ABSTRACT
Objective
Paraoxonase 1 (PON1) polymorphisms are associated with an increased susceptibility to cardiovascular disease. PON1 Q192R polymorphism (rs662) partially determine PON1 hydrolytic activity and protect against oxidation of LDL and HDL. This study aimed to delineate the association of PON1 status (functional 192 genotype and plasma activity levels) and atherogenicity in urbans residents aged 40 years or more.
Materials and methods
Anthropometric data, lipid profiles, the atherogenic index of the plasma (AIP) and Framingham score risk were measured. Three kinetic assays were conducted to assay PON1 status using phenylacetate and 4-(chloromethyl)phenyl acetate as substrates.
Results
Smoking per se did not significantly impact the AIP but the interaction PON1 genotype by smoking significantly increased the AIP. In subjects with the RR genotype smoking increased the AIP index from (estimated mean ± SEM) -0.038 ± 0.039 to 0.224 ± 0.094. The QR genotype increased the Framingham risk index by around 1.3 points. Smoking by RR genotype carriers significantly increased the Framingham risk score (17.23 ± 2.04) as compared to smoking (13.00 ± 1.06) and non-smoking (7.79 ± 0.70) by QQ+QR genotype carriers. The interaction RR genotype by smoking was a more important predictor (odds ratio = 7.90) of an increased Framingham risk score (> 20) than smoking per se (odds ratio = 2.73). The interaction smoking by RR genotype carriers significantly increased triglycerides and lowered HDL cholesterol.
Conclusion
Smoking per se has no (AIP) or a mild (Framingham risk score) effect on atherogenicity, while the interaction smoking by PON1 RR genotype has a clinically highly significant impact on atherogenicity.
Atherogenic index; cardiovascular risk; Framingham score risk; lipid profile; paraoxonase 1; smoking
INTRODUCTION
Human serum paraoxonase 1 enzyme (PON1; EC 3.1.8.1) is a glycoprotein synthesized in the liver and mostly bound to high density lipoproteins (HDL) particles in plasma. PON1 protects low density lipoproteins (LDL) (11. Mackness MI, Arrol S, Durrington PN. Paraoxonase prevents accumulation of lipoperoxides in low-density lipoprotein. FEBS Lett. 1991;286:152-4.) and HDL (22. Aviram M, Rosenblat M, Bisgaier CL, Newton RS, Primo-Parmo SL, La Du BN. Paraoxonase inhibits high-density lipoprotein oxidation and preserves its functions. A possible peroxidative role for paraoxonase. J Clin Invest. 1998;101(8):1581-90.) from oxidation possibly by hydrolyzing phospholipid or cholesteryl ester hydroperoxides (33. Watson AD, Berliner JA, Hama SY, La Du BN, Faull KF, Fogelman AM, et al. Protective effect of high density lipoprotein associated paraoxonase. Inhibition of the biological activity of minimally oxidized low density lipoprotein. J Clin Invest. 1995;96(6):2882-91.). Evidence for the role of PON1 in the antioxidant property of HDL is provided by the findings that PON1 knockout mice are more susceptible to atherosclerosis when fed on a fat-rich diet (44. Shih DM, Gu L, Xia YR, Navab M, Li WF, Hama S, et al. Mice lacking serum paraoxonase are susceptible to organophosphate toxicity and atherosclerosis. Nature 1998;394(6690):284-7.).
PON1 is polymorphic and single nucleotides polymorphisms (SNP) have been described both in the promoter and coding regions of the PON1 gene. The most studied polymorphism in the coding region (rs 662) results in an exchange of a glutamine (Q) by an arginine (R) at position 192 of the amino acid sequence. This PON1 Q192R polymorphism modulates the catalytic activity of PON1 but the direction of this change is substrate-dependent (55. Li WF, Costa LG, Richter RJ, Hagen T, Shih DM, Tward A, et al. Catalytic efficiency determines the in-vivo efficacy of PON1 for detoxifying organophosphorus compounds. Pharmacogenetics 2000;10(9):767-79.,66. Mackness B, Davies GK, Turkie W, Lee E, Roberts DH, Hill E, et al. Paraoxonase status in coronary heart disease: are activity and concentration more important than genotype? Arterioscler Thromb Vasc Biol. 2001;21(9):1451-7.).
Different PON1 genotypes have been investigated as markers of susceptibility to cardiovascular disease (77. Costa LG, Cole TB, Furlong CE. Paraoxonase (PON1): from toxicology to cardiovascular medicine. Acta Biomed. 2005;76 Suppl 2(10):50-7.). The PON1 Q192R polymorphism determines in part PON1 hydrolytic activity and therefore plays a role in the protection of LDL and HDL against oxidation. These effects are, however, heavily debated since both the PON1*192Q allele (88. Bhattacharyya T, Nicholls SJ, Topol EJ, Zhang R, Yang X, Schmitt D, et al. Relationship of paraoxonase 1 (PON1) gene polymorphisms and functional activity with systemic oxidative stress and cardiovascular risk. JAMA. 2008;299(11):1265-76.
9. Fujihara J, Yasuda T, Kawai Y, Morikawa N, Arakawa K, Koda Y, et al. First survey of the three gene polymorphisms (PON1 Q192R, eNOS E298D and eNOS C-786T) potentially associated with coronary artery spasm in African populations and comparison with worldwide data. Cell Biochem Funct. 2011;29(2):156-63.-1010. Regieli JJ, Jukema JW, Doevendans PA, Zwinderman AH, Kastelein JJ, Grobbee DE, et al. Paraoxonase variants relate to 10-year risk in coronary artery disease: impact of a high-density lipoprotein-bound antioxidant in secondary prevention. J Am Coll Cardiol. 2009;54(14):1238-45.) and the PON1*192R allele (1111. Voetsch B, Benke KS, Damasceno BP, Siqueira LH, Loscalzo J. Paraoxonase 192 Gln-->Arg polymorphism: an independent risk factor for nonfatal arterial ischemic stroke among young adults. Stroke. 2002;33(6):1459-64.,1212. Mohamed RH, Mohamed RH, Karam RA, Abd El-Aziz TA. The relationship between paraoxonase1-192 polymorphism and activity with coronary artery disease. Clin Biochem. 2010;43(6):553-8.) have been associated with increased risk for cardiovascular disease, while there are also studies describing no association (1313. Robertson KS, Hawe E, Miller GJ, Talmud PJ, Humphries SE; Northwick Park Heart Study II. Human paraoxonase gene cluster polymorphisms as predictors of coronary heart disease risk in the prospective Northwick Park Heart Study II. Biochim Biophys Acta. 2003;1639(3):203-12.). One factor that may explain the contradictory results is the approach used to evaluate PON1. Most studies have investigated PON1 genotype and did not include the measurement of PON1 plasmatic activity. Total PON1 plasmatic activity is negatively related to increased risk for cardiovascular disease (88. Bhattacharyya T, Nicholls SJ, Topol EJ, Zhang R, Yang X, Schmitt D, et al. Relationship of paraoxonase 1 (PON1) gene polymorphisms and functional activity with systemic oxidative stress and cardiovascular risk. JAMA. 2008;299(11):1265-76.,1414. Yildiz A, Gur M, Yilmaz R, Demirbag R, Polat M, Selek S, et al. Association of paraoxonase activity and coronary blood flow. Atherosclerosis. 2008;197(1):257-63.
15. Granér M, James RW, Kahri J, Nieminen MS, Syvänne M, Taskinen M-R. Association of paraoxonase-1 activity and concentration with angiographic severity and extent of coronary artery disease. J Am Coll Cardiol. 2006;47(12):2429-35.-1616. Tang WH, Hartiala J, Fan Y, Wu Y, Stewart AF, Erdmann J, et al. Clinical and genetic association of serum paraoxonase and arylesterase activities with cardiovascular risk. Arterioscler Thromb Vasc Biol. 2012;32(11):2803-12.). Therefore, the measurement of PON1 status, that provides a functional assignment of an individual’s PON1 Q192R polymorphism and their total total PON1 plasma activity (1717. Li W, Costa LG, Furlong CE. Serum paraoxonase status: a major factor in determining resistance to organophosphates. J Toxicol Environ Health. 1993;40(2-3):337-46.) is a more reliable marker than genotype or plasma activity measurement alone (77. Costa LG, Cole TB, Furlong CE. Paraoxonase (PON1): from toxicology to cardiovascular medicine. Acta Biomed. 2005;76 Suppl 2(10):50-7.,1818. Jarvik GP, Rozek LS, Brophy VH, Hatsukami TS, Richter RJ, Schellenberg GD, et al. Paraoxonase (PON1) phenotype is a better predictor of vascular disease than is PON1(192) or PON1(55) genotype. Arterioscler Thromb Vasc Biol. 2000;20(11):2441-7.).
Dyslipidemia is one of the major risk factors for cardiovascular disorders. Increased levels of triglycerides and total and LDL cholesterol are positively associated with cardiovascular disorders whereas high HDL levels have a protective effect. Different combinations of lipid profile parameters and other risk factors can be used to identify high risk individuals (1919. Bhardwaj S, Bhattacharjee J, Bhatnagar MK, Tyagi S, Delhi N. Atherogenic index of plasma, castelli risk index and atherogenic coefficient - new parameters in assessing cardiovascular risk. Int J Pharm Biol Sci. 2013;3(3):359-64.). One of these indexes is the atherogenic index of plasma (AIP), which is the relationship between log triglycerides and HDL cholesterol and correlates closely with LDL particle size (2020. Dobiásová M, Frohlich J. The plasma parameter log (TG/HDL-C) as an atherogenic index: correlation with lipoprotein particle size and esterification rate in apoB-lipoprotein-depleted plasma (FER(HDL)). Clin Biochem. 2001;34(7):583-8.). AIP has been considered a reliable index for atherosclerosis because it is known that the smaller the HDL-c particle, the higher the risk of esterification by lecithin cholesterol acyltransferase and consequently atherosclerosis development (2020. Dobiásová M, Frohlich J. The plasma parameter log (TG/HDL-C) as an atherogenic index: correlation with lipoprotein particle size and esterification rate in apoB-lipoprotein-depleted plasma (FER(HDL)). Clin Biochem. 2001;34(7):583-8.). Another index is the Framingham risk score, which takes into consideration variables such as age, sex, smoking status, lipid profile and hypertension to estimate a 10-year risk of a cardiovascular disease event (2121. D’Agostino RB Sr, Vasan RS, Pencina MJ, Wolf PA, Cobain M, Massaro JM, et al. General cardiovascular risk profile for use in primary care: the Framingham Heart Study. Circulation 2008;117(6):743-53.).
Considering the controversy related to the influence of PON1 Q192R on lipid profile and that no study has examined whether this polymorphism is associated with the AIP and Framingham index, this study aimed to delineate the association of PON1 status with those indexes in randomly selected urban residents aged 40 years or more.
MATERIALS AND METHODS
Subjects
This study is part of a main study on cardiovascular health (cross sectional, population based study) conducted by the Public Health Department, State University of Londrina, Brazil. The study participants were urban residents, aged 40 years or more, randomly selected from the city of Cambe, Parana State, Brazil. The study population consisted of 1180 participants (with standardized interviews) and 967 participants (interviews and blood samplings) (2222. Souza RKT De, Bortoletto MSS, Loch MR, González AD, Matsuo T, Cabrera MAS, et al. Prevalence of cardiovascular risk factors in people aged 40 years or more from the city of Cambé, PR, Brazil (2011): a population-based study. Epidemiol Serv Saúde. 2013;22(3):435-44.). Serum samples were available from 700 individuals. We excluded subjects who used multiple antiviral (1 individual) or immunossupressant (4 individuals) drugs, fish oil or omega-3 (3 individuals), lithium (4 individuals) and allopurinol (3 individuals). Based on these exclusion criteria a subset of 685 individuals was selected for this study. The study was approved by the local Ethics Committee on Human Research (CAAE: 0192.0.268.000-10). All subjects gave written informed consent to participate in the study.
Demographic and anthropometric data
Subjects were interviewed before blood sampling in order to collect socio-demographic data (sex, age, smoking and use of medication) and anthropometric variables (weight, height and waist circumference). Blood pressure was verified using the Omron HEM-742INT after 10 min of rest while the subject was seated, and the mean of two recordings was used. Waist circumference was measured using a tape with the subject standing at the level of midway between the lower rib margin and the iliac crest. Body mass index (BMI) was calculated dividing the weight in kg by the height in meters squared. Subjects were divided into non smokers (n = 564) and current smokers (n = 121). The latter group comprised 109 current smokers who smoked daily and 12 subjects who smoked regularly but not daily. The mean number of cigarettes/day was 15.2 (±10.2, standard deviation).
Laboratory measurements
After an overnight fast, blood samples were collected by venipuncture into tubes without anticoagulant. The samples were immediately centrifuged and the serum was aliquoted and stored at -80°C until processing. The lipid profile was assessed by enzymatic colorimetric methods in an automated clinical chemistry system (Dimension RXL, Siemens, USA). LDL cholesterol was calculated using Friedewald formula (2323. Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem. 1972;18(6):499-502.). LDL was not estimated in 9 individuals with triglyceride values higher than 400 mg/dL. The interassay coefficients of variation for all lipid markers were lower than 10%.
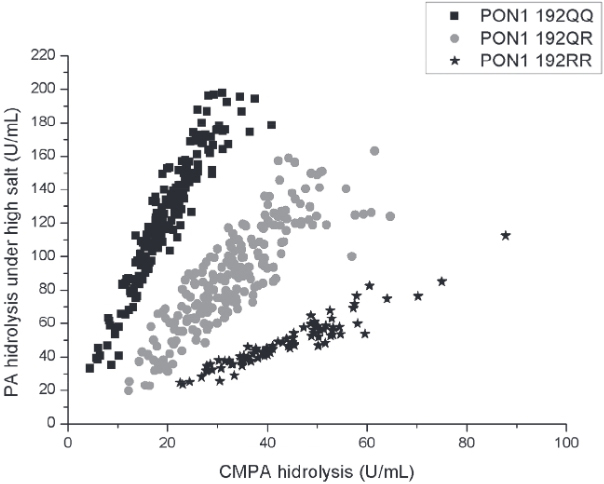
PON1 status was determined through three kinetic assays (2424. Richter RJ, Jarvik GP, Furlong CE. Determination of paraoxonase 1 status without the use of toxic organophosphate substrates. Circ Cardiovasc Genet. 2008;1(2):147-52.). To stratify individuals in the functional genotypes for the PON1 Q192R polymorphism (PON1192Q/Q, PON1192Q/R, PON1192R/R) the substrates used were phenylacetate (PA, Sigma, USA) and 4-(chloromethyl)phenyl acetate (CMPA, Sigma, USA). Q allozyme presents low efficiency to metabolize CMPA whereas both alloforms hydrolyze PA with approximately the same efficiency. The reaction with PA is conducted under high salt condition in order to partially inhibit the activity of the R allozyme thus providing a better resolution of the three PON1192 functional genotypes. The analysis was conducted in a spectrophotometer microplate reader (EnSpire, Perkin Elmer, USA). All assays were carried out in triplicate and replicates that varied by 10% or greater were repeated. Briefly, CMPA hydrolysis was measured at 280 nm for 4 min at 25ºC using 20 µL of plasma diluted 1:40 in dilution buffer [20 mmol/L Tris-HCl (pH 8.0), 1.0 mmol/L CaCl2]. PA hydrolysis under high salt conditions were measured at 270 nm for 4 min at 25ºC using 20 µL of plasma diluted 1:40 in dilution buffer. High salt media was composed by PA added to 2 mol/L NaCl, 20 mmol/L Tris-HCl (pH 8.0), 1.0 mmol/L CaCl2. The results obtained with these two assays were used to plot a 2-dimensional enzyme activity graphic that displays rates of PA hydrolysis under high salt conditions versus CMPA hydrolysis. Figure 1 shows the classification of the subjects from this study. A third assay that measures rates of PA hydrolysis at low salt concentration reveals plasma PON1 activity since under this assay condition, the PON1 Q192R polymorphism does not influence PON1 catalytic activity against PA (2525. Furlong CE, Holland N, Richter RJ, Bradman A, Ho A, Eskenazi B. PON1 status of farmworker mothers and children as a predictor of organophosphate sensitivity. Pharmacogenet. Genomics. 2006;16(3):183-90.). For this assay, rates of hydrolysis of PA were measured at 270 nm for 4 min at 25ºC using 20 µL of plasma diluted 1:80 in dilution buffer. Only the linear initial rates of substrate hydrolysis were measured for all calculations.
Population distribution plot of hydrolysis of 4-(chloromethyl)phenyl acetate versus phenylacetate. Each data point indicates one individual.
Indexes of cardiovascular disease risk
AIP was calculated as log triglycerides/HDL cholesterol (2020. Dobiásová M, Frohlich J. The plasma parameter log (TG/HDL-C) as an atherogenic index: correlation with lipoprotein particle size and esterification rate in apoB-lipoprotein-depleted plasma (FER(HDL)). Clin Biochem. 2001;34(7):583-8.). Framingham score was calculated taking into consideration the algorithms sex, age, concentrations of total cholesterol and HDL, smoking, systolic blood pressure, and self-declared treatment for hypertension status. The obtained Framingham score was then converted into the Framingham absolute risk to have a cardiovascular disease in 10 years (2626. Sposito AC, Caramelli B, Fonseca FAH, Bertolami MC, et al. IV Diretriz Brasileira sobre Dislipidemias e Prevenção da Aterosclerose. Departamento de Aterosclerose da Sociedade Brasileira de Cardiologia. Arq Bras Cardiol. 2007;88(1):2-19.).
Statistics
We used analyses of variance to check differences in continuous variables between subjects allocated to different study groups (e.g. AIP groups). If significant, the Tukey test was employed to check planned comparisons among multiple treatment means. Multivariate and univariate general linear model (GLM) analyses were used to examine the effects of explanatory variables, either categories or continuous variables, on the AIP or Framingham risk score. Pairwise comparisons among estimated marginal means were performed using the Bonferroni method. Correlations between variables were calculated using Pearson’s correlation coefficients. Analyses of contingence tables (Chi-square test) were used to ascertain the distribution of variables among study groups. We used binary logistic regression analyses to check the association between an increased AIP and Framingham risk index and explanatory variables including PON1 Q192R genotypes, smoking (and their interactions), sex, abdominal circumference, age, etc. We log-transformed variables when necessary in order to normalize data distribution. The SPSS (Windows version 19) was employed to analyze all data. Statistical significance was set at p = 0.05, two tailed.
RESULTS
PON1 status
From the 685 participants, plasma from 484 was available for PON1 status determination. Functional PON1 Q192R genotyping failed to unveil the genotype of 8 participants. According to their PON1 Q192R polymorphism, 476 participants were classified as homozygous for PON1*192Q allele (186 subjects; 39.1%); heterozygous (212 subjects; 44.5%) or homozygous for the PON1*192R allele (78 subjects; 16.4%). This population is in Hardy-Weinberg equilibrium. Regarding PON1 activity, the data in Figure 1 show the large variability of this measure (arylesterase activity in U/mL) in each functional genotype: 46.9 to 377.3 for PON1 192QQ individuals, 45.3 to 348.6 for PON1 192QR individuals and 81.29 to 269.8 for PON1 192RR individuals.
Socio-demographic and clinical data
Table 1 shows the socio-demographic and clinical data of the subjects in this study. The subjects were divided in three groups, those with normal AIP (< 0.11), medium AIP (0.11 - 0.21), and high AIP (> 0.21). There were no significant differences in age, PON1 Q192R genotypes, PON1 total activity, between the three study groups. There were significant more males in the high AIP group. BMI was significantly higher in subjects with a medium and high AIP as compared to those with a normal AIP. Waist circumference was significantly different among the three groups and increased from the normal, medium to high AIP group. All pairwise comparisons performed on total cholesterol, HDL cholesterol, triglyceride levels and AIP were significantly different between the three groups. The Framingham score was significantly higher in subjects with medium and high AIP values than in those with normal values. The Framingham risk index was significantly higher in subjects with high AIP values than in the two other subgroups. There were significant correlations between the AIP and Framingham score (r = 0.314, p < 0.001, n = 678) and Framingham risk (r = 0.277, p < 0.001, n = 678). The Framingham score and risk were significantly intercorrelated (r = 0.586, p < 0.001, n = 678). In the current smokers, there were no significant associations between the number of cigarettes smoked per day and the Framingham score, Framingham risk index, AIP, lipid levels, PON1 Q192R genotypes and PON1 total activity.
Logistic regression of odds of AIP > 0.21 versus lower AIP
Table 2 shows the results of a logistic regression analysis with individuals with an increased AIP (that is > 0.21) versus those with a normal or medium index as reference group. Automatic stepwise binary regression analysis showed that subjects with high AIP values were significantly distinguished (c2 = 66.23, df = 3, p < 0.001; Nagelkerke = 0.188) from those with a normal/medium index. Waist circumference and the interaction term RR genotype x smoking were positively associated with the high AIP group, whereas female sex was negatively associated. The significant interaction pattern between PON1 RR genotype by smoking shows that smoking by RR carriers increases the odds to belong to the high AIP group. Entering smoking (Wald = 0.34, df = 1, p = 0.558) separately showed that smoking alone was not associated with a high AIP and that the effects of the interaction RR genotype x smoking remained significant (Wald = 6.18, df = 1, p = 0.013).
AIP and HDL cholesterol and triglyceride levels
Table 3 shows the results of multivariate GLM analysis with AIP and HDL cholesterol and triglycerides levels as dependent variables and waist circumference, sex, age, plasma PON1 activity, smoking and the interaction term RR x smoking as explanatory variables (entered as factors or covariates). Multivariate tests showed that sex, age, waist circumference, total PON1 activity and the interaction RR x smoking were significantly associated with AIP, HDL cholesterol and triglycerides. Tests of between subjects’ effects showed that sex was significantly associated with AIP (higher in men); waist circumference positively with AIP and triglycerides, but negatively with HDL cholesterol; and the interaction RR x smoking positively with AIP and triglycerides, but negatively with HDL cholesterol. There was also a significant inverse correlation between age and HDL cholesterol and a positive between total PON1 activity and triglycerides. We found that 20.1% of the variance in AIP was explained (F = 16.67, df = 7/464, p < 0.001) by the regression on sex, waist circumference and the interaction between smoking and RR genotype. In subjects with the RR genotype, smoking increased the AIP index from -0.038 ± 0.039 to 0.224 ± 0.094 (estimated marginal means ± standard error). Seventeen point three percent of the variance in triglycerides (F = 13.88, df = 1, p < 0.001) was explained by the regression on waist circumference, total PON1 activity and the interaction smoking x RR genotype. Thirteen point six percent of the variance in HDL cholesterol was explained by the regression on age, waist circumference and the interaction smoking x RR genotype.
Results of multivariate general linear model (GLM) analyses with atherogenic index of plasma (AIP), high density lipoprotein cholesterol (HDL) and triglycerides (TG) as dependent variables
We have also examined the possible intervening effects of the use of different medications on the AIP index. Table 4 shows the effects of the different drugs used by the participants on the AIP. Subjects using oral hypoglycemics, oral hypoglycemics and/or insulin, hypolipidemics (fibrates and/or statins) and antihypertensives showed higher AIP values than subjects who did not take these medications. Forced entry of those 5 different medication variables in the multivariate GLM analysis displayed in Table 3 shows that after considering the effects of these 5 drugs, age (F = 7.28, df = 2/458, p = 0.001), sex (F = 4.86, df = 2/458, p = 0.008), waist circumference (F = 33.90, df = 2/458, p < 0.001), PON1 activity (F = 5.59, df = 2/458, p = 0.004) and the smoking x RR interaction (F = 3.29, df = 4/918, p = 0.011) remained significant. Only use of antihypertensives (F = 4.50, df = 2/258, p = 0.012) was significant in this multivariate GLM analysis, whereas the other 4 drugs were not significant.
Framingham risk index
Table 5 shows that a higher waist circumference, QR genotype and groups according to RR and smoking were significantly associated with the Framingham risk index after considering the effects of the significant drug variables, i.e., use of hypoglycemics, dipyrone and aspirin. The same table also shows the estimated marginal means in the genotypic groups, i.e. QR genotype and the groups divided according to smoking and the RR genotype. Thus, The QR genotype increases the Framingham risk index by around 1.3 points, whereas smoking by RR genotype carriers increases the index by 4.2 points versus smoking QQ+QR genotype carriers and 9.44 points when compared to non-smoking QQ+QR carriers. The use of dipyrone lowered the index from 13.56 (±0.95) to 11.79 (±1.09), whereas aspirin increased the index from 11.26 (±0.94) to 14.09 (±1.23). The use of hypoglycemics was associated with a higher Framingham risk index (11.29 ± 0.86 versus 14.05 ± 1.34).
Table 6 shows the results of a logistic regression analysis with the group of subjects with a high Framingham index (> 20) as dependent variable and the other subjects as reference group. Waist circumference, the QR genotype, use of hypoglycemics and groups divided according to smoking and the RR genotype were significantly associated with a higher Framingham risk index (c2 = 35.44, df = 6, p < 0.001; Nagelkerke = 0.147), whereas dipyrone was inversely associated (c2 = 35.44, df = 6, p < 0.001; Nagelkerke = 0.147). The RR genotype in combination with smoking resulted in a much higher odds ratio (i.e. 7.90) than smoking alone (i.e. 2.73).
DISCUSSION
To the best of our knowledge, this is the first study examining the association between PON1-Q192R polymorphism and atherogenic indexes, i.e. AIP and Framingham score risk, in a general population. The major finding of this study is that the PON1 Q192R polymorphism is associated with increased atherogenicity, i.e. the PON1-QR genotype increases the Framingham risk score and the interaction between smoking by PON1-RR carriers is associated with an increased AIP and Framingham score risk and by lowered HDL-cholesterol and increased triglyceride levels.
An atherogenic lipid profile and increased incidence of cardiovascular disease have been described in individuals homozygous to the PON1*192R allele (1212. Mohamed RH, Mohamed RH, Karam RA, Abd El-Aziz TA. The relationship between paraoxonase1-192 polymorphism and activity with coronary artery disease. Clin Biochem. 2010;43(6):553-8.,2727. Gluba A, Pietrucha T, Banach M, Piotrowski G, Rysz J. The role of polymorphisms within paraoxonases (192 Gln/Arg in PON1 and 311Ser/Cys in PON2) in the modulation of cardiovascular risk: a pilot study. Angiology. 2010;61(2):157-65.,2828. Hassan MA, Al-Attas OS, Hussain T, Al-Daghri NM, Alokail MS, Mohammed AK, et al. The Q192R polymorphism of the paraoxonase 1 gene is a risk factor for coronary artery disease in Saudi subjects. Mol Cell Biochem. 2013;380(1-2):121-8.). In a study performed on non-cardiovascular patients, PON1-RR smokers had a more atherogenic lipid profile (2929. Haj Mouhamed D, Ezzaher A, Mechri A, Neffati F, Omezzine A, Bouslama A, et al. Effect of cigarette smoking on paraoxonase 1 activity according to PON1 L55M and PON1 Q192R gene polymorphisms. Environ. Environ Health Prev Med. 2012;17(4):316-21.). The latter could be attributed to a lower hydrolytic activity of the PON1 R192 allozyme towards lipid peroxides as compared to the Q192 isoform (66. Mackness B, Davies GK, Turkie W, Lee E, Roberts DH, Hill E, et al. Paraoxonase status in coronary heart disease: are activity and concentration more important than genotype? Arterioscler Thromb Vasc Biol. 2001;21(9):1451-7.). On the other hand, smoking induces oxidative stress and is a known risk factor for dyslipidemias. Therefore, the pro-atherogenic state observed in PON1-RR smokers may reflect a synergism between an increased oxidative stress status induced by smoking and a decreased ability to hydrolyze lipid peroxides, assigned by the R192 allozyme. Moreover, the decreased HDL cholesterol could also have resulted from a decrease in its synthesis since the enzyme lecithin-cholesterol acyltransferase can be inactivated in the presence of higher levels of lipid hydroperoxides (3030. Bielicki JK, Forte TM. Evidence that lipid hydroperoxides inhibit plasma lecithin:cholesterol acyltransferase activity. J Lipid Res. 1999;40(5):948-54.).
It is noteworthy that even though smoking has been reported to be associated with an elevated AIP (3131. Arumalla V, Vani N, Ramarao J. Dose-dependent impacts on the diagnostic efficacies of atherogenic lipids in adult Indian smokers. J Clin Diagnostic Res. 2011;5(7):1352-5.,3232. Venkatesan A, Hemalatha A, Bobby Z, Selvaraj N, Sathiyapriya V. Effect of smoking on lipid profile and lipid peroxidation in normal subjects. Indian J Physiol Pharmacol. 2006;50(3):273-8.), in the present study AIP was not affected by smoking per se but rather by the interaction between smoking and PON1 192QR genotypes. Moreover, the impact of smoking on the Framingham risk index was much higher in PON1-RR individuals as compared to non-smoking PON1-QQ/QR individuals. Atherogenic indexes are considered to be better predictors of increased risk to cardiovascular diseases than each of the lipids (HDL-cholesterol triglycerides, total cholesterol) separately (1919. Bhardwaj S, Bhattacharjee J, Bhatnagar MK, Tyagi S, Delhi N. Atherogenic index of plasma, castelli risk index and atherogenic coefficient - new parameters in assessing cardiovascular risk. Int J Pharm Biol Sci. 2013;3(3):359-64.,2020. Dobiásová M, Frohlich J. The plasma parameter log (TG/HDL-C) as an atherogenic index: correlation with lipoprotein particle size and esterification rate in apoB-lipoprotein-depleted plasma (FER(HDL)). Clin Biochem. 2001;34(7):583-8.,3333. Dobiasova M, Frohlich J. The new atherogenic plasma index reflects the triglyceride and HDL-cholesterol ratio, the lipoprotein particle size and the cholesterol esterification rate: changes during lipanor therapy. Vnitr Lek. 2000;46(3):152-6.). The AIP for example is considered an indicator of atherogenic dyslipidemia, i.e., the combined occurrence of high fasting blood concentrations of triglycerides and low levels of HDL particles. We found that PON1-RR smokers presented a clinically highly relevant increase of 9.4 points on the Framingham risk index as compared to non-smoking PON1-QQ/QR individuals and thus almost a 10% higher risk for having a myocardial infarction in 10 years.
In the present study there was a positive correlation between PON1 plasmatic activity (arylesterase activity) and triglycerides but not with HDL. Regarding HDL cholesterol, a positive relationship was expected since the majority of PON1 proteins are bound to these particles. Roest and cols. (3434. Roest M, van Himbergen TM, Barendrecht AB, Peeters PH, van der Schouw YT, Voorbij HA. Genetic and environmental determinants of the PON-1 phenotype. Eur J Clin Invest. 2007;37(3):187-96.) described a weak association between PON1 activity (determined using PA) and HDL. These authors offered two possible explanations for this lack of/weak association: a) PON1 may be associated only with specific subspecies of HDL; b) the low saturation rate of HDL with PON1 (there is a sevenfold excess of HDL particles to bind only one PON1 molecule) which would explain the limited impact of fluctuations in HDL on PON1 concentration in blood. The association between PON1 and lipid metabolism is rather complex and the mechanisms underlying this association remain to be elucidated. At the same time that PON1, due to its protective effect on HDL and LDL, influences lipid metabolism and serum lipoproteins, lipid metabolism can also influence PON1 activity modulating its expression (3535. Rainwater DL, Rutherford S, Dyer TD, Rainwater ED, Cole SA, Vandeberg JL, et al. Determinants of variation in human serum paraoxonase activity. Heredity (Edinb). 2009;102(2):147-54.,3636. Turk R, Podpečan O, Mrkun J, Kosec M, Flegar-Meštrić Z, Perkov S, et al. Lipid mobilisation and oxidative stress as metabolic adaptation processes in dairy heifers during transition period. Anim Reprod Sci. 2013;141(3-4):109-15.). Interestingly, van Himbergen and cols. (3737. van Himbergen TM, van Tits LJ, Ter Avest E, Roest M, Voorbij HA, de Graaf J, et al. Paraoxonase (PON1) is associated with familial combined hyperlipidemia. Atherosclerosis 2008;199(1):87-94.) reported a remarkably different association between PON1 arylesterase activity and lipid profile when compared familial combined hyperlipidemia patients with their unaffected relatives. In the relatives, PON1 associated with higher levels of apoliprotein B (apoB), HDL- and LDL-cholesterol. In the patients, conversely, it associated with higher levels of VLDL-cholesterol and triglycerides. Our results indicate that a positive association between PON1 arylesterase and triglycerides may not be restricted to familial combined hyperlipidemia patients. Moreover, our results reinforce the concept that PON1 status determination is a better approach to investigate PON1 influence on lipid profile once activity reached significance as an explanatory variable to triglycerides levels whereas the interaction between PON1 Q192R polymorphism and smoking was one of the explanatory variables to AIP, Framingham score risk, triglycerides and HDL. Having both datasets in multivariate models was a step forward for meaningful interpretation of the results. As expected, male gender significantly predicted increased AIP values whereas waist circumference predicted increased AIP and triglycerides and decreased HDL. These findings reflect the higher levels of HDL in women and the negative impact central obesity has on lipid profile.
Another finding of the present study is that subjects who took drugs to treat cardiometabolic diseases presented higher AIP values. Even though statistical significance was observed for oral hypoglycemics, fibrates and antihypertensives, a similar trend could also be observed for statins and aspirin (Table 4). This finding may be reflecting the influence of cardiometabolic diseases on AIP and suggests that the use of medication does not necessarily bring AIP values to normal levels. For example, even though treatment with fibrates (2020. Dobiásová M, Frohlich J. The plasma parameter log (TG/HDL-C) as an atherogenic index: correlation with lipoprotein particle size and esterification rate in apoB-lipoprotein-depleted plasma (FER(HDL)). Clin Biochem. 2001;34(7):583-8.) and statins (3838. Kucera M, Oravec S, Hirnerova E, Huckova N, Celecova Z, Gaspar L, et al. Effect of atorvastatin on low-density lipoprotein subpopulations and comparison between indicators of plasma atherogenicity: a pilot study. Angiology. 2014;65(9):794-9.) decreases AIP when comparing pre- and post-treatment values, control values are not reached. Moreover, it has been described that in more than 70% of the patients atherogenic risk persists despite of the treatment with hypolipidemics (3939. Rizzo M, Barylski M, Rizvi A, Montalto G, Mikhailidis D, Banach M. Combined dyslipidemia: should the focus be LDL cholesterol or atherogenic dyslipidemia? Curr Pharm Des. 2013;19(21):3858-68.). Our data also show a positive association between the Framingham risk index and use of aspirin and hypoglycemic, but a negative association with dipyrone. Moreover, aspirin has cardioprotective activities (4040. Strully KW, Fowler JH, Murabito JM, Benjamin EJ, Levy D, Christakis NA. Aspirin use and cardiovascular events in social networks. Soc Sci Med. 2012;74(7):1125-9.
41. Dorresteijn JA, Visseren FL, Ridker PM, Paynter NP, Wassink AM, Buring JE, et al. Aspirin for primary prevention of vascular events in women: individualized prediction of treatment effects. Eur Heart J. 2011;32(23):2962-9.-4242. Dallmeier D, Larson MG, Vasan RS, Keaney JF Jr, Fontes JD, Meigs JB, et al. Metabolic syndrome and inflammatory biomarkers: a community-based cross-sectional study at the Framingham Heart Study. Diabetol Metab Syndr. 2012;4(1):28.). Our study, however, was not designed to examine the effects of drugs on the atherogenic indexes and data interpretation is complex since some subjects presented more than one cardiometabolic disease and various subjects were polymedicated. Most importantly is that after adjusting our data for drug use, AIP remained positively and significantly associated with PON1*192R allele carrier smokers.
Finally, supporting the high influence of European colonization in the south of Brazil, the present study as well as a study published in 2002 (4343. Allebrandt KV, Souza RL, Chautard-Freire-Maia EA. Variability of the paraoxonase gene (PON1) in Euro- and Afro-Brazilians. Toxicol Appl Pharmacol. 2002;180(3):151-6.) describe a higher frequency of PON1192Q/Q and heterozygous than PON1192R/R among the subjects. Population-based studies and meta-analysis have shown that the PON1*192R allele is more common in African population, whereas Q isoform is more frequent in Caucasians (66. Mackness B, Davies GK, Turkie W, Lee E, Roberts DH, Hill E, et al. Paraoxonase status in coronary heart disease: are activity and concentration more important than genotype? Arterioscler Thromb Vasc Biol. 2001;21(9):1451-7.).
This study has strengths and limitations that must be considered for the interpretation of the results. Firstly, considering the factors that have been described to influence PON1 activity (for a review, see [4444. Costa LG, Giordano G, Furlong CE. Pharmacological and dietary modulators of paraoxonase 1 (PON1) activity and expression: the hunt goes on. Biochem Pharmacol. 2011;81(3):337-44.]) limitations of our study include lack of information on nutrition and alcohol consumption. Secondly, this is a cross-sectional study and therefore we cannot assert causality. Strengths are that our results were adjusted for many potential confounders, including age, sex, waist circumference, smoking and use of drugs. Most importantly, we included the functional measure of PON1 total plasmatic activity for each subject within each PON1 Q192R phenotypic group. The large variability observed among individuals represent important differences in the individual’s rates of detoxification of endogenous toxic metabolites as well as xenobiotics.
This study shows an important interaction between PON1 192RR functional genotype and smoking. Smoking by PON1 RR carriers was associated by significantly increased AIP, triglyceride levels and a Framingham score risk and by significantly lower levels of HDL cholesterol. The findings provide an example of gene-environment interactions that increase cardiovascular risk.
REFERENCES
-
1Mackness MI, Arrol S, Durrington PN. Paraoxonase prevents accumulation of lipoperoxides in low-density lipoprotein. FEBS Lett. 1991;286:152-4.
-
2Aviram M, Rosenblat M, Bisgaier CL, Newton RS, Primo-Parmo SL, La Du BN. Paraoxonase inhibits high-density lipoprotein oxidation and preserves its functions. A possible peroxidative role for paraoxonase. J Clin Invest. 1998;101(8):1581-90.
-
3Watson AD, Berliner JA, Hama SY, La Du BN, Faull KF, Fogelman AM, et al. Protective effect of high density lipoprotein associated paraoxonase. Inhibition of the biological activity of minimally oxidized low density lipoprotein. J Clin Invest. 1995;96(6):2882-91.
-
4Shih DM, Gu L, Xia YR, Navab M, Li WF, Hama S, et al. Mice lacking serum paraoxonase are susceptible to organophosphate toxicity and atherosclerosis. Nature 1998;394(6690):284-7.
-
5Li WF, Costa LG, Richter RJ, Hagen T, Shih DM, Tward A, et al. Catalytic efficiency determines the in-vivo efficacy of PON1 for detoxifying organophosphorus compounds. Pharmacogenetics 2000;10(9):767-79.
-
6Mackness B, Davies GK, Turkie W, Lee E, Roberts DH, Hill E, et al. Paraoxonase status in coronary heart disease: are activity and concentration more important than genotype? Arterioscler Thromb Vasc Biol. 2001;21(9):1451-7.
-
7Costa LG, Cole TB, Furlong CE. Paraoxonase (PON1): from toxicology to cardiovascular medicine. Acta Biomed. 2005;76 Suppl 2(10):50-7.
-
8Bhattacharyya T, Nicholls SJ, Topol EJ, Zhang R, Yang X, Schmitt D, et al. Relationship of paraoxonase 1 (PON1) gene polymorphisms and functional activity with systemic oxidative stress and cardiovascular risk. JAMA. 2008;299(11):1265-76.
-
9Fujihara J, Yasuda T, Kawai Y, Morikawa N, Arakawa K, Koda Y, et al. First survey of the three gene polymorphisms (PON1 Q192R, eNOS E298D and eNOS C-786T) potentially associated with coronary artery spasm in African populations and comparison with worldwide data. Cell Biochem Funct. 2011;29(2):156-63.
-
10Regieli JJ, Jukema JW, Doevendans PA, Zwinderman AH, Kastelein JJ, Grobbee DE, et al. Paraoxonase variants relate to 10-year risk in coronary artery disease: impact of a high-density lipoprotein-bound antioxidant in secondary prevention. J Am Coll Cardiol. 2009;54(14):1238-45.
-
11Voetsch B, Benke KS, Damasceno BP, Siqueira LH, Loscalzo J. Paraoxonase 192 Gln-->Arg polymorphism: an independent risk factor for nonfatal arterial ischemic stroke among young adults. Stroke. 2002;33(6):1459-64.
-
12Mohamed RH, Mohamed RH, Karam RA, Abd El-Aziz TA. The relationship between paraoxonase1-192 polymorphism and activity with coronary artery disease. Clin Biochem. 2010;43(6):553-8.
-
13Robertson KS, Hawe E, Miller GJ, Talmud PJ, Humphries SE; Northwick Park Heart Study II. Human paraoxonase gene cluster polymorphisms as predictors of coronary heart disease risk in the prospective Northwick Park Heart Study II. Biochim Biophys Acta. 2003;1639(3):203-12.
-
14Yildiz A, Gur M, Yilmaz R, Demirbag R, Polat M, Selek S, et al. Association of paraoxonase activity and coronary blood flow. Atherosclerosis. 2008;197(1):257-63.
-
15Granér M, James RW, Kahri J, Nieminen MS, Syvänne M, Taskinen M-R. Association of paraoxonase-1 activity and concentration with angiographic severity and extent of coronary artery disease. J Am Coll Cardiol. 2006;47(12):2429-35.
-
16Tang WH, Hartiala J, Fan Y, Wu Y, Stewart AF, Erdmann J, et al. Clinical and genetic association of serum paraoxonase and arylesterase activities with cardiovascular risk. Arterioscler Thromb Vasc Biol. 2012;32(11):2803-12.
-
17Li W, Costa LG, Furlong CE. Serum paraoxonase status: a major factor in determining resistance to organophosphates. J Toxicol Environ Health. 1993;40(2-3):337-46.
-
18Jarvik GP, Rozek LS, Brophy VH, Hatsukami TS, Richter RJ, Schellenberg GD, et al. Paraoxonase (PON1) phenotype is a better predictor of vascular disease than is PON1(192) or PON1(55) genotype. Arterioscler Thromb Vasc Biol. 2000;20(11):2441-7.
-
19Bhardwaj S, Bhattacharjee J, Bhatnagar MK, Tyagi S, Delhi N. Atherogenic index of plasma, castelli risk index and atherogenic coefficient - new parameters in assessing cardiovascular risk. Int J Pharm Biol Sci. 2013;3(3):359-64.
-
20Dobiásová M, Frohlich J. The plasma parameter log (TG/HDL-C) as an atherogenic index: correlation with lipoprotein particle size and esterification rate in apoB-lipoprotein-depleted plasma (FER(HDL)). Clin Biochem. 2001;34(7):583-8.
-
21D’Agostino RB Sr, Vasan RS, Pencina MJ, Wolf PA, Cobain M, Massaro JM, et al. General cardiovascular risk profile for use in primary care: the Framingham Heart Study. Circulation 2008;117(6):743-53.
-
22Souza RKT De, Bortoletto MSS, Loch MR, González AD, Matsuo T, Cabrera MAS, et al. Prevalence of cardiovascular risk factors in people aged 40 years or more from the city of Cambé, PR, Brazil (2011): a population-based study. Epidemiol Serv Saúde. 2013;22(3):435-44.
-
23Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem. 1972;18(6):499-502.
-
24Richter RJ, Jarvik GP, Furlong CE. Determination of paraoxonase 1 status without the use of toxic organophosphate substrates. Circ Cardiovasc Genet. 2008;1(2):147-52.
-
25Furlong CE, Holland N, Richter RJ, Bradman A, Ho A, Eskenazi B. PON1 status of farmworker mothers and children as a predictor of organophosphate sensitivity. Pharmacogenet. Genomics. 2006;16(3):183-90.
-
26Sposito AC, Caramelli B, Fonseca FAH, Bertolami MC, et al. IV Diretriz Brasileira sobre Dislipidemias e Prevenção da Aterosclerose. Departamento de Aterosclerose da Sociedade Brasileira de Cardiologia. Arq Bras Cardiol. 2007;88(1):2-19.
-
27Gluba A, Pietrucha T, Banach M, Piotrowski G, Rysz J. The role of polymorphisms within paraoxonases (192 Gln/Arg in PON1 and 311Ser/Cys in PON2) in the modulation of cardiovascular risk: a pilot study. Angiology. 2010;61(2):157-65.
-
28Hassan MA, Al-Attas OS, Hussain T, Al-Daghri NM, Alokail MS, Mohammed AK, et al. The Q192R polymorphism of the paraoxonase 1 gene is a risk factor for coronary artery disease in Saudi subjects. Mol Cell Biochem. 2013;380(1-2):121-8.
-
29Haj Mouhamed D, Ezzaher A, Mechri A, Neffati F, Omezzine A, Bouslama A, et al. Effect of cigarette smoking on paraoxonase 1 activity according to PON1 L55M and PON1 Q192R gene polymorphisms. Environ. Environ Health Prev Med. 2012;17(4):316-21.
-
30Bielicki JK, Forte TM. Evidence that lipid hydroperoxides inhibit plasma lecithin:cholesterol acyltransferase activity. J Lipid Res. 1999;40(5):948-54.
-
31Arumalla V, Vani N, Ramarao J. Dose-dependent impacts on the diagnostic efficacies of atherogenic lipids in adult Indian smokers. J Clin Diagnostic Res. 2011;5(7):1352-5.
-
32Venkatesan A, Hemalatha A, Bobby Z, Selvaraj N, Sathiyapriya V. Effect of smoking on lipid profile and lipid peroxidation in normal subjects. Indian J Physiol Pharmacol. 2006;50(3):273-8.
-
33Dobiasova M, Frohlich J. The new atherogenic plasma index reflects the triglyceride and HDL-cholesterol ratio, the lipoprotein particle size and the cholesterol esterification rate: changes during lipanor therapy. Vnitr Lek. 2000;46(3):152-6.
-
34Roest M, van Himbergen TM, Barendrecht AB, Peeters PH, van der Schouw YT, Voorbij HA. Genetic and environmental determinants of the PON-1 phenotype. Eur J Clin Invest. 2007;37(3):187-96.
-
35Rainwater DL, Rutherford S, Dyer TD, Rainwater ED, Cole SA, Vandeberg JL, et al. Determinants of variation in human serum paraoxonase activity. Heredity (Edinb). 2009;102(2):147-54.
-
36Turk R, Podpečan O, Mrkun J, Kosec M, Flegar-Meštrić Z, Perkov S, et al. Lipid mobilisation and oxidative stress as metabolic adaptation processes in dairy heifers during transition period. Anim Reprod Sci. 2013;141(3-4):109-15.
-
37van Himbergen TM, van Tits LJ, Ter Avest E, Roest M, Voorbij HA, de Graaf J, et al. Paraoxonase (PON1) is associated with familial combined hyperlipidemia. Atherosclerosis 2008;199(1):87-94.
-
38Kucera M, Oravec S, Hirnerova E, Huckova N, Celecova Z, Gaspar L, et al. Effect of atorvastatin on low-density lipoprotein subpopulations and comparison between indicators of plasma atherogenicity: a pilot study. Angiology. 2014;65(9):794-9.
-
39Rizzo M, Barylski M, Rizvi A, Montalto G, Mikhailidis D, Banach M. Combined dyslipidemia: should the focus be LDL cholesterol or atherogenic dyslipidemia? Curr Pharm Des. 2013;19(21):3858-68.
-
40Strully KW, Fowler JH, Murabito JM, Benjamin EJ, Levy D, Christakis NA. Aspirin use and cardiovascular events in social networks. Soc Sci Med. 2012;74(7):1125-9.
-
41Dorresteijn JA, Visseren FL, Ridker PM, Paynter NP, Wassink AM, Buring JE, et al. Aspirin for primary prevention of vascular events in women: individualized prediction of treatment effects. Eur Heart J. 2011;32(23):2962-9.
-
42Dallmeier D, Larson MG, Vasan RS, Keaney JF Jr, Fontes JD, Meigs JB, et al. Metabolic syndrome and inflammatory biomarkers: a community-based cross-sectional study at the Framingham Heart Study. Diabetol Metab Syndr. 2012;4(1):28.
-
43Allebrandt KV, Souza RL, Chautard-Freire-Maia EA. Variability of the paraoxonase gene (PON1) in Euro- and Afro-Brazilians. Toxicol Appl Pharmacol. 2002;180(3):151-6.
-
44Costa LG, Giordano G, Furlong CE. Pharmacological and dietary modulators of paraoxonase 1 (PON1) activity and expression: the hunt goes on. Biochem Pharmacol. 2011;81(3):337-44.
-
Acknowledgment of grant support: this work was supported by SETI/Fundação Araucária (PPSUS 200/210). André de Souza-Nogueira received a Master fellowhip from Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (Capes). Estefania Gastaldello Moreira and Décio Sabbatini Barbosa are senior research fellows from SETI/Fundação Araucária. Michael Maes is supported by a Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) PVE fellowship at the Health Sciences Graduation Program, Londrina State University (UEL).
Publication Dates
-
Publication in this collection
25 Aug 2016 -
Date of issue
Oct 2016
History
-
Received
15 Sept 2015 -
Accepted
16 Dec 2015