Abstract
Background:
HAS-BLED score was developed to assess 1-year major bleeding risk in patients anticoagulated with vitamin K antagonists (VKA) due to atrial fibrillation (AF).
Objective:
Of this study was to assess the ability of HAS-BLED score and its components to predict major bleeding in patients treated in an anticoagulation outpatient clinic of a tertiary hospital.
Methods:
A retrospective cohort study on AF patients treated with VKA was conducted. Logistic regression analysis was performed to evaluate the ability of individual score components to predict major bleeding. The significance level adopted in all tests was 5%.
Results:
We studied 263 patients with a mean age of 71.1 ± 10.5 years over a period of 237.6 patients-year. Median HAS-BLED score was 2 (1-3). The overall incidence of major bleeding was 5.7%, and it was higher among high-risk HAS-BLED score patients than in low risk patients (9.6 vs. 3.1%; p = 0.052). Area under the ROC curve was 0.70 (p = 0.01). Cut-off point ≥ 3 showed sensibility of 66.7%, specificity of 62.1%, positive predictive value of 9.6% and negative predictive value of 96.9%. Major bleeding-free survival was lower in high-risk group (p = 0.017). In multivariate analysis, concurrent antiplatelet use was the only independent predictor of major bleeding among score components (OR 5.13, 95%CI: 1.55-17.0; p = 0.007).
Conclusion:
HAS-BLED score was able to predict major bleeding in this cohort of AF patients. Among score components, special attention should be given for concomitant antiplatelet use, which was independently associated to this outcome. Antiplatelets in AF patients under VKA anticoagulation should be used in selected patients with favorable risk-benefit assessment.
Keywords:
Atrial Fibrillation; Hemorrhage; Outpatient Clinics; Hospital
Resumo
Fundamento:
O escore HAS-BLED foi desenvolvido para avaliar o risco em um ano de sangramento maior em pacientes com fibrilação atrial (FA) anticoagulados com antagonistas da vitamina K (AVK).
Objetivo:
O objetivo deste estudo foi avaliar a capacidade do escore HAS-BLED e de seus componentes em predizer sangramento maior em pacientes atendidos em um ambulatório de anticoagulação de um hospital terciário.
Métodos:
Foi realizado um estudo coorte retrospectivo com pacientes com FA tratados com AVK. Análise de regressão logística foi realizada para avaliar a capacidade de cada componente do escore em predizer sangramento maior. O nível de significância adotado em todos os testes foi de 5%.
Resultados:
Foram estudados 263 pacientes com média de idade de 71,1 ± 10,5 anos ao longo de um período de tratamento de 237,6 pacientes-ano. A mediana do escore HAS-BLED foi de 2 (1-3). A incidência de sangramento maior foi de 5,7%, sendo mais elevada nos pacientes de alto risco que nos pacientes de baixo risco (9,6 vs. 3,1%; p = 0,052). A área sob a curva ROC foi de 0,70 (p = 0,01). Um ponto de corte ≥ 3 mostrou sensibilidade de 66,7%, especificidade de 62,1%, valor preditivo positivo de 9,6% e valor preditivo negativo de 96,9%. Sobrevida livre de sangramento maior foi menor no grupo de alto risco (p = 0,017). Na análise multivariada, o único preditor independente de sangramento maior entre os componentes do escore foi o uso concomitante de antiplaquetários (OR 5,13, IC95%: 1,55-17,0; p = 0,007).
Conclusão:
O escore HAS-BLED foi capaz de prever sangramento maior na população de pacientes com FA estudada. Entre os componentes do escore, atenção especial deve ser dada para o uso concomitante de antiplaquetários, que mostrou associação independente. Em pacientes com FA em uso de AVK como terapia anticoagulante, o uso de antiplaquetários deve ser realizado somente naqueles pacientes com avaliação risco-benefício favorável.
Palavras-chave:
Fibrilação Atrial; Hemorragia; Ambulatório Hospitalar
Introduction
Atrial fibrillation (AF) is a common cardiac rhythm disturbance that increases in prevalence with advancing age: approximately 1% of patients with AF are under 60 years of age, whereas up to 12% of patients with AF are 75 to 84 years of age.11 January CT, Wann LS, Alpert JS, Calkins H, Cigarroa JE, Cleveland JC Jr, et al; ACC/AHA Task Force Members. 2014 AHA/ACC/HRS Guideline for the management of patients with atrial fibrillation: a report of the American College of Cardiology / American Heart Association Task Force on practice guidelines and the Heart Rhythm Society. Circulation. 2014;130(23):e199-267. doi: 10.1161/CIR.0000000000000041. Erratum in: Circulation. 2014;130(23):e272-4.
https://doi.org/10.1161/CIR.000000000000...
AF is associated with a broad spectrum of negative outcomes, such as heart failure and major cardiovascular events, including ischemic stroke, with increased cardiovascular and all-cause mortality.22 Odutayo A, Wong CX, Hsiao AJ, Hopewell S, Altman DG, Emdin CA. Atrial fibrillation and risks of cardiovascular disease, renal disease, and death: systematic review and meta-analysis. BMJ. 2016;354:i4482. doi: 10.1136/bmj.i4482.
https://doi.org/10.1136/bmj.i4482...
Ischemic strokes related to AF usually result from cardioembolism of a large cerebral artery, and therefore tend to be larger, more frequently fatal or associated with greater disability than strokes from other causes.33 Freedman B, Potpara TS, Lip GY. Stroke prevention in atrial fibrillation. Lancet. 2016;388(10046):806-17. doi: 10.1016/S0140-6736(16)31257-0.
https://doi.org/10.1016/S0140-6736(16)31...
Studies with oral anticoagulants (OAC) have shown reduction in all-cause mortality by 26% and in systemic thromboembolism by 64% with the use of vitamin K antagonists (VKA) compared with placebo or untreated controls.44 Hart RG, Pearce LA, Aguilar MI. Meta-analysis: antithrombotic therapy to prevent stroke in patients who have nonvalvular atrial fibrillation. Ann Intern Med. 2007;146(12):857-67. PMID: 17577005. On the other hand, VKA are associated with increased risk of bleeding.55 Bengtson LG, Lutsey PL, Chen LY, MacLehose RF, Alonso A. Comparative effectiveness of dabigatran and rivaroxaban versus warfarin for the treatment of non-valvular atrial fibrillation. J Cardiol. 2017;69(6):868-876. doi: 10.1016/j.jjcc.2016.08.010.
https://doi.org/10.1016/j.jjcc.2016.08.0...
Gomes et al66 Gomes T, Mamdani MM, Holbrook AM, Paterson JM, Hellings C, Juurlink DN. Rates of hemorrhage during warfarin therapy for atrial fibrillation. CMAJ. 2013;185(2):E121-7. doi: 10.1503/cmaj.121218.
https://doi.org/10.1503/cmaj.121218...
demonstrated a 3.8% risk of hemorrhage per person-year with warfarin, especially within the first 30 days of therapy (11.8%), with almost 1% of all new users being admitted to hospital due to hemorrhagic complications during this period.
There are several risk scores available to assess bleeding risk in patients in treatment with VKA.(7-12) HAS-BLED score [Hypertension (uncontrolled), Abnormal renal/liver function, Stroke, Bleeding history or predisposition (anemia), Labile INR, Elderly (> 65 years), Drugs/alcohol concomitantly] offers a better prediction of bleeding compared with many other bleeding risk scores for AF patients, and has also been validated in several different cohorts, including large real-world and clinical trial populations.1313 Lane DA, Lip GY. Use of the CHA(2)DS(2)-VASc and HAS-BLED scores to aid decision making for thromboprophylaxis in nonvalvular atrial fibrillation. Circulation. 2012;126(7):860-5. doi: 10.1161/CIRCULATIONAHA.111.060061.
https://doi.org/10.1161/CIRCULATIONAHA.1...
The score ranges from 0 to 9, with scores ≥ 3 indicating higher risk of bleeding. In this situation, thorough and regular patient follow-up is recommended.1313 Lane DA, Lip GY. Use of the CHA(2)DS(2)-VASc and HAS-BLED scores to aid decision making for thromboprophylaxis in nonvalvular atrial fibrillation. Circulation. 2012;126(7):860-5. doi: 10.1161/CIRCULATIONAHA.111.060061.
https://doi.org/10.1161/CIRCULATIONAHA.1...
In addition, HAS-BLED score should be used to identify modifiable bleeding risk factors that might be addressed, but should not be used to exclude patients from OAC therapy, as suggested by CHA2DS2-VASc score [Congestive heart failure, Hypertension, Age ≥ 75 years, Diabetes, Stroke, Vascular disease, Age 65-74 years, Sex category (i.e., female gender)].33 Freedman B, Potpara TS, Lip GY. Stroke prevention in atrial fibrillation. Lancet. 2016;388(10046):806-17. doi: 10.1016/S0140-6736(16)31257-0.
https://doi.org/10.1016/S0140-6736(16)31...
,1414 Lip GY. Stroke and bleeding risk assessment in atrial fibrillation: when, how, and why? Eur Heart J. 2013;34(14):1041-9. doi: 10.1093/eurheartj/ehs435.
https://doi.org/10.1093/eurheartj/ehs435...
,1515 Olesen JB, Lip GY, Lindhardsen J, Lane DA, Ahlehoff O, Hansen ML, et al. Risks of thromboembolism and bleeding with thromboprophylaxis in patients with atrial fibrillation: a net clinical benefit analysis using a 'real world' nationwide cohort study. Thromb Haemost. 2011;106(4):739-49. doi: 10.1160/TH11-05-0364.
https://doi.org/10.1160/TH11-05-0364...
The objective of this study was to assess the ability of HAS-BLED score and its components to predict major bleeding in patients treated at a anticoagulation outpatient clinic of a tertiary hospital.
Methods
A retrospective cohort study was conducted on patients on oral anticoagulation with VKA attending the outpatient anticoagulation clinic at Hospital de Clínicas de Porto Alegre (HCPA). This hospital is a tertiary care teaching hospital located in southern Brazil and most patients are low-income. All decisions regarding the management of anticoagulation are based on a protocol published by Kim et al.1616 Kim YK, Nieuwlaat R, Connolly SJ, Schulman S, Meijer K, Raju N, et al. Effect of a simple two-step warfarin dosing algorithm on anticoagulant control as measured by time in therapeutic range: a pilot study. J Thromb Haemost. 2010;8(1):101-6. doi: 10.1111/j.1538-7836.2009.03652.x
https://doi.org/10.1111/j.1538-7836.2009...
All patients who visited the clinic from January to March 2014 were screened for inclusion in the study. Non-valvular AF patients under VKA anticoagulation were included. Valvular AF was defined as AF associated to moderate to severe mitral stenosis or prosthetic heart valve.1717 Camm AJ, Lip GY, De Caterina R, Savelieva I, Atar D, Hohnloser SH, et al. 2012 focused update of the ESC Guidelines for the management of atrial fibrillation: an update of the 2010 ESC Guidelines for the management of atrial fibrillation. Eur Heart J. 2012;33(21):2719-47. doi: 10.1093/eurheartj/ehs253. Erratum in: Eur Heart J. 2013;34(10):790. Eur Heart J. 2013;34(36):2850-1.
https://doi.org/10.1093/eurheartj/ehs253...
This study was approved by local Research Ethics Committee.
Ischemic stroke risk was estimated with the scores CHADS2 (Congestive heart failure, Hypertension, Age > 75, Diabetes, Stroke) and CHA2DS2-VASc.1818 Gage BF, Waterman AD, Shannon W, Boechler M, Rich MW, Radford MJ. Validation of clinical classification schemes for predicting stroke: results from the National Registry of Atrial Fibrillation. JAMA. 2001;285(22):2864-70. PMID: 11401607.,1919 Lip GY, Nieuwlaat R, Pisters R, Lane DA, Crijns HJ. Refining clinical risk stratification for predicting stroke and thromboembolism in atrial fibrillation using a novel risk factor-based approach: the euro heart survey on atrial fibrillation. Chest. 2010;137(2):263-72. doi: 10.1378/chest.09-1584.
https://doi.org/10.1378/chest.09-1584...
High-risk for ischemic stroke was defined as a score ≥ 2. As for the risk of major bleeding, patients were stratified into 2 groups according to individual HAS-BLED scores: (1) low-moderate risk group (score 0-2) and (2) high risk group (score ≥ 3).1212 Pisters R, Lane DA, Nieuwlaat R, de Vos CB, Crijns HJ, Lip GY. A novel user-friendly score (HAS-BLED) to assess 1-year risk of major bleeding in patients with atrial fibrillation: the Euro Heart Survey. Chest. 2010;138(5):1093-100. doi: 10.1378/chest.10-0134.
https://doi.org/10.1378/chest.10-0134...
The acronym HAS-BLED represents each of the bleeding risk factors and assigns 1 point for each of the following: uncontrolled hypertension, abnormal renal and/or liver function, previous stroke, bleeding history or predisposition, labile international normalized ratios, elderly, and concomitant drugs and/or alcohol abuse.1313 Lane DA, Lip GY. Use of the CHA(2)DS(2)-VASc and HAS-BLED scores to aid decision making for thromboprophylaxis in nonvalvular atrial fibrillation. Circulation. 2012;126(7):860-5. doi: 10.1161/CIRCULATIONAHA.111.060061.
https://doi.org/10.1161/CIRCULATIONAHA.1...
Data were obtained by reviewing electronic health records during outpatient clinic visits, emergency visits and hospital admissions during the study period. Patients who were lost to follow-up, who died or whose anticoagulation therapy was discontinued were equally included in the analysis. Time in therapeutic range (TTR) was estimated though Rosendaal's linear interpolation method.2020 Rosendaal FR, Cannegieter SC, van der Meer FJ, Briet E. A method to determine the optimal intensity of oral anticoagulant therapy. Thromb Haemost. 1993;69(3):236-9. PMID: 8470047.
Laboratory test results and left ventricular ejection fraction (preferably assessed by echocardiogram) at the beginning of follow-up period were recorded. Anemia was defined by hemoglobin (Hb) below 13.0 g/L in men and 12.0 g/L in women.2121 World Health Organization. (WHO). Department of Nutrition for Health and Development. Iron deficiency anaemia: assessment, prevention and control: a guide for programme managers. Geneva; 2001. Uncontrolled hypertension was defined as systolic blood pressure > 160 mmHg1212 Pisters R, Lane DA, Nieuwlaat R, de Vos CB, Crijns HJ, Lip GY. A novel user-friendly score (HAS-BLED) to assess 1-year risk of major bleeding in patients with atrial fibrillation: the Euro Heart Survey. Chest. 2010;138(5):1093-100. doi: 10.1378/chest.10-0134.
https://doi.org/10.1378/chest.10-0134...
. Major bleeding was defined as any bleeding requiring hospitalization and/or causing a decrease in hemoglobin level > 2 g/dL and/or requiring blood transfusion.1212 Pisters R, Lane DA, Nieuwlaat R, de Vos CB, Crijns HJ, Lip GY. A novel user-friendly score (HAS-BLED) to assess 1-year risk of major bleeding in patients with atrial fibrillation: the Euro Heart Survey. Chest. 2010;138(5):1093-100. doi: 10.1378/chest.10-0134.
https://doi.org/10.1378/chest.10-0134...
Presence of chronic dialysis, renal transplantation or serum creatinine ≥ 200 µmol/L (2.26 mg/dL) was classified as abnormal renal function.1212 Pisters R, Lane DA, Nieuwlaat R, de Vos CB, Crijns HJ, Lip GY. A novel user-friendly score (HAS-BLED) to assess 1-year risk of major bleeding in patients with atrial fibrillation: the Euro Heart Survey. Chest. 2010;138(5):1093-100. doi: 10.1378/chest.10-0134.
https://doi.org/10.1378/chest.10-0134...
Abnormal liver function was defined as previous clinical diagnosis of chronic hepatic disease (e.g., cirrhosis) or biochemical evidence of significant hepatic derangement (e.g., bilirubin > 2x upper limit of normal, in association with AST/ALT/ALP > 3 x upper limit of normal).1212 Pisters R, Lane DA, Nieuwlaat R, de Vos CB, Crijns HJ, Lip GY. A novel user-friendly score (HAS-BLED) to assess 1-year risk of major bleeding in patients with atrial fibrillation: the Euro Heart Survey. Chest. 2010;138(5):1093-100. doi: 10.1378/chest.10-0134.
https://doi.org/10.1378/chest.10-0134...
Statistical analysis
Descriptive analyses were performed by absolute and relative frequency for qualitative variables, and mean ± standard deviation and median (interquartile range, 25-75th percentile) for quantitative variables with symmetric and asymmetric distribution, respectively. Normality of the distribution of each variable was evaluated using Shapiro-Wilk test. Comparison between the groups was performed by Chi-square test. Fisher exact test was used in situations of low frequency. Area under the Receiver Operating Characteristic (ROC) curve was calculated to evaluate the ability of HAS-BLED score to predict major bleeding. Major bleeding-free survival between HAS-BLED groups was analyzed by Kaplan-Meier curves and compared using log-rank test. Logistic regression was performed to detect the adjusted association of HAS-BLED score components with the outcome major bleeding. Relation between candidate variables and the outcome was initially investigated in a series of bivariate analyses (p < 0.10 for selection of candidate variables). Next, we used stepwise forward selection for variable inclusion until all retained variables were statistically significant at a p < 0.05. The significance level adopted in all tests was 5%. Data were analyzed in Statistical Package for Social Sciences (SPSS) software, version 21.0 (IBM, Armonk, NY, USA).
Results
A total of 263 patients were included in the study, corresponding to 38.5% of individuals attending in outpatient anticoagulation clinic. Demographic characteristics are detailed in Table 1. There were no patients using nonsteroidal anti-inflammatory drug (NSAIDs). Indications for anticoagulation among patients not included in the current cohort were: mechanical cardiac prosthesis (21.2%), deep vein thrombosis / pulmonary thromboembolism (19.0%), valvular AF (8.6%) and thrombus in the left ventricle (2.0%).
Mean follow-up was 329.9 ± 81.9 days. Of the 263 patients, 205 (77.9%) completed the follow-up, 25 (9.5%) were lost to follow-up, 21 (8.0%) stopped the anticoagulation and 12 (4.6%) died. Overall, 2,754 INR tests were performed during the study period, with a median of 10 INR tests per patient (range 7-13). Of these, 1,270 (46.1%) presented values between 2.0 and 3.0 over a treatment time of 237.6 patients-year. Median TTR was 62.5% (44.2-79.5%). Patients were under-anticoagulated (i.e., INR < 2.0) during a median of 18.9% of time (6.1-36.6%) and over-anticoagulated (i.e., INR > 3.0) during a median 9.6% of time (1.6-23.3%).
The incidence of major bleeding in all patients was 5.7% (n = 15), which represents 6.3 major bleedings/100-patients-year. The major bleeding sites were mucocutaneous (n = 4; 26.7%), genitourinary (n = 4; 26.7%), gastrointestinal (n = 3; 20.0%), intracranial (n = 3; 20.0%) and retroperitoneal (n = 1; 6.7%).
"Elderly" was the most prevalent HAS-BLED score component (72.2%), followed by "Labile INR" (47.1%). When the patients were divided according the HAS-BLED score in high and low-moderate risk for major bleeding, we observed that 104 patients (39.5%) were classified as high risk (HAS-BLED ≥ 3). This group had a higher incidence of major bleeding compared with the low-moderate (9.6 versus 3.1%; p = 0.052).
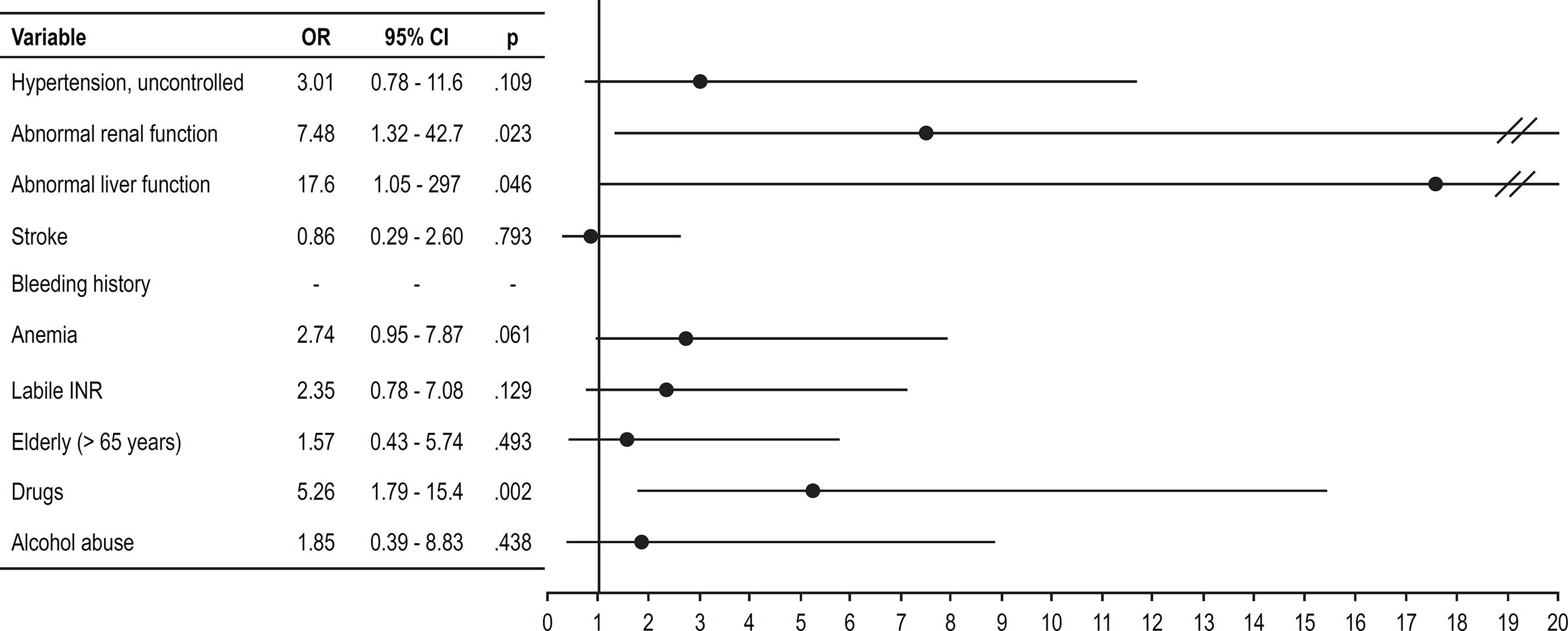
The association of HAS-BLED score components with major bleeding incidence is described in Table 2. In multivariate logistic regression analysis, concurrent antiplatelet use was the only independent predictor of major bleeding among score components (OR 5.13, 95%CI: 1.55-17.0; p = 0.007).
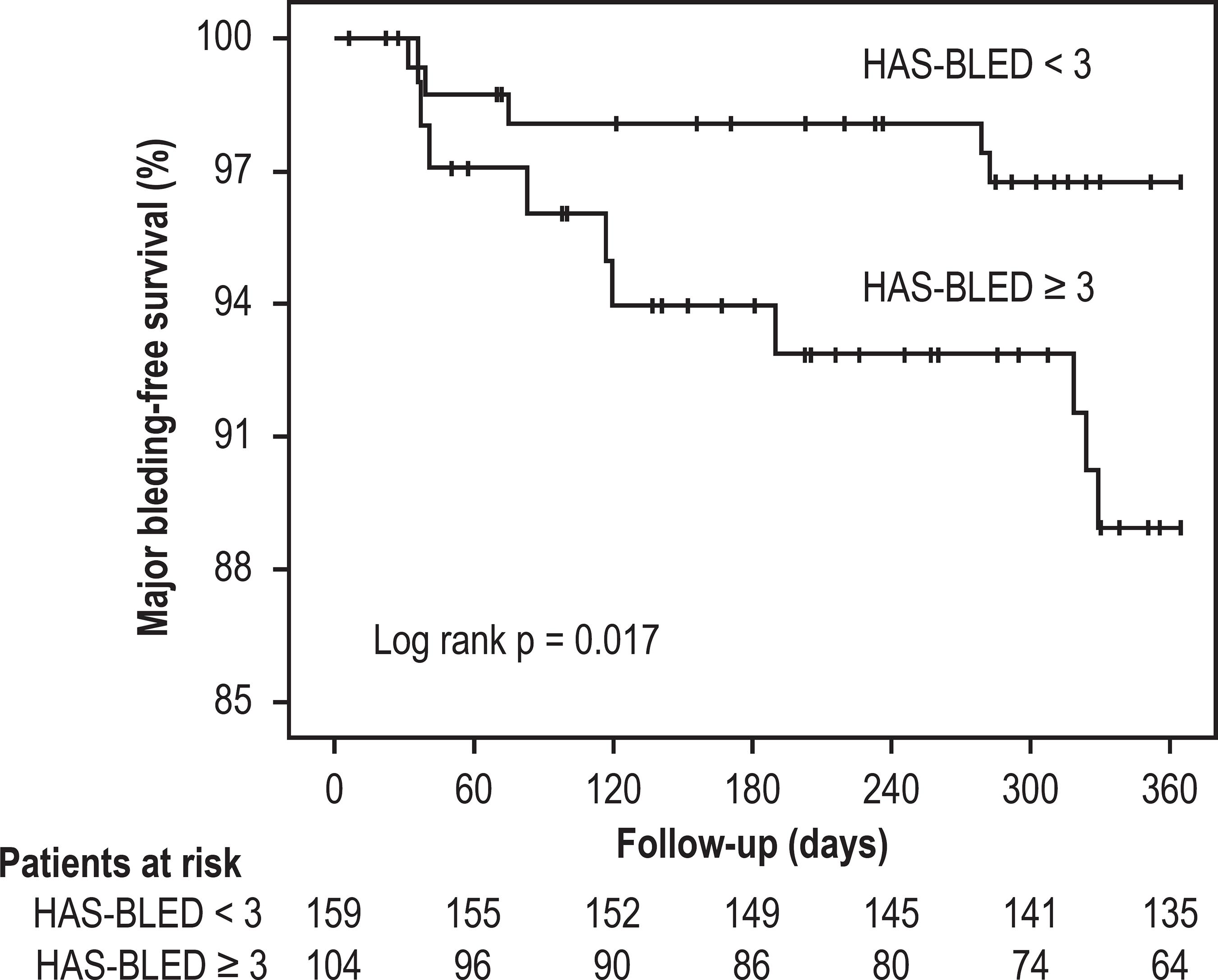
The area under the ROC curve for HAS-BLED score ability to predict major bleeding was 0.70 (95%CI: 0.53-0.86; p = 0.01). A score cut-off point ≥ 3 presented sensibility of 66.7%, specificity of 62.1%, positive predictive value of 9.6% and negative predictive value of 96.9%. Major bleeding-free survival Kaplan-Meier curves according HAS-BLED score categories are shown in Figure 1. High-risk patients, i.e. HAS-BLED score ≥ 3, had a significantly lower major bleeding-free survival (p = 0.017).
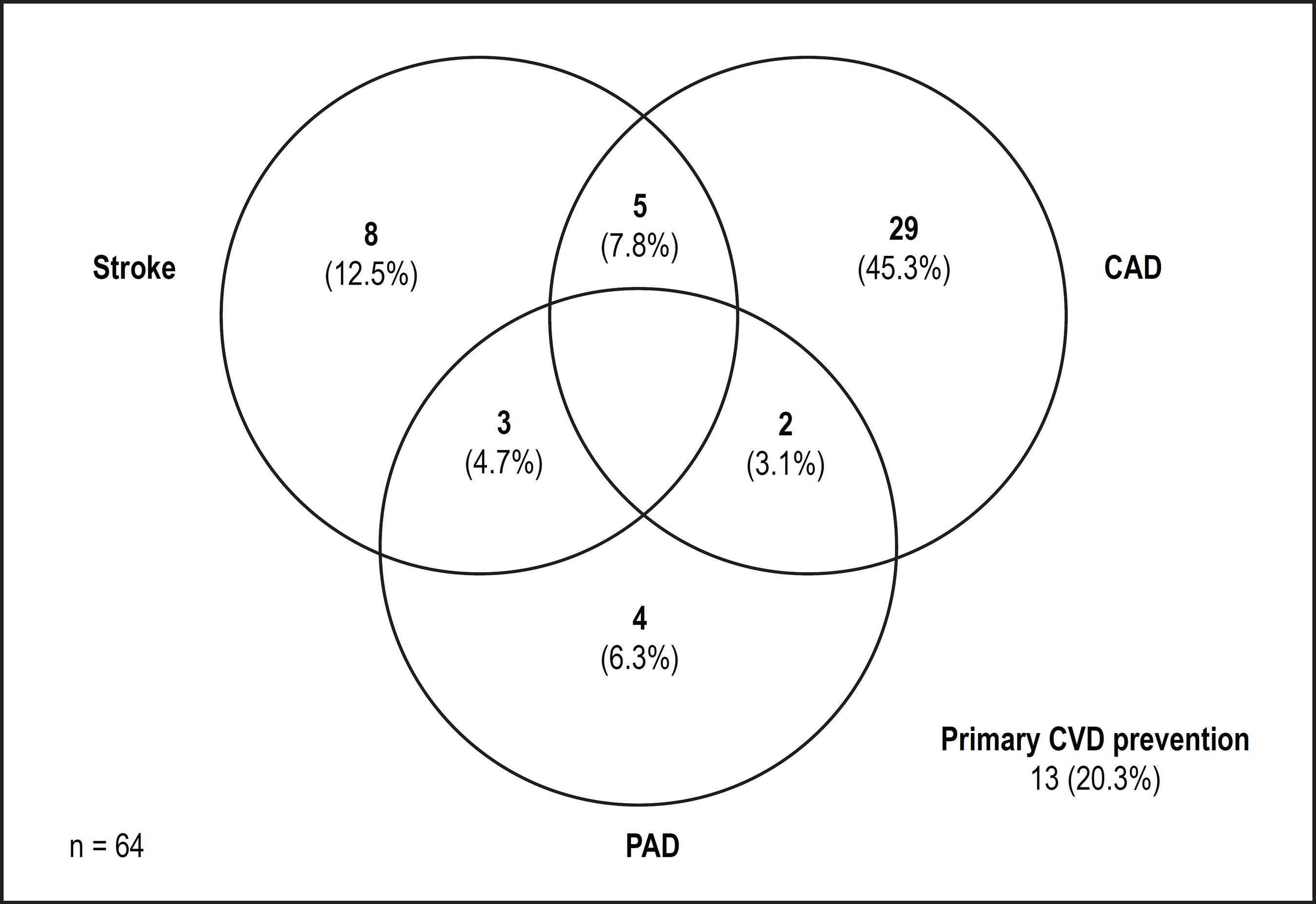
The magnitude of the risks associated to each score component is presented in Figures 2 and 3 in univariate and multivariate analysis, respectively. In multivariate analysis, only the concurrent use of antiplatelet agents independently predicted the occurrence of major bleeding among the score components. Antiplatelets used during follow-up and their indications are described in Figure 4 and Table 3.
OR: odds ratio; CI: confidence interval; INR: international normalized ratio. HAS-BLED components and associated major bleeding risk in univariate analysis.
OR: odds ratio; CI: confidence interval; INR: international normalized ratio. HAS-BLED components and major bleeding risk associated in multivariate analysis.
Venn diagram of indication of concomitant use of drugs. CAD: coronary artery disease; PAD: peripheral artery disease; CVD: cardiovascular disease.
Discussion
In this study, a significant association between high HAS-BLED scores (i.e, score ≥ 3) and major bleeding during follow-up was observed. The only independent variable of HAS-BLED score associated with this adverse event was concomitant use of antiplatelet agents. Our findings corroborate the use of HAS-BLED score as a predictor of bleeding, and the importance of an adequate assessment of modifiable risk factors for bleeding.
Current literature suggest that increments in HAS-BLED score values are associated with progressively increased risk of bleeding, with little difference in hemorrhage rates between patients in use of OAC and controls. Because many risk factors for bleeding are also risk factors for stroke, the use of OACs is wrongly contraindicated for many patients with high risk of stroke (and consequently high risk of bleeding),33 Freedman B, Potpara TS, Lip GY. Stroke prevention in atrial fibrillation. Lancet. 2016;388(10046):806-17. doi: 10.1016/S0140-6736(16)31257-0.
https://doi.org/10.1016/S0140-6736(16)31...
thereby losing benefit of stroke prevention and maintaining a high risk of bleeding even without use of OAC.
The rate of major bleeding found in our study (6.3/100 patients-year) was higher than previously reported (1.4-2.4/100 patients-year).2222 Ogilvie IM, Welner SA, Cowell W, Lip GY. Ischaemic stroke and bleeding rates in 'real-world' atrial fibrillation patients. Thromb Haemost. 2011;106(1):34-44. doi: 10.1160/TH10-10-0674.
https://doi.org/10.1160/TH10-10-0674...
This may be explained by the median TTR found in this cohort (62.5%), which is in the lower limit of 60-65% of currently accepted rate.2323 Connolly SJ, Pogue J, Eikelboom J, Flaker G, Commerford P, Franzosi MG, et al; ACTIVE W Investigators. Benefit of oral anticoagulant over antiplatelet therapy in atrial fibrillation depends on the quality of international normalized ratio control achieved by centers and countries as measured by time in therapeutic range. Circulation. 2008;118(20):2029-37. doi: 10.1161/CIRCULATIONAHA.107.750000.
https://doi.org/10.1161/CIRCULATIONAHA.1...
Connolly et al.2323 Connolly SJ, Pogue J, Eikelboom J, Flaker G, Commerford P, Franzosi MG, et al; ACTIVE W Investigators. Benefit of oral anticoagulant over antiplatelet therapy in atrial fibrillation depends on the quality of international normalized ratio control achieved by centers and countries as measured by time in therapeutic range. Circulation. 2008;118(20):2029-37. doi: 10.1161/CIRCULATIONAHA.107.750000.
https://doi.org/10.1161/CIRCULATIONAHA.1...
suggest that this is the minimal threshold in which there is significant benefit of using OAC as compared with double antiplatelet therapy, with lower TTR being associated with both stroke and hemorrhage rates that are comparable to those observed in AF patients treated with clopidogrel plus acetylsalicylic acid (ASA).
Another possible explanation for the high bleeding rate among our patients was the proportion of patients under concomitant use of antiplatelet drugs (24.3%). The majority of them had no clear indication to maintain antiplatelet use once OAC therapy has been initiated. As shown in Figure 4, the reasons for concurrent use of antiplatelets were coronary artery disease (CAD, 56.2%), stroke (25.0%), primary cardiovascular disease (CVD) prevention and peripheral artery disease (PAD, 14.1%). Current guidelines recommend discontinuation of antiplatelet therapy in most cases when OAC is indicated; exceptions include acute coronary syndrome (ACS) and stent implantation, though even in these cases antiplatelet drugs should be discontinued as soon as possible.2424 Camm AJ, Kirchhof P, Lip GY, Schotten U, Savelieva I, Ernst S, et al; European Heart Rhythm Association; European Association for Cardio-Thoracic Surgery. Guidelines for the management of atrial fibrillation: the Task Force for the Management of Atrial Fibrillation of the European Society of Cardiology (ESC). Eur Heart J. 2010;31(19):2369-429. doi: 10.1093/eurheartj/ehq278. Erratum in: Eur Heart J. 2011;32(9):1172.
https://doi.org/10.1093/eurheartj/ehq278...
This is based on low quality evidence that suggest that patients with stable CAD have increased bleeding risk with OAC plus antiplatelet agent without further decrease in major adverse cardiovascular events in comparison with OAC alone.1515 Olesen JB, Lip GY, Lindhardsen J, Lane DA, Ahlehoff O, Hansen ML, et al. Risks of thromboembolism and bleeding with thromboprophylaxis in patients with atrial fibrillation: a net clinical benefit analysis using a 'real world' nationwide cohort study. Thromb Haemost. 2011;106(4):739-49. doi: 10.1160/TH11-05-0364.
https://doi.org/10.1160/TH11-05-0364...
,2525 You JJ, Singer DE, Howard PA, Lane DA, Eckman MH, Fang MC, et al. Antithrombotic therapy for atrial fibrillation: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 Suppl):e531S-75S. doi: 10.1378/chest.11-2304.
https://doi.org/10.1378/chest.11-2304...
According to the 2016 US Preventive Services Task Force review,2626 Final Recommendation Statement: aspirin use to prevent cardiovascular disease and colorectal cancer: preventive medication. U.S. Preventive Services Task Force. [Access in 2016 Nov 10]. Available from: http://www.uspreventiveservicestaskforce.org
http://www.uspreventiveservicestaskforce...
ASA for the primary prevention of cardiovascular events has been shown to be safe and effective in the very strict group of adults ranging 50-59 years old who have an estimated cardiovascular risk > 10% and a low bleeding risk according to American Heart Association (AHA) score. Regarding PAD and stroke,2727 Anand S, Yusuf S, Xie C, Pogue J, Eikelboom J, Budaj A, et al; Warfarin Antiplatelet Vascular Evaluation Trial Investigators. Oral anticoagulant and antiplatelet therapy and peripheral arterial disease. N Engl J Med. 2007;357(3):217-27. doi: 10.1056/NEJMoa065959.
https://doi.org/10.1056/NEJMoa065959...
28 Fischer M. Does the combination of warfarin and aspirin have a place in secondary stroke prevention? No. Stroke. 2009;40(5):1944-5. doi: 10.1161/STROKEAHA.108.537670.
https://doi.org/10.1161/STROKEAHA.108.53...
-2929 Furlan AJ. Does the combination of warfarin and aspirin have a place in secondary stroke prevention? Yes. Stroke. 2009;40(5):1942-3. doi: 10.1161/STROKEAHA.108.537662.
https://doi.org/10.1161/STROKEAHA.108.53...
current evidence indicates no benefit of adding ASA to VKA.
There are some limitations in our study that deserve to be mentioned. First, retrospective design may have influenced the quality and consistency of the data collected. Besides, review of the medical records could only identify events (cardiovascular and bleeding) that occurred at our hospital or that have been spontaneously reported by the patients during clinical care. This may underestimate adverse events occurrence. Second, the relative small sample size associated with the short follow-up period may have underestimated the occurrence of adverse events, especially events related to AF, which have a longer latency period. Yet, we believe that this may be a conservative bias, given the elevated event rates observed in this limited sample. Third, the higher rates of bleeding found in this study could have overestimated the prediction power of the score. Finally, the fact that the study was conducted at a single center can limit external validity of our findings.
Conclusion
HAS-BLED may be a useful and applicable tool to assess bleeding risk in our population, particularly to aid the identification of potentially modifiable factors such as concomitant use of antiplatelet agents. We highlight that HAS-BLED should not be used as the single instrument to either initiate OAC or exclude patients from OAC therapy. Hospitals and clinical care facilities should implement protocols for regular assessment of patients using OAC, and HAS-BLED may have a major role helping to reduce complications associated with this therapy. Further research is needed to assess the impact of score-based protocols for modifying bleeding risk factors and rates in anticoagulation outpatient clinics.
-
Sources of FundingThere were no external funding sources for this study.
-
Study AssociationThis study is not associated with any thesis or dissertation work.
References
-
1January CT, Wann LS, Alpert JS, Calkins H, Cigarroa JE, Cleveland JC Jr, et al; ACC/AHA Task Force Members. 2014 AHA/ACC/HRS Guideline for the management of patients with atrial fibrillation: a report of the American College of Cardiology / American Heart Association Task Force on practice guidelines and the Heart Rhythm Society. Circulation. 2014;130(23):e199-267. doi: 10.1161/CIR.0000000000000041. Erratum in: Circulation. 2014;130(23):e272-4.
» https://doi.org/10.1161/CIR.0000000000000041 -
2Odutayo A, Wong CX, Hsiao AJ, Hopewell S, Altman DG, Emdin CA. Atrial fibrillation and risks of cardiovascular disease, renal disease, and death: systematic review and meta-analysis. BMJ. 2016;354:i4482. doi: 10.1136/bmj.i4482.
» https://doi.org/10.1136/bmj.i4482 -
3Freedman B, Potpara TS, Lip GY. Stroke prevention in atrial fibrillation. Lancet. 2016;388(10046):806-17. doi: 10.1016/S0140-6736(16)31257-0.
» https://doi.org/10.1016/S0140-6736(16)31257-0 -
4Hart RG, Pearce LA, Aguilar MI. Meta-analysis: antithrombotic therapy to prevent stroke in patients who have nonvalvular atrial fibrillation. Ann Intern Med. 2007;146(12):857-67. PMID: 17577005.
-
5Bengtson LG, Lutsey PL, Chen LY, MacLehose RF, Alonso A. Comparative effectiveness of dabigatran and rivaroxaban versus warfarin for the treatment of non-valvular atrial fibrillation. J Cardiol. 2017;69(6):868-876. doi: 10.1016/j.jjcc.2016.08.010.
» https://doi.org/10.1016/j.jjcc.2016.08.010 -
6Gomes T, Mamdani MM, Holbrook AM, Paterson JM, Hellings C, Juurlink DN. Rates of hemorrhage during warfarin therapy for atrial fibrillation. CMAJ. 2013;185(2):E121-7. doi: 10.1503/cmaj.121218.
» https://doi.org/10.1503/cmaj.121218 -
7Fang MC, Go AS, Chang Y, Borowsky LH, Pomernacki NK, Udaltsova N, et al. A new risk scheme to predict warfarin-associated hemorrhage: the ATRIA (Anticoagulation and Risk Factors in Atrial Fibrillation) Study. J Am Coll Cardiol. 2011;58(4):395-401. doi: 10.1016/j.jacc.2011.03.031.
» https://doi.org/10.1016/j.jacc.2011.03.031 -
8Shireman TI, Mahnken JD, Howard PA, Kresowik TF, Hou Q, Ellerbeck EF. Development of a contemporary bleeding risk model for elderly warfarin recipients. Chest. 2006;130(5):1390-6. doi: 10.1378/chest.130.5.1390.
» https://doi.org/10.1378/chest.130.5.1390 -
9Gage BF, Yan Y, Milligan PE, Waterman AD, Culverhouse R, Rich MW, et al. Clinical classification schemes for predicting hemorrhage: results from the National Registry of Atrial Fibrillation (NRAF). Am Heart J. 2006;151(3):713-9. doi: 10.1016/j.ahj.2005.04.017.
» https://doi.org/10.1016/j.ahj.2005.04.017 -
10Kuijer PM, Hutten BA, Prins MH, Büller HR. Prediction of the risk of bleeding during anticoagulant treatment for venous thromboembolism. Arch Intern Med. 1999(5);159:457-60. PMID: 10074953.
-
11Beyth RJ, Quinn LM, Landefeld CS. Prospective evaluation of an index for predicting the risk of major bleeding in outpatients treated with warfarin. Am J Med. 1998;105(2):91-9. PMID: 9727814.
-
12Pisters R, Lane DA, Nieuwlaat R, de Vos CB, Crijns HJ, Lip GY. A novel user-friendly score (HAS-BLED) to assess 1-year risk of major bleeding in patients with atrial fibrillation: the Euro Heart Survey. Chest. 2010;138(5):1093-100. doi: 10.1378/chest.10-0134.
» https://doi.org/10.1378/chest.10-0134 -
13Lane DA, Lip GY. Use of the CHA(2)DS(2)-VASc and HAS-BLED scores to aid decision making for thromboprophylaxis in nonvalvular atrial fibrillation. Circulation. 2012;126(7):860-5. doi: 10.1161/CIRCULATIONAHA.111.060061.
» https://doi.org/10.1161/CIRCULATIONAHA.111.060061 -
14Lip GY. Stroke and bleeding risk assessment in atrial fibrillation: when, how, and why? Eur Heart J. 2013;34(14):1041-9. doi: 10.1093/eurheartj/ehs435.
» https://doi.org/10.1093/eurheartj/ehs435 -
15Olesen JB, Lip GY, Lindhardsen J, Lane DA, Ahlehoff O, Hansen ML, et al. Risks of thromboembolism and bleeding with thromboprophylaxis in patients with atrial fibrillation: a net clinical benefit analysis using a 'real world' nationwide cohort study. Thromb Haemost. 2011;106(4):739-49. doi: 10.1160/TH11-05-0364.
» https://doi.org/10.1160/TH11-05-0364 -
16Kim YK, Nieuwlaat R, Connolly SJ, Schulman S, Meijer K, Raju N, et al. Effect of a simple two-step warfarin dosing algorithm on anticoagulant control as measured by time in therapeutic range: a pilot study. J Thromb Haemost. 2010;8(1):101-6. doi: 10.1111/j.1538-7836.2009.03652.x
» https://doi.org/10.1111/j.1538-7836.2009.03652.x -
17Camm AJ, Lip GY, De Caterina R, Savelieva I, Atar D, Hohnloser SH, et al. 2012 focused update of the ESC Guidelines for the management of atrial fibrillation: an update of the 2010 ESC Guidelines for the management of atrial fibrillation. Eur Heart J. 2012;33(21):2719-47. doi: 10.1093/eurheartj/ehs253. Erratum in: Eur Heart J. 2013;34(10):790. Eur Heart J. 2013;34(36):2850-1.
» https://doi.org/10.1093/eurheartj/ehs253 -
18Gage BF, Waterman AD, Shannon W, Boechler M, Rich MW, Radford MJ. Validation of clinical classification schemes for predicting stroke: results from the National Registry of Atrial Fibrillation. JAMA. 2001;285(22):2864-70. PMID: 11401607.
-
19Lip GY, Nieuwlaat R, Pisters R, Lane DA, Crijns HJ. Refining clinical risk stratification for predicting stroke and thromboembolism in atrial fibrillation using a novel risk factor-based approach: the euro heart survey on atrial fibrillation. Chest. 2010;137(2):263-72. doi: 10.1378/chest.09-1584.
» https://doi.org/10.1378/chest.09-1584 -
20Rosendaal FR, Cannegieter SC, van der Meer FJ, Briet E. A method to determine the optimal intensity of oral anticoagulant therapy. Thromb Haemost. 1993;69(3):236-9. PMID: 8470047.
-
21World Health Organization. (WHO). Department of Nutrition for Health and Development. Iron deficiency anaemia: assessment, prevention and control: a guide for programme managers. Geneva; 2001.
-
22Ogilvie IM, Welner SA, Cowell W, Lip GY. Ischaemic stroke and bleeding rates in 'real-world' atrial fibrillation patients. Thromb Haemost. 2011;106(1):34-44. doi: 10.1160/TH10-10-0674.
» https://doi.org/10.1160/TH10-10-0674 -
23Connolly SJ, Pogue J, Eikelboom J, Flaker G, Commerford P, Franzosi MG, et al; ACTIVE W Investigators. Benefit of oral anticoagulant over antiplatelet therapy in atrial fibrillation depends on the quality of international normalized ratio control achieved by centers and countries as measured by time in therapeutic range. Circulation. 2008;118(20):2029-37. doi: 10.1161/CIRCULATIONAHA.107.750000.
» https://doi.org/10.1161/CIRCULATIONAHA.107.750000 -
24Camm AJ, Kirchhof P, Lip GY, Schotten U, Savelieva I, Ernst S, et al; European Heart Rhythm Association; European Association for Cardio-Thoracic Surgery. Guidelines for the management of atrial fibrillation: the Task Force for the Management of Atrial Fibrillation of the European Society of Cardiology (ESC). Eur Heart J. 2010;31(19):2369-429. doi: 10.1093/eurheartj/ehq278. Erratum in: Eur Heart J. 2011;32(9):1172.
» https://doi.org/10.1093/eurheartj/ehq278 -
25You JJ, Singer DE, Howard PA, Lane DA, Eckman MH, Fang MC, et al. Antithrombotic therapy for atrial fibrillation: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 Suppl):e531S-75S. doi: 10.1378/chest.11-2304.
» https://doi.org/10.1378/chest.11-2304 -
26Final Recommendation Statement: aspirin use to prevent cardiovascular disease and colorectal cancer: preventive medication. U.S. Preventive Services Task Force. [Access in 2016 Nov 10]. Available from: http://www.uspreventiveservicestaskforce.org
» http://www.uspreventiveservicestaskforce.org -
27Anand S, Yusuf S, Xie C, Pogue J, Eikelboom J, Budaj A, et al; Warfarin Antiplatelet Vascular Evaluation Trial Investigators. Oral anticoagulant and antiplatelet therapy and peripheral arterial disease. N Engl J Med. 2007;357(3):217-27. doi: 10.1056/NEJMoa065959.
» https://doi.org/10.1056/NEJMoa065959 -
28Fischer M. Does the combination of warfarin and aspirin have a place in secondary stroke prevention? No. Stroke. 2009;40(5):1944-5. doi: 10.1161/STROKEAHA.108.537670.
» https://doi.org/10.1161/STROKEAHA.108.537670 -
29Furlan AJ. Does the combination of warfarin and aspirin have a place in secondary stroke prevention? Yes. Stroke. 2009;40(5):1942-3. doi: 10.1161/STROKEAHA.108.537662.
» https://doi.org/10.1161/STROKEAHA.108.537662
Publication Dates
-
Publication in this collection
25 Sept 2017 -
Date of issue
Nov-Dec 2017
History
-
Received
11 Feb 2017 -
Reviewed
06 Apr 2017 -
Accepted
05 June 2017