Abstracts
AIM: We evaluated the composition and community attributes of invertebrates associated to Eichhornia azurea at Cascalho Lagoon, Upper Paraná River, Mato Grosso do Sul State, Brazil, over a hydrological cycle, as well the possible influence of abiotic factors upon these attributes. METHODS: The samplings were conducted during 2010 in the rainy and dry periods at stands of E. azurea. The attributes evaluated were abundance, richness, diversity, evenness and dominance. The abiotic factors, temperature, dissolved oxygen, pH and turbidity were summarized by a Principal Component Analysis (PCA). In order to verify possible differences between the mean values of the community attributes and the scores of the PCA axis in different periods, we employed null models analysis of variance. The influence of abiotic factors on each attribute was evaluated through Pearson correlations. RESULTS: We captured 3,052 individuals, distributed into 32 taxa, belonging to the phyllum Mollusca, Annelida, Nematoda and Arthropoda. Among the assessed attributes, only abundance and richness varied significantly between periods, with higher values during the rainy period. Chironomidade was dominant in both periods, whereas Notonectidae and Cyclopoida were rare in the rainy, and Bivalve, Decapoda, Haliplidae, Trichoptera and Pyralidae, in the dry period. A temporal distinction was evident only for the PCA axis 1, which represented gradients in temperature, dissolved oxygen and pH. Among the community attributes, only abundance was significant and negatively correlated with this axis. CONCLUSION: We attested that: i) the rainy period should add favorable conditions for invertebrates' higher richness and abundance in this macrophyte; ii) only the later attribute was influenced by limnological gradients.
aquatic macrophytes; Chironomidae; limnological gradients; hydrological cycle; lentic environment
OBJETIVO: Nós avaliamos a composição e atributos da comunidade de invertebrados associados à Eichhornia azurea na lagoa do Cascalho, alto rio Paraná, MS, Brasil, durante um ciclo hidrológico, bem como a possível influência de fatores abióticos sobre estes atributos. MÉTODOS: Amostragens foram realizadas em bancos de E. azurea durante os períodos chuvoso e seco em 2010. Os atributos avaliados foram abundância, riqueza, diversidade, equitabilidade e dominância. As variáveis limnológicas temperatura, oxigênio dissolvido, pH e turbidez foram sumarizadas através da Análise de Componentes Principais (PCA). Para verificar possíveis diferenças entre as médias dos atributos e dos escores do eixo 1 da PCA nos diferentes períodos utilizou-se análises de variância de modelos nulos. A influência dos fatores abióticos sobre cada atributo foi avaliada através de correlações de Pearson. RESULTADOS: Nós capturamos 3052 indivíduos, distribuídos em 32 táxons, pertencentes aos filos Mollusca, Annelida, Nematoda e Arthropoda. Dentre os atributos avaliados, apenas a abundância e riqueza variaram significativamente entre os períodos, com maiores valores obtidos para o chuvoso. Chironomidade foi dominante em ambos os períodos enquanto Notonectidae e Cyclopoida foram raros no período chuvoso e Bivalve, Decapoda, Haliplidae, Trichoptera e Pyralidae no período seco. Distinção temporal foi evidente apenas para o eixo 1 da PCA, o qual representou gradientes de temperatura, oxigênio dissolvido e pH. Dentre os atributos avaliados, somente a abundância apresentou correlação significativa e negativa com este eixo. CONCLUSÃO: Ficou evidente que: i) o período chuvoso deve agregar condições favoráveis para a ocorrência de maior riqueza e elevada abundância de invertebrados nesta macrófita; ii) somente este último atributo foi influenciado pelos gradientes limnológicos.
macrófitas aquáticas; Chironomidae; gradientes limnológicos; ciclo hidrológico; ambiente lêntico
Invertebrates associated to Eichhornea azurea Kunth in a lagoon of the Upper Paraná River: composition, community attributes and influence of abiotic factors
Invertebrados associados à Eichhornea azurea Kunth em uma lagoa do alto rio Paraná: composição, atributos da comunidade e influência de fatores abióticos
Valéria Flávia Batista-SilvaI, II; Daiane Dias BonetoI, III; Dayani BaillyI, II; Milza Celi Fedatto AbelhaI, II; Elaine Antoniassi Luiz KashiwaquiI, II
IGrupo de Estudos em Ciências Ambientais e Educação - GEAMBE, Universidade Estadual de Mato Grosso do Sul - UEMS, BR 163, km 20,2, CEP 79980-000, Mundo Novo, MS, Brazil
IIUniversidade Estadual de Mato Grosso do Sul - UEMS, BR 163, km 20,2, CEP 79980-000, Mundo Novo, MS, Brazil
IIIGraduada em Ciências Biológicas, Universidade Estadual de Mato Grosso do Sul - UEMS, BR 163, km 20,2, CEP 79980-000, Mundo Novo, MS, Brazil e-mail: vfb_silva@uems.br; daibiologia1@hotmail.com; dayanibailly@gmail.com; mcfabelha@yahoo.com.br; elainealk@yahoo.com.br
ABSTRACT
AIM: We evaluated the composition and community attributes of invertebrates associated to Eichhornia azurea at Cascalho Lagoon, Upper Paraná River, Mato Grosso do Sul State, Brazil, over a hydrological cycle, as well the possible influence of abiotic factors upon these attributes.
METHODS: The samplings were conducted during 2010 in the rainy and dry periods at stands of E. azurea. The attributes evaluated were abundance, richness, diversity, evenness and dominance. The abiotic factors, temperature, dissolved oxygen, pH and turbidity were summarized by a Principal Component Analysis (PCA). In order to verify possible differences between the mean values of the community attributes and the scores of the PCA axis in different periods, we employed null models analysis of variance. The influence of abiotic factors on each attribute was evaluated through Pearson correlations.
RESULTS: We captured 3,052 individuals, distributed into 32 taxa, belonging to the phyllum Mollusca, Annelida, Nematoda and Arthropoda. Among the assessed attributes, only abundance and richness varied significantly between periods, with higher values during the rainy period. Chironomidade was dominant in both periods, whereas Notonectidae and Cyclopoida were rare in the rainy, and Bivalve, Decapoda, Haliplidae, Trichoptera and Pyralidae, in the dry period. A temporal distinction was evident only for the PCA axis 1, which represented gradients in temperature, dissolved oxygen and pH. Among the community attributes, only abundance was significant and negatively correlated with this axis.
CONCLUSION: We attested that: i) the rainy period should add favorable conditions for invertebrates' higher richness and abundance in this macrophyte; ii) only the later attribute was influenced by limnological gradients.
Keywords: aquatic macrophytes, Chironomidae, limnological gradients, hydrological cycle, lentic environment.
RESUMO
OBJETIVO: Nós avaliamos a composição e atributos da comunidade de invertebrados associados à Eichhornia azurea na lagoa do Cascalho, alto rio Paraná, MS, Brasil, durante um ciclo hidrológico, bem como a possível influência de fatores abióticos sobre estes atributos.
MÉTODOS: Amostragens foram realizadas em bancos de E. azurea durante os períodos chuvoso e seco em 2010. Os atributos avaliados foram abundância, riqueza, diversidade, equitabilidade e dominância. As variáveis limnológicas temperatura, oxigênio dissolvido, pH e turbidez foram sumarizadas através da Análise de Componentes Principais (PCA). Para verificar possíveis diferenças entre as médias dos atributos e dos escores do eixo 1 da PCA nos diferentes períodos utilizou-se análises de variância de modelos nulos. A influência dos fatores abióticos sobre cada atributo foi avaliada através de correlações de Pearson.
RESULTADOS: Nós capturamos 3052 indivíduos, distribuídos em 32 táxons, pertencentes aos filos Mollusca, Annelida, Nematoda e Arthropoda. Dentre os atributos avaliados, apenas a abundância e riqueza variaram significativamente entre os períodos, com maiores valores obtidos para o chuvoso. Chironomidade foi dominante em ambos os períodos enquanto Notonectidae e Cyclopoida foram raros no período chuvoso e Bivalve, Decapoda, Haliplidae, Trichoptera e Pyralidae no período seco. Distinção temporal foi evidente apenas para o eixo 1 da PCA, o qual representou gradientes de temperatura, oxigênio dissolvido e pH. Dentre os atributos avaliados, somente a abundância apresentou correlação significativa e negativa com este eixo.
CONCLUSÃO: Ficou evidente que: i) o período chuvoso deve agregar condições favoráveis para a ocorrência de maior riqueza e elevada abundância de invertebrados nesta macrófita; ii) somente este último atributo foi influenciado pelos gradientes limnológicos.
Palavras-chave: macrófitas aquáticas, Chironomidae, gradientes limnológicos, ciclo hidrológico, ambiente lêntico.
1. Introduction
Lentic environments, specifically marginal lagoons, usually possess at their littoral regions extensive macrophyte stands (Schreiber and Brauns, 2010), which constitute abundant substrate for aquatic invertebrates (Albertoni et al., 2007; Ohtaka et al., 2011). The association of invertebrates to aquatic macrophytes creates numerous benefits for these, as direct or indirect obtaining of food, shelter, oxygen and other favorable conditions for their growth (Padial et al., 2009; Ohtaka et al., 2011).
Compounding this type of vegetation, Eichhornia azurea Kunth, a rooted aquatic macrophyte with long stems and emerged floating leaves (Padial et al., 2009), stands out by the efficiency in colonizing lentic waters, and is frequently pointed out as abundant in Brazilian lakes (Esteves, 1998; Thomaz et al., 2004). In the littoral region of this type of environment the invertebrates are prone to present higher diversity and abundance when associated to E. azurea because vegetated areas typically create more favorable conditions for organisms' survival than open water zone (Bazzanti et al., 2010). Besides the benefits above mentioned, aquatic macrophytes provide more heterogeneous habitats and stability to environmental conditions (Padial et al., 2009; Ferreiro et al., 2011).
In freshwater environments, invertebrates have important ecological role in fragmentation and decomposition of organic matter, establishing a strategic link for the transfer of energy and nutrients between macrophytes and organisms at upper trophic levels (Esteves, 1998). In this way, the maintenance of this community structure is essential for the balanced functioning of aquatic environments.
In recent decades several studies on invertebrates associated to aquatic macrophytes have been developed in different aquatic environments, focusing mainly the community structure (Thomaz et al., 2008; Ferreiro et al., 2011; Ohtaka et al., 2011). Our work aggregates information to this context through the description of macrophytes-invertebrates association in a floodplain lagoon unprecedentedly studied. Specifically, our study aimed at evaluating the composition and community attributes (abundance, richness, diversity, evenness and dominance) of invertebrates associated to Eichhornia azurea at Cascalho Lagoon, Mato Grosso do Sul State, over a rainy and a dry period. We tested the hypothesis that: i) the community attributes (abundance, richness, diversity and evenness) change in the rainy and dry periods; ii) the abiotic factors prevailing in the different periods influence these attributes.
2. Material and Methods
2.1.Study area
The Cascalho Lagoon (24° 02' 28.4" S and 54° 14' 00.2" W) has permanent connection with the right margin of the Upper Paraná River and is located next to the Ilha Grande National Park (Figure 1). The region corresponds to the beginning of the riverine zone of Itaipu Reservoir (see Oliveira et al., 2004), distant around 20 km from Mundo Novo Municipality, Mato Grosso do Sul State. It is inserted in the Upper Paraná River floodplain and consists in an artificial environment built in 1986 from excavations to create an area for recreation and for boat anchoring. This environment presents about 148.0 m length, 108.0 m width, and 733.0 m perimeter. The margins are colonized by grass and other aquatic macrophytes, such as Eichhornia crassipes, Utricularia gibba, Salvinia sp. and, mostly, Eichhornia azurea.
2.2.Samplings
To cover the main phases of the hydrological cycle, we conducted two samplings in the rainy period (February and March 2010) and other two in the dry period (June and August 2010) in two stands of Eichhornia azurea located at the end of the Cascalho Lagoon, in relation to the stretch of connection with the Paraná River. In each macrophyte stand, we collected two samples, each one compounded by vegetative structures (set of leaves, petiole and roots) of E. azurea, for the determination of associate invertebrates. Each sample was kept in plastic bags labeled and preserved in 75% alcohol.
In the laboratory, the samples were washed with running water onto a sieve (0.5 mm mesh size) and scraped with blades aiming the detachment of invertebrates associated to vegetative structures. The material retained on the sieve was preserved in 75% alcohol.
The samples were sorted using stereoscopic microscope and the invertebrates were identified with the aid of specialized literature (Merritt and Cummins, 1996; Nieser and Melo, 1997).
2.3.Data analysis
The community attributes evaluated were abundance, richness, diversity, evenness and dominance. To standardize the numerical abundances, these were expressed in grams per macrophyte dry weight. Here we explained in details the methodology used to estimate dry weight. To do that, after the material washing, the vegetative structure of each sample was fragmented into small pieces and homogenized. From this material, we removed, at random, 100 g of wet weight to determine the dry weight. These subsamples were wrapped in aluminum foil and taken to oven drying at 60 °C until the weight stabilization given by complete dehydration. Final weight of each subsample corresponded to the dry weight value.
Thus, it was possible to estimate the dry weight value of each sample through the expression (Equation 1):
where DWStotal = total dry weight of the sample, WWStotal = total wet weight of the sample, and DWSS = dry weight of the subsample. The invertebrate abundance in each sample was calculated by dividing the number of individuals by the value of DWStotal, expressed in number of individuals per 100 g of dry weight.
The richness, diversity and evenness were obtained according to Pielou (1969). The richness (S) was defined as the number of taxa captured. The diversity was calculated through the Shannon-Wiener index (H') defined as (Equation 2):
where ni = number of individuals of the taxa i and N = total number of individuals. The evenness (E) was calculated according to Equation 3:
where H' = Shannon-Wiener index and S = number of taxa. These attribute were estimated using the software PC-ORD 4.0 (McCune and Mefford, 2007).
The pattern of species dominance for the rainy and dry periods was evaluated through species-abundance relationship curves (Magurran, 1988). To obtain these curves, the species were ranked in decreasing abundances log-transformed (log10 = x + 1). It was established arbitrarily that rare species would be those with abundance equal to or lower to 1.0 individual per 100 g of dry weight.
Concurrently to the invertebrate samplings, water samples were taken to measure some abiotic variables such as temperature (°C), dissolved oxygen (mg/l), pH and turbidity (NTU).
The matrix of abiotic data was summarized through a Principal Component Analysis (PCA), using the software PC-ORD 4.0 (McCune and Mefford, 2007). The axes with eigenvalues higher than 1 (Kaiser-Guttman criterion - Jackson, 1993) were retained to interpret the ordination of abiotic variables in the different phases of the hydrological cycle.
Considering 16 as the total number of observations (4 samples in each month, totalizing 8 samples in each period), in order to check possible differences between the mean values of abundance, richness, diversity and evenness and the PCA scores axes in the different periods (rainy and dry) we employed one-factor analysis of variance (ANOVA null models - Gotelli and Graves, 1996; Gotelli and Entsmiger, 2007). In this analysis, each attribute was the dependent variable, and the periods, the independent variable. In the ANOVAs we used the EcoSim software (Gotelli and Entsmiger, 2007) and entailed significant differences at p < 0.05, after 10.000 randomizations.
The possible influence of abiotic factors on the community attributes was examined through Pearson correlations between the scores of the significant axes and the abundance, richness, diversity and evenness, using the Statistica 7.0 software (Statsoft, 2005).
3. Results
In this study, 3.052 individuals were collected, distributed into 32 taxa. Among these, 2 belonged to the phylum Mollusca (Bivalve and Gastropoda), 1 to phylum Annelida (Oligochaeta) and Nematoda, and 28 to the phylum Arthropoda, distributed among Crustacea (Decapoda, Chydoridae, Cyclopoida, Calanoida), Arachnida (Acarina) and Insecta. In general, the insects were the most representative in richness, with the identification of 6 orders and 22 families (Table 1). The most abundant taxa were Chironomidae (1,541.6 ind/100 g) and Gastropoda (474.1 ind/g) that together corresponded to 55.2% of the total collected.
The mean abundance varied significantly (OI = 16.3 and pobs> pexp = 0.003) between the phases of the hydrological cycle, with higher values in the rainy period (Figure 2a). In relation to the richness (Figure 2b), diversity (Figure 2c) and evenness (Figure 2d), only the first presented mean values significantly different (OI = 11.1 and pobs> pexp = 0.009) between rainy and dry periods.
Despite the occurrence of 32 taxa during the study period, the dominance was restricted to few groups. In this way, the rainy periods with 26 taxa presented four dominant groups: Chironomidade (1,101.8 ind/100 g), Gastropoda (391.2 ind/100 g), Oligochaeta (347.4 ind/100 g) and Ceratopogonidae (313.4 ind/100 g). Notonectidae and Cyclopoida were rare (Figure 3a), these added to Belostomatidae, Chaoboridae, Elmidae, Noteridae, Acarina and Aeshinidae corresponded to groups exclusive to this period.
During the dry period, 24 taxa were recorded, with dominance again of Chironomidae (439.8 ind/100 g), but with expressive decrease in abundance compared to previous period. Rare taxa were represented by Bivalve, Decapoda, Haliplidae, Trichoptera and Pyralidae (Figure 3b). Except the last, these taxa added to Velidae and Calanoida were exclusives of this period.
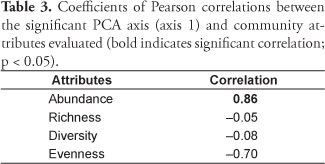
The first two PCA axes were retained for interpretation (both > 1.0) and together explained 92.64% of the data variability. The first axis accumulated greater percentage of explanation (61.3%), representing a gradient in temperature, dissolved oxygen and pH. Dissolved oxygen and pH were positively correlated with the axis 1 (r = 0.59 and r = 0.57, respectively), whereas temperature was negatively correlated to it (r = -0.57). For the axis 2, only a negative correlation with turbidity was found (r = -0.88). Meanwhile, the temporal distinction was evident only for the axis 1 (Figure 4), observation confirmed by the analysis of variance (OI = 32.38 and pobs> pexp = 0.01 for the axis 1, and OI = 0.35 and pobs> pexp = 0.57 for the axis 2). The mean values of abiotic factors are listed in Table 2. Among evaluated attributes, only the abundance (r = -0.86, p < 0.05) presented a significant correlation with the PCA axis 1 (Table 3).
4. Discussion
The composition and number of taxa of invertebrates associated to E. azurea at Cascalho Lagoon were similar to those often associated to aquatic vegetation from lentic environments (Trivinho-Strixino et al., 2000; Albertoni et al., 2007; Souza-Franco et al., 2009; Kouamé et al., 2011). However the number of taxa found in these studies was higher (on Family level) than those pointed out by investigations that disregard the association macrophytes-invertebrates (Marques et al., 1998; Lucca et al., 2010). This finding could be an indication of aquatic macrophytes as good samplers of invertebrate community in this type of environment.
The high insect taxonomic representativeness and abundance of this group was a pattern also observed in other studies, regardless the type of sampled substrate (Mormul et al., 2006; Albertoni et al., 2007; Thomaz et al., 2008; Souza-Franco et al., 2009; Lucca et al., 2010). Among other aspects, the morphological, physiological and behavioral adaptations provides to the group a versatility, both in colonization of sediments and of different types of aquatic vegetation (Merritt and Cummins, 1996; Nessimian and Carvalho, 1998).
The species composition in any environment is ruled by prevailing environmental conditions (Begon et al., 2007). Specifically, marginal lagoons are subjected to hydrological inflows variable in intensity and frequency, depending on their location on the floodplain and on their connectivity degree with the water course (Henry and Costa, 2003). Considering that the Cascalho Lagoon presents immediate connection with the channel of the Paraná River, it is expected that the aquatic organisms present would be closely linked to the dynamics of this river.
With respect to the abundance, the obtained result of few dominant species and many rare ones followed the general pattern of biological communities described by Begon et al. (2007). However, the dominance's higher values found in the rainy period can be justified by two main factors. The first refers to the type of marginal vegetation studied, which provides greater resistance to the flood dilution effect [see Junk et al. (1989) and Neiff (1990) for more details about flood dilution effect], because they are rooted and provide broad spatial structuration. Accordingly to this last factor, small spaces between the ramifications of the macrophytes roots may serve as refuge during floods and stronger water currents (Mello et al., 2004). The second concerns to the intensification of the macrophytes decay process, which increases the palatability and nutritional value of its detritus (Mormul et al., 2006; Stripari and Henry, 2002; Ferreiro et al., 2011), making this an abundant and feasible resource for the increase of invertebrate populations that forage on them. This is due to the fact that during warmer and rainy months, the macrophytes stands in lentic environments are prone to increase in biomass resulting in a greater availability of organic matter (Esteves, 1998). Although we did not quantify the macrophytes biomass in Cascalho Lagoon in both periods, visually we observed the increase of this vegetation during the rainy period.
Considering that the highest availability of food resource tends to increase the richness by reducing interspecific competition (Gotelli and Graves, 1996), it is assumed that higher mean values of this attribute recorded in the rainy period were governed by the intensification of macrophytes decay process and its availability as food resource, as it was pointed out in the above paragraph. Nevertheless, the similarity in the total richness between periods (28-rainy and 26-dry) could be explained by the remarkable replacement of taxa between the months of the dry period.
Regarding the influence of limnological factors on the invertebrates' community attributes, our results suggested an important role of temperature, pH and oxygen on the organisms' abundance, whereas richness, diversity and evenness were not associated with the limnological gradients. The temperature affects ectothermic organisms like the invertebrates in a way that their physiology and, consequently, their distribution and abundance are temperature dependent (Silveira, 2004). Also, the temperature influences negatively oxygen (and other gases) solubility in the water and may alter its pH when the decomposition of organic matter is accelerated by temperature rises (Esteves, 1998).
In this way, the increase in abundance with the reduction in the values of oxygen and pH in the rainy period could be linked, among other factors, to the effect of temperature on these limnological variables. This may be due to the increase in the organic matter input coming from the water inflow and flooding of the margins during the rainy period, which presents higher temperatures, leading to an increase of decay rate, and consequent decrease of dissolved oxygen and pH (Simião-Ferreira and Carvalho, 2008).
With respect to the dominance of Chironomidae in the studied periods, it should be related to the wide tolerance of some species to extreme conditions which enable them to effectively colonize different environments (Di Giovani et al., 1996; Sanseverino and Nessimian, 1998; Marques et al., 1999). The dominance of this family among aquatic invertebrates has been observed in several studies, both in natural (Lucca et al., 2010), and degraded environments (Bieger et al., 2010; Paula and Fonseca-Gessner, 2010). Additionally, Chironomidae forages on detritus and periphyton (Marques et al., 1999; Trivinho-Strixino et al., 2000) and this fact allows them to find in the aquatic macrophytes stands abundant food source since these plants act as substrates that accumulate organic matter and periphyton, besides themselves being an important source of detritus in the ecosystem (Minshall, 1984; Rodrigues et al., 2003).
In addition to Chironomidae, the high abundance that we found for groups such as Gastropoda, Oligochaeta and Ceratopogonidae, especially in the rainy period, was coherent to literature data. These taxa were reported as the dominant ones in the same period in a French lake (Kouamé et al., 2011) and a high abundance of Gastropoda, Oligochaeta and Ceratopogonidae was also detected in Brazilian lagoons where they were described as common members of the macrophyte associated communities (Takeda et al., 2003; Takeda and Fujita, 2004; Campos, 2010; Ohtaka, et al., 2011). The high surface area of E. azurea was poited out as a favorable environment for Gastropoda (Takeda et al., 2003), since these organisms scrape the epidermis of aquatic plants (Mormul et al., 2006). The taxon Oligochaeta includes individuals that are detritivorous and tolerant to anoxic conditions (Goulart and Callisto, 2003). These features should be related to the dominance of this group in the rainy period, due the intense decay and lower values of dissolved oxygen as mentioned earlier. In relation to Ceratopogonidae, despites the prevalence of a predator behavior in the adult, immature of some species ingest algae (Merritt and Cummins, 1996) and this might be the reason of its association to macrophytes (Campos, 2010), which are commonly colonized by abundant periphyton (Rodrigues et al., 2003; Molina et al., 2011).
With respect to the observed rare taxa (Notonectidae and Cyclopoida in the rainy and Bivalve, Haliplidae, Trichoptera and Pyralidae in the dry period), they were similarly registered in other lentic environments colonized by macrophytes (Velho et al., 2001; Takeda et al., 2003; Mormul et al., 2006; Albertoni et al., 2007). However, most of this information was restricted to occurrence, preventing comparisons of rarity.
Therefore, answering the established hypotheses, our results allowed to partially accept them: i) among the investigated attributes only abundance and richness were significantly different between the periods, indicating that the rainy period should add favorable conditions for high richness and abundance in E. azurea stands at Cascalho Lagoon; ii) only the abundance was significantly correlated with limnological gradient, suggesting that the process of species substitution in the dry period was intense enough to maintain the other attributes (richness, diversity and evenness) relatively constanty.
Acknowledgements
The authors are grateful to the Universidade Estadual de Mato Grosso do Sul (UEMS, Mundo Novo/MS) for logistic support and Dr. Carlos Alexandre Fernandes for elaboration of the map.
Received: 11 August 2011
Accepted: 13 April 2012
- ALBERTONI, EF., PRELLVITZ, LJ. and PALMA-SILVA, C. 2007. Macroinvertebrate fauna associated with Pistia stratiotes and Nymphoides indica in subtropical lakes (South Brazil). Brazilian Journal of Biology, vol. 67, p. 499-507. http://dx.doi.org/10.1590/S1519-69842007000300015
- BAZZANTI, M., COCCIA, C. and DOWGIALLO, MG. 2010. Microdistribution of macroinvertebrates in a temporary pond of Central Italy: taxonomic and functional analyses. Limnologica Ecology and Management of Inland Waters, vol. 40, p. 291-299. http://dx.doi.org/10.1016/j.limno.2009.10.006
- BEGON, M., TOWNSEND, CR. and HARPER, JR. 2007. Ecologia: de indivíduos a ecossistemas. 4th ed. Porto Alegre: Artmed. 752 p.
- BIEGER, L., CARVALHO, ABP., STRIEDER, MN, MALTCHIK, L. and STENERT, C. 2010. Are the streams of the Sinos River basin of good water quality? Aquatic macroinvertebrates may answer the question. Brazilian Journal of Biology, vol. 70, no. 4, p. 1207-1215. http://dx.doi.org/10.1590/S1519-69842010000600010
- CAMPOS, RLE. 2010. Eryngium (Apiaceae) phytotelmata and their macroinvertebrate communities, including a review and bibliography. Hydrobiologia, vol. 652, p. 311-328. http://dx.doi.org/10.1007/s10750-010-0364-y
- DI GIOVANI, MV., GORETTI, E. and TAMANTI, V. 1996. Macrobenthos in Montedoglio Reservoir, central Italy. Hydrobiologia, vol. 321, p. 17-28. http://dx.doi.org/10.1007/BF00018673
- ESTEVES, FA. 1998. Fundamentos de limnologia 2nd ed. Rio de Janeiro: Interciência. 575 p.
- FERREIRO, N., FEIJOÓ, C., GIORGI, A. and LEGGIERI, L. 2011. Effects of macrophyte heterogeneity and food availability on structural parameters of the macroinvertebrate community in a Pampean stream. Hydrobiologia, vol. 664, p. 199-211. http://dx.doi.org/10.1007/s10750-010-0599-7
- GOTELLI, NJ. and ENTSMINGER, GL. 2007. EcoSim: null models software for ecology. version 7.0. Jericho, Vermont, Acquired Intelligence Inc. & Kesey-Bear. Available from: <http://garyentsminger.com/ecosim.htm>
- GOTELLI, NJ. and GRAVES, GR. 1996. Null models in ecology Washington: Smithsonian Institution Press. 368 p.
- GOULART, M. and CALLISTO, M. 2003. Bioindicadores de qualidade de água como ferramenta em estudos de impacto ambiental. Revista da FAPAM, vol. 2, no. 2, p. 53-164.
- HENRY, R. and COSTA, MLR. 2003. As macrófitas como fator de heterogeneidade espacial: um estudo em três lagoas com diferentes conectividades com o rio Paranapanema. In THOMAZ, SM. and BINI, LM., orgs. Ecologia e manejo de macrófitas aquáticas. Maringá: EDUEM. 341 p.
- JACKSON, DA. 1993. Stopping rules in principal components analysis: a comparison of heuristical and statistical approaches. Ecology, vol. 74, p. 2204-2214. http://dx.doi.org/10.2307/1939574
- JUNK, WJ., BAYLEY, PB. and SPARKS, RE. 1989. The flood pulse concept in river floodplain systems. Canadian Journal of Fisheries and Aquatic Science, vol. 106, p. 110-127.
- KOUAMÉ, MK., DIETOA, MY., EDIA, EO., COSTA, SK., OUATTARA, A. and GOURÈNE, G. 2011. Macroinvertebrate communities associated with macrophyte habitats in a tropical man-made lake (Lake Taabo, Côte d'Ivoire). Knowledge and Management of Aquatic Ecosystems, vol. 400, no. 3, p. 3-18.
- LUCCA, JV., PAMPLIN, P., GESSNER, AF., TRIVINHO-STRIXINO, S., SPADANO-ALBUQUERQUE, AL. and ROCHA, O. 2010. Benthic macroinvertebrates of a tropical lake: Lake Caçó, MA, Brazil. Brazilian Journal of Biology, vol. 70, no. 3, p. 593-600. PMid:20730346.
- MAGURRAN, AE. 1998. Ecological diversity and its measurement London: Chapman e Hall. 178 p.
- MARQUES, MMGSM., BARBOSA, FAR. and CALLISTO, M. 1999. Distribution and abundance of Chironomidae (Diptera, Insecta) in an impacted watershed in South-East Brazil. Brazilian Journal of Biology, vol. 59, no. 4, p. 553-561.
- MARQUES, MMGSM., FERREIRA, RL. and BARBOSA, FAR. 1998. A comunidade de macroinvertebrados aquáticos e características limnológicas das lagoas Carioca e da Barra, Parque Estadual do Rio Doce, MG. Revista Brasileira de Biologia, vol. 59, no. 2, p. 203-210.
- MINSHALL, GW. 1984. Aquatic insect-substratum relationships. In RESH, VH. and ROSENBERG, DM., orgs. The ecology of aquatic insects New York: Praeger Publishers. 625 p.
- McCUNE, B. and MEFFORD, MJ. 2007. PC-ORD: multivariate analysis of ecological data. version 4.0. Oregon: MjM Software Design.
- MELLO, SM., TAKEDA, AM., GRZYBKOWSKA, M. and MONKOLSKI, A. 2004. Distribution of ephemeropteran nymphs associated with different stolon sections of Eichhornia azurea (Schwartz) in two floodplain lakes of the Upper Paraná River (Brazil). Polish Journal of Ecology, vol. 52, no. 3, p. 369-376.
- MERRITT, RW. and CUMMINS, KW. 1996. An introduction to the aquatic insects of North America 3rd ed. Dubuque: Kendal/Hunt. 862 p.
- MOLINA, CI., GIBON, FM., OBERDORFF, T., DOMINGUEZ, E., PINTO, J., MARÍN, R. and ROULET, M. 2011. Macroinvertebrate food web structure in a floodplain lake of the Bolivian Amazon. Hydrobiologia, vol. 663, p. 135-153. http://dx.doi.org/10.1007/s10750-010-0565-4
- MORMUL, RP., VIEIRA, LA., PRESSINATTE JÚNIOR, S., MONKOLSKI, A. and SANTOS, AM. 2006. Sucessão de invertebrados durante o processo de decomposição de duas plantas aquáticas (Eichhornia azurea e Polygonum ferrugineum). Acta Scientiarum Biological Sciences, vol. 28, no. 2, p. 109-115.
- NEIFF, JJ. 1990. Ideas para la interpretacion ecologica del Paraná. Interciência, vol. 15, p. 424-441.
- NESSIMIAN, JL. and CARVALHO, AL. 1998. Ecologia de insetos aquáticos Rio de Janeiro: PPGE-UFRJ. 309 p.
- NIESER, N. and MELO, AL. 1997 Os heterópteros aquáticos de Minas Gerais: guia introdutório com chave de identificação para as espécies de Nepomorpha e Gerromorpha. Belo Horizonte: Editora UFMG. 180 p.
- OHTAKA, A., NARITA, T., KAMIYA, T., KATAKURA, H., ARAKI, Y., IM, S., CHHAY, R. and TSUKAWAKI, S. 2011. Composition of aquatic invertebrates associated with macrophytes in Lake Tonle Sap, Cambodia. Limnology, vol. 12, p. 137-144. http://dx.doi.org/10.1007/s10201-010-0330-4
- OLIVEIRA, EF., GOULART, E. and MINTE-VERA, CV. 2004. Fish diversity along spatial gradients in the Itaipu Reservoir, Paraná, Brazil. Brazilian Journal of Biology, vol. 64, no. 3a, p. 447-458. PMid:15622842.
- PADIAL, A., THOMAZ, S. and AGOSTINHO, AA. 2009. Effects of structural heterogeneity provided by the ûoating macrophyte Eichhornia azurea on the predation efûciency and habitat use of the small Neotropical ûsh Moenkhausia sanctaeûlomenae Hydrobiologia, vol. 624, p. 161-170. http://dx.doi.org/10.1007/s10750-008-9690-8
- PAULA, MC. and FONSECA-GESSNER, AA. 2010. Macroinvertebrates in low-order streams in two fragments of Atlantic Forest in different states of conservation, in the State of São Paulo. Brazilian Journal Biology, vol. 70, no. 3, p. 899-909. PMid:21085795.
- PIELOU, EC. 1969. Association tets versus homogeneity test: their use in subdividing quadrats into groups. Vegetatio, vol. 19, p. 4-18.
- RODRIGUES, L., BICUDO, DC. and MOSCHINI-CARLOS, V. 2003. O papel do perifíton em áreas alagáveis e nos diagnósticos ambientais. In THOMAZ, SM. and BINI, LM., orgs. Ecologia e manejo de macrófitas aquáticas. Maringá: EDUEM. 341 p.
- SANSEVERINO, AM. and NESSIMIAN, J L. 1998. Habitat preference of Chironomidae larvae in an upland stream of Atlantic Forest, Rio de Janeiro State, Brazil. Verhandlungen der Interna-tioalen Vereinigunf für theoretische und angewandre Limnologie, vol. 26, p. 2141-2144.
- SCHREIBER, J. and BRAUNS, M. 2010. How much is enough? Adequate sample size for littoral macroinvertebrates in lowland lakes. Hydrobiologia, vol. 649, p. 365-373. http://dx.doi.org/10.1007/s10750-010-0284-x
- SILVEIRA, MP. 2004. Aplicação do biomonitoramento para avaliação da qualidade da água em rios Jaguariúna: Embrapa Meio Ambiente. 68 p. (Documentos, no. 26).
- SIMIÃO-FERREIRA, J. and CARVALHO, AR. 2008. Biomonitoramento do efluente da ETE do distrito agroindustrial de Anápolis, Goiás (DAIA). Health and Environment Journal, vol. 9, no. 1, p. 47-56.
- SOUZA-FRANCO, GM., ANDRIAN, LF. and FRANCO, RM. 2009. Comunidade de insetos aquáticos associados à Eichhornia azurea (Schwartz) Kunth, em uma lagoa de várzea na planície de inundação do Alto Rio Paraná, Mato Grosso do Sul, MS, Brasil. Biológico, vol. 71, no. 1, p. 83-91.
- STASOFT. 2005. Statistica version 7.0. Tulsa: StatSoft Inc.
- STRIPARI, NL. and HENRY, R. 2002. The invertebrate colonization during decomposition of Eichhornia azurea Kunth in a lateral lake in the mouth zone of Paranapanema River into Jurumirim Resevoir (São Paulo, Brazil). Brazilian Journal of Biology, vol. 62, no. 2, p. 293-310. PMid:12494917.
- TAKEDA, AM. and FUJITA, DS. 2004. Benthic invertebrates. In THOMAZ, SM., AGOSTINHO, AA. and HAHN, NS., orgs. The Upper Paraná River and its floodplain: physical aspects, ecology and conservation. Leiden: Blackhuys Publishers. 393 p.
- TAKEDA, AM., SOUZA-FRANCO, GM., MELO, SM. and MONKOLSKI, A. 2003. Invertebrados associados às macrófitas aquáticas da planície de inundação do alto rio Paraná (Brasil). In THOMAZ, SM. and BINI, LM., orgs. Ecologia e manejo de macrófitas aquáticas. Maringá: EDUEM. 341 p.
- THOMAZ, SM., BINI, LM., PAGIORO, TA., MURPHY, KJ., SANTOS, AM. and SOUZA, DC. 2004. Aquatic macrophytes: diversity, biomass and decomposition. In THOMAZ, SM, AGOSTINHO, AA, and HAHN, NS., orgs. The Upper Paraná River and its floodplain: physical aspects, ecology and conservation. Leiden: Backhuys Publishers. 393 p.
- THOMAZ, SM., DIBBLE, ED., EVANGELISTA, LR., HIGUTI, J. and BINI, LM. 2008. Influence of aquatic macrophyte habitat complexity on invertebrate abundance and richness in tropical lagoons. Freshwater Biology, vol. 53, p. 358-367.
- TRIVINHO-STRIXINO, S., CORREIA, LCS. and SONODA, K. 2000. Phytophilous Chironomidae (Diptera) and other macroinvertebrates in the ox-bow Infernão Lake (Jataí Ecological Station, Luiz Antônio, SP, Brazil). Revista Brasileira de Biologia, vol. 60, no. 3, p. 527-535. PMid:11188879. http://dx.doi.org/10.1590/S0034-71082000000300018
- VELHO, LFM., LANSAC-TÔHA, FA., TAKEDA, AM., HIGUTI, J. and FRANCO, GMS. 2001. Structure and dynamics of the cyclopoid copepod (Crustacea) assemblage associated with aquatic macrophytes in two lotic environments of the Upper Paraná River basin, Brazil. Acta Scientiarum, v. 23, no. 2, p. 349-356.
Publication Dates
-
Publication in this collection
14 June 2012 -
Date of issue
Dec 2011
History
-
Received
11 Aug 2011 -
Accepted
13 Apr 2012