ABSTRACT
Prolactinomas are the most common pituitary adenomas (approximately 40% of cases), and they represent an important cause of hypogonadism and infertility in both sexes. The magnitude of prolactin (PRL) elevation can be useful in determining the etiology of hyperprolactinemia. Indeed, PRL levels > 250 ng/mL are highly suggestive of the presence of a prolactinoma. In contrast, most patients with stalk dysfunction, drug-induced hyperprolactinemia or systemic diseases present with PRL levels < 100 ng/mL. However, exceptions to these rules are not rare. On the other hand, among patients with macroprolactinomas (MACs), artificially low PRL levels may result from the so-called “hook effect”. Patients harboring cystic MACs may also present with a mild PRL elevation. The screening for macroprolactin is mostly indicated for asymptomatic patients and those with apparent idiopathic hyperprolactinemia. Dopamine agonists (DAs) are the treatment of choice for prolactinomas, particularly cabergoline, which is more effective and better tolerated than bromocriptine. After 2 years of successful treatment, DA withdrawal should be considered in all cases of microprolactinomas and in selected cases of MACs. In this publication, the goal of the Neuroendocrinology Department of the Brazilian Society of Endocrinology and Metabolism (SBEM) is to provide a review of the diagnosis and treatment of hyperprolactinemia and prolactinomas, emphasizing controversial issues regarding these topics. This review is based on data published in the literature and the authors' experience.
Keywords
Hyperprolactinemia; prolactinomas; pseudoprolactinomas; macroprolactin; hook-effect; dopamine agonists; pituitary surgery; temozolomide
INTRODUCTION
Hyperprolactinemia has multiple etiologies (Table 1) and is the most common endocrine disorder of the hypothalamic-pituitary axis (11. Vilar L, Fleseriu M, Bronstein MD. Pitfalls in the diagnosis of hyperprolactinemia. Arq Bras Endocrinol Metab. 2014;58(1):9-22.–33. Bronstein MD. Disorders of prolactin secretion and prolactinomas. In: DeGroot LJ, Jameson JL, editors. Endocrinology. 6th ed. Philadelphia: Saunders/Elsevier; 2010. p. 333-57.). A prolactinoma is the most common cause of chronic hyperprolactinemia once pregnancy, primary hypothyroidism, and drugs that raise serum prolactin (PRL) levels have been ruled out (44. Melmed S, Casanueva FF, Hoffman AR, Kleinberg DL, Montori VM, Schlechte JA, et al. Diagnosis and treatment of hyperprolactinemia: An Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(2):273-88.–66. Glezer A, Bronstein MD. Prolactinomas. Endocrinol Metab Clin N Am. 2015;44(1):71-8.).
Prolactinomas are the most common hormone-secreting pituitary tumors, accounting for approximately 40% of all pituitary tumors (22. Vilar L, Naves LA, Fleseriu M. Avaliação diagnóstica da hiperprolactinemia (Diagnostic evaluation of hyperprolactinemia). In: Vilar L, editor. Endocrinologia Clínica, 6a ed. (Clinical Endocrinology, 6th ed.). Rio de Janeiro: Guanabara Koogan; 2016. p. 3-14.,66. Glezer A, Bronstein MD. Prolactinomas. Endocrinol Metab Clin N Am. 2015;44(1):71-8.) In adults, prolactinomas have an estimated prevalence of 60-100 per million population (77. Colao A, Lombardi G. Growth hormone and prolactin excess. Lancet. 1998;352:1455-61.,88. Ciccarelli A, Daly AF, Beckers A. The epidemiology of prolactinomas. Pituitary 2005;8(1):3-6.), and in a population from three different districts of Belgium, prolactinomas have been reported to represent 73.3% of all pituitary adenomas, with a higher prevalence in women (78.2%) (99. Daly AF, Rixhon M, Adam C, Dempegioti A, Tichomirowa MA, Beckers A. High prevalence of pituitary adenomas: a cross-sectional study in the province of Liege, Belgium. J Clin Endocrinol Metab. 2006;91(12):4769-75.). Between the age of 20 and 50 years, the ratio between women and men is estimated to be 10:1, whereas after the fifth decade of life, both genders are equally affected (1010. Auriema R, Pivonello R, Colao A. Prolactinomas. In: Vilar L (editor). In: Vilar L, editor. Endocrinologia Clínica, 6a ed. (Clinical Endocrinology, 6th ed.). Rio de Janeiro: Guanabara Koogan; 2016. p. 15-29.,1111. Colao A, Sarno AD, Cappabianca P, Briganti F, Pivonello R, Di Somma C, et al. Gender differences in the prevalence, clinical features and response to cabergoline in hyperprolactinemia. Eur J Endocrinol. 2003;148(3):325-31.). Although prolactinomas are rare at the pediatric and adolescent ages, they account for approximately half of all pituitary adenomas in that population (1212. Fideleff HL, Boquete HR, Suárez MG, Azaretzky M. Prolactinoma in children and adolescents. Horm Res. 2009;72(4):197-205.). PRL-secreting carcinomas are extremely rare (1313. Heaney AP. Pituitary carcinoma: difficult diagnosis and treatment. J Clin Endocrinol Metab. 2011;96(12):3649-60.).
The most characteristic signs and symptoms found in patients with hyperprolactinemia are those related to hypogonadotropic hypogonadism and galactorrhea (11. Vilar L, Fleseriu M, Bronstein MD. Pitfalls in the diagnosis of hyperprolactinemia. Arq Bras Endocrinol Metab. 2014;58(1):9-22.,33. Bronstein MD. Disorders of prolactin secretion and prolactinomas. In: DeGroot LJ, Jameson JL, editors. Endocrinology. 6th ed. Philadelphia: Saunders/Elsevier; 2010. p. 333-57.,77. Colao A, Lombardi G. Growth hormone and prolactin excess. Lancet. 1998;352:1455-61.). Increased PRL levels decrease gonadotropin pulsatile secretion through inhibition of hypothalamic GnRH release (1414. Milenković L, D'Angelo G, Kelly PA, Weiner RI. Inhibition of gonadotropin hormone-releasing hormone release by prolactin from GT1 neuronal cell lines through prolactin receptors. Proc Natl Acad Sci USA. 1994;91(4):1244-7.). In addition, there may be direct effects of hyperprolactinemia on testes and ovaries. Hypogonadism can cause menstrual irregularity and amenorrhea in women, sexual dysfunction, infertility, and loss of bone mineral mass in both genders (1515. Buvat J, Lemaire A, Buvat-Herbaut M, Fourlinnie JC, Racadot A, Fossati P. Hyperprolactinemia and sexual function in men. Horm Res. 1985;22(3):196-203.,1616. Corona G, Mannucci E, Fisher AD, Lotti F, Ricca V, Balercia G, et al. Effect of hyperprolactinemia in male patients consulting for sexual dysfunction. J Sex Med. 2007;4(5):1485-93.). Hyperprolactinemia can also reduce the libido independently of testosterone levels (1717. Huang W, Molitch M. Evaluation and management of galactorrhea. Am Fam Physician. 2012;85(11):1073-80.). In patients harboring macroprolactinomas, tumor mass effect symptoms, such as headache, visual changes, and, more rarely, cerebrospinal fluid (CSF) rhinorrhea, hydrocephalus and seizures, can also occur (11. Vilar L, Fleseriu M, Bronstein MD. Pitfalls in the diagnosis of hyperprolactinemia. Arq Bras Endocrinol Metab. 2014;58(1):9-22.–33. Bronstein MD. Disorders of prolactin secretion and prolactinomas. In: DeGroot LJ, Jameson JL, editors. Endocrinology. 6th ed. Philadelphia: Saunders/Elsevier; 2010. p. 333-57.). Hypopituitarism beyond hypogonadism can occur if there is compression of the pituitary stalk or destruction of normal pituitary tissue (33. Bronstein MD. Disorders of prolactin secretion and prolactinomas. In: DeGroot LJ, Jameson JL, editors. Endocrinology. 6th ed. Philadelphia: Saunders/Elsevier; 2010. p. 333-57.,66. Glezer A, Bronstein MD. Prolactinomas. Endocrinol Metab Clin N Am. 2015;44(1):71-8.,77. Colao A, Lombardi G. Growth hormone and prolactin excess. Lancet. 1998;352:1455-61.). It is worth commenting that some women present with non-puerperal galactorrhea in the presence of regular menstrual cycles and normal PRL levels (1818. Sakiyama R, Quan M. Galactorrhea and hyperprolactinemia. Obstet Gynecol Surv. 1983;38(12):689-700.,1919. Romijn JA. Hyperprolactinemia and prolactinoma. Handb Clin Neurol. 2014;124:185-95.). This so-called “idiopathic galactorrhea” is estimated to be present in up to 40-50% of all women with non-puerperal galactorrhea (1919. Romijn JA. Hyperprolactinemia and prolactinoma. Handb Clin Neurol. 2014;124:185-95.,2020. Suh HK, Frantz AG. Size heterogeneity of human prolactin in plasma and pituitary extracts. J Clin Endocrinol Metab. 1974;39(5):928-35.). In contrast, the finding of galactorrhea in men is highly suggestive of a prolactinoma (22. Vilar L, Naves LA, Fleseriu M. Avaliação diagnóstica da hiperprolactinemia (Diagnostic evaluation of hyperprolactinemia). In: Vilar L, editor. Endocrinologia Clínica, 6a ed. (Clinical Endocrinology, 6th ed.). Rio de Janeiro: Guanabara Koogan; 2016. p. 3-14.,1818. Sakiyama R, Quan M. Galactorrhea and hyperprolactinemia. Obstet Gynecol Surv. 1983;38(12):689-700.).
In this publication, the goal of the Neuroendocrinology Department of the Brazilian Society of Endocrinology and Metabolism (SBEM) is to provide a review on the diagnosis and treatment of hyperprolactinemia and prolactinomas, emphasizing controversial issues regarding these topics.
PROLACTIN SERUM ISOFORMS
The PRL size is heterogeneous in terms of circulating molecular forms. The predominant form in healthy subjects and in patients with prolactinomas is monomeric PRL (molecular weight of 23 kDa), while dimeric or big PRL (45-60 kDa) and big-big PRL or macroprolactin (150-170 kDa) correspond to less than 20% of the total PRL (2020. Suh HK, Frantz AG. Size heterogeneity of human prolactin in plasma and pituitary extracts. J Clin Endocrinol Metab. 1974;39(5):928-35.,2121. Sinha YN. Structural variants of prolactin: occurrence and physiological significance. Endocr Rev. 1995;16(3):354-69.). When the serum of a patient with hyperprolactinemia contains mostly macroprolactin, the condition is termed macroprolactinemia (2222. Jackson RD, Wortsman J, Malarkey WB. Characterization of a large molecular weight prolactin in women with idiopathic hyperprolactinemia and normal menses. J Clin Endocrinol Metab. 1985;61(2):258-64.,2323. Jackson RD, Wortsman J, Malarkey WB. Macroprolactinemia presenting like a pituitary tumor. Am J Med. 1985;78(2):346-50.). In up to 90% of cases, macroprolactin is composed of a complex formed by an IgG and monomeric PRL (22. Vilar L, Naves LA, Fleseriu M. Avaliação diagnóstica da hiperprolactinemia (Diagnostic evaluation of hyperprolactinemia). In: Vilar L, editor. Endocrinologia Clínica, 6a ed. (Clinical Endocrinology, 6th ed.). Rio de Janeiro: Guanabara Koogan; 2016. p. 3-14.,2424. Hattori N. The frequency of macroprolactinemia in pregnant women and the heterogeneity of its etiologies. J Clin Endocrinol Metab. 1996;81(2):586-90.–2929. Kasum M, Oreskovic S, Zec I, Jezek D, Tomic V, Gall V, et al. Macroprolactinemia: new insights in hyperprolactinemia. Biochem Med (Zagreb). 2012;22(2):171-9.).
DIAGNOSTIC EVALUATION
For the correct identification of the etiology of hyperprolactinemia, some parameters must be taken into account: medical history, physical examination, clinical features, laboratory findings (especially PRL serum levels), and imaging studies of the pituitary and sella turcica (11. Vilar L, Fleseriu M, Bronstein MD. Pitfalls in the diagnosis of hyperprolactinemia. Arq Bras Endocrinol Metab. 2014;58(1):9-22.,33. Bronstein MD. Disorders of prolactin secretion and prolactinomas. In: DeGroot LJ, Jameson JL, editors. Endocrinology. 6th ed. Philadelphia: Saunders/Elsevier; 2010. p. 333-57.,55. Glezer A, Bronstein MD. Approach to the patient with persistent hyperprolactinemia and negative sellar imaging. J Clin Endocrinol Metab. 2012;97(7):2211-6.). Furthermore, the screening for macroprolactinemia should often be considered, particularly in cases of asymptomatic hyperprolactinemia (33. Bronstein MD. Disorders of prolactin secretion and prolactinomas. In: DeGroot LJ, Jameson JL, editors. Endocrinology. 6th ed. Philadelphia: Saunders/Elsevier; 2010. p. 333-57.–55. Glezer A, Bronstein MD. Approach to the patient with persistent hyperprolactinemia and negative sellar imaging. J Clin Endocrinol Metab. 2012;97(7):2211-6.).
In addition to PRL determination, TSH, free T4, and creatinine levels should be obtained to rule out secondary causes of hyperprolactinemia (11. Vilar L, Fleseriu M, Bronstein MD. Pitfalls in the diagnosis of hyperprolactinemia. Arq Bras Endocrinol Metab. 2014;58(1):9-22.,33. Bronstein MD. Disorders of prolactin secretion and prolactinomas. In: DeGroot LJ, Jameson JL, editors. Endocrinology. 6th ed. Philadelphia: Saunders/Elsevier; 2010. p. 333-57.,66. Glezer A, Bronstein MD. Prolactinomas. Endocrinol Metab Clin N Am. 2015;44(1):71-8.). Moreover, acromegaly must be investigated by measuring IGF-1 in all patients with a macroadenoma, even when there are no manifestations of this disease (3030. Vilar L, Czepielewski MA, Naves LA, Rollin GA, Casulari LA, Coelho CE. Substantial shrinkage of adenomas cosecreting growth hormone and prolactin with use of cabergoline therapy. Endocr Pract. 2007;13(4):396-402.). Finally, β-hCG measurement is mandatory in any woman of childbearing age with amenorrhea (11. Vilar L, Fleseriu M, Bronstein MD. Pitfalls in the diagnosis of hyperprolactinemia. Arq Bras Endocrinol Metab. 2014;58(1):9-22.,33. Bronstein MD. Disorders of prolactin secretion and prolactinomas. In: DeGroot LJ, Jameson JL, editors. Endocrinology. 6th ed. Philadelphia: Saunders/Elsevier; 2010. p. 333-57.).
1. CONTROVERSIAL ISSUES REGARDING DIAGNOSIS
1.1. Environmental influences on PRL secretion
Prolactin is secreted in a pulsatile manner, and serum levels can vary greatly throughout the day, with higher levels during sleep, a morning peak and a gradual decline after awakening, but without a typical circadian rhythm. Under normal conditions, ~50% of the total daily production of PRL occurs during the sleep period. Thus, samples should be collected up to 3 hours after awakening, preferably while the patient is fasted (11. Vilar L, Fleseriu M, Bronstein MD. Pitfalls in the diagnosis of hyperprolactinemia. Arq Bras Endocrinol Metab. 2014;58(1):9-22.,44. Melmed S, Casanueva FF, Hoffman AR, Kleinberg DL, Montori VM, Schlechte JA, et al. Diagnosis and treatment of hyperprolactinemia: An Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(2):273-88.).
Stress from any source, whether psychological, induced by exercise or due to other acute illness, leads to the physiological elevation of PRL levels (22. Vilar L, Naves LA, Fleseriu M. Avaliação diagnóstica da hiperprolactinemia (Diagnostic evaluation of hyperprolactinemia). In: Vilar L, editor. Endocrinologia Clínica, 6a ed. (Clinical Endocrinology, 6th ed.). Rio de Janeiro: Guanabara Koogan; 2016. p. 3-14.,33. Bronstein MD. Disorders of prolactin secretion and prolactinomas. In: DeGroot LJ, Jameson JL, editors. Endocrinology. 6th ed. Philadelphia: Saunders/Elsevier; 2010. p. 333-57.,55. Glezer A, Bronstein MD. Approach to the patient with persistent hyperprolactinemia and negative sellar imaging. J Clin Endocrinol Metab. 2012;97(7):2211-6.). However, supine rest is not necessary prior to sampling, contrary to what was believed in the past (3131. Vieira JGH, Oliveira JH, Tachihana T, Maciel RMB, Hauache OM. Evaluation of plasma prolactin levels: is it necessary to rest before the collection? Arq Bras Endocrinol Metab. 2006;50(3):569-70.). Venipuncture stress may cause an elevation in the PRL level, but it is usually mild (< 40-60 ng/mL) (3232. Ferriani RA, Sá MFS. Effect of venipuncture stress on plasma prolactin levels. Int J Gynaecol Obstet. 1985;23(6):459-62.). The same is true for breast stimulation (11. Vilar L, Fleseriu M, Bronstein MD. Pitfalls in the diagnosis of hyperprolactinemia. Arq Bras Endocrinol Metab. 2014;58(1):9-22.,22. Vilar L, Naves LA, Fleseriu M. Avaliação diagnóstica da hiperprolactinemia (Diagnostic evaluation of hyperprolactinemia). In: Vilar L, editor. Endocrinologia Clínica, 6a ed. (Clinical Endocrinology, 6th ed.). Rio de Janeiro: Guanabara Koogan; 2016. p. 3-14.,3333. Molitch ME. Disorders of prolactin secretion. Endocrinol Metab Clin North Am. 2001;30(3):585-610.). Moreover, as PRL is secreted episodically, its levels measured during the day may possibly be beyond the upper limit of normality for a particular laboratory in healthy individuals (11. Vilar L, Fleseriu M, Bronstein MD. Pitfalls in the diagnosis of hyperprolactinemia. Arq Bras Endocrinol Metab. 2014;58(1):9-22.,33. Bronstein MD. Disorders of prolactin secretion and prolactinomas. In: DeGroot LJ, Jameson JL, editors. Endocrinology. 6th ed. Philadelphia: Saunders/Elsevier; 2010. p. 333-57.). Therefore, an elevated PRL level should be confirmed at least once (3333. Molitch ME. Disorders of prolactin secretion. Endocrinol Metab Clin North Am. 2001;30(3):585-610.) unless the PRL levels are clearly elevated (> 80-100 ng/mL) (11. Vilar L, Fleseriu M, Bronstein MD. Pitfalls in the diagnosis of hyperprolactinemia. Arq Bras Endocrinol Metab. 2014;58(1):9-22.). Nevertheless, according to the guidelines of the Endocrine Society, a single PRL level above the upper limit of normal confirms the diagnosis of hyperprolactinemia, as long as the serum sample was obtained without excessive venipuncture stress (44. Melmed S, Casanueva FF, Hoffman AR, Kleinberg DL, Montori VM, Schlechte JA, et al. Diagnosis and treatment of hyperprolactinemia: An Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(2):273-88.).
COMMENT 1: As PRL is secreted in a pulsatile manner and as venipuncture stress can increase PRL levels, we suggest that an elevated PRL level should be confirmed at least once, unless the PRL levels are clearly elevated (> 80-100 ng/mL).
COMMENT 2: Vigorous exercise and nipple stimulation should be avoided for at least 30 minutes before checking PRL levels as they may result in PRL elevation.
1.2 Accuracy of prolactin levels
The magnitude of PRL elevation can be useful in determining the etiology of hyperprolactinemia because the highest values are observed in patients with prolactinomas (11. Vilar L, Fleseriu M, Bronstein MD. Pitfalls in the diagnosis of hyperprolactinemia. Arq Bras Endocrinol Metab. 2014;58(1):9-22.–55. Glezer A, Bronstein MD. Approach to the patient with persistent hyperprolactinemia and negative sellar imaging. J Clin Endocrinol Metab. 2012;97(7):2211-6.,3333. Molitch ME. Disorders of prolactin secretion. Endocrinol Metab Clin North Am. 2001;30(3):585-610.,3434. Vilar L, Freitas MC, Naves LA, Casulari LA, Azevedo M, Montenegro R Jr, et al. Diagnosis and management of hyperprolactinemia: results of a Brazilian multicenter study with 1234 patients. J Endocrinol Invest. 2008;31(5):436-44.). For example, levels > 250 ng/mL are highly suggestive of the presence of a prolactinoma (33. Bronstein MD. Disorders of prolactin secretion and prolactinomas. In: DeGroot LJ, Jameson JL, editors. Endocrinology. 6th ed. Philadelphia: Saunders/Elsevier; 2010. p. 333-57.–55. Glezer A, Bronstein MD. Approach to the patient with persistent hyperprolactinemia and negative sellar imaging. J Clin Endocrinol Metab. 2012;97(7):2211-6.), although they may occasionally be found in other conditions (11. Vilar L, Fleseriu M, Bronstein MD. Pitfalls in the diagnosis of hyperprolactinemia. Arq Bras Endocrinol Metab. 2014;58(1):9-22.,3434. Vilar L, Freitas MC, Naves LA, Casulari LA, Azevedo M, Montenegro R Jr, et al. Diagnosis and management of hyperprolactinemia: results of a Brazilian multicenter study with 1234 patients. J Endocrinol Invest. 2008;31(5):436-44.), as commented on below. In contrast, most patients with stalk dysfunction (pseudoprolactinomas), drug-induced hyperprolactinemia or systemic diseases present with PRL levels < 100 ng/mL (11. Vilar L, Fleseriu M, Bronstein MD. Pitfalls in the diagnosis of hyperprolactinemia. Arq Bras Endocrinol Metab. 2014;58(1):9-22.,44. Melmed S, Casanueva FF, Hoffman AR, Kleinberg DL, Montori VM, Schlechte JA, et al. Diagnosis and treatment of hyperprolactinemia: An Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(2):273-88.,3434. Vilar L, Freitas MC, Naves LA, Casulari LA, Azevedo M, Montenegro R Jr, et al. Diagnosis and management of hyperprolactinemia: results of a Brazilian multicenter study with 1234 patients. J Endocrinol Invest. 2008;31(5):436-44.). However, exceptions to these rules are not rare (11. Vilar L, Fleseriu M, Bronstein MD. Pitfalls in the diagnosis of hyperprolactinemia. Arq Bras Endocrinol Metab. 2014;58(1):9-22.,3434. Vilar L, Freitas MC, Naves LA, Casulari LA, Azevedo M, Montenegro R Jr, et al. Diagnosis and management of hyperprolactinemia: results of a Brazilian multicenter study with 1234 patients. J Endocrinol Invest. 2008;31(5):436-44.).
In patients with prolactinomas, circulating PRL levels usually parallel the tumor size (11. Vilar L, Fleseriu M, Bronstein MD. Pitfalls in the diagnosis of hyperprolactinemia. Arq Bras Endocrinol Metab. 2014;58(1):9-22.,44. Melmed S, Casanueva FF, Hoffman AR, Kleinberg DL, Montori VM, Schlechte JA, et al. Diagnosis and treatment of hyperprolactinemia: An Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(2):273-88.,77. Colao A, Lombardi G. Growth hormone and prolactin excess. Lancet. 1998;352:1455-61.). Indeed, microprolactinomas (MIC) (diameter < 10 mm) usually result in PRL levels of 100-200 ng/mL, but not infrequently, they may be < 100 ng/mL, and occasionally reach 500 ng/mL or more (11. Vilar L, Fleseriu M, Bronstein MD. Pitfalls in the diagnosis of hyperprolactinemia. Arq Bras Endocrinol Metab. 2014;58(1):9-22.,3333. Molitch ME. Disorders of prolactin secretion. Endocrinol Metab Clin North Am. 2001;30(3):585-610.,3434. Vilar L, Freitas MC, Naves LA, Casulari LA, Azevedo M, Montenegro R Jr, et al. Diagnosis and management of hyperprolactinemia: results of a Brazilian multicenter study with 1234 patients. J Endocrinol Invest. 2008;31(5):436-44.). Macroprolactinomas (MACs) (diameter ≥ 10 mm) are typically associated with PRL values > 250 ng/mL (44. Melmed S, Casanueva FF, Hoffman AR, Kleinberg DL, Montori VM, Schlechte JA, et al. Diagnosis and treatment of hyperprolactinemia: An Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(2):273-88.–77. Colao A, Lombardi G. Growth hormone and prolactin excess. Lancet. 1998;352:1455-61.). In the vast majority of patients with giant prolactinomas (maximum diameter ≥ 4 cm), PRL levels will be > 1000 ng/mL (3535. Rahmanian M, Meybodi HA, Larijani B, Mohajeri-Tehrani MR. Giantprolactinoma: case report and review of literature. J Diabetes Metab Disord. 2013;12(1):3.,3636. Acharya SV, Gopal RA, Menon PS, Bandgar TR, Shah NS. Giantprolactinoma and effectiveness of medical management. Endocr Pract. 2010;16(1):42-6.). On the other hand, artificially low PRL levels may result from the so-called “hook effect”, which should be considered in all cases of large (≥ 3 cm) pituitary macroadenomas associated with normal or mildly elevated PRL levels (< 200 ng/mL) (11. Vilar L, Fleseriu M, Bronstein MD. Pitfalls in the diagnosis of hyperprolactinemia. Arq Bras Endocrinol Metab. 2014;58(1):9-22.,3737. Fleseriu M, Lee M, Pineyro MM, Skugor M, Reddy SK, Siraj ES, et al. Giant invasive pituitary prolactinoma with falsely low serum prolactin: the significance of ‘hook effect’. J Neurooncol. 2006;79(1):41-3.–3939. Frieze TW, Mong DP, Koops MK. “Hook effect” in prolactinomas: case report and review of literature. Endocr Pract. 2002;8(4):296-303.), as commented on below. Patients harboring cystic MACs may also present with mild PRL elevation (11. Vilar L, Fleseriu M, Bronstein MD. Pitfalls in the diagnosis of hyperprolactinemia. Arq Bras Endocrinol Metab. 2014;58(1):9-22.–33. Bronstein MD. Disorders of prolactin secretion and prolactinomas. In: DeGroot LJ, Jameson JL, editors. Endocrinology. 6th ed. Philadelphia: Saunders/Elsevier; 2010. p. 333-57.).
The Brazilian Multicenter Study on Hyperprolactinemia (BMSH) analyzed 1234 patients whose results are presented in Figure 1 and Table 2 (3434. Vilar L, Freitas MC, Naves LA, Casulari LA, Azevedo M, Montenegro R Jr, et al. Diagnosis and management of hyperprolactinemia: results of a Brazilian multicenter study with 1234 patients. J Endocrinol Invest. 2008;31(5):436-44.). In this study, only patients harboring prolactinomas presented with PRL values ≥ 500 ng/nL (3434. Vilar L, Freitas MC, Naves LA, Casulari LA, Azevedo M, Montenegro R Jr, et al. Diagnosis and management of hyperprolactinemia: results of a Brazilian multicenter study with 1234 patients. J Endocrinol Invest. 2008;31(5):436-44.).
PRL levels according to the etiology of the hyperprolactinemia in the Brazilian multicenter study on hyperprolactinemia (Adapted from Ref. 34).
Prolactin levels (ng/mL) according to the etiology of the hyperprolactinemia in the Brazilian Multicenter Study on Hyperprolactinemia
1.2.1 How do PRL levels behave in cases of pseudoprolactinomas?
In patients with “pseudoprolactinomas”, whose main etiology isa nonfunctioning pituitary adenoma (NFPA), hyperprolactinemia results from compression of the pituitary stalk (22. Vilar L, Naves LA, Fleseriu M. Avaliação diagnóstica da hiperprolactinemia (Diagnostic evaluation of hyperprolactinemia). In: Vilar L, editor. Endocrinologia Clínica, 6a ed. (Clinical Endocrinology, 6th ed.). Rio de Janeiro: Guanabara Koogan; 2016. p. 3-14.,4040. Randall RV, Scheithauer BW, Laws, ER Jr, Abboud CF. Pseudoprolactinomas. Trans Am Clin Climatol Assoc. 1983; 94:114-21.). In that situation, this so-called disconnection hyperprolactinemia is thought to result from loss of the inhibitory effect of dopamine on PRL secretion (1919. Romijn JA. Hyperprolactinemia and prolactinoma. Handb Clin Neurol. 2014;124:185-95.). NFPAs represent the principal differential diagnosis of macroprolactinomas, as they require distinct treatments and have a distinct natural history and prognosis (11. Vilar L, Fleseriu M, Bronstein MD. Pitfalls in the diagnosis of hyperprolactinemia. Arq Bras Endocrinol Metab. 2014;58(1):9-22.,4040. Randall RV, Scheithauer BW, Laws, ER Jr, Abboud CF. Pseudoprolactinomas. Trans Am Clin Climatol Assoc. 1983; 94:114-21.). The term pseudoprolactinomas also includes other conditions such as craniopharyngiomas, Rathke's cleft cysts, sarcoidosis, Langerhans-cell histiocytosis and metastasis (1919. Romijn JA. Hyperprolactinemia and prolactinoma. Handb Clin Neurol. 2014;124:185-95.,4040. Randall RV, Scheithauer BW, Laws, ER Jr, Abboud CF. Pseudoprolactinomas. Trans Am Clin Climatol Assoc. 1983; 94:114-21.).
Based on a large series of histologically confirmed cases (n = 226) with NFPA, serum PRL > 2000 mIU/L (> 95 ng/mL) is almost never (< 2%) encountered in these patients (4141. Karavitaki N, Thanabalasingham G, Shore HC, Trifanescu R, Ansorge O, Meston N, et al. Do the limits of serum prolactin in disconnection hyperprolactinaemia need re-definition? A study of 226 patients with histologically verified non-functioning pituitary macroadenoma. Clin Endocrinol (Oxf). 2006;65(4):524-9.). Accordingly, in a recent study, among 64 patients with immunohistochemically confirmed NFPAs, PRL levels ranged from 33 to 250 ng/mL (~80% < 100 ng/mL) (4242. Vilar L, Albuquerque JL, Vilar CF, Ferreira LA, Cardoso IRA, Inacio IS, et al. Demographic, clinical, and laboratorial features of 770 patients with hyperprolactinemia. Arch Endocrinol Metab. 2017; 61 (Suppl. 3):S11.) By contrast, in BMSH, among 82 patients with NFPA, PRL levels ranged from 28 to 490 ng/mL (< 100 ng/mL in 82%); however, not all patients had been submitted to immunohistochemical evaluation (Table 3) (3434. Vilar L, Freitas MC, Naves LA, Casulari LA, Azevedo M, Montenegro R Jr, et al. Diagnosis and management of hyperprolactinemia: results of a Brazilian multicenter study with 1234 patients. J Endocrinol Invest. 2008;31(5):436-44.).
COMMENT 3: In cases of non-functioning pituitary adenomas, hyperprolactinemia results from stalk compression, and thus, prolactin (PRL) levels are modestly elevated (< 100 ng/mL) in the great majority of cases. Values > 250 ng/mL are exceedingly rare. By contrast, in patients with macroprolactinomas, PRL levels are usually > 250 ng/mL, and not infrequently, they exceed 1000 ng/mL. However, PRL levels may be misleadingly low due to the hook effect or in patients with cystic macroprolactinomas.
1.2.2 How do PRL levels behave in cases of drug-induced hyperprolactinemia?
The most common cause of non-physiological hyperprolactinemia is the use of drugs, which act through different mechanisms: increased transcription of the PRL gene (estrogens), antagonism of the dopamine receptor (risperidone, haloperidol, metoclopramide, domperidone, sulpiride, etc.), dopamine depletion (reserpine, methyldopa), inhibition of hypothalamic dopamine production (verapamil, heroin, morphine, enkephalin analogs, etc.), inhibition of dopamine reuptake (tricyclic antidepressants, cocaine, amphetamine, monoamine oxidase inhibitors), inhibition of serotonin reuptake (opiates, fenfluramine, fluoxetine, sibutramine), etc. (11. Vilar L, Fleseriu M, Bronstein MD. Pitfalls in the diagnosis of hyperprolactinemia. Arq Bras Endocrinol Metab. 2014;58(1):9-22.,22. Vilar L, Naves LA, Fleseriu M. Avaliação diagnóstica da hiperprolactinemia (Diagnostic evaluation of hyperprolactinemia). In: Vilar L, editor. Endocrinologia Clínica, 6a ed. (Clinical Endocrinology, 6th ed.). Rio de Janeiro: Guanabara Koogan; 2016. p. 3-14.,4242. Vilar L, Albuquerque JL, Vilar CF, Ferreira LA, Cardoso IRA, Inacio IS, et al. Demographic, clinical, and laboratorial features of 770 patients with hyperprolactinemia. Arch Endocrinol Metab. 2017; 61 (Suppl. 3):S11.–4747. Ajmal A, Joffe H, Nachtigall LB. Psychotropic-induced hyperprolactinemia: a clinical review. Psychosomatics. 2014;55(1):29-36) (Table 3).
In the Brazilian Multicenter Study on Hyperprolactinemia (BMSH), antidepressants and neuroleptics (in monotherapy or in combination) were the culprits in a large majority of the cases (82.2%) (3434. Vilar L, Freitas MC, Naves LA, Casulari LA, Azevedo M, Montenegro R Jr, et al. Diagnosis and management of hyperprolactinemia: results of a Brazilian multicenter study with 1234 patients. J Endocrinol Invest. 2008;31(5):436-44.). Among antipsychotics, the most frequently involved drugs were haloperidol, phenothiazines, and risperidone, while tricyclic drugs were the main representants among the antidepressants (3434. Vilar L, Freitas MC, Naves LA, Casulari LA, Azevedo M, Montenegro R Jr, et al. Diagnosis and management of hyperprolactinemia: results of a Brazilian multicenter study with 1234 patients. J Endocrinol Invest. 2008;31(5):436-44.). Other studies found the following rates of hyperprolactinemia associated with each therapeutic drug class: 31% for neuroleptics, 28% for neuroleptic-like drugs, 26% for antidepressants, 5% for H2-receptor antagonists, and 10% for other drugs (4545. Petit A, Piednoir D, Germain ML, Trenque T. Drug-induced hyperprolactinemia: a case-non-case study from the national pharmacovigilance database. Therapie. 2003;58(2):159-63.). In one group of 106 patients receiving antipsychotics, hyperprolactinemia was present in 81%, 35%, 29%, and 38% of patients taking risperidone, olanzapine, ziprasidone, and typical antipsychotics, respectively (4646. Johnsen E, Kroken RA, Abaza M, Olberg H, Jørgensen HA. Antipsychotic- induced hyperprolactinemia: a cross-sectional survey. J Clin Psychopharmacol. 2008;28(6):686-90.).
The newer atypical antipsychotics (AAPs) are characterized by increased antipsychotic efficacy and fewer neurological and endocrine related side-effects compared to classical antipsychotic drugs (4545. Petit A, Piednoir D, Germain ML, Trenque T. Drug-induced hyperprolactinemia: a case-non-case study from the national pharmacovigilance database. Therapie. 2003;58(2):159-63.,4646. Johnsen E, Kroken RA, Abaza M, Olberg H, Jørgensen HA. Antipsychotic- induced hyperprolactinemia: a cross-sectional survey. J Clin Psychopharmacol. 2008;28(6):686-90.). With the exception of risperidone, amisulpride and molindone, which are often associated with high PRL levels (4545. Petit A, Piednoir D, Germain ML, Trenque T. Drug-induced hyperprolactinemia: a case-non-case study from the national pharmacovigilance database. Therapie. 2003;58(2):159-63.), most of the AAPs elicit a poor hyperprolactinemic response or no hyperprolactinemia at all (4343. Voicu V, Medvedovici A, Ranetti AE, Rădulescu FŞ. Drug-induced hypo- and hyperprolactinemia: mechanisms, clinical and therapeutic consequences. Expert Opin Drug Metab Toxicol. 2013;9(8):955-68.,4545. Petit A, Piednoir D, Germain ML, Trenque T. Drug-induced hyperprolactinemia: a case-non-case study from the national pharmacovigilance database. Therapie. 2003;58(2):159-63.,4646. Johnsen E, Kroken RA, Abaza M, Olberg H, Jørgensen HA. Antipsychotic- induced hyperprolactinemia: a cross-sectional survey. J Clin Psychopharmacol. 2008;28(6):686-90.). Furthermore, the use of drugs such as quetiapine and aripiprazole (a dopamine partial agonist) was shown to be associated with resolution of the hyperprolactinemia induced by other AAPs (4848. Kunwar AR, Megna JL. Resolution of risperidone-induced hyperprolactinemia with substitution of quetiapine. Ann Pharmacother. 2003;37(2):206-8.). Moreover, decreased PRL levels were also reported when aripiprazole was used as adjunct therapy to risperidone (4949. Shim JC, Shin JG, Kelly DL, Jung DU, Seo YS, Liu KH, et al. Adjunctive treatment with a dopamine partial agonist, aripiprazole, for antipsychotic-induced hyperprolactinemia:a placebo-controlled trial. Am J Psychiatry. 2007;164(9):1404-10.).
Antidepressants induce hyperprolactinemia in a small proportion of patients, but they rarely elevate PRL to a significant degree (4646. Johnsen E, Kroken RA, Abaza M, Olberg H, Jørgensen HA. Antipsychotic- induced hyperprolactinemia: a cross-sectional survey. J Clin Psychopharmacol. 2008;28(6):686-90.). Among 80 patients treated with fluoxetine, only 10 (12.5% developed hyperprolactinemia, with 38 ng/mL being the highest PRL level (5050. Papakostas GI, Miller KK, Petersen T, Sklarsky KG, Hilliker SE, Klibanski A, et al. Serum prolactin levels among outpatients with major depressive disorders during the acute phase of treatment with fluoxetine. J Clin Psychiatry. 2006;67(6):952-7.). Atypical antidepressants, including bupropion and mirtazapine, appear to have no effect on PRL levels (4444. La Torre D, Falorni A. Pharmacological causes of hyperprolactinemia. Ther Clin Risk Manag. 2007;3(5):929-51.,4747. Ajmal A, Joffe H, Nachtigall LB. Psychotropic-induced hyperprolactinemia: a clinical review. Psychosomatics. 2014;55(1):29-36).
Although PRL elevation is usually mild (25-100 ng/mL) in cases of drug-induced hyperprolactinemia, it is also highly variable. Indeed, metoclopramide, risperidone, and phenothiazines can lead to prolactin levels > 200 ng/mL (11. Vilar L, Fleseriu M, Bronstein MD. Pitfalls in the diagnosis of hyperprolactinemia. Arq Bras Endocrinol Metab. 2014;58(1):9-22.,4343. Voicu V, Medvedovici A, Ranetti AE, Rădulescu FŞ. Drug-induced hypo- and hyperprolactinemia: mechanisms, clinical and therapeutic consequences. Expert Opin Drug Metab Toxicol. 2013;9(8):955-68.–4646. Johnsen E, Kroken RA, Abaza M, Olberg H, Jørgensen HA. Antipsychotic- induced hyperprolactinemia: a cross-sectional survey. J Clin Psychopharmacol. 2008;28(6):686-90.). Among 180 cases enrolled in the BMSH, most (64%) presented with PRL levels < 100 ng/mL, but in 5%, they exceeded 250 ng/mL (range, 28-380; mean, 105.1 ± 73.2) (Table 3) (3434. Vilar L, Freitas MC, Naves LA, Casulari LA, Azevedo M, Montenegro R Jr, et al. Diagnosis and management of hyperprolactinemia: results of a Brazilian multicenter study with 1234 patients. J Endocrinol Invest. 2008;31(5):436-44.). Interestingly, PRL levels of 720 ng/mL were recently reported in a young lady who had been treated with domperidone for 3 months. Following domperidone discontinuation, PRL fell to the normal range (5151. Vilar L, Lyra R, Albuquerque JL, Lyra R, Vilar CF, Diniz ET, et al. Very high prolactin levels associated to chronic therapy with domperidone. Arch Endocrinol Metab. 2016;60 (Suppl. 3):S22.).
COMMENT 4: Although drug-induced hyperprolactinemia is usually associated with PRL levels < 100 ng/mL, they are largely variable and may overlap those found in patients with prolactinomas.
1.2.3 How do the hook effect and linearity issues in PRL assays impact our practice?
Immunometric assays have greatly improved the sensitivity of PRL and other hormone measurements. They are usually performed through capture antibodies that are immobilized in a solid phase, and a second antibodyis labeled a signal generator. These antibodies bind to different epitopes of the PRL to be quantified, thus forming a “sandwich” test using either a fluorescent or chemiluminescent marker. The relative antigen-to-antibody proportion influences its interaction and may compromise the appropriate formation of the immunocomplexes. Thus, extremely high concentrations of PRL can simultaneously saturate both the capture and the labeled antibody when only a few PRL molecules are actually bound in a sandwich complex to be quantified by the test. In that situation, most of the PRL molecules are bound to just one antibody instead and are subsequently washed away (Figure 2). Therefore, falsely low results are reported, and the correct result for the PRL concentration is much higher than reported. This artifact is called a high-dose hook effect, also known as the prozone phenomenon, and the reported results are usually within or, more often, slightly above the manufacturer's reference range (5252. St-Jean E, Blain F, Comtois R. High prolactin levels may be missed by immunoradiometric assay in patients with macroprolactinomas. Clin Endocrinol (Oxf). 1996;44(3):305-9.,5353. Stenman UH. Pitfalls in hormone determinations. Best Pract Res Clin Endocrinol Metab. 2013;27(6):743-4.).
Schematic depiction of “hook effect.” Left, Extremely high antigen concentrations saturate both capture and signal antibodies and prevent “sandwich” formation. Right, When liquid phase is discarded, most of the antigen is lost with the signal antibody; thus, antigen concentration is measured as low (Adapted from Ref. 39).
The hook effect differs between the different assay systems used in clinical practice. Dilution of the sample at 1:100 is the test of choice to unmask this hook effect (11. Vilar L, Fleseriu M, Bronstein MD. Pitfalls in the diagnosis of hyperprolactinemia. Arq Bras Endocrinol Metab. 2014;58(1):9-22.,44. Melmed S, Casanueva FF, Hoffman AR, Kleinberg DL, Montori VM, Schlechte JA, et al. Diagnosis and treatment of hyperprolactinemia: An Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(2):273-88.). Indeed, this step will result in a dramatic rise in PRL levels if the patient has a macroprolactinoma, remaining low in cases of non-functioning pituitary adenomas (11. Vilar L, Fleseriu M, Bronstein MD. Pitfalls in the diagnosis of hyperprolactinemia. Arq Bras Endocrinol Metab. 2014;58(1):9-22.,44. Melmed S, Casanueva FF, Hoffman AR, Kleinberg DL, Montori VM, Schlechte JA, et al. Diagnosis and treatment of hyperprolactinemia: An Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(2):273-88.,3737. Fleseriu M, Lee M, Pineyro MM, Skugor M, Reddy SK, Siraj ES, et al. Giant invasive pituitary prolactinoma with falsely low serum prolactin: the significance of ‘hook effect’. J Neurooncol. 2006;79(1):41-3.,3838. Barkan AL, Chandler WF. Giant pituitary prolactinoma with falsely low serum prolactin: the pitfall of the “high-dose hook effect”: case report. Neurosurgery. 1998;42(4):913-5.).
Physicians should keep in mind that laboratories cannot dilute all samples on a routine basis to rule out the hook effect. Thus, it is extremely important that they are aware of the phenomenon so they do not forget to order dilution for all PRL samples suspected to be overconcentrated. In clinical practice, this means that dilutions must be ordered for PRL measurements in all patients with macroadenomas ³ 3 cm and initial PRL levels < 200 ng/mL, even if the PRL levels are normal (11. Vilar L, Fleseriu M, Bronstein MD. Pitfalls in the diagnosis of hyperprolactinemia. Arq Bras Endocrinol Metab. 2014;58(1):9-22.,22. Vilar L, Naves LA, Fleseriu M. Avaliação diagnóstica da hiperprolactinemia (Diagnostic evaluation of hyperprolactinemia). In: Vilar L, editor. Endocrinologia Clínica, 6a ed. (Clinical Endocrinology, 6th ed.). Rio de Janeiro: Guanabara Koogan; 2016. p. 3-14.,3838. Barkan AL, Chandler WF. Giant pituitary prolactinoma with falsely low serum prolactin: the pitfall of the “high-dose hook effect”: case report. Neurosurgery. 1998;42(4):913-5.). However, it is relevant to mention that dilutions ordered by physicians are performed at the discretion of laboratory workers. This means that, according to the reportable reference range of an assay and its reported levels of hook effect by the respective manufacturer, dilutions may occasionally start at 1:10. For instance, considering an assay in which, according to its manufacturer, the hook effect is not supposed to be induced up to a PRL value of 20,680 ng/mL, this theoretically means that a starting dilution of 1:10 should be sufficient enough to detect the phenomenon, as it is very improbable to have a PRL value higher than 200,680 ng/mL (5353. Stenman UH. Pitfalls in hormone determinations. Best Pract Res Clin Endocrinol Metab. 2013;27(6):743-4.). In this regard, it is noteworthy that unnecessary dilutions usually cause a loss of accuracy in measurements. Fortunately, with the newer assays, extremely high levels of PRL are usually necessary to hook the assay, and this fact has dramatically decreased the incidence of this phenomenon (5353. Stenman UH. Pitfalls in hormone determinations. Best Pract Res Clin Endocrinol Metab. 2013;27(6):743-4.,5454. do Carmo Dias Gontijo M, de Souza Vasconcellos L, Ribeiro-Oliveira A Jr. Hook effect and linear range in prolactin assays: distinct confounding entities. Pituitary. 2016;19(4):458-9.).
Interestingly, the hook effect has often been confused with assay linearity problems by clinicians (5454. do Carmo Dias Gontijo M, de Souza Vasconcellos L, Ribeiro-Oliveira A Jr. Hook effect and linear range in prolactin assays: distinct confounding entities. Pituitary. 2016;19(4):458-9.). In a given assay with a reportable PRL ranging from 0.25-200 ng/mL according to the manufacturer, PRL samples coming from patients with untreated macroprolactinomas may often fall out of this reportable range, even when an automatic dilution of 1:10 is superimposed. In this case, it is common that the laboratory releases a result of “> 2000 ng/mL”, and it seems that they are not aware of the importance of exact quantification of the result. Therefore, clinicians must refrain from starting treatment until the laboratory re-assays the sample at further dilutions, even if it has to be performed manually, and up to the point that the linear range of the assay is reached. Otherwise, the observed effect of a treatment may be misled due to an inexact reported measurement (5353. Stenman UH. Pitfalls in hormone determinations. Best Pract Res Clin Endocrinol Metab. 2013;27(6):743-4.,5454. do Carmo Dias Gontijo M, de Souza Vasconcellos L, Ribeiro-Oliveira A Jr. Hook effect and linear range in prolactin assays: distinct confounding entities. Pituitary. 2016;19(4):458-9.).
COMMENT 5: The hook effect should be considered in every patient presenting with a large (≥ 3 cm) pituitary macroadenoma and prolactin levels within the normal range or only modestly elevated.
1.3 Macroprolactinemia screening: routinely or just in asymptomatic individuals?
Macroprolactinemia is a condition where more than 60% of circulating PRL is made up of macroprolactin (11. Vilar L, Fleseriu M, Bronstein MD. Pitfalls in the diagnosis of hyperprolactinemia. Arq Bras Endocrinol Metab. 2014;58(1):9-22.,2929. Kasum M, Oreskovic S, Zec I, Jezek D, Tomic V, Gall V, et al. Macroprolactinemia: new insights in hyperprolactinemia. Biochem Med (Zagreb). 2012;22(2):171-9.). In most of the in vitro studies, macroprolactin was shown to display low biological activity (2828. Thirunavakkarasu K, Dutta P, Sridhar S, Dhaliwal L, Prashad GR, Gainder S, et al. Macroprolactinemia in hyperprolactinemic infertile women. Endocrine. 2013;44(3):750-5.,2929. Kasum M, Oreskovic S, Zec I, Jezek D, Tomic V, Gall V, et al. Macroprolactinemia: new insights in hyperprolactinemia. Biochem Med (Zagreb). 2012;22(2):171-9.,5555. Glezer A, Soares CR, Vieira JG, Giannella-Neto D, Ribela MT, Goffin V, et al. Human macroprolactin displays low biological activity via its homologous receptor in a new sensitive bioassay. J Clin Endocrinol Metab. 2006;91(3):1048-55.). This is corroborated by the finding that in most series with macroprolactinemia, individuals are pauci- or asymptomatic (5656. Gibney J, Smith TP, McKenna TJ. The impact on clinical practice of routine screening for macroprolactin. J Clin Endocrinol Metab. 2005;90(7):3927-32.,5757. Leite V, Cosby H, Sobrinho LG, Fresnoza MA, Santos MA, Friesen HG. Characterization of big, big prolactin in patients with hyperprolactinaemia. Clin Endocrinol (Oxf). 1992;37(4):365-72.), with no need to perform sellar imaging (5858. Hauache OM, Rocha AJ, Maia AC Jr, Maciel RM, Vieira JG. Screening for macroprolactinaemia and pituitary imaging studies. Clin Endocrinol (Oxf). 2002;57(3):327-31.) or specific treatments (5858. Hauache OM, Rocha AJ, Maia AC Jr, Maciel RM, Vieira JG. Screening for macroprolactinaemia and pituitary imaging studies. Clin Endocrinol (Oxf). 2002;57(3):327-31.,5959. Bronstein MD. Editorial: is macroprolactinemia just a diagnostic pitfall? Endocrine. 2012;41(2):169-70.). Others argued that the binding of PRL to their receptor could be blocked by modification of the tertiary structure of the original molecule (6060. Hattori N, Nakayama Y, Kitagawa K, Ishihara T, Saiki Y, Inagaki C. Anti-prolactin (PRL) autoantibody-binding sites (epitopes) on PRL molecule in macroprolactinemia. J Endocrinol. 2006;190(2):287-93.).
However, there are individuals who, despite increased macroprolactin, also present with high levels of monomeric PRL, leading to “true” hyperprolactinemia with clinical symptoms and the need foran etiologic diagnosis for the proper management of hyperprolactinemia (5959. Bronstein MD. Editorial: is macroprolactinemia just a diagnostic pitfall? Endocrine. 2012;41(2):169-70.). Moreover, the presence of symptoms could result from the concomitance of macroprolactinemia with other conditions, such as polycystic ovary syndrome (6161. Hayashida SA, Marcondes JA, Soares JM Jr, Rocha MP, Barcellos CR, Kobayashi NK, et al. Evaluation of macroprolactinemia in 259 women under investigation for polycystic ovary syndrome. Clin Endocrinol (Oxf). 2014;80(4):616-8.), idiopathic galactorrhea (11. Vilar L, Fleseriu M, Bronstein MD. Pitfalls in the diagnosis of hyperprolactinemia. Arq Bras Endocrinol Metab. 2014;58(1):9-22.,22. Vilar L, Naves LA, Fleseriu M. Avaliação diagnóstica da hiperprolactinemia (Diagnostic evaluation of hyperprolactinemia). In: Vilar L, editor. Endocrinologia Clínica, 6a ed. (Clinical Endocrinology, 6th ed.). Rio de Janeiro: Guanabara Koogan; 2016. p. 3-14.), or psychogenic erectile dysfunction (6262. Guay AT, Sabharwal P, Varma S, Malarkey WB. Delayed diagnosis of psychological erectile dysfunction because of the presence of macroprolactinemia. J Clin Endocrinol Metab. 1996;81(7):2512-4.).
Assaying serum PRL before and after precipitation with polyethylene glycol (PEG) is the most used method for screening macroprolactinemias due to its low cost and easy workability (6363. Vieira JGH, Tachibana TT, Obara LH, Maciel RMB. Extensive experience and validation of polyethylene glycol precipitation as a screening method for macroprolactinemia. Clin Chem. 1998;44(8):1758-9.). Theoretically, macroprolactin is precipitated with PEG, and only monomeric PRL will be recovered in the supernatant. However, some monomeric PRL also suffers precipitation, hence the need for the standardization of normal monomeric PRL values after PEG precipitation (5858. Hauache OM, Rocha AJ, Maia AC Jr, Maciel RM, Vieira JG. Screening for macroprolactinaemia and pituitary imaging studies. Clin Endocrinol (Oxf). 2002;57(3):327-31.,6060. Hattori N, Nakayama Y, Kitagawa K, Ishihara T, Saiki Y, Inagaki C. Anti-prolactin (PRL) autoantibody-binding sites (epitopes) on PRL molecule in macroprolactinemia. J Endocrinol. 2006;190(2):287-93.). Recoveries < 40% are indicative of the predominance of macroprolactin, whereas recoveries > 60% point to the diagnosis of monomeric hyperprolactinemia (5656. Gibney J, Smith TP, McKenna TJ. The impact on clinical practice of routine screening for macroprolactin. J Clin Endocrinol Metab. 2005;90(7):3927-32.,5858. Hauache OM, Rocha AJ, Maia AC Jr, Maciel RM, Vieira JG. Screening for macroprolactinaemia and pituitary imaging studies. Clin Endocrinol (Oxf). 2002;57(3):327-31.). Overall, PEG precipitation enables the correct diagnosis of macroprolactinemia in at least 80% of cases (5656. Gibney J, Smith TP, McKenna TJ. The impact on clinical practice of routine screening for macroprolactin. J Clin Endocrinol Metab. 2005;90(7):3927-32.,6363. Vieira JGH, Tachibana TT, Obara LH, Maciel RMB. Extensive experience and validation of polyethylene glycol precipitation as a screening method for macroprolactinemia. Clin Chem. 1998;44(8):1758-9.). The gold standard diagnostic test is the separation of isoforms by gel filtration, which correlates well with the PEG precipitation and is the only way of assessment when screening with PEG is inconclusive (5858. Hauache OM, Rocha AJ, Maia AC Jr, Maciel RM, Vieira JG. Screening for macroprolactinaemia and pituitary imaging studies. Clin Endocrinol (Oxf). 2002;57(3):327-31.,6363. Vieira JGH, Tachibana TT, Obara LH, Maciel RMB. Extensive experience and validation of polyethylene glycol precipitation as a screening method for macroprolactinemia. Clin Chem. 1998;44(8):1758-9.). However, it is an expensive and time-consuming method that cannot be used routinely.
It is noteworthy that different assays recognize macroprolactin differently (6464. Smith TP, Suliman AM, Fahie-Wilson MN, McKenna TJ. Gross variability in the detection of prolactin in sera containing big big prolactin (macroprolactin) by commercial immunoassays. J Clin Endocrinol Metab. 2002;87(12):5410-5.). It has been demonstrated that some of the new assays show lower cross-reactivity with macroprolactin; however, the number of samples defined as macroprolactin is still significant (6565. Vieira JGH, Tachibana TT, Ferrer CM, de Sá J, Biscolla RP, Ana Hoff AO, et al. Hyperprolactinemia: new assay more specific for the monomeric form does not eliminate screening for macroprolactin with polyethylene glycol precipitation. Arq Bras Endocrinol Metab. 2010;54(9):856-62.).
Hyperprolactinemia related to macroprolactin may be due to its lower renal clearance, longer half-life and lower capability to activate hypothalamic dopaminergic tone, which negatively regulates the secretion of pituitary prolactin (55. Glezer A, Bronstein MD. Approach to the patient with persistent hyperprolactinemia and negative sellar imaging. J Clin Endocrinol Metab. 2012;97(7):2211-6.). The frequency of macroprolactinemia in the general population was shown to be 0.2% in women from Scandinavia (6666. Bjøro T, Mørkrid L, Wergeland R, Turter A, Kvistborg A, Sand T, Torjesen P. Frequency of hyperprolactinaemia due to large molecular weight prolactin (150-170 kD PRL). Scand J Clin Lab Invest. 1995;55(2):139-47.) and 3.7% in a total of 1330 Japanese hospital workers of both genders (6767. Hattori N, Ishihara T, Saiki Y. Macroprolactinaemia: prevalence and aetiologies in a large group of hospital workers. Clin Endocrinol (Oxf). 2009;71(5):702-8.), whereas among hyperprolactinemic individuals, it ranged from 8 to 42%, with a mean of 19.6%, in 8 European series (5656. Gibney J, Smith TP, McKenna TJ. The impact on clinical practice of routine screening for macroprolactin. J Clin Endocrinol Metab. 2005;90(7):3927-32.,6868. Fahie-Wilson MN, Soule SG. Macroprolactinemia contribution to hyperprolactinemia in a district general hospital and evaluation of a screening test based on precipitation with polyethylene glycol. Ann Clin Biochem. 1997;34:252-8.–7575. Parlant-Pinet L, Harthé C, Roucher F, Morel Y, Borson-Chazot F, Raverot G, et al. Macroprolactinaemia: a biological diagnostic strategy from the study of 222 patients. Eur J Endocrinol. 2015;172(6):687-95.). The study population may explain the variation in the frequency of macroprolactinemia in hyperprolactinemic individuals. As an example, two Brazilian studies have shown frequencies of 16.5% in 115 consecutive patients with hyperprolactinemia (7676. Vilar L, Moura E, Canadas V, Gusmão A, Campos R, Leal E, et al. Prevalence of macroprolactinemia among 115 patients with hyperprolactinemia. Arq Bras Endocrinol Metab. 2007;51(1):86-91.) and 46% in 113 cases from a reference laboratory (5858. Hauache OM, Rocha AJ, Maia AC Jr, Maciel RM, Vieira JG. Screening for macroprolactinaemia and pituitary imaging studies. Clin Endocrinol (Oxf). 2002;57(3):327-31.). This high frequency of macroprolactinemia becomes an important issue in clinical practice: what is the probability with an additional assessment and treatment in an individual presenting with macroprolactinemia? Should all individuals with hyperprolactinemia be actively investigated for the presence of macroprolactinemia?
The request for serum PRL assessment occurs in two scenarios. At first, there are complaints related to hyperprolactinemia, such as galactorrhea, hypogonadism and infertility, leading to serum PRL measurement. If laboratory tests confirm the clinical suspicion of monomeric hyperprolactinemia, macroprolactinemia screening is not indicated, and it is recommended to proceed to the usual investigation of physiological, pharmacological and pathological causes of hyperprolactinemia for proper handling of the case. In the second scenario, serum PRL evaluation is requested in the absence of complaints related to hyperprolactinemia. In this situation, facing hyperprolactinemia in an asymptomatic individual, macroprolactinemia screening is always indicated. If positive and monomeric PRL levels are normal, it should guide the patient that there is no need for further investigation, follow-up or treatment, due to the benign nature of the condition. If the screening with PEG is inconclusive, one may proceed to gel-filtration chromatography, or if the latter is unavailable, be guided by the clinical picture. If the macroprolactin result is negative, investigate hyperprolactinemia as usual. A flowchart for the management of macroprolactinemic individuals is proposed in Figure 3.
Flowchart to the management of patients with macroprolactinemia (mPRL: monomeric prolactin) (Adapted from Ref. 6).
Some authors advocate routine macroprolactin screening as a cost-effective procedure (5656. Gibney J, Smith TP, McKenna TJ. The impact on clinical practice of routine screening for macroprolactin. J Clin Endocrinol Metab. 2005;90(7):3927-32.,7676. Vilar L, Moura E, Canadas V, Gusmão A, Campos R, Leal E, et al. Prevalence of macroprolactinemia among 115 patients with hyperprolactinemia. Arq Bras Endocrinol Metab. 2007;51(1):86-91.–7878. Shimatsu A, Hattori A. Macroprolactinemia: diagnostic, clinical, and pathogenic significance. Clin Dev Immunol. 2012;2012:167132.), and others allow screening to rule out macroprolactinemia and investigate other conditions that justify symptoms (7979. Leaños-Miranda A, Ramírez-Valenzuela KL, Campos-Galicia I, Chang-Verdugo R, Chinolla-Arellano LZ. Frequency of macroprolactinemia in hyperprolactinemic women presenting with menstrual irregularities, galactorrhea, and/or infertility: etiology and clinical manifestations. Int J Endocrinol. 2013;2013:478282.). In a study conducted in a Brazilian reference laboratory, there was more cost in searching for individuals with true hyperprolactinemia, but screening macroprolactinemia did not prevent investigation and inappropriate treatment, pointing to the need for the dissemination of medical knowledge about macroprolactinemia (8080. de Soárez PC, Souza SC, Vieira JG, Ferraz MB. The effect of identifying macroprolactinemia on health-care utilization and costs in patients with elevated serum prolactin levels. Value Health. 2009;12(6):930-4.). By contrast, according to some studies, the detection of macroprolactin may change the initial diagnosis in a significant proportion of patients. Indeed, in three series (7373. Donadio F, Barbieri A, Angioni R, Mantovani G, Beck-Peccoz P, Spada A, et al. Patients with macroprolactinaemia: clinical and radiological features. Eur J Clin Invest. 2007;37(7):552-7.,7474. Isik S, Berker D, Tutuncu YA, Ozuguz U, Gokay F, Erden G, et al. Clinical and radiological findings in macroprolactinemia. Endocrine. 2012;41(2):327-33.,8181. Vilar L, Gadelha PS, Rangel Filho F, Siqueira A, Viana KF, Fonseca MM, et al. Prevalence of macroprolactinemia among patients with idiopathic hyperprolactinemia. Arq Bras Endocrinol Metab. 2014;58 (Suppl 1):S17-18.), macroprolactinemia was encountered in 25 to 68.3% (mean, 42.3%) of patients with apparent idiopathic hyperprolactinemia (IH). Moreover, the diagnosis of PRL-secreting pituitary microadenoma shifted to non-secreting pituitary microadenoma in 10 of 49 patients (20%) reported by Donadio and cols. (7373. Donadio F, Barbieri A, Angioni R, Mantovani G, Beck-Peccoz P, Spada A, et al. Patients with macroprolactinaemia: clinical and radiological features. Eur J Clin Invest. 2007;37(7):552-7.). Thus, macroprolactinemia may occasionally represent a relevant cause of misdiagnosis, unnecessary investigation and inappropriate treatment (7373. Donadio F, Barbieri A, Angioni R, Mantovani G, Beck-Peccoz P, Spada A, et al. Patients with macroprolactinaemia: clinical and radiological features. Eur J Clin Invest. 2007;37(7):552-7.,7474. Isik S, Berker D, Tutuncu YA, Ozuguz U, Gokay F, Erden G, et al. Clinical and radiological findings in macroprolactinemia. Endocrine. 2012;41(2):327-33.,8181. Vilar L, Gadelha PS, Rangel Filho F, Siqueira A, Viana KF, Fonseca MM, et al. Prevalence of macroprolactinemia among patients with idiopathic hyperprolactinemia. Arq Bras Endocrinol Metab. 2014;58 (Suppl 1):S17-18.). Conversely, PRL should never be measured in asymptomatic patients, in order to avoid the unnecessary detection of macroprolactinemia cases (11. Vilar L, Fleseriu M, Bronstein MD. Pitfalls in the diagnosis of hyperprolactinemia. Arq Bras Endocrinol Metab. 2014;58(1):9-22.,55. Glezer A, Bronstein MD. Approach to the patient with persistent hyperprolactinemia and negative sellar imaging. J Clin Endocrinol Metab. 2012;97(7):2211-6.).
Concerning the natural history of macroprolactinemia, macroprolactinemic subjects usually display persistent macroprolactinemia without the development of raised free PRL (8282. Hattori N, Adachi T, Ishihara T, Shimatsu A. The natural history of macroprolactinaemia. Eur J Endocrinol. 2012;166(4):625-9.). However, during follow-up, hyperprolactinemia may develop in macroprolactinemic subjects who were initially normoprolactinemic along with an increase in anti-PRL autoantibody titers (8282. Hattori N, Adachi T, Ishihara T, Shimatsu A. The natural history of macroprolactinaemia. Eur J Endocrinol. 2012;166(4):625-9.).
Overall, symptoms related to hyperprolactinemia (galactorrhea, menstrual disorders and sexual dysfunction) have been reported in up to 45% of patients with macroprolactinemia (5656. Gibney J, Smith TP, McKenna TJ. The impact on clinical practice of routine screening for macroprolactin. J Clin Endocrinol Metab. 2005;90(7):3927-32.,5858. Hauache OM, Rocha AJ, Maia AC Jr, Maciel RM, Vieira JG. Screening for macroprolactinaemia and pituitary imaging studies. Clin Endocrinol (Oxf). 2002;57(3):327-31.,6969. Vallette-Kasic S, Morange-Ramos I, Selim A, Gunz G, Morange S, Enjalbert A, et al. Macroprolactinemia revisited: a study on 106 patients. J Clin Endocrinol Metab. 2002;87(2):581-8.,7070. Strachan MW, Teoh WL, Don-Wauchope AC, Seth J, Stoddart M, Beckett GJ. Clinical and radiological features of patients with macroprolactinaemia. Clin Endocrinol (Oxf). 2003;59(3):339-46.,7373. Donadio F, Barbieri A, Angioni R, Mantovani G, Beck-Peccoz P, Spada A, et al. Patients with macroprolactinaemia: clinical and radiological features. Eur J Clin Invest. 2007;37(7):552-7.,7474. Isik S, Berker D, Tutuncu YA, Ozuguz U, Gokay F, Erden G, et al. Clinical and radiological findings in macroprolactinemia. Endocrine. 2012;41(2):327-33.,7676. Vilar L, Moura E, Canadas V, Gusmão A, Campos R, Leal E, et al. Prevalence of macroprolactinemia among 115 patients with hyperprolactinemia. Arq Bras Endocrinol Metab. 2007;51(1):86-91.,8383. Vilar L, Naves LA, Freitas MC, Lima M, Canadas V, Albuquerque JL, et al. Clinical and laboratory features greatly overlap in patients with macroprolactinemia or monomeric hyperprolactinemia. Minerva Endocrinol. 2007;2(2):79-86.,8484. Vilar L, Vilar CF, Albuquerque JL, Gadelha PS, Thé AC, Trovão E, et al. Clinical and MRI findings among 120 patients with macroprolactinemia: results from a prospective study. Arch Endocrinol Metab. 2017;61 (Suppl. 3):S8.). As mentioned, this would mostly result from the concomitance with monomeric hyperprolactinemia (7474. Isik S, Berker D, Tutuncu YA, Ozuguz U, Gokay F, Erden G, et al. Clinical and radiological findings in macroprolactinemia. Endocrine. 2012;41(2):327-33.,7575. Parlant-Pinet L, Harthé C, Roucher F, Morel Y, Borson-Chazot F, Raverot G, et al. Macroprolactinaemia: a biological diagnostic strategy from the study of 222 patients. Eur J Endocrinol. 2015;172(6):687-95.,8585. Elenkova A, Genov N, Abadzhieva Z, Kirilov G, Vasilev V, Kalinov K, et al. Macroprolactinemia in patients with prolactinomas: prevalence and clinical significance. Exp Clin Endocrinol Diabetes. 2013;121(4):201-5.) or other disorders, such as polycystic ovary syndrome (11. Vilar L, Fleseriu M, Bronstein MD. Pitfalls in the diagnosis of hyperprolactinemia. Arq Bras Endocrinol Metab. 2014;58(1):9-22.,6161. Hayashida SA, Marcondes JA, Soares JM Jr, Rocha MP, Barcellos CR, Kobayashi NK, et al. Evaluation of macroprolactinemia in 259 women under investigation for polycystic ovary syndrome. Clin Endocrinol (Oxf). 2014;80(4):616-8.,6262. Guay AT, Sabharwal P, Varma S, Malarkey WB. Delayed diagnosis of psychological erectile dysfunction because of the presence of macroprolactinemia. J Clin Endocrinol Metab. 1996;81(7):2512-4.). Of note, the finding of both galactorrhea and menstrual disorders is rare in macroprolactinemia (8383. Vilar L, Naves LA, Freitas MC, Lima M, Canadas V, Albuquerque JL, et al. Clinical and laboratory features greatly overlap in patients with macroprolactinemia or monomeric hyperprolactinemia. Minerva Endocrinol. 2007;2(2):79-86.,8484. Vilar L, Vilar CF, Albuquerque JL, Gadelha PS, Thé AC, Trovão E, et al. Clinical and MRI findings among 120 patients with macroprolactinemia: results from a prospective study. Arch Endocrinol Metab. 2017;61 (Suppl. 3):S8.).
In most patients with macroprolactinemia, PRL levels are < 100 ng/mL, but they are highly variable: from 20-663 ng/mL (mean, 61 ± 66; < 100 ng/mL in approximately 91% of cases) (8383. Vilar L, Naves LA, Freitas MC, Lima M, Canadas V, Albuquerque JL, et al. Clinical and laboratory features greatly overlap in patients with macroprolactinemia or monomeric hyperprolactinemia. Minerva Endocrinol. 2007;2(2):79-86.), to 119.5 ± 112.9 (range, 32.5-404; < 100 mg/L in 74%) among 115 patients in the BMSH (3333. Molitch ME. Disorders of prolactin secretion. Endocrinol Metab Clin North Am. 2001;30(3):585-610.). In most studies, PRL levels were lower in macroprolactinemic patients than in those with monomeric hyperprolactinemia, but there was a great overlap between groups (3434. Vilar L, Freitas MC, Naves LA, Casulari LA, Azevedo M, Montenegro R Jr, et al. Diagnosis and management of hyperprolactinemia: results of a Brazilian multicenter study with 1234 patients. J Endocrinol Invest. 2008;31(5):436-44.,5656. Gibney J, Smith TP, McKenna TJ. The impact on clinical practice of routine screening for macroprolactin. J Clin Endocrinol Metab. 2005;90(7):3927-32.,6363. Vieira JGH, Tachibana TT, Obara LH, Maciel RMB. Extensive experience and validation of polyethylene glycol precipitation as a screening method for macroprolactinemia. Clin Chem. 1998;44(8):1758-9.,6969. Vallette-Kasic S, Morange-Ramos I, Selim A, Gunz G, Morange S, Enjalbert A, et al. Macroprolactinemia revisited: a study on 106 patients. J Clin Endocrinol Metab. 2002;87(2):581-8.,8383. Vilar L, Naves LA, Freitas MC, Lima M, Canadas V, Albuquerque JL, et al. Clinical and laboratory features greatly overlap in patients with macroprolactinemia or monomeric hyperprolactinemia. Minerva Endocrinol. 2007;2(2):79-86.). Moreover, MRI abnormalities (e.g., macroadenomas and, mostly, microadenomas or an empty sella) may be found in approximately 20-25% of macroprolactinemic patients (6969. Vallette-Kasic S, Morange-Ramos I, Selim A, Gunz G, Morange S, Enjalbert A, et al. Macroprolactinemia revisited: a study on 106 patients. J Clin Endocrinol Metab. 2002;87(2):581-8.,7070. Strachan MW, Teoh WL, Don-Wauchope AC, Seth J, Stoddart M, Beckett GJ. Clinical and radiological features of patients with macroprolactinaemia. Clin Endocrinol (Oxf). 2003;59(3):339-46.,7373. Donadio F, Barbieri A, Angioni R, Mantovani G, Beck-Peccoz P, Spada A, et al. Patients with macroprolactinaemia: clinical and radiological features. Eur J Clin Invest. 2007;37(7):552-7.,7474. Isik S, Berker D, Tutuncu YA, Ozuguz U, Gokay F, Erden G, et al. Clinical and radiological findings in macroprolactinemia. Endocrine. 2012;41(2):327-33.,8484. Vilar L, Vilar CF, Albuquerque JL, Gadelha PS, Thé AC, Trovão E, et al. Clinical and MRI findings among 120 patients with macroprolactinemia: results from a prospective study. Arch Endocrinol Metab. 2017;61 (Suppl. 3):S8.).
COMMENT 6: Macroprolactinemia is, in most cases, a laboratory diagnostic pitfall, with a mean frequency of ~20% among hyperprolactinemic subjects. Clinical, radiological and laboratory features cannot be used reliably to differentiatemonomeric hyperprolactinemia from macroprolactinemia. The screening for macroprolactin is mostly indicated for asymptomatic hyperprolactinemic patients, subjects with apparent idiopathic hyperprolactinemia, and any patient without an obvious cause for the hyperprolactinemia.
TREATMENT OF HYPERPROLACTINEMIA AND PROLACTINOMAS
The ideal treatment of hyperprolactinemia depends on its etiology and may include, for instance, L-thyroxine replacement in patients with primary hypothyroidism, dopamine agonists (DAs) for prolactinomas, and drug withdrawal in cases of drug-induced hyperprolactinemia (22. Vilar L, Naves LA, Fleseriu M. Avaliação diagnóstica da hiperprolactinemia (Diagnostic evaluation of hyperprolactinemia). In: Vilar L, editor. Endocrinologia Clínica, 6a ed. (Clinical Endocrinology, 6th ed.). Rio de Janeiro: Guanabara Koogan; 2016. p. 3-14.–77. Colao A, Lombardi G. Growth hormone and prolactin excess. Lancet. 1998;352:1455-61.). By contrast, macroprolactinemia does not need to be treated (55. Glezer A, Bronstein MD. Approach to the patient with persistent hyperprolactinemia and negative sellar imaging. J Clin Endocrinol Metab. 2012;97(7):2211-6.,2424. Hattori N. The frequency of macroprolactinemia in pregnant women and the heterogeneity of its etiologies. J Clin Endocrinol Metab. 1996;81(2):586-90.,2525. Cavaco B, Leite V, Santos MA, Arranhado E, Sobrinho LG. Some forms of big big prolactin behave as a complex of monomeric prolactin with an immunoglobulin G in patients with macroprolactinemia or prolactinoma. J Clin Endocrinol Metab. 1995;80(8):2342-6.).
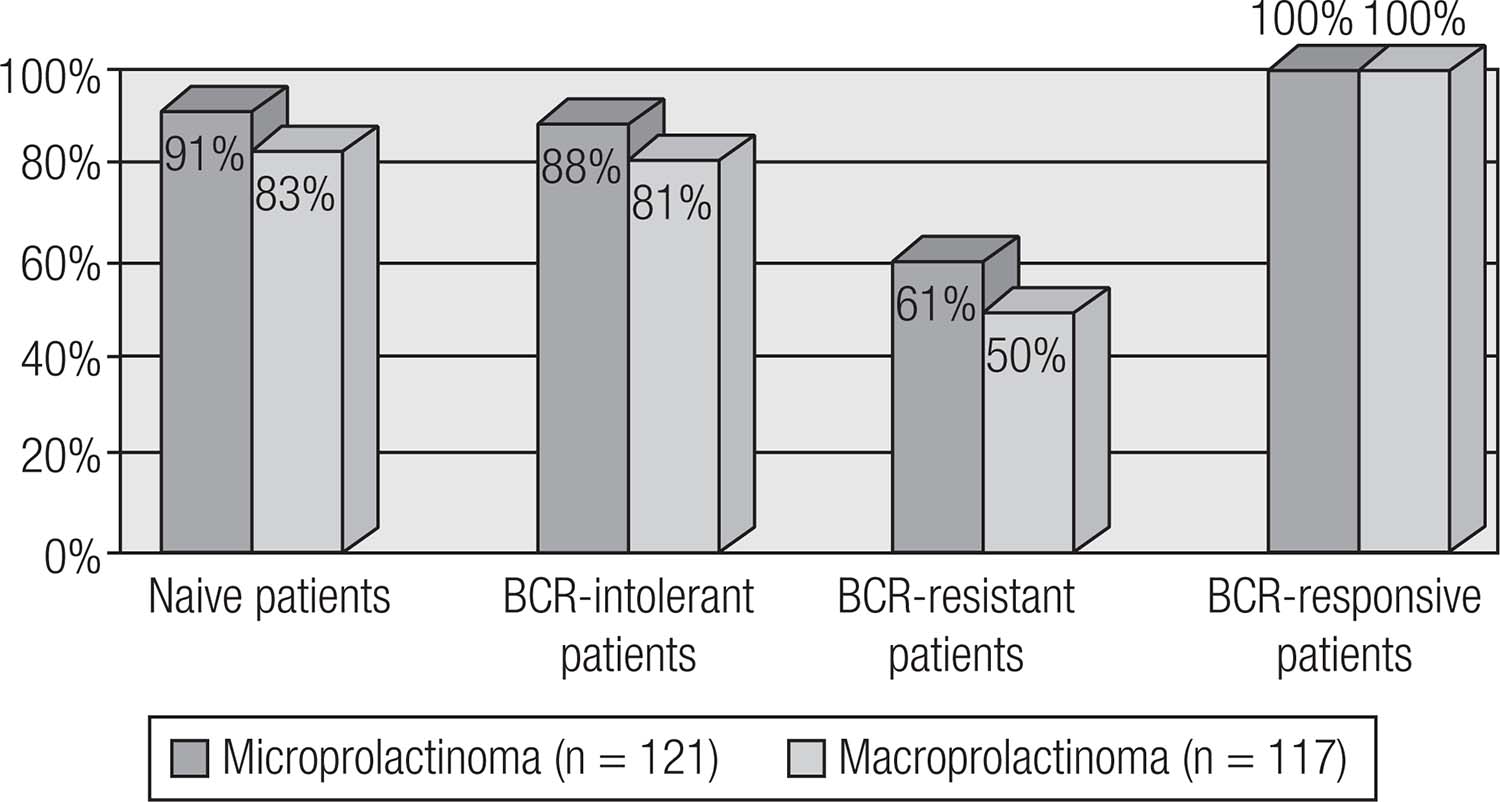
Current available therapeutic options for prolactinomas include surgery, pituitary radiation therapy and pharmacotherapy with dopamine agonists (DAs) (1919. Romijn JA. Hyperprolactinemia and prolactinoma. Handb Clin Neurol. 2014;124:185-95.). DAs are the gold standard treatment for prolactinomas, as their use controls hormonal secretion and tumor growth in approximately 80% of cases (88. Ciccarelli A, Daly AF, Beckers A. The epidemiology of prolactinomas. Pituitary 2005;8(1):3-6.). Among DAs, bromocriptine (BRC) and cabergoline (CAB), both of which are ergot derivatives, are the most commonly used worldwide. Quinagolide is available in some European countries. Cabergoline (CAB), a specific agonist of the dopamine receptor type 2 (D2R), is the first choice because of its better tolerability and greater efficacy in inducing PRL normalization and tumor shrinkage (Figures 4 and 5) (Table 4) (1919. Romijn JA. Hyperprolactinemia and prolactinoma. Handb Clin Neurol. 2014;124:185-95.,8686. dos Santos Nunes V, El Dib R, Boguszewski CL, Nogueira CR. Cabergoline versus bromocriptine in the treatment of hyperprolactinemia: a systematic review of randomized controlled trials and meta-analysis. Pituitary. 2011;14(3):259-65.–8989. Molitch ME. Pharmacologic resistance in prolactinoma patients. Pituitary. 2005;8(1):43-52.). Bromocriptine use leads to normal serum PRL levels in 80% of microprolactinomas and 70% of macroprolactinomas, whereas with CAB, this goal is achieved in 85% of patients (33. Bronstein MD. Disorders of prolactin secretion and prolactinomas. In: DeGroot LJ, Jameson JL, editors. Endocrinology. 6th ed. Philadelphia: Saunders/Elsevier; 2010. p. 333-57.,8686. dos Santos Nunes V, El Dib R, Boguszewski CL, Nogueira CR. Cabergoline versus bromocriptine in the treatment of hyperprolactinemia: a systematic review of randomized controlled trials and meta-analysis. Pituitary. 2011;14(3):259-65.–8888. Gillam MP, Molitch MP, Lombardi G, Colao A. Advances in the treatment of prolactinomas. Endocr Rev. 2006;27(5):485-534.). CAB effectiveness is higher in naïve patients, but the drug is also very effective in patients with intolerance or resistance to BCR (Figures 6 and 7) (3434. Vilar L, Freitas MC, Naves LA, Casulari LA, Azevedo M, Montenegro R Jr, et al. Diagnosis and management of hyperprolactinemia: results of a Brazilian multicenter study with 1234 patients. J Endocrinol Invest. 2008;31(5):436-44.,8888. Gillam MP, Molitch MP, Lombardi G, Colao A. Advances in the treatment of prolactinomas. Endocr Rev. 2006;27(5):485-534.). The better performance of CAB probably results from its better tolerance and higher affinity for D2R (1010. Auriema R, Pivonello R, Colao A. Prolactinomas. In: Vilar L (editor). In: Vilar L, editor. Endocrinologia Clínica, 6a ed. (Clinical Endocrinology, 6th ed.). Rio de Janeiro: Guanabara Koogan; 2016. p. 15-29.,1919. Romijn JA. Hyperprolactinemia and prolactinoma. Handb Clin Neurol. 2014;124:185-95.,8888. Gillam MP, Molitch MP, Lombardi G, Colao A. Advances in the treatment of prolactinomas. Endocr Rev. 2006;27(5):485-534.).
Comparison of cabergoline (CAB) and bromocriptine (BCR), concerning efficacy and tolerability (Adapted from Ref. 34).
Comparative efficacy of CAB and BCR in inducing tumor shrinkage in naïve patients with macroprolactinomas (Adapted from Ref. 34).
Efficacy of cabergoline on the normalization of PRL levels in 238 patients with prolactinomas. BCR: bromocriptine (Adapted from Ref. 34).
Concerning macroprolactinoma shrinkage, cabergoline is effective not only in naive patients but also in those previoiusly treated with bromocriptine (BCR) (Adapted from Ref. 34).
Comparative efficacy of cabergoline (CAB) and bromocriptine (BCR) among patients with macroprolactinomas from the Brazilian Multicenter Study on Hyperprolactinemia
2. CONTROVERSIAL ISSUES REGARDING TREATMENT
In this topic, we will cover challenging or controversial aspects related to the treatment of prolactinomas and the management of psychotropic-induced hyperprolactinemia.
2.1 How to manage the resistance to dopamine agonists?
Different arbitrary concepts have been proposed for the definition of resistance to dopamine agonist (DA) therapy (22. Vilar L, Naves LA, Fleseriu M. Avaliação diagnóstica da hiperprolactinemia (Diagnostic evaluation of hyperprolactinemia). In: Vilar L, editor. Endocrinologia Clínica, 6a ed. (Clinical Endocrinology, 6th ed.). Rio de Janeiro: Guanabara Koogan; 2016. p. 3-14.,44. Melmed S, Casanueva FF, Hoffman AR, Kleinberg DL, Montori VM, Schlechte JA, et al. Diagnosis and treatment of hyperprolactinemia: An Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(2):273-88.,8888. Gillam MP, Molitch MP, Lombardi G, Colao A. Advances in the treatment of prolactinomas. Endocr Rev. 2006;27(5):485-534.–9191. Vilar L, Burke CW. Quinagolide efficacy and tolerability in hyperprolactinaemic patients who are resistant to or intolerant of bromocriptine. Clin Endocrinol (Oxf). 1994;41(6):821-6.). Currently many experts have adopted the definition suggested by Molitch, which includes failure to normalize PRL levels and to decrease macroprolactinoma size by ≥ 50% with maximal conventional doses of medication (7.5 mg/day of bromocriptine or 2.0 mg/week of cabergoline) (8989. Molitch ME. Pharmacologic resistance in prolactinoma patients. Pituitary. 2005;8(1):43-52.,9090. Molitch ME. Management of medically refractory prolactinoma. J Neurooncol. 2014;117(3):421-8.). Bromocriptine (BCR) fails to normalize prolactin levels in approximately one-quarter of patients; cabergoline (CAB), in 10-15%. BCR fails to decrease prolactinoma size by at least 50% in approximately one-third of the patients; CAB, in 10-15% (3434. Vilar L, Freitas MC, Naves LA, Casulari LA, Azevedo M, Montenegro R Jr, et al. Diagnosis and management of hyperprolactinemia: results of a Brazilian multicenter study with 1234 patients. J Endocrinol Invest. 2008;31(5):436-44.,8888. Gillam MP, Molitch MP, Lombardi G, Colao A. Advances in the treatment of prolactinomas. Endocr Rev. 2006;27(5):485-534.–9191. Vilar L, Burke CW. Quinagolide efficacy and tolerability in hyperprolactinaemic patients who are resistant to or intolerant of bromocriptine. Clin Endocrinol (Oxf). 1994;41(6):821-6.).
Pathogenetic mechanisms
There are a number of potential mechanisms to explainresistance to DAs. Drug absorption or drug affinity to D2R are not involved (9090. Molitch ME. Management of medically refractory prolactinoma. J Neurooncol. 2014;117(3):421-8.). DA resistance is rather associated with a decrease in D2R gene transcription, resulting in a 4-fold decrease in the number of D2Rs on the cell membrane (9292. Passos VQ, Fortes MAHZ, Giannella-Neto D, Bronstein MD. Genes differentially expressed in prolactinomas responsive and resistant to dopamine agonists. Neuroendocrinology. 2009;89(2):163-70.,9393. Shimazu S, Shimatsu A, Yamada S, Inoshita N, Nagamura Y, Usui T, et al. Resistance to dopamine agonists in prolactinoma is correlated with reduction of dopamine D2 receptor long isoform mRNA levels. Eur J Endocrinol. 2012;166(3):383-90.). Moreover, there is a decrease in the G protein that couples the D2R to adenylyl cyclase, further decreasing the ability of dopamine to inhibit PRL secretion (9090. Molitch ME. Management of medically refractory prolactinoma. J Neurooncol. 2014;117(3):421-8.).
Patients who initially respond to a DA may rarely become resistant to these drugs at a later point in time (9090. Molitch ME. Management of medically refractory prolactinoma. J Neurooncol. 2014;117(3):421-8.). Most commonly, this is due to noncompliance. Rarely, there may be malignant transformation of a prolactinoma (9494. Hurel SJ, Harris PE, McNicol AM, Foster S, Kelly WF, Baylis PH. Metastatic prolactinoma: effect of octreotide, cabergoline, carboplatin and etoposide; immunocytochemical analysis of proto-oncogene expression. J Clin Endocrinol Metab. 1997;82(9):2962-5.). In some cases, the development of DA resistance is due to the concomitant use of hormone replacement therapy with estrogen or testosterone (9595. Prior JC, Cox TA, Fairholm D, Kostashuk E, Nugent R. Testosterone-related exacerbation of a prolactin-producing macroadenoma: possible role for estrogen. J Clin Endocrinol Metab. 1987;64(2):391-4.).
Treatment
The approaches for patients with resistance to DA therapy include (11. Vilar L, Fleseriu M, Bronstein MD. Pitfalls in the diagnosis of hyperprolactinemia. Arq Bras Endocrinol Metab. 2014;58(1):9-22.) switching to another DA; (22. Vilar L, Naves LA, Fleseriu M. Avaliação diagnóstica da hiperprolactinemia (Diagnostic evaluation of hyperprolactinemia). In: Vilar L, editor. Endocrinologia Clínica, 6a ed. (Clinical Endocrinology, 6th ed.). Rio de Janeiro: Guanabara Koogan; 2016. p. 3-14.) raising the dose of the DA beyond conventional doses if the patient continues to respond and tolerate; (33. Bronstein MD. Disorders of prolactin secretion and prolactinomas. In: DeGroot LJ, Jameson JL, editors. Endocrinology. 6th ed. Philadelphia: Saunders/Elsevier; 2010. p. 333-57.) surgical tumor resection; (44. Melmed S, Casanueva FF, Hoffman AR, Kleinberg DL, Montori VM, Schlechte JA, et al. Diagnosis and treatment of hyperprolactinemia: An Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(2):273-88.) radiotherapy; and (55. Glezer A, Bronstein MD. Approach to the patient with persistent hyperprolactinemia and negative sellar imaging. J Clin Endocrinol Metab. 2012;97(7):2211-6.) experimental treatments with other drugs (3434. Vilar L, Freitas MC, Naves LA, Casulari LA, Azevedo M, Montenegro R Jr, et al. Diagnosis and management of hyperprolactinemia: results of a Brazilian multicenter study with 1234 patients. J Endocrinol Invest. 2008;31(5):436-44.,8888. Gillam MP, Molitch MP, Lombardi G, Colao A. Advances in the treatment of prolactinomas. Endocr Rev. 2006;27(5):485-534.,9090. Molitch ME. Management of medically refractory prolactinoma. J Neurooncol. 2014;117(3):421-8.,9696. Olafsdottir A, Schlechte J. Management of resistant prolactinomas. Nat Clin Pract Endocrinol Metab. 2006;2(10):552-61.,9797. Colao A, DiSarno A, Sarnacchiaro F, Ferone D, DiRenzo G, Merola B, et al. Prolactinomas resistant to standard dopamine agonists respond to chronic cabergoline treatment. J Clin Endocrinol Metab. 1997;82(3):876-83.).
Switching to another DA
Most of the data regarding switching dopamine agonists in resistant patients address switching from BCR to CAB. CAB is effective in normalizing PRL levels in approximately 50-80% of patients resistant to BCR, and up to 70% respond with some tumor size change (3030. Vilar L, Czepielewski MA, Naves LA, Rollin GA, Casulari LA, Coelho CE. Substantial shrinkage of adenomas cosecreting growth hormone and prolactin with use of cabergoline therapy. Endocr Pract. 2007;13(4):396-402.,9090. Molitch ME. Management of medically refractory prolactinoma. J Neurooncol. 2014;117(3):421-8.,9696. Olafsdottir A, Schlechte J. Management of resistant prolactinomas. Nat Clin Pract Endocrinol Metab. 2006;2(10):552-61.,9797. Colao A, DiSarno A, Sarnacchiaro F, Ferone D, DiRenzo G, Merola B, et al. Prolactinomas resistant to standard dopamine agonists respond to chronic cabergoline treatment. J Clin Endocrinol Metab. 1997;82(3):876-83.). It is not clear why CAB should be so effective in patients resistant to BCR, but this may be due to cabergoline's possessing a higher affinity for dopamine binding sites, a longer time occupying the receptor, and a slower elimination from the pituitary (9090. Molitch ME. Management of medically refractory prolactinoma. J Neurooncol. 2014;117(3):421-8.,9797. Colao A, DiSarno A, Sarnacchiaro F, Ferone D, DiRenzo G, Merola B, et al. Prolactinomas resistant to standard dopamine agonists respond to chronic cabergoline treatment. J Clin Endocrinol Metab. 1997;82(3):876-83.). By contrast, the response to BCR in a patient resistant to CAB is much more rare and has only been reported twice (9090. Molitch ME. Management of medically refractory prolactinoma. J Neurooncol. 2014;117(3):421-8.,9898. Iyer P, Molitch ME. Positive prolactin response to bromocriptine in two patients with cabergoline resistant prolactinomas. Endocr Pract. 2011;17(3):e55-8.).
Raising the dose of the DA beyond conventional doses
As long as there is a continued response and no adverse effects from higher doses, there is no reason not to continue to increase the CAB dose (9090. Molitch ME. Management of medically refractory prolactinoma. J Neurooncol. 2014;117(3):421-8.). There is, however, a concern regarding the risk of cardiac valvulopathy induced by high doses of CAB (see below) (8888. Gillam MP, Molitch MP, Lombardi G, Colao A. Advances in the treatment of prolactinomas. Endocr Rev. 2006;27(5):485-534.,9090. Molitch ME. Management of medically refractory prolactinoma. J Neurooncol. 2014;117(3):421-8.).
Using an individualized, stepwise approach of dose titration of CAB, Ono and cols. (9999. Ono M, Miki N, Kawamata T, Makino R, Amano K, Seki T, et al. Prospective study of high-dose cabergoline treatment of prolactinomas in 150 patients. J Clin Endocrinol Metab. 2008; 93(12):4721-7.) documented that 25 of 26 patients (96.1%) considered to have DA resistance, achieved normalization of the PRL levels within 12 months, with a mean dose of CAB of 5.2 ± 0.6 mg per week (range 3-12 mg/week). The rate of PRL normalization gradually increased to 35, 73, and 89% at 3, 6, and 9 mg/week, respectively, finally reaching 96% at the highest dose of 12 mg/week (9999. Ono M, Miki N, Kawamata T, Makino R, Amano K, Seki T, et al. Prospective study of high-dose cabergoline treatment of prolactinomas in 150 patients. J Clin Endocrinol Metab. 2008; 93(12):4721-7.). DiSarno and cols. (100100. Di Sarno A, Landi ML, Marzullo P, Di Somma C, Pivonello R, Cerbone G, et al. The effect of quinagolide and cabergoline, two selective dopamine receptor type 2 agonists, in the treatment of prolactinomas. Clin Endocrinol (Oxf). 2000; 53(1):53-60.) found that doses > 2.0 mg/week (up to 7 mg/week) of CAB werestill unable to normalize PRL levels in 18% of patients with macroadenomas and 10% of those with microadenomas. More recently, Vilar and cols. (101101. Vilar L, Vilar C, Albuquerque JL, Gadelha P,Thé AC, Trovão E, et al. The use of increasing doses of cabergoline in the management of cabergoline-resistant prolactinomas. Endocrine Abstracts. 2017;49:EP975.) prospectively evaluated the management of 25 prolactinomas refractory to CAB 3 mg/week by progressively increasing the CAB dose as needed and tolerated, every 3 months, up to 9 mg/week. Overall, normalization of PRL levels was achieved in 18 patients (72%), as follows: in 3 (12%) patients, with up to 4 mg/week; in 9 (36%) patients, with 5 mg/week; and in 6 (24%) patients, with 6-7 mg/week. No patients benefitted from doses > 7 mg/week. CAB was well tolerated, and no significant echocardiographic valve abnormalities were detected (101101. Vilar L, Vilar C, Albuquerque JL, Gadelha P,Thé AC, Trovão E, et al. The use of increasing doses of cabergoline in the management of cabergoline-resistant prolactinomas. Endocrine Abstracts. 2017;49:EP975.).
Debulking surgery
Patients can always undergo transsphenoidal surgery if their tumor is potentially resectable and an experienced neurosurgeon is available (44. Melmed S, Casanueva FF, Hoffman AR, Kleinberg DL, Montori VM, Schlechte JA, et al. Diagnosis and treatment of hyperprolactinemia: An Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(2):273-88.,9090. Molitch ME. Management of medically refractory prolactinoma. J Neurooncol. 2014;117(3):421-8.). There is also some evidence that debulking surgery may improve the response to DA (66. Glezer A, Bronstein MD. Prolactinomas. Endocrinol Metab Clin N Am. 2015;44(1):71-8.,102102. Primeau V, Raftopoulos C, Maiter D. Outcomes of transsphenoidal surgery in prolactinomas: improvement of hormonal control in dopamine agonist-resistant patients. Eur J Endocrinol. 2012;166(5):779-86.–104104. Vrooner L, Jaffrain-Rea M-L, Petrossians P, Tamagno G, Chanson P, Vilar L, et al. Prolactinomas resistant to standard doses of cabergoline: a multicenter study of 92 patients. Eur J Endocrinol. 2012;167(5):651-62.). Of 61 patients resistant to either BCR or CAB, Hamilton and cols. (102102. Primeau V, Raftopoulos C, Maiter D. Outcomes of transsphenoidal surgery in prolactinomas: improvement of hormonal control in dopamine agonist-resistant patients. Eur J Endocrinol. 2012;166(5):779-86.) reported that surgery resulted in a normalization of PRL in 36% without dopamine agonist medication and in 15% with medication. Similarly, Primeau and cols. (102102. Primeau V, Raftopoulos C, Maiter D. Outcomes of transsphenoidal surgery in prolactinomas: improvement of hormonal control in dopamine agonist-resistant patients. Eur J Endocrinol. 2012;166(5):779-86.) found in 26 patients resistant to BCR, quinagolide or CAB that surgery resulted in a normalization of PRL in 42% without medication and in 27% with medication. In the European Multicenter Study, the rate of postoperative normalization of PRL was only 7.8% without medication and 5.3% with medication (104104. Vrooner L, Jaffrain-Rea M-L, Petrossians P, Tamagno G, Chanson P, Vilar L, et al. Prolactinomas resistant to standard doses of cabergoline: a multicenter study of 92 patients. Eur J Endocrinol. 2012;167(5):651-62.).
Radiotherapy
Among the functioning pituitary tumors, prolactinomas seem to be the less responsive to radiotherapy (see below). Indeed, although it can also be effective in controlling tumor growth, its efficacy in restoring PRL levels to normal is limited (44. Melmed S, Casanueva FF, Hoffman AR, Kleinberg DL, Montori VM, Schlechte JA, et al. Diagnosis and treatment of hyperprolactinemia: An Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(2):273-88.,8989. Molitch ME. Pharmacologic resistance in prolactinoma patients. Pituitary. 2005;8(1):43-52.,9090. Molitch ME. Management of medically refractory prolactinoma. J Neurooncol. 2014;117(3):421-8.).
Other drugs
Of note, if one is dealing with microprolactinomas resistant to DAs, hormonal replacement therapy is often all that is necessary for men and premenopausal women not willing to conceive (44. Melmed S, Casanueva FF, Hoffman AR, Kleinberg DL, Montori VM, Schlechte JA, et al. Diagnosis and treatment of hyperprolactinemia: An Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(2):273-88.,1919. Romijn JA. Hyperprolactinemia and prolactinoma. Handb Clin Neurol. 2014;124:185-95.,9090. Molitch ME. Management of medically refractory prolactinoma. J Neurooncol. 2014;117(3):421-8.).
Estrogens may cause a decrease in the effectiveness of dopamine agonists through multiple mechanisms, including direct effects on PRL gene transcription (105105. Maurer RA. Estradiol regulates the transcription of the prolactin gene. J Biol Chem. 1982;257(5):2133-6.), stimulation of mitotic activity (106106. Lloyd GM, Meares JD, Jacobi J. Effects of oestrogen and bromocriptine on in vivo secretion and mitosis in prolactin cells. Nature. 1975;255(5508):497-8.), and a decrease in the number of D2 receptors on the lactotroph cell membrane (107107. Pasqualini C, Bojda F, Kerdelhue B. Direct effect of estradiol on the number of dopamine receptors in the anterior pituitary of ovariectomized rats. Endocrinology. 1986;119(6):2484-9.). Moreover, estrogen may block apoptosis (108108. Aoki MDP, Aoki A, Maldonado CA. Sexual dimorphism of apoptosis in lactotrophs induced by bromocryptine. Histochem Cell Biol. 2001;116(3):215-22.). Thus, reducing endogenous estrogens in resistant prolactinomas through the use of SERMs in women or aromatase inhibitors in men might be an interesting experimental approach. Accordingly, a few patients with “bromocriptine resistant” invasive macroprolactinomas have been shown to respond with a decrease in tumor size and a lowering of PRL levels when tamoxifen was added (9090. Molitch ME. Management of medically refractory prolactinoma. J Neurooncol. 2014;117(3):421-8.,109109. Lamberts SWJ, Verleun T, Oosterom R. Effect of tamoxifen administration on prolactin release by invasive prolactin-secreting pituitary adenomas. Neuroendocrinology. 1982;34(5):339-42.). The use of the SERM clomiphene was also shown to be effective in the recovery of hypogonadism in prolactinoma patients who persisted with low testosterone levels despite maximal doses of DA (110110. Vilar L, Vilar CF, Aroucha PT, Albuquerque JL, Gadelha PS, Thé AC, et al. The role ofclomiphene citrate in the resolution of hypogonadism in male patients with prolactinomas under cabergoline therapy. Arch Endocrinol Metab. 2017;61 (Suppl. 3):S28.,111111. Ribeiro RS, Abucham J. Recovery of persistent hypogonadism by clomiphene in males with prolactinomas under dopamine agonist treatment. Eur J Endocrinol. 2009;161(1):163-9.). In addition, the successful use of anastrozole, an aromatase inhibitor, was recently reported in two males with DA-resistant prolactinomas (9090. Molitch ME. Management of medically refractory prolactinoma. J Neurooncol. 2014;117(3):421-8.,112112. Gillam MP, Middler S, Freed DJ, Molitch ME. The novel use of very high doses of cabergoline and a combination of testosterone and an aromatase inhibitor in the treatment of a giant prolactinoma. J Clin Endocrinol Metab. 2002;87(10):4447-51.).
Somatostatin analogs generally are not useful for PRL-secreting tumors (9090. Molitch ME. Management of medically refractory prolactinoma. J Neurooncol. 2014;117(3):421-8.), and there have been few reports on the successful combination therapy of octreotide (113113. Fusco A, Lugli F, Sacco E, Tilaro L, Bianchi A, Angelini F, et al. Efficacy of the combined cabergoline and octreotide treatment in a case of a dopamine-agonist resistant macroprolactinoma. Pituitary. 2011;14(4):351-7.) or lanreotide (114114. Vilar L, Vilar C, Albuquerque JL, Gadelha P, Thé AC, Trovão E, et al. Effect of the addition of lanreotide autogel to the treatment of an aggressive prolactinoma – a case report. Endocrine Abstracts. 2017;49:EP841.) and cabergoline in cases of DA-resistant tumors. The somatostatin receptor subtype 5 (SSTR5) is the most important with respect to the regulation of PRL secretion (115115. Jaquet P, Qouafik L, Saveanu A, Gunz G, Fina F, Dufour H, et al. Quantitative and functional expression of somatostatin receptor subtypes in human prolactinomas. J Clin Endocrinol Metab. 1999;84(9):3268-76.). Therefore, as pasireotide only has substantial activity at SSTR5 (116116. Bruns C, Lewis I, Briner U, Meno-Tetang G, Weckbecker G. SOM230: a novel somatostatin peptidomimetic with broad somatotropin release inhibiting factor (SRIF) receptor binding and a unique antisecretory profile. Eur J Endocrinol. 2002;146(5):707-16.), it would be potentially more useful than the SSTR2-agonists octreotide and lanreotide in patients with aggressive DA-resistant prolactinomas (9090. Molitch ME. Management of medically refractory prolactinoma. J Neurooncol. 2014;117(3):421-8.).
Temozolomide, an oral alkylating agent, has been successfully used in the treatment of aggressive or malignant pituitary tumors since 2006 (117117. Halevy C, Whitelaw BC. How effective is temozolomide for treating pituitary tumours and when should it be used? Pituitary.2017;20(2):261-6.,118118. Ji Y, Vogel RI, Lou E. Temozolomide treatment of pituitary carcinomas and atypical adenomas: systematic review of case reports. Neurooncol Pract. 2016;3(3):188-95.). It has also been found to be moderately successful in some very aggressive, large DA-resistant prolactinomas. Reviewing such cases, Whitelaw and cols. (119119. Whitelaw BC, Dworakowska D, Thomas NQ, Barazi S, Riordan-Eva P, King AP, et al. Temozolomide in the management of dopamine agonist-resistant prolactinomas. Clin Endocrinol (Oxf). 2012;76(6):877-86.) found that 12 of 20 (75%) resistant PRL-secreting macroadenomas responded to temozolomide. Given the toxicity of the drug, its use is generally regarded as the therapy of last resort and should only be performed after the failure of DAs, surgery and radiotherapy for tumor control (9090. Molitch ME. Management of medically refractory prolactinoma. J Neurooncol. 2014;117(3):421-8.,119119. Whitelaw BC, Dworakowska D, Thomas NQ, Barazi S, Riordan-Eva P, King AP, et al. Temozolomide in the management of dopamine agonist-resistant prolactinomas. Clin Endocrinol (Oxf). 2012;76(6):877-86.). Unfortunately, many of these very aggressive tumors escape from the suppressive effects of temozolomide after 0.5-2.5 years (9090. Molitch ME. Management of medically refractory prolactinoma. J Neurooncol. 2014;117(3):421-8.,119119. Whitelaw BC, Dworakowska D, Thomas NQ, Barazi S, Riordan-Eva P, King AP, et al. Temozolomide in the management of dopamine agonist-resistant prolactinomas. Clin Endocrinol (Oxf). 2012;76(6):877-86.).
Other treatment strategies undergoing clinical trials are the use of chimeric molecules (somatostatin analogues and dopamine D2 receptors) (22. Vilar L, Naves LA, Fleseriu M. Avaliação diagnóstica da hiperprolactinemia (Diagnostic evaluation of hyperprolactinemia). In: Vilar L, editor. Endocrinologia Clínica, 6a ed. (Clinical Endocrinology, 6th ed.). Rio de Janeiro: Guanabara Koogan; 2016. p. 3-14.,33. Bronstein MD. Disorders of prolactin secretion and prolactinomas. In: DeGroot LJ, Jameson JL, editors. Endocrinology. 6th ed. Philadelphia: Saunders/Elsevier; 2010. p. 333-57.), PRL receptor antagonists (33. Bronstein MD. Disorders of prolactin secretion and prolactinomas. In: DeGroot LJ, Jameson JL, editors. Endocrinology. 6th ed. Philadelphia: Saunders/Elsevier; 2010. p. 333-57.), and antiblastic drugs, such as mTOR and tyrosine kinase inhibitors (33. Bronstein MD. Disorders of prolactin secretion and prolactinomas. In: DeGroot LJ, Jameson JL, editors. Endocrinology. 6th ed. Philadelphia: Saunders/Elsevier; 2010. p. 333-57.,120120. Jouanneau E, Wierinckx A, Ducray F, Favrel V, Borson-Chazot F, Honnorat J, et al. New targeted therapies in pituitary carcinoma resistant to temozolomide. Pituitary. 2012;15(1):37-43.).
COMMENT 7: Patients with resistance to bromocriptine should be switched to treatment with cabergoline (CAB), which leads to prolactin normalization in at least 50% of cases. Facedwith resistance to CAB, a stepwise increase in the dose should be performed first, as long as there is a continued response and no adverse effects. This approach is successful in at least 70% of patients. In our experience, doses > 7 mg/week do not provide additional benefits. Debulking surgery may improve a response to the dopamine agonist (DA) when the medical treatment has failed. Radiation and temozolomide can be indicated to control aggressive tumor growth even under DA therapy.
2.2 Are there differences in the safety profile of DAs?
DA therapy often precipitates a broad spectrum of side effects, more frequently gastrointestinal symptoms (e.g., constipation, nausea, vomiting, etc.) but also postural hypotension, dizziness and headaches, which may range from mild to severe (3434. Vilar L, Freitas MC, Naves LA, Casulari LA, Azevedo M, Montenegro R Jr, et al. Diagnosis and management of hyperprolactinemia: results of a Brazilian multicenter study with 1234 patients. J Endocrinol Invest. 2008;31(5):436-44.,8888. Gillam MP, Molitch MP, Lombardi G, Colao A. Advances in the treatment of prolactinomas. Endocr Rev. 2006;27(5):485-534.,121121. Auriemma RS, Grasso LF, Pivonello R, Colao A. The safety of treatments for prolactinomas. Expert Opin Drug Saf. 2016;15(4):503-12.). The most common adverse events include nausea or vomiting (~35%), headache (~30%), and dizziness or vertigo (~25%) (121121. Auriemma RS, Grasso LF, Pivonello R, Colao A. The safety of treatments for prolactinomas. Expert Opin Drug Saf. 2016;15(4):503-12.). The safety profile of CAB is similar to that reported for BRC, but CAB adverse effects are generally less frequent, less severe, and of shorter duration (3434. Vilar L, Freitas MC, Naves LA, Casulari LA, Azevedo M, Montenegro R Jr, et al. Diagnosis and management of hyperprolactinemia: results of a Brazilian multicenter study with 1234 patients. J Endocrinol Invest. 2008;31(5):436-44.,8888. Gillam MP, Molitch MP, Lombardi G, Colao A. Advances in the treatment of prolactinomas. Endocr Rev. 2006;27(5):485-534.), and they resolve with dose reduction or continued use in many patients (3434. Vilar L, Freitas MC, Naves LA, Casulari LA, Azevedo M, Montenegro R Jr, et al. Diagnosis and management of hyperprolactinemia: results of a Brazilian multicenter study with 1234 patients. J Endocrinol Invest. 2008;31(5):436-44.,8888. Gillam MP, Molitch MP, Lombardi G, Colao A. Advances in the treatment of prolactinomas. Endocr Rev. 2006;27(5):485-534.,121121. Auriemma RS, Grasso LF, Pivonello R, Colao A. The safety of treatments for prolactinomas. Expert Opin Drug Saf. 2016;15(4):503-12.). The rate of treatment discontinuation due to complete intolerance is also significantly higher with BCR, compared to CAB (~12% vs ~4%) (8787. Webster J, Piscitelli G, Polli A, Ferrari CI, Ismail I, Scanlon MF. A comparison of cabergoline and bromocriptine in the treatment of hyperprolactinemic amenorrhea. Cabergoline Comparative Study Group. N Engl J Med. 1994;331(14):904-9.).
Whether CAB is associated with an increased risk of clinically relevant cardiac valvulopathy in patients with prolactinomas as in those with Parkinson's disease is still debated (see below) (1010. Auriema R, Pivonello R, Colao A. Prolactinomas. In: Vilar L (editor). In: Vilar L, editor. Endocrinologia Clínica, 6a ed. (Clinical Endocrinology, 6th ed.). Rio de Janeiro: Guanabara Koogan; 2016. p. 15-29.,121121. Auriemma RS, Grasso LF, Pivonello R, Colao A. The safety of treatments for prolactinomas. Expert Opin Drug Saf. 2016;15(4):503-12.).
2.3 Do dopamine agonists cause psychiatric disorders?
Dopamine antagonists (e.g., haloperidol, olanzapine, quetiapine, sulpiride) are classically used for the treatment of psychiatric disorders such as psychosis and schizophrenia due to the supposed dopaminergic hyperactivity that occurs in these cases (4343. Voicu V, Medvedovici A, Ranetti AE, Rădulescu FŞ. Drug-induced hypo- and hyperprolactinemia: mechanisms, clinical and therapeutic consequences. Expert Opin Drug Metab Toxicol. 2013;9(8):955-68.,4646. Johnsen E, Kroken RA, Abaza M, Olberg H, Jørgensen HA. Antipsychotic- induced hyperprolactinemia: a cross-sectional survey. J Clin Psychopharmacol. 2008;28(6):686-90.,4747. Ajmal A, Joffe H, Nachtigall LB. Psychotropic-induced hyperprolactinemia: a clinical review. Psychosomatics. 2014;55(1):29-36). DA treatment exerts the opposite effect to that of the dopamine antagonists (77. Colao A, Lombardi G. Growth hormone and prolactin excess. Lancet. 1998;352:1455-61.). These agents can rarely cause psychiatric adverse side effects, including depression, somnolence, anorexia, anxiety, insomnia, impaired concentration, nervousness, hallucinations, nightmares, psychosis, impulsive control disorders (ICDs), and mania (33. Bronstein MD. Disorders of prolactin secretion and prolactinomas. In: DeGroot LJ, Jameson JL, editors. Endocrinology. 6th ed. Philadelphia: Saunders/Elsevier; 2010. p. 333-57.,55. Glezer A, Bronstein MD. Approach to the patient with persistent hyperprolactinemia and negative sellar imaging. J Clin Endocrinol Metab. 2012;97(7):2211-6.,122122. National Library of Medicine. Daily Med Current Medication Information. 2010 Available from: http://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=31309. Accessed on: April 7, 2017.
http://dailymed.nlm.nih.gov/dailymed/dru...
–125125. Rovera C, Cremaschi L, Thanju A, Fiorentini A, Mauri MC, Serati M. Cabergoline can induce mania with psychotic features in bipolar I disorder: a case report. Asian J Psychiatr. 2016;22:94-5.). The exact incidence of these side effects in DA-treated patients is not known but is thought to range from less than 1% to 3% (122122. National Library of Medicine. Daily Med Current Medication Information. 2010 Available from: http://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=31309. Accessed on: April 7, 2017.
http://dailymed.nlm.nih.gov/dailymed/dru...
,123123. Yüksel RN, Elyas Kaya Z, Dilbaz N, Cingi Yirün M. Cabergoline-induced manic episode: case report. Ther Adv Psychopharmacol. 2016;6(3):229-23.).
ICDs are characterized by difficulties in resisting urges to engage in behaviors that are excessive and/or ultimately harmful to oneself or others and can include gambling, kleptomania, intermittent explosive disorder, compulsive sexual behavior and compulsive buying (126126. Bilal L, Ching C. Cabergoline-induced psychosis in a patient with undiagnosed depression. J Neuropsychiatry Clin Neurosci. 2012;24(4):E54.).
Few reports have been published focusing on ICDs and other psychiatric disorders in prolactinoma patients treated with CAB (123123. Yüksel RN, Elyas Kaya Z, Dilbaz N, Cingi Yirün M. Cabergoline-induced manic episode: case report. Ther Adv Psychopharmacol. 2016;6(3):229-23.–133133. Almanzar S, Zapata-Vega MI, Raya JA. Dopamine agonist-induced impulse control disorders in a patient with prolactinoma. Psychosomatics. 2013;54(4):387-91.). These reports have described disorders such as a first episode of mania (123123. Yüksel RN, Elyas Kaya Z, Dilbaz N, Cingi Yirün M. Cabergoline-induced manic episode: case report. Ther Adv Psychopharmacol. 2016;6(3):229-23.,129129. Harris YT, Harris AZ, Deasis JM, Ferrando SJ, Reddy N, Young RC. Cabergoline associated with first episode mania. Psychosomatics. 2012;53(6):595-600.); mania with psychotic features in a subject with bipolar disorder after starting CAB (125125. Rovera C, Cremaschi L, Thanju A, Fiorentini A, Mauri MC, Serati M. Cabergoline can induce mania with psychotic features in bipolar I disorder: a case report. Asian J Psychiatr. 2016;22:94-5.); psychosis in a patient with undiagnosed depression (126126. Bilal L, Ching C. Cabergoline-induced psychosis in a patient with undiagnosed depression. J Neuropsychiatry Clin Neurosci. 2012;24(4):E54.); compulsive gambling one year after starting CAB (130130. Davie M. Pathological gambling associated with cabergoline therapy in a patient with a pituitary prolactinoma. J Neuropsychiatry Clin Neurosci. 2007;19(4):473-4.); gambling and compulsive sexual behavior just after starting CAB (131131. Falhammar H, Yarker JY. Pathological gambling and hypersexuality in cabergoline-treated prolactinoma. Med J Aust. 2009;190(2):97.); psychosis in a patient without a previous history of psychiatric disorder three months after starting CAB (132132. Bilal L, Ching C. Cabergoline-induced psychosis in a patient with undiagnosed depression. J Neuropsychiatry Clin Neurosci. 2012;24(4):E54.); and depression and compulsive buying (133133. Almanzar S, Zapata-Vega MI, Raya JA. Dopamine agonist-induced impulse control disorders in a patient with prolactinoma. Psychosomatics. 2013;54(4):387-91.).
In 2011, Martinkova and cols. (128128. Martinkova J, Trejbalova L, Sasikova M, Benetin J, Valkovic P. Impulse control disorders associated with dopaminergic medication in patients with pituitary adenomas. Clin Neuropharmacol. 2011;34(5):179-81.) published a review reporting a 10% prevalence of ICD in patients on CAB for prolactinoma treatment. In another study, males with prolactinomas treated with DAs have been reported to be 9.9 times more likely to develop an impulse control disorder compared to their counterparts with nonfunctioning pituitary adenomas (134134. Bancos I, Nannenga MR, Bostwick JM, Silber MH, Erickson D, Nippoldt TB. Impulse control disorders in patients with dopamine agonist-treated prolactinomas and nonfunctioning pituitary adenomas: a case-control study. Clin Endocrinol (Oxf). 2014;80(6):863-8.). More recently, Barake and cols. (135135. Barake M, Evins AE, Stoeckel L, Pachas GN, Nachtigall LB, Miller KK, et al. Investigation of impulsivity in patients on dopamine agonist therapy for hyperprolactinemia: a pilot study. Pituitary. 2014;17(2):150-6.) conducted the only randomized study aiming to assess the impact of CAB on the appearance of ICDs and concluded that patients on CAB (particularly with higher dosages) were more susceptible to impulsivity disorders than those not taking the drug.
Psychiatric effects of CAB can be modulated by P-glycoprotein (P-gp). This protein, encoded by the ABCB1 gene (136136. Hoffmeyer S, Burk O, von Richter O, Arnold HP, Brockmoller J, Johne A, et al. Functional polymorphisms of the human multidrug-resistance gene: multiple sequence variations and correlation of one allele with P-glycoprotein expression and activity in vivo. Proc Natl Acad Sci U S A. 2000;97(7):3473-8.), is a membrane transporter expressed in tumor cells and in normal tissues that acts as an efflux transporter in order to transport certain substances out of the brain and protect it against harmful substances. A recent study showed that CAB is a P-gp substrate (137137. Athanasoulia AP, Sievers C, Ising M, Brockhaus AC, Yassouridis A, Stalla GK, et al. Polymorphisms of the drug transporter gene ABCB1 predict side effects of treatment with cabergoline in patients with PRL adenomas. Eur J Endocrinol. 2012;167(3):327-35.). This study demonstrated that ABCB1 gene polymorphisms (with loss of function) could explain individual differences in the occurrence of central nervous system side-effects, demonstrating a future perspective to select patients for CAB treatment based on this gene study (137137. Athanasoulia AP, Sievers C, Ising M, Brockhaus AC, Yassouridis A, Stalla GK, et al. Polymorphisms of the drug transporter gene ABCB1 predict side effects of treatment with cabergoline in patients with PRL adenomas. Eur J Endocrinol. 2012;167(3):327-35.).
COMMENT 8: In susceptible subjects, psychotic symptoms and impulsive control disorders may be triggered or aggravated by dopamine agonist (DA) therapy. It seems therefore important to exclude psychiatric disorders before prescribing DAs and to carefully follow up patients prone to or with a history of psychiatric disorders.
2.4 Risk of heart valvular disease associated with dopamine agonists – what is its relevance?
The main warning signs that cause concerns among endocrinologists about a possible association between the therapeutic use of dopamine agonists (DAs) with heart valvular disease (HVD) emerged from two articles published in 2007 involving patients with Parkinson's disease (138138. Zanettini R, Antonini A, Gatto G, Gentile R, Tesei S, Pezzoli G. Valvular heart disease and the use of dopamine agonists for Parkinson's disease. N Engl J Med. 2007;356(1):39-46.,139139. Schade R, Andersohn F, Suissa S, Haverkamp W, Garbe E. Dopamine agonists and the risk of cardiac-valve regurgitation. N Engl J Med. 2007;356(1):29-38.). In this group of patients, there is now strong evidence that treatment with high doses of CAB and pergolide (PGL) is associated with a higher risk of cardiac valvular regurgitation and moderate evidence that treatment with CAB, and the non-ergot-derived DAs pramipexole is associated with a higher risk of heart failure (140140. Tran T, Brophy JM, Suissa S, Renoux C. Risks of cardiac valve regurgitation and heart failure associated with ergot- and non-ergot-derived dopamine agonist use in patients with Parkinson's disease: asystematic review of observational studies. CNS Drugs. 2015;29(12):985-98.). The underlying mechanism involves the activation of the 5-hydroxytryptamine 2B (5-HT2B) receptors, abundantly expressed on heart valves, which induces a cascade of events involving mitogenesis, fibroblast proliferation, valve thickening and, finally, valvular dysfunction (141141. Roth BL. Drugs and valvular heart disease. N Engl J Med. 2007;356(1):6-9.,142142. Elangbam, C.S. Drug-induced valvulopathy: an update. Toxicol Pathol. 2010;38(6):837-48.). DAs bind to 5-HT2B receptors with different affinities; for instance, CAG and PGL show high affinity and act as a potent agonist, whereas bromocriptine (BRC) has a lower affinity compared to other DAs (141141. Roth BL. Drugs and valvular heart disease. N Engl J Med. 2007;356(1):6-9.,142142. Elangbam, C.S. Drug-induced valvulopathy: an update. Toxicol Pathol. 2010;38(6):837-48.). However, there have been case reports and clinical studies in Parkinson's disease reporting on an increased risk of abnormal valvular regurgitation and cardiac and non-cardiac fibrotic reactions in patients treated with BRC (143143. Serratrice J, Disdier P, Habib G, Viallet F, Weiller PJ. Fibrotic valvular heart disease subsequent to bromocriptine treatment. Cardiol Rev. 2002;10(6):334-6.,144144. Tan LC, Ng KK, Au WL, Lee RK, Chan YH, Tan NC. Bromocriptine use and the risk of valvular heart disease. Mov Disord. 2009;24(3):344-9.).
As expected, the findings in patients with Parkinson's disease brought an immediate concern about the risk of HVD in endocrine patients treated with DAs, especially CAB, despite the striking differences between these groups that must be highlighted. First, the daily and cumulative doses of DAs used in neurology (at least 3 mg of CAB per day) are approximately 10-fold higher than those used in endocrinology (rarely more than 3 mg of CAB per week) (145145. Valassi E, Klibanski A, Biller BM. Potential cardiac valve effects of dopamine agonists in hyperprolactinemia. J Clin Endocrinol Metab. 2010;95(3):1025-33.). Moreover, most studies in Parkinson's disease predominantly include men older than 60 years of agewho are usually on therapy with multiple drugs, while patients with endocrine abnormalities treated with DAs are predominantly young or middle-age women (146146. Choong CY, Abascal VM, Weyman J, Levine RA, Gentile F, Thomas JD, et al. Prevalence of valvular regurgitation by Doppler echocardiography in patients with structurally normal hearts by two-dimensional echocardiography. Am Heart J. 1989;117(3):636-42.). This is important because the prevalence of valvular regurgitation increases with age and is influenced by gender, body mass index and hypertension (146146. Choong CY, Abascal VM, Weyman J, Levine RA, Gentile F, Thomas JD, et al. Prevalence of valvular regurgitation by Doppler echocardiography in patients with structurally normal hearts by two-dimensional echocardiography. Am Heart J. 1989;117(3):636-42.,147147. Okura H, Takada Y, Yamabe A, Ozaki T, Yamagishi H, Toda I, et al. Prevalence and correlates of physiological valvular regurgitation in healthy subjects. Circ J. 2011;75(11):2699-704.). Another factor to be considered is the impact of examiner bias in the results, as shown in the study by Gu and cols. (148148. Gu H, Luck S, Carroll PV, Powrie J, Chambers J. Cardiac valve disease and low-dose dopamine agonist therapy: an artefact of reporting bias? Clin Endocrinol (Oxf). 2011;74(5):608-10.).
In patients with hyperprolactinemia, most cross-sectional studies were dedicated to investigating the role of CAB in HVD. A meta-analysis of seven observational studies with 1,398 individuals found an increased risk of mild-to-moderate tricuspid valve regurgitation in hyperprolactinemic patients taking CAB. No significant differences were observed in mitral or aortic valve regurgitation (149149. De Vecchis R, Esposito C, Ariano C. Cabergoline use and risk of fibrosis and insufficiency of cardiac valves. Meta-analysis of observational studies. Herz. 2013;38(8):868-80.). Another recent systematic review analyzed data from 21 studies and from 40 patients followed by the authors (150150. Caputo C, Prior D, Inder WJ. The need for annual echocardiography to detect cabergoline-associated valvulopathy in patients with prolactinoma: a systematic review and additional clinical data. Lancet Diabetes Endocrinol. 2015;3(11):906-13.). Utilizing the precise definition of CAB-associated valvulopathy as the triad of moderate or severe regurgitation associated with a restricted and thickened valve, clinically relevant disease was identified in only two out of 1,811 patients, resulting in a prevalence of 0.11% (150150. Caputo C, Prior D, Inder WJ. The need for annual echocardiography to detect cabergoline-associated valvulopathy in patients with prolactinoma: a systematic review and additional clinical data. Lancet Diabetes Endocrinol. 2015;3(11):906-13.). The authors of this study also prospectively assessed 40 patients with prolactinoma taking cabergoline. Cardiovascular examination before echocardiography detected an audible systolic murmur in 10% of cases (all were functional murmurs), and no clinically significant valvular lesion was shown on echocardiogram in the 90% of patients without a murmur (150150. Caputo C, Prior D, Inder WJ. The need for annual echocardiography to detect cabergoline-associated valvulopathy in patients with prolactinoma: a systematic review and additional clinical data. Lancet Diabetes Endocrinol. 2015;3(11):906-13.).
A Brazilian study by Boguszewski and cols. (151151. Boguszewski CL, dos Santos CM, Sakamoto KS, Marini LC, de Souza AM, Azevedo M. A comparison of cabergoline and bromocriptine on the risk of valvular heart disease in patients with prolactinomas. Pituitary. 2012;15(1):44-9.) was the first to include a group of prolactinoma patients treated with BRC for comparison. Notably, a higher prevalence of trace tricuspid regurgitation was observed in patients taking BRC, while more prevalent trace-to-mild tricuspid and trace mitral regurgitation and a higher mitral tent area were observed in CAB users; nevertheless, all these echocardiographic findings were not clinically significant (151151. Boguszewski CL, dos Santos CM, Sakamoto KS, Marini LC, de Souza AM, Azevedo M. A comparison of cabergoline and bromocriptine on the risk of valvular heart disease in patients with prolactinomas. Pituitary. 2012;15(1):44-9.). This observation was confirmed in a subsequent study in which an increased prevalence of subclinical HVD in patients with prolactinomas on long-term treatment with either BRC or CAB was reported (152152. Elenkova A, Shabani R, Kalinov K, Zacharieva S. Increased prevalence of subclinical cardiac valve fibrosis in patients with prolactinomas on long-term bromocriptine and cabergoline treatment. Eur J Endocrinol. 2012;167(1):17-25.).
Beyond the cross-sectional investigations, longitudinal studies have shed light on the potential damage caused by DAs to cardiac valves. Delgado and cols. (153153. Delgado V, Biermasz NR, van Thiel SW, Ewe SH, Marsan NA, Holman ER, et al. Changes in heart valve structure and function in patients treated with dopamine agonists for prolactinomas, a 2-year follow-up study. Clin Endocrinol (Oxf). 2012;77(1):99-105.) followed patients with prolactinomas treated (n = 45) or not treated (n = 29) with CAB for 2 years and found an increased prevalence of valvular calcification in CAB users that was not accompanied by a higher prevalence of valvular dysfunction. Auriemma and cols. (154154. Auriemma RS, Pivonello R, Perone Y, Grasso LF, Ferreri L, Simeoli C, et al. Safety of long-term treatment with cabergoline on cardiac valve disease in patients with prolactinomas. Eur J Endocrinol. 2013;169(3):359-66.) concluded that 5 years of CAB therapy in prolactinomas does not increase the risk of significant cardiac valve regurgitation. Similarly, 19 patients with prolactinomas were followed in Curitiba, Brazil, during 5 years of continuous treatment with DAs. Neither a significant change in valvular regurgitation grades nor symptomatic disease was observed in the follow-up (155155. Zaccaron BA, Sena RH, Souza AM, Boguszewski CL. Safety of long-term treatment with dopamine agonists on valvular heart disease in patients with prolactinomas. Arq Bras Endocrinol Metab. 2014;58(Suppl 5):S172.). Population-based studies have also confirmed the safety of CAB in endocrine patients. A nationwide cohort study carried out in Denmark did not support that hyperprolactinemia or its treatment is associated with an increased risk of clinically significant HVD (156156. Steffensen C, Maegbaek ML, Laurberg P, Andersen M, Kistorp CM, Norrelund H, et al. Heart valve disease among patients with hyperprolactinemia: a nationwide population-based cohort study. J Clin Endocrinol Metab. 2012;97(5):1629-34.). In a multi-country, nested case-control study involving data from the United Kingdom, Italy, and the Netherlands, CAB was associated with an increased risk of HVD in Parkinson's disease but not in hyperprolactinemia (157157. Trifirò G, Mokhles MM, Dieleman JP, van Soest EM, Verhamme K, Mazzaglia G, et al. Risk of cardiac valve regurgitation with dopamine agonist use in Parkinson's disease and hyperprolactinaemia: a multi-country, nested case-control study. Drug Saf. 2012;35(2):159-71.), confirming the role of high cumulative doses in the development of valvular dysfunction and the safety of long-term use of low cumulative doses.
In two studies involving 51 prolactinoma patients considered to be resistant to standard doses of CAB, no significant echocardiographic valve abnormalities were detected despite the use of doses ranging from 3 to 12 mg/week (9999. Ono M, Miki N, Kawamata T, Makino R, Amano K, Seki T, et al. Prospective study of high-dose cabergoline treatment of prolactinomas in 150 patients. J Clin Endocrinol Metab. 2008; 93(12):4721-7.,110110. Vilar L, Vilar CF, Aroucha PT, Albuquerque JL, Gadelha PS, Thé AC, et al. The role ofclomiphene citrate in the resolution of hypogonadism in male patients with prolactinomas under cabergoline therapy. Arch Endocrinol Metab. 2017;61 (Suppl. 3):S28.).
The absence of a detrimental effect of CAB therapy on cardiac valves has also been noticed in a longitudinal study in patients with acromegaly (158158. Maione L, Garcia C, Bouchachi A, Kallel N, Maison P, Salenave S, et al. No evidence of a detrimental effect of cabergoline therapy on cardiac valves in patients with acromegaly. J Clin Endocrinol Metab. 2012;97(9):E1714-E19.).
The findings of the abovementioned studies challenge the US Food and Drug Administration (FDA) advice that patients with prolactinoma treated with CAB should have an annual echocardiogram to screen for valvular heart disease. Of note, with conventional doses of cabergoline, i.e., up to 2 mg/week, usually used in patients with prolactinomas, there does not appear to be any increased risk of cardiac valve abnormalities (9090. Molitch ME. Management of medically refractory prolactinoma. J Neurooncol. 2014;117(3):421-8.,145145. Valassi E, Klibanski A, Biller BM. Potential cardiac valve effects of dopamine agonists in hyperprolactinemia. J Clin Endocrinol Metab. 2010;95(3):1025-33.,156156. Steffensen C, Maegbaek ML, Laurberg P, Andersen M, Kistorp CM, Norrelund H, et al. Heart valve disease among patients with hyperprolactinemia: a nationwide population-based cohort study. J Clin Endocrinol Metab. 2012;97(5):1629-34.). However, as it is uncertain at what dose level these valve effects become significant, some experts recommend assessing all patients receiving > 2 mg/week with an echocardiogram on a yearly basis (9090. Molitch ME. Management of medically refractory prolactinoma. J Neurooncol. 2014;117(3):421-8.). More recently, based on the findings of their systematic review, Caputo and cols. (150150. Caputo C, Prior D, Inder WJ. The need for annual echocardiography to detect cabergoline-associated valvulopathy in patients with prolactinoma: a systematic review and additional clinical data. Lancet Diabetes Endocrinol. 2015;3(11):906-13.) proposed that such patients should be screened by a clinical cardiovascular examination and that echocardiograms should be reserved for patients with an audible murmur, those treated for more than 5 years at a dose of more than 3 mg per week, or those who maintain cabergoline treatment after the age of 50 years. Nevertheless, special attention should be given in interpreting echocardiographic findings in older individuals, as highlighted by some authors (146146. Choong CY, Abascal VM, Weyman J, Levine RA, Gentile F, Thomas JD, et al. Prevalence of valvular regurgitation by Doppler echocardiography in patients with structurally normal hearts by two-dimensional echocardiography. Am Heart J. 1989;117(3):636-42.,147147. Okura H, Takada Y, Yamabe A, Ozaki T, Yamagishi H, Toda I, et al. Prevalence and correlates of physiological valvular regurgitation in healthy subjects. Circ J. 2011;75(11):2699-704.).
Subclinical valvular abnormalities detected by echocardiography are not an indication for cessation of the therapy (121121. Auriemma RS, Grasso LF, Pivonello R, Colao A. The safety of treatments for prolactinomas. Expert Opin Drug Saf. 2016;15(4):503-12.,154154. Auriemma RS, Pivonello R, Perone Y, Grasso LF, Ferreri L, Simeoli C, et al. Safety of long-term treatment with cabergoline on cardiac valve disease in patients with prolactinomas. Eur J Endocrinol. 2013;169(3):359-66.). Importantly, if valve lesions are detected or progress during follow-up, further evaluation is indicated to distinguish CAB-induced etiologies from other causes of valvulopathies. In such cases, therapy may be interrupted, reduced or maintained based on clinical judgment and discussion with the patient (121121. Auriemma RS, Grasso LF, Pivonello R, Colao A. The safety of treatments for prolactinomas. Expert Opin Drug Saf. 2016;15(4):503-12.,150150. Caputo C, Prior D, Inder WJ. The need for annual echocardiography to detect cabergoline-associated valvulopathy in patients with prolactinoma: a systematic review and additional clinical data. Lancet Diabetes Endocrinol. 2015;3(11):906-13.,154154. Auriemma RS, Pivonello R, Perone Y, Grasso LF, Ferreri L, Simeoli C, et al. Safety of long-term treatment with cabergoline on cardiac valve disease in patients with prolactinomas. Eur J Endocrinol. 2013;169(3):359-66.).
COMMENT 9: Current available data do not support major concerns about the risk of valvulopathy in patients with hyperprolactinemia or other endocrine diseases who are chronically treated with DAs in standard doses. Thus, an echocardiogram evaluation would not be recommended for patients receiving cabergoline at doses up to 2 mg/week.
2.5 Withdrawal of the dopamine agonist – Why, when, how and how often?
Why, when and how?
Although it is well known that prolactinomas respond very well to DAs, the optimal duration of treatment is still not clear (44. Melmed S, Casanueva FF, Hoffman AR, Kleinberg DL, Montori VM, Schlechte JA, et al. Diagnosis and treatment of hyperprolactinemia: An Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(2):273-88.,66. Glezer A, Bronstein MD. Prolactinomas. Endocrinol Metab Clin N Am. 2015;44(1):71-8.,1010. Auriema R, Pivonello R, Colao A. Prolactinomas. In: Vilar L (editor). In: Vilar L, editor. Endocrinologia Clínica, 6a ed. (Clinical Endocrinology, 6th ed.). Rio de Janeiro: Guanabara Koogan; 2016. p. 15-29.). Despite the need for long-term therapy, withdrawal of treatment should be considered because of the adverse effects of medical treatment, potential long-term consequences in cardiac valves, and treatment costs (44. Melmed S, Casanueva FF, Hoffman AR, Kleinberg DL, Montori VM, Schlechte JA, et al. Diagnosis and treatment of hyperprolactinemia: An Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(2):273-88.,66. Glezer A, Bronstein MD. Prolactinomas. Endocrinol Metab Clin N Am. 2015;44(1):71-8.,8888. Gillam MP, Molitch MP, Lombardi G, Colao A. Advances in the treatment of prolactinomas. Endocr Rev. 2006;27(5):485-534.).
Studies on persistent normoprolactinemia after DA withdrawal have been published since 1979 (159159. Eversmann T, Fahlbusch R, Rjosk HK, von Werder K. Persisting suppression of prolactin secretion after long-term treatment with bromocriptine in patients with prolactinomas. Acta Endocrinol (Copenh). 1979;92(3):413-27.). In 2002, a Brazilian study by Passos and cols. (160160. Passos VQ, Souza JJ, Musolino NR, Bronstein MD. Long-term follow-up of prolactinomas: normoprolactinemia after bromocriptine withdrawal. J Clin Endocrinol Metab. 2002; 87(8):3578-82.) showed a remission rate of 20.6% after BCR discontinuation. In 2003, a landmark prospective study by Colao and cols. (161161. Colao A, Di Sarno A, Cappabianca P, Di Somma C, Pivonello R, Lombardi G. Withdrawal of long-term cabergoline therapy for tumoral and nontumoral hyperprolactinemia. N Engl J Med. 2003;349(21):2023-33.) demonstrated that CAB could be successfully withdrawn in 70% of patients with a MIC and 64% of those harboring a MAC (161161. Colao A, Di Sarno A, Cappabianca P, Di Somma C, Pivonello R, Lombardi G. Withdrawal of long-term cabergoline therapy for tumoral and nontumoral hyperprolactinemia. N Engl J Med. 2003;349(21):2023-33.). An extension of this study to 8 years documented remission rates of 66 and 47%, respectively (162162. Colao A, Di Sarno A, Guerra E, Pivonello R, Cappabianca P, Caranci F, et al. Predictors of remission of hyperprolactinaemia after long-term withdrawal of cabergoline therapy. Clin Endocrinol (Oxf). 2007;67(3):426-33.). The higher success rates of this study were attributed to stricter selection criteria for CAB withdrawal, including a prolonged period of normoprolactinemia during treatment and a significant tumor size reduction (161161. Colao A, Di Sarno A, Cappabianca P, Di Somma C, Pivonello R, Lombardi G. Withdrawal of long-term cabergoline therapy for tumoral and nontumoral hyperprolactinemia. N Engl J Med. 2003;349(21):2023-33.,162162. Colao A, Di Sarno A, Guerra E, Pivonello R, Cappabianca P, Caranci F, et al. Predictors of remission of hyperprolactinaemia after long-term withdrawal of cabergoline therapy. Clin Endocrinol (Oxf). 2007;67(3):426-33.). Since 2003, several studies have evaluated the recurrence rates of hyperprolactinemia after DA withdrawal with variable results (162162. Colao A, Di Sarno A, Guerra E, Pivonello R, Cappabianca P, Caranci F, et al. Predictors of remission of hyperprolactinaemia after long-term withdrawal of cabergoline therapy. Clin Endocrinol (Oxf). 2007;67(3):426-33.–170170. Watanabe S, Akutsu H, Takano S, Yamamoto T, Ishikawa E, Suzuki H, et al. Long-term results of cabergoline therapy for macroprolactinomas and analysis of factors associated with remission after withdrawal. Clin Endocrinol (Oxf). 2017;86(2):207-13.).
In a meta-analysis of 19 studies with a total of 743 patients, the overall remission rate after the withdrawal of DA therapy was only 21%, 32% for idiopathic hyperprolactinemia, 21% for microadenoma and 16% for macroprolactinomas (171171. Dekkers OM, Lagro J, Burman P, Jørgensen JO, Romijn JA, Pereira AM. Recurrence of hyperprolactinemia after withdrawal of dopamine agonists: systematic review and meta-analysis. J Clin Endocrinol Metab. 2010; 95(1):43-51.). Furthermore, a higher rate of remission was found in studies in which CAB was used (35% in 4 studies) versus BRC (20% in 12 studies) (p = 0.07) (171171. Dekkers OM, Lagro J, Burman P, Jørgensen JO, Romijn JA, Pereira AM. Recurrence of hyperprolactinemia after withdrawal of dopamine agonists: systematic review and meta-analysis. J Clin Endocrinol Metab. 2010; 95(1):43-51.). In studies lasting over 24 months of treatment, the remission was higher (34%) in comparison to studies with a shorter treatment duration (16%) (p = 0.01) (153153. Delgado V, Biermasz NR, van Thiel SW, Ewe SH, Marsan NA, Holman ER, et al. Changes in heart valve structure and function in patients treated with dopamine agonists for prolactinomas, a 2-year follow-up study. Clin Endocrinol (Oxf). 2012;77(1):99-105.). The remission rate was also higher in studies in which agreater than 50% reduction of the tumor was achieved in all patients prior to discontinuation of therapy (171171. Dekkers OM, Lagro J, Burman P, Jørgensen JO, Romijn JA, Pereira AM. Recurrence of hyperprolactinemia after withdrawal of dopamine agonists: systematic review and meta-analysis. J Clin Endocrinol Metab. 2010; 95(1):43-51.).
The guidelines provided by the Endocrine Society in 2011 recommended that patients who have attained normoprolactinemia for at least 2 years and who have no visible tumor remnant on MRI may be candidates for a gradual DA withdrawal (44. Melmed S, Casanueva FF, Hoffman AR, Kleinberg DL, Montori VM, Schlechte JA, et al. Diagnosis and treatment of hyperprolactinemia: An Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(2):273-88.).
A meta-analysis from 2015 that only evaluated patients submitted to CAB withdrawal found that the hyperprolactinemia recurrence rate was 65% by a random effects meta-analysis (172172. Hu J, Zheng X, Zhang W, Yang H. Current drug withdrawal strategy in prolactinoma patients treated with cabergoline: a systematic review and meta-analysis. Pituitary. 2015;18(5):745-51.). In a random effects meta-regression adjusting for optimal withdrawal strategies, a CAB dose reduced to the lowest level before withdrawal was associated with treatment success (p = 0.006), whereas CAB treatment longer than 2 years showed no trend of effect (p = 0.587). Patients who received the lowest CAB dose and presented a significant reduction in tumor size before withdrawal were more likely to benefit from CAB withdrawal (p < 0.001) (172172. Hu J, Zheng X, Zhang W, Yang H. Current drug withdrawal strategy in prolactinoma patients treated with cabergoline: a systematic review and meta-analysis. Pituitary. 2015;18(5):745-51.).
In three recent studies, the recurrence rates after CAB withdrawal ranged from 27% to 54% (168168. Sala E, Bellaviti Buttoni P, Malchiodi E, Verrua E, Carosi G, et al. Recurrence of hyperprolactinemia following dopamine agonist withdrawal and possible predictive factors of recurrence in prolactinomas. J Endocrinol Invest. 2016;39(12):1377-82.–170170. Watanabe S, Akutsu H, Takano S, Yamamoto T, Ishikawa E, Suzuki H, et al. Long-term results of cabergoline therapy for macroprolactinomas and analysis of factors associated with remission after withdrawal. Clin Endocrinol (Oxf). 2017;86(2):207-13.). Among 74 patients (19 MACs and 55 MICs) treated with CAB for ≥ 3 years, recurrences occurred within 12 months in 34 (45.9%), regardless of the previous duration of CAB therapy (up to 3 yrs, 3 to 5 yrs or > 5 yrs) or initial adenoma size (168168. Sala E, Bellaviti Buttoni P, Malchiodi E, Verrua E, Carosi G, et al. Recurrence of hyperprolactinemia following dopamine agonist withdrawal and possible predictive factors of recurrence in prolactinomas. J Endocrinol Invest. 2016;39(12):1377-82.). Among 67 patients (23 MACs and 44 MICs), the overall recurrence rate was 54% (35% in cases of MIC and 64% in subjects with MAC) (169169. Dogansen SC, Selcukbiricik OS, Tanrikulu S, Yarman S. Withdrawal of dopamine agonist therapy in prolactinomas: In which patients and when? Pituitary. 2016;19(3):303-10.). A higher remission rate was found with CAB, compared to BCR (55% vs. 36%) (169169. Dogansen SC, Selcukbiricik OS, Tanrikulu S, Yarman S. Withdrawal of dopamine agonist therapy in prolactinomas: In which patients and when? Pituitary. 2016;19(3):303-10.). More recently, treatment discontinuation was evaluated in 11 patients with macroprolactinomas treated with CAB for at least 5 years (170170. Watanabe S, Akutsu H, Takano S, Yamamoto T, Ishikawa E, Suzuki H, et al. Long-term results of cabergoline therapy for macroprolactinomas and analysis of factors associated with remission after withdrawal. Clin Endocrinol (Oxf). 2017;86(2):207-13.). Recurrences of hyperprolactinemia were observed in 3 (27%) post-withdrawal patients at a median time of 3.0 (range; 2.9-11.2) months, indicating that a high percentage (73%) maintained remission for at least 12 months after CAB cessation (170170. Watanabe S, Akutsu H, Takano S, Yamamoto T, Ishikawa E, Suzuki H, et al. Long-term results of cabergoline therapy for macroprolactinomas and analysis of factors associated with remission after withdrawal. Clin Endocrinol (Oxf). 2017;86(2):207-13.).
How often?
Two independent studies (173173. Vilar L, Albuquerque JL, Gadelha PS, Rangel Filho F, Siqueira AM, da Fonseca MM, et al. Second attempt of cabergoline withdrawal in patients with prolactinomas after a failed first attempt: is it worthwhile? Front Endocrinol (Lausanne). 2015;6:11.,174174. Kwancharoen R, Auriemma RS, Yenokyan G, Wand GS, Colao A, Salvatori R. Second attempt to withdraw cabergoline in prolactinomas: a pilot study. Pituitary. 2014;17(5):451-6.) have investigated the outcome of a second attempt at CAB withdrawal in patients with recurrence of hyperprolactinemia after the first withdrawal, receiving additional CAB therapy for at least 2 years. The results of these studies have demonstrated that a second attempt at CAB withdrawal after 2 additional years of therapy may be successful in approximately 30% of patients (173173. Vilar L, Albuquerque JL, Gadelha PS, Rangel Filho F, Siqueira AM, da Fonseca MM, et al. Second attempt of cabergoline withdrawal in patients with prolactinomas after a failed first attempt: is it worthwhile? Front Endocrinol (Lausanne). 2015;6:11.,174174. Kwancharoen R, Auriemma RS, Yenokyan G, Wand GS, Colao A, Salvatori R. Second attempt to withdraw cabergoline in prolactinomas: a pilot study. Pituitary. 2014;17(5):451-6.).
What are the predictors of remission?
Several studies in the literature sought the predictors of remission after stopping DAs, but the results of these studies have been contradictory (44. Melmed S, Casanueva FF, Hoffman AR, Kleinberg DL, Montori VM, Schlechte JA, et al. Diagnosis and treatment of hyperprolactinemia: An Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(2):273-88.,66. Glezer A, Bronstein MD. Prolactinomas. Endocrinol Metab Clin N Am. 2015;44(1):71-8.,1010. Auriema R, Pivonello R, Colao A. Prolactinomas. In: Vilar L (editor). In: Vilar L, editor. Endocrinologia Clínica, 6a ed. (Clinical Endocrinology, 6th ed.). Rio de Janeiro: Guanabara Koogan; 2016. p. 15-29.,1919. Romijn JA. Hyperprolactinemia and prolactinoma. Handb Clin Neurol. 2014;124:185-95.). Overall, parameters more often associated with a successful DA withdrawal include suppressed PRL levels, the use of low doses of CAB, and lack of a visible tumor on MRI before withdrawal. However, hyperprolactinemia recurrence was observed even in patients who presented with these parameters (168168. Sala E, Bellaviti Buttoni P, Malchiodi E, Verrua E, Carosi G, et al. Recurrence of hyperprolactinemia following dopamine agonist withdrawal and possible predictive factors of recurrence in prolactinomas. J Endocrinol Invest. 2016;39(12):1377-82.–172172. Hu J, Zheng X, Zhang W, Yang H. Current drug withdrawal strategy in prolactinoma patients treated with cabergoline: a systematic review and meta-analysis. Pituitary. 2015;18(5):745-51.).
In the great majority of studies, patients harboring MACs presented a lower risk of hyperprolactinemia remission than those with MICs when treatment is discontinued (161161. Colao A, Di Sarno A, Cappabianca P, Di Somma C, Pivonello R, Lombardi G. Withdrawal of long-term cabergoline therapy for tumoral and nontumoral hyperprolactinemia. N Engl J Med. 2003;349(21):2023-33.–169169. Dogansen SC, Selcukbiricik OS, Tanrikulu S, Yarman S. Withdrawal of dopamine agonist therapy in prolactinomas: In which patients and when? Pituitary. 2016;19(3):303-10.). The same was true when CAB and BCR were compared (167167. Anagnostis P, Adamidou F, Polyzos SA, Efstathiadou Z, Karathanassi E, Kita M. Long term follow-up of patients with prolactinomas and outcome of dopamine agonist withdrawal: a single center experience. Pituitary. 2012;15(1):25-9.–169169. Dogansen SC, Selcukbiricik OS, Tanrikulu S, Yarman S. Withdrawal of dopamine agonist therapy in prolactinomas: In which patients and when? Pituitary. 2016;19(3):303-10.). In the series by Dogansen and cols. (169169. Dogansen SC, Selcukbiricik OS, Tanrikulu S, Yarman S. Withdrawal of dopamine agonist therapy in prolactinomas: In which patients and when? Pituitary. 2016;19(3):303-10.), the overall remission rate was 46% (65% in cases of MIC and 36% in subjects with MAC). A higher remission rate was experienced by patients treated with CAB vs. BCR, for both MIC (86% vs. 56%) and MAC (45% vs. 27%), respectively (169169. Dogansen SC, Selcukbiricik OS, Tanrikulu S, Yarman S. Withdrawal of dopamine agonist therapy in prolactinomas: In which patients and when? Pituitary. 2016;19(3):303-10.).
Menopause might be considered a factor that influences the reduction of PRL levels (175175. Karunakaran S, Page RC, Wass JA. The effect of the menopause on prolactin levels in patients with hyperprolactinaemia. Clin Endocrinol (Oxf). 2001;54(3):295-300.). Nevertheless, Colao and cols. (163163. Biswas M, Smith J, Jadon D, McEwan P, Rees DA, Evans LM, et al. Long-term remission following withdrawal of dopamine agonist therapy in subjects with microprolactinomas. Clin Endocrinol (Oxf). 2005;63(1):26-31.) showed that the rate of recurrence of hyperprolactinemia was similar among premenopausal and postmenopausal patients.
There are few studies showing that some patients may spontaneously normalize serum PRL concentrations after pregnancy. Auriemma and cols. (176176. Auriemma RS, Perone Y, Di Sarno A, Grasso LF, Guerra E, Gasperi M, et al. Results of a single-center observational 10-year survey study on recurrence of hyperprolactinemia after pregnancy and lactation. J Clin Endocrinol Metab. 2013;98(1):372-9.) reported that pregnancy was associated with normalization of PRL levels in 68% of patients. Moreover, breastfeeding did not increase the recurrence rate of hyperprolactinemia (177177. Domingue ME, Devuyst F, Alexopoulou O, Corvilain B, Maiter D. Outcome of prolactinoma after pregnancy and lactation: a study on 73 patients. Clin Endocrinol (Oxf). 2014;80(5):642-8.). Thus, DA withdrawal has been suggested after pregnancy to assess the possibility of hyperprolactinemia remission (176176. Auriemma RS, Perone Y, Di Sarno A, Grasso LF, Guerra E, Gasperi M, et al. Results of a single-center observational 10-year survey study on recurrence of hyperprolactinemia after pregnancy and lactation. J Clin Endocrinol Metab. 2013;98(1):372-9.,177177. Domingue ME, Devuyst F, Alexopoulou O, Corvilain B, Maiter D. Outcome of prolactinoma after pregnancy and lactation: a study on 73 patients. Clin Endocrinol (Oxf). 2014;80(5):642-8.). Previous pituitary surgery and radiotherapy also favor persistent normal PRL levels after DA withdrawal (44. Melmed S, Casanueva FF, Hoffman AR, Kleinberg DL, Montori VM, Schlechte JA, et al. Diagnosis and treatment of hyperprolactinemia: An Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(2):273-88.,66. Glezer A, Bronstein MD. Prolactinomas. Endocrinol Metab Clin N Am. 2015;44(1):71-8.,8888. Gillam MP, Molitch MP, Lombardi G, Colao A. Advances in the treatment of prolactinomas. Endocr Rev. 2006;27(5):485-534.).
How long should patients be followedup after withdrawal?
The follow-up after DA withdrawal has varied in different studies. Generally, the patients have been observed for 1 to 5 years after cessation of treatment (171171. Dekkers OM, Lagro J, Burman P, Jørgensen JO, Romijn JA, Pereira AM. Recurrence of hyperprolactinemia after withdrawal of dopamine agonists: systematic review and meta-analysis. J Clin Endocrinol Metab. 2010; 95(1):43-51.,172172. Hu J, Zheng X, Zhang W, Yang H. Current drug withdrawal strategy in prolactinoma patients treated with cabergoline: a systematic review and meta-analysis. Pituitary. 2015;18(5):745-51.). Most patients relapse within the first year, during which the patient should be subjected to a stricter monitoring of PRL levels (e.g., every 3 months) (164164. Kharlip J, Salvatori R, Yenokyan G, Wand GS. Recurrence of hyperprolactinemia after withdrawal of long-term cabergoline therapy. J Clin Endocrinol Metab. 2009;94(7):2428-36.–169169. Dogansen SC, Selcukbiricik OS, Tanrikulu S, Yarman S. Withdrawal of dopamine agonist therapy in prolactinomas: In which patients and when? Pituitary. 2016;19(3):303-10.). In virtually all studies, all recurrences were observed within 24 months of DA discontinuation (162162. Colao A, Di Sarno A, Guerra E, Pivonello R, Cappabianca P, Caranci F, et al. Predictors of remission of hyperprolactinaemia after long-term withdrawal of cabergoline therapy. Clin Endocrinol (Oxf). 2007;67(3):426-33.–173173. Vilar L, Albuquerque JL, Gadelha PS, Rangel Filho F, Siqueira AM, da Fonseca MM, et al. Second attempt of cabergoline withdrawal in patients with prolactinomas after a failed first attempt: is it worthwhile? Front Endocrinol (Lausanne). 2015;6:11.). In cases of hyperprolactinemia recurrence, the risk of tumor growth is very low (< 10%) (8888. Gillam MP, Molitch MP, Lombardi G, Colao A. Advances in the treatment of prolactinomas. Endocr Rev. 2006;27(5):485-534.,160160. Passos VQ, Souza JJ, Musolino NR, Bronstein MD. Long-term follow-up of prolactinomas: normoprolactinemia after bromocriptine withdrawal. J Clin Endocrinol Metab. 2002; 87(8):3578-82.,162162. Colao A, Di Sarno A, Guerra E, Pivonello R, Cappabianca P, Caranci F, et al. Predictors of remission of hyperprolactinaemia after long-term withdrawal of cabergoline therapy. Clin Endocrinol (Oxf). 2007;67(3):426-33.,169169. Dogansen SC, Selcukbiricik OS, Tanrikulu S, Yarman S. Withdrawal of dopamine agonist therapy in prolactinomas: In which patients and when? Pituitary. 2016;19(3):303-10.).
COMMENT 10: Following two years of treatment, cabergoline (CAB) withdrawal should be strongly considered in all patients with microprolactinomas and those with macroprolactinomas without visible tumor or harboring small tumor remnants, in the presence of PRL levels < 10 ng/mL and the use of the lowest dose of CAB. Before drug withdrawal, it is recommended to gradually taper the CAB dose.
2.6 What is the role of surgery for prolactinomas?
Transsphenoidal surgery (TSS) represents the golden standard of surgical approach for both microprolactinomas and most macroprolactinomas (8888. Gillam MP, Molitch MP, Lombardi G, Colao A. Advances in the treatment of prolactinomas. Endocr Rev. 2006;27(5):485-534.), whereas craniotomy must be reserved for tumors inaccessible via the transsphenoidal approach and currently is indicated in extremely rare cases (66. Glezer A, Bronstein MD. Prolactinomas. Endocrinol Metab Clin N Am. 2015;44(1):71-8.,8888. Gillam MP, Molitch MP, Lombardi G, Colao A. Advances in the treatment of prolactinomas. Endocr Rev. 2006;27(5):485-534.,178178. Smith TR, Hulou MM, Huang KT, Gokoglu A, Cote DJ, Woodmansee WW, Laws ER Jr. Current indications for the surgical treatment of prolactinomas. J Clin Neurosci. 2015;22(11):1785-91).
Additional indications for TSS surgery are complications of the prolactinomas, particularly the management of symptomatic apoplexy (178178. Smith TR, Hulou MM, Huang KT, Gokoglu A, Cote DJ, Woodmansee WW, Laws ER Jr. Current indications for the surgical treatment of prolactinomas. J Clin Neurosci. 2015;22(11):1785-91) and the surgical repair of CSF leaks (179179. Lund VJ. Endoscopic management of cerebrospinalfluid leaks. Am J Rhinol. 2002;16(1):17-23.). The latter can occur spontaneously due to tumor invasion of the sphenoid sinus or result from a rapid tumor shrinkage induced by DA therapy (180180. Mankia SK, Weerakkody RA, Wijesuriya S, Kandasamy N, Finucane F, Guilfoyle M, et al. Spontaneous cerebrospinalfluid rhinorrhoea as the presenting feature of an invasive macroprolactinoma. BMJ Case Rep. 2009;2009. pii: bcr12.2008.1383–182182. Lam G, Mehta V, Zada G. Spontaneous and medically induced cerebrospinal fluid leakage in the setting of pituitary adenomas: review of the literature. Neurosurg Focus. 2012;32(6):E2.). In a recent review, more than 90% of cases of cerebrospinal fluid leakage were related to the use of DA, with a mean time of 3.3 months between the start of drug administration and the diagnosis of rhinorrhea (182182. Lam G, Mehta V, Zada G. Spontaneous and medically induced cerebrospinal fluid leakage in the setting of pituitary adenomas: review of the literature. Neurosurg Focus. 2012;32(6):E2.). However, this complication can also occur during long-term treatment (183183. Bronstein MD, Musolino NR, Benabou S, Marino R Jr. Cerebrospinal fluid rhinorrhea occurring in long-term bromocriptine treatment for macroprolactinomas. Surg Neurol. 1989;32(5):346-9.). An additional strategy to CSF leakage would be the withdrawal of DA to allow tumor re-growth to stop the leak (181181. Leong KS, Foy PM, Swift AC, Atkin SL, Hadden DR, MacFarlane IA. CSF rhinorrhoea following treatment with dopamine agonists for massive invasive prolactinomas. Clin Endocrinol (Oxf). 2000;52(1):43-9.). As β2-transferrin is only found in CSF, its detection in nasal secretions is a very accurate tool for confirming the diagnosis of CSF rhinorrhea (179179. Lund VJ. Endoscopic management of cerebrospinalfluid leaks. Am J Rhinol. 2002;16(1):17-23.,184184. Anani WQ, Ojerholm E, Shurin MR. Resolving transferrin isoforms via agarose gel electrophoresis. Lab Med. 2015;46(1):26-33.).
Efficacy
The most important determinants of successful surgery for prolactinoma treatment are the experience of the neurosurgeon, moderately increased serum PRL levels (< 200 ng/mL), and tumor size and invasiveness (66. Glezer A, Bronstein MD. Prolactinomas. Endocrinol Metab Clin N Am. 2015;44(1):71-8.,121121. Auriemma RS, Grasso LF, Pivonello R, Colao A. The safety of treatments for prolactinomas. Expert Opin Drug Saf. 2016;15(4):503-12.). In a literature review involving more than 50 series, initial surgical remission, defined as the normalization of PRL levels, occurred on average in 74.7% and 34% of patients with microprolactinomas (MICs) and macroprolactinomas (MACs), with a recurrence rate of 18% and 23%, respectively (8888. Gillam MP, Molitch MP, Lombardi G, Colao A. Advances in the treatment of prolactinomas. Endocr Rev. 2006;27(5):485-534.). More recently, the analysis of surgical results from 13 published series, including at least 100 patients, has shown the control of PRL levels to be achieved in approximately 73% of 1211 microprolactinomas and 38% of 1480 macroprolactinomas (185185. Colao A. The prolactinoma. Best Pract Res Clin Endocrinol Metab. 2009;23(5):575-96.).
Notably, as shown in two recent studies (102102. Primeau V, Raftopoulos C, Maiter D. Outcomes of transsphenoidal surgery in prolactinomas: improvement of hormonal control in dopamine agonist-resistant patients. Eur J Endocrinol. 2012;166(5):779-86.,104104. Vrooner L, Jaffrain-Rea M-L, Petrossians P, Tamagno G, Chanson P, Vilar L, et al. Prolactinomas resistant to standard doses of cabergoline: a multicenter study of 92 patients. Eur J Endocrinol. 2012;167(5):651-62.), many patients with partial resistance to CAB achieve PRL normalization after surgical debulking, using a lower dose of CAB than the presurgical one.
Safety
Complications from TSS for MIC are infrequent, the mortality rate being at most 0.6% and the major morbidity rate being approximately 3.4% (8888. Gillam MP, Molitch MP, Lombardi G, Colao A. Advances in the treatment of prolactinomas. Endocr Rev. 2006;27(5):485-534.,121121. Auriemma RS, Grasso LF, Pivonello R, Colao A. The safety of treatments for prolactinomas. Expert Opin Drug Saf. 2016;15(4):503-12.). In patients with large tumors, particularly those with giant prolactinomas, the mortality rate ranges from 3.3% to 31.2% (8888. Gillam MP, Molitch MP, Lombardi G, Colao A. Advances in the treatment of prolactinomas. Endocr Rev. 2006;27(5):485-534.,121121. Auriemma RS, Grasso LF, Pivonello R, Colao A. The safety of treatments for prolactinomas. Expert Opin Drug Saf. 2016;15(4):503-12.). Visual loss, stroke/vascular injury, meningitis/abscess, oculomotor palsy and cerebrospinal fluid rhinorrhea have been reported to occur in 0.1%, 0.2%, 0.1%, 0.1% and 1.9% of cases, respectively (121121. Auriemma RS, Grasso LF, Pivonello R, Colao A. The safety of treatments for prolactinomas. Expert Opin Drug Saf. 2016;15(4):503-12.,186186. Barker 2nd FG, Klibanski A, Swearingen B. Transsphenoidal surgery for pituitary tumors in the United States, 1996–2000: mortality, morbidity, and the effects of hospital and surgeon volume. J Clin Endocrinol Metab. 2003;88(10):4709-19.–188188. Nelson Jr AT, Tucker Jr HS, Becker DP. Residual anterior pituitary function following transsphenoidal resection of pituitary macroadenomas. J Neurosurg 1984;61(3):577-80.). Transient diabetes insipidus (DI) is quite common with TSS for both micro- and macroadenomas (121121. Auriemma RS, Grasso LF, Pivonello R, Colao A. The safety of treatments for prolactinomas. Expert Opin Drug Saf. 2016;15(4):503-12.). In contrast, permanent DI is rare and has been reported to occur in approximately 1% of surgeries on MACs (121121. Auriemma RS, Grasso LF, Pivonello R, Colao A. The safety of treatments for prolactinomas. Expert Opin Drug Saf. 2016;15(4):503-12.,187187. Sudhakar N, Ray A, Vafidis JA. Complications after trans-sphenoidal surgery: our experience and a review of the literature. Br J Neurosurg. 2004;18(5):507-12.,188188. Nelson Jr AT, Tucker Jr HS, Becker DP. Residual anterior pituitary function following transsphenoidal resection of pituitary macroadenomas. J Neurosurg 1984;61(3):577-80.). Hypopituitarism, commonly found in patients with macroadenomas before surgery as a consequence of mass effects, has been reported either to further worsen or to improve after surgery (102102. Primeau V, Raftopoulos C, Maiter D. Outcomes of transsphenoidal surgery in prolactinomas: improvement of hormonal control in dopamine agonist-resistant patients. Eur J Endocrinol. 2012;166(5):779-86.–104104. Vrooner L, Jaffrain-Rea M-L, Petrossians P, Tamagno G, Chanson P, Vilar L, et al. Prolactinomas resistant to standard doses of cabergoline: a multicenter study of 92 patients. Eur J Endocrinol. 2012;167(5):651-62.).
COMMENT 11: Surgery, usually by the transsphenoidal approach, is indicated for patients with resistance or intolerance to dopamine agonists; macroprolactinomas with chiasmal compression and visual impairment without fast improvement by medical treatment; and acute tumor complications, such as symptomatic apoplexy or cerebrospinal fluid leakage.
2.7 What is the role of radiotherapy for prolactinomas?
As prolactinomas are among the most radioresistant pituitary tumors, PRL normalization occurs in only approximately one-third of patients submitted to radiotherapy (RT) (8888. Gillam MP, Molitch MP, Lombardi G, Colao A. Advances in the treatment of prolactinomas. Endocr Rev. 2006;27(5):485-534.,189189. Sheplan Olsen LJ, Robles Irizarry L, Chao ST, Weil RJ, Hamrahian AH, Hatipoglu B, Suh JH. Radiotherapy for prolactin-secreting pituitary tumors. Pituitary. 2012;15(2):135-45.,190190. Wan H, Chihiro O, Yuan S. MASEP gamma knife radiosurgery for secretory pituitary adenomas: experience in 347 consecutive cases. J Exp Clin Cancer Res. 2009;28:36.). Moreover, it is associated with a high risk of radiation-induced hypopituitarism at 10-20 years (121121. Auriemma RS, Grasso LF, Pivonello R, Colao A. The safety of treatments for prolactinomas. Expert Opin Drug Saf. 2016;15(4):503-12.,191191. Kong DS, Lee JI, Lim do H, Kim KW, Shin HJ, Nam DH, et al. The efficacy of fractionated radiotherapy and stereotactic radiosurgery for pituitary adenomas: long-term results of 125 consecutive patients treated in a single institution. Cancer. 2017;110(4):854-60.). Therefore, RT is only indicated to control tumor growth in DA-resistant cases not controlled by surgery, as well as in the very rare cases of malignant prolactinomas (44. Melmed S, Casanueva FF, Hoffman AR, Kleinberg DL, Montori VM, Schlechte JA, et al. Diagnosis and treatment of hyperprolactinemia: An Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(2):273-88.,66. Glezer A, Bronstein MD. Prolactinomas. Endocrinol Metab Clin N Am. 2015;44(1):71-8.,8888. Gillam MP, Molitch MP, Lombardi G, Colao A. Advances in the treatment of prolactinomas. Endocr Rev. 2006;27(5):485-534.).
Historically, patients have been treated with both external beam radiotherapy and stereotactic techniques (189189. Sheplan Olsen LJ, Robles Irizarry L, Chao ST, Weil RJ, Hamrahian AH, Hatipoglu B, Suh JH. Radiotherapy for prolactin-secreting pituitary tumors. Pituitary. 2012;15(2):135-45.). External beam radiotherapy, also referred to as “conventional radiotherapy” (CRT), involves the use of multiple non-overlapping beams of X-rays that intersect over the target area (191191. Kong DS, Lee JI, Lim do H, Kim KW, Shin HJ, Nam DH, et al. The efficacy of fractionated radiotherapy and stereotactic radiosurgery for pituitary adenomas: long-term results of 125 consecutive patients treated in a single institution. Cancer. 2017;110(4):854-60.). Stereotactic RT is a form of RT that uses stereotactic guidance to deliver high doses of radiation in a single fraction (radiosurgery) or in multiple fractions (191191. Kong DS, Lee JI, Lim do H, Kim KW, Shin HJ, Nam DH, et al. The efficacy of fractionated radiotherapy and stereotactic radiosurgery for pituitary adenomas: long-term results of 125 consecutive patients treated in a single institution. Cancer. 2017;110(4):854-60.).
Efficacy
A comprehensive review of the literature regarding hormonal normalization in patients with prolactinomas treated with CRT or radiosurgery concluded that the overall normalization rate of both approaches seems to be similar (34.1% for CRT vs. 31.4% for radiosurgery) (8888. Gillam MP, Molitch MP, Lombardi G, Colao A. Advances in the treatment of prolactinomas. Endocr Rev. 2006;27(5):485-534.). In the series by Wan and cols. (190190. Wan H, Chihiro O, Yuan S. MASEP gamma knife radiosurgery for secretory pituitary adenomas: experience in 347 consecutive cases. J Exp Clin Cancer Res. 2009;28:36.), of the 176 patients with prolactinomas submitted to gamma-knife radiosurgery, 23.3% achieved PRL normalization, whereas in 90.3%, the tumor volume decreased or remained unchanged.
Stereotactic techniques deliver less radiation to normal brain and nerve tissue in the tumor vicinity. Thus, radiation-induced complications are thought to be less frequent with radiosurgery than with CRT (8888. Gillam MP, Molitch MP, Lombardi G, Colao A. Advances in the treatment of prolactinomas. Endocr Rev. 2006;27(5):485-534.,189189. Sheplan Olsen LJ, Robles Irizarry L, Chao ST, Weil RJ, Hamrahian AH, Hatipoglu B, Suh JH. Radiotherapy for prolactin-secreting pituitary tumors. Pituitary. 2012;15(2):135-45.,190190. Wan H, Chihiro O, Yuan S. MASEP gamma knife radiosurgery for secretory pituitary adenomas: experience in 347 consecutive cases. J Exp Clin Cancer Res. 2009;28:36.). Moreover, a shorter time to hormonal normalization with radiosurgery has been reported in some series (189189. Sheplan Olsen LJ, Robles Irizarry L, Chao ST, Weil RJ, Hamrahian AH, Hatipoglu B, Suh JH. Radiotherapy for prolactin-secreting pituitary tumors. Pituitary. 2012;15(2):135-45.,191191. Kong DS, Lee JI, Lim do H, Kim KW, Shin HJ, Nam DH, et al. The efficacy of fractionated radiotherapy and stereotactic radiosurgery for pituitary adenomas: long-term results of 125 consecutive patients treated in a single institution. Cancer. 2017;110(4):854-60.), but not in all (192192. Castinetti F, Nagai M, Morange I, Dufour H, Caron P, Chanson P, et al. Long-term results of stereotactic radiosurgery in secretory pituitary adenomas. J Clin Endocrinol Metab. 2009;94(9):3400-7.).
Currently, radiosurgery has been the recommended treatment option unless the tumor is larger than 3-4 cm, or within 3 mm of the optic nerves, chiasm or tracts (192192. Castinetti F, Nagai M, Morange I, Dufour H, Caron P, Chanson P, et al. Long-term results of stereotactic radiosurgery in secretory pituitary adenomas. J Clin Endocrinol Metab. 2009;94(9):3400-7.). In this situation, patients should be offered CRT or, whenever possible, stereotactic RT in multiple fractions (8888. Gillam MP, Molitch MP, Lombardi G, Colao A. Advances in the treatment of prolactinomas. Endocr Rev. 2006;27(5):485-534.,189189. Sheplan Olsen LJ, Robles Irizarry L, Chao ST, Weil RJ, Hamrahian AH, Hatipoglu B, Suh JH. Radiotherapy for prolactin-secreting pituitary tumors. Pituitary. 2012;15(2):135-45.–191191. Kong DS, Lee JI, Lim do H, Kim KW, Shin HJ, Nam DH, et al. The efficacy of fractionated radiotherapy and stereotactic radiosurgery for pituitary adenomas: long-term results of 125 consecutive patients treated in a single institution. Cancer. 2017;110(4):854-60.).
Safety
The use of convention radiotherapy (CRT) is associated with the development of several severe complications (121121. Auriemma RS, Grasso LF, Pivonello R, Colao A. The safety of treatments for prolactinomas. Expert Opin Drug Saf. 2016;15(4):503-12.). More than 50% of patients receiving pituitary radiotherapy will develop at least one anterior pituitary hormone deficiency within the following decade (193193. Tsang RW, Brierley JD, Panzarella T, Gospodarowicz MK, Sutcliffe SB, Simpson WJ. Radiation therapy for pituitary adenoma: treatment outcome and prognostic factors. Int J Radiat Oncol Biol Phys. 1994;30(3):557-65.,194194. Littley MD, Shalet SM, Beardwell CG, Robinson EL, Sutton ML Radiation-induced hypopituitarism is dose-dependent. Clin Endocrinol (Oxf). 1989;31(3):363-73.). Although hypopituitarism tends to arise in the first 5 years after radiation treatment, new deficiencies can appear even 20 years later (103103. Hamilton DK, Vance ML, Roulos PT, Laws ER. Surgical outcomes in hyporesponsive prolactinomas: analysis of patients with resistance or intolerance to dopamine agonists. Pituitary. 2005;8(1):53-60.,176176. Auriemma RS, Perone Y, Di Sarno A, Grasso LF, Guerra E, Gasperi M, et al. Results of a single-center observational 10-year survey study on recurrence of hyperprolactinemia after pregnancy and lactation. J Clin Endocrinol Metab. 2013;98(1):372-9.,177177. Domingue ME, Devuyst F, Alexopoulou O, Corvilain B, Maiter D. Outcome of prolactinoma after pregnancy and lactation: a study on 73 patients. Clin Endocrinol (Oxf). 2014;80(5):642-8.). Additional complications of CRT include cerebrovascular accidents (CVAs), a second brain tumor and optic nerve injury. The incidence of CVA has been found to increase from the time of radiation, from 4% at 5 years to 11% at 10 years and 21% at 20 years (121121. Auriemma RS, Grasso LF, Pivonello R, Colao A. The safety of treatments for prolactinomas. Expert Opin Drug Saf. 2016;15(4):503-12.,195195. Brada M, Burchell L, Ashley S, Traish D. The incidence of cerebrovascular accidents in patients with pituitary adenoma. Neurooncol Pract. 2014;1(1):22-8.). The cumulative risk of second brain tumors has been demonstrated to range from 2.0% at 10 yr, to 2.4% at 20 yr, and 8.5% at 30 yr (196196. Minniti G, Traish D, Ashley S, Gonsalves A, Brada M. Risk of second brain tumor after conservative surgery and radiotherapy for pituitary adenoma: update after an additional 10 years. J Clin Endocrinol Metab. 2005;90(2):800-4.). The rate of presumed radiation-induced optic neuropathy has been estimated to be 0.8% at 10 years (197197. Erridge SC, Conkey DS, Stockton D, Strachan MW, Statham PF, Whittle IR, et al. Radiotherapy for pituitary adenomas: long-term efficacy and toxicity. Radiother Oncol. 2009;93(3):597-601.). Another rare complication is radiation-induced necrosis of the surrounding brain tissue, with a prevalence of approximately 0.2-0.8% (8888. Gillam MP, Molitch MP, Lombardi G, Colao A. Advances in the treatment of prolactinomas. Endocr Rev. 2006;27(5):485-534.,121121. Auriemma RS, Grasso LF, Pivonello R, Colao A. The safety of treatments for prolactinomas. Expert Opin Drug Saf. 2016;15(4):503-12.).
In patients receiving radiosurgery, hypopituitarism has been found to be the most common complication, occurring in approximately one-third of patients (121121. Auriemma RS, Grasso LF, Pivonello R, Colao A. The safety of treatments for prolactinomas. Expert Opin Drug Saf. 2016;15(4):503-12.,198198. Cohen-Inbar O, Xu Z, Schlesinger D, Vance ML, Sheehan JP. Gamma Knife radiosurgery for medically and surgically refractory prolactinomas: long-term results. Pituitary. 2015;18(6):820-30.). Clinical deterioration due to visual field loss and worsened facial sensory loss, as well as a handful of cases of second brain malignancies have also been reported (8888. Gillam MP, Molitch MP, Lombardi G, Colao A. Advances in the treatment of prolactinomas. Endocr Rev. 2006;27(5):485-534.,191191. Kong DS, Lee JI, Lim do H, Kim KW, Shin HJ, Nam DH, et al. The efficacy of fractionated radiotherapy and stereotactic radiosurgery for pituitary adenomas: long-term results of 125 consecutive patients treated in a single institution. Cancer. 2017;110(4):854-60.,199199. Liu X, Kano H, Kondziolka D, Park KJ, Iyer A, Shin S, et al. Gamma knife stereotactic radiosurgery for drug resistant or intolerant invasive prolactinomas. Pituitary. 2013;16(1):68-75.).
These data suggest the greater safety of radiosurgery compared to CRT. However, further studies of large series and with long follow-ups are required to draw definitive conclusions (121121. Auriemma RS, Grasso LF, Pivonello R, Colao A. The safety of treatments for prolactinomas. Expert Opin Drug Saf. 2016;15(4):503-12.).
COMMENT 12: Due to its low efficacy and potentially severe side-effects, radiation therapy is only indicated to control tumor growth in DA-resistant cases not controlled by surgery. Whenever possible, preference should be given to stereotactic techniques.
2.8 How giant prolactinomas should be managed?
Although there is no consensus on the definition of giant prolactinomas (GPs), they are usually defined as tumors with a maximum diameter greater than or equal to 4 cm (200200. Moraes AB, Silva CM, Vieira Neto L, Gadelha MR. Giant prolactinomas: the therapeutic approach. Clin Endocrinol (Oxf). 2013;79(4):447-56.–202202. Maiter D, Delgrange E. Therapy of endocrine disease: the challenges in managing giant prolactinomas. Eur J Endocrinol. 2014;170(6):R213-27.).
GPs represent only 2-3% of all prolactinomas, and they are more commonly found in middle-aged men (200200. Moraes AB, Silva CM, Vieira Neto L, Gadelha MR. Giant prolactinomas: the therapeutic approach. Clin Endocrinol (Oxf). 2013;79(4):447-56.,201201. Delgrange E, Raverot G, Bex M, Burman P, Decoudier B, Devuyst F, et al. Giant prolactinomas in women. Eur J Endocrinol. 2013;170(1):31-8.). These tumors are usually more aggressive in men than in women (203203. Delgrange E, Trouillas J, Maiter D, Donckier J, Tourniaire J. Sex-related difference in the growth of prolactinomas: a clinical and proliferation marker study. J Clin Endocrinol Metab. 1997;82(7):2102-7.). GPs often cause visual field defects and/or ophthalmoplegia due to compression of the optic chiasm and/or cranial nerves, respectively, and headaches, as well as other neurological alterations and other atypical manifestations for a pituitary adenoma, such as orbital invasion, epistaxis or obstructive hydrocephalus (204204. Shrivastava RK, Arginteanu MS, King WA, Post KD. Giant prolactinomas: clinical management and long-term follow up. J Neurosurg. 2002;97(2):299-306.–207207. Alkatari S, Aljohani N. Obstructive hydrocephalus, fifth nerve and hypothalamus involvement: acute presentation of a giant prolactinoma. Clin Med Insights Case Rep. 2012;5:115-8.). Panhypopituitarism may also be present. Therefore, formal visual testing is usually necessary, and pituitary function evaluation is mandatory (200200. Moraes AB, Silva CM, Vieira Neto L, Gadelha MR. Giant prolactinomas: the therapeutic approach. Clin Endocrinol (Oxf). 2013;79(4):447-56.,203203. Delgrange E, Trouillas J, Maiter D, Donckier J, Tourniaire J. Sex-related difference in the growth of prolactinomas: a clinical and proliferation marker study. J Clin Endocrinol Metab. 1997;82(7):2102-7.).
Different therapeutic approaches, such as dopamine agonists (DAs) alone or in combination with surgery and radiotherapy, may be necessary to reach the therapeutic goals in patients with GPs, which include tumor volume control, normalization of the PRL level and restoration of eugonadism (200200. Moraes AB, Silva CM, Vieira Neto L, Gadelha MR. Giant prolactinomas: the therapeutic approach. Clin Endocrinol (Oxf). 2013;79(4):447-56.). Temozolomide (TMZ) has been used in aggressive prolactinomas that are unresponsive to the abovementioned therapies (117117. Halevy C, Whitelaw BC. How effective is temozolomide for treating pituitary tumours and when should it be used? Pituitary.2017;20(2):261-6.–119119. Whitelaw BC, Dworakowska D, Thomas NQ, Barazi S, Riordan-Eva P, King AP, et al. Temozolomide in the management of dopamine agonist-resistant prolactinomas. Clin Endocrinol (Oxf). 2012;76(6):877-86.). Due to the large tumor volume and commonly invasive behavior, control of mass effects should be a priority in the management of patients with giant prolactinomas (8888. Gillam MP, Molitch MP, Lombardi G, Colao A. Advances in the treatment of prolactinomas. Endocr Rev. 2006;27(5):485-534.).
Dopamine agonists
As medical therapy in most cases causes a marked and rapid reduction of the tumor size, it is considered the first-line treatment option in patients with GPs, even in the presence of visual abnormalities. However, close monitoring of the visual field is mandatory (200200. Moraes AB, Silva CM, Vieira Neto L, Gadelha MR. Giant prolactinomas: the therapeutic approach. Clin Endocrinol (Oxf). 2013;79(4):447-56.–202202. Maiter D, Delgrange E. Therapy of endocrine disease: the challenges in managing giant prolactinomas. Eur J Endocrinol. 2014;170(6):R213-27.). In this setting, early surgery (e.g., within 15 to 30 days) may occasionally be necessary if the tumors do not shrink and if severe visual field defects do not improve or even get worse (200200. Moraes AB, Silva CM, Vieira Neto L, Gadelha MR. Giant prolactinomas: the therapeutic approach. Clin Endocrinol (Oxf). 2013;79(4):447-56.–202202. Maiter D, Delgrange E. Therapy of endocrine disease: the challenges in managing giant prolactinomas. Eur J Endocrinol. 2014;170(6):R213-27.).
The use of DA for GPs seems to reduce significantly the tumor mass (> 30% decrease in tumor diameter or > 65% reduction in tumor volume) in 74% of the cases and to normalize the PRL level in 60% of the cases (200200. Moraes AB, Silva CM, Vieira Neto L, Gadelha MR. Giant prolactinomas: the therapeutic approach. Clin Endocrinol (Oxf). 2013;79(4):447-56.). Cabergoline (CAB) is the preferred DA for the medical management of GPs due to its greater efficacy and better tolerability compared to BCR (3434. Vilar L, Freitas MC, Naves LA, Casulari LA, Azevedo M, Montenegro R Jr, et al. Diagnosis and management of hyperprolactinemia: results of a Brazilian multicenter study with 1234 patients. J Endocrinol Invest. 2008;31(5):436-44.,8888. Gillam MP, Molitch MP, Lombardi G, Colao A. Advances in the treatment of prolactinomas. Endocr Rev. 2006;27(5):485-534.). In the series by Espinosa and cols. (208208. Espinosa E, Sosa E, Mendoza V, Ramírez C, Melgar V, Mercado M. Giant prolactinomas: are they really different from ordinary macroprolactinomas? Endocrine. 2015;52(3):652-9.), CAB treatment resulted in the normalization of PRL levels in 68% and in the reduction of > 50% in tumor volume in 87% of the GP patients. The composite goal of PRL normalization and > 50% tumor reduction was achieved by 55% of patients with GPs (n = 26) and by 66% of patients with no giant macroprolactinomas (n = 100) (p = 0.19) (208208. Espinosa E, Sosa E, Mendoza V, Ramírez C, Melgar V, Mercado M. Giant prolactinomas: are they really different from ordinary macroprolactinomas? Endocrine. 2015;52(3):652-9.). Among GPs ≥ 6 cm, DA treatment achieved PRL normalization in 11/18 (61%) patients within a median interval of 20 months (209209. Shimon I, Sosa E, Mendoza V, Greenman Y, Tirosh A, Espinosa E et al. Giant prolactinomas larger than 60 mm in size: a cohort of massive and aggressive prolactin-secreting pituitary adenomas. Pituitary. 2016;19(4):429-36.). In the series by Vilar and cols. (210210. Vilar L, Albuquerque JL, Gomes B, Gadelha PS, Lopes IGA, Thé AC, et al. Giant prolactinomas: clinical manifestations and response to treatment in 16 cases. Arch Endocrinol Metab. 2015; 59 (Suppl. 1):S21.), 10 of 16 patients (62.5%) reached PRL normalization at weekly doses ranging from 2.5 to 7 mg. Regarding DA withdrawal, there are no data in the literature about CAB withdrawal in patients with GPs. Concerning BRC withdrawn in these patients, some authors described the persistence of normoprolactinemia and tumor volume shrinkage in only a minority of patients (211211. Saeki N, NakamuraM, Sunami K, Yamaura A. Surgical indication after bromocriptine therapy on giant prolactinomas: effects and limitations of the medical treatment. Endocr J. 1998;45(4):529-37.–213213. Kyung Rae C, Kyung-Il J, Hyung Jin S. Bromocriptine therapy for the treatment of invasive prolactinoma: the single institute experience. Brain Tumor Res Treat. 2013; 1(2):71–7).
Surgery
Neurosurgery should be considered as part of the multidisciplinary approach to patients with DA-resistant prolactinoma (debulking surgery) or should be restricted to some acute complications such as apoplexy or leakage of cerebrospinal fluid during DA therapy (44. Melmed S, Casanueva FF, Hoffman AR, Kleinberg DL, Montori VM, Schlechte JA, et al. Diagnosis and treatment of hyperprolactinemia: An Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(2):273-88.,206206. Chaurasia PK, Singh D, Meher S, Saran RK, Singh H. Epistaxis as first clinical presentation in a child with giant prolactinoma: Case report and review of literature. J Pediatr Neurosci. 2011;6(2):134-7.,214214. Komotar RJ, Starke RM, Raper DM, Anand VK, Schwartz TH. Endoscopic endonasal compared with microscopic transsphenoidal and open transcranial resection of giant pituitary adenomas. Pituitary. 2012;15(2):150-9.). Neurosurgery could also be considered for patients with severe visual field defects that do not show any improvement or even worsen during DA treatment. The transsphenoidal approach is usually the first choice, even for GPs (200200. Moraes AB, Silva CM, Vieira Neto L, Gadelha MR. Giant prolactinomas: the therapeutic approach. Clin Endocrinol (Oxf). 2013;79(4):447-56.). In patients with giant and invasive prolactinomas, surgery is hardly curative, regardless of the surgical technique employed or the neurosurgeon's experience. Therefore, in these cases, the goal of surgery is to debulk the tumor in order to improve the symptoms related to mass effects (8888. Gillam MP, Molitch MP, Lombardi G, Colao A. Advances in the treatment of prolactinomas. Endocr Rev. 2006;27(5):485-534.,121121. Auriemma RS, Grasso LF, Pivonello R, Colao A. The safety of treatments for prolactinomas. Expert Opin Drug Saf. 2016;15(4):503-12.). Debulking surgery may also improve the tumor response to DA therapy (102102. Primeau V, Raftopoulos C, Maiter D. Outcomes of transsphenoidal surgery in prolactinomas: improvement of hormonal control in dopamine agonist-resistant patients. Eur J Endocrinol. 2012;166(5):779-86.,104104. Vrooner L, Jaffrain-Rea M-L, Petrossians P, Tamagno G, Chanson P, Vilar L, et al. Prolactinomas resistant to standard doses of cabergoline: a multicenter study of 92 patients. Eur J Endocrinol. 2012;167(5):651-62.).
Radiotherapy
Radiotherapy should be used in patients with GPs who do not achieve disease control with DA therapy and surgery, particularly regarding mass effects (200200. Moraes AB, Silva CM, Vieira Neto L, Gadelha MR. Giant prolactinomas: the therapeutic approach. Clin Endocrinol (Oxf). 2013;79(4):447-56.–202202. Maiter D, Delgrange E. Therapy of endocrine disease: the challenges in managing giant prolactinomas. Eur J Endocrinol. 2014;170(6):R213-27.).
Temozolomide
Temozolomide is an oral alkylating chemotherapeutic agent that exerts cytotoxic effects through methylation of DNA and can be used in aggressive giant prolactinomas that remain uncontrolled in terms of mass effects despite the multiple treatment modalities previously mentioned. The standard dose is 150–200 mg/m2 for 5 days, repeated every 28-days in a cycle (117117. Halevy C, Whitelaw BC. How effective is temozolomide for treating pituitary tumours and when should it be used? Pituitary.2017;20(2):261-6.,119119. Whitelaw BC, Dworakowska D, Thomas NQ, Barazi S, Riordan-Eva P, King AP, et al. Temozolomide in the management of dopamine agonist-resistant prolactinomas. Clin Endocrinol (Oxf). 2012;76(6):877-86.).
COMMENT 13: Cabergoline is the therapy of choice for patients with giant prolactinomas (GPs), as this drug enables normalization of prolactin levels and significant tumor reduction in the majority of cases, but higher doses are often required, compared to those for smaller macroprolactinomas. Surgery should be considered as part of the management of these patients, especially in patients who still have a large tumor load despite dopamine agonist therapy. Radiotherapy and/or temozolomide should be reserved for the subgroup of GPs that are not controlled in terms of mass effects regardless of the multiple conventional treatment modalities.
2.9 How to manage the challenges involving prolactinomas and pregnancy?
Chronic anovulation and infertility are very frequent among women with prolactinomas, mostly due to the hypogonadotropic hypogonadism related to hyperprolactinemia (66. Glezer A, Bronstein MD. Prolactinomas. Endocrinol Metab Clin N Am. 2015;44(1):71-8.,1010. Auriema R, Pivonello R, Colao A. Prolactinomas. In: Vilar L (editor). In: Vilar L, editor. Endocrinologia Clínica, 6a ed. (Clinical Endocrinology, 6th ed.). Rio de Janeiro: Guanabara Koogan; 2016. p. 15-29.,8888. Gillam MP, Molitch MP, Lombardi G, Colao A. Advances in the treatment of prolactinomas. Endocr Rev. 2006;27(5):485-534.). Hyperprolactinemia decreases the luteinizing hormone (LH) pulse amplitude and frequency through the suppression of GnRH (1414. Milenković L, D'Angelo G, Kelly PA, Weiner RI. Inhibition of gonadotropin hormone-releasing hormone release by prolactin from GT1 neuronal cell lines through prolactin receptors. Proc Natl Acad Sci USA. 1994;91(4):1244-7.). This effect appears to be mediated by an earlier step of suppressing the generation of kisspeptin, a protein made by neurons in the arcuate and periventricular nuclei of the hypothalamus, which stimulates GnRH release (215215. Brown RS, Herbison AE, Grattan DR. Prolactin regulation of kisspeptin neurons in the mouse brain and its role in the lactation-induced suppression of kisspeptin expression. J Neuroendocrinol. 2014:26(12):898-908.). PRL can also decrease estrogen and progesterone production through direct effects on the ovaries (216216. Molitch ME. Endocrinology in pregnancy: management of the pregnant patient with a prolactinoma. Eur J Endocrinol. 2015;172(5):R205-13.). Fertility is, however, restored in most women with the use of DA. In the absence of hormonal control in cases with microprolactinomas, clomiphene citrate or recombinant gonadotropins may be used for ovulation induction (216216. Molitch ME. Endocrinology in pregnancy: management of the pregnant patient with a prolactinoma. Eur J Endocrinol. 2015;172(5):R205-13.,217217. Glezer A, Jallad RS, Machado MC, Fragoso MC, Bronstein MD. Pregnancy and pituitary adenomas. Minerva Endocrinol. 2016;41(3):341-50.).
During pregnancy, the primary concern is growth of the tumor because of high levels of estrogens, leading to visual disturbances and headache. The second point of concern is the fetal exposure to DA in early embryogenesis and the potential risk of malformations (216216. Molitch ME. Endocrinology in pregnancy: management of the pregnant patient with a prolactinoma. Eur J Endocrinol. 2015;172(5):R205-13.,217217. Glezer A, Jallad RS, Machado MC, Fragoso MC, Bronstein MD. Pregnancy and pituitary adenomas. Minerva Endocrinol. 2016;41(3):341-50.).
Tumor growth
Prolactinomas can enlarge during pregnancy as a result of both the stimulatory effect of these high estrogen levels and the discontinuation of the dopamine agonist that might have caused tumor shrinkage (216216. Molitch ME. Endocrinology in pregnancy: management of the pregnant patient with a prolactinoma. Eur J Endocrinol. 2015;172(5):R205-13.). In microprolactinomas, the chance of clinically significant tumor growth is less than 5%; therefore, after pregnancy confirmation, DA can be withdrawn, and the patient should be monitored clinically every trimester. In contrast, in patients with macroadenomas, the risk of tumor growth with clinical repercussion is up to 35% (217217. Glezer A, Jallad RS, Machado MC, Fragoso MC, Bronstein MD. Pregnancy and pituitary adenomas. Minerva Endocrinol. 2016;41(3):341-50.). However, Holmgren and cols. (218218. Holmgren U, Bergstrand G, Hagenfeldt K, Werner S. Women with prolactinoma—effect of pregnancy and lactation on serum prolactin and on tumour growth. Acta Endocrinol (Copenh). 1986;111(4):452-9.) noted a reduced risk of tumor growth during pregnancy in patients treated with BRC for at least 12 months. Thus, in patients with expansive macroprolactinomas, it is mandatory to observe a tumor within the sellar boundaries and usually to wait at least 1 year under treatment with a DA (217217. Glezer A, Jallad RS, Machado MC, Fragoso MC, Bronstein MD. Pregnancy and pituitary adenomas. Minerva Endocrinol. 2016;41(3):341-50.,219219. Bronstein MD, Paraiba DB, Jallad RS. Management of pituitary tumors in pregnancy. Nat Rev Endocrinol. 2011;7(5):301-10.).
DAs safety in the fetus
BRC has been shown to cross the placenta in human studies (103103. Hamilton DK, Vance ML, Roulos PT, Laws ER. Surgical outcomes in hyporesponsive prolactinomas: analysis of patients with resistance or intolerance to dopamine agonists. Pituitary. 2005;8(1):53-60.); CAB has been shown to do so in animal models, but such data are lacking in humans (121121. Auriemma RS, Grasso LF, Pivonello R, Colao A. The safety of treatments for prolactinomas. Expert Opin Drug Saf. 2016;15(4):503-12.,216216. Molitch ME. Endocrinology in pregnancy: management of the pregnant patient with a prolactinoma. Eur J Endocrinol. 2015;172(5):R205-13.). As a general approach in pregnancy, DAsare discontinued as soon as pregnancy is confirmed (216216. Molitch ME. Endocrinology in pregnancy: management of the pregnant patient with a prolactinoma. Eur J Endocrinol. 2015;172(5):R205-13.). Nevertheless, taking into account that CAB has a long half-life and can be detected in the circulation within 30 days after drug withdrawal, early fetal exposure is unavoidable (217217. Glezer A, Jallad RS, Machado MC, Fragoso MC, Bronstein MD. Pregnancy and pituitary adenomas. Minerva Endocrinol. 2016;41(3):341-50.). Moreover, the experience with this drug is still limited compared to BRC: ~950 vs. ~6200 pregnancies induced by CAB and BRC, respectively (216216. Molitch ME. Endocrinology in pregnancy: management of the pregnant patient with a prolactinoma. Eur J Endocrinol. 2015;172(5):R205-13.). These facts are the rational for BRC preference in hyperprolactinemic women willing to become pregnant (217217. Glezer A, Jallad RS, Machado MC, Fragoso MC, Bronstein MD. Pregnancy and pituitary adenomas. Minerva Endocrinol. 2016;41(3):341-50.), as recommended by the Guidelines of the Endocrine Society, with BRC being the only authorized DA for pregnancy induction (44. Melmed S, Casanueva FF, Hoffman AR, Kleinberg DL, Montori VM, Schlechte JA, et al. Diagnosis and treatment of hyperprolactinemia: An Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(2):273-88.). However, in clinical practice, CAB has often been used for this purpose with no apparent damage to the fetus (216216. Molitch ME. Endocrinology in pregnancy: management of the pregnant patient with a prolactinoma. Eur J Endocrinol. 2015;172(5):R205-13.).
The potential fetal malformation and impairment of the dopaminergic brain circuitry under DA use are a concern (216216. Molitch ME. Endocrinology in pregnancy: management of the pregnant patient with a prolactinoma. Eur J Endocrinol. 2015;172(5):R205-13.,217217. Glezer A, Jallad RS, Machado MC, Fragoso MC, Bronstein MD. Pregnancy and pituitary adenomas. Minerva Endocrinol. 2016;41(3):341-50.,219219. Bronstein MD, Paraiba DB, Jallad RS. Management of pituitary tumors in pregnancy. Nat Rev Endocrinol. 2011;7(5):301-10.). Addressing this issue, Molitch (216216. Molitch ME. Endocrinology in pregnancy: management of the pregnant patient with a prolactinoma. Eur J Endocrinol. 2015;172(5):R205-13.) has compared outcomes data from 6239 pregnancies induced by BCR with 968 pregnancies induced by CAB and pregnancies in the US general population, regarding miscarriages, births at term, premature births, multiple pregnancies, and malformed newborns. Overall, no difference was found among the three groups (216216. Molitch ME. Endocrinology in pregnancy: management of the pregnant patient with a prolactinoma. Eur J Endocrinol. 2015;172(5):R205-13.).
In the vast majority of cases, the DA was withdrawn at up to 6 weeks of pregnancy. In the literature, there are only data on approximately 100 cases of BRC use throughout pregnancy, which have disclosed one case of undescended testis and one case of talipes deformity (217217. Glezer A, Jallad RS, Machado MC, Fragoso MC, Bronstein MD. Pregnancy and pituitary adenomas. Minerva Endocrinol. 2016;41(3):341-50.,220220. Raymond JP, Goldstein E, Konopka P, Leleu MR, Merceron LE, Loria Y. Follow-up of children born of bromocriptine-treated mothers. Horm Res. 1985;22(3):239-46.,221221. Canales ES, Garcia IC, Ruiz JE, Zárate A. Bromocriptine as prophylactic therapy in prolactinoma during pregnancy. Fertil Steril. 1981;36(4):524-6.). Recently, a compilation of 15 pregnancies with the use of CAB during the entire pregnancy has shown 12 healthy babies and one fetal death by severe pre-eclampsia (222222. Glezer A, Bronstein MD. Prolactinomas, cabergoline, and pregnancy. Endocrine. 2014;47(1):64-9.).
Concerning the long follow-up of children conceived during BRC treatment, Bronstein followed up 70 children, during 12 to 240 months, and there was one case of idiopathic hydrocephalus, one child with tuberous sclerosis and another one with precocious puberty (223223. Bronstein MD. Prolactinomas and pregnancy. Pituitary. 2005;8(1):31-8.). In two other studies (217217. Glezer A, Jallad RS, Machado MC, Fragoso MC, Bronstein MD. Pregnancy and pituitary adenomas. Minerva Endocrinol. 2016;41(3):341-50.,224224. Krupp P, Monka C, Richter K. The safety aspects of infertility treatments. Program of the Second World Congress of Gynecology and Obstetrics 1988. Rio de Janeiro, Brazil. p. 9.), no impairment of physical development was observed.
In four studies, the follow-ups of children conceived on CAB were described. Bronstein (223223. Bronstein MD. Prolactinomas and pregnancy. Pituitary. 2005;8(1):31-8.) and Ono and cols. (225225. Ono M, Miki N, Amano K, Kawamata T, Seki T, Makino R, et al. High-dose cabergoline therapy for hyperprolactinemic infertility in women with micro- and macroprolactinomas. J Clin Endocrinol Metab. 2010;95(6):2672-9.) did not find any abnormalities in five and 83 children followed for 41 months and 12 years, respectively. Lebbe and cols. (226226. Lebbe M, Hubinont C, Bernard P, Maiter D. Outcome of 100 pregnancies initiated under treatment with cabergoline in hyperprolactinaemic women. Clin Endocrinol (Oxf). 2010;73(2):236-42.) following 88 children described two cases of slight delay in verbal fluency and one case of difficulty in achieving complete continence. Moreover, Stalldecker and cols. (227227. Stalldecker G, Gil MSM, Guitelman MA, Alfieri A, Ballarino MC, Boero L, et al. Effects of cabergoline on pregnancy and embryo-fetal development: retrospective study on 103 pregnancies and a review of the literature. Pituitary. 2010;13(4):345-50.) followed 61 children and found two cases of seizures and two cases of pervasive developmental disorder.
In summary, BCR and CAB seem to be equally safe for the fetus; none of these drugs are apparently associated with increased risk for abortions or malformations, when used for pregnancy induction in infertile hyperprolactinemic women. Ideally, CAB should be discontinued 30 days before the conception (216216. Molitch ME. Endocrinology in pregnancy: management of the pregnant patient with a prolactinoma. Eur J Endocrinol. 2015;172(5):R205-13.,218218. Holmgren U, Bergstrand G, Hagenfeldt K, Werner S. Women with prolactinoma—effect of pregnancy and lactation on serum prolactin and on tumour growth. Acta Endocrinol (Copenh). 1986;111(4):452-9.,223223. Bronstein MD. Prolactinomas and pregnancy. Pituitary. 2005;8(1):31-8.).
The best approach
An algorithm proposed by Glezer and cols. (217217. Glezer A, Jallad RS, Machado MC, Fragoso MC, Bronstein MD. Pregnancy and pituitary adenomas. Minerva Endocrinol. 2016;41(3):341-50.) for the management of prolactinoma during pregnancy is shown is Figure 8. The DA should be withdrawn as soon as pregnancy is confirmed in patients with microadenomas or enclosed macroadenomas. Assessment of tumor shrinkage in macroadenomas is mandatory before pregnancy allowance. In previously expanded or invasive tumors, the choice of BRC maintenance throughout pregnancy or its reintroduction if needed, or else the indication of previous surgical debulking, depends on the physician's and patient's decision (217217. Glezer A, Jallad RS, Machado MC, Fragoso MC, Bronstein MD. Pregnancy and pituitary adenomas. Minerva Endocrinol. 2016;41(3):341-50.).
Algorithm suggested for the prolactinoma management during pregnancy (PRL: prolactin; BCR: bromocriptine; MRI: magnetic resonance imaging) (Adapted from Ref. 217).
Patients with microprolactinomas (MICs) or enclosed macroprolactinomas (MACs) should be followed each trimester, with attention to headache and visual impairment (217217. Glezer A, Jallad RS, Machado MC, Fragoso MC, Bronstein MD. Pregnancy and pituitary adenomas. Minerva Endocrinol. 2016;41(3):341-50.). In the presence of those complaints, sellar MRI without contrast, preferably after the first trimester, should be performed, and BRC reintroduction is indicated if tumor growth is related to the clinical findings (217217. Glezer A, Jallad RS, Machado MC, Fragoso MC, Bronstein MD. Pregnancy and pituitary adenomas. Minerva Endocrinol. 2016;41(3):341-50.). In cases of BCR intolerance, CAB, although off-label, could be used instead (217217. Glezer A, Jallad RS, Machado MC, Fragoso MC, Bronstein MD. Pregnancy and pituitary adenomas. Minerva Endocrinol. 2016;41(3):341-50.,219219. Bronstein MD, Paraiba DB, Jallad RS. Management of pituitary tumors in pregnancy. Nat Rev Endocrinol. 2011;7(5):301-10.,222222. Glezer A, Bronstein MD. Prolactinomas, cabergoline, and pregnancy. Endocrine. 2014;47(1):64-9.). On the other hand, in patients with expanding MACs, DA maintenance throughout pregnancy should be evaluated by an expert (217217. Glezer A, Jallad RS, Machado MC, Fragoso MC, Bronstein MD. Pregnancy and pituitary adenomas. Minerva Endocrinol. 2016;41(3):341-50.,219219. Bronstein MD, Paraiba DB, Jallad RS. Management of pituitary tumors in pregnancy. Nat Rev Endocrinol. 2011;7(5):301-10.). Clinical evaluation monthly and neuroophthalmological evaluation each trimester are indicated. In patients with MICs or MACs experiencing symptoms related to mass effects and without improvement with clinical treatment, neurosurgery should be performed, preferably during the second trimester (216216. Molitch ME. Endocrinology in pregnancy: management of the pregnant patient with a prolactinoma. Eur J Endocrinol. 2015;172(5):R205-13.,217217. Glezer A, Jallad RS, Machado MC, Fragoso MC, Bronstein MD. Pregnancy and pituitary adenomas. Minerva Endocrinol. 2016;41(3):341-50.). An alternative approach might be delivery of the baby, if the pregnancy is sufficiently advanced (e.g., > 37 weeks) (216216. Molitch ME. Endocrinology in pregnancy: management of the pregnant patient with a prolactinoma. Eur J Endocrinol. 2015;172(5):R205-13.,217217. Glezer A, Jallad RS, Machado MC, Fragoso MC, Bronstein MD. Pregnancy and pituitary adenomas. Minerva Endocrinol. 2016;41(3):341-50.,219219. Bronstein MD, Paraiba DB, Jallad RS. Management of pituitary tumors in pregnancy. Nat Rev Endocrinol. 2011;7(5):301-10.).
Notably, during pregnancy, periodic checking of PRL levels is of no diagnostic benefit and can be misleading (216216. Molitch ME. Endocrinology in pregnancy: management of the pregnant patient with a prolactinoma. Eur J Endocrinol. 2015;172(5):R205-13.,217217. Glezer A, Jallad RS, Machado MC, Fragoso MC, Bronstein MD. Pregnancy and pituitary adenomas. Minerva Endocrinol. 2016;41(3):341-50.). Indeed, in normal women, PRL levels rise with gestation, but PRL levels do not always rise with tumor enlargement, and tumor enlargement can occur without a change in the PRL concentration (216216. Molitch ME. Endocrinology in pregnancy: management of the pregnant patient with a prolactinoma. Eur J Endocrinol. 2015;172(5):R205-13.). A rise in PRL may well not indicate tumor enlargement and therefore may cause unnecessary worry. By contrast, the lack of a rise in PRL may be falsely reassuring in a patient with headaches or other evidence of tumor enlargement (216216. Molitch ME. Endocrinology in pregnancy: management of the pregnant patient with a prolactinoma. Eur J Endocrinol. 2015;172(5):R205-13.).
Follow-up
After pregnancy, PRL levels and tumor size should be reassessed because reduction, or even complete tumor disappearance, after pregnancy may occur. Moreover, asymptomatic tumor enlargement may occur during pregnancy (216216. Molitch ME. Endocrinology in pregnancy: management of the pregnant patient with a prolactinoma. Eur J Endocrinol. 2015;172(5):R205-13.,217217. Glezer A, Jallad RS, Machado MC, Fragoso MC, Bronstein MD. Pregnancy and pituitary adenomas. Minerva Endocrinol. 2016;41(3):341-50.). It has been suggested that high levels of estrogen during pregnancy can induce areas of tumor necrosis and apoptosis. The median rate of hyperprolactinemia remission after pregnancy in eight studies was 27%, ranging from 10 to 68% (217217. Glezer A, Jallad RS, Machado MC, Fragoso MC, Bronstein MD. Pregnancy and pituitary adenomas. Minerva Endocrinol. 2016;41(3):341-50.).
Breastfeeding does not seem to be associated with tumor growth risk, and it is allowed in patients who did not require DA during pregnancy (216216. Molitch ME. Endocrinology in pregnancy: management of the pregnant patient with a prolactinoma. Eur J Endocrinol. 2015;172(5):R205-13.–219219. Bronstein MD, Paraiba DB, Jallad RS. Management of pituitary tumors in pregnancy. Nat Rev Endocrinol. 2011;7(5):301-10.).
COMMENT 14: In the large majority of cases, a dopamine agonist is only necessary to induce pregnancy and should be discontinued as soon as pregnancy is confirmed. Nevertheless, in selected cases harboring invasive macroprolactinomas or in patients with marked growth and visual and/or headache complaints, the drug may be used throughout pregnancy.
COMMENT 15: Bromocriptine and cabergoline seem to be equally safe for the fetus, when used for pregnancy induction. Indeed, short-term exposure to these drugs for less than 6 weeks of gestation has not been found to cause any increase in spontaneous abortions, ectopic pregnancies, trophoblastic disease, multiple pregnancies, or congenital malformations.
2.10 How to manage the psychotropic-induced hyperprolactinemia?
It should always be kept in mind that patients with apparent psychotropic-induced hyperprolactinemia may have an alternative etiology for the PRL elevation, such prolactinomas, primary hypothyroidism, macroprolactinemia, or pseudo-prolactinoma (11. Vilar L, Fleseriu M, Bronstein MD. Pitfalls in the diagnosis of hyperprolactinemia. Arq Bras Endocrinol Metab. 2014;58(1):9-22.). Ideally, a baseline PRL level should be obtained before starting psychotropic medications known to increase PRL, but in the majority of cases, this is not practically possible, particularly when these medications are started urgently (44. Melmed S, Casanueva FF, Hoffman AR, Kleinberg DL, Montori VM, Schlechte JA, et al. Diagnosis and treatment of hyperprolactinemia: An Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(2):273-88.,4444. La Torre D, Falorni A. Pharmacological causes of hyperprolactinemia. Ther Clin Risk Manag. 2007;3(5):929-51.). Endocrine Society Guidelines suggest discontinuing the drug for 3 days or substituting with an alternative drug if psychotropic-induced hyperprolactinemia is suspected and then remeasuring PRL (44. Melmed S, Casanueva FF, Hoffman AR, Kleinberg DL, Montori VM, Schlechte JA, et al. Diagnosis and treatment of hyperprolactinemia: An Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(2):273-88.). Nevertheless, the decision to withdraw or substitute the medication should always be made in consultation with the patient's psychiatrist (44. Melmed S, Casanueva FF, Hoffman AR, Kleinberg DL, Montori VM, Schlechte JA, et al. Diagnosis and treatment of hyperprolactinemia: An Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(2):273-88.,4646. Johnsen E, Kroken RA, Abaza M, Olberg H, Jørgensen HA. Antipsychotic- induced hyperprolactinemia: a cross-sectional survey. J Clin Psychopharmacol. 2008;28(6):686-90.). If the drug cannot be safely withdrawn due to the risk of a relapse of psychiatric symptoms and if the onset of hyperprolactinemia does not coincide with the initiation of therapy, then pituitary MRI should be performed to exclude or diagnose a sellar region lesion (44. Melmed S, Casanueva FF, Hoffman AR, Kleinberg DL, Montori VM, Schlechte JA, et al. Diagnosis and treatment of hyperprolactinemia: An Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(2):273-88.,4646. Johnsen E, Kroken RA, Abaza M, Olberg H, Jørgensen HA. Antipsychotic- induced hyperprolactinemia: a cross-sectional survey. J Clin Psychopharmacol. 2008;28(6):686-90.).
The different approaches proposed for symptomatic patients with confirmed psychotropic-induced hyperprolactinemia include (i) switching the offending drug to aripiprazole or other atypical antipsychotics (AAPs) with a low effect on PRL levels (4141. Karavitaki N, Thanabalasingham G, Shore HC, Trifanescu R, Ansorge O, Meston N, et al. Do the limits of serum prolactin in disconnection hyperprolactinaemia need re-definition? A study of 226 patients with histologically verified non-functioning pituitary macroadenoma. Clin Endocrinol (Oxf). 2006;65(4):524-9.,209209. Shimon I, Sosa E, Mendoza V, Greenman Y, Tirosh A, Espinosa E et al. Giant prolactinomas larger than 60 mm in size: a cohort of massive and aggressive prolactin-secreting pituitary adenomas. Pituitary. 2016;19(4):429-36.–211211. Saeki N, NakamuraM, Sunami K, Yamaura A. Surgical indication after bromocriptine therapy on giant prolactinomas: effects and limitations of the medical treatment. Endocr J. 1998;45(4):529-37.) and (ii) adding DA therapy (4141. Karavitaki N, Thanabalasingham G, Shore HC, Trifanescu R, Ansorge O, Meston N, et al. Do the limits of serum prolactin in disconnection hyperprolactinaemia need re-definition? A study of 226 patients with histologically verified non-functioning pituitary macroadenoma. Clin Endocrinol (Oxf). 2006;65(4):524-9.).
Switching to aripiprazole or other AAPs
Because the atypical antipsychotic aripiprazole is a partial D2 receptor agonist, it has a neutral effect on PRL levels and may often decrease them (4646. Johnsen E, Kroken RA, Abaza M, Olberg H, Jørgensen HA. Antipsychotic- induced hyperprolactinemia: a cross-sectional survey. J Clin Psychopharmacol. 2008;28(6):686-90., 228228. Yoon HW, Lee JS, Park SJ, Lee SK, Choi WJ, Kim TY, et al. Comparing the effectiveness and safety of the addition of and switching to aripiprazole for resolving antipsychotic-induced hyperprolactinemia: a multicenter, open-label, prospective study. Clin Neuropharmacol. 2016;39(6):288-94.). Thus, it may be used as alternative therapy for patients with antipsychotic-induced hyperprolactinemia (4646. Johnsen E, Kroken RA, Abaza M, Olberg H, Jørgensen HA. Antipsychotic- induced hyperprolactinemia: a cross-sectional survey. J Clin Psychopharmacol. 2008;28(6):686-90.,228228. Yoon HW, Lee JS, Park SJ, Lee SK, Choi WJ, Kim TY, et al. Comparing the effectiveness and safety of the addition of and switching to aripiprazole for resolving antipsychotic-induced hyperprolactinemia: a multicenter, open-label, prospective study. Clin Neuropharmacol. 2016;39(6):288-94.). Other atypical antipsychotics, such as quetiapine, olanzapine, lurasidone, asenapine, or clozapine, cause only a mild elevation in PRL levels, and only in a subgroup of those treated (4343. Voicu V, Medvedovici A, Ranetti AE, Rădulescu FŞ. Drug-induced hypo- and hyperprolactinemia: mechanisms, clinical and therapeutic consequences. Expert Opin Drug Metab Toxicol. 2013;9(8):955-68.,4646. Johnsen E, Kroken RA, Abaza M, Olberg H, Jørgensen HA. Antipsychotic- induced hyperprolactinemia: a cross-sectional survey. J Clin Psychopharmacol. 2008;28(6):686-90.). Therefore, these drugs may also serve as alternatives to antipsychotics with robust effects on PRL (e.g., risperidone) (4343. Voicu V, Medvedovici A, Ranetti AE, Rădulescu FŞ. Drug-induced hypo- and hyperprolactinemia: mechanisms, clinical and therapeutic consequences. Expert Opin Drug Metab Toxicol. 2013;9(8):955-68.,4646. Johnsen E, Kroken RA, Abaza M, Olberg H, Jørgensen HA. Antipsychotic- induced hyperprolactinemia: a cross-sectional survey. J Clin Psychopharmacol. 2008;28(6):686-90.).
However, in patients who are clinically very stable on antipsychotic treatment, switching to a different antipsychotic such as aripiprazole or other atypical antipsychotics may be inappropriate (4646. Johnsen E, Kroken RA, Abaza M, Olberg H, Jørgensen HA. Antipsychotic- induced hyperprolactinemia: a cross-sectional survey. J Clin Psychopharmacol. 2008;28(6):686-90.–4848. Kunwar AR, Megna JL. Resolution of risperidone-induced hyperprolactinemia with substitution of quetiapine. Ann Pharmacother. 2003;37(2):206-8.). In these cases, using aripiprazole as adjunctive therapy may be useful. Indeed, in a placebo-controlled trial, adjunctive aripiprazole normalized drug-induced hyperprolactinemia in more than 80% of patients with schizophrenia (229229. Raghuthaman G, Venkateswaran R, Krishnadas R. Adjunctive aripiprazole in risperidone-induced hyperprolactinaemia: double-blind, randomised, placebo-controlled trial. B J Psych Open. 2015;1(2):172-7.). More recently, in a double-blind, randomized, placebo-controlled trial involving patients with risperidone-induced hyperprolactinemia, normalization of PRL levels was encountered in 46% of patients who received adjunctive aripiprazole therapy (4949. Shim JC, Shin JG, Kelly DL, Jung DU, Seo YS, Liu KH, et al. Adjunctive treatment with a dopamine partial agonist, aripiprazole, for antipsychotic-induced hyperprolactinemia:a placebo-controlled trial. Am J Psychiatry. 2007;164(9):1404-10.).
Very interestingly, aripiprazole therapy was recently shown to be effective in normalizing PRL levels and in reducing tumor size in two patients with microprolactinomas and psychotic symptoms (230230. Burback L. Management of a microprolactinoma with aripiprazole in a woman with cabergoline-induced mania. Endocrinol Diabetes Metab Case Rep. 2015;2015:150100.,231231. Bakker IC, Schubart CD, Zelissen PM. Successful treatment of a prolactinoma with the antipsychotic drug aripiprazole. Endocrinol Diabetes Metab Case Rep. 2016;2016:160028.).
Adding DA therapy
The use of DA in the treatment of antipsychotic-induced hyperprolactinemia is not well studied (4646. Johnsen E, Kroken RA, Abaza M, Olberg H, Jørgensen HA. Antipsychotic- induced hyperprolactinemia: a cross-sectional survey. J Clin Psychopharmacol. 2008;28(6):686-90.,4747. Ajmal A, Joffe H, Nachtigall LB. Psychotropic-induced hyperprolactinemia: a clinical review. Psychosomatics. 2014;55(1):29-36). DA and antipsychotic medications may have opposing mechanisms of action. Indeed, the therapeutic effects of most antipsychotics come from D2 receptor antagonism (232232. Yanofski J. The dopamine dilemma. Psychiatry (Edgmont). 2010;7(6):18-23.,233233. Owen R, Owen F, Poulter M, Crow TJ. Dopamine D2 receptors in substantia nigra in schizophrenia. Brain Res. 1994;299(1):152-4.). Thus, there is a concern that DA therapy may reduce the effectiveness of antipsychotic drugs and trigger relapse or exacerbation of psychosis (4646. Johnsen E, Kroken RA, Abaza M, Olberg H, Jørgensen HA. Antipsychotic- induced hyperprolactinemia: a cross-sectional survey. J Clin Psychopharmacol. 2008;28(6):686-90.). There is, however, little evidence to support this concern. For instance, Chang and cols. (234234. Chang SC, Chen CH, Lu ML. Cabergoline-induced psychotic exacerbation in schizophrenic patients. Gen Hosp Psychiatry. 2008;30(4):378-80.) reported a cabergoline-induced psychotic exacerbation in three schizophrenic patients when CAB was introduced for the treatment of the hyperprolactinemic symptoms. Likewise, Santos Andrade and cols. (235235. Santos Andrade EH, Pan PM, da Silva PF, Gadelha A. New insights in the management of antipsychotics in the treatment of schizophrenia in a patient with prolactinoma: a case report and review of the literature. Case Rep Med. 2010;2010:573252.) described a similar situation when BCR was added for macroprolactinoma treatment in a patient with schizophrenia on haloperidol. By contrast, in 5 studies that involved 174 schizophrenic and bipolar patients and had a duration ranging from 8 weeks to 6 months, the association of dopamine antagonists with low doses of CAB or BCR for the treatment of hyperprolactinemic symptoms apparently did not impair the underlying psychiatric disorder (236236. Pollice R, Di Giovambattista E, Tomassini A, Di Pucchio A, Mazza M, Di Michele V, et al. Risperidone-induced symptomatic hyperprolactinemia in youth with schizophrenia: efficacy and tolerability of cabergoline treatment. Clin Ter. 2007;158(2):121-6.–240240. Lee MS, Song HC, Na H, Yang J, Ko YH, Jung IK, Joe SH. Effect of bromocriptine on antipsychotic drug-induced hyperprolactinemia: eight-week randomized, single-blind, placebo-controlled, multicenter study. Psychiatry Clin Neurosci. 2010;64 (1):19-27.).
DA therapy should therefore be reserved for patients with severe hyperprolactinemia who fail to respond to aripiprazole as an alternative or adjunctive treatment. Due to its higher specificity for the D2 receptor, CAB would be preferable to BCR in the management of antipsychotic-induced hyperprolactinemia. Nevertheless, it should be used cautiously, in doses as low as possible (4646. Johnsen E, Kroken RA, Abaza M, Olberg H, Jørgensen HA. Antipsychotic- induced hyperprolactinemia: a cross-sectional survey. J Clin Psychopharmacol. 2008;28(6):686-90.).
COMMENT 16: The best approach for symptomatic antipsychotic-induced hyperprolactinemia would be the use of aripiprazole as an alternative or adjunctive therapy. DA therapy at low doses can be used cautiously in patients who do not respond to the first approach.
REFERENCES
-
1Vilar L, Fleseriu M, Bronstein MD. Pitfalls in the diagnosis of hyperprolactinemia. Arq Bras Endocrinol Metab. 2014;58(1):9-22.
-
2Vilar L, Naves LA, Fleseriu M. Avaliação diagnóstica da hiperprolactinemia (Diagnostic evaluation of hyperprolactinemia). In: Vilar L, editor. Endocrinologia Clínica, 6a ed. (Clinical Endocrinology, 6th ed.). Rio de Janeiro: Guanabara Koogan; 2016. p. 3-14.
-
3Bronstein MD. Disorders of prolactin secretion and prolactinomas. In: DeGroot LJ, Jameson JL, editors. Endocrinology. 6th ed. Philadelphia: Saunders/Elsevier; 2010. p. 333-57.
-
4Melmed S, Casanueva FF, Hoffman AR, Kleinberg DL, Montori VM, Schlechte JA, et al. Diagnosis and treatment of hyperprolactinemia: An Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(2):273-88.
-
5Glezer A, Bronstein MD. Approach to the patient with persistent hyperprolactinemia and negative sellar imaging. J Clin Endocrinol Metab. 2012;97(7):2211-6.
-
6Glezer A, Bronstein MD. Prolactinomas. Endocrinol Metab Clin N Am. 2015;44(1):71-8.
-
7Colao A, Lombardi G. Growth hormone and prolactin excess. Lancet. 1998;352:1455-61.
-
8Ciccarelli A, Daly AF, Beckers A. The epidemiology of prolactinomas. Pituitary 2005;8(1):3-6.
-
9Daly AF, Rixhon M, Adam C, Dempegioti A, Tichomirowa MA, Beckers A. High prevalence of pituitary adenomas: a cross-sectional study in the province of Liege, Belgium. J Clin Endocrinol Metab. 2006;91(12):4769-75.
-
10Auriema R, Pivonello R, Colao A. Prolactinomas. In: Vilar L (editor). In: Vilar L, editor. Endocrinologia Clínica, 6a ed. (Clinical Endocrinology, 6th ed.). Rio de Janeiro: Guanabara Koogan; 2016. p. 15-29.
-
11Colao A, Sarno AD, Cappabianca P, Briganti F, Pivonello R, Di Somma C, et al. Gender differences in the prevalence, clinical features and response to cabergoline in hyperprolactinemia. Eur J Endocrinol. 2003;148(3):325-31.
-
12Fideleff HL, Boquete HR, Suárez MG, Azaretzky M. Prolactinoma in children and adolescents. Horm Res. 2009;72(4):197-205.
-
13Heaney AP. Pituitary carcinoma: difficult diagnosis and treatment. J Clin Endocrinol Metab. 2011;96(12):3649-60.
-
14Milenković L, D'Angelo G, Kelly PA, Weiner RI. Inhibition of gonadotropin hormone-releasing hormone release by prolactin from GT1 neuronal cell lines through prolactin receptors. Proc Natl Acad Sci USA. 1994;91(4):1244-7.
-
15Buvat J, Lemaire A, Buvat-Herbaut M, Fourlinnie JC, Racadot A, Fossati P. Hyperprolactinemia and sexual function in men. Horm Res. 1985;22(3):196-203.
-
16Corona G, Mannucci E, Fisher AD, Lotti F, Ricca V, Balercia G, et al. Effect of hyperprolactinemia in male patients consulting for sexual dysfunction. J Sex Med. 2007;4(5):1485-93.
-
17Huang W, Molitch M. Evaluation and management of galactorrhea. Am Fam Physician. 2012;85(11):1073-80.
-
18Sakiyama R, Quan M. Galactorrhea and hyperprolactinemia. Obstet Gynecol Surv. 1983;38(12):689-700.
-
19Romijn JA. Hyperprolactinemia and prolactinoma. Handb Clin Neurol. 2014;124:185-95.
-
20Suh HK, Frantz AG. Size heterogeneity of human prolactin in plasma and pituitary extracts. J Clin Endocrinol Metab. 1974;39(5):928-35.
-
21Sinha YN. Structural variants of prolactin: occurrence and physiological significance. Endocr Rev. 1995;16(3):354-69.
-
22Jackson RD, Wortsman J, Malarkey WB. Characterization of a large molecular weight prolactin in women with idiopathic hyperprolactinemia and normal menses. J Clin Endocrinol Metab. 1985;61(2):258-64.
-
23Jackson RD, Wortsman J, Malarkey WB. Macroprolactinemia presenting like a pituitary tumor. Am J Med. 1985;78(2):346-50.
-
24Hattori N. The frequency of macroprolactinemia in pregnant women and the heterogeneity of its etiologies. J Clin Endocrinol Metab. 1996;81(2):586-90.
-
25Cavaco B, Leite V, Santos MA, Arranhado E, Sobrinho LG. Some forms of big big prolactin behave as a complex of monomeric prolactin with an immunoglobulin G in patients with macroprolactinemia or prolactinoma. J Clin Endocrinol Metab. 1995;80(8):2342-6.
-
26Hattori N, Ikekubo K, Nakaya Y, Kitagawa K, Inagaki C. Immunoglobulin G subclasses and prolactin (PRL) isoforms in macroprolactinemia due to anti-PRL autoantibodies. J Clin Endocrinol Metab. 2005;90(5):3036-44.
-
27Heffner LJ, Gramates LS, Yuan RW. A glycosylated prolactin species is covalently bound to immunoglobulin in human amniotic fluid. Biochem Biophys Res Commun. 1989;165(1):299-305.
-
28Thirunavakkarasu K, Dutta P, Sridhar S, Dhaliwal L, Prashad GR, Gainder S, et al. Macroprolactinemia in hyperprolactinemic infertile women. Endocrine. 2013;44(3):750-5.
-
29Kasum M, Oreskovic S, Zec I, Jezek D, Tomic V, Gall V, et al. Macroprolactinemia: new insights in hyperprolactinemia. Biochem Med (Zagreb). 2012;22(2):171-9.
-
30Vilar L, Czepielewski MA, Naves LA, Rollin GA, Casulari LA, Coelho CE. Substantial shrinkage of adenomas cosecreting growth hormone and prolactin with use of cabergoline therapy. Endocr Pract. 2007;13(4):396-402.
-
31Vieira JGH, Oliveira JH, Tachihana T, Maciel RMB, Hauache OM. Evaluation of plasma prolactin levels: is it necessary to rest before the collection? Arq Bras Endocrinol Metab. 2006;50(3):569-70.
-
32Ferriani RA, Sá MFS. Effect of venipuncture stress on plasma prolactin levels. Int J Gynaecol Obstet. 1985;23(6):459-62.
-
33Molitch ME. Disorders of prolactin secretion. Endocrinol Metab Clin North Am. 2001;30(3):585-610.
-
34Vilar L, Freitas MC, Naves LA, Casulari LA, Azevedo M, Montenegro R Jr, et al. Diagnosis and management of hyperprolactinemia: results of a Brazilian multicenter study with 1234 patients. J Endocrinol Invest. 2008;31(5):436-44.
-
35Rahmanian M, Meybodi HA, Larijani B, Mohajeri-Tehrani MR. Giantprolactinoma: case report and review of literature. J Diabetes Metab Disord. 2013;12(1):3.
-
36Acharya SV, Gopal RA, Menon PS, Bandgar TR, Shah NS. Giantprolactinoma and effectiveness of medical management. Endocr Pract. 2010;16(1):42-6.
-
37Fleseriu M, Lee M, Pineyro MM, Skugor M, Reddy SK, Siraj ES, et al. Giant invasive pituitary prolactinoma with falsely low serum prolactin: the significance of ‘hook effect’. J Neurooncol. 2006;79(1):41-3.
-
38Barkan AL, Chandler WF. Giant pituitary prolactinoma with falsely low serum prolactin: the pitfall of the “high-dose hook effect”: case report. Neurosurgery. 1998;42(4):913-5.
-
39Frieze TW, Mong DP, Koops MK. “Hook effect” in prolactinomas: case report and review of literature. Endocr Pract. 2002;8(4):296-303.
-
40Randall RV, Scheithauer BW, Laws, ER Jr, Abboud CF. Pseudoprolactinomas. Trans Am Clin Climatol Assoc. 1983; 94:114-21.
-
41Karavitaki N, Thanabalasingham G, Shore HC, Trifanescu R, Ansorge O, Meston N, et al. Do the limits of serum prolactin in disconnection hyperprolactinaemia need re-definition? A study of 226 patients with histologically verified non-functioning pituitary macroadenoma. Clin Endocrinol (Oxf). 2006;65(4):524-9.
-
42Vilar L, Albuquerque JL, Vilar CF, Ferreira LA, Cardoso IRA, Inacio IS, et al. Demographic, clinical, and laboratorial features of 770 patients with hyperprolactinemia. Arch Endocrinol Metab. 2017; 61 (Suppl. 3):S11.
-
43Voicu V, Medvedovici A, Ranetti AE, Rădulescu FŞ. Drug-induced hypo- and hyperprolactinemia: mechanisms, clinical and therapeutic consequences. Expert Opin Drug Metab Toxicol. 2013;9(8):955-68.
-
44La Torre D, Falorni A. Pharmacological causes of hyperprolactinemia. Ther Clin Risk Manag. 2007;3(5):929-51.
-
45Petit A, Piednoir D, Germain ML, Trenque T. Drug-induced hyperprolactinemia: a case-non-case study from the national pharmacovigilance database. Therapie. 2003;58(2):159-63.
-
46Johnsen E, Kroken RA, Abaza M, Olberg H, Jørgensen HA. Antipsychotic- induced hyperprolactinemia: a cross-sectional survey. J Clin Psychopharmacol. 2008;28(6):686-90.
-
47Ajmal A, Joffe H, Nachtigall LB. Psychotropic-induced hyperprolactinemia: a clinical review. Psychosomatics. 2014;55(1):29-36
-
48Kunwar AR, Megna JL. Resolution of risperidone-induced hyperprolactinemia with substitution of quetiapine. Ann Pharmacother. 2003;37(2):206-8.
-
49Shim JC, Shin JG, Kelly DL, Jung DU, Seo YS, Liu KH, et al. Adjunctive treatment with a dopamine partial agonist, aripiprazole, for antipsychotic-induced hyperprolactinemia:a placebo-controlled trial. Am J Psychiatry. 2007;164(9):1404-10.
-
50Papakostas GI, Miller KK, Petersen T, Sklarsky KG, Hilliker SE, Klibanski A, et al. Serum prolactin levels among outpatients with major depressive disorders during the acute phase of treatment with fluoxetine. J Clin Psychiatry. 2006;67(6):952-7.
-
51Vilar L, Lyra R, Albuquerque JL, Lyra R, Vilar CF, Diniz ET, et al. Very high prolactin levels associated to chronic therapy with domperidone. Arch Endocrinol Metab. 2016;60 (Suppl. 3):S22.
-
52St-Jean E, Blain F, Comtois R. High prolactin levels may be missed by immunoradiometric assay in patients with macroprolactinomas. Clin Endocrinol (Oxf). 1996;44(3):305-9.
-
53Stenman UH. Pitfalls in hormone determinations. Best Pract Res Clin Endocrinol Metab. 2013;27(6):743-4.
-
54do Carmo Dias Gontijo M, de Souza Vasconcellos L, Ribeiro-Oliveira A Jr. Hook effect and linear range in prolactin assays: distinct confounding entities. Pituitary. 2016;19(4):458-9.
-
55Glezer A, Soares CR, Vieira JG, Giannella-Neto D, Ribela MT, Goffin V, et al. Human macroprolactin displays low biological activity via its homologous receptor in a new sensitive bioassay. J Clin Endocrinol Metab. 2006;91(3):1048-55.
-
56Gibney J, Smith TP, McKenna TJ. The impact on clinical practice of routine screening for macroprolactin. J Clin Endocrinol Metab. 2005;90(7):3927-32.
-
57Leite V, Cosby H, Sobrinho LG, Fresnoza MA, Santos MA, Friesen HG. Characterization of big, big prolactin in patients with hyperprolactinaemia. Clin Endocrinol (Oxf). 1992;37(4):365-72.
-
58Hauache OM, Rocha AJ, Maia AC Jr, Maciel RM, Vieira JG. Screening for macroprolactinaemia and pituitary imaging studies. Clin Endocrinol (Oxf). 2002;57(3):327-31.
-
59Bronstein MD. Editorial: is macroprolactinemia just a diagnostic pitfall? Endocrine. 2012;41(2):169-70.
-
60Hattori N, Nakayama Y, Kitagawa K, Ishihara T, Saiki Y, Inagaki C. Anti-prolactin (PRL) autoantibody-binding sites (epitopes) on PRL molecule in macroprolactinemia. J Endocrinol. 2006;190(2):287-93.
-
61Hayashida SA, Marcondes JA, Soares JM Jr, Rocha MP, Barcellos CR, Kobayashi NK, et al. Evaluation of macroprolactinemia in 259 women under investigation for polycystic ovary syndrome. Clin Endocrinol (Oxf). 2014;80(4):616-8.
-
62Guay AT, Sabharwal P, Varma S, Malarkey WB. Delayed diagnosis of psychological erectile dysfunction because of the presence of macroprolactinemia. J Clin Endocrinol Metab. 1996;81(7):2512-4.
-
63Vieira JGH, Tachibana TT, Obara LH, Maciel RMB. Extensive experience and validation of polyethylene glycol precipitation as a screening method for macroprolactinemia. Clin Chem. 1998;44(8):1758-9.
-
64Smith TP, Suliman AM, Fahie-Wilson MN, McKenna TJ. Gross variability in the detection of prolactin in sera containing big big prolactin (macroprolactin) by commercial immunoassays. J Clin Endocrinol Metab. 2002;87(12):5410-5.
-
65Vieira JGH, Tachibana TT, Ferrer CM, de Sá J, Biscolla RP, Ana Hoff AO, et al. Hyperprolactinemia: new assay more specific for the monomeric form does not eliminate screening for macroprolactin with polyethylene glycol precipitation. Arq Bras Endocrinol Metab. 2010;54(9):856-62.
-
66Bjøro T, Mørkrid L, Wergeland R, Turter A, Kvistborg A, Sand T, Torjesen P. Frequency of hyperprolactinaemia due to large molecular weight prolactin (150-170 kD PRL). Scand J Clin Lab Invest. 1995;55(2):139-47.
-
67Hattori N, Ishihara T, Saiki Y. Macroprolactinaemia: prevalence and aetiologies in a large group of hospital workers. Clin Endocrinol (Oxf). 2009;71(5):702-8.
-
68Fahie-Wilson MN, Soule SG. Macroprolactinemia contribution to hyperprolactinemia in a district general hospital and evaluation of a screening test based on precipitation with polyethylene glycol. Ann Clin Biochem. 1997;34:252-8.
-
69Vallette-Kasic S, Morange-Ramos I, Selim A, Gunz G, Morange S, Enjalbert A, et al. Macroprolactinemia revisited: a study on 106 patients. J Clin Endocrinol Metab. 2002;87(2):581-8.
-
70Strachan MW, Teoh WL, Don-Wauchope AC, Seth J, Stoddart M, Beckett GJ. Clinical and radiological features of patients with macroprolactinaemia. Clin Endocrinol (Oxf). 2003;59(3):339-46.
-
71Theunissen C, De Schepper J, Schiettecatte J, Verdood P, Hooghe-Peeters EL, Velkeniers B. Macroprolactinemia: clinical significance and characterization of the condition. Acta Clin Belg. 2005;60(4):190-7.
-
72Bağdatoğlu C, Bağdatoğlu OT, Muslu N, Köseoğlu A, Dağtekin A, Polat G, et al. The importance of macroprolactinemia in the differential diagnosis of hyperprolactinemic patients. Turk Neurosurg. 2008;18(3):223-7.
-
73Donadio F, Barbieri A, Angioni R, Mantovani G, Beck-Peccoz P, Spada A, et al. Patients with macroprolactinaemia: clinical and radiological features. Eur J Clin Invest. 2007;37(7):552-7.
-
74Isik S, Berker D, Tutuncu YA, Ozuguz U, Gokay F, Erden G, et al. Clinical and radiological findings in macroprolactinemia. Endocrine. 2012;41(2):327-33.
-
75Parlant-Pinet L, Harthé C, Roucher F, Morel Y, Borson-Chazot F, Raverot G, et al. Macroprolactinaemia: a biological diagnostic strategy from the study of 222 patients. Eur J Endocrinol. 2015;172(6):687-95.
-
76Vilar L, Moura E, Canadas V, Gusmão A, Campos R, Leal E, et al. Prevalence of macroprolactinemia among 115 patients with hyperprolactinemia. Arq Bras Endocrinol Metab. 2007;51(1):86-91.
-
77Fahie-Wilson M. The macroprolactinemiaproblem and its solution. South Med J. 2006;99(11):1206.
-
78Shimatsu A, Hattori A. Macroprolactinemia: diagnostic, clinical, and pathogenic significance. Clin Dev Immunol. 2012;2012:167132.
-
79Leaños-Miranda A, Ramírez-Valenzuela KL, Campos-Galicia I, Chang-Verdugo R, Chinolla-Arellano LZ. Frequency of macroprolactinemia in hyperprolactinemic women presenting with menstrual irregularities, galactorrhea, and/or infertility: etiology and clinical manifestations. Int J Endocrinol. 2013;2013:478282.
-
80de Soárez PC, Souza SC, Vieira JG, Ferraz MB. The effect of identifying macroprolactinemia on health-care utilization and costs in patients with elevated serum prolactin levels. Value Health. 2009;12(6):930-4.
-
81Vilar L, Gadelha PS, Rangel Filho F, Siqueira A, Viana KF, Fonseca MM, et al. Prevalence of macroprolactinemia among patients with idiopathic hyperprolactinemia. Arq Bras Endocrinol Metab. 2014;58 (Suppl 1):S17-18.
-
82Hattori N, Adachi T, Ishihara T, Shimatsu A. The natural history of macroprolactinaemia. Eur J Endocrinol. 2012;166(4):625-9.
-
83Vilar L, Naves LA, Freitas MC, Lima M, Canadas V, Albuquerque JL, et al. Clinical and laboratory features greatly overlap in patients with macroprolactinemia or monomeric hyperprolactinemia. Minerva Endocrinol. 2007;2(2):79-86.
-
84Vilar L, Vilar CF, Albuquerque JL, Gadelha PS, Thé AC, Trovão E, et al. Clinical and MRI findings among 120 patients with macroprolactinemia: results from a prospective study. Arch Endocrinol Metab. 2017;61 (Suppl. 3):S8.
-
85Elenkova A, Genov N, Abadzhieva Z, Kirilov G, Vasilev V, Kalinov K, et al. Macroprolactinemia in patients with prolactinomas: prevalence and clinical significance. Exp Clin Endocrinol Diabetes. 2013;121(4):201-5.
-
86dos Santos Nunes V, El Dib R, Boguszewski CL, Nogueira CR. Cabergoline versus bromocriptine in the treatment of hyperprolactinemia: a systematic review of randomized controlled trials and meta-analysis. Pituitary. 2011;14(3):259-65.
-
87Webster J, Piscitelli G, Polli A, Ferrari CI, Ismail I, Scanlon MF. A comparison of cabergoline and bromocriptine in the treatment of hyperprolactinemic amenorrhea. Cabergoline Comparative Study Group. N Engl J Med. 1994;331(14):904-9.
-
88Gillam MP, Molitch MP, Lombardi G, Colao A. Advances in the treatment of prolactinomas. Endocr Rev. 2006;27(5):485-534.
-
89Molitch ME. Pharmacologic resistance in prolactinoma patients. Pituitary. 2005;8(1):43-52.
-
90Molitch ME. Management of medically refractory prolactinoma. J Neurooncol. 2014;117(3):421-8.
-
91Vilar L, Burke CW. Quinagolide efficacy and tolerability in hyperprolactinaemic patients who are resistant to or intolerant of bromocriptine. Clin Endocrinol (Oxf). 1994;41(6):821-6.
-
92Passos VQ, Fortes MAHZ, Giannella-Neto D, Bronstein MD. Genes differentially expressed in prolactinomas responsive and resistant to dopamine agonists. Neuroendocrinology. 2009;89(2):163-70.
-
93Shimazu S, Shimatsu A, Yamada S, Inoshita N, Nagamura Y, Usui T, et al. Resistance to dopamine agonists in prolactinoma is correlated with reduction of dopamine D2 receptor long isoform mRNA levels. Eur J Endocrinol. 2012;166(3):383-90.
-
94Hurel SJ, Harris PE, McNicol AM, Foster S, Kelly WF, Baylis PH. Metastatic prolactinoma: effect of octreotide, cabergoline, carboplatin and etoposide; immunocytochemical analysis of proto-oncogene expression. J Clin Endocrinol Metab. 1997;82(9):2962-5.
-
95Prior JC, Cox TA, Fairholm D, Kostashuk E, Nugent R. Testosterone-related exacerbation of a prolactin-producing macroadenoma: possible role for estrogen. J Clin Endocrinol Metab. 1987;64(2):391-4.
-
96Olafsdottir A, Schlechte J. Management of resistant prolactinomas. Nat Clin Pract Endocrinol Metab. 2006;2(10):552-61.
-
97Colao A, DiSarno A, Sarnacchiaro F, Ferone D, DiRenzo G, Merola B, et al. Prolactinomas resistant to standard dopamine agonists respond to chronic cabergoline treatment. J Clin Endocrinol Metab. 1997;82(3):876-83.
-
98Iyer P, Molitch ME. Positive prolactin response to bromocriptine in two patients with cabergoline resistant prolactinomas. Endocr Pract. 2011;17(3):e55-8.
-
99Ono M, Miki N, Kawamata T, Makino R, Amano K, Seki T, et al. Prospective study of high-dose cabergoline treatment of prolactinomas in 150 patients. J Clin Endocrinol Metab. 2008; 93(12):4721-7.
-
100Di Sarno A, Landi ML, Marzullo P, Di Somma C, Pivonello R, Cerbone G, et al. The effect of quinagolide and cabergoline, two selective dopamine receptor type 2 agonists, in the treatment of prolactinomas. Clin Endocrinol (Oxf). 2000; 53(1):53-60.
-
101Vilar L, Vilar C, Albuquerque JL, Gadelha P,Thé AC, Trovão E, et al. The use of increasing doses of cabergoline in the management of cabergoline-resistant prolactinomas. Endocrine Abstracts. 2017;49:EP975.
-
102Primeau V, Raftopoulos C, Maiter D. Outcomes of transsphenoidal surgery in prolactinomas: improvement of hormonal control in dopamine agonist-resistant patients. Eur J Endocrinol. 2012;166(5):779-86.
-
103Hamilton DK, Vance ML, Roulos PT, Laws ER. Surgical outcomes in hyporesponsive prolactinomas: analysis of patients with resistance or intolerance to dopamine agonists. Pituitary. 2005;8(1):53-60.
-
104Vrooner L, Jaffrain-Rea M-L, Petrossians P, Tamagno G, Chanson P, Vilar L, et al. Prolactinomas resistant to standard doses of cabergoline: a multicenter study of 92 patients. Eur J Endocrinol. 2012;167(5):651-62.
-
105Maurer RA. Estradiol regulates the transcription of the prolactin gene. J Biol Chem. 1982;257(5):2133-6.
-
106Lloyd GM, Meares JD, Jacobi J. Effects of oestrogen and bromocriptine on in vivo secretion and mitosis in prolactin cells. Nature. 1975;255(5508):497-8.
-
107Pasqualini C, Bojda F, Kerdelhue B. Direct effect of estradiol on the number of dopamine receptors in the anterior pituitary of ovariectomized rats. Endocrinology. 1986;119(6):2484-9.
-
108Aoki MDP, Aoki A, Maldonado CA. Sexual dimorphism of apoptosis in lactotrophs induced by bromocryptine. Histochem Cell Biol. 2001;116(3):215-22.
-
109Lamberts SWJ, Verleun T, Oosterom R. Effect of tamoxifen administration on prolactin release by invasive prolactin-secreting pituitary adenomas. Neuroendocrinology. 1982;34(5):339-42.
-
110Vilar L, Vilar CF, Aroucha PT, Albuquerque JL, Gadelha PS, Thé AC, et al. The role ofclomiphene citrate in the resolution of hypogonadism in male patients with prolactinomas under cabergoline therapy. Arch Endocrinol Metab. 2017;61 (Suppl. 3):S28.
-
111Ribeiro RS, Abucham J. Recovery of persistent hypogonadism by clomiphene in males with prolactinomas under dopamine agonist treatment. Eur J Endocrinol. 2009;161(1):163-9.
-
112Gillam MP, Middler S, Freed DJ, Molitch ME. The novel use of very high doses of cabergoline and a combination of testosterone and an aromatase inhibitor in the treatment of a giant prolactinoma. J Clin Endocrinol Metab. 2002;87(10):4447-51.
-
113Fusco A, Lugli F, Sacco E, Tilaro L, Bianchi A, Angelini F, et al. Efficacy of the combined cabergoline and octreotide treatment in a case of a dopamine-agonist resistant macroprolactinoma. Pituitary. 2011;14(4):351-7.
-
114Vilar L, Vilar C, Albuquerque JL, Gadelha P, Thé AC, Trovão E, et al. Effect of the addition of lanreotide autogel to the treatment of an aggressive prolactinoma – a case report. Endocrine Abstracts. 2017;49:EP841.
-
115Jaquet P, Qouafik L, Saveanu A, Gunz G, Fina F, Dufour H, et al. Quantitative and functional expression of somatostatin receptor subtypes in human prolactinomas. J Clin Endocrinol Metab. 1999;84(9):3268-76.
-
116Bruns C, Lewis I, Briner U, Meno-Tetang G, Weckbecker G. SOM230: a novel somatostatin peptidomimetic with broad somatotropin release inhibiting factor (SRIF) receptor binding and a unique antisecretory profile. Eur J Endocrinol. 2002;146(5):707-16.
-
117Halevy C, Whitelaw BC. How effective is temozolomide for treating pituitary tumours and when should it be used? Pituitary.2017;20(2):261-6.
-
118Ji Y, Vogel RI, Lou E. Temozolomide treatment of pituitary carcinomas and atypical adenomas: systematic review of case reports. Neurooncol Pract. 2016;3(3):188-95.
-
119Whitelaw BC, Dworakowska D, Thomas NQ, Barazi S, Riordan-Eva P, King AP, et al. Temozolomide in the management of dopamine agonist-resistant prolactinomas. Clin Endocrinol (Oxf). 2012;76(6):877-86.
-
120Jouanneau E, Wierinckx A, Ducray F, Favrel V, Borson-Chazot F, Honnorat J, et al. New targeted therapies in pituitary carcinoma resistant to temozolomide. Pituitary. 2012;15(1):37-43.
-
121Auriemma RS, Grasso LF, Pivonello R, Colao A. The safety of treatments for prolactinomas. Expert Opin Drug Saf. 2016;15(4):503-12.
-
122National Library of Medicine. Daily Med Current Medication Information. 2010 Available from: http://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=31309 Accessed on: April 7, 2017.
» http://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=31309 -
123Yüksel RN, Elyas Kaya Z, Dilbaz N, Cingi Yirün M. Cabergoline-induced manic episode: case report. Ther Adv Psychopharmacol. 2016;6(3):229-23.
-
124Voon V, Reynolds B, Brezing C, Gallea C, Skaljic M, Ekanayake V, et al. Impulsive choice and response in dopamine agonist-related impulse control behaviors. Psychopharmacology (Berl). 2010;207(4):645-59.
-
125Rovera C, Cremaschi L, Thanju A, Fiorentini A, Mauri MC, Serati M. Cabergoline can induce mania with psychotic features in bipolar I disorder: a case report. Asian J Psychiatr. 2016;22:94-5.
-
126Bilal L, Ching C. Cabergoline-induced psychosis in a patient with undiagnosed depression. J Neuropsychiatry Clin Neurosci. 2012;24(4):E54.
-
127Samuel M, Rodriguez-Oroz M, Antonini A, Brotchie JM, Ray Chaudhuri K, Brown RG, et al. Management of impulse control disorders in Parkinson's disease: Controversies and future approaches. Mov Disord. 2015;30(2):150-9.
-
128Martinkova J, Trejbalova L, Sasikova M, Benetin J, Valkovic P. Impulse control disorders associated with dopaminergic medication in patients with pituitary adenomas. Clin Neuropharmacol. 2011;34(5):179-81.
-
129Harris YT, Harris AZ, Deasis JM, Ferrando SJ, Reddy N, Young RC. Cabergoline associated with first episode mania. Psychosomatics. 2012;53(6):595-600.
-
130Davie M. Pathological gambling associated with cabergoline therapy in a patient with a pituitary prolactinoma. J Neuropsychiatry Clin Neurosci. 2007;19(4):473-4.
-
131Falhammar H, Yarker JY. Pathological gambling and hypersexuality in cabergoline-treated prolactinoma. Med J Aust. 2009;190(2):97.
-
132Bilal L, Ching C. Cabergoline-induced psychosis in a patient with undiagnosed depression. J Neuropsychiatry Clin Neurosci. 2012;24(4):E54.
-
133Almanzar S, Zapata-Vega MI, Raya JA. Dopamine agonist-induced impulse control disorders in a patient with prolactinoma. Psychosomatics. 2013;54(4):387-91.
-
134Bancos I, Nannenga MR, Bostwick JM, Silber MH, Erickson D, Nippoldt TB. Impulse control disorders in patients with dopamine agonist-treated prolactinomas and nonfunctioning pituitary adenomas: a case-control study. Clin Endocrinol (Oxf). 2014;80(6):863-8.
-
135Barake M, Evins AE, Stoeckel L, Pachas GN, Nachtigall LB, Miller KK, et al. Investigation of impulsivity in patients on dopamine agonist therapy for hyperprolactinemia: a pilot study. Pituitary. 2014;17(2):150-6.
-
136Hoffmeyer S, Burk O, von Richter O, Arnold HP, Brockmoller J, Johne A, et al. Functional polymorphisms of the human multidrug-resistance gene: multiple sequence variations and correlation of one allele with P-glycoprotein expression and activity in vivo. Proc Natl Acad Sci U S A. 2000;97(7):3473-8.
-
137Athanasoulia AP, Sievers C, Ising M, Brockhaus AC, Yassouridis A, Stalla GK, et al. Polymorphisms of the drug transporter gene ABCB1 predict side effects of treatment with cabergoline in patients with PRL adenomas. Eur J Endocrinol. 2012;167(3):327-35.
-
138Zanettini R, Antonini A, Gatto G, Gentile R, Tesei S, Pezzoli G. Valvular heart disease and the use of dopamine agonists for Parkinson's disease. N Engl J Med. 2007;356(1):39-46.
-
139Schade R, Andersohn F, Suissa S, Haverkamp W, Garbe E. Dopamine agonists and the risk of cardiac-valve regurgitation. N Engl J Med. 2007;356(1):29-38.
-
140Tran T, Brophy JM, Suissa S, Renoux C. Risks of cardiac valve regurgitation and heart failure associated with ergot- and non-ergot-derived dopamine agonist use in patients with Parkinson's disease: asystematic review of observational studies. CNS Drugs. 2015;29(12):985-98.
-
141Roth BL. Drugs and valvular heart disease. N Engl J Med. 2007;356(1):6-9.
-
142Elangbam, C.S. Drug-induced valvulopathy: an update. Toxicol Pathol. 2010;38(6):837-48.
-
143Serratrice J, Disdier P, Habib G, Viallet F, Weiller PJ. Fibrotic valvular heart disease subsequent to bromocriptine treatment. Cardiol Rev. 2002;10(6):334-6.
-
144Tan LC, Ng KK, Au WL, Lee RK, Chan YH, Tan NC. Bromocriptine use and the risk of valvular heart disease. Mov Disord. 2009;24(3):344-9.
-
145Valassi E, Klibanski A, Biller BM. Potential cardiac valve effects of dopamine agonists in hyperprolactinemia. J Clin Endocrinol Metab. 2010;95(3):1025-33.
-
146Choong CY, Abascal VM, Weyman J, Levine RA, Gentile F, Thomas JD, et al. Prevalence of valvular regurgitation by Doppler echocardiography in patients with structurally normal hearts by two-dimensional echocardiography. Am Heart J. 1989;117(3):636-42.
-
147Okura H, Takada Y, Yamabe A, Ozaki T, Yamagishi H, Toda I, et al. Prevalence and correlates of physiological valvular regurgitation in healthy subjects. Circ J. 2011;75(11):2699-704.
-
148Gu H, Luck S, Carroll PV, Powrie J, Chambers J. Cardiac valve disease and low-dose dopamine agonist therapy: an artefact of reporting bias? Clin Endocrinol (Oxf). 2011;74(5):608-10.
-
149De Vecchis R, Esposito C, Ariano C. Cabergoline use and risk of fibrosis and insufficiency of cardiac valves. Meta-analysis of observational studies. Herz. 2013;38(8):868-80.
-
150Caputo C, Prior D, Inder WJ. The need for annual echocardiography to detect cabergoline-associated valvulopathy in patients with prolactinoma: a systematic review and additional clinical data. Lancet Diabetes Endocrinol. 2015;3(11):906-13.
-
151Boguszewski CL, dos Santos CM, Sakamoto KS, Marini LC, de Souza AM, Azevedo M. A comparison of cabergoline and bromocriptine on the risk of valvular heart disease in patients with prolactinomas. Pituitary. 2012;15(1):44-9.
-
152Elenkova A, Shabani R, Kalinov K, Zacharieva S. Increased prevalence of subclinical cardiac valve fibrosis in patients with prolactinomas on long-term bromocriptine and cabergoline treatment. Eur J Endocrinol. 2012;167(1):17-25.
-
153Delgado V, Biermasz NR, van Thiel SW, Ewe SH, Marsan NA, Holman ER, et al. Changes in heart valve structure and function in patients treated with dopamine agonists for prolactinomas, a 2-year follow-up study. Clin Endocrinol (Oxf). 2012;77(1):99-105.
-
154Auriemma RS, Pivonello R, Perone Y, Grasso LF, Ferreri L, Simeoli C, et al. Safety of long-term treatment with cabergoline on cardiac valve disease in patients with prolactinomas. Eur J Endocrinol. 2013;169(3):359-66.
-
155Zaccaron BA, Sena RH, Souza AM, Boguszewski CL. Safety of long-term treatment with dopamine agonists on valvular heart disease in patients with prolactinomas. Arq Bras Endocrinol Metab. 2014;58(Suppl 5):S172.
-
156Steffensen C, Maegbaek ML, Laurberg P, Andersen M, Kistorp CM, Norrelund H, et al. Heart valve disease among patients with hyperprolactinemia: a nationwide population-based cohort study. J Clin Endocrinol Metab. 2012;97(5):1629-34.
-
157Trifirò G, Mokhles MM, Dieleman JP, van Soest EM, Verhamme K, Mazzaglia G, et al. Risk of cardiac valve regurgitation with dopamine agonist use in Parkinson's disease and hyperprolactinaemia: a multi-country, nested case-control study. Drug Saf. 2012;35(2):159-71.
-
158Maione L, Garcia C, Bouchachi A, Kallel N, Maison P, Salenave S, et al. No evidence of a detrimental effect of cabergoline therapy on cardiac valves in patients with acromegaly. J Clin Endocrinol Metab. 2012;97(9):E1714-E19.
-
159Eversmann T, Fahlbusch R, Rjosk HK, von Werder K. Persisting suppression of prolactin secretion after long-term treatment with bromocriptine in patients with prolactinomas. Acta Endocrinol (Copenh). 1979;92(3):413-27.
-
160Passos VQ, Souza JJ, Musolino NR, Bronstein MD. Long-term follow-up of prolactinomas: normoprolactinemia after bromocriptine withdrawal. J Clin Endocrinol Metab. 2002; 87(8):3578-82.
-
161Colao A, Di Sarno A, Cappabianca P, Di Somma C, Pivonello R, Lombardi G. Withdrawal of long-term cabergoline therapy for tumoral and nontumoral hyperprolactinemia. N Engl J Med. 2003;349(21):2023-33.
-
162Colao A, Di Sarno A, Guerra E, Pivonello R, Cappabianca P, Caranci F, et al. Predictors of remission of hyperprolactinaemia after long-term withdrawal of cabergoline therapy. Clin Endocrinol (Oxf). 2007;67(3):426-33.
-
163Biswas M, Smith J, Jadon D, McEwan P, Rees DA, Evans LM, et al. Long-term remission following withdrawal of dopamine agonist therapy in subjects with microprolactinomas. Clin Endocrinol (Oxf). 2005;63(1):26-31.
-
164Kharlip J, Salvatori R, Yenokyan G, Wand GS. Recurrence of hyperprolactinemia after withdrawal of long-term cabergoline therapy. J Clin Endocrinol Metab. 2009;94(7):2428-36.
-
165Huda MS, Athauda NB, Teh MM, Carroll PV, Powrie J. Factors determining the remission of microprolactinomas after dopamine agonist withdrawal. Clin Endocrinol (Oxf). 2010;72(4):507-11.
-
166Barber TM, Kenkre J, Garnett C, Scott RV, Byrne JV, Wass JA. Recurrence of hyperprolactinaemia following discontinuation of dopamine agonist therapy in patients with prolactinoma occurs commonly especially in macroprolactinoma. Clin Endocrinol (Oxf). 2011;75(6):819-24.
-
167Anagnostis P, Adamidou F, Polyzos SA, Efstathiadou Z, Karathanassi E, Kita M. Long term follow-up of patients with prolactinomas and outcome of dopamine agonist withdrawal: a single center experience. Pituitary. 2012;15(1):25-9.
-
168Sala E, Bellaviti Buttoni P, Malchiodi E, Verrua E, Carosi G, et al. Recurrence of hyperprolactinemia following dopamine agonist withdrawal and possible predictive factors of recurrence in prolactinomas. J Endocrinol Invest. 2016;39(12):1377-82.
-
169Dogansen SC, Selcukbiricik OS, Tanrikulu S, Yarman S. Withdrawal of dopamine agonist therapy in prolactinomas: In which patients and when? Pituitary. 2016;19(3):303-10.
-
170Watanabe S, Akutsu H, Takano S, Yamamoto T, Ishikawa E, Suzuki H, et al. Long-term results of cabergoline therapy for macroprolactinomas and analysis of factors associated with remission after withdrawal. Clin Endocrinol (Oxf). 2017;86(2):207-13.
-
171Dekkers OM, Lagro J, Burman P, Jørgensen JO, Romijn JA, Pereira AM. Recurrence of hyperprolactinemia after withdrawal of dopamine agonists: systematic review and meta-analysis. J Clin Endocrinol Metab. 2010; 95(1):43-51.
-
172Hu J, Zheng X, Zhang W, Yang H. Current drug withdrawal strategy in prolactinoma patients treated with cabergoline: a systematic review and meta-analysis. Pituitary. 2015;18(5):745-51.
-
173Vilar L, Albuquerque JL, Gadelha PS, Rangel Filho F, Siqueira AM, da Fonseca MM, et al. Second attempt of cabergoline withdrawal in patients with prolactinomas after a failed first attempt: is it worthwhile? Front Endocrinol (Lausanne). 2015;6:11.
-
174Kwancharoen R, Auriemma RS, Yenokyan G, Wand GS, Colao A, Salvatori R. Second attempt to withdraw cabergoline in prolactinomas: a pilot study. Pituitary. 2014;17(5):451-6.
-
175Karunakaran S, Page RC, Wass JA. The effect of the menopause on prolactin levels in patients with hyperprolactinaemia. Clin Endocrinol (Oxf). 2001;54(3):295-300.
-
176Auriemma RS, Perone Y, Di Sarno A, Grasso LF, Guerra E, Gasperi M, et al. Results of a single-center observational 10-year survey study on recurrence of hyperprolactinemia after pregnancy and lactation. J Clin Endocrinol Metab. 2013;98(1):372-9.
-
177Domingue ME, Devuyst F, Alexopoulou O, Corvilain B, Maiter D. Outcome of prolactinoma after pregnancy and lactation: a study on 73 patients. Clin Endocrinol (Oxf). 2014;80(5):642-8.
-
178Smith TR, Hulou MM, Huang KT, Gokoglu A, Cote DJ, Woodmansee WW, Laws ER Jr. Current indications for the surgical treatment of prolactinomas. J Clin Neurosci. 2015;22(11):1785-91
-
179Lund VJ. Endoscopic management of cerebrospinalfluid leaks. Am J Rhinol. 2002;16(1):17-23.
-
180Mankia SK, Weerakkody RA, Wijesuriya S, Kandasamy N, Finucane F, Guilfoyle M, et al. Spontaneous cerebrospinalfluid rhinorrhoea as the presenting feature of an invasive macroprolactinoma. BMJ Case Rep. 2009;2009. pii: bcr12.2008.1383
-
181Leong KS, Foy PM, Swift AC, Atkin SL, Hadden DR, MacFarlane IA. CSF rhinorrhoea following treatment with dopamine agonists for massive invasive prolactinomas. Clin Endocrinol (Oxf). 2000;52(1):43-9.
-
182Lam G, Mehta V, Zada G. Spontaneous and medically induced cerebrospinal fluid leakage in the setting of pituitary adenomas: review of the literature. Neurosurg Focus. 2012;32(6):E2.
-
183Bronstein MD, Musolino NR, Benabou S, Marino R Jr. Cerebrospinal fluid rhinorrhea occurring in long-term bromocriptine treatment for macroprolactinomas. Surg Neurol. 1989;32(5):346-9.
-
184Anani WQ, Ojerholm E, Shurin MR. Resolving transferrin isoforms via agarose gel electrophoresis. Lab Med. 2015;46(1):26-33.
-
185Colao A. The prolactinoma. Best Pract Res Clin Endocrinol Metab. 2009;23(5):575-96.
-
186Barker 2nd FG, Klibanski A, Swearingen B. Transsphenoidal surgery for pituitary tumors in the United States, 1996–2000: mortality, morbidity, and the effects of hospital and surgeon volume. J Clin Endocrinol Metab. 2003;88(10):4709-19.
-
187Sudhakar N, Ray A, Vafidis JA. Complications after trans-sphenoidal surgery: our experience and a review of the literature. Br J Neurosurg. 2004;18(5):507-12.
-
188Nelson Jr AT, Tucker Jr HS, Becker DP. Residual anterior pituitary function following transsphenoidal resection of pituitary macroadenomas. J Neurosurg 1984;61(3):577-80.
-
189Sheplan Olsen LJ, Robles Irizarry L, Chao ST, Weil RJ, Hamrahian AH, Hatipoglu B, Suh JH. Radiotherapy for prolactin-secreting pituitary tumors. Pituitary. 2012;15(2):135-45.
-
190Wan H, Chihiro O, Yuan S. MASEP gamma knife radiosurgery for secretory pituitary adenomas: experience in 347 consecutive cases. J Exp Clin Cancer Res. 2009;28:36.
-
191Kong DS, Lee JI, Lim do H, Kim KW, Shin HJ, Nam DH, et al. The efficacy of fractionated radiotherapy and stereotactic radiosurgery for pituitary adenomas: long-term results of 125 consecutive patients treated in a single institution. Cancer. 2017;110(4):854-60.
-
192Castinetti F, Nagai M, Morange I, Dufour H, Caron P, Chanson P, et al. Long-term results of stereotactic radiosurgery in secretory pituitary adenomas. J Clin Endocrinol Metab. 2009;94(9):3400-7.
-
193Tsang RW, Brierley JD, Panzarella T, Gospodarowicz MK, Sutcliffe SB, Simpson WJ. Radiation therapy for pituitary adenoma: treatment outcome and prognostic factors. Int J Radiat Oncol Biol Phys. 1994;30(3):557-65.
-
194Littley MD, Shalet SM, Beardwell CG, Robinson EL, Sutton ML Radiation-induced hypopituitarism is dose-dependent. Clin Endocrinol (Oxf). 1989;31(3):363-73.
-
195Brada M, Burchell L, Ashley S, Traish D. The incidence of cerebrovascular accidents in patients with pituitary adenoma. Neurooncol Pract. 2014;1(1):22-8.
-
196Minniti G, Traish D, Ashley S, Gonsalves A, Brada M. Risk of second brain tumor after conservative surgery and radiotherapy for pituitary adenoma: update after an additional 10 years. J Clin Endocrinol Metab. 2005;90(2):800-4.
-
197Erridge SC, Conkey DS, Stockton D, Strachan MW, Statham PF, Whittle IR, et al. Radiotherapy for pituitary adenomas: long-term efficacy and toxicity. Radiother Oncol. 2009;93(3):597-601.
-
198Cohen-Inbar O, Xu Z, Schlesinger D, Vance ML, Sheehan JP. Gamma Knife radiosurgery for medically and surgically refractory prolactinomas: long-term results. Pituitary. 2015;18(6):820-30.
-
199Liu X, Kano H, Kondziolka D, Park KJ, Iyer A, Shin S, et al. Gamma knife stereotactic radiosurgery for drug resistant or intolerant invasive prolactinomas. Pituitary. 2013;16(1):68-75.
-
200Moraes AB, Silva CM, Vieira Neto L, Gadelha MR. Giant prolactinomas: the therapeutic approach. Clin Endocrinol (Oxf). 2013;79(4):447-56.
-
201Delgrange E, Raverot G, Bex M, Burman P, Decoudier B, Devuyst F, et al. Giant prolactinomas in women. Eur J Endocrinol. 2013;170(1):31-8.
-
202Maiter D, Delgrange E. Therapy of endocrine disease: the challenges in managing giant prolactinomas. Eur J Endocrinol. 2014;170(6):R213-27.
-
203Delgrange E, Trouillas J, Maiter D, Donckier J, Tourniaire J. Sex-related difference in the growth of prolactinomas: a clinical and proliferation marker study. J Clin Endocrinol Metab. 1997;82(7):2102-7.
-
204Shrivastava RK, Arginteanu MS, King WA, Post KD. Giant prolactinomas: clinical management and long-term follow up. J Neurosurg. 2002;97(2):299-306.
-
205Levy RA, Quint DJ. Giant pituitary adenoma with unusual orbital and skull base extension. AJR Am J Roentgenol. 1998;170(1):194-6.
-
206Chaurasia PK, Singh D, Meher S, Saran RK, Singh H. Epistaxis as first clinical presentation in a child with giant prolactinoma: Case report and review of literature. J Pediatr Neurosci. 2011;6(2):134-7.
-
207Alkatari S, Aljohani N. Obstructive hydrocephalus, fifth nerve and hypothalamus involvement: acute presentation of a giant prolactinoma. Clin Med Insights Case Rep. 2012;5:115-8.
-
208Espinosa E, Sosa E, Mendoza V, Ramírez C, Melgar V, Mercado M. Giant prolactinomas: are they really different from ordinary macroprolactinomas? Endocrine. 2015;52(3):652-9.
-
209Shimon I, Sosa E, Mendoza V, Greenman Y, Tirosh A, Espinosa E et al. Giant prolactinomas larger than 60 mm in size: a cohort of massive and aggressive prolactin-secreting pituitary adenomas. Pituitary. 2016;19(4):429-36.
-
210Vilar L, Albuquerque JL, Gomes B, Gadelha PS, Lopes IGA, Thé AC, et al. Giant prolactinomas: clinical manifestations and response to treatment in 16 cases. Arch Endocrinol Metab. 2015; 59 (Suppl. 1):S21.
-
211Saeki N, NakamuraM, Sunami K, Yamaura A. Surgical indication after bromocriptine therapy on giant prolactinomas: effects and limitations of the medical treatment. Endocr J. 1998;45(4):529-37.
-
212Sieck JO, Niles NL, Jinkins JR, Al-Mefty O, el-Akkad S, Woodhouse N. Extrasellar prolactinomas: successful management of 24 patients using bromocriptine. Horm Res.1986;23(3):167-76.
-
213Kyung Rae C, Kyung-Il J, Hyung Jin S. Bromocriptine therapy for the treatment of invasive prolactinoma: the single institute experience. Brain Tumor Res Treat. 2013; 1(2):71–7
-
214Komotar RJ, Starke RM, Raper DM, Anand VK, Schwartz TH. Endoscopic endonasal compared with microscopic transsphenoidal and open transcranial resection of giant pituitary adenomas. Pituitary. 2012;15(2):150-9.
-
215Brown RS, Herbison AE, Grattan DR. Prolactin regulation of kisspeptin neurons in the mouse brain and its role in the lactation-induced suppression of kisspeptin expression. J Neuroendocrinol. 2014:26(12):898-908.
-
216Molitch ME. Endocrinology in pregnancy: management of the pregnant patient with a prolactinoma. Eur J Endocrinol. 2015;172(5):R205-13.
-
217Glezer A, Jallad RS, Machado MC, Fragoso MC, Bronstein MD. Pregnancy and pituitary adenomas. Minerva Endocrinol. 2016;41(3):341-50.
-
218Holmgren U, Bergstrand G, Hagenfeldt K, Werner S. Women with prolactinoma—effect of pregnancy and lactation on serum prolactin and on tumour growth. Acta Endocrinol (Copenh). 1986;111(4):452-9.
-
219Bronstein MD, Paraiba DB, Jallad RS. Management of pituitary tumors in pregnancy. Nat Rev Endocrinol. 2011;7(5):301-10.
-
220Raymond JP, Goldstein E, Konopka P, Leleu MR, Merceron LE, Loria Y. Follow-up of children born of bromocriptine-treated mothers. Horm Res. 1985;22(3):239-46.
-
221Canales ES, Garcia IC, Ruiz JE, Zárate A. Bromocriptine as prophylactic therapy in prolactinoma during pregnancy. Fertil Steril. 1981;36(4):524-6.
-
222Glezer A, Bronstein MD. Prolactinomas, cabergoline, and pregnancy. Endocrine. 2014;47(1):64-9.
-
223Bronstein MD. Prolactinomas and pregnancy. Pituitary. 2005;8(1):31-8.
-
224Krupp P, Monka C, Richter K. The safety aspects of infertility treatments. Program of the Second World Congress of Gynecology and Obstetrics 1988. Rio de Janeiro, Brazil. p. 9.
-
225Ono M, Miki N, Amano K, Kawamata T, Seki T, Makino R, et al. High-dose cabergoline therapy for hyperprolactinemic infertility in women with micro- and macroprolactinomas. J Clin Endocrinol Metab. 2010;95(6):2672-9.
-
226Lebbe M, Hubinont C, Bernard P, Maiter D. Outcome of 100 pregnancies initiated under treatment with cabergoline in hyperprolactinaemic women. Clin Endocrinol (Oxf). 2010;73(2):236-42.
-
227Stalldecker G, Gil MSM, Guitelman MA, Alfieri A, Ballarino MC, Boero L, et al. Effects of cabergoline on pregnancy and embryo-fetal development: retrospective study on 103 pregnancies and a review of the literature. Pituitary. 2010;13(4):345-50.
-
228Yoon HW, Lee JS, Park SJ, Lee SK, Choi WJ, Kim TY, et al. Comparing the effectiveness and safety of the addition of and switching to aripiprazole for resolving antipsychotic-induced hyperprolactinemia: a multicenter, open-label, prospective study. Clin Neuropharmacol. 2016;39(6):288-94.
-
229Raghuthaman G, Venkateswaran R, Krishnadas R. Adjunctive aripiprazole in risperidone-induced hyperprolactinaemia: double-blind, randomised, placebo-controlled trial. B J Psych Open. 2015;1(2):172-7.
-
230Burback L. Management of a microprolactinoma with aripiprazole in a woman with cabergoline-induced mania. Endocrinol Diabetes Metab Case Rep. 2015;2015:150100.
-
231Bakker IC, Schubart CD, Zelissen PM. Successful treatment of a prolactinoma with the antipsychotic drug aripiprazole. Endocrinol Diabetes Metab Case Rep. 2016;2016:160028.
-
232Yanofski J. The dopamine dilemma. Psychiatry (Edgmont). 2010;7(6):18-23.
-
233Owen R, Owen F, Poulter M, Crow TJ. Dopamine D2 receptors in substantia nigra in schizophrenia. Brain Res. 1994;299(1):152-4.
-
234Chang SC, Chen CH, Lu ML. Cabergoline-induced psychotic exacerbation in schizophrenic patients. Gen Hosp Psychiatry. 2008;30(4):378-80.
-
235Santos Andrade EH, Pan PM, da Silva PF, Gadelha A. New insights in the management of antipsychotics in the treatment of schizophrenia in a patient with prolactinoma: a case report and review of the literature. Case Rep Med. 2010;2010:573252.
-
236Pollice R, Di Giovambattista E, Tomassini A, Di Pucchio A, Mazza M, Di Michele V, et al. Risperidone-induced symptomatic hyperprolactinemia in youth with schizophrenia: efficacy and tolerability of cabergoline treatment. Clin Ter. 2007;158(2):121-6.
-
237Tollin SR. Use of the dopamine agonists bromocriptine and cabergoline in the management of risperidone-induced hyperprolactinemia in patients with psychotic disorders. J Endocrinol Invest. 2000;23(11):765-70.
-
238Cavallaro R, Cocchi F, Angelone SM, Lattuada E, Smeraldi E. Cabergoline treatment of risperidone-induced hyperprolactinemia: a pilot study. J Clin Psychiatry. 2004;65(2):187-90.
-
239Kalkavoura CS, Michopoulos I, Arvanitakis P, Theodoropoulou P, Dimopoulou K, Tzebelikos E, Lykouras L. Effects of cabergoline on hyperprolactinemia, psychopathology, and sexual functioning in schizophrenic patients. Exp Clin Psychopharmacol. 2013;21(4):332-41.
-
240Lee MS, Song HC, Na H, Yang J, Ko YH, Jung IK, Joe SH. Effect of bromocriptine on antipsychotic drug-induced hyperprolactinemia: eight-week randomized, single-blind, placebo-controlled, multicenter study. Psychiatry Clin Neurosci. 2010;64 (1):19-27.
Publication Dates
-
Publication in this collection
Mar-Apr 2018
History
-
Received
03 Feb 2017 -
Accepted
09 Aug 2017