Abstract
This study aimed at evaluating the bacteriological effects of the treatment of sheep meat contaminated with total coliforms, coliforms at 45 °C and Salmonella spp. by using irradiation at doses of 3 kGy and 5 kGy. Thirty sheep meat samples were collected from animals located in Rio de Janeiro State, Brazil, and then grouped in three lots including 10 samples: non-irradiated (control); irradiated with 3 kGy; and irradiated with 5 kGy. Exposure to gamma radiation in a 137Cs source-driven irradiating facility was perfomed at the Nuclear Defense Section of the Brazilian Army Technological Center (CTEx) in Rio de Janeiro. The samples were kept under freezing temperature (-18 °C) until the analyses, which occurred in two and four months after irradiation. The results were interpreted by comparison with the standards of the current legislation and demonstrated that non-irradiated samples were outside the parameters established by law for all groups of bacteria studied. Gamma irradiation was effective in inactivating those microorganisms at both doses tested and the optimal dose was achieved at 3 kGy. The results have shown not only the need for sanitary conditions improvements in slaughter and processing of sheep meat but also the irradiation effectiveness to eliminate coliform bacteria and Salmonella spp.
food irradiation; microbiology; sheep meat quality
Elimination of coliforms and Salmonella spp. in sheep meat by gamma irradiation treatment
Luciana Salles Vasconcelos HenriquesI; Fábio da Costa HenryII; João Batista BarbosaII; Silvania Alves LadeiraII; Silvia Menezes de Faria PereiraII; Isabela Maria da Silva AntonioIII; Gina Nunes TeixeiraIII; Meire Lelis Leal MartinsII; Helio de Carvalho VitalIV; Dália dos Prazeres RodriguesV; Eliane Moura Falavina dos ReisV
ILaboratório de Zootecnia e Nutrição Animal, Universidade Estadual do Norte Fluminense Darcy Ribeiro, Campos dos Goytacazes, RJ, Brazil
IILaboratório de Tecnologia de Alimentos, Universidade Estadual do Norte Fluminense Darcy Ribeiro, Campos dos Goytacazes, RJ, Brazil
IIILaboratório de Sanidade Animal, Universidade Estadual do Norte Fluminense Darcy Ribeiro, Campos dos Goytacazes, RJ, Brazil
IVSeção de Defesa Nuclear da Divisão de Defesa Química, Biológica e Nuclear, Centro Tecnológico do Exército, Rio de Janeiro, RJ, Brazil
VLaboratório de Enterobactérias, Instituto Oswaldo Cruz, Rio de Janeiro, RJ, Brazil
Send correspondence to Send correspondence to L.S.V. Henriques. Laboratório de Zootecnia e Nutrição Animal Universidade Estadual do Norte Fluminense Darcy Ribeiro Campos dos Goytacazes RJ, Brasil. E-mail: luciana.salles@gmail.com.
ABSTRACT
This study aimed at evaluating the bacteriological effects of the treatment of sheep meat contaminated with total coliforms, coliforms at 45 °C and Salmonella spp. by using irradiation at doses of 3 kGy and 5 kGy. Thirty sheep meat samples were collected from animals located in Rio de Janeiro State, Brazil, and then grouped in three lots including 10 samples: non-irradiated (control); irradiated with 3 kGy; and irradiated with 5 kGy. Exposure to gamma radiation in a 137Cs source-driven irradiating facility was perfomed at the Nuclear Defense Section of the Brazilian Army Technological Center (CTEx) in Rio de Janeiro. The samples were kept under freezing temperature (-18 °C) until the analyses, which occurred in two and four months after irradiation. The results were interpreted by comparison with the standards of the current legislation and demonstrated that non-irradiated samples were outside the parameters established by law for all groups of bacteria studied. Gamma irradiation was effective in inactivating those microorganisms at both doses tested and the optimal dose was achieved at 3 kGy. The results have shown not only the need for sanitary conditions improvements in slaughter and processing of sheep meat but also the irradiation effectiveness to eliminate coliform bacteria and Salmonella spp.
Key words: food irradiation, microbiology, sheep meat quality.
Introduction
The sheep industry is expanding as a result of changes in dietary habits of consumers, who have been seeking for products with higher quality, flavor, tenderness and lower fat contents (Neres et al., 2001). Such factors justify the need for more researches investigating new conservation methods, since foods rich in protein and fatty acids, such as meats and its derivatives, are more susceptible to quality loss during storage (Leonel, 2008).
Food preservation methods have been improving together with sciences advances. Among the alternative technologies that are being adopted for food treatment worldwide, irradiation should be highlighted. Researches show that this conservation method could apply to both industry and consumers interests (Silva, 2008).
Data about the gamma radiation effects on microorganisms found in sheep meat are still limited, especially those who present standards set by a Board Resolution called "Resolução da Diretoria Colegiada" (RDC) N°12, dated 02/01/2001 (Brasil, 2001). This study aimed at evaluating the bacteriological effects of irradiation at gamma doses of 3 kGy and 5 kGy in the treatment of sheep meat contaminated with coliform bacteria and Salmonella spp. Its findings might help the control and prevention of foodborne diseases.
Material and Methods
Sampling
Thirty sheep meat samples (250 g each) were randomly collected from animals located in properties in Rio de Janeiro State, Brazil. The samples were removed immediately after slaughter and chilling of carcasses, individually vacuum packed and identified according to the treatment to be applied as follows: control (non-irradiated), irradiated with 3 kGy and irradiated with 5 kGy. All samples were then transported under refrigeration to the Universidade Estadual do Norte Fluminense Darcy Ribeiro (UENF), where they were kept at freezing temperature (-18 °C) until irradiation.
Irradiation and storage of frozen meat
The samples exposure to gamma radiation from a 137Cs source was performed in a research irradiator at the Nuclear Defense Section of the Army's Technological Center in Guaratiba -RJ. The 137Cs source had 47 kCi of activity and the average dose rate of 1.3 kGy/h, where the irradiation of the samples lasted 2.3 and 3.8 h at doses of 3 kGy and 5 kGy, respectively. Transportation to CTEx and return to UENF were made under refrigeration.
The control and irradiated samples were kept at -18 °C at UENF. Prior to analyses, they were subjected to slow thawing at a temperature of 4 °C in refrigerator for 12 h. Two and four months after irradiation, five samples from each treatment were analyzed at a time. The analyses followed the methodology described by Silva et al. (2001) and the results were interpreted by comparison with the standards of the RDC N°12, dated 02/01/2001 (Brasil, 2001).
Preparation of dilutions and culture media
For the dilutions preparation, the vacuum packages were aseptically opened. Each analytical unit had 25 g, which was aseptically removed from different points of each sample and added to 225 mL of sterile 0.1% peptone water for future analyses of total coliforms and coliforms at 45 °C. For the Salmonella spp. analyses, another 25 g analytical unit of each sample was removed by using the same procedure and added to 225 mL of Lactose Broth (pre-enrichment broth). The homogenization of the analytical unit and the diluent was performed in a Stomacher (60"/230 rpm) (Seward®), where the initial dilution was obtained (10-1). For the preparation of the second dilution (10-2), 1.0 mL of previous dilution (10-1) was aseptically transferred into a tube containing 9 mL of the same diluent. The process was repeated until the 10-3 dilution was obtained. For the Salmonella spp. analyses, only a 10-1 dilution was prepared. The preparation of culture media followed the manufacturer recommendations.
MPN/g determination of total coliforms
An inoculation of 1.0 mL of the dilutions 10-1,10-2 and 10-3 was performed in three sets of three tubes containing 8.0 mL of Lauryl Sulfate Tryptose Broth (LST) which were incubated at 35 °C for 48 h. After this time, the tubes that showed turbidity and gas production (positive) in the interior of the Durham tubes were transferred to the analyses of total coliforms and coliforms at 45 °C. For total coliforms confirmation, a aliquot was transferred to tubes containing 8.0 mL of Brilliant Green Bile Broth (BG), which were then incubated at 35 °C for 24-48 h. Positive results were interpreted in the appropriate table of MPN considering the inoculated dilutions and then the MPN/g was calculated.
MPN/g determination of coliforms at 45 °C
An aliquot was transferred from LST Broth tubes with positive results to the corresponding tubes containing 8.0 mL of Escherichia coli Broth (EC). Incubation was performed in water bath (Tecnal®) at 45.5 °C for 24 h. The number of tubes with gas production in Durham tubes (positives) was recorded and compared with an appropriate MPN table of inoculated dilutions.
Salmonella spp. analysis
After Lactose Broth incubation at 35 °C for 20 h, 1.0 mL was transferred from this broth into two tubes (previously boiled for 10 min with the following media): one containing 10 mL of Tetrathionate Broth (TT), and another containing 10 mL of Selenite Cystine Broth (SC). By using the TT Broth, 0.2 mL of iodine solution was added to each tube. Both broths were incubated at 35 °C for 24 h. Differential plating was performed by shaking the tubes in a shaker vortex (Quimis®) and then by sowing using exhaustion from an aliquot of TT Broth on Hektoen Enteric Agar plates (HE) and Xylose Lysine Deoxycholate Agar (XLD) to obtain pure colonies. The same procedure was repeated with the SC Broth in the same media previously cited. The plates were inverted and incubated at 35 °C for 24 h in an incubator.
The api® 20 E test (bioMérieux®) was performed. Strains with Salmonella spp. biochemical characteristics were sent for serotyping confirmation to Laboratório de Enterobacterias / Instituto Oswaldo Cruz / FIOCRUZ, Rio de Janeiro. Their antigenic characteristics were analyzed with somatic and flagellar antisera according Grimont and Weill (2007).
Statistical analysis
Statistical tests included variance analysis (ANOVA) with a 5% significance level and mean comparisons according to Tukey's test (SAS, 1999).
Results and Discussion
MPN/g determination of total coliforms and coliforms at 45 °C
The results for the total coliforms analyses of sheep meat performed after two and four months of storage counted in the control (non-irradiated) and in samples treated with 3 and 5 kGy are shown in Table 1.
RDC N°12 does not establish microbiological standards for total coliforms in any food category. However, in the present investigation, it was observed that all nonirradiated sheep meat samples were contaminated by total coliforms, detected as colony growth and gas production in the culture media, both in two and four months. It was also observed that there was no significant change in counts for this microorganism group between the two studied periods (p > 0.05).
No growth and/or gas production by total coliforms were observed in the samples irradiated with 3 and 5 kGy doses on both dates (Table 1), evidencing the effectiveness of irradiation for the control of total coliforms in sheep meat. Because it is more economical, faster to apply and has lower chances to cause sensory changes in the meat, the dose of 3 kGy should be preferred.
The presence of high concentrations of coliforms in foods is indicative of failures during processing, heat treatment or inadequate hygiene (Pardi et al., 2001). However within the vast group of total coliforms, which includes about 20 species, not only bacteria from the gastrointestinal tract of humans and warm-blooded animals are present, but also non-enteric species like Serratia and Aeromonas genera. Thus, their presence in foods is less representative as an indicator of fecal contamination than coliforms at 45 °C or E. coli (Silva et al., 2001).
The results from the analyses of coliforms at 45 °C in the non-irradiated samples of sheep meat and samples treated with doses of 3 and 5 kGy are shown in Table 2.
According to RDC No. 12, in a group analysis of five not matured vacuum packed meat samples, only two may present coliforms at 45 °C counting between 103 and 104 to be considered acceptable. In this study, of five non-irradiated samples, evaluated in two months, three had scores within this range, whereas in four months, four samples were between these values (Table 2). Therefore, these results demonstrate that the non-irradiated samples were outside the parameters established by current legislation and inadequate for human consumption from a microbiological standpoint. There was no significant difference in the microorganisms counting from this group between the two periods studied (p > 0.05).
In contrast, there was no growth or gas production of coliforms at 45 °C in samples irradiated with 3 kGy and 5 kGy in both analyzed time intervals, thus, the effectiveness of the treatment of sheep meat by exposure to gamma radiation for the control of such microorganisms has been clearly observed.
The presence of coliforms at 45 °C in foods suggests the presence of fecal contamination, since there is a high incidence of E. coli in this group, which has as its primary habitat humans and animals intestines. However, coliforms at 45 °C also include some strains that can be found in other habitats (plants and soil) like Enterobacter and Klebsiella. For that reason, the direct correlation between this group and the one originating from fecal contamination should be considered cautiously (Silva et al., 2006).
Information about the effect of gamma radiation on microbial population as well as shelf life extension of sheep meat are still limited, especially by taking into account microorganisms that have standards established by the RDC N°12. Therefore, for comparison, the results of this study are consistent with those reported for other species.
Sedeh et al. (2007) conducted a survey in which beef samples were irradiated with doses of 0.5; 1.0; 2.0 and 3.0 kGy and then kept under refrigeration (4-7 °C) for three weeks and then frozen (-18 °C) for 8 months. Microbiological analyses showed that the number of coliforms decreased with increasing doses for both refrigerated and frozen samples, but this effect was more highlighted in those that were kept frozen. It was also observed that the optimal dose found was 3 kGy, i.e. equal to the lower dose tested in this study.
Oliveira et al. (2009) tested the effect of irradiation (1.5 and 3.0 kGy) and two package types (conventional or vacuum ones) in MPN/g determination of total coliforms and coliforms at 45 °C in chicken meat chilled for 30 days. Irradiation at both doses was found to be efficient at both doses in reducing microbial population, both in the group of total coliforms and coliforms at 45 °C, although no significant differences were found among the treatments of thermo tolerant coliforms group. The survey also found that there was no effect between the irradiation dose and the package used. Mantilla et al. (2010) also tested the effect of irradiation with doses of 3 kGy and a modified atmosphere (80% CO2 / 20% N2) on the growth of coliforms bacteria in chicken meat. It was observed that total coliforms only developed in samples packed in air and in non-irradiated and non-modified atmosphere. Coliforms at 45 °C, however, only grew in the non-irradiated air packages, which showed that, regardless of the atmosphere used, there was no growth of coliforms in irradiated samples.
In tests with camel meat samples, Al-Bachir and Zeinou (2009) showed that irradiation extended the shelf life of the samples from less than two weeks (in nonirradiated ones) to over six weeks (in those irradiated with doses of 2, 4 or 6 kGy) by taking into account the group of total coliforms.
Byun et al. (2001) reported that the washing process and irradiation with doses of 3 kGy eliminated coliforms bacteria in natural sausages casings made from lamb and pigs intestines. In addition a 5 kGy dose sufficed to destroy such microorganisms without the need of the washing procedure.
Kanatt et al. (2010) used a variety of meat products ready for preparation and available in Indian grocery stores such as mutton and chicken bites, and reported that the samples irradiated with 2.5 kGy had their shelf life significantly extended and that coliforms at 45°C were destroyed by the irradiation treatment.
Yildirim et al. (2005) tested the effect of irradiation (2.4 and 7 kGy) in a group of coliforms bacteria present in a traditional Turkish dish (Cig Köfte), commonly consumed as an appetizer, having raw beef or lamb meat in its composition. Their study showed that doses of 2 kGy were sufficient to eliminate these microorganisms in that type of food.
Salmonella spp. analyses
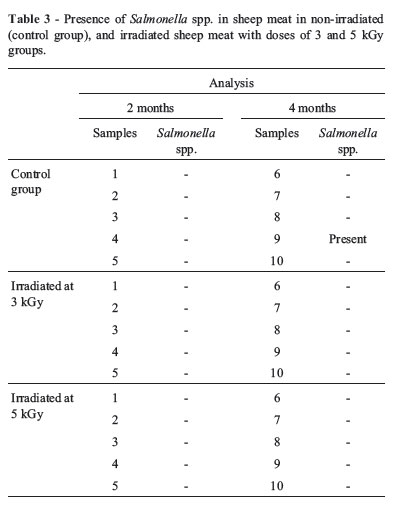
The results obtained for Salmonella spp. analyses of sheep meat performed after two and four months of storages both for control samples and samples irradiated with doses of 3 and 5 kGy, are described in Table 3.
The analyses performed after two months of storage both for control and samples irradiated with doses of 3 and 5 kGy showed that there were no contamination by Salmonella spp. Thus, all samples met the standards established by the RDC N° 12, which determines the absence of this microorganism in a 25 g sample. However, in the analyses performed after four months of storage, in spite of the absence of Salmonella spp. in irradiated samples, both Salmonella enterica subsp. houtenae and Salmonella ser. Albany were isolated in a non-irradiated sample.
Salmonella spp. could cause serious food poisoning. Its presence in non-irradiated samples indicates the need of control measures and food security guarantee.
Jakabi et al. (1999) and Jones et al. (2000) stated that S. enterica subsp. houtenae has cold-blooded animals and the environment as a natural habitat. Some researchers, however, describe some findings that contradict such statements. As mentioned by Tabarani et al. (2010), this serotype has been isolated from foods of animal origin, pets, wild birds, mammals including wild possums and many reptile species.
Salmonella enterica subsp. houtenae was isolated for the first time in 1978 in an avian species (Phillips and Hatkin, 1978). Runkel et al. (1991) established that wild possums are reservoirs of this subspecies, since it corresponds to all isolates of the biliary tract of these animals. Ma et al. (2003) believe they have described the first report of human infection by S. enterica subsp. houtenae and Lourenço et al. (2004) described the first human bacteremia caused by S. enterica subsp. houtenae in Brazil associated with a patient infected by HIV. The latter authors claim that it is important to consider the fact that the patient used to live with birds and dogs, which may have served as a reservoir for Salmonella spp., and also could not discard the possibility of foodborne contamination. In a contamination assessment of chilled gooat meat sold in natura in the city of Recife, Brazil, it was observed that among seven samples contaminated by Salmonella spp., four were identified as S. enterica subsp. houtenae (Moura et al., 2007). In supermarkets and butcher shops in Porto Alegre, Brazil, from 333 pork sausage samples analyzed, the presence of Salmonella spp. was detected in 82 (24.4%). In this study, however, only one isolate did not belong to Salmonella enterica subspecies, but the Salmonella houtenae subspecies. The authors also noted that human infections associated with this subspecies are rare (Mürmann et al., 2009). This fact was also observed in a study conducted by Tavechio et al. (2002). From approximately 123 different serotypes of Salmonella spp. observed in various food sources, less than 1% corresponded to S. enterica subsp. houtenae.
Regarding Salmonella ser. Albany, literature reports that this serovar is not often correlated to infections in humans and animals. However, it has been isolated from chickens, as described in the studies by Luiz et al. (2004), Moreira et al. (2008) and Brito et al. (2010). Besides those correlations, Zaidi et al. (2006) monitored the Salmonella serotypes most commonly isolated from infected people and food supplies in Yucatan, Mexico, between 2000 and 2002. Their observations showed that Salmonella ser. Albany was the first serotype most isolated in poultry, the sixth in asymptomatic children, and the eighth in patients with diarrhea and also in beef. In pork, however, the isolation was not observed. Despite the few isolation reports of these strains in humans, molecular studies of Salmonella ser. Albany in food (Doublet et al., 2003) demonstrated the presence of genes that confer multiple resistance to antibiotics in those strains, which according to the authors, can difficult the treatment of intestinal infections caused by this microorganism.
In Salmonella spp. analysis in beef, the optimal dose of gamma irradiation to eliminate this microorganism was 3 kGy (Sedeh et al., 2007). Costa et al. (2004) observed that the dose of 2.5 kGy was sufficient for effective control of Salmonella spp., regardless of the presence of fat and oxygen. Oliveira et al. (2009) reported that the control samples of chicken meat, both in conventional and vacuum packages, were contaminated by Salmonella spp. and that the dose of 1.5 kGy was sufficient for its elimination, regardless of the atmosphere packing used. However, Santos et al. (2003) stated that the recommended dose to ensure the safety of chicken meat against the presence of Salmonella spp. is 3.8 kGy. In chilled rabbit meat samples, Salmonella spp. was detected in control and irradiated samples at 1.5 kGy, but not at 3 kGy (Badr, 2004).
Some studies, however, did not detect the presence of Salmonella spp. both in irradiated and control samples, as observed by Yildirim et al. (2005) and Parlato et al. (2007), in a typical Turkish dish (Cig Köfte) and chicken, respectively.
The results obtained in the present study, it has been found the need of improvements in the sanitary conditions during slaughter and processing of sheep meat and the effectiveness of the irradiation treatment in the elimination of coliform bacteria and Salmonella spp.
Submitted: May 09, 2011;
Approved: April 04, 2013.
All the content of the journal, except where otherwise noted, is licensed under a Creative Commons License CC BY-NC.
- Al-Bachir M, Zeinou R (2009) Effect of gamma irradiation on microbial load and quality characteristics of minced camel meat. Meat Sci 82:119-124.
- Badr HM (2004) Use of irradiation to control foodborne pathogens and extend the refrigerated market life of rabbit meat. Meat Sci 67:541-548.
- Brasil, ANVISA, Agência Nacional de Vigilância Sanitária (2001) Resolução RDC N° 12, de 02 de Janeiro de 2001. Regulamento técnico sobre padrões microbiológicos para alimentos. Available at: http://www.anvisa.gov.br/legis/resol/12_01rdc.htm Accessed 29 Oct 2010.
- Brito DAP, Alves LMC, Costa FN (2010) Detecção de Salmonella Albany, Staphylococcus coagulase positivos e micro-organismos mesófilos em carcaças de frango in natura Arq Inst Biol (Sao Paulo) 77:149-152.
- Byun MW, Lee JW, Jo C, Yook HS (2001) Quality properties of sausage made with gamma-irradiated natural pork and lamb casing. Meat Sci 59:223-228.
- Costa CS, Alfaro AT, Carro SBT, Antunez HCS, Silva WP, Soares GJD (2004) Efeito do teor de gordura, vácuo e dose de radiação gama na sobrevivência de Salmonella Typhimurium ATCC 14028 em carne bovina moída resfriada. Alim Nutr 15:139-144.
- Doublet B, Lailler R, Meunier D, Brisabois A, Boyd D, Mulvey MR, Chaslus-Dancla E, Cloeckaert A (2003) Variant Salmonella genomic island 1 antibiotic resistence gene cluster in Salmonella enterica serovar Albany. Emerg Infect Dis 9:585-591.
- Grimont PAD, Weill FX (2007) Antigenic formulae of the Salmonella serovars. 9. ed. WHO Collaborating Centre for Reference and Research on Salmonella, Paris, França.
- Jakabi M, Buzzo AA, Ristori CA, Tavechio AT, Sakuma H, Paula AMR, Gelli DS (1999) Observações laboratoriais sobre surtos alimentares de Salmonella spp., ocorridos na grande São Paulo, no período de 1994 a 1997. Rev Inst Adolfo Lutz 58:47-51.
- Jones YE, McLaren IM, Wray C (2000) Laboratory Aspects of Salmonella In: Wray C, Wray A (eds). Salmonella in Domestic Animals. CABI Publishing, Oxon, USA, 393-405.
- Kanatt SR, Shobita Rao M, Chawla SP, Sharma A (2010) Shelflife extension of convenience meat products sold in Indian supermarkets by radiation processing. Radiat Phys Chem 79:1259-1263.
- Leonel FR (2008) Irradiação e qualidade da carne de frango congelada e embalada a vácuo. D.Sc. Thesis, Faculdade de Ciências Agrárias e Veterinárias, Jaboticabal, 88 pp.
- Lourenço MCS, Reis EFM, Valls R, Asensi MD, Hofer E (2004) Salmonella enterica susp. houtenae serogroup O:16 in a HIV positive patient: case report. Rev Inst Med Trop Sao Paulo 46:169-170.
- Luiz AF, Moreira FC, Corrêa EF, Falcão DP (2004) Monitoring of the dissemination of Salmonella in the chicken Frankfurt-sausage production line of a sausage factory in the state of São Paulo, Brazil. Mem Inst Oswaldo Cruz 99:477-480.
- Ma JS, Chen PY, Lau YJ, Chi CS (2003) Brain abscess caused by Salmonella enterica subspecies houtenae in a patient with chronic granulomatous disease. J Microbiol Immunol Infect 36:282-284.
- Mantilla SPS, Santos EB, Vital EC, Mano SB, Freitas MQ, Franco RB (2010) Efeito combinado da embalagem em atmosfera modificada e radiação gama na microbiologia e na aceitação sensorial de filés de peito de frango resfriados. Biotemas 23:149-155.
- Moreira GN, Rezende CSM, Carvalho RN, Mesquita SQP, Oliveira NA, Arruda MLT (2008) Ocorrência de Salmonella sp. em carcaças de frangos abatidos e comercializados em municípios do estado de Goiás. Rev Inst Adolf Lutz 67:126-130.
- Moura APBL, Pinheiro Júnior JW, Oliveira RBA, Duarte DAM, Ribeiro AR, Reis EMF (2007) Pesquisa de coliformes termotolerantes, totais e Salmonella spp. em carnes caprinas comercializadas na cidade do Recife, Pernambuco. Arq Inst Biol (Sao Paulo) 74:293-299.
- Mürmann L, Santos MC, Cardoso M (2009) Prevalence, genetic characterization and antimicrobial resistance of Salmonella isolated from fresh pork sausages in Porto Alegre, Brazil. Food Control 20:191-195.
- Neres MA, Monteiro ALG, Garcia CA, Costa C, Arrigone MB, Rosa TJM (2001) Forma física da ração e pesos de abate nas características de carcaça de cordeiros em creep feeding Rev Bras Zootecn 30:948-954.
- Oliveira AL, Pereira MT, Bueno PHS, Oliveira RBP, Pinto FC, Correira RF, Machado MM (2009) Qualidade microbiológica da carne de frango irradiada em embalagem convencional e a vácuo. Arq Bras Med Vet Zootec 61:1210-1217.
- Parlato A, Calderaro E, Bartolotta A, D'oca MC, Giuffrida AS, Brai M, Tranchina L, Agozzino P, Avellone G, Ferrugia M, Di Noto AM Caracappa S (2007) Gas chromatographic/mass spectrometric and microbiological analysis on irradiated chicken. Radiat Phys Chem 76:1463-1465.
- Phillips WEJr, Hatkin JM (1978) Isolation of Salmonella houtenae from a cockatiel. Avian Dis 22:350-353.
- Runkel NS, Rodriguez LF, Moody FG, Larocco MT, Blasdel T (1991) Salmonella infection of the biliary and intestinal tract of wild opossums. Lab Anim Sci 41:54-56.
- Santos AF, Vizeu DM, Destro MT, Franco BDGM, Landgraf M (2003) Determinação da dose de radiação gama para reduzir a população de Salmonella spp em carne de frango. Ciênc Tecnol Aliment 23:200-205.
- Pardi MC, Santos IF, Souza ER, Pardi HS (2001) Ciência, higiene e tecnologia da carne. Editora UFG, Goiânia, Brasil.
- SAS (1999) User's guide statistics. Cary, NC: SAS Institute, Inc.
- Sedeh FM, Arbabi K, Fatolahi H, Abhari M (2007) Using gamma irradiation and low temperature on microbial decontamination of red meat in Iran. Indian J Microbiol 47:72-76.
- Silva N, Junqueira VCA, Silveira NFA (2001) Manual de Métodos de Análise Microbiológica de Alimentos. 2. ed. Varela, São Paulo, Brasil.
- Silva MP, Cavalli DR, Oliveira TCRM (2006) Avaliação do padrão coliformes a 45 °C e comparação da eficiência das técnicas dos tubos múltiplos e petrifilm EC na detecção de coliformes totais e Escherichia coli em alimentos. Ciênc Tecnol Aliment 26:352-359.
- Silva ACO (2008) Efeito da radiação gama sobre lipídios, microbiota contaminante e validade comercial do leite cru integral refrigerado e sobre características sensoriais do leite pasteurizado integral refrigerado. D.Sc. Thesis, Curso de Pós-Graduação em Ciências Veterinárias, Niterói, 89 pp.
- Tabarani CM, Bennett NJ, Kiska DL, Riddell SW, Botash AS, Domachowske JB (2010) Empyema of preexisting subdural hemorrhage caused by a rare Salmonella species after exposure to bearded dragons in a foster home. J Pediatrics 156:322-323.
- Tavechio AT, Ghilardi AC, Peresi JT, Fuzihara TO, Yonamine EK, Jakabi M, Fernandes SA (2002) Serotypes isolated from nonhuman sources in São Paulo, Brazil, from 1996 through 2000. J Food Prot 65:1041-1044.
- Yildirim I, Uzunlu S, Topuz A (2005) Effect of gamma irradiation on some principle microbiological and chemical quality parameters of raw Turkish meat ball. Food Control 16:363-367.
- Zaidi MB, Mcdermott PF, Fedorka-Cray P, Leon V, Canche C, Hubert SK, Abbott J, León M, Zhao S, Headrich M, Tollefson L (2006) Nontyphoidal Salmonella from human clinical cases, asymptomatic children, and raw retail meats in Yucatan, Mexico. Clin Infect Dis 42:21-28.
Send correspondence to
Publication Dates
-
Publication in this collection
24 Jan 2014 -
Date of issue
Dec 2013
History
-
Received
09 May 2011 -
Accepted
04 Apr 2013