Abstracts
Solitary fibrous tumor (SFT) is an uncommon spindle-cell neoplasm that most often involves the pleura, rarely occurring in extra-thoracic locations. Twenty-six cases of SFT arising in the thyroid gland have been described. We report a case of a 60-year-old woman presenting an 8-month history of enlargement of the neck associated with dysphagia. The patient underwent a right hemithyroidectomy and SFT of the thyroid was diagnosed. Immunohistochemistry showed positivity for CD34 marker, and the high number of mitoses and the presence of cellular atypia suggested that the tumor was malignant. To our knowledge, this is the second case of malignant SFT of the thyroid gland ever reported. Due to the rarity of these tumors, the indication of adjuvant therapy and prognosis are uncertain. Long-term follow-up after surgical resection seems to be advisable.
O tumor fibroso solitário (SFT) é uma neoplasia rara de células fusiformes que mais frequentemente envolve a pleura, raramente ocorrendo em áreas extratorácicas. Já foram descritos 26 casos de SFT da tiroide. Relatamos o caso de uma paciente de 60 anos de idade com um histórico de 8 meses de aumento do pescoço associado à disfagia. A paciente foi submetida a uma hemitiroidectomia direita e foi diagnosticado um SFT de tiroide. A imuno-histoquímica mostrou resultados positivos para o marcador CD34, e o grande número de mitoses e a presença de atipia celular sugerem que o tumor era maligno. Em nosso conhecimento, este é o segundo caso de STF da tiroide maligno já relatado. Dada a rara ocorrência desses tumores, a indicação de tratamento adjuvante e o prognóstico são incertos. Recomenda-se o acompanhamento de longo prazo depois da ressecção cirúrgica.
INTRODUCTION
Solitary fibrous tumor (SFT) is a rare spindle-cell neoplasm, originally described by Klemperer and Rabin in 1931 as a pleural tumor (11 Klemperer P, Rabin CB. Primary neoplasm of the pleura: a report of five cases. Arch Pathol. 1931;11:385-412.). Albeit it has been originally recognized as a localized form of mesothelioma (22 Larsen SR, Godballe C, Krogdahl A. Solitary fibrous tumor arising in an intrathoracic goiter. Thyroid. 2010;20(4):435-7.), some studies have reported extrapleural sites in the body (33 Chan JKC. Solitary fibrous tumor – everywhere, and a diagnosis in vogue. Histopathology. 1997;31:568-76.,44 Kie JH, Kim JY, Park YN, Lee MK, Yang WI, Park JS. Solitary fibrous tumor of the thyroid. Histopathology. 1997;30:365-8.), such as thyroid, salivary glands, tongue, respiratory tract, orbit, liver, meninges and retroperitoneum (33 Chan JKC. Solitary fibrous tumor – everywhere, and a diagnosis in vogue. Histopathology. 1997;31:568-76.,55 Parwani AV, Galindo R, Steinberg DM, Zeiger MA, Westra WH, Ali SZ. Solitary fibrous tumor of the thyroid: cytopathologic findings and differential diagnosis. Diagn Cytopathol. 2003;28:213-6.). They are very rare in the thyroid gland, with only 26 cases described after the original report in 1993 (66 Verdi D, Pennelli G, Pelizzo MR, Toniato A. Solitary fibrous tumor of the thyroid gland: a report of two cases with an analysis of their clinical and pathological features. Endocr Pathol. 2011;22:165-9.,77 Taccagni G, Sambade C, Nesland J, Terreni MR, Sobrinho-Simoes M. Solitary fibrous tumour of the thyroid: clinicopathological, immunohistochemical and ultrastructural study of three cases. Virchows Arch A Pathol Anat Histopathol. 1993;422:491-7.). Among those, only one met the criteria for malignant SFT (88 Ning S, Song X, Xiang L, Chen Y, Cheng Y, Chen H. Malignant solitary fibrous tumor of the thyroid gland. Diagn Cytopathol. 2010;39:694-9.), proposed by Vallat-Decouvelaere and cols. in 1998 (99 Vallat-Decouvelaere AV, Dry SM, Fletcher CDM. Atypical and malignant solitary fibrous tumors in extrathoracic locations: evidence of their comparability to intra-thoracic tumors. Am J Surg Pathol. 1998;22:1501-11.). We present what appears to be the second case described of a malignant solitary fibrous tumor of the thyroid.
CASE REPORT
A 60-year-old woman presented an 8-month history of enlargement of the neck associated with dysphagia to solids. She also complained of voice hoarseness and mild respiratory symptoms, mostly at night. The patient was referred to the Department of Head and Neck Surgery of the Hospital das Clínicas of the University of Sao Paulo, School of Medicine, in November 2012. At the physical examination, an enlargement of the right side of the neck was observed, with a homogeneous and tender consistency at palpation. The right vocal fold had reduced mobility, with an underlying submucosal mass. Thyroid function tests were normal, and the ultrasonography (US) demonstrated a right-side thyroid mass, with an intrathoracic component and reduction of the lumen of the trachea. The volume of the gland was 283.6 cc, and the left thyroid lobe appeared normal. Computed tomography (CT) findings were in accordance with the US data (Figure 1). An US-guided fine-needle aspiration biopsy (FNAB) was performed, with a benign cytopathologic result.
Tumor section surface in the thyroid gland. The tumor is irregular, with multinodular areas and white-tan color. The section surface is firm and curled.
The patient underwent right hemithyroidectomy in January 2013, with a frozen-section analysis suspicious for thyroid lymphoma or chronic thyroiditis. The right recurrent laryngeal nerve (RLN), as well as both right-side parathyroid glands were preserved. Right after extubation in the operative room, the patient developed severe dyspnea refractory to non-invasive positive pressure ventilation. A tracheostomy was then performed.
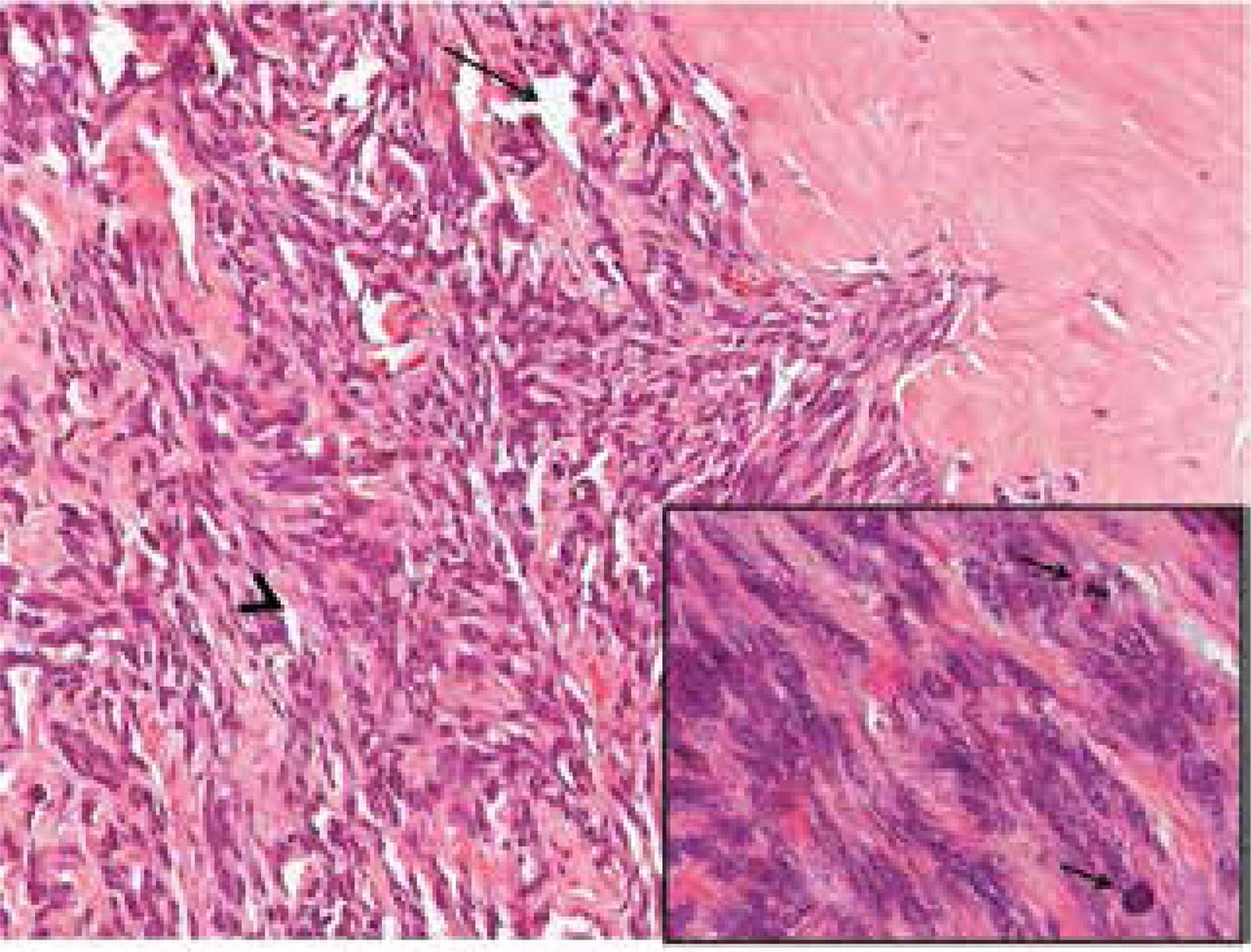
Histopathological findings included SFT with 13.8 x 8.5 cm, without necrotic foci and with 06/10 mitoses per high-power fields (HPF). Immunohistochemistry staining showed that tumor cells strongly expressed CD34 marker, with negative expression of desmin, S100 protein, cytokeratin 35BH11, beta-catenina, and calcitonin (Figures 2 and 3).
Histological architecture with “patternless” growth pattern. Spindle cells are separated by thick hyalinized collagen with cracking artifact (arrowhead) and hemangiopericytoma-like vessels (arrow). Note the infiltrative tumor edge (hematoxilin and eosin, 200X). Inset: spindle cells with moderate atypia and mitotic figures (arrows) (hematoxilin and eosin, 1000X).
The patient was discharged from the hospital on the fifth postoperative day, and the tracheostomy tube was removed three weeks after the operation. She persisted with right vocal fold paralysis, with normal speech and respiration.
DISCUSSION
SFTs are rare ubiquitous neoplasms of mesenchymal origin. The etiology is unknown (1010 Papi G, Corrado S, Uberti ED, Roti E. Solitary fibrous tumor of the thyroid gland. Thyroid. 2007;17:119-26.). It was described as a pleural-based lesion (11 Klemperer P, Rabin CB. Primary neoplasm of the pleura: a report of five cases. Arch Pathol. 1931;11:385-412.), but there were subsequent reports on other anatomic sites as well (33 Chan JKC. Solitary fibrous tumor – everywhere, and a diagnosis in vogue. Histopathology. 1997;31:568-76.).
Less than 0.1% of all tumors appear on the head and neck area (22 Larsen SR, Godballe C, Krogdahl A. Solitary fibrous tumor arising in an intrathoracic goiter. Thyroid. 2010;20(4):435-7.). The first report of a SFT of the thyroid (SFT-T) was published in 1993 by Taccagni and cols. (77 Taccagni G, Sambade C, Nesland J, Terreni MR, Sobrinho-Simoes M. Solitary fibrous tumour of the thyroid: clinicopathological, immunohistochemical and ultrastructural study of three cases. Virchows Arch A Pathol Anat Histopathol. 1993;422:491-7.). Since then, to our knowledge, only 26 cases were described in the English-language literature (66 Verdi D, Pennelli G, Pelizzo MR, Toniato A. Solitary fibrous tumor of the thyroid gland: a report of two cases with an analysis of their clinical and pathological features. Endocr Pathol. 2011;22:165-9.). A review of these articles is summarized in table 1.
According to cumulative data, the mean age for appearance of SFT-T is 50 years (ranging from 28 to 68 years), with no difference in gender. The size of the tumor can range from 2 to 10 cm, and there is equal incidence for both thyroid lobes (1111 Santeusanio G, Schiaroli S, Ortenzi A, Mulè A, Perone G, Fadda G. Solitary fibrous tumor of thyroid: report of two cases with immunohistochemical features and literature review. Head Neck Pathol. 2008;2:231-5.). The clinical presentation is similar to regular thyroid neoplasms (1010 Papi G, Corrado S, Uberti ED, Roti E. Solitary fibrous tumor of the thyroid gland. Thyroid. 2007;17:119-26.). The patient usually refers to a slowly growing painless mass, but, occasionally, there may be a rapid increase in volume (77 Taccagni G, Sambade C, Nesland J, Terreni MR, Sobrinho-Simoes M. Solitary fibrous tumour of the thyroid: clinicopathological, immunohistochemical and ultrastructural study of three cases. Virchows Arch A Pathol Anat Histopathol. 1993;422:491-7.,88 Ning S, Song X, Xiang L, Chen Y, Cheng Y, Chen H. Malignant solitary fibrous tumor of the thyroid gland. Diagn Cytopathol. 2010;39:694-9.). The present case, a 60-year-old female patient with a 13.8 x 8.5 cm tumor arising from the right thyroid lobe, had the largest SFT-T ever described.
SFT-T can only be diagnosed after exclusion of other thyroid tumors that exhibit spindle-cell morphology (1212 Babouk NL. Solitary fibrous tumor of the thyroid gland. Saudi Med J. 2004;25:805-7.). Among the possible differential diagnosis are hemangiopericytomas, Riedel’s thyroiditis, medullary carcinomas, anaplastic carcinomas, sarcomas, leiomyomas, neurofibromas, schwanomas, and lymphomas (1010 Papi G, Corrado S, Uberti ED, Roti E. Solitary fibrous tumor of the thyroid gland. Thyroid. 2007;17:119-26.).
Although FNAB is a standard procedure in the evaluation of thyroid nodules, its efficacy in the diagnosis of SFT-T is limited (1313 Farrag TY, Micchelli S, Tufano RP. Solitary fibrous tumor of the thyroid gland. Laryngoscope. 2009;119:2306-8.). Only two reports showed a diagnosis of spindle-cell neoplasm using cytology combined with immunocytochemistry, but a definitive confirmation of SFT-T was not possible using this method alone (55 Parwani AV, Galindo R, Steinberg DM, Zeiger MA, Westra WH, Ali SZ. Solitary fibrous tumor of the thyroid: cytopathologic findings and differential diagnosis. Diagn Cytopathol. 2003;28:213-6.,1313 Farrag TY, Micchelli S, Tufano RP. Solitary fibrous tumor of the thyroid gland. Laryngoscope. 2009;119:2306-8.). The FNAB of the present patient showed a benign lesion, most likely granulomatous thyroiditis. Immunocytochemistry was not performed in this case, since cytology demonstrated a benign lesion. Because of FNAB limitations, proper diagnosis can only be ensured by combination of histopathological and immunohistochemistry evaluation.
Microscopically, SFT-T exhibits a unique morphology, often referred as a “patternless” pattern, with spindle cell proliferation, intermingled with hypercellular and hypocellular areas (1414 Deshmukh NS, Mangham DC, Warfield AT, Watkinson JC. Solitary fibrous tumor of the thyroid gland. J Laryngol Otol. 2001;115:940-2.). At immunohistochemistry, the tumor cells revealed positivity for CD34, bcl-2, CD99, and vimentin, but not for epithelial markers (such as keratins, thyroglobulin), desmin, and S100 protein (1010 Papi G, Corrado S, Uberti ED, Roti E. Solitary fibrous tumor of the thyroid gland. Thyroid. 2007;17:119-26.,1111 Santeusanio G, Schiaroli S, Ortenzi A, Mulè A, Perone G, Fadda G. Solitary fibrous tumor of thyroid: report of two cases with immunohistochemical features and literature review. Head Neck Pathol. 2008;2:231-5.).
Extra-pleural SFT-Ts are mostly benign neoplasms, and there are no distinct pathologic features to help differentiating benign from malignant lesions (33 Chan JKC. Solitary fibrous tumor – everywhere, and a diagnosis in vogue. Histopathology. 1997;31:568-76.,1515 Song Z, Yu C, Song X, Wei L, Liu A. Primary solitary fibrous tumor of the thyroid – report of a case and a review of the literature. J Cancer. 2011;2:206-9.). Some characteristics suggesting a more aggressive behavior were proposed in 1998 by Vallat-Decouvelaere and cols., and included (i) high cellularity, (ii) cytological atypia, (iii) higher frequency of mitoses (> 4/10 HPF), (iv) evidence of tumor necrosis or infiltrating margins (99 Vallat-Decouvelaere AV, Dry SM, Fletcher CDM. Atypical and malignant solitary fibrous tumors in extrathoracic locations: evidence of their comparability to intra-thoracic tumors. Am J Surg Pathol. 1998;22:1501-11.). So far, there is only one report of malignant SFT-T, which already presented pulmonary metastasis when diagnosed (88 Ning S, Song X, Xiang L, Chen Y, Cheng Y, Chen H. Malignant solitary fibrous tumor of the thyroid gland. Diagn Cytopathol. 2010;39:694-9.). In the present case, even without distant metastasis, the tumor presented two of the above-cited criteria for malignancy (cytological atypia and > 4/10 mitoses per HPF). With these findings, we believe that our case is the second report of malignant SFT-T.
The management of SFT-T is primarily surgical, which also provides a definitive diagnosis (1010 Papi G, Corrado S, Uberti ED, Roti E. Solitary fibrous tumor of the thyroid gland. Thyroid. 2007;17:119-26.,1313 Farrag TY, Micchelli S, Tufano RP. Solitary fibrous tumor of the thyroid gland. Laryngoscope. 2009;119:2306-8.). If there is no suspicion of malignancy, such as invasion of surrounding structures, a thyroid lobectomy may be sufficient. In the present case, we did not find any intraoperative sings of malignancy. Due to the preoperative right vocal fold paresis, combined with the intraoperative finding of a normal left lobe, we chose to perform a right hemithyroidectomy. In such cases of preoperative vocal fold impairment, it is recommended that the surgical procedure starts by the exploration of the side of the neck that is ipsilateral to the paresis, as long as this does not affect the oncologic aspects of the operation (i.e., removal of the lobe containing the lesion). Even with an intact left RLN, the patient developed important dyspnea in the recovery room, and a tracheostomy was then performed. The patient received steroids in the postoperative period, and the tracheostomy tube was uneventfully withdrawn after 3 weeks.
TSH suppression therapy with hormones has no rationale in the management of SFT-T, as the origin of tumor cells is stromal (88 Ning S, Song X, Xiang L, Chen Y, Cheng Y, Chen H. Malignant solitary fibrous tumor of the thyroid gland. Diagn Cytopathol. 2010;39:694-9.). The indication of chemotherapy or external radiation therapy is controversial, even in malignant tumors (88 Ning S, Song X, Xiang L, Chen Y, Cheng Y, Chen H. Malignant solitary fibrous tumor of the thyroid gland. Diagn Cytopathol. 2010;39:694-9.,1010 Papi G, Corrado S, Uberti ED, Roti E. Solitary fibrous tumor of the thyroid gland. Thyroid. 2007;17:119-26.).
Because of the rarity of these tumors, especially in cases of malignancy, not much is known about its behavior or prognosis. Close follow-up after surgical resection of lesions seems to be the best management.
In conclusion, this is the 27th reported case of SFT-T in the English-language literature, being the second malignant tumor reported, and the larger lesion described so far.
REFERENCES
-
1Klemperer P, Rabin CB. Primary neoplasm of the pleura: a report of five cases. Arch Pathol. 1931;11:385-412.
-
2Larsen SR, Godballe C, Krogdahl A. Solitary fibrous tumor arising in an intrathoracic goiter. Thyroid. 2010;20(4):435-7.
-
3Chan JKC. Solitary fibrous tumor – everywhere, and a diagnosis in vogue. Histopathology. 1997;31:568-76.
-
4Kie JH, Kim JY, Park YN, Lee MK, Yang WI, Park JS. Solitary fibrous tumor of the thyroid. Histopathology. 1997;30:365-8.
-
5Parwani AV, Galindo R, Steinberg DM, Zeiger MA, Westra WH, Ali SZ. Solitary fibrous tumor of the thyroid: cytopathologic findings and differential diagnosis. Diagn Cytopathol. 2003;28:213-6.
-
6Verdi D, Pennelli G, Pelizzo MR, Toniato A. Solitary fibrous tumor of the thyroid gland: a report of two cases with an analysis of their clinical and pathological features. Endocr Pathol. 2011;22:165-9.
-
7Taccagni G, Sambade C, Nesland J, Terreni MR, Sobrinho-Simoes M. Solitary fibrous tumour of the thyroid: clinicopathological, immunohistochemical and ultrastructural study of three cases. Virchows Arch A Pathol Anat Histopathol. 1993;422:491-7.
-
8Ning S, Song X, Xiang L, Chen Y, Cheng Y, Chen H. Malignant solitary fibrous tumor of the thyroid gland. Diagn Cytopathol. 2010;39:694-9.
-
9Vallat-Decouvelaere AV, Dry SM, Fletcher CDM. Atypical and malignant solitary fibrous tumors in extrathoracic locations: evidence of their comparability to intra-thoracic tumors. Am J Surg Pathol. 1998;22:1501-11.
-
10Papi G, Corrado S, Uberti ED, Roti E. Solitary fibrous tumor of the thyroid gland. Thyroid. 2007;17:119-26.
-
11Santeusanio G, Schiaroli S, Ortenzi A, Mulè A, Perone G, Fadda G. Solitary fibrous tumor of thyroid: report of two cases with immunohistochemical features and literature review. Head Neck Pathol. 2008;2:231-5.
-
12Babouk NL. Solitary fibrous tumor of the thyroid gland. Saudi Med J. 2004;25:805-7.
-
13Farrag TY, Micchelli S, Tufano RP. Solitary fibrous tumor of the thyroid gland. Laryngoscope. 2009;119:2306-8.
-
14Deshmukh NS, Mangham DC, Warfield AT, Watkinson JC. Solitary fibrous tumor of the thyroid gland. J Laryngol Otol. 2001;115:940-2.
-
15Song Z, Yu C, Song X, Wei L, Liu A. Primary solitary fibrous tumor of the thyroid – report of a case and a review of the literature. J Cancer. 2011;2:206-9.
-
16Papi G, Corrado S, Ruggiero C, LiVolsi VA. Solitary fibrous tumor of the thyroid gland associated with papillary thyroid carcinoma. Thyroid. 2006;16:319-20.
-
17Bohorquez CL, Gonzalez-Campora R, Loscertales MC, Escudero AG, Mezquita JC. Solitary fibrous tumor of the thyroid with capsular invasion. Pathol Res Pract. 2003;199:687-90.
-
18Rodriguez I, Ayala E, Caballero C, De Miguel C, Matias-Guiu X, Cubilla AL, et al. Solitary fibrous tumor of the thyroid gland. Am J Surg Pathol. 2001;25:1424-8.
-
19Brunnemann RB, Ro JY, Ordonez NG, Mooney J, El-Naggar AK, Ayala AG. Extrapleural solitary fibrous tumor: a clinicopathologic study of 24 cases. Mod Pathol. 1999;12:1034-42.
-
20Cameselle-Teijeiro J, Varela-Duran J, Fonseca E, Villanueva JP, Sobrinho-Simoes M. Solitary fibrous tumor of the thyroid. Am J Clin Pathol. 1994;101:535-8.
Publication Dates
-
Publication in this collection
June 2014
History
-
Received
15 Jan 2014 -
Accepted
27 Feb 2014