Abstracts
Inula aucheriana DC is a member of the family Asteraceae which is known to produce cytotoxic secondary metabolites noted as sesquiterpene lactones. In the present study, sesquiterpene lactones inuchinenolide B, 6-deoxychamissonolide (stevin) and 14-acetoxy-1β,5α,7αH-4β-hydroxy-guai-9(10),11(13)-dien-12,8α-olide were isolated from I. aucheriana. Inuchinenolide B and 14-acetoxy-1β,5α,7αH-4β-hydroxy-guai-9(10),11(13)-dien-12,8α-olide were further evaluated by the MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide) assay to demonstrate cytotoxic activity with IC50 values of (56.6, 19.0), (39.0, 11.8), and (55.7, 15.3) μg/mL against HepG-2, MCF-7 and A-549 cells, respectively. The cytotoxic activity of the two evaluated sesquiterpene lactones partly explains the cytotoxic activity that was previously observed for the extracts of Inula aucheriana. The isolated compounds could be further investigated in cancer research studies.
cytotoxicity; Inula aucheriana; MTT assay; sesquiterpene lactones
Inula aucheriana DC, uma espécie da família Asteraceae, é conhecida pela produção de lactonas sesquiterpênicas que são metabólitos secundários cititóxicos. No presente estudo, as seguintes lactonas sesquiterpênicas foram isoladas de I. aucheriana: inuchinenolida B, 6-desoxichamissonolida (estevina) e 14-acetoxi-1β,5α,7αH-4β-hidroxi-guai-9(10, 11(13)-dien-12,8α-olida. A atividade citotóxica da inuchinenolida B e da 14-acetoxi-1β,5α,7αH-4β-hidroxi-guai-9(10),11(13)-dien-12,8α-olida foi avaliada pelo ensaio do MTT (brometo de 3-(4,5 dimetiltiazol-2il)-2,5- difeniltetrazólio) em culturas de células HepG-2, MCF-7 e A-549 tendo sido determinados valores de IC50 de (56.6, 19.0 μg/mL), (39.0, 11.8 μg/mL), e (55.7, 15.3 μg/mL), respectivamente. A atividade citotóxica das duas lactonas sesquiterpênicas avaliadas explica a atividade citotóxica previamente observada para os extratos de Inula aucheriana. As substâncias isoladas merecem ser investigadas em estudos direcionados para pesquisas em câncer.
citotoxicidade; Inula aucheriana; ensaio do MTT; lactonas sesquiterpênicas
INTRODUCTION
Inula aucheriana DC. a perennial species of Asteraceae grows in the West Azerbaijan province, Iran. It is a member of the Inula genus which has about 100 species of mainly perennial and subshrubs, distributed across warm and temperate parts of Europe, Asia and Africa (Bown 2002BOWN D. 2002. Encyclopedia of Herbs and Their Uses. London: Dorling Kindersley.). Some species of this genus have been used in traditional medicine by Greek, Roman and Chinese healers for their expectorants, antitussives, diaphoretics, antiemetics, and antibacterial properties (Seca et al. 2014SECA AM, GRIGORE A, PINTO DC and SILVA AM . 2014. The genus Inula and their metabolites: from ethnopharmacological to medicinal uses. J Ethnopharmacol 154(2): 286-310. ). Com pounds like monoterpenes, sesquiterpenes, diterpenes, flavonoids, and glycolipids have been isolated from Inula while several biological effects like antiproliferative, antibacterial and hepatoprotective activity have been reported from this genus (Zhao et al. 2006ZHAO YM, ZHANG ML, SHI QW and KIYOTA H. 2006. Chemical constituents of plants from the genus Inula. Chem Biodivers 3(4): 371-384.). Some phytochemical studies about different species of Inula have introduced several cytotoxic and apoptotic compounds, most of these isolated materials are sesquiterpene lactones of different structure types. There has been limited research about the cytotoxicity of Inula aucheriana; however, our previous study demonstrated that the extracts from the aerial parts of this species exhibit cytotoxicity to several cancerous cell lines (Hamzeloo-Moghadam et al. 2012HAMZELOO-MOGHADAM M, HAJIMEHDIPOOR H, SAIEDNIA S, ATOOFI A, SHAHRESTANI R, READ RW and MOSADDEGH M. 2012. Anti-proliferative Activity and Apoptotic Potential of Britannin, a Sesquiterpene Lactone from Inula aucheriana. Nat Prod Commun 7(8): 979-980.); thus the present study was focused on the isolation of compounds from the aerial parts of this Asteraceae species as well as on the evaluation of their cytotoxic activity to find the compound/compounds that are at least partially responsible for the previously observed cytotoxic results.
EXPERIMENTAL
GENERAL EXPERIMENTAL PROCEDURES
IR (Infrared) spectra were recorded on a Nicolet Maga 550, and NMR spectra on a Bruker instrument, operating at 500 MHz for 1H and 125 MHz for 13C. TMS (Tetramethylsilane) was used as internal standard. HRMS (High Resolution Mass Spectroscopy) were obtained with an Orbitrap LTQ XL (Thermo Fisher Scientific, San Jose Ca, USA) ion trap mass spectrometer using a nanospray (nanoelectrospray) ionization source to generate ions from methanol solutions.
CHEMICALS AND REAGENTS
RPMI 1640 medium, penicillin-streptomycin, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) from Sigma, DMEM medium and FBS (Gibco). Organic solvents (analytical grade) were purchased from Merck.
PLANT MATERIAL
Inula aucheriana was collected from the West Azerbaijan province, Iran (July 2010) and was au thenticated by botanists of the Traditional Medicine and Materia Medica Research Center (TMRC), Shahid Beheshti University of Medical Sciences, Tehran, Iran. A voucher specimen was deposited at TMRC Herbarium for future reference (voucher no. TMRC 3173). The aerial parts of the species were dried in shade and ground for extraction.
EXTRACTION AND ISOLATION
Dried aerial parts of I. aucheriana (500 g) were successively extracted with n-hexane and chloroform (each solvent for 3 days), every day the solvent was replaced with fresh solvent to yield n-hexane (9.4 g) and chloroform (8.9 g) extracts. The chloroform extract (5 g) was subjected to Vacuum Liquid Chromatography [(VLC) silica gel, 40-63 Nm] using sequential washings with EtOAc, EtOAc -MeOH (2:1)-(1:1) and MeOH as eluent to give two fractions (F1-F2). Fraction F1 (1600 mg) was fractionated by silica gel SPE (7.5 × 2.5 cm, 40-63 μm) with CH2Cl2, CH2Cl2- EtOAc (10:1)- (1:1), EtOAc , EtOAc- MeOH (3:1)-(1:1) and MeOH respectively, to afford 17 fractions (Fr1-Fr17). Fraction Fr8 (139 mg) was further subjected to semi preparative HPLC (Shimadzu, SPD 20A :ODS, PDA detector) with water-acetonitrile (75:25) as the mobile phase and flow rate of 8 mL/min, to finally afford compound A (8 mg, 0.0016%), B (26 mg, 0.0052%) and C (5 mg, 0.0010%).
PREPARATION FOR MTT ASSAY
The compounds were dissolved in DMSO (5 mg/500 μL). The final concentrations (100 µg/mL, 50 µg/mL, 25 µg/mL, 12.5 µg/mL, 6.25 µg/mL and 3.125 µg/mL) were provided from the above solution (final DMSO concentration was 1%).
CELL LINES
The human tumor cell lines were obtained from the Pasteur Institute, Tehran, Iran: HepG-2 (human hepatocellular carcinoma), MCF-7 (human breast adenocarcinoma), and A-549 (Non-small cell lung carcinoma). MCF-7 cells were cultured in DMEM medium with 5% FBS, while the other two cell lines were maintained in RPMI 1640 medium with 10% FBS to obtain the desired growth. All cell lines were treated with 1% penicillin-streptomycin, in a humidified incubator at 37°C in an atmosphere of 5% CO2.
MTT ASSAY
The assessment of the viability of the cells was carried out in a micro culture tetrazolium/formazan assay (MTT assay) (Mosaddegh et al. 2006MOSADDEGH M, HAMZELOO-MOGHADAM M, GHAFARI S, NAGHIBI F, OSTAD SN and READ RW. 2010. Sesquiterpene lactones from Inula oculus-christi. Nat Prod Commun 5(4): 511-514., 2010). Ninety six-well plates were used and HepG-2 cells 15 × 103, MCF-7 cells 8 × 103 and A-549 cells 8 × 103 were seeded in each well. They were then incubated at 37°C. After 24h the medium was replaced with fresh medium containing different concentrations of the compounds to be tested. After 72h exposure of the cells at 37°C to each sample, the medium was replaced with fresh medium containing MTT, with a final concentration of 0.5 mg/mL. The cells were incubated for another 4h in a humidified atmosphere at 37°C, then the medium containing MTT was removed and the remaining formazan crystals dissolved in DMSO. The absorbance was recorded at 570 nm with an ELISA reader (TECAN). Tamoxifen was used as positive control. The relative cell viability (%) related to control wells containing cells, cell culture medium and DMSO 1%, was calculated by [A]samples /[A]control × 100 where [A]samples was the absorbance of test sample and [A]control was the absorbance of wells containing cells + medium+DMSO1%. To calculate IC50, viability (%) versus log concentrations was graphed by Microsoft Excel program, and the concentration at 50% viability was determined.
RESULTS AND DISCUSSION
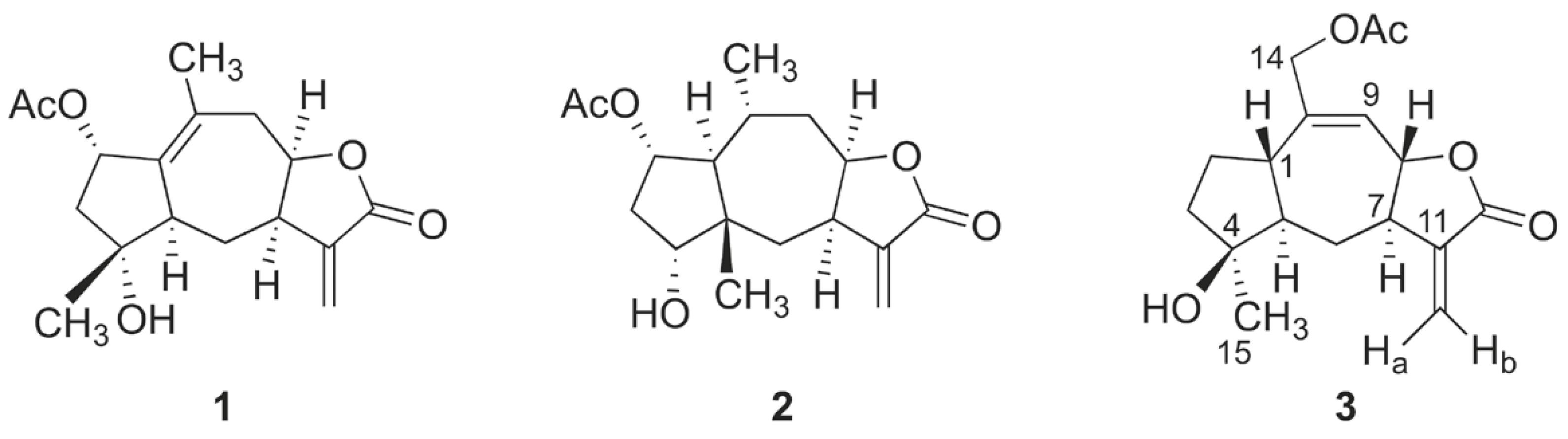
Compounds A, B and C, reported for the first time from Inula aucheriana were identified as inuchinenolide B 1, 14-acetoxy-1β,5α,7αH-4β-hydroxy-guai-9(10),11(13)-dien-12,8α- olide 2 (here named aucherinolide) and 6- deoxychamissonolide 3 (Fig. 1), respectively.
Although they were known compounds (Ito and Iida 1981ITO K and IIDA T. 1981. Seven sesquiterpene lactones from Inula britannica var. chinensis Phytochem 20: 271-273., Cheng et al. 2012CHENG XR, LI WW, REN J, ZENG Q, ZHANG SD, SHEN YH, YAN SK, YE J, JIN HZ and ZHANG WD. 2012. Sesquiterpene Lactones from Inula hookeri. Planta Med 78(5): 465-471., Willuhn et al. 1981WILLUHN G, PRETZSCH G and WENDISCH D. 1981. Zwei neue pseudoguaianolide aus den blüten von Arnica chamissonis less. Subsp. Genuina Maguire. Tetrahedron 37: 773-776.), their 2D NMR data were not available in the literature, and for 6-deoxychamissonolide 2, the previously reported NMR spectra was obtained in acetone-D6, while those in this report were obtained in CDCl3. The 1D and 2D NMR data for inuchinenolide B and 6-deoxychamissonolide are reported in Table I. The molecular formulae of C17H22O5 for inuchinenolide B (m/z: 329.1357 [M+Na]+) and C17H24O5 for 6-deoxychamissonolide (m/z: 331.1515 [M+Na]+) were revealed by high-resolution QTOF mass spectrometric analysis.
1H and 13C NMR chemical shifts (d, ppm), 1H multiplicities (mult.) and coupling constants (J, Hz), 1H-13C correlations in HMBC spectra and 1H- 1H correlations for compounds A and C in CDCl3.
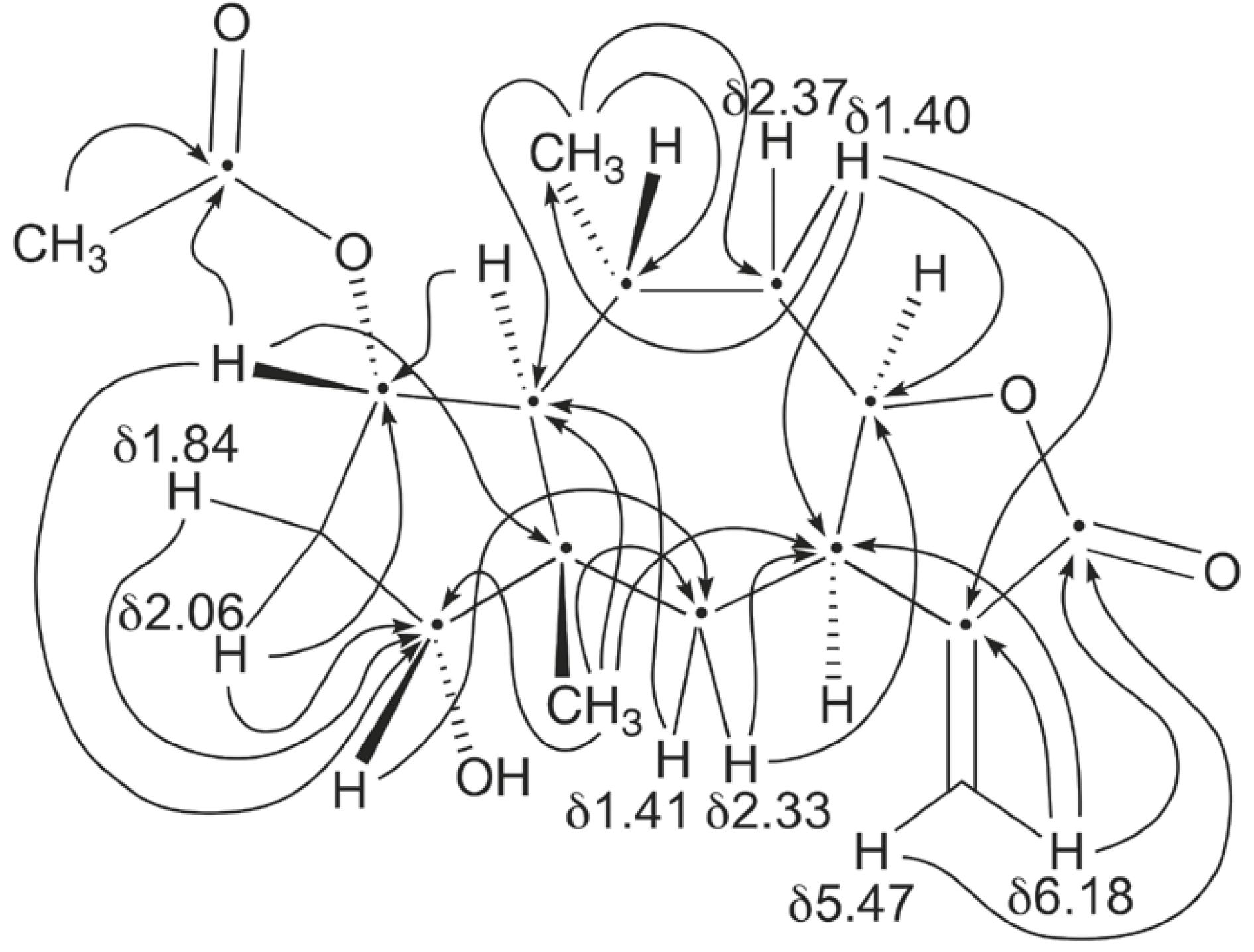
Compound B, which we name aucherinolide, was unusual. The molecular formula of C17H22O5 (m/z: 345.1310 [M+K]+) was obtained by high-resolution QTOF mass spectrometry. The 1H- and 13C-NMR spectra and the key HMBC correlations (Fig. 2) proved it to be a 12,8-guaianolide sesquiterpene lactone with an acetate group at C-14. The structure was concluded as being 14-acetoxy-1β,5α,7αH-4β-hydroxy-guai-9(10),11(13)-dien- 12,8α-olide 2 (Fig. 1) based on this information and after comparison of the spectroscopic data with those of a new substance introduced by Cheng et al. (2012) for Inula hookeri. In particular, relative stereochemistry was able to be assigned by Cheng et al. (2012) through NOESY experiments. The NMR data are given in Table II and agree very closely with those of Cheng et al. (2012). The only exception is for the assigned proton chemical shift for Ha2, for which we see clear correlation evidence for assignment at δ1.78, overlapped with the signal for Hb3, and not at δ1.40, as reported.
1H and 13C NMR chemical shifts (d, ppm), 1H multiplicities (mult.) and coupling constants (J, Hz), 1H-13C correlations in HMBC spectra and 1H-1H correlations for compounds A and C in CDCl3.
There are several reports of cytotoxic and apoptotic sesquiterpene lactones from the genus Inula. A brief report follows. 1,6-O,O-diacetylbritannilactone from Inula britannica L. demonstrated strong cytotoxic activity and induction of apoptosis in HL-60 cells through the activation of caspase-8, -9, and -3, phos phorylation of Bcl-2 and Bid, and by augmenting the release of cytochrome c from mitochondria (Pan et al. 2007PAN MH, CHIOU YS, CHENG AC, BAI N, LO CY, TAN D and HO CT. 2007. Involvement of MAPK, Bcl-2 family, cytochrome c, and caspases in induction of apoptosis by 1,6 ,Odiacetylbritannilactone in human leukemia cells. Mol Nutr Food Res 51(2): 229-238.). Acetylbritannilactone, also from Inula britannica, has enforced apoptosis in VSMC cells as shown by induction of a greater ratio of Bax/Bcl-2, activation of caspase-9, caspase-3, and the breakdown of Poly (ADP-ribose) polymerase (Liu et al. 2011LIU B, HAN M, SUN RH, WANG JJ, LIU YP and WEN JK . 2011. Acetylbritannilactone induces G1 arrest and apoptosis in vascular smooth muscle cells. Int J Cardiol 149(1): 30-38.). It has also hindered the growth via impelling cell cycle arrest in G0/G1 phase of HT-29 cancer cells (Fang et al. 2011FANG XM, LIU B, LIU Y, WANG JJ, WEN JK, LI BH and HAN M. 2011. Acetylbritannilactone suppresses growth via upregulation of krüppel-like transcription factor 4 expression in HT-29 colorectal cancer cells. Oncol Rep 26(5): 1181-1187.). Eupatolide, another sesquiterpene from the same species has been shown to induce apoptosis in human breast cancer cells through tumor necrosis factorrelated apoptosis-inducing ligand (Lee et al. 2010LEE J, HWANGBO C, LEE JJ, SEO J and LEE JH. 2010. The sesquiterpene lactone eupatolide sensitizes breast cancer cells to TRAIL through down-regulation of c- FLIP expression. Oncol Rep 23(1): 229-237.). Ergolide has also been isolated from Inula britannica. Studies about the potential of this cytotoxic sesquiterpene lactone have proposed the mechanism for its inducing apoptosis through down regulation of cell survival signal molecules which occur as a consequence of inhibiting the NFkappaB signaling pathway (Song et al. 2005SONG YJ, LEE DY, KIM SN, LEE KR, LEE HW, HAN JW, KANG DW, LEE HY and KIM YK . 2005. Apoptotic potential of sesquiterpene lactone ergolide through the inhibition of NF-kappaB signaling pathway. J Pharm Pharmacol 57(12): 1591-1597.).
There has also been an effort to synthesize derivatives of Inula britannica sesquiterpene lactones through a study by Liu et al. (2005); derivatives of 1-O-acetylbritannilactone, (2- Oalkyloxime-3-phenyl)-propionyl-1-O-acetylbritannilactone esters which are sesquiterpene lactones of the above mentioned species, have been synthesized and four of them have exhibited antiproliferative effects on HL-60 and Bel-7402 cell lines (Liu et al. 2005). Dimeric sesquiterpene lactones japonicones A-D have been previously isolated from Inula japonica Thunb., among which japonicone A has displayed the most potent cytotoxic activity on A-549, LOVO, CEM and MDA-MB-435 cells (Qin et al. 2009QIN JJ, JIN HZ, FU JJ, HU XJ, WANG Y, YAN SK and ZHANG WD. 2009. Japonicones A-D, bioactive dimeric sesquiterpenes from Inula japonica Thunb. Bioorg Med Chem Lett 19(3): 710-713.). Germacranolide inulacappolide from Inula cappa DC, has been evaluated for cytotoxicity and has shown strong activity against HeLa, K-562 and KB cell lines (Xie et al. 2007XIE HG, CHEN H, CAO B, ZHANG HW and ZOU ZM. 2007. Cytotoxic Germacranolide Sesquiterpene from Inula cappa. Chem Pharm Bull 55(8): 1258-1260.) and bigelovin isolated from Inula helianthus-aquatica has been cytotoxic to U937 cells and it has also instigated apoptosis and arrested the cell cycle at G(0)/G(1) phase (Zeng et al. 2009ZENG GZ, TAN NH, JI CJ, FAN JT, HUANG HQ, HAN HJ and ZHOU GB. 2009. Apoptosis inducement of bigelovin from Inula helianthus-aquatica on human Leukemia U937 cells. Phytother Res 23(6): 885-891.). Pseudoguaianolides and guaianolides from Inula hookeri C. B. Clarke have been likewise investigated for antiproliferative activity on HepG-2, HeLa, PC-3, and MGC-803 cell lines and have demonstrated cytotoxic results (Cheng et al. 2012). The sesquiterpene lactone isocostunolide from the roots of Inula helenium has strongly enforced cytotoxic activity in A-2058, HT-29, and HepG-2 cell lines. It has also been reported that isocostunolide could induce apoptosis in A-2058 cells via a mitochondria-dependent pathway (Chen et al. 2007CHEN CN, HUANG HH, WU CL, LIN CP, HSU JT, HSIEH HP, CHUANG SE and LAI GM. 2007. Isocostunolide, a sesquiterpene lactone, induces mitochondrial membrane depolarization and caspasedependent apoptosis in human melanoma cells. Cancer Lett 246(1-2): 237-252.) and finally, gaillardin a sesquiterpene lactone isolated from the aerial parts of Inula oculus-christi has demonstrated cytotoxic activity and apoptotic induction in MCF-7 cells (Hamzeloo-Moghadam et al. 2013).
We have previously reported the apoptotic sesquiterpene lactone britannin from Inula aucheriana and in the present study three other sesquiterpene lactones have been isolated from the species. The amount of 6-deoxychamissonolide was not enough for biologic assay so the cytotoxic activities of inuchinenolide B and aucherinolide were evaluated and the results are shown in Figures 3-5 and Table III. Qin and colleagues have examined the cytotoxicity of inuchinenolide B in MCF-7 cells and have reported the IC50 value as 15.5 μM (Qin et al. 2013); however, we have determined the IC50 value to be 39.0 μg/mL (118 μM) in MCF-7 cells. The values were higher in HepG-2 and A-549 cell lines but still revealed the cytotoxicity of the compound to these cells (56.6 μg/mL equal to 172 μM) and (55.7 μg/mL equal to 169 μM) in HepG-2 and A-549 cells, respectively. Aucherinolide demonstrated to be more potent in the above mentioned cells with IC50 values of 19.0 μg/mL (55 μM), 15.3 μg/mL (44 μM) and 11.8 μg/mL (34 μM) in HepG-2, A-549 and MCF-7 cells, respectively. The results could explain the previously reported cytotoxic results of Inula aucheriana extracts.
Viability of inuchinenolide B and aucherinolide in HepG-2 cells. Values represent the mean ±SD of 3 separate experiments.
Viability of inuchinenolide B and aucherinolide in A-549 cells. Values represent the mean ±SD of 3 separate experiments.
Viability of inuchinenolide B and aucherinolide in MCF-7 cells. Values represent the mean ±SD of 3 separate experiments.
Surveying the literature about the cytotoxicity of various sesquiterpene lactones which have been evaluated through MTT assay, reveals different ranges of IC50 for these compounds. Examples include deoxyelephantopin, a sesquiterpene lac tone from Elephantopus scaber which has been evaluated in A-549 cell line to show cytotoxicity with IC50 12.287 μg/mL (Kabeer et al. 2013KABEER FA, SREEDEVI GB, NAIR MS, RAJALEKSHMI DS, GOPALAKRISHNAN LP, KUNJURAMAN S and PRATHAPAN R. 2013. Antineoplastic effects of deoxyelephantopin, a sesquiterpene lactone from Elephantopus scaber, on lung adenocarcinoma (A549) cells. J Integr Med 11(4): 269-277. ). In another study, IC50 values of 6.37, 6.20 and 4.76 μg/mL have been reported for sesquiterpene lactone gaillardin from the aerial parts of Inula oculus-christi in MCF-7, HepG-2 and A-549 cell lines, repectively (Hamzeloo-Moghadam et al. 2013), while sesquiterpene lactones from Laserpitium species have exhibited cytotoxicity with IC50 values of 4.32-97.54 μM against MCF 7/6 cell line (Popović et al. 2013). Moreover, Rosselli et al. (2012)ROSSELLI S, BRUNO M, RAIMONDO FM, SPADARO V, VAROL M, KOPARAL AT and MAGGIO A. 2012. Cytotoxic Effect of Eudesmanolides Isolated from Flowers of Tanacetum vulgare ssp. Siculum Molecules 17: 8186-8195 have demonstrated IC50 values of five sesquiterpene lactones isolated from flowers of Tanacetum vulgare against A-549 cells to be 15.3-59.4 μM (Rosselli et al. 2012). Considering the outcomes of the present study, IC50 values of inuchinenolide B seemed rather high; however, the results of MTT assay for aucherinolide were comparable with some published reports about the in vitro toxcicity of sesquiterpene lactones mentioned above.
CONCLUSIONS
As mentioned earlier there are several reports about the cytotoxicity and apoptotic inducing properties of sesquiterpene lactones of the genus Inula. The sesquiterpene lactones of the present study, especially the two that have demonstrated cytotoxic activity somewhat explain the previously reported cytotoxicity demonstrated by extracts of Inula aucheriana. These compounds might execute their cytotoxic properties through apoptotic induction; however, this assumption needs further investigations about the molecular mechanisms of their cytotoxic activity.
ACKNOWLEDGMENTS
The project was granted by Traditional Medicine and Materia Medica Research Center (TMRC), Shahid Beheshti University of Medical Sciences (Grant No. 118). The authors also express their gratitude to Mr. Hamid Moazzeni, (TMRC) for assistance in collecting the plant. Mass spectrometric analysis for this work was carried out at the Bioanalytical Mass Spectrometry Facility, UNSW.
REFERENCES
- BOWN D. 2002. Encyclopedia of Herbs and Their Uses. London: Dorling Kindersley.
- CHEN CN, HUANG HH, WU CL, LIN CP, HSU JT, HSIEH HP, CHUANG SE and LAI GM. 2007. Isocostunolide, a sesquiterpene lactone, induces mitochondrial membrane depolarization and caspasedependent apoptosis in human melanoma cells. Cancer Lett 246(1-2): 237-252.
- CHENG XR, LI WW, REN J, ZENG Q, ZHANG SD, SHEN YH, YAN SK, YE J, JIN HZ and ZHANG WD. 2012. Sesquiterpene Lactones from Inula hookeri. Planta Med 78(5): 465-471.
- FANG XM, LIU B, LIU Y, WANG JJ, WEN JK, LI BH and HAN M. 2011. Acetylbritannilactone suppresses growth via upregulation of krüppel-like transcription factor 4 expression in HT-29 colorectal cancer cells. Oncol Rep 26(5): 1181-1187.
- HAMZELOO-MOGHADAM M, HAJIMEHDIPOOR H, SAIEDNIA S, ATOOFI A, SHAHRESTANI R, READ RW and MOSADDEGH M. 2012. Anti-proliferative Activity and Apoptotic Potential of Britannin, a Sesquiterpene Lactone from Inula aucheriana. Nat Prod Commun 7(8): 979-980.
- HAMZELOO-MOGHADAM M, NAGHIBI F, ATOOFI A, ASGHARIAN REZAIE M, IRANI M and MOSADDEGH M. 2013. Cytotoxic Activity and Apoptosis Induction by Gaillardin. Z Naturforsch 68c: 108-112.
- ITO K and IIDA T. 1981. Seven sesquiterpene lactones from Inula britannica var. chinensis Phytochem 20: 271-273.
- KABEER FA, SREEDEVI GB, NAIR MS, RAJALEKSHMI DS, GOPALAKRISHNAN LP, KUNJURAMAN S and PRATHAPAN R. 2013. Antineoplastic effects of deoxyelephantopin, a sesquiterpene lactone from Elephantopus scaber, on lung adenocarcinoma (A549) cells. J Integr Med 11(4): 269-277.
- LEE J, HWANGBO C, LEE JJ, SEO J and LEE JH. 2010. The sesquiterpene lactone eupatolide sensitizes breast cancer cells to TRAIL through down-regulation of c- FLIP expression. Oncol Rep 23(1): 229-237.
- LIU B, HAN M, SUN RH, WANG JJ, LIU YP and WEN JK . 2011. Acetylbritannilactone induces G1 arrest and apoptosis in vascular smooth muscle cells. Int J Cardiol 149(1): 30-38.
- LIU S, LIU H, YAN W, ZHANG L, BAI N and HO CT. 2005. Design, synthesis, and anti-tumor activity of (2-Oalkyloxime-3-phenyl)-propionyl-1-Oacetylbritannilactone esters. Bioorg Med Chem 13(8): 2783-2789.
- MOSADDEGH M, HAMZELOO-MOGHADAM M, GHAFARI S, NAGHIBI F, OSTAD SN and READ RW. 2010. Sesquiterpene lactones from Inula oculus-christi. Nat Prod Commun 5(4): 511-514.
- MOSADDEGH M, OSTAD SN, NAGHIBI F and HAMZELOO-MOGHADAM M. 2006. Cytotoxic effects of five species of Inula against some tumor cell lines. Iranian J Pharmaceut Sci 2: 203-208.
- PAN MH, CHIOU YS, CHENG AC, BAI N, LO CY, TAN D and HO CT. 2007. Involvement of MAPK, Bcl-2 family, cytochrome c, and caspases in induction of apoptosis by 1,6 ,Odiacetylbritannilactone in human leukemia cells. Mol Nutr Food Res 51(2): 229-238.
- POPOVIĆ V, HEYERICK A, PETROVIĆ S, VAN CALENBERGH S, KARALIĆ I, NIKETIĆ M and DEFORCE D. 2013. Sesquiterpene lactones from the extracts of two Balkan endemic Laserpitium species and their cytotoxic activity. Phytochemistry 87: 102-111.
- QIN JJ, JIN HZ, FU JJ, HU XJ, WANG Y, YAN SK and ZHANG WD. 2009. Japonicones A-D, bioactive dimeric sesquiterpenes from Inula japonica Thunb. Bioorg Med Chem Lett 19(3): 710-713.
- QIN JJ, JIN HZ, HUANG Y, ZHANG SD, SHAN L, VORUGANTI S, NAG S, WANG W, ZHANG WD and ZHANG R. 2013. Selective cytotoxicity, inhibition of cell cycle progression, and induction of apoptosis in human breast cancer cells by sesquiterpenoids from Inula lineariifolia Turcz. Eur J Med Chem 68: 473-481.
- ROSSELLI S, BRUNO M, RAIMONDO FM, SPADARO V, VAROL M, KOPARAL AT and MAGGIO A. 2012. Cytotoxic Effect of Eudesmanolides Isolated from Flowers of Tanacetum vulgare ssp. Siculum Molecules 17: 8186-8195
- SECA AM, GRIGORE A, PINTO DC and SILVA AM . 2014. The genus Inula and their metabolites: from ethnopharmacological to medicinal uses. J Ethnopharmacol 154(2): 286-310.
- SONG YJ, LEE DY, KIM SN, LEE KR, LEE HW, HAN JW, KANG DW, LEE HY and KIM YK . 2005. Apoptotic potential of sesquiterpene lactone ergolide through the inhibition of NF-kappaB signaling pathway. J Pharm Pharmacol 57(12): 1591-1597.
- WILLUHN G, PRETZSCH G and WENDISCH D. 1981. Zwei neue pseudoguaianolide aus den blüten von Arnica chamissonis less. Subsp. Genuina Maguire. Tetrahedron 37: 773-776.
- XIE HG, CHEN H, CAO B, ZHANG HW and ZOU ZM. 2007. Cytotoxic Germacranolide Sesquiterpene from Inula cappa. Chem Pharm Bull 55(8): 1258-1260.
- ZENG GZ, TAN NH, JI CJ, FAN JT, HUANG HQ, HAN HJ and ZHOU GB. 2009. Apoptosis inducement of bigelovin from Inula helianthus-aquatica on human Leukemia U937 cells. Phytother Res 23(6): 885-891.
- ZHAO YM, ZHANG ML, SHI QW and KIYOTA H. 2006. Chemical constituents of plants from the genus Inula. Chem Biodivers 3(4): 371-384.
Publication Dates
-
Publication in this collection
08 May 2015 -
Date of issue
Apr-Jun 2015
History
-
Received
13 Feb 2014 -
Accepted
24 Oct 2014