ABSTRACT
Clupeomorphs are found in many assemblages of the northeastern Brazil, whose ages range from the Neocomian to Paleogene. Ten species were described. Among them †Scutatuspinosus itapagipensis, a torpedo-like fish found in shales of the Marfim Formation from Bahia, remains poorly known. At first it was positioned within †Scutatuspinosinae and indicated as closely related to †Diplomystus. Diagnoses for the subfamily and genus were based on certain characters masking relationships. Recently it was placed in †Ellimmichthyiformes, but its systematic position remains controversial. We furnish additional data and restorations together with a parsimony analysis with TNT program, using 60 unordered and unweighted characters from selected species of 13 genera. Our analysis produced 12 trees with 158 steps, a consistency index (CI) of 0.44 and retention index (RI) of 0.52. Low support indices still indicate insufficient data for many taxa and uncertain status for clades. According to the majority rule consensus, †Scutatuspinosus itapagipensis is placed within a “†paraclupeine” group with †Ezkutuberezi carmenae, †Ellimma branneri, †Ellimmichthys longicostatus, and †Paraclupea chetunguensis. Its minimum age and position among †paraclupeids indicate an early history of the group older than it was thought to be, supporting a biogeographical hypothesis based on generalized track from China to northeastern Brazil during the Lower Cretaceous.
Key words:
†Scutatuspinosus itapagipensis; osteology; systematics; Recôncavo Basin; Lower Cretaceous
INTRODUCTION
The Brazilian Cretaceous is very rich in deposits containing vertebrate remains, particularly bony fishes. Among them clupeomorphs are common members of many fossiliferous assemblages, mainly from northeastern Brazil (Gallo and Figueiredo 2002GALLO V and FIGUEIREDO FJ. 2002. Clupeomorph fishes from the Lower Cretaceous of the Recôncavo Basin, Northeastern Brazil. J Vert Paleontol 22(3): 55A.). At least eight genera and 10 species have been described to date (Figueiredo and Ribeiro 2016). Of them, †Scutatuspinosus itapagipensis, a double-armored herring-like fish known from the Neocomian of Recôncavo Basin, matters as a puzzling taxon whose systematic position remains unclear.
Santos and Corrêa (1985SANTOS RS and CORRÊA VLS. 1985. Contribuição ao conhecimento da paleoictiofaúnula do Cretáceo do Brasil. In: Campos DA et al. (Orgs), Coletânea de Trabalhos Paleontológicos, Brasília: Ministério das Minas e Energia e Departamento Nacional da Produção Mineral, p. 169-174.) originally described †Scutatuspinosus itapagipensis from greyish lacustrine shales of the Marfim Formation (Ilhas Group) cropping out in the locality of Itapagipe, Salvador. As far as known, this fish is the most ancient †ellimmichthyiform known, dated from the Hauterivian-Barremian interval (Patterson 1993PATTERSON C. 1993. Osteichthyes: Teleostei. In: Benton M (Ed), The Fossil Record 2, London: Chapman and Hall, p. 621-656., Murray and Wilson 2013MURRAY AM and WILSON MVH. 2013. Two new paraclupeid fishes (Clupeomorpha: Ellimmichthyiformes) from the Upper Cretaceous of Morocco. In: Arratia G et al. (Eds), Mesozoic Fishes 5 - Global Diversity and Evolution, München: Verlag Dr. Pfeil, Germany, p. 267-290.). Although Santos and Corrêa (1985) used many specimens in the original description (n=72), the skeletal anatomy was only superficially studied. †Scutatuspinosus itapagipensis was placed within a new subfamily †Scutatuspinosinae within Clupeidae and considered closely related to †Diplomystus, which was assumed by past authors (e.g., Schaeffer 1947SCHAEFFER B. 1947. Cretaceous and Tertiary actinopterygian fishes from Brazil. Bull Amer Mus Nat Hist 141(3): 357-474.) to be a primitive taxon of this family. The diagnosis of this subfamily was based on a set of generalized characters for clupeomorphs thus masking putative affinities.
Grande (1982GRANDE L. 1982. A revision the fossil genus Diplomystus, with comments on the interrelationships of clupeomorph fishes. Am Mus Novit 2728: 1-34., 1985) set forth a cladistic basis for clupeomorph systematics characterizing the group as a whole and discovering many monophyletic subunities. In presenting his review of clupeomorphs, he recognized the monophyletic assemblage †Ellimmichthyiformes diagnosed based on derived broader than long predorsal scutes and postulated as sister group of the speciose Clupeiformes (Grande 1982).
Afterwards, Chang and Maisey (2003CHANG MM and MAISEY JG. 2003. Redescription of †Ellimma branneri and †Diplomystus shengliensis, and relationships of some basal clupeomorphs. Am Mus Novit 3404: 1-35. ) performed a cladistic analysis for selected †ellimmichthyiform fishes and, although not including †Scutatuspinosus, indicated this taxon as a putative member of the group. This claim was accepted by Hay et al. (2007HAY MJ, CUMBAA SL, MURRAY AM and PLINT AG. 2007. A new paraclupeid fish (Clupeomorpha, Ellimmichthyiformes) from a muddy marine prodelta environment: middle Cenomanian Dunvegan Formation, Alberta, Canada. Canad J Earth Sci 44(6): 775-790.) without any justifying character. In recent years, great advances in deciphering early clupeomorph relationships have been achieved but a consensus has not emerged. The main cause seems to be that there are several cases of insufficient information about taxa. Thus †Scutatuspinosus has been either omitted (Forey 2004FOREY PL. 2004. A three-dimensional skull of a primitive clupeomorph from the English Chalk and implications for the evolution of the clupeomorph acusticolateralis system. In: Arratia G and Tintori A (Eds), Mesozoic Fishes 3 - Systematics, Paleoenvironments and Biodiversity, München: Verlag Dr. Friedrich Pfeil, Germany , p. 405-427., Zaragüeta-Bagils 2004) or included (i.e., Alvarado-Ortega et al. 2008, Murray and Wilson 2013MURRAY AM and WILSON MVH. 2013. Two new paraclupeid fishes (Clupeomorpha: Ellimmichthyiformes) from the Upper Cretaceous of Morocco. In: Arratia G et al. (Eds), Mesozoic Fishes 5 - Global Diversity and Evolution, München: Verlag Dr. Pfeil, Germany, p. 267-290., Figueiredo and Ribeiro 2016FIGUEIREDO FJ and RIBEIRO DRM. 2016. Relationships of †Codoichthys carnavalii Santos, 1994 (Teleostei, Clupeomorpha, †Ellimmichthyiformes) from the late Aptian of São Luís-Grajaú basin, NE Brazil. An Acad Bras Cienc 88: 1277-1307.) in cladistic analyses. Examining those analyses that included this taxon we noted that authors have used utterly doubtful or controversial anatomical data taken from the published literature. Taking into account its minimum age and primitiveness, †Scutatuspinosus shows key and putative characters to decipher early clupeomorph relationships. Thus it makes clear that a redescription of this taxon is of critical importance before any attempt is made to decipher its affinities. So, in this paper we furnish a detailed reassessment of the osteology of †Scutatuspinosus as the preservation of specimens permit, and explore its relationships using cladistic methodology.
GEOLOGICAL SETTING
The Recôncavo Basin encompasses an exposed area of about 11,500 km2 in northeastern Brazil corresponding to the southern part of a NE-SW trending intracontinental rift formed by a series of asymmetrical grabens (Caixeta et al. 1994CAIXETA JM, BUENO GV, MAGNAVITA LP and FEIJÓ FJ. 1994. Bacias do Recôncavo, Tucano e Jatobá. Bol Geoci PETROBRAS 8(1): 163-172., Maisey 2000MAISEY JG. 2000. Continental break up and the distribution of fishes of Western Gondwana during the Early Cretaceous. Cretaceous Res 21: 281-314.). It is limited on the eastern margin by the Salvador fault and to west by Maragogipe fault and limited to north and northwest with Tucano basin through the Aporá structural high, while to the south its exposed portion is delimited by the Itacaré structural high. The rock sequence reaches about 7000 m in height (Caixeta et al. 1994).
Since 1937 Recôncavo Basin has been studied taking into account petroleum exploration purposes (Rocha et al. 2002ROCHA PS, SOUZA AO DE AB, and CÂMARA RJB. 2002. O futuro da bacia do Recôncavo, a mais antiga província petrolífera brasileira. Bahia Análise & Dados 11(4): 32-44.). In addition, this basin is also important to decipher many aspects of the puzzling early Atlantic Ocean history, inasmuch as Recôncavo together with Tucano and Jatobá basins, in association with the Sergipe-Alagoas Basin, are the result of the crustal stretching process responsible for the Gondwana break-up and consequently separation of Africa and South America (Arai 2014ARAI M. 2014. Aptian/Albian (Early Cretaceous) paleogeography of the South Atlantic: a paleontological perspective. Brazilian J Geol 44(2): 339-350.). In fact, it comprises informative clastic sediments deposited in eolian, lacustrine, deltaic and fluvial environments during the Early Cretaceous rifting process at the beginning of the Gondwana break-up (Maisey 2000MAISEY JG. 2000. Continental break up and the distribution of fishes of Western Gondwana during the Early Cretaceous. Cretaceous Res 21: 281-314.).
Recôncavo Basin is basally filled with a stratigraphic sucession ranging from Upper Jurassic to Lower Cretaceous, representing two major sedimentary sequences: the first, called pre-rift, constitutes an arid alluvial fan system and associated facies, represented by Afligidos, Aliança, Sergi, Itaparica, and Água Doce formations; the second syn-rift, is composed of alluvial, fluvial, and deltaic lacustrine sediments corresponding to the complete record of infilling of a tectonically active lake basin, represented by the Lower Cretaceous formations Candeias, Maracangalha, Marfim, Pojuca and São Sebastião (Falcão and Della Favera 2012FALCÃO LC and DELLA FAVERA JC. 2012. Sublacustrine channel-levee system of the Caruaçu member, southwest Recôncavo Basin, Bahia, Brazil. Rev Brasil Geoci 42(3): 539-546.). Apparently, the syn-rift phase finished in the Lower Aptian and a post-rift (Sag) phase began with the deposit of sandstones, shales and limestones (as in the case of the Marizal Formation). Nowadays, a thin package of Upper Cretaceous and Tertiary post-rift clastics (e.g., Miocene marine deposits of Sabiá Formation and Pliocene deposits of the Barreiras Formation) partially covers the basin (Magnavita et al. 2005MAGNAVITA LP, SILVA RS and SANCHES CP. 2005. Roteiros geológicos e guia de campo da Bacia do Recôncavo, NE do Brasil. Bol Geoci PETROBRAS 13: 301-334.).
The Candeias Formation was deposited in a fresh deep-water lake after a deltaic complex represented by Maracangalha and Marfim formations (Falcão and Della Favera 2012FALCÃO LC and DELLA FAVERA JC. 2012. Sublacustrine channel-levee system of the Caruaçu member, southwest Recôncavo Basin, Bahia, Brazil. Rev Brasil Geoci 42(3): 539-546.). Regarding fossils, Allport (1860ALLPORT S. 1860. On the discovery of some fossil remains near Bahia in South America. Quart J Geol Soc 16: 263-268.) pointed out the occurrence of fish scales, crocodile teeth, a toothed lower jaw assigned to †Cladocyclus, and a probable dinosaur vertebrae in an outcrop of the Candeias Formation in the locality of Almeida Brandão. Cope (1886COPE ED. 1886. A contribution to the vertebrate paleontology of Brazil. Proc Am Philos Soc 23(121): 3-4.) registered the occurrence of the †paraclupeid †Ellimmichthys longicostatus in the localities of Itacaranha, Almeida Brandão, and Simões Filho (formerly Água Comprida). For this latter locality, he pointed out occurrence of fragments of †Cladocyclus mawsoni. After the death of Cope, Woodward (1908WOODWARD AS. 1908. On some Fossil Fishes discovered by Ennes de Souza in the Cretaceous Formation of Ilhéos (State of Bahia), Brazil. Quartl J Geol Soc London 64: 358-362.) received part of his large collection and identified and described semionotids (i.e., species of †Lepidotes), teleosts (†Cladocyclus, †Ellimmichthys), coelacanth (†Mawsonia gigas), crocodile (†Goniopholis hartii), and pterosaurs as well as dinosaur remains.
Silva (1993SILVA VG. 1993. Taxonômica e posicionamento estratigráfico de paleovertebrados da Bacia do Recôncavo. Acta Geol Leopoldensia 16(38): 41-58. ), while recovering data from Cope’s collection housed in the Museu Nacional do Rio de Janeiro, revisited many outcrops of Cretaceous formations in Recôncavo Basin, furnishing a detailed description of the main fish-bearing sections. She pointed out the presence of scales of †Lepidotes in the lacustrine shales of Maracangalha and Candeias formations in Santo Amaro. Even in the Itaparica island, in which Candeias shales crops out, particularly in Manguinhos and Bom Despacho beaches, many fish remains were collected. Other fossiliferous outcrops of the Candeias Formation were explored at Itacaranha, Mapele, and Simões Filho. In those lacustrine deposits she confirmed the presence of many scattered fish remains (e.g., †Lepidotes scales, pycnodont teeth). In spite of the ocurrence of many taxa in assemblages of Recôncavo Basin, correlation is very difficult due to lack of putative index fossils.
MATERIALS AND METHODS
Fossils and preparation. All material of †Scutatuspinosus itapagipensis belongs to the paleontological collection of the Museu de Ciências da Terra, Departamento Nacional de Produção Mineral, Rio de Janeiro, and is referred herein by the abbreviation DGM followed by the register number. The specimens were collected by the geologist José Lino de Mello Junior, in 1935, during a field trip the neighboring Salvador, State of Bahia (Santos and Corrêa 1985SANTOS RS and CORRÊA VLS. 1985. Contribuição ao conhecimento da paleoictiofaúnula do Cretáceo do Brasil. In: Campos DA et al. (Orgs), Coletânea de Trabalhos Paleontológicos, Brasília: Ministério das Minas e Energia e Departamento Nacional da Produção Mineral, p. 169-174.).
We have used selected specimens from the 72 listed in the original description. Measurements follow Forey et al. (2003FOREY PL, YI L, PATTERSON C and DAVIES CE. 2003. Fossil Fishes from the Cenomanian (Upper Cretaceous) of Namoura, Lebanon. J Syst Palaeontol 1(4): 227-330. ) and they are listed in the Table I. Silicone peels were prepared of negative imprints on shales and magnesium oxide smoke was applied to the surface of specimens for enhancing skeletal details and reducing brightness in the photographs.
Morphometrical data of selected specimens of †Scutatuspinosus itapagipensis, in millimeters. An asterisk indicates estimated measurement.
Drawings were made under Motic stereomicroscope with tube drawing attachement. USB digital camera and MIAS ®software for PC computer were used to capture and to enhance images, particularly of predorsal scutes.
Phylogenetic analytical methods. The phylogenetic relationships as well as the relevance of some characters used in the diagnosis of †Scutatuspinosus were tested through a cladistic analysis performed with the program TNT (Goloboff et al. 2008GOLOBOFF P, FARRIS J and NIXON K. 2008. T.N.T, a free program for phylogenetic analysis. Cladistics 24: 774-786. ). We used a matrix of 60 unordered, independent, and unweighted osteological characters taken of 13 ellimmichthyiform taxa (Table II, Appendix - Supplementary Material). The taxa (including major references from which anatomical data are taken) are the following: †Codoichthys carnavalii (fideSantos 1994SANTOS RS. 1994. Ictiofáunula da Formação Codó, Cretáceo Inferior, com a descrição de um novo táxon - Codoichthys carnavalii (Pisces-Teleostei). An Acad Bras Cienc 66: 131-143., Figueiredo and Ribeiro 2016FIGUEIREDO FJ and RIBEIRO DRM. 2016. Relationships of †Codoichthys carnavalii Santos, 1994 (Teleostei, Clupeomorpha, †Ellimmichthyiformes) from the late Aptian of São Luís-Grajaú basin, NE Brazil. An Acad Bras Cienc 88: 1277-1307.), †Scutatuspinosus itapagipensis (see specimens listed below), †Diplomystus dentatus (fideGrande 1982GRANDE L. 1982. A revision the fossil genus Diplomystus, with comments on the interrelationships of clupeomorph fishes. Am Mus Novit 2728: 1-34.), †Ellimma branneri (fideChang and Maisey 2003CHANG MM and MAISEY JG. 2003. Redescription of †Ellimma branneri and †Diplomystus shengliensis, and relationships of some basal clupeomorphs. Am Mus Novit 3404: 1-35. , and DGM 142 to 160), †Ellimmichthys longicostatus (Grande 1982, Chang and Grande 1997, and MN 4428-V, DGM 32-P, DGM 64-P, DGM 58-P, BMNH 7109, BMNH 1035), †Ezkutuberezi carmenae (fide Poyato-Ariza et al. 2000), †Armigatus namourensis (fideForey et al. 2003FOREY PL, YI L, PATTERSON C and DAVIES CE. 2003. Fossil Fishes from the Cenomanian (Upper Cretaceous) of Namoura, Lebanon. J Syst Palaeontol 1(4): 227-330. ), †Triplomystus noorae (fide Forey et al. 2003), †Paraclupea chetungensis (fide Chang and Grande 1997), †Rhombichthys intocabilis (fideKhalloufi et al. 2010KHALLOUFI B, ZARAGÜETA-BAGILS R and LELIÈVRE H. 2010. Rhombichthys intoccabilis, gen. et sp. nov. (Ellimmichthyiformes, Clupeomorpha, Teleostei), from the Cenomanian (Upper Cretaceous) of Ein Yabrud, Middle East: anatomical description and phylogenetic implications. J Vert Paleontol 30: 57-67.), †Sorbinichthys africanus (fideMurray and Wilson 2011MURRAY AM and WILSON MVH. 2011. A new species of Sorbinichthys (Teleostei: Clupeomorpha: Ellimmichthyiformes) from the Late Cretaceous of Moroccco. Can J Earth Sci 48: 1-9.), †Thorectichthys marocensis (fide Murray and Wilson 2013), and †Tycheroichthys dunveganensis (fideHay et al. 2007HAY MJ, CUMBAA SL, MURRAY AM and PLINT AG. 2007. A new paraclupeid fish (Clupeomorpha, Ellimmichthyiformes) from a muddy marine prodelta environment: middle Cenomanian Dunvegan Formation, Alberta, Canada. Canad J Earth Sci 44(6): 775-790.).
Further comments are needed ad interim. We opted to exclude the monotypical †Horseshoeichthys armaserratus (Newbrey et al. 2010NEWBREY MG, MURRAY AM, BRINKMAN DB, WILSON MVH and NEWMAN AG. 2010. A new articulated freshwater fish (Clupeomorpha, Ellimmichthyiformes) from the Late Cretaceous Horseshoe Canyon Formation, Alberta, Canada. Can J Earth Sci 47: 1183-1196.) from the taxa list because its inclusion caused many missing data in the matrix. The Brazilian taxa †Ellimma cruzae (see Santos, 1990SANTOS RS. 1990. Clupeiformes e Gonorhychiformes do Cretáceo Inferior (Aptiano) da Formação Cabo, NE do Brasil. An Acad Bras Cienc 62: 261-268.) and †Ellimmichthys maceioensis (see Malabarba et al. 2004MALABARBA MC, DO CARMO DA, GÓMEZ-PÉREZ I and DE QUEIROZ-NETO JV. 2004. A new clupeomorph fish from the Cretaceous Maceió Formation, Alagoas Basin, NE Brasil. Neues Jahrb Geol Paläont 233(2): 255-274.) are not included in our analysis because they certainly do not belong to these genera and anatomical information is hitherto insufficient. A critical review of both taxa will be presented elsewhere.
Regarding living species, we used skeletons of Elops saurus (AO.UERJ 25, n=1, 150 mm SL; AO.UERJ 96, n=3, 130-152 mm SL; AO.UERJ 132, n=4, 210-250 mm SL; AO.UERJ 136, n=1, 250 mm SL, D.UERJ 190, n=15, 27-81 mm TL) together with data from the literature for the genus (i.e., Nybelin 1968NYBELIN O. 1968. The dentition in the mouth cavity of Elops. In: Orvig T (Ed), Current problems of Lower Vertebrate Phylogeny, Stockholm: Nobel Symposium 4, p. 439-443., 1971, Forey 1973bFOREY PL. 1973b. A revision of the elopiform fishes, fossil and Recent. Bull Br Mus Nat Hist Geol Suppl 10: 1-222., Taverne 1974TAVERNE L. 1974. L’ostéologie d’Elops Linné, C., 1766 (Pisces: Elopiformes) et son intérêt phylogénétique. Acad R Belg Mém Cl Sci 41: 1-96., Arratia 1997ARRATIA G. 1997. Basal teleosts and teleostean phylogeny. Paleo Ichthyologica 7: 5-168. ). Also, this species was used for rooting the tree. We included data from the living clupeiforms Denticeps clupeoides (fideGreenwood 1968GREENWOOD PH. 1968. The osteology and relationships of the Denticipitidae, a family of clupeomorph fishes. Bull Br Mus Nat Hist Zool 16: 215-273. , Di Dario and De Pinna 2006), Odaxothrissa vitatta (fideGrande 1985GRANDE L. 1985. Recent and fossil Clupeomorph fishes with material for revision of the subgroups of Clupeoids. Bull Am Mus Nat Hist 181: 231-372.), and Chirocentrus dorab (fideCavender 1966CAVENDER T. 1966. The caudal skeleton of the Cretaceous teleosts Xiphactinus, Ichthyodectes, and Gillicus, its bearing on their relationship with Chirocentrus. Occas Pap Mus Zool Univ Mich 650(29): 1-14., Grande 1985, Di Dario 2009). In addition, we included the enigmatic and primitive †Ornategulum sardinioides with data taken of Forey (1973a). A list of references used for taxa and discussion, coding and changing of characters are all taken of Alvarado-Ortega et al. (2008) with corrections from Murray and Wilson (2013MURRAY AM and WILSON MVH. 2013. Two new paraclupeid fishes (Clupeomorpha: Ellimmichthyiformes) from the Upper Cretaceous of Morocco. In: Arratia G et al. (Eds), Mesozoic Fishes 5 - Global Diversity and Evolution, München: Verlag Dr. Pfeil, Germany, p. 267-290.).
ANATOMICAL ABBREVIATIONS
We used r and l before anatomical abbreviations to indicate right and left side, respectively. AA, angulo-articular; ACH, anterior ceratohyal; ANT, antorbital; ASPH, autosphenotic; ber. f, beryciform foramen; BR, branchiostegal ray (numbered); CL, cleithrum; COR, coracoid; D, dentary; d.cd.sc, dorsal caudal scute; DH, dorsal hypohyal; ECPT, ectopterygoid; ENPT, endopterygoid; EP, epural (numbered); EPN, epineural; EPO, epioccipital; f.r, fin rays; FR, frontal; H, hypural; HM, hyomandibula; HV, ventral hypohyal; IH, inter-hyal; IO, infraorbital (numbered); io. c, infraorbital sensory canal; l.l.c, lateral sensory line canal; l. pr, lateral process; LET, lateral ethmoid; md. c, mandibular sensory canal; MES, mesethmoid; MPT, metapterygoid; MX, maxilla; n.a. U1, neural arch of the first ural centrum; NA, nasal; n.s, neural spine; n.sp. PU1, neural spine of first preural centrum; o.otcn, opening of otic sensory canal; OP, opercle; op. pr. HM, opercular process of hyomandibula; otcn, otic sensory canal; PA, parietal; PAL, palatine; PAS, parasphenoid; PCH, posterior ceratohyal; PCL, postcleithrum; PH, parhypural; PMX, premaxilla; POP, preopercle; pl. r, pleural rib; pop. c, preopercular sensory canal; pr.r, precurrent ray (numbered); PTG, proximal pterygiophore (numbered); PTM, posttemporal; PTO, pterotic; PU, preural centrum; Q, quadrate; RAR, retroarticular; rd, radial; S, symplectic; SCA, scapula; sca. f; scapular foramen; SCL, supracleithrum; SMX, supramaxilla (numbered); SN, supraneural (numbered); SORB, supraorbital; sorb. c, supraorbital sensory canal; st. c, supratemporal commissural sensory canal; U, ural centrum; UN, uroneural; VC, vertebral centrum; VH, ventral hypohyal; v.cd.sc, ventral caudal scute.
MEASUREMENTS
BD, body depth (measured as the greatest vertical distance of the body); HD, head depth (vertical distance taken at the mid-line of occiput to ventral border of head); HL, head length (measured as the horizontal distance between tip of premaxilla and posterior limit of opercle); PAL, preanal length (taken from the anteriormost point of the head to the origin of anal fin); PDL, predorsal length (distance between anterior end of the head to the origin of dorsal fin); PVL, prepelvic length (taken from the anterior end of head and the origin of pelvic fin); SL, standard length (distance between anterior end of head to the posteriormost end of hypural); TL, total length (distance between anterior end of head to the posteriormost fin ray of caudal fin).
INSTITUTIONAL ABBREVIATIONS
AMNH - Department of Vertebrate Paleontology, American Museum of Natural History, New York.
AO.UERJ - Vertebrate collection (fish dry skeletons) of the Departamento de Zoologia in Universidade do Estado do Rio de Janeiro.
BMNH - Department of Paleontology, Natural History Museum (formely British Museum of Natural History), London.
D.UERJ - Cleared and stained vertebrate collection of the Departamento de Zoologia of Universidade do Estado do Rio de Janeiro.
DGM-P - Museu de Ciências da Terra, Divisão de Geologia e Mineralogia, Departamento Nacional de Produção Mineral, Rio de Janeiro.
MN-V - Paleovertebrate Collection of the Museu Nacional, Rio de Janeiro.
Pz.UERJ - Paleozoological Collection of Departamento de Zoologia of Universidade do Estado do Rio de Janeiro.
SYSTEMATIC PALEONTOLOGY
Subdivision TELEOSTEI Müller, 1846
Subcoort OTOCEPHALA Johnson and Patterson, 1996
Superorder CLUPEOMORPHA Greenwood et al., 1966
Order †ELLIMMICHTHYIFORMES Grande, 1982GRANDE L. 1982. A revision the fossil genus Diplomystus, with comments on the interrelationships of clupeomorph fishes. Am Mus Novit 2728: 1-34.
Family †PARACLUPEIDAE Chang and Chou, 1977
Genus †ScutatuspinosusSantos and Corrêa, 1985SANTOS RS and CORRÊA VLS. 1985. Contribuição ao conhecimento da paleoictiofaúnula do Cretáceo do Brasil. In: Campos DA et al. (Orgs), Coletânea de Trabalhos Paleontológicos, Brasília: Ministério das Minas e Energia e Departamento Nacional da Produção Mineral, p. 169-174.
Revised Diagnosis. Torpedo-like †paraclupeid fish reaching 68 mm TL, with the following unique combination of features: head length contained about four times in TL; reduced orbit (*), in which diameter equals one fourth of HL; parietals joined medially, not separated by supraoccipital; toothless parasphenoid; minute conical teeth in a single row on jaws; elongate and straight maxilla with prominent cranial process (*); two small supramaxillae backwardly positioned on the posterior half of the maxilla (*); quadrate-mandibular articulation in the midpoint of the orbit; complete circumorbital series, with expanded laminate infraorbital bones; supraorbital and antorbital bones present; posttemporal bone with long and subrectangular anterodorsal process and membranous outgrowth of crenulated border; hyomandibula with a single expanded articular head; large L-shaped preopercular bone with preopercular sensory canal giving off atrophied tubules; large and subrectangular opercle ornamented with parallel striations on lower half; origin of the dorsal fin a little behind the middle point of SL; six-rayed pelvic fin anterior to dorsal fin; S-like cleithrum; postcleithra absent; 10 predorsal scutes slightly ornamented with straight radiating striations, each one bearing a median keel finishing in a posterior spine-like process; anteriormost predorsal scutes ellipsoid and posteriormost ones heart-shaped; 10 long and slender supraneural bones; short-based and posteriorly positioned anal fin; 17 prepelvic ventral scutes with rounded dorsal border (*); eight squarish postpelvic scutes bearing pungent posterior process (*); 32 vertebrae, 19 abdominal and 13 caudal; strongly keeled pleural ribs inserted in excavations of abdominal centra and thin posteriormost ones on parapophyses; six hypurals; three epurals; three uroneurals, the first one reaching the first preural centrum; parhypural fused to the first preural centrum; long equal-sized neural spine of the first and second preural centra; sharp and short neural spine of the first preural centrum; first hypural attached to first ural centrum; third hypural hypertrophied; caudal diastema absent; ural and preural centra almost equal-sized.
(*) denotes uniquely derived features for †Ellimmichthyiformes.
Content. Only one species known, that is †Scutatuspinosus itapagipensis.
Remarks. We note some disagreements regarding the original diagnosis of †Scutatuspinosus by Santos and Corrêa (1985SANTOS RS and CORRÊA VLS. 1985. Contribuição ao conhecimento da paleoictiofaúnula do Cretáceo do Brasil. In: Campos DA et al. (Orgs), Coletânea de Trabalhos Paleontológicos, Brasília: Ministério das Minas e Energia e Departamento Nacional da Produção Mineral, p. 169-174.). For instance, they pointed out a count of conical teeth on premaxilla (n=6), maxilla (n=28) and dentary (n=38) as relevant taxonomic characters. Apparently, these counts were taken from the type specimen because the quantity of teeth varies so much in examined specimens. Unlike these authors, we assumed these counts were not relevant characters due to high individual variability. Furthermore, although Santos and Corrêa (1985) claimed the presence of the lateroparietal condition of skull roof, we agree with most recent authors (e.g., Alvarado-Ortega et al. 2008, Murray and Wilson 2013MURRAY AM and WILSON MVH. 2013. Two new paraclupeid fishes (Clupeomorpha: Ellimmichthyiformes) from the Upper Cretaceous of Morocco. In: Arratia G et al. (Eds), Mesozoic Fishes 5 - Global Diversity and Evolution, München: Verlag Dr. Pfeil, Germany, p. 267-290., Figueiredo and Ribeiro 2016FIGUEIREDO FJ and RIBEIRO DRM. 2016. Relationships of †Codoichthys carnavalii Santos, 1994 (Teleostei, Clupeomorpha, †Ellimmichthyiformes) from the late Aptian of São Luís-Grajaú basin, NE Brazil. An Acad Bras Cienc 88: 1277-1307.) who noted the parietals medially in contact. This primitive condition is shared with all †Elimmichthyiformes. Regarding the caudal skeleton, Santos and Corrêa (1985) indicated the presence of six hypurals, all autogenous. Rather, we note the second hypural is fused to the first ural centrum as in other †Ellimmichthyiformes and advanced Clupeiformes.
† Scutatuspinous itapagipensisSantos and Corrêa, 1985SANTOS RS and CORRÊA VLS. 1985. Contribuição ao conhecimento da paleoictiofaúnula do Cretáceo do Brasil. In: Campos DA et al. (Orgs), Coletânea de Trabalhos Paleontológicos, Brasília: Ministério das Minas e Energia e Departamento Nacional da Produção Mineral, p. 169-174.
(Figs. 1-11, Tables I and II)
Synonyms. None.
Diagnosis. As for genus (monotypic).
Types. Holotype (DGM 1164-P, fish complete and articulated); Paratypes: DGM 1165-P to 1176-P (formerly DGM 540-P to 552-P); DGM 1207-P to1264-P (formerly DGM 553-P to 610-P).
Locality and geological time. Península de Itapagipe, Bairro do Bonfim; Salvador; Marfim Formation (Ilhas Group); Neocomian (?Hauterivian) of Recôncavo Basin.
Remarks. Many specimens are preserved closely associated in the same bedding planes suggesting that †Scutatuspinosus was a schooling fish (Santos and Corrêa 1985SANTOS RS and CORRÊA VLS. 1985. Contribuição ao conhecimento da paleoictiofaúnula do Cretáceo do Brasil. In: Campos DA et al. (Orgs), Coletânea de Trabalhos Paleontológicos, Brasília: Ministério das Minas e Energia e Departamento Nacional da Produção Mineral, p. 169-174., p. 170). Also, all specimens are complete and articulated indicating death in a lentic environment. There is no evidence of postmortem scavenger activity. A lower jaw slightly projecting, minute gnathal teeth, and shallow Meckelian fossa indicate that †Scutatuspinosus was an epipelagic filter-feeding fish.
DESCRIPTIVE MORPHOLOGY
Overall form of the body. †Scutatuspinosus itapagipensis has a torpedo-like body (Figs. 1a and b). All examined specimens are preserved in lateral view in the bedding planes indicating a laterally compressed body in life. The HL corresponds to 30-36% of SL. The predorsal profile and ventral outline are slightly convex. The BD is 29-32% SL and ratio of BD/SL ranges from 0.25 to 0.32. The PDL is 56-63% SL. The dorsal fin is short and placed slightly behind the midpoint of the body, posterior to vertical line crossing the origin of the pelvic fin. The dorsal fin depth is almost equal to the fin base length. The PVL is 48-51% SL and the ratio of PVL/SL ranges from 0.43 to 0.5. The anal fin is short-based. The PAL is 74-83% SL. The caudal fin is forked, deeply notched, with upper and lower lobes almost equal-sized. The dorsal surface of the body shows an excess of dark pigment whereas the ventral one lacks it. Apparently, it is not a carbon film from the tissues. Rather, it corresponds to an obliterative counter-shading camouflage strategy in life as seen in many hodiernal sardines and anchovies, and other epipelagic fishes.
†Scutatuspinosus itapagipensisSantos and Corrêa 1985SANTOS RS and CORRÊA VLS. 1985. Contribuição ao conhecimento da paleoictiofaúnula do Cretáceo do Brasil. In: Campos DA et al. (Orgs), Coletânea de Trabalhos Paleontológicos, Brasília: Ministério das Minas e Energia e Departamento Nacional da Produção Mineral, p. 169-174.: (a) photograph of the holotype (DGM 1164-P); (b) restoration of skeleton mainly based on DGM 1164-P.
Cranial bones. The skull roof of †Scutatuspinosus is moderately elongate and most of dermal bones are smooth, except for the contacting region between frontal and parietal (Figs. 2b, 3-5) in which the ornamentation is noted as a form of radiating and fading striations. It differs from that coarse and heavy sculptured skull roof found in †Ellimmichthys (Grande 1982GRANDE L. 1982. A revision the fossil genus Diplomystus, with comments on the interrelationships of clupeomorph fishes. Am Mus Novit 2728: 1-34.) or adults of †Ellimma (Chang and Maisey 2003CHANG MM and MAISEY JG. 2003. Redescription of †Ellimma branneri and †Diplomystus shengliensis, and relationships of some basal clupeomorphs. Am Mus Novit 3404: 1-35. ). Fontanels, fossae, and a recessus lateralis, usually present in clupeiform fishes, are lacking. Unfortunately, the skull in most of specimens is compressed and crushed, and dermal bones are mostly eroded and damaged. This has caused considerable difficulty for accurate reconstruction of the skeleton. The endocranium is badly preserved or completely hidden by dermal bones in all specimens examined.
†Scutatuspinosus itapagipensisSantos and Corrêa 1985SANTOS RS and CORRÊA VLS. 1985. Contribuição ao conhecimento da paleoictiofaúnula do Cretáceo do Brasil. In: Campos DA et al. (Orgs), Coletânea de Trabalhos Paleontológicos, Brasília: Ministério das Minas e Energia e Departamento Nacional da Produção Mineral, p. 169-174.: (a) head in left lateral view (DGM 1164-P); (b) interpretative line drawing of DGM 1164-P.
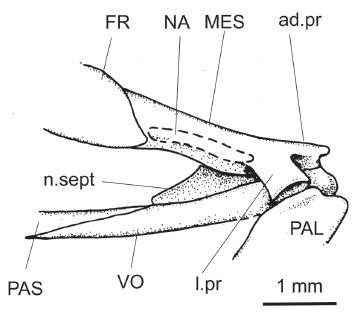
†Scutatuspinosus itapagipensisSantos and Corrêa 1985SANTOS RS and CORRÊA VLS. 1985. Contribuição ao conhecimento da paleoictiofaúnula do Cretáceo do Brasil. In: Campos DA et al. (Orgs), Coletânea de Trabalhos Paleontológicos, Brasília: Ministério das Minas e Energia e Departamento Nacional da Produção Mineral, p. 169-174.: restoration of ethmoidal region based on DGM 1261-P.
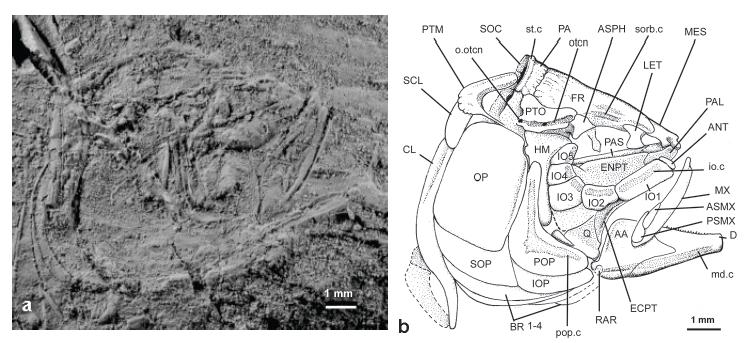
†Scutatuspinosus itapagipensisSantos and Corrêa 1985SANTOS RS and CORRÊA VLS. 1985. Contribuição ao conhecimento da paleoictiofaúnula do Cretáceo do Brasil. In: Campos DA et al. (Orgs), Coletânea de Trabalhos Paleontológicos, Brasília: Ministério das Minas e Energia e Departamento Nacional da Produção Mineral, p. 169-174.: (a) photograph of DGM 1253-P; (b) interpretative line-drawing of skull and pectoral girdle of DGM 1253-P.
†Scutatuspinosus itapagipensisSantos and Corrêa 1985SANTOS RS and CORRÊA VLS. 1985. Contribuição ao conhecimento da paleoictiofaúnula do Cretáceo do Brasil. In: Campos DA et al. (Orgs), Coletânea de Trabalhos Paleontológicos, Brasília: Ministério das Minas e Energia e Departamento Nacional da Produção Mineral, p. 169-174.: (a) photograph of DGM 1255-P; (b) interpretative line-drawing of skull and pectoral girdle of DGM 1255-P.
As far as can be seen, the mesethmoid (Figs. 2b, 3-5, MES) is an elongate and low bone corresponding to ¼ of the frontal length. In all examined specimens it is either poorly preserved or broken, particularly due to its flimsy condition. There is a reduced median anterodorsal process (Fig. 3, ad.pr) bearing an adjacent small surface for receiving the ascending process of premaxilla. Also, there are two deep lateral excavations flanking this process for accommodating the rounded articular head of maxilla.
The ventral surface of the mesethmoid extends back to produces the median bony keel of the nasal septum (Fig. 3, n.sept). It is thin and probably completed posteriorly with cartilage in life. There is a short and cup-like lateral process (Fig. 3, l. pr) inclined antero-ventrally for receiving the reduced anterodorsal condyle of the palatine bone. Posteriorly, the mesethmoid is partially covered by the anteriormost portion of the frontal.
The nasal (Fig. 3, NA) is a short tube-like bone practically reduced to its neurodermal component. It lies on the underlying dorsolateral surface of the mesethmoid and receives the anterior extension of the supraorbital sensory canal.
The lateral ethmoid (Figs. 2b, 4 and 5, LET) is a small subrectangular shield of perichondral bone that contributes to the anterodorsal limit of the orbit, but not reaching the parasphenoid ventrally. Between mesethmoid and lateral ethmoid there is a great space probably filled with cartilage in life. The lateral border of the lateral ethmoid produces an oblique prominent crest for the first infraorbital bone.
The frontal (Figs. 2b, 3-5, FR) is by far the largest bone of the skull roof, contributing with about 2/3 HL, and covering most of the orbit and ethmoidal regions. The anteriormost portion of this bone above the orbit is narrow but at the level of otic region it expands laterally a little, thus reaching its greatest width. In some specimens (e.g., DGM 1255-P, Fig. 5) there is along the medial meeting of frontals, just above the orbit, a weak transverse bony ridge apparently separating low anterior and posterior grooves for the supraorbital sensory canal as in the well-preserved skull of †Diplomystus from the Upper Cretaceous (Cenomanian) of the English Chalk (Forey 2004FOREY PL. 2004. A three-dimensional skull of a primitive clupeomorph from the English Chalk and implications for the evolution of the clupeomorph acusticolateralis system. In: Arratia G and Tintori A (Eds), Mesozoic Fishes 3 - Systematics, Paleoenvironments and Biodiversity, München: Verlag Dr. Friedrich Pfeil, Germany , p. 405-427.).
The posterior border of the frontal is ornamented with fine striations fading out at the level of the orbit. The frontal meets the parietal through a crenulated and arched suture.
The supraorbital sensory canal (Figs. 2b and 4, sorb.c) runs its trajectory on the frontal mainly enclosed in bone, hardly visible on the surface except for uncovered bony sites of selected specimens. It opens on surface in a deep and elongate groove anterior to the dilatator fossa but any additional opening is absent. In some specimens this canal seems to produce an obscure branch towards parietal.
The parietal (Figs. 2b, 4 and 5, PA) is crescent-shaped and transversely elongate. Each bone meets its antimere in the midline anterior to supraoccipital. Its surface is ornamented with almost straight striations radiating from the growth center. A supratemporal commissural sensory canal (Figs. 2b, 4 and 5, st. c) pierces the bone in an arched crest on the posterior border. Behind the commissure there is a deep parietal excavation that extends posteriorly through the neighboring epioccipital and supraoccipital. As far as is known, such an excavation has only been described by Forey (2004FOREY PL. 2004. A three-dimensional skull of a primitive clupeomorph from the English Chalk and implications for the evolution of the clupeomorph acusticolateralis system. In: Arratia G and Tintori A (Eds), Mesozoic Fishes 3 - Systematics, Paleoenvironments and Biodiversity, München: Verlag Dr. Friedrich Pfeil, Germany , p. 405-427.) for †Diplomystus, †Triplomystus, and †Armigatus.
The supraoccipital (Figs. 2b, 4 and 5, SOC) is a small triangular bone in lateral view. It is placed behind the parietals and is articulated to each epioccipital lateroventrally. It bears a low and short occipital crest posteriorly. We are unable to confirm the presence of an associated pre-epioccipital fossa.
As usual for clupeomorphs, the oto-occipital region is shorter than the orbit and ethmoidal regions. Unfortunately, the orbitosphenoid and pterosphenoid are not preserved well enough to permit a description in any specimen observed.
The autosphenotic (Figs. 4 and 5, ASPH) is a subtriangular bone in lateral view that produces a moderate postorbital spine-like process a little inclined lateroventrally. This bone meets the frontal dorsally, the pterotic posterodorsally, and the prootic (not preserved) ventrally. It contributes to the facet for the anterior half of the articular head of the hyomandibula.
The pterotic (Figs. 2b, 4 and 5, PTO) is a large trapezoid bone. Posteriorly it produces a very short and rounded spine-like process. Most of lateral wall of this bone contributes to the articular facet for the hyomandibula. Together with autosphenotic and part of frontal, this bone forms a shallow and unroofed dilatator fossa. The otic sensory canal (Fig. 4, otcn) runs within a straight tube along most of the dorsolateral border of the bone; its posterior end opens in a minute pore (Fig. 4, o.otcn). Also, at the level of the hyomandibula and opercle there is a minute pore for the preopercular sensory canal. Posterior to the pterotic there is a deep excavation probably corresponding to a posttemporal fossa. Between the pterotic and posttemporal bones of most of the specimens examined there is a regular space that we think this was occupied by a laminate lateral extrascapular (not preserved).
Noteworthy, the outline of the pterotic is very similar to that of †Armigatus (cf.Patterson 1967PATTERSON C. 1967. Are the teleosts a polyphyletic group? In: Problémes actuales de paleontology (evolution des vertébrès). Colloques int CNRS, Paris 163: 93-109.) and †Triplomystus (cf.Forey et al. 2003FOREY PL, YI L, PATTERSON C and DAVIES CE. 2003. Fossil Fishes from the Cenomanian (Upper Cretaceous) of Namoura, Lebanon. J Syst Palaeontol 1(4): 227-330. ) in which a lateral dilation of the otic sensory canal and consequently formation of recessus lateralis are lacking.
The parasphenoid (Figs. 2b, 3-5, PAS) is an elongate shafted bone that crosses the orbit in the ventral midline. Anteriorly, this bone meets the vomer at the level of the lateral ethmoid but its end is unknown. Teeth are lacking. We are unable to confirm the presence of a basipterygoid process, an anatomical feature commonly found in other †Ellimmichthyiformes.
The vomer (Fig. 3, VO) is an elongate, well-ossified, and toothless bone. Its anterior end is expanded and rounded. It is placed in the basicranium forming most of the anterior floor of ethmoidal region. This bone tapers backwardly and meets parasphenoid anterior to lateral ethmoid.
Circumorbital bones and eye-ball. Sclerotic bones are poorly preserved so that only uninformative fragments remain; probably they were small or not ossified. The circumorbital bones are very thin and bear expanded membranous components whose limits are difficult to determine in most of the specimens examined. Often, they are damaged or loss. Posterior to the orbit the infraorbital bones completely covers the cheek. The orbit is relatively small, corresponding about ¼ HL. The infraorbital sensory canal runs through the infraorbital bones in a simple bony tube near orbit rim without giving off secondary tubules.
A small tube-like antorbital (Fig. 4, ANT) is seen in DGM 1253-P abutting against the anterior margin of the lachrymal. Notably, its presence among †Ellimmichthyiformes is unusual and that of †Scutatuspinosus lacks a sensory canal as commonly found in modern Clupeiformes.
The first infraorbital or lachrymal (Fig. 4, IO1) is a large laminate and trapezoid bone that covers most of the palatine bone laterally. The second infraorbital bone (Figs. 2b and 4, IO2) is short and subrectangular. It is placed upon the anteroventral rim of the orbit in opposite to the supraorbital bone. The infraorbital sensory canal crosses the bone in an elongate tubule at the midline.
The third infraorbital (Fig. 3, IO3) is trapezoid and the largest from the circumorbital series. It is placed at the posteroventral corner of the orbit and its anterodorsal border followed the curvature of the eye ball. The posteroventral border expands a little as a lamina ventrally that covers most of the ventral process of the hyomandibula and metapterygoid.
The fourth and fifth infraorbital bones (Fig. 3, IO4 and IO5) are very thin and flimsy bones, rarely preserved. They are subrectangular, longer antero-posteriorly than dorso-ventrally, and expanded posteriorly to cover most of cheek at the level of the hyomandibula.
The dermosphenotic is preserved in DGM 1262-P (not figured). It is a small and sickle-shaped bone, as in †Armigatus (see Patterson 1967PATTERSON C. 1967. Are the teleosts a polyphyletic group? In: Problémes actuales de paleontology (evolution des vertébrès). Colloques int CNRS, Paris 163: 93-109.), that covers most of the anterodorsal border of the autosphenotic at the level of the shallow dilatator fossa.
The supraorbital (Fig. 5, SORB) is a short, elongate, and fusiform bone lying on the lateral border of the frontal in the anterior half of the orbit, above the lateral ethmoid. Although originally considered absent in †ellimmichthyiform fishes, this anamestic bone is present in †Ellimma branneri (cf.Chang and Maisey 2003CHANG MM and MAISEY JG. 2003. Redescription of †Ellimma branneri and †Diplomystus shengliensis, and relationships of some basal clupeomorphs. Am Mus Novit 3404: 1-35. , fig. 2a) and †Rhombichthys intoccabilis (Khalloufi et al. 2010KHALLOUFI B, ZARAGÜETA-BAGILS R and LELIÈVRE H. 2010. Rhombichthys intoccabilis, gen. et sp. nov. (Ellimmichthyiformes, Clupeomorpha, Teleostei), from the Cenomanian (Upper Cretaceous) of Ein Yabrud, Middle East: anatomical description and phylogenetic implications. J Vert Paleontol 30: 57-67.).
Upper jaw. It contains premaxilla, maxilla and two supramaxillae. The premaxilla (Figs. 2b and 5, PMX) is a short, elongate, and triangular bone. It corresponds to a third of maxilla length. The medial portion of this bone forms a low ascending process while the main body tapers backwards in a sharp posterior process. There is a single row of minute conical teeth along oral border. Santos and Corrêa (1985SANTOS RS and CORRÊA VLS. 1985. Contribuição ao conhecimento da paleoictiofaúnula do Cretáceo do Brasil. In: Campos DA et al. (Orgs), Coletânea de Trabalhos Paleontológicos, Brasília: Ministério das Minas e Energia e Departamento Nacional da Produção Mineral, p. 169-174.) counted six teeth, probably in the holotype, but the number is variable in other specimens, we counted from six up to ten or even more.
The maxilla (Figs. 2b, 4 and 5, MX) is a shafted, massive and elongate bone, almost straight. Although superficially very similar to that of †Santanaclupea silvasantosi (see Maisey 1993MAISEY JG. 1993. A new clupeomorph fish from the Santana Formation (Albian) of NE Brazil. Am Mus Novit 3076: 1-15.), it does not extend posteriorly beyond the posterior limit of the orbit or quadrate. As in the premaxilla, the oral border is garnished of a single row of minute conical teeth. Anteriorly, the maxilla produces an elongate and digit-shaped cranial process (better seen in DGM 1228-P) similar to that of Elops.
Two small and equal-sized ipsilateral supramaxillae are positioned posteriorly on the dorsal blade of the posterior half of the maxilla. The anterior supramaxilla (Figs. 4 and 5, ASMX) is elongate, fusiform and smooth. The posterior supramaxilla (Figs. 4 and 5, PSMX) is a low drop-shaped bone, with a long and sharp anterodorsal process covering most of the dorsal border of the anterior supramaxilla. In some specimens there is a weak keel crossing obliquely the main body of the bone.
Lower jaw. The lower jaw is moderately deep and elongate, very compressed laterally. In lateral view it is composed of dentary, anguloarticular, and retroarticular. From the symphysis backwards the lower jaw ascends gradually to produce a moderate coronoid process on its posterior third. The lower jaw reaches the middle of the orbit.
The dentary (Figs. 2b, 4 and 5, D) occupies about ¾ of the lower jaw length. It is a triangular and elongate bone. The symphyseal margin is truncated and low. Posteriorly the bone has a deep incisura dividing the main body into upper and lower limbs. The ventral border of the bone is a little incurved. The presence of a shallow Meckelian fossa suggests a low volume for the adductor mandibulae muscle. There is a single row of minute conical teeth on the oral border anterior to the coronoid process. The mandibular sensory canal (Figs. 2b and 4, md.c) runs bone-enclosed in a strong straight tubule. Pores are not clear on the surface.
The anguloarticular (Figs. 4 and 5, AA) is coarsely triangular in lateral view. Its strengthened superior crest grows up gradually to form a moderate coronoid process. Its anterior process is short and obtuse in lateral view, fitting between the two limbs of the dentary. The prearticular fossa is very shallow. The mandibular sensory canal runs along the bone enclosed in a short tubule. A pore from this canal is seen externally beneath articular facet for quadrate. The post-articular process is short and digit-like.
Near the posterior corner of the angulo-articular, there is a short and subrectangular retroarticular (Figs. 4 and 5, RAR) lodged in a ventral recess.
Suspensorium. The articulation between the lower jaw and the quadrate lies beneath the midpoint of the orbit. The hyomandibula (Figs. 4 and 5, HM) is a large axe-shaped bone with ventral shaft vertically positioned. There is a broad, single, articulatory head for autosphenotic and pterotic bones. An expanded anterior membranous process abuts against the posterior end of endopterygoid and partially covers the metapterygoid. The opercular process is short and rounded, and placed posterodorsally.
The toothless palatine (Figs. 3-5, PAL) is an enlarged and quadrangular bone. It seems to be a complete chondral bone, without any dermal component. There is a reduced anterodorsal process for meeting the maxilla and a small facet for the lateral process of the mesethmoid and vomer anterodorsally.
The endopterygoid (Figs. 2b, 4 and 5, ENPT) is an elliptical shield-like bone forming most of the orbit floor. Its posterior half is deeply excavated. Apparently teeth are lacking on the medial surface because sockets for teeth are absent.
The ectopterygoid (Figs. 2b, 4 and 5, ECPT) is an edentulous boomerang-shaped bone. Anteriorly, the ectopterygoid meets the palatine through an oblique suture. At the midline and along all extension of the bone there is a well-developed lateral crest. Posterodorsally, this bone produces a well-developed flange and its sharp posteroventral process abuts firmly against the anterodorsal border of the quadrate.
The metapterygoid (Fig. 5, MPT) is a large trapezoidal bone. It is firmly attached to the laminate anteroventral process of the hyomandibula. It is placed far from the parasphenoid so that a medial process for meeting basipterygoid process is apparently lacking. Its ventral border is almost straight and meets the quadrate with intervening cartilage. Its anterior border lies on the hind edge of the ectopterygoid.
The quadrate (Figs. 2b, 4-6, Q) is the usual triangular bone bearing a stout articular head a little inclined forwards. The posteroventral process is short, sharp and slightly inclined, and the posterodorsal notch for lodging the short and club-like symplectic (Figs. 2b and 5) is somewhat shallow.
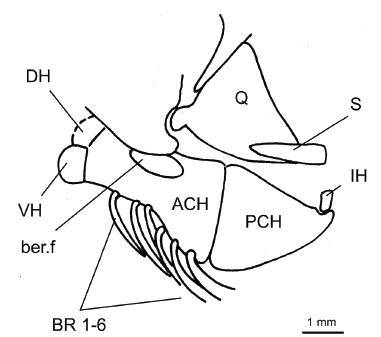
†Scutatuspinosus itapagipensisSantos and Corrêa 1985SANTOS RS and CORRÊA VLS. 1985. Contribuição ao conhecimento da paleoictiofaúnula do Cretáceo do Brasil. In: Campos DA et al. (Orgs), Coletânea de Trabalhos Paleontológicos, Brasília: Ministério das Minas e Energia e Departamento Nacional da Produção Mineral, p. 169-174.: hyoidean bones and associated structures of DGM 1262-P.
Hyoidean bones. The anterior ceratohyal (Fig. 6, ACH) is short, deep and subrectangular. There is a moderate elliptical beryciform foramen (Fig. 6, ber.f) near slender dorsal border of the bone. A subtriangular posterior ceratohyal (Fig. 6, PCH) meets the anterior ceratohyal in a straight interspace of cartilage. At the posterodorsal corner there is a shallow notch for lodging a nodular interhyal (Fig. 6, IH).
There are 10 branchiostegal rays (Figs. 2b, 4-6, BR). The six anteriormost ones are very slender, acinaciform, and have an enlarged head proximally. They are attached to the ventral border of the anterior ceratohyal. The remaining branchiostegals are stronger, falcate and long. They lie in a series below the subopercle attached to the ventral border of the posterior ceratohyal.
The dorsal and ventral hypohyals (Fig. 6, DH and VH) are small, spleen-shaped and subequal-sized. They are attached to smooth and obtuse surface of the anterior ceratohyal.
Santos and Corrêa (1985SANTOS RS and CORRÊA VLS. 1985. Contribuição ao conhecimento da paleoictiofaúnula do Cretáceo do Brasil. In: Campos DA et al. (Orgs), Coletânea de Trabalhos Paleontológicos, Brasília: Ministério das Minas e Energia e Departamento Nacional da Produção Mineral, p. 169-174.) originally indicated the presence of an elongate and leaf-like urohyal in the DGM 541-P and we confirm it.
Opercular series. The opercular series is complete, composed of opercle, subopercle, interopercle and preopercle.
The preopercle (Figs. 2b, 4 and 5, POP) is L-shaped with dorsal and ventral branches forming an almost right angle in the confluence of which there is a small membranous outgrowth. The dorsal limb is slightly larger than the ventral. It shows a well-developed posterior membranous outgrowth. The ventral margin is large and slightly convex. The medial laminate process for meeting the quadrate is short and the contact between both bones is not firm.
The preopercular sensory canal (Figs. 2b, 4 and 5, pop. c) crosses the bone in a bony tubule close to the anterior margin and gives off four to five secondary very atrophied tubules.
The opercle (Figs. 2b, 4 and 5, OP) is large and subrectangular. Its anterior border is strengthened by a vertical crest. The lower half of the bone is slightly ornamented with parallel striation as in adults of †Ellimma branneri (see Chang and Maisey 2003CHANG MM and MAISEY JG. 2003. Redescription of †Ellimma branneri and †Diplomystus shengliensis, and relationships of some basal clupeomorphs. Am Mus Novit 3404: 1-35. ). The articular facet for the hyomandibula is placed near the anterodorsal corner. It is cup-shaped and extends backwards in a longitudinal flange on the inner surface of the bone.
The subopercle (Figs. 2b, 4 and 5, SOP) is low, elongate and subrectangular. Its major depth is of about a fifth that of the opercle. Anterodorsally, this bone bears a reduced digit-like process abutting the anteroventral corner of the opercle.
The interopercle (Figs. 2b and 4, IOP) is an elongate triangular bone mostly hidden by the ventral margin of the preopercle. It is a little longer than the preopercle and its posterior end abuts firmly against the subopercle.
Pectoral girdle and fin. The post-temporal (Figs. 2b, 4-5, 7 and 8b, PTM) is a large bone in comparison with that of ordinary clupeomorphs. In lateral view, it has two long limbs. The dorsal limb, corresponding to an anterodorsal process for contacting the epioccipital, is long, subrectangular, with a rounded anterior end. The trajectory of the lateral line on the main body of the post-temporal bone is unclear. The thin ventral limb for articulation with the intercalar is elongate and extends antero-ventrally. It is slightly shorter than the dorsal arm. In the confluence of the limbs, the main body of the bone is flatted and expanded, bearing an obscure tubule for the lateral sensory canal. The posterior margin of the bone is crenulated as in some species of †Armigatus (see Forey et al. 2003FOREY PL, YI L, PATTERSON C and DAVIES CE. 2003. Fossil Fishes from the Cenomanian (Upper Cretaceous) of Namoura, Lebanon. J Syst Palaeontol 1(4): 227-330. ).
†Scutatuspinosus itapagipensisSantos and Corrêa 1985SANTOS RS and CORRÊA VLS. 1985. Contribuição ao conhecimento da paleoictiofaúnula do Cretáceo do Brasil. In: Campos DA et al. (Orgs), Coletânea de Trabalhos Paleontológicos, Brasília: Ministério das Minas e Energia e Departamento Nacional da Produção Mineral, p. 169-174.: pectoral girdle and fin of DGM 1261-P.
†Scutatuspinosus itapagipensisSantos and Corrêa 1985SANTOS RS and CORRÊA VLS. 1985. Contribuição ao conhecimento da paleoictiofaúnula do Cretáceo do Brasil. In: Campos DA et al. (Orgs), Coletânea de Trabalhos Paleontológicos, Brasília: Ministério das Minas e Energia e Departamento Nacional da Produção Mineral, p. 169-174.: (a) vertebral column and associated structures of DGM 1164-P; (b) pectoral, vertebral column, supraneural bones and associated structures of DGM 1268-P.
The supracleithrum (Figs. 2b, 4-5, 7, 8b, SCL) is short and spatulate. It is obliquely pierced by an inconspicuous bony tube for the lateral line (Fig. 7, l.l.c). It lies on the pointed end of the slender and sigmoid cleithrum (Figs. 4, 5, 7 and 8, CL). Whereas the dorsal limb of the cleithrum is long and slender, the ventral one is short and arched, producing an anterior convex margin that meets the opercular bones in the posterior wall of the branchial chamber.
The presence of a postcleithrum is uncertain, although Santos and Corrêa (1985SANTOS RS and CORRÊA VLS. 1985. Contribuição ao conhecimento da paleoictiofaúnula do Cretáceo do Brasil. In: Campos DA et al. (Orgs), Coletânea de Trabalhos Paleontológicos, Brasília: Ministério das Minas e Energia e Departamento Nacional da Produção Mineral, p. 169-174., fig. 2, p. 173) indicated the presence of a spatulate and narrow bony element attached to the cleithrum above the pectoral fin origin. All specimens that we examined lack this bone. Thus we interpreted that case as a posterior lamina of the cleithrum.
Of the endoskeletal girdle, the scapula (Fig. 7, SCA) is almost hidden by fin-rays or completely eroded in examined specimens. It was a small squarish bone bearing a large scapular foramen (Fig. 7, sca.f). An axillary process is lacking.
The large coracoid (Fig. 7, CO) is a laminate boot-shaped bone mirrored horizontally. It projects forwards to contact the anterior end of the cleithrum. There is a large fenestra between both bones. In most of the examined specimens the radials are hidden by leading fin-rays or are not preserved. We counted four baculiform proximal radials (Fig. 7, rd) and 12 slender and long associated fin-rays.
Vertebrae, ribs and intermuscular bones. The vertebral column (Figs. 8a, b) is composed of 32 preural vertebrae, of which 19 are abdominal. There are 17 pairs of pleural ribs with proximal articular heads inserted in deep cavities of the abdominal centra (Fig. 8b), unlike the two (or three) posteriormost ones that articulate with parapophyses. The remaining 13 vertebrae are from the caudal region.
Each vertebra is well-ossified and spool-shaped in lateral view. The outer surface is smooth and the notochordal pit very narrow. The neural arches are co-ossified to centra and those from the abdominal region bear long and bifid neural spines (Fig. 8b, n.sp). In the caudal region, both fuse to form a single element. The hemal spine of the eleventh preural vertebra is associated with the first pterygiophore of the anal fin.
The epineural intermuscular bones (Fig. 8b, EPN) are long, slender and arched. They are fused to the base of neural arch. Placed high in the flank of the anterior half of caudal region there is a framework of four to five detached and hypsiloid epineurals.
Each pleural rib (Fig. 8b, pl.r) is long, arched and shows a median crest along most of its length. Its proximal end is dilated. Each rib touches tip of ventral (=abdominal) scutes ventrally.
There are at least 10 slender, sigmoidal, and anteroventrally-posterodorsally inclined supraneurals (=predorsal bones), each one associated with a corresponding predorsal scute (Fig. 8b, SN).
Dorsal and anal fins and endoskeleton supports. The dorsal fin (Fig. 9a) originates at the interval between the sixteenth and seventeenth abdominal vertebrae, counted in cranio-caudal direction. It has a small first ray, unsegmented and not branched, followed by a long leading ray segmented, but not branched distally. The nine remaining ones are segmented and distally ramified (Fig 9a, f.r). They show dimerous pattern.
†Scutatuspinosus itapagipensisSantos and Corrêa 1985SANTOS RS and CORRÊA VLS. 1985. Contribuição ao conhecimento da paleoictiofaúnula do Cretáceo do Brasil. In: Campos DA et al. (Orgs), Coletânea de Trabalhos Paleontológicos, Brasília: Ministério das Minas e Energia e Departamento Nacional da Produção Mineral, p. 169-174.: (a) dorsal fin and endoskeletal support of DGM 1262-P; (b) anal fin and endoskeletal support of DGM 1254-P.
The dorsal fin is supported by 10 proximal pterygiophores (Fig. 9a, PTG). The first one is composed of two branches in an acute angle with the confluence united by a continuous laminate outgrowth. Other pterygiophores are elongate and pipe-like. Medial and distal pterygiophores are not preserved and a postdorsal spur is lacking too.
The anal fin (Fig. 9b) is short-based and is placed spanning across four vertebrae. It is composed of an unbranched and unsegmented fin-ray followed of one segmented but not distally branched. All seven remaining are distally branched and segmented (Fig. 9b, f.r). They are supported by eight slender and long proximal pterygiophores (Fig. 9b, PTG).
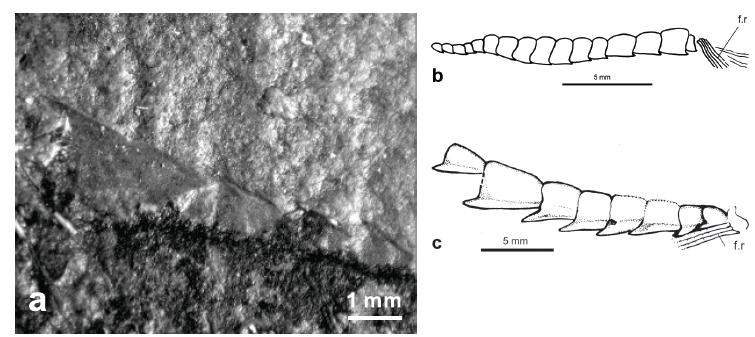
Pelvic girdle and fin. The pelvic bone is completely hidden by ventral scutes whereas the pelvic fin is small and composed of six fin-rays (Figs. 11b and c, f.r). It is placed a little behind a vertical line at the origin of the dorsal fin.
†Scutatuspinosus itapagipensisSantos and Corrêa 1985SANTOS RS and CORRÊA VLS. 1985. Contribuição ao conhecimento da paleoictiofaúnula do Cretáceo do Brasil. In: Campos DA et al. (Orgs), Coletânea de Trabalhos Paleontológicos, Brasília: Ministério das Minas e Energia e Departamento Nacional da Produção Mineral, p. 169-174.: (a) caudal skeleton of DGM 1253-P; (b) restoration of caudal skeleton based on DGM 1253-P. Arrows indicate leading upper and lower principal caudal rays.
†Scutatuspinosus itapagipensisSantos and Corrêa 1985SANTOS RS and CORRÊA VLS. 1985. Contribuição ao conhecimento da paleoictiofaúnula do Cretáceo do Brasil. In: Campos DA et al. (Orgs), Coletânea de Trabalhos Paleontológicos, Brasília: Ministério das Minas e Energia e Departamento Nacional da Produção Mineral, p. 169-174.: (a) posteriormost predorsal scutes of DGM 1164-P; (b) pre-pelvic scutes of DGM 1224-P; (c). Post-pelvic scutes of DGM 1164-P.
Caudal fin and endoskeleton support. The caudal fin is deeply forked and equal-lobed. It is supported by structures associated with three preural centra and two ural centra (Fig. 10). All neural and hemal arches of the preural vertebrae are fused to centra. Dorsal (Fig. 10, d.cd.sc) and ventral caudal scutes (Fig. 10, v.cd.sc) are small, elongate, and fusiform. They are positioned anterior to the leading precurrent rays.
There are eight dorsal and ventral precurrent rays (Fig. 10, pr.r). The anteriormost precurrent rays are unsegmented, whereas the posteriormost ones show segmentation. There follows 10 principal fin-rays in the upper lobe and nine in the lower lobe. The principal fin-rays show a dimerous pattern. A first branching occurs in the half of the length of the main ray followed of new branching more distally to produce ray tips.
The third preural centrum (Fig. 10, PU3) has long neural and hemal spines. The second one (Fig. 10, PU2) shows equally long dorsal and ventral spines that reach the precurrent rays.
The first preural centrum (Fig. 10, PU1) has an elongate and pointed neural spine (Fig. 10, n.sp. PU1). The parhypural (Fig. 10, PH) is attached closely to the ventral margin of the first hypural and its proximal end is fused to a first preural centrum. A parhypurapohysis is lacking.
The first ural centrum (Fig. 10, U1) is equal-sized to preural centra and bears a low, expanded and laminate neural arch.
The first hypural (Fig. 10, H1) is triangular, long and large. Its proximal end is attached to the base of the large and cylindrical first ural centrum. Also, the first hypural shows an oblique crest.
Between the proximal ends of the parhypural and first hypural there is a hypural foramen in some specimens (e.g., DGM 1250-P).
The second hypural (Fig. 10, H2) is long, slender, and low; its proximal end is co-ossified to first ural centrum.
The third hypural (Fig. 10, H3) is a broad triangular plate, very large, bearing a longitudinal keel. This bone extends below the main body axis occupying completely the region where a diastema is found in some other taxa. It bears the expanded proximal ends of the main central fin-rays. Its anterior end is short and acetabular, fitting to the surface of a bell-shaped second ural centrum (Fig. 10, U2).
The fourth and fifth hypurals (Fig. 10, H4 and H5) are subrectangular, low, and elongate; they are above the second ural centrum. Agreeing with Santos and Corrêa (1985SANTOS RS and CORRÊA VLS. 1985. Contribuição ao conhecimento da paleoictiofaúnula do Cretáceo do Brasil. In: Campos DA et al. (Orgs), Coletânea de Trabalhos Paleontológicos, Brasília: Ministério das Minas e Energia e Departamento Nacional da Produção Mineral, p. 169-174.) we noted a reduced sixth hypural (Fig. 10, H6) in DGM 1253-P.
There are three uroneurals; the first one (Fig. 10, UN1) is the longest of the set, reaching half of the first preural centrum anteriorly. It has a low anterior laminate process. Lying on the posterior half of the first uroneural and above the second ural centrum there is a second uroneural (Fig. 10, UN2).
The third uroneural (Fig. 10, UN3) is small and elliptical, and reaches the dorsal end of the second uroneural. It is well-preserved in DGM 1250-P.
We counted three long and slender epurals (Fig. 10, EP) occupying the area above the atrophied and laminate neural arch of the first preural centrum.
Predorsal and abdominal scutes. †Scutatuspinosus shows apparently a complete series of predorsal scutes comprising at least 10 elements (Fig. 11a). This series extends from the occiput to the origin of the dorsal fin. Anteriormost scutes are ovoid, longer than wide, whereas posteriormost ones are a little larger and heart-shaped, as in †Armigatus. Each scute is medially keeled and produces a posterior spine-like process. The first scute anterior to the dorsal fin is the largest of the set but also the most problematic. Santos and Corrêa (1985SANTOS RS and CORRÊA VLS. 1985. Contribuição ao conhecimento da paleoictiofaúnula do Cretáceo do Brasil. In: Campos DA et al. (Orgs), Coletânea de Trabalhos Paleontológicos, Brasília: Ministério das Minas e Energia e Departamento Nacional da Produção Mineral, p. 169-174.) described this scute based on the type specimen taking into account its state of preservation and named †Scutatuspinosus based on this feature. They claimed that the largest scute is very similar to that of †Diplomystus, with three secondary spines and a larger median one on the posterior border. But Alvarado-Ortega et al. (2008) described it as showing smooth border. Examining holotype we agree with them in this case. The so-called spinous border is probably an artefact due to broken state taking into account the outline preserved in the rock (Fig. 11a). But unlike Alvarado-Ortega et al. (2008), we noted that this scute and all the others of the series are clearly ornamented with fading and straight radiating striations (cf. Santos and Corrêa 1985, fig. 2, p.171).
There are 17 prepelvic (Fig. 11b) and 8 postpelvic abdominal scutes (Fig. 11c) all keeled ventrally. The prepelvic scutes are small and quadrangular, with a convex dorsal border and reduced posterior spine-like process. They gradually increase in size towards the tail.
The pattern of abdominal scutes of †Scutatuspinosus is derived within †Ellimmichthyiformes because all other taxa have triangular scutes, deeper than long. The series reaches the antero-ventral end of the pectoral girdle. The postpelvic scutes are large and bear pungent spine-like processes, as commonly found in †Ellimmichthyiformes. The penultimate scute is the largest of the set. The series of abdominal scutes is similar to a saw and finishes at the origin of the anal fin, but does not reach it.
Squamation. The body is completely covered by obscure and imbricated cycloid scales. As stated by Santos and Corrêa (1985SANTOS RS and CORRÊA VLS. 1985. Contribuição ao conhecimento da paleoictiofaúnula do Cretáceo do Brasil. In: Campos DA et al. (Orgs), Coletânea de Trabalhos Paleontológicos, Brasília: Ministério das Minas e Energia e Departamento Nacional da Produção Mineral, p. 169-174.), they are large, a little deeper than long (at least in the trunk). They were thin and deciduous, so that they are absent in most of the specimens. Radii are not seen.
RESULTS
Our analysis generated 12 most parsimonious (MP) trees (tree length = 158; CI = 0.44; RI = 0.52; HI=0.557; Rescaled Consistency Index 0.23). The strict consensus tree shows a basal polytomy for all †Ellimmichthyiformes. Applying a 50% majority consensus tree (Fig. 12), †Sorbinichthys appears as sister-group of remaining taxa. The bootstrap and Bremer values for this group as a whole are weak so that they omitted in the figure 12. The highest bootstrap value (36) corresponds to Node 2 (†Ellimmichthyiformes more advanced than †Sorbinichthys). Only character 4 (i.e., ornamentation of skull roof) is uniquely derived for this group.
The majority rule consensus of 12 shortest trees obtained. Uniquely derived character is indicated with an asterisk symbol (*). The percentage of occurrence of arrangements are exposed together with change of character states. Synapomorphies supporting nodes are: Node 1 (†Ellimmichthyiformes, 75%): 10[01], 39 [01]; Node 2 (100%): 4*[01], 7[02], 12[01], 42[01]; Node 3 (66%): 29[01], 50[01], Node 4 (66%): 16[10], 36[10], 49[01]; Node 5 (66%): 12[10], 49[01], 53[01]; Node 6 (58%): 1 [01], 30[10]; Node 7 (75%): 5[01], 19[01], 60[01].
Although weakly supported, †Ellimmichthyiformes is placed clearly separated from the well supported Clupeiformes. †Thorectichthys, †Codoichthys, †Diplomystus, appear in a basal polytomy with two major well-defined groups remaining from a dismantled †Paraclupeidae, one composed of †Tycheiroichthys + †Rhombichthys + †Triplomystus and another formed by †Ellimma + †Ellimmichthys + †Paraclupea + †Ezkutuberezi + †Scutatuspinosus corresponding to a †Paraclupeinae clade. Most of MP trees place †Scutatuspinosus as a sister group of formed by †Ezkutuberezi, †Paraclupea, †Ellimmichthys, and †Ellimma, supported by two unambiguous derived features (Fig. 12, Node 27, characters 5, 19 and 60).
Exploring the MP trees, we noted different arrangements for †Scutatuspinosus itapagipensis depending on the existence of a †Paraclupeinae group. Either this taxon may be linked to †Ezkutuberezi or appearing as sister group of remaining †paraclupeines. In the first case, the characters 16 (teeth on endopterygoid, 1→0) and 52 (number of abdominal scutes, 2→1) support the clade. In the second case, the character 16 (teeth on endopterygoid, 1→0), and 49 (prepelvic and postpelvic scutes, 0→1), 53 (postcleithra, 0→1) and 55 (third hypural, 0→1) appeared to support the clade. In another condition, when †Thorectichthys appears as sister group of †paraclupeines, †Scutatuspinosus and † Ezkutuberezi are closely linked based on two reversals of characters 5 (dermal roof sculptured, 1→0) and 19 (epineural and epipleural bones invading caudal region, 1→0). In some cases, †paraclupeines (as stated above) do not form a clade, particularly when †Rhombichthys is included. In this, †Scutatuspinosus is linked with †Rhombichthys based on the characters 30 (proximal extremity of first uroneural, 0→1) and 56 (third hypural with upward process, 0→1).
DISCUSSION AND CONCLUSIONS
As in all previous analyses (e.g., Alvarado-Ortega et al. 2008, Murray and Wilson 2013MURRAY AM and WILSON MVH. 2013. Two new paraclupeid fishes (Clupeomorpha: Ellimmichthyiformes) from the Upper Cretaceous of Morocco. In: Arratia G et al. (Eds), Mesozoic Fishes 5 - Global Diversity and Evolution, München: Verlag Dr. Pfeil, Germany, p. 267-290.) the monophyly of †Ellimmichthyiformes is weakly supported. But our phylogenetic scheme differs from that of Zaragüeta-Bagils (2004) in which the group was revealed paraphyletic and instead agree with Alvarado-Ortega et al. (2008), in which monophyly is indicated. However, the other groupings are also weakly supported. An heuristic search made for all trees suggested unstable phylogenetic positions of many †ellimmichthyiform taxa such as †Codoichthys, †Sorbinichthys, †Thorectichthys, and †Diplomystus. The lower Bremer decay index for the †Ellimmichthyiformes indicates that further morphological studies should be done concerning other poorly known taxa to permit reassessment of the monophyly or paraphyly of the group as a whole.
Since Grande’s hand-drawn analysis (Grande 1985GRANDE L. 1985. Recent and fossil Clupeomorph fishes with material for revision of the subgroups of Clupeoids. Bull Am Mus Nat Hist 181: 231-372.), the monophyly of †Ellimmichthyiformes has been accepted a priori based on selected characters. But the reality of the group seems to be better supported due to the members lacking evidences for close affinities with outside taxa (e.g., clupeiforms, ostariophysans, and elopomorphs).
Taking characters of Grande (1985GRANDE L. 1985. Recent and fossil Clupeomorph fishes with material for revision of the subgroups of Clupeoids. Bull Am Mus Nat Hist 181: 231-372.) and adding new others, Chang and Maisey (2003CHANG MM and MAISEY JG. 2003. Redescription of †Ellimma branneri and †Diplomystus shengliensis, and relationships of some basal clupeomorphs. Am Mus Novit 3404: 1-35. ) used parsimony analysis to test the monophyly of †Ellimmichthyiformes by the first time. They used a data matrix containing 30 characters and 11 taxa. As a result, they recognized a clade containing †Armigatus, †Diplomystus, †Ellimma, †Ellimmichthys, and †Paraclupea. Furthermore, they discovered two subclades, one for †Armigatus plus †Diplomystus and other containing †Ellimma, †Ellimmichthys and †Paraclupea. Although not included in their analysis, they noted †Scutatuspinosus as a putative member of †Ellimmichthyiformes closely related to †paraclupeines.
Later, Zaragüeta-Bagils (2004), using a large data matrix including 56 characters and 15 terminal taxa dismantled †Ellimmichthyiformes, indicating it is a group of convenience only. In addition, he indicated that †Armigatus brevissimus, †Diplomystus birdi, and †D. dentatus form a group outside clupeiforms and †ellimmichthyiforms. Unfortunately, he omitted †Scutatuspinosus from his analysis.
In another hand-draw phylogenetic scheme, Forey (2004FOREY PL. 2004. A three-dimensional skull of a primitive clupeomorph from the English Chalk and implications for the evolution of the clupeomorph acusticolateralis system. In: Arratia G and Tintori A (Eds), Mesozoic Fishes 3 - Systematics, Paleoenvironments and Biodiversity, München: Verlag Dr. Friedrich Pfeil, Germany , p. 405-427.) argued that †Ellimmichthyiformes is monophyletic and diagnosed by a sigmoidal cleithrum. Also, he recognized two major subclades: a node D (for †Armigatus, †Triplomystus, †Diplomystus) and node C (for †Paraclupea and †Ellimmichthys). He indicated Node D diagnosed by enlarged endopterygoid teeth, elongate anal fin, and deep and narrow opercle, while his node C was characterized by selected characters of Grande (1982GRANDE L. 1982. A revision the fossil genus Diplomystus, with comments on the interrelationships of clupeomorph fishes. Am Mus Novit 2728: 1-34., 1985). Like Zaragüeta-Bagils (2004), Forey (2004) omitted †Ellimma, †Scutatuspinosus, and †Codoichthys, all yet known at that time.
In a parsimony analysis, Alvarado-Ortega et al. (2008) using a data matrix of 28 clupemorphs and 58 characters put †Armigatus outside from the †Ellimmichthyiformes. They used mainly characters taken from Grande (1982GRANDE L. 1982. A revision the fossil genus Diplomystus, with comments on the interrelationships of clupeomorph fishes. Am Mus Novit 2728: 1-34., 1985) and Zaragüeta-Bagils (2004) and divided the group in two clades: †Sorbinichthyidae and †Paraclupeidae. According to them, †Sorbinichthys is a member of a †Diplomystus clade and †Paraclupeidae contains all other †ellimmichthyiform fishes. However, some taxa were omitted in the analysis of Alvarado-Ortega et al. (2008) as the case of †Codoichthys but others (e.g., †Tycheroichthys, †Rhombichthys, †Thorecticthys) were only described in recent years. Problems with definition of characters and codification of states were detected so that they were critically reassessed by Murray and Wilson (2013MURRAY AM and WILSON MVH. 2013. Two new paraclupeid fishes (Clupeomorpha: Ellimmichthyiformes) from the Upper Cretaceous of Morocco. In: Arratia G et al. (Eds), Mesozoic Fishes 5 - Global Diversity and Evolution, München: Verlag Dr. Pfeil, Germany, p. 267-290.).
More recently, Murray and Wilson (2013MURRAY AM and WILSON MVH. 2013. Two new paraclupeid fishes (Clupeomorpha: Ellimmichthyiformes) from the Upper Cretaceous of Morocco. In: Arratia G et al. (Eds), Mesozoic Fishes 5 - Global Diversity and Evolution, München: Verlag Dr. Pfeil, Germany, p. 267-290.) using 28 taxa and 62 characters obtained a consensus tree of three shorest trees (176 steps, CI=0.44, RI=0.57). They indicated †Armigatoidei (†Armigatus plus †Diplomystus) was found as sister of all other †ellimmichthyiforms. The strict and majority-role consensus trees were identical but neither decay (Bremer support) nor bootstrap analyses indicated strong support for any branch except for certain pairs of species. †Ellimmichthyoidei contained †Sorbinichthyidae and †Paraclupeidae (including †scutatuspinosines, †thorectichthyines, and †paraclupeines). They detected many problems (i.e., many missing data) with taxa so that many of them were excluded from their analysis, for instance, †Ornategulum, †Ellimmichthys longicostatus, †Ezkutuberezi, and †Horseshoeichthys. Certain taxa show doubtful status, as is the case of †Ellimmichthys maceioensis (i.e., neither related to †E. longicostatus nor †Ellimma cruzi). The anatomical data for †Scutatuspinosus were taken from Alvarado-Ortega et al. (2008).
Figueiredo and Ribeiro (2016FIGUEIREDO FJ and RIBEIRO DRM. 2016. Relationships of †Codoichthys carnavalii Santos, 1994 (Teleostei, Clupeomorpha, †Ellimmichthyiformes) from the late Aptian of São Luís-Grajaú basin, NE Brazil. An Acad Bras Cienc 88: 1277-1307.), while revisiting †Codoichthys, listed previous preliminary efforts not cited in recent papers to decipher †Ellimmichthyiformes relationships, mostly derived from Figueiredo’s Thesis (unpublished data 2006). The results are almost equivalent to Murray and Wilson (2013MURRAY AM and WILSON MVH. 2013. Two new paraclupeid fishes (Clupeomorpha: Ellimmichthyiformes) from the Upper Cretaceous of Morocco. In: Arratia G et al. (Eds), Mesozoic Fishes 5 - Global Diversity and Evolution, München: Verlag Dr. Pfeil, Germany, p. 267-290.) and a corrected list of characters are furnished and discussed by them. Figueiredo and Ribeiro (2016) and Murray and Wilson (2013) show a weakly supported †Ellimmichthyiformes mainly based on S-shaped cleithrum and predorsal series of scutes. The same is the case for subgroups, but additionally †Ellimma cruzae was placed outside †Ellimma, and †Codoichthys was formally included within †Ellimmichthyiformes.
Although highlighting character conflict, Figueiredo and Ribeiro (2016FIGUEIREDO FJ and RIBEIRO DRM. 2016. Relationships of †Codoichthys carnavalii Santos, 1994 (Teleostei, Clupeomorpha, †Ellimmichthyiformes) from the late Aptian of São Luís-Grajaú basin, NE Brazil. An Acad Bras Cienc 88: 1277-1307.) feel the urgency of review for characters and reassessing taxa (e.g., †Ellimma cruzae, †Ellimmichthys maceioensis, †Ellimmichthys longicostatus). More informative specimens are needed too.
The analysis provided by Murray and Wilson (2013MURRAY AM and WILSON MVH. 2013. Two new paraclupeid fishes (Clupeomorpha: Ellimmichthyiformes) from the Upper Cretaceous of Morocco. In: Arratia G et al. (Eds), Mesozoic Fishes 5 - Global Diversity and Evolution, München: Verlag Dr. Pfeil, Germany, p. 267-290.) agreed with our results herein in the case of determination of the most basal clade of †Ellimmichthyiformes, that is †Sorbinichthyidae.
Although ancient, †Scutatuspinosus shows many skeletal specializations, some are homoplastically found outside †Ellimmichthyiformes, such as non-triangular abdominal scutes (shared with †Santanaclupea, from the Lower Cretaceous of Araripe Basin, see Maisey 1993MAISEY JG. 1993. A new clupeomorph fish from the Santana Formation (Albian) of NE Brazil. Am Mus Novit 3076: 1-15.) and straight and elongate maxilla (see Di Dario 2009). According to our results, the basal position of the †Scutatuspinosus within †Paraclupeidae permits the inference of a minimum age for that group corresponding to the Hauterivian-Barremian interval. If so, the early history of †Ellimmichthyiformes is a more ancient journey than hitherto imagined. Perhaps it is dated from the Upper Jurassic, when clupeomorphs were already divided in two major lineages: †Ellimmichthyiformes and Clupeiformes.
Recently, Vernygora et al. (2016VERNYGORA O, MURRAY AM and WILSON MVH. 2016. A primitive clupeomorph form the Albian Loon River Formation (Northwest Territories, Canada). Can J Earth Sci 53: 331-342.) described a new torpedo-like †ellimmichthyiform fish, †Foreyclupea loonensis, from the Albian Loon Formation, Canada. They claimed that this fish is closely related to †Scutatuspinosus itapagipensis and †Ranulfoichthys dorsonudum (Alvarado-Ortega 2014) and adopted a new family †Scutatuspinosidae for them considered sister group of †Paraclupeidae. This claim was based on the derived presence of a smooth and slightly rounded anterior dorsal margin of the body and absence of diastema between second and third hypurals (also shared with †Armigatus and †Diplomystus). However, the characters are doubtful per se. The first character is also found in †Codoichthys (not included in their analyses) and seems to be linked to the absence of predorsal series of scutes in †Foreyclupea and †Ranulfoichthys (as for many non-†ellimmichthyiform taxa). In this way, Vernygora et al. (2016) interpreted the absence as a case of secondary loss rather than primitive condition because it is present in †Scutatuspinosus. Within the group, the position of the pelvic fin was used to highlight affinities. But although †Scutatuspinosus and †Foreyclupea show a derived pelvic fin anterior to the origin of the dorsal fin, it is homoplastically found in †Ellimmichthys longicostatus.
Even, the ventral scutes of †Ranulfoichthys and †Scutatuspinosus are dorsally rounded unlike those of †Foreyclupea that are very similar to that of †Codoichthys. If so, only more characters can highlight putative affinities among these taxa.
As for previous works on the phylogeny of †ellimmichthyiforms, the obtained trees by Vernygora et al. (2016VERNYGORA O, MURRAY AM and WILSON MVH. 2016. A primitive clupeomorph form the Albian Loon River Formation (Northwest Territories, Canada). Can J Earth Sci 53: 331-342.) are weakly supported. But, excluding †Foreyclupea and †Ranulfoichthys, their phylogenetic scheme agrees with our majority rule consensus (Fig. 12) in which †Scutatuspinosus appears as sister group of remaining †paraclupeines.
The distribution of †paraclupeines such as †Paraclupea from China, and †Scutatuspinosus, †Ellimma plus †Ellimmichthys from northeastern Brazil suggests a complex biogeographical history in the early history of †Ellimmichthyiformes (Chang and Maisey 2003CHANG MM and MAISEY JG. 2003. Redescription of †Ellimma branneri and †Diplomystus shengliensis, and relationships of some basal clupeomorphs. Am Mus Novit 3404: 1-35. ). We think that this history is probably associated with a large geodispersal event and punctuated extinction events along a large remaining Tethyan corridor (Arai 2014ARAI M. 2014. Aptian/Albian (Early Cretaceous) paleogeography of the South Atlantic: a paleontological perspective. Brazilian J Geol 44(2): 339-350.). But to date approaches for an empirical test using current techniques of historical biogeography (e.g., cladistic biogeography and panbiogeography) are wanting.
ACKNOWLEDGMENTS
We are indebted to Dr. Valéria Gallo for reading a draft of this paper and for offering many helpful criticism and suggestions. We are grateful to Maria Regina Salgado de Melo (PROATEC-UERJ) for producing high resolution photographs. Acknowledgement is also made to Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ) for a grant-in-aid that enabled the junior author to work with fish fossil material in 2012. We are grateful to the reviewers Dr. Márcia Polck (DNPM) and Dr. Alison Murray (University of Alberta) for profitable comments and suggestions. Also, we thank the staff of the Departamento Nacional de Produção Mineral for hospitality during visits and loan of specimens.
REFERENCES
- ALLPORT S. 1860. On the discovery of some fossil remains near Bahia in South America. Quart J Geol Soc 16: 263-268.
- ALVARADO-ORTEGA J. 2014. Ancient herring fromt he Tlayúa Quarry (Cretaceous, Albian) near Tepexi de Rodrigues, Puebla State, central Mexico, closing the gap in the early diversification of Clupeomorpha. Cretaceous Res 50: 171-186.
- ALVARADO-ORTEGA J, OVALLES-DAMIAN E and ARRATIA G. 2008. A review of the interrelationships of the order Ellimmichthyiformes (Teleostei: Clupeomorpha). In: Arratia G, Schultze H-P and Wilson MVH (Eds), Mesozoic Fishes 4 - Homology and Phylogeny, München: Verlag Dr. Friedrich Pfeil, Germany, p. 257-278.
- ARAI M. 2014. Aptian/Albian (Early Cretaceous) paleogeography of the South Atlantic: a paleontological perspective. Brazilian J Geol 44(2): 339-350.
- ARRATIA G. 1997. Basal teleosts and teleostean phylogeny. Paleo Ichthyologica 7: 5-168.
- CAIXETA JM, BUENO GV, MAGNAVITA LP and FEIJÓ FJ. 1994. Bacias do Recôncavo, Tucano e Jatobá. Bol Geoci PETROBRAS 8(1): 163-172.
- CAVENDER T. 1966. The caudal skeleton of the Cretaceous teleosts Xiphactinus, Ichthyodectes, and Gillicus, its bearing on their relationship with Chirocentrus. Occas Pap Mus Zool Univ Mich 650(29): 1-14.
- CHANG MM and GRANDE L. 1997. Redescription of †Paraclupea chetungensis, an early Clupeomorph from the Lower Cretaceous of Southeastern China. Fieldiana Geol 37: 1-19.
- CHANG MM and MAISEY JG. 2003. Redescription of †Ellimma branneri and †Diplomystus shengliensis, and relationships of some basal clupeomorphs. Am Mus Novit 3404: 1-35.
- COPE ED. 1886. A contribution to the vertebrate paleontology of Brazil. Proc Am Philos Soc 23(121): 3-4.
- DI DARIO F. 2009. Chirocentrids as engrauloids: evidence from suspensorium, branchial arches, and infraorbital bones (Clupeomorpha, Teleostei). Zool J Linn Soc Lond 156: 363-383.
- DI DARIO F and DE PINNA MCC. 2006. The supratemporal system and the pattern of ramification of cephalic sensory canals in Denticeps clupeoides (Denticipitoidei, Teleostei): additional evidence for monophyly of Clupeiformes and Clupeoidei. Pap Avulsos Zool 46(10): 107-123.
- FALCÃO LC and DELLA FAVERA JC. 2012. Sublacustrine channel-levee system of the Caruaçu member, southwest Recôncavo Basin, Bahia, Brazil. Rev Brasil Geoci 42(3): 539-546.
- FIGUEIREDO FJ and RIBEIRO DRM. 2016. Relationships of †Codoichthys carnavalii Santos, 1994 (Teleostei, Clupeomorpha, †Ellimmichthyiformes) from the late Aptian of São Luís-Grajaú basin, NE Brazil. An Acad Bras Cienc 88: 1277-1307.
- FOREY PL. 1973a. A primitive clupeomorph fish from the Middle Cenomanian of Hakel, Lebanon. Can J Earth Sci 10(8): 1302-1318.
- FOREY PL. 1973b. A revision of the elopiform fishes, fossil and Recent. Bull Br Mus Nat Hist Geol Suppl 10: 1-222.
- FOREY PL. 2004. A three-dimensional skull of a primitive clupeomorph from the English Chalk and implications for the evolution of the clupeomorph acusticolateralis system. In: Arratia G and Tintori A (Eds), Mesozoic Fishes 3 - Systematics, Paleoenvironments and Biodiversity, München: Verlag Dr. Friedrich Pfeil, Germany , p. 405-427.
- FOREY PL, YI L, PATTERSON C and DAVIES CE. 2003. Fossil Fishes from the Cenomanian (Upper Cretaceous) of Namoura, Lebanon. J Syst Palaeontol 1(4): 227-330.
- GALLO V and FIGUEIREDO FJ. 2002. Clupeomorph fishes from the Lower Cretaceous of the Recôncavo Basin, Northeastern Brazil. J Vert Paleontol 22(3): 55A.
- GOLOBOFF P, FARRIS J and NIXON K. 2008. T.N.T, a free program for phylogenetic analysis. Cladistics 24: 774-786.
- GRANDE L. 1982. A revision the fossil genus Diplomystus, with comments on the interrelationships of clupeomorph fishes. Am Mus Novit 2728: 1-34.
- GRANDE L. 1985. Recent and fossil Clupeomorph fishes with material for revision of the subgroups of Clupeoids. Bull Am Mus Nat Hist 181: 231-372.
- GREENWOOD PH. 1968. The osteology and relationships of the Denticipitidae, a family of clupeomorph fishes. Bull Br Mus Nat Hist Zool 16: 215-273.
- HAY MJ, CUMBAA SL, MURRAY AM and PLINT AG. 2007. A new paraclupeid fish (Clupeomorpha, Ellimmichthyiformes) from a muddy marine prodelta environment: middle Cenomanian Dunvegan Formation, Alberta, Canada. Canad J Earth Sci 44(6): 775-790.
- KHALLOUFI B, ZARAGÜETA-BAGILS R and LELIÈVRE H. 2010. Rhombichthys intoccabilis, gen. et sp. nov. (Ellimmichthyiformes, Clupeomorpha, Teleostei), from the Cenomanian (Upper Cretaceous) of Ein Yabrud, Middle East: anatomical description and phylogenetic implications. J Vert Paleontol 30: 57-67.
- MAGNAVITA LP, SILVA RS and SANCHES CP. 2005. Roteiros geológicos e guia de campo da Bacia do Recôncavo, NE do Brasil. Bol Geoci PETROBRAS 13: 301-334.
- MAISEY JG. 1993. A new clupeomorph fish from the Santana Formation (Albian) of NE Brazil. Am Mus Novit 3076: 1-15.
- MAISEY JG. 2000. Continental break up and the distribution of fishes of Western Gondwana during the Early Cretaceous. Cretaceous Res 21: 281-314.
- MALABARBA MC, DO CARMO DA, GÓMEZ-PÉREZ I and DE QUEIROZ-NETO JV. 2004. A new clupeomorph fish from the Cretaceous Maceió Formation, Alagoas Basin, NE Brasil. Neues Jahrb Geol Paläont 233(2): 255-274.
- MURRAY AM and WILSON MVH. 2011. A new species of Sorbinichthys (Teleostei: Clupeomorpha: Ellimmichthyiformes) from the Late Cretaceous of Moroccco. Can J Earth Sci 48: 1-9.
- MURRAY AM and WILSON MVH. 2013. Two new paraclupeid fishes (Clupeomorpha: Ellimmichthyiformes) from the Upper Cretaceous of Morocco. In: Arratia G et al. (Eds), Mesozoic Fishes 5 - Global Diversity and Evolution, München: Verlag Dr. Pfeil, Germany, p. 267-290.
- NEWBREY MG, MURRAY AM, BRINKMAN DB, WILSON MVH and NEWMAN AG. 2010. A new articulated freshwater fish (Clupeomorpha, Ellimmichthyiformes) from the Late Cretaceous Horseshoe Canyon Formation, Alberta, Canada. Can J Earth Sci 47: 1183-1196.
- NYBELIN O. 1968. The dentition in the mouth cavity of Elops. In: Orvig T (Ed), Current problems of Lower Vertebrate Phylogeny, Stockholm: Nobel Symposium 4, p. 439-443.
- NYBELIN O. 1971. On the caudal skeleton in Elops with remarks on the other teleostean fishes. Acta R Soc Sci Litt Gothobg Zool 7: 1-52.
- PATTERSON C. 1967. Are the teleosts a polyphyletic group? In: Problémes actuales de paleontology (evolution des vertébrès). Colloques int CNRS, Paris 163: 93-109.
- PATTERSON C. 1993. Osteichthyes: Teleostei. In: Benton M (Ed), The Fossil Record 2, London: Chapman and Hall, p. 621-656.
- POYATO-ARIZA FJ, LÓPEZ-HORGUE MA and GARCÍA-GARMILLA F. 2000. A new early Cretaceous clupeomorph fish from the Arratia Valley, Basque Country, Spain. Cretaceous Res 21: 571-585.
- ROCHA PS, SOUZA AO DE AB, and CÂMARA RJB. 2002. O futuro da bacia do Recôncavo, a mais antiga província petrolífera brasileira. Bahia Análise & Dados 11(4): 32-44.
- SANTOS RS. 1990. Clupeiformes e Gonorhychiformes do Cretáceo Inferior (Aptiano) da Formação Cabo, NE do Brasil. An Acad Bras Cienc 62: 261-268.
- SANTOS RS. 1994. Ictiofáunula da Formação Codó, Cretáceo Inferior, com a descrição de um novo táxon - Codoichthys carnavalii (Pisces-Teleostei). An Acad Bras Cienc 66: 131-143.
- SANTOS RS and CORRÊA VLS. 1985. Contribuição ao conhecimento da paleoictiofaúnula do Cretáceo do Brasil. In: Campos DA et al. (Orgs), Coletânea de Trabalhos Paleontológicos, Brasília: Ministério das Minas e Energia e Departamento Nacional da Produção Mineral, p. 169-174.
- SCHAEFFER B. 1947. Cretaceous and Tertiary actinopterygian fishes from Brazil. Bull Amer Mus Nat Hist 141(3): 357-474.
- SILVA VG. 1993. Taxonômica e posicionamento estratigráfico de paleovertebrados da Bacia do Recôncavo. Acta Geol Leopoldensia 16(38): 41-58.
- TAVERNE L. 1974. L’ostéologie d’Elops Linné, C., 1766 (Pisces: Elopiformes) et son intérêt phylogénétique. Acad R Belg Mém Cl Sci 41: 1-96.
- VERNYGORA O, MURRAY AM and WILSON MVH. 2016. A primitive clupeomorph form the Albian Loon River Formation (Northwest Territories, Canada). Can J Earth Sci 53: 331-342.
- WOODWARD AS. 1908. On some Fossil Fishes discovered by Ennes de Souza in the Cretaceous Formation of Ilhéos (State of Bahia), Brazil. Quartl J Geol Soc London 64: 358-362.
- ZARAGÜETA-BAGILS R. 2004. Basal clupeomorphs and ellimmichthyiform phylogeny. In: Arratia G and Tintori A (Eds), Mesozoic Fishes 3 - Systematics, Paleoenvironments and Biodiversity. München: Verlag Dr. Friedrich Pfeil, Germany , p. 391-404.
Publication Dates
-
Publication in this collection
06 Feb 2017 -
Date of issue
Apr-Jun 2017
History
-
Received
30 May 2016 -
Accepted
23 Sept 2016