Abstracts
The aim of this study was to prepare enzymatic hydrolysates from whey protein concentrate with a nutritionally adequate peptide profile and the ability to inhibit angiotensin-converting enzyme (ACE) activity. The effects of the type of enzyme used (pancreatin or papain), the enzyme:substrate ratio (E:S ratio=0.5:100, 1:100, 2:100 and 3:100) and the use of ultrafiltration (UF) were investigated. The fractionation of peptides was performed by size-exclusion-HPLC, and the quantification of the components of the chromatographic fractions was carried out by a rapid Corrected Fraction Area method. The ACE inhibitory activity (ACE-IA) was determined by Reverse Phase-HPLC. All parameters tested affected both the peptide profile and the ACE-IA. The best peptide profile was achieved for the hydrolysates obtained with papain, whereas pancreatin was more advantageous in terms of ACE-IA. The beneficial effect of using a lower E:S ratio on the peptide profile and ACE-IA was observed for both enzymes depending on the conditions used to prepare the hydrolysates. The beneficial effect of not using UF on the peptide profile was observed in some cases for pancreatin and papain. However, the absence of UF yielded greater ACE-IA only when using papain.
Whey proteins; Protein hydrolysates; Peptide profile; Inhibitory activity; Angiotensin-converting enzyme
O objetivo deste estudo foi preparar hidrolisados enzimáticos do concentrado proteico do soro de leite com perfil peptídico nutricionalmente adequado e com capacidade para inibir a atividade da enzima conversora da angiotensina (ECA). Os efeitos do tipo de enzima usado (pancreatina ou papaína), da relação enzima:substrato (E:S=0,5:100, 1:100, 2:100 e 3:100) e do uso da ultrafiltração (UF) foram investigados. O fracionamento dos peptídeos foi feito por CLAE de exclusão molecular e a quantificação dos componentes das frações cromatográficas foi realizada pelo método da Área Corrigida da Fração. A atividade inibitória da ECA (AI-ECA) foi determinada por CLAE de fase reversa. Todos os parâmetros testados afetaram tanto o perfil peptídico quanto a AI-ECA. O melhor perfil peptídico foi atingido para os hidrolisados obtidos com papaína, enquanto a pancreatina foi mais vantajosa em termos da AI-ECA. O efeito benéfico do uso de menor relação E:S sobre o perfil peptídico e a AI-ECA foi observado para ambas as enzimas dependendo das condições usadas para o preparo dos hidrolisados. O efeito benéfico da ausência da UF sobre o perfil peptídico foi observado em alguns casos para pancreatina e papaína. No entanto, a ausência da UF produziu maior AI-ECA somente quando a papaína foi usada.
Proteínas do soro de leite; Hidrolisados proteicos; Perfil peptídico; Atividade inibitória; Enzima conversora de angiotensina
ARTICLES
Analysis of whey protein hydrolysates: peptide profile and ACE inhibitory activity
Marialice Pinto Coelho SilvestreI,II,* * Correspondence: M.P.C. Silvestre. Edetec Food Industry S/A. Av. José Cândido da Silveira, 2100, 31035-536 - Belo Horizonte - MG, Brazil. E-mail: malice@ufmg.br ; Mauro Ramalho SilvaI,II; Viviane Dias Medeiros SilvaII; Mariana Wanessa Santana de SouzaII; Carlos de Oliveira Lopes JuniorII; Wendel de Oliveira AfonsoII
IFaculty of Pharmacy, Federal University of Minas Gerais
IIEdetec Food Industry S/A
ABSTRACT
The aim of this study was to prepare enzymatic hydrolysates from whey protein concentrate with a nutritionally adequate peptide profile and the ability to inhibit angiotensin-converting enzyme (ACE) activity. The effects of the type of enzyme used (pancreatin or papain), the enzyme:substrate ratio (E:S ratio=0.5:100, 1:100, 2:100 and 3:100) and the use of ultrafiltration (UF) were investigated. The fractionation of peptides was performed by size-exclusion-HPLC, and the quantification of the components of the chromatographic fractions was carried out by a rapid Corrected Fraction Area method. The ACE inhibitory activity (ACE-IA) was determined by Reverse Phase-HPLC. All parameters tested affected both the peptide profile and the ACE-IA. The best peptide profile was achieved for the hydrolysates obtained with papain, whereas pancreatin was more advantageous in terms of ACE-IA. The beneficial effect of using a lower E:S ratio on the peptide profile and ACE-IA was observed for both enzymes depending on the conditions used to prepare the hydrolysates. The beneficial effect of not using UF on the peptide profile was observed in some cases for pancreatin and papain. However, the absence of UF yielded greater ACE-IA only when using papain.
Uniterms: Whey proteins. Protein hydrolysates. Peptide profile. Inhibitory activity. Angiotensin-converting enzyme.
RESUMO
O objetivo deste estudo foi preparar hidrolisados enzimáticos do concentrado proteico do soro de leite com perfil peptídico nutricionalmente adequado e com capacidade para inibir a atividade da enzima conversora da angiotensina (ECA). Os efeitos do tipo de enzima usado (pancreatina ou papaína), da relação enzima:substrato (E:S=0,5:100, 1:100, 2:100 e 3:100) e do uso da ultrafiltração (UF) foram investigados. O fracionamento dos peptídeos foi feito por CLAE de exclusão molecular e a quantificação dos componentes das frações cromatográficas foi realizada pelo método da Área Corrigida da Fração. A atividade inibitória da ECA (AI-ECA) foi determinada por CLAE de fase reversa. Todos os parâmetros testados afetaram tanto o perfil peptídico quanto a AI-ECA. O melhor perfil peptídico foi atingido para os hidrolisados obtidos com papaína, enquanto a pancreatina foi mais vantajosa em termos da AI-ECA. O efeito benéfico do uso de menor relação E:S sobre o perfil peptídico e a AI-ECA foi observado para ambas as enzimas dependendo das condições usadas para o preparo dos hidrolisados. O efeito benéfico da ausência da UF sobre o perfil peptídico foi observado em alguns casos para pancreatina e papaína. No entanto, a ausência da UF produziu maior AI-ECA somente quando a papaína foi usada.
Unitermos: Proteínas do soro de leite. Hidrolisados proteicos. Perfil peptídico. Atividade inibitória. Enzima conversora de angiotensina.
INTRODUCTION
Whey is currently a major by-product of the modern cheese and casein production industries and represents an important source of environmental pollution due to its large volume, production rate and high organic matter content (Román et al., 2009). This dairy stream also represents an excellent source of functional proteins and peptides, which have helped to transform whey from a waste material to a valuable dairy stream for use in the agri-food, biotechnology, medical and related markets (Smithers, 2008).
The use of whey in natura is limited due to its perishable nature and the excessive dilution of its components. However, certain membrane separation technologies can be used to partially concentrate the whey, giving rise to whey protein concentrate (WPC), which consists of 35-80% protein (Brans et al., 2004).
Enzymatic hydrolysis of WPC is a well-known method for increasing the aggregate value of its proteins by modifying their solubility, viscosity and emulsifying and foaming properties and, more importantly, for improving their nutritional properties. WPC hydrolysates are considered ideal ingredients in the formulation of human milk substitutes due to their high nutritional value, low bitterness and low antigenicity (Guadix, Camacho, Guadix, 2004). The hydrolytic reaction must be strictly controlled to produce hydrolysates with the appropriate peptide profiles for nutritional purposes. These protein hydrolysates may have several applications in specialty foods, such as those designed to provide nutritional support to individuals with particular physiological or nutritional needs not covered by conventional diets (Pacheco, Amaya-Farfan, Sgarbieri, 2002).
As the functional and nutritional properties of protein hydrolysates are affected by peptides of different sizes, it is important to understand the molecular weight distribution profile, which is commonly measured using Size Exclusion Chromatography. An efficient method was previously developed by Silvestre, Hamon and Yvon (1994a,b) using this principle, which enabled the separation of peptides with molecular masses lower than 1,000 Da, and this is the method used in the current study. This method was used for the characterisation of the peptide profiles of hydrolysates obtained from different protein sources (Carreira et al., 2004; Lopes et al., 2005, 2008; Morais et al., 2005; Silva et al., 2007).
Protein hydrolysates are also a source of bioactive peptides that are inactive in the intact protein but become active after hydrolysis. These peptides have many beneficial actions in the organism, such as antihypertensive properties (Hartmann, Meisel, 2007). Angiotensin-converting enzyme (ACE) plays a crucial role in the regulation of blood pressure by converting angiotensin I into angiotensin II, a potent vasoconstrictor, and by simultaneously inactivating bradykinin, a vasodilator (Simões Silva, Flynn, 2012). Hydrolysates and peptides derived from milk proteins may exhibit inhibitory activity (IA) against ACE. Thus, these proteins could be introduced into the diet as an alternative, non-pharmacological approach to preventing and treating arterial hypertension (Costa, Gontijo, Netto, 2007; Jiang et al., 2007; Miguel et al., 2007; Otte et al., 2007).
This work focused on the study of the action of pancreatin and papain in obtaining WPC hydrolysates with peptide profiles that were nutritionally appropriate and conferred ACE inhibitory activity. The effects of several parameters, such as enzyme type, E:S ratio and the use of UF, were evaluated.
MATERIAL AND METHODS
Material
Powdered whey protein concentrate (WPC - Kerrylac 750) was kindly furnished by Kerry do Brazil Ltd (Três Corações, MG, Brazil). Pancreatin (Corolase PP® from porcine pancreas, EC 3.4.21.4, activity of 34.7 U mL-1) and papain (Corolase L10®, EC 3.4.22.2, activity of 31.6 U mL-1) were kindly provided by AB Enzymes (Barueri, SP, Brazil). In this study, enzyme activity was defined as the amount of enzyme required to liberate 1 g of tyrosine in one min at 37 ºC. Formic acid was purchased from Merck (Whitehouse Station, NJ, USA). The polyvinylidene fluoride membranes (0.22 mm for samples and 0.45 mm for solvents) and the tangential flow filtration system of 10 kDa porosity were purchased from Millipore (São Paulo, SP, Brazil). The HPLC system used for fractionating the protein hydrolysates consisted of one isocratic pump and a UV-Vis detector (1200 Series, Agilent, Santa Clara, CA, USA) coupled to a computer with ChemStation software for LC Systems (Agilent, Santa Clara, CA, USA). A poly(2-hydroxyethylaspartamide)-silica (PHEA) column (250 x 9.4 mm, 5 mm and 200 Å pore size) was used for HPLC. Angiotensin-converting enzyme (ACE) from rabbit lung, 0.25 units mg-1 of protein, EC 3.4.15.1, Hippuryl-histidyl-leucine (HHL) and hippuric acid were purchased from Sigma (St. Louis, MO, USA). HPLC-grade trifluoroacetic acid was purchased from Vetec (Duque de Caxias, RJ, Brazil). HPLC-grade acetonitrile was acquired from J.T. Backer (Phillipsburg, NJ, USA). The reversed-phase high performance liquid chromatography (RP-HPLC) system consisted of a GraceSmart chromatography column RP-18, 150 x 46 mm, 5 mm, 120 Å (Grace Davison, Deerfield, IL, USA), a quaternary pump and a UV-VIS spectrophotometer (HP series 1100, Waldbronn, Germany) coupled to a computer with analytical software (HP chemstation, Avondale, USA). The water for HPLC was purified by passage through an Aries Vaponics water purification system (Rockland, USA). All of the solvents used in this study were analytical grade.
Preparation of enzymatic hydrolysates from whey protein concentrate
Sixteen hydrolysates were prepared by varying the following parameters: type of enzyme, enzyme:substrate ratio (E:S) and the use of UF. The conditions used for preparing these hydrolysates are displayed in Table I. Solutions of whey protein concentrate (10% w/v) were prepared with distilled water; the concentration of protein in each solution was 3.42%, and the pH was adjusted to 7.0 with a 3 M NaOH solution. The solutions were heated in a Vaseline bath under constant agitation by a magnetic stirrer (752A model, Fisatom, São Paulo, SP, Brazil) at the optimal temperature for each enzyme (50 ºC for pancreatin and 55 ºC for papain), followed by the addition of these enzymes to obtain the desired E:S ratio. The total hydrolysis time was 5 h, and after this time, the enzymes were inactivated by heating in a water bath at 75 ºC for 15 sec. The samples were subsequently lyophilised (Freeze Dry System / Freezone 4.5, model 77500, Labconco, Kansas City, MO, USA).
Ultrafiltration of protein hydrolysates
Some samples of protein hydrolysates (Table I) were submitted to UF and diafiltration using a volume of water equivalent to ten times the initial volume. A system of tangential flow with a 10 kDa cut-off membrane coupled to a peristaltic pump was used. The samples were subsequently lyophilised.
Characterisation of peptide profiles from WPC hydrolysates
The characterisation of peptide profiles was performed in two stages, which included the fractionation of peptides according to size and their subsequent quantification. The fractionation of WPC hydrolysates was conducted by size exclusion (SE) HPLC on a PHEA column, according to the method previously developed by Silvestre, Hamon and Yvon (1994a), using 0.05 M formic acid (pH 2.5) as the mobile phase, with isocratic conditions at a flow rate of 0.5 mL min-1 for 35 min. The samples were dissolved in the mobile phase (0.1%) and filtered through a 0.22 mm membrane, and 50 mL of these solutions was injected into the column. The mobile phase was filtered through a 0.45 mm membrane and degassed by sonication in an ultrasound bath (USC1400 model, Unique, Santo Amaro, SP, Brazil) for 30 min before use.
The rapid method of Correct Fraction Area (CFA) that was previously developed by Silvestre, Hamon and Yvon (1994b) was used for quantifying the peptides and free amino acids in the SE-HPLC fractions of the WPC hydrolysates. Briefly, five whey standard hydrolysates (two using trypsin and three using pancreatin) were prepared and then fractionated into four fractions by SE-HPLC, as described above. The four fractions were collected (Fraction Collector, CF-1 model, Spectrum Chrom, Houston, TX, USA) and submitted to amino acid analysis following solvent removal (Centrivap, 78100-00D model, Labconco, Kansas City, MO, USA). The calculation of CFA was performed using specific formulas after detection at three wavelengths (230, 280 and 300 nm) to remove the contribution of aromatic amino acids. A standard curve was drawn to correlate CFA with the amino acid contents of the fractions.
Evaluation in vitro of the ACE-inhibitory activity of the protein hydrolysates
Evaluation of the ACE-inhibitory activity of the WPC hydrolysates was performed according to the method developed by Wu, Aluko and Muir (2002) using RP-HPLC. Initially, a volume of 12.5 mL of the substrate hippuryl-histidyl-leucine (HHL) (2.17 mM) was mixed with 50 mL of hydrolysed WPC (10 mg mL-1), both prepared with a 100 mM borate buffer, pH 8.3, containing 300 mM NaCl, and the mixture was incubated at 37 ºC for 10 min. A volume of 200 mL of ACE (4 mU), prepared in the same buffer, was subjected to a similar treatment. The two solutions were subsequently mixed, and after incubation at 37 ºC for 30 min, the reaction was stopped by the addition of 125 mL of HCl (1 M). Next, the mixture was filtered through a 0.22 mm membrane for analysis by RP-HPLC.
For this analysis, a GraceSmart RP-18 column was used, and the hippuric acid and HHL were detected at 228 nm. The elution flux was of 0.5 mL min-1 with a two-solvent system: (A) 0.05% TFA in water and (B) 0.05% TFA in acetonitrile. The solvent B was used as following: for the first 10 min, in a gradient from 5 to 60%; during the following 2 min it was kept at 60% and for an additional 1 min at 5%. This procedure was followed by isocratic elution for 4 min at the constant flow rate of 0.5 mL min-1, using the solvent A.
The ACE inhibitory activity was expressed in two ways: as a percentage of inhibition and as an IC50 value, which is defined as the concentration of hydrolysate (mg mL-1) necessary to reduce the activity of the enzyme by 50%.
Evaluation of the effects of certain parameters
The effects of the type of enzyme, E:S ratio and UF on the peptide profile and ACE-inhibitory activity of WPC hydrolysates were evaluated. Two enzymes (pancreatin and papain) were used for preparing these hydrolysates with E:S ratios of 0.5:100, 1:100, 2:100 and 3:100. For each of these cases, certain samples were submitted to UF, and others were not.
Statistical analysis
All experiments and measurements were performed in triplicate. A completely randomised factorial design was adopted to investigate the presence of significant effects among the various treatments (p<0.05). The results were analysed using Statistica software (Statsoft, 2000). An analysis of variance was performed to evaluate the peptide and free amino acid contents of the chromatographic fractions at each condition, as well as the effects of enzyme type, E:S ratio and UF. Duncan's test was applied to evaluate differences between the means (Pimentel-Gomes, 2000).
RESULTS AND DISCUSSION
Peptide and amino acid contents
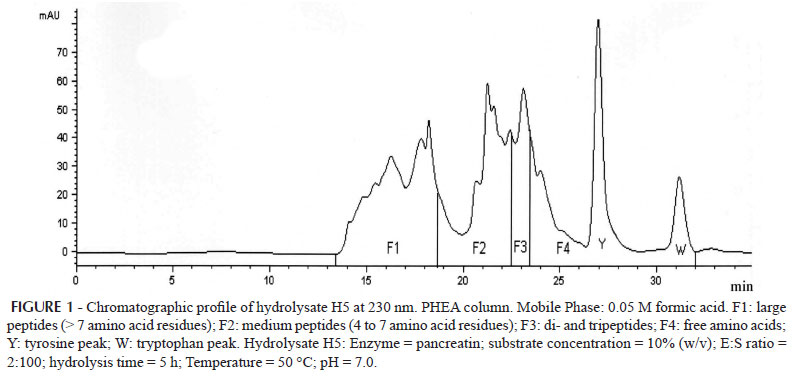
As illustrated in Figure 1, the hydrolysates were resolved in four fractions: F1, from 13.5 to 18.5 min (large peptides with more than 7 amino acid residues); F2, from 18.5 to 22.5 min (medium peptides with 4 to 7 amino acid residues); F3, from 22.5 to 23.5 min (di- and tripeptides); and F4, from 23.5 to 32.0 min (free amino acids).
The peptide and free amino acid contents of the WPC hydrolysates are presented in Table II, where a significant variation in the peptide and free amino acid contents among the different samples is observed. To choose the most appropriate hydrolysates for the development of nutritional supplements for clinical use, previous studies were considered. According to Frenhani and Burini (1999), during the metabolism of proteins, the first stage of hydrolysis leads to the formation of oligopeptides that contain 2 to 6 amino acid residues and free amino acids. Next, these peptides are broken into di- and tripeptides, and the proteins are finally absorbed in the form of di- and tripeptides and free amino acids. According to the same authors, the di- and tripeptides are absorbed more efficiently than the free amino acids, which, in turn, have greater absorption than the tetra- and larger peptides. For equivalent quantities of di- and tripeptides and mixtures of amino acids, the di- and tripeptides exhibited absorption rates that were approximately 10 times faster. Schaafsma (2009) also reported the advantage of di- and tripeptides over free amino acids with regard to the rate of absorption. In this context, the criteria for choosing the most advantageous hydrolysates in the present work were associated with the presence of high amounts of di- and tripeptides (fraction F3) and of free amino acids (fraction F4), as well as a low content of large peptides (fraction F1).
Thus, it can be inferred that, from a nutritional standpoint, the best peptide profile was achieved for the hydrolysate H14, as it displayed one of the greatest contents of di- and tripeptides (15.29%) and free amino acids (47.83%), as well as the least amount of large peptides (25.73%). The peptide profiles of hydrolysates H7 and H8 come just after H14, with the only disadvantage concerning their reduced free amino acid contents (26.89% and 28.56%, respectively). Hydrolysate H8 demonstrated an advantage over H14 regarding its smaller amount of large peptides (21.20%).
The effect of enzymatic hydrolysis on the peptide profile of WPC hydrolysates was considered in three studies. In the first study, the action of a subtilisin (protease from Bacillus subtilis, Protemax N200, Prozyn, São Paulo, Brazil) under different conditions led to a better peptide profile than the best results of the present study (H14, H8 and H7), with regard to the smaller large peptide content (12.28%) (Afonso et al., 2008). In the second study, several WPC hydrolysates were prepared using the same pancreatin that was used in the present study, but the lots and hydrolytic conditions of the WPC were different. Most of the hydrolysates exhibited appropriate peptide profiles from a nutritional standpoint, with low amounts of large peptides (12.80%, on average) and high amounts of di- and tripeptides (12.11% on average) and free amino acids (49.06% on average) (Afonso et al., 2009). A comparison with hydrolysate H14 led to the conclusion that these peptide profiles were advantageous due to their lower amounts of large peptides, but they were poorer due to their lower di- and tripeptide contents. In the third study, different enzymes and reaction conditions from those used in the current study were used to hydrolyse WPC. The best peptide profile was obtained from the action of Aspergillus oryzae protease (E:S 1:100, pH 7, 50 ºC, 5 h), which was better than that of H14 in terms of the amount of di- and tripeptides (16.14%), in addition to the lower large peptide content (18.76%) (Silva et al., 2009).
ACE-inhibitory activity of WPC hydrolysates
Among the sixteen hydrolysates evaluated (Table III), seven demonstrated high ACE-IA (> 80%: H1, H2, H3, H5, H6, H7 and H8), and four demonstrated greater activity (> 70%: H4, H13, H14 and H16) than the range of values reported in the literature (Otte et al., 2007). Three hydrolysates (H9, H11 and H15) displayed moderate inhibitory activity (40-60%) and only two samples (H10 and H12) demonstrated a low ability to inhibit ACE (<20%). It is noteworthy that the greatest ACE-IA results were obtained when pancreatin was used at E:S ratios of 0.5:100 (in the presence or absence of UF) and 3:100 (in the presence of UF).
Moreover, no ACE-IA was found for WPC, indicating that enzymatic treatment, which gives rise to molecules of small sizes, is required for the manifestation of this bioactive property. A similar phenomenon has already been reported in the literature for WPC (Mullally, Meisel, Fitzgerald, 1997; Guo, Pan, Tanokura, 2009) and other intact proteins (Mullally, Meisel, Fitzgerald, 1997; Jiang et al., 2007).
The fact that the whey protein hydrolysates prepared in this study had lower ACE inhibitory activity (higher IC50) than the synthetic antihypertensive Captopril (IC50 = 0.005 mg mL-1) does not hinder the application of whey protein-derived peptides in the treatment/prevention of hypertension. In fact, it is expected that milk protein-derived ACE inhibitory peptides, unlike Captopril, would have no undesirable side-effects (Hayes et al., 2007).
Some studies on the ACE-inhibitory activity of enzymatic hydrolysates of WPC were found in the literature. Mullally, Meisel and Fitzgerald (1997), using five different proteases, obtained hydrolysates with ACE-IA values that ranged from 60.8% to 88.6%, which are similar to those found for several hydrolysates in the present study. Guo, Pan and Tanokura (2009) employed only one enzyme to hydrolyse WPC (a protease from L. helveticus) and used different temperatures, pHs, E:S ratios and reaction times; these researchers obtained hydrolysates with ACE-IA values that ranged from 15% to 63%, which are similar to those of five hydrolysates in this study that demonstrated moderate or low ACE-IA. Tavares et al. (2011) used an aqueous extracts of Cynara cardunculus for the hydrolysis of WPC, and they noted that the IC50 values of the hydrolysates ranged from 0.072 to 0.197 mg mL-1, a range that covers most of the values observed in the current study.
Effects of certain parameters on the peptide profile and ACE inhibitory activity
The effects of enzyme type, E:S ratio and the use of UF were evaluated based on three factors. The first factor involved the production of an appropriate peptide profile from a nutritional point of view; the second involved the production of hydrolysates with high ACE-IA and the third involved the reduction of costs for scaling up the process (use of low E:S ratio and absence of UF).
Effect of type of enzyme
The influence of the type of enzyme on the peptide profile of protein hydrolysates can be seen in Table II. To keep the other parameters constant, analysis of the data had to consider the following comparisons: 1. with UF: H2 with H10, H4 with H12, H6 with H14 and H8 with H16; 2. without UF: H1 with H9, H3 with H11, H5 with H13 and H7 with H15.
The action of papain produced the best peptide profile among all of the hydrolysates (H14). In addition, using UF, the action of papain was more advantageous than pancreatin in terms of the number of cases (four) while the pancreatin was more advantageous than papain in only two cases. In the absence of UF, this situation was reversed, and pancreatin produced beneficial results in a greater number of cases (four) than papain (two).
According to the specificities of the enzymes used in the current study, some of the results were expected, such in the case of the pancreatin which is an enzymatic mixture containing both endopeptidases (trypsin and chymotrypsin) and exopeptidases (carboxypeptidases A and B) that can lead to the cleavage of protein molecules at specific points, yielding the release of a greater amount of smaller peptides. Along the same reasoning, being an endopeptidase, papain is expected to produce higher large peptide content than pancreatin.
Few reports were found in the literature that have addressed the effect of enzyme type on the peptide profiles of WPC hydrolysates. Regarding the results of other authors, Pacheco and Sgarbieri (2005) reported the effect of the actions of three enzymes (a different pancreatin from that used in the current study and two proteases, one from Bacillus sp. and the other from Bacillus licheniformis) on the free amino acid content of WPC, evaluated by ion exchange chromatography with derivatization by ninhydrin. As in the present work, the pancreatin was the most beneficial enzyme due to its release of larger amounts of free amino acids, which was followed by the protease from Bacillus sp. and then from Bacillus licheniformis.
Silvestre et al. (2011) evaluated the effects of a subtilisin and the same type of pancreatin used in the current work, but from a different lot and under different hydrolytic conditions (E:S=4:100; reaction time=10 h). A similarity was observed between the peptide profiles obtained by these two enzymes. Comparing the best peptide profiles obtained using pancreatin in the previous study with those of the current work, a nutritional superiority may be observed for the former results, due to the lower large peptide content (12.80% and 21.20%, respectively) and greater amount of free amino acids (49.06% and 28.56%, respectively). However, the di- and tripeptide content obtained in the present study was slightly greater than in the previous study (15.97% and 12.11%, respectively). The superiority of the peptide profile of the previous study may be associated with the greater E:S ratio and the reaction time used, as the enzyme activity was not determined in the previous work.
In another study, seven enzymes were used to hydrolyse WPC, of which one corresponded to the pancreatin that was employed in the present study; however, it was from a different lot (enzyme activity = 4.71 U mL-1), and the hydrolysis was performed under different conditions than those used here (E:S=1:100; WPC concentration=7%) (Silva et al., 2009). The peptide profiles obtained using A. oryzae protease and pancreatin were similar but superior to the other enzymes from a nutritional standpoint. Comparing the best peptide profiles using pancreatin obtained in the previous study with those of the current work, a nutritional similarity can be observed; the former produced a greater amount of free amino acids (38.04% and 28.56%, respectively), whereas the di- and tripeptide contents of the second study were greater (9.12% and 15.97%, respectively). The large peptide contents were very close in both studies (21.35% and 21.20% for the first and second studies, respectively. These results indicate that for the same enzyme the use of greater E:S ratio and substrate concentration, as well as higher-activity enzyme favored in the current work the production of small peptides but reduced the amount of amino acids released during the hydrolysis.
The influence of enzyme type on the ACE-inhibitory activity of protein hydrolysates can be observed in Table III. To keep the other parameters constant, analysis of the data must consider the following comparisons: 1. with UF: H2 with H10, H4 with H12, H6 with H14 and H8 with H16; 2. without UF: H1 with H9, H3 with H11, H5 with H13 and H7 with H15. Pancreatin was more advantageous than papain for all cases.
The choice of the enzyme used for protein hydrolysis can affect the ACE-inhibitory activity of the hydrolysates because the presence of certain amino acids at the C- and N-terminal positions influences this property (Costa, Gontijo, Netto, 2007; Hernández-Ledesma et al., 2002). The most potent inhibitors consist of peptides containing either dicarboxylic or branched amino acid residues, such as valine and isoleucine, at the N-terminal position. Furthermore, the presence of hydrophobic amino acids, such as tryptophan, tyrosine, phenylalanine and proline, at the C-terminal portion is necessary (Costa, Gontijo, Netto, 2007; Tavares et al., 2011).
The statemens above help to explain, at least in part, the superior performance of pancreatin; the action of chymotrypsin, one of the enzymes found in this enzymatic complex, is associated with the breaking of peptide bonds at the C-terminal position of aromatic amino acids, releasing peptides with ACE-inhibitory activity. In contrast, papain, which demonstrates only endopeptidase activity and acts in the cleavage of substrates containing amino acid residues of lysine, arginine and valine, produces less potent peptides (Beynon, Bond, 2001).
The influence of the type of enzyme on the ACE-inhibitory activity of protein hydrolysates has been previously studied by other researchers, but only one study used WPC as a substrate. Mullally, Meisel and Fitzgerald (1997) evaluated the effects of five enzymes and demonstrated that the most efficient enzymes were trypsin (88.6%) and chymotrypsin (87.7%), followed by corolase PP (78.2%), which is a pancreatin, PTN 3.0S (a commercial enzyme preparation enriched with trypsin; 60.8%), and finally, elastase (35.5%).
Effects of the enzyme: substrate ratio
The effects of the E:S ratio (0.5:100; 1:100; 2:100 and 3:100) on the peptide profiles of WPC hydrolysates are displayed in Table II. To keep the other parameters constant, analysis of the data must consider the comparisons of two groups for each enzyme. For pancreatin, Group 1 (without UF) included H1 with H3 with H5 and with H7, and Group 2 (with UF) included H2 with H4 with H6 and with H8. For papain, Group 1 included H9 with H11 with H13 and with H15, and Group 2 included H10 with H12 with H14 and with H16. As indicated in this table, an advantage of using a lower E:S ratio was observed in some cases for pancreatin. For papain, the use of a lower E:S ratio was advantageous only after UF.
The beneficial effect of using a lower E:S ratio on the peptide profiles of WPC hydrolysates has already been demonstrated in two previous studies. Using the same pancreatin as the current work but under different hydrolytic conditions (E:S = 1:100; WPC concentration = 7%), it was possible to obtain lower amounts of the large peptides and greater amounts of free amino acids when the E:S ratio decreased from 4:100 to 2:100 (Silva et al., 2009). The action of a subtilisin in E:S ratios of 1:100, 2:100 and 4:100 also previously demonstrated, in some cases, that the use of a lower E:S ratio was advantageous as it was decreased from 4:100 to 2:100 and from 2:100 to 1:100; this decrease led to a reduction in the amount of large peptides and an increase in the free amino acid content (Afonso et al., 2009).
The influence of the E:S ratio (0.5:100, 1:100, 2:100 and 3:100) on the ACE-inhibitory activity of enzymatic hydrolysates of WPC can be viewed in Table III. To keep the other parameters constant, analysis of the data must consider the comparisons of two groups for each enzyme. For pancreatin, Group 1 (without UF) included H1 with H3 with H5 and with H7, and Group 2 (with UF) included H2 with H4 with H6 and with H8. For papain, Group 1 included H9 with H11 with H13 and with H15, and Group 2 included H10 with H12 with H14 and with H16.
As displayed in this table, the value of the E:S ratio of 0.5:100 produced the greatest ACE-inhibitory activities for pancreatin, with no statistical differences between the values found with or without UF. For papain, the greatest ACE-inhibitory activities were obtained with an E:S ratio of 2:100 and 3:100, without and with UF, respectively. The use of a lower E:S ratio can be more advantageous depending on the conditions used to prepare the protein hydrolysates, a finding that may be related to the existence of optimal conditions for hydrolysis. Outside of these conditions, ACE-inhibitory peptides may degrade rather than form, which would decrease inhibitory activity (Raghavan, Kristinsson, 2009).
In the only study of the effect of the E:S ratio on the ACE-inhibitory activity of protein hydrolysates, Guo, Pan and Tanokura (2009) demonstrated that the action of a protease from Lactobacillus helveticus on WPC led to an increase in the ACE-IA when the E:S moved from 0.2:100 to 0.8:100, where it reached its maximum value (63%) and subsequently remained unchanged until an E:S of 1.2:100, a value that is far inferior to the maximum found in the present study.
Effect of ultrafiltration
The influence of UF on the peptide profile of WPC enzymatic hydrolysates is presented in Table II. To keep the other parameters constant, analysis of the data must consider the following comparisons: for pancreatin, H1 with H2, H3 with H4, H5 with H6, H7 with H8; and for papain, H9 with H10, H11 with H12, H13 with H14 and H15 with H16.
As displayed in this table, the beneficial effect of not using UF was observed for pancreatin and papain. For pancreatin, an advantage was observed in the absence of UF regarding the higher di- and tripeptide contents of H3 and H5 in comparison to H4 and H6, respectively. For papain, the only advantage of not using UF was observed regarding the lower amount of large peptides when comparing H9 to H10. In fact, this result was expected because the membrane used here for the UF retains molecules with molecular masses greater than 10,000 Da.
No other reports on the effect of UF on the peptide profile of WPC hydrolysates were found in the literature. In a study previously conducted in our laboratory for whey hydrolysates obtained using a pancreatin different from that used here, the best peptide profiles were obtained using UF (Delvivo et al., 2005).
The effect of UF on the ACE-inhibitory activity of protein hydrolysates is displayed in Table III. To keep the other parameters constant, analysis of the data must consider the following comparisons: for pancreatin, H1 with H2, H3 with H4, H5 with H6, H7 with H8; and for papain, H9 with H10, H11 with H12, H13 with H14 and H15 with H16
The absence of UF was not beneficial when pancreatin was used, as the absence of UF resulted in protein hydrolysates with either lower ACE-IA values than those obtained in the presence of UF (H5 with H6 and H7 with H8) or with no significant differences between the ACE-IA values for both types of treatment (H1 with H2, H3 with H4). However, when using papain in the absence of UF, it was possible to obtain hydrolysates with greater ACE-IA than that obtained in the presence of UF in three cases (H9 with H10, H11 with H12 and H13 with H14). In only one case (H15 with H16) was this situation reversed. Our findings indicate that the absence of UF can be advantageous depending on the hydrolytic conditions used. The greater ACE-IA values obtained in the absence of UF may be explained by two factors. First, according to Raghavan and Kristinsson (2009), there is a synergistic action between all peptides found in a sample of whole hydrolysates (not subjected to any process of separation of peptides). Second, depending on the conditions employed, the UF process may cause the retention of peptides with structures (aromatic amino acids or proline at the C-terminal position) that favour ACE-inhibitory activity.
Only one study examined the influence of UF on the ACE-inhibitory activity of WPC hydrolysates. Mullally, Meisel and Fitzgerald (1997) hydrolysed WPC by trypsin and subsequently subjected it to UF. The authors demonstrated that no change in ACE-IA was observed when a 10 KDa cut-off membrane was used.
When taken as a whole, the results obtained both here and by other authors indicate that although UF is considered by some to be a method for enriching protein hydrolysates with peptides exhibiting ACE-inhibitory activity (Jiang et al., 2007), this finding is not always observed, and depending on the conditions employed, especially in the preparation of protein hydrolysates, the absence of this process can favour ACE-IA, which may reduce the costs of scaling up the process.
CONCLUSION
The type of enzyme, E:S ratio and the use of UF affected the peptide profiles and the ACE inhibitory activity of the WPC hydrolysates. The best peptide profile was obtained using papain with an E:S ratio of 2:100 and UF, whereas pancreatin produced the greatest ACE inhibitory activity with an E:S ratio of 0.5:100 (in the presence or absence of UF) or 3:100 (in the presence of UF).
ACKNOWLEDGMENTS
The authors thank the Financiadora de Estudos e Projetos (FINEP - contract nº 01.10.0005.00), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and the Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG) for financial support.
Received for publication on 09th May 2012
Accepted for publication on 13th September 2012
- AFONSO, W.O.; BIASUTTI, E.A.R.; CASTRO, V.M.; SILVA, V.D.M.; SILVESTRE, M.P.C. Utilização do soro de leite visando reduzir a poluição ambiental: hidrólise pela pancreatina. Tecno-log, v.12, p.7-16, 2008.
- AFONSO, W.O.; BIASUTTI, E.A.R.; GERALDI, L.M.; SILVA, V.D.M.; CAPOBIANGO, M.; SILVESTRE, M.P.C. Emprego da hidrólise enzimática utilizando subtilisina para elevar o valor nutricional do soro de leite. Nutrire, v.34, p.97-114, 2009.
- BEYNON, R.; BOND, J.S. Proteolytic Enzymes. 2.ed. Oxford: Oxford University Press, 2001. 340 p.
- BRANS, G.; SCHROËN, C.G.P.H.; VAN DER SMAN, R.G.M.; BOOM, R.M. Membrane fractionation of milk: state of the art and challenges. J. Memb. Sci, v.243, p.263-272, 2004.
- CARREIRA, R.L.; DE MARCO, L.M.; DIAS, D.R.; MORAIS, H.A.; SILVESTRE, M.P.C. Analysis of peptide profiles of casein hydrolysates prepared with pepsin, trypsin and subtilisin. Acta Farm. Bon., v.23, p.17-25, 2004.
- COSTA, E.L.; GONTIJO, J.A.R.; NETTO, F.M. Effect of heat and enzymatic treatment on the antihypertensive activity of whey protein hydrolysates. Int. Dairy J, v.17, p.632-640, 2007.
- DELVIVO, F.M.; SILVA, V.D.M.; MORAIS, H.A.; FIGUEIREDO, A.F.S.; AGUIAR, M.J.B.; COELHO, J.V.; SILVESTRE, M.P.C. Desenvolvimento de formulação dietética para fenilcetonúricos à base de hidrolisados de soro de leite. Rev. Bras. Nutr. Clin, v.20, p.117-126, 2005.
- FRENHANI, P.B.; BURINI, R.B. Mechanisms of amino acids and oligopeptides absorption in humans. Arq. Gastroenterol, v.36, p.227-237, 1999.
- GUADIX, A.; CAMACHO, F.; GUADIX, E.M. Production of whey protein hydrolysates with reduced allergenicity in a stable membrane reactor. J. Food Engineer, v.72, p.398-405, 2006.
- GUO, Y.; PAN, D.; TANOKURA, M. Optimisation of hydrolysis conditions for the production of the angiotensin-I-converting enzyme (ACE) inhibitory peptides from whey protein using response surface methodology. Food Chem, v.114, p.328-333, 2009.
- HARTMANN, R.; MEISEL, H. Food-derived peptides with biological activity: from research to food applications. Curr. Opin. Biotechnol, v.18, p.163-169, 2007.
- HAYES, M.; STANTON, C.; SLATTERY, H.; O'SULLIVAN, O.; HILL, C.; FITZGERALD, G.F.; ROSS, R.P. Casein fermentate of Lactobacillus animalis DPC6134 contains a range of novel propeptide angiotensin-converting enzyme inhibitors. Appl. Environ. Microbiol., v.73, p.4658-4667, 2007.
- HERNÁNDEZ-LEDESMA, B.; RECIO, I.; RAMOS, M.; AMIGO, L. Preparation of ovine and caprine β-lactoglobulin hydrolysates with ACE-inhibitory activity. Identification of active peptides from caprine β-lactoglobulin hydrolysed with thermolysin. Int. Dairy J., v.12, p.805-812, 2002.
- JIANG, J.; CHEN, S.; REN, F.; LUO, Z.; ZENG, S.S. Yak Milk Casein as a functional ingredient: Preparation and identification of angiotensin-I-converting enzyme inhibitory peptides. J. Dairy Res, v.74, p.18-25, 2007.
- LOPES, D.C.F.; BIZZOTTO, C.S.; SILVA, V.D.M.; AFONSO, W.O.; LOPES JR, C.O.; SILVESTRE, M.P.C. Obtention of low-phenylalanine protein hydrolysates from rice: use of two pancreatins. J. Food Technol, v.6, p.57-65, 2008.
- LOPES, D.C.F.; DELVIVO, F.M.; SILVESTRE, M.P.C. Hydrolysates of skim milk powder: peptide profiles for dietetic purposes. Brit. Food J., v.107, p.42-53, 2005.
- MIGUEL, M.; LOPEZ-FANDINO, R.; ALONSO, M.J.; SALAICES, M. Vascular effects and antihypertensive properties of k-casein macropeptide. Int. Dairy J., v.17, p.1473-1477, 2007.
- MORAIS, H.A.; DE MARCO, L.M.; OLIVEIRA, M.C.; SILVESTRE, M.P.C. Casein hydrolysates using papain: peptide profile and encapsulation in liposomes. Acta Alim, v.34, p.59-69, 2005.
- MULLALLY, M.M.; MEISEL, H.; FITZGERALD, R.J. Angiotensin-I-converting enzyme inhibitory activities of gastric and pancreatic proteinase digests of whey proteins. Int. Dairy J., v.7, p.299-303, 1997.
- OTTE, J.; SHALABY, S.M.A.; ZAKORA, M.; NIELSEN, M.S. Fractionation and identification of ACE-inhibitory peptides from α-lactalbumin and β-casein produced by thermolysin-catalysed hydrolysis. Int. Dairy J, v.17, p.1460-1472, 2007.
- PACHECO, M.T.B.; AMAYA-FARFAN, J.; SGARBIERI, V.C. Partial characterization of a whey protein concentrate and its enzyme hydrolysates. J. Food Biochem, v.26, p.327-338, 2002.
- PACHECO, M.T.B.; SGARBIERI, V.C. Effect of different hydrolysates of whey protein on hepatic glutathione content in mice. J. Medic. Food, v.8, p.337-342, 2005.
- PIMENTEL-GOMES, F. Curso de estatística experimental. 14.ed. Piracicaba: Livraria Nobel, 2000. 477 p.
- RAGHAVAN, S.; KRISTINSSON, H.G. ACE-inhibitory activity of tilapia protein hydrolysates. Food Chem, v.117, p.582-588, 2009.
- ROMÁN, A.; WANG, J.; CSANÁDI, J.; HODÚR, C.; VATAI, G. Partial demineralization and concentration of acid whey by nanofiltration combined with diafiltration. Desalination, v.241, p.288-295, 2009.
- SCHAAFSMA, G. Safety of protein hydrolysates, fractions thereof and bioactive peptides in human nutrition. Eur. J. Clin. Nutr, v.63, p.1161-1168, 2009.
- SILVA, M.C.; SILVA, V.D.M.; LANA, A.M.Q.; SILVESTRE, M.P.C. Grau de hidrólise e perfil peptídico de hidrolisados enzimáticos obtidos a partir de concentrado protéico do soro de leite. Alim. Nutr, v.20, p.395-402, 2009.
- SILVA, V.D.M.; DE MARCO, L.M.; AFONSO, W.O.; LOPES, D.C.F.; JANUÁRIO, J.N.; AGUIAR, M.J.B.; SILVESTRE, M.P.C. Preparation of low phenylalanine whey hydrolysates, using papain and pancreatin immobilized on activated carbon and alumina. Am. J. Food Technol, v.2, p.327-341, 2007.
- SILVESTRE, M.P.C.; AFONSO, W.O.; LOPES JR, C.O.; SILVA, V.D.M.; MORAIS, H.A.; SOUZA, M.W.S.; SILVA, M.R. Use of subtilisin and pancreatin for hydrolyzing whey protein concentrate. Am. J. Food Technol., v.6, p.647-660, 2011.
- SILVESTRE, M.P.C.; HAMON, M.; YVON, M. Analyses of protein hydrolysates. 1. Use of poly (2-hydroxyethyl-aspartamide)-silica column in size-exclusion chromatography for the fractionation of casein hydrolysates. J. Agric. Food Chem, v.42, p.2778-2782, 1994a.
- SILVESTRE, M.P.C.; HAMON, M.; YVON, M. Analyses of protein hydrolysates. 2. Characterization of casein hydrolysates by a rapid peptide quantification method. J. Agric. Food Chem, v.42, p.2783-2789, 1994b.
- SIMÕES E SILVA, A.C.; FLYNN, J.T. The rennin-angiotensin-aldosterone system in 2011: role in hypertension and chronic kidney disease. Pediatr. Nephrol, v.27, p.1835-1845, 2012.
- SMITHERS, G.W. Whey and whey proteins - From 'gutter-to-gold'. Int. Dairy J., v.18, p.695-704, 2008.
- STATSOFT. Statistic for Windows [Computer program manual]. StatSoft, Inc. Tulsa, 2000.
- TAVARES, T.G.; CONTRERAS, M.M.; AMORIM, M.; MARTÍN-ÁLVAREZ, P.J.; PINTADO, M.E.; RECIO, I.; MALCATA, F.X. Optimisation, by response surface methodology, of degree of hydrolysis and antioxidant and ACE-inhibitory activities of whey protein hydrolysates obtained with cardoon extract. Int. Dairy J., v.21, p.926-933, 2011.
- WU, J.; ALUKO, R.E.; MUIR, A.D. Improved method for direct high-performance liquid chromatography assay of angiotensin-I-converting enzyme-catalyzed reactions. J. Chromatogr. A, v.950, p.125-130, 2002.
Publication Dates
-
Publication in this collection
14 Feb 2013 -
Date of issue
Dec 2012
History
-
Received
09 May 2012 -
Accepted
13 Sept 2012