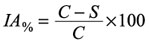Abstracts
In order to treat hyperpigmentation-related problems, there has been a global trend in developing cosmetics claiming to have skin-whitening properties, which act by inhibiting melanin biosynthesis. The objective of this work was to evaluate the in vitro mushroom tyrosinase inhibitory activity of five Amazonian native flora oils, and so to verify the possibility of their incorporation into cosmetic products. In addition, the fatty acid composition of the essential oils was determined by gas chromatography-flame ionisation detection in order to determine the main components of these oils. The tyrosinase inhibitory activity of the tested oils was found to be in the following order: açaí (IA50 = 66.08 µg mL-1) > tucumã > patauá > pracaxi > castanha do Brasil. This study suggests that açaí oil has great potential in the treatment of hyperpigmentation and other related disorders, due to its considerable tyrosinase inhibitory activity.
Amazonian plants; Amazonian plants; Skin lightening agents; Tyrosinase; Tyrosinase; Melanin; Hyperpigmentation
Com o intuito de se tratar problemas dermatológicos de hiperpigmentação, há uma tendência mundial no desenvolvimento de cosméticos que possuam propriedades despigmentantes, os quais agem inibindo a biossíntese de melanina. O objetivo deste trabalho foi avaliar in vitro a atividade de inibição da tirosinase de cogumelo de cinco óleos de plantas nativas da Amazônia e, desta forma, verificar a possibilidade de sua incorporação em produtos cosméticos. Ainda, a composição de ácidos graxos dos óleos foi determinada por cromatografia gasosa com detecção por ionização de chama, no intuito de determinar os principais componentes destes óleos. A atividade de inibição da tirosinase dos óleos testados foi encontrada na seguinte ordem: açaí (IA50 = 66,08 µg mL-1) > tucumã > patauá > pracaxi > castanha do Brasil. Este estudo sugere que o óleo de açaí possui grande potencial para o tratamento da hiperpigmentação cutânea e doenças correlatas, devido à sua considerável atividade de inibição da tirosinase.
Plantas amazônicas; Plantas amazônicas; Despigmentantes; Tirosinase; Tirosinase; Melanina; Hiperpigmentação cutânea
ARTICLE
Mushroom tyrosinase inhibitory activity and major fatty acid constituents of Amazonian native flora oils
Raquel da Silva TeixeiraI; Paula Rafaela RochaI; Hudson Caetano PoloniniI, II; Marcos Antônio Fernandes BrandãoI, II; Maria das Graças Afonso Miranda ChavesII; Nádia Rezende Barbosa RaposoI, II
INIQUA, Federal University of Juiz de Fora
IINUPICS, Federal University of Juiz de Fora
Correspondence Correspondence: N. R. B. Raposo Universidade Federal de Juiz de Fora, Campus Universitário Rua José Lourenço Kelmer, s/n, São Pedro 36036-900 - Juiz de Fora - MG, Brazil Tel/fax: 55 32 3229 3809 E-mail: nadiafox@gmail.com
ABSTRACT
In order to treat hyperpigmentation-related problems, there has been a global trend in developing cosmetics claiming to have skin-whitening properties, which act by inhibiting melanin biosynthesis. The objective of this work was to evaluate the in vitro mushroom tyrosinase inhibitory activity of five Amazonian native flora oils, and so to verify the possibility of their incorporation into cosmetic products. In addition, the fatty acid composition of the essential oils was determined by gas chromatography-flame ionisation detection in order to determine the main components of these oils. The tyrosinase inhibitory activity of the tested oils was found to be in the following order: açaí (IA50 = 66.08 µg mL-1) > tucumã > patauá > pracaxi > castanha do Brasil. This study suggests that açaí oil has great potential in the treatment of hyperpigmentation and other related disorders, due to its considerable tyrosinase inhibitory activity.
Uniterms: Amazonian plants/evaluation/dermatological use. Skin lightening agents. Tyrosinase/inhibition/in vitro evaluation. Melanin. Hyperpigmentation/treatment.
RESUMO
Com o intuito de se tratar problemas dermatológicos de hiperpigmentação, há uma tendência mundial no desenvolvimento de cosméticos que possuam propriedades despigmentantes, os quais agem inibindo a biossíntese de melanina. O objetivo deste trabalho foi avaliar in vitro a atividade de inibição da tirosinase de cogumelo de cinco óleos de plantas nativas da Amazônia e, desta forma, verificar a possibilidade de sua incorporação em produtos cosméticos. Ainda, a composição de ácidos graxos dos óleos foi determinada por cromatografia gasosa com detecção por ionização de chama, no intuito de determinar os principais componentes destes óleos. A atividade de inibição da tirosinase dos óleos testados foi encontrada na seguinte ordem: açaí (IA50 = 66,08 µg mL-1) > tucumã > patauá > pracaxi > castanha do Brasil. Este estudo sugere que o óleo de açaí possui grande potencial para o tratamento da hiperpigmentação cutânea e doenças correlatas, devido à sua considerável atividade de inibição da tirosinase.
Unitermos: Plantas amazônicas/avaliação/uso dermatológico. Despigmentantes. Tirosinase/inibição/avaliação in vitro. Melanina. Hiperpigmentação cutânea/tratamento.
INTRODUCTION
The major determinant of the colour of mammalian skin and hair is the content and distribution in keratinocytes of the melanins, coloured polymorphous biopolymers produced from complex and multi-step oxidative reactions involving the amino acid L-tyrosine (Adhikari et al., 2008; Lin et al., 2011). This pigment acts primarily by defending animals from ultraviolet (UV) radiation. However, its abnormal production is related to an increase in the number of melanocytes and/or increased activity of melanogenic enzymes. This can result in considerable aesthetic problems due to hyperpigmentation, namely freckles, melasma, liver spots, age spots and others (Adhikari et al., 2008).
In order to treat hyperpigmentation-related problems, there has been a global trend in developing cosmetics claiming to have skin-whitening properties, which act by inhibiting melanin biosynthesis (Lin et al., 2011). This can be achieved by protecting the skin from solar light exposure, removing melanin by corneal ablation, blocking or diminishing the proliferation of melanocytes or inhibiting tyrosinase activity, since melanin is derived from tyrosine and its biosynthesis is to a large extent regulated by this copper-containing enzyme (Wang et al., 2006; Ha et al., 2005).
Current research is focused on both synthetic and natural sources for reducing melanin formation (Arung et al., 2011; Park et al.., 2011). With respect to natural substances, the Amazon biome is a major source of new species and therefore molecules that may have pharmacological activity, skin-whitening included. In this light, the objective of this work was to evaluate the mushroom tyrosinase inhibitory activity of five Amazonian native flora oils, and so to verify the possibility of their incorporation into cosmetic products. In addition, the fatty acid composition of the essential oils was determined by gas chromatography in order to determine the main components of these oils.
MATERIAL AND METHODS
Plant material
Vegetable oils of Astrocaryum vulgare Mart. (Arecaceae, popular name tucumã), Bertholletia excelsa Bonpl. (Lecythidaceae, popular name castanha do Brasil). Euterpe oleraceae M. (Areaceae, popular name açaí) Oenocarpus bataua Mart. (Arecaceae, popular name patauá) and Pentaclethra macroloba (Willd.) Kuntze (Mimosaceae, popular name pracaxi) were acquired from the Amazonian oil industry (Pará, Brazil).
Tyrosinase inhibition: qualitative enzymatic reaction screening
The assay was performed according to the protocol described by Macrini et al. (2009), with some modifications. Aliquots of 10 µL of a solution composed of 125 U mL-1 of mushroom tyrosinase (Sigma-Aldrich, USA) were added to 96-well microplates, and then 70 µL of pH 6.8 phosphate buffer solution and 60 µL of the Amazonian native flora oils (350 µg mL-1, in 25% dimethyl sulphoxide, DMSO) were also added. For the positive control, 60 µL of kojic acid (17.5 µg mL-1 in 25% DMSO) was used instead of the Amazonian oil, and for the negative control, 60 µL of 2.5% DMSO was added. To the resultant mixture, 70 µL of L-tyrosine (Sigma-Aldrich, USA) were added at a concentration of 0.3 mg mL-1 in distilled water (the final volume in the wells was 210 µL).
The absorbance of the microplate wells was read using a microplate spectrophotometer (SpectraCount, Packard, USA) at 510 nm (T0). Then, the microplates were incubated at 30 ± 1ºC for 60 min and the absorbance was measured again (T1). An additional incubation period of 60 min at 30 ± 1ºC was done and, after this period, a new spectrophotometric reading was taken (T2).
The inhibitory percentage at the two time points (T1 and T2) was obtained according to the formula:
where IA% = inhibitory activity; C = negative control absorbance; S = sample or positive control absorbance (absorbance at time T1 or T2 minus the absorbance at time T0).
Tyrosinase inhibition: quantitative enzymatic reaction assay
For the samples that reached an IA% greater than 35%, a quantitative assay was conducted. For this purpose, the above experimental protocol was followed, with modifications (a 500 U mL-1 tyrosinase solution was used instead of 125 U mL-1 and the optical densities were measured every 10 min for 1 h).
The quantitative determination was obtained through an analytical curve and the respective equation of the line. For this, the oils were diluted in the microplate wells to concentrations of 100, 80, 40, 20 and 10 µg mL-1 with 25% DMSO, and the kojic acid was diluted to concentrations of 10, 5, 2.5, 1.25 and 0.625 µg mL-1. The analytical curve was plotted between tyrosinase inhibition activity percentages at each time point and the concentrations of the oils/ positive control. Using the equation of the line, the inhibitory activity at 50% (IA50, in µg mL-1) was calculated.
Fatty acids assay
For essential oil lipid hydrolysis, 10 mg of the oils were dissolved in 100 µL of a mixture of ethanol and 1 mol L-1 potassium hydroxide (95:5, v/v) in 2 mL cryogenic tubes. After 10 s of vigorous mechanical agitation, the oils were hydrolysed in a microwave (Panasonic, Japan), using 80 W of power for 5 min; they were then cooled and the following reagents were added to the tubes, in this order: 400 µL of 20% hydrochloric acid, one spatula tip of sodium chloride and 600 µL of ethyl acetate. Sample were agitated for a further 10 s, followed by a 5 min rest period. A 300 µL aliquot from the organic layer was transferred to a microcentrifuge tube and dried in an oven in order to obtain the free fatty acids. The residues were methylated with 14% boron trifluoride in methanol and heated in a water bath for 10 min at 80 ºC. The resulting solution was injected into an HP5890 gas chromatograph (HP, USA) with a flame ionisation detector. The instrument was equipped with a 15 m x 0.25 mm HP-INNOWax column, 1 µL injection (1/50 split) with hydrogen as the carrier gas (2 mL min-1). The temperature of both the detector and the injector was maintained at 250 ºC, and the column was subjected to a temperature gradient (initial = 80 ºC, then an increase of 7 ºC min-1 to 240 ºC). The identification of the peaks was made by comparison with Supelco 37 methylated fatty acids (Supelco, USA) using the same conditions described above.
Statistical analysis
All data were reported as mean (n=3). The comparison between the values obtained with the different oils and the reference drug (kojic acid) was achieved through a Student's t-test to calculate p-values (α = 0.05), using the software Statistical Package for the Social Sciences® (SPSS) v. 14.0.
RESULTS AND DISCUSSION
Oils from plants are increasingly gaining more attention from pharmacists, physicians and chemists due to their great scope of applications in different fields such as cosmetics, perfumery, pharmaceuticals and the food industry. Since they have a wide range of pharmacological applications due to their highly complex chemical composition, currently, there is a global trend toward replacing synthetic substances with natural extracts (Al-Mamary et al., 2011).
One of the abilities of plant materials currently being researched is the inhibition of the tyrosinase enzyme, but these studies have mainly been focused on crude drugs or plant extracts, and not on plant oils (Adhikari et al., 2008). Since the Brazilian Amazon is renowned worldwide for its great plant diversity and potential for new pharmaceuticals, this study selected five well-known Amazonian species for screening their capability in being used as raw materials for skin-whitening agents, which are sought after by the cosmetic market.
According to Momtaz et al. (2008), vegetable oils are rich in compounds containing hydrophobic components, which can act as competitive inhibitors for the enzyme tyrosinase, and so on melanin synthesis. According to these authors, plant oils have great reducing power, this being the principle for exploring the tyrosinase inhibitory potential of plants, which may also have phytoconstituents that chelate the copper ions present in the active site of the tyrosinase enzyme. It is also important to discover new skin-whitening agents because some of the currently used substances are characterised by high toxicity, as hydroquinone (Macrini et al., 2009).
To our knowledge, this is the first report of such activity for these species, and all of the five Amazonian vegetable oils screened for their tyrosinase inhibition activity showed some degree of activity, as can be seen in Table I. The tyrosinase inhibitory activity of tested oils was found to be in the following order: açaí > tucumã > patauá > pracaxi > castanha do Brasil. Thus, the natural oil obtained from the açaí was the most active in the present study, as it had an IA50 close to kojic acid, the gold standard for this activity.
Our methodology included two screenings and a final quantitative step. The tyrosinase concentration of 125 U mL-1 in the first test was adopted to increase the chances of discovering an oil with potential skin-whitening activity. As all oils seemed to possess such activity (p < 0.05), they were submitted to the second screening, and as they continued to show some activity, they were all subjected to the quantitative analysis in order to discover which was the most effective and therefore has the greatest potential for the cosmetic/pharmaceutical industry.
According to Table I, it can be noted that the oils maintained their 60 min activity in the second screening, and so one can infer that they did not lose their tyrosinase inhibition activity. Conversely, kojic acid went from 73.13% inhibition in the first hour to 42.77% in the second hour. Açaí oil, with a good IA50, had the longest duration of action, compared to kojic acid, which may be a desirable characteristic for a skin-whitening product as it decreases the need for constant reapplication of the product to the skin.
In order to know the composition of the studied oils, a gas chromatography analysis was also performed, as can be seen in Table II and Figure 1. The results show that the oils possessed both saturated and unsaturated fatty acids, the latter being considered essential fatty acids. This, coupled with the tyrosinase inhibition results, led to the inference that they can be included in functional foods and cosmetics.
Theoretically, one can imagine that the high skin-whitening property found in açaí oil may be due to the presence of oleic acid (C18:1) in its composition, since this was the main component in the chromatographic analysis. However, further studies must be conducted with its components separately in order to determine which specific compound is responsible for enzyme inhibition. However, it has been noted that natural products generally exert their activity by synergy between their various compounds, and a single substance is generally not responsible for the biological effects found in natural products.
CONCLUSIONS
This study suggests that açaí oil has great potential for the treatment of hyperpigmentation and other related disorders due to its considerable tyrosinase inhibitory activity. Thus, this may be an interesting candidate for evaluation in more complex biological assays such as melanocyte cultures and eventually in animal/human assays. However, for the raw material be effectively incorporated into the cosmetic market, it is now required to develop an adequate delivery system/formulation for its topical use in humans and for this activity be assessed in animal cells.
ACKNOWLEDGEMENTS
This research was supported by FAPEMIG, CNPq, CAPES and PROPESQ.
Received for publication on 10th February 2012
Accepted for publication on 3rd May 2012
- ADHIKARI, A.; DEVOTKA, H.P.; TAKANO, K.; NAKANE, T.; BASNET, P.; SKALKO-BASNET, N. Screening of Nepalese crude drugs traditionally used to treat hyperpigmentation: in vitro tyrosinase inhibition. Int. J. Cosm. Sci., v.30, p.353-360, 2008.
- AL-MAMARY, M.; ABDELWAHAB, S.I.; AL-GHALIBI, S.; AL-GHASANI, E. The antioxidant and tyrosinase inhibitory activities of some essential oils obtained from aromatic plants grown and used in Yemen. Sci. Res. Essays, v.6, n.34, p.6840-6845, 2011.
- ARUNG, E.T.; SHIMIZU, K.; KONDO, R. Artocarpus plants as a potential source of skin whitening agents. Nat. Prod. Commun., v.6, n.9, p.1397-1402, 2011.
- HA, S.K.; KOKETSU, M.; LEE, K.; CHOI, S.Y.; PARK, J.H.; ISHIHARA, H.; KIM, S.Y. Inhibition of tyrosinase activity by N,N-unsubstituted selenourea derivatives. Biol. Pharm. Bull., v.28, p.838-840, 2005.
- LIN, V.C.; DING, H.; KUO, S.; CHIN, L.; WU, J.; CHANG, T. Evaluation of in vitro and in vivo depigmenting activity of raspberry ketone from Rheum officinale. Int. J. Mol. Sci., v.12, n.8, p.4819-4835, 2011.
- MACRINI, D.J.; SUFFREDINI, I.B.; VARELLA, A.D.; YOUNES, R.N.; OHARA, M.T. Extracts from Amazonian plants have inhibitory activity against tyrosinase: an in vitro evaluation. Braz. J. Pharm. Sci., v.45, n.2, p.715-21, 2009.
- MOMTAZ, S.; MAPUNYA, B.M.; HOUGHTON, P.J.; EDGERLY, C.; HUSSEIN, A.; NAIDOO, S.; LALL, N. Tyrosinase inhibition by extracts and constituents of Sideroxylon inerme L. stem bark, used in South Africa for skin lightening. J. Ethnopharmacol, v.119, n.3, p.507-512, 2008.
- PARK, S.A.; OH, T.; KIM, S.S.; KIM, J.E.; LEE, S.J.; LEE, N.H. Constituents with tyrosinase inhibitory activities from branches of Ficus erecta var. sieboldii King. J. Enzyme. Inhib. Med. Chem, v.27, n.3, p.390-394, 2011.
- WANG, K.H.; LIN, R.; HSU, F.L.; HUANG, Y.H.; CHANG, H.C.; HUANG, C.Y.; LEE, M.H. Cosmetic applications of selected traditional Chinese herbal medicines. J. Ethnopharmacol, v.106, p.353-359, 2006.
Publication Dates
-
Publication in this collection
18 Oct 2012 -
Date of issue
Sept 2012
History
-
Received
10 Feb 2012 -
Accepted
03 May 2012