ABSTRACT
In recent years, there has been growing interest in clarifying the pathogenesis of some chronic diseases, such as obesity and type 2 diabetes mellitus. Metabolic alterations in these diseases are characterized by chronic hyperglycemia and insulin resistance. Studies have demonstrated the participation of minerals in the pathogenesis of insulin resistance, more specifically their involvement in the synthesis and regulation of insulin. Selenium is an anti-inflammatory and antioxidant micronutrient that is essential for the activity of selenoproteins. Two selenoproteins (glutathione peroxidase and selenoprotein P) are known to be involved in the insulin signaling pathway. The aim of this review is to provide an update on the role of selenium in insulin resistance mechanisms. Evidence shows that adequate concentrations of selenium play a key role in the secretion and action of insulin, but an excess of selenium in the body is associated with the pathogenesis of insulin resistance and the development of diabetes mellitus.
Keywords:
Diabetes mellitus; Selenium/insulin resistance; Insulin
INTRODUCTION
In recent years, the pathogenesis of some chronic diseases has attracted interest, such as obesity and type 2 diabetes mellitus. Metabolic alterations in these diseases are characterized by chronic hyperglycemia and insulin resistance (Rains, Jain, 2011Rains JL, Jain SK. Oxidative stress, insulin signaling, and diabetes. Free Radic Biol Med. 2011;50(5):567-75.; Steinbrenner, 2013Steinbrenner, H. Interference of selenium and selenoproteins with the insulin-regulated carbohydrate and lipid metabolism. Free Radic Biol Med. 2013;65:1538-47.).
Recent studies have attempted to elucidate the molecular alterations involved in the development of insulin resistance. These include reductions in the number and activity of relevant receptors in peripheral tissues, and changes to intracellular insulin signaling pathways (Tencerova et al., 2015Tencerova M, Aouadi M, Vangala P, Nicoloro SM, Yawe JC, Cohen JL, et al. Activated Kupffer cells inhibit insulin sensitivity in obese mice. FASEB J. 2015;29(7):2959-69.; Winnier et al., 2015Winnier DA, Fourcaudot M, Norton L, Abdul-Ghani MA, Hu SL, Farook VS, et al. Transcriptomic identification of ADH1B as a novel candidate gene for obesity and insulin resistance in human adipose tissue in Mexican Americans from the Veterans Administration Genetic Epidemiology Study (VAGES). PLoS One. 2015;10(4):e0119941.). Accordingly, minerals have been the subject of several studies, focusing specifically on their involvement in the synthesis and regulation of insulin (Cruz et al., 2014Cruz KJ, Oliveira AR, Pinto DP, Morais JB, Lima FS, Colli C, et al. Influence of magnesium on insulin resistance in obese women. Biol Trace Elem Res. 2014;160(3):305-10.; Garcia et al., 2013García OP, Ronquillo D, Caamaño MC, Martínez G, Camacho M, López V, et al. Zinc, iron and vitamins A, C and E are associated with obesity, inflammation, lipid profile and insulin resistance in mexican school-aged children. Nutrients. 2013;5(12):5012-30.).
Selenium is an anti-inflammatory and antioxidant micronutrient essential for the activity of selenoproteins, such as glutathione peroxidase (GPx) and selenoprotein P (Sepp). These enzymes are also involved in insulin regulation (Cominetti et al., 2011Cominetti C, Bortoli MC, Abdalla DSP, Cozzolino SMF. Considerations about oxidative stress, selenium and nutrigenetics. Rev Nutr. 2011;36(3):131-53.; Steinbrenner et al., 2013Steinbrenner, H. Interference of selenium and selenoproteins with the insulin-regulated carbohydrate and lipid metabolism. Free Radic Biol Med. 2013;65:1538-47.; Wei et al., 2014Wei Z, Yao M, Li Y, He X, Yang Z. Dietary selenium deficiency exacerbates lipopolysaccharide-induced inflammatory response in mouse mastitis models. Inflammation. 2014;37(6):1925-31.; Zou et al., 2016Zou C, Qiu Q, Chen H, Dou L, Liang, J. Hepatoprotective effects of selenium during diabetes in rats. Hum Exp Toxicol. 2016;35(2):114-23.). Several studies have revealed selenium to be an insulin-mimetic, because it plays roles in the regulation of enzymes in the insulin signaling cascade, the expression of lipogenic enzymes, and in carbohydrate metabolism in the liver (Chen et al., 2015Chen H, Qiu Q, Zou C, Dou L, Liang J. Regulation of hepatic carbohydrate metabolism by selenium during diabetes. Chem Biol Interact. 2015;232:1-6.; Iizuka et al., 2010Iizuka Y, Ueda Y, Yagi Y, Sakurai E. Significant improvement of insulin resistance of GK rats by treatment with sodium selenate. Biol Trace Elem Res. 2010;138(1-3):265-71.; Mao, Teng, 2013Mao J, Teng W. The relationship between selenoprotein P and glucose metabolism in experimental studies. Nutrients. 2013;5(6):1937-1948.).
However, it has been emphasized that chronic ingestion of large amounts of selenium promotes the formation of antioxidant selenoproteins, leading to reduced concentrations of reactive oxygen species, which are required in physiological amounts for insulin signaling (Labunskyy et al., 2011Labunskyy VM, Lee BC, Handy DE, Loscalzo J, Hatfield DL, Gladyshev VN. Both maximal expression of selenoproteins and selenoprotein deficiency can promote development of type 2 diabetes-like phenotype in mice. Antioxid Redox Signal. 2011;14(12):2327-36.; Wang et al., 2014Wang X, Zhang W, Chen H, Liao N, Wang Z, Zhang X et al. High selenium impairs hepatic insulin sensitivity through opposite regulation of ROS. Toxicol Lett. 2014;224(1):16-23.; Zhou et al., 2015Zhou J, Xu G, Bai Z, Li K, Yan J, Li F, et al. Selenite exacerbates hepatic insulin resistance in mouse model of type 2 diabetes through oxidative stress-mediated JNK pathway. Toxicol Appl Pharmacol. 2015;289(3):409-18.).
Given that insulin resistance is a key metabolic disorder that can result in the development of chronic diseases such as type 2 diabetes mellitus, and that selenium participates in the prevention of this disease, we have reviewed the participation of this micronutrient in insulin resistance.
Etiologic factors of insulin resistance
Insulin has a crucial function in glucose homeostasis; it regulates the balance between glucose production by the liver, and glucose uptake by muscle and adipose tissue. In myocytes and adipocytes, insulin regulates glucose transport by controlling the translocation of a glucose transporter (GLUT4) to the plasma membrane (Bruce, Hanson, 2010Bruce KD, Hanson MA. The developmental origins, mechanisms, and implications of metabolic syndrome. J Nutr. 2010;140(3):648-52.; Kim, Feldman, 2015Kim B, Feldman EL. Insulin resistance as a key link for the increased risk of cognitive impairment in the metabolic syndrome. Exp Mol Med. 2015;47(3):e149.).
The insulin receptor is a tetramer composed of two extracellular α-subunits and two transmembrane β-subunits. The action of insulin is initiated through binding to the α-subunits, which alters the conformation of the β-subunits. This results in autophosphorylation and activation of its tyrosine kinase function. The β-subunit can autophosphorylate, and it can phosphorylate other proteins or intracellular substrates, such as insulin receptor substrate 1 (IRS-1), insulin receptor substrate 2 (IRS-2), and the Src homology collagen (Shc) family of proteins (Hirabara et al., 2014; Litvinova et al., 2014Litvinova LS, Kirienkova EV, Mazunin IO, Vasilenko MA, Fattakhov NS. Pathogenesis of insulin resistance in metabolic obesity. Biochemistry (Moscow). 2014;8(3):191-202.).
The action of insulin is mediated by three main signaling cascades: the phosphatidyl inositol 3-kinase (PI3K)/protein kinase B (Akt) pathway, the Cbl associated protein (CAP)/Cbl pathway, and the Ras/mitogen-activated protein kinase (MAPK) pathway. These pathways regulate relevant cellular processes, such as glucose uptake, protein synthesis, and the expression of genes involved in cellular proliferation and differentiation (Litvinova et al., 2014Litvinova LS, Kirienkova EV, Mazunin IO, Vasilenko MA, Fattakhov NS. Pathogenesis of insulin resistance in metabolic obesity. Biochemistry (Moscow). 2014;8(3):191-202.).
Phosphorylation of IRS-1 (a process performed by various protein kinases, particularly PI3K) results in the formation of binding sites for proteins containing the Src homology 2 domain (SH2). This activation is required for stimulation of glucose transport by insulin, and is sufficient to induce GLUT4 translocation to the plasma membrane, at least partially. After phosphorylation by PI3K, IRS-1 begins to activate other cytoplasmic substrates, such as serine kinases, protein kinase B (Akt), and atypical isoforms of protein kinase C (aPKCζ and aPKCι/λ). Once phosphorylated, these proteins participate in insulin signal transduction pathways for glucose transport (Kwon, Pessin, 2013Kwon H, Pessin JE. Adipokines mediate inflammation and insulin resistance. Front Endocrinol (Lausanne). 2013;4:71.).
In addition to PI3K activation, other signals are required for insulin-stimulated glucose transport. In the second pathway, activation of the insulin receptor leads to phosphorylation of the Cbl oncogene. In most insulin-sensitive tissues, Cbl is associated with the adapter protein CAP. After phosphorylation, the Cbl-CAP complex migrates to the cell membrane and interacts with the CrkII protein, which in turn is associated with the C3G protein. C3G is a nucleotide exchanger that catalyzes the conversion of GDP to GTP in the TC10 protein, thus activating it. Once activated, TC10 generates a second signal for translocation of the GLUT4 protein, in parallel with the activation of PI3K (Hirabara et al., 2012Hirabara SM, Gorjão R, Vinolo MA, Rodrigues AC, Nachbar RT, Curi, R. Molecular targets related to inflammation and insulin resistance and potential interventions. J Biomed Biotechnol. 2012;2012:379024.; Litvinova et al., 2014Litvinova LS, Kirienkova EV, Mazunin IO, Vasilenko MA, Fattakhov NS. Pathogenesis of insulin resistance in metabolic obesity. Biochemistry (Moscow). 2014;8(3):191-202.).
In a similar manner to other growth factors, insulin stimulates MAPK. This pathway begins with the phosphorylation of IRS and/or Shc proteins, which interact with growth factor receptor binding protein 2 (Grb2). The latter is constitutively associated with Son of Sevenless (SOS), which is a protein that exchanges GDP for GTP in Ras (a GTP-binding switch protein), thus activating it. Activation of Ras requires the participation of Src homology phosphatase 2 (SHP2). Once activated, Ras stimulates serine phosphorylation of the MAPK cascade, which promotes cellular proliferation and/or differentiation. Pharmacological blockade of this pathway prevents the effect of insulin on cell growth, but has no effect on insulin’s metabolic properties (Hirabara et al., 2012Hirabara SM, Gorjão R, Vinolo MA, Rodrigues AC, Nachbar RT, Curi, R. Molecular targets related to inflammation and insulin resistance and potential interventions. J Biomed Biotechnol. 2012;2012:379024.).
The term insulin resistance refers to the reduced sensitivity of peripheral tissues to the action of insulin, which is caused by changes in insulin-mediated signaling pathways, and results in systemic hyperglycemia. Causes of insulin resistance include various metabolic aberrations that reduce the capacity of tissues to respond to insulin, especially in the liver, adipose tissue, and muscle (Tencerova et al., 2015Tencerova M, Aouadi M, Vangala P, Nicoloro SM, Yawe JC, Cohen JL, et al. Activated Kupffer cells inhibit insulin sensitivity in obese mice. FASEB J. 2015;29(7):2959-69.; Winnier et al., 2015Winnier DA, Fourcaudot M, Norton L, Abdul-Ghani MA, Hu SL, Farook VS, et al. Transcriptomic identification of ADH1B as a novel candidate gene for obesity and insulin resistance in human adipose tissue in Mexican Americans from the Veterans Administration Genetic Epidemiology Study (VAGES). PLoS One. 2015;10(4):e0119941.).
At various stages of insulin resistance, some tissues, such as skeletal muscle and adipose tissues, regulate GLUT4 expression differently. In the adipocytes of obese individuals, the concentration of GLUT4 is reduced, while concentrations in the skeletal muscle are normal. Since skeletal muscle is the main site for insulin-stimulated glucose consumption, changes in systemic sensitivity to insulin in obese and diabetic individuals cannot be explained by the reduced production of GLUT4. Instead, insulin resistance in these patients is explained by the low glucose uptake in skeletal muscle, which is due to the reduced translocation of GLUT4 from intracellular vesicles to the cell membrane (Muntoni, Muntoni, 2011Muntoni S, Muntoni, S. Insulin resistance: pathophysiology and rationale for treatment. Ann Nutr Metab. 2011;58(1):25-36.; Slattery et al., 2014Slattery MJ, Bredella MA, Thakur H, Torriani M, Misra M. Insulin resistance and impaired mitochondrial function in obese adolescent girls. Metab Syndr Relat Disord. 2014;12(1):56-61.).
It is important to note that regulation of the cellular redox balance is necessary to maintain and adapt insulin secretion, and to promote sensitivity of the target tissues to insulin. In this sense, oxidative stress is related to the multifactorial etiology of insulin resistance, with effects mainly observed in skeletal muscle tissues (Steinbrenner, 2013Steinbrenner, H. Interference of selenium and selenoproteins with the insulin-regulated carbohydrate and lipid metabolism. Free Radic Biol Med. 2013;65:1538-47.; Henriksen, Diamond-Stanic, Marchionne, 2011Henriksen EJ, Diamond-Stanic MK, Marchionne EM. Oxidative stress and the etiology of insulin resistance and type 2 diabetes. Free Radic Biol Med. 2011;51(5):993-9.).
Several systemic and cellular problems may contribute to increased production of reactive oxygen species, such as hyperglycemia, dyslipidemia, endoplasmic reticulum stress, advanced glycation end products, and lipid peroxides. These conditions can activate factors that are associated with reduced insulin action. These factors include the impairment of insulin signaling by phosphorylation of IRS on serine and threonine residues, alteration of cellular redistribution of insulin signaling components, decreased transcription of the GLUT4 gene, and changes in mitochondrial activity (Henriksen, Diamond-Stanic, Marchionne, 2011Henriksen EJ, Diamond-Stanic MK, Marchionne EM. Oxidative stress and the etiology of insulin resistance and type 2 diabetes. Free Radic Biol Med. 2011;51(5):993-9.; Rains, Jain, 2011Rains JL, Jain SK. Oxidative stress, insulin signaling, and diabetes. Free Radic Biol Med. 2011;50(5):567-75.; Steinbrenner, 2013Steinbrenner, H. Interference of selenium and selenoproteins with the insulin-regulated carbohydrate and lipid metabolism. Free Radic Biol Med. 2013;65:1538-47.).
In this regard, two mechanisms of reactive oxygen species production appear to be most relevant to the etiology of insulin resistance: the excess mitochondrial production of hydrogen peroxide (H2O2), and the increased activation of NADPH oxidase. Excess H2O2 production promotes mitochondrial dysfunction, which can affect insulin secretion in pancreatic β-cells. Excess H2O2 production is also involved in insulin resistance in skeletal muscle, particularly among individuals with high-energy expenditure, such as increased lipid intake. The increase in NADPH oxidase activation is mediated by angiotensin II receptors, which generate superoxide anions and impair insulin signaling by disrupting the translocation of GLUT4 (Henriksen, Diamond-Stanic, Marchionne, 2011Henriksen EJ, Diamond-Stanic MK, Marchionne EM. Oxidative stress and the etiology of insulin resistance and type 2 diabetes. Free Radic Biol Med. 2011;51(5):993-9.; Rains, Jain, 2011Rains JL, Jain SK. Oxidative stress, insulin signaling, and diabetes. Free Radic Biol Med. 2011;50(5):567-75.).
Selenium and insulin resistance
Changes in selenium status appear to contribute significantly to the development of insulin resistance (Feng et al., 2015Feng W, Cui X, Liu B, Liu C, Xiao Y, Lu W, et al. Association of urinary metal profiles with altered glucose levels and diabetes risk: a population-based study in china. PLoS One. 2015;10(4):e0123742.; Kim, Song, 2014Kim HN, Song SW. Concentrations of chromium, selenium, and copper in the hair of viscerally obese adults are associated with insulin resistance. Biol Trace Elem Res. 2014;158(2):152-7.; Pounis et al., 2014Pounis G, Costanzo S, Persichillo M, Curtis A, Sieri S, Vinceti M et al. Mushroom and dietary selenium intakes in relation to fasting glucose levels in a free-living Italian adult population: The Moli-sani Project. Diabetes Metab. 2014;40(1):34-42.). Previous research has shown that dietary supplementation with selenium can reduce fasting serum insulin concentrations and the insulin resistance index (Alizadeh et al., 2012Alizadeh M, Safaeiyan A, Ostadrahimi A, Estakhri R, Daneghian S, Ghaffari A, et al. Effect of L-arginine and selenium added to a hypocaloric diet enriched with legumes on cardiovascular disease risk factors in women with central obesity: a randomized, double-blind, placebo-controlled trial. Ann Nutr Metab. 2012;60(2):157-68.; Bahmani et al., 2016Bahmani F, Kia M, Soleimani A, Asemi Z, Esmaillzadeh A. Effect of selenium supplementation on glycemic control and lipid profiles in patients with diabetic nephropathy. Biol Trace Elem. 2016;172(2):282-9.; Farrokhian et al., 2016Farrokhian A, Bahmani F, Taghizadeh M, Mirhashemi SM, Aarabi MH, Raygan F, et al. Selenium supplementation affects insulin resistance and serum hs-CRP in patients with type 2 diabetes and coronary heart disease. Horm Metab Res. 2016;48(4):263-268.). The main findings of studies that have investigated the role of selenium on insulin resistance are described in Tables I and II.
Selenium’s antidiabetic action appears to result from its behaviour as an insulin-mimetic and as an antioxidant nutrient, since insulin signaling, and secretion is associated with the cellular redox state (Mao, Teng, 2013Mao J, Teng W. The relationship between selenoprotein P and glucose metabolism in experimental studies. Nutrients. 2013;5(6):1937-1948.; Rayman, Stranges, 2013Rayman MP, Stranges S. Epidemiology of selenium and type 2 diabetes: Can we make sense of it? Free Radic Biol Med. 2013;65:1557-64.; Steinbrenner, 2013Steinbrenner, H. Interference of selenium and selenoproteins with the insulin-regulated carbohydrate and lipid metabolism. Free Radic Biol Med. 2013;65:1538-47.). In a study performed by Barakat, Moustafa and Bikhazi (2012Barakat GM, Moustafa ME, Bikhazi AB. Effects of selenium and exendin-4 on glucagon-like peptide-1 receptor, IRS-1, and Raf-1 in the liver of diabetic rats. Biochem Genet. 2012;50(11-12):922-35.), it was found that 4 weeks of treatment with sodium selenite (5 ppm in drinking water) increased mRNA and protein levels of glucagon-like peptide-1 receptor (GLP-1R) in the liver of diabetic rats, which may have favored the reduction of blood glucose levels. It is worth mentioning that GLP-1 appears to reduce hepatic glucose production by promoting glycogen synthesis (D’Alessio, Vahl, Prigeon, 2004D'Alessio D, Vahl T, Prigeon R. Effects of Glucagon-like Peptide 1 on the hepatic glucose metabolism. Horm Metab Res. 2004;36(11-12):837-41.).
In a subsequent study using the same experimental model, these researchers investigated the effects of selenium treatment in the pancreas of diabetic rats. Increases in GLP-1R, IRS-1 and preproinsulin mRNA levels were found. Moreover, selenium treatment improved the number of endocrine islets, with restoration of normal cellularity and viable cells in the pancreatic tissue (Barakat et al., 2016Barakat GM, Moustafa ME, Khalifeh I, Hodroj MH, Bikhazi A, Rizk S. Effects of exendin-4 and selenium on the expression of GLP-1R, IRS-1, and preproinsulin in the pancreas of diabetic rats. J Physiol Biochem. 2016;73(3):387-94.). The effects of GLP-1 on the biosynthesis and secretion of insulin, as well as the proliferation, differentiation and apoptosis of pancreatic β cells, are well known (Lee, Jun, 2014Lee YS, Jun HS. Anti-diabetic actions of glucagon-like peptide-1 on pancreatic beta-cells. Metabolism. 2014;63(1):9-19.). In this regard, these results suggest a possible role of selenium in the physiological actions of incretin hormones.
Similarly, Campbell et al. (2008Campbell SC, Aldibbiat A, Marriott CE, Landy C, Ali T, Ferris WF, et al. Selenium stimulates pancreatic beta-cell gene expression and enhances islet function. FEBS Lett. 2008;582(15):2333-7.) found that selenium participates in insulin’s action on β-pancreatic cells, through its ability to regulate the gene expression of insulin promoter factor 1, and to increase the mRNA expression of insulin in the mouse beta cell line Min6. Selenium promotes the increased production and secretion of insulin in these cells.
In a study by Iukuza et al. (2010), 0.173 mg/kg of sodium selenite (Na2SeO3) was administered intraperitoneally to diabetic rats for 14 days. The result was increased glucose uptake by peripheral tissues and adipocytes. This was probably due to the action of selenium on the insulin receptor, thus increasing sensitivity to insulin and alleviating hyperglycemia, for regulation of hepatic glucose production.
It is important to mention selenium’s role in the regulation of the hepatic metabolism of carbohydrates. Chen et al. (2015Chen H, Qiu Q, Zou C, Dou L, Liang J. Regulation of hepatic carbohydrate metabolism by selenium during diabetes. Chem Biol Interact. 2015;232:1-6.) found that the administration of selenium to diabetic rats (through a 1 ppm dose in their drinking water) increased cellular glucose uptake and restored the activity of glucose-6-phosphatase and glycogen phosphorylase (enzymes involved in the hepatic glucose production) to normal levels.
Pancreatic β-cells are known for their reduced antioxidant protection, probably due to the important role played by physiological concentrations of reactive oxygen species in insulin signaling pathways. Research has revealed lower expression of GPx selenoproteins, and higher expression of Sepp1 in these cells. These findings are suggestive of a possible role of selenium in the regulation of the redox state of pancreatic β-cells (Lei, Vatamaniuk, 2011Lei XG, Vatamaniuk MZ. Two tales of antioxidant enzymes on ß cells and diabetes. Antioxid Redox Signal. 2011;14(3):489-503.; Steinbrenner et al., 2013Steinbrenner, H. Interference of selenium and selenoproteins with the insulin-regulated carbohydrate and lipid metabolism. Free Radic Biol Med. 2013;65:1538-47.).
A study by Steinbrenner et al. (2013Steinbrenner, H. Interference of selenium and selenoproteins with the insulin-regulated carbohydrate and lipid metabolism. Free Radic Biol Med. 2013;65:1538-47.) showed that Sepp1 can provide additional antioxidant protection to the islets of Langerhans, serving as a selenium donor for the function of other antioxidant selenoproteins such as Gpx, and participating in the protection of cell membranes from oxidative damage. Sepp1 is also located in glucagon-secreting cells. Nevertheless, elevated glucose concentrations (11-22 mM) appear to dysregulate the expression of Sepp1 in β-cells, which can reduce antioxidant function and trigger endocrine dysfunction.
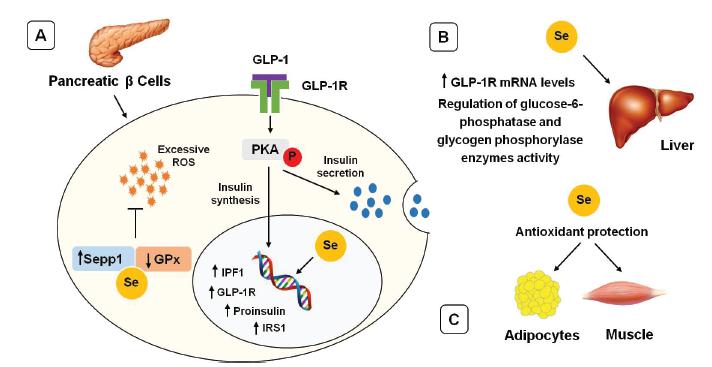
Labunskyy et al. (2011Labunskyy VM, Lee BC, Handy DE, Loscalzo J, Hatfield DL, Gladyshev VN. Both maximal expression of selenoproteins and selenoprotein deficiency can promote development of type 2 diabetes-like phenotype in mice. Antioxid Redox Signal. 2011;14(12):2327-36.) found that a deficiency of Sepp1 and methionine sulfoxide reductase 1 (MsrB1) in transgenic mice induces glucose intolerance and hyperinsulinemia. Research by Zou et al. (2016Zou C, Qiu Q, Chen H, Dou L, Liang, J. Hepatoprotective effects of selenium during diabetes in rats. Hum Exp Toxicol. 2016;35(2):114-23.) showed that selenium treatment (a 1 ppm dose in drinking water) in diabetic rats reduces serum glucose concentration, lipid peroxidation, and the activity of the enzymes aspartate aminotransferase, alanine aminotransferase, and alkaline phosphatase. The authors also demonstrated that selenium supplementation increased the activity of major antioxidant enzymes, such as GPx, superoxide dismutase, and catalase. Figure 1 shows the participation of selenium in protection from insulin resistance.
The role of selenium in protection from insulin resistance. Overall, selenium appears to improve insulin resistance through three mechanisms. A) Firstly, in β-pancreatic cells, selenoproteins such as Sepp1 and Gpx exert antioxidant action, thereby inhibiting the excessive production of reactive oxygen species, which may impair the insulin signaling pathway. Moreover, selenium promotes the transcription and synthesis of proinsulin, insulin promoter factor 1, and GLP-1R. In the figure, we highlight the participation of the GLP-1-activated protein kinase A pathway in the regulation of insulin secretion and synthesis. B) Secondly, in the liver, selenium influences hepatic glucose production, by regulating glucose-6-phosphatase and glycogen phosphorylase enzymatic activity, and increasing GLP-1R expression. C) Thirdly, in peripheral organs, selenium improves insulin sensitivity due to its antioxidant action. GLP-1: glucagon-like peptide 1; GLP-1R: GLP-1 receptor; GPx: glutathione peroxidase; IPF1: insulin promoter factor 1; IRS-1: insulin receptor substrate 1; P: phosphorylation; PKA: protein kinase A; ROS: reactive oxygen species; Se: selenium; Sepp1: selenoprotein P1.
On the other hand, selenium may also impair insulin’s action in target cells. Some studies have shown an association of high dietary selenium intake with increased risk of diabetes and insulin resistance (Table I and II) (Pounis et al., 2014Pounis G, Costanzo S, Persichillo M, Curtis A, Sieri S, Vinceti M et al. Mushroom and dietary selenium intakes in relation to fasting glucose levels in a free-living Italian adult population: The Moli-sani Project. Diabetes Metab. 2014;40(1):34-42.; Stranges et al., 2007Stranges S, Marshall JR, Natarajan R, Donahue RP, Trevisan M, Combs GF, et al. Effects of long-term selenium supplementation on the incidence of type 2 diabetes: a randomized trial. Ann Intern Med. 2007;147(4):217-23.). The negative action of selenium on insulin signaling pathways appears to relate to dysregulation of the cellular redox state (Labunskyy et al., 2011Labunskyy VM, Lee BC, Handy DE, Loscalzo J, Hatfield DL, Gladyshev VN. Both maximal expression of selenoproteins and selenoprotein deficiency can promote development of type 2 diabetes-like phenotype in mice. Antioxid Redox Signal. 2011;14(12):2327-36.; Wang et al., 2014Wang X, Zhang W, Chen H, Liao N, Wang Z, Zhang X et al. High selenium impairs hepatic insulin sensitivity through opposite regulation of ROS. Toxicol Lett. 2014;224(1):16-23.).
In this context, the overexpression of antioxidant selenium-dependent enzymes promotes the removal of intracellular hydrogen peroxide, which is a reactive oxygen species that regulates key enzymes for insulin signaling. In turn, this promotes the dephosphorylation of the enzymes of insulin signaling, thereby inactivating them and changing the action of insulin (Steinbrenner, 2013Steinbrenner, H. Interference of selenium and selenoproteins with the insulin-regulated carbohydrate and lipid metabolism. Free Radic Biol Med. 2013;65:1538-47.; Wang et al., 2014Wang X, Zhang W, Chen H, Liao N, Wang Z, Zhang X et al. High selenium impairs hepatic insulin sensitivity through opposite regulation of ROS. Toxicol Lett. 2014;224(1):16-23.; Zhou, Huang, Lei, 2013Zhou J, Huang K, Lei XG. Selenium and diabetes - evidence from animal studies. Free Radic Biol Med. 2013;65:1548-56.). In particular, some investigators have described the inhibition of phosphorylation of specific enzymes of the insulin signaling pathway, such as Akt and 5’-adenosine monophosphate-activated protein kinase (AMPK), caused by the elevated expression of GPx or Sepp1. Sepp1 also inhibits phosphorylation of the tyrosine residue on hepatocyte insulin receptors, and promotes the phosphorylation of a serine residue in IRS-1 (Misu et al., 2010Misu H, Takamura T, Takayama H, Hayashi H, Matsuzawa-Nagata N, Kurita S, et al. A liver-derived secretory protein, selenoprotein P, causes insulin resistance. Cell Metab. 2010;12(5):483-95.; Steinbrenner, 2013; Wang et al., 2014).
Zeng et al. (2012Zeng MS, Li X, Liu Y, Zhao H, Zhou JC, Li K, et al. A high-selenium diet induces insulin resistance in gestating rats and their offspring. Free Radic Biol Med. 2012;52(8):1335-42.) found that in rats, a diet rich in selenium (3.0 mg/kg per day) can induce hyperinsulinemia, insulin resistance, and glucose intolerance. High selenium intake also reduces the mRNA expression or protein levels of six factors that participate in insulin signaling pathways in the liver and muscle, and increases the activity of GPx1 in the pancreas, liver, and erythrocytes.
Similarly, Labunskyy et al. (2011Labunskyy VM, Lee BC, Handy DE, Loscalzo J, Hatfield DL, Gladyshev VN. Both maximal expression of selenoproteins and selenoprotein deficiency can promote development of type 2 diabetes-like phenotype in mice. Antioxid Redox Signal. 2011;14(12):2327-36.) found that under conditions of high selenium intake (a 0.4 ppm dose of sodium selenite), there is increased expression of the antioxidant selenoproteins GPx1 and MsrB1, and a positive correlation between GPx1 overexpression and the expression of other selenoproteins, such as SelS and SelT. These findings suggest that the factors described may participate in the impairment of insulin signaling. The authors suggest that the cellular dysregulation of the redox state, caused by high selenoprotein expression, can compromise the protein folding of insulin and its receptor.
Pinto et al. (2011Pinto A, Speckmann B, Heisler M, Sies H, Steinbrenner, H. Delaying of insulin signal transduction in skeletal muscle cells by selenium compounds. J Inorg Biochem. 2011;105(6):812-20.) demonstrated the action of various compounds containing selenium (sodium selenite, sodium selenate, selenomethionine, and methylseleninic acid) on insulin sensitivity in vitro in myocytes. The authors found that > 1 μM doses of sodium selenite and methylseleninic acid were capable of inhibiting Akt phosphorylation, and attenuated the glucose uptake by decreasing the concentration of intracellular reactive oxygen species. This result can be attributed to the differences in bioavailability and metabolism of the selenium-containing compounds.
In addition, selenium may contribute to the induction of insulin resistance because of its role in the metabolism of carbohydrates and lipids (Steinbrenner, 2013Steinbrenner, H. Interference of selenium and selenoproteins with the insulin-regulated carbohydrate and lipid metabolism. Free Radic Biol Med. 2013;65:1538-47.). In a study on mice, Wang et al. (2014Wang X, Zhang W, Chen H, Liao N, Wang Z, Zhang X et al. High selenium impairs hepatic insulin sensitivity through opposite regulation of ROS. Toxicol Lett. 2014;224(1):16-23.) found that high doses of selenium (200 mg/kg) can promote increased expression of carboxykinase phosphoenolpyruvate and glucose-6-phosphatase, a key enzyme for gluconeogenesis. The authors also found that selenium promotes the accumulation and metabolism of fatty acids in the liver; this effect is related to the increased production of reactive oxygen species originating from mitochondrial oxidative phosphorylation, and hence the development of hepatic insulin resistance.
In this scenario, it is noteworthy that hyperglycemia favors hepatic Sepp1 transcription by activating the forkhead box class O 1α (FoxO1α) and hepatocyte nuclear factor 4α (HNF-4α) transcription factors, through the action of peroxisomal proliferator-activated receptor gamma coactivator 1α (PCG-1α). These transcription factors bind to a functional site in the Sepp1 promoter and upregulate gene expression. They are also involved in regulating the expression of hepatic glucose metabolism enzymes, and it is known that insulin promotes the inactivation of these enzymes. Therefore, under insulin resistance conditions, there may be an increase in Sepp1 concentrations concomitant with the increase in the activity of gluconeogenic enzymes. This situation may partially explain the associations between selenoprotein concentrations and the risk of type 2 diabetes mellitus (Jung et al., 2013Jung TW, Choi HY, Lee SY, Hong HC, Yang SJ, Yoo HJ, et al. Salsalate and adiponectin improve palmitate-induced insulin resistance via inhibition of selenoprotein P through the AMPK-FOXO1a pathway. PLoS One. 2013;8(6):e66529.; Mao, Teng, 2013Mao J, Teng W. The relationship between selenoprotein P and glucose metabolism in experimental studies. Nutrients. 2013;5(6):1937-1948.; Steinbrenner et al., 2011Steinbrenner H, Speckmann B, Pinto A, Sies H. High selenium intake and increased diabetes risk: experimental evidence for interplay between selenium and carbohydrate metabolism. J Clin Biochem Nutr. 2011;48(1):40-5.; Steinbrenner, 2013).
A study by Jung et al. (2013Jung TW, Choi HY, Lee SY, Hong HC, Yang SJ, Yoo HJ, et al. Salsalate and adiponectin improve palmitate-induced insulin resistance via inhibition of selenoprotein P through the AMPK-FOXO1a pathway. PLoS One. 2013;8(6):e66529.) showed that induction of diabetes in rats by palmitate increases the expression of Sepp1 in HepG2 cells, thereby exacerbating insulin resistance. The activation of AMPK, induced by either salicylate or adiponectin, could reverse the increase in Sepp1 expression through phosphorylation of FoxO1α. Similar results were obtained by Takayama et al. (2014Takayama H, Misu H, Iwama H, Chikamoto K, Saito Y, Murao K, et al. Metformin suppresses expression of the selenoprotein P gene via an AMP-activated kinase (AMPK)/FoxO3a pathway in H4IIEC3 hepatocytes. J Biol Chem. 2014;289(1):335-45.), wherein metformin inhibited the AMPK-mediated increased expression of Sepp1 in the H4IIEC3 hepatocytes of diabetic rats. On the other hand, coadministration of an AMPK antagonist reversed these results, indicating that this pathway can be a possible therapeutic target of insulin resistance and diabetes.
CONCLUSION
The action of selenium in the pathogenesis of insulin resistance is not fully elucidated, but our review has revealed that the main mechanisms involve the regulation of intracellular concentrations of reactive oxygen species, and carbohydrate and lipid metabolism. When ingested in sufficient amounts to maintain plasma concentrations in the range of 80-120 µg/L (Rayman, 2012Rayman MP. Selenium and human health. Lancet. 2012;379(9822):1256-68.), selenium acts as an antioxidant and insulin-mimetic nutrient, favoring the synthesis and action of insulin. However, when selenium intake results in plasma levels above 120 µg/L (Rayman, 2012), the cellular redox state may become dysregulated, which can compromise the chemical interactions involved in the insulin signaling cascade. Nevertheless, it is noteworthy that the effects of selenium on insulin resistance are dependent on the chemical form (organic or inorganic), dose, and exposure time; these factors are not yet fully established. Further studies are needed to clarify the relation between selenium and insulin resistance in humans.
REFERENCES
- Alizadeh M, Safaeiyan A, Ostadrahimi A, Estakhri R, Daneghian S, Ghaffari A, et al. Effect of L-arginine and selenium added to a hypocaloric diet enriched with legumes on cardiovascular disease risk factors in women with central obesity: a randomized, double-blind, placebo-controlled trial. Ann Nutr Metab. 2012;60(2):157-68.
- Bahmani F, Kia M, Soleimani A, Asemi Z, Esmaillzadeh A. Effect of selenium supplementation on glycemic control and lipid profiles in patients with diabetic nephropathy. Biol Trace Elem. 2016;172(2):282-9.
- Barakat GM, Moustafa ME, Bikhazi AB. Effects of selenium and exendin-4 on glucagon-like peptide-1 receptor, IRS-1, and Raf-1 in the liver of diabetic rats. Biochem Genet. 2012;50(11-12):922-35.
- Barakat GM, Moustafa ME, Khalifeh I, Hodroj MH, Bikhazi A, Rizk S. Effects of exendin-4 and selenium on the expression of GLP-1R, IRS-1, and preproinsulin in the pancreas of diabetic rats. J Physiol Biochem. 2016;73(3):387-94.
- Bruce KD, Hanson MA. The developmental origins, mechanisms, and implications of metabolic syndrome. J Nutr. 2010;140(3):648-52.
- Campbell SC, Aldibbiat A, Marriott CE, Landy C, Ali T, Ferris WF, et al. Selenium stimulates pancreatic beta-cell gene expression and enhances islet function. FEBS Lett. 2008;582(15):2333-7.
- Chen H, Qiu Q, Zou C, Dou L, Liang J. Regulation of hepatic carbohydrate metabolism by selenium during diabetes. Chem Biol Interact. 2015;232:1-6.
- Cominetti C, Bortoli MC, Abdalla DSP, Cozzolino SMF. Considerations about oxidative stress, selenium and nutrigenetics. Rev Nutr. 2011;36(3):131-53.
- Cruz KJ, Oliveira AR, Pinto DP, Morais JB, Lima FS, Colli C, et al. Influence of magnesium on insulin resistance in obese women. Biol Trace Elem Res. 2014;160(3):305-10.
- Czernichow S, Couthouis A, Bertrais S, Vergnaud AC, Dauchet L, Galan P, et al. Antioxidant supplementation does not affect fasting plasma glucose in the Supplementation with Antioxidant Vitamins and Minerals (SU.VI.MAX) study in France: association with dietary intake and plasma concentrations. Am J Clin Nutr. 2006;84(2):395-9.
- D'Alessio D, Vahl T, Prigeon R. Effects of Glucagon-like Peptide 1 on the hepatic glucose metabolism. Horm Metab Res. 2004;36(11-12):837-41.
- Faghihi T, Radfar M, Barmal M, Amini P, Qorbani M, Abdollahi M, et al. A randomized, placebo-controlled trial of selenium supplementation in patients with type 2 diabetes: effects on glucose homeostasis, oxidative stress, and lipid profile. Am J Ther. 2014;21(6):491-495.
- Farrokhian A, Bahmani F, Taghizadeh M, Mirhashemi SM, Aarabi MH, Raygan F, et al. Selenium supplementation affects insulin resistance and serum hs-CRP in patients with type 2 diabetes and coronary heart disease. Horm Metab Res. 2016;48(4):263-268.
- Feng W, Cui X, Liu B, Liu C, Xiao Y, Lu W, et al. Association of urinary metal profiles with altered glucose levels and diabetes risk: a population-based study in china. PLoS One. 2015;10(4):e0123742.
- Gao H, Hägg S, Sjögren P, Lambert PC, Ingelsson E, Van Dam RM. Serum selenium in relation to measures of glucose metabolism and incidence of Type 2 diabetes in an older Swedish population. Diabetic Med. 2014;31(7):787-793.
- García OP, Ronquillo D, Caamaño MC, Martínez G, Camacho M, López V, et al. Zinc, iron and vitamins A, C and E are associated with obesity, inflammation, lipid profile and insulin resistance in mexican school-aged children. Nutrients. 2013;5(12):5012-30.
- Henriksen EJ, Diamond-Stanic MK, Marchionne EM. Oxidative stress and the etiology of insulin resistance and type 2 diabetes. Free Radic Biol Med. 2011;51(5):993-9.
- Hirabara SM, Gorjão R, Vinolo MA, Rodrigues AC, Nachbar RT, Curi, R. Molecular targets related to inflammation and insulin resistance and potential interventions. J Biomed Biotechnol. 2012;2012:379024.
- Iizuka Y, Ueda Y, Yagi Y, Sakurai E. Significant improvement of insulin resistance of GK rats by treatment with sodium selenate. Biol Trace Elem Res. 2010;138(1-3):265-71.
- Jung TW, Choi HY, Lee SY, Hong HC, Yang SJ, Yoo HJ, et al. Salsalate and adiponectin improve palmitate-induced insulin resistance via inhibition of selenoprotein P through the AMPK-FOXO1a pathway. PLoS One. 2013;8(6):e66529.
- Kim B, Feldman EL. Insulin resistance as a key link for the increased risk of cognitive impairment in the metabolic syndrome. Exp Mol Med. 2015;47(3):e149.
- Kim HN, Song SW. Concentrations of chromium, selenium, and copper in the hair of viscerally obese adults are associated with insulin resistance. Biol Trace Elem Res. 2014;158(2):152-7.
- Kwon H, Pessin JE. Adipokines mediate inflammation and insulin resistance. Front Endocrinol (Lausanne). 2013;4:71.
- Labunskyy VM, Lee BC, Handy DE, Loscalzo J, Hatfield DL, Gladyshev VN. Both maximal expression of selenoproteins and selenoprotein deficiency can promote development of type 2 diabetes-like phenotype in mice. Antioxid Redox Signal. 2011;14(12):2327-36.
- Lee YS, Jun HS. Anti-diabetic actions of glucagon-like peptide-1 on pancreatic beta-cells. Metabolism. 2014;63(1):9-19.
- Lei XG, Vatamaniuk MZ. Two tales of antioxidant enzymes on ß cells and diabetes. Antioxid Redox Signal. 2011;14(3):489-503.
- Litvinova LS, Kirienkova EV, Mazunin IO, Vasilenko MA, Fattakhov NS. Pathogenesis of insulin resistance in metabolic obesity. Biochemistry (Moscow). 2014;8(3):191-202.
- Mao J, Teng W. The relationship between selenoprotein P and glucose metabolism in experimental studies. Nutrients. 2013;5(6):1937-1948.
- Misu H, Takamura T, Takayama H, Hayashi H, Matsuzawa-Nagata N, Kurita S, et al. A liver-derived secretory protein, selenoprotein P, causes insulin resistance. Cell Metab. 2010;12(5):483-95.
- Muntoni S, Muntoni, S. Insulin resistance: pathophysiology and rationale for treatment. Ann Nutr Metab. 2011;58(1):25-36.
- Mutakin, Meiliana A, Wijaya A, Kobayashi K, Yamazaki C, Kameo S, et al. Association between selenium nutritional status and metabolic risk factors in men with visceral obesity. J Trace Elem Med Biol. 2013;27(2):112-116.
- Pinto A, Speckmann B, Heisler M, Sies H, Steinbrenner, H. Delaying of insulin signal transduction in skeletal muscle cells by selenium compounds. J Inorg Biochem. 2011;105(6):812-20.
- Pounis G, Costanzo S, Persichillo M, Curtis A, Sieri S, Vinceti M et al. Mushroom and dietary selenium intakes in relation to fasting glucose levels in a free-living Italian adult population: The Moli-sani Project. Diabetes Metab. 2014;40(1):34-42.
- Rains JL, Jain SK. Oxidative stress, insulin signaling, and diabetes. Free Radic Biol Med. 2011;50(5):567-75.
- Rayman MP. Selenium and human health. Lancet. 2012;379(9822):1256-68.
- Rayman MP, Stranges S. Epidemiology of selenium and type 2 diabetes: Can we make sense of it? Free Radic Biol Med. 2013;65:1557-64.
- Slattery MJ, Bredella MA, Thakur H, Torriani M, Misra M. Insulin resistance and impaired mitochondrial function in obese adolescent girls. Metab Syndr Relat Disord. 2014;12(1):56-61.
- Steinbrenner H, Hotze AL, Speckmann B, Pinto A, Sies H, Schott M, et al. Localization and regulation of pancreatic selenoprotein P. J Mol Endocrinol. 2013;50(1):31-42.
- Steinbrenner H, Speckmann B, Pinto A, Sies H. High selenium intake and increased diabetes risk: experimental evidence for interplay between selenium and carbohydrate metabolism. J Clin Biochem Nutr. 2011;48(1):40-5.
- Steinbrenner, H. Interference of selenium and selenoproteins with the insulin-regulated carbohydrate and lipid metabolism. Free Radic Biol Med. 2013;65:1538-47.
- Stranges S, Marshall JR, Natarajan R, Donahue RP, Trevisan M, Combs GF, et al. Effects of long-term selenium supplementation on the incidence of type 2 diabetes: a randomized trial. Ann Intern Med. 2007;147(4):217-23.
- Takayama H, Misu H, Iwama H, Chikamoto K, Saito Y, Murao K, et al. Metformin suppresses expression of the selenoprotein P gene via an AMP-activated kinase (AMPK)/FoxO3a pathway in H4IIEC3 hepatocytes. J Biol Chem. 2014;289(1):335-45.
- Tencerova M, Aouadi M, Vangala P, Nicoloro SM, Yawe JC, Cohen JL, et al. Activated Kupffer cells inhibit insulin sensitivity in obese mice. FASEB J. 2015;29(7):2959-69.
- Wang X, Zhang W, Chen H, Liao N, Wang Z, Zhang X et al. High selenium impairs hepatic insulin sensitivity through opposite regulation of ROS. Toxicol Lett. 2014;224(1):16-23.
- Wei Z, Yao M, Li Y, He X, Yang Z. Dietary selenium deficiency exacerbates lipopolysaccharide-induced inflammatory response in mouse mastitis models. Inflammation. 2014;37(6):1925-31.
- Winnier DA, Fourcaudot M, Norton L, Abdul-Ghani MA, Hu SL, Farook VS, et al. Transcriptomic identification of ADH1B as a novel candidate gene for obesity and insulin resistance in human adipose tissue in Mexican Americans from the Veterans Administration Genetic Epidemiology Study (VAGES). PLoS One. 2015;10(4):e0119941.
- Zeng MS, Li X, Liu Y, Zhao H, Zhou JC, Li K, et al. A high-selenium diet induces insulin resistance in gestating rats and their offspring. Free Radic Biol Med. 2012;52(8):1335-42.
- Zhou J, Huang K, Lei XG. Selenium and diabetes - evidence from animal studies. Free Radic Biol Med. 2013;65:1548-56.
- Zhou J, Xu G, Bai Z, Li K, Yan J, Li F, et al. Selenite exacerbates hepatic insulin resistance in mouse model of type 2 diabetes through oxidative stress-mediated JNK pathway. Toxicol Appl Pharmacol. 2015;289(3):409-18.
- Zou C, Qiu Q, Chen H, Dou L, Liang, J. Hepatoprotective effects of selenium during diabetes in rats. Hum Exp Toxicol. 2016;35(2):114-23.
Publication Dates
-
Publication in this collection
2018
History
-
Received
22 Nov 2016 -
Accepted
30 Aug 2017