Abstract
Species richness and geographical distribution of Cyclopoida freshwater copepods were analyzed along the "La Plata" River basin. Ninety-six samples were taken from 24 sampling sites, twelve sites for zooplankton in open waters and twelve sites for zooplankton within macrophyte stands, including reservoirs and lotic stretches. There were, on average, three species per sample in the plankton compared to five per sample in macrophytes. Six species were exclusive to the plankton, 10 to macrophyte stands, and 17 were common to both. Only one species was found in similar proportions in plankton and macrophytes, while five species were widely found in plankton, and thirteen in macrophytes. The distinction between species from open water zooplankton and macrophytes was supported by nonmetric multidimensional analysis. There was no distinct pattern of endemicity within the basin, and double sampling contributes to this result. This lack of sub-regional faunal differentiation is in accordance with other studies that have shown that cyclopoids generally have wide geographical distribution in the Neotropics and that some species there are cosmopolitan. This contrasts with other freshwater copepods such as Calanoida and some Harpacticoida. We conclude that sampling plankton and macrophytes together provided a more accurate estimate of the richness and geographical distribution of these organisms than sampling in either one of those zones alone.
La Plata River basin; reservoirs; rivers; zooplankton
ECOLOGY
Estimating cyclopoid copepod species richness and geographical distribution (Crustacea) across a large hydrographical basin: comparing between samples from water column (plankton) and macrophyte stands
Gilmar Perbiche-NevesI; Carlos E.F. da RochaI; Marcos G. NogueiraII
IDepartamento de Zoologia, Instituto de Biociências, Universidade de São Paulo. Rua do Matão, travessa 14, 321, 05508-900 São Paulo, SP, Brazil. Email: gilmarpneves@yahoo.com.br
IIDepartamento de Zoologia, Instituto de Biociências, Universidade Estadual Paulista. Distrito de Rubião Júnior, 18618-970 Botucatu, SP, Brazil
ABSTRACT
Species richness and geographical distribution of Cyclopoida freshwater copepods were analyzed along the "La Plata" River basin. Ninety-six samples were taken from 24 sampling sites, twelve sites for zooplankton in open waters and twelve sites for zooplankton within macrophyte stands, including reservoirs and lotic stretches. There were, on average, three species per sample in the plankton compared to five per sample in macrophytes. Six species were exclusive to the plankton, 10 to macrophyte stands, and 17 were common to both. Only one species was found in similar proportions in plankton and macrophytes, while five species were widely found in plankton, and thirteen in macrophytes. The distinction between species from open water zooplankton and macrophytes was supported by nonmetric multidimensional analysis. There was no distinct pattern of endemicity within the basin, and double sampling contributes to this result. This lack of sub-regional faunal differentiation is in accordance with other studies that have shown that cyclopoids generally have wide geographical distribution in the Neotropics and that some species there are cosmopolitan. This contrasts with other freshwater copepods such as Calanoida and some Harpacticoida. We conclude that sampling plankton and macrophytes together provided a more accurate estimate of the richness and geographical distribution of these organisms than sampling in either one of those zones alone.
Key words: La Plata River basin; reservoirs; rivers, zooplankton
Freshwater copepods are a link in the trophic web, connecting producers to consumers (PERBICHE-NEVES et al. 2007); they inhabit lakes, rivers, pools, caves, humid rocks, etc. (BOXSHALL & DEFAYE 2008), where it is easy to find free living copepods of the order Cyclopoida.
In large spatial scales, cyclopoids are less endemic than other copepods, for instance diaptomids. Many cyclopoid species in Mesocyclops, Metacyclops, Eucyclops, etc. are widely distributed in the Neotropical region and in the world (REID 1985, SILVA 2008). However, there are exceptions to this rule, for instance species of Thermocyclops (REID 1989), which occur in the South hemisphere, and differ between the Afrotropical and Neotropical biogeographical regions. In contrast to the patterns of distribution of cyclopoids in large bio geographical areas, there are no clear patterns in the spatial distribution of these organisms among river basins in South America, as observed for diaptomids (SUÁREZ-MORALES et al. 2005). The low endemism of cyclopoids can be in part explained by their efficient dispersion (e.g., by birds, fishes, humans) and their recent colonization of many parts of the world (BOXSHALL & JAUME 2000, SUÁREZ-MORÁLES et al. 2004). Comparing among the main rivers of the La Plata Basin, the composition of cyclopoid species is similar (PAGGI & JOSÉ DE PAGGI 1990, LANSAC-TÔHA et al. 2002).
Cyclopoid copepods of inland waters are more diverse than in the littoral, which can be colonized by aquatic macrophytes. For example, two studies sampling the two types of habitats, open water and macrophyte stands have documented this trend for lakes in Brazil (LANSAC-TÔHA et al. 2002, MAIABARBOSA et al. 2008). The habitat complexity provided by aquatic macrophytes (GENKAI-KATO 2007, LUCENA-MOYA & DUGGAN 2011) also allow several species to be more abundant in them (GERALDES & BOAVIDA 2004).
Most of zooplankton horizontal migration between limnetic zones and macrophyte stands in lentic environments can be attributed to predation pressure by planktivorous fish (GENKAI-KATO 2007, FANTIN-CRUZ et al. 2008). Lower richness of cyclopoid species tends to be found in limnetic waters, where generally few abundant species dominate. Clear tendencies in some ecological attributes can be observed for copepods and other crustaceans in reservoirs (SILVA & MATSUMURA-TUNDISI 2002, NOGUEIRA et al. 2008).
There are no comparative studies of copepod richness between habitats in large geographical scales, only in lotic stretches or in lakes (LANSAC-TÔHA et al. 2002, MAIA-BARBOSA et al. 2008). This study is the first to compare copepod species richness in a large hydrographic basin, the fourth largest in the world. We simultaneously sampled plankton and macrophyte stands in rivers and reservoirs. Based on references, we tested the alternative hypotheses that in limnetic zones there is greater richness of species in macrophytes than in zooplankton. Additionally, we tried to pinpoint particular species in each kind of habitat, and to ascertain how sampling effort can determine the species that are found.
MATERIAL AND METHODS
The "La Plata" River basin crosses Argentina, Brazil, Bolivia, Paraguay and Uruguay. Samples were taken in the summer (from January to March 2010) and in the winter (from June to July 2010), periods that have different mean temperatures and precipitation. It was established that a minimum of two sampling trips were necessary to estimate richness. Altogether 24 sites were sampled, which included 12 reservoirs in the high Paraná and Uruguay rivers (because they are dominant in this stretch), and 12 lotic stretches (in Paraguay, middle and low Paraná and Uruguay rivers (Fig. 1).
The main rivers of the La Plata" River are the Paraná, Paraguay and Uruguay rivers. In each river, we chose sampling sites that were deemed representative of three stretches of the river: high, middle and low. In the main river of the basin, the Paraná River, we sampled the first reservoir (Ilha Solteira Reservoir) after it had been built. We also sampled one more reservoir (Itaipu Reservoir) at the end of the high stretch (680 km apart from each other), and another reservoir at the end of the Paraná River in the beginning of the middle stretch (1,000 away from the first sampling site). After this last reservoir (Yacyreta Reservoir), at the middle stretch of the river, there was a long lotic stretch, where we sampled six sites, each being approximately 250 km apart from the other, until we reached the mouth of the "La Plata" River, between Buenos Aires and Uruguay.
Beyond the three main rivers of the "La Plata" basin (Paraná, Paraguay and Uruguay), we sampled five main tributary rivers of the Paraná River (Grande, Paranaiba, Tiete, Paranapanema, and Iguaçu rivers), because collectively they amount to a large area of the basin in the high stretch. All tributaries are totally dammed, with a long cascade formed by a series of reservoirs. In each tributary river, we sampled the first and the last large reservoir. In general, the first reservoir of these tributaries has a dendritic shape, a large area (more than 200 km2), is deep and has high volume and high water retention time, functioning as a regulator for the downstream reservoir series (AGOSTINHO et al. 2007, NOGUEIRA et al. 2012). In the Uruguay River, many large reservoirs have been constructed, especially in the high stretch, but there is an old reservoir in the low stretch. We sampled two reservoirs (the first and the last reservoirs in this river), and three sites in lotic stretches (two in the middle stretch, 260 km away from each other), and one in the low stretch, 5 km from the delta of Paraná River.
In the Paraguay River there are no reservoirs, only lotic stretches. Thus, the high, middle (250 km apart) and low stretches (650 km below the middle station) were sampled in the main channel. In each sampling site, we obtained zooplankton samples from open waters (limnetic region) and also zooplankton samples within macrophyte stands. In total, ninety-six samples were obtained.
As far as zooplankton are concerned, the sampling was obtained from the main river channels and from upstream zones of the reservoir, all of these with water retention time (WRT) longer than 15 days (see AGOSTINHO et al. 2007, ZALOCAR DE DOMITROVIC et al. 2007, BOLTOVSKOY et al. 2013), which is considered as the lower time threshold for the development of a copepod life cycle (RIETZLER et al. 2002). The approximate WRT of each reservoir is indicated in Fig. 1, to highlight differences between storage reservoirs and run-of-river or intermediate reservoirs. Water flow values in lotic sites are also shown in Fig. 1.
Zooplankton samples were taken by vertical hauls through water column (from close to the bottom to the surface) at each station. Values of water filtered varied between 706 L and 2,826 L, and the average value was 1,766 L. Conical plankton nets, 0.30 m mouth diameter per 0.90m side length, and 68 ìm mesh size, were used after being modified with a mouth reducing cone (TRANTER & SMITH 1979), a kind of anti-reflux bulkhead with another 0.50 m diameter circle. In deeper stations the vertical hauls were extended to a depth of 40m. In rivers, vertical hauls were taken from a drifting boat in order to ensure that the hauls were not excessively oblique. The volume of water filtered was estimated by the cylinder volume formula, using: pi * radius of the mouth net2 * the length of the haul.
For samples obtained inside macrophyte stands, organisms were sampled with conical plankton nets of 68 ìm of mesh size, adapted with a 2 m drive cable of aluminum and with a steel screen to avoid excessive macrophyte intake. This net was passed between and alongside the macrophyte banks at standardized time of five minutes to obtain good amount of qualitative material.
Samples were fixed with 4% formalin solution. In the laboratory, they were analysed in their totality. Male and female copepods were identified to species. Copepods were examined under stereo- and compound microscopes, and identified using specialized taxonomic references (REID 1985, EINSLE 1996, ROCHA 1998, KARAYTUG 1999, ALEKSEEV 2002, UEDA & REID 2003, SILVA & MATSUMURA-TUNDISI 2005). Species richness was considered according to the number of species, in the most robust way as possible. The samples obtained in this study are deposited in the "Collection of microcrustaceans of continental waters" of "Universidade Estadual Paulista" (in Botucatu, Brazil). Vouchers are also deposited in at the Museu de Zoologia da Universidade de São Paulo MZUSP) (e.g., Macrocyclops albidus (Jurine, 1820) (MZUSP30601), Eucyclops neumani (Pesta, 1927) (MZUSP30602), and Microcyclops ceibaensis (Marsh, 1919) (MZUSP30603).
The non-parametric Man Whitney U test (for non-parametric data) was used to compare species richness between zooplankton and macrophytes stand. We used R Cran Project (R DEVELOPMENT CORE TEAM 2012) for this test.
A non-metric multidimensional scaling (NMDS) analysis using Bray-Curtis dissimilarity was applied for spatial ordination of the data, aiming to verify differences among sampling sites, and taking the species into consideration. We used the Vegan and Mass packages for software R Cran Project (R DEVELOPMENT CORE TEAM 2012), according to OKSANEN (2013). The iterative search was carried out using the "meta MDS" function, by several random starts, and selecting among similar solutions with the smallest stresses. For scaling we used centering, PC rotation and half change scaling.
RESULTS
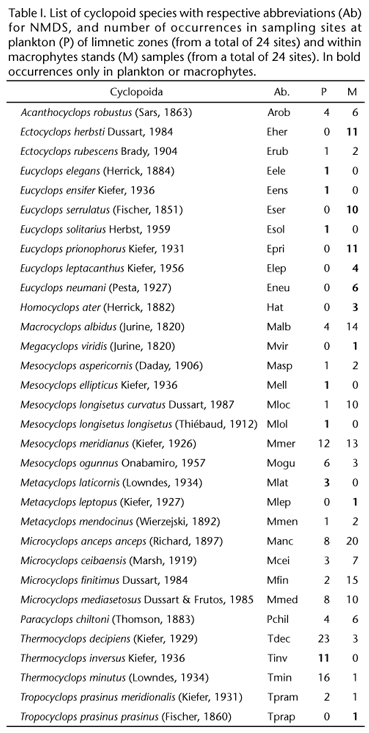
We identified 32 species of cyclopoid copepods (Table I), 23 of which were found in zooplankton and 26 in macrophytes. Six species were exclusive to plankton, 10 to macrophyte stands, and 17 were common to both. Only Mesocyclops meridianus (Kiefer, 1926) was found in similar proportions in plankton (50% from total samples) and in macrophytes (54%), while five species (e.g., Thermocyclops) were widely found in plankton, and thirteen (e.g., Microcyclops and Eucyclops) in macrophytes.
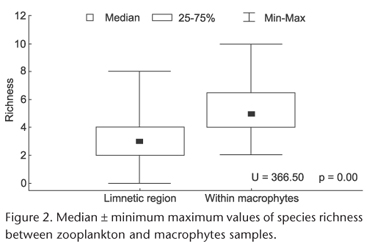
In general, higher median value of species richness was found in macrophyte than in plankton samples (Fig. 2), with a significant difference. For zooplankton in the summer we found a mean ± standard-deviation of 2.75 ± 1.03 species and 3.12 ± 1.74 species. For macrophytes, we found 5.08 ± 1.62 species in the summer and 5.41 ± 2.20 species per sample.
After 20 attempts we found two convergent solutions for NMDS. This analysis (Fig. 3) classified the kind of habitat sample in basically three groups: zooplankton, macrophytes, and a median line where zooplankton and macrophyte samples were located close to each other, supporting the result shown in Table I. Geographical distribution was strongly affected by the habitat sampled. There was no clear spatial pattern of endemicity for cyclopoid species within the La Plata basin.
DISCUSSION
Our results were similar to those obtained by MAIA-BARBOSA et al. (2008), which found major richness of zooplanktonic species in places with aquatic macrophytes, pointing to the heterogeneity of habitats as the responsible factor. Thus, the hypothesis tested in our study was corroborated. The model of GENKAI-KATO (2007) supports this statement, pointing out macrophyte areas as a refuge for zooplankton species to avoid predation of planktivorous fish.
The greatest number of exclusive species was found in macrophytes. Our results show that some genera, as Eucyclops and Ectocyclops are more frequent in macrophytes than in zooplankton, where Thermocyclops is common. This result agrees with LANSAC-TOHA et al. (2002) and MAIA-BARBOSA et al. (2008). LANSAC-TOHA et al. (2002) found 12 species of cyclopoids in the flood plain of the upper Paraná River in aquatic macrophytes. Of these species, a few were in plankton, being found more frequently and sometimes exclusively in macrophytes, similar to the findings of MAIA-BARBOSA et al. (2008). These authors found13 species of cyclopoid copepods in litoranean regions with aquatic macrophytes in Lake Dom Helvécio (State of Minas Gerais, Brazil), but only 7 of these were found in areas without macrophytes.
We did not find any clear pattern of geographical distribution for the cyclopoid species of our study. Many species are well distributed and some of them are cosmopolitan (UEDA & REID 2003, BOXSHALL & DEFAYE 2008). According to SELDEN et al. (2010) the recent evolution of the order Cyclopoida in the Neogene (± 15 M.A.) is another possible factor for its wide distribution. The results for Cyclopoida contrasts with the results of other freshwater copepods such as diaptomids and some harpacticoids, according to the literature.
In our results, the most widely distributed species were Ectocyclops herbsti Dussart, 1984, Eucyclops serrulatus (Fischer, 1851), Macrocyclops albidus (Jurine, 1820), Microcyclops anceps (Richard, 1897), Microcyclops finitimus Dussart, 1984, Microcyclops ceibaensis (Marsh, 1919), Thermocyclops decipiens (Kiefer, 1929), Thermocyclops minutus (Lowndes, 1934), and Thermocyclops inversus Kiefer, 1936. It is important to highlight that some species, for instance Mesocyclops aspericornis (Daday, 1906) and Mesocyclops ogunnus Onabamiro, 1957, are invasive from Africa (UEDA & REID 2003).The species cited above are widely distributed in the Neotropical Region (REID 1985). The most common species, in addition to those that occur in both environments sampled, are adapted to different ecosystems. This contrasts with species that are from litoranean zones or macrophyte stands. Examples are Homocyclops ater (Herrick, 1882) and E. herbsti.
Compared to other studies in the La Plata basin (e.g., PAGGI & JOSÉ DE PAGGI 1990, LANSAC-TOHA et al. 2002, SILVA & MATSUMURA-TUNDISI 2002, NOGUEIRA et al. 2008), the number of species found in this study is considerably high, and this was attributed to the combination of sampling in a large area and in two types of habitat (plankton and macrophytes), allowing the capture of exclusive species from specific habitats. Our richness of cyclopoids is also high if compared with other basins in the world (TASH 1971, KOBAYASHI et al. 1998).
The geographical distribution of cyclopoid copepods was highly influenced by the type of habitat sampled, as shown by the NMDS. This result confirms that some species are almost restricted to open waters or to macrophytes stands. Other species are common in several kinds of habitats, or can be found accidently in them, when removed by the water flow, for example. For a more accurate estimate of the diversity of these copepods it is necessary to sample at least two kinds of habitats, open waters and macrophyte stands.
ACKNOWLEDGEMENTS
We thank FAPESP (2008/02015-7; 2009/00014-6; 2011/ 18358-3) for financial support.
LITERATURE CITED
Submitted: 17.X.2013;
Accepted: 12.III.2014.
Editorial responsibility: Marcos D.S. Tavares
All content of the journal, except where identified, is licensed under a Creative Commons attribution-type BY-NC.
- AGOSTINHO, A.A.; L.C. GOMES & F.M. PELICICE. 2007. Ecologia e manejo de recursos pesqueiros em reservatórios do Brasil Maringá, Eduem, 501p.
- ALEKSEEV, V.R. 2002. Copepoda, p. 123-188. In: C.H. FERNANDO (Ed.). A guide to tropical freshwater zooplankton: Identification, ecology and impact on fisheries London, Backhuys Publishers.
- BOLTOVSKOY, D.; N. CORREA; F. BORDET; V. LEITES & D. CATALDO. 2013. Toxic Microcystis (cyanobacteria) inhibit recruitment of the bloom-enhancing invasive bivalve Limnoperna fortunei Freshwater Biology 58 (9): 1968-1981. doi:10.1111/ fwb.12184
- BOXSHALL, G.A. & D. DEFAYE. 2008. Global diversity of Copepods (Crustacea: Copepoda) in freshwater. Hydrobiologia 595: 195-207.
- BOXSHALL, G.A. & D. JAUME. 2000. Making Waves: The Repeated Colonization of Fresh Water by Copepod Crustaceans. Advances in Ecological Research 31: 61-79.
- EINSLE, U. 1996. Copepoda Cyclopoida: Genera Cyclops, Megacyclops, Acanthocyclops, p. 1-82. In: H.J.F. DUMONT (Ed.). Guides to the identification of the microinvertebrates of the continental waters of the world The Netherlands, Backhuys Publishers.
- FANTIN-CRUZ, I.; K.K. TONDATO; J.M.F. PENHA; L.A.F. MATEUS; P. GIRARD & R. FANTIN-CRUZ. 2008. Influence of fish abundance and macrophyte cover on microcrustacean density in temporary lagoons of the Northern Pantanal-Brazil. Acta Limnologica Brasiliensia 20 (4): 339-344.
- GENKAI-KATO, M. 2007. Macrophyte refuges, prey behaviour and trophic interactions: consequences for lake water clarity. Ecology Letters 10: 105-114.
- GERALDES, A.M. & M.J. BOAVIDA. 2004. Do Littoral Macrophytes Influence Crustacean Zooplankton distribution? Limnetica 23 (1-2): 57-64.
- KARAYTUG, S. 1999. Genera Paracyclops, Ochridacyclops and key to the Eucyclopinae. In: H.J.F. DUMONT (Ed.). Guides to the identification of the microinvertebrates of the continental waters of the world The Netherlands, Backhuys Publishers.
- KOBAYASHI, T.; R.J. SHIEL; P. GIBBS & P.I. DIXON. 1998. Freshwater zooplankton in the Hawkesbury-Nepean River: comparison of community structure with other rivers. Hydrobiologia 377: 133-145.
- LANSAC-TÔHA, F.A.; L.F.M. VELHO; J. HIGUTI & E.M. TAKAHASHI. 2002. Cyclopidae (Crustacea, Copepoda) from the upper Paraná River floodplain, Brazil. Brazilian Journal of Biology 62 (1): 125-133.
- LUCENA-MOYA, P. & I.C. DUGGAN. 2011. Macrophyte architecture affects the abundance and diversity of littoral microfauna. Aquatic Ecology 45: 279-287.
- MAIA-BARBOSA, P. M.; R.S. PEIXOTO & A.S. GUIMARÃES. 2008. Zooplankton in littoral Waters of a tropical lake: a revisited biodiversity. Brazilian Journal of Biology 68 (4): 1061-1067.
- NOGUEIRA, M.G.; P.C. REIS-OLIVEIRA & Y.T. BRITTO. 2008. Zooplankton assemblages (Copepoda and Cladocera) in a cascade of reservoirs of a large tropical river (SE Brazil). Limnetica 27 (1): 151-170.
- NOGUEIRA, M.G.; G. PERBICHE-NEVES & D.A.O. NALIATO. 2012. Limnology of two contrasting hydroelectric reservoirs (storage and run-of-river) in southeast Brazil, p. 167-184. In: H. SAMADI-BOROUJENI (Ed.). Hydropower Croatia, Intech.
- OKSANEN, J. 2013. Multivariate Analysis of Ecological Communities in R: vegan tutorial Available online at: http://cc.oulu.fi/~jarioksa/opetus/metodi/vegantutor.pdf [Accessed: 14/VII/2013]
- PAGGI, J.C. & S. JOSÉ DE PAGGI. 1990. Zooplankton de ambientes lóticos e lênticas do rio Paraná médio. Acta Limnologica Brasiliensia 3: 685-719.
- PERBICHE-NEVES, G.; M. SERAFIM-JÚNIOR; A.R. GHIDINI & L. BRITO. 2007. Spatial and temporal distribution of Copepoda (Cyclopoida and Calanoida) of an eutrophic reservoir in the basin of upper Iguaçu River, Paraná, Brazil. Acta Limnologica Brasiliensia 19 (4): 393-406.
- R DEVELOPMENT CORE TEAM. 2012. A language and environment for statistical computing. Vienna, Austria, R Foundation for Statistical Computing ISBN 3-900051-07-0, URL. Available online at: http://www.R-project.org [Accessed10.XII.2012]
- REID, J.W. 1985. Chave de identificação e lista de referências bibliográficas para as species continentais sulamericanas de vida livre da Ordem Cyclopoida (Crustacea, Copepoda). Boletim de Zoologia, Universidade de São Paulo 9: 17-143.
- REID, J.W. 1989. The distribution of species of the genus Thermocyclops (Copepoda, Cyclopoida) in the western hemisphere, with description of T. parvus, new species. Hydrobiologia 175: 149-174.
- RIETZLER, A.C.; T. MATSUMURA-TUNDISI & J.G. TUNDISI. 2002. Life cycle, feeding and adaptive strategy implications on the cooccurence of Argyrodiaptomus furcatus and Notodiaptomus iheringi in Lobo-Broa Reservoir (SP, Brazil). Brazilian Journal of Biology 62: 93-105.
- ROCHA, C.E.F. 1998. New morphological characters useful for the taxonomy of genus Microcyclops (Copepoda, Cyclopoida). Journal of Marine Systems 15: 425-431.
- SELDEN, P.A.; R. HUYS; M.H. STEPHENSON; A.P. HEWARD & P.N. TAYLOR. 2010. Crustaceans from bitumen clast in Carboniferous glacial diamictite extend fossil record of copepods. Nature Communications 1 (50): 1-6.
- SILVA, W.M. & T. MATSUMURA-TUNDISI. 2002. Distribution and abundance of Cyclopoida populations in a cascade of reservoir of the Tietê River (São Paulo State, Brazil).Verhandlungen Internationale Vereinigung für Theoretische und Angewandte Limnologie 28: 667-670.
- SILVA, W.M. & T. MATSUMURA-TUNDISI. 2005. Taxonomy, ecology, and geographical distribution of the species of the genus Thermocyclops Kiefer, 1927 (Copepoda, Cyclopoida) in São Paulo State, Brazil, with description of a new species. Brazilian Journal of Biology 65 (3): 521-31.
- SILVA, W.M. 2008. Diversity and distribution of the free-living freshwater Cyclopoida (Copepoda:Crustacea) in the Neotropics. Brazilian Journal of Biology 68 (4): 1099-1106.
- SUÁREZ-MORALES, E.; J.W. REID; F. FIERS & T.M. ILIFFE. 2004. Historical biogeography and distribution of the freshwater cyclopine copepods (Copepoda, Cyclopoida, Cyclopinae) of the Yucatan Peninsula, Mexico. Journal of Biogeography 31 (7): 1051-1063.
- SUÁREZ-MORALES, E.; J.W. REID & M. ELÍAS-GUTIÉRREZ. 2005. Diversity and Distributional Patterns of Neotropical Freshwater Copepods (Calanoida: Diaptomidae). International Review of Hydrobiology 90 (1): 71-83.
- TASH, J.C. 1971. Some Cladocera and Copepoda from the Upper Klamath River Basin. Northwest Science 45 (4): 239-243.
- TRANTER, D.J. & P.E. SMITH. 1979. Filtration performance, p. 27-56. In: D.J. TRANTER (Ed.). Zooplankton sampling Paris, Imprimerie Rolland, 3rd ed.
- UEDA, H. & J.W. REID. 2003. Copepoda: Cyclopoida Genera Mesocyclops and Thermocyclops, p. 1-316. In: H.J.F. DUMONT (Ed.). Guides to the identification of the microinvertebrates of the continental waters of the world The Netherlands, Backhuys Publishers.
- ZALOCAR DE DOMITROVIC, Y.; A.S.G. POI DE NEIFF & S.L. CASCO. 2007. Abundance and diversity of phytoplankton in the Paraná River (Argentina) 220 km downstream of the Yacyretá Reservoir. Brazilian Journal of Biology 67 (1): 53-63.
Publication Dates
-
Publication in this collection
04 July 2014 -
Date of issue
June 2014
History
-
Accepted
12 Mar 2014 -
Received
17 Oct 2013