Abstract
The appropriate feeding regime for larvae and post-larvae of crustacean decapods is essential for successful larval culture. Reports on the development and morphology of the mouthparts and foregut of these crustaceans have aided in the selection of appropriate larval foodstuffs and consequently increased larval survival and growth rate during development. In the present study, the functional morphology of foregut and mouthparts was investigated in larvae and post-larvae of the freshwater prawn M. amazonicum (Heller, 1862). From observations gathered on both the outer and inner feeding apparati the first stage larvae have obligatory lecithotrophy and feeding behaviour is initiated after molting to the second stage. The foregut of the larvae undergoes diverse morphological changes during larval development and the larval foregut of this species is primarily a mixing organ due to the absence of gastric mills and similar structures. After metamorphosis into post-larvae, drastic morphological changes occur in the foregut and mouthparts to adapt the animals to feed on the greater diversity of foods that are available in their new benthic habitat.
Foregut; larval development; mouthparts; neotropical species
MORPHOLOGY AND PHYSIOLOGY
Ontogenesis and functional morphology of the digestive system of the freshwater prawn, Macrobrachium amazonicum (Decapoda: Palaemonidae)
Luciano D. Queiroz; Fernando A. Abrunhosa; Cristiana R. Maciel
Instituto de Estudos Costeiros, Universidade Federal do Pará. Campus Universitário de Bragança, Alameda Leandro Ribeiro, Aldeia, 68600-000 Bragança, PA, Brazil, E-mail: faraujo@ufpa.br
ABSTRACT
The appropriate feeding regime for larvae and post-larvae of crustacean decapods is essential for successful larval culture. Reports on the development and morphology of the mouthparts and foregut of these crustaceans have aided in the selection of appropriate larval foodstuffs and consequently increased larval survival and growth rate during development. In the present study, the functional morphology of foregut and mouthparts was investigated in larvae and post-larvae of the freshwater prawn M. amazonicum (Heller, 1862). From observations gathered on both the outer and inner feeding apparati the first stage larvae have obligatory lecithotrophy and feeding behaviour is initiated after molting to the second stage. The foregut of the larvae undergoes diverse morphological changes during larval development and the larval foregut of this species is primarily a mixing organ due to the absence of gastric mills and similar structures. After metamorphosis into post-larvae, drastic morphological changes occur in the foregut and mouthparts to adapt the animals to feed on the greater diversity of foods that are available in their new benthic habitat.
Key words: Foregut; larval development; mouthparts; neotropical species.
Detailed studies on the digestive system of the larvae underscore the importance of such information for understanding larval feeding biology and the development of feeding strategies in larval culture and for stock enhancement (FACTOR 1989, NISHIDA et al. 1990, MINAGAWA & TAKASHIMA 1994, ABRUNHOSA & KITTAKA 1997a, ABRUNHOSA et al. 2003). Detailed information on the development of the foregut and the feeding appendages has, for example, contributed to the understanding of suitable food selection for crustacean culture or has even indicated the non- addition of food in cases of larval lecithotrophy.
Most of the literature shows that decapods crustacean larvae possess well-developed mandibles and setose mouthparts. These features strongly suggest a larval feeding behaviour. On the other hand, evidence of non-feeding behaviour has also been reported in transitory stages such as puerulus in spiny lobster, nisto in slipper lobster, glaucothoe in anomurans, and zoeal stages in callianissids (NISHIDA et al. 1990, KITTAKA 1994, LEMMENS & KNOTT 1994, NISHIDA et al. 1995, ABRUNHOSA & KITTAKA, 1997a,b, ABRUNHOSA et al. 2006). Non-feeding behaviour of these decapods is consistent with multiple morphological characteristics of the digestive system including reduced mandibles, reduced mouthpart setae number, and poorly developed foreguts.
However, even in decapods species in which a developed digestive system is observed, in cases in which food is temporarily unavailable, these larvae have been reported as having a facultative lecithotrophic capability, relying on yolk stores or nutritional reserves accumulated during earlier development stages (ANGER 1995, ABRUNHOSA et al. 2003).
This study describes the development of the feeding appendages (mouthparts and foregut) of cultured larvae of the freshwater prawn M. amazonicum that inhabit the Amazonian areas. The functional ability of these structures was observed in the zoeal and post-larval stages and was analysed and compared with other feeding studies that have been previously published.
MATERIAL AND METHODS
Egg-bearing females were captured by the use of traps in a tidal channel of an estuary in the northeastern region of the State of Pará. After hatching, larvae and post-larvae were placed in a rectangular tank of synthetic material (pvc) of black colour with volume of 50-litter capacity located in the laboratory. A dynamic closed system with constant aeration where the culture water circulates through biological filters was used in the larval culture. The temperature ranged 30 ± 0.5ºC, pH 8.0 and salinity 10ppt, and the larvae were fed with Artemia nauplii (density 6 nauplii/mL). The larval stages were identified following GUEST (1979).
Ten individuals of the 1st (ZI), 2nd (Z2), last zoea (ZIX) and post-larva stages were sampled and used for dissection and illustrations. The samples were fixed in formalin solution 10% (v/v) for a 24 hours period before being immersed in an aqueous solution of potassium hydroxide 5% (KOH) (p/v) and heated to 80ºC for 30 minutes for ZI and ZII and 60 minutes for ZIX and post-larvae, respectively. Later, the samples were washed with distilled water and immersed in ethylic alcohol 70% + glycerine solution (1:1).
The larvae of the respective stages were placed on a slide with ethylic alcohol 70% + glycerol solution (1:1). Dissection was undertaken with the use of an optic microscopy (ZEISS) using fine needles (BD Ultra-Fine®, 12.7 x 0.33mm). The foreguts were extracted and transferred to another slide where they were stained with a solution of methylene blue 1%.
RESULTS
MOUTHPARTS
Mandibles
The mandibles are placed in the posterior portion of the mouth. It consists of molar and incisive processes.
Zoea I (Fig. 1): mandible very simple, incisive process with two short teeth; molar process rounded without protuberances bearing short seta; presence of tooth between both processes.
Zoea II (Fig. 2): incisive process with three teeth, one bifurcated; molar process with one strong tooth.
Zoea IX (Fig. 3): incisive process with three long, strong teeth; molar process with one strong tooth and some denticles; three serrated and two smooth teeth between incisive and molar processes.
Post larva (Fig. 4): incisive and molar processes separated by Y-shaped incision; incisive process with three teeth more developed; molar process larger than incisive process, showing prominent teeth with sharpen denticles distally.
Maxillule
Zoea I (Fig. 5): endopod with two short simple setae apically; basal endite with four short setae apically; coxal endite with four short setae apically,1 simple, subdistal seta.
Zoea II (Fig. 6): endopod with two simple apical setae; basal endite with six cuspidate and one simple setae; coxal endite with 5 simple setae, and one simple subdistal seta.
Zoea IX (Fig. 7): endopod with two simple apical setae; basal endite with five cuspidate, four small simple setae distally, and one simple subdistal setae; coxal endite with six plumodenticulate setae and 2+1 simple subdistal setae.
Post larvae (Fig. 8): endopod hook-shaped with one seta on inner proximal margin; basal endite with about 11 cuspidate distal setae, one plumose, and four simple subdistal setae; coxal endite with seven distal and seven subdistal plumodenticulate setae.
Maxilla
Zoea I (Fig. 9): Scaphognathite with 4+1 plumose setae; endopod with one plumose seta distally, 2+3 simple setae sub-distally; basal endite with distal and proximal lobes bearing three simple setae each; coxal endite with proximal and distal lobes indistinctly fused, bearing two simple setae each.
Zoea II (Fig. 10): scaphognathite with 6+1 plumose se-tae; endopod with one plumose seta distally, two long simple setae subdistally; coxal and basal endites larger than in zoea I, simple setae more elongated.
Zoea IX (Fig. 11): scaphognathite with 40-43 plumose setae; endopod similar to that of Zoea II; basal endite with proximal and distal lobes separated, bearing five and four simple setae, respectively; coxal endite with distal and proximal lobes fused, bearing four simple setae each.
Post larva (Fig. 12): scaphognathite with 41-43 plumose setae; endopod unarmed; basal endite with proximal and distal lobes separated, bearing 8 and 6 simple setae, respectively; coxal endite absent.
Maxilliped I
Zoea I (Fig. 13): basipod with three simple setae on inner margin; endopod short, with one subterminal and three simple distal setae.
Zoea II (Fig. 14): basipod with six simple setae on inner margin; endopod short with 2+3 simple setae.
Zoea IX (Fig. 15): basipod with 8-16 simple setae; endopod 2-segmented with four simple setae distally, one plumodenticulate setae subdistally; exopodal lobe present, with 10 plumose setae.
Post larva (Fig. 16): basipod with 31-33 plumose distal setae and six simple marginal setae; endopod with one plumose distal seta; exopodal lobe with 12 plumose and five simple subdistal setae.
Maxilliped II
Zoea I (Fig. 17): basipod with one simple seta on inner margin; endopod 4-segmented, segment 1 and 2 lacking setae, segment 3 with one simple and one denticulate setae, segment 4 with one simple seta and one strong curved denticulate spine on apex.
Zoea II (Fig. 18): basipod with one simple setae on inner margin; endopod 4-segmented, segment 1 and 2 lacking setae, segment 3 with two simple and one strong denticulate setae, segment 4 with two simple and one strong denticulate spine.
Zoea IX (Fig. 19): basipod with 1-2 simple setae; endopod 4-segmentes with 0, 0, 0, 1+2, 2+2 simple setae and a strong denticulate curved spine.
Post larva (Fig. 20): basipod with four simple setae; endopod 5-segmented, enlarged and curved, segments 1 and 2 with few simple setae, segments 4 and 5 showing rows of strong denticulate setae.
Maxilliped III
Zoea I: (Fig. 21) basipod with one simple seta on inner margin; endopod 5-segmented, segments 1, 2 and 3 with 1, 1, 0 simple setae, respectively, segment 4 with a denticulate setae distally, segment 5 with one simple setae and a one strong curved denticulate spine on apex.
Zoea II (Fig. 22): basipod with one simple seta on inner margin; endopod 4-segmented, segments 1 and 2 with 2, 0 setae, segment 3 with one denticulate and one simple seta distally, segment 4 with three simple subdistal setae and one strong denticulate spine apically.
Zoea IX (Fig. 23): basipod with 1-2 simple setae; endopod 4-segmented, segment 1 with three simple setae, segment 2 with two simple subdistal and two denticulate distal setae, segment 3 with three denticulate distal setae, segment 1 with three simple setae and one strong denticulate curved spine.
Post larva (Fig. 24): basipod with two simple setae; endopod 4-segmented with numerous setae on all segments, segments 1, 2 and 3 with simple setae, segment 4 with denticulate setae ending in strong curved denticulate spine.
FOREGUTS
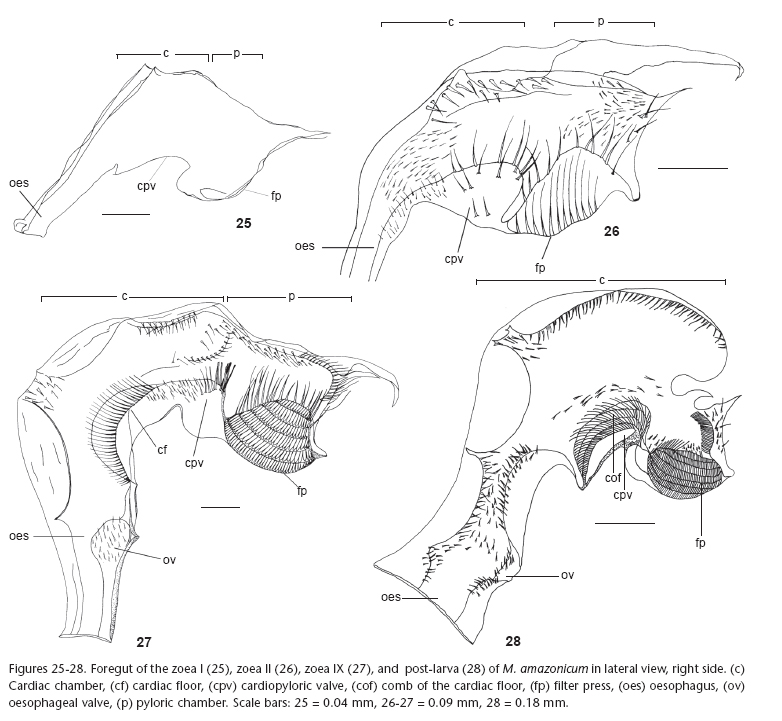
The main morphological changes found in the M. amazonicum foreguts during the larval development include the expansion of the cardiac chamber, mainly in the post-larva, appearance and increasing of the cardiopyloric valve, as well as the increase of the complexity of the filter press. The morphology of the foregut structures at each stage and main changes observed are described as follows.
First zoeal stage (Zoea I)
Foregut undeveloped; cardiac and pyloric chambers lacking setae and other structures (ossicles and teeth); cardiopyloric valve lacking setae; filter press undeveloped, lacking setae (Fig. 25).
Second zoea (Zoea II)
Foregut simple, lacking ossicles and lateral and dorsal teeth; cardiopyloric valve little developed showing few setae; filter press developed (Fig. 26).
Cardiac chamber: narrow, similar in size to pyloric chamber with numerous fine setae on cardiac floor; cardiac wall slightly wrinkled, bearing setules; cardiopyloric valve rounded, enlarged, robust, bearing strong setae on the posterior portion; medium and short setae on anterior, superior, and lateral portions.
Pyloric chamber: enlarged, showing elongate and medium setae on the pyloric roof; filter press enlarged, occupying the entire inferior pyloric chamber.
Last zoeal stage (Zoea IX)
Foregut increasing in complexity compared to those of Zoea I and II; oesophageal valve well-developed; foregut chambers lacking ossicles and lateral and dorsal teeth (Fig. 27.)
Cardiac chamber: about twice the pyloric chamber; cardiac floor with elongate and medium setae laterally distributed; cardiac roof showing medium setae; median depression with numerous setae placed mesoposteriorly; cardiopyloric valve similar in shape to that of Zoea II, showing increasing in the setal number.
Pyloric chamber: bearing numerous setae in the pyloric roof; pyloric wall with setules concentrated in the posterior portion; filter press large and well-developed.
Post larva
Foregut undergoes drastic changes compared to those of previous zoeal stages (Fig. 28).
Cardiac chamber: enlarged, longer than pyloric chamber; presence of frontal plate and floor comb relatively rigid; lacking setae, gastric mills, and lateral teeth.
Pyloric chamber: short; setae filling entire posterior portion; filter press well-developed.
DISCUSSION
Studies have reported that the number of larval stages for the freshwater prawn M. amazonicum may vary between different populations of this species. Thus, the last zoeal stage may arise in the zoea VIII or IX for individuals collected in the northeast of Brazil and in the zoea XII for larvae of females caught in Venezuela and central Amazonia, according to GAMBA (1984) and MAGALHÃES (1985), respectively. In the present study the larvae showed a similar pattern to that described by GUEST (1979), characterizing the population of the coastal zone of the Amazonian region.
Though these works have been concerned with general descriptions of the external morphology of larval stage, studies on the functional importance of the feeding apparatus, specifically the mouthparts of different larval stages are not discussed. However, all the descriptions mentioned have showed the species to have rudimentary structures of the mouthparts in the first zoeal stage. The structure and setation of the various mouthparts play an important role in detection, capture and mastication of the prey before it is guided into the foregut for further processing. From the morphological analyses of the mouthparts of M. amazonicum, it is suggested that zoea I are non- feeding larvae. On the contrary, examination of the mouthparts and foregut of the zoea larvae and postlarvae of M. amazonicum indicates that substantial morphological alterations occur during their development. Such changes are observed in the feeding appendages during the transition of the first zoeal stage to zoea II with abrupt morphological transformations taking place in all of these feeding structures.
The zoea I mandibles are very simple and other mouthpart appendages (first and second maxillae and also, first, second and third maxillipeds) partially developed. The cardiac and pyloric chambers as well as the cardiopyloric valve of the foregut are entirely unarmed of setation and present an undeveloped filter press. Such features strongly indicate that food processing does not occur in this zoea I larval stage and consequently, these larvae show obligatory lecithotrophic behaviour. These results are consistent to those previously reported by ARAUJO & VALENTI (2007), in which these authors suggested lecithotrophic behaviour for M. amazonicum after observations on the feeding behaviour in experimental cultures.
From present observations and from studies of the early life history of the decapods species, it can be deduced that larval development in the palaemonid species may be either completely lecithotrophic - MAGALHÃES (2000) for M. jelskii (Miers, 1877) - or may have non-feeding larval stage(s). The studies on starvation treatments in larvae of some palaemonids have revealed that early larval stages may undergo successive moults in a complete absence of external food (ARAUJO & VALENTI 2007, ANGER 2001) even when the larvae bear well developed feeding appendages. These larvae have a nutritional yolk reserve and therefore, an external food source is not essential.
The non-feeding or facultative lecithotrophic behaviour in zoea I of palaemonids may be related to feeding strategy. Many freshwater prawns have their larval development in brackish water where plankton production is generally higher. Thus, first larvae that hatch in freshwater have sufficient time to be transported by outflowing currents from the parental freshwater habitat towards adjacent estuaries or coastal marine waters (ANGER 2001).
Other palaemonid species of the Amazonian region, such as Pseudopalaemon chryseus Kensey & Walker, 1982 (MAGALHÃES 1986), Palaemonetes ivonicus Holthuis, 1950 (MAGALHÃES 1986), Palaemonetes mercedae Pereira, 1986 (MAGALHÃES 1988), Macrobrachium nattereri (Heller, 1862) (MAGALHÃES 1989) e Pseudopalaemon amazonensis Branch-Port, 1979 (MAGALHÃES & MEDEIROS 1998) have larger eggs in comparison with M. amazonicum. They have also a rich presence of yolk reserve and, also a shortened development, with few larval stages and rudimentary mouthparts. According to ANGER (2001) all of these species follows export strategy and they have at least facultative lecithotrophy in the zoea I stage, whereas, other species, in which, the export strategy are not observed, the larval development is abbreviated, benthic and lecithotrophic.
Contrary to the non-feeding behaviour in one or more of the zoeal stages found in the M. amazonicum, complete lecithotrophic development has been reported. Larvae of Lepidophthalmus siriboia Felder & Rodrigues, 1993 (Callianassidae) were successfully developed from hatching to the metamorphic stage without an external food source. The zoeal feeding apparatus of this species were reported as showing rudimentary and poorly developed setae.
For other decapods, however, evidence of non-feeding is demonstrated by reduced and uncalcified mandibles. However, reduction in number of setae in the mouthpart appendages and poorly developed foregut are observed in the metamorphic stage only (NISHIDA et al. 1990, WOLFE & FELGENHAUER 1991, LEMMENS & NOTT 1994, ABRUNHOSA & KITTAKA 1997a,b).
Significant morphological and anatomical changes in the mouthparts and foregut take place after moulting to zoea II in
M. amazonicum and these changes are observed primarily in the foregut structures. The cardiac and pyloric chambers, as well as, the cardiopyloric valve and the filter press become very setose and functional. Therefore, an analysis of the mouthparts and foregut of the zoea II of M. amazonicum shows that they are suited for feeding both on the phytoplankton as well as other soft food particles. No other major changes in the structure of the external and inner feeding appendages were evident during the development from zoea II to last zoeal stage.
The functional morphology of the foregut has already been reported in previous studies for other palaemonids, including the giant freshwater prawn M. rosenbergii (De Man, 1879) (ABRUNHOSA & MELO 2002). However, contrary to our findings, no significant changes were observed in all zoeal development stages of this species. For M. rosenbergii, the zoea I foregut structures are clearly functional beginning at zoea I, or at least in this stage the feeding appendages are already setose. These facts suggest that further studies on the digestive system of palaemonids are required to clarify the feeding behaviour of freshwater prawns.
Post-larvae are benthic feeding animals with well developed mandibles specialized for the cutting and tearing of large soft food particles. The transformation to post-larvae corresponds with changes in the environment and feeding habits of most decapods. NISHIDA et al. (1990) suggested that in puerulus and post puerulus of the spiny lobster Jasus edwardsii (Hutton, 1875) the feeding appendages of the first moult juvenile are capable of masticating large food particles.
In M. amazonicum the feeding appendages undergo noticeable morphological changes during metamorphosis from zoea IX to post-larvae. The inner structures of the maxillipeds increase in size while the exopods undergo slight reduction in size. In carid decapods, the exopods of the maxillipeds are reported as having a natatory function, while the endopod have developed apprehension ability for the gathering of external food (BAUER 2004, FELGENHAUER 1992). Therefore, the reduction of the exopods may be related to loss or reduction of the correspond ing functions due to the benthonic habit after metamorphosis. The foregut of the newly moulted juvenile of M. amazonicum is very complex, showing gastric plates and lack gastric mills or lateral and dorsal teeth. The foregut is very simi lar to that reported for the first post larvae of M. rosenbergii (ABRUNHOSA & MELO 2002).
ACKNOWLEDGMENTS
The present study was supported by the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and Fundação de Amparo à Pesquisa do Estado do Pará (Fapespa).
LITERATURE CITED
Abrunhosa, F. A. & J. Kittaka. 1997a.The morphological development of juvenile western rock lobster Panulirus cygnus George, 1962 (Decapoda: Palinuridae) reared in the laboratory. Bulletin of Marine Science 61 (1) 81-96.
Abrunhosa, F.A. & J. Kittaka. 1997b. Functional morphology of mouthparts and foregut of the last zoea, glaucothoe and first juvenile of the king crabs Paralithodes camtschaticus, P. brevipes and P. platypus. Fisheries Science 63 (6): 923-930.
Abrunhosa, F.A. & M.A. Melo. 2002. Morfologia comparativa do estômago do primeiro e último estágios zoea e juvenil I de Macrobrachium rosenbergii (Decapoda: Palaemonidae). Revista Ciência Agronômica 33 (2): 65-68.
Abrunhosa, F.A.; M.A. Melo & J.P. Abrunhosa. 2003. Development and functional morphology of the foregut of larvae and post-larva of Ucides cordatus (Decapoda, Ocypodidae). Nauplius 11 (1): 37-43.
Abrunhosa, F.; M. Melo; J. LIMA & J. ABRUNHOSA. 2006. Developmental morphology of mouthparts and foregut of the larvae and postlarvae of Lepidophthalmus siriboia Felder and Rodrigues, 1993 (Decapoda: Callianassidae). Acta Amazonica 36: 335- 342.
Anger, K. 1995. Starvation resistance in larvae of a semi terrestrial crab, Sesarma curacaoense (Decapoda: Grapsidae). Journal of Experimental Marine Biology and Ecology 187: 161-174.
Anger, K. 2001. The biology of decapod crustacean larvae. Lisse, A.A. Balkema, Crustacean Issues 14, 419p. ARAUJO, M.C. & W.C. VALENTI. 2007. Feeding habit of the Amazon river prawn Macrobrachium amazonicum larvae. Aquaculture 265: 187- 193.
Bauer, R.T. 2004. Remarkable shrimps: adaptations and natural history of the carideans. Norman, University of Oklaroma Press, 282p.
Factor, J. R. 1989. Development of the feeding apparatus in decapod crustaceans, p. 185-203. In: B.E. FELGENHAUER; L. WATLING & A.B. THISTLE (Eds). Crustacean issues. Rotterdam, A.A. Balkema, VI+219p.
Felgenhauer, B.E. 1992. External anatomy and integumentary structures, p. 7-43. In: F.W. HARRISON (Ed.). Microscopic Anatomy of Invertebrates. New York, Wiley-Liss, 480p.
Gamba, A.L. 1984. Different egg-associated and larval development characteristics of Macrobrachium jelskii and Macrobrachium amazonicum (Arthropoda: Crustacea) in Venezuelan continental lagoon. International Journal of Invertebrate Reproduction and Development 7: 135-142.
Guest, W.C. 1979. Laboratory life history of the Palaemonid shrimp Macrobrachium amazonicum (Heller, 1862) (Decapoda, Palaemonidae). Crustaceana 37 (2): 141-151.
Kittaka, J. 1994. Larval rearing, p. 402-423. In: B.F. PHILLIPS; J.S. COBB & J. KITTAKA (Eds). Spiny Lobster Management. Oxford, Fishing News Books, XXIII+550p.
Lemmens , J.W.T.J. & B. KNOTT. 1994. Morphological changes in external and internal feeding structures during the transition phyllosoma-puerulus-juvenile in the western rock lobster (Panulirus cygnus: Decapoda:Palinuridae). Journal of Morphology 220: 271-280.
Magalhães, C. 1985. Desenvolvimento larval obtido em laboratório de palemonídeos da região Amazônica: I. Macrobrachium amazonicum (Heller, 1862) (Crustacea, Decapoda). Amazoniana 9 (2): 247-274.
Magalhães, C. 1986. The larval development of palaemonid shrimps from the Amazon Region reared in the laboratory. IV. The abbreviated development of Palaemonetes ivonicus Holthuis, 1950 (Crustacea: Decapoda). Amazoniana 10 (1): 63-78.
Magalhães, C. 1987. The larval development of palaemonid shrimps from the Amazon Region reared in the laboratory. V. The abbreviated development of Pseudopalaemon chryseus Kensley & Walker, 1982 (Crustacea: Decapoda: Palaemonidae). Acta Amazonica 16: 95-108.
Magalhães, C. 1988. The larval development of palaemonid shrimps from the Amazon Region reared in the laboratory. III. The Extremely Abbreviated Development of Palaemonetes (Palaemonetes) mercedae Pereira, 1986 (Crustacea, Decapoda). Studies on Neotropical Fauna and Environment 23 (1): 1-8.
Magalhães, C. 1989. The larval development of palaemonid shrimps from the Amazon Region reared in the laboratory. VI. Abbreviated development of Macrobrachium nattereri (Heller, 1862) (Crustacea: Decapoda). Amazoniana 10 (4): 379-392.
Magalhães, C. 2000. Abbreviated development of Macrobrachuium jelskii (Miers, 1977) (Crustacea: Decapoda: Palaemonidae) from the Rio Solimões floodplain, Brazil, reared in the laboratory. Nauplius 8 (1): 1-14.
Magalhães, C. & N. Medeiros. 1998. The larval development of palaemonid shrimps from the Amazon Region reared in the laboratory. VII. Abbreviated development of Pseudopalaemon amazonensis Ramos-Porto, 1979 (Crustacea: Decapoda: Caridea). Acta Amazonica 28 (4): 433-448.
Minagawa, M. & F. Takashima. 1994. Developmental changes in larval mouthparts and foregut in the red frog crab, Ranina ranina (Decapoda: Raninidae). Aquaculture 126: 61-71.
Nishida, S.; B.D. Quigley; T. Nemoto & J. Kittaka. 1990. Comparative morphology of the mouthparts and foregut of the final-stage phyllosoma, puerulus, and post puerulus of the rock lobster Jasus edwardssii (Decapoda: Palinuridae). Journal of Crustacean Biology 10: 293-305.
Nishida, S.; Y. Takahashi & J. Kittaka. 1995. Structural changes in the hepatopancreas of the rock lobster Jasus edwardsii (Crustacea: Palinuridae) during development from the puerulus to post-puerulus. Marine Biology 123: 837-844.
Wolfe, S.H. & B.E. Felgenhauer. 1991. Mouthpart and foregut ontogeny in larval, post larval, and juvenile spiny lobster, Panulirus argus Latreille (Decapoda: Palinuridae). Zoologica Scripta 20: 57-75.
Submitted: 20.XI.2009;
Accepted: 14.V.2010.
Editorial responsibility: Marcos D.S. Tavares
- Abrunhosa, F. A. & J. Kittaka. 1997a.The morphological development of juvenile western rock lobster Panulirus cygnus George, 1962 (Decapoda: Palinuridae) reared in the laboratory. Bulletin of Marine Science 61 (1) 81-96.
- Abrunhosa, F.A. & J. Kittaka. 1997b. Functional morphology of mouthparts and foregut of the last zoea, glaucothoe and first juvenile of the king crabs Paralithodes camtschaticus, P. brevipes and P. platypus Fisheries Science 63 (6): 923-930.
- Abrunhosa, F.A. & M.A. Melo. 2002. Morfologia comparativa do estômago do primeiro e último estágios zoea e juvenil I de Macrobrachium rosenbergii (Decapoda: Palaemonidae). Revista Ciência Agronômica 33 (2): 65-68.
- Abrunhosa, F.A.; M.A. Melo & J.P. Abrunhosa. 2003. Development and functional morphology of the foregut of larvae and post-larva of Ucides cordatus (Decapoda, Ocypodidae). Nauplius 11 (1): 37-43.
- Abrunhosa, F.; M. Melo; J. LIMA & J. ABRUNHOSA. 2006. Developmental morphology of mouthparts and foregut of the larvae and postlarvae of Lepidophthalmus siriboia Felder and Rodrigues, 1993 (Decapoda: Callianassidae). Acta Amazonica 36: 335- 342.
- Anger, K. 1995. Starvation resistance in larvae of a semi terrestrial crab, Sesarma curacaoense (Decapoda: Grapsidae). Journal of Experimental Marine Biology and Ecology 187: 161-174.
- Anger, K. 2001. The biology of decapod crustacean larvae. Lisse, A.A. Balkema, Crustacean Issues 14, 419p.
- ARAUJO, M.C. & W.C. VALENTI. 2007. Feeding habit of the Amazon river prawn Macrobrachium amazonicum larvae. Aquaculture 265: 187- 193.
- Bauer, R.T. 2004. Remarkable shrimps: adaptations and natural history of the carideans. Norman, University of Oklaroma Press, 282p.
- Factor, J. R. 1989. Development of the feeding apparatus in decapod crustaceans, p. 185-203. In: B.E. FELGENHAUER; L. WATLING & A.B. THISTLE (Eds). Crustacean issues. Rotterdam, A.A. Balkema, VI+219p.
- Felgenhauer, B.E. 1992. External anatomy and integumentary structures, p. 7-43. In: F.W. HARRISON (Ed.). Microscopic Anatomy of Invertebrates. New York, Wiley-Liss, 480p.
- Gamba, A.L. 1984. Different egg-associated and larval development characteristics of Macrobrachium jelskii and Macrobrachium amazonicum (Arthropoda: Crustacea) in Venezuelan continental lagoon. International Journal of Invertebrate Reproduction and Development 7: 135-142.
- Guest, W.C. 1979. Laboratory life history of the Palaemonid shrimp Macrobrachium amazonicum (Heller, 1862) (Decapoda, Palaemonidae). Crustaceana 37 (2): 141-151.
- Kittaka, J. 1994. Larval rearing, p. 402-423. In: B.F. PHILLIPS; J.S. COBB & J. KITTAKA (Eds). Spiny Lobster Management. Oxford, Fishing News Books, XXIII+550p.
- Lemmens , J.W.T.J. & B. KNOTT. 1994. Morphological changes in external and internal feeding structures during the transition phyllosoma-puerulus-juvenile in the western rock lobster (Panulirus cygnus: Decapoda:Palinuridae). Journal of Morphology 220: 271-280.
- Magalhães, C. 1985. Desenvolvimento larval obtido em laboratório de palemonídeos da região Amazônica: I. Macrobrachium amazonicum (Heller, 1862) (Crustacea, Decapoda). Amazoniana 9 (2): 247-274.
- Magalhães, C. 1986. The larval development of palaemonid shrimps from the Amazon Region reared in the laboratory. IV. The abbreviated development of Palaemonetes ivonicus Holthuis, 1950 (Crustacea: Decapoda). Amazoniana 10 (1): 63-78.
- Magalhães, C. 1987. The larval development of palaemonid shrimps from the Amazon Region reared in the laboratory. V. The abbreviated development of Pseudopalaemon chryseus Kensley & Walker, 1982 (Crustacea: Decapoda: Palaemonidae). Acta Amazonica 16: 95-108.
- Magalhães, C. 1988. The larval development of palaemonid shrimps from the Amazon Region reared in the laboratory. III. The Extremely Abbreviated Development of Palaemonetes (Palaemonetes) mercedae Pereira, 1986 (Crustacea, Decapoda). Studies on Neotropical Fauna and Environment 23 (1): 1-8.
- Magalhães, C. 1989. The larval development of palaemonid shrimps from the Amazon Region reared in the laboratory. VI. Abbreviated development of Macrobrachium nattereri (Heller, 1862) (Crustacea: Decapoda). Amazoniana 10 (4): 379-392.
- Magalhães, C. 2000. Abbreviated development of Macrobrachuium jelskii (Miers, 1977) (Crustacea: Decapoda: Palaemonidae) from the Rio Solimões floodplain, Brazil, reared in the laboratory. Nauplius 8 (1): 1-14.
- Magalhães, C. & N. Medeiros. 1998. The larval development of palaemonid shrimps from the Amazon Region reared in the laboratory. VII. Abbreviated development of Pseudopalaemon amazonensis Ramos-Porto, 1979 (Crustacea: Decapoda: Caridea). Acta Amazonica 28 (4): 433-448.
- Minagawa, M. & F. Takashima. 1994. Developmental changes in larval mouthparts and foregut in the red frog crab, Ranina ranina (Decapoda: Raninidae). Aquaculture 126: 61-71.
- Nishida, S.; B.D. Quigley; T. Nemoto & J. Kittaka. 1990. Comparative morphology of the mouthparts and foregut of the final-stage phyllosoma, puerulus, and post puerulus of the rock lobster Jasus edwardssii (Decapoda: Palinuridae). Journal of Crustacean Biology 10: 293-305.
- Nishida, S.; Y. Takahashi & J. Kittaka. 1995. Structural changes in the hepatopancreas of the rock lobster Jasus edwardsii (Crustacea: Palinuridae) during development from the puerulus to post-puerulus. Marine Biology 123: 837-844.
- Wolfe, S.H. & B.E. Felgenhauer. 1991. Mouthpart and foregut ontogeny in larval, post larval, and juvenile spiny lobster, Panulirus argus Latreille (Decapoda: Palinuridae). Zoologica Scripta 20: 57-75.
Publication Dates
-
Publication in this collection
25 July 2011 -
Date of issue
June 2011
History
-
Received
20 Nov 2009 -
Accepted
14 May 2010