km2), Jardim (2,0 km2), Urussanga (12,6 km2) and Jaconé (4 km2). The former lagoon (Fora) communicates with the sea by a permanent channel, opened in 2001 in order to increase tidal exchange and flushing, whereas the latter (Urussanga) is most influenced by the drainage basin with numerous rivers discharges allied to restricted circulation. The Urussanga lagoon is connected to the isolated and more pristine Jaconé lagoon by a long and narrow canal (Salgado).
The climate of the region is humid with a mean annual precipitation of 1100 mm (Kjerfve & Knoppers 1999). During the wet season, the precipitation is about twice to three times higher than in the dry season. According to Wasserman (2000), salinity ranges from 40 to 5 in the dry season to 27 to 5 in the wet season, showing a clear decrease gradient from the tidal channel to the innermost compartment of the lagoonal system. All five lagoons are shallow, with mean depth around 1.15 m, and tidal amplitudes ranging from 0.01 to 0.04 m (Wasserman 2000).
The SJLS drainage basin (244.6 km2) receives input of nutrients from three major sources: (1) marine intrusions; (2) riverine discharge and rainfall; and (3) agricultural and domestic effluents (Carmouze et al. 1991). Increasing urban occupation of its watersheds, mainly for fishery and tourism, has contributed with higher loads of organic matter through wastewaters. As a result, the SJLS represents a complex eutrophic system, undergoing frequent anoxic events with consequences to its biota.
The present work aims to assess the relationship between several environmental variables and the spatial-temporal distribution of the benthic macrofauna in SJLS, identifying key factors affecting the structure and composition of the benthic community. Studies on the macrozoobenthos of SJLS are scarce and ecological information needs updating. We tested the hypothesis that the organic matter is the main responsible for the faunistic pattern observed. Consequently, our findings could make a valuable contribution to the future development of an integrated economic, social and environmental management policy for the lagoon consistent with its conservation.
MATERIAL AND METHODS
Seasonal samplings were conducted in August-September 2007 (Dry I), April 2008 (Wet I), September 2008 (Dry II) and March 2009 (Wet II) in seven marginal sites along the lagoonal system, representing each location (Fora: F1 and F2, Boqueirão: B1; Jardim: J1; Urussanga: U1; Salgado: S1; and Jaconé: Jc1) (Fig. 1). Five replicate samples were taken at each site for macrofauna analysis using a 15 cm diameter x 30 cm high PVC core (total area of 0.18 m2). Replicate core size and number have been determined from pilot studies developed a priori in the ecosystem. The core area and number of replicates used also facilitated comparisons with published literature (Wasserman 2000, Pagliosa & Barbosa 2006).
The samples were sieved through a 0.5 mm mesh and preserved in 5% neutralized formalin. Additional sediment (~300 g) was sampled in triplicates per site for organic matter, carbonate contents, chlorophyll-a and phaeopigments (within 1 cm surface layer), grain size distribution and heavy metals. Samples were kept frozen until laboratorial analyzes. Undisturbed sediment redox potential, water salinity and dissolved oxygen were measured in the field by specific probes (Analyser 300M and YSI 33).
In the laboratory, the macrozoobenthos were sorted, identified to the species level, when possible, counted and preserved in 75% ethanol. Sediment samples were oven-dried at 60°C for 48 hours. Total organic matter was calculated after ignition at 495-500°C for two hours using ash-free dry weights. Sub-samples were attacked by 10% HCl for one hour in order to obtain carbonate content. Pigments quantification followed adapted methods of Plante-Cuny (1978) and Dalto & Gremare (2006) and wet-weight concentrations were calculated using Lorenzen (1967) equations. Sediment samples for grain size fractions were oven-dried at 100°C for 24 hours and then processed through mechanical dry sieving and decantation, according to methods described in Suguio (1973). The metals (Al, Cd, Cr, Cu, Fe, Mn, Ni Pb and Zn) were determined after acid digestion with an Ultima 2 sequential inductively coupled plasma optical emission spectrometer (ICP-OES, Jobin-Yvon, France).
The validity of grouping sampling sites into sectors, according to a putative physicochemical gradient, was tested by one-way analysis of similarities (ANOSIM) per period. Thus, the factor consisted of three levels: the outer sector represented by sites F1, F2 and B1; the intermediary sector, with sites J1 and U1; and, the inner sector, including sites S1 and Jc1, with minimum or no marine influence. Abiotic variables per period were analyzed separately by plotting means and standard deviations to observe general trends. The significance of seasonal differences in abiotic data was also tested by ANOSIM, using the Euclidean distance of normalized data. The Kruskal-Wallis ANOVA by rank analysis was performed to test which environmental variables accounted for seasonal differences. Log-transformed data was subjected to ordination by means of Principal Component Analysis (PCA). Pearson correlations were employed to identify significant affinities between heavy metals and several abiotic variables (salinity, redox potential, organic matter and grain size fractions).
Macrobenthic community structure was analyzed in terms of total number of individuals (N), total number of species (s), Pielou's Evenness (J') and Shannon-Wiener diversity (H'). Species dominance and frequency were calculated considering the total abundance per sampling period and sampling sites, respectively. Biological data was analyzed by means of multivariate techniques after forth root transformation. Differences in macrozoobenthic assemblages were investigated using cluster analysis (group average method) and non-metric multidimensional scaling ordinations (nMDS) based on Bray-Curtis similarity index. ANOSIM permutation test was applied to test for differences between seasons. Species contributing mostly to the dissimilarity among sampling seasons were investigated using the SIMPER procedure. Environmental variables best correlated with the multivariate patterns of the macrobenthic community were identified by means of Spearman coefficient (Bio-Env analyses). Statistics employed are contained in the Primer® v. 6 and STATISTICA 7® software packages.
RESULTS
Environmental variables
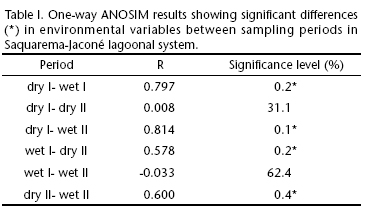
The grouping of sampling sites into an outer, intermediary and inner sector does not seem to be a consistent analytical strategy once ANOSIM results did not reveal any significant differences in environmental variables among the sectors in either the dry period of 2007 (Global R = 0.275, p = 0.13), wet period of 2008 (Global R = 0.375, p = 0.06), dry period of 2008 (Global R = 0.025, p = 0.44) or wet period of 2009 (Global R = 0.35, p = 0.11). Therefore, the results presented herein are not based on a strict division of the lagoonal system into sectors although the clear influence of marine waters on different locations will be pointed out whenever relevant. On the other hand, the analysis of similarity highlighted seasonal differences on the abiotic variables among the group of sites per sampling period (Global R = 0.462, p = 0.001). Table I shows that the dry periods (I and II) were significantly different from both wet periods.
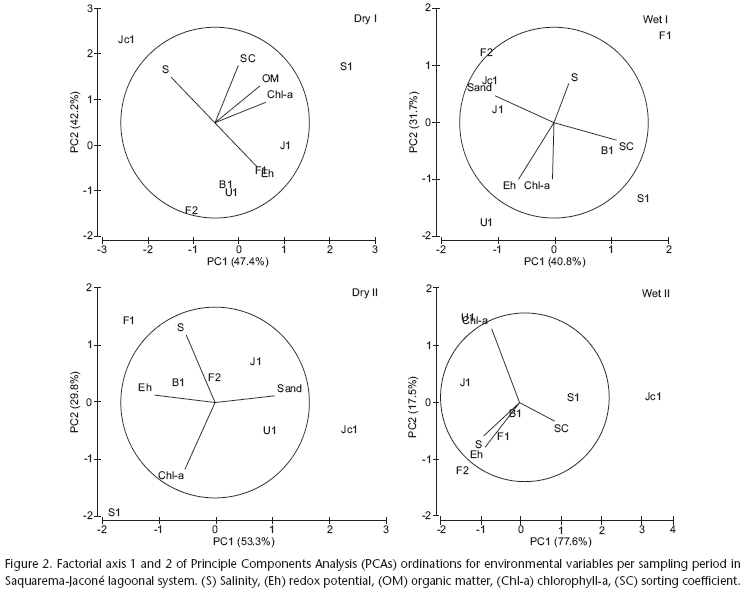
Prior to the exploratory analysis, the twelve environmental variables (salinity, redox potential, dissolved oxygen, organic matter, carbonates, chlorophyll-a, pheopigments, mean grain size, pebble, sand, silt+clay and sorting coefficient) were correlated in order to identify redundant variables in determining the spatial distribution of sampling sites per period. After excluding such variables, clearer and more consistent representations were obtained using Principal Component Analysis (PCA), indicated by the high contributions of factorial axis 1 and 2. The sum of both axes explained more than 83% of the variations in most of the ordinations (period wet I H < 73%). In the period wet II, the ordination accounted for 95% of the variations. The high contributions comprise a total of four or five main components in every ordination, signaling for the importance of salinity, chlorophyll-a and redox potential (Fig. 2). However, the sampling sites of SJLS did not show a clear pattern of spatial distribution. The results point to a stronger temporal influence rather than a spatial heterogeneity in SJLS.
Spatial variation in environmental variables can be compared in figure 3. A decreasing salinity gradient (30 to 10) was observed from sites located near the tidal channel (F1 and F2) to the innermost sites (S1 and Jc1), signaling for the reduction of marine influence. Although mean dissolved oxygen values ranged from 10 to 15 mg.L-1, concentrations showed great spatial variability, reaching hypoxic levels in F1, J1 and Jc1. The lagoon sediments revealed very reducing conditions (mean redox potential around -200 mV) and greater intra-spatial heterogeneity. Salgado canal (S1) presented the highest organic matter (~15%) and carbonate content (~8%), followed by Jardim (J1) with ~7% and ~5%, respectively. Sites in the inlet presented higher carbonate content and lower organic matter concentrations. Phytopigments presented similar mean values, approximately 20 µg.g-1. However, in S1 a higher contribution of phaeopigments (~60 µg.g-1) indicates a high degradation level of the microphytobenthic/phytoplanctonic material and consequently its low quality as food for the macrobenthos. All locations were characterized by sandy bottoms (more than 90%) with low fractions of pebble and silt+clay. In S1, better sorted sediments and greater mean grain size were found.
According to temporal patterns (Tab. II), some abiotic gradients could be described. Higher values in salinity range were observed in dry I, with rather lower and relative constant mean values (16 to 19) in the following periods. Extremely reducing sediments were encountered in the wet periods (minimum values around -300 mV), with wet II presenting higher variability. Dissolved oxygen concentration dropped to hypoxic conditions in dry I (2.7 mg.L-1). In the dry periods, organic matter showed higher mean concentrations (5-7.2%) as a result of approximately 20% maximum input. Mean carbonate contents varied from 5.5% to 3.6% throughout the studied period with higher contributions (~4-11%) in dry I. Chlorophyll-a mean concentrations were about two times greater in the first year of sampling (dry I and wet I) with a maximum value of 104 µg.g-1 in the latter, decreasing substantially in the second year (dry II and wet II). Phaeopigments attained ca. 90 µg.g-1 in the wet periods. However, mean chlorophyll-a/pheopigments ratio suggest larger amount of fresh algal input (2.8) probably through riverine origin in the same periods (wet I and wet II). Granulometric parameters of mean grain size, sand and selection coefficient assumed constant mean values and range throughout the studied period. However, pebble fraction showed higher contributions in dry I and wet II (approximately 21%) whilst silt and clay reached maximum values in dry I (~8%). SJLS can be characterized by sandy bottoms with very reducing conditions and an episodic hypoxic event detected in the period dry I due to enriched mean levels of organic matter and phaeopigments.
SJLS was quite homogeneous along the studied period in terms of the environmental variables considered. A greater stability was observed for the granulometric variables (mean grain size, pebble, sand, silt+clay and sorting coefficient) whereas a marked spatial and temporal variability was noticed for redox potential. Indeed, Kruskal-Wallis analysis of variance attributed the seasonal differences to redox potential values, between dry I and the periods wet I (K = 2.97, p = 0.004) and wet II (K = 2.65, p = 0.04).
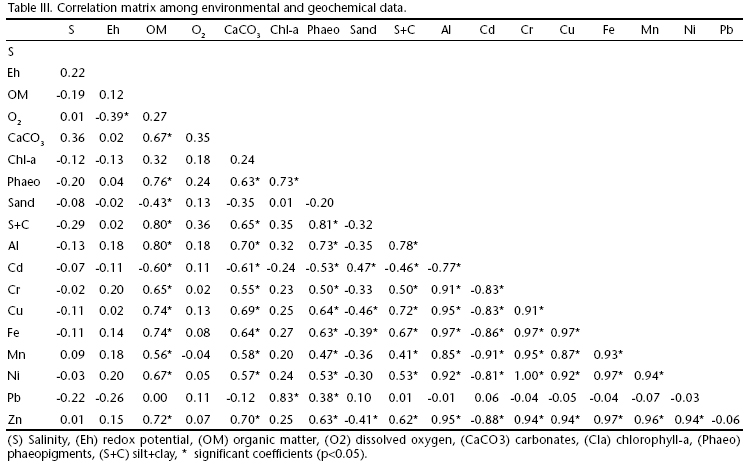
Heavy metals concentrations showed no seasonal differences according to the ANOSIM test (Global R = -0.094, p = 0.98). Except for Cd and Pb, positive significant correlations (Tab. III) between all metals and the organic matter as well as the fine sediment fraction (silt and clay) indicate that metals are trapped physically and/or chemically in detritus particles and minerals. Indeed, phaeopigments and carbonates were also strongly correlated with heavy metals and the abiotic variables mentioned. In contrast, salinity and redox potential showed no relation whatsoever with any of the heavy metals analyzed.
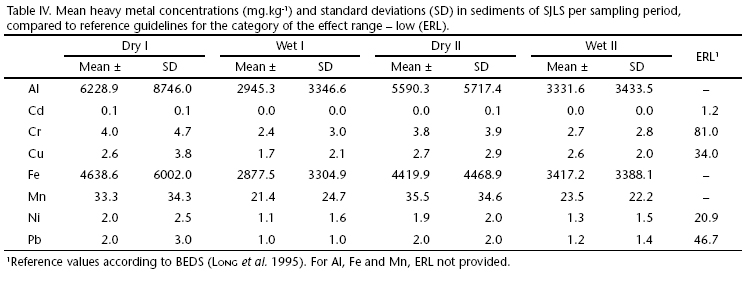
Unfortunately, Brazil still lacks a regional reference guideline for sedimentary metal contents. However, adopting the biological effects database for sediments (BEDS), determined by Long et al. (1995), Cd, Cr, Cu, Ni, Pb and Zn mean concentrations in SJLS fall way below the category of the effect range-low (ERL) (Tab, IV), which means that estimated effects would be rarely observed in the biota, suggesting no environmental contamination of these metals. Therefore, geochemical results show that although the majority of metals are bond to sediments, they offer no risk of contamination in the benthic macrofauna.
Macrobenthic community
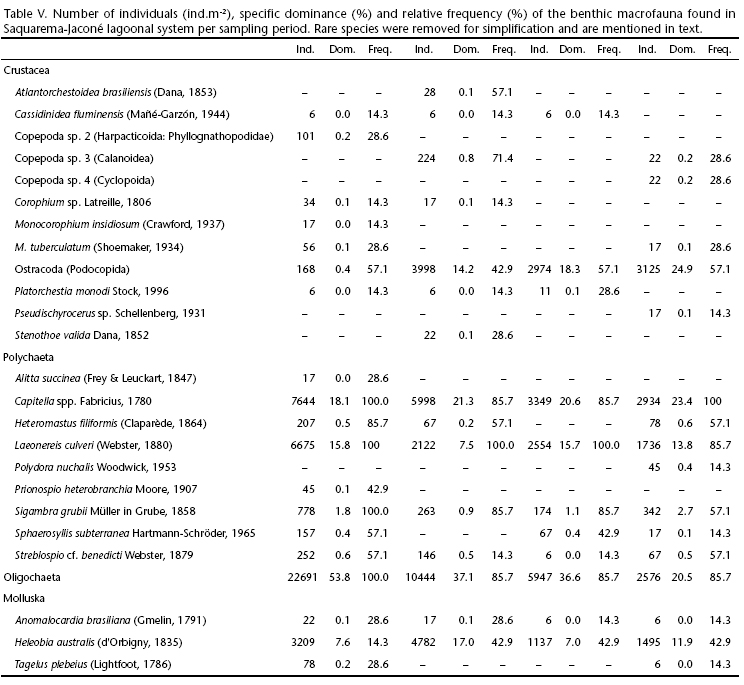
In SJLS the macrofauna totalized 17.705 specimens, of which 43% were collected in the period dry I, 28% in wet I, 16% in dry II and 13% in wet II- showing a gradual decrease in abundance along the studied period. From the 37 identified taxa in this system, 62% were crustaceans, 30% polychaetes and 8% mollusks. Details on the composition and structure of the macrozoobenthic community of SJLS in terms of number of individuals, specific dominance and relative frequency per sampling period are shown in Table V. Rare taxa represented by one or two specimens were excluded from the list. These species were Lumbrineris atlantica Kinberg, 1865, Pedicorophium sp. Karaman, 1981 and a cumacean species from the dry period of 2007; Amphitoe marcuzzii Ruffo, 1954 and Caprella equilibra Say, 1818 from the wet period of 2008; and, Uca rapax (Smith, 1870) and Polydora cornuta Bosc, 1802 from the dry period of 2008; Amphitoe sp. Leach, 1814, a cladoceran and Monocorophium acherusicum (Costa, 1857) from the wet period of 2009 (two individuals each). In addition, Amphitoe ramondi Audouin, 1826 was removed from the wet periods and Copepoda sp. 1 (Harpacticoida: Harpaticidae) from the dry periods, both species with one single specimen in each seasonal replicate; and Kalliapseudes schubartii (Mañé-Garzón, 1969) from the dry and wet seasons of 2008 (Wet I and Dry II).
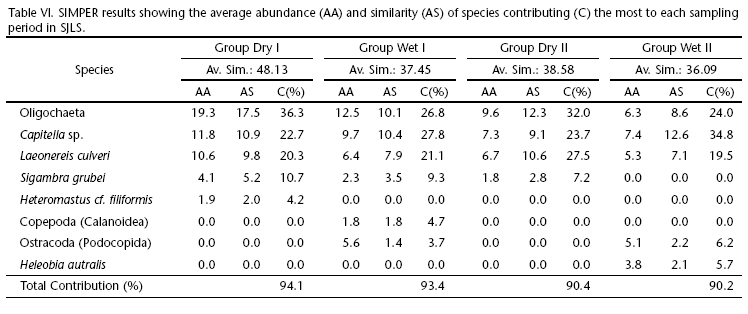
Analysis of similarities performed on Bray-Curtis resemblance matrixes of abundance data (square root transformed) revealed that only four to six taxa contributed to more than 90% to the total average similarities per sampling period (Tab. VI). Among the taxa with the highest contributions, Oligochaeta, Capitella spp. Blainville, 1828, and Laeonereis culveri (Webster, 1880) accounted for more than 20% of average similarities each, alternating in average abundance among the three most important taxa explaining faunistic patterns of temporal distribution. Average similarities were particularly higher in the dry period of 2007 (dry I = 48%) and were slightly lower in the following periods, ranging from 36 to 38%.
Comparisons between groups (sampling periods) showed that the wet periods were slightly more dissimilar than the dry ones, representing around 62% and 56% of average dissimilarities, respectively. Oligochaeta and Capitella spp. were the main responsible taxa accounting for this difference, with a total cumulative contribution of 42% in the dry periods and 37% in the wet periods. These taxa were followed by L. culveri and ostracodes, together contributing with H"20% to the average dissimilarity between sampling groups.
Analysis of similarities (ANOSIM) revealed no seasonal differences among the number of species (S), number of individuals (N), Pielou's Evenness (J') and Shannon-Wiener diversity (H') (Global R = -0.064, p = 0.9), suggesting greater homogeneity in the distribution of biological attributes. In general, the Saquarema-Jaconé lagoonal system showed an increasing trend in the number of individuals from the outer to intermediary sites (up to J1), probably due to the dominance of oligochaetes and Capitella spp. (Fig. 4). The site located in Salgado canal (S1) was the poorest regarding number of taxa and abundance, being evenly represented by oligochaetes and Streblospio cf. benedicti Webster, 1879. In Jaconé (Jc1), the wide range in the number of individuals may be attributed to the shift in oligochaetes, Heleobia australis (d'Orbigny, 1835) and ostracodes dominance. Overall diversity and evenness indices did not result in any clear pattern. Although values seemed variable within sampling sites, the system can be characterized by diversity levels around 0.8-1.20 with medium homogeneity in the distribution of species (J' = 0.5-0.6).
Spatial and seasonal differences in the distribution pattern of the benthic macrofaunal assemblages in SJLS were confirmed by multidimensional scale ordinations (MDS) and cluster analysis (Fig. 5). In every representation, site S1 was distinctly separated from the other sampling sites, with only 10-30% of similarity with all other sites. The other locations accounted for about 40% of faunistic affinities. In the dry period of 2008 (dry II), a greater uniformity was observed in the structure and composition of the macrozoobenthic community. It is interesting to notice the grouping of sites located near the sea connection (F1 and F2) and those from the intermediary sector (J1 and U1), while B1, located in between, presented alternatively biological features from both sectors.
Regarding the environmental variables best correlated with the distribution of the macrofauna, Bio-Env results did not point out any single variable as key structuring factor. Instead, usually the interaction among groups of three to five variables accounted for the strongest correlations. In the period dry I, silt+clay fraction appeared in the five best rank correlations either with salinity at higher rank (rw = 0.70) or in a series of different combinations including redox potential, dissolved oxygen, mean grain size, organic matter and/or carbonates content at rather lower ranks (rw ranging from 0.66- 0.68). Dissolved oxygen, organic matter, mean grain size, silt+clay and chlorophyll-a or pheopigments performed collaboratively a key role in determining the faunistic pattern described in the period wet I, with correlations varying from 0.805 to 0.87, the highest throughout the studied period. In the period dry II, the highest correlations (rwH < 0.69) always comprised salinity and redox potential grouped with organic matter, phytopigments and/or silt+clay fraction. Redox potential also performed a key role in structuring the macrozoobenthic community in the period wet II, together with organic matter and secondarily with coarser grain fractions (pebble or sand) and sorting coefficient, accounting for correlations between 0.73-0.78. Heavy metals were not included in the bioenvironmental analyses because the abiotic variables themselves accounted for correlations above 0.6 and little or no contribution would be expected from them.
DISCUSSION
Based on the environment and biological community patterns obtained by multivariate analyzes, no clear sectors could be distinguished in the SJLS. In fact, a single macrobenthic assemblage was apparent throughout the lagoon system. Little or no significant spatial and temporal differences were observed, with the exception of Salgado canal (S1), always distinctly separated from the other sites, which attained highest homogeneity during the dry period of 2008 (dry II). This structure is consistent with the dynamic character of the habitat, characterized as a typical brackish system (salinity around 20) with low average depth and limited penetration of ocean waters through the tidal channel. As a result, a single macrobenthic community was observed, dominated by a few estuarine species, resistant to both marine and freshwater inputs. These findings are not in agreement with other studies in which two or three zones with distinguishable macrobenthic assemblages could be clearly observed from the mouth of the lagoonal system (e.g. Koutsoubas et al. 2000), following a salinity gradient.
Many environmental variables have been correlated with the temporal and spatial distribution patterns of the macrobenthic community in lagoonal systems, which could either be purely physical and chemical (Blanchet et al. 2005, Magni et al. 2009, Mikac et al. 2007) or associated with food supply (Colling et al. 2007, Magni et al. 2008). In SJLS, the composition and structure of the macrobenthic community seems to be determined by complex combinations of physical and chemical factors, mainly by the fine sediment fraction (silt+clay) with salinity, redox potential, dissolved oxygen, organic matter and phytopigments. SJLS is dominated by sandy bottoms (>90%) with high sedimentary organic content, reducing conditions and dominance of tolerant species, what is typically described in muddy bottoms of low-energy areas, with low permeability to oxygen. According to McLachlan & Brown (2006), these sediments can similarly accumulate toxic byproducts such as sulphides and ammonia due to increased microbial oxygen uptake, resulting in anoxic and reducing conditions which may lead to major impoverishment in species richness and dominance of few opportunistic taxa. Therefore, the great number of variables involved in the explanation of the macrozoobenthic community pattern is reasonable taking into account the complex nature of physicochemical and biological processes. Other investigations have highlighted the interaction of many key structuring factors (e.g. Blanchet et al. 2005, Magni et al. 2009). Regarding salinity, its influence in the composition and number of species is well known as it imposes severe conditions to organisms, rendering difficulties in maintaining osmoregulation (Little 2000). Indeed, Lardicci et al. (2001) pointed out that salinity below 18-20, as found in the present work, is considered to be a threshold affecting the structure of the benthic assemblages in lagoon habitats.
Heavy metals concentration levels were always lower when compared to other studies in the region (Lacerda et al. 1992, Wasserman et al. 1998, Patchineelam 1999) and reference backgrounds from other Brazilian coastal lagoons (Santos et al. 2003, Sabadini-Santos et al. 2009). Low concentrations in SJLS did not signal for anthropogenic pollution, considering its increasing urban occupation.
The bioavailability of sediment-bound trace metals is dependent not only on the total metal concentration but also on the strength of the association between the metal and sediment compounds. Metals adsorbed to biogenic carbonates and organic matter are much more available than metals strongly bound to other sediment compounds, such as iron oxides and sulfides (Lacerda et al. 1992). According to the authors, anoxic sediments coupled with high organic matter content due to extreme eutrophication will favor metal precipitation as sulphides and immobilization with refractory organic compounds, as shown in reduced coastal environments such as salt marshes and mangroves. Although SJLS was characterized with such harsh conditions, correlations between heavy metal concentrations and environmental variables were solely significant for organic matter and silt+clay fraction rather than redox potential. This study lacks further assessment on the specificity of metal binding by different sediment compounds. However, preliminary results suggest that metal concentrations in sediments of SJLS are bound to humic substances and, therefore, fairly available to organisms. What is more, concentration levels in this study met the primary standard criteria for biota contamination (below ERL) established in Long et al. (1995), not representing intoxication risks to the benthic macrofauna of SJLS. The sandy nature of the SJLS, with fewer fine particles capable of binding heavy metals may have resulted in the low concentrations of heavy metals found and thus representing a minor risk factor to benthic impairment. Our data update and complement the existing literature concerning heavy metals in the region. The absence of evidences of metallic contamination in SJLS sediments make it a reference ecosystem for further biogeochemical and ecotoxicological studies in similar environments.
Despite the fact that the macrofauna summed up to 37 species, the benthos of SJLS is characterized by a selection of species within location (around 2 to 14) which are able to cope with such adverse conditions. Species in this study outnumbered in more than two fold the species richness in other similar choked coastal lagoons in Brazil: Imboassica Lagoon (Figueiredo-Barros et al. 2006) and Patos Lagoon (Colling et al. 2007). Noticeable variability in the structure and composition of the community is attributed to the occurrence of 13 rare species (85% crustaceans) and to shifts in abundance of frequent taxa. In particular, the numerically dominant species in this study were mainly the opportunistic polychaetes Capitella spp. and L. culveri, and oligochaetes, accounting for more than 70% of the entire lagoon's abundance per sampling period. Polychaete species, such as capitellids, are the most abundant taxonomic group in lagoons with urbanized catchment (i.e. organically polluted environments) whereas melitid crustaceans and hydrobiid mollusks predominate in lagoons with forested catchments (i.e., pristine environments) (Mikac et al. 2007). This inference was true for the preserved and isolated Jaconé lagoon (Jc1), where the detritivore hydrobiid H. australis was particularly dominant, probably playing an important role in nutrient cycling in the lagoon (Figueiredo-Barros et al. 2006).
Population dynamics of deposit-feeding polychaetes, such as Capitella spp., are well known to respond to changes in sediment organic content. Ramskov & Forbes (2008), using biological laboratory assays, suggested that the minimum high quality organic matter concentration necessary for survival and growth of this species is 0.2% and for reproduction is 0.5%. In SJLS, the average organic matter content ranged from 5.0 to 7.2% in the dry periods and from 2.7 to 3.3% in the wet periods. These far more enriched concentrations can be related to the incorporation of refractory humic material and suffice to warrant the successful thriving of Capitella spp. in this habitat, explaining its high dominance. Oligochaetes and L. culveri, other subsurface deposit-feeders, are also associated with organically-enriched sediments worldwide (Magni et al. 2005, Pagliosa & Barbosa 2006). Observed shifts in the contribution of each taxa per sampling period could be attributed to competition among them for the same food resource (i.e. detritus) or space (Como & Magni 2009).
The dominant taxa reported herein are known to be highly resilient after a disturbance, representing the early stage of community recovery following dystrophic crises (Como & Magni 2009). During the wet seasons (summer) in SJLS, it could be argued that the system was experiencing stressful conditions due to extremely reduced sediment surface (-178 to -238 mV). Nereids, such as L. culveri, are reportedly tolerant to hypoxia and sulphides during summer, reaching deeper depths in the sediments to stay away from adverse environmental conditions (Rossi et al. 2001, Pagliosa & Barbosa 2006, Como & Magni 2009). In SJLS, the bioturbation of these large nereids may contribute to the reoxidation and detoxification of sulphidic sediments, directly or indirectly favoring macrofaunal succession. Similarly, Ramskov & Forbes (2008) highlighted the important role performed by the highly opportunistic Capitella spp. in recolonization and bioremediation of organically polluted sediments, providing habitat amelioration for the establishment of other fauna species. In the first dry period of the present study, it is important to highlight the concomitant presence of another capitellid, Heteromastus filiformis (Claparède, 1864), in much lower abundance when compared to Capitella spp. but disappearing in the following sampling periods. Koutsoubas et al. (2000) also found coexistence of both Capitellidae sustaining the ecological role of Capitella spp. However, according to Pearson & Rosenberg (1978) and confirmed by Shirosi et al. (2010), H. filiformis is a typical species appearing in the second stage of the ecological succession. This finding allows drawing the conclusion that the macrobenthic community of SJLS may have undergone regular dystrophic conditions along the studied period, not even having sufficient time to reach the second stage of recovery before the occurrence of another unfavorable event.
In conclusion, SJLS is characterized by persistent physicochemical stress, dominated by first-order opportunistic species linked to organically enriched and disturbed sediments. The macrobenthic community was reported to respond to habitat perturbations over a spatial and temporal scale in terms of biological indicators, mainly in abundance and number of species. The anthropogenic influence on its watersheds was noticed through the accumulation of organic matter in sediments rather than the concentration of trace heavy metals (e.g. Cu, Ni, Zn). The complex interaction of environmental variables influenced by natural or human-induced processes renders assessment of key community structuring factors difficult. The present study provides updated knowledge regarding the macrobenthic community of SJLS and its determining environmental factors which may contribute to the establishment of conservational measures and monitoring programs in order to cope with population growth in the region.
ACKNOWLEDGMENTS
This study was supported by Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ). The authors are grateful to Ricardo E. Santelli and Eliane P. Oliveira from the Department of Geochemistry (UFF) for conducting heavy metals analyzes. We thank Cinthya Santos and André Senna with taxonomic identification of polychaetes and crustaceans, respectively. Our appreciation also goes to Wilson Machado for providing constructive suggestions.
LITERATURE CITED
Submitted: 18.I.2011;
Accepted: 17.IV.2011.
Editorial responsibility: Rosana M. da Rocha
- Bird, E.C.F. 1994. Physical setting and geomorphology of coastal lagoons, p. 9-39. In: B. Kjerfve (Ed.). Coastal Lagoon Processes. New York, Elsevier, 598p.
- Blanchet, H.; X. de Montaudouin; P. Chardy & G. Blachelet. 2005. Structuring factors and recent changes in subtidal macrozooobenthic communities of a coastal lagoon, Arcachon Bay (France). Estuarine Coastal and Shelf Science 64: 561-576.
- Carmouze, J.P.; B. Knoppers & P. Vasconcelos. 1991. Metabolism of a subtropical Brazilian lagoon. Biogeochemistry 14: 129-148.
- Colling, L.A.; C.E. Bemvenutti & M.S. Gandra. 2007. Seasonal variability on the structure of sublittoral macrozoobenthic association in the Patos Lagoon estuary, southern Brazil. Iheringia, Série Zoologica, 97 (3): 257-262.
- Como, S. & P. Magni. 2009. Temporal changes of a macrobenthic assemblage in harsh lagoon sediments. Estuarine Coastal and Shelf Science 83: 638-646.
- Costanza, R.; R. d'Arge; R. de Groot,; S. Farber; M. Grasso; B. Hannon; K. Limburg; S. Naeem; R.V. O'Neill; J. Paruelo; R.G. Raskin; P. Sutton & M. van der Belt. 1997. The value of the world's ecosystem services and natural capital. Nature 387: 253-260.
- Dalto, A.G. & A. Gremare. 2006. Muddy-bottom meiofauna responses to metal concentrations and organic enrichment in New Caledonia South-West Lagoon. Estuarine Coastal and Shelf Science 67 (4): 629-644.
- Figueiredo-Barros, M.P.; J.J.F. Leal; F.A. Esteves; A.M. Rocha & R.L. Bozelli. 2006. Life cycle, production and nutrient stock in Heleobia autralis d'Orbigny, 1835 (Gastropoda: Hydrobiidae) in a tropical coastal lagoon. Estuarine Coastal Shelf Science 69: 87-95.
- Gamito, S. 2006. Benthic ecology of semi-natural coastal lagoons, in the Ria Formosa (Southern Portugal), exposed to different water renewal regimes. Hydrobiologia 555: 75-87.
- Kjerfe, B. 1994. Coastal Lagoons Processes. Elsevier, Amsterdam, 598p.
- Kjerfve, B. & B.A. Knoppers. 1999. Physical characteristics of lagoons of the East Fluminense Coast, State of Rio de Janeiro, Brazil, p. 57-67. In: B. Knoppers; E.D. Bidone & J.J. Abrão (Eds.). Environmental Geochemistry of Coastal Lagoon Systems. Rio de Janeiro, Brazil, Serie Geoquímica Ambiental, VI+210p.
- Kjerfve, B. & K.E. Magill. 1989. Geographic and hydrodynamic characteristics of shallow coastal lagoons. Marine Geology 88: 187-199.
- Knoppers, B.; B. Kjerfve & J.P. Carmouze. 1991. Trophic state and hydrodynamic turn-over time in six choked lagoons of Brazil. Biogeochemistry 14: 149-166.
- Koutsoubas, D.; C. Dounas; C. Arvanitidis; S. Kornilios; G. Petihakis; G. Triantafyllou & A. Eleftheriou. 2000. Macrobenthic community structure and disturbance assessment in Giolova Lagoon, Ionian Sea. Journal of Marine Science 57: 1472-1480.
- Lacerda, L.D.; M.A. Fernandez; C.F. Calazans & K.F. Tanizaki. 1992. Bioavailability of heavy metals in sediments of two coastal lagoons in Rio de Janeiro, Brazil. Hydrobiologia 228: 65-70.
- Lardicci, C.S.; S.C. Como & F. Rossi. 2001. Recovery of macrozoobenthic community after severe dystrophic crisis in a Mediterranean coastal lagoon (Orbetello, Italy). Marine Pollution Bulletin 42 (3): 202-214.
- Little, C. 2000. The Biology of Soft Shores and Estuaries. New York, Oxford University, 252p.
- Long, E.R.; D.D. MacDonald; S.L. Smith & F.D. Calder. 1995. Incidence of adverse biological effects within ranges of chemical concentrations in marine and estuarine sediments. Environmental Management 19 (1): 81-97.
- Lorenzen, C. 1967. Determination of chlorophyll and pheopigments: Spectrophotometric equations. Limnology and Oceanography 12: 343-346.
- McLachlan, A. & A. Brown 2006. The Ecology of Sandy Shores. Amsterdam, Academic Press, 373p.
- Magni, P.; S. Micheletti; D. Casu; A. Floris; G. Giordani; A.N. Petrov; G. de Falco & A. Castelli. 2005. Relationships between chemical characteristics and macrofaunal communities in the Cabras lagoon (Western Mediterranean, Italy). Hydrobiologia 550: 105-109.
- Magni, P.; G. de Falco; S. Como; D. Casu; A. Floris; A.N. Petrov; A. Castelli & A. Perilli. 2008. Distribution and ecological relevance of fine sediments in organic-enriched lagoons: The case study of the Cabras lagoon (Sardinia, Italy). Marine Pollution Bulletin 56: 549-564.
- Magni, P.; D. Tagliapietra; C. Lardicci; L. Balthis; S. Castelli; S. Como; G. Frangipane; G. Giordani; J. Hyland; F. Maltagliati; G. Pessa; A. Rismondo; J. Tattaranni; P. Tomassetti & P. Varioli. 2009. Animal-sediment relationships: Evaluating the `Pearson-Rosenberg paradigm' in Mediterranean coastal lagoons. Marine Pollution Bulletin 58: 478-486.
- Mikac, K.M.; W.A. Maher & A.R. Jones. 2007. Do physicochemical sediment variables and their soft sediment macrofauna differ among microsize coastal lagoons with forested and urbanized catchments? Estuarine Coastal and Shelf Science 72: 308-318.
- Moreno, I.M.; A. Ávila & M.A. Losada. 2010. Morphodynamics of intermittent coastal lagoons in Southern Spain: Zahara de los Atunes. Geomorphology 121: 305-316.
- Munari, C. & M. Mistri. 2010. Towards the application of the Water Framework Directive in Italy: assessing the potential of benthic tools in Adriatic coastal transitional ecosystems. Marine Pollution Bulletin 60: 1040-1050.
- Pagliosa, P.R. & F.A.R. Barbosa. 2006. Assessing the environment-benthic fauna coupling in protected and urban areas of southern Brazil. Biological Conservation 129: 408-417.
- Patchineelam, S.R. 1999. Human impact detected by geochemical signatures in recent sediments of Jaconé Lagoon, Rio de Janeiro, Brazil, p. 203-210. In: B. Knoppers; E.D. Bidone & J.J. Abrão (Eds). Environmental Geochemistry of Coastal Lagoon Systems. Rio de Janeiro, Serie Geoquimica Ambiental, VI+210p.
- Pearson, T.H. & R. Rosenberg. 1978. Macrobenthic succession in relation to organic enrichment and pollution of the marine environment. Oceanography and Marine Biology Annual Review 16: 229-311.
- Pérez-Ruzafa, A.; M. Concepción; I.M. Pérez-Ruzafa & M. Pérez-Marcos. 2010. Coastal lagoons: "Transitional ecosystems" between transitional and coastal waters. Journal of Coastal Conservation. doi: 10.1007/s11852-010-0095-2.
- Plante-Cuny, M.R. 1978. Pigments photosynthétiques et production primaire des fonds meubles néritiques d'une région tropicale (Nosy-Bé, Madagascar). Travaux et documents de l'ORSTOM 96: 1-359.
- Ramskov, T. & V.E. Forbes. 2008. Life history and population dynamics of the opportunistic polychaete Capitella spp. I in relation to sediment organic matter. Marine Ecology Progress Series 369: 181-192.
- Rossi, F.; S. Como; S. Corti & C. Lardicci. 2001. Seasonal variation of a deposit-feeders assemblage and sedimentary organic matter in a brackish basin mudflat (Western Mediterranean, Italy). Estuarine Coastal and Shelf Science 53: 181-191.
- Sabadini-Santos, E.; B.A. Knoppers; E.P. Oliveira; T. Leipe & R.E. Santelli. 2009. Regional geochemical baselines for sedimentary metals of the tropical São Francisco estuary, NE-Brazil. Marine Pollution Bulletin 58: 601-634.
- Santos, I.R.; P. Baisch & G.T.N.P. Lima. 2003. Metais pesados em sedimentos superficiais da Lagoa Mirim, Fronteira Brasil-Uruguai. Geochimica Brasil 17 (1): 37-47.
- Schirosi, R.; L. Musco & A. Giangrande. 2010. Benthic assemblage of Acquatina Lake South Adriatic Sea: Present state and long-term faunistic changes. Scientia Marina 74 (2): 235-246.
- Suguio, K. 1973. Introdução à Sedimentalogia. São Paulo, Edgard Blücher, 317p.
- Wasserman, J.C. 2000. Estudo do impacto ambiental da Barra Franca na Lagoa de Saquarema-RJ Rio de Janeiro, 317p.
- Wasserman, J.C.; F.B.L. Oliveira & M. Bidarra. 1998. Cu and Fe associated with humic acids in sediments of a tropical coastal lagoon. Organic Geochemistry 28 (12): 813- 822.
Publication Dates
-
Publication in this collection
25 July 2011 -
Date of issue
June 2011
History
-
Received
18 Jan 2011 -
Accepted
17 Apr 2011