Abstracts
Complex multiple emulsions have an excellent ability to fill large volumes of functional cosmetic agents. This study was aimed to encapsulate large volume of green tea in classical multiple emulsion and to compare its stability with a multiple emulsion without green tea extract. Multiple emulsions were developed using Cetyl dimethicone copolyol as lipophilic emulsifier and classic polysorbate-80 as hydrophilic emulsifier. Multiple emulsions were evaluated for various physicochemical aspects like conductivity, pH, microscopic analysis, rheology and these characteristics were followed for a period of 30 days in different storage conditions. In vitro and in vivo skin protection tests were also performed for both kinds of multiple emulsions i.e. with active (MeA) and without active (MeB). Both formulations showed comparable characteristics regarding various physicochemical characteristics in different storage conditions. Rheological analysis showed that formulations showed pseudo plastic behavior upon continuous shear stress. Results of in vitro and in vivo skin protection data have revealed that the active formulation has comparable skin protection effects to that of control formulation. It was presumed that stable multiple emulsions could be a promising choice for topical application of green tea but multiple emulsions presented in this study need improvement in the formula, concluded on the basis of pH, conductivity and apparent viscosity data.
Cosmetics; Green tea; Multiple emulsion; Rheology; Skin; Stability
Emulsões múltiplas complexas possuem excelente habilidade de agregar grandes quantidades de agentes cosméticos funcionais. Este estudo teve por objetivo encapsular grandes volumes de chá verde em uma emulsão múltipla clássica e comparar sua estabilidade com a emulsão múltipla sem o extrato do chá verde. Emulsões múltiplas são desenvolvidas usando cetil dimeticona copoliol como emulsificante lipofílico e o clássico polissorbato-80 como emulsificante hidrofílico. As emulsões múltiplas foram avaliadas por meio de vários aspectos fisico-químicos como condutividade, pH, análise microscópica e reologia. Estas características foram observadas por um período de 30 dias sob diferentes condições de armazenamento. Testes de proteção da pele in vivo e in vitro foram realizados para ambos os tipos de emulsões testadas, i.e. com o ativo em estudo (MeA) e sem ativo (MeB). Ambas as formulações apresentaram características comparáveis no que diz respeito aos diferentes fatores físico-químicos avaliados sob diferentes condições de armazenamento. A análise reológica mostrou que as formulações apresentaram comportamento pseudo-plástico sob contínuo estresse de cisalhamento. Os resultados dos testes in vivo e in vitro sobre a proteção da pele revelaram que a formulação ativa promoveu efeitos comparáveis à formulação controle. Nossos dados mostraram que emulsões múltiplas estáveis poderiam ser escolhas promissoras para a aplicação tópica do chá verde. Entretanto, a fórmula das emulsões múltiplas apresentadas neste estudo precisam ser melhoradas no que diz respeito ao pH, condutividade e viscosidade aparente.
Cosméticos; Chá verde; Múltipla emulsão; Reologia; Pele; Estabilidade
ARTICLE
Fabrication, physicochemical characterization and preliminary efficacy evaluation of a W/O/W multiple emulsion loaded with 5% green tea extract
Tariq Mahmood; Naveed Akhtar; Barkat Ali Khan; Akhtar Rasul; Haji M. Shoaib Khan
Department of Pharmacy, Faculty of Pharmacy and Alternative Medicine, The Islamia University of Bahawalpur, Pakistan
Correspondence Correspondence: Tariq Mahmood Department of Pharmacy Faculty of Pharmacy and Alternative Medicine The Islamia University of Bahawalpur 63100, Pakistan E-mail: tariqmahmood750@gmail.com
ABSTRACT
Complex multiple emulsions have an excellent ability to fill large volumes of functional cosmetic agents. This study was aimed to encapsulate large volume of green tea in classical multiple emulsion and to compare its stability with a multiple emulsion without green tea extract. Multiple emulsions were developed using Cetyl dimethicone copolyol as lipophilic emulsifier and classic polysorbate-80 as hydrophilic emulsifier. Multiple emulsions were evaluated for various physicochemical aspects like conductivity, pH, microscopic analysis, rheology and these characteristics were followed for a period of 30 days in different storage conditions. In vitro and in vivo skin protection tests were also performed for both kinds of multiple emulsions i.e. with active (MeA) and without active (MeB). Both formulations showed comparable characteristics regarding various physicochemical characteristics in different storage conditions. Rheological analysis showed that formulations showed pseudo plastic behavior upon continuous shear stress. Results of in vitro and in vivo skin protection data have revealed that the active formulation has comparable skin protection effects to that of control formulation. It was presumed that stable multiple emulsions could be a promising choice for topical application of green tea but multiple emulsions presented in this study need improvement in the formula, concluded on the basis of pH, conductivity and apparent viscosity data.
Uniterms: Cosmetics. Green tea. Multiple emulsion. Rheology. Skin. Stability.
RESUMO
Emulsões múltiplas complexas possuem excelente habilidade de agregar grandes quantidades de agentes cosméticos funcionais. Este estudo teve por objetivo encapsular grandes volumes de chá verde em uma emulsão múltipla clássica e comparar sua estabilidade com a emulsão múltipla sem o extrato do chá verde. Emulsões múltiplas são desenvolvidas usando cetil dimeticona copoliol como emulsificante lipofílico e o clássico polissorbato-80 como emulsificante hidrofílico. As emulsões múltiplas foram avaliadas por meio de vários aspectos fisico-químicos como condutividade, pH, análise microscópica e reologia. Estas características foram observadas por um período de 30 dias sob diferentes condições de armazenamento. Testes de proteção da pele in vivo e in vitro foram realizados para ambos os tipos de emulsões testadas, i.e. com o ativo em estudo (MeA) e sem ativo (MeB). Ambas as formulações apresentaram características comparáveis no que diz respeito aos diferentes fatores físico-químicos avaliados sob diferentes condições de armazenamento. A análise reológica mostrou que as formulações apresentaram comportamento pseudo-plástico sob contínuo estresse de cisalhamento. Os resultados dos testes in vivo e in vitro sobre a proteção da pele revelaram que a formulação ativa promoveu efeitos comparáveis à formulação controle. Nossos dados mostraram que emulsões múltiplas estáveis poderiam ser escolhas promissoras para a aplicação tópica do chá verde. Entretanto, a fórmula das emulsões múltiplas apresentadas neste estudo precisam ser melhoradas no que diz respeito ao pH, condutividade e viscosidade aparente.
Uniterms: Cosméticos. Chá verde. Múltipla emulsão. Reologia. Pele. Estabilidade.
INTRODUCTION
Since ancient times, natural extracts from animal, botanical or mineral origin have been used as "active ingredients" of drugs or cosmetics. (Elsner, Maibach, 2000). Herbal extracts have been used by cosmetic science in order to beautify and maintain the physiological balance of the human skin. A number of plant products described in scientific literature shows distinct activities on the skin, such as moisturizing, antioxidant, sunscreen and depigmentation (Magalhães et al., 2011).
Emulsified systems and particularly double emulsions water in oil in water emulsion (W/O/W) are known to be thermodynamically instable (Benna-Zayani et al., 2008). Both types of emulsions i.e., W/O and O/W exist simultaneously. They combine the properties of both W/O and O/W emulsions. These have been described as heterogeneous system of one immiscible liquid dispersed in another liquid in the form of fine droplets which have diameter greater than 1 micron. Multiple emulsions have very low thermodynamic stability. The main advantages of (W/O/W) multiple emulsions are the protection of entrapped substances, their capacity to incorporate several actives in the different emulsion compartments, and their sustained release effects (Akhtar et al., 2008; Morais et al., 2008; Akhtar, Yazan, 2005).
Most literature data relates to multiple emulsions based on conventional non-ionic surfactants. However, most of these surfactant systems were reported to produce multiple emulsions with a limited shelf-life. The most often used primary lipophilic polymeric emulsifier for the preparation of W/O/W emulsions is cetyl dimethicone copolyol. This emulsifier is used in W/O/W emulsions usually at a concentration of about 4% (in the primary W/O emulsions formulation); however, the use of higher concentrations (i.e., 5-15%) has also been reported. Low emulsifier concentrations are advantageous and preferred in pharmaceutical and cosmetic applications with respect to relevant toxicological, economic and environmental issues (Vasiljevic et al., 2009). Creams are well tolerated formulations because they interact mildly with the skin. They provide a barrier effect and improve permeation of active compounds into the human epidermis (Oliveira et al., 2011).
Through this work we aimed to develop, characterize and conduct a preliminary efficacy study of multiple emulsion encapsulated with 5% green tea extract, so that the many cosmeceutical benefits it could offer may be determined in future studies involving large population groups.
MATERIAL AND METHODS
Material
We have used the following substances to develop multiple emulsions: extra pure paraffin oil as oil phase for primary emulsion formation (Merck, Germany). Cetyl dimethicone copolyol (Abil® EM 90) has been used as lipophilic emulsifier supplied by Franken (Franken, Germany) and Polysorbate-80 (Tween® 80) supplied by (Merck, Germany) was used as hydrophilic emulsifier for secondary emulsification. Sodium chloride was used as conductimetric tracer. Standardized, ethanolic green tea extract prepared in our lab was encapsulated as functional cosmetic agent in the inner aqueous phase of the primary emulsion. Crystal violet dye was employed as dyeing agent in the in vivo skin protection test. All other chemicals used in this study were of analytical grade.
Preparation of multiple emulsions
Multiple emulsions were prepared using a two-step emulsification procedure (Akhtar et al., 2010). Primary emulsion was prepared by emulsifying both oil phase and aqueous phase in the presence of lipophilic surfactant while heating both phases at 75 °C in a digital water bath (Heidolph, Germany). Green tea extract and conductimetric tracer were incorporated into the internal aqueous phase of the primary emulsion. Homogenization of the primary emulsion was accomplished with IKA Mixing Overhead Stirrer, Eurostar (IKA, Werke, Germany) at 2000 rpm for 5 minutes and then reduced to 1000 rpm for 15 minutes and finally the emulsion was cooled to room temperature while maintaining a stirring speed of 500 rpm for a further 5 minutes. The primary emulsion obtained was subjected to second stage emulsification, in which the primary emulsion was added up slowly to the aqueous phase containing hydrophilic emulsifier at a stirring speed of 700 rpm for 40 minutes. At this time, multiple emulsions were confirmed by microscopic analysis. A similar procedure was revised to formulate a base (MeB) emulsion without green tea extract. The composition of multiple emulsions (w/w) is shown in (Table I).
Microscopic Analysis
Multiple characteristics of the prepared emulsions were revealed by the microscopic analysis, through an optical microscope (Nikon E200, Nikon, Japan) with a camera (DCM-35 USB 2.0 and Minisee Image software). Observations were made at 100 X magnification after suspending the samples in the external phase of the emulsion.
Conductimetric Analysis
Conductimetric analysis of undiluted samples was performed in order to examine the release of the electrolyte initially entrapped in the internal water phase (Vasiljevic et al., 2009; Akhtar et al., 2008). The specific conductivity of the emulsions was measured directly using a digital Conductivity Meter, (WTW- Tetracon®, Germany) at 25±2 °C. Conductivity tests were performed for multiple emulsion formulations immediately after preparation and after 24 h, 48 h, day 07, 15, 30 for samples kept in different storage conditions (08 ± 1 °C, 25 ± 1 °C, 40 ± 1 °C, 40 ± 1 °C + 75% RH) by using digital conductivity meter. All the measurements were performed in triplicate.
pH determination
The pH of fresh samples and samples kept in different storage conditions (08 ± 1 °C, 25 ± 1 °C, 40 ± 1 °C, 40 ± 1 °C + 75% RH) was determined by a digital pH meter ProfiLine pH 197 (WTW, Germany). The pH measurements were also taken for the samples after 24 h, 48 h and day 7, 15, 30. All the measurements were performed in triplicate.
Centrifugation and phase separation
The emulsions were centrifuged at 25 °C with centrifuge machine (Hettich EBA 20, Germany) at 5000 rpm for 10 minutes. Tests were performed for each prepared formulation kept in different storage conditions (08 ± 1 °C, 25 ± 1 °C, 40 ± 1 °C, 40 ± 1 °C+75% RH) at specific intervals (24 h, 48 h, day 7, 15 and 30).
Rheological examination
Rheological properties were determined, using a Brookfield programmable rheometer (Model DV.III; Brookfield engineering laboratories Inc.). Rheocalc V 2.6 (Microsoft Corporation) was used as a support program during the measurements. Of each sample an amount of 0.5 g was applied in a sample holder. The viscosities of the samples were determined at 25 °C and spindle (CP41) speeds ranging from 100 to 200 rpm. The measurements were performed in triplicate.
For evaluation, the results of viscosity measurements were fitted to the Power Law, known as:
Τ = kDn (1)
In vitro Occlusion Test
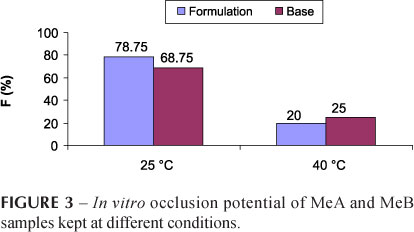
This test was performed to compare the in vitro occlusive capacities of the active and base formulation using the modified method of Erdal and Araman (Erdal, Araman, 2006). The test was conducted using 10 mL vials, filled with 5 mL distilled water and sealed with cellulose acetate filter with a pore size of 0.45 micrometer (Sartorius AG, Germany) for the in vitro occlusion test. A sample amount 0.5 mg was spread over the filter and stored at 25 ± 1 °C and 40 ± 1 °C for 48 h. The occlusion factor F was calculated using the following equation.
F= 100 x [(A-B)/A] (2)
where A is the amount of water evaporated through the cellulose acetate membrane without applying a formulation and B is the amount of water evaporated through the cellulose acetate membrane after applying a sample.
In vitro and In vivo Skin Protection Test
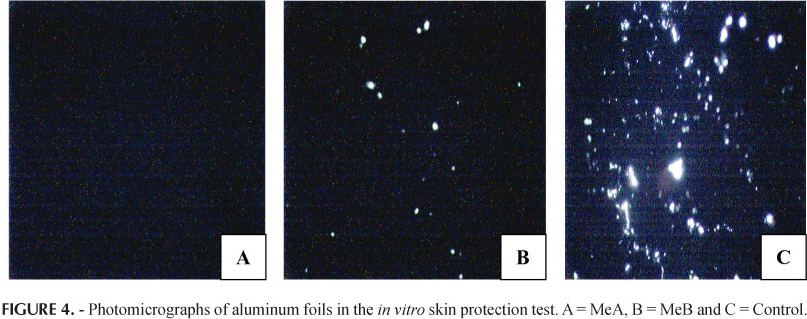
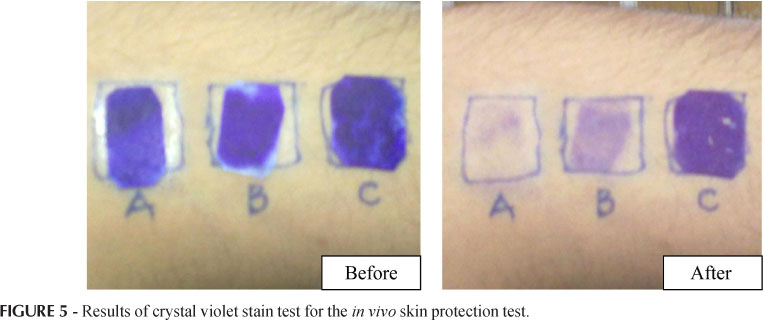
The in vitro/in vivo skin protecting effects of both MeA and MeB were assessed by the modification of the aluminum - foil deterioration test and the crystal violet stain test (Wille, 2006). The in vitro test was carried out by cutting 3 pieces of aluminum foil. Foil 1 was coated with about 50 µL of sample MeA and, similarly, foil 2 was coated with the same amount of MeB formulation. Both foils were air dried for 10 minutes. The 3rd foil was kept as control without any formulation coated on it. The coated and un-coated foils were exposed to one drop of 3 N HCl acid for 30 minutes. HCl treated foils were observed through an optical microscope (Nikon E200, Nikon, Japan) with a camera (DCM-35 USB 2.0 and Minisee Image software) attached. Observations were recorded at 10X magnification. Images were taken for the light passing through any holes formed in the aluminum foils. For the in vivo skin protection test 3 squares were drawn on the volar arm of a human subject (n=1) and these areas were then coated with MeA, MeB; the third area was preserved as control. Afterwards, filter papers were cut in appropriate size, dipped in 0.2% crystal violet stain, drained of excess dye and applied to the treated and un-treated area for 5 minutes. These filter papers were then removed, excess dye washed off by several rinses, and results for stained skin areas were photographed. In vivo skin protection study was conducted in our cosmetics lab, approved by the Board of Advanced Studies and Research from the Islamia University of Bahawalpur (No. 942/Acad). The study was conducted in accordance with the ethics principles of the Declaration of Helsinki and was consistent with Good Clinical Practice guidelines.
RESULTS AND DISCUSSION
Microscopic analysis
At the time of preparation multiple emulsion formulations were found to be homogeneous, creamy with glossy appearance. The glossy appearance may be due to silicone emulsifier. MeA was yellowish green while MeB was white in color as no green tea extract was present in it. Multiple emulsion formation was confirmed by the microscopic analysis and results are shown in Figure 1.
pH Analysis
pH results revealed that no obvious chemical degradation occurred in any of the samples of MeA and MeB, kept in different storage conditions. In general, the pH values decreased slightly at higher temperatures in formulations containing green tea extract (Table II). These variations may be due to hydrolysis reactions or even oxidation of the preparation components which commonly occurs in accelerated stability studies. However formulation MeB samples without active ingredients showed no change in pH at all storage conditions. Since these changes were observed mainly in the formulations containing the active ingredients, this suggests that instability was due to these components, which affected the integrity of the formulation (Gonçalves et al., 2011).
Conductivity Analysis
The conductivity values of drug loaded emulsions were higher than unloaded formulations and it has been reported previously that conductivity increases in drug loaded formulations (Kantarci et al., 2007). Conductimetric analysis was carried out in order to measure the entrapped conductimetric tracer in the inner aqueous phase of the primary emulsion in order to detect any leakage of internal aqueous phase of the primary emulsion to the outer aqueous phase. This phenomenon was more pronounced as the temperature increased. It should be noted that the more a conductimetric tracer is released, the more active substance is free to move in external aqueous phase, and thus the effects of the active substance will be less sustained. It was obvious from the results that the sample of MeA formulation kept at 25 °C was stable enough against any leakage due to diffusion or rupturing of oil membrane. The results of the conductimetric analysis are shown in Table II and Table III.
Centrifugation and Phase separation
We also performed centrifugation test at 5000 rpm for 20 minutes for each sample at different time intervals and we did not found any phase separation in any of the samples of MeA and MeB upon centrifugation test. Similar results were observed in a previous study of centrifugation at 6000 rpm for 15 min (Deccache et al., 2010). Although conductimetric tracer leaked from some of the samples kept at higher temperatures, the results of centrifugation indicate that emulsions were stable enough against any phase separation. However, leakage of internal aqueous phase into the external phase caused thinning of the multiple emulsion samples at higher temperatures. No phase separation occurred and no sudden fall in conductivity values was observed, as it has been reported that too much reduction in conductivity values has been attributed to phase separation (Kantarci et al., 2007).
Rheological analysis
We determined multiple emulsion flow parameters using Power low equation:
τ = K γ n
where τ is shear stress, γ is shear rate, K is consistency index and n is flow behavior index.
Consistency index K is a measure of the system consistency and it is in relation with apparent viscosity. Flow behavior index n determines the degree of non-Newtonian behavior and varies in the range between 0 and 1. The non-Newtonian character of the investigated system is more pronounced for smaller values of the constant n (Krstonošić et al., 2011).
The viscosities of multiple emulsion samples were measured at a speed of 100 to 200 rpm (with 20 increments) while applying shear rates from 200 to 400 (with 20 increments) on each sample. High shear rates were applied for quality assurance of emulsions by applying high shear stresses. Results for the rheology of the fresh sample and for samples kept in different conditions of storage (followed for 30 days) are shown in (Table IV) and (Table V) and samples of both MeA and MeB have shown shear thinning pseudo plastic behaviors on varying shear rates. It was observed that viscosities of multiple emulsions decreased continuously during storage with the passage of time. There may be two reasons behind this phenomenon; a) diffusion of water molecules from the internal to the external aqueous phase or b) bursting of multiple globules due to osmotic pressure (Tirnaksiz, Kalsin, 2005). Though a decrease in viscosity has been observed with passage of time for the samples of MeA and MeB, this appears to be due to the migration of internal water globules through the oil (paraffin oil) layer, as an increase in conductivity values is indicative of no phase separation. Further accelerated centrifugation tests at different time intervals have not revealed any phase separation in any of the samples from MeA and MeB. However, it was obvious that MeA and MeB fresh samples and samples stored in different conditions have shown some unusual non-newtonian "dilatant" behavior (Figure 2) which is rarely seen in cosmetic emulsions. Power Law calculations clearly indicated flow index (n) values greater than 1, which means samples have shown "dilatant" behavior. More recently it was investigated that due to high salt concentration, flocculation occurs and this flocculation leads to "dilatant" behavior and, ultimately, viscosity of the dispersion is reduced (Zhang et al., 2012).
In vitro Occlusion Test
This test was performed to compare the in vitro occlusive capacities of active and base formulation i.e. MeA and MeB. Occlusion is a desired phenomenon in cosmetic formulations intended for moisturizing purpose, and thus to prevent any excessive evaporation of water from the stratum corneum of the skin. When results of MeA and MeB samples were compared for their occlusive effects, apparently no variation in occlusion was observed for MeA and MeB for two storage conditions i.e. 25 ºC and 40 ºC. However, it was evident that the occlusion potential of both formulations was higher at a lower temperature. These similar effects seem due to same ratio of oily phase in both MeA and MeB. This explanation has also been offered previously, stating that the value of F depends upon the volume of oily phase (Erdal, Araman, 2006). The results are presented in Figure 3.
In vitro and In vivo Skin Protection Test
The in vitro/in vivo skin protecting effects of both MeA and MeB were tested. A typical result for the in vitro skin protection test is shown in Figure 4, in which the control piece (C) of aluminum foil developed holes in it, indicating that it offered no resistance to acid corrosion. The active formulation (A) with 5% green tea extract offered comparable resistance against acid corrosion with respect to MeB formulation shown in (B). Similarly, results for the in vivo skin protection test are shown in Figure 5, which indicate that MeA offered more resistance to penetration of crystal violet stain than MeB and MeB offered more resistance to penetration of crystal violet stain than the control.
CONCLUSION
In recent years there has been a quest for aesthetic cosmetic formulations with multifunctional properties. Since multiple emulsions have presented some properties that individualize them from others, we have tried to fabricate and characterize multiple emulsions using classical hydrophilic emulsifier Polysorbate-80, loading this system with high volume of green tea extract. We subjected these formulations to the evaluation of different physico-chemical properties under different conditions of storage. We concluded that:
· The emulsions were stable enough against any phase separation but pH, conductivity and apparent viscosity data was not impressive enough to assume multiple emulsions long-term stability.
· No apparent variation in occlusion was observed for MeA and MeB for two storage conditions i.e. 25 ºC and 40 ºC.
· In the in vivo skin protection test MeA offered more resistance to penetration of crystal violet stain than MeB and MeB offered more resistance to penetration of crystal violet stain than the control.
The formulations presented some good stability characteristics, but the composition of the multiple emulsions need improvement to achieve long-term stability.
CONFLICT OF INTEREST POLICY
Authors have no competitive interests for this manuscript.
ACKNOWLEDGMENT
Authors are highly thankful to Higher Education Commission of Pakistan for providing Scholarship. Authors are highly obliged with efforts of Assistant Professor Mirela Moldovan at University of Medicine and Pharmacy Iuliu Hatieganu of Cluj-Napoca, Romania. Her preliminary review of this manuscript before submitting to Brazilian Journal of Pharmaceutical Sciences improved this manuscript scientifically.
Received for publication on 05th May 2012
Accepted for publication on 26th March 2013
- AKHTAR, N.; YAZAN, Y. Formulation and characterization of a cosmetic multiple emulsion system containing macadamia nut oil and two antiaging agents. Turk. J. Pharm. Sci., v.2, p.173-185, 2005.
- AKHTAR, N.; AHMAD, M.; GULFISHAN, M.; MASOOD, M.I.; ALEEM, M. Formulation and in vitro evaluation of a cosmetic emulsion from almond oil. Pak. J. Pharm. Sci., v.21, p.430-437, 2008.
- AKHTAR, N.; AHMAD, M.; KHAN, H.M.S.; AKRAM, J.; GULFISHAN, M.; MAHMOOD, A.; UZAIR, M. Formulation and characterization of a multiple emulsion containing 1% L-ascorbic acid. Bull. Chem. Soc. Ethiop, v.24, p.1-10, 2010.
- BENNA-ZAYANI, M.; KBIR-ARIGUIB, N.; TRABELSI-AYADI, M.; GROSSIORD, J.-L. Stabilisation of W/O/W double emulsion by polysaccharides as weak gels. Colloids Surf. A: Physicochem. Eng. Aspects, v.316, p.46-54, 2008.
- DECCACHE, D.S.; SANTOS, E.P.; CABRAL, L.M.; RODRIGUES, C.R.; SOUSA, V.P. Development of methodologies for dimethylaminoethanol glycolate assay in association with sunscreens in dermocosmetic formulation. Braz. J. Pharm. Sci., v.46, p.705-713, 2010.
- ELSNER, P.; MAIBACH, H.I. Cosmeceuticals New York: Marcel Dekker, 2000. 110 p.
- ERDAL, M.S.; ARAMAN, A. Development and evaluation of multiple emulsions systems containing cholesterol and squalene. Tur. J. Pharm. Sci., v.3, p.105-121, 2006.
- GONÇALVES, G.M.S.; SREBERNICH, S.M.; SOUZA, J.A.M. Stability and sensory assessment of emulsions containing propolis extract and/or tocopheryl acetate. Braz. J. Pharm. Sci., v.47, p.585-592, 2011.
- KANTARCI, G.; OZGÜNEY, I.; KARASULU, H.Y.; ARZIK, S.; GÜNERI, T. Comparison of different water/oil microemulsions containing diclofenac sodium: preparation, characterization, release rate, and skin irritation studies. AAPS Pharm. Sci. Tech., v.8, p.E91, 2007.
- KRSTONOŠIĆ, V.; DOKIĆ, L.; NIKOLIĆ, I.; DAPČEVIĆ, T.; HADNAĐEV, M. Influence of sodium dodecyl sulphate concentration on disperse and rheological characteristics of oil - in - water emulsions stabilized by OSA starch - SDS mixtures. J. Serb. Chem. Soc., v.76, p.1-15, 2011.
- MAGALHÃES, W.V.; BABY, R.; VELASCO, M.V.R.; PEREIRA, D.M.M.; KANEKO, T.M. Patenting in the cosmetic sector: study of the use of herbal extracts. Braz. J. Pharm. Sci., v.47, p.693-700, 2011.
- OLIVEIRA, L.A.; SOUZA-MOREIRA, T.M.; CEFALI, L.C.; CHIARI, B.G.; CORRÊA, M.A.; ISAAC, V.L.B.; SALGADO, H.R.N.; PIETRO, R.C.L.R. Design of antiseptic formulations containing extract of Plinia cauliflora. Braz. J. Pharm. Sci., v.47, p.525-534, 2011.
- MORAIS, M.; SANTOS, O.D.H.; NUNES, J.R.L.; ZANATTA, C.F.; ROCHA-FILHO, P.A. W/O/W Multiple Emulsions Obtained by One-Step Emulsification Method and Evaluation of the Involved Variables Jacqueline. J. Disp. Sci. Technol., v.29, p.63-69, 2008.
- TIRNAKSIZ, F.; KALSIN, O. A topical w/o/w multiple emulsions prepared with tetronic 908 as a hydrophilic surfactant: formulation, characterization and release study. J. Pharm. Pharm. Sci., v.8, p.299-315, 2005.
- VASILJEVIĆ, D.D.; PAROJČIĆ, J.V.; PRIMORAC, M.M.; VULETA, G.M. Rheological and droplet size analysis of W/O/W multiple emulsions containing low concentrations of polymeric emulsifiers. J. Serb. Chem. Soc., v.74, p.801-816, 2009.
- WILLE, J.J. Skin delivery systems, transdermals, dermatologicals and cosmetic actives USA: Blackwell Publishing, 2006. 392 p.
- ZHANG, J.; LI, L.; WANG, J.; SUN, H.; XU, J.; SUN, D. Double inversion of emulsions induced by salt concentration. Langmuir, v.28, p.6769-6775, 2012.
Correspondence:
Publication Dates
-
Publication in this collection
25 July 2013 -
Date of issue
June 2013
History
-
Received
May 2012 -
Accepted
Mar 2013