ABSTRACT
Saccharum spontaneum has been used for the development of energy cane a crop aimed to be used for the production of second-generation ethanol, or lignocellulosic ethanol. Lignin is a main challenge in the conversion of cell wall sugars into ethanol. In our studies to isolate the genes the lignin biosynthesis in S. spontaneum we have had great difficulty in RT-PCR reactions. Thus, we evaluated the effectiveness of different additives in the amplification of these genes. While COMT and CCoAOMT genes did not need any additives for other genes there was no amplification (HCT, F5H, 4CL and CCR) or the yield was very low (CAD and C4H). The application of supplementary cDNA was enough to overcome the non-specificity and low yield for C4H and C3H, while the addition of 0.04% BSA + 2% formamide was effective to amplify 4CL, CCR, F5H and CCR. HCT was amplified only by addition of 0.04% BSA + 2% formamide + 0.1 M trehalose and amplification of PAL was possible with addition of 2% of DMSO. Besides optimization of expression assays, the results show that additives can act independently or synergistically.
Key words:
biomass; cell wall; energy cane; lignin; lignocellulosic ethanol; sugarcane
INTRODUCTION
Due to the increasing demand for clean energy, there has been a growing interest in biofuels obtained from biomass. In the production of the second-generation ethanol (E2G) or lignocellulosic ethanol the cell wall polymeric sugars cellulose and hemicellulose are hydrolyzed to simpler units which become fermentable (Ruth and Thomas 2003RUTH MF AND THOMAS SR. 2003. Effect of Corn Stover Composition on Ethanol Process Economics. 25th Symp. Biotechnol. Fuels Chem NREL Publications, Breckenridge, Colorado, p. 22., Aden and Foust 2009ADEN A AND FOUST T. 2009. Technoeconomic analysis of the dilute sulfuric acid and enzymatic hydrolysis process for the conversion of corn stover to ethanol. Cellulose 16: 535-545., Huang et al. 2009HUANG HJ, RAMASWAMY S, AL-DAJANI W, TSCHIRNER U AND CAIRNCROSS RA. 2009. Effect of biomass species and plant size on cellulosic ethanol: A comparative process and economic analysis. Biomass and Bioenergy 33: 234-246.).
Lignin is a bottleneck for E2G production in several plants. This phenolic polymer chemically binds to cellulose and hemicellulose in a systematic arrangement, increasing the recalcitrance of the biomass (Boerjan et al. 2003BOERJAN W, RALPH J AND BAUCHER M. 2003. Lignin biosynthesis. Annu Rev Plant Biol 54: 519-546., Vanholme et al. 2010VANHOLME R ET AL. 2010. Engineering traditional monolignols out of lignin by concomitant up-regulation of F5H1 and down-regulation of COMT in Arabidopsis. Plant J 64: 885-897.). As a consequence, gene manipulation has been tried aiming to decrease or change lignin composition (Chapple and Carpita 1998CHAPPLE C AND CARPITA N. 1998. Plant cell walls as targets for biotechnology. Curr Opin Plant Biol 1: 179-185.). The degree of reduction or modification of lignin depends on the position of the enzyme in the biosynthetic pathway and the pattern of expression (Vanholme et al. 2012, Poovaiah et al. 2014POOVAIAH CR, NAGESWARA-RAO M, SONEJI JR, BAXTER HL AND STEWART CN. 2014. Altered lignin biosynthesis using biotechnology to improve lignocellulosic biofuel feedstocks. Plant Biotechnol J 12: 1163-1173.). Change in the expression of genes encoding enzymes operating in the earlier part of the route usually changes the total lignin content (Bonawitz and Chapple 2010BONAWITZ ND AND CHAPPLE C. 2010. The genetics of lignin biosynthesis: connecting genotype to phenotype. Annu Rev Genet 44: 337-363.), while genes positioned later lead to changes in the lignin type (Li et al. 2003, Ralph et al. 2006RALPH J, AKIYAMA T, KIM H, LU F, SCHATZ PF, MARITA JM, RALPH SA, REDDY MSS, CHEN F AND DIXON RA. 2006. Effects of coumarate 3-hydroxylase down-regulation on lignin structure. J Biol Chem 281: 8843-8853., Chen and Dixon 2007CHEN F AND DIXON RA. 2007. Lignin modification improves fermentable sugar yields for biofuel production. Nat Biotechnol 25: 759-761.).
Sugarcane bagasse is promising lignocellulosic material for E2G production. It is composed primarily of cellulose (39%), hemicellulose (25%) and lignin (23%) (Carroll and Somerville 2009CARROLL A AND SOMERVILLE C. 2009. Cellulosic Biofuels. Annu Rev Plant Biol 60: 165-182., Rezende et al. 2011REZENDE CA, DE LIMA MA, MAZIERO P, RIBEIRO DE AZEVEDO E, GARCIA W AND POLIKARPOV I. 2011. Chemical and morphological characterization of sugarcane bagasse submitted to a delignification process for enhanced enzymatic digestibility. Biotechnol Biofuels 4: 54., Szczerbowski et al. 2014SZCZERBOWSKI D, PITARELO AP, ZANDONÁ FILHO A AND RAMOS LP. 2014. Sugarcane biomass for biorefineries: comparative composition of carbohydrate and non-carbohydrate components of bagasse and straw. Carbohydr Polym 114: 95-101.). Few studies were conducted with lignin biosynthesis in sugarcane (Cesarino et al. 2012aCESARINO I, ARAÚJO P, DOMINGUES JÚNIOR AP AND MAZZAFERA P. 2012a. An overview of lignin metabolism and its effect on biomass recalcitrance. Brazilian J Bot 35: 303-311., b, 2013, Bottcher et al. 2013BOTTCHER A ET AL. 2013. Lignification in sugarcane: biochemical characterization, gene discovery, and expression analysis in two genotypes contrasting for lignin content. Plant Physiol 163: 1539-1557., Santos et al. 2015SANTOS AB, BOTTCHER A, VICENTINI R, MAYER JLS, KIYOTA E, LANDELL MGA, CRESTE S AND MAZZAFERA P. 2015. Lignin biosynthesis in sugarcane is affected by low temperature. Environ Exp Bot 120: 31-42.a, b, Vicentini et al. 2015VICENTINI R, BOTTCHER A, SANTOS BRITO M, SANTOS AB, CRESTE S, DE ANDRADE LANDELL MG, CESARINO I AND MAZZAFERA P. 2015. Large-scale transcriptome analysis of two sugarcane genotypes contrasting for lignin content. PLoS ONE 10: e0134909., Ferreira et al. 2016FERREIRA SS, HOTTA CT, DE CARLI POELKING VG, LEITE DCC, BUCKERIDGE MS, LOUREIRO ME, BARBOSA MHP, CARNEIRO MS AND SOUZA GM. 2016. Co-expression network analysis reveals transcription factors associated to cell wall biosynthesis in sugarcane. Plant Mol Biol 91: 15-35.) but transgenic plants were already produced (Jung et al. 2012JUNG JH, FOUAD WM, VERMERRIS W, GALLO M AND ALTPETER F. 2012. RNAi suppression of lignin biosynthesis in sugarcane reduces recalcitrance for biofuel production from lignocellulosic biomass. Plant Biotechnol J 10: 1067-1076., Bewg et al. 2016BEWG WP, POOVAIAH C, LAN W, RALPH J AND COLEMAN HD. 2016. RNAi downregulation of three key lignin genes in sugarcane improves glucose release without reduction in sugar production. Biotechnol Biofuels 9: 270., Jung and Altpeter 2016).
Recently, breeders are transferring and increasing the number of Saccharum spontaneum alleles in commercial sugarcane hybrids, aiming to increase fiber content in the bagasse and, consequently, the potential for E2G generation (Carvalho-Netto et al. 2014). S. spontaneum culms are rich in lignin and other polyphenolic compounds (Devi and Muthu 2014DEVI JAI AND MUTHU AK. 2014. Evaluation of in vitro free Radical Scavenging Potential of Whole Plant of S. spontaneum (Linn). Evaluation 6: 1436-1440.) which can negatively interfere in RT-PCR reactions during analysis of gene expression. Additionally, the genomes of the Saccharum genus are polyploid, redundant, and present a high GC content as verified in other grasses (Šmarda et al. 2014ŠMARDA P, BUREŠ P, HOROVÁ L, LEITCH IJ, MUCINA L, PACINI E, TICHÝ L, GRULICH V AND ROTREKLOVÁ O. 2014. Ecological and evolutionary significance of genomic GC content diversity in monocots. Proc Natl Acad Sci 111: E4096-E4102.), which often leads to the observation of non-specific bands or smears in the agarose gel as a result of RT-PCR analysis. In our case, the difficulty to amplify cDNA of genes of the lignin biosynthesis route in S. spontaneum internodes, using good quality (non-degraded) RNA, led us to test different additives in RT-PCR reactions.
RT-PCR is an essential technique in gene expression and other biotechnological studies (Farell and Alexandre 2012FARELL EM AND ALEXANDRE G. 2012. Bovine serum albumin further enhances the effects of organic solvents on increased yield of polymerase chain reaction of GC-rich templates. BMC Res Notes 5: 1.) but it is prone to several problems caused by inhibitors in the samples (Samarakoon et al. 2013SAMARAKOON T, WANG SY AND ALFORD MH. 2013. Enhancing PCR amplification of DNA from recalcitrant plant specimens using a trehalose-based additive. Appl Plant Sci 1: 1200236.). Additionally, some nucleotide regions in primers with high melting temperature (Tm) or with high GC content (>60%) can form secondary structures “interrupting” the reaction (Zeng et al. 2006ZENG ZS, YAN HT, ZHENG XD, HU GZ, CHEN Y AND DING M. 2006. High GC amplification: a comparative study of betaine, DMSO, formamide and glycerol as additives. Life Sci J 3: 67-71., Mamedov et al. 2008MAMEDOV TG, PIENAAR E, WHITNEY SE, TERMAAT JR, CARVILL G, GOLIATH R, SUBRAMANIAN A AND VILJOEN HJ. 2008. A fundamental study of the PCR amplification of GC-rich DNA templates. Comput Biol Chem 32: 452-457., Farell and Alexandre 2012). Several alternatives have been proposed to minimize these problems, such as the addition of substances of varied molecular nature (Simonović et al. 2012SIMONOVIĆ A, TRIFUNOVIC M, RASPOR M, CINGEL A, BOGDANOVIC M, DRAGICEVIC M AND SUBOTIC A. 2012. Dimethyl sulfoxide improves sensitivity and specificity of RT-PCR and QRT-PCR amplification of low-expressed transgenes. Arch Biol Sci 64: 865-876.) that can act synergistically (Ralser et al. 2006RALSER M, QUERFURTH R, WARNATZ HJ, LEHRACH H, YASPO ML AND KROBITSCH S. 2006. An efficient and economic enhancer mix for PCR. Biochem Biophys Res Commun 347: 747-751., Farell and Alexandre 2012) or independently (Chakrabarti and Schutt 2001CHAKRABARTI R AND SCHUTT CE. 2001. The enhancement of PCR amplification by low molecular-weight sulfones. Gene 274: 293-298.) in increasing the amplification process and the specificity of the reaction. Formamide (Sarkar et al. 1990SARKAR G, KAPELNER S AND SOMMER SS. 1990. Formamide can dramatically improve the specificity of PCR. Nucleic Acids Res 18: 7465., Chakrabarti and Schutt 2001), dimethyl sulfoxide - DMSO (Winship 1989WINSHIP PR. 1989. An improved method for directly sequencing PCR amplified material using dimethyl sulphoxide. Nucleic Acids Res 17: 1266., Varadaraj and Skinner 1994VARADARAJ K AND SKINNER DM. 1994. Denaturants or cosolvents improve the specificity of PCR amplification of a G+C-rich DNA using genetically engineered DNA polymerases. Gene 140: 1-5., Chakrabarti and Schutt 2002), non-ionic detergents (Demeke and Adams 1992DEMEKE T AND ADAMS RP. 1992. The effects of plant polysaccharides and buffer additives on PCR. Biotechniques 12: 332-334.), betaine (Baskaran et al. 1996BASKARAN N, KANDPAL RP, BHARGAVA AK, GLYNN MW, BALE A AND WEISSMAN SM. 1996. Uniform amplification of a mixture of deoxyribonucleic acids with varying GC content. Genome Res 6: 633-638., Weissensteiner and Lanchbury 1996WEISSENSTEINER T AND LANCHBURY JS. 1996. Strategy for controlling preferential amplification and avoiding false negatives in PCR typing. Biotechniques 21: 1102-1109., Henke et al. 1997HENKE W, HERDEL K, JUNG K, SCHNORR D AND LOENING SA. 1997. Betaine improves the PCR amplification of GC-rich DNA sequences. Nucleic Acids Res 25: 3957-3958.), trehalose (Spiess et al. 2004SPIESS AN, MUELLER N AND IVELL R. 2004. Trehalose is a potent PCR enhancer: lowering of DNA melting temperature and thermal stabilization of Taq polymerase by the disaccharide trehalose. Clin Chem 50: 1256-1259., Samarakoon et al. 2013) and other compatible solutes have been used to solve the problems of formation of secondary structures in the template of cDNA (Ralser et al. 2006, Farell and Alexandre 2012). BSA (bovine serum albumin) has a high content of lysine which binds to phenolic compounds (Katcher and Schwartz 1994KATCHER HL AND SCHWARTZ I. 1994. A distinctive property of Tth DNA polymerase: enzymatic amplification in the presence of phenol. Biotechniques 16: 84-92.) and polysaccharides (Demeke and Adams 1992), avoiding polymerase inactivation (Kreader 1996KREADER CA. 1996. Relief of amplification inhibition in PCR with bovine serum albumin or T4 gene 32 protein. Appl Environ Microbiol 62: 1102-1106.). The beneficial effects of these additives on PCR reactions are often specific to the type of cDNA template and primer and they have to be empirically determined (Simonović et al. 2012). Commercially available ready-to-use solutions, containing additives, have led to good results, but they possess the disadvantage of having unknown composition and the lack of flexibility to adjust the concentration of individual components (Ralser et al. 2006).
Aiming to isolate genes of the lignin biosynthesis in S. spontaneum for further studies on gene expression and sugarcane transformation, we used formamide, BSA, trehalose, betaine, DMSO and PEG-8000 as additives in RT-PCR reactions. We were able to amplify all genes of the lignin biosynthesis route.
MATERIALS AND METHODS
PLANT MATERIAL
The plants used in the study were clones obtained from tissue culture material provided by the Agronomic Institute of Campinas (IAC), Ribeirão Preto, São Paulo-Brazil. Culms were cut in segments containing two nodes and were planted in plastic trays containing vermiculite and kept in the greenhouse, with daily irrigation. After 40 days, the seedlings obtained were transplanted to plastic vessels of 50 L (3 plants per vessel) containing organic substrate (nitrogen= 0.5%, moisture = 50%, organic carbon = 15%, pH = 6, C/N = 130 mmol/Kg, CIC/C ratio= 8%, Genefertil-Genesol) and kept in the greenhouse under sprinkler irrigation (30 minutes, twice a day) for a period of approximately one year. After this period, the vessels were transferred to full sun, out of the greenhouse and plants were left to grow for another four months, under sprinkler irrigation (30 minutes, three times a day). Plant material was harvested in June 2014, with 5 replicates (5 plants). Healthy culms were collected from internodes 2nd+3rd (young) and 8th (mature), not including the node. With the aid of a blade the culms were cut into small pieces, frozen in liquid nitrogen and freeze-dried. The lyophilized material was ground to a powder in sterile mortars and cooled with liquid nitrogen and then stored in freezer -80°C.
RNA EXTRACTION AND CONSTRUCTION OF THE FIRST cDNA STRAND
RNA extraction was made with Trizol (Reagent Tri-Phasis - Bio Agency). Approximately 1/3 of the volume of an Eppendorf of 2 mL was filled with a mixture 1/1 (v/v) of young (2°+3°) and mature (8°) internode powders (in order to identify as many isoforms possible) from the five replicates and 1.5 mL of Tri-Phasis reagent was added. Further on the extraction followed the manufacturer recommendations. The quantification of total RNA was made in Nanodrop spectrophotometer (Thermo Scientific) at 260 nm and the purity evaluated by 260 nm/280 nm ratio. The RNA quality was checked by 1% agarose electrophoresis with ethidium bromide and further visualization under UV light (Gel Doc 2000, BioRad). RNA samples were treated with DNAse (Turbo DNAse-free kit, Ambion) and submitted to reverse transcription using the RevertedAid First Strand cDNA Synthesis kit, according to manufacturer’s instructions (Thermo Scientific).
In silico ANALYSIS OF DATABASE AND DESIGN OF PRIMERS
The sequences of the genes coding for the enzymes: 4-hydroxicinnamoyl CoA: ligase (4CL), cinnamoyl CoA reductase (CCR), ferulate 5-hydroxylase (F5H), caffeate O-methyltransferase (COMT) cinnamyl alcohol dehydrogenase (CAD), L- phenylalanine ammonia- lyase (PAL), caffeoyl CoA 3-O-methyltransferase (CCoAOMT), p-coumaroylshikimate 3’-hydroxylase (C3’H), cinnamate 4- hydroxylase (C4H), hydroxycinnamoyl-CoA: shikimate/quinate p-hydroxycinnamoyl-transferase (HCT) were carried out in the NCBI, Gene Index and Phytozome databases. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) = positive control. We used as bait the sequences more expressed in hybrid sugarcane tissues identified by Bottcher et al. (2013BOTTCHER A ET AL. 2013. Lignification in sugarcane: biochemical characterization, gene discovery, and expression analysis in two genotypes contrasting for lignin content. Plant Physiol 163: 1539-1557.). Among the sequences returned from the databases we selected those from sorghum (Sorghum bicolor), rice (Oryza sativa), corn (Zea mays), wheat (Triticum aestivum), rye grass (Lolium perenne) and Arabidopsis thaliana. Only sequences with full CDS, low e-value (10-6) and verified by BLASTx were used (Altschul et al. 1997ALTSCHUL SF, MADDEN TL, SCHÄFFER AA, ZHANG J, ZHANG Z, MILLER W AND LIPMAN DJ. 1997. Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25: 3389-3402.). These sequences were aligned in the BioEDIT software (Hall 1999HALL TA. 1999. BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp Ser 41: 95-98.) and from the alignment, primers were designed in conserved regions with Primer 3 (Rozen and Skaletsky 2000), using as parameters Tm in the range of 57°C - 60°C, 2°C of maximum difference between forward and reverse primers and % GC between 55 and 60. When this was not possible, degenerate primers were designed. The avoid formation of secondary internal structures and dimers the primers were verified by the OligoAnalizer 3.1 software. The primer sequences, GC content and the expected size of the amplicon are in Table I.
Primers used in the RT-PCR reactions, expected amplicon size (base pairs) and % of G+C in the amplicon. Non-specific bases (degenerate primers) are indicated in parenthesis.
RT-PCR REACTIONS
RT-PCR reactions were carried out in a thermal cycler Veriti 96-Well Thermal Cycler (AB Applied Biosystems). The standard conditions for RT-PCR were 40 cycles, each cycle consisting of a denaturing step at 94°C for 30 s, ringing at 57°C for 1 min, extension at 72°C for 2 min, with an initial denaturation step of 94°C for 5 min and a final extension of 72°C for 5 min. The final concentrations of 25 µL reactions were: 200 µM of each dNTP (dNTP mix Quiagen - 10 mM of each dNTP), 1.5 mM of MgCl2, 1 U of Taq-polymerase (Phoneutria 500 U) in buffer 1x Qiagen (Tris-Cl, KCl, (NH4)2SO4, 15 mM MgCl2; pH 8.7), 0.4 µM of each primer, 1 µL of a cDNA solution (corresponding to 3.5 ng of total RNA) and nuclease-free water. Additives were added independently or mixed in at the following final concentrations in 25 µL of RT-PCR reaction: 1 M betaine (Sigma), 2% formamide (Pharmacia Biotech, 99%), 2% DMSO (Merck, max. 0.03% H2O), 0.1 M trehalose [D-(+)-trehalose dehydrate, Sigma], 8% PEG-8000 (Affymetrix) and 0.04% BSA (Sigma). Betaine, trehalose and PEG-8000 were initially prepared as 5X concentrated stock solutions and BSA 25X. DMSO and formamide were considered as 100% stocks. The amplified bands were separated in 1% agarose gel containing ethidium bromide and displayed in photo-documenter-Gel Doc 2000 BioRad.
RESULTS AND DISCUSSION
Preliminarily, RT-PCR reactions were carried out without additives (Figure 1). Eight of the 10 genes analyzed did not have good amplification or they did not amplify. The amplicons were nonspecific to PAL and C3H; they were not observed with HCT, F5H, 4CL and CCR, or showed low yield with CAD and C4H.
RT-PCR amplification of lignin biosynthesis genes using standard reaction conditions as described in the text. cDNA was produced from a pool of young (2°+3°) and mature (8°) internodes from S. spontaneum. The numbers indicate the base pairs of the expected amplicon and arrows indicate weak bands. bp = base pairs (DNA 1 kb ladder New England Biolabs). GAPDH = positive control. Negative control reactions (template omitted) did not amplify any bands (not shown).
Only COMT and CCoAOMT genes presented good amplification without additives, observing intense and specific bands at the expected size of 756 and 649 bp, respectively. C4H and C3H reactions produced bands with the expected size of 1281 and 1343 bp, respectively, but with low specificity and low yield. The genes coding for these two enzymes could be simply and satisfactorily amplified when 2 µL of cDNA was used in the RT-PCR reactions, increasing yield and specificity of the bands (Figures 2a and 2b).
RT-PCR amplification of C4H (a) and C3H (b) using standard reaction conditions as described in the text but 1 or 2 µl of cDNA were included in the reaction mixture. cDNA was produced from a pool of young (2°+3°) and mature (8°) internodes from S. spontaneum. The numbers indicate the base pairs of the expected amplicon and arrows indicate weak bands. bp = base pairs [DNA 1 kb ladder from Thermo Scientific (a) and from New England Biolabs (b)]. Negative control reactions (template omitted) did not amplify any bands (not shown).
For CAD gene amplification, all PCR reactions were initially set up with 1 µL of cDNA and, preliminarily, additives were tested solely and at fixed concentrations, according to the literature (Lorenz 2011LORENZ TC. 2011. Polymerase chain reaction: basic protocol plus troubleshooting and optimization strategies. J Vis Exp JoVE 63: e3998., Simonović et al. 2012SIMONOVIĆ A, TRIFUNOVIC M, RASPOR M, CINGEL A, BOGDANOVIC M, DRAGICEVIC M AND SUBOTIC A. 2012. Dimethyl sulfoxide improves sensitivity and specificity of RT-PCR and QRT-PCR amplification of low-expressed transgenes. Arch Biol Sci 64: 865-876.). Only treatments 0.04% BSA and 0.04% BSA + 2% formamide allowed CAD gene amplification, and the combination of both additives showed better results in relation to the standard reaction, without additives (see Figure 1), generating a specific band with 943 bp, as expected (Figure 3a). The low efficiency with BSA may be due to its sensitivity to high temperatures used in the RT-PCR reaction, suggesting that its role would be more like a co-additive in the amplification of “templates” containing high GC % and PCR inhibitors (Farell and Alexandre 2012FARELL EM AND ALEXANDRE G. 2012. Bovine serum albumin further enhances the effects of organic solvents on increased yield of polymerase chain reaction of GC-rich templates. BMC Res Notes 5: 1.). The combination of BSA with formamide seemed to proportionate a synergistic effect, stabilizing Taq polymerase by inhibitors binding (mainly phenols) to BSA (Woide et al. 2010WOIDE D, ZINK A AND THALHAMMER S. 2010. Technical note: PCR analysis of minimum target amount of ancient DNA. Am J Phys Anthropol 142: 321-327., Samarakoon et al. 2013SAMARAKOON T, WANG SY AND ALFORD MH. 2013. Enhancing PCR amplification of DNA from recalcitrant plant specimens using a trehalose-based additive. Appl Plant Sci 1: 1200236.), and formamide-caused destabilization of GC rich regions, thereby decreasing Tm and facilitating the extension step in the reaction. In other reactions using BSA and formamide, but varying template amount, 1.5µL or 2 µL of cDNA proved most efficient for CAD gene amplification (Figure 3b).
RT-PCR amplification of CAD (a) using different adjuvants at varied concentrations and with BSA + formamide and varied amount of cDNA in the reaction mixture (b). cDNA was produced from a pool of young (2°+3°) and mature (8°) internodes from S. spontaneum. Expected CAD amplicon was 979 bp. 1 = 2% formamide, 2 = 4% DMSO, 3 = 2% formamide + 2% DMSO, 4 = 2% DMSO + 1 M betaine, 5 = 1 M betaine, 6 = 1 M betaine + 2% formamide, 7 = 0.04% BSA, 8 = 0.04% BSA + 4% DMSO, 9 = 2% formamide + 0.04% BSA, 10 = 0.04% BSA + 1 M betaine, 11 = GAPDH (positive control). Arrows indicate weak bands. bp = base pairs [DNA 1 kb ladder from Thermo Scientific (a) and from New England Biolabs(b)]. Reactions in B were carried out with 0.04% BSA + 2% formamide and different cDNA volumes were included. Negative control reactions (template omitted) did not amplify any bands (not shown).
No amplification products were observed for F5H, 4CL, HCT and CCR genes without additives (see Figure 1). Taking as reference the results with CAD, reactions were carried out using 0.04% BSA + 2% formamide (Figure 4a). Although specific bands were observed for F5H, CCR, and 4CL genes (625, 823 and 1340 bp, respectively), the yield was low. When more cDNA was added to the reactions the combination 3 µl of cDNA + 0.04% BSA + 2% formamide gave good results for these genes (Figure 4b).
RT-PCR amplification of HCT, F5H, PAL, CCR and 4CL using 0.04% BSA + 2% formamide and 1 µl cDNA in the reaction mixture (a) and F5H, 4CL and CCR using BSA and formamide but 3 µl cDNA in the reaction mixture (b). cDNA was produced from a pool of young (2°+3°) and mature (8°) internodes from S. spontaneum. The numbers indicate the base pairs of the expected amplicon and arrows indicate weak bands. bp = base pairs (DNA 1 kb ladder from New England Biolabs). Negative control reactions (template omitted) did not amplify any bands (not shown).
The need to employ additives in reactions with CAD, F5H, CCR, and 4CL may be related to the fact that we used degenerate primers. Thus, the primers would be in fact a “pool” of oligomers and could be leading to the formation of secondary structures, “hairpin loops” or dimers, which would be competing with the annealing process in the RT-PCR reaction, decreasing amplification. (Singh et al. 2000SINGH VK, GOVINDARAJAN R, NAIK S AND KUMAR A. 2000. The effect of hairpin structure on PCR amplification efficiency. Mol Biol Today 1: 67-69., Linhart and Shamir 2002LINHART C AND SHAMIR R. 2002. The degenerate primer design problem. Bioinformatics 18: S172-S181., Strien et al. 2013STRIEN J, SANFT J AND MALL G. 2013. Enhancement of PCR amplification of moderate GC-containing and highly GC-rich DNA sequences. Mol Biotechnol 54: 1048-1054.). Formamide decreases the Tm of cDNA template and consequently optimizes the annealing process of the primer (Simonović et al. 2012SIMONOVIĆ A, TRIFUNOVIC M, RASPOR M, CINGEL A, BOGDANOVIC M, DRAGICEVIC M AND SUBOTIC A. 2012. Dimethyl sulfoxide improves sensitivity and specificity of RT-PCR and QRT-PCR amplification of low-expressed transgenes. Arch Biol Sci 64: 865-876.). Formamide is much more efficient as an additive when used at a concentrations up to 5%, its effectiveness being abolished at 10% (Chakrabarti and Schutt 2010CHAKRABARTI R AND SCHUTT C. 2010. Chemical PCR: Compositions for enhancing polynucleotide amplification reactions. U.S. Patent No. 7,772,383. Aug 10, 2010. ), due to inhibition of Taq polymerase activity (Varadaraj and Skinner 1994VARADARAJ K AND SKINNER DM. 1994. Denaturants or cosolvents improve the specificity of PCR amplification of a G+C-rich DNA using genetically engineered DNA polymerases. Gene 140: 1-5.). For this reason, we used an intermediate concentration (2%), but the yield was improved in the reactions of CAD, CCR, 4CL and F5H genes only with cDNA supplementation (Figure 4b).
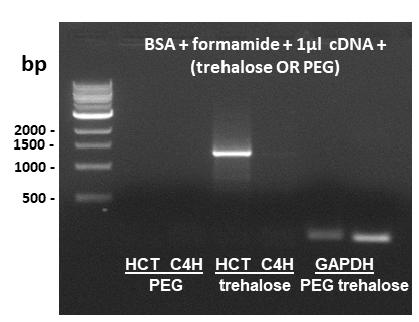
BSA + formamide combination was not effective for amplification of HCT gene, which has the highest % GC among genes herein studied (Table I). No amplification band was observed in the expected size of 1317 bp (Figure 4a). Others additives with unknown or not well established function are being successfully tested in various experiments of gene amplification by RT-PCR (Nordstrom et al. 2006NORDSTROM LJ, CLARK CA, ANDERSEN B, CHAMPLIN SM AND SCHWINEFUS JJ. 2006. Effect of ethylene glycol, urea, and N-methylated glycines on DNA thermal stability: the role of DNA base pair composition and hydration. Biochemistry 45: 9604-9614., Spink et al. 2007SPINK CH, GARBETT N AND CHAIRES JB. 2007. Enthalpies of DNA melting in the presence of osmolytes. Biophys Chem 126: 176-185.). Trehalose (0.1 M) and 8% PEG-8000 were chosen as additives to overcome the amplification problem of HCT in reactions containing 0.04% BSA + 2% formamide. BSA + formamide + trehalose allowed the amplification of HCT, resulting in a band with 1317 bp, as expected (Figure 5). PEG-8000 did not generate positive results. Additional cDNA was not needed to obtain a good yield and specificity.
RT-PCR amplification of HCT and C4H using 0.04% BSA + 2% formamide and 1 µl cDNA in the reaction mixture, supplemented either with 8% PEG-8000 or 0.1 M trehalose. GAPDH was used as a control with PEG-8000 and trehalose. The expected amplicons of HCT and C4H were 1317 and 1281 base pairs. cDNA was produced from a pool of young (2°+3°) and mature (8°) internodes from S. spontaneum. bp = base pairs (DNA 1 kb ladder from New England Biolabs). Negative control reactions (template omitted) did not amplify any bands (not shown).
It has been reported that besides decreasing the Tm of cDNA, trehalose also has a thermal stabilizer effect of Taq polymerase. (Weissensteiner and Lanchbury 1996WEISSENSTEINER T AND LANCHBURY JS. 1996. Strategy for controlling preferential amplification and avoiding false negatives in PCR typing. Biotechniques 21: 1102-1109., Carninci et al. 1998CARNINCI P, NISHIYAMA Y, WESTOVER A, ITOH M, NAGAOKA S, SASAKI N, OKAZAKI Y, MURAMATSU M AND HAYASHIZAKI Y. 1998. Thermostabilization and thermoactivation of thermolabile enzymes by trehalose and its application for the synthesis of full length cDNA. Proc Natl Acad Sci 95: 520-524., Spiess et al. 2004SPIESS AN, MUELLER N AND IVELL R. 2004. Trehalose is a potent PCR enhancer: lowering of DNA melting temperature and thermal stabilization of Taq polymerase by the disaccharide trehalose. Clin Chem 50: 1256-1259., Samarakoon et al. 2013SAMARAKOON T, WANG SY AND ALFORD MH. 2013. Enhancing PCR amplification of DNA from recalcitrant plant specimens using a trehalose-based additive. Appl Plant Sci 1: 1200236.). The fact that trehalose is an innocuous chemical compound and does not inhibit Taq polymerase suggests that in the reactions with HCT it would be acting in synergism with formamide in the annealing process, resulting in minimal competition between the annealing process and the formation of hairpin-loops or dimers in the pool of primers used in this experiment.
Little success was observed in the amplification of PAL gene using the basic conditions of RT-PCR (see Figure 1), with 3 diffuse and non-specific bands, below the expected size of 2033 bp. The addition of BSA and formamide also did not result in success (Figure 4a). PAL was amplified with the addition of 2% DMSO, presenting intense bands on expected 2033 bp position (Figures 6a and 6b). DMSO can stimulate RT-PCR reactions within the final concentration range of 1% to 10% (Jung et al. 2001JUNG M, MUCHE JM, LUKOWSKY A, JUNG K AND LOENING SA. 2001. Dimethyl sulfoxide as additive in ready-to-use reaction mixtures for real-time polymerase chain reaction analysis with SYBR Green I dye. Anal Biochem 289: 292-295., Kitade et al. 2003KITADE Y, OOTSUKA S, IITSUKA O AND SAGA N. 2003. Effect of DMSO on PCR of Porphyra yezoensis (Rhodophyta) gene. J Appl Phycol 15: 555-557., Simonović et al. 2012SIMONOVIĆ A, TRIFUNOVIC M, RASPOR M, CINGEL A, BOGDANOVIC M, DRAGICEVIC M AND SUBOTIC A. 2012. Dimethyl sulfoxide improves sensitivity and specificity of RT-PCR and QRT-PCR amplification of low-expressed transgenes. Arch Biol Sci 64: 865-876.). However, other studies have shown that above 2% this solvent can inhibit the activity of Taq polymerase (Lorenz 2011LORENZ TC. 2011. Polymerase chain reaction: basic protocol plus troubleshooting and optimization strategies. J Vis Exp JoVE 63: e3998.), which defined the concentration used in our study.
Taken together, our data shows that the appropriate use of single or combined additives in RT-PCR reaction mixtures can significantly improve the expression effectiveness of genes of lignin biosynthesis in sugarcane. The guidelines for optimizing amplification may be applied to other recalcitrant plant genes.
RT-PCR amplification of PAL using standard reaction conditions as described in the text (a) or including 2% DMSO in the reaction mixture (b). cDNA was produced from a pool of young (2°+3°) and mature (8°) internodes from S. spontaneum. Arrows indicate weak bands. bp = base pairs (DNA 1 kb ladder from New England Biolabs). Negative control reactions (template omitted) did not amplify any bands (not shown).
ACKNOWLEDGMENTS
JPPL thanks Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) and Fundação de Amparo à Pesquisa do estado de São Paulo (FAPESP, grant 2014/25994-1) for MSc fellowships; PM thanks Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for a research fellowship and FAPESP (2008/58035-6) for grant support. The authors thank Dr. Silvana Creste of Centro de Cana - Instituto Agronômico de Campinas (IAC) for providing the S. spontaneum clones.
REFERENCES
- ADEN A AND FOUST T. 2009. Technoeconomic analysis of the dilute sulfuric acid and enzymatic hydrolysis process for the conversion of corn stover to ethanol. Cellulose 16: 535-545.
- ALTSCHUL SF, MADDEN TL, SCHÄFFER AA, ZHANG J, ZHANG Z, MILLER W AND LIPMAN DJ. 1997. Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25: 3389-3402.
- BASKARAN N, KANDPAL RP, BHARGAVA AK, GLYNN MW, BALE A AND WEISSMAN SM. 1996. Uniform amplification of a mixture of deoxyribonucleic acids with varying GC content. Genome Res 6: 633-638.
- BEWG WP, POOVAIAH C, LAN W, RALPH J AND COLEMAN HD. 2016. RNAi downregulation of three key lignin genes in sugarcane improves glucose release without reduction in sugar production. Biotechnol Biofuels 9: 270.
- BOERJAN W, RALPH J AND BAUCHER M. 2003. Lignin biosynthesis. Annu Rev Plant Biol 54: 519-546.
- BONAWITZ ND AND CHAPPLE C. 2010. The genetics of lignin biosynthesis: connecting genotype to phenotype. Annu Rev Genet 44: 337-363.
- BOTTCHER A ET AL. 2013. Lignification in sugarcane: biochemical characterization, gene discovery, and expression analysis in two genotypes contrasting for lignin content. Plant Physiol 163: 1539-1557.
- CARNINCI P, NISHIYAMA Y, WESTOVER A, ITOH M, NAGAOKA S, SASAKI N, OKAZAKI Y, MURAMATSU M AND HAYASHIZAKI Y. 1998. Thermostabilization and thermoactivation of thermolabile enzymes by trehalose and its application for the synthesis of full length cDNA. Proc Natl Acad Sci 95: 520-524.
- CARROLL A AND SOMERVILLE C. 2009. Cellulosic Biofuels. Annu Rev Plant Biol 60: 165-182.
- CARVALHO-NETTO CV, BRESSIANI JA, SORIANO HL, FIORI CS, SANTOS JM, BARBOSA GVS, XAVIER MA, LANDELL MGA AND PEREIRA GAG. 2014. The potential of the energy cane as the main biomass crop for the cellulosic industry. Chem Biol Technol Agric 1: 1-8.
- CESARINO I, ARAÚJO P, DOMINGUES JÚNIOR AP AND MAZZAFERA P. 2012a. An overview of lignin metabolism and its effect on biomass recalcitrance. Brazilian J Bot 35: 303-311.
- CESARINO I, ARAÚJO P, LEME AFP, CRESTE S AND MAZZAFERA P. 2013. Suspension cell culture as a tool for the characterization of class III peroxidases in sugarcane. Plant Physiol Biochem 62: 1-10.
- CESARINO I, ARAÚJO P, MAYER JLS, LEME AFP AND MAZZAFERA P. 2012b. Enzymatic activity and proteomic profile of class III peroxidases during sugarcane stem development. Plant Physiol Biochem 55: 66-76
- CHAKRABARTI R AND SCHUTT C. 2010. Chemical PCR: Compositions for enhancing polynucleotide amplification reactions. U.S. Patent No. 7,772,383. Aug 10, 2010.
- CHAKRABARTI R AND SCHUTT CE. 2001. The enhancement of PCR amplification by low molecular-weight sulfones. Gene 274: 293-298.
- CHAKRABARTI R AND SCHUTT CE. 2002. Novel sulfoxides facilitate GC-rich template amplification. Biotechniques 32: 866-875.
- CHAPPLE C AND CARPITA N. 1998. Plant cell walls as targets for biotechnology. Curr Opin Plant Biol 1: 179-185.
- CHEN F AND DIXON RA. 2007. Lignin modification improves fermentable sugar yields for biofuel production. Nat Biotechnol 25: 759-761.
- DEMEKE T AND ADAMS RP. 1992. The effects of plant polysaccharides and buffer additives on PCR. Biotechniques 12: 332-334.
- DEVI JAI AND MUTHU AK. 2014. Evaluation of in vitro free Radical Scavenging Potential of Whole Plant of S. spontaneum (Linn). Evaluation 6: 1436-1440.
- FARELL EM AND ALEXANDRE G. 2012. Bovine serum albumin further enhances the effects of organic solvents on increased yield of polymerase chain reaction of GC-rich templates. BMC Res Notes 5: 1.
- FERREIRA SS, HOTTA CT, DE CARLI POELKING VG, LEITE DCC, BUCKERIDGE MS, LOUREIRO ME, BARBOSA MHP, CARNEIRO MS AND SOUZA GM. 2016. Co-expression network analysis reveals transcription factors associated to cell wall biosynthesis in sugarcane. Plant Mol Biol 91: 15-35.
- HALL TA. 1999. BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp Ser 41: 95-98.
- HENKE W, HERDEL K, JUNG K, SCHNORR D AND LOENING SA. 1997. Betaine improves the PCR amplification of GC-rich DNA sequences. Nucleic Acids Res 25: 3957-3958.
- HUANG HJ, RAMASWAMY S, AL-DAJANI W, TSCHIRNER U AND CAIRNCROSS RA. 2009. Effect of biomass species and plant size on cellulosic ethanol: A comparative process and economic analysis. Biomass and Bioenergy 33: 234-246.
- JUNG JH AND ALTPETER F. 2016. TALEN mediated targeted mutagenesis of the caffeic acid O-methyltransferase in highly polyploid sugarcane improves cell wall composition for production of bioethanol. Plant Mol Biol 92: 131-142.
- JUNG JH, FOUAD WM, VERMERRIS W, GALLO M AND ALTPETER F. 2012. RNAi suppression of lignin biosynthesis in sugarcane reduces recalcitrance for biofuel production from lignocellulosic biomass. Plant Biotechnol J 10: 1067-1076.
- JUNG M, MUCHE JM, LUKOWSKY A, JUNG K AND LOENING SA. 2001. Dimethyl sulfoxide as additive in ready-to-use reaction mixtures for real-time polymerase chain reaction analysis with SYBR Green I dye. Anal Biochem 289: 292-295.
- KATCHER HL AND SCHWARTZ I. 1994. A distinctive property of Tth DNA polymerase: enzymatic amplification in the presence of phenol. Biotechniques 16: 84-92.
- KITADE Y, OOTSUKA S, IITSUKA O AND SAGA N. 2003. Effect of DMSO on PCR of Porphyra yezoensis (Rhodophyta) gene. J Appl Phycol 15: 555-557.
- KREADER CA. 1996. Relief of amplification inhibition in PCR with bovine serum albumin or T4 gene 32 protein. Appl Environ Microbiol 62: 1102-1106.
- LI Y, KAJITA S, KAWAI S, KATAYAMA Y AND MOROHOSHI N. 2003. Down-regulation of an anionic peroxidase in transgenic aspen and its effect on lignin characteristics. J Plant Res 116: 175-182.
- LINHART C AND SHAMIR R. 2002. The degenerate primer design problem. Bioinformatics 18: S172-S181.
- LORENZ TC. 2011. Polymerase chain reaction: basic protocol plus troubleshooting and optimization strategies. J Vis Exp JoVE 63: e3998.
- MAMEDOV TG, PIENAAR E, WHITNEY SE, TERMAAT JR, CARVILL G, GOLIATH R, SUBRAMANIAN A AND VILJOEN HJ. 2008. A fundamental study of the PCR amplification of GC-rich DNA templates. Comput Biol Chem 32: 452-457.
- NORDSTROM LJ, CLARK CA, ANDERSEN B, CHAMPLIN SM AND SCHWINEFUS JJ. 2006. Effect of ethylene glycol, urea, and N-methylated glycines on DNA thermal stability: the role of DNA base pair composition and hydration. Biochemistry 45: 9604-9614.
- POOVAIAH CR, NAGESWARA-RAO M, SONEJI JR, BAXTER HL AND STEWART CN. 2014. Altered lignin biosynthesis using biotechnology to improve lignocellulosic biofuel feedstocks. Plant Biotechnol J 12: 1163-1173.
- RALPH J, AKIYAMA T, KIM H, LU F, SCHATZ PF, MARITA JM, RALPH SA, REDDY MSS, CHEN F AND DIXON RA. 2006. Effects of coumarate 3-hydroxylase down-regulation on lignin structure. J Biol Chem 281: 8843-8853.
- RALSER M, QUERFURTH R, WARNATZ HJ, LEHRACH H, YASPO ML AND KROBITSCH S. 2006. An efficient and economic enhancer mix for PCR. Biochem Biophys Res Commun 347: 747-751.
- REZENDE CA, DE LIMA MA, MAZIERO P, RIBEIRO DE AZEVEDO E, GARCIA W AND POLIKARPOV I. 2011. Chemical and morphological characterization of sugarcane bagasse submitted to a delignification process for enhanced enzymatic digestibility. Biotechnol Biofuels 4: 54.
- ROZEN S AND SKALETSKY H. 1999. Primer3 on the WWW for general users and for biologist programmers. Bioinforma methods Protoc: 365-386.
- RUTH MF AND THOMAS SR. 2003. Effect of Corn Stover Composition on Ethanol Process Economics. 25th Symp. Biotechnol. Fuels Chem NREL Publications, Breckenridge, Colorado, p. 22.
- SAMARAKOON T, WANG SY AND ALFORD MH. 2013. Enhancing PCR amplification of DNA from recalcitrant plant specimens using a trehalose-based additive. Appl Plant Sci 1: 1200236.
- SANTOS AB, BOTTCHER A, KIYOTA E, MAYER JLS, VICENTINI R, SANTOS BRITO M, CRESTE S, LANDELL MGA AND MAZZAFERA P. 2015. Water stress alters lignin content and related gene expression in two sugarcane genotypes. J Agric Food Chem 63: 4708-4720.
- SANTOS AB, BOTTCHER A, VICENTINI R, MAYER JLS, KIYOTA E, LANDELL MGA, CRESTE S AND MAZZAFERA P. 2015. Lignin biosynthesis in sugarcane is affected by low temperature. Environ Exp Bot 120: 31-42.
- SARKAR G, KAPELNER S AND SOMMER SS. 1990. Formamide can dramatically improve the specificity of PCR. Nucleic Acids Res 18: 7465.
- SIMONOVIĆ A, TRIFUNOVIC M, RASPOR M, CINGEL A, BOGDANOVIC M, DRAGICEVIC M AND SUBOTIC A. 2012. Dimethyl sulfoxide improves sensitivity and specificity of RT-PCR and QRT-PCR amplification of low-expressed transgenes. Arch Biol Sci 64: 865-876.
- SINGH VK, GOVINDARAJAN R, NAIK S AND KUMAR A. 2000. The effect of hairpin structure on PCR amplification efficiency. Mol Biol Today 1: 67-69.
- ŠMARDA P, BUREŠ P, HOROVÁ L, LEITCH IJ, MUCINA L, PACINI E, TICHÝ L, GRULICH V AND ROTREKLOVÁ O. 2014. Ecological and evolutionary significance of genomic GC content diversity in monocots. Proc Natl Acad Sci 111: E4096-E4102.
- SPIESS AN, MUELLER N AND IVELL R. 2004. Trehalose is a potent PCR enhancer: lowering of DNA melting temperature and thermal stabilization of Taq polymerase by the disaccharide trehalose. Clin Chem 50: 1256-1259.
- SPINK CH, GARBETT N AND CHAIRES JB. 2007. Enthalpies of DNA melting in the presence of osmolytes. Biophys Chem 126: 176-185.
- STRIEN J, SANFT J AND MALL G. 2013. Enhancement of PCR amplification of moderate GC-containing and highly GC-rich DNA sequences. Mol Biotechnol 54: 1048-1054.
- SZCZERBOWSKI D, PITARELO AP, ZANDONÁ FILHO A AND RAMOS LP. 2014. Sugarcane biomass for biorefineries: comparative composition of carbohydrate and non-carbohydrate components of bagasse and straw. Carbohydr Polym 114: 95-101.
- VANHOLME R ET AL. 2010. Engineering traditional monolignols out of lignin by concomitant up-regulation of F5H1 and down-regulation of COMT in Arabidopsis. Plant J 64: 885-897.
- VANHOLME R, STORME V, VANHOLME B, SUNDIN L, CHRISTENSEN JH, GOEMINNE G, HALPIN C, ROHDE A, MORREEL K AND BOERJAN W. 2012. A systems biology view of responses to lignin biosynthesis perturbations in Arabidopsis. Plant Cell 24: 3506-3529.
- VARADARAJ K AND SKINNER DM. 1994. Denaturants or cosolvents improve the specificity of PCR amplification of a G+C-rich DNA using genetically engineered DNA polymerases. Gene 140: 1-5.
- VICENTINI R, BOTTCHER A, SANTOS BRITO M, SANTOS AB, CRESTE S, DE ANDRADE LANDELL MG, CESARINO I AND MAZZAFERA P. 2015. Large-scale transcriptome analysis of two sugarcane genotypes contrasting for lignin content. PLoS ONE 10: e0134909.
- WEISSENSTEINER T AND LANCHBURY JS. 1996. Strategy for controlling preferential amplification and avoiding false negatives in PCR typing. Biotechniques 21: 1102-1109.
- WINSHIP PR. 1989. An improved method for directly sequencing PCR amplified material using dimethyl sulphoxide. Nucleic Acids Res 17: 1266.
- WOIDE D, ZINK A AND THALHAMMER S. 2010. Technical note: PCR analysis of minimum target amount of ancient DNA. Am J Phys Anthropol 142: 321-327.
- ZENG ZS, YAN HT, ZHENG XD, HU GZ, CHEN Y AND DING M. 2006. High GC amplification: a comparative study of betaine, DMSO, formamide and glycerol as additives. Life Sci J 3: 67-71.
Publication Dates
-
Publication in this collection
Jan-Mar 2018
History
-
Received
31 Mar 2017 -
Accepted
19 May 2017