Abstract
Introduction : This paper presents a detection method for upper limb movement intention as part of a brain-machine interface using EEG signals, whose final goal is to assist disabled or vulnerable people with activities of daily living.
Methods
EEG signals were recorded from six naïve healthy volunteers while performing a motor task. Every volunteer remained in an acoustically isolated recording room. The robot was placed in front of the volunteers such that it seemed to be a mirror of their right arm, emulating a Brain Machine Interface environment. The volunteers were seated in an armchair throughout the experiment, outside the reaching area of the robot to guarantee safety. Three conditions are studied: observation, execution, and imagery of right arm’s flexion and extension movements paced by an anthropomorphic manipulator robot. The detector of movement intention uses the spectral F test for discrimination of conditions and uses as feature the desynchronization patterns found on the volunteers. Using a detector provides an objective method to acknowledge for the occurrence of movement intention.
Results
When using four realizations of the task, detection rates ranging from 53 to 97% were found in five of the volunteers when the movement was executed, in three of them when the movement was imagined, and in two of them when the movement was observed.
Conclusions
Detection rates for movement observation raises the question of how the visual feedback may affect the performance of a working brain-machine interface, posing another challenge for the upcoming interface implementation. Future developments will focus on the improvement of feature extraction and detection accuracy for movement intention using EEG data.
Keywords:
Movement intention; Objective response detection; Statistical F test; Event-related desynchronization; Brain-machine interface
Introduction
Brain interfaces are commonly considered as the ultimate means of interaction between humans and machines or computers (Allison et al., 2012Allison BZ, Brunner C, Altstätter C, Wagner IC, Grissmann S, Neuper C. A hybrid ERD/SSVEP BCI for continuous simultaneous two dimensional cursor control. Journal of Neuroscience Methods. 2012; 209(2):299-307. http://dx.doi.org/10.1016/j.jneumeth.2012.06.022. PMid:22771715.
http://dx.doi.org/10.1016/j.jneumeth.201...
; Doud et al., 2011Doud AJ, Lucas JP, Pisansky MT, He B. Continuous three dimensional control of a virtual helicopter using a motor imagery based brain-computer interface. PLoS One. 2011; 6(10):e26322. http://dx.doi.org/10.1371/journal.pone.0026322. PMid:22046274.
http://dx.doi.org/10.1371/journal.pone.0...
; Hochberg et al., 2012Hochberg LR, Bacher D, Jarosiewicz B, Masse NY, Simeral JD, Vogel J, Haddadin S, Liu J, Cash SS, van der Smagt P, Donoghue JP. Reach and grasp by people with tetraplegia using a neurally controlled robotic arm. Nature. 2012; 485(7398):372-5. http://dx.doi.org/10.1038/nature11076. PMid:22596161.
http://dx.doi.org/10.1038/nature11076...
; Sirvent Blasco et al., 2012Sirvent Blasco J, Iáñez E, Úbeda A, Azorín J. Visual evoked potential-based brain-machine interface applications to assist disabled people. Expert Systems with Applications. 2012; 39(9):7908-18. http://dx.doi.org/10.1016/j.eswa.2012.01.110.
http://dx.doi.org/10.1016/j.eswa.2012.01...
). The idea of controlling our environment using brain activity opens the possibility for many potential applications using these interfaces. For instance, a potential use of a Brain-Machine Interface (BMI) is the assistance of severely disabled individuals in order to improve their communication skills, mobility, and general wellbeing (Hochberg et al., 2012Hochberg LR, Bacher D, Jarosiewicz B, Masse NY, Simeral JD, Vogel J, Haddadin S, Liu J, Cash SS, van der Smagt P, Donoghue JP. Reach and grasp by people with tetraplegia using a neurally controlled robotic arm. Nature. 2012; 485(7398):372-5. http://dx.doi.org/10.1038/nature11076. PMid:22596161.
http://dx.doi.org/10.1038/nature11076...
; Lebedev and Nicolelis, 2006Lebedev MA, Nicolelis MAL. Brain-machine interfaces: past, present and future. Trends in Neurosciences. 2006; 29(9):536-46. http://dx.doi.org/10.1016/j.tins.2006.07.004. PMid:16859758.
http://dx.doi.org/10.1016/j.tins.2006.07...
; Nicolelis, 2003Nicolelis MAL. Brain-machine interfaces to restore motor function and probe neural circuits. Nature Reviews. Neuroscience. 2003; 4(5):417-22. http://dx.doi.org/10.1038/nrn1105. PMid:12728268.
http://dx.doi.org/10.1038/nrn1105...
; Pfurtscheller et al., 2003Pfurtscheller G, Müller GR, Pfurtscheller J, Gerner HJ, Rupp R. ‘Thought’ - Control of functional electrical stimulation to restore hand grasp in a patient with tetraplegia. Neuroscience Letters. 2003; 351(1):33-6. http://dx.doi.org/10.1016/S0304-3940(03)00947-9. PMid:14550907.
http://dx.doi.org/10.1016/S0304-3940(03)...
; Santos-Couto-Paz et al., 2013Santos-Couto-Paz CC, Teixeira-Salmela LF, Tierra-Criollo CJ. The addition of functional task-oriented mental practice to conventional physical therapy improves motor skills in daily functions after stroke. Brazilian Journal of Physical Therapy. 2013; 17(6):564-71. http://dx.doi.org/10.1590/S1413-35552012005000123. PMid:24271094.
http://dx.doi.org/10.1590/S1413-35552012...
).
A significant amount of BMI research is based on non-invasive methods for the measurement of brain activity (Cecotti, 2011Cecotti H. Spelling with non-invasive brain-computer interfaces: current and future trends. The Journal of Physiology. 2011; 105(1-3):106-14. PMid:21911058.). In particular, electroencephalogram (EEG) is a method to measure overall cortical activity that is easy to setup, operate, and does not require a surgical intervention. However, the measurements EEG provide are polluted with noise coming from brain activity of no interest, as well as noise coming from other sources like muscle activity or external interferences. Hence, it is difficult to extract meaningful information from these measurements (Nicolelis, 2003Nicolelis MAL. Brain-machine interfaces to restore motor function and probe neural circuits. Nature Reviews. Neuroscience. 2003; 4(5):417-22. http://dx.doi.org/10.1038/nrn1105. PMid:12728268.
http://dx.doi.org/10.1038/nrn1105...
; Nicolelis and Ribeiro, 2006Nicolelis MAL, Ribeiro S. Seeking the neural code. Scientific American. 2006; 295(6):70-7. http://dx.doi.org/10.1038/scientificamerican1206-70. PMid:17144353.
http://dx.doi.org/10.1038/scientificamer...
). In fact, extracting specific information from EEG signals (e.g., the movement intention of a person) is a challenge that depends on the latest instruments, filtering, and processing methods available.
With focus on EEG data processing, this paper presents the development of a method for the detection of arm movement intention in a BMI setup. In order to do so, an experiment with six volunteers standing in front of a manipulator robot emulated a BMI environment. The purpose of this setup is to test the interference of a moving manipulator robot over the electrophysiological responses measured by EEG. Moreover, our goal is to test the detection algorithm over EEG data obtained within this environment and to identify further challenges towards the implementation of a BMI for self-feeding (Lana et al., 2013bLana EP, Adorno BV, Tierra-Criollo CJ. Assistance task using a manipulator robot and user kinematics feedback. In: 11 Simpósio Brasileiro de Automação Inteligente; 2013 Oct 13-17; Fortaleza, Brazil. Fortaleza: Universidade Federal do Ceará; 2013b. p. 1-6.).
Usually, the detection of movement intention is performed using classifiers that are parameterized, validated and tested in order to obtain the classification accuracy (Bai et al., 2007Bai O, Lin P, Vorbach S, Li J, Furlani S, Hallett M. Exploration of computational methods for classification of movement intention during human voluntary movement from single trial EEG. Clinical Neurophysiology. 2007; 118(12):2637-55. http://dx.doi.org/10.1016/j.clinph.2007.08.025. PMid:17967559.
http://dx.doi.org/10.1016/j.clinph.2007....
). In BMI, the most commonly used classifiers include linear discriminant analysis (Kamavuako et al., 2015Kamavuako EN, Jochumsen M, Niazi IK, Dremstrup K. Comparison of features for movement prediction from single-trial movement-related cortical potentials in healthy subjects and stroke patients. Computational Intelligence and Neuroscience. 2015; 2015:1-8. http://dx.doi.org/10.1155/2015/858015. PMid:26161089.
http://dx.doi.org/10.1155/2015/858015...
; Lew et al., 2012Lew EYL, Chavarriaga R, Silvoni S, Millán JR. Detection of self-paced reaching movement intention from EEG signals. Frontiers in neuroengineering. 2012; 5:1-17. PMid:23055968.; 2014Lew EYL, Chavarriaga R, Silvoni S, Millán JR. Single trial prediction of self-paced reaching directions from EEG signals. Frontiers in Neuroscience. 2014; 8:1-13. http://dx.doi.org/10.3389/fnins.2014.00222. PMid:25136290.
http://dx.doi.org/10.3389/fnins.2014.002...
), matched filters (Jiang et al., 2015Jiang N, Gizzi L, Mrachacz-Kersting N, Dremstrup K, Farina D. A brain-computer interface for single-trial detection of gait initiation from movement related cortical potentials. Clinical Neurophysiology. 2015; 126(1):154-9. http://dx.doi.org/10.1016/j.clinph.2014.05.003. PMid:24910150.
http://dx.doi.org/10.1016/j.clinph.2014....
), support vector machines (Bhagat, 2014Bhagat NA. Detecting movement intent from scalp EEG in a novel upper limb robotic rehabilitation system for stroke. In: 36th Annual International Conference of the IEEE Engineering in Medicine and Biology Society; 2014 Aug 26-30; Chicago, USA. Chigago: IEEE; 2014. p. 4127-30. http://dx.doi.org/10.1109/EMBC.2014.6944532.
http://dx.doi.org/10.1109/EMBC.2014.6944...
), common spatial patterns (Nikulin et al., 2008Nikulin VV, Hohlefeld FU, Jacobs AM, Curio G. Quasi-movements: a novel motor-cognitive phenomenon. Neuropsychologia. 2008; 46(2):727-42. http://dx.doi.org/10.1016/j.neuropsychologia.2007.10.008. PMid:18035381.
http://dx.doi.org/10.1016/j.neuropsychol...
) multivariate linear classifiers (Bai et al., 2011Bai O, Rathi V, Lin P, Huang D, Battapady H, Fei D-Y, Schneider L, Houdayer E, Chen X, Hallett M. Prediction of human voluntary movement before it occurs. Clinical Neurophysiology. 2011; 122(2):364-72. http://dx.doi.org/10.1016/j.clinph.2010.07.010. PMid:20675187.
http://dx.doi.org/10.1016/j.clinph.2010....
), among others. For successful classification, it is required to have a training record available in order to determine the working parameters of the classifier. In this sense, classifiers present at least two limitations: (1) the training data increase the number of sessions required for a particular study, and (2) the classifier works at the specified conditions of the experiment, with its capabilities limited with respect to known variability sources (e.g., inter-subject, day-to-day, or even inter-trial variability).
We propose a detector in order to exploit some advantages over classifiers: (1) its parameters are objectively obtained based on known electrophysiological responses, avoiding the need for training data, and (2) the parameters of the detector are assumed generally applicable to any individual without modification, thus overcoming the limitation with respect to variability that is present in most classifiers.
Two characteristic features in EEG signals related to movement intention are the Movement-Related Cortical Potentials (MRCP) and Event-Related Desynchronization (ERD) at the alpha and beta bands (Giuliana et al., 2011Giuliana G, Mario M, Yassin J. A quality parameter for the detection of the intentionality of movement in patients with neurological tremor performing a finger-to-nose test. In: 33rd Annual International Conference of the IEEE EMBS; 2011 Aug 30-Sept 03; Boston, USA. Boston: IEEE; 2011. p. 7707-10. http://dx.doi.org/10.1109/IEMBS.2011.6091899.
http://dx.doi.org/10.1109/IEMBS.2011.609...
; Jiang et al., 2015Jiang N, Gizzi L, Mrachacz-Kersting N, Dremstrup K, Farina D. A brain-computer interface for single-trial detection of gait initiation from movement related cortical potentials. Clinical Neurophysiology. 2015; 126(1):154-9. http://dx.doi.org/10.1016/j.clinph.2014.05.003. PMid:24910150.
http://dx.doi.org/10.1016/j.clinph.2014....
; Li et al., 2012Li J, Wang Y, Zhang L, Jung TP. Combining ERPs and EEG spectral features for decoding intended movement direction. In: Annual International Conference of the IEEE Engineering in Medicine and Biology Society; 2012 Aug 28-Sep 01; San Diego, USA. San Diego: IEEE; 2012. p. 1769-72.; Morash et al., 2008Morash V, Bai O, Furlani S, Lin P, Hallett M. Classifying EEG signals preceding right hand, left hand, tongue, and right foot movements and motor imageries. Clinical Neurophysiology. 2008; 119(11):2570-8. http://dx.doi.org/10.1016/j.clinph.2008.08.013. PMid:18845473.
http://dx.doi.org/10.1016/j.clinph.2008....
; Niazi et al., 2011Niazi IK, Jiang N, Tiberghien O, Nielsen JF, Dremstrup K, Farina D. Detection of movement intention from single-trial movement-related cortical potentials. Journal of Neural Engineering. 2011; 8(6):1-10. http://dx.doi.org/10.1088/1741-2560/8/6/066009. PMid:22027549.
http://dx.doi.org/10.1088/1741-2560/8/6/...
). As eye movements caused by observing the manipulator robot generate signals that directly interfere with MRCP, as shown in more detail in the Results section, we base our detection algorithm on ERD features.
We analyzed a motor task that consists of a flexion and extension of the volunteer’s right elbow, as a kinematic abstraction of the movements needed for a self-feeding task. A manipulator robot directed the task and three conditions were tested: the observation, execution, and imagery of the movement paced by the robot. The objective is to determine the detection rates for movement imagery, the condition that would be eventually used in the BMI to control the manipulator robot during a self-feeding task (Lana et al., 2013bLana EP, Adorno BV, Tierra-Criollo CJ. Assistance task using a manipulator robot and user kinematics feedback. In: 11 Simpósio Brasileiro de Automação Inteligente; 2013 Oct 13-17; Fortaleza, Brazil. Fortaleza: Universidade Federal do Ceará; 2013b. p. 1-6.). The observation and movement conditions are considered as lower and upper limits for detection, respectively. For imagery to be a valid control signal, it is desired that its detection rates be closer to those of movement than to those of observation.
In this paper, the detection algorithm provides an objective tool to discriminate whether the intention of movement was present or not. The results obtained are not completely reliable for a single-trial detection, but they clearly improve when using just a few trials for detection (around 4-8 trials). However, unlike most single-trial detection approaches, one clear advantage of the detector presented here is the independence on training sessions and specific parameterizations, constituting in this sense a more general and robust approach if compared to classifiers.
Methods
Experiment setup
Six neurologically healthy volunteers (S1 to S6) with no prior experience in motor imagery participated in the study. Subjects were not under the influence of any medication that could interfere with EEG. Volunteers were all male, right-handed, age ranging from 23 to 32. All volunteers read and signed an informed consent form. The experiment took place at the Biomedical Engineering Laboratory of the Federal University of Minas Gerais and was approved by the local Ethics Committee.
For the experiment, every volunteer remained in an acoustically isolated recording room with temperature at 25 ± 1.5 °C. In addition, we placed an anthropomorphic manipulator robot in front of the volunteers such that it seemed to be a mirror of their right arm, with the purpose of emulating a BMI environment resembling the one that will be used in a future application of self-feeding (Lana et al., 2013bLana EP, Adorno BV, Tierra-Criollo CJ. Assistance task using a manipulator robot and user kinematics feedback. In: 11 Simpósio Brasileiro de Automação Inteligente; 2013 Oct 13-17; Fortaleza, Brazil. Fortaleza: Universidade Federal do Ceará; 2013b. p. 1-6.). The volunteers were seated in an armchair throughout the experiment, outside the reaching area of the robot to guarantee safety. A picture of the experimental setup can be found on (Lana et al., 2013aLana EP, Adorno BV, Tierra-Criollo CJ. An ERD/ERS analysis of the relation between human arm and robot manipulator movements. In: IEEE/ISSNIP Biosignals and Biorobotics Conference; 2013 Feb 18-20; Rio de Janeiro, Brazil. Rio de Janeiro: IEEE; 2013a. p. 1-7. http://dx.doi.org/10.1109/BRC.2013.6487461.
http://dx.doi.org/10.1109/BRC.2013.64874...
). Every volunteer was asked not to perform any movement other than the required during the recording sessions, and to blink only in the interval between movements.
Since the role of the manipulator robot is to direct the motor task of the experiment, we generated a movement sequence for the robot to execute. The sequence consisted of two types of movement: a flexion (UP) and an extension (DW) representing the movements for self-feeding. Figure 1 shows the movement sequence generated for the experiment. The robot waits a random time from 8 to 12 seconds between movements, following a uniform distribution. It performs the flexion at time 0UP, holds that position during the random time, performs the extension at time 0DW, holds that position again during the random time, and repeats the sequence. The execution of the movements lasts approximately 2.5 s each.
Experiment protocol. Movement sequence of the robot manipulator. 0UP and 0DW indicate the starting instants for flexion (UP) and extension (DW) movements, respectively (Lana et al., 2013aLana EP, Adorno BV, Tierra-Criollo CJ. An ERD/ERS analysis of the relation between human arm and robot manipulator movements. In: IEEE/ISSNIP Biosignals and Biorobotics Conference; 2013 Feb 18-20; Rio de Janeiro, Brazil. Rio de Janeiro: IEEE; 2013a. p. 1-7. http://dx.doi.org/10.1109/BRC.2013.6487461.
http://dx.doi.org/10.1109/BRC.2013.64874... ).
The experiment consisted of five fifteen-minute sessions, with three-minute resting intervals between them. We explored three conditions in the study: observation, execution, and imagery of the robot’s movement.
For the first session, we asked the volunteer to just stare (ST) at the moving robot as it repeated the sequence over the session span. For the two subsequent sessions, we asked the volunteer to replicate the movement (MV) of the robot, as a respective flexion or extension of the elbow. Finally, for the last two sessions, we asked and explained the volunteer to perform kinesthetic imagery (IM) paced by the robot’s movement.
Data collection
Brain activity was recorded from the scalp using a 17 channel EEG (F7, F3, Fz, F4, F8, T3, C3, Cz, C4, T4, T5, P3, Pz, P4, T6, O1 and O2) according to the International 10-20 System, with reference at earlobes ([A1 + A2]/2). A cap with Ag/AgCl electrodes was used to record the activity from the scalp. The 36-channel BrainNet BNT-36 biological amplifier (EMSA, Rio de Janeiro, Brazil) was used for amplification, prefiltering (including a notch filter at 60 Hz), digitization and acquisition of the EEG. EEG signals were prefiltered using an analogic bandpass filter between 0.1 and 100 Hz and then digitized at a sampling frequency of 600 Hz.
For the MV condition, the movement of the volunteer's arm was recorded using an accelerometer, which was placed at the distal end of the volunteer's radius, close to the wrist, with the recorded axis parallel to the direction of movement. The movement was performed in a semi-pronation of the forearm, starting with the elbow flexed at 90°. The accelerometer data was used in a posterior EEG data analysis to make trial segmentation according to the arm movement. We also recorded the movement of the robot.
The volunteer remained at 80 cm from the robot, outside its reachable space. EOG registered the eye movements by using two Ag/AgCl electrodes positioned above and below the volunteer's right eye. EOG data was used to estimate the influence of eye movements in the EEG measurements.
The digital signal processing, analyses, and detection algorithm were implemented using the Matlab® software. The detection algorithm was developed as a pseudo-online implementation, that is, first data were collected and then the algorithm was executed over the data record as a stream, which provides an indication that the proposed detector is suitable for use in online implementations.
ERP and Event Related Desynchronization/Synchronization (ERD/ERS)
In order to obtain brain patterns related to the conditions of the study, we performed an ERP and ERD/ERS analysis. EEG signals were split into trials corresponding to flexion and extension movements. Every trial consisted of the 3.5 seconds before and 6 seconds after movement initiation (0UP and 0DW in Figure 1). Given that ERD/ERS are expected in the alpha band and frequencies above, no thorough artifact rejection was considered, since most of the artifacts are in frequencies below 4 Hz (Fatourechi et al., 2007Fatourechi M, Bashashati A, Ward RK, Birch GE. EMG and EOG artifacts in brain computer interface systems: a survey. Clinical Neurophysiology. 2007; 118(3):480-94. http://dx.doi.org/10.1016/j.clinph.2006.10.019. PMid:17169606.
http://dx.doi.org/10.1016/j.clinph.2006....
; Jeon et al., 2011Jeon Y, Nam CS, Kim YJ, Whang MC. Event-related (De)synchronization (ERD/ERS) during motor imagery tasks: implications for brain-computer interfaces. International Journal of Industrial Ergonomics. 2011; 41(5):428-36. http://dx.doi.org/10.1016/j.ergon.2011.03.005.
http://dx.doi.org/10.1016/j.ergon.2011.0...
; Santos et al., 2009Santos S Fo, Tierra-Criollo CJ, Souza AP, Pinto MAS, Lima MLC, Manzano GM. Magnitude squared of coherence to detect imaginary movement. EURASIP Journal on Advances in Signal Processing. 2009; 2009(1):534536. http://dx.doi.org/10.1155/2009/534536.
http://dx.doi.org/10.1155/2009/534536...
). A visual inspection of the EEG signals, which were band-pass filtered in the frequencies of interest (specially alpha band), was sufficient to remove the remaining highly polluted trials.
We used the coherent mean to estimate the ERP and intertrial variance method to estimate the ERD/ERS (Kalcher and Pfurtscheller, 1995Kalcher J, Pfurtscheller G. Discrimination between phaselocked and non-phase-locked event-related EEG activity. Electroencephalography and Clinical Neurophysiology. 1995; 94(5):381-4. http://dx.doi.org/10.1016/0013-4694(95)00040-6. PMid:7774524.
http://dx.doi.org/10.1016/0013-4694(95)0...
; Kotchoubey, 2006Kotchoubey B. Event-related potentials, cognition, and behavior: a biological approach. Neuroscience and Biobehavioral Reviews. 2006; 30(1):42-65. http://dx.doi.org/10.1016/j.neubiorev.2005.04.002. PMid:16033699.
http://dx.doi.org/10.1016/j.neubiorev.20...
). The trials presenting artifacts were rejected for the ERP estimation. More detailed explanation, analysis, and results of ERD/ERS for this study were reported by Lana et al. (2013a)Lana EP, Adorno BV, Tierra-Criollo CJ. An ERD/ERS analysis of the relation between human arm and robot manipulator movements. In: IEEE/ISSNIP Biosignals and Biorobotics Conference; 2013 Feb 18-20; Rio de Janeiro, Brazil. Rio de Janeiro: IEEE; 2013a. p. 1-7. http://dx.doi.org/10.1109/BRC.2013.6487461.
http://dx.doi.org/10.1109/BRC.2013.64874...
.
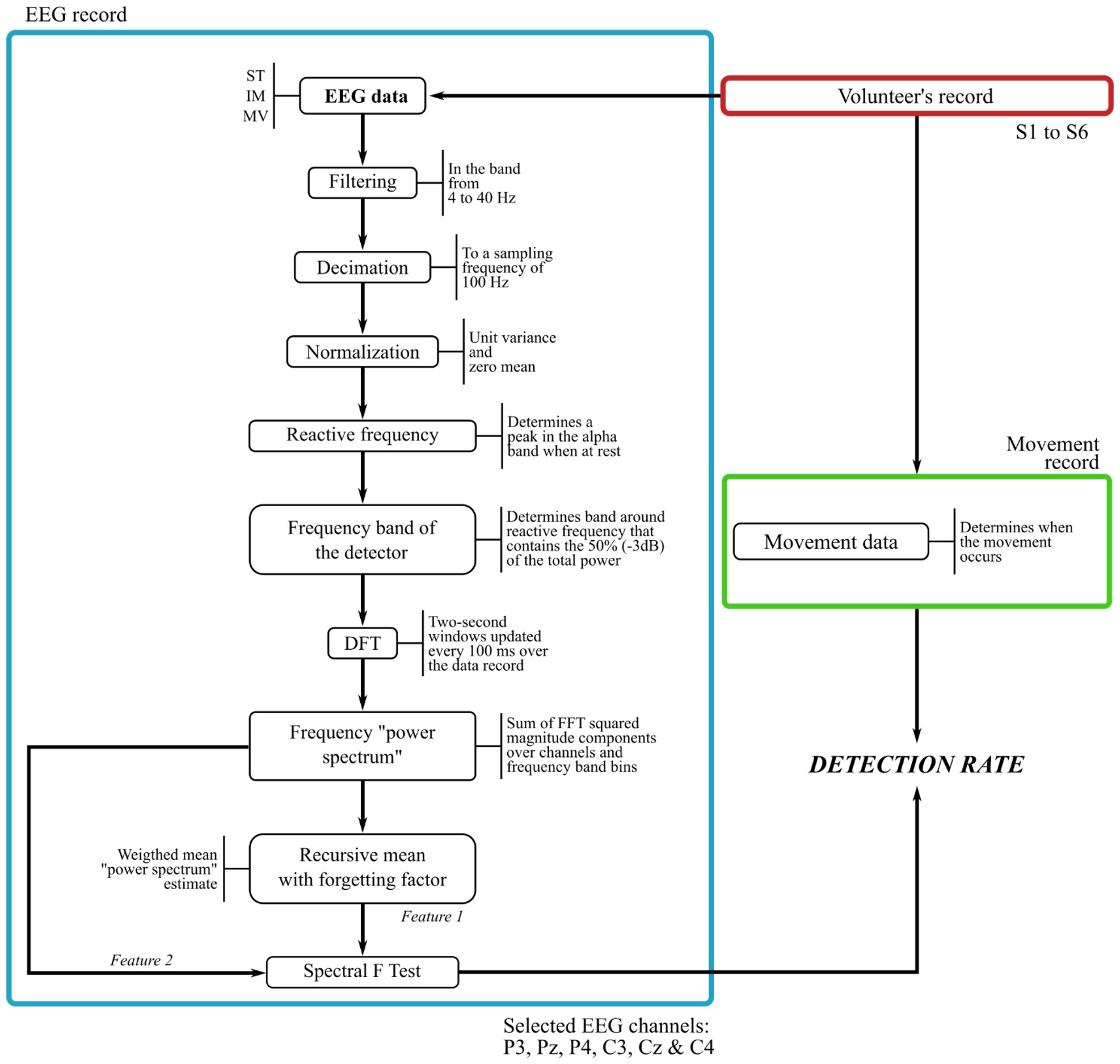
Spectral F Test (SFT) detector
After obtaining the ERD/ERS patterns, we implemented a detector based in the alpha band (around 8-13 Hz) desynchronization related to the brain responses to movement intention. The SFT detector is a statistical test that compares two power signals and determines significant differences among them. Figure 2 describes the procedure that yields the movement intention detector. Based on the brain activity patterns found in the ERD/ERS analysis (Lana et al., 2013aLana EP, Adorno BV, Tierra-Criollo CJ. An ERD/ERS analysis of the relation between human arm and robot manipulator movements. In: IEEE/ISSNIP Biosignals and Biorobotics Conference; 2013 Feb 18-20; Rio de Janeiro, Brazil. Rio de Janeiro: IEEE; 2013a. p. 1-7. http://dx.doi.org/10.1109/BRC.2013.6487461.
http://dx.doi.org/10.1109/BRC.2013.64874...
), we chose six EEG channels for the detector: P3, Pz, P4, C3, Cz, and C4.
A band-pass filter between 4 and 40 Hz was applied to the EEG signals and then decimated to a sampling frequency of 100 Hz. The decimation was performed in order to have faster computations when applying the pseudo-online algorithm for detection. In addition, the EEG signals were normalized to have zero mean and unit variance.
We determined the reactive frequency for every volunteer as the peak in the frequency power spectrum located in the alpha band when the volunteer is at rest (Pfurtscheller and Silva, 1999Pfurtscheller G, Silva FHL. Event-related EEG/MEG synchronization and desynchronization: Basic principles. Clinical Neurophysiology. 1999; 110(11):1842-57. http://dx.doi.org/10.1016/S1388-2457(99)00141-8. PMid:10576479.
http://dx.doi.org/10.1016/S1388-2457(99)...
). A frequency band for the detector was established as a threshold comprising the 50% (-3dB) of the power at while the volunteer was in resting state.
For the detector features, we computed the Discrete Fourier Transform (DFT) squared magnitude of two-second windows for every selected channel of EEG (i.e., P3, Pz, P4, C3, Cz, and C4) and the analysis window was updated every 100 ms, over the whole record span for the pseudo-online algorithm.
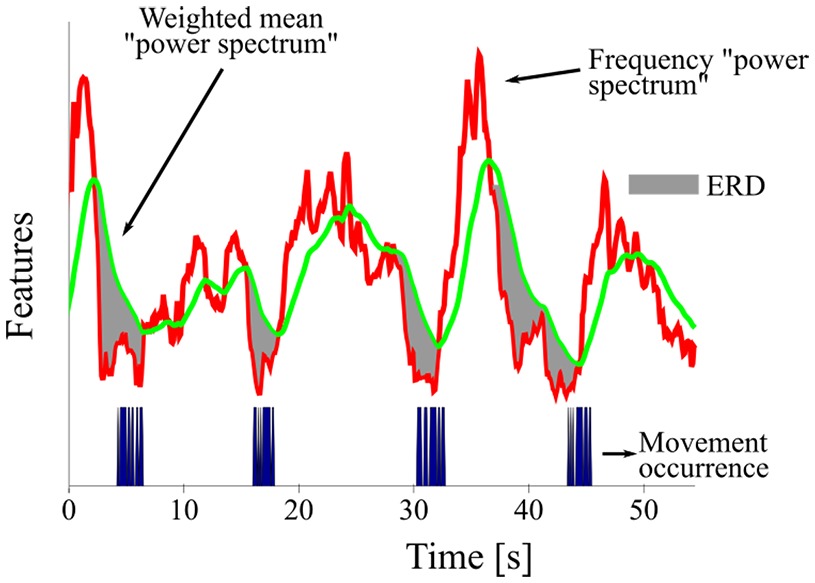
The feature containing the movement information, (frequency “power spectrum”), was computed as the sum over estimated DFT bins and the six EEG channels considered for the detector. The feature for comparison, (weighted mean “power spectrum”), was computed as a recursive mean with forgetting factor given by:
where is the recursive mean of the “power spectrum” at window , with , is the current “power spectrum” sample being processed, is the forgetting factor (Tierra-Criollo, 2001Tierra-Criollo CJ. Monitorização objetiva da resposta à estimulação somato-sensitiva utilizando parâmetros espectrais [thesis]. Rio de Janeiro: Universidade Federal do Rio de Janeiro; 2001.), is the number of EEG channels considered for the detector, and is the number of frequency bins used to compute the features. We use the forgetting factor to estimate a variable-base EEG power. If the value of is , the recursive mean equally weighs the history as well as the current value of the “power spectrum”, whereas if its value is the recursive mean considers only the current value of the “power spectrum”. Therefore, we set a value of that is adequate to keep information of the history of the “power spectrum”. This history accounts for variations at the EEG power that is unrelated to the movement. Since brain activity varies over time, the forgetting factor allows having an updated base measure for comparison, assuming that the time interval of resting is greater than that of the task performance.
For our detector, the forgetting factor was applied to the data from all the volunteers. Figure 3 shows an example of the two features, frequency “power spectrum” and weighted mean “power spectrum”.
Detector features of a segment of volunteer S1’s record under condition MV. The shaded areas correspond to an ERD when compared to the mean “power spectrum”.
The two features extracted from the EEG signals were compared using the SFT detector, and a function for every analyzed window is defined as (Infantosi and Sá, 2007Infantosi AF, Sá AM. A statistical test for evaluating the event-related synchronization/desynchronization and its potential use in brain-computer-interfaces. IFMBE Proceedings. 2007; 18:1122-6. http://dx.doi.org/10.1007/978-3-540-74471-9_260.
http://dx.doi.org/10.1007/978-3-540-7447...
; Simpson et al., 2000Simpson DM, Tierra-Criollo CJ, Leite RT, Zayen EJ, Infantosi AF. Objective response detection in an electroencephalogram during somatosensory stimulation. Annals of Biomedical Engineering. 2000; 28(6):691-8. http://dx.doi.org/10.1114/1.1305530. PMid:10983714.
http://dx.doi.org/10.1114/1.1305530...
; Tierra-Criollo, 2001Tierra-Criollo CJ. Monitorização objetiva da resposta à estimulação somato-sensitiva utilizando parâmetros espectrais [thesis]. Rio de Janeiro: Universidade Federal do Rio de Janeiro; 2001.)
The ratio in Equation 2 corresponds to an F distribution, whose detection threshold at a significance level is defined by with which corresponds to the value of an F distribution with , degrees of freedom at significance level (Tierra-Criollo, 2001Tierra-Criollo CJ. Monitorização objetiva da resposta à estimulação somato-sensitiva utilizando parâmetros espectrais [thesis]. Rio de Janeiro: Universidade Federal do Rio de Janeiro; 2001.). Movement intention activity is detected whenever .
An n-trial detector was defined in analogous way, with an F distribution with , degrees of freedom. The detection was tested using a significance level of 5% for all the analyzed cases.
Results
Our first observation was that eye movements highly influenced the EEG records, therefore preventing us from obtaining the ERP associated with the motor task of our experiment and from using the MRCP as feature for the detector. Figure 4 shows the clear correlation between the coherent mean of EEG trials with the corresponding EOG mean. The low frequency of the eye movements masked the low frequency ERP, possibly because the proximity of the robot to the volunteers caused them to perform extensive eye movements.
For the detector, Lana et al. (2013a)Lana EP, Adorno BV, Tierra-Criollo CJ. An ERD/ERS analysis of the relation between human arm and robot manipulator movements. In: IEEE/ISSNIP Biosignals and Biorobotics Conference; 2013 Feb 18-20; Rio de Janeiro, Brazil. Rio de Janeiro: IEEE; 2013a. p. 1-7. http://dx.doi.org/10.1109/BRC.2013.6487461.
http://dx.doi.org/10.1109/BRC.2013.64874...
reported the desynchronization patterns in the alpha band in the context of the current experiment. For conditions MV and IM, a clear desynchronization is present in the occipital (O1 and O2), parietal (P3, Pz and P4) and central (C3, Cz, C4) areas, if compared to other areas. We found similar activity patterns for condition ST but the activity in central areas is not as clear as for the other two conditions, movement MV and imagery IM. It is also noticeable that the desynchronization in the responsive areas lasts approximately the same time as the movement, around 2.5 seconds. The parietal areas show the most intense desynchronization among the responsive areas. In addition, there are no notorious differences between the conditions of flexion (UP) and extension (DW), therefore discrimination of movements UP and DW cannot be obtained using event related desynchronization, a fact discussed in (Tehovnik et al., 2013Tehovnik EJ, Woods LC, Slocum WM. Transfer of information by BMI. Neuroscience. 2013; 255:134-46. http://dx.doi.org/10.1016/j.neuroscience.2013.10.003. PMid:24120558.
http://dx.doi.org/10.1016/j.neuroscience...
).
We made a comparison between the frequency power spectrum (Welch periodogram) of segments of the EEG signals at rest and the EEG signals when performing the task, as shown in Figure 5. The spectrum obtained presents the same pattern to that obtained by the ERD computation. The cortex area that presents the more intense desynchronization is the area underlying Pz. The occipital, parietal, and central areas also present a desynchronization related to the task. On the other hand, reactive frequency appears like a clear peak in the alpha band when the volunteer is at rest (Pfurtscheller and Silva, 1999Pfurtscheller G, Silva FHL. Event-related EEG/MEG synchronization and desynchronization: Basic principles. Clinical Neurophysiology. 1999; 110(11):1842-57. http://dx.doi.org/10.1016/S1388-2457(99)00141-8. PMid:10576479.
http://dx.doi.org/10.1016/S1388-2457(99)...
). This pattern was present among all the volunteers. The values of the reactive frequency were determined for every volunteer. These values vary among subjects, but all are within the alpha band. Table 1 presents the reactive frequencies for all the volunteers.
Power spectrum comparison between rest and task states for volunteer S6 under condition MV.
Figure 6a shows a typical for a case when detection occurs, in a time scale between the three seconds before and five seconds after the task initiation, whereas Figure 6b shows the same function for a case when no detection occurs with the execution of the task.
Function with (a) detection and (b) no detection of movement intention. The figure shows the results using five realizations of the task for volunteer S1 under condition MV.
In order to consider a particular detection as valid, we defined a time interval when the task condition must be detected. The detection range comprised the half of second before task execution (movement preparation), the actual time of execution, and the half-second after performing the task (latency) as depicted in Figure 6a.
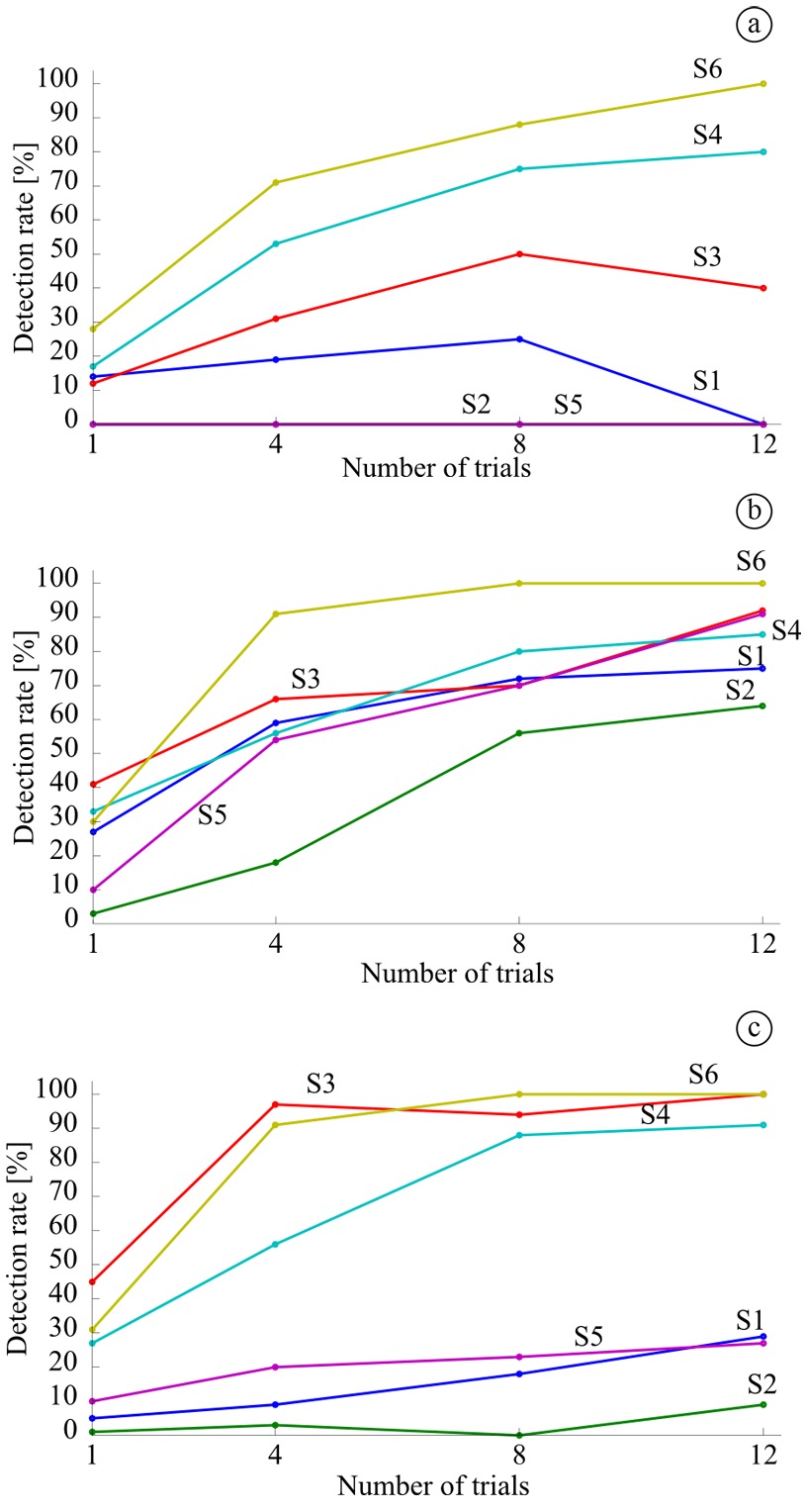
We computed for all the volunteers and conditions using 1 (single trial), 4, 8, and 12 trials for the detector. We determined the detection rate for every volunteer based on the time interval to consider a detection as valid (Figure 6a). The false positives were computed as the detections outside this interval.
We determined the detection rate for all the volunteers and conditions of the study as the rate between the amount of valid detections over the total number of expected detections (Figure 7). In general, the highest detection rate obtained was for condition MV (Figure 7b), followed by IM (Figure 7c) and finally ST (Figure 7a). We obtained detection rates ranging from 53 to 97%, when using four realizations of the task, in five of the volunteers for condition MV, in three of them for IM, and in two of them for ST.
For imagery (IM), which is the condition to be used to command the BMI, the detection rates for three volunteers (S1, S2 and S5) are much lower than for the others (S3, S4 and S6). Furthermore, the detection rate does not improve significantly with the number of trials used in the detector for the volunteers with low detection rate. False positives varied among subjects, but all rates remained below 5% when using more than one trial for the detector.
Discussion
This paper presented the implementation of a detection algorithm for movement intention based on EEG signals. The detection is based on ERD patterns that allow comparing the power in the alpha band to a base power estimative.
The widely spread ERD patterns suggest high cognitive activity related to the task performed during the experiment. This activity could be caused by the attention to some aspects of the task, such as the movement initiation, extent, duration, and type. It is also remarkable the presence of a more intense alpha band desynchronization in the parietal area, possibly due to the level of attention and association between visual and motor stimuli caused by the robot movement (Behrmann et al., 2004Behrmann M, Geng JJ, Shomstein S. Parietal cortex and attention. Current Opinion in Neurobiology. 2004; 14(2):212-7. http://dx.doi.org/10.1016/j.conb.2004.03.012. PMid:15082327.
http://dx.doi.org/10.1016/j.conb.2004.03...
; Li et al., 2012Li J, Wang Y, Zhang L, Jung TP. Combining ERPs and EEG spectral features for decoding intended movement direction. In: Annual International Conference of the IEEE Engineering in Medicine and Biology Society; 2012 Aug 28-Sep 01; San Diego, USA. San Diego: IEEE; 2012. p. 1769-72.; Nair et al., 2003Nair DG, Purcott KL, Fuchs A, Steinberg F, Kelso JAS. Cortical and cerebellar activity of the human brain during imagined and executed unimanual and bimanual action sequences: a functional MRI study. Brain Research. Cognitive Brain Research. 2003; 15(3):250-60. http://dx.doi.org/10.1016/S0926-6410(02)00197-0. PMid:12527099.
http://dx.doi.org/10.1016/S0926-6410(02)...
). The desynchronization responses differ from other studies where the central areas are considered the most responsive to motor tasks (Jeon et al., 2011Jeon Y, Nam CS, Kim YJ, Whang MC. Event-related (De)synchronization (ERD/ERS) during motor imagery tasks: implications for brain-computer interfaces. International Journal of Industrial Ergonomics. 2011; 41(5):428-36. http://dx.doi.org/10.1016/j.ergon.2011.03.005.
http://dx.doi.org/10.1016/j.ergon.2011.0...
; Pineda et al., 2000Pineda JA, Allison BZ, Vankov A. The effects of self-movement, observation, and imagination on alpha rhythms and readiness potentials (RP’s): toward a brain-computer interface (BCI). IEEE Transactions on Rehabilitation Engineering. 2000; 8(2):219-22. http://dx.doi.org/10.1109/86.847822. PMid:10896193.
http://dx.doi.org/10.1109/86.847822...
; Sirvent Blasco et al., 2012Sirvent Blasco J, Iáñez E, Úbeda A, Azorín J. Visual evoked potential-based brain-machine interface applications to assist disabled people. Expert Systems with Applications. 2012; 39(9):7908-18. http://dx.doi.org/10.1016/j.eswa.2012.01.110.
http://dx.doi.org/10.1016/j.eswa.2012.01...
). The ERD found for the three conditions of the study suggest similar electrophysiological patterns for the movement observation, execution, and imagery. Similar results were also reported by Pineda et al. (2000)Pineda JA, Allison BZ, Vankov A. The effects of self-movement, observation, and imagination on alpha rhythms and readiness potentials (RP’s): toward a brain-computer interface (BCI). IEEE Transactions on Rehabilitation Engineering. 2000; 8(2):219-22. http://dx.doi.org/10.1109/86.847822. PMid:10896193.
http://dx.doi.org/10.1109/86.847822...
, Nikulin et al. (2008)Nikulin VV, Hohlefeld FU, Jacobs AM, Curio G. Quasi-movements: a novel motor-cognitive phenomenon. Neuropsychologia. 2008; 46(2):727-42. http://dx.doi.org/10.1016/j.neuropsychologia.2007.10.008. PMid:18035381.
http://dx.doi.org/10.1016/j.neuropsychol...
, Yuan et al. (2010)Yuan H, Perdoni C, He B. Relationship between movement speed and EEG activity during imagined and executed hand movements. Journal of Neural Engineering. 2010; 7(2):26001. http://dx.doi.org/10.1088/1741-2560/7/2/026001. PMid:20168002.
http://dx.doi.org/10.1088/1741-2560/7/2/...
, and Bhagat (2014)Bhagat NA. Detecting movement intent from scalp EEG in a novel upper limb robotic rehabilitation system for stroke. In: 36th Annual International Conference of the IEEE Engineering in Medicine and Biology Society; 2014 Aug 26-30; Chicago, USA. Chigago: IEEE; 2014. p. 4127-30. http://dx.doi.org/10.1109/EMBC.2014.6944532.
http://dx.doi.org/10.1109/EMBC.2014.6944...
. The relation between movement and observation in the motor cortex was discussed by Tehovnik et al. (2013)Tehovnik EJ, Woods LC, Slocum WM. Transfer of information by BMI. Neuroscience. 2013; 255:134-46. http://dx.doi.org/10.1016/j.neuroscience.2013.10.003. PMid:24120558.
http://dx.doi.org/10.1016/j.neuroscience...
.
There is a weaker response in the central areas when movement is only observed. This may suggest that the observation elicits similar responses to movement or imagery but without activating the primary motor cortex, that is, without reaching the area where the movement generation is commanded. This is somehow expected given that the observation is a passive task, if compared to motion or imagery.
More research on different types of movements and the effects of visuomotor feedback in a working BMI must be performed. This kind of research may help to unveil deeper insights about the physiological basis of motion and may help in the development of BMIs. So far, the discrimination of movement types between flexion and extension has been achieved in the work by Silva et al. (2015)Silva AST, Sá AMFLM, Tierra-Criollo CJ. Functional connectivity during elbow movements: comparison between motor and non-motor task. In: Proceedings of the 6th European Conference of the International Federation for Medical and Biological Engineering; 2014 Sept 7-11; Dubrovnik, Croatia. Heidelberg: Springer; 2015. p. 938-41. http://dx.doi.org/10.1007/978-3-319-11128-5_233.
http://dx.doi.org/10.1007/978-3-319-1112...
.
The implemented SFT-based algorithm has proved to be a suitable option for objective detection of movement intention. However, improvements are needed, especially those related to feature extraction, as well as adaptive and optimal parameterization. The reactive frequency is already automatically determined as the peak of frequency at resting state, but optimization is still required for the selection of the window size for spectrum computation and to obtain the value for the forgetting factor .
Three of the volunteers performed the motor imagery worse. This is noted if we compare the detection rates for movement execution and imagery in Figure 7. A possible cause for the poor imagery performance can be related to the novelty of the experiment for those specific volunteers. Studies about ERD modulation have reported the importance of task training to improve the brain responses (Doud et al., 2011Doud AJ, Lucas JP, Pisansky MT, He B. Continuous three dimensional control of a virtual helicopter using a motor imagery based brain-computer interface. PLoS One. 2011; 6(10):e26322. http://dx.doi.org/10.1371/journal.pone.0026322. PMid:22046274.
http://dx.doi.org/10.1371/journal.pone.0...
; Jeon et al., 2011Jeon Y, Nam CS, Kim YJ, Whang MC. Event-related (De)synchronization (ERD/ERS) during motor imagery tasks: implications for brain-computer interfaces. International Journal of Industrial Ergonomics. 2011; 41(5):428-36. http://dx.doi.org/10.1016/j.ergon.2011.03.005.
http://dx.doi.org/10.1016/j.ergon.2011.0...
; Pfurtscheller et al., 1998Pfurtscheller G, Neuper C, Schlögl A, Lugger K. Separability of EEG signals recorded during right and left motor imagery using adaptive autoregressive parameters. IEEE Transactions on Rehabilitation Engineering. 1998; 6(3):316-25. http://dx.doi.org/10.1109/86.712230. PMid:9749909.
http://dx.doi.org/10.1109/86.712230...
; Pfurtscheller and Neuper, 2001Pfurtscheller G, Neuper C. Motor imagery and direct brain-computer communication. Proceedings of the IEEE. 2001; 89(7):1123-34. http://dx.doi.org/10.1109/5.939829.
http://dx.doi.org/10.1109/5.939829...
). This training would increase the difference between spontaneous and movement-related conditions and consequently the chance of successful detections. In addition, some of the volunteers reported difficulty to imagine the movement of their own arms while staring at the moving robot.
In conclusion, the main contribution of this paper is the method for objective detection of movement intention. Different from most classifiers (Bai et al., 2007; 2011Bai O, Lin P, Vorbach S, Li J, Furlani S, Hallett M. Exploration of computational methods for classification of movement intention during human voluntary movement from single trial EEG. Clinical Neurophysiology. 2007; 118(12):2637-55. http://dx.doi.org/10.1016/j.clinph.2007.08.025. PMid:17967559.
http://dx.doi.org/10.1016/j.clinph.2007....
; Bhagat, 2014Bhagat NA. Detecting movement intent from scalp EEG in a novel upper limb robotic rehabilitation system for stroke. In: 36th Annual International Conference of the IEEE Engineering in Medicine and Biology Society; 2014 Aug 26-30; Chicago, USA. Chigago: IEEE; 2014. p. 4127-30. http://dx.doi.org/10.1109/EMBC.2014.6944532.
http://dx.doi.org/10.1109/EMBC.2014.6944...
; Jiang et al., 2015Jiang N, Gizzi L, Mrachacz-Kersting N, Dremstrup K, Farina D. A brain-computer interface for single-trial detection of gait initiation from movement related cortical potentials. Clinical Neurophysiology. 2015; 126(1):154-9. http://dx.doi.org/10.1016/j.clinph.2014.05.003. PMid:24910150.
http://dx.doi.org/10.1016/j.clinph.2014....
; Kamavuako et al., 2015Kamavuako EN, Jochumsen M, Niazi IK, Dremstrup K. Comparison of features for movement prediction from single-trial movement-related cortical potentials in healthy subjects and stroke patients. Computational Intelligence and Neuroscience. 2015; 2015:1-8. http://dx.doi.org/10.1155/2015/858015. PMid:26161089.
http://dx.doi.org/10.1155/2015/858015...
; Lew et al., 2012Lew EYL, Chavarriaga R, Silvoni S, Millán JR. Detection of self-paced reaching movement intention from EEG signals. Frontiers in neuroengineering. 2012; 5:1-17. PMid:23055968.; 2014Lew EYL, Chavarriaga R, Silvoni S, Millán JR. Single trial prediction of self-paced reaching directions from EEG signals. Frontiers in Neuroscience. 2014; 8:1-13. http://dx.doi.org/10.3389/fnins.2014.00222. PMid:25136290.
http://dx.doi.org/10.3389/fnins.2014.002...
; Nikulin et al., 2008Nikulin VV, Hohlefeld FU, Jacobs AM, Curio G. Quasi-movements: a novel motor-cognitive phenomenon. Neuropsychologia. 2008; 46(2):727-42. http://dx.doi.org/10.1016/j.neuropsychologia.2007.10.008. PMid:18035381.
http://dx.doi.org/10.1016/j.neuropsychol...
), the detector does not need training data to determine its parameters. The only previous knowledge needed for the SFT detector is the electrophysiological response, i.e., the only characteristic needed is the desynchronization present as a persistent feature related to movement intention, which is used to detect whether the volunteer intended a movement or not. This characteristic may be considered invariant among subjects and experiment conditions.
The BMI paradigm we use has two focuses: the triggering of high-level tasks by the electrophysiological activity of the BMI user obtained through EEG signals, and the execution and solution of several kinds of task by using robotic systems. Both of these aspects are treated as separate parts of our current research. However, we always keep a path that will allow the appropriate integration of these aspects into a single BMI.
As future work, we intend to extend and improve the proposed algorithm in order to detect more types of movement intention. In addition, we will explore other biosignal sources to increase the amount and robustness of control commands, as done in hybrid interfaces research (Allison et al., 2012Allison BZ, Brunner C, Altstätter C, Wagner IC, Grissmann S, Neuper C. A hybrid ERD/SSVEP BCI for continuous simultaneous two dimensional cursor control. Journal of Neuroscience Methods. 2012; 209(2):299-307. http://dx.doi.org/10.1016/j.jneumeth.2012.06.022. PMid:22771715.
http://dx.doi.org/10.1016/j.jneumeth.201...
; Fazli et al., 2012Fazli S, Mehnert J, Steinbrink J, Curio G, Villringer A, Müller KR, Blankertz B. Enhanced performance by a hybrid NIRS-EEG brain computer interface. NeuroImage. 2012; 59(1):519-29. http://dx.doi.org/10.1016/j.neuroimage.2011.07.084. PMid:21840399.
http://dx.doi.org/10.1016/j.neuroimage.2...
; Pfurtscheller et al., 2010Pfurtscheller G, Allison BZ, Brunner C, Bauernfeind G, Solis-Escalante T, Scherer R, Zander TO, Mueller-Putz G, Neuper C, Birbaumer N. The hybrid BCI. Frontiers in Neuroscience. 2010; 4:1-11. PMid:20582271.).
Acknowledgements
This work has been supported by the Brazilian agencies CAPES, CNPq and FAPEMIG.
References
- Allison BZ, Brunner C, Altstätter C, Wagner IC, Grissmann S, Neuper C. A hybrid ERD/SSVEP BCI for continuous simultaneous two dimensional cursor control. Journal of Neuroscience Methods. 2012; 209(2):299-307. http://dx.doi.org/10.1016/j.jneumeth.2012.06.022. PMid:22771715.
» http://dx.doi.org/10.1016/j.jneumeth.2012.06.022 - Bai O, Lin P, Vorbach S, Li J, Furlani S, Hallett M. Exploration of computational methods for classification of movement intention during human voluntary movement from single trial EEG. Clinical Neurophysiology. 2007; 118(12):2637-55. http://dx.doi.org/10.1016/j.clinph.2007.08.025. PMid:17967559.
» http://dx.doi.org/10.1016/j.clinph.2007.08.025 - Bai O, Rathi V, Lin P, Huang D, Battapady H, Fei D-Y, Schneider L, Houdayer E, Chen X, Hallett M. Prediction of human voluntary movement before it occurs. Clinical Neurophysiology. 2011; 122(2):364-72. http://dx.doi.org/10.1016/j.clinph.2010.07.010. PMid:20675187.
» http://dx.doi.org/10.1016/j.clinph.2010.07.010 - Behrmann M, Geng JJ, Shomstein S. Parietal cortex and attention. Current Opinion in Neurobiology. 2004; 14(2):212-7. http://dx.doi.org/10.1016/j.conb.2004.03.012. PMid:15082327.
» http://dx.doi.org/10.1016/j.conb.2004.03.012 - Bhagat NA. Detecting movement intent from scalp EEG in a novel upper limb robotic rehabilitation system for stroke. In: 36th Annual International Conference of the IEEE Engineering in Medicine and Biology Society; 2014 Aug 26-30; Chicago, USA. Chigago: IEEE; 2014. p. 4127-30. http://dx.doi.org/10.1109/EMBC.2014.6944532.
» http://dx.doi.org/10.1109/EMBC.2014.6944532 - Cecotti H. Spelling with non-invasive brain-computer interfaces: current and future trends. The Journal of Physiology. 2011; 105(1-3):106-14. PMid:21911058.
- Doud AJ, Lucas JP, Pisansky MT, He B. Continuous three dimensional control of a virtual helicopter using a motor imagery based brain-computer interface. PLoS One. 2011; 6(10):e26322. http://dx.doi.org/10.1371/journal.pone.0026322. PMid:22046274.
» http://dx.doi.org/10.1371/journal.pone.0026322 - Fatourechi M, Bashashati A, Ward RK, Birch GE. EMG and EOG artifacts in brain computer interface systems: a survey. Clinical Neurophysiology. 2007; 118(3):480-94. http://dx.doi.org/10.1016/j.clinph.2006.10.019. PMid:17169606.
» http://dx.doi.org/10.1016/j.clinph.2006.10.019 - Fazli S, Mehnert J, Steinbrink J, Curio G, Villringer A, Müller KR, Blankertz B. Enhanced performance by a hybrid NIRS-EEG brain computer interface. NeuroImage. 2012; 59(1):519-29. http://dx.doi.org/10.1016/j.neuroimage.2011.07.084. PMid:21840399.
» http://dx.doi.org/10.1016/j.neuroimage.2011.07.084 - Giuliana G, Mario M, Yassin J. A quality parameter for the detection of the intentionality of movement in patients with neurological tremor performing a finger-to-nose test. In: 33rd Annual International Conference of the IEEE EMBS; 2011 Aug 30-Sept 03; Boston, USA. Boston: IEEE; 2011. p. 7707-10. http://dx.doi.org/10.1109/IEMBS.2011.6091899.
» http://dx.doi.org/10.1109/IEMBS.2011.6091899 - Hochberg LR, Bacher D, Jarosiewicz B, Masse NY, Simeral JD, Vogel J, Haddadin S, Liu J, Cash SS, van der Smagt P, Donoghue JP. Reach and grasp by people with tetraplegia using a neurally controlled robotic arm. Nature. 2012; 485(7398):372-5. http://dx.doi.org/10.1038/nature11076. PMid:22596161.
» http://dx.doi.org/10.1038/nature11076 - Infantosi AF, Sá AM. A statistical test for evaluating the event-related synchronization/desynchronization and its potential use in brain-computer-interfaces. IFMBE Proceedings. 2007; 18:1122-6. http://dx.doi.org/10.1007/978-3-540-74471-9_260.
» http://dx.doi.org/10.1007/978-3-540-74471-9_260 - Jeon Y, Nam CS, Kim YJ, Whang MC. Event-related (De)synchronization (ERD/ERS) during motor imagery tasks: implications for brain-computer interfaces. International Journal of Industrial Ergonomics. 2011; 41(5):428-36. http://dx.doi.org/10.1016/j.ergon.2011.03.005.
» http://dx.doi.org/10.1016/j.ergon.2011.03.005 - Jiang N, Gizzi L, Mrachacz-Kersting N, Dremstrup K, Farina D. A brain-computer interface for single-trial detection of gait initiation from movement related cortical potentials. Clinical Neurophysiology. 2015; 126(1):154-9. http://dx.doi.org/10.1016/j.clinph.2014.05.003. PMid:24910150.
» http://dx.doi.org/10.1016/j.clinph.2014.05.003 - Kalcher J, Pfurtscheller G. Discrimination between phaselocked and non-phase-locked event-related EEG activity. Electroencephalography and Clinical Neurophysiology. 1995; 94(5):381-4. http://dx.doi.org/10.1016/0013-4694(95)00040-6. PMid:7774524.
» http://dx.doi.org/10.1016/0013-4694(95)00040-6 - Kamavuako EN, Jochumsen M, Niazi IK, Dremstrup K. Comparison of features for movement prediction from single-trial movement-related cortical potentials in healthy subjects and stroke patients. Computational Intelligence and Neuroscience. 2015; 2015:1-8. http://dx.doi.org/10.1155/2015/858015. PMid:26161089.
» http://dx.doi.org/10.1155/2015/858015 - Kotchoubey B. Event-related potentials, cognition, and behavior: a biological approach. Neuroscience and Biobehavioral Reviews. 2006; 30(1):42-65. http://dx.doi.org/10.1016/j.neubiorev.2005.04.002. PMid:16033699.
» http://dx.doi.org/10.1016/j.neubiorev.2005.04.002 - Lana EP, Adorno BV, Tierra-Criollo CJ. An ERD/ERS analysis of the relation between human arm and robot manipulator movements. In: IEEE/ISSNIP Biosignals and Biorobotics Conference; 2013 Feb 18-20; Rio de Janeiro, Brazil. Rio de Janeiro: IEEE; 2013a. p. 1-7. http://dx.doi.org/10.1109/BRC.2013.6487461.
» http://dx.doi.org/10.1109/BRC.2013.6487461 - Lana EP, Adorno BV, Tierra-Criollo CJ. Assistance task using a manipulator robot and user kinematics feedback. In: 11 Simpósio Brasileiro de Automação Inteligente; 2013 Oct 13-17; Fortaleza, Brazil. Fortaleza: Universidade Federal do Ceará; 2013b. p. 1-6.
- Lebedev MA, Nicolelis MAL. Brain-machine interfaces: past, present and future. Trends in Neurosciences. 2006; 29(9):536-46. http://dx.doi.org/10.1016/j.tins.2006.07.004. PMid:16859758.
» http://dx.doi.org/10.1016/j.tins.2006.07.004 - Lew EYL, Chavarriaga R, Silvoni S, Millán JR. Detection of self-paced reaching movement intention from EEG signals. Frontiers in neuroengineering. 2012; 5:1-17. PMid:23055968.
- Lew EYL, Chavarriaga R, Silvoni S, Millán JR. Single trial prediction of self-paced reaching directions from EEG signals. Frontiers in Neuroscience. 2014; 8:1-13. http://dx.doi.org/10.3389/fnins.2014.00222. PMid:25136290.
» http://dx.doi.org/10.3389/fnins.2014.00222 - Li J, Wang Y, Zhang L, Jung TP. Combining ERPs and EEG spectral features for decoding intended movement direction. In: Annual International Conference of the IEEE Engineering in Medicine and Biology Society; 2012 Aug 28-Sep 01; San Diego, USA. San Diego: IEEE; 2012. p. 1769-72.
- Morash V, Bai O, Furlani S, Lin P, Hallett M. Classifying EEG signals preceding right hand, left hand, tongue, and right foot movements and motor imageries. Clinical Neurophysiology. 2008; 119(11):2570-8. http://dx.doi.org/10.1016/j.clinph.2008.08.013. PMid:18845473.
» http://dx.doi.org/10.1016/j.clinph.2008.08.013 - Nair DG, Purcott KL, Fuchs A, Steinberg F, Kelso JAS. Cortical and cerebellar activity of the human brain during imagined and executed unimanual and bimanual action sequences: a functional MRI study. Brain Research. Cognitive Brain Research. 2003; 15(3):250-60. http://dx.doi.org/10.1016/S0926-6410(02)00197-0. PMid:12527099.
» http://dx.doi.org/10.1016/S0926-6410(02)00197-0 - Niazi IK, Jiang N, Tiberghien O, Nielsen JF, Dremstrup K, Farina D. Detection of movement intention from single-trial movement-related cortical potentials. Journal of Neural Engineering. 2011; 8(6):1-10. http://dx.doi.org/10.1088/1741-2560/8/6/066009. PMid:22027549.
» http://dx.doi.org/10.1088/1741-2560/8/6/066009 - Nicolelis MAL, Ribeiro S. Seeking the neural code. Scientific American. 2006; 295(6):70-7. http://dx.doi.org/10.1038/scientificamerican1206-70. PMid:17144353.
» http://dx.doi.org/10.1038/scientificamerican1206-70 - Nicolelis MAL. Brain-machine interfaces to restore motor function and probe neural circuits. Nature Reviews. Neuroscience. 2003; 4(5):417-22. http://dx.doi.org/10.1038/nrn1105. PMid:12728268.
» http://dx.doi.org/10.1038/nrn1105 - Nikulin VV, Hohlefeld FU, Jacobs AM, Curio G. Quasi-movements: a novel motor-cognitive phenomenon. Neuropsychologia. 2008; 46(2):727-42. http://dx.doi.org/10.1016/j.neuropsychologia.2007.10.008. PMid:18035381.
» http://dx.doi.org/10.1016/j.neuropsychologia.2007.10.008 - Pfurtscheller G, Allison BZ, Brunner C, Bauernfeind G, Solis-Escalante T, Scherer R, Zander TO, Mueller-Putz G, Neuper C, Birbaumer N. The hybrid BCI. Frontiers in Neuroscience. 2010; 4:1-11. PMid:20582271.
- Pfurtscheller G, Müller GR, Pfurtscheller J, Gerner HJ, Rupp R. ‘Thought’ - Control of functional electrical stimulation to restore hand grasp in a patient with tetraplegia. Neuroscience Letters. 2003; 351(1):33-6. http://dx.doi.org/10.1016/S0304-3940(03)00947-9. PMid:14550907.
» http://dx.doi.org/10.1016/S0304-3940(03)00947-9 - Pfurtscheller G, Neuper C, Schlögl A, Lugger K. Separability of EEG signals recorded during right and left motor imagery using adaptive autoregressive parameters. IEEE Transactions on Rehabilitation Engineering. 1998; 6(3):316-25. http://dx.doi.org/10.1109/86.712230. PMid:9749909.
» http://dx.doi.org/10.1109/86.712230 - Pfurtscheller G, Neuper C. Motor imagery and direct brain-computer communication. Proceedings of the IEEE. 2001; 89(7):1123-34. http://dx.doi.org/10.1109/5.939829.
» http://dx.doi.org/10.1109/5.939829 - Pfurtscheller G, Silva FHL. Event-related EEG/MEG synchronization and desynchronization: Basic principles. Clinical Neurophysiology. 1999; 110(11):1842-57. http://dx.doi.org/10.1016/S1388-2457(99)00141-8. PMid:10576479.
» http://dx.doi.org/10.1016/S1388-2457(99)00141-8 - Pineda JA, Allison BZ, Vankov A. The effects of self-movement, observation, and imagination on alpha rhythms and readiness potentials (RP’s): toward a brain-computer interface (BCI). IEEE Transactions on Rehabilitation Engineering. 2000; 8(2):219-22. http://dx.doi.org/10.1109/86.847822. PMid:10896193.
» http://dx.doi.org/10.1109/86.847822 - Santos S Fo, Tierra-Criollo CJ, Souza AP, Pinto MAS, Lima MLC, Manzano GM. Magnitude squared of coherence to detect imaginary movement. EURASIP Journal on Advances in Signal Processing. 2009; 2009(1):534536. http://dx.doi.org/10.1155/2009/534536.
» http://dx.doi.org/10.1155/2009/534536 - Santos-Couto-Paz CC, Teixeira-Salmela LF, Tierra-Criollo CJ. The addition of functional task-oriented mental practice to conventional physical therapy improves motor skills in daily functions after stroke. Brazilian Journal of Physical Therapy. 2013; 17(6):564-71. http://dx.doi.org/10.1590/S1413-35552012005000123. PMid:24271094.
» http://dx.doi.org/10.1590/S1413-35552012005000123 - Silva AST, Sá AMFLM, Tierra-Criollo CJ. Functional connectivity during elbow movements: comparison between motor and non-motor task. In: Proceedings of the 6th European Conference of the International Federation for Medical and Biological Engineering; 2014 Sept 7-11; Dubrovnik, Croatia. Heidelberg: Springer; 2015. p. 938-41. http://dx.doi.org/10.1007/978-3-319-11128-5_233.
» http://dx.doi.org/10.1007/978-3-319-11128-5_233 - Simpson DM, Tierra-Criollo CJ, Leite RT, Zayen EJ, Infantosi AF. Objective response detection in an electroencephalogram during somatosensory stimulation. Annals of Biomedical Engineering. 2000; 28(6):691-8. http://dx.doi.org/10.1114/1.1305530. PMid:10983714.
» http://dx.doi.org/10.1114/1.1305530 - Sirvent Blasco J, Iáñez E, Úbeda A, Azorín J. Visual evoked potential-based brain-machine interface applications to assist disabled people. Expert Systems with Applications. 2012; 39(9):7908-18. http://dx.doi.org/10.1016/j.eswa.2012.01.110.
» http://dx.doi.org/10.1016/j.eswa.2012.01.110 - Tehovnik EJ, Woods LC, Slocum WM. Transfer of information by BMI. Neuroscience. 2013; 255:134-46. http://dx.doi.org/10.1016/j.neuroscience.2013.10.003. PMid:24120558.
» http://dx.doi.org/10.1016/j.neuroscience.2013.10.003 - Tierra-Criollo CJ. Monitorização objetiva da resposta à estimulação somato-sensitiva utilizando parâmetros espectrais [thesis]. Rio de Janeiro: Universidade Federal do Rio de Janeiro; 2001.
- Yuan H, Perdoni C, He B. Relationship between movement speed and EEG activity during imagined and executed hand movements. Journal of Neural Engineering. 2010; 7(2):26001. http://dx.doi.org/10.1088/1741-2560/7/2/026001. PMid:20168002.
» http://dx.doi.org/10.1088/1741-2560/7/2/026001
Publication Dates
-
Publication in this collection
27 Nov 2015 -
Date of issue
Oct-Dec 2015
History
-
Received
10 May 2015 -
Accepted
02 Sept 2015