Abstract
Introduction:
Agonistic behaviors help to ensure survival, provide advantage in competition, and communicate social status. The resident-intruder paradigm, an animal model based on male intraspecific confrontations, can be an ethologically relevant tool to investigate the neurobiology of aggressive behavior.
Objectives:
To examine behavioral and neurobiological mechanisms of aggressive behavior in male Swiss mice exposed to repeated confrontations in the resident intruder paradigm.
Methods:
Behavioral analysis was performed in association with measurements of plasma corticosterone of mice repeatedly exposed to a potential rival nearby, but inaccessible (social instigation), or to 10 sessions of social instigation followed by direct aggressive encounters. Moreover, corticotropin-releasing factor (CRF) and brain-derived neurotrophic factor (BNDF) were measured in the brain of these animals. Control mice were exposed to neither social instigation nor aggressive confrontations.
Results:
Mice exposed to aggressive confrontations exhibited a similar pattern of species-typical aggressive and non-aggressive behaviors on the first and the last session. Moreover, in contrast to social instigation only, repeated aggressive confrontations promoted an increase in plasma corticosterone. After 10 aggressive confrontation sessions, mice presented a non-significant trend toward reducing hippocampal levels of CRF, which inversely correlated with plasma corticosterone levels. Conversely, repeated sessions of social instigation or aggressive confrontation did not alter BDNF concentrations at the prefrontal cortex and hippocampus.
Conclusion:
Exposure to repeated episodes of aggressive encounters did not promote habituation over time. Additionally, CRF seems to be involved in physiological responses to social stressors.
Keywords:
Aggression; social instigation; corticosterone; CRF; BDNF
Resumo
Introdução:
Comportamentos agonísticos ajudam a garantir a sobrevivência, oferecem vantagem na competição e comunicam status social. O paradigma residente-intruso, modelo animal baseado em confrontos intraespecíficos entre machos, pode ser uma ferramenta etológica relevante para investigar a neurobiologia do comportamento agressivo.
Objetivos:
Analisar os mecanismos comportamentais e neurobiológicos do comportamento agressivo em camundongos Swiss machos expostos a confrontos repetidos no paradigma residente-intruso.
Métodos:
A análise comportamental foi realizada em associação com medidas de corticosterona plasmática em camundongos expostos repetidamente a um rival em potencial próximo, porém inacessível (instigação social), ou a 10 sessões de instigação social seguidas de encontros agressivos diretos. Além disso, o fator de liberação de corticotrofina (CRF) e o fator neurotrófico derivado do cérebro (BNDF) foram medidos no encéfalo desses animais. Camundongos controles não foram expostos à instigação social ou confrontos agressivos.
Resultados:
Os camundongos expostos a confrontos agressivos exibiram um padrão semelhante de comportamentos agressivos e não agressivos típicos da espécie na primeira e na última sessão. Em contraste com instigação social apenas, confrontos agressivos repetidos promoveram aumento na corticosterona plasmática. Após 10 sessões de confrontos agressivos, os camundongos apresentaram uma tendência não significativa de redução dos níveis de CRF no hipocampo, que se correlacionaram inversamente com os níveis plasmáticos de corticosterona. Por outro lado, sessões repetidas de instigação social ou confronto agressivo não alteraram as concentrações de BDNF no córtex pré-frontal e hipocampo.
Conclusão:
A exposição a episódios repetidos de encontros agressivos não promoveu habituação ao longo do tempo. Adicionalmente, o CRF parece estar envolvido nas respostas fisiológicas aos estressores sociais.
Descritores:
Agressão; instigação social; corticosterona; CRF; BDNF
Introduction
Social systems and social stressors differ between species but in general social animals form dominance-based hierarchies.11. Blanchard RJ, McKittrick CR, Blanchard DC. Animal models of social stress: effects on behavior and brain neurochemical systems. Physiol Behav. 2001;73:261-71. In this context, aggression is an adaptive behavioral trait important for establishing dominance as well as competition for mating partners, food, and territories.22. Koolhaas JM, Bohus B. Animal models of human aggression. In: Boulton AA, Baker GB, Martin-Iverson MT, editors. Animal models in psychiatry. II. New York: Humana Press; 1992. p. 249-71. While a certain level of aggression is supposed to be beneficial for survival, abnormal aggression can be detrimental. In fact, increase in aggressiveness is a common occurrence in a variety of psychiatric disorders.33. Réale,D, Martin J, Coltman DW, Poissant J, Festa-Bianchet M. Male personality, life-history strategies and reproductive success in a promiscuous mammal. J Evol Biol. 2009;22:1599-607.,44. Watters J, Sih A. The mix matters: behavioural types and group dynamics in water striders. Behaviour. 2005;142:1417-31.
Laboratory models of social stress include the visible burrow system, crowding stress, and the resident-intruder paradigm.55. Armario A, Castellanos JM, Balasch J. Effect of crowding on emotional reactivity in male rats. Neuroendocrinology. 1984;39:330-3.–77. Tornatzky W, Miczek KA. Long-term impairment of autonomic circadian rhythms after brief intermittent social stress. Physiol Behav. 1993;53:983-93. Social stress models are particularly useful because they are considered ethologically relevant.88. Miczek KA, Yap JJ, Covington HE 3rd. Social stress, therapeutics and drug abuse: preclinical models of escalated and depressed intake. Pharmacol Ther. 2008;120:102-28. Furthermore, most of the stress faced by humans occurs in a social context.99. Haller J, Harold G, Sandi C, Neumann ID. Effects of adverse early-life events on aggression and anti-social behaviours in animals and humans. J Neuroendocrinol. 2014;26:724-38. Although preclinical research has produced important descriptions of aggression and provided a solid basis for analysis of the neurobiology of aggressive behavior, the degree of similarity across species, neural systems, behavioral expression, and outcomes of aggression remain unclear.
Limbic regions such as the prefrontal cortex (PFC) and hippocampus are involved in innate social behaviors and response to social stress.1010. Lopez NL, Vazquez DM, Olson SL. An integrative approach to the neurophysiological substrates of social withdrawal and aggression. Dev Psychopathol. 2004;16:69-93. These areas seem to be critical for emotional and cognitive functions such as social recognition, fighting, mating, fear, or motivated behaviors.1010. Lopez NL, Vazquez DM, Olson SL. An integrative approach to the neurophysiological substrates of social withdrawal and aggression. Dev Psychopathol. 2004;16:69-93. The PFC and hippocampus have been identified as particularly important in the modulatory control of subcortical circuits that mediate aggressive and impulsive behaviors1111. Blair RJR. The roles of orbital frontal cortex in the modulation of antisocial behavior. Brain Cogn. 2004;55:198-208.,1212. de Almeida RMM, Ferrari PF, Parmigiani S, Miczek KA. Escalated aggressive behavior: dopamine, serotonin and GABA. Eur J Pharmacol. 2005;526:51-64.; the components of these circuits include the medial amygdala, hypothalamus and the periaqueductal grey.1313. Adamec RE, Stark-Adamec CI. Limbic control of aggression in the cat. Prog Neuro-Psychoph. 1983;7:505-12.–2222. Siegel A, Roeling TA, Gregg TR, Kruk MR. Neuropharmacology of brain-stimulation-evoked aggression. Neurosci Biobehav Rev. 1999;23:359-89. Indeed, PFC lesions promote an increase in aggressive behavior in rats.2323. de Bruin JP, van Oyen HG, Van de Poll N. Behavioural changes following lesions of the orbital prefrontal cortex in male rats. Behav Brain Res. 1983;10:209-32. Similarly, lesions involving frontal and temporal brain areas have been demonstrated to dramatically increase aggressiveness in humans.2424. Hawkins KA, Trobst, KK. Frontal lobe dysfunction and aggression: conceptual issues and research findings. Aggress Violent Beh. 2000;5:147-57.
It is already known that repeated episodes of social confrontation promote long-lasting neuroadaptation in rodents that are defeated in the resident-intruder paradigm.2525. Boyson CO, Holly EN, Shimamoto A, Albrechet-Souza L, Weiner LA, DeBold JF, Miczek KA. Social stress and CRF-dopamine interactions in the VTA: role in long-term escalation of cocaine self-administration. J Neurosci. 2014;34:6659-67. However, less is known about functional, as well as neuroadaptive, changes that occur in the brain of aggressive residents exposed to repeated episodes of social interactions. In this study, male resident Swiss mice were repeatedly exposed to either a potential rival nearby, but inaccessible (social instigation),1212. de Almeida RMM, Ferrari PF, Parmigiani S, Miczek KA. Escalated aggressive behavior: dopamine, serotonin and GABA. Eur J Pharmacol. 2005;526:51-64. or to 10 sessions of social instigation followed by direct aggressive encounters. Controls were exposed to neither social instigation nor aggressive confrontations. After the last session, we measured plasma corticosterone (CORT), the stress neuropeptide corticotropin-releasing factor (CRF) and brain-derived neurotrophic factor (BDNF) in limbic brain areas. CRF was measured in the hippocampus and hypothalamus, areas previously related to aggression and behavioral and physiological responses to stress.2626. Smagin DA, Park JH, Michurina TV, Peunova N, Glass Z, Sayed K, Bondar NP, Kovalenko IN, Kudryavtseva NN, Enikolopov G. Altered hippocampal neurogenesis and amygdalar neuronal activity in adult mice with repeated experience of aggression. Front Neurosci. 2015;9:443.,2727. Vale W, Spiess J, Rivier C, Rivier J. Characterization of a 41-residue ovine hypothalamic peptide that stimulates secretion of corticotropin and beta-endorphin. Science. 1981;213:1394-7. BDNF was measured in the PFC and hippocampus, brain regions associated with behavioral planning and affective behaviors.2828. Ninan I. Synaptic regulation of affective behaviors; role of BDNF. Neuropharmacology. 2014;76:684-95.
The primary role of CRF is to activate the hypothalamic-pituitary-adrenal (HPA) axis by acting on receptors in the pituitary and promoting the release of adrenocorticotropic hormone (ACTH) into the portal blood system.2727. Vale W, Spiess J, Rivier C, Rivier J. Characterization of a 41-residue ovine hypothalamic peptide that stimulates secretion of corticotropin and beta-endorphin. Science. 1981;213:1394-7. ACTH stimulates the release of CORT from the adrenal glands – CORT plays several roles in mediating appropriate responses to stress and also exerts a negative feedback control of the HPA axis.2929. Bale TL, Vale WW. CRF and CRF receptors: role in stress responsivity and other behaviors. Annu Rev Pharmacol Toxicol. 2004;44:525-57. Extrahypothalamic distribution of CRF includes neuronal populations in the amygdala,3030. Koob GF. A role for brain stress systems in addiction. Neuron. 2008;59:11-34.,3131. Swanson LW, Sawchenko PE, Rivier J, Vale WW. Organization of ovine corticotropin-releasing factor immunoreactive cells and fibers in the rat brain: an immunohistochemical study. Neuroendocrinology. 1983;36:165-86. hippocampus,3232. Chen Y, Bender RA, Frotscher M, Baram TZ. Novel and transient populations of corticotropin-releasing hormone-expressing neurons in developing hippocampus suggest unique functional roles: a quantitative spatiotemporal analysis. J Neurosci. 2001;21:7171-81. and locus coeruleus.3333. Valentino RJ, Van Bockstaele E. Convergent regulation of locus coeruleus activity as an adaptive response to stress. Eur J Pharmacol. 2008;583:194-203. CRF and its related peptides exert central function and mediate several behavioral and physiological responses to stress,3434. Britton KT, Lee G, Vale W, Rivier J, Koob GF. Corticotropin releasing factor (CRF) receptor antagonist blocks activating and ‘anxiogenic’ actions of CRF in the rat. Brain Res. 1986;369:303-6.–3838. Sahuque LL, Kullberg EF, McGeehan AJ, Kinder JR, Hicks MP, Blanton MG, Janak PH, Olive MF. Anxiogenic and aversive effects of corticotropin-releasing factor (CRF) in the bed nucleus of the stria terminalis in the rat: role of CRF receptor subtypes. Psychopharmacology (Berl). 2006;186:122-32. including anxiety-like behavior and some aspects of aggressiveness.3535. Coste SC, Heard AD, Phillips TJ, Stenzel-Poore MP. Corticotropin-releasing factor receptor type 2-deficient mice display impaired coping behaviors during stress. Genes Brain Behav. 2006;5:131-8.,3939. Backström T, Winberg S. Central corticotropin releasing factor and social stress. Front Neurosci. 2013;7:117.–4343. Holly EN, Boyson CO, Montagud-Romero S, Stein DJ, Gobrogge KL, DeBold JF, et al. Episodic social stress-escalated cocaine self-administration: role of phasic and tonic corticotropin releasing factor in the anterior and posterior ventral tegmental area. Journal of Neuroscience. 2016;36:4093-105.
The present study tested the hypothesis that BDNF may underlie, at least in part, experience-induced neuroplasticity in resident mice exposed to repeated sessions of agonistic interactions. BDNF is a molecule involved in the regulation of diverse biological functions, ranging from neuronal survival and differentiation during development to synaptic plasticity and cognitive behavior in the adult4444. Maynard KR, Hill JL, Calcaterra NE, Palko ME, Kardian A, Paredes D, et al. Functional role of BDNF production from unique promoters in aggression and serotonin signaling. Neuropsychopharmacology. 2016;41:1943-55.; it has also been demonstrated to be a critical mediator of changes in social motivation.4545. Berton O, McClung CA, Dileone RJ, Krishnan V, Renthal W, Russo SJ, Graham D, Tsankova NM, Bolanos CA, Rios M, Monteggia LM, Self DW, Nestler EJ. Essential role of BDNF in the mesolimbic dopamine pathway in social defeat stress. Science. 2006;311:864-8. In both rodents and humans, BDNF disruption is associated with neurobehavioral alterations and psychiatric disorders.4444. Maynard KR, Hill JL, Calcaterra NE, Palko ME, Kardian A, Paredes D, et al. Functional role of BDNF production from unique promoters in aggression and serotonin signaling. Neuropsychopharmacology. 2016;41:1943-55.
Methods
Subjects
Adult Swiss mice weighed 25-30 g (8 weeks old) upon arrival were housed in polycarbonate cages (30 × 19 × 15 cm) with pine shavings as bedding. Rodent laboratory chow and water were available ad libitum through stainless steel wire mesh lids. Male mice were assigned as residents (n = 24) or intruders (n = 20). Each resident mouse was pair-housed with a female Swiss mouse (n = 24), whereas intruders were kept in groups of 6 per cage. The vivarium of the Animal Experimentation Unit at Hospital de Clínicas de Porto Alegre (Porto Alegre, RS, Brazil) was maintained on a 12h light/dark cycle, 22±2°C temperature, and 50-60% humidity. The experiments were performed during the light phase, between 09:00 and 12:00 a.m. Experimental procedures were conducted in accordance with Brazilian Federal Law no.11.794/2008, which regulates the scientific use of animals. The project was approved by the Ethics Committee on Animal Use of the Animal Experimentation Unit at Hospital de Clínicas de Porto Alegre.
Tubal-ligation surgery
Female mice were tubally ligated using antiseptic techniques and standard surgical procedure.4646. Remie R. Experimental surgery. In: Krinke GJ, editor. The laboratory rat. London: Academic Press; 2000. p. 523-68. Briefly, mice were anesthetized with ketamine (120 mg/kg) + xylazine (30 mg/kg, intraperitoneally [i.p.]) and placed in the right lateral decubitus position. Then, a dorsal incision (approximately 1.0 cm) was made, the ovary was located, and the ends of the uterine horn were tied off using absorbable sutures. The oviduct was located and severed using a micro-scissor. All reproductive structures were repositioned back in the abdominal cavity, and the abdominal incision was closed with absorbable sutures and the skin with non-absorbable sutures.4747. Harris BN, Saltzman W. Effect of reproductive status on hypothalamic-pituitary-adrenal (HPA) activity and reactivity in male Californiamice (Peromyscus californicus). Physiol Behav. 2013;112−113:70−6. The same procedure was performed on the left side. Mice were injected with tramadol (10 mg/kg, i.p.) immediately after the surgery and during the next 3 consecutive days (12/12h) to provide analgesia. Female mice were single-housed and allowed to recover for 7 days before being paired with a resident male. Upon termination of the experiment, females were euthanized with an overdose of ketamine (300 mg/kg) + xylazine (30 mg/kg, i.p.).
Experimental design
The experimental design is shown in Figure 1. Male resident mice were tested after being pair-housed with a female for 3 weeks. Before the sessions, the female cage mate was removed from the resident's cage and kept in a holding cage. Resident mice were divided into the following experimental groups: 1) controls (CT): an empty perforated acrylic tube (18×6 cm) was placed into the resident's cage for 5 min; 2) social instigation group (SI): a perforated acrylic tube (18 ×6 cm) containing an intruder mouse was placed into the resident's cage for 5 min. Mice had visual, auditory, and olfactory contact, but the resident had no direct access to the intruder; 3) social instigation + aggressive confrontation group (SI+AC): a perforated acrylic tube (18 ×6 cm) containing an intruder mouse was placed into the resident's cage for 5 min, followed by actual confrontation without any protection. The aggressive encounter was terminated 5 min after the resident initiated the first attack bite or at 5 min if the resident failed to attack.4848. Miczek KA, O’Donnell JM. Alcohol and chlordiazepoxide increase suppressed aggression in mice. Psychopharmacology (Berl). 1980;69:39-44. Residents were exposed to 10 sessions of social instigation or social instigation + aggressive confrontation, twice in a week, with a minimum interval of 72 h between sessions. Mice that did not present bite attacks against the intruder in the first two sessions were excluded from the experiment.
Experimental design. Aggressive and non-aggressive behaviors were assessed during the first (T1) and last (T10) resident-intruder encounters. Blood samples were collected 5 days before the first resident-intruder encounter (baseline) and immediately after T10 for plasma corticosterone analysis. After the last blood collection, brains were extracted for CRF and BDNF measurements.
Aggressive and non-aggressive behaviors exhibited by resident mice during the first (T1) and last (T10) agonistic encounters
T1 and T10 were video-recorded and later coded by two independent researchers using the Observer XT software (Noldus, v.9.0.436, Wageningen, The Netherlands). The measurements assigned by the two observers agreed with each other (r22. Koolhaas JM, Bohus B. Animal models of human aggression. In: Boulton AA, Baker GB, Martin-Iverson MT, editors. Animal models in psychiatry. II. New York: Humana Press; 1992. p. 249-71. = 0.94). The frequency of aggressive behaviors was measured, including attack bites, chasing, lateral threats, sniffing, and tail rattling, as well as the latency of the first attack bite. The frequency or duration of non-aggressive behaviors, including grooming, rearing, and walking, was also evaluated.
Plasma CORT analysis before the resident-intruder encounters (baseline) and after T10
Resident mice had blood samples collected from the submandibular vein 5 days before the resident-intruder encounters (baseline) and immediately after T10, using disposable sterile lancets. Samples were centrifuged for 10 min at 4°C and 4,000 revolutions/min. Blood plasma was extracted and frozen at −80ºC for subsequent dosages. Plasma CORT was assessed using commercial ELISA kits (Enzo Life Science, Farmingdale, NY, USA). Detection levels were 32-20 pg/ml, according to the manufacturer.
Brain CRF and BDNF measured after T10
After the last blood collection, resident mice were rapidly anesthetized with isoflurane and euthanized by decapitation. Brains were quickly removed, thoroughly washed in isotonic saline solution, and dissected on ice. The PFC, hippocampus, and hypothalamus were localized according to a brain atlas4949. Paxinos G, Franklin K. The mouse brain in stereotaxic coordinates. 2nd ed. San Diego: Elsevier Academic; 2001. and removed. Brain tissue samples were homogenized (weight/volume, 1:10) with ice-cold 0.1 M phosphate buffer (pH 7.4), with the addition of protease inhibitor cocktail (Sigma-Aldrich, St. Louis, MO, USA). Homogenates were centrifuged at 2,000 g for 5 min, and aliquots of supernatants were separated and stored at −80°C until further analyses. CRF in the hippocampus and hypothalamus were determined using a CRF mouse ELISA assay kit (My ELISA kits, St. Petersburg, FL, USA). BDNF in the PFC and hippocampus were measure by sandwich-ELISA, according to the manufacturer's instructions using specific monoclonal antibodies (R&D Systems Inc., Minneapolis, MN, USA).
Statistical analyses
Statistical analyses were performed using STATISTICA version 6.0. The data are reported as mean ± standard error of mean. Frequency and duration of aggressive and non-aggressive behaviors on T1 and T10 were analyzed with the Student t-test for dependent samples. Plasma CORT levels were analyzed using two-way analysis of variance (ANOVA) with repeated measures followed by post-hoc comparisons using the Newman-Keuls multiple-range test. CRF and BDNF concentrations were analyzed with one-way ANOVA. A linear least-square regression was conducted to determine the relationship between plasma CORT (ng/ml) and hippocampal CRF (pg/ml). Statistical significance was set at p = 0.05. For the behavioral experiments, each group contained eight animals, and six-seven were randomly used for hormonal and neurochemical measurements.
Results
The frequency or duration of aggressive and non-aggressive behaviors exhibited by male resident mice during T1 and T10 is shown in Table 1. The Student t-test for dependent samples revealed a significant decrease in the frequency of bites during T10 compared to T1 (t= 3.11, p < 0.05). No significant differences were observed between T1 and T10 in any other behavioral category evaluated (values of tvarying from 0.05 to 2.21; p > 0.05 in all cases).
Aggressive and non-aggressive behaviors exhibited by male resident mice during the first (T1) and the last (T10) social instigation + aggressive confrontation session.
Plasma CORT measurements obtained at baseline and after T10 are shown in Figure 2. Two-way ANOVA with repeated measures revealed significant differences between groups (F2,17 = 4.55, p < 0.05), sessions (F1,17 = 35.85, p < 0.0001), and the interaction between these factors (F2,17 = 4.36, p < 0.05). Post-hoc analyses indicated a significant increase in plasma CORT in mice exposed to aggressive confrontations (SI+AC), compared to baseline levels, control animals, and mice exposed to social instigation only (SI).
Plasma corticosterone levels (ng/ml) measured in controls (CT), mice exposed to social instigation only (SI), or social instigation + aggressive confrontations (SI+AC). Blood samples were collected before the resident-intruder encounters (baseline) and after the last session (T10). Data presented as mean ± standard error of mean. * Compared to baseline, CT and SI in the same session. p < 0.05, n = 6-7 mice per group.
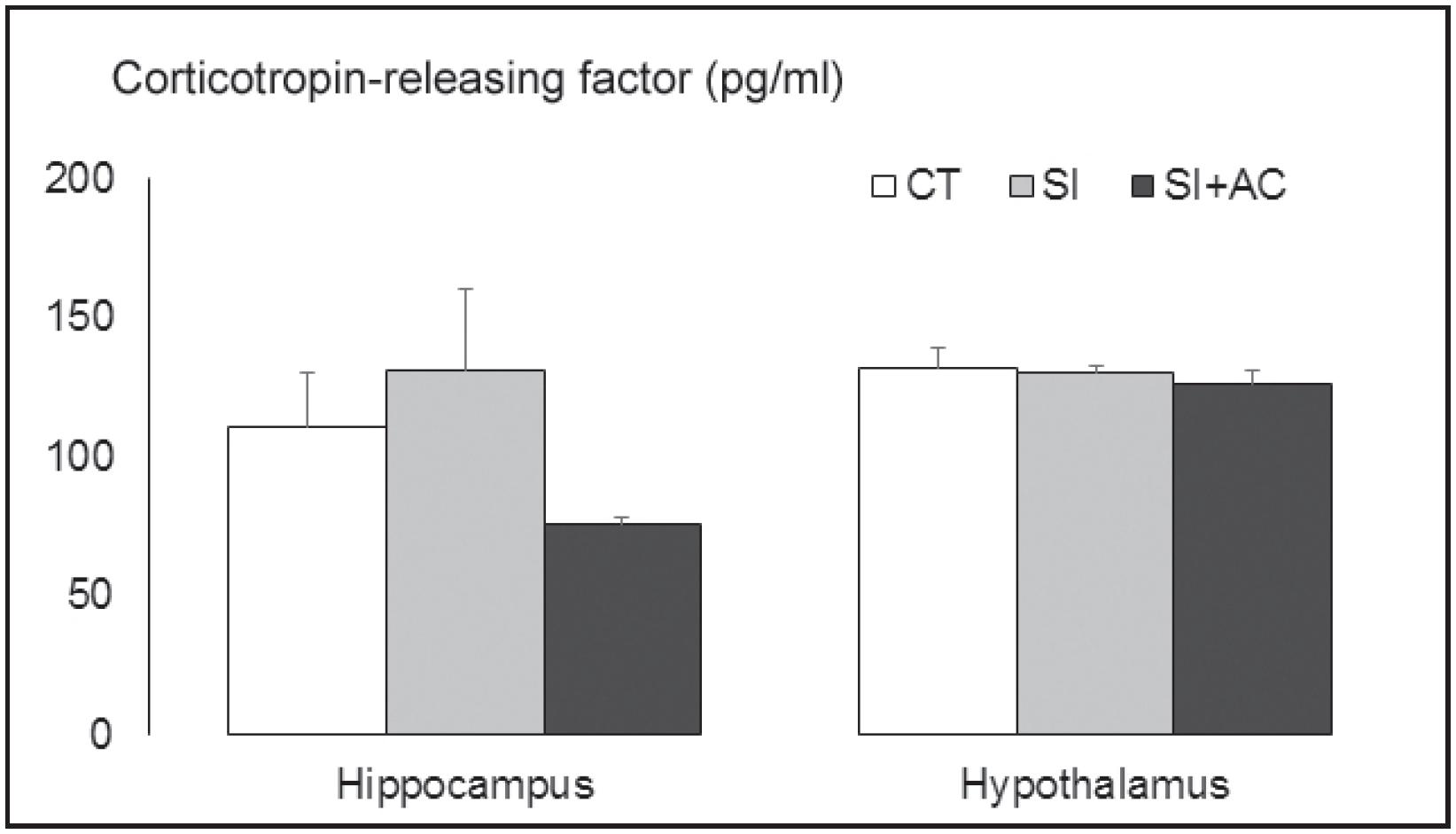
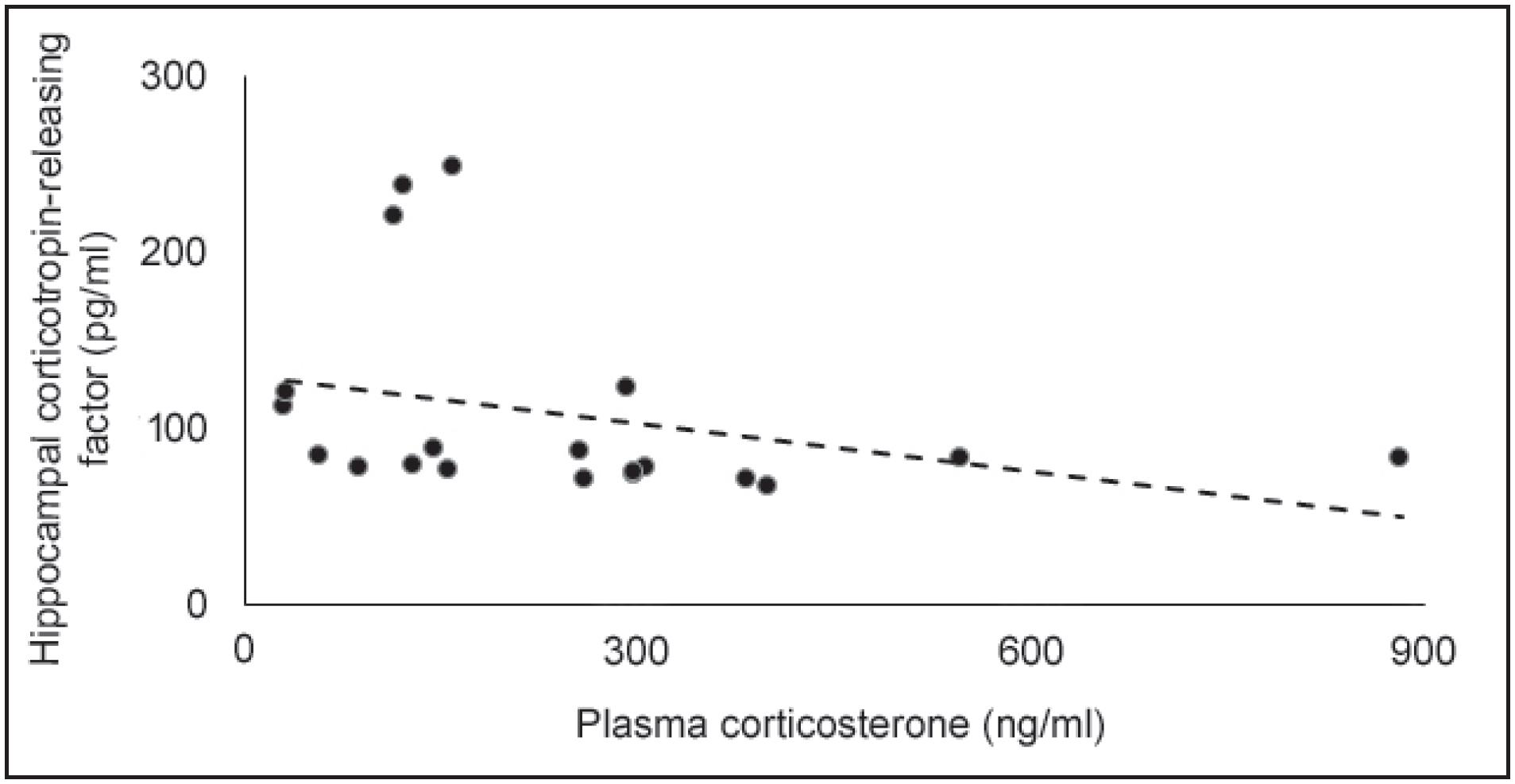
CRF levels in the hippocampus and hypothalamus after T10 are presented in Figure 3. One-way ANOVA revealed no significant differences between the groups in both areas (F2,17 = 1.66 and 0.37, respectively; p > 0.05 in both cases). However, in the hippocampus, the SI+AC group presented a decrease of 32% in CRF levels compared to control mice, and 42.5% compared to SI. Also, there was a significant negative linear relationship between plasma CORT and hippocampal CRF levels (r22. Koolhaas JM, Bohus B. Animal models of human aggression. In: Boulton AA, Baker GB, Martin-Iverson MT, editors. Animal models in psychiatry. II. New York: Humana Press; 1992. p. 249-71.= −0.52, p < 0.05, Figure 4).
Corticotropin-releasing factor levels (pg/ml) measured in the hippocampus and hypothalamus of controls (CT), mice exposed to social instigation only (SI), or social instigation + aggressive confrontations (SI+AC). Data presented as mean ± standard error of mean, n = 6-7 mice per group.
Plasma corticosterone (ng/ml) plotted as a function of hippocampal corticotropin-releasing factor (pg/ml); the dotted line represents the linear fit (r22. Koolhaas JM, Bohus B. Animal models of human aggression. In: Boulton AA, Baker GB, Martin-Iverson MT, editors. Animal models in psychiatry. II. New York: Humana Press; 1992. p. 249-71. = −0.52). p > 0.05, n = 6-7 mice per group.
BDNF levels in the PFC and hippocampus after T10 are presented in Figure 5. One-way ANOVA revealed no significant differences between the groups in both areas (F2,17 = 1.07 and 1.23, respectively; p > 0.05 in both cases).
Brain-derived neurotrophic factor levels (pg/ml) measured on the prefrontal cortex and hippocampus of controls (CT), mice exposed to social instigation only (SI), or social instigation + aggressive confrontations (SI+AC). Data presented as mean ± standard error of mean, n = 6-7 mice per group.
Discussion
In this study, male Swiss mice exposed to repeated aggressive inter-male confrontations exhibited a similar pattern of species-typical aggressive and non-aggressive behavior on the first and last sessions. Moreover, the current procedures showed that direct confrontations engendered an activation of the HPA axis during T10, suggesting that exposure to repeated episodes of aggressive encounters does not promote habituation over time. Additionally, after the last aggressive confrontation, mice presented a non-significant trend toward reducing hippocampal levels of CRF, which was negatively correlated with plasma CORT.
The activation of the HPA axis found in the present study is not a surprise, since aggressive encounters involve considerable risks for both resident and intruder. Either of the fighting parties can suffer injuries, and both loose energy reserves that may become crucial in a subsequent challenge.5050. Haller J. Related Biochemical background for an analysis of cost-benefit interrelations in aggression. Neurosci Biobehav Rev. 1995;19:599-604. In agreement with our neuroendocrinal results, male mice exposed to a paradigm of repeated experience of winning in a social conflict have been demonstrated to present increased levels of anxiety in the elevated plus-maze test.2626. Smagin DA, Park JH, Michurina TV, Peunova N, Glass Z, Sayed K, Bondar NP, Kovalenko IN, Kudryavtseva NN, Enikolopov G. Altered hippocampal neurogenesis and amygdalar neuronal activity in adult mice with repeated experience of aggression. Front Neurosci. 2015;9:443.
Increases in plasma glucocorticoids during a confrontation have been suggested to facilitate behaviors that are predominant for the animal in that specific context.5151. Haller J, Millar S, Kruk MR. Mineralocorticoid receptor blockade inhibits aggressive behaviour in male rats. Stress. 1998;2:2017. Indeed, brain mineralocorticoid receptor blockade during the first aggressive encounter inhibits subsequent propensity for violence in rats.5252. Kruk MR, Haller J, Meelis W, de Kloet ER. Mineralocorticoid receptor blockade during a rat's first violent encounter inhibits its subsequent propensity for violence. Behav Neurosci. 2013;127:505-14. Thus, both offensive and defensive forms of aggression might respond to reductions in the glucocorticoid release normally associated with the stress of either challenge or conspecific attack, raising the possibility that treatments reducing the systemic activation of the HPA axis may affect aggressiveness.
Repeated sessions of social instigation, which is assumed to enhance the “aggressive arousal” of the resident male,5353. Fish EW, Faccidomo S, Miczek KA. Aggression heightened by alcohol or social instigation in mice: reduction by the 5-HT(1B) receptor agonist CP-94,253. Psychopharmacology (Berl). 1999;146: 391-9. did not induce significant increases in CORT levels, suggesting that the mobilization of the HPA axis depends on direct confrontations, at least in the protocol used here. Importantly, the basal concentration of plasma CORT in this study (mean = 31.3 ng/mL) is consistent with others.5454. Finn DA, Sinnott RS, Ford MM, Long SL, Tanchuck MA, Phillips TJ. Sex differences in the effect of ethanol injection and consumption on brain allopregnanolone levels in C57BL/6 mice. Neuroscience. 2004;123:813-9.
In a study using male rats defeated in the resident-intruder paradigm and measuring several neuropeptides in the brain, the authors found a decrease in CRF levels only in the hippocampus, suggesting a depressive-like state in submissive animals.5555. Panksepp J, Burgdorf J, Beinfeld MC, Kroes RA, Moskal JR. Brain regional neuropeptide changes resulting from social defeat. Behav Neurosci. 2007;121:1364-71. In the present study, even though aggressive confrontations did not promote a significant main effect on brain CRF, resident mice showed a trend toward reducing this neuropeptide in the hippocampus when compared to controls and mice exposed to social instigation only (reduction of 35.5% and 45.5%, respectively). Thus, it is possible that CRF in the hippocampus may be more involved in emotional and cognitive functions rather than in submissive or aggressive behavior. Interestingly, we found a negative correlation between hippocampal CRF and plasma CORT, suggesting that CRF may be involved in physiological responses to social stressors. Further investigation, however, is required before a mechanistic explanation of hippocampal CRF and plasma CORT can be proposed.
The hypothalamus, more specifically its paraventricular nucleus, is the origin of the HPA axis, whose activation culminates with the release of glucocorticoids from the adrenal glands.2727. Vale W, Spiess J, Rivier C, Rivier J. Characterization of a 41-residue ovine hypothalamic peptide that stimulates secretion of corticotropin and beta-endorphin. Science. 1981;213:1394-7. Interestingly, in our experiment, after the last aggressive confrontation, mice presented an increase of plasma CORT without changing hypothalamic levels of CRF. A possible explanation for this apparent discrepancy could be that CRF and CORT present distinct temporal profiles of release. Thus, we may have collected the blood samples and extracted the brains at a time point when hypothalamic CRF had already stimulated ACTH release, but returned to basal levels. The stress response has classically been characterized by two temporal “waves” of stress mediator actions. The first one includes rapid actions of noradrenaline, serotonin, dopamine and CRF, promoting vigilance, alertness, appraisal of the situation and the choice of an optimal strategy to face the challenge. These events are followed by alterations of gene expression and cell function promoting sustained and adaptive stress responses attributed to glucocorticoids.5656. Joëls M, Baram TZ. The neuro-symphony of stress. Nat Rev Neurosci. 2009;10:459-66.
Finally, increase in hippocampal levels of BDNF in resident hamsters has been suggested to evidence that behaviors associated with aggression and with winning a fight involve plastic mechanisms important to encode spatial representations.5757. Taylor SL, Stanek LM, Ressler KJ, Huhman KL. Differential brain-derived neurotrophic factor expression in limbic brain regions following social defeat or territorial aggression. Behav Neurosci. 2011;125:911-20. Differently from our study, however, those animals were submitted to a single social interaction session. Thus, changes in BDNF levels may occur after the initial agonistic encounters, which may help to explain the lack of changes in BDNF in the present study. Moreover, aggression-induced cortical activation seems to be especially strong in mice selected for high aggressive behavior.1616. Halász J, Tóth M, Kalló I, Liposits Z, Haller J. The activation of prefrontal cortical neurons in aggression—a double labeling study. Behav Brain Res. 2006;175:166-75.,5858. Haller J, Kruk MR. Normal and abnormal aggression: human disorders and novel laboratory models. Neurosci Biobehav Rev. 2006;30:292-303. A limitation of this study is that we measured brain BDNF only after 10 resident-intruder sessions (social instigation only or social instigation + aggressive confrontation), when neuronal adaptations may be already stablished.
Conclusion
The present results extend our current knowledge about the neurobiology of aggressiveness by evaluating neurobiological mechanisms of species-typical aggressive behavior in male Swiss mice exposed to repeated sessions Trends Psychiatry Psychother. 2017;00(0) – 5 of agonistic encounters. The behavioral repertoire and the increase in plasma CORT after 10 aggressive confrontations indicate that resident mice do not present habituation over time. These findings support the idea that treatments reducing the systemic activation of the HPA axis may affect aggressiveness.5151. Haller J, Millar S, Kruk MR. Mineralocorticoid receptor blockade inhibits aggressive behaviour in male rats. Stress. 1998;2:2017. Contrary to our expectation, resident mice presented a non-statistically significant but substantial decrease of hippocampal CRF after the last confrontation, which correlated negatively with plasma CORT. Understanding the neuroadaptations that occur after successive episodes of social conflict may provide valuable insights into normal and abnormal forms of aggression and, ultimately, lead to effective approaches to control inappropriate aggressiveness in humans and other animals.
Acknowledgements
This research was supported by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq). Lucas Albrechet-Souza was supported by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES; CSF-PAJT 88887.096822/2015-00). The authors would like to thank Dr. Flávio Kapczinski, Fabiola Meyer, Tuane Garcez and Marta Cioato for providing helpful assistance during the development of this study.
References
-
1Blanchard RJ, McKittrick CR, Blanchard DC. Animal models of social stress: effects on behavior and brain neurochemical systems. Physiol Behav. 2001;73:261-71.
-
2Koolhaas JM, Bohus B. Animal models of human aggression. In: Boulton AA, Baker GB, Martin-Iverson MT, editors. Animal models in psychiatry. II. New York: Humana Press; 1992. p. 249-71.
-
3Réale,D, Martin J, Coltman DW, Poissant J, Festa-Bianchet M. Male personality, life-history strategies and reproductive success in a promiscuous mammal. J Evol Biol. 2009;22:1599-607.
-
4Watters J, Sih A. The mix matters: behavioural types and group dynamics in water striders. Behaviour. 2005;142:1417-31.
-
5Armario A, Castellanos JM, Balasch J. Effect of crowding on emotional reactivity in male rats. Neuroendocrinology. 1984;39:330-3.
-
6Blanchard RJ, Blanchard DC. Antipredator defensive behaviors in a visible burrow system. J Comp Psychol. 1989;103:70-82.
-
7Tornatzky W, Miczek KA. Long-term impairment of autonomic circadian rhythms after brief intermittent social stress. Physiol Behav. 1993;53:983-93.
-
8Miczek KA, Yap JJ, Covington HE 3rd. Social stress, therapeutics and drug abuse: preclinical models of escalated and depressed intake. Pharmacol Ther. 2008;120:102-28.
-
9Haller J, Harold G, Sandi C, Neumann ID. Effects of adverse early-life events on aggression and anti-social behaviours in animals and humans. J Neuroendocrinol. 2014;26:724-38.
-
10Lopez NL, Vazquez DM, Olson SL. An integrative approach to the neurophysiological substrates of social withdrawal and aggression. Dev Psychopathol. 2004;16:69-93.
-
11Blair RJR. The roles of orbital frontal cortex in the modulation of antisocial behavior. Brain Cogn. 2004;55:198-208.
-
12de Almeida RMM, Ferrari PF, Parmigiani S, Miczek KA. Escalated aggressive behavior: dopamine, serotonin and GABA. Eur J Pharmacol. 2005;526:51-64.
-
13Adamec RE, Stark-Adamec CI. Limbic control of aggression in the cat. Prog Neuro-Psychoph. 1983;7:505-12.
-
14Adams DB, Boudreau W, Cowan CW, Kokonowski C, Oberteuffer K, Yohay K. Offense produced by chemical stimulation of the anterior hypothalamus of the rat. Physiol Behav. 1993;53:1127 32.
-
15Delville Y, De Vries GJ, Ferris CF. Neural connections of the anterior hypothalamus and agonistic behavior in golden hamsters. Brain Behav Evol. 2000;55:53-76.
-
16Halász J, Tóth M, Kalló I, Liposits Z, Haller J. The activation of prefrontal cortical neurons in aggression—a double labeling study. Behav Brain Res. 2006;175:166-75.
-
17Joppa MA, Meisel RL, Garber MA. -Fos expression in female hamster brain following sexual and aggressive behaviors. Neuroscience. 1995;68:783-92.
-
18Kollack-Walker S, Watson SJ, Akil H. Social stress in hamsters: defeat activates specific neurocircuits within the brain. J Neurosci. 1997;17:8842-55.
-
19Kruk MR. Ethology and pharmacology of hypothalamic aggression in the rat. Neurosci Biobehav Rev. 1991;15:527-38.
-
20Luiten PG, Koolhaas JM, de Boer S, Koopmans SJ. The corticomedial amygdala in the central nervous system organization of agonistic behavior. Brain Res. 1985;332:283-97.
-
21Martinez M, Phillips PJ, Herbert J. Adaptation in patterns of c-fos expression in the brain associated with exposure to either single or repeated social stress in male rats. Eur J Neurosci. 1998;10:20 33.
-
22Siegel A, Roeling TA, Gregg TR, Kruk MR. Neuropharmacology of brain-stimulation-evoked aggression. Neurosci Biobehav Rev. 1999;23:359-89.
-
23de Bruin JP, van Oyen HG, Van de Poll N. Behavioural changes following lesions of the orbital prefrontal cortex in male rats. Behav Brain Res. 1983;10:209-32.
-
24Hawkins KA, Trobst, KK. Frontal lobe dysfunction and aggression: conceptual issues and research findings. Aggress Violent Beh. 2000;5:147-57.
-
25Boyson CO, Holly EN, Shimamoto A, Albrechet-Souza L, Weiner LA, DeBold JF, Miczek KA. Social stress and CRF-dopamine interactions in the VTA: role in long-term escalation of cocaine self-administration. J Neurosci. 2014;34:6659-67.
-
26Smagin DA, Park JH, Michurina TV, Peunova N, Glass Z, Sayed K, Bondar NP, Kovalenko IN, Kudryavtseva NN, Enikolopov G. Altered hippocampal neurogenesis and amygdalar neuronal activity in adult mice with repeated experience of aggression. Front Neurosci. 2015;9:443.
-
27Vale W, Spiess J, Rivier C, Rivier J. Characterization of a 41-residue ovine hypothalamic peptide that stimulates secretion of corticotropin and beta-endorphin. Science. 1981;213:1394-7.
-
28Ninan I. Synaptic regulation of affective behaviors; role of BDNF. Neuropharmacology. 2014;76:684-95.
-
29Bale TL, Vale WW. CRF and CRF receptors: role in stress responsivity and other behaviors. Annu Rev Pharmacol Toxicol. 2004;44:525-57.
-
30Koob GF. A role for brain stress systems in addiction. Neuron. 2008;59:11-34.
-
31Swanson LW, Sawchenko PE, Rivier J, Vale WW. Organization of ovine corticotropin-releasing factor immunoreactive cells and fibers in the rat brain: an immunohistochemical study. Neuroendocrinology. 1983;36:165-86.
-
32Chen Y, Bender RA, Frotscher M, Baram TZ. Novel and transient populations of corticotropin-releasing hormone-expressing neurons in developing hippocampus suggest unique functional roles: a quantitative spatiotemporal analysis. J Neurosci. 2001;21:7171-81.
-
33Valentino RJ, Van Bockstaele E. Convergent regulation of locus coeruleus activity as an adaptive response to stress. Eur J Pharmacol. 2008;583:194-203.
-
34Britton KT, Lee G, Vale W, Rivier J, Koob GF. Corticotropin releasing factor (CRF) receptor antagonist blocks activating and ‘anxiogenic’ actions of CRF in the rat. Brain Res. 1986;369:303-6.
-
35Coste SC, Heard AD, Phillips TJ, Stenzel-Poore MP. Corticotropin-releasing factor receptor type 2-deficient mice display impaired coping behaviors during stress. Genes Brain Behav. 2006;5:131-8.
-
36Klampfl SM, Neumann ID, Bosch OJ. Reduced brain corticotropin-releasing factor receptor activation is required for adequate maternal care and maternal aggression in lactating rats. Eur J Neurosci. 2013;38:2742-50.
-
37Klampfl SM, Brunton PJ, Bayerl DS, Bosch OJ. Hypoactivation of CRF receptors, predominantly type 2, in the medial-posterior BNST is vital for adequate maternal behavior in lactating rats. J Neurosci. 2014;34:9665-76.
-
38Sahuque LL, Kullberg EF, McGeehan AJ, Kinder JR, Hicks MP, Blanton MG, Janak PH, Olive MF. Anxiogenic and aversive effects of corticotropin-releasing factor (CRF) in the bed nucleus of the stria terminalis in the rat: role of CRF receptor subtypes. Psychopharmacology (Berl). 2006;186:122-32.
-
39Backström T, Winberg S. Central corticotropin releasing factor and social stress. Front Neurosci. 2013;7:117.
-
40Farrokhi C, Blanchard DC, Griebel, G, Yang M, Gonzales C, Markham C, Blanchard RJ. Effects of the CRF1 antagonist SSR125543A on aggressive behaviors in hamsters. Pharmacol Biochem Behav. 2004;77:465-9.
-
41Gammie SC, Hasen NS, Stevenson SA, Bale TL, D’Anna KL. Elevated stress sensitivity in corticotropin-releasing factor receptor 2 deficient mice decreases maternal, but not intermale aggression. Behav Brain Res. 2005;160:169-77.
-
42Mele A, Cabib S, Oliverio A, Melchiorri P, Puglisi-Allegra S. Effects of corticotropin releasing factor and sauvagine on social behavior of isolated mice. Peptides. 1987;8:935-8.
-
43Holly EN, Boyson CO, Montagud-Romero S, Stein DJ, Gobrogge KL, DeBold JF, et al. Episodic social stress-escalated cocaine self-administration: role of phasic and tonic corticotropin releasing factor in the anterior and posterior ventral tegmental area. Journal of Neuroscience. 2016;36:4093-105.
-
44Maynard KR, Hill JL, Calcaterra NE, Palko ME, Kardian A, Paredes D, et al. Functional role of BDNF production from unique promoters in aggression and serotonin signaling. Neuropsychopharmacology. 2016;41:1943-55.
-
45Berton O, McClung CA, Dileone RJ, Krishnan V, Renthal W, Russo SJ, Graham D, Tsankova NM, Bolanos CA, Rios M, Monteggia LM, Self DW, Nestler EJ. Essential role of BDNF in the mesolimbic dopamine pathway in social defeat stress. Science. 2006;311:864-8.
-
46Remie R. Experimental surgery. In: Krinke GJ, editor. The laboratory rat. London: Academic Press; 2000. p. 523-68.
-
47Harris BN, Saltzman W. Effect of reproductive status on hypothalamic-pituitary-adrenal (HPA) activity and reactivity in male Californiamice (Peromyscus californicus). Physiol Behav. 2013;112−113:70−6.
-
48Miczek KA, O’Donnell JM. Alcohol and chlordiazepoxide increase suppressed aggression in mice. Psychopharmacology (Berl). 1980;69:39-44.
-
49Paxinos G, Franklin K. The mouse brain in stereotaxic coordinates. 2nd ed. San Diego: Elsevier Academic; 2001.
-
50Haller J. Related Biochemical background for an analysis of cost-benefit interrelations in aggression. Neurosci Biobehav Rev. 1995;19:599-604.
-
51Haller J, Millar S, Kruk MR. Mineralocorticoid receptor blockade inhibits aggressive behaviour in male rats. Stress. 1998;2:2017.
-
52Kruk MR, Haller J, Meelis W, de Kloet ER. Mineralocorticoid receptor blockade during a rat's first violent encounter inhibits its subsequent propensity for violence. Behav Neurosci. 2013;127:505-14.
-
53Fish EW, Faccidomo S, Miczek KA. Aggression heightened by alcohol or social instigation in mice: reduction by the 5-HT(1B) receptor agonist CP-94,253. Psychopharmacology (Berl). 1999;146: 391-9.
-
54Finn DA, Sinnott RS, Ford MM, Long SL, Tanchuck MA, Phillips TJ. Sex differences in the effect of ethanol injection and consumption on brain allopregnanolone levels in C57BL/6 mice. Neuroscience. 2004;123:813-9.
-
55Panksepp J, Burgdorf J, Beinfeld MC, Kroes RA, Moskal JR. Brain regional neuropeptide changes resulting from social defeat. Behav Neurosci. 2007;121:1364-71.
-
56Joëls M, Baram TZ. The neuro-symphony of stress. Nat Rev Neurosci. 2009;10:459-66.
-
57Taylor SL, Stanek LM, Ressler KJ, Huhman KL. Differential brain-derived neurotrophic factor expression in limbic brain regions following social defeat or territorial aggression. Behav Neurosci. 2011;125:911-20.
-
58Haller J, Kruk MR. Normal and abnormal aggression: human disorders and novel laboratory models. Neurosci Biobehav Rev. 2006;30:292-303.
Publication Dates
-
Publication in this collection
12 June 2017 -
Date of issue
Apr-Jun 2017
History
-
Received
24 Oct 2016 -
Accepted
15 Feb 2017