ABSTRACT
Ocimum is one of the most important genera of the Lamiaceae family. Several studies about basil and its popular use reveal many characteristics of the herb, including its use as antioxidant, anti-aging, anti-inflammatory, anti-carcinogenic, anti-microbial, and cardiovascular agents, among others. In this paper, we evaluated genotoxic, oxidative, and anti-inflammatory parameters from the extract of Ocimum basilicum in different concentrations, using human leukocytes cultures exposed to challenging agents. Our results confirm that the O. basilicum extract acts as an antioxidant and effectively reverts or subjugates the effects of high oxidizing agents such as hydrogen peroxide. These actions are attributed to its composition, which is rich in polyphenols and flavonoids as well as compounds such as rosmarinic acid, all of which have well-known antioxidant activity. We also show that our basil extract presents anti-inflammatory properties, the mechanism of which is a composed interaction between the inhibition of pro-inflammatory mediator and the stimulation of anti-inflammatory cytokines. Although pharmacodynamics studies are necessary to evaluate the activities in vivo, our results demonstrated that basil could act as an antioxidant and anti-inflammatory and a possible alternative for medicinal treatment.
Uniterms:
Ocimum basilicum/evaluation; Ocimum basilicum/genotoxic effects; Ocimum basilicum/anti-inflammatory properties; Medicinal plants/study.
INTRODUCTION
The free radicals are oxidizing agents, having one or more unpaired electrons in their outer orbit, making them highly reactive species that act as electrophiles (Wills, Thomas, Gillham, 1989WILLS, E.D.; THOMAS, J.H.; GILLHAM, B. Wills' biochemical basis of medicine. 2.ed. [S.l.]: Wright, 1989. 590p.). The reactions of free radicals with the organism result in cell and tissue damage that contributes to the development of pathologies. The free radicals act on cellular components by oxidizing lipids, proteins, nucleic acids, and carbohydrates (Choi et al., 2002CHOI, C.W.; KIM, S.C.; HWANG, S.S.; CHOI, B.K.; AHN, H.J.; LEE, M.Y.; PARK, S.H.; KIM, S.K. Antioxidant activity and free radical scavenging capacity between Korean medicinal plants and flavonoids by assay-guided comparison. Plant Sci., v.163, n.6, p.1161-1168, 2002.).
The genus Ocimum L. includes approximately 150 species, possessing a great variation in plant morphology and biology, essential oil content, and chemical composition (Danesi et al., 2008DANESI, F.; ELEMENTI, S.; NERI, R.; MARANESI, M.; D'ANTUONO, L.F.; BORDONI, A. Effect of cultivar on the protection of cardiomyocytes from oxidative stress by essential oils and aqueous extracts of basil (Ocimum basilicum L.). J. Agric. Food Chem., v.56, n.21, p.9911-9917, 2008.). Ocimum basilicum, popular known as Basil or Sweet Basil, is a common herb that belongs to Lamiaceae family. Studies have shown many pharmacological effects in several diseases, with potent antioxidant, anti-aging, anticancer, antiviral, and antimicrobial properties (Sakr, Al-Amoudi, 2012SAKR, S.A.; AL-AMOUDI, W.M. Effect of leave extract of Ocimum basilicum on deltamethrin induced nephrotoxicity and oxidative stress in albino rats. J. Appl. Pharm. Sci., v.2, n.5, 2012.). Studies have reported that rosmarinic acid (RA) is the most biologically active compound present in Basil related to this activities (Javanmardi et al., 2002JAVANMARDI, J.; KHALIGHI, A.; KASHI, A.; BAIS, H.P.; VIVANCO, J.M. Chemical characterization of basil (Ocimum basilicum L.) found in local accessions and used in traditional medicines in Iran. J. Agric. Food Chem., v.50, n.21, p.5878-5883, 2002.; Lee, Scagel, 2009LEE, J.; SCAGEL, C.F. Chicoric acid found in basil (Ocimum basilicum L.) leaves. Food Chem., v.115, n.2, p.650-656, 2009.; Shiga et al., 2009SHIGA, T.; SHOJI, K.; SHIMADA, H.; HASHIDA, S.-N.; GOTO, F.; YOSHIHARA, T. Effect of light quality on rosmarinic acid content and antioxidant activity of sweet basil, Ocimum basilicum L. Plant Biotechnol., v.26, n.2, p.255-259, 2009. (Short Communication).).
Traditionally, basil has been used as a medicinal and aromatic herb, to add aroma and flavor to food (Vieira, Simon, 2000VIEIRA, R.F.; SIMON, J.E. Chemical characterization of basil (Ocimum spp.) found in the markets and used in traditional medicine in Brazil. Econ. Bot., v.54, n.2, p.207-216, 2000. ) basil contains several secondary metabolites like polyphenols, flavonoids, and terpenes, with recognized potential biologic effects that have been identified in this species (Lee, Scagel, 2009LEE, J.; SCAGEL, C.F. Chicoric acid found in basil (Ocimum basilicum L.) leaves. Food Chem., v.115, n.2, p.650-656, 2009.). Researchers have also tried to establish a standard of composition for basil, both in the extract and in oil, but the existence of more than 25 different types of O. basilicum, with many constitutional differences (Lee et al., 2005; Liber et al., 2011LIBER, Z.; CAROVIĆ-STANKO, K.; POLITEO, O.; STRIKIĆ, F.; KOLAK, I.; MILOS, M.; SATOVIC, Z. Chemical characterization and genetic relationships among Ocimum basilicum L. cultivars. Chem. Biodivers., v.8, n.11, p.1978-1989, 2011.) makes the task impossible.
The economic importance and global dissemination of basil, with its many uses in cooking and folk medicine, make it important to investigate its pharmacological and toxicological effects in order to ensure its efficacy and safety. Toxicity studies for these species are rare and do not focus on genetic aspects of toxicity to human cells. The main purpose of the present study was to evaluate the oxidative, genotoxic, and anti-inflammatory parameters that may be present in the extract from Basil leaves using human leukocyte cell cultures.
MATERIAL AND METHODS
Chemical, apparatus and general procedures
All chemicals were of analytical grade. Methanol, acetic acid, gallic acid, chlorogenic acid, and caffeic acid purchased from Merck (Darmstadt, Germany). Quercetin, rutin, rosmarinic acid, and kaempferol were acquired from Sigma Chemical Co. (St. Louis, MO, USA). High performance liquid chromatography (HPLC-DAD) was performed with a Shimadzu Prominence Auto Sampler (SIL-20A) HPLC system (Shimadzu, Kyoto, Japan) equipped with Shimadzu LC-20AT reciprocating pumps connected to a DGU 20A5 degasser with a CBM 20A integrator, SPD-M20A diode array detector, and LC solution 1.22 SP1 software.
Plant material
The commercial dry leaves of O. basilicum L., variety Genovese (Chá & Cia Produtos Naturais(r), Jacareí, SP), were purchased from a local market in Uruguaiana, RS, Brazil (latitude 29°45'17''; longitude 57°05'18''). The leaves were triturated and macerated at room temperature in hydroalcoholic solution (30 H2O: 70 ethanol v/v) at concentration of 20 g per 100 mL of solvent for one week under daily shaking. The maceration process was repeated for two more weeks to exhaustion of the vegetable material. At the end of three weeks, the filtrates were pooled and evaporated under reduced pressure in a rotary evaporator in order to remove ethanol and water. The dry extract of O. basilicum (ObE) was used in the following tests.
Determination of total polyphenolic contents
The total polyphenol concentration in the crude extract was measured spectrophotometrically using a modified Folin-Ciocalteau method (Chandra, Gonzalez De Mejia, 2004CHANDRA, S.; GONZALEZ DE MEJIA, E. Polyphenolic compounds, antioxidant capacity, and quinone reductase activity of an aqueous extract of Ardisia compressa in comparison to mate (Ilex paraguariensis) and green (Camellia sinensis) teas. J. Agric. Food Chem., v.52, n.11, p.3583-3589, 2004.). Briefly, we added 0.5 mL of 2 N Folin-Ciocalteau reagent to 1 mL of each sample (0.15 mg/mL) and allowed this mixture to stand for 5 min before adding 2 mL of 20% Na2CO3. Next we allowed the solution to stand for 10 min before reading it in a Shimadzu-UV-1201 spectrophotometer (Shimadzu, Kyoto, Japan) at 730 nm. We estimated the phenolic compounds in the crude extract in triplicate. Because of the chemical heterogeneity of plant products and the specificity of phenolic reagents, it is very difficult to choose suitable standards for identifying all phenols in a plant extract. Aqueous solutions of gallic acid in the range of 0.001-0.2 mg/mL as references was used. Thus, it is only possible to get relative equivalents with the standard used. The total polyphenol content was expressed as milligram equivalents of gallic acid per milliliter of the extract.
Determination of total flavonoids
Methanolic solutions of quercetin in the range of 4.0-12.0 µg/mL as references was used. To 2 mL of each reference solution, we added 20 mL of methanol and 1 mL of 5% methanolic solution of AlCl3 (w/v) and produced a total volume of 50 mL with methanol at 20 ºC. After 30 min, we measured the absorbance in a Shimadzu-UV-1201 spectrophotometer at 425 nm. We used the same procedure to analyze the crude extract. The blank was 5% AlCl3 (w/v) (Woisky, Salatino, 1998WOISKY, R.G.; SALATINO, A. Analysis of propolis: Some parameters and procedures for chemical quality control. J. Apic. Res., v.37, n.2, p.99-105, 1998.). We estimated the flavonoids in crude extract in triplicate. The contents were expressed as milligram equivalents of quercetin/ mL of the extracts.
High performance liquid chromatography
Apparatus: The extracts were examined with high performance liquid chromatography (HPLC) using an HPLC system (Shimadzu, Kyoto, Japan), Prominence auto sampler (SIL-20A), which was equipped with Shimadzu LC-20 AT reciprocating pumps connected to a degasser DGU 20A5 with an integrator CBM 20A, a UV-VIS detector DAD (diode) SPD-M20A, and the Software LC solution 1.22 SP1.
Analysis of phenolic compounds
Reverse phase chromatographic analyses were performed under gradient conditions using a C18 column (4.6 mm × 150 mm) packed with 5-μm diameter particles. The mobile phase was water containing 2% acetic acid (A) and methanol (B). The composition gradient was 5% of B to 2 min, when it was changed to obtain 25%, 40%, 50%, 60%, 70%, and 100% B at 10, 20, 30, 40, 50, and 80 min, respectively, following the method described by Laghari et al. (2011LAGHARI, A.H.; MEMON, S.; NELOFAR, A.; KHAN, K.M.; YASMIN, A. Determination of free phenolic acids and antioxidant activity of methanolic extracts obtained from fruits and leaves of Chenopodium album. Food Chem., v.126, n.4, p.1850-1855, 2011.) with slight modifications. The extracts were analyzed at a concentration of 0.150 mg/mL. We investigated the presence of the following six antioxidants: gallic acid, chlorogenic acid, caffeic acid, quercetin, rutin, and kaempferol. These compounds were identified by comparing their retention times and UV absorption spectra with those of the commercial standards. The flow rate was 0.7 mL/min and the injection volume was 40 μL. The wavelengths were 254 nm for gallic acid; 327 nm for caffeic and chlorogenic acids; and 365 nm for quercetin, rutin, and kaempferol. The samples and mobile phase were filtered through a 0.45-μm membrane filter (Millipore) and then degassed in an ultrasonic bath before use. Stock solutions of reference standards were prepared in the HPLC mobile phase for quercetin, rutin, and kaempferol at 0.020-0.200 mg/mL and for gallic, caffeic, and chlorogenic acids at 0.050-0.250 mg/mL. A chromatography peak was confirmed by DAD spectra (200-500 nm) and by comparing its retention time with that of a reference standard.
Cytotoxicity curve in leukocytes
Initially, the dose-effect cytotoxicity curve was determined in leukocytes using ObE dissolved in PBS Buffer pH 7.2 at doses ranging from 0.0001 mg/mL to 100 mg/mL, to determine the lethal dose 50% (LD50). Human leukocytes cultures were prepared using 0.5 mL of venous blood collected by venipuncture from a male volunteer (survey approved by the Ethics Committee of the Federal University of Santa Maria, approval letter number 23,081). They were centrifuged to separate leukocytes and immediately transferred to RPMI 1640 medium supplemented with 10% fetal bovine serum, 1% streptomycin/penicillin, and phytohemagglutinin according to a previous study described by Montagner et al. (2010MONTAGNER, G.F.F.S.; SAGRILLO, M.; MACHADO, M.M.; ALMEIDA, R.C.; MOSTARDEIRO, C.P.; DUARTE, M.M.; CRUZ, I.B. Toxicological effects of ultraviolet radiation on lymphocyte cells with different manganese superoxide dismutase Ala16Val polymorphism genotypes. Toxicol. In Vitro, v.24, n.5, p.1410-1416, 2010.). Cells were kept at 37 °C for 72 h. The analyzed parameter was cell proliferation according to Burow et al. (1998BUROW, M.E.; WELDON, C.B.; TANG, Y.; NAVAR, G.L.; KRAJEWSKI, S.; REED, J.C.; HAMMOND, T.G.; CLEJAN, S.; BECKMAN, B.S. Differences in susceptibility to tumor necrosis factor α-induced apoptosis among MCF-7 breast cancer cell variants. Cancer Res., v.58, n.21, p.4940-4946, 1998.).
Leukocytes culture sample preparations
To assess the anti-oxidative status and anti-genotoxic profiles, the leukocyte cultures were divided in six groups. The groups were a negative control (phosphate buffer pH 7.2); a positive control (hydrogen peroxide 100 μM); a group with rosmarinic acid at the concentration previously found in the plant by the phytochemical analysis; and three groups with different extract concentrations obtained from toxicity studies; that is, the LD50, LD50/10, and LD50/100. Groups with rosmarinic acid and extracts have also received 100 µM H2O2 to induce oxidation. To assess the inflammatory parameters the same groups division described above were used, with exception of positive control that has incorporated ibuprofen 100 μM. Additionally, all groups except the negative control received dextran 1% to induce the inflammatory process. The concentrations were selected to establish effective doses and exhibit low toxicity to leukocytes. All tests were performed in triplicate.
Evaluation of antioxidants parameters of ObE in human leukocytes cultures
To analyze the oxidative parameters, we used classical techniques as lipid peroxidation, protein carbonylation, ascorbic acid content, and superoxide dismutase and catalase activities. The assays were conducted in triplicates.
Lipid peroxidation
The extent of lipid peroxidation was estimated as the concentration of thiobarbituric acid reactive products (malondialdehyde) according to Ohkawa, Ohishi, Yagi (1979OHKAWA, H.; OHISHI, N.; YAGI, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem., v.95, n.2, p.351-358, 1979.). The method spectrophotometrically measures the color produced by the reaction of TBA with malondialdehyde (MDA) at 532 nm.
Protein carbonylation
The protocol was performed according to Morabito et al. (2004MORABITO, F.; CRISTANI, M.; SAIJA, A.; STELITANO, C.; CALLEA, V.; TOMAINO, A.; MINCIULLO, P.L.; GANGEMI, S. Lipid peroxidation and protein oxidation in patients affected by Hodgkin's lymphoma. Mediators Inflamm., v.13, n.5/6, p.381-383, 2004.). In this technique, carbonyl (CO) groups (aldehydes and ketones) are produced on protein side chains when they are oxidized, reacting with 2,4-dinitrophenylhydrazine, forming a color complex.
Ascorbic acid content
According to the method of Jacques-Silva et al. (2001) the curve of ascorbic acid was taken as a reference and the reference samples were mixed with trichloroacetic acid 13.3% and 2,4-dinitrophenylhydrazine. After incubation period, the samples were measured at 520 nm in spectrophotometer. The extracts were measured using the same procedure.
Catalase activity
Catalase activity was determined from the rate of decomposition of H2O2 (Aebi, 1984AEBI, H. Catalase in vitro. Methods Enzymol., v.105, p.121-126, 1984.). One unit of catalase activity was defined as the required activity to degrade one mol of hydrogen peroxide in 60s.
Superoxide dismutase activity
Superoxide dismutase (SOD) (E.C.1.15.1.1) activity was measured spectrophotometrically according to Boveris, Cadenas (1997BOVERIS, A.; CADENAS, E. Cellular sources and steady-state levels of reactive oxygen species. Lung Biol. Health Dis., v.105, p.1-26, 1997.). The technique is based on the inhibition of the reaction of superoxide anion with epinephrine. The oxidation reaction of epinephrine produces adrenocromo, which can be detected spectrophotometrically (480 nm). The enzyme activity was determined by measuring the rate of formation of adrenocromo. The reaction medium contains glycine-NaOH and epinephrine. A unit of enzyme activity is defined as the amount of enzymes required to inhibit the rate of epinephrine autoxidation by 50%.
Evaluation of anti-genotoxic parameters of ObE in human leukocytes cultures
The techniques used to evaluate the anti-genotoxic parameters were cell proliferation and unviability, DNA damage, chromosomal instability, and micronuclei frequency.
Cellular proliferation and viability
Viability is assessed by the loss of membrane integrity, using trypan blue (Burow et al., 1998BUROW, M.E.; WELDON, C.B.; TANG, Y.; NAVAR, G.L.; KRAJEWSKI, S.; REED, J.C.; HAMMOND, T.G.; CLEJAN, S.; BECKMAN, B.S. Differences in susceptibility to tumor necrosis factor α-induced apoptosis among MCF-7 breast cancer cell variants. Cancer Res., v.58, n.21, p.4940-4946, 1998.). In this technique, the same samples and respective concentrations are combined with Turk's solution (acetic acid 3% plus gentian violet 1% in water), and after three min, the sample is placed in a Neubauer chamber. The differentiation of living and dead cells is observed by the blue coloration of dead cells. A total of 300 cells are counted and the amount of total leukocytes (proliferation) is achieved through counting in a Neubauer chamber.
Alkaline comet DNA assay
This test was assayed following procedures of Singh et al. (1988SINGH, N.P.; MCCOY, M.T.; TICE, R.R.; SCHNEIDER, E.L. A simple technique for quantitation of low levels of DNA damage in individual cells. Exp. Cell Res., v.175, n.1, p.184-191, 1988.). After incubation, the samples (leukocytes) were mixed with low-melting point agarose and placed on a microscope slide pre-coated with normal melting point agarose. The slides were immersed in a lysis solution, and an electrophoresis was performed (20 min at 300 mA and 25 V). In the end, the slides were neutralized and left to dry overnight at room temperature. The dry slides were re-hydrated and then fixed for 10 min, and allowed to dry again. The last stage was the coloring and the use of stop solution. The slides were analyzed under blind conditions. DNA damage was given as DNA damage index (DI). The DNA damage was calculated from cells in different damage classes.
Chromosomal instability
Colcemid was added in each leukocyte culture and incubated at 37 °C during 60 min. After this, the cells were centrifuged at 1,800 rpm for 10 min. The cell pellets were re-suspended in hypotonic solution and incubated at 37 °C for 16 min. After a new centrifugation, the cell pellets were re-suspended in acetic acid: methanol (3:1) and poured into a flask containing a fixative solution (acetic acid and methanol 3:1) followed by centrifugation. The slides were prepared by dropping 3 or 4 drops of cell suspension into a cold, wet slide, which then was air dried. The cells were analyzed with 10× magnification to verify the density and distribution of metaphase chromosomes (Yunis, 1976YUNIS, J.J. High resolution of human chromosomes. Science, v.191, n.4233, p.1268-1270, 1976.).
Micronuclei frequency
The cells were placed in a conic tube with saline and centrifuged in 1,000 rpm for 5 min (this procedure was repeated). One milliliter with the cell pellet was kept in the tubes mixed with the pipette and spread over the slide (two per sample) and left to dry in room temperature. Slides were stained by panoptic method and then analyzed under optical microscopy in immersion lens. For each slide, 1,000 cells were counted (Schmid, 1975SCHMID, W. The micronucleus test. Mutat. Res., v.31, n.1, p.9-15, 1975.).
Evaluation of anti-inflammatory parameters of ObE in human leukocytes cultures
For the determination of tumor necrosis factor-α (TNF-α), interleukine-10 (IL-10), interleukine-6 (IL-6), inhibition of COX-2 activities, and nitric oxide production, measurements were made using ELISA kits according to the manufacturer's instructions. All tests were performed in triplicate. The results of these tests were expressed in percentage of production in relation to the negative control.
Statistical analysis
Data were expressed as mean ± standard deviation (SD). Comparisons between groups were performed using one-way analysis of variance (ANOVA), followed by post hoc of Bonferroni for multiple comparison tests. Nonlinear regression analysis was used to determine LD50. Results were considered statistically significant when p<0.05.
RESULTS AND DISCUSSION
Medicinal plants are rich sources of natural antioxidants and represent a promising perspective in the discovery of new drugs in the therapeutic arena. Most members of the Lamiaceae family have exhibited interesting biological effects due to their antioxidant compounds (Schofield, Mbugua, Pell, 2001SCHOFIELD, P.; MBUGUA, D.; PELL, A. Analysis of condensed tannins: a review. Anim. Feed Sci. Technol., v.91, n.1, p.21-40, 2001.).
Phenolic compounds are broadly distributed in the plant kingdom and are the most abundant secondary metabolites found in plants (Komali et al., 1999KOMALI, A.S.; ZHENG, Z.; SHETTY, K. A mathematical model for the growth kinetics and synthesis of phenolics in oregano (Origanum vulgare) shoot cultures inoculated with Pseudomonas species. Process Biochem., v.35, n.3, p.227-235, 1999.). The key role of phenolic compounds as antibacterial is emphasized in several reports (Møller et al., 1999MØLLER, J.K.S.; MADSEN, H.L.; AALTONEN, T.; SKIBSTED, L.H Dittany (Origanum dictamnus) as a source of water-extractable antioxidants. Food Chem., v.64, n.2, p.215-219, 1999.; Schofield, Mbugua, Pell, 2001SCHOFIELD, P.; MBUGUA, D.; PELL, A. Analysis of condensed tannins: a review. Anim. Feed Sci. Technol., v.91, n.1, p.21-40, 2001.). Flavonoids compounds occur naturally in plant foods and are a common component of our diet. Table I shows the data from analysis by HPLC-DAD method (Rice-Evans, Miller, Paganga, 1996) to determine the concentration of totals polyphenol and flavonoids and some compounds present in O. basilicum L, all described in literature as biologically active drugs.
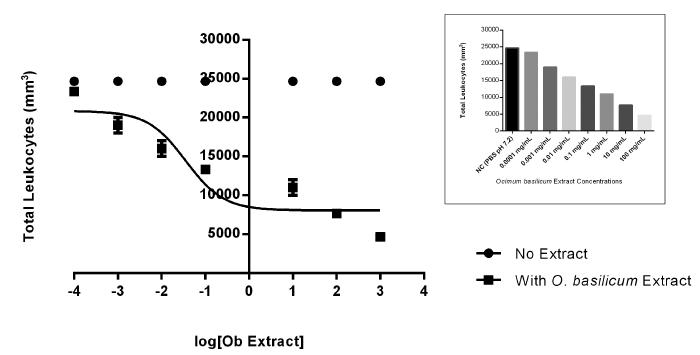
In Figure 1, we show the dose-effect curve. As shown below, when the dose of the extract is increased, the total number of leukocytes decreases, showing a dose-dependent effect. It is important to note that in LD50 concentration (35.44 µg/mL), the cellular unviability and DNA damage showed no significant changes (data not shown), which agrees with the results by Gomez-Flores et al. (2008). This result allowed us to establish the doses that were used in our protocols. Aiming to search for a dose with high affectivities and low toxicity, we determine the work doses for this protocol: 35.44 µg/mL (LD50), 3.544 µg/mL (LD50/10), and 0.3544 µg/mL (LD50/100).
Dose-effect curve using the crude extract of Ocimum basilicum in different doses (0.0001 mg/mL to 100 mg/mL) to determine the lethal dose 50 (LD50) in human leukocytes. The inset shows the total amount of leukocytes versus the tests concentrations of O. basilicum extract.
The accumulations of free radicals in organs or tissues are strongly associated with oxidative damages in biomolecules and cell membranes. This can lead to many chronic diseases, such as inflammatory, cancer, diabetes, aging, cardiac dysfunction, and other degenerative diseases (Wang et al., 2004WANG, S.; KONOREV, E.A.; KOTAMRAJU, S.; JOSEPH, J.; KALIVENDI, S.; KALYANARAMAN, B. Doxorubicin induces apoptosis in normal and tumor cells via distinctly different mechanisms intermediacy of H2O2-and p53-dependent pathways. J. Biol. Chem., v.279, n.24, p.25535-25543, 2004.). Figure 2 demonstrates the results found for oxidative parameters.
Effects of O. basilicum extract in oxidative parameters of human leukocytes in culture subject to hydrogen peroxide. In A: lipid peroxidation; B; carbonyl contents; C: ascorbic acid; D: catalase activity; E: superoxide dismutase activity. NC: negative control; PC: positive control; RA: rosmarinic acid; LD50: concentration equals to LD50 (35.44 µg/mL); LD50/10: 10 times less the concentration of LD50 (3.544 µg/mL); LD50/100: One hundred times less the concentration of LD50 (0.3544 µg/mL). Data are expressed as mean ± S.D. Results were confirmed by an experiment that was repeated three times. Different letters represent statistically different results among columns (p<0.05).
The involvement of reactive oxygen species in interactions with polyunsaturated fatty acids in cell membranes generates lipid peroxidation. The result is the formation of highly reactive hydro or lipoperoxides that may trigger the oxidative cascade, resulting in damage to membrane integrity (Tsang, Chung, 2009TSANG, A.H.; CHUNG, K.K. Oxidative and nitrosative stress in Parkinson's disease. Biochim. Biophys. Acta, v.1792, n.7, p.643-650, 2009.). The results found in lipid peroxidation show that LD50 and LD50/10 obtained values are close to the control of rosmarinic acid, a well-known antioxidant agent. Moreover, the values of peroxidation in LD50 3,576 nMol of MDA/mL in erythrocytes and LD50/10 4,226 nMol of MDA/mL in erythrocytes, were 41.81% and 31.29% lower when compared to the positive control, respectively. The content of protein carbonyl is the most general indicator and the most commonly used marker of protein oxidation, and its accumulation has been observed in several human diseases. The values found in protein carbonyl show that LD50 was 28% and LD50/10 18.8% lower when compared to the positive control, and approximate to negative control and rosmarinic acid control. Vitamin C or ascorbic acid has been implicated in different biological processes and plays an important role in oxidant defense. Ascorbic acid functions in several enzymatic steps, acting like a cofactor in the synthesis of collagen, monoamines, amino acids, peptide hormones, and carnitine. Samples with higher concentrations of extract showed higher concentrations of ascorbic acid. This fact is directly related to the concentrations of polyphenols and antioxidant action. The most concentrated samples, LD50 and DL50/10, have higher levels of polyphenols. Polyphenols are a first line of defense against oxidative action of hydrogen peroxide. When polyphenols decrease their concentrations, other non-enzymatic antioxidants come into play, thereby reducing the concentration of vitamin C (Kelm et al., 2000KELM, M.; NAIR, M.G.; STRASBURG, G.M.; DEWITT, D.L. Antioxidant and cyclooxygenase inhibitory phenolic compounds from Ocimum sanctum Linn. Phytomedicine, v.7, n.1, p.7-13, 2000.; Petersen, Simmonds, 2003PETERSEN, M.; SIMMONDS, M.S.J. Rosmarinic acid. Phytochemistry, v.62, n.2, p.121-125, 2003.).
Mammalian cells have elaborate antioxidant defense mechanisms to control damage effects of reactive oxygen species and the catalase enzyme is one of these, protecting cells against the toxic effects of hydrogen peroxide (Goyal, Basak, 2010GOYAL, M.M.; BASAK, A. Human catalase: looking for complete identity. Protein Cell, v.1, n.10, p.888-897, 2010.). The results of catalase analysis show the ability of O. basilicum extract in neutralizing the effects of hydrogen peroxide. The results were dose-related and show a better effect at LD50 concentration. Superoxide has been implicated in reactions associated with aging and in pathophysiological processes due to its transformation into more reactive species such as hydroxyl radical, which initiates lipid peroxidation.
According to our results, the activity of SOD enzyme was not affected in the three concentrations of O. basilicum extract. All these results are related to the presence of antioxidant compounds present in the extract (Table I), as the groups of polyphenols and flavonoids. This information confirms the data from the literature that phenolic compounds, especially flavonoids act in the oxidative metabolism, not by changing the enzymatic defenses, but by directing neutralizing of reactive species in order to stabilize them (Liu et al., 2008LIU, X.; CUI, C.; ZHAO, M.; WANG, J.; LUO, W.; YANG, B.; JIANG, Y. Identification of phenolics in the fruit of emblica (Phyllanthus emblica L.) and their antioxidant activities. Food Chem., v.109, n.4, p.909-915, 2008.).
We also evaluated the effects of hydrogen peroxide and the counter effects of O. basilicum extract on human leukocytes cells. The results of these markers are show in Figure 3.
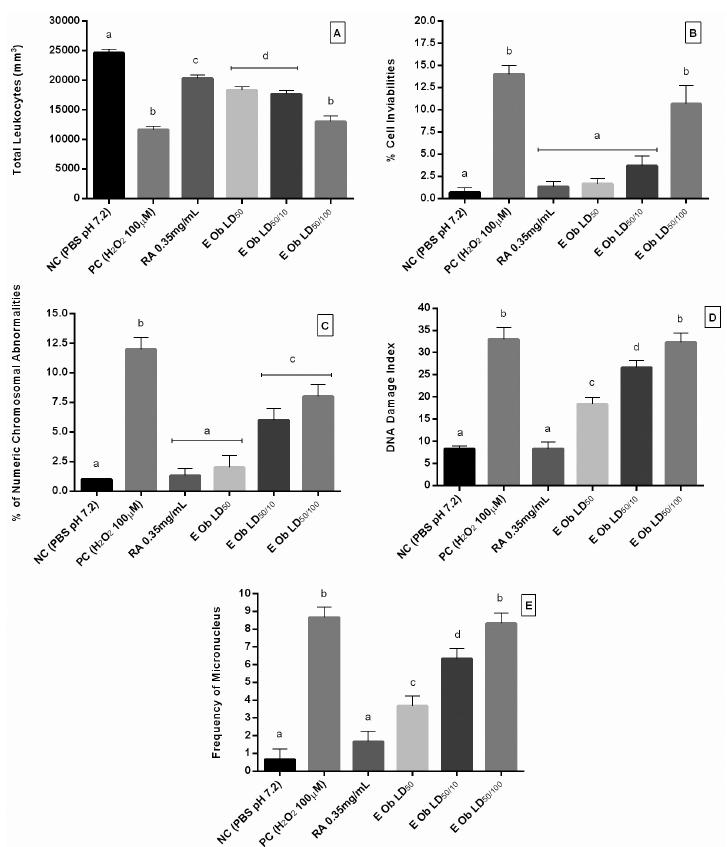
Effects of O. basilicum extract in anti-genotoxic parameters of human leukocytes in culture subject to hydrogen peroxide. In A: cell proliferation; B; cell inviability; C: percentage of abnormal chromossomics; D: DNA damage index; E: frequency of micronuclei. NC: negative control; PC: positive control; RA: rosmarinic acid; LD50: concentration equals to LD50 (35.44 µg/mL); LD50/10: 10 times less the concentration of LD50 (3.544 µg/mL); LD50/100: One hundred times less the concentration of LD50 (0.3544 µg/mL). Data are expressed as mean ± S.D. Results were confirmed by an experiment that was repeated three times. Different letters represent statistically different results among columns (p < 0.05).
In cellular proliferation (Figure 3A), cellular increase of the concentrations of LD50 and LD50/100 was similar to the rosmarinic acid control and different from the positive control, whereas the lowest concentration LD50/100 presented a decrease in cellular proliferation, which may be compared to the positive control. Figure 3B shows the percentage of unviable cells for both controls groups and for the three concentrations groups tested. The negative control presented only 0.667%, whereas positive control was 14% of unviable cells. Rosmarinic acid was 1.33% and the other three concentrations of O. basilicum: LD50: 1.667%; LD50/10: 3.67%; LD50/100: 10.67% of unviable cells. Shimizu et al. (2013SHIMIZU, T.; TORRES, M.P.; CHAKRABORTY, S.; SOUCHEK, J.J.; RACHAGANI, S.; KAUR, S.; MACHA, M.; GANTI, A.K.; HAUKE, R.J.; BATRA, S.K. Holy Basil leaf extract decreases tumorigenicity and metastasis of aggressive human pancreatic cancer cells in vitro and in vivo: potential role in therapy. Cancer Lett., v.336, n.2, p.270-280, 2013.) in a pancreatic cancer model, shows that Ocimum sanctum, belonging to the same plant family tested here, leaves inhibit the proliferation, migration, invasion, and induce apoptosis of cells in vitro. It is important to note that these results were obtained in cancer cells and it would be inaccurate to extrapolate this statement for normal leukocytes, having a functional metabolism.
According to Collins et al. (2008COLLINS, A.R.; OSCOZ, A.A.; BRUNBORG, G.; GAIVÃO, I.; GIOVANNELLI, L.; KRUSZEWSKI, M.; SMITH, C.C.; STETINA, R. The comet assay: topical issues. Mutagenesis, v.23, n.3, p.143-151, 2008.), the high viability of cells is required as a previous condition for the performance of the comet assay. Chromosomal instability results in numerical and structural chromosomal complexity and several studies associated these instabilities with poor prognosis in solid tumors (Birkbak et al., 2011BIRKBAK, N.J.; EKLUND, A.C.; LI, Q.; MCCLELLAND, S.E.; ENDESFELDER, D.; TAN, P.; TAN, I.B.; RICHARDSON, A.L.; SZALLASI, Z.; SWANTON, C. Paradoxical relationship between chromosomal instability and survival outcome in cancer. Cancer Res., v.71, n.10, p.3447-3452, 2011.).
The results demonstrate that all concentrations of extract of basil have presented percentages of chromosomal abnormalities (Figure 3C) similar to the negative control, and even the lowest concentration presented 33.334% less mitotic index when compared to positive control.
The comet assay is one of the most promising genotoxicity tests developed to measure and analyze DNA damage in single cells (Mukhopadhyay et al., 2004MUKHOPADHYAY, I.; CHOWDHURI, D.K.; BAJPAYEE, M.; DHAWAN, A. Evaluation of in vivo genotoxicity of cypermethrin in Drosophila melanogaster using the alkaline Comet assay. Mutagenesis, v.19, n.2, p.85-90, 2004.). The test was used as a parameter for assessing the DNA damage index (Figure 3D). As we can see at the lowest concentration, LD50/100, the cells presented the highest damage index compared with positive control, whereas the other two concentrations of extract, LD50 and LD50/10, showed damage indexes of 44.45% and 19.18% lower when compared with the positive control, respectively.
The micronuclei assay provides a convenient and reliable index of both chromosome breakage and chromosome loss. The micronuclei are expressed in dividing cells that either contain chromosome breaks lacking centromeres (acentric fragments) and/or whole chromosomes that are unable to migrate to the poles during mitosis (Fenech, 2000FENECH, M. The in vitro micronucleus technique. Mutat. Res., v.455, n.1/2, p.81-95, 2000.). The results show the micronucleus frequency (MN) found at the control groups and three concentrations of plant extract under analysis (Figure 3E). The frequency of MN was dose-dependent. The lowest concentration of the extract has shown the highest frequency of cells with this alteration.
Basil was frequently reported as a common anti-inflammatory (Narayanaswamy, Duraisamy, Balakrishnan, 2011NARAYANASWAMY, N.; DURAISAMY, A.; BALAKRISHNAN, K.P. Screening of some medicinal plants for their antityrosinase and antioxidant activities. Int. J. PharmTech. Res., v.3, p.1107-1112, 2011.). Aiming to confirm this activity and evaluate the mechanism involved, we perform a series of tests in human leukocytes cell exposed to a pro-inflammatory agent. The results are presented in Figure 4.
Effects of O. basilicum extract in inflammatory parameters of human leukocytes in culture subject to dextran solution. All the results are presented as percentage in relation to the negative control. In A: tumoral necrose factor-α (TNF-α); B; interleukine-10 (IL-10); C: interleukine-6 (IL-6); D: Ciclooxigenase type 2 activity (COX2 activity); E: nitric oxide (NO) production NC: negative control; PC: positive control; RA: rosmarinic acid; LD50: concentration equals to LD50 (35.44 µg/mL); LD50/10: 10 times less the concentration of LD50 (3.544 µg/mL); LD50/100: One hundred times less the concentration of LD50 (0.3544 µg/mL). Data are expressed as mean ± S.D. Results were confirmed by an experiment that was repeated three times. Different letters represent statistically different results among columns (p < 0.05).
Cytokines are subdivided in proinflammatory (initiate defense against pathogens) and anti-inflammatory (regulate the inflammatory process helping to balance the inflammatory response) acting with an important role in inflammatory response. The proinflammatory cytokines includes IL-1, IL-2, IL-6, IL-8, and TNF-α, and the anti-inflammatory cytokines includes IL-1 antagonist receptor, IL-4, IL-10, and IL-13 (Goldstein, Leung, Silverstein, 2006GOLDSTEIN, S.L.; LEUNG, J.C.; SILVERSTEIN, D.M. Pro-and anti-inflammatory cytokines in chronic pediatric dialysis patients: effect of aspirin. Clin. J. Am. Soc. Nephrol., v.1, n.5, p.979-986, 2006.). The results show that the production of the proinflammatory cytokines such as TNF-α and IL-6 was not affected in the three different doses of O. basilicum extract. However, the percentage of production of IL-10, the anti-inflammatory cytokine, shows an increase in the percentage of more than 60% at the highest dose of the extract when compared with the positive control (ibuprofen). The percentage of production of IL-10 also demonstrated to be dose-dependent, but in all concentrations, this production was higher than the negative and positive controls. The cyclooxygenases (COX) have been identified in three different isoforms. COX-2 acts like the inducible isoform, which is regulated by growth factors and different cytokines (IL1β, IL6, or TNFα) (Sobolewski et al., 2010SOBOLEWSKI, C.; CERELLA, C.; DICATO, M.; GHIBELLI, L.; DIEDERICH, M. The role of cyclooxygenase-2 in cell proliferation and cell death in human malignancies. Int. J. Cell Biol., v.2010, art.215158, p.1-21, 2010.). The ability of the extract from O. basilicum to inhibit COX-2 was determined and the results show that the extract was not capable to reduce the activity of the cyclooxygenase like the positive control (ibuprofen) but his activity decrease when compared to the negative control (100%) and was dose-dependent. The inhibition of COX-2 in LD50 and LD50/10 approximates to the inhibition caused by rosmarinic acid and several studies report many properties of rosmarinic acid including cyclooxygenase inhibition (Kelm et al., 2000KELM, M.; NAIR, M.G.; STRASBURG, G.M.; DEWITT, D.L. Antioxidant and cyclooxygenase inhibitory phenolic compounds from Ocimum sanctum Linn. Phytomedicine, v.7, n.1, p.7-13, 2000.; Petersen, Simmonds, 2003PETERSEN, M.; SIMMONDS, M.S.J. Rosmarinic acid. Phytochemistry, v.62, n.2, p.121-125, 2003.).
The percentage of nitric oxide generated was measured and cells pretreated with the three plant extracts showed a dose-dependent inhibition. The negative control was considered 100%. The maximal NO inhibitory effect was exhibited by O. basilicum at dose of LD50. Comparing to the negative control, the percentage of reduction was higher than 30% (34.67%). In a similar study using three plants of Lamiaceae family, O. basilicum extract demonstrated the higher content of phenols and maximal levels of DNA protection and free radical scavenging against toxicity induced by cadmium chloride (Thirugnanasampandan, Jayakumar, 2011THIRUGNANASAMPANDAN, R.; JAYAKUMAR, R. Protection of cadmium chloride induced DNA damage by Lamiaceae plants. Asian Pacific J. Trop. Biomed., v.1, n.5, p.391-394, 2011.). The results suggested that the anti-inflammatory activities of these extracts could be explained, at least in part, by their antioxidant properties. Rosmarinic acid was one of the most abundant caffeic acid esters present in O. basilicum (Jayasinghe et al., 2003JAYASINGHE, C.; GOTOH, N.; AOKI, T.; WADA, S. Phenolics composition and antioxidant activity of sweet basil (Ocimum basilicum L.). J. Agric. Food Chem., v.51, n.15, p.4442-4449, 2003.). It has been related that this compound has antioxidant, anti-HIV, and anti-inflammatory or cyclooxygenase and lipoxygenases inhibitory activities (Petersen, Simmonds, 2003PETERSEN, M.; SIMMONDS, M.S.J. Rosmarinic acid. Phytochemistry, v.62, n.2, p.121-125, 2003.).
CONCLUSIONS
It is possible to verify that O. basilicum extract acts as an antioxidant and effectively reverts or subjugates the effects of a high oxidizing agent such as hydrogen peroxide. These actions are explained by its composition, which is rich in polyphenols and flavonoids as well as compounds such as rosmarinic acid, which have a well-known antioxidant activity.
Our study shows that Basil extract possesses anti-inflammatory properties, and the mechanism involved is a composed interaction between the inhibition of pro-inflammatory mediator and the stimulation of anti-inflammatory cytokines.
Although pharmacodynamic studies are necessary to evaluate the activities in vivo, our results demonstrated that Basil could act as antioxidant and anti-inflammatory, becoming a possible alternative for medicinal treatment.
ACKNOWLEDGMENTS
The authors are thankful to the financial support of Fundação de Amparo a Pesquisa do Rio Grande do Sul (FAPERGS) and to Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and to Wiener Lab Group by the donation of kits for evaluations of laboratory parameters.
REFERENCES
- AEBI, H. Catalase in vitro. Methods Enzymol., v.105, p.121-126, 1984.
- BIRKBAK, N.J.; EKLUND, A.C.; LI, Q.; MCCLELLAND, S.E.; ENDESFELDER, D.; TAN, P.; TAN, I.B.; RICHARDSON, A.L.; SZALLASI, Z.; SWANTON, C. Paradoxical relationship between chromosomal instability and survival outcome in cancer. Cancer Res., v.71, n.10, p.3447-3452, 2011.
- BOVERIS, A.; CADENAS, E. Cellular sources and steady-state levels of reactive oxygen species. Lung Biol. Health Dis., v.105, p.1-26, 1997.
- BUROW, M.E.; WELDON, C.B.; TANG, Y.; NAVAR, G.L.; KRAJEWSKI, S.; REED, J.C.; HAMMOND, T.G.; CLEJAN, S.; BECKMAN, B.S. Differences in susceptibility to tumor necrosis factor α-induced apoptosis among MCF-7 breast cancer cell variants. Cancer Res., v.58, n.21, p.4940-4946, 1998.
- CHANDRA, S.; GONZALEZ DE MEJIA, E. Polyphenolic compounds, antioxidant capacity, and quinone reductase activity of an aqueous extract of Ardisia compressa in comparison to mate (Ilex paraguariensis) and green (Camellia sinensis) teas. J. Agric. Food Chem., v.52, n.11, p.3583-3589, 2004.
- CHOI, C.W.; KIM, S.C.; HWANG, S.S.; CHOI, B.K.; AHN, H.J.; LEE, M.Y.; PARK, S.H.; KIM, S.K. Antioxidant activity and free radical scavenging capacity between Korean medicinal plants and flavonoids by assay-guided comparison. Plant Sci., v.163, n.6, p.1161-1168, 2002.
- COLLINS, A.R.; OSCOZ, A.A.; BRUNBORG, G.; GAIVÃO, I.; GIOVANNELLI, L.; KRUSZEWSKI, M.; SMITH, C.C.; STETINA, R. The comet assay: topical issues. Mutagenesis, v.23, n.3, p.143-151, 2008.
- DANESI, F.; ELEMENTI, S.; NERI, R.; MARANESI, M.; D'ANTUONO, L.F.; BORDONI, A. Effect of cultivar on the protection of cardiomyocytes from oxidative stress by essential oils and aqueous extracts of basil (Ocimum basilicum L.). J. Agric. Food Chem., v.56, n.21, p.9911-9917, 2008.
- FENECH, M. The in vitro micronucleus technique. Mutat. Res., v.455, n.1/2, p.81-95, 2000.
- GOLDSTEIN, S.L.; LEUNG, J.C.; SILVERSTEIN, D.M. Pro-and anti-inflammatory cytokines in chronic pediatric dialysis patients: effect of aspirin. Clin. J. Am. Soc. Nephrol., v.1, n.5, p.979-986, 2006.
- GOMEZ-FLORES, R.; VERÁSTEGUI-RODRÍGUEZ, L.; QUINTANILLA-LICEA, R.; TAMEZ-GUERRA, P.; TAMEZ-GUERRA, R.; RODRÍGUEZ-PADILLA, C. In vitro rat lymphocyte proliferation induced by Ocinum basilicum, Persea americana, Plantago virginica, and Rosa spp. extracts. J. Med. Plants Res., v.2, n.1, p.5-10, 2008.
- GOYAL, M.M.; BASAK, A. Human catalase: looking for complete identity. Protein Cell, v.1, n.10, p.888-897, 2010.
- JACQUES-SILVA, M.C.; NOGUEIRA, C.W.; BROCH, L.C.; FLORES, E.M.; ROCHA, J.B. Diphenyl diselenide and ascorbic acid changes deposition of selenium and ascorbic acid in liver and brain of mice. Pharmacol. Toxicol., v.88, n.3, p.119-125, 2001.
- JAVANMARDI, J.; KHALIGHI, A.; KASHI, A.; BAIS, H.P.; VIVANCO, J.M. Chemical characterization of basil (Ocimum basilicum L.) found in local accessions and used in traditional medicines in Iran. J. Agric. Food Chem., v.50, n.21, p.5878-5883, 2002.
- JAYASINGHE, C.; GOTOH, N.; AOKI, T.; WADA, S. Phenolics composition and antioxidant activity of sweet basil (Ocimum basilicum L.). J. Agric. Food Chem., v.51, n.15, p.4442-4449, 2003.
- KELM, M.; NAIR, M.G.; STRASBURG, G.M.; DEWITT, D.L. Antioxidant and cyclooxygenase inhibitory phenolic compounds from Ocimum sanctum Linn. Phytomedicine, v.7, n.1, p.7-13, 2000.
- KOMALI, A.S.; ZHENG, Z.; SHETTY, K. A mathematical model for the growth kinetics and synthesis of phenolics in oregano (Origanum vulgare) shoot cultures inoculated with Pseudomonas species. Process Biochem., v.35, n.3, p.227-235, 1999.
- LAGHARI, A.H.; MEMON, S.; NELOFAR, A.; KHAN, K.M.; YASMIN, A. Determination of free phenolic acids and antioxidant activity of methanolic extracts obtained from fruits and leaves of Chenopodium album. Food Chem., v.126, n.4, p.1850-1855, 2011.
- LEE, J.; SCAGEL, C.F. Chicoric acid found in basil (Ocimum basilicum L.) leaves. Food Chem., v.115, n.2, p.650-656, 2009.
- LEE, S.-J.; UMANO, K.; SHIBAMOTO, T.; LEE, K.-G. Identification of volatile components in basil (Ocimum basilicum L.) and thyme leaves (Thymus vulgaris L.) and their antioxidant properties. Food Chem., v.91, n.1, p.131-137, 2005.
- LIBER, Z.; CAROVIĆ-STANKO, K.; POLITEO, O.; STRIKIĆ, F.; KOLAK, I.; MILOS, M.; SATOVIC, Z. Chemical characterization and genetic relationships among Ocimum basilicum L. cultivars. Chem. Biodivers., v.8, n.11, p.1978-1989, 2011.
- LIU, X.; CUI, C.; ZHAO, M.; WANG, J.; LUO, W.; YANG, B.; JIANG, Y. Identification of phenolics in the fruit of emblica (Phyllanthus emblica L.) and their antioxidant activities. Food Chem., v.109, n.4, p.909-915, 2008.
- MØLLER, J.K.S.; MADSEN, H.L.; AALTONEN, T.; SKIBSTED, L.H Dittany (Origanum dictamnus) as a source of water-extractable antioxidants. Food Chem., v.64, n.2, p.215-219, 1999.
- MONTAGNER, G.F.F.S.; SAGRILLO, M.; MACHADO, M.M.; ALMEIDA, R.C.; MOSTARDEIRO, C.P.; DUARTE, M.M.; CRUZ, I.B. Toxicological effects of ultraviolet radiation on lymphocyte cells with different manganese superoxide dismutase Ala16Val polymorphism genotypes. Toxicol. In Vitro, v.24, n.5, p.1410-1416, 2010.
- MORABITO, F.; CRISTANI, M.; SAIJA, A.; STELITANO, C.; CALLEA, V.; TOMAINO, A.; MINCIULLO, P.L.; GANGEMI, S. Lipid peroxidation and protein oxidation in patients affected by Hodgkin's lymphoma. Mediators Inflamm., v.13, n.5/6, p.381-383, 2004.
- MUKHOPADHYAY, I.; CHOWDHURI, D.K.; BAJPAYEE, M.; DHAWAN, A. Evaluation of in vivo genotoxicity of cypermethrin in Drosophila melanogaster using the alkaline Comet assay. Mutagenesis, v.19, n.2, p.85-90, 2004.
- NARAYANASWAMY, N.; DURAISAMY, A.; BALAKRISHNAN, K.P. Screening of some medicinal plants for their antityrosinase and antioxidant activities. Int. J. PharmTech. Res., v.3, p.1107-1112, 2011.
- OHKAWA, H.; OHISHI, N.; YAGI, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem., v.95, n.2, p.351-358, 1979.
- PETERSEN, M.; SIMMONDS, M.S.J. Rosmarinic acid. Phytochemistry, v.62, n.2, p.121-125, 2003.
- RICE-EVANS, C.A.; MILLER, N.J.; PAGANGA, G. Structure-antioxidant activity relationships of flavonoids and phenolic acids. Free Radicals Biol. Med., v.20, n.7, p.933-956, 1996.
- SAKR, S.A.; AL-AMOUDI, W.M. Effect of leave extract of Ocimum basilicum on deltamethrin induced nephrotoxicity and oxidative stress in albino rats. J. Appl. Pharm. Sci., v.2, n.5, 2012.
- SCHMID, W. The micronucleus test. Mutat. Res., v.31, n.1, p.9-15, 1975.
- SCHOFIELD, P.; MBUGUA, D.; PELL, A. Analysis of condensed tannins: a review. Anim. Feed Sci. Technol., v.91, n.1, p.21-40, 2001.
- SHIGA, T.; SHOJI, K.; SHIMADA, H.; HASHIDA, S.-N.; GOTO, F.; YOSHIHARA, T. Effect of light quality on rosmarinic acid content and antioxidant activity of sweet basil, Ocimum basilicum L. Plant Biotechnol., v.26, n.2, p.255-259, 2009. (Short Communication).
- SHIMIZU, T.; TORRES, M.P.; CHAKRABORTY, S.; SOUCHEK, J.J.; RACHAGANI, S.; KAUR, S.; MACHA, M.; GANTI, A.K.; HAUKE, R.J.; BATRA, S.K. Holy Basil leaf extract decreases tumorigenicity and metastasis of aggressive human pancreatic cancer cells in vitro and in vivo: potential role in therapy. Cancer Lett., v.336, n.2, p.270-280, 2013.
- SINGH, N.P.; MCCOY, M.T.; TICE, R.R.; SCHNEIDER, E.L. A simple technique for quantitation of low levels of DNA damage in individual cells. Exp. Cell Res., v.175, n.1, p.184-191, 1988.
- SOBOLEWSKI, C.; CERELLA, C.; DICATO, M.; GHIBELLI, L.; DIEDERICH, M. The role of cyclooxygenase-2 in cell proliferation and cell death in human malignancies. Int. J. Cell Biol., v.2010, art.215158, p.1-21, 2010.
- THIRUGNANASAMPANDAN, R.; JAYAKUMAR, R. Protection of cadmium chloride induced DNA damage by Lamiaceae plants. Asian Pacific J. Trop. Biomed., v.1, n.5, p.391-394, 2011.
- TSANG, A.H.; CHUNG, K.K. Oxidative and nitrosative stress in Parkinson's disease. Biochim. Biophys. Acta, v.1792, n.7, p.643-650, 2009.
- VIEIRA, R.F.; SIMON, J.E. Chemical characterization of basil (Ocimum spp.) found in the markets and used in traditional medicine in Brazil. Econ. Bot., v.54, n.2, p.207-216, 2000.
- WANG, S.; KONOREV, E.A.; KOTAMRAJU, S.; JOSEPH, J.; KALIVENDI, S.; KALYANARAMAN, B. Doxorubicin induces apoptosis in normal and tumor cells via distinctly different mechanisms intermediacy of H2O2-and p53-dependent pathways. J. Biol. Chem., v.279, n.24, p.25535-25543, 2004.
- WILLS, E.D.; THOMAS, J.H.; GILLHAM, B. Wills' biochemical basis of medicine. 2.ed. [S.l.]: Wright, 1989. 590p.
- WOISKY, R.G.; SALATINO, A. Analysis of propolis: Some parameters and procedures for chemical quality control. J. Apic. Res., v.37, n.2, p.99-105, 1998.
- YUNIS, J.J. High resolution of human chromosomes. Science, v.191, n.4233, p.1268-1270, 1976.
Publication Dates
-
Publication in this collection
2017
History
-
Received
28 May 2015 -
Accepted
18 Nov 2016