Abstracts
Since its discovery, myostatin (MSTN) has been at the forefront of muscle therapy research because intrinsic mutations or inhibition of this protein, by either pharmacological or genetic means, result in muscle hypertrophy and hyperplasia. In addition to muscle growth, MSTN inhibition potentially disturbs connective tissue, leads to strength modulation, facilitates myoblast transplantation, promotes tissue regeneration, induces adipose tissue thermogenesis and increases muscle oxidative phenotype. It is also known that current advances in gene therapy have an impact on sports because of the illicit use of such methods. However, the adverse effects of these methods, their impact on athletic performance in humans and the means of detecting gene doping are as yet unknown. The aim of the present review is to discuss biosynthesis, genetic variants, pharmacological/genetic manipulation, doping and athletic performance in relation to the MSTN pathway. As will be concluded from the manuscript, MSTN emerges as a promising molecule for combating muscle wasting diseases and for triggering wide-ranging discussion in view of its possible use in gene doping.
Myostatin; Myostatin; Myostatin; Gene doping; Gene therapy; Physical performance; Skeletal muscle
Desde sua descoberta, a miostatina (MSTN) entrou na linha de frente em pesquisas relacionadas às terapias musculares porque mutações intrínsecas ou inibição desta proteína tanto por abordagens farmacológicas como genéticas resultam em hipertrofia muscular e hiperplasia. Além do aumento da massa muscular, a inibição de MSTN potencialmente prejudica o tecido conectivo, modula a força muscular, facilita o transplante de mioblastos, promove regeneração tecidual, induz termogênese no tecido adiposo e aumenta a oxidação na musculatura esquelética. É também sabido que os atuais avanços em terapia gênica têm uma relação com o esporte devido ao uso ilícito de tal método. Os efeitos adversos de tal abordagem, seus efeitos no desempenho de atletas e métodos para detectar doping genético são, contudo, desconhecidos. O objetivo da presente revisão de literatura foi discutir biossíntese, variantes genéticas, manipulação genética e farmacológica, e doping relacionado à via da MSTN. Como será concluído do manuscrito, a MSTN emerge como uma molécula promissora para combater doenças atróficas musculares e para gerar muitas discussões devido à sua possível utilização em doping genético.
Miostatina; Miostatina; Miostatina; Doping genético; Desempenho atlético; Músculo esquelético
REVIEW
Myostatin: genetic variants, therapy and gene doping
André Katayama YamadaI; Rozangela VerlengiaII; Carlos Roberto Bueno JuniorIII
IDepartment of Physiological Sciences, Federal University of São Carlos
IICollege of Health Sciences, University Methodist of Piracicaba
IIIInstitute of Biosciences, University of São Paulo
Correspondence Correspondence: C. R. Bueno Junior Institute of Biosciences., University of São Paulo Rua do Matão, Travessa 13, 106, Cidade Universitária 05508-090 - São Paulo-SP, Brazil E-mail: buenojr@usp.br
ABSTRACT
Since its discovery, myostatin (MSTN) has been at the forefront of muscle therapy research because intrinsic mutations or inhibition of this protein, by either pharmacological or genetic means, result in muscle hypertrophy and hyperplasia. In addition to muscle growth, MSTN inhibition potentially disturbs connective tissue, leads to strength modulation, facilitates myoblast transplantation, promotes tissue regeneration, induces adipose tissue thermogenesis and increases muscle oxidative phenotype. It is also known that current advances in gene therapy have an impact on sports because of the illicit use of such methods. However, the adverse effects of these methods, their impact on athletic performance in humans and the means of detecting gene doping are as yet unknown. The aim of the present review is to discuss biosynthesis, genetic variants, pharmacological/genetic manipulation, doping and athletic performance in relation to the MSTN pathway. As will be concluded from the manuscript, MSTN emerges as a promising molecule for combating muscle wasting diseases and for triggering wide-ranging discussion in view of its possible use in gene doping.
Uniterms: Myostatin. Myostatin/genetic variants. Myostatin/pharmacological inhibitors. Gene doping. Gene therapy. Physical performance. Skeletal muscle.
RESUMO
Desde sua descoberta, a miostatina (MSTN) entrou na linha de frente em pesquisas relacionadas às terapias musculares porque mutações intrínsecas ou inibição desta proteína tanto por abordagens farmacológicas como genéticas resultam em hipertrofia muscular e hiperplasia. Além do aumento da massa muscular, a inibição de MSTN potencialmente prejudica o tecido conectivo, modula a força muscular, facilita o transplante de mioblastos, promove regeneração tecidual, induz termogênese no tecido adiposo e aumenta a oxidação na musculatura esquelética. É também sabido que os atuais avanços em terapia gênica têm uma relação com o esporte devido ao uso ilícito de tal método. Os efeitos adversos de tal abordagem, seus efeitos no desempenho de atletas e métodos para detectar doping genético são, contudo, desconhecidos. O objetivo da presente revisão de literatura foi discutir biossíntese, variantes genéticas, manipulação genética e farmacológica, e doping relacionado à via da MSTN. Como será concluído do manuscrito, a MSTN emerge como uma molécula promissora para combater doenças atróficas musculares e para gerar muitas discussões devido à sua possível utilização em doping genético.
Unitermos: Miostatina. Miostatina/variantes genéticas. Miostatina/inibidores farmacológicos. Doping genético. Desempenho atlético. Músculo esquelético.
INTRODUCTION
Myostatin (MSTN), or growth and differentiation factor 8 (GDF-8), is a member of the transforming growth factor (TGF-β) family which acts at physiological levels to limit muscle mass (Lee, 2010; Lee, Glass, 2011; Wang, McPherron, 2012). Genomic and molecular studies in multiple species have demonstrated that mutations in the MSTN gene promote dramatic increase in muscle mass as a result of the combination of hypertrophy and hyperplasia (McPherron, Lee, 1997; Zimmers et al., 2002; Schuelke et al., 2004; Moscher et al., 2007). MSTN inhibition by genetic manipulation and/or drugs also results in hypertrophic phenotype, and this can be a promising strategy to counteract muscle wasting in a diversity of settings (Gilson et al., 2009; Murphy et al., 2010; Heineke et al., 2010; Bish et al., 2011).
MSTN is widely studied in animal models (Mosher et al., 2007; Kota et al., 2009; Bish et al., 2011; Welle et al., 2011; Busquets et al., 2012) but less so in humans for ethical reasons (Wagner et al., 2008). For example, in 1999, an 18-year-old volunteer in a phase trial died after undergoing reactive attachment disorder treatment (RAD) to deliver in vivo a gene encoding a deficient liver enzyme - ornithine transcarbamilase (Raper et al., 2003). Another study reported that the insertion of retrovirus resulted in one death among three children with X-linked severe combined immunodeficiency (Cavazzana-Calvo et al., 2005). It is therefore a complex challenge to translate animal therapies to human clinical trials, for reasons including: differences in the size of the muscles, the nature of the vector, differences in immune responses between species and differences in metabolism (Baotina et al., 2007).
In addition, MSTN manipulation has major implications for the treatment of muscle wasting diseases. Gene therapy techniques can, however, enable the manipulation of biotechnology for illicit use in sport (Mansour, Azzazy, 2009; McKanna, Toriello, 2010; Oliveira et al., 2011). Such a scenario is theoretically possible, but it is not known whether any athletes have already resorted to it. A much debated issue is whether gene doping can enhance human performance among athletes. As mentioned, the available data are limited to animal models.
It is also important to be concerned about adverse effects: would MSTN inhibition or knock-out have deleterious effects on tissues such as bone and tendons because of the disproportional amount of muscle mass? It is known that contracting muscle transfers tension to skeletal elements through connective tissues, and it has already been demonstrated (Elashry et al., 2012) that MSTN null mice (mice with supra-muscular phenotype) present decreased connective tissue content, which can predispose them to injury. Another issue in the scientific community and sports organizations is the concern to control this type of high-tech doping in competitions. As yet there are no anti-doping methods available to screen and identify transgenic athletes, but some studies are underway to develop these (Bogani et al., 2011; Scarano et al., 2011; Carter, Flueck, 2012).
Thus, the aim of the present review is to explore the state of the art of MSTN cellular mechanisms, genetic variants and gene/pharmacological manipulation for therapeutic purposes and the implications of gene doping for athletes. Highwire and PubMed databases were consulted for this review using the following terms: gene doping, athletes, genes, gene therapy, myostatin and skeletal muscle.
MYOSTATIN BIOSYNTHESIS AND PATHWAY
In 1997, a group led by Se-Jin Lee at Johns Hopkins School of Medicine described MSTN for the first time as a negative regulator of muscle mass (McPherron et al., 1997) during screening for novel members of the Transforming Growth Factor Beta (TGF-β) in mammals. With the purpose of determining MSTN function, the authors disrupted the gene in null mutation mice, which then presented significant increase in muscle mass (hypertrophy and hyperplasia) when compared to a control group. MSTN mutations in cattle (Belgian Blue and Piedmontese) associated with the deletion of this protein also result in incredible hypertrophy (Buske et al., 2011).
MSTN circulates in the bloodstream and its gene encodes a small N-terminal sequence, which is followed by a pro-peptide region (latent region) that controls biochemical MSTN action, and a small C-terminal region (mature region) that makes the link to Activin Receptor II (ACTRIIB) (Lee, 2010). ACTRIIB has a transmembranic characteristic, belongs to the serine/treonine kinase family and phosphorylates activin receptor I (ACTRI), initiating a cascade of intracellular signals by regulatory proteins SMAD2 and SMAD3, which, when activated, are translocated to the nucleus, thus regulating gene transcription (Han, Mitch, 2011). When SMAD2 and 3 are phosphorylated, they promote MSTN action through muscle hypertrophy inhibition. MSTN blocks the growth of myoblasts and inhibits the expression of myogenic regulatory factors (MRFs), such as myoblast determination protein 1 (MYOD) (McFarlane et al., 2011). Moreover, classic studies show that MSTN negatively regulates the activation of satellite cells and self-renewal (McCroskery et al., 2004). In contrast, a recent study reported that inhibiting MSTN produces significant hypertrophy, but without early activation of satellite cells (Wang, McPherron, 2012).
NATURAL GENETIC VARIANTS
The first case of a human mutation in the MSTN gene was described in 2004 (Schuelke et al., 2004), when a German newborn presented extraordinary musculature (mainly in the arms and legs) compared to a child of the same age. By 4.5 years of age, the child was able to hold two/three kg dumbbells with his arms extended, and muscle hypertrophy was analyzed by ultrasound at six years of age, when the cross sectional area was also 2.5 times greater than that of a normal child. It is worth mentioning that the child's mother had previously been an athlete with considerable strength (Schuelke et al., 2004). A set of tests demonstrated that the child was clinically normal, and a molecular readout of the MSTN gene showed a mutation in the intron located between the exons 1 and 2, resulting in a premature termination codon (Schuelke et al., 2004). This finding was important in future studies to understanding the function of MSTN in the human organism and to developing potential therapies or studying the athletic phenotypes.
Another study reported a mutation in whippets that results in an athletic advantage associated with increased muscle mass in this group (Mosher et al., 2007). In this study, three groups of racing whippets were assessed: dogs who have two copies of the wild type allele (+/+), heterozygous with one wild type allele and one mutant allele (+/mh) and homozygous for the mutant allele (mh/mh). The authors found that 8 out of 12 heterozygous dogs and one homozygous mutant had the highest racing grade among 85 whippets analyzed. Meanwhile, 12 out of 72 wild-type individuals performed to this level. The heterozygous dogs were the fastest, thereby demonstrating the existence of an athletic phenotype for sprint performance, whereas the homozygous dogs presented the most robust muscle mass phenotype, similarly to other double muscling species.
In 2010, a genetic variant was identified for the first time in racehorses, which seems potentially to influence athletic performance (muscle power) (Hill et al., 2010) and there is evidence showing that thoroughbred horses have more muscle mass and present phenotypes related to increased strength in comparison to other horse species. The authors investigated the sequence variation of the MSTN gene, which contains three exons and spans 6,172 bp in chromosome 18 (Hill et al., 2010). Future functional genomics studies may reveal whether the mutation has a significant impact on MSTN gene expression and this finding may be a first step towards selecting athletes based on genetics and also to boosting performance through genetic manipulation.
GENE THERAPY AND MYOSTATIN INHIBITING DRUGS
Progress in molecular biology has enabled researchers to manipulate genes for therapeutic purposes. Human gene therapy is the transference of genetic material to human cells in order to treat or prevent diseases (Tilemann et al., 2012). Gene therapy uses genetic material, such as RNA and DNA, or even genetically modified cells (Kole et al., 2012; Gambari, 2012). It consists in artificial gene transfection to the organism, whereby the inserted gene ultimately leads to the increase and/or reduction in expression of one or more genes (Gaffney, Parisotto, 2007). The genetic material can be transferred by both direct (in vivo) and indirect (ex vivo) methods (Zhang et al., 2012; Gregory-Evans et al., 2012). The in vivo method consists in inserting the gene directly into the organism by subcutaneous, intramuscular, intravenous or intravascular injection (Zhang et al., 2012), while the ex vivo method consists of withdrawing cells from the patient, modifying them in the laboratory and reintroducing them into the patient. It seems that gene therapy will emerge as a promising way to treat some diseases, but some limitations associated with its adverse effects have to be addressed (Gregory-Evans et al., 2012).
In vivo injections of MSTN blockade antibody (JA16 neutralizing monoclonal antibody) in MDX mice increase both skeletal muscle hypertrophy and strength, and can be used as a therapeutic strategy (Bogdanovich et al., 2002). Another neutralizing MSTN antibody called MYO-029 was developed by Wyeth Pharmaceuticals to treat muscular dystrophies. In 2008, a clinical trial with MYO-029 was conducted on patients with different types of muscular dystrophies (Wagner et al., 2008). This "giant collaborative" project comprised a double-blind, placebo-controlled and randomized study with analysis of clinical, functional and serum parameters. Despite having no effect on muscle hypertrophy and function, treatment with MYO-029 proved to be quite safe in those patients. In another study, the contractile properties of single muscle fibers were evaluated using MYO-029 and muscle improvement occurred in four of the five patients treated, despite the fact that no quantitative improvement in muscle strength was observed (Krivickas et al., 2009).
An alternative strategy to the antibodies is a soluble version of the activin type II receptor (ACTRIIB/Fc), which induces dramatic muscle growth (Lee et al., 2010). Previous findings have shown, for example, that the injection of two other proteins (a control mouse monoclonal antibody and the myostatin propeptide fused to an Fc domain) had no effect compared to the PBS group (control). However, ACTRIIB/Fc caused growth of 32-40%, which was paralleled by an increase in the cross-sectional area of the gastrocnemius and plantaris muscles (Lee et al., 2005). In addition, this study found that the effect of the soluble receptor is attenuated but not eliminated in MSTN-/- mice, suggesting that at least one other ligand (GDF-11) in addition to MSTN normally functions to limit muscle growth.
Another elegant study reported that ACTRIIB/Fc treatment improved the success of human myoblast transplantation more than threefold, as evaluated by the number of human dystrophin-positive myofibers (Fakhfakh et al., 2012). In vitro experiments showed that this approach increased the differentiation of human myoblasts through an up-regulation of the regulatory genes responsible for myogenesis such as MyoD, resulting in myotube fusion and maturation. In order to better demonstrate the underlying mechanisms, forced swimming was performed and ACTRII2B treatment combined with exercise. Finally, it is important to point out that some clinical trials using ACTRIIB are already underway (Rahimov et al., 2011).
The protein follistatin was first identified as a single chain-polipeptide with weak inhibitory activity toward follicle-stimulating hormone secretion by anterior pituitary cells and was later demonstrated to be an activin-binding protein (Rodino-Kaplac et al., 2009). It is an MSTN binding protein that can inhibit MSTN and activin both in vitro and in vivo. However, due to its effects on the suppression of the hypothalamic-pituitary-gonadal axis, a spliced form of Follistatin was developed, which can be delivered by adeno associated virus (AAV) into muscles. Furthermore, no adverse effect has been detected using this approach (Kota et al., 2009). Another study described a powerful method using a one-time gene administration of MSTN inhibitor protein, which enhanced muscle mass and strength in normal and dystrophic mouse models for more than two years (Haidet et al., 2008).
Studies with RNA interference have come to the fore in recent years, and they consist in utilizing small fragments of RNA that bind to larger RNAs, promoting their inhibition (Kohler et al., 2011). Recently, it was reported that transgenic -zebra-fish receiving a microinjection of double-stranded biological RNA MSTN silencing presented hypertrophy and hyperplasia. RNAi for MSTN was also able to reduce MSTN mRNA and protein, increasing muscle mass and fiber size in vivo (Acosta et al., 2005).
A more recent study reported the use of antisense oligonucleotides to manipulate MSTN pre-RNA splicing and knock-down MSTN expression - this study provided proof
of principle of how MSTN knockout can be achieved by exon skipping (Kang et al., 2011). In comparison to virus mediated approaches, the use of these tools has advantages relating, for example, to the safety of uncontrolled insertion into the genome.
GENE DOPING AND PHYSICAL PERFORMANCE
According to World Anti-Doping Agency (WADA), gene doping is the non therapeutic manipulation of cells, genes, genetic elements or modulation of gene expression with the aim of increasing athletic ability (Gaffney, Parizzoto, 2007).
MSTN inhibition or deletion by gene therapy, drugs, genetic manipulation and mutations can result in hypertrophy and some studies have demonstrated enhancement of physical performance (Mosher et al., 2007; Lebrasseur et al., 2009; Santiago et al., 2011; Busquets et al., 2012). A question that remains to be answered is whether the increase in performance seen in animal models could be translated to humans. At the moment, the data show that MSTN plays a prominent role in muscle mass, but to associate this with improved performance is quite speculative.
In order to test the hypothesis that inhibition of MSTN preserves muscles from sarcopenia (Lenk et al., 2011), MSTN inhibition was investigated in old mice. Mice received an MSTN antibody (PF-354) and body composition, muscle weight and physical capacity were evaluated by means of various tests, including the time in treadmill until exhaustion test, ambulatory activity, grip strength and tetanic force. Glucose tolerance, insulin sensitivity, oxygen consumption and levels of protein expression and phosphorylation were also investigated to identify the mechanisms involved in the MSTN pathway under these experimental conditions. The anabolic effect of PF-354 was associated with a reduction in SMAD3 phosphorylation (LeBrasseur et al., 2009). Exercise training associated with PF-354 increased physical performance significantly, showing the potential impact of both strategies in counteracting sarcopenia.
Another elegant study published in the journal Science Translational Medicine demonstrated the efficiency of inhibiting MSTN in primates (Kota et al., 2009). It was developed a Follistatin isoform (AVV1-FS344) that prevents MSTN from binding to ACTRIIB receptors on muscle cells and induces increased muscle mass and strength, without affecting the morphology and function of major organs. Thus, AAV1-FS344 would be of interest to the athletic community, probably because it is safer than other methods of inhibiting MSTN.
Meanwhile, Morine et al. (2010) described a method in which they used adeno-associated virus vector (AVV) to over-express dominant negative MSTN exclusively on the liver of mice. This MSTN inhibition resulted in increased muscle mass and strength in control mice and in Duchenne muscle dystrophy models. The increase in muscle mass was pronounced in the soleus, quadriceps, tibialis anterior and extensor digitorum longus, although no effect on diaphragm and ventricular hypertrophy were observed (Morine et al., 2010). Transfection into the liver is an interesting strategy for inhibiting MSTN in a range of pathologies because transferring to the liver has a series of advantages compared to other experimental approaches. The use of adeno-associated virus (AVV) transfected to the liver requires only one application to cause considerable transgene expression, for example. Accordingly, the authors explain that this method would be efficient in large animals and is very difficult to detect, which could attract interest from athletes.
Some recent studies have demonstrated that MSTN can function not only as a modulator of muscle mass, but can also play a role in metabolism or even in endurance phenotype. It is well established that skeletal muscle has different fiber types which exhibit specific contractile and metabolic characteristics. In a well designed study, Candena et al. (2010) investigated the effects of a hypertrophy-inducing substance called ACE-031 (a soluble receptor form of activin IIB). In the soleus muscle, the total number (type I and II) of each fiber type was similar between the control and treated groups. The new finding was that both fast and slow fibers hypertrophied, providing evidence that blocking the MSTN pathway can improve strength, hypertrophy and resistance to fatigue concomitantly. This study was the first to demonstrate the versatility of this molecule not only in enhancing strength, but also in improving endurance capacity.
Another study reinforces the view that other phenotypes in addition to increased muscle mass might occur in skeletal muscle. MSTN null mice (MSTN-/-) display increased myofibre hypertrophy and hyperplasia, but compromised muscle force. In this study, the mice were subjected to swimming and treadmill exercise in order to evaluate morphological and molecular parameters (Matsakas et al., 2012). Exercise restored muscle function, increased muscle capillary density and the expression of key genes involved in oxidative metabolism. This finding is important because MSTN inhibition may induce growth of skeletal muscles without improving function, but exercise training may reprogram muscle to increase its strength, and this could be an attractive prospect for athletes.
Beyond skeletal muscle mass regulation, MSTN plays a potential role in the modulation of adipose tissue phenotype and metabolism. An interesting study demonstrated that pharmacological inhibition of ACTRIIB induced an increase in brown adipose tissue (BAT) through a fine-tuning mechanism that involves activation of myoglobin expression and PGC-1α in BAT. In addition, MSTN inhibition enhances mitochondrial function and uncoupled respiration, thereby increasing energy expenditure (Fournier et al., 2012). Another study found that suppressed ACTRIIB can treat diet-induced obesity and induce a brown fat-like thermogenic gene program in white adipose tissue (Koncarevic et al., 2012). The results of these studies can have major implications for sports that require a low percentage of adipose tissue and for the treatment of obesity.
CONCLUSION
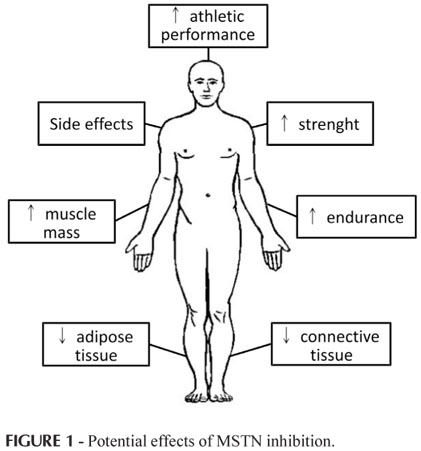
MSTN was discovered 15 years ago and, since then, important advances have been made in an attempt to develop outstanding therapeutic strategies (pharmacological or/and genetic manipulation). Increase in muscle mass is the most evident phenotype triggered by MSTN inhibition. Most of the elegant studies were carried out in animals and it is questionable both whether the findings observed in animals could be translated to humans, and whether MSTN inhibition results in improved physical performance, as demonstrated in Figure 1.
Beyond improving power/strength phenotypes in models of muscle dystrophies and other conditions, MSTN inhibition can potentially induce thermogenesis and fat loss. However, connective tissue is affected by this approach, and it is a major challenge to increase muscle mass while preserving connective tissue. At the same time, the development of these concepts raises the possibility of genetic manipulation in the world of sport. In this scenario, a good many athletes would no doubt subject themselves to this kind of illegal procedure, even bearing in mind that transfection may produce a number of adverse effects. Consequently, the organizations that regulate amateur and professional sports need to be prepared to discuss these issues.
Received for publication on 06th December 2011
Accepted for publication on 22nd May 2012
- ACOSTA, J.; CARPIO, Y.; BORROTO, I.; GONZALEZ, O.; ESTRADA, M.P. Myostatin gene silenced by RNAi show a zebrafish giant phenotype. J. Biotechnol, v.119, p.324-331, 2005.
- BAOUTINA, A.; ALEXANDER, I.E.; RASKO, J.E.J. Potential use of gene transfer in athletic performance enhancement. Mol. Ther., v.15, p.1751-1766, 2007.
- BISH, L.T.; SLEEPER, M.M.; FORBES, S.C.; MORINE, K.J.; REYNOLDS, C.; SINGLETARY, G..E.; TRAFNY, D.; PHAM, J.; BOGAN, J.; KORNEGAY, J.N.; VANDENBORNE, K.; WALTER, G..A.; SWEENEY, H.L. Long-term systemic myostatin inhibition via liver-targeted gene transfer in golden retriever muscular dystrophy. Hum. Gene. Ther., v.22, p.1499-1509, 2011.
- BOGANI, P.; SPIRITI, M.M.; LAZZARANO, S.; ARCANGELI, A.; BUIATTI, M.; MINUNNI, M. Transgene traceability in transgenic mice: a bioanalytical approach for potential gene-doping. Bioanalysis, v.3, p.2523-2531, 2011.
- BOGDANOVICH, S.; KRAG.; T.O.; BARTON, R.E.; MORRIS, L.D.; WHITTEMORE, L.A.; AHIMA, R.S.; KHURANA, T.S. Functional improvement of dystrophic muscle by myostatin blockade. Nature, v.420, p.418-421, 2002.
- BUSKE, B.; GENGLER, N.; SOYEURT, H. Short-communication: influence of the muscle hypertrophy mutation of the myostatin gene on milk production traits and milk fatty acid composition in dual-purpose Belgian Blue dairy cattle. J. Dairy Sci, v.94, p.3687-3692, 2011.
- BUSQUETS, S.; TOLEDO, M.; ORPÍ, M.; MASSA, D.; PORTA, M.; CAPDEVILLA, E.; PADILLA, N.; FRAILIS, V.; LÓPEZ-SORIANO, F.J.; HAN, H.Q.; ARGILÉS, J.M. Myostatin blockage using actRIIB antagonism in mice bearing in Lewis lung carcinoma results in the improvement of muscle wasting and physical performance. J. Cachex. Sarcopenia. Muscle, v.3, p.37-43, 2012.
- CANDENA, S.M.; TOMKINSON, K.N.; MONNELL, T.E.; SPAITS, M.S.; KUMAR, R.; UNDERWOOD, K.W.; PEARSALL, R.S.; LANCHEY, J.L. Administration of a soluble activin type II B receptor promotes skeletal muscle growth independent of fiber type. J. Appl. Physiol., v.109, p.635-642, 2010.
- CARTER, A.; FLUECK, M. A polymerase chain reaction-based methodology to detect gene doping. Eur. J. Appl. Physiol., v.112, p.1527-1536, 2012.
- CAVAZZANA-CALVO, M.; LAGRESLE, C.; HACEIN-BEY-ABINA, S.; FISCHER, A. Gene therapy for severe combined immunodeficiency. Annu. Rev. Med., v.56, p.585-602, 2005.
- ELASHRY, M.I.; COLLINS-HOOPER, H.; VAIYAPURI, S.; PATEL, K. Characterization of connective tissue from the hypertrophic skeletal muscle of myostatin null mice. J. Anatom., v.220, p.603-611, 2012.
- FOURNIER, B.; MURRAY, B.; GUTZWILLER, S.; MARCALETTI, S.; MARCELLIN, D.; BERGLING, S.; BRACHAT, S.; PERSOHN, E.; PIERREL, E.; BOMBARD, F.; HATAKEYAMA, S.; TRENDELENBURG, A.U.; MORVAN, F.; RICHARDSON, B.; GLASS, D.J.; LACH-TRIFIELIFF, E.; FEIGE, J.N. Blockade of the activin receptor IIB activates functional brown adipogenesis and thermogenesis by inducing mitochondrial oxidative metabolism. Mol. Cell. Biol., v.32, p.2871-2879, 2012.
- GAFFNEY, G.R.; PARISOTTO, R.P. Gene doping: a review of performance-enhancing genetics. Pediatr. Clin. North Am., v.54, p.807-822, 2007.
- GAMBARI, R. Alternative options for DNA-based experimental therapy of β-thalassemia. Expert Opin. Biol. Ther., v.12, p.443-462, 2012.
- GILSON, H.; SCHAKMAN, O.; KALISTA, S.; LAUSE, P.; TSUCHIDA, K.; THISSEN, J.P. Follistatin induces muscle hypertrophy through satellite cell proliferation and inhibition of both myostatin and activin. Am. J. Physiol. Endocrinol. Metab., v.297, p.E157-E164, 2009.
- GREGORY-EVANS, K.; BASHAR, A.M.; TAN, M. Ex vivo gene therapy and vision. Curr. Gene Ther., v.12, p.103-115, 2012.
- HAIDET, A.M.; RIZO, L.; HANDY, C.; UMAPATHI, P.; EAGLE, A.; SHILLING, C.; BOUE, D.; MARTIN, P.T.; SAHENK, Z.; MENDELL, J.R.; KASPAR, B.K. Long-term enhancement of skeletal muscle mass and strength by single gene administration of myostatin inhibitors. Proc. Natl. Acad. Sci., v.105, p.4318-4322, 2008.
- HAN, H.Q.; MITCH, W.E. Targeting the myostatin signaling pathway to treat muscle wasting diseases. Curr. Opin. Support Palliat. Care, v.5, p.334-341, 2011.
- HEINEKE, J.; AUGER-MESSIER, M.; XU, J.; SARGENT, M.; YORK, A.; WELLE, S.; MOLKETIN, J.D. Genetic deletion of myostatin from the heart prevents skeletal muscle atrophy in heart failure. Circulation, v.121, p.419-425, 2010.
- HILL, E.W.; GU, J.; EIVERS, S.S.; FONSECA, R.G.; MCGIVNEY, B.A.; GOVINDARAJAN, P.; ORR, N.; KATZ, L.M.; MACHUGH, D.E. A sequence polymorphism in MSTN predicts sprinting ability and racing stamina in thoroughbred horses. PloS One, v.5, e8645, 2010.
- KANG, J.K.; MALERBA, A.; POPPLEWELL, L.; FOSTER, K.; DICKSON, G. Antisense-induced myostatin exon skipping leads to muscle hypertrophy in mice following octa-guanodine morpholino oligomer treatment. Mol. Ther., v.19, p.159-164, 2011.
- KOHLER, M.; SCHANZER, W.; THEVIS, M. RNA interference for performance enhancement and detection in doping control. Drug. Test. Anal., v.3, p.661-667, 2011.
- KOLE, R.; KRAINER, A.R.; ALTMAN, S. RNA therapeutics: beyond RNA interference and antisense oligonucleotides. Nat. Rev. Drug Discov., v.11, p.125-140, 2012.
- KONCAREVIC, A.; KAJIMURA, S.; CORNWALL-BRADY, M.; ANDREUCCI, A.; PULLEN, A.; SAKO, D.; KUMAR, R.; GRINBERG, A. V.; LIHARSA, K.; UCRAN, J. A.; HOWARD, E.; SPIEGELMAN, B.M.; SEEHRA, J.; LACHEY, J. A novel therapeutic approach to treating obesity through modulation of TGFβ signaling. Endocrinology, v.153, p.3133-3146, 2012. May 1.
- KOTA, J.; HANDY, C.R.; HAIDET, A.M.; MONTGOMERY, C.L.; EAGLE, A.; RODINO-KLAPAC, L.R.; TUCKER, D.; CHILLING, C.J.; THERFALL, D.R.; WALKER, C.M.; WEISBRODE, S.E.; LANSSEN, P.M.L.; REED CLARCK, R.; SAHENK, Z.; MENDELL, J.R.; KASPAR, B.K. Follistatin gene delivery enhances muscle growth and strength in nonhuman primates. Sci. Transl. Med., v.1, n.6, 6ra15, 2009.
- KRIVICKAS, L.S.; WALSH, R.; AMATO, A.A. Single muscle fiber contractile properties in adults with muscular dystrophy treated with MYO-029. Muscle Nerve, v.39, p.2-9, 2009.
- LEBRASSEUR, N.K.; SHELHORN, T.M.; BERNARDO, B.L.; COSGROVE, P.G.; LORIA, P.M.; BROWN, T.A. Myostatin inhibition enhances the effect of exercise on performance and metabolic outcomes in aged mice. J. Gerontol. A. Biol. Sci. Med. Sci., v.64, p.940-948, 2009.
- LEE, S.J.; GLASS, D.J. Treating cancer cachexia to treat cancer. Skeletal Muscle, v.1, 2011. Availabe at: <http://www.skeletalmusclejournal.com/content/pdf/2044-5040-1-2.pdf>. Acessed on: 20 Sep. 2011.
- LEE, S.J. Extracellular regulation of myostatin: a molecular rheostat for muscle mass. Immunol. Endocr. Metab. Agents Med. Chem., v.10, p.183-194, 2010.
- LEE, S.J.; LEE, Y.S.; ZIMMERS, T.A.; SOLEIMANI, A.; MATZUK, M.M.; TSUCHIDA, K.; COHN, R.D.; BARTON, E.R. Regulation of muscle mass by follistatin and activins. Mol. Endocrinol., v.24, p.1998-2008, 2010.
- LEE, S.J.; REED, L.A.; DAVIES, M.V.; GIRGENRATH, S.; GOAD, M.E.; TOMKINSON, K.M.; WRIGHT, J.F.; BARKER, C.; EHRMANTRAUT, C.; HOLMSTROM, J.; TROWELL, B.; GERTZ, B.; JIANG, M.S.; SEBALD, S.M.; MATZUK, M.; LI, E.; LIANG, L.F.; QUATTLEBAUM, E.; STOTISH, R.L.; WOLFMAN, N.M. Regulation of muscle growth by multiple ligands signaling through activin type II receptors. Proc. Natl. Acad. Sci., v.102, p.18117-18122, 2005.
- LENK, K.; SCHULER, G.; ADAMS, V. Skeletal muscle wasting in cachexia and sarcopenia: molecular pathophysiology and impact of exercise training. J. Cachex. Sarcopenia Muscle, v.1, p.9-21, 2010.
- MANSOUR, M.M.; AZZAZY, H.M. The hunt for gene dopers. Drug Test Anal., v.1, p.311-322, 2009.
- MATSAKAS, A.; MACHARIA, R.; OTTO, A.; ELASHRY, M.I.; MOUISEL, E.; ROMANELLO, V.; SARTORI, R.; AMTHOR, H.; SANDRI, M.; NARKAR, V.; PATEL, K. Exercise training attenuates the hypermuscular phenotype and restores skeletal muscle function in myostatin null mouse. Exp. Physiol., v.97, p.125-140, 2012.
- MCCROSKERY, S.; THOMAS, M.; MAXWELL, L.; SHARMA, M.; KAMBADUR, R. Myostatin negatively regulates satellite cell activation and self-renewal. J. Cell. Biol., v.162, p.1135-1147, 2003.
- MCFARLANE, C.; HUI, G..Z.; AMANDA, W.Z.; LAU, H.Y.; LOKIREDDY, S.; XIAOJIA, G.; MBOULY, V.; BUTLER-BROWNE, G.; GLUCKMAN, P.D.; MCKANNA, T.A.; TORIELLO, H.V. Gene doping: the hype and the harm. Ped. Clin. North Am., v.57, p.719-727, 2010.
- MCPHERRON, A.C.; LEE, S.J. Double muscling in cattle due to mutations in the myostatin gene. Proc. Natl. Acad. Sci., v. 9423, p.12457-12461, 1997.
- MCPHERRON, A.C.; LAWLER, A.M.; LEE, S.J. Regulation of skeletal muscle mass in mice by a new TGF-beta superfamily member. Nature, v.387, p.83-90, 1997.
- MORINE, K.J.; BISH, L.T.; PENDRAK, K.; SLEEPER, M.M.; BARTON, E.R.; SWEENEY, H.L. Systemic myostatin inhibition via liver-targeted gene transfer in normal and distrophic mice. PLoS One, v.5, e9176, 2010.
- MOSHER, D.S.; QUIGNON, P.; BUSTAMANTE, C.D.; SUTTER, N.B.; MELLERSH, C.S.; PARKER, H.G.; OSTRANDER, E.A. A mutation in the myostatin gene increases muscle mass and enhances racing performance in heterozygote dogs. PLoS Genet., v.3, e79, 2007.
- MURPHY, K.T.; KOOPMAN, R.; NAIM, T.; LÉGER, B.; TRIEU, J.; IBEBUNJO, C.; LYNCH, G..S. Antibody-directed myostatin inhibition in 21-mo-old mice reveals novel roles for myostatin signaling in skeletal muscle structure and function. FASEB J., v.24, p.4433-4442, 2010.
- OLIVEIRA, R.S.; COLLARES, T.S.; SMITH, K.R.; COLLARES, T.V.; SEIXAS, F.K. The use of genes for performance enhancement: doping or therapy? Braz. J. Med. Biol Res., v.44, p.1194-1201, 2011.
- RAHIMOV, F.; KING, O.D.; WARSING, L.C.; POWELL, R.E.; EMERSON, C.P.; KUNKEL, L.M.; WAGNER, K.R. Gene expression profiling of skeletal muscles treated with a soluble activin type IIB receptor. Physiol. Genomics., v.43, p.398-407, 2011.
- RAPER, S.; CHIRMULE, S.; LEE, F.S.; WIVEL, N.A.; BAGG, A.; GAO, G.P. Fatal systemic inflammatory response syndrome in a ornithine transcarbamylase deficient patient following adenovirus gene transfer. Mol. Genet. Metab., v.80, p.248-158, 2003.
- RODINO-KAPLAC, L.R.; HAIDET, A.M.; KOTA, J.; HANDY, C.; KASPAR, B.K.; MENDELL, J.R. Inhibition of myostatin with emphasis on follistatin as a therapy for muscle disease. Muscle Nerve, v.39, p.283-296, 2009.
- SANTIAGO, C.; RUIZ, J.R.; RODRÍGUEZ-ROMO, G..; FIUZA-LUCES, C.; YVERT, T.; GONZALEZ-FREIRE, M.; GÓMEZ-GALLEGO, F.; MORÁN, M.; LUCIA, A. The K153R polymorphism in the myostatin gene and muscle power phenotypes in young, non-athletic men. PloS One, v.6, e16323, 2011.
- SCARANO, S.; ERMINI, M.L.; SPIRITI, M.M.; MASCINI, M.; BOGANI, P.; MINUNNI, M. Simultaneous detection of transgenic DNA by surface plasmon resonance imaging with potential application to gene doping detection. Anal. Chem., v.83, p.6245-6353, 2011.
- SCHUELKE, M.; WAGNER, K.R.; STOLZ, L.E.; HÜBNER, C.; RIEBEL, T.; KÖMEN, W.; BRAUN, T.; TOBIN, J.F.; LEE, S.J. Myostatin mutation associated with gross muscle hypertrophy in a child. N. Engl. J. Med., v.350, p.2682-2688, 2004.
- SHARMA, M.; KAMBADUR, R. Human myostatin negatively regulates human myoblast growth and differentiation. Am. J. Physiol. Cell Physiol., v.301, p.C195-C203, 2011.
- TILEMANN, L.; ISHIKAWA, K.; WEBER, T.; HAJJAR, R.J. Gene therapy for heart failure. Circ. Res., v.110, p.777-793, 2012.
- WAGNER, K.R.; FLECKENSTEIN, J.L.; AMATO, A.A.; BAROHN, R.J.; BUSHBY, K.; ESCOLAR, D.M.; FLANIGAN, K.M.; PESTRONK, A.; TAWIL, R.; WOLFE, G.I.; EAGLE, M.; FLORENCE, J.M.; KING, W.M.; PANDYA, S.; STRAUB, V.; JUNEAU, P.; MEYERS, K.; CSIMMA, C.; ARAUJO, T.; ALLEN, R.; PARSONS, S.A.; WOZNEY, J.M.; LAVALLIE, E.R.; MENDELL, J.R.. A phase I/II trial of MYO-029 in adult subjects with muscular dystrophy. Ann. Neurol., v.63, p.561-571, 2008.
- WANG, Q.; MCPHERRON, A. Myostatin inhibition induces muscle fibre hypertrophy prior to satellite cell activation. J. Physiol., v.590, p.2151-2165, 2012.
- WELLE, S.; MEHTA, S.; BURGESS, K. Effect of postdevelopmental myostatin depletion on myofibrillar protein metabolism. Am. J. Physiol. Endocrinol. Metab., v.300, p.E993-E1001, 2011.
- ZHANG, Y.; SATTERLEE, A.; HUANG, L. In vivo gene delivery by nonviral vectors: overcoming hurdles? Mol. Ther., v.20, n.7, p.1298-1304, 2012.
- ZIMMERS, T.A.; DAVIES, M.V.; KONIARIS, L.G.; HAYNES, P.; ESQEULA, A.F.; TOMIKINSON, K.N.; MCPHERNON, A.C.; WOLFMAN, N.M.; LEE, S.J. Induction of cachexia in mice by systemically administered myostatin. Science, v.29, p.1486-1488, 2002.
Publication Dates
-
Publication in this collection
18 Oct 2012 -
Date of issue
Sept 2012
History
-
Received
06 Dec 2011 -
Accepted
22 May 2012