Abstracts
Gunnera perpensa (Gunneraceae) is an African plant widely used in traditional medicine. This species is known for its activity involving the female reproductive system, such as inducing or increasing labor, treating female infertility, expelling the placenta and/or preventing post-partum hemorrhage. These properties are probably due to (z)-venusol, a majoritary compound, and its action in conjunction with substances in the whole extract and other natural products. In southern Brazil, a native species Gunnera manicata L. that also belongs to Gunneraceae can be found. In spite of the traditional use of G. perpensa, there is no pharmacological and phytochemical information regarding the South American Gunnera species. Therefore, the aim of this study was to investigate the activity of Brazilian G. manicata aqueous extracts on the reproductive system of immature female Wistar rats through a uterotrophic assay and to verify the presence of (z)-venusol by liquid chromatography coupled with tandem mass spectrometry (LC-MS/MS). Data were analyzed by analysis of variance (ANOVA) and Bonferroni´s post-hoc test (p< 0.01). Results obtained shown that G. manicata extracts did not present in vivo anti or estrogenic activity. Furthermore, (z)-venusol compound was not found. This study represents the first preliminary screening done on the South American G. manicata species.
Gunnera manicata L.; Gunneraceae; Medicinal plants; Uterotrophic assay
Gunnera perpensa (Gunneraceae) é uma planta de origem africana extensamente utilizada na medicina tradicional do país. Esta espécie é conhecida por suas atividades no sistema reprodutor feminino, como indução ou aumento do trabalho de parto, tratamento da infertilidade em mulheres, expulsão da placenta e/ou impedimento de hemorragia pós-parto. Tais atividades devem-se, provavelmente, ao sinergismo existente entre o (z)-venusol, composto majoritário, e outros compostos presentes na planta. No sul do Brasil, encontra-se uma espécie nativa, Gunnera manicata L., pertencente à família Gunneraceae. Apesar do uso tradicional de G. perpensa, não há informações farmacológicas e fitoquímicas a respeito da espécie sul Americana de Gunnera. Assim, o objetivo deste estudo foi investigar a atividade de extratos aquosos da espécie brasileira G. manicata no sistema reprodutor de ratas Wistar imaturas através de ensaio uterotrófico e verificar a presença do composto (z)-venusol utilizando-se cromatografia líquida acoplada a espectrômetro de massas em tandem (CL-EM/EM). Para a análise estatística, utilizou-se ANOVA/Bonferroni (p<0,01). Os resultados obtidos demonstraram que os extratos de G. manicata testados não apresentaram atividade anti ou estrogênica in vivo. Na análise química não foi verificada a presença do composto (z)-venusol. Este estudo representa o primeiro screening realizado com a espécie sul-americana G. manicata.
Gunnera manicata L.; Gunneraceae; Plantas medicinais; Ensaio uterotrófico
ARTICLE
Evaluation of anti-estrogenic or estrogenic activities of aqueous root extracts of Gunnera manicata L.
Kristiane de Cássia MariottiI; Gabriela Cristina SchmittI; Fabiano BarretoII; Rodrigo E. FortunatoII; Rodrigo B. SingerIII; Eliane DallegraveIV; Mirna Bainy LealV; Renata Pereira LimbergerI, * * Correspondence: R. P. Limberger. Laboratório de Análises e Pesquisas Toxicológicas, Departamento de Análises, Faculdade de Farmácia, Universidade Federal do Rio Grande do Sul. Av. Ipiranga, 2752 - 90610-000 - Porto Alegre - RS, Brazil. E-mail address: renata@ufrgs.br
ILaboratory of Analysis and Toxicological Research, Department of Analysis, Faculty of Pharmacy, Federal University of Rio Grande do Sul
IINational Agricultural Laboratory of Rio Grande do Sul, LANAGRO/RS
IIIDepartment of Botany, Federal University of Rio Grande do Sul
IVToxicology Information Center of Rio Grande do Sul, State Foundation of Production and Research in Health
VLaboratory of Pharmacology and Toxicology of Natural Products, Department of Pharmacology, Institute of Basic Health Sciences, Federal University of Rio Grande do Sul
ABSTRACT
Gunnera perpensa (Gunneraceae) is an African plant widely used in traditional medicine. This species is known for its activity involving the female reproductive system, such as inducing or increasing labor, treating female infertility, expelling the placenta and/or preventing post-partum hemorrhage. These properties are probably due to (z)-venusol, a majoritary compound, and its action in conjunction with substances in the whole extract and other natural products. In southern Brazil, a native species Gunnera manicata L. that also belongs to Gunneraceae can be found. In spite of the traditional use of G. perpensa, there is no pharmacological and phytochemical information regarding the South American Gunnera species. Therefore, the aim of this study was to investigate the activity of Brazilian G. manicata aqueous extracts on the reproductive system of immature female Wistar rats through a uterotrophic assay and to verify the presence of (z)-venusol by liquid chromatography coupled with tandem mass spectrometry (LC-MS/MS). Data were analyzed by analysis of variance (ANOVA) and Bonferroni´s post-hoc test (p< 0.01). Results obtained shown that G. manicata extracts did not present in vivo anti or estrogenic activity. Furthermore, (z)-venusol compound was not found. This study represents the first preliminary screening done on the South American G. manicata species.
Uniterms:Gunnera manicata L./pharmacognosy. Gunneraceae/pharmacognosy. Medicinal plants. Uterotrophic assay/experimental study.
RESUMO
Gunnera perpensa (Gunneraceae) é uma planta de origem africana extensamente utilizada na medicina tradicional do país. Esta espécie é conhecida por suas atividades no sistema reprodutor feminino, como indução ou aumento do trabalho de parto, tratamento da infertilidade em mulheres, expulsão da placenta e/ou impedimento de hemorragia pós-parto. Tais atividades devem-se, provavelmente, ao sinergismo existente entre o (z)-venusol, composto majoritário, e outros compostos presentes na planta. No sul do Brasil, encontra-se uma espécie nativa, Gunnera manicata L., pertencente à família Gunneraceae. Apesar do uso tradicional de G. perpensa, não há informações farmacológicas e fitoquímicas a respeito da espécie sul Americana de Gunnera. Assim, o objetivo deste estudo foi investigar a atividade de extratos aquosos da espécie brasileira G. manicata no sistema reprodutor de ratas Wistar imaturas através de ensaio uterotrófico e verificar a presença do composto (z)-venusol utilizando-se cromatografia líquida acoplada a espectrômetro de massas em tandem (CL-EM/EM). Para a análise estatística, utilizou-se ANOVA/Bonferroni (p<0,01). Os resultados obtidos demonstraram que os extratos de G. manicata testados não apresentaram atividade anti ou estrogênica in vivo. Na análise química não foi verificada a presença do composto (z)-venusol. Este estudo representa o primeiro screening realizado com a espécie sul-americana G. manicata.
Unitermos:Gunnera manicata L./fitoquímica. Gunneraceae/fitoquímica. Plantas medicinais. Ensaio uterotrófico/estudo experimental.
INTRODUCTION
Gunnera (Gunneraceae) species are mainly found in the Southern hemisphere. In Southern Africa, G. perpensa is a medicinal plant used by traditional healers for the treatment of venereal diseases (Buwa et al., 2006). It is also a traditional herbal remedy used by many South African women as antenatal medications or to induce or augment labor, to treat female infertility, expel the placenta and/or prevent post-partum hemorrhage (Kaido et al., 1996; Khan et al., 2004). Kahn et al. (2004) showed that aqueous extract of G. perpensa directly stimulates a contractile response for both the ileum and uterus muscle in isolated muscle obtained from a Sprague-Dawley rat organ assay. These properties with an effect on the female reproductive system are probably due to the (z)-venusol compound. The results found by Khan and co-workers suggest that (z)-venusol acts in conjunction with substances in the whole extract (Khan et al., 2004). Venusol is a phenylpropanoid glycoside (Vionery et al,. 2000) isolated previously by Proliac et al. (1981) and Pagani (1990). Both the z and e isomers were obtained by Viornery et al. (2000). In all these instances the plant source was Umbilicus pendulinus or Umbilicus ruperstris (Crassulaceae) (Khan et al., 2004).
The Gunnera manicata L. species is a native plant from Southern Brazil, growing at high altitudes (from 900 m upwards) on the border between the States of Rio Grande do Sul and Santa Catarina, in a region locally known as "Aparados da Serra". Despite being appreciated as an ornamental plant and belonging to the same family (Gunneraceae) of G. perpensa (extensively studied species), there is scant data about the pharmacology and phytochemical properties of G. manicata in the scientific literature. Therefore, new approaches for this species are necessary.
Considering the extensive traditional use and proven activities on the female reproductive system of G. perpensa, the aim of this study was to investigate the activity of Brazilian G. manicata extracts on the reproductive system of immature Wistar rats through uterotrophic assays and verify the presence of (z)-venusol by liquid chromatography coupled with tandem mass spectrometry (LC-MS/MS). This study represents one of the first analyses of the pharmacological and phytochemical properties of G. manicata.
MATERIAL AND METHODS
Plant material
Roots of G. manicata were collected in the Aparados da Serra region, Southern Brazil, and identified by Rodrigo B. Singer. A voucher specimen (R.B. Singer s. n., Serra da Rocinha, January 15th, 2009) was deposited at the ICN herbarium - Department of Botany - UFRGS in Porto Alegre, Brazil.
Preparation of extracts
The dried roots of G. manicata were extracted in a water bath at 50 °C for 3 hours (ARE aqueous root extracts). These extracts were collected, filtered, concentrated to dryness under reduced pressure and the residue was refrigerated until later use in the experiments. The ARE were dissolved in water at doses of 500.0 and 1500.0 mg/ml.
Uterotrophic assay
Animals
Immature female Wistar rats (21 days) obtained from the Fundação Estadual de Produção e Pesquisa em Saúde (FEPPS, Porto Alegre, RS, Brazil) were used. The animals were housed in polyethylene cages under standard conditions of temperature (22 ± 2 ºC), controlled humidity and 12 h-light/dark cycles. Standard pellet food and tap water were available ad libitum. The experiments were performed after approval of the protocol by the FEPPS Ethics Committees (numbers 04/2008) and were carried out in accordance with current guidelines for the care of laboratory animals.
Biological assay
The test compounds were given daily for 3 consecutive days by oral gavage (po) to the immature female rats (6-7 animals/group). ARE at doses of 500.0 and 1500.0 mg/kg/day were used to assess possible estrogenic activity. For detection of antiestrogenicity, tamoxifen 20.0 mg/kg/day and a dose of 1500.0 mg/kg/day were administered to estrogen-treated females (estradiol cypionate 0.4 mg/kg/day). The vehicle (corn oil 10 mL/kg/day) was administered as a negative control while estradiol cypionate (0.4 mg/kg/day) was used as a positive control for estrogenicity and tamoxifen (20.0 mg/kg/day) as a positive control for antiestrogenicity. The dosing volume for all solutions was 10 mL/kg body weight. Animals were weighed every day and killed by cervical dislocation 24 h after the final dose. After euthanasia, the rats were necropsied and analyzed for macroscopic alterations in liver, kidneys, adrenals, uterus and ovaries. The uterus was excised, trimmed free of fat, pierced, and dried to remove fluid. The body of the uterus was cut just above its junction with the cervix and at the junction of the uterine horns with the ovaries (Odum et al., 1997; Arbo et al., 2009). The weight of the organs was determined and expressed as relative weight (organ mass/body weight x 100).
Data analysis
Data were analyzed by analysis of variance (ANOVA). Differences between groups were determined by Bonferroni´s post-hoc test and level of significance was set at 1% (p< 0.01).
LC-MS/MS analysis
The analysis was performed by direct infusion into a Sciex API 5000 triple stage quadrupole mass spectrometer (Applied Biosystems, Foster City, CA, USA). The mobile phase used in the infusion procedure was mixed with solvent A (0.1% formic acid) and solvent B (acetonitrile). The flow rate of the syringe was 10 µl min-1. The ions were monitored in full scan in positive and negative modes, while nitrogen was used for both nebulizer and collision gas. The (z)-venusol was acetylated and analyzed according to Kahn and co-workers (Khan et al., 2004).
RESULTS
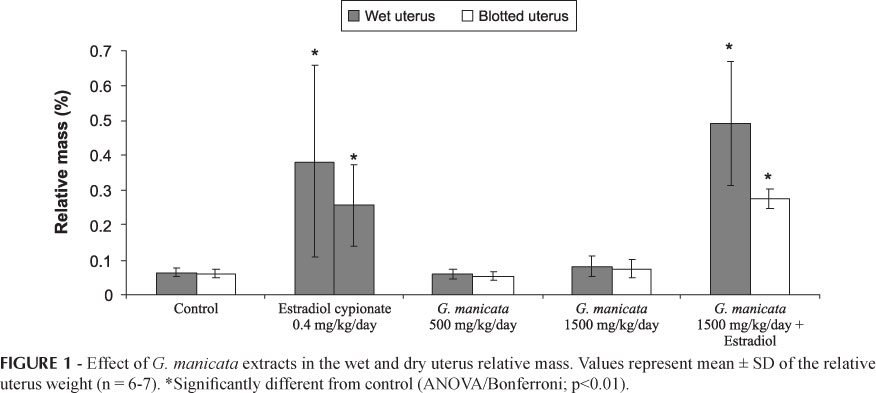
On the uterotrophic assay, no abnormal macroscopic findings or body weight changes were detected in the rats given the test extracts. The wet and dry relative weights of the uterus of rats given estradiol cypionate (0.4 mg/kg/day) were significantly increased compared to the vehicle. Tamoxifen 20.0 mg/kg/day significantly reverted the estradiol effect. The relative uterine mass of rats treated with extracts was not significantly different from control (Figure 1).
The weight of the liver, kidneys and adrenals was determined and no statistically significant changes were observed in the assessment of the relative mass of organs of necropsied animals (Table I).
Studies concerning isolation or characterization of secondary metabolites are necessary to elucidate the phytochemical profile of species. Therefore, chemical analysis was performed focusing on the presence or absence of (z)-venusol, the majoritary compound of G. perpensa. The LC-MS/MS analysis did not yield the fragmentation profile of (z)-venusol in the G. manicata species.
In the investigation for the presence of phenolic compounds: rutin, quercetin, chlorogenic acid and gallic acid by LC-MS/MS, only gallic acid was founded in the analyzed samples.
DISCUSSION
Gunnera (Gunneraceae) comprises more than forty species occurring mainly in the Southern Hemisphere (Wanntorp, 2003). Considering this large occurrence, this study sought to draw a parallel between the South-African G. perpensa and South American G. manicata, focusing on the activities and chemical profile of the latter. Therefore, the aim of this study was to elucidate the possible effect of G. manicata on the female reproductive system and to verify the presence of a secondary metabolite, (z)-venusol, in aqueous root extracts.
The results obtained in a uterotrophic assay demonstrated that G. manicata aqueous root extracts do not exert estrogen-like effects or anti-estrogenic-like effects on immature female rats. These data were verified by the absence of abnormal macroscopic findings and lack of statistical significance in the relative uterine weight of rats given the test extracts versus the negative controls. These results were concordant with the phytochemical analysis that showed absence of (z)-venusol. This phenylpropanoid glycoside is the majoritary compound of Gunnera perpensa extract and directly stimulates a contractile response in both the ileum and uterus muscle. These activities are probably due to the presence of (z)-venusol that induces a state of spontaneous contractility in the organ bath test. It is possible that the (z)-venusol acts in conjunction with substances present in the whole extract.
Thus, our findings are consistent with those presented by Khan and co-workers suggesting that the activities on the female reproductive system are due to a synergism between (z)-venusol and other compounds present in whole extract. Consequently, the lack of (z)-venusol promoted an absence of activities.
ACKNOWLEDGMENTS
This study was supported by the CNPq. The authors would like to acknowledge FEPPS for donation of the animals.
Received for publication on 19th October 2010
Accepted for publication on 30th May 2011
- ARBO, M. D.; FRANCO, M. T.; LARENTIS, E. R.; GARCIA, S. C.; SEBBEN, V.C.; LEAL, M. B.; DALLEGRAVE, E.; LIMBERGER, R. P. Screening for in vivo (anti)estrogenic activity of ephedrine and p-synephrine and their natural sources Ephedra sinica Stapf. (Ephedraceae) and Citrus aurantium L. (Rutaceae) in rats. Arch. Toxicol., v.83, n.1, p.95-99, 2009.
- BUWA, L.V.; VAN STADEN, J. Antibacterial and antifungal activity of traditional medicinal plants used against venereal diseases in South África. J. Ethnopharmacol., v.103, n.1, p.139-142, 2006.
- KAIDO, T. L.; VEALE, D. J. H.; HAVLIK, I.; RAMA, D. B. K. Preliminary screening of plants used in South Africa as traditional herbal remedies during pregnancy and labour. J. Ethnopharmacol, v.55, n.3, p.185-191, 1996.
- KHAN, F.; PETER, X. K.; MACKENZIE, R. M.; KATSOULIS, L.; GEHRING, R.; MUNRO, O. Q.; HEERDEN, F. R. VAN.; DREWES, S. E. Venusol from Gunnera perpensa: structural and activity studies. Phytochemistry, v.65, n.8, p.1117-1121, 2004.
- ODUM, J.; LEFEVRE, P.A.; TITTENSOR, S.; PATON, D.; ROUTLEDGE, E.J.; BERESFORD, N.A.; SUMPTER, J.P.; ASHBY, J. The rodent uterotrophic assay: Critical protocol features, studies with phenols, and comparison with a yeast estrogenicity assay. Regul. Toxicol. Pharmacol, v.25, n.2, p.176-188, 1997.
- VIORNERY, L.; SALIBA, C.; DASKIEWICZ, JB.; BAYET, C.; COMTE, G.; FENET, B.; GUTIERREZ, G.; BARRON, D. Phenylpropanoids from Umbilicus pendulinus Chem. Pharm. Bull. (Tokyo), v.48, n.11, p.1768-1770, 2000.
- WANNTORP, L.; WANNTORP, H. E. The biogeography of Gunnera L.: vicariance and dispersal. J. Biogeogr., v.30, n.7, p. 979-987, 2003.
Publication Dates
-
Publication in this collection
24 Oct 2011 -
Date of issue
Sept 2011
History
-
Received
19 Oct 2010 -
Accepted
30 May 2011