ABSTRACT
Thirteen pentacyclic triterpenes, methyl 3-oxours-12-en-23-oate, marsformosanone, taraxerone, β-amyrenone, α-amyrenone, lupenone, 24-methylencycloartan-3-one, moretenol acetate, β-amyrin acetate, germanicol acetate, 24-methylencycloartanyl acetate, β-amyrin, and α-amyrin were identified in a chloroform-methanol propolis extract from Melipona beecheii. Additionally, were identified in this propolis, hexadecanoic acid, methyl ester, octadecanoic acid, methyl ester and 1-triacontanol. The purification of the propolis extract was carried out using different chromatographic techniques, including vacuum liquid chromatography, gravity column chromatography and gel filtration chromatography Sephadex LH-20. The identification of the metabolites was performed using mass spectrometry.
Keywords:
Stingless bee; Propolis; Pentacyclic triterpenes; β-Amyrin acetate; Germanicol acetate; Lupenone
Introduction
The Meliponini tribe, the members of which are also known as stingless bees, belongs to the Apidae family, and Meliponinae subfamily. These are tropical bees, among which Trigona and Melipona are the most well-known (Heard, 1999Heard, T.A., 1999. The role of stingless bees in crop pollination. Annu. Rev. Entomol. 44, 183-206.; Ferreira et al., 2017aFerreira, R.G.S., Silva-Junior, W.F., Veiga-Junior, V.F., Lima, A.A.N., Lima, E.S., 2017. Physicochemical characterization and biological activities of the triterpenic mixture α,β-amyrenone. Molecules 22, 298-306.,bFerreira, J.M., Fernandes-Silva, C.C., Salatino, A., Message, D., Negri, G., 2017. Antioxidant activity of a geopropolis from northeast Brazil: chemical characterization and likely botanical origin. Evid. Based Complement. Alternat. Med., http://dx.doi.org/10.1155/2017/4024721.
http://dx.doi.org/10.1155/2017/4024721...
). For millennia, the inhabitants of the Southern region of Mexico and neighboring countries have maintained the tradition of breeding and raising melipona bees, from which various products have been obtained, such as honey for food and medicine and beeswax used as a sealant and an art tool. (National Research and Council, 2007National Research Council, 2007. Status of Pollinators in North America. The National Academies Press, United States of America, Washington, DC, http://dx.doi.org/10.17226/11761.
http://dx.doi.org/10.17226/11761...
).
The Melipona beecheii species has been of great importance and widely cultivated since the pre-Columbian era, in particular by Mesoamerican cultures, such as the Maya. M. beecheii has been practically the only bee species domesticated by the Mayan culture due to its nest size, the excellent flavor, therapeutic properties attractive golden appearance of its honey, also the tameness of the bees (Quezada-Euán et al., 2001Quezada-Euán, J.G., May-Itzá, W., González-Acereto, J.A., 2001. Meliponiculture in Mexico: problems and perspective for development. Bee World 82, 160-167.).
Besides the honey and wax, M. beecheii produces propolis, an apiarian product with a bitter flavor and a resinous aspect, which presents a color variance from greenish-yellow to reddish-brown. Propolis consists basically of a mix of wax and resin exudates from different plants which the bee gathers to utilize as auxiliary material for hive protection (Bracho Pérez et al., 2009Bracho Pérez, J.C., Rodríguez, B., Llanes, F., 2009. Triterpenos pentacíclicos en propóleo. Rev. Soc. Quim. Perú 75, 439-452.). Propolis is a product with commercial interest due to the practice of meliponiculture and it is appreciated for its qualities in the treatment of respiratory illnesses, inflammatory diseases, fatigue, hemorrhoids and gastritis, plus, it can also be used as a base for the elaboration of remedies and food products in traditional indigenous medicine in Mexico. (Guzmán et al., 2011Guzmán, M., Balboa, C., Vandame, R., Albores, M.L., González-Acereto, J., 2011. Manejo de las abejas nativas sin aguijón en México: Melipona beecheii y Scaptotrigona mexicana. El Colegio de la Frontera Sur, San Cristóbal de Las Casas, Chiapas, México, 59 p.).
Various studies carried out on different propolis have demonstrated that its chemical composition and biological activity depend on the vegetal species, resin sources and balm that the pollinizing bees collect (Bankova, 2005Bankova, V., 2005. Chemical diversity of propolis and the problem of standardization. J. Ethnopharmacol. 100, 114-117.). With this in mind, diverse secondary metabolites have been identified, such as flavonoids, caffeic acid esters, diterpenes, benzophenones and volatile elements including sesquiterpenes (Demestre et al., 2009Demestre, M., Messerli, S.M., Celli, N., Shahhossini, M., Kluwe, L., Mautner, V., Maruta, H., 2009. CAPE (caffeic acid phenyl ester)-based propolis extract (Bio30) suppresses the growth of human neurofibromatosis (NF) tumor xenografts in mice. Phytother. Res. 23, 226-230.; Bankova et al., 2000Bankova, V.S., De Castro, S.L., Marcucci, M.C., 2000. Propolis recent advances in chemistry and plant origin. Apidologie 31, 3-15.; Tomás-Barberán et al., 1993Tomás-Barberán, F., García-Viguera, C., Vit-Oliver, P., Ferreres, F., Tomás-Lorente, F., 1993. Phytochemical evidence for the botanical origin of tropical propolis from Venezuela. Phytochemistry 34, 191-196.).
The chemical composition of M. beecheii propolis, as with all other melipone bees, is highly variable and, despite the availability of studies such as those of Pino et al. (2006)Pino, J.A., Marbot, R., Delgado, A., Zumárraga, C., Sauri, E., 2006. Volatile constituents of propolis from honey bees and stingless bees from Yucatan. J. Essent. Oil Res. 18, 53-56., Torres-González et al. (2016)Torres-González, A., López-Rivera, P., Duarte-Lisci, G., López-Ramírez, Á., Correa-Benítez, A., Rivero-Cruz, J.F., 2016. Analysis of volatile components from Melipona beecheii geopropolis from Southeast Mexico by headspace solid-phase microextraction. Nat. Prod. Res. 30, 237-240., and Fonte-Carballo et al. (2016)Fonte-Carballo, L., Milián-Rodríguez, Y.E., Díaz-Solares, M., 2016. Potencialidad antimicrobiana y caracterización de propóleos de Melipona beecheii provenientes de dos fincas agroenergéticas. Pastos Forrajes 39, 149-156. in which volatile metabolites were identified; as well as the main families of metabolites present in its methanol and ethanol extracts, current data is still scarce, especially in comparison with chemical component studies of Apis mellifera propolis.
Based on the above, and with the purpose of contributing to the available information on M. beecheii chemical composition, the main objective of this research was to identify the secondary metabolites present in the propolis of this species, starting with GC-MS and IR analysis, with which a pentacyclic triterpenes predominance was found, as well as other components including two fatty acids, aliphatic esters and a long chain alcohol.
Material and methods
General experimental procedures
Vacuum Liquid Chromatography (VLC) and column chromatography purifications were performed using E.M. Merck TLC-grade silica gel 60GF and E.M. Merck silica gel (70–230 mesh), respectively. Gel permeation column chromatography purifications were carried out using Sephadex LH-20 (Sigma, size 25–100). Analytical TLC experiments were carried out using aluminum-backed silica gel (60F254) plates (E.M. Merck, 0.2 mm thickness); the various components in the chromatograms were visualized by dipping the plates in a solution of phosphomolybdic acid (20 g) and ceric sulfate (2.5 g) in 500 ml of sulfuric acid (5%), followed by drying and gentle heating. GC-MS analyses were run on a Hewlett Packard 5890 gas chromatograph connected to a mass selective detector (MSD) (model 5975) [GC conditions: Split injection of 1 ml of sample; Ultra 1 column (25 m × 0.2 mm i.d.), flow rate 1.0 ml/min (Nitrogen); oven temperature program T 1 = 100 °C (3 min), T 2 = 280 °C (30 min), gradient 10 °C/min, injector 300° and detector (FID) 300 °C]. The components of each fraction were identified by comparing the MS spectra with those previously reported, by matching fragmentation patterns with those in the NIST05 library.
Propolis from Melipona beecheii
The propolis samples from Melipona beecheii (352.38 g) were collected in the meliponary "Flor de Mayo" in Maní, Yucatán, Mexico (20.3931° N, 89.3918° W) by the scraping technique of the upper parts of the beehive.
Extraction and isolation
The ground propolis (335.4 g) was extracted twice with a mixture of CHCl3:MeOH 1:1 at room temperature (24 h); the solution was filtered and evaporated to produce 2.12 g (0.63%) of crude extract, which was subjected to VLC purification, eluting with increasing amounts of ethyl acetate in hexane to produce seven main fractions (A–G). Fraction A was purified using a combination of Sephadex LH-20 (CHCl3:MeOH 1:1) and gravity column chromatography (hexane-acetone 9:1), with which it was possible to obtain two semi-pure fractions, I (2.4 mg) and II (6.7 mg). On the other hand, fraction B was purified using gravity column chromatography (hexane/ethyl acetate 9:1), which resulted in the isolation of the fractions III (1 mg), IV (1.9 mg) and V (9.8 mg) in a semi-pure form.
Results
The yield of crude M. beecheii propolis extract was 0.54%. Successive chromatographic purifications of the CHCl3:MeOH 1:1 crude extract of M. beecheii propolis, using a combination of VLC, gel filtration on Sephadex LH-20 and gravity column chromatography, resulted in the isolation of five semi-pure fractions, I–V. The GC-MS analysis of the five fractions allowed the detection of thirteen pentacyclic triterpenes and other constituents. The identification of the different kinds of metabolites was obtained based on the molecular weight and its fragmentation pattern in mass spectrometry.
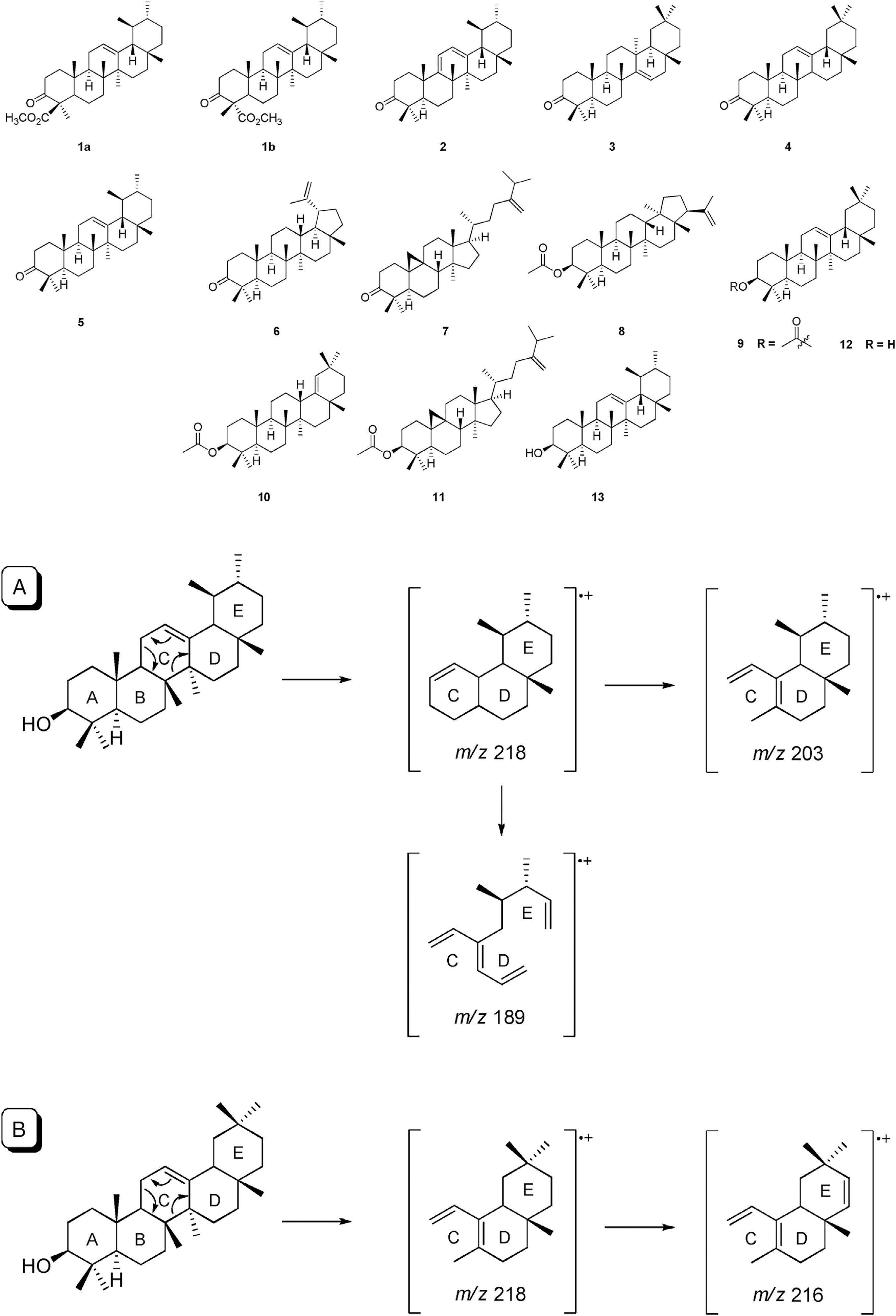
The chromatographic profile in the CG-MS analysis (Fig. 1A) of the fraction I showed the presence of one pentacyclic triterpenes identified as methyl 3-oxours-12-en-23-oate (1, t R 30.11 min). The mass spectrum of metabolite 1 showed a parent ion peak at m/z 468 suggesting the molecular formula C30H48O3. It is not possible to distinguish by mass spectrometry in the case of the isomeric forms (1a and 1b) for metabolite 1. Additionally in the fraction I were found two metabolites with a parent ion peak at m/z 270 (C17H34O2) and m/z 298 (C19H38O2), identified as hexadecanoic acid, methyl ester (a, t R 13.77 min) and octadecanoic acid, methyl ester (b, t R 15.67 min) respectively.
GC profile of the fractions I–V of propolis from Melipona beecheii. A. Fraction I; B. Fraction II; C. Fraction III; D. Fraction IV; and E. Fraction V.
On the other hand, the GC-MS analysis of the fraction II (Fig. 1B) allowed the identification of seven pentacyclic triterpenes (2–8). The signal by GC at t
R 26.56 min showed a mass spectrum, which indicated a mixture of two metabolites, observing two parent ion peak at m/z 422 and m/z 424, suggesting a molecular formula of C30H46O and C30H48O, identified as marsformosanone (2) and taraxerone (3), respectively (Paul et al., 1974Paul, B.D., Subba-Rao, G., Govind, J.K., 1974. Isolation of myricardiol, myricitrin, taraxerol, and taraxerone from Myrica cerifera L. root bark. J. Pharm. Sci. 63, 958-959.; Oladoye et al., 2015Oladoye, S.O., Ayodele, E.T., Abdul-Hammed, M., Idowu, O.T., 2015. Characterisation and identification of taraxerol and taraxer-14-en-3-one from Jatropha tanjorensis (Ellis and Saroja) leaves. Pak. J. Sci. Res. Ind. Res. 58, 46-50.). The metabolites 4 (t
R 27.03 min), 5 (t
R 24.93 min) and 6 (t
R 28.08) presented the same parent ion peak at m/z 424 (C30H48O). The metabolites 4 and 5 presented a base ion peak at m/z 218 and the fragment ions at m/z 203 and m/z 189 which allowed identified as β-amyrenone (4) and α-amyrenone (5) (Mathe et al., 2004Mathe, C., Culioli, G., Archier, P., Vieillescazes, C., 2004. Characterization of archaeological frankincense by gas chromatography-mass spectrometry. J. Chromatogr. A. 1023, 277-285.; Cely-Veloza et al., 2014Cely-Veloza, W.F., Matulevich, J.A., Castrillón, W.F., 2014. Triterpenos y esteroles de Salvia leucantha (Lamiaceae) y evaluación de su actividad antioxidante. Rev. Facultad Cien. Bas. 10, 68-79.; Ferreira et al., 2017aFerreira, R.G.S., Silva-Junior, W.F., Veiga-Junior, V.F., Lima, A.A.N., Lima, E.S., 2017. Physicochemical characterization and biological activities of the triterpenic mixture α,β-amyrenone. Molecules 22, 298-306.,bFerreira, J.M., Fernandes-Silva, C.C., Salatino, A., Message, D., Negri, G., 2017. Antioxidant activity of a geopropolis from northeast Brazil: chemical characterization and likely botanical origin. Evid. Based Complement. Alternat. Med., http://dx.doi.org/10.1155/2017/4024721.
http://dx.doi.org/10.1155/2017/4024721...
). The metabolite 6 presented a base ion peak at m/z 205 and an ion peak at 189 characteristic of a lupane triterpen skeleton, which allowed to identify it as lupenone (6) (Prashant and Krupadanam, 1993Prashant, A., Krupadanam, G.L.D., 1993. Dehydro-6-hydroxyrotenoid and lupenone from Tephrosia villosa. Phytochemistry 32, 484-486.). The metabolites 7 (t
R 29.02 min) and 8 (t
R 30.29 min) presented parent ion peak at m/z 438 (C31H50O) and m/z 468 (C32H52O2) respectively and according to their fragmentation patterns were identified as 24-methylencycloartan-3-one (7) (Alsaadi and Al-Maliki, 2015Alsaadi, J.H.H., Al-Maliki, A.D.M., 2015. Hypoglycemic effect of 24-methylencycloartan-3-one isolated from Prosopis juliflora Pods in alloxan induced diabetic rabbits. World. J. Exp. Biosci. 31, 6-13.) and moretenol acetate (8) (Abdel El-Fattah et al., 1992Abdel El-Fattah, H., Khalil, A.T., Mansour, E.S., Waight, E.S., 1992. Triterpenes and triterpene esters from Lactuta saligna. Int. J. Pharmacogn. 30, 21-26.).
The GC-MS analysis of fraction III (Fig. 1C) resulted in the identification of three pentacyclic triterpenes (9–11). The mass spectrum of 9 and 10 showed that these metabolites have parent ion peak at m/z 468 and a molecular formula C32H52O2 for both. These metabolites were identified based on their fragmentation pattern and by comparison in the literature as β-amyrin acetate (9) and germanicol acetate (10) (da Silva et al., 2018da Silva, U.P., Furlani, G.M., Demuner, J.A., da Silva, O.L.M., Varejao, E.V.V., 2018. Allelopathic activity and chemical constituents ox extracts from roots of Euphorbia heterophylla L.. Nat. Prod. Res., http://dx.doi.org/10.1080/14786419.2018.1460829.
http://dx.doi.org/10.1080/14786419.2018....
). The metabolite 11 (m/z 482, C33H54O2) was identified as 24-methylencycloartanyl acetate (De Pascual et al., 1987De Pascual, T., Urones, J.G., Marcos, I.S., Basame, P., Sexmero-Cuadrado, M.J., Fernández-Moro, R., 1987. Triterpenes from Euphorbia broteri. Phytochemistry 26, 1767-1776.). In addition, 1-triacontanol (c, t
R 28.64 min) was identified in the fraction III.
In the GC-MS analysis of fraction IV (Fig. 1D), were found the metabolites 5 and 6. Finally, in the GC-MS analysis of fraction V (Fig. 1E), a triterpenes mixture was found, whose mass spectra showed the same molecular ion value at m/z 426 for both cases, suggesting a molecular formula of C30H50O and based on its fragmentation pattern, these triterpenes were identified as β-amyrin (12) at t R 27.29 min and α-amyrin (13) at t R 28.11 min (Scheme 1) (Estrada-Vielma, 2003Estrada-Vielma, A., 2003. Determinación de los componentes mayoritarios del propóleo que se produce en la zona sur del estado de Yucatán (Tesis de Licenciatura). Facultad de Química, Universidad Autónoma de Yucatán, pp. 83–87.; Furukawa et al., 2002Furukawa, S., Takagi, N., Ikeda, T., Ono, M., Nafady, A., Nohara, T., Sugimoto, H., Doi, S., Yamada, H., 2002. Two novel long-chain alkalonic acid esters of lupeol from Alecrim-propolis. Chem. Pharm. Bull. 50, 439-440.). All identified pentacyclic triterpenes are shown in Table 1.
Retro Diels–Alder fragmentation in GC-MS analysis in ursane and oleanane-type triterpenes. A. α-amyrin and B. β-amyrin.
Discussion
Many of the metabolites identified in the propolis from M. beecheii have been reported in literature as possessing different biological activities, including antimicrobial, anti-inflammatory, cytotoxic, antioxidant, hepatoprotective and antiulcer, among others (Freire et al., 2002Freire, M.D.F., Carvalho, M.G., Berbara, R.L.L., Freire, R.B., 2002. Atividade antimicrobiana de acetato de lupeol isolado de Vernonia scorpioides (Lam.) Pers., Asteraceae. Rev. Bras. Farm. 83, 83-87.; Pinto et al., 2008Pinto, S.H., Pinto, L.M.S., Guedes, M.A., Cunha, G.M.A., Chaves, M.H., Santos, F.A., Rao, V.S., 2008. Antinoceptive effect of triterpenoid α, β-amyrin in rats on orofacial pain induced by formalin and capsaicin. Phytomedicine 15, 630-634.; Abbas et al., 2009Abbas, M., Disi, A., Al-Khalil, S., 2009. Isolation and Identification of anti-ulcer components from Anchusa strigosa root. Jordan J. Pharm. Sci. 2, 131-139.; Nikkon et al., 2010Nikkon, F., Salam, K.A., Yeasmin, T., Mosaddik, A., Khondkar, P., Haque, M.E., 2010. Mosquitocidal triterpenes from the stem of Duranta repens. Pharm. Biol. 48, 264-268.; Lucetti et al., 2010Lucetti, D.L., Lucetti, E.C., Bandeira, M.A., Veras, H.N., Silva, A.H., Leal, L.K., Lopes, A.A., Alves, C.C.V., Silva, S.G., Brito, G.A., Viana, G.B., 2010. Anti-inflammatory effects and possible mechanism of action of lupeol acetate isolated from Himatanthus drasticus (Mart.) Plumel. J. Inflamm. , 7-60.; Duan et al., 2011Duan, D.D., Bu, C.Y., Cheng, J., Wang, Y.N., Shi, G.L., 2011. Isolation and identification of acaricidal compounds in Inula japonica (Asteraceae). J. Econ. Entomol. 104, 375-378.; Yoon et al., 2015Yoon, Y.P., Lee, H.J., Lee, D.U., Lee, S.K., Hong, J.H., Lee, C.J., 2015. Effects of lupenone, lupeol, and taraxerol derived from Adenophora triphylla on the gene expression and production of airway MUC5AC mucin. Tuberc. Respir. Dis. 78, 210-217.). Among the majoritarian pentacyclic triterpenes present in the propolis, lupenone (6), α-amiryn (13) and β-amyrin (12) were identified (Fig. 1D and E). Aragao et al. (2008)Aragao, G.F., Pinheiro, M.C.C., Bandeira, P.N., Lemos, T.L.G., Viana, G.S.D.B., 2008. Analgesic and anti-inflammatory activities of the isomeric mixture of alpha-and beta-amyrin from Protium heptaphyllum (Aubl.) March. J. Herb. Pharmacother. 7, 31-47. reported that the mixture of α- and β-amyrin isomers, of ursane and oleanane skeleton, respectively, possess anti-inflammatory and analgesic activities which, together with the antiviral and anti-inflammatory properties of lupenone, could be linked with the mitigation of the symptoms of respiratory diseases experienced by people with these conditions, when consuming propolis (Yoon et al., 2015Yoon, Y.P., Lee, H.J., Lee, D.U., Lee, S.K., Hong, J.H., Lee, C.J., 2015. Effects of lupenone, lupeol, and taraxerol derived from Adenophora triphylla on the gene expression and production of airway MUC5AC mucin. Tuberc. Respir. Dis. 78, 210-217.). In addition to the reported biological activity of the α- and β-amyrin isomers, gastroprotective activity in rats, exposed to the irritant effect of capsaicin, was found to reduce the damage to the gastric mucosa to insignificant effects at concentrations of 100 mg/kg of this pair of isomers, as has been reported by Navarrete et al. (2002)Navarrete, A., Trejo-Miranda, J.L., Reyes-Trejo, L., 2002. Principles of root bark of Hippocratea excels (Hippocrataceae) with gastroprotective activity. J. Ethnopharmacol. 79, 383-388.. Other bioactive properties of these triterpenes are the hepatoprotective, antiulcer and insecticidal effects (Pinto et al., 2008Pinto, S.H., Pinto, L.M.S., Guedes, M.A., Cunha, G.M.A., Chaves, M.H., Santos, F.A., Rao, V.S., 2008. Antinoceptive effect of triterpenoid α, β-amyrin in rats on orofacial pain induced by formalin and capsaicin. Phytomedicine 15, 630-634.; Abbas et al., 2009Abbas, M., Disi, A., Al-Khalil, S., 2009. Isolation and Identification of anti-ulcer components from Anchusa strigosa root. Jordan J. Pharm. Sci. 2, 131-139.; Nikkon et al., 2010Nikkon, F., Salam, K.A., Yeasmin, T., Mosaddik, A., Khondkar, P., Haque, M.E., 2010. Mosquitocidal triterpenes from the stem of Duranta repens. Pharm. Biol. 48, 264-268.; Duan et al., 2011Duan, D.D., Bu, C.Y., Cheng, J., Wang, Y.N., Shi, G.L., 2011. Isolation and identification of acaricidal compounds in Inula japonica (Asteraceae). J. Econ. Entomol. 104, 375-378.). In addition to β-amyrin (12), its antioxidant, cytotoxic and anti-inflammatory activity has been reported (Manrique and Santana, 2008Manrique, A.J., Santana, W.C., 2008. Flavonoids, antibacterial and antioxidant activities of propolis of stingless bees, Melipona quadrifasciata, Melipona compressipes, Tetragonisca angustula, and Nannotrigona sp. from Brazil and Venezuela. Zootecnia Trop. 26, 157-166.).
Taking into account that the chemical composition of propolis varies according to the flora from which it is obtained (Manrique and Santana, 2008Manrique, A.J., Santana, W.C., 2008. Flavonoids, antibacterial and antioxidant activities of propolis of stingless bees, Melipona quadrifasciata, Melipona compressipes, Tetragonisca angustula, and Nannotrigona sp. from Brazil and Venezuela. Zootecnia Trop. 26, 157-166.), these same metabolites have been found in the propolis of other bee species in the American continent (Bracho Pérez et al., 2009Bracho Pérez, J.C., Rodríguez, B., Llanes, F., 2009. Triterpenos pentacíclicos en propóleo. Rev. Soc. Quim. Perú 75, 439-452.) as is the case of the β-amyrin acetate (9), which is known to have anti-inflammatory, antinociceptive, antioxidant and cytotoxic activity, among others, the presence of which has been reported in the propolis of bees from other parts of the American continent and now in the propolis from M. beecheii.
Among other important metabolites which were found in the propolis from M. beecheii, mention can be made of the germanicol acetate (10), and the 24-methylencicloartan-3-one (7) among those that have been reported with different biological activities, including antibacterial, anticancer and anti-inflammatory (Shimizu et al., 1996Shimizu, N., Itoh, T., Ichinohe, Y., Matsumoto, T., 1996. Cyclosadol, isocyclosadol, and other related triterpene alcohols obtained from 24-methylenecycloartanol by acid-catalyzed isomerization. Bull. Chem. Soc. Jpn. 57, 1425-1426.; Yasukawa et al., 2000Yasukawa, K.E.N., Akihisa, T., Yoshida, Z.Y., Takido, M., 2000. Inhibitory effect of euphol, a triterpene alcohol from the roots of Euphorbia kansui, on tumour promotion by 12-O-tetradecanoylphorbol-13-acetate in two-stage carcinogenesis in mouse skin. J. Pharm. Pharmacol. 52, 119-124.; Ragasa et al., 2011Ragasa, C.Y., Espineli, D.L., Shen, C.C., 2011. New triterpenes from Barringtonia asiatica. Chem. Pharm. Bull. 59, 778-782.).
In contrast, no information has been published on the biological activity of the methyl 3-oxours-12-en-23-oate (1a and 1b); however, it is known to have similarities with β-boswellic acid, the properties of which have been studied along with those of its derivatives, among which are anti-inflammatory, anti-cancer, immunomodulatory and neuroprotective activities, among others (Mehta et al., 2014Mehta, M., Satija, S., Nanda, A., Garg, M., 2014. Nanotechnologies for boswellic acids. Am. J. Drug Discov. Dev. 4, 1-11.).
Conclusion
The investigation of the chemical composition of propolis from various species of bees is of utmost importance, given that, in different parts of the world, several studies have been conducted relating to the chemical composition of the substances with the biological properties they possess, an aspect that has great value and interest for science. Motivated by this, the analysis of the medium-low polarity fraction of propolis of M. beecheii was carried out, providing its chemical composition and the assumption of some of its biological properties. A total of thirteen pentacyclic triterpenes were identified by GC-MS in a CHCl3-MeOH extract of propolis from M. beecheii, among which can be mentioned, α-amyrin, β-amyrin, lupenone, germanicol acetate and β-amyrin acetate, among others, of which a great variety of biological properties have been reported. Many of the pentacyclic triterpenes found in the propolis of M. beecheii have been reported in propolis from other areas of the world, illustrating that these components contribute to the properties of this propolis and to increasing the knowledge of the varieties of plants that are involved in the formation of the same.
Acknowledgement
Authors thank the financing from Ciencia Básica CONACyT, corresponding to the project: "Caracterización del efecto antimicrobiano (S. aureus, E. coli y C. albicans) y antioxidante de los componentes de la miel de Melipona beecheii" folio 221624.
Appendix A Supplementary data
Supplementary data associated with this article can be found, in the online version, at doi:10.1016/j.bjp.2019.01.006.
References
- Abbas, M., Disi, A., Al-Khalil, S., 2009. Isolation and Identification of anti-ulcer components from Anchusa strigosa root. Jordan J. Pharm. Sci. 2, 131-139.
- Alsaadi, J.H.H., Al-Maliki, A.D.M., 2015. Hypoglycemic effect of 24-methylencycloartan-3-one isolated from Prosopis juliflora Pods in alloxan induced diabetic rabbits. World. J. Exp. Biosci. 31, 6-13.
- Aragao, G.F., Pinheiro, M.C.C., Bandeira, P.N., Lemos, T.L.G., Viana, G.S.D.B., 2008. Analgesic and anti-inflammatory activities of the isomeric mixture of alpha-and beta-amyrin from Protium heptaphyllum (Aubl.) March. J. Herb. Pharmacother. 7, 31-47.
- Bankova, V., 2005. Chemical diversity of propolis and the problem of standardization. J. Ethnopharmacol. 100, 114-117.
- Bankova, V.S., De Castro, S.L., Marcucci, M.C., 2000. Propolis recent advances in chemistry and plant origin. Apidologie 31, 3-15.
- Bracho Pérez, J.C., Rodríguez, B., Llanes, F., 2009. Triterpenos pentacíclicos en propóleo. Rev. Soc. Quim. Perú 75, 439-452.
- Cely-Veloza, W.F., Matulevich, J.A., Castrillón, W.F., 2014. Triterpenos y esteroles de Salvia leucantha (Lamiaceae) y evaluación de su actividad antioxidante. Rev. Facultad Cien. Bas. 10, 68-79.
- De Pascual, T., Urones, J.G., Marcos, I.S., Basame, P., Sexmero-Cuadrado, M.J., Fernández-Moro, R., 1987. Triterpenes from Euphorbia broteri Phytochemistry 26, 1767-1776.
- Demestre, M., Messerli, S.M., Celli, N., Shahhossini, M., Kluwe, L., Mautner, V., Maruta, H., 2009. CAPE (caffeic acid phenyl ester)-based propolis extract (Bio30) suppresses the growth of human neurofibromatosis (NF) tumor xenografts in mice. Phytother. Res. 23, 226-230.
- Duan, D.D., Bu, C.Y., Cheng, J., Wang, Y.N., Shi, G.L., 2011. Isolation and identification of acaricidal compounds in Inula japonica (Asteraceae). J. Econ. Entomol. 104, 375-378.
- Abdel El-Fattah, H., Khalil, A.T., Mansour, E.S., Waight, E.S., 1992. Triterpenes and triterpene esters from Lactuta saligna Int. J. Pharmacogn. 30, 21-26.
- Estrada-Vielma, A., 2003. Determinación de los componentes mayoritarios del propóleo que se produce en la zona sur del estado de Yucatán (Tesis de Licenciatura). Facultad de Química, Universidad Autónoma de Yucatán, pp. 83–87.
- Ferreira, R.G.S., Silva-Junior, W.F., Veiga-Junior, V.F., Lima, A.A.N., Lima, E.S., 2017. Physicochemical characterization and biological activities of the triterpenic mixture α,β-amyrenone. Molecules 22, 298-306.
- Ferreira, J.M., Fernandes-Silva, C.C., Salatino, A., Message, D., Negri, G., 2017. Antioxidant activity of a geopropolis from northeast Brazil: chemical characterization and likely botanical origin. Evid. Based Complement. Alternat. Med., http://dx.doi.org/10.1155/2017/4024721
» http://dx.doi.org/10.1155/2017/4024721 - Fonte-Carballo, L., Milián-Rodríguez, Y.E., Díaz-Solares, M., 2016. Potencialidad antimicrobiana y caracterización de propóleos de Melipona beecheii provenientes de dos fincas agroenergéticas. Pastos Forrajes 39, 149-156.
- Freire, M.D.F., Carvalho, M.G., Berbara, R.L.L., Freire, R.B., 2002. Atividade antimicrobiana de acetato de lupeol isolado de Vernonia scorpioides (Lam.) Pers., Asteraceae. Rev. Bras. Farm. 83, 83-87.
- Furukawa, S., Takagi, N., Ikeda, T., Ono, M., Nafady, A., Nohara, T., Sugimoto, H., Doi, S., Yamada, H., 2002. Two novel long-chain alkalonic acid esters of lupeol from Alecrim-propolis. Chem. Pharm. Bull. 50, 439-440.
- Guzmán, M., Balboa, C., Vandame, R., Albores, M.L., González-Acereto, J., 2011. Manejo de las abejas nativas sin aguijón en México: Melipona beecheii y Scaptotrigona mexicana El Colegio de la Frontera Sur, San Cristóbal de Las Casas, Chiapas, México, 59 p.
- Heard, T.A., 1999. The role of stingless bees in crop pollination. Annu. Rev. Entomol. 44, 183-206.
- Lucetti, D.L., Lucetti, E.C., Bandeira, M.A., Veras, H.N., Silva, A.H., Leal, L.K., Lopes, A.A., Alves, C.C.V., Silva, S.G., Brito, G.A., Viana, G.B., 2010. Anti-inflammatory effects and possible mechanism of action of lupeol acetate isolated from Himatanthus drasticus (Mart.) Plumel. J. Inflamm. , 7-60.
- Manrique, A.J., Santana, W.C., 2008. Flavonoids, antibacterial and antioxidant activities of propolis of stingless bees, Melipona quadrifasciata, Melipona compressipes, Tetragonisca angustula, and Nannotrigona sp. from Brazil and Venezuela. Zootecnia Trop. 26, 157-166.
- Mathe, C., Culioli, G., Archier, P., Vieillescazes, C., 2004. Characterization of archaeological frankincense by gas chromatography-mass spectrometry. J. Chromatogr. A. 1023, 277-285.
- Mehta, M., Satija, S., Nanda, A., Garg, M., 2014. Nanotechnologies for boswellic acids. Am. J. Drug Discov. Dev. 4, 1-11.
- National Research Council, 2007. Status of Pollinators in North America. The National Academies Press, United States of America, Washington, DC, http://dx.doi.org/10.17226/11761
» http://dx.doi.org/10.17226/11761 - Navarrete, A., Trejo-Miranda, J.L., Reyes-Trejo, L., 2002. Principles of root bark of Hippocratea excels (Hippocrataceae) with gastroprotective activity. J. Ethnopharmacol. 79, 383-388.
- Nikkon, F., Salam, K.A., Yeasmin, T., Mosaddik, A., Khondkar, P., Haque, M.E., 2010. Mosquitocidal triterpenes from the stem of Duranta repens Pharm. Biol. 48, 264-268.
- Oladoye, S.O., Ayodele, E.T., Abdul-Hammed, M., Idowu, O.T., 2015. Characterisation and identification of taraxerol and taraxer-14-en-3-one from Jatropha tanjorensis (Ellis and Saroja) leaves. Pak. J. Sci. Res. Ind. Res. 58, 46-50.
- Paul, B.D., Subba-Rao, G., Govind, J.K., 1974. Isolation of myricardiol, myricitrin, taraxerol, and taraxerone from Myrica cerifera L. root bark. J. Pharm. Sci. 63, 958-959.
- da Silva, U.P., Furlani, G.M., Demuner, J.A., da Silva, O.L.M., Varejao, E.V.V., 2018. Allelopathic activity and chemical constituents ox extracts from roots of Euphorbia heterophylla L.. Nat. Prod. Res., http://dx.doi.org/10.1080/14786419.2018.1460829
» http://dx.doi.org/10.1080/14786419.2018.1460829 - Pino, J.A., Marbot, R., Delgado, A., Zumárraga, C., Sauri, E., 2006. Volatile constituents of propolis from honey bees and stingless bees from Yucatan. J. Essent. Oil Res. 18, 53-56.
- Pinto, S.H., Pinto, L.M.S., Guedes, M.A., Cunha, G.M.A., Chaves, M.H., Santos, F.A., Rao, V.S., 2008. Antinoceptive effect of triterpenoid α, β-amyrin in rats on orofacial pain induced by formalin and capsaicin. Phytomedicine 15, 630-634.
- Prashant, A., Krupadanam, G.L.D., 1993. Dehydro-6-hydroxyrotenoid and lupenone from Tephrosia villosa Phytochemistry 32, 484-486.
- Quezada-Euán, J.G., May-Itzá, W., González-Acereto, J.A., 2001. Meliponiculture in Mexico: problems and perspective for development. Bee World 82, 160-167.
- Ragasa, C.Y., Espineli, D.L., Shen, C.C., 2011. New triterpenes from Barringtonia asiatica Chem. Pharm. Bull. 59, 778-782.
- Shimizu, N., Itoh, T., Ichinohe, Y., Matsumoto, T., 1996. Cyclosadol, isocyclosadol, and other related triterpene alcohols obtained from 24-methylenecycloartanol by acid-catalyzed isomerization. Bull. Chem. Soc. Jpn. 57, 1425-1426.
- Tomás-Barberán, F., García-Viguera, C., Vit-Oliver, P., Ferreres, F., Tomás-Lorente, F., 1993. Phytochemical evidence for the botanical origin of tropical propolis from Venezuela. Phytochemistry 34, 191-196.
- Torres-González, A., López-Rivera, P., Duarte-Lisci, G., López-Ramírez, Á., Correa-Benítez, A., Rivero-Cruz, J.F., 2016. Analysis of volatile components from Melipona beecheii geopropolis from Southeast Mexico by headspace solid-phase microextraction. Nat. Prod. Res. 30, 237-240.
- Yasukawa, K.E.N., Akihisa, T., Yoshida, Z.Y., Takido, M., 2000. Inhibitory effect of euphol, a triterpene alcohol from the roots of Euphorbia kansui, on tumour promotion by 12-O-tetradecanoylphorbol-13-acetate in two-stage carcinogenesis in mouse skin. J. Pharm. Pharmacol. 52, 119-124.
- Yoon, Y.P., Lee, H.J., Lee, D.U., Lee, S.K., Hong, J.H., Lee, C.J., 2015. Effects of lupenone, lupeol, and taraxerol derived from Adenophora triphylla on the gene expression and production of airway MUC5AC mucin. Tuberc. Respir. Dis. 78, 210-217.
Publication Dates
-
Publication in this collection
26 Aug 2019 -
Date of issue
Mar-Apr 2019
History
-
Received
30 Sept 2018 -
Accepted
24 Jan 2019 -
Published
22 Mar 2019