Abstract
Stroke is the third leading cause of mortality and disability in industrial countries. Treatment with herbs with antioxidant properties has been reported to be an alternative to the conventional treatments. This study was conducted to investigate the effect of Anchusa italica extract on hippocampal injury induced by transient global cerebral ischemia and reperfusion in the rat. To do so, 50 rats were randomly assigned to five groups; control, sham, ischemia, and 50 or 100 mg/kg of Anchusa italica treated animals. Ischemia was induced by occlusion of carotid artery for 30 minutes. Afterward, behavioral tests and biochemical analyses were conducted. Induction of ischemia/reperfusion caused a decline in learning and passive avoidance memory in rats. Moreover, Anchusa italica caused an increase in learning and improved the passive avoidance memory. Induction of ischemia/reperfusion caused a decrease in the antioxidant capacity of the brain and serum as well as an increase in the malondialdehyde of the brain and serum. Anchusa italica led to an increase in the antioxidant capacity of the brain and serum and decrease in the malondialdehyde of the brain and serum. Overall, because of its protective effects on spatial memory, passive avoidance learning, antioxidant capacity, and lipid peroxidation during ischemia/reperfusion, Anchusa italica might be beneficial in ischemic patients.
Keywords:
Boraginaceae/phytochemistry; Brain ischemia; Spatial memory/drug effects; Malondialdehyde/pharmacology; Neuroprotection/drug effects; Lipidic peroxidation/drug effects; Antioxidant capacity; Passive avoidance memory.
INTRODUCTION
Cerebral ischemia is one of the most debilitating brain injuries and the most important cause of mortality worldwide particularly in people over 65 years. Different factors are effective in inducing cerebral ischemia including cardiac arrest, atherosclerosis, thromboembolitic events, vasoconstriction, congenital heart disorders, low blood pressure, and some types of tumors (Thom et al., 2006Thom T, Haase N, Rosamond W, Howard VJ, Rumsfeld J, Manolio T, et al. Heart disease and stroke statistics-2006 update a report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation. 2006;113(6):e85-e151.; Bernard et al., 2002Bernard SA, GrayTW, Buist MD, Jones BM, Silvester W, Gutteridge G, et al. Treatment of comatose survivors of out-of-hospital cardiac arrest with induced hypothermia. N Engl J Med. 2002;346:557-63.; Pourheydar et al., 2014Pourheydar B, Shahi M, Farjah GH, Javanmard M, Karimipour M, Atabaki F. Evaluation of apoptosis in hippocampal cells of rat following intravenous injection of bone marrow stromal cells in ischemia-reperfusion model. Urmia Med J. 2014;25:586-97.). Neurological events due to cerebral ischemia include sensory, movement, and visual disorders as well as aphasia, agnosia, amnesia, and impaired spatial learning and memory (Andrews et al., 2008Andrews E, Tsai SY, Johnson S, Farrer J, Wagner J, Kopen G, et al. Human adult bone marrow-derived somatic cell therapy results in functional recovery and axonal plasticity following stroke in the rat. Exp Neurol. 2008;211(2):588-92.; Bokura, Robinson, 1997Bokura H, Robinson RG. Long-term cognitive impairment associated with caudate stroke. Stroke. 1997;28(5):970-5.).
Hippocampus, as an organ involved in the formation of new memory and processing spatial information, is the most sensitive area of the brain during ischemia, trauma, and degenerative diseases (Zhang, Piantadosi, 1994Zhang J, Piantadosi CA. Prolonged production of hydroxyl radical in rat hippocampus after brain ischemia-reperfusion is decreased by 21-aminosteroids. Neurosci Lett 1994;177(1-2):127-30.; Deckert, Jorgensen, 1988Deckert J, Jorgensen MB. Evidence for pre-and postsynaptic localization of adenosine A1 receptors in the CA1 region of rat hippocampus: a quantitative autoradiographic study. Brain Res. 1988;446(1):161-4.). During cerebral ischemia, generation of free radicals including reactive oxygen (ROS) and nitrogen species (RNS) brings damage to lipids, proteins, and desoxyribonucleic acid, resulting in neuronal death. Free radicals play an important role in breaking the blood-brain barrier and inducing cerebral edema (Wolff, Dean, 1987Wolff SP, Dean R. Glucose autoxidation and protein modification: the potential role of 'autoxidative glycosylation' in diabetes. Biochem J. 1987;245(1):243-50.). Indeed, oxidative stress extensively influences cell membrane system particularly membrane bilayer phospholipids. Therefore, it can be argued that lipids peroxidation is oxidative damage to lipids with carbon double bonds (White et al., 2000White BC, Sullivan JM, DeGracia DJ, O'Neil BJ, Neumar RW, Grossman LI, et al. Brain ischemia and reperfusion: molecular mechanisms of neuronal injury. J Neurol Sci. 2000;179(S 1-2):1-33.; Oruc et al., 2004Oruc EO, Sevgiler Y, Uner N. Tissue-specific oxidative stress responses in fish exposed to 2, 4-D and azinphosmethyl. Comp Biochem Physiol, Part C: Toxicol Pharmacol. 2004;137(1):43-51.). The final product of lipids peroxidation is malondialdehyde (MDA), which is a mutagenic, carcinogenic, and highly reactive compound (Janero, 1990Janero DR. Malondialdehyde and thiobarbituric acid-reactivity as diagnostic indices of lipid peroxidation and peroxidative tissue injury. Free Radic Biol Med. 1990;9(6):515-40.).
Use of antioxidants is an approach to reduce adverse effects of cerebral ischemia (Dekanski et al., 2011Dekanski D, Selaković V, Piperski V, Radulović Ž, Korenić A, Radenović L. Protective effect of olive leaf extract on hippocampal injury induced by transient global cerebral ischemia and reperfusion in Mongolian gerbils. Phytomedicine. 2011;18(13):1137-43.). Recently, many synthetic antioxidants have been found to be carcinogenic and toxic and therefore some efforts have been made to identify nature-based antioxidants (Zhang et al., 2006Zhang H, Chen F, Wang X, Yao HY. Evaluation of antioxidant activity of parsley (Petroselinum crispum) essential oil and identification of its antioxidant constituents. Food Res Int. 2006;39(8):833-9.; Gulluce et al., 2007Gulluce M, Sahin F, Sokmen M, Ozer H, Daferera D, Sokmen A, et al. Antimicrobial and antioxidant properties of the essential oils and methanol extract from Mentha longifolia L. ssp. longifolia. Food Chem. 2007;103(4):1449-56.).
Anchusa italica, locally called as “Gav zaban” in Persian, is one of the important genus of family Boraginaceae. Five species of genus Anchusa have been found in Iran (Mozaffarian, 1996Mozaffarian V. A dictionary of Iranian plant names: Latin, English, Persian. Tehran: Farhang Mo'aser; 1996.; Kazemi, 2013Kazemi M. Essential oil composition of Anchusa italica from Iran. Chem Nat Compd. 2013;49(2):369-70.). A. italica has been demonstrated to contain alkaloids, tannins, saponins, triterpenes, and polyphenols such as anthocyanins and flavonoids (Barbakadze et al., 2010Barbakadze V, Gogilashvili L, Amiranashvili L, Merlani M, Mulkijanyan K, Churadze M, et al. Poly [3-(3, 4-dihydroxyphenyl) glyceric acid] from Anchusa italica roots. Nat Prod Commun. 2010;5(7):1091-5.). In traditional medicine, A. italica extract is used as a stimulant, tonic, palliative in biliary diseases, diuretic, sedative, antiarthritic, anti-asthma, and laxative. It is also used to reduce fever and cough (Khare, 2008Khare CP. Indian medicinal plants: an illustrated dictionary. Heidelberg: Springer Science & Business Media; 2008.; Kebriaee-Zadeh, 2010Kebriaee-Zadeh A. Overview of national drug policy of Iran. Iran J Pharm Res. 2010;2:1-2.). A. italica has been reported to have antioxidant, anticancer, and antiviral effects as well as certain effects on the central nervous system and endogenous glands (Al-Snafi, 2014Al-Snafi A. The pharmacology of Anchusa italica and Anchusa strigosa: a review. Int J Pharm Pharm Sci. 2014;6(4):7-10.). In the light of A. italica antioxidant effects, this study was conducted to investigate neuroprotective effects of hydroalcoholic extract from A. italica flowers on hippocampal injury induced by transient global cerebral ischemia and reperfusion in the rat.
MATERIAL AND METHODS
Plant material and extraction
A. italica flowers were gathered and botanically confirmed by a botanist and deposited as specimens in Herbarium of Medical Plants Research Center in Shahrekord University of Medical Sciences, Shahrekord, Iran (SKUMS-504). The dried and pulverized sample was extracted using ethanol 70%. The resulting extract was filtered and concentrated using a rotary evaporator. To conduct final drying, the concentrated extract was incubated at 37 ℃.
Determining antioxidant capacity of the extract and measuring total phenol and flavonoid content
After the stock of extract and DPPH was prepared, the sample was incubated in the dark for 15 minutes and the samples’ absorbance was read at 517 nm wavelength. Methanol was used as blank and methanol plus DPPH was considered a control. After calculating the inhibition rate of free radicals by the extract, the antioxidant activity was reported as IC50 (McDonald et al., 2001McDonald S, Prenzler PD, Antolovich M, Robards K. Phenolic content and antioxidant activity of olive extracts. Food Chem. 2001;73(1):73-84.).
The total phenol of A. italica was measured by Folin-Ciocalteau reagent and aluminum chloride colorimetric method (Chang et al., 2002Chang CC, Yang MH, Wen HM, Chern JC. Estimation of total flavonoid content in propolis by two complementary colorimetric methods. J Food Drug Anal. 2002;10(3):178-82.; Asgari et al., 2012Asgari S, Setorki M, Rafieian-Kopaei M, Shahinfard N, Ansari R, Forouz Z. Postprandial hypolipidemic and hypoglycemic effects of Allium hertifolium and Sesamum indicum on hypercholesterolemic rabbits. J Pharm Pharmacol. 2012;6(15):1131-5.). The experiments were conducted in triplicate and the total phenol and flavonoid content of the extract was measured and reported in mg/g dried extract.
Animals
The investigation conforms to the Guide for the Care and Use of Laboratory Animal and ethical guidelines of IR. SKUMS Bioethics Committee (Rec.2015.141). In this study, 50 male adult Wistar rats weighing 250-300 g were used. The rats were kept at 21±2 ℃ temperature, under 12-h dark/12-h light cycle and had free access to the same food and water. The rats were assigned to five groups as follows: 1) Control group that underwent no surgery and received distilled water alone, 2) Sham group that underwent surgery without drug administration and occlusion of carotid arteries. This group also received distilled water alone, 3) Ischemia group that underwent ischemia without drug administration received distilled water alone, and Groups 4) and 5) received A. italica extract (50 and 100 mg/kg, respectively) with induction of ischemia. The extract was administered intraperitoneally (IP) (Mrudula, 2014Mrudula G. Antistress and antioxidant effect of Borago officinalis leaves. J Drug Discovery Ther. 2014;2(18):1-9.; Ghahremanitamadon et al., 2014Ghahremanitamadon F, Shahidi S, Zargooshnia S, Nikkhah A, Ranjbar, A, Soleimani Asl S. Protective effects of Borago officinalis extract on amyloid ß-peptide (25-35)-induced memory impairment in male rats: a behavioral study. Biomed Res Int. 2014;2014:798535.). In intraperitoneal injection, the rate of absorption of the drug was higher than oral administration. Drugs were administered after the induction of ischemia model.
Induction of ischemia
The global cerebral ischemia is created with severe blood reduction or occlusion to the brain. The ischemia model in this study was performed with general carotid arteries occlusion of bilateral vessels (Kawai et al., 1992Kawai K, Nitecka L, Ruetzler CA, Nagashima G, Joo F, Mies G, et al. Global cerebral ischemia associated with cardiac arrest in the rat: I. Dynamics of early neuronal changes. J Cereb Blood Flow Metab. 1992;12(2):238-49.; Lipton, 1999Lipton P. Ischemic cell death in brain neurons. Physiol Rev. 1999;79(4):1431-568.). After induction of anesthesia using 400 mg/kg of chloral hydrate, the neck anterior lateral region underwent surgery after carotid sheath was determined, and common carotid arteries were carefully identified from vagosympathetic nerve and separated. Then, carotid arteries were occluded for 30 minutes and then reperfusion was performed. Behavioral tests were performed one week after induction of ischemia. After performing the behavioral tests and inducing anesthesia for the second time, the blood samples were taken from the hearts. Finally, hippocampus, cortex, and sub-cortex that were already taken out from the brain were separated on the ice and analyzed biochemically. The blood samples were centrifuged and then the serum isolated and used for biochemical analyses.
Morris water maze test and shuttle box test
In this test, each rat was given an opportunity of 60 seconds to find the platform. If the rat could not find the platform, the researcher would guide it to the platform. Between each pair of the trials, the rats could rest for 30 seconds to examine the surrounding. Between two blocks, the rats were taken out from water for 10 minutes and left to rest in the cage. Each rat was trained four times per day for four days and the test was repeated on day 5 without the platform that was considered “probe day” (Dominique et al., 1998Dominique JF, Roozendaal B, McGaugh JL. Stress and glucocorticoids impair retrieval of long-term spatial memory. Nature. 1998;394(6695):787-90.).
The shuttle box test lasted for four days for each rat. On days 1 and 2 of the test, each rat was placed inside the instrument to adapt to it. On day 3, the acquisition was examined by putting them separately into a bright room. After a 2-minute period of acculturation, the guillotine door was opened and after the rat entered the dark room it was closed and an electrical shock (1 mA, 1 second, once) was applied on the rat such that it just paddled. In this test, the primary latency to enter the dark room was recorded on the day 3 and the latency during passing was recorded on the day 4 (Khalili, Roghani, Ekhlasi, 2010Khalili M, Roghani M, Ekhlasi M. The effect of aqueous crocus sativus L. extract on intracerebroventricular streptozotocin-induced cognitive deficits in rat: a behavioral analysis. Iran J Pharm Res. 2010;8(3):185-91.).
Measuring antioxidant capacity of the serum and the brain
For measuring antioxidant capacity of the serum and the brain, three solutions were used: a buffer (1.55 mL sodium acetate, 8 mL concentrated acetic acid reaching 500 mL by adding distilled water), a ferrous chloride solution (270 mg FeCl3 (6 H2O) reaching 50 mL by adding distilled water), and triazine solution (47 mg triazine dissolved in 40 mL of 40 mM HCl). The labor solution was prepared by addition of 10 mL of the first solution, 1 ml of the second solution, and 1 mL of the third solution. Next, 25 µL of the serum sample and 25 µL of the homogenized brain sample were added to 1.5 mL of the labor solution and kept at 37 ℃. Optical absorbance was read at 593 nm wavelength (Benzie, Strain, 1999Benzie IF, Strain J. [2] Ferric reducing/antioxidant power assay: direct measure of total antioxidant activity of biological fluids and modified version for simultaneous measurement of total antioxidant power and ascorbic acid concentration. Meth Enzymol. 1999;299:15-27.).
Measuring serum and brain MDA
To measure the amount of the serum MDA, 0.5 g of thiobarbituric acid was mixed with 80 mL acetic acid 20% and then the pH was set at 3.5 using NaOH and its volume reached 100 mL by adding acetic acid 20%. After that, 100 µL of the serum sample was mixed with 100 µL of SDS 8.1% and 2.5 mL of the labor solution. The samples were placed in a bain-marie of boiling water for 60 minutes and then cooled and centrifuged at 4000 rpm. The optical absorbance of the supernatant was read at 523 nm wavelength.
To measure the brain MDA, 1 g of the brain tissue was cooled with KCL 2.5% and homogenized at 10% (weight-volume) proportion, and then incubated at 37±1 ℃ for 60 minutes in a metabolic shaker. Then, 1 ml of trichloroacetic acid 5% and 1 mL of thiobarbituric acid 67% were added to it and mixed well after each step. The content of each vial was transferred to the centrifugation tube and centrifuged at 2000 rpm for 15 minutes. Then, the supernatant was transferred to another tube and placed in a bain-marie. After 10 minutes, the tubes were cooled, and the optical absorbance read at 535 nm wavelength (Karatas, Karatepe, Baysar, 2002Karatas F, Karatepe M, Baysar A. Determination of free malondialdehyde in human serum by high-performance liquid chromatography. Anal Biochem. 2002;311(1):76-9.).
Data analysis
The data were expressed as mean ± SEM and analyzed by one-way ANOVA and Tukey’s post-hoc tests in GraphPad Prism Software version 5. The level of significance was considered at P<0.05.
RESULTS
Total phenol and flavonoid content
The total phenol and flavonoid contents of A. italica extract were derived 62.2±0.3 and 47.06±0.4 mg/g of the dried extract, respectively. Scavenging capacity of radicals and antioxidant activity of A. italica extract (IC50) was derived about 90.52±0.3 µg/mL.
The effect of A. italica extract on learning and spatial memory in Morris Water Maze test
The findings of Morris Water Maze test demonstrated that latency to find the platform was the highest in the ischemic group and lowest in the control group. More clearly, ischemia caused learning and memory deficit. In the extract-treated groups, memory and learning improved. Intraperitoneal administration of 50 and 100 mg/kg of the extract caused a significant decrease in the latency to find the platform compared to the ischemia group (p<0.001). There was no significant difference in the latency among the control, sham, and the extract (50 and 100 mg/kg) groups (p>0.05). Also, there was no significant difference between the two extract (50 and 100 mg/kg) treated groups (p>0.05) (Figure 1).
The effect of A. italica extract on learning and spatial memory in Morris water maze test on hippocampal injury induced by transient global cerebral ischemia and reperfusion. time: latency to find the platform on day 3, 4 and duration of swimming in target zone on the probe day. ISC 100 and ISC 50: treated with 100 and 50 mg/kg of Anchusa italica extract, respectively. (***: significant difference between the all groups with ischemia group, <0.001). The number of samples, n=7.
Latency to find the platform was highest in the ischemic group and lowest in the control group. Intraperitoneal administration of 50 and 100 mg/kg of the extract caused a significant decrease in the latency to find the platform compared to the ischemic group (p<0.001). Similarly, there was no significant difference in the latency time among the control, sham, and the extract 100 mg/kg groups (p>0.05). There was a significant difference between two extract-treated (50 and 100 mg/kg) groups (p<0.001) (Figure 1).
Duration of swimming in target zone on the probe day was longest in the control group and shortest in the ischemia group. Intraperitoneal administration of 50 and 100 mg/kg of the extract caused a significant increase in the duration of swimming in target zone compared to the ischemia group (p<0.001). However, there was no significant difference in the duration of swimming in target zone among the control, sham, and the extract (50 and 100 mg/kg) groups (p>0.05). Also, there was no significant difference between the two extract (50 and 100 mg/kg) treated groups (p>0.05) (Figure 1).
The effect of A. italica extract on passive avoidance memory in Shuttle Box test
The findings demonstrated that the time difference between the primary and secondary latency was highly marked in the control group. This difference in the ischemia group was negligible. In the extract-treated groups, the time difference decreased with an increase in the extract dose. The secondary latency was highest in the control group and lowest in the ischemia group. Intraperitoneal administrations of 50 and 100 mg/kg caused a significant increase in the secondary latency compared to the ischemia group (p<0.001). There was no significant difference in the secondary latency among the control, sham, and the extract 100 mg/kg groups (p>0.05). Moreover, there was a significant difference between the two extract (50 and 100 mg/kg) treated groups (p<0.05) (Figure 2). In this test, the latency during passing was considered as the secondary latency time.
The effect of A. italica extract on passive avoidance memory in shuttle box test on hippocampal injury induced by transient global cerebral ischemia and reperfusion. t1: the primary latency to enter the dark room and t2: the latency during passing. ISC 100 and ISC 50: treated with 100 and 50 mg/kg of Anchusa italica extract, respectively. (***: significant difference between the all groups with ischemia group, P < 0.001). The number of samples, n=7.
The effect of A. italica extract on antioxidant capacity of serum and brain
The antioxidant capacity of the serum was significantly higher in the rats treated with 50 and 100 mg/kg of the A. italica extract than in the ischemic group (p<0.01 and p<0.001, respectively). Moreover, the antioxidant capacity of the brain was significantly higher in the rats treated with 50 and 100 mg/kg of the A. italica extract than in the ischemia group (p<0.05 and p<0.001). The antioxidant capacity of the serum and brain was not significantly different between the control, sham and the extract 100 mg/kg groups (p>0.05). There was a significant difference between the two treated groups (p<0.001) (Figure 3).
The effect of A. italica extract on antioxidant capacity of serum and brain on hippocampal injury induced by transient global cerebral ischemia and reperfusion. ISC 100 and ISC 50: treated with 100 and 50 mg/kg of A. italica extract, respectively. (***: significant difference between the all groups with ischemia group, P<0.001). The number of samples, n=7.
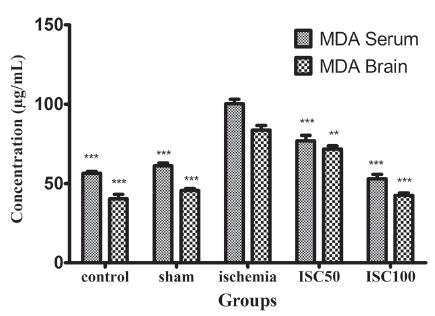
The effect o f A. italica extract on MDA of serum and brain
The serum MDA level was significantly lower in the rats treated with 50 and 100 mg/kg of the A. italica extract than in the ischemia group (p<0.001). Moreover, the brain MDA level was significantly lower in the rats treated with 50 and 100 mg/kg of the A. italica extract than in the ischemia group (p < 0.05 and p < 0.001). The serum and brain MDA levels were not significantly different among the control, sham and the extract (100 mg/kg) groups (p>0.05); however, there was a significant difference between the two extract (50 and 100 mg/kg) treated groups (p<0.001) (Figure 4).
The effect of A. italica extract on MDA of serum and brain on hippocampal injury induced by transient global cerebral ischemia and reperfusion. ISC 100 and ISC 50: treated with 100 and 50 mg/kg of A. italica extract, respectively. (*** and **: significant difference between the all groups with ischemia group, P<0.001, P<0.01). The number of samples, n=7.
DISCUSSION
The importance of oxidative stress in cerebral ischemia
Regarding the significance of stroke as one of the important health issues worldwide, many studies have been conducted to investigate the factors involved in pathogenesis as well as prevention and treatment of stroke. The positive effects of herbal drugs in mitigating adverse effects of ischemia/reperfusion have already been demonstrated. Medicinal plants and some of their active compounds have been demonstrated to minimize brain lesion volume, cerebral edema, neuronal injury, and ischemia-induced consequences. The action mechanisms of protective effects of medicinal plants include a decrease in oxidative and nitrative stress, decrease in lipids peroxidation, decrease in the activities of microglia and astrocytes, inhibition of apoptotic proteins expression, increase in expression of mitochondrial genes, prevention of fragmentation and oxidative damage to DNA, increase the expression of antiapoptotic proteins, and decrease in expression of inflammation mediators (Rabiei, Bigdeli, Lorigooini, 2015Rabiei Z, Bigdeli M, Lorigooini Z. A Review of medicinal herbs with antioxidant properties in the treatment ?of cerebral ischemia and reperfusion. J Babol Univ Med Sci. 2015;17(12):47-56.).
Cerebral ischemia causes initiation of signaling cascade that leads to the production of free radicals and oxidative stress depending on the severity and duration of the complication. Oxidative stress is an important factor for cerebral dysfunction in neurodegenerative diseases such as ischemia (Lipton, 1999Lipton P. Ischemic cell death in brain neurons. Physiol Rev. 1999;79(4):1431-568.). Oxidative stress due to ischemia can enhance the production of free radicals and lipid peroxidation. Lipid peroxidation has been reported to increase in the early stages of ischemia (El Kossi, Zakhary, 2000El Kossi MMH, Zakhary MM. Oxidative stress in the context of acute cerebrovascular stroke. Stroke. 2000;31(8):1889-92.; Ozkul et al., 2007Ozkul A, Akyol A, Yenisey C, Arpaci E, Kiylioglu N, Tataroglu C. Oxidative stress in acute ischemic stroke. J Clin Neurosci. 2007;14(11):1062-6.; Aygul et al., 2006Aygul R, Kotan D, Demirbas F, Ulvi H, Deniz O. Plasma oxidants and antioxidants in acute ischaemic stroke. J Int Med Res. 2006;34(4):413-8.). Oxidative stress not only causes an increase in production of free radicals but also reduces the cellular antioxidant mechanisms (Açikgöz et al., 1998Açikgöz O, Gönenç S, Kayatekin BM, Uysal N, Pekçetin Ç, Şemin İ, et al. Methamphetamine causes lipid peroxidation and an increase in superoxide dismutase activity in the rat striatum. Brain Res. 1998;813(1):200-2.).
Antioxidants have attracted much attention in the development of neuroprotective agents to treat stroke. The physiological role of antioxidants is seen in scavenging free radicals (Köksal, Gülçin, 2008Köksal E, Gülçin I. Antioxidant activity of cauliflower (Brassica oleracea L.). Turk J Agric For. 2008;32:65-78.). Plant antioxidants have been recommended as potential agents to prevent cerebral damage or to reduce and treat cerebral damage due to ischemia/reperfusion. Therefore, studying the effects of antioxidants and free radical-trapping agents is important since they are fundamentally protective agents against various brain damage such as cerebral damage due to ischemia/reperfusion (Baradaran, Nasri, Rafieian-Kopaei, 2013Rafieian-Kopaei M, Baradaran A, Rafieian M. Oxidative stress and the paradoxical effects of antioxidants. J Res Med Sci. 2013;18(7):628.).
The antioxidant effect of A. italica extract
In the current study, the brain and serum antioxidant capacities in the ischemia group decreased compared to the control group. Moreover, in the A. italica-treated groups, the brain and serum antioxidant capacities increased compared to the ischemia group, suggesting an improvement of these variables after administration with the extract.
This study demonstrated that the serum and brain MDA was higher in the ischemia group compared to the control group. Besides, in the extract-treated groups, the serum and brain MDA decreased compared to the ischemia group. It seems that this finding is due to the protective effect of A. italica extract components in reducing lipid peroxidation. The role of plant flavonoids has been demonstrated in reducing lipid peroxidation. Akhlaghi and Bandy (2009Akhlaghi M, Bandy B. Mechanisms of flavonoid protection against myocardial ischemia-reperfusion injury. J Mol Cell Cardiol. 2009;46(3):309-7.) reported that the use of flavonoids caused a decrease in production of MDA and free radicals such as superoxide, pyroxyl, and peroxynitrite during ischemia. Pataki et al. (2002Pataki T, Bak I, Kovacs P, Bagchi D, Das DK, Tosaki A. Grape seed proanthocyanidins improved cardiac recovery during reperfusion after ischemia in isolated rat hearts. Am J Clin Nutr. 2002;75(5):894-9.) studied the effect of flavonoids on ischemia and found that these compounds decrease the production of ROS during ischemia-reperfusion.
A. italica extract was reported to have a wide spectrum of pharmacological and therapeutic effects including antioxidant and anti-inflammatory activities (Al-Snafi, 2014Al-Snafi A. The pharmacology of Anchusa italica and Anchusa strigosa: a review. Int J Pharm Pharm Sci. 2014;6(4):7-10.). Another study has indicated that borage leaf extract is able to eliminate ROS and DPPH radicals (Wettasinghe, Shahidi, 2000Wettasinghe M, Shahidi F. Scavenging of reactive-oxygen species and DPPH free radicals by extracts of borage and evening primrose meals. Food Chem. 2000;70(1):17-26.). Elsewhere, the anti-oxidative activity decreased with a decrease in phenolic compounds (Oms-Oliu et al., 2008Oms-Oliu G, Odriozola-Serrano I, Soliva-Fortuny R, Martín-Belloso O. The role of peroxidase on the antioxidant potential of fresh-cut 'Piel de Sapo'melon packaged under different modified atmospheres. Food Chem. 2008;106(3):1085-1092.). In the current study, A. italica extract displayed acceptable antioxidant activity to inhibit DPPH radicals. Therefore, this ability appears to be related to the antioxidant property of phenolic and flavonoid compounds present in A. italica leaf extract.
In this study, A. italica extract was found to contain high amounts of phenolic and flavonoid contents. The effect of A. italica extract in controlling ischemia seems to be because of the presence of these compounds.
Antioxidants activities are due to secondary metabolites such as polyphenols (Baradaran, Nasri, Rafieian-Kopaei, 2013Rafieian-Kopaei M, Baradaran A, Rafieian M. Oxidative stress and the paradoxical effects of antioxidants. J Res Med Sci. 2013;18(7):628.). Antioxidant activities of the flavonoids are likely to be much more important. Flavonoids exert antioxidant activity against free radicals and have been known as important factors for inhibiting lipids peroxidation (Middleton, Kandaswami, Theoharides, 2000Middleton E, Kandaswami C, Theoharides TC. The effects of plant flavonoids on mammalian cells: implications for inflammation, heart disease, and cancer. Pharmacol Rev. 2000;52(4):673-751.; Rafieian-Kopaei, Baradaran, Rafieian, 2013).
Zhang et al. (2013Zhang S, Qi Y, Xu Y, Han X, Peng J, Liu K, et al. Protective effect of flavonoid-rich extract from Rosa laevigata Michx on cerebral ischemia-reperfusion injury through suppression of apoptosis and inflammation. Neurochem Int. 2013;63(5):522-32.) studied flavonoids effects on ischemia/reperfusion-induced damage and found that these compounds prevented the fragmentation of DNA and increased expression of Bcl-2. In addition, they caused a decrease in expression of p53, Apaf1, fas, fasL, Bax, Bid, cytochrome C, and caspase 3, 8, and 9. The findings of this study revealed that flavonoid compounds cause a decrease in expression of NF-KB, iNos, MMP9, Cox-2, TNF-α, IL-1β, IL-4, and IL-6. Therefore, it seems that flavonoids have potential to treat ischemia because of their antioxidant, antiapoptotic, and anti-inflammatory properties.
Hi et al. (2012) demonstrated that saponins could exert protective effects against cerebral ischemia through decreasing MDA, lactate dehydrogenase, xanthine oxidase, caspase 3, and Bax gene and increasing SOD and glutathione peroxidase. A study showed that saponins could also exert antiapoptotic effects in ischemia through inhibiting P13K pathway (Chen et al., 2011Chen S, Liu J, Liu X, Fu Y, Zhang M, Lin Q, et al. Panax notoginseng saponins inhibit ischemia-induced apoptosis by activating PI3K/Akt pathway in cardiomyocytes. J Ethnopharmacol. 2011;137(1):263-70.). A. italica has been shown to contain saponins (Al-Snafi, 2014Al-Snafi A. The pharmacology of Anchusa italica and Anchusa strigosa: a review. Int J Pharm Pharm Sci. 2014;6(4):7-10.); therefore, it seems that protective effect of this plant on cerebral ischemia is due to the presence of these compounds.
Morris Water Maze test revealed that in the ischemia group, latency to find the platform in target zone was much longer than that of the control group. This duration decreased in the extract-treated groups compared to the ischemia group. Duration of swimming in target zone on the probe day in the ischemia group was much shorter than that of the control group and increased in the extract-treated groups compared to the ischemia group. This finding can represent the improvement of spatial memory and learn to recall the platform location in A. italica extract-treated groups. Pu et al. (2007Pu F, Mishima K, Irie K, Motohashi K, Tanaka Y, Orito K, et al. Neuroprotective effects of quercetin and rutin on spatial memory impairment in an 8-arm radial maze task and neuronal death induced by repeated cerebral ischemia in rats. J Pharmacol Sci. 2007;104:329-34.) investigated the effects of four flavonoids in cerebral ischemia model. These flavonoid compounds caused improvement of spatial memory deficit and neuronal death induced by cerebral ischemia (Pu et al., 2007). Shang et al. (2006Shang YZ, Miao H, Cheng JJ, Qi JM. Effects of amelioration of total flavonoids from stems and leaves of Scutellaria baicalensis Georgi on cognitive deficits, neuronal damage and free radicals disorder induced by cerebral ischemia in rats. Biol Pharm Bull. 2006;29(4):805-10.) induced ischemia in rats to study the effects of flavonoids in treating this disorder and found that these compounds led to an improvement in learning and spatial memory of these rats.
Shuttle Box test demonstrated that the difference between t1 and t2 in the ischemia group was slight, which indicates avoidance memory deficit is due to ischemia. Moreover, this difference in A. italica-treated groups was much more marked than that of the ischemia group, which indicates the optimal effect of this extract on passive avoidance memory.
CONCLUSION
The findings of this study showed that inducing ischemia-reperfusion in Wistar rats caused spatial and passive avoidance memory deficit in these rats decreased the antioxidant capacity of the serum and the brain, and increased the brain and serum MDA. Intraperitoneal administration of A. italica hydroalcoholic extract for 14 days could significantly improve ischemia/reperfusion-induced injury on memory behaviors and the brain and serum biochemical changes. In the light of these findings, it seems that A. italica hydroalcoholic extract caused improvement of ischemia-induced injuries through scavenging free radicals derived from ischemia/reperfusion process owing to containing abundant antioxidant compounds (phenolic and flavonoid compounds and saponins).
ACKNOWLEDGMENT
This work was supported by grant No. 1937 from Shahrekord University of Medical Sciences. The authors thank the Research Council of Shahrekord University of Medical Sciences and Shahrekord University, Iran for all supports provided.
REFERENCES
- Açikgöz O, Gönenç S, Kayatekin BM, Uysal N, Pekçetin Ç, Şemin İ, et al. Methamphetamine causes lipid peroxidation and an increase in superoxide dismutase activity in the rat striatum. Brain Res. 1998;813(1):200-2.
- Akhlaghi M, Bandy B. Mechanisms of flavonoid protection against myocardial ischemia-reperfusion injury. J Mol Cell Cardiol. 2009;46(3):309-7.
- Al-Snafi A. The pharmacology of Anchusa italica and Anchusa strigosa: a review. Int J Pharm Pharm Sci. 2014;6(4):7-10.
- Andrews E, Tsai SY, Johnson S, Farrer J, Wagner J, Kopen G, et al. Human adult bone marrow-derived somatic cell therapy results in functional recovery and axonal plasticity following stroke in the rat. Exp Neurol. 2008;211(2):588-92.
- Asgari S, Setorki M, Rafieian-Kopaei M, Shahinfard N, Ansari R, Forouz Z. Postprandial hypolipidemic and hypoglycemic effects of Allium hertifolium and Sesamum indicum on hypercholesterolemic rabbits. J Pharm Pharmacol. 2012;6(15):1131-5.
- Aygul R, Kotan D, Demirbas F, Ulvi H, Deniz O. Plasma oxidants and antioxidants in acute ischaemic stroke. J Int Med Res. 2006;34(4):413-8.
- Baradaran A, Nasri H, Rafieian-Kopaei M. Comment on: anti-oxidative stress activity of Stachys lavanduli-folia aqueous extract in humans. Cell J. 2013;15(3):272-3.
- Barbakadze V, Gogilashvili L, Amiranashvili L, Merlani M, Mulkijanyan K, Churadze M, et al. Poly [3-(3, 4-dihydroxyphenyl) glyceric acid] from Anchusa italica roots. Nat Prod Commun. 2010;5(7):1091-5.
- Benzie IF, Strain J. [2] Ferric reducing/antioxidant power assay: direct measure of total antioxidant activity of biological fluids and modified version for simultaneous measurement of total antioxidant power and ascorbic acid concentration. Meth Enzymol. 1999;299:15-27.
- Bernard SA, GrayTW, Buist MD, Jones BM, Silvester W, Gutteridge G, et al. Treatment of comatose survivors of out-of-hospital cardiac arrest with induced hypothermia. N Engl J Med. 2002;346:557-63.
- Bokura H, Robinson RG. Long-term cognitive impairment associated with caudate stroke. Stroke. 1997;28(5):970-5.
- Chang CC, Yang MH, Wen HM, Chern JC. Estimation of total flavonoid content in propolis by two complementary colorimetric methods. J Food Drug Anal. 2002;10(3):178-82.
- Chen S, Liu J, Liu X, Fu Y, Zhang M, Lin Q, et al. Panax notoginseng saponins inhibit ischemia-induced apoptosis by activating PI3K/Akt pathway in cardiomyocytes. J Ethnopharmacol. 2011;137(1):263-70.
- Deckert J, Jorgensen MB. Evidence for pre-and postsynaptic localization of adenosine A1 receptors in the CA1 region of rat hippocampus: a quantitative autoradiographic study. Brain Res. 1988;446(1):161-4.
- Dekanski D, Selaković V, Piperski V, Radulović Ž, Korenić A, Radenović L. Protective effect of olive leaf extract on hippocampal injury induced by transient global cerebral ischemia and reperfusion in Mongolian gerbils. Phytomedicine. 2011;18(13):1137-43.
- Dominique JF, Roozendaal B, McGaugh JL. Stress and glucocorticoids impair retrieval of long-term spatial memory. Nature. 1998;394(6695):787-90.
- El Kossi MMH, Zakhary MM. Oxidative stress in the context of acute cerebrovascular stroke. Stroke. 2000;31(8):1889-92.
- Ghahremanitamadon F, Shahidi S, Zargooshnia S, Nikkhah A, Ranjbar, A, Soleimani Asl S. Protective effects of Borago officinalis extract on amyloid ß-peptide (25-35)-induced memory impairment in male rats: a behavioral study. Biomed Res Int. 2014;2014:798535.
- Gulluce M, Sahin F, Sokmen M, Ozer H, Daferera D, Sokmen A, et al. Antimicrobial and antioxidant properties of the essential oils and methanol extract from Mentha longifolia L. ssp. longifolia. Food Chem. 2007;103(4):1449-56.
- He H, Xu J, Xu Y, Zhang C, Wang H, He Y, et al. Cardioprotective effects of saponins from Panax japonicus on acute myocardial ischemia against oxidative stress-triggered damage and cardiac cell death in rats. J Ethnopharmacol. 2012;140(1):73-82.
- Janero DR. Malondialdehyde and thiobarbituric acid-reactivity as diagnostic indices of lipid peroxidation and peroxidative tissue injury. Free Radic Biol Med. 1990;9(6):515-40.
- Karatas F, Karatepe M, Baysar A. Determination of free malondialdehyde in human serum by high-performance liquid chromatography. Anal Biochem. 2002;311(1):76-9.
- Kawai K, Nitecka L, Ruetzler CA, Nagashima G, Joo F, Mies G, et al. Global cerebral ischemia associated with cardiac arrest in the rat: I. Dynamics of early neuronal changes. J Cereb Blood Flow Metab. 1992;12(2):238-49.
- Kazemi M. Essential oil composition of Anchusa italica from Iran. Chem Nat Compd. 2013;49(2):369-70.
- Kebriaee-Zadeh A. Overview of national drug policy of Iran. Iran J Pharm Res. 2010;2:1-2.
- Khalili M, Roghani M, Ekhlasi M. The effect of aqueous crocus sativus L. extract on intracerebroventricular streptozotocin-induced cognitive deficits in rat: a behavioral analysis. Iran J Pharm Res. 2010;8(3):185-91.
- Khare CP. Indian medicinal plants: an illustrated dictionary. Heidelberg: Springer Science & Business Media; 2008.
- Köksal E, Gülçin I. Antioxidant activity of cauliflower (Brassica oleracea L.). Turk J Agric For. 2008;32:65-78.
- Lipton P. Ischemic cell death in brain neurons. Physiol Rev. 1999;79(4):1431-568.
- McDonald S, Prenzler PD, Antolovich M, Robards K. Phenolic content and antioxidant activity of olive extracts. Food Chem. 2001;73(1):73-84.
- Middleton E, Kandaswami C, Theoharides TC. The effects of plant flavonoids on mammalian cells: implications for inflammation, heart disease, and cancer. Pharmacol Rev. 2000;52(4):673-751.
- Mozaffarian V. A dictionary of Iranian plant names: Latin, English, Persian. Tehran: Farhang Mo'aser; 1996.
- Mrudula G. Antistress and antioxidant effect of Borago officinalis leaves. J Drug Discovery Ther. 2014;2(18):1-9.
- Oms-Oliu G, Odriozola-Serrano I, Soliva-Fortuny R, Martín-Belloso O. The role of peroxidase on the antioxidant potential of fresh-cut 'Piel de Sapo'melon packaged under different modified atmospheres. Food Chem. 2008;106(3):1085-1092.
- Oruc EO, Sevgiler Y, Uner N. Tissue-specific oxidative stress responses in fish exposed to 2, 4-D and azinphosmethyl. Comp Biochem Physiol, Part C: Toxicol Pharmacol. 2004;137(1):43-51.
- Ozkul A, Akyol A, Yenisey C, Arpaci E, Kiylioglu N, Tataroglu C. Oxidative stress in acute ischemic stroke. J Clin Neurosci. 2007;14(11):1062-6.
- Pataki T, Bak I, Kovacs P, Bagchi D, Das DK, Tosaki A. Grape seed proanthocyanidins improved cardiac recovery during reperfusion after ischemia in isolated rat hearts. Am J Clin Nutr. 2002;75(5):894-9.
- Pourheydar B, Shahi M, Farjah GH, Javanmard M, Karimipour M, Atabaki F. Evaluation of apoptosis in hippocampal cells of rat following intravenous injection of bone marrow stromal cells in ischemia-reperfusion model. Urmia Med J. 2014;25:586-97.
- Pu F, Mishima K, Irie K, Motohashi K, Tanaka Y, Orito K, et al. Neuroprotective effects of quercetin and rutin on spatial memory impairment in an 8-arm radial maze task and neuronal death induced by repeated cerebral ischemia in rats. J Pharmacol Sci. 2007;104:329-34.
- Rabiei Z, Bigdeli M, Lorigooini Z. A Review of medicinal herbs with antioxidant properties in the treatment ?of cerebral ischemia and reperfusion. J Babol Univ Med Sci. 2015;17(12):47-56.
- Rafieian-Kopaei M, Baradaran A, Rafieian M. Oxidative stress and the paradoxical effects of antioxidants. J Res Med Sci. 2013;18(7):628.
- Shang YZ, Miao H, Cheng JJ, Qi JM. Effects of amelioration of total flavonoids from stems and leaves of Scutellaria baicalensis Georgi on cognitive deficits, neuronal damage and free radicals disorder induced by cerebral ischemia in rats. Biol Pharm Bull. 2006;29(4):805-10.
- Thom T, Haase N, Rosamond W, Howard VJ, Rumsfeld J, Manolio T, et al. Heart disease and stroke statistics-2006 update a report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation. 2006;113(6):e85-e151.
- Wettasinghe M, Shahidi F. Scavenging of reactive-oxygen species and DPPH free radicals by extracts of borage and evening primrose meals. Food Chem. 2000;70(1):17-26.
- White BC, Sullivan JM, DeGracia DJ, O'Neil BJ, Neumar RW, Grossman LI, et al. Brain ischemia and reperfusion: molecular mechanisms of neuronal injury. J Neurol Sci. 2000;179(S 1-2):1-33.
- Wolff SP, Dean R. Glucose autoxidation and protein modification: the potential role of 'autoxidative glycosylation' in diabetes. Biochem J. 1987;245(1):243-50.
- Zhang H, Chen F, Wang X, Yao HY. Evaluation of antioxidant activity of parsley (Petroselinum crispum) essential oil and identification of its antioxidant constituents. Food Res Int. 2006;39(8):833-9.
- Zhang J, Piantadosi CA. Prolonged production of hydroxyl radical in rat hippocampus after brain ischemia-reperfusion is decreased by 21-aminosteroids. Neurosci Lett 1994;177(1-2):127-30.
- Zhang S, Qi Y, Xu Y, Han X, Peng J, Liu K, et al. Protective effect of flavonoid-rich extract from Rosa laevigata Michx on cerebral ischemia-reperfusion injury through suppression of apoptosis and inflammation. Neurochem Int. 2013;63(5):522-32.
Publication Dates
-
Publication in this collection
2018
History
-
Received
06 May 2017 -
Accepted
07 Aug 2017