Abstracts
In this study the K, U and Th radionuclide concentrations were determined in a sugar cane plantation that received large quantities of fertilizer and gypsum. Geochemical data were obtained for the clay, iron and organic matter in the soil samples, taken from depths of 0-20 and 20-40 cm. The average radionuclide concentrations were 71 Bq Kg-1 for K; 5.97 Bq Kg-1 for U, and 11.26 Bq Kg-1 for Th, and the uranium was variable (56%). The radionuclide average concentrations for the different fertilizer compositions varied in the range of 603.5 - 8855.0 Bq Kg-1 for K; 27.3 - 684.4 Bq Kg-1 for U, and 58 - 788.5 Bq Kg-1 for Th, and it was most variable for K (53%). Gypsum average concentrations were 82.6 Bq Kg-1 for K, 641.5 Bq Kg-1 for U, and 201.7 Bq Kg-1 for Th, and it was most variable for K (46%). Average organic matter, clay and iron concentrations for both the depths were 0.71 and 0.68 %; 16.2 and 16.9 %, and 29.0 and 26.8 mg/l respectively.
Fertilizers; soils; radionuclides; gamma-ray spectrometry
Determinou-se as concentrações dos radionuclídeos K, U e Th em área cultivada com cana-de-açúcar que recebe quantidades expressivas de adubos e gesso. Os radionuclídeos apresentaram teores médios de 71 Bq Kg-1 de K, 5,97 Bq Kg-1 de U e 11,26 Bq Kg-1 de Th, sendo a maior variabilidade detectada para o urânio (56%). Os teores médios dos radionuclídeos para as diferentes formulações dos adubos encontram-se na faixa de 603,5 - 8855 Bq Kg-1 de K, 27,3 - 684,4 Bq Kg-1 de U e 58 - 788,5 Bq Kg-1 de Th, com maior variação para o K (53%). O gesso apresentou valores médios 82,6 Bq Kg-1 de K, 641,5 Bq Kg-1 de U e 201,7 Bq Kg-1 de Th, cuja maior variabilidade também foi para o K (46%). Os teores médios da matéria orgânica, argila e ferro para as duas profundidades foram respectivamente de: 0,71 e 0,68 %; 16,2 e 16,9 %; 29,0 e 26,8 mg/l.
AGRICULTURE, AGRIBUSINESS AND BIOTECHNOLOGY
Gamma-ray spectrometry sensor and geochemical prospecting in an area of sugar cane plantation
Valter Antonio BecegatoI, * * Author for correspondence ; Francisco José Fonseca FerreiraII; João Batista Pereira CabralIII; Sílvio Luis Rafaelli NetoI
IDepartamento de Engenharia Rural; CAV; Universidade do Estado de Santa Catarina; C.P. 281; 88520-000; Lages - SC - Brasil
IILaboratório de Geofísica Aplicada; Universidade Federal do Paraná; C.P. 19001; Curitiba - PR - Brasil
IIIUniversidade Federal de Goiás; 7580-000; Jataí; Goiás - GO - Brasil
ABSTRACT
In this study the K, U and Th radionuclide concentrations were determined in a sugar cane plantation that received large quantities of fertilizer and gypsum. Geochemical data were obtained for the clay, iron and organic matter in the soil samples, taken from depths of 0-20 and 20-40 cm. The average radionuclide concentrations were 71 Bq Kg-1 for K; 5.97 Bq Kg-1 for U, and 11.26 Bq Kg-1 for Th, and the uranium was variable (56%). The radionuclide average concentrations for the different fertilizer compositions varied in the range of 603.5 - 8855.0 Bq Kg-1 for K; 27.3 - 684.4 Bq Kg-1 for U, and 58 - 788.5 Bq Kg-1 for Th, and it was most variable for K (53%). Gypsum average concentrations were 82.6 Bq Kg-1 for K, 641.5 Bq Kg-1 for U, and 201.7 Bq Kg-1 for Th, and it was most variable for K (46%). Average organic matter, clay and iron concentrations for both the depths were 0.71 and 0.68 %; 16.2 and 16.9 %, and 29.0 and 26.8 mg/l respectively.
Key words: Fertilizers, soils, radionuclides, gamma-ray spectrometry
RESUMO
Determinou-se as concentrações dos radionuclídeos K, U e Th em área cultivada com cana-de-açúcar que recebe quantidades expressivas de adubos e gesso. Os radionuclídeos apresentaram teores médios de 71 Bq Kg-1 de K, 5,97 Bq Kg-1 de U e 11,26 Bq Kg-1 de Th, sendo a maior variabilidade detectada para o urânio (56%). Os teores médios dos radionuclídeos para as diferentes formulações dos adubos encontram-se na faixa de 603,5 - 8855 Bq Kg-1 de K, 27,3 - 684,4 Bq Kg-1 de U e 58 - 788,5 Bq Kg-1 de Th, com maior variação para o K (53%). O gesso apresentou valores médios 82,6 Bq Kg-1 de K, 641,5 Bq Kg-1 de U e 201,7 Bq Kg-1 de Th, cuja maior variabilidade também foi para o K (46%). Os teores médios da matéria orgânica, argila e ferro para as duas profundidades foram respectivamente de: 0,71 e 0,68 %; 16,2 e 16,9 %; 29,0 e 26,8 mg/l.
INTRODUCTION
Modern agriculture adopts high technology, aiming at increasing the productivity. Cultures such as soybean, corn and sugar cane use cultivars with great genetic potential that allied with fertilizers, result in the high productivity. It is impossible to imagine the Brazilian agriculture in the last 20 years without the utilization of large amounts of the fertilizers. This, however interferes with the environment (Guiraud et al., 2004), but the consequences are still poorly known (Andrello et al., 2003). There are several formula and trademarks of the fertilizers in the market, with variable nitrogen, phosphorous and potassium contents. Gypsum, a by-product of the phosphate fertilizers, has been used recently in agriculture. Calcium sulfate dehydrate, derived from the (phosphoric acid) contains calcium, sulfur and water in varied concentrations. This is used in order to supply calcium and sulfur to deeper soil horizons, so that the plant root system, in search of nutrients and water, can develop by exploring a larger volume of soil (Nuernberg et al., 2002).
Radioactivity is a natural phenomenon, typical of elements with the large and unstable nucleus. It is an irreversible mass and energy-loss process: the atoms disintegrate until a stable condition is reached. Marques (1988), studyng acid, intermediate and basic rocks from the Paraná Basin, found varied uranium and thorium contents, the maximum corresponding to rhyolite floods. Dickson and Scott (1977) noted that for the same type of rock, variations in the U, Th and K radionuclide concentrations can occur. According to these authors, the average content of uranium in the crust is approximately 3 ppm and of the two valences, U+4 and U+6, the more reduced is found in insoluble minerals, and U+6 is complexed with anions, sulfates and phosphates, forming some soluble species. The average thorium content is approximately 12 ppm, with Th+3 and Th+4 valences (Boyle, 1982).
There are several fertilizer trademarks and commercial formula. Pfister et al., (1976) observed the higher radionuclide concentration in the triple superphosphate in relation to simple super results from the manufacturing process. To obtain the triple superphosphate, the rock is treated with the phosphoric acid, retaining uranium as a dissolved uranil complex. Monoammonium phosphate and diammonium phosphate are obtained by dosing the phosphoric acid and ammonium.
The soils differ in their capacity of cation retention. The cation change capacity (CCC) depends on the organic matter texture and content. More clayey soils retain more cations, whereas in sandy soils, where the water percolation is faster, chemical elements in solution in the soil are leached. CCC can vary between 10 and 150 cmomc Kg-1. Organic matter can increase the cation change variation from 200 to 400 cmolc Kg-1 (Santos and Camargo, 1999).
This work aimed at determining the radionuclide concentrations in the soils, fertilizers and gypsum, in order to understand the K, U and Th dynamics in sandy soils bearing sugar cane plantation.
MATERIALS AND METHODS
The study area was a sugar cane plantation for the production of sugar and alcohol. According to the climatic chart of the State of Paraná (Iapar, 1978) and adopting Köeppens classification, it is a mesothermal, humid subtropical climate (Cfa), with hot summer, rains tending to concentrate in the summer months. The annual average precipitation is 1200 mm. The area is set in the third Paranaense high plateau - Bauru Basin (Fig. 1), Caiuá Group - Rio Paraná Formation. It is constituted by well-selected sandstones in lenses or strata, with well-rounded grains in the coarser fractions and scarce silt-argillaceous matrix (Fernandes and Coimbra, 1994).
According to Fernandes and Coimbra (2000), the depositional context encompasses large-sized eolic constructions and dune complexes of amalgamated winding crests, the former built from fossil dune eolic sandstones and the latter corresponding to subaqueous deposits. The sedimentation environment is characterized by the severe, arid to semi-arid climatic conditions. The sandstone lies discordantly on the Serra Geral Formation. Gimenez Filho et al., (1983) described the landscape as dominated by ample, short-amplitude hills with straight and continuous convex profiles and extensive planar tops. The ramps are long and slope gently to steeply; close to the drainage channels, the declivity is higher. The predominant soil is Distrophic Quartzarenic Neosoil of very sandy texture.
A Syntrex/Geofyzika GS-512 spectrometer was used to collect the gamma-ray radiation produced by K, eU and eTh. The data in the cps were automatically converted to %K, and eU and eTh ppm. The equipment was calibrated in the Institute of Radioprotection and Dosimetry (IRD) of the National Commission of Nuclear Energy (CNEN), following the procedures described in Barreto et al., (1986). The data were directly obtained from the points on the soils (Fig. 2), as well as in the fertilizer and gypsum samples used in the sugar cane plantation.
The radioactivity was determined by the detection of gamma rays. The main gamma radiation sources are potassium (K), uranium (U) and thorium (Th), which emit gamma quanta in the energy interval above 2615 KeV. The potassium spectrometry is based on the detection of the 40K isotope, whose natural abundance is 0.0012% of total K. The 40K isotope emits gamma rays of 1461 KeV. The determination is direct and the results are expressed in % of total K. To determine uranium concentrations, gamma rays coming from 214Bi, 1764 KeV, a product of the 238U disintegration series, were detected in the field. Thus, the determination was indirect and the data were expressed in ppm eU. An analogous procedure was followed for the thorium determination.
The gamma rays coming from 208Tl, 2615KeV, a product of the 232Th disintegration series, were detected in the field and the results expressed in ppm eTh. Soil samples were collected 79 points, at depths of 0-20 and 20-40 cm, totaling 158 samples. In the laboratory, the following analyses took place: organic matter, iron and clay, following the procedures described by Tedesco et al., (1995). The points had been interpolated by the method of the Kriging to generate the surface maps.
RESULTS AND DISCUSSION
The average contents for the 79 points were 71 Bq Kg-1 K, 5.97 Bq Kg-1 eU and 11.26 Bq Kg-1 eTh (Table 1). Souza (1998) got average eU content of 86.1 Bq Kg-1 for the clay soils; Santos Júnior et al., (2005) 1.827 Bq Kg-1 de K; Baixeras et al., (2001), 98.89 Bq Kg-1 eU and 47.90 Bq Kg-1 eTh; Kumru and Bakaç (2003), average values of 135 Bq Kg-1 eU and 17.05 Bq Kg-1 eTh; Kannan et al., (2002), 16 Bq Kg-1 to 359.65 Bq Kg-1 eU in soil samples from India and Boyle (1982), who obtained 359.65 Bq Kg-1 eU.
The largest variation coefficient was 56% for the uranium and the smallest 27% for the potassium. The uranium distribution was slightly positively skewness, whereas thorium showed a strong positive skewness.
The radionuclide contents of the fertilizers used in the study area are presented in Table 3. Table 4 shows 238U values for the phosphate fertilizers from the various countries, plus those of the study area.
The mean radionuclide contents found in the gypsum samples were 82 Bq Kg-1 K, 641 Bq Kg-1 U and 201 Bq Kg-1 Th (Table 3), comparatively with those of Bolivar et al., (1995) 600 Bq Kg-1 U in the gypsum deposits from the fertilizer factories, Aguirre et al., (1995) with intervals from 4.3 to 502 Bq Kg-1 U and 2.4 to 156 Bq Kg-1 Th in the river sediments close to the fertilizer factories.
The fact that higher U and Th concentrations were not detected in the soils was possibly due to a leaching process favored by the soil sandy texture and the low organic matter content (Table 2). The low cationic chance capacity did not promote the radionuclide retention and this was corroborated by Pfister et al., (1976). The effect of the texture in the mobility of the radionuclides was similar to that of the divalent cations Ca2+ and Mg2+ (Morstvedt, 1992). The divalent cations were adsorbed by clays in the soil by cation exchange. When organic matter lacks in the sandy soils, U is usually mobile and transported as hexavalent carbonate complex or a divalent uranil ion (Rothbaum et al., 1979). Low concentrations could also be related to the radionuclide losses and the laminar erosion was not only due to the strong superficial erosion during the soil preparation, but also due to the intense movement of the farming machinery when the sugar cane plantation was installed. Guimond and Hardin (1989) reported that the Mississippi river and its banks received large amounts of the radionuclides coming from the fertilized areas that underwent laminar erosion, once the river cross ten American states that largely consumed phosphate fertilizers.
The organic matter can raise the CCC of the soils to 400 cmolc Kg-1 (Santos and Camargo, 1999). The low organic matter contents, 0.71 and 0.68% (Table 2), in both investigated depths were the consequence of the procedure adopted before the harvest, the burning of the cane remain in order to eliminate the part of them and made the harvest easier, increasing the efficiency and the mechanical storage (Mendonza et al., 2000).
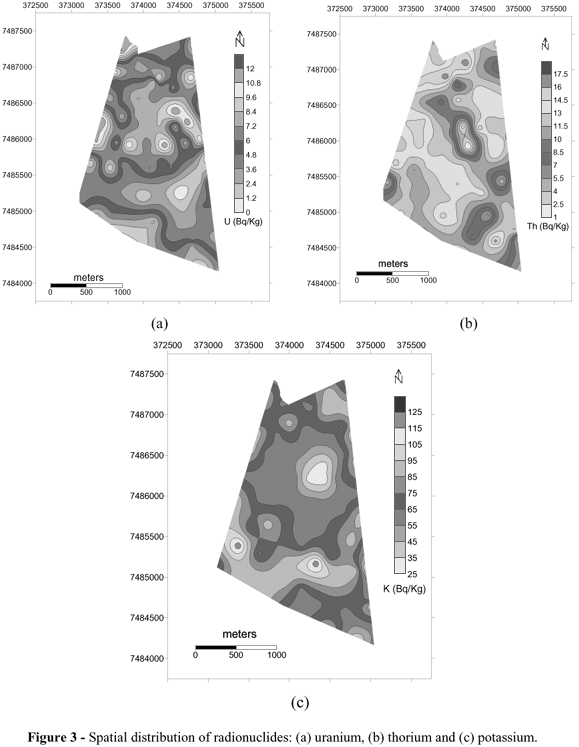
The spatial distribution of the radionuclides (Fig. 3) and the geochemical attributes could be seen in Table 2 for the two depths. The variation coefficients for the clay were 19 and 18% for the organic matter, 25 and 22% for the iron, variable between 95 and 72%. The clay spatial distribution indicated in Figures 4a and 4b was positive skewness (Table 2). The predominating values fell into the 12-18% interval for both the depths.
The organic matter spatial distribution is represented in Figures 4c and 4d. Higher contents should be expected for the 0-20 cm depth in horizon A. Besides the burning before the harvest, high temperatures and rain led to the fast decomposition, caused by the soil microbial activity, which did not favor accumulation of the organic matter. But taking into consideration the mean values in Table 2, a small difference was noted for both the depths.
Clay is an important variable in the correlation between the geophisycal and geochemical data, mainly in what concerns the dinamycs of the radionuclides in the soils. Meaningful correlations were noticed between uranium and clay (r=0.90), thorium and clay (r=0.84), that was corroborated by Echeverraria (2001).
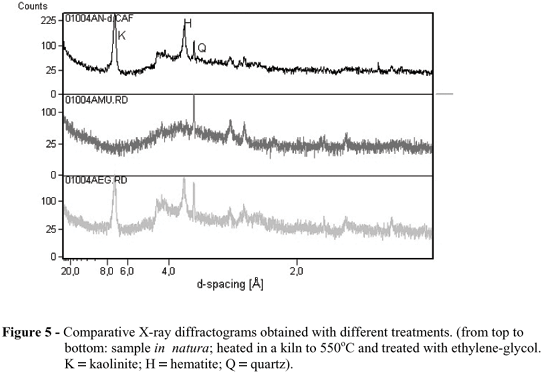
Fig. 5 represented the X-ray diffractogram, showing the presence of Kaolinite and Quartz, an evidence of the presence of the sandy soils, confirmed by the low content of clay 16.2%. The values for iron, mainly in the form of hematite and represented by Figures 6a and 6b, showed a slight difference for both the depths, the highest concentrations occurring between 0-20 cm (Table 2). It was at this depth that the highest variation occurred, 95% against 72% for the 20-40 cm depth.
Received: November 24, 2005;
Revised: October 04, 2006;
Accepted: July 19, 2007.
- Aguirre, A.M.; Léon, M. G. and Ivanovich, M. (1995), U and Th speciation in river sediments. The Science of Total Environment, 173/174, 203-209.
- Andrello, A.C.; Guimarães, M.F.; Appoloni, C.R. and Nascimento Filho, V.F. (2003), Use of Cesium-137 Methodology in the Evaluation of Superficial Erosive Processes. Brazilian Archives of Biology and Technology., 46, (3), 307-314.
- Baixeras, C.; Erlandsson, B.; Font L. and Jonsson G. (2001), Radon emanation from soil samples. Radiation Measurements, 34, 441-443.
- Barisic, D.; Lulic, S. and Miletic, P. (1992), Radium and uranium in phosphate fertilizers and their impact on the radioactivity of waters. Water Res, 26 (5), 607-611.
- Barreto, P.M.C.; Austerlitz, C.; Malheiros, T. and Lovborg, L. (1986), Radioactive concrete sources at IRD/CNEN, Brazil, for the calibration of uranium exploration and environmental field instruments Relatório IRD/DEX-3/CNEN, 66.
- Bolivar, J.P.; Tenório, R.G. and Léon, G. (1995), Fluxes and distribution of natural radionuclides in the production and use of fertilizers. Applied Radiation and Isotopes, 46, (6/7), 717-718.
- Boyle, R.U. (1982), Geochemical prospecting for thorium and uranium deposits. Developments in economic geology, 16, 71-78.
- Dickson, B.L. and Scott, K.M. (1997), Interpretation of aerial gamma-ray surveys - adding the geochemical factors. AGSO Journal of Australian Geology and Geophysics, Austrália, 17, (2), 187-200.
- Echevarria, G.; Sheppard, M.I. and Morel, J.L. (2001), Effect of pH on the sorption of uranium in soils. Journal of Environmental Radioactivity, 53 (2), 257-264.
- Fernandes, L.A. and Coimbra, A.M. (1994), O Grupo Caiuá (Ks): revisão estratigráfica e contexto deposicional. Revista Brasileira de Geociências, 24 (4):164-176.
- Fernandes, L.A. and Coimbra, A.M. (2000), Revisão estratigráfica da parte oriental da bacia Bauru (Neocretáceo). Revista Brasileira de Geociências, 30, (4), 723-734.
- Gimenez, Filho A.; Pires Neto, A. G. and Riccomini, C. (1981), Mapeamento geológico do bloco SF-22-T (ACS-73) São Paulo; Instituto de Pesquisas Tecnológicas, 69 p. (relatório n. 14845).
- Guimond, R.J. and Hardin, J. M. (1989), Radioactivity released from phosphate-containing fertilizers and from gypsum. J. Radiat. Appl. Instrum. Radiat. Phys. Chem, 34, (2), 369-315.
- Guiraud, D.M.C.; Lenzi, E.; Luchese, E.B. and Fávero, L.O.B. (2005), Loss of Macronutrientes (N, P, K) in the Hydrographic Basin of the River Ivaí, an Affluent of the River Paraná. Brazilian Archives of Biology and Technology, 47, (4), 649-658.
- Hermamo, H.; Landsberger, S.; Harbotile, G. and Panno, S. (1995), Studies of radioactivity and heavy metals in phosphate fertilizer. J. Radioanal. Nucl. Chem, 194, 331-336.
- Iapar. (1978), Cartas climáticas básicas do Estado do Paraná Londrina, 41 p.
- Kannan, V.; Rajan, M.P. and Iyengar M.A.A.R.; Ramesh R. (2002), Distribution of natural and anthropogenic radionuclides in soil and beach samples of Kalpakkam (India) using hyper pure germanium (HPGe) gamma ray spectrometry. Applied Radiation and Isotopes, 57, 109-119.
- Kumru, M.N. and Bakaç. M. (2003), R-mode factor analysis applied to the distribution of elements in soils from the Aydin basin, Turkey. Journal of Geochemical Exploration, 77, 81-91.
- Marques, L.S. (1988), Rare elements In: Piccirillo, E.M.; Melfi, A. J. The Mesozoic flood Volcanism of The Paraná Basin. São Paulo: Universidade de São Paulo, Instituto Astronômico e Geosfísico, 499-504.
- Mendonza, H.N.S.; Lima, E.; Anjos, L.H.C.; Silva, L.A.; Ceddia, M.B. and Antunes, M.V.M. (2000), Propriedades químicas e biológicas de solo de tabuleiro cultivado com cana-de-açúcar com e sem queima da palhada. R. bras. Ci. Solo, 24, 201-207.
- Morstvedt, J. J. (1986), Effects of calcium silicate slag application on radium-226 concentrations in plant tissues. Commun. In Soil Sci. Plant. Anal, 17, 75-84.
- Nuernberg, N.J.; Rech, T.D. and Basso, C. (2002), Usos do gesso agrícola Florianópolis: Epagri, 31p. Boletim Técnico, 122.
- Pfister, H.; Philipp, G. and Paul, Y.H. (1976), Population dose from natural radionuclides in phosphate fertilizers. Radiation and Environmental Biophysics, 13, 247-261.
- Rothbaum, H.P.; Mcgaveston, D.A.; Wall, T.; Johnston, A.E. and Mattingly, G.E.G. (1979), Uranium acumulation in soils from long-continued applications of superphosphate. Journal of Soil Science, 30, (1), 147-153.
- Santos Júnior, J.A.; Cardoso, J.J.R.F.; Silva, C.M.; Silveira, S.V. and Amaral, R.S. (2005), Analysis of the 40K Levels in Soil using Gamma Spectrometry. Brazilian Archives of Biology and Technology, 48, (Special), 221-228.
- Santos, G. de A.; Camargo, F.A. de O. (1999), Fundamentos da matéria orgânica no solo: ecossistemas tropicais e subtropicais. Editores - Porto Alegre: Genesis, 508p.
- Souza, J.L. (1998), Anomalias aerogamaespectrométricas (K, U e Th) da quadrícula de Araras (SP) e sua relações com processos pedogenéticos e fertilizantes fosfatados, Dissertação de Mestrado, UFPR, Curitiba, Brasil.
- Tedesco, J.M.; Gianello, C.; Bissani, C. A.; Bohnen, H. and Volkweiss, S. J. (1995), Análises de solo, plantas e outros materiais Porto Alegre: Departamento de solos. UFRGS. 174p.
- Vucic, N. and Ilic, Z. (1989), Extraction and spectrophotometric determination of uranium in phosphate fertilizers. Journal Radioanal. Nucl. Chem , 129, 113-120.
- Yamazaki, I.M. and Geraldo, L. P. (2003), Uranium content in phosphate fertilizers commercially produced in Brazil. Applied Radiation and Isotopes, 59, 133-136.
Publication Dates
-
Publication in this collection
11 Apr 2008 -
Date of issue
Feb 2008
History
-
Accepted
19 July 2007 -
Reviewed
04 Oct 2006 -
Received
24 Nov 2005