Abstracts
Biological assays that have been performed on different types of Brazilian propolis have shown that type 6 propolis (G6) has a strong antimicrobial activity and a low flavonoid content. This study aimed to evaluate the correlation between the phenolic composition and the biological activities displayed by propolis G6 from the state of Bahia and green propolis, also known as type 12 (G12). The values of the flavonoids and the total phenolics in propolis G6 were different than those in propolis G12. Although the G12 variety presented greater antioxidant activity, propolis G6 proved to have greater antimicrobial and cytotoxic activities. The results showed that the phenolic compounds may not be the only compounds responsible for the biological activity. More detailed studies of the chemical composition and an assessment of the biological activity are required to establish the quality of propolis.
Brazilian própolis; Flavonoids; Phenolic compounds; Propolis
Ensaios biológicos realizados com diferentes tipos de própolis brasileira têm mostrado que a própolis do tipo 6 (G6) tem forte atividade antimicrobiana e menor teor de flavonóides. Este trabalho teve como objetivo avaliar a correlação entre a composição fenólica e atividades biológicas apresentadas pela própolis G6, do Estado da Bahia, e a própolis verde, do tipo 12 (G12). Os teores de flavonóides e fenólicos totais na própolis G6 foram diferentes dos teores da própolis G12. Apesar da G12 apresentar maior atividade antioxidante, a própolis G6 apresenta maior atividade antimicrobiana e citotóxica. Os resultados mostraram que os compostos fenólicos não são os únicos compostos responsáveis pela atividade biológica da própolis. Estudos mais específicos da composição química, em adição à avaliação das atividades biológicas, são requeridos para determinar a qualidade da própolis.
Própolis brasileira; Flanovóides; Compostos fenólicos; Própolis
ARTICLE
The correlation between the phenolic composition and biological activities of two varieties of Brazilian propolis (G6 and G12)
Ingridy Simone Ribeiro CabralI; Tatiane Luiza Cadorin OldoniII; Severino Matias de AlencarIII; Pedro Luiz RosalenIV; Masaharu IkegakiV
ICenter of Nuclear Energy in Agriculture, University of São Paulo
IIDepartment of Chemistry, Federal Technological University of Paraná
IIIDepartment of Agri-Food Industry, Food and Nutrition, "Luiz de Queiroz" College of Agriculture, University of São Paulo
IVDepartment of Physiological Sciences, Piracicaba Dental School, University of Campinas
VFaculty of Pharmaceutical Sciences, Federal University of Alfenas
Correspondence Correspondence: M. Ikegaki Faculdade de Ciências Farmacêuticas, Universidade Federal de Alfenas Rua Gabriel Monteiro da Silva, 700. Centro 37130-000 - Alfenas MG, Brasil E-mail: masaharu.ikegaki@unifal-mg.edu.br
ABSTRACT
Biological assays that have been performed on different types of Brazilian propolis have shown that type 6 propolis (G6) has a strong antimicrobial activity and a low flavonoid content. This study aimed to evaluate the correlation between the phenolic composition and the biological activities displayed by propolis G6 from the state of Bahia and green propolis, also known as type 12 (G12). The values of the flavonoids and the total phenolics in propolis G6 were different than those in propolis G12. Although the G12 variety presented greater antioxidant activity, propolis G6 proved to have greater antimicrobial and cytotoxic activities. The results showed that the phenolic compounds may not be the only compounds responsible for the biological activity. More detailed studies of the chemical composition and an assessment of the biological activity are required to establish the quality of propolis.
Uniterms: Brazilian própolis/evaluation. Flavonoids/determination in propolis. Phenolic compounds/determination in propolis. Propolis/quality control.
RESUMO
Ensaios biológicos realizados com diferentes tipos de própolis brasileira têm mostrado que a própolis do tipo 6 (G6) tem forte atividade antimicrobiana e menor teor de flavonóides. Este trabalho teve como objetivo avaliar a correlação entre a composição fenólica e atividades biológicas apresentadas pela própolis G6, do Estado da Bahia, e a própolis verde, do tipo 12 (G12). Os teores de flavonóides e fenólicos totais na própolis G6 foram diferentes dos teores da própolis G12. Apesar da G12 apresentar maior atividade antioxidante, a própolis G6 apresenta maior atividade antimicrobiana e citotóxica. Os resultados mostraram que os compostos fenólicos não são os únicos compostos responsáveis pela atividade biológica da própolis. Estudos mais específicos da composição química, em adição à avaliação das atividades biológicas, são requeridos para determinar a qualidade da própolis.
Unitermos: Própolis brasileira/avaliação. Flanovóides/determinação no própolis. Compostos fenólicos/determinação no própolis. Própolis/controle de qualidade.
INTRODUCTION
Propolis is a resinous substance with a complex chemical composition that is collected from various species of plants by Apis mellifera bees, which results in a material of different colors and consistencies (Guisalberti, 1979; Burdock, 1998; Castaldo, Capasso, 2002; Alencar et al., 2007).
Propolis has been used in folk medicine for centuries. Pharmacological properties such as antimicrobial, antioxidant (Oldoni et al., 2011; Cabral et al., 2009; Alencar et al., 2007), anticarcinogenic (Duarte et al., 2006; Castro et al., 2007; Castro et al., 2009) and anti-inflammatory (Park et al., 1998) activities have been described for propolis.
Because propolis is widely used, it has become necessary to establish quality criteria for this product. The total content of flavonoids and phenolic compounds are among the criteria used to evaluate the quality of propolis extracts intended for national and international trade. These criteria are described in the Regulation of Identity and Quality of Propolis that was proposed by the Agriculture Ministry (Instruction Number 3; January 19, 2001). In crude propolis, the minimum required content of phenolic compounds is 5% (w/w), and the minimum for flavonoids is 0.5% (w/w) (Brazil, 2001). The absorption spectrum in the ultraviolet and visible region is also evaluated and must have the "characteristic peaks" between 200 and 400 nm of the major classes of flavonoids.
Different varieties of Brazilian propolis were classified into 12 types according to the chemical profile and the evaluation of antimicrobial and antioxidant activities. The propolis types that showed increased biological activities were types 3 (from Rio Grande do Sul), 6 (from Bahia) and 12 (from Minas Gerais) (Park et al., 2000). Propolis type 12, known as green propolis, is the most popular type sold in the Japanese market, and the composition of type 12 includes flavonoids and cinnamic acid derivatives (Park et al., 2002). However, other authors found that Bahia propolis, classified as type 6, has a distinct chemical composition with a low content of flavonoids and a greater amount of apolar compounds such as fatty acids and prenylated benzophenone (Duarte et al., 2006; Castro et al., 2007; Castro et al., 2009). Additionally, type 6 propolis shows a high level of antimicrobial activity (Castro et al., 2007). Therefore, this study aimed to verify whether there is positive correlation between the identity and quality criteria for propolis and the biological activities exhibited by the extracts of propolis.
MATERIAL AND METHODS
Propolis samples
Crude samples of bee (Apis mellifera) propolis were obtained in Jan Feb, 2008. Type 6 was obtained from the Atlantic forest in the state of Bahia, which is located in the northeastern region of Brazil (SL11º56'31" and WL38º05'04", city of "Entre Rios") (Park et al., 2000). Type 12 was obtained in the state of Minas Gerais, which is located in the southeast region of Brazil (SL21º25'40.56" and WL45º56'46.84", city of "Alfenas") (Park et al., 2000). The propolis samples were maintained at 4 ºC in black polyethylene bags.
Preparation of ethanolic extracts
The preparation of the ethanolic extracts of propolis (EEPs) was based on the methodology described by Ikegaki (2001). Briefly, 2 g of propolis were weighed, and 15 mL of 80% ethanol (v/v) was added to each sample. The samples were placed in a water bath at 70 ºC for 30 min. The mixture was then centrifuged, and the procedure was repeated, using a second extraction of 10 mL 80% ethanol (v/v). The supernatants from the two extractions were mixed and transferred to a glass tube. The EEP was analyzed for chemical composition and biological activity.
Spectrophotometric analysis
The determination of the absorption spectrum was carried out according to the methodology described by Ikegaki (2001). An aliquot of 25 mL of each EEP was diluted with 30 mL of 95% ethanol (v/v) to determine the absorption spectrum in the region in the UV-Vis region from 200 to 400 nm.
Total phenolic compounds
The analysis of the total phenolic compounds was carried out according to the spectrophotometric method described by Folin-Ciocalteau (Woisky; Salatino, 1998). Gallic acid was used as a standard. Extract solutions (0.5 mL) at a concentration of 0.8% (w/v) were mixed with 2.5 mL of Folin-Ciocalteau reagent (1:10) and 2.0 mL of 4% Na2CO3 (w/v). The absorbance was measured at 740 nm after 2 hours of incubation in the dark at room temperature. A control study was conducted under the same conditions. The content of the total phenolic compounds in the samples was determined based on the standard curve of gallic acid. The Folin-Ciocalteau method was validated, and the limits of detection (LOD) and limits of quantification (LOQ) were 2.14 µg/mL and 6.49 µg/mL, respectively. The results of the content of total phenols are expressed as gallic acid equivalents (mg GA/g of propolis).
Total flavonoid content
The total flavonoid content of the EEPs was determined according to the method described by Park et al. (1995) and Alencar et al. (2007). An aliquot of each EEP (0.5 mL) at a concentration of 8% (w/v) was mixed with 4.3 mL of 80% ethanol (v/v), 0.1 mL of 10% Al(NO3)3 (w/v) and 0.1 mL of 1 M potassium acetate. A control series was prepared in parallel, and ethanol was added instead of aluminum nitrate. After 40 minutes at room temperature, the absorbance was measured at 415 nm. The total flavonoid content was calculated as quercetin (mg/g) equivalents from a calibration curve.
Reversed-phase high performance liquid chromatography (RP-HPLC)
The analysis of the ethanolic extracts of propolis by reversed-phase high performance liquid chromatography (HPLC-RP) was performed according to the methodology described by Ikegaki (2001). A 20 mL aliquot of each extract was injected into a reverse phase column REGIS ODS (250 x 4.6 mm), and the column was then installed into a liquid chromatography system (Shimadzu Co.) composed of two LC-6AD pumps, a degasser DGU-2A, a SIL-10AF autosampler, a photodiode array detector SPD-M10Avp, a fraction collector FRC-10A and a column oven CTO-10ASvp maintained at 35 ºC. The mobile phase consisted of water/acetic acid (19:1, v/v) (solvent A) and methanol (solvent B) with a solvent flow rate of 1 mL/min. The initial solvent concentration was 30% solvent B, and the gradient was increased up to 40% B in 15 minutes, 50% B in 30 minutes, 60% B in 45 minutes, 75% B in 65 minutes, 85% B in 85 minutes, and 90% B in 95 minutes and finally decreased to 30% B in 105 minutes. In this study, we investigated the following authentic standards of phenolic acids and flavonoids (Extrasynthese Co.): quercetin, kaempferol, apigenin, pinocembrine, chrysin, acacetin, galangin, canferide, isosacuranetin, sacuranetin, ramnetin, isoramnetin, rutin, gallic acid, p-coumaric acid, cinnamic acid and ferulic acid.
Susceptibility test
The antimicrobial activity of the EEPs was determined as the minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) against Staphylococcus aureus ATCC 6538. Isolated colonies were suspended in 4.5 mL of sterile 0.9% NaCl until a turbidity with an absorbance of approximately 0.102 to 660 nm was reached, which is equivalent to a bacterial growth on the agar surface of 2 x 107 CFU/mL. The MIC was determined according to a technique described by Duarte et al. (2003). A volume of 1.4 mL of the bacterial suspension was inoculated into 140 mL of nutrient broth to obtain a bacterial concentration of 2 x 105 CFU mL. After homogenization, a volume of 4,950 mL of inoculated culture medium was placed in each tube. A volume of 50 µL of ethanolic extract of propolis (final concentrations ranging from 320 µg/mL to 5 µg/mL) was added to each tube, resulting in a final volume of 5 mL. A tube with the inoculated culture medium and 50 µL of 80% ethanol (v/v) served as a control. Then, the tubes were agitated and incubated at 37 ºC for 24 h. After incubation, the tubes were agitated and the absorbance was measured in a spectrophotometer at 660 nm. The MIC was defined as the lowest concentration range of EEP in which no visible bacterial growth occurred.
Based on the results obtained in the MIC test, the tubes that showed inhibitory activity were used as inoculum suspensions. The nutrient agar plates were inoculated using 50 mL of inoculum and subsequently incubated at 37ºC for 24 h. The MBC was defined as the lowest concentration of EEP in which no cell growth occurred on the surface of the inoculated medium.
Free radical-scavenging activity on DPPH
The effect of DPPH radical scavenging was carried out according to Brand-Williams et al. (1995). The reaction mixture contained 0.5 mL of the ethanolic extract of propolis at a concentration of 90 µg/mL, 3 mL of ethanol P.A. and 0.3 mL of 0.5 mM DPPH radical (1,1-diphenyl-2-picrilidrazil) ethanolic solution. The mixture was incubated for 45 minutes at room temperature and in the dark. The absorbance was recorded at 517 nm in a spectrophotometer. The radicalscavenging activity was determined as the antioxidant activity (AA), which was calculated using the rate of decline in absorbance of the DPPH solution, samples and standards after 45 minutes of reaction (steady state) relative to the reference solution (DPPH in ethanol), according to the formula:
% Antioxidant activity = 100 - [(Asample- Ablank)*100)/Acontrol]
where:
Asample = absorbance of DPPH solution (samples)
Ablank = absorbance of the solution of the samples without the addition of DPPH
Acontrol = absorbance of the reference solution of DPPH (ethanol)
Cytotoxic activity determination in vitro
The HeLa tumor cell line was cultivated in DMEM (Dulbecco's Modified Eagle's Medium - Gibco/Life Technologies) culture medium, supplemented with 10% (v/v) fetal bovine serum (Cultilab, Campinas, SP) and incubated at 37 ºC with an atmosphere of 5% CO2 and 95% O2. Penicillin and streptomycin (Amresco) were used as antibiotics. After trypsinization, the cells were suspended in DMEM containing 10% (v/v) fetal bovine serum, distributed into 24-well plates at a density of 2.5 x 104 cells/well and incubated at 37ºC under the same conditions described above. After reaching 60-70% confluence, the cells were exposed to the vehicle or EEP at various concentrations for a period of 24 h or 48 h. After the exposure time, 100 µL of methyl thiazolyl tetrazolium chloride solution (MTT) (5 mg /mL in PBS) was added to the each well, and the cells were incubated for another 3 h. The culture medium was removed, the plate was washed two times with PBS, and 200 µL of isopropanol solution acidified in 0.04 M HCl was added. Cellular viability was determined by the absorbance at 560 nm with reference to 655 nm (Mosmann, 1983). The data obtained in the cell proliferation and cytotoxicity assays were analyzed using the GraphPad Prism program (GraphPad Software, San Diego, CA).
Statistical analysis
Statistical analysis of the results was performed using the SAS 9.2 software. Analysis of variance (ANOVA) and Tukey's test were applied to detect significant differences between mean values (p <0.05).
RESULTS AND DISCUSSION
For many years, Brazilian green propolis prevailed in the Brazilian propolis market. The botanical origin of Brazilian green propolis is rosemary, Baccharis dracunculifolia (Park et al., 2002). Because of the popularity of green propolis, the quality parameters established both by the market and by official organizations referred to this type of propolis. The establishment of quality parameters made the Brazilian propolis trade increase. Because the primary market is in Japan, propolis trade has brought in foreign currency and has contributed significantly to the development of apiculture in Brazil.
Brazil has a large diversity of vegetation from which bees can collect resins, which causes great chemical diversity among the propolis collected in different regions and in different seasons. The biological properties of propolis are related to its chemical composition, and it became necessary to classify the different types of propolis found in Brazil. Park et al. (2000) ranked most prevalent types of propolis in Brazil in 12 groups or types. The green propolis has been classified as group 12 (G12), and the propolis of Bahia has been classified as group 6 (G6).
The organoleptic characteristics, which are intrinsic to the botanical origin of propolis, may represent obstacles to the acceptance and consumption of propolis. The G6 and G12 propolis types have been sensorially analyzed. The G6 propolis was brown with a hard consistency and a woody odor that is similar to certain types of wine. The G12 propolis was green in color with a hard consistency and had the characteristic odor of propolis. The ethanol extracts showed the same differences in their color and odor characteristics when compared.
In addition to the organoleptic characteristics, other parameters are considered important for determining propolis quality. According to the Technical Regulation of Identity and Quality of the Propolis set forth by the Agriculture Ministry (Brazil, 2001), the ultraviolet and visible spectrum absorption, the content of total flavonoids and phenolic compounds are considered important in determining propolis quality.
Phenolic compounds generally exhibit an absorption peak in the ultraviolet light range of 250 and 350 nm during spectrophotometric analysis (Mabry et al., 1970). In our study, we examined the spectroscopic profiles of different propolis samples. The G6 propolis showed the highest peak absorbance at a wavelength of 280 nm, while the G12 propolis presented the highest peak absorbance at 306 nm. The absorption maximum of G6 was significantly lower than the absorption of G12 propolis, indicating a lower phenolic compound concentration (Figure 1).
The total phenolic and flavonoid contents also varied between the samples. The G6 propolis had a phenolic content of 14.8 mg GA/g of propolis (1.48%) and a flavonoid content of 0.32%. The G12 propolis showed a phenolic content of 169.6 mg GA/g of propolis (16.96%) and a flavonoid content of 6.15%. The minimum requirements according to the Regulation of Identify and Quality of Propolis are 5% (w/w) for phenolic compounds and 0.5% (w/w) for flavonoids (Brazil, 2001). The contents for total phenolics and flavonoids for propolis G6 are lower than those required by Brazilian regulations and are also lower than those reported in the literature for propolis from temperate climates (Ahn et al., 2004; Kumazawa et al., 2004). Despite the low concentrations of these chemical compounds, propolis G6 showed biological activity, which suggests that chemicals other than phenolics and flavonoids in the propolis possess similar biological activities. Therefore, propolis G6 cannot be discriminated against by any legislation because of its low phenolic and flavonoid contents.
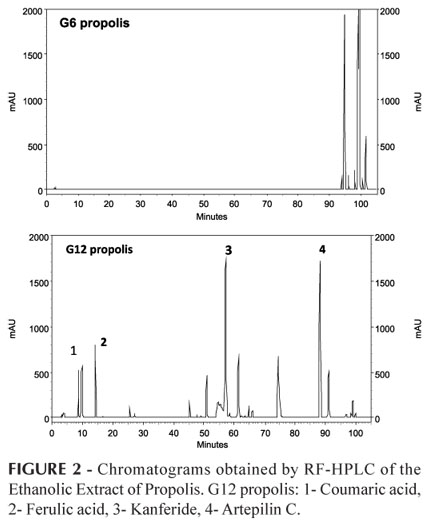
These results were confirmed by the analysis of HPLC chromatograms. It was observed that propolis G6 has a chemical profile with a few apolar peaks. Based on the phenolic and flavonoid standards, it was not possible to identify any particular substance. However, the G12 propolis showed a profile with a greater variety of substances of different polarities. We were able to identify certain compounds, including cinnamic acid, ferulic acid, kanferide, and Artepilin C. Peaks 3 (kanferide) and 4 (artepilin-C) made up the majority of the composition of G12 (Figure 2).
Artepilin C has demonstrated several biological activities, including anti-inflammatory properties (Paulino et al., 2008), the inhibition of cell proliferation and angiogenesis (Keshavarz et al., 2009), antibacterial and antitumor properties (Salatino et al., 2005) and antioxidant properties (Shimizu, 2004). Castro et al. (2009) isolated and identified a prenylated benzophenone, Hyperibone A, from propolis G6. This compound has demonstrated high antimicrobial activity and cytotoxicity against HeLa tumor cells.
Our results confirmed that G6 propolis does not reach the minimum criteria required by the Regulation in any of the parameters. This propolis did not show an absorption spectrum with characteristic peaks because of the low flavonoid and phenolic compound contents. G12 propolis showed a higher flavonoid and total phenolic compound content, and a characteristic absorption spectrum. These results show that the green propolis has excellent quality according to the Technical Regulation (Instruction Nº 3 of January 19, 2001).
With regard to the biological activities of the propolis samples, we observed that the G6 propolis had more effective antimicrobial and cytotoxic activities. The MIC results showed that both types of propolis were able to inhibit the growth of S. aureus ATCC 6538; however, G6 propolis showed much higher antimicrobial activity compared to G12 propolis. The minimum inhibitory concentration of propolis G6 was between 5 and 10 µg/mL, while G12 propolis showed values between 40 and 80 µg/mL. Only G6 showed bactericidal activity, with a minimum bactericidal concentration between 160 and 320 µg/mL. In the antioxidant activity studies (Figure 3), G12 propolis showed higher DPPH radical-scavenging activity (53.0%) than G6 propolis (21.7%). This result was expected because phenolic compounds are known to have antioxidant properties (Park et al., 2002), and G12 propolis presented higher levels of these compounds, as described above.
According to Figure 4, G6 propolis showed higher cytotoxicity to HeLa tumor cells than G12 propolis at a concentration of 200 µg/mL. At this concentration, cell viability was less than 25% for the G6 propolis and greater than 75% for G12 propolis. With these results, it was possible to trace the cell viability curve for different concentrations of G6 propolis. The IC50 value obtained was 60 µg/mL, which is the propolis concentration necessary to reduce the number of tumor cells by 50% in this experiment.
The correlations between phenolic compounds and antioxidant activity and between the antioxidant activity and the flavonoid content were positive, indicating that phenolic compounds and flavonoids have an important role in the antioxidant activity of propolis extracts. This result corroborates previously published data (Park et al., 2002). However, different substances may be involved in the biological activities that we analyzed (antimicrobial and cytotoxic), including Hyperibone A (Castro et al., 2009), because we observed that G6 propolis, which has the lowest phenolic compound and flavonoid contents, showed the best antibacterial and cytotoxic effects.
Silva et al. (2006) established the correlation between the levels of phenolics and flavonoids with antioxidant and antimicrobial activities of 49 samples of Brazilian propolis. They demonstrated that the level of these compounds does not directly reflect the biological activity of the propolis extracts. They also suggested that tests of the biological activities should be included in the quality control of the propolis extracts.
The colorimetric method of determining total flavonoids is not an accurate method because the bathochromic effect of the absorption caused by complexation with aluminum is not uniform for all flavonoids. The chelation between aluminum and flavonoids occurs differently between flavonoids, and an increased degree of hydroxylation of the nucleus leads to an increase in the bathochromic effect, which causes the spectra to shift toward longer wavelengths (Jurdi, Geissman, 1995; Oldoni, 2007). Thus, depending on the composition of the flavonoids, the results can be under- or overestimated.
Ito et al. (2001) observed that the propolis classified as group 1 (G1) from Bage - RS showed anti-HIV activity in in vitro assays. However, Ikegaki (2001) analyzed six samples from the same city and found that there are no flavonoids present in the composition of this propolis and that the characteristic peaks are absent in the spectrophotometric analysis.
Because propolis is a product of animal origin and must be registered as food in the Agriculture Ministry, it is appropriate that quality control does not include the examination of biological activities. However, propolis is used by people for therapeutic purposes and not as a food. According to Castaldo and Capasso (2002), propolis has been used for centuries to relieve symptoms of many diseases, and several studies are published every year in which the biological activity of propolis is evaluated. However, the levels of phenolic compounds and flavonoids are not always measured or, when they are evaluated, the correlation between these compounds and biological activity is negative (Cabral, 2008; Li et al., 2008; Awale et al., 2008).
It would be interesting to establish chemical markers for propolis through the isolation and identification of biologically active compounds. These studies would allow people to accurately assess the quality of Brazilian propolis and to determine the types of propolis that are appropriate for medical use. This research might cause other varieties of propolis to be considered of good quality, which could stimulate beekeepers in different regions of the country to produce propolis and increase their income and the Brazilian market share of propolis.
CONCLUSION
It was established that propolis G12 has a high content of total flavonoid and phenolic compounds, which gives it an excellent quality according to the Technical Regulation of Identity and Quality of Propolis from the Agriculture Ministry. However, propolis G6, which did not reach the minimum levels of phenolics and flavonoids required, shows bacteriostatic and bactericidal efficiency and is cytotoxic to HeLa tumor cells. The results of this study demonstrate that the concentration of phenolics and flavonoids does not directly reflect all of the biological activities of propolis extracts.
Received for publication on 19th August 2011
Accepted for publication on 18th June 2012
- AHN, M.R.; KUMAZAWA, S.; HAMASAKA, T.; BANG, K.S.; NAKAYAMA, T. Antioxidant activity and constituents of propolis collected in various areas of Korea. J. Agric. Food Chem., v.52, n.1, p.7286-7292, 2004.
- ALENCAR, S.M.; OLDONI, T.L.C.; CASTRO, M.L.; CABRAL, I.S.R.; COSTA-NETO, C.M.; CURY, J.A.; ROSALEN, P.L.; IKEGAKI, M. Chemical composition and biological activity of a new type of Brazilian propolis: red propolis. J. Ethnopharmacol, v.113, n.1, p.278-283, 2007.
- AWALE, S.; LI, F.; ONOZUKA, H.; ESUMI, H.; TEKUZA, Y, KADOTA, S. Constituents of Brazilian red propolis and their preferential cytotoxic activity against human pancreatic PANC-1 cancer cell line in nutrient-deprived condition. Bioorg. Med. Chem., v.16, n.1, p.181-189, 2008.
- BANKOVA, V.; POPOV, S.; MAREKOV, N.L. Isopentenyl cinnamates from poplar buds and propolis. Phytochem, v.28, n.3, p.871-873, 1989.
- BRAND, W.; CUVELIER, M.E.; BERSET, C. Use of a free radical method to evaluate antioxidant activity. LWT - Food Sci. Technol., v.28, n.1, p.25-30, 1995.
- BRASIL. Ministério da Agricultura, Pecuária e do Abastecimento. Instrução Normativa n.3, de 19 de janeiro de 2001. Regulamentos técnicos de identidade e qualidade de apitoxina, cera de abelha, geléia real, geléia real liofilizada, pólen apícola, própolis e extrato de própolis. Diário Oficial da União, Brasília, 23 Jan. 2001a. Seção I, p.18-23.
- BURDOCK, G.A. Review of the biological properties and toxicity of bee propolis. Food Chem.Toxicol, v.36, n.4, p.347-363, 1998.
- CABRAL, I.S.R. Isolamento e identificação de compostos com atividade antibacteriana da própolis vermelha brasileira. Piracicaba, 2008. 94 p. [Dissertation of Master degree. "Luiz de Queiroz" College of Agriculture, University of São Paulo]
- CABRAL, I.S.R.; OLDONI, T.L.C.; PRADO, A.; BEZERRA, R.M.N.; ALENCAR, S.M.; IKEGAKI, M.; ROSALEN, P.L. Composição fenólica, atividade antibacteriana e antioxidante da própolis vermelha brasileira. Quím. Nova, v.32, n.1, p.1523-1527, 2009.
- CASTALDO, S.; CAPASSO, S. Propolis, an old remedy used in modern medicine. Fitoterapia, v.73, n.1, p.S1-S6, 2002.
- CASTRO, M.L.; CURY, J.A.; ROSALEN, S.M.; IKEGAKI, M.; DUARTE, S.; KOO, H. Própolis do sudeste e nordeste do Brasil: influência da sazonalidade na atividade antibacteriana e composição fenólica. Quím. Nova, v.30, n.7, p.1512-1516, 2007.
- CASTRO, M.L.; NASCIMENTO, A.M.; IKEGAKI, M.; COSTA-NETO, C.M.; ALENCAR, S.M.; ROSALEN, P. Identification of a bioactive compound isolated from Brazilian própolis type 6. Bioorg. Med. Chem., v.17, n.14, p.5332-5335, 2009.
- CHEN, C.N.; WU, C.L.; SHY, H.S.; LIN, J.K. Cytotoxic Prenylflavanones from Taiwanese propolis. J. Nat. Prod., v.66, n.4, p.503-506, 2003.
- DUARTE, S.; KOO, H.; BOWEN, W.H.; HAYACIBARA, M.F.; CURY, J.A; IKEGAKI, M.; PARK, Y.K.; ROSALEN, P.L. Effect of a novel type of propolis and its chemical fractions on glucosyltransferases and on growth and adherence of mutans streptococci. Biol. Pharm. Bull., v.26, n.4, p.527-531, 2003.
- DUARTE, S.; ROSALEN, P.L.; HAYACIBARA, M.F.; CURY, J.A.; BOWEN, W.H.; MARQUIS, R.E.; REHDER, V.L.; SARTORATTO, A.; IKEGAKI, M.; KOO, H. The influence of a novel propolis on mutans streptococci biofilms and caries development in rats. Arch. Oral Biol, v.51, n.1, p.15-22, 2006.
- GHISALBERTI, E.L. Propolis: a review. Bee World, v.60, n.2, p.59-84, 1979.
- IKEGAKI, M. Determinação de qualidade de propolis de Apis mellifera africanizada da região sul do Brasil: avaliação de algumas propriedades físico-químicas e biológicas da própolis. Campinas, 2001. 74 p. [Thesis of PhD degree. Faculty of Pharmaceutical Sciences. University of São Paulo]
- ITO, J.; CHANG, F.R.; WANG, H.K.; PARK, Y.K.; IKEGAKI, M.; KILGORE, N.; LEE, K.H. Anti-AIDS agents. 48. Anti-HIV activity of moronic acid derivatives and the new melliferone-related triterpenoid isolated from brazilian propolis. J. Nat. Prod., v.64, n.1, p.1278-1281, 2001.
- JURD, L.; GEISSMAN, T.A. Absorption spectra of metal complexes of flavonoid compounds. J. Org. Chem, v.21, n.12, p.1395-1401, 1956.
- KESHAVARZ, M.; MOSTAFAIE, A.; MANSOURI, K.; SHAKIBA, Y.; MOTLANGH, H.R. Inhibition of corneal neovascularization with propolis extract. Arch. Med. Res., v.40, n.1, p.59-61, 2009.
- KUMAZAWA, S.; HAMASAKA, T.; NAKAYAMA, T. Antioxidant activity of propolis of various geographic origins. Food Chem. v.84, n.1, p.329-339, 2004.
- LI, F.; AWALE, S.; TEKUZA, Y.; KADOTA, S. Cytotoxic constituents from Brazilian red propolis and their structure-activity relationship. Bioorg. Med. Chem, v.16, n.10, p.181-189, 2008.
- MABRY, T.J.; MARKHAM, K.R.; THOMAS, M.B. The systematic identification of flavonoids Berlin: Springer-Verlag, 1970. 354 p.
- MOSMANN, T. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J. Immunol. Methods, v.65, n.1, p.55-63, 1983.
- OLDONI, T.L.C. Isolamento e identificação de compostos com atividade antioxidante de uma nova variedade de própolis brasileira produzida por abelhas da espécie Apis mellifera Piracicaba, 2007. 104 p. [Dissertation of Master degree. "Luiz de Queiroz" College of Agriculture, University of São Paulo]
- OLDONI, T.L.C.; CABRAL, I.S.R; REGITANO-d'ARCE, M.A.B.R.; ROSALEN, P.L.; IKEGAKI, M.; NASCIMENTO, A.M.; ALENCAR, S.M. Isolation and analysis of bioactive isoflavonoids and chalcone from a new type of brazilian propolis. Sep. Pur. Technol, v.77, n.2, p.208-213, 2011.
- PAULINO, N.; ABREU, S.R.L.; UTO, Y.; KOYAMA, D.; NAGASAWA, H.; HORI, H.; DIRSCH, V.M.; VOLLMAR, A.M.; SREMIN, A.; BRETZ, W.A. Anti-inflammatory effects of a bioavailable compound, Artepillin C, in Brazilian propolis. Eur. J. Pharmacol., v.587, n.1, p.296-301, 2008.
- PARK, Y.K.; ALENCAR, S.M.; AGUIAR, C.L. Botanical origin and chemical composition of brazilian propolis. J. Agric. Food Chem, v.50, n.9, p.2502-2506, 2002.
- PARK, Y.K.; IKEGAKI, M.; ALENCAR, S.M.; MOURA, F.F. Evaluation of brazilian propolis by both physicochemical methods and biological activity. Honey Bee Sci, v.21, n.2, p.85-90, 2000.
- PARK, Y.K.; KOO, M.H.; SATO, H.H. Estudo de alguns componentes da própolis coletada por Apis mellifera no Brasil. Arq. Biol. Tecnol, v.38, n.1, p.1253-1259, 1995.
- SALATINO, A.; TEIXEIRA, E.W.; NEGRI, G.; MESSAGE, D. Origin and chemical variation of brazilian propolis. E-CAM, v.2, n.1, p.33-38, 2005.
- SFORCIN, J.L.; FERNANDES JR., A.; LOPES, C.A.M.; BANKOVA, V.; FUNARI, S.R.C. Seasonal effect on brazilian propolis antibacterial activity. J. Ethnopharmacol, v.73, n.1, p.243-249, 2000.
- SHIMIZU, K. Antioxidative bioavailability of artepillin C in brazilian propolis. Arch. Biochem. Biophys., v.424, n.1, p.181-188, 2004.
- SILVA, J.F.M.; SOUZA, M.C.; MATTA, S.R.; ANDRADE, M.R.; VIDAL, V.N. Correlation analysis between phenolic levels of Brazilian propolis extracts and their antimicrobial and antioxidant activities. Food Chem, v.99, n.1, p.431-435, 2006.
- WOISKY, R.G.; SALATINO, A. Analysis of propolis: some parameters and procedures for chemical quality control. J. Apicult. Res., v.37, n.2, p.99-105, 1998.
Publication Dates
-
Publication in this collection
18 Oct 2012 -
Date of issue
Sept 2012
History
-
Received
19 Aug 2011 -
Accepted
18 June 2012