Abstracts
Impression cytology (IC) has been widely used as a method for evaluating the ocular surface and superficial cells layers in the diagnosis and follow-up after treatment of several ocular surface tumors of both epithelial and melanocytic origin. Information regarding this can be found in the English-language literature since 1992. Using either cellulose acetate or Biopore membranes for specimen collection, a high correlation has been found between IC and tissue histology. Compared with exfoliative cytology with spatula, IC is less traumatic to the patient’s eye, provides a precise location of the area being studied, and allows accurate observation of the cells the way they exist in vivo. The additional advantage of IC is the preservation of limbal stem cells responsible for continuous corneal epithelium renewal; these can be affected after incisional or excisional biopsy at the corneoscleral limbus, which is the most frequent site of appearance of tumors in the stratified epithelium. Treatment for ocular surface squamous neoplasia has historically included surgery, but nonsurgical interventions have also been adopted. Hence, in certain cases, ophthalmologists may prefer interventions less invasive than surgical biopsy such as of impression cytology for both initial diagnosis and therapeutic monitoring of treatment for ocular surface lesions. Nevertheless, it should be considered that IC may be less helpful if the results conflict with the clinical picture or if the clinical diagnosis is uncertain and results are negative. In such cases, surgical biopsy is required for accurate diagnosis. The purpose of this review is to examine the published literature on the utilization of IC for the diagnosis and management of ocular surface tumors and to discuss the requirement for further investigation on the subject.
Conjunctiva; Cornea; Limbus corneae; Conjunctival neoplasms/diagnosis; Eye neoplasms; Cytodiagnosis; Cytological techniques; Diagnostic techniques, ophthalmological; Review
A citologia de impressão (CI) tem sido amplamente utilizada como um método de avaliação da superfície ocular e das camadas de células superficiais no diagnóstico e no seguimento após tratamento de vários tumores da superfície ocular de origem epitelial ou melanocítica. As informações podem sem encontradas na literatura em língua inglesa desde 1992. Utilizando-se de membranas de acetato de celulose ou Biopore na coleta dos espécimes, uma alta correlação tem sido encontrada entre a CI e a histologia do tecido. Comparando-se com a citologia esfoliativa, a citologia de impressão é menos traumática para o olho do paciente, fornece uma localização precisa da área estudada e permite ver as células da forma como elas organizam-se in vivo. A vantagem adicional da citologia de impressão é a preservação das células- tronco germinativas responsáveis pela renovação contínua do epitélio da córnea. Elas podem ser afetadas após biópsia cirúrgica na região do limbo que é o sítio mais frequentemente acometido pelos tumores do epitélio estratificado. O tratamento para a neoplasia escamosa da superfície ocular tem sido historicamente a cirurgia, mas intervenções não cirúrgicas também foram adotadas. Por esta razão, em certos casos, oftalmologistas podem recorrer a formas menos invasivas que a biópsia cirúrgica (como a citologia de impressão) tanto para o diagnóstico inicial quanto para o monitoramento terapêutico das lesões da superfície ocular. No entanto, deve-se ter em mente que a citologia de impressão deixa de ser útil quando seu resultado não coincide com o quadro clínico ou quando o diagnóstico clínico é incerto e o resultado da citologia de impressão negativo. Nesses casos, a biópsia cirúrgica deve ser realizada para o diagnóstico. O objetivo desta revisão é examinar a literatura sobre a utilização da citologia de impressão no diagnóstico e tratamento dos tumores da superfície ocular bem como discutir a necessidade de uma investigação mais aprofundada sobre o assunto.
Túnica conjuntiva; Córnea; Limbo da córnea; Neoplasias da túnica conjuntiva/diagnóstico; Neoplasias oculares; Citodiagnóstico; Técnicas citológicas; Técnicas de diagnóstico oftalmológico; Revisão
Introduction
Ocular surface tumors encompass malignant, premalignant, and benign lesions arising from the conjunctiva, limbus, or cornea. These neoplasms may originate mainly from squamous epithelia, melanocytes, or lymphocyte cells(11 Varde MA, Biswas J. Ocular surface tumors. Oman J Ophthalmol. 2009;2(1):1-2.).
Clinical examination of the tumors based on slit-lamp biomicroscopy by a trained professional frequently yields a correct diagnosis, if the clinician is familiar with the clinical characteristics. However, in some instances, only a broad differential diagnosis is possible, and slit-lamp biomicroscopy cannot reliably exclude uncommon diagnosis such as amelanotic malignant melanoma, highlighting the importance of acquiring a clinical diagnosis before administering a treatment. The gold standard is obtaining a biopsy, either incisional or excisional, for histopathology. The main risk of clinical misdiagnosis of an excised benign lesion is exposing the patient to unnecessary surgery; to prevent this, adjunctive diagnostic tests can be performed(22 Rudkin AK, Dodd T, Muecke JS. The differential diagnosis of localised amelanotic limbal lesions: a review of 162 consecutive excisions. Br J Ophthalmol. 2011:95(3):350-4.).
Diagnosis may be improved by cytological examination, high-resolution anterior segment ultrasound (UBM), in vivo confocal microscopy, and optical coherence tomography. Cytological sampling is a relatively noninvasive method and is thereby preferable when treatment with nonsurgical techniques such as administration of a topical chemotherapeutic agent with an antineoplastic drug is considered(33 Lee GA, Hirst LW. Ocular surface squamous neoplasia. Surv Ophthalmol. 1995;39(6):429-50.). It may also assist in evaluating lesions in cases where surgery may not be appropriate, including patients not medically fit for surgical biopsy(44 Barros JN, Lowen MS, Ballalai PL, Mascaro VL, Gomes JA, Martins MC. Predictive index to differentiate invasive squamous cell carcinoma from preinvasive ocular surface lesions by impression cytology. Br J Ophthalmol. 2009:93(2);209-14.). In 1954, Larmande and Timsit were the first to use cytodiagnosis in ophthalmology to assist in the evaluation of tumors of the sclerocorneal limbus(55 Larmande A, Timsit E. [Importance of cytodiganosis in ophthalmology: preliminar reporto f 8 cases of tumors of the sclero-corneal limbus]. Bull Soc Ophtalmol Fr. 1954;5:415-9. French.).
Ocular surface cytology can be performed by several methods, including spatula scraping, brush cytology, and impression cytology (IC). IC is a well-established technique for collecting superficial epithelial layers by applying collecting devices (either cellulose acetate filter papers or Biopore membrane device), so that cells adhere to their surface and are removed from the eye to be processed further for analysis by various appropriate methods. IC was first developed to diagnose dry-eye status, and it is now used to diagnose various ocular surface disorders, including neoplasia. It represents a non- or minimally invasive biopsy technique applicable to the conjunctiva, cornea, and limbal area for both diagnosis and follow-up after treatment of tumors(66 Tananuvat N, Lertprasertsuk N, Mahanupap P, Noppanakeepong P. Role of impression cytology in diagnosis of ocular surface neoplasia. Cornea. 2008;27(3):269-74.). Because repeated surgical biopsies of suspicious ocular surface lesions may cause complications such as scarring, lid deformity, limbal stem cell deficiency (LSCD), and great discomfort to the patient, IC can assist in the evaluation(44 Barros JN, Lowen MS, Ballalai PL, Mascaro VL, Gomes JA, Martins MC. Predictive index to differentiate invasive squamous cell carcinoma from preinvasive ocular surface lesions by impression cytology. Br J Ophthalmol. 2009:93(2);209-14.).
The present review examines and updates the published literature on the utilization of IC for the diagnosis and management of ocular surface tumors and discusses the requirement for further investigation on the subject.
IC technique
After a complete ophthalmological examination, including slit-lamp biomicroscopy, IC can be performed according to methods previously described(44 Barros JN, Lowen MS, Ballalai PL, Mascaro VL, Gomes JA, Martins MC. Predictive index to differentiate invasive squamous cell carcinoma from preinvasive ocular surface lesions by impression cytology. Br J Ophthalmol. 2009:93(2);209-14.). A drop of topical anesthesia is consistently used. Then, the collection of the superficial cell layers of the ocular surface is performed by forceps-assisted application of a membrane with submicroscopic pores, such as MF-Millipore, onto the patient’s lesion. Membranes are often precut in different shapes and sizes for orientation purposes during processing. Most authors agree to using membranes with pore sizes ranging 0.025-0.45 µm. It is essential to consider the pore size because it affects the consistency of cell collection (the larger the pore size, greater the cellularity) and the resolution of the details under the microscope (morphology was better preserved in the smaller pore size papers). The membrane is firmly pressed against the area to be sampled with the aid of a swab or a solid rod for some seconds and then peeled off using the forceps. Whenever needed, more samples can be collected. They are immediately transferred to be fixed in a solution containing glacial acetic acid, 37% formaldehyde, and ethyl alcohol in a 1:1:20 volume ratio, taking care to completely immerse the membranes. After samples have been fixed, different staining techniques can be performed laboratory analysis. The most used stains include periodic acid-Schiff (PAS), hematoxylin-eosin, Gill’s hematoxylin, and Papanicolaou. The cells can be mounted on a slide after fixation and staining ready for interpretation. PAS is used to stain goblet cells and their secretions and hematoxylin as a counterstain to stain epithelial cells. Papanicolaou helps to better interpret the epithelial changes of squamous metaplasia and the distinct nuclear patterns. These stains have also been used together. Although over the last decade several techniques have used IC samples, light microscopy remains the most used method. To evaluate IC specimens by light microscopy, several features are universally evaluated: the morphology of the epithelial cells, the degree of squamous metaplasia, the nuclear to cytoplasmic (N/C) ratio; the density, shape, and PAS intensity of goblet cells present; and the presence of nonepithelial cells, including inflammatory cells, melanocytic cells, and microorganisms. Atypical cells are identified by the presence of nuclear enlargement, hyperchromasia, irregular nuclear outline, and coarse nuclear chromatin and eventually by the presence of prominent nucleoli, under magnifications of 100×, 200×, and 400×. If different types of atypical cells are observed in the same specimen, more severe stage is considered.
Application of IC in the evaluation of lesions of melanocytic origin
Lesions of melanocytic origin are as common as epithelial tumors and include conjunctival racial melanosis, primary acquired melanosis (PAM), secondary melanosis, nevus, and melanoma. Although majority of the melanocytic lesions are benign, some can be malignant; therefore, distinguishing various conjunctival lesions is crucial(77 Shields CL, Schields JA. Tumors of the conjunctiva and cornea. Surv Ophthalmol. 2004;49(1):3-24.).
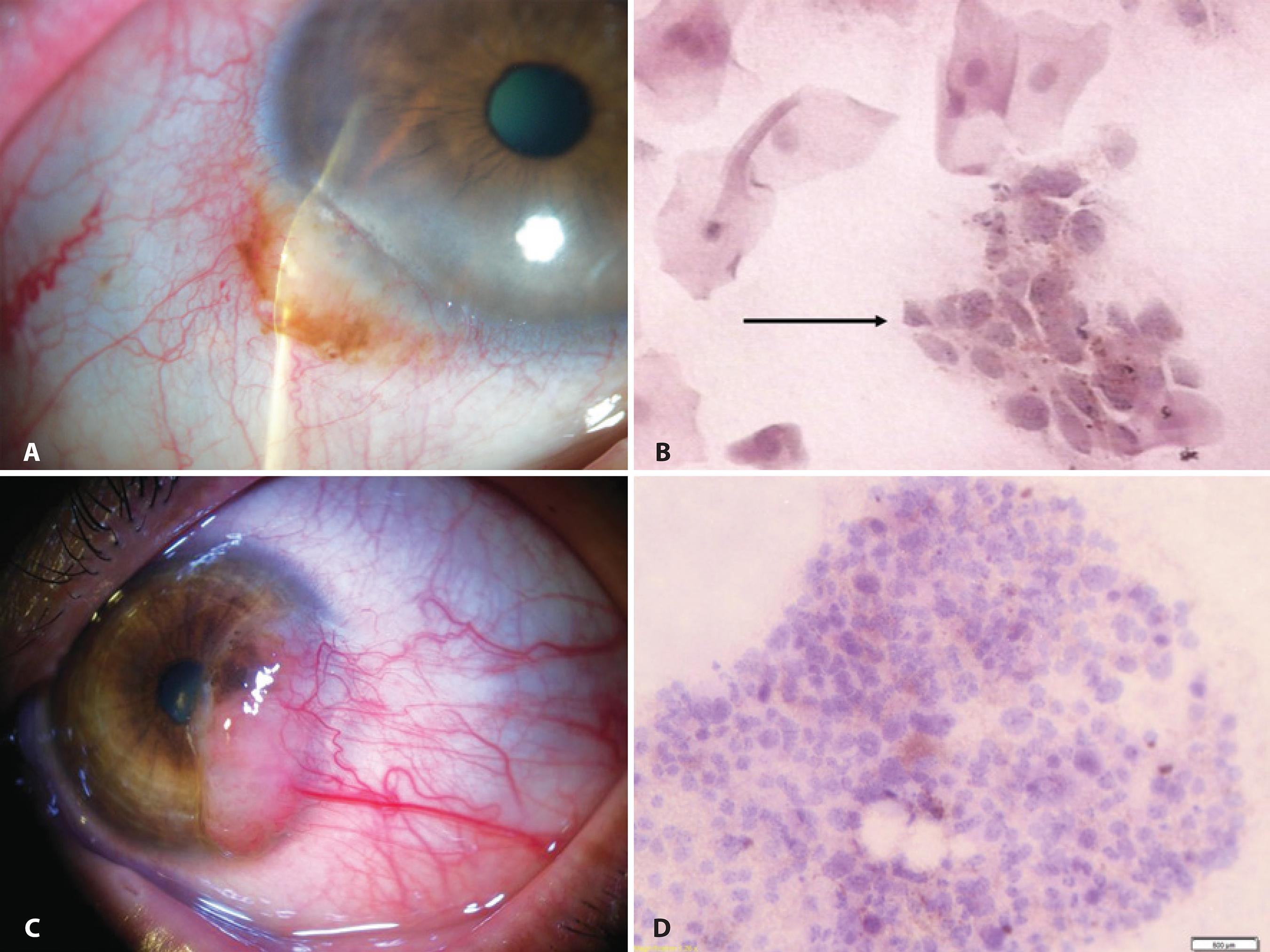
The first IC study of pigmented lesions from the conjunctiva was published in 1992(88 Paridaens AD, McCartney AC, Curling OM, Lyons CJ, Hungerford JL. Impression cytology of conjunctival melanosis and melanoma. Br J Ophthalmol. 1992;76(4):198-201.). A 73% correlation between IC and histopathology was observed in the diagnosis of 24 tumors, of which three were nevi, nine were melanomas, 10 were cases of PAM, and two were cases of secondary melanosis; examples are shown in figure 1. An increased nuclear-to-cytoplasmic (NC) ratio, an irregular nuclear chromatin pattern, the presence of large nucleoli, and the observation of mitosis and anisokaryosis were regarded as cytological features of malignancy in cells containing melanin. When the relative proportion of atypical melanocytes was low, lesions were cytologically diagnosed as premalignant melanosis equivalent to the histological diagnosis PAM with atypia. If cancerous cells were abundant, the diagnosis was suggestive of melanoma. The authors reported that repeated examinations may increase the sensitivity of the cytological technique. Authors stated that although a diagnostic biopsy may remain necessary for determination of the origin and extent of those lesions, recurrent tumors or suspicious areas may be biopsied less frequently using IC, thus reducing the risk of side effects and patient discomfort(88 Paridaens AD, McCartney AC, Curling OM, Lyons CJ, Hungerford JL. Impression cytology of conjunctival melanosis and melanoma. Br J Ophthalmol. 1992;76(4):198-201.).
Examples of IC in the evaluation of lesions of melanocytic origin: A) Anterior segment slit-lamp photograph demonstrating a conjunctival nevus. B) IC obtained from the same lesion demonstrating a cluster of nevus cells (arrow) among epithelial cells (Hematoxylin-Eosin staining; original magnification, 400×). C) Anterior segment slit-lamp photograph of malignant melanoma. D) IC obtained from the sample depicted in (C) demonstrated clusters of pleomorphic atypical tumor-dissociated cells with different sizes and anisokaryosis characterized by large and irregular nuclei in a cytomorphology not resembling epithelial cells. Brown melanin granules can be seen inside the cytoplasm of the malignant melanocytes (Hematoxylin-Eosin staining; original magnification, 200×).
In 2007, a study revealed 68 melanocytic conjunctival lesions, of which 31 were nevi, nine were melanoma, and 28 were PAM. The authors compared the Biopore membrane IC (referred to as “Biopore”) with exfoliative cytology (EC) in these lesions. Twenty-three of the 26 samples analyzed by Biopore and 20 of the 24 samples analyzed by EC correlated with the corresponding histology. Biopore accurately predicted the outcome in 88% and EC in 83% of the lesions. The authors concluded that Biopore could be used in cytology of melanocytic lesions and was easier and faster to interpret than EC. If difficult with Biopore, sampling of the fornix, caruncula, and ocular material in children could be performed by EC. Because some melanocytic lesions will be covered with one or more layers of normal epithelium, cytology could only provide a realistic picture of a lesion when it was able to sample deeper than the most superficial layer of epithelial cells. Biopore, however, may sample only the first layer of cells on the conjunctiva, unless it is repeated several times to acquire cells of deeper layers(99 Keijser S, Missotten GS, De Wolff-Rouendaal D, Verbeke SL, Van Luijk CM, Veselic- Charvat M, et al. Impression cytology of melanocytic conjunctival tumours using the Biopore membrane. Eur J Ophthalmol. 2007;17(4):501-6.). Similarly, IC with cellulose acetate filters was able to sample deeper layers when performed repeatedly(1010 Singh R, Joseph A, Umapathy T, Tint NL, Dua HS. Impression cytology of the ocular surface. Br J Ophthalmol. 2005;89(12):1655-9. Comment in: Br J Ophthalmol. 2008; 92(1):157-8.).
A case of a patient with an irregular pigmented lesion of the lower eyelid margin simulating malignant tumor, which was treated based on the results of IC and diagnosed with secondary melanosis by histology, was presented in 2009. The importance of IC was emphasized as an effective and safe method circumventing unnecessary and extensive procedures(1111 Oliveira CS, Barros J de N, Souza S de B, Cvintal T, Schellini SA. [Melanosis in eyelid margin with malignancy suspect: case report]. Arq Bras Oftalmol. 2009;72(5):706-9. Portuguese.).
A few melanocytic lesions, including four nevi and one melanoma, were examined in another study, and for such cases, results of both IC and histopathological features correlated(55 Larmande A, Timsit E. [Importance of cytodiganosis in ophthalmology: preliminar reporto f 8 cases of tumors of the sclero-corneal limbus]. Bull Soc Ophtalmol Fr. 1954;5:415-9. French.).
IC features of 35 conjunctival nevi from children and adults referred to as more noticeable were described in 2009. Approximately 26% were amelanotic but could be identified as localized areas of hyperemia. Using criteria derived from histology, IC was reported for conjunctival nevi when nests or clusters of nevus cells were observed within the epithelium layer containing or not containing mucous-secreting goblet cells. Epithelial cell layers demonstrated normal morphology, or, when the lesion was elevated, showed signs of squamous metaplasia (SM). IC confirmed the clinical diagnosis by demonstrating typical histopathological features of the superficial layers of conjunctival nevi in 91.4% of the cases. For amelanotic nevi, IC also allows differential diagnosis from other non-pigmented lesions(1212 Barros J de N, Lowen MS, Mascaro VLD, Andrade TP, Martins MC. Impression cytology features of conjunctival nevi referred as more noticeable. Arq Bras Oftalmol. 2009;72(2):205-10.).
Recently, a case of an amelanotic corneally displaced malignant conjunctival melanoma was described. The authors showed that IC performed prior to the treatment provided the first clue for the diagnosis later confirmed by histopathology. IC samples revealed abundant clusters of pleomorphic atypical tumor-dissociated cells with different sizes and anisokaryosis characterized by large and irregular nuclei with occasionally prominent nucleoli in a cytomorphology not resembling epithelial cells. Some of the atypical cells were spindle-shaped. Melanin pigment was absent. A few nonneoplastic squamous epithelial cells were also observed. Clinical diagnosis of amelanotic melanoma is challenging, and IC can assist in supporting the initial diagnosis when interpreted by a trained cytologist or under guidance of an ocular pathologist. For amelanotic melanoma, IC enables differential diagnosis from other nonpigmented lesions(1313 Barros J de N, Motono M, Costa FD, Cunha MC, Chojniak MM. Amelanotic corneally displaced malignant conjunctival melanoma: a case report evaluated with impression cytology. Arq Bras Oftalmol. 2014;77(1):57-9.). In addition, incisional biopsy of melanoma should be avoided because of the risk of local tumor dissemination(1414 Lim L, Madigan MC, Conway RM. Conjunctival melanoma: a review of conceptual and treatment advances. Clin Ophthalmol. 2013;6:521-31.).
Application of IC in the evaluation of lesions of epithelial origin
Ocular surface squamous neoplasia (OSSN) is the most common tumor of the ocular surface. The spectrum of OSSN ranges from mild to severe dysplasia, through full-thickness epithelial involvement, to invasive squamous cell carcinoma (SCC). Although the clinical appearance of a lesion can be suggestive of OSSN, tissue biopsy is necessary to confirm the diagnosis because the different stages of OSSN are extremely difficult to distinguish by slit-lamp biomicroscopy, with an accuracy of clinical diagnosis by experienced clinicians of approximately 40%(33 Lee GA, Hirst LW. Ocular surface squamous neoplasia. Surv Ophthalmol. 1995;39(6):429-50.).
It has been reported that IC immunostained with cytokeratin antibodies and HMB-45 was useful to differentiate a pigmented conjunctival seborrheic keratosis masquerading as malignant melanoma. IC disclosed basaloid cells intermixing with squamoid cells, and these cells demonstrated positive immunoreactivity to cytokeratin and no reactivity to HMB-45 and therefore were proven to represent an epithelium-derived tumor despite of being pigmented. This report illustrated that IC combined with immunocytochemical staining may be a valuable diagnostic aid in the differentiation of pigmented conjunctival tumors prior to treatment(1515 Tseng SH, Chen YT, Huang FC, Jin YT. Seborreic keratosis of conjunctiva simulating a malignant melanoma: an immunocytochemical study with impression cytology. Ophthalmology. 1999;106(8):1516-20.).
The published correlation rate with IC for predicting the subsequent histological
findings ranged between 77% (55/71) and 80% (20/25), and both cellulose
acetate(1616 Nolan GR, Hirst, LW, Wright RG, Bancroft BJ. Application of impression
cytology to the diagnosis of conjunctival neoplasms. Diagn Cytopathol.
1994;11(3):246-9.) and
Biopore membranes(1717 Tole DM, McKelvie PA, Daniell M. Reliability of impression cytology for
the diagnosis of ocular surface squamous neoplasia employing the biopore membrane. Br
J Ophthalmol. 2001;85(2):154-8. Comment in: Br J Ophthalmol.
2001;85(7):888.)
have been successfully used. The difficulty in interpreting these IC specimens caused
by the paucity of published criteria was overcome with the publication by Nolan et
al., who described in detail the cytomorphology of OSSN based on a high number of
cases. The following cytological criteria were used to diagnose intraepithelial OSSN:
nuclear enlargement (more than two times the dimensions of the nucleus of normal
conjunctival cells), presence of irregular nuclear contour, coarsely clumped
chromatin, nuclear pleomorphism, binucleation or multinucleation, and evident
nucleoli. When nuclear enlargement was less than twice the dimensions in normal
conjunctival cells or when it was limited to only few squamous cells, the specimen
was categorized as having atypical squamous cells indefinite for dysplasia. If none
of the abovementioned characteristics was observed, the specimen was regarded
negative. The finding of syncytia-like groupings, intraepithelial infiltration of
inflammatory cells, and macronucleoli may be suggestive of SCC in some
samples(1818 Nolan GR, Hirst, LW, Bancroft BJ. The citomorphology of ocular surface
squamous neoplasia by using impression cytology. Cancer.
2001;93(1):60-7.).
Nevertheless, at present, no unique specific cytological feature to differentiate SCC
from intraepithelial lesions in IC specimens has been identified. According to these
reports, there were no false-positives identified by IC(1616 Nolan GR, Hirst, LW, Wright RG, Bancroft BJ. Application of impression
cytology to the diagnosis of conjunctival neoplasms. Diagn Cytopathol.
1994;11(3):246-9.
17 Tole DM, McKelvie PA, Daniell M. Reliability of impression cytology for
the diagnosis of ocular surface squamous neoplasia employing the biopore membrane. Br
J Ophthalmol. 2001;85(2):154-8. Comment in: Br J Ophthalmol.
2001;85(7):888.
18 Nolan GR, Hirst, LW, Bancroft BJ. The citomorphology of ocular surface
squamous neoplasia by using impression cytology. Cancer.
2001;93(1):60-7.-1919 McKelvie PA, Daniell M, McNab A, Loughnan M, Santamaria JB. Squamous
cell carcinoma of the conjunctiva: a series of 26 cases. Br J Ophthalmol.
2002;86(2):168-73. Comment in: Br J Ophthalmol. 2002;86(12):1462.). Examples are shown in figure 2.
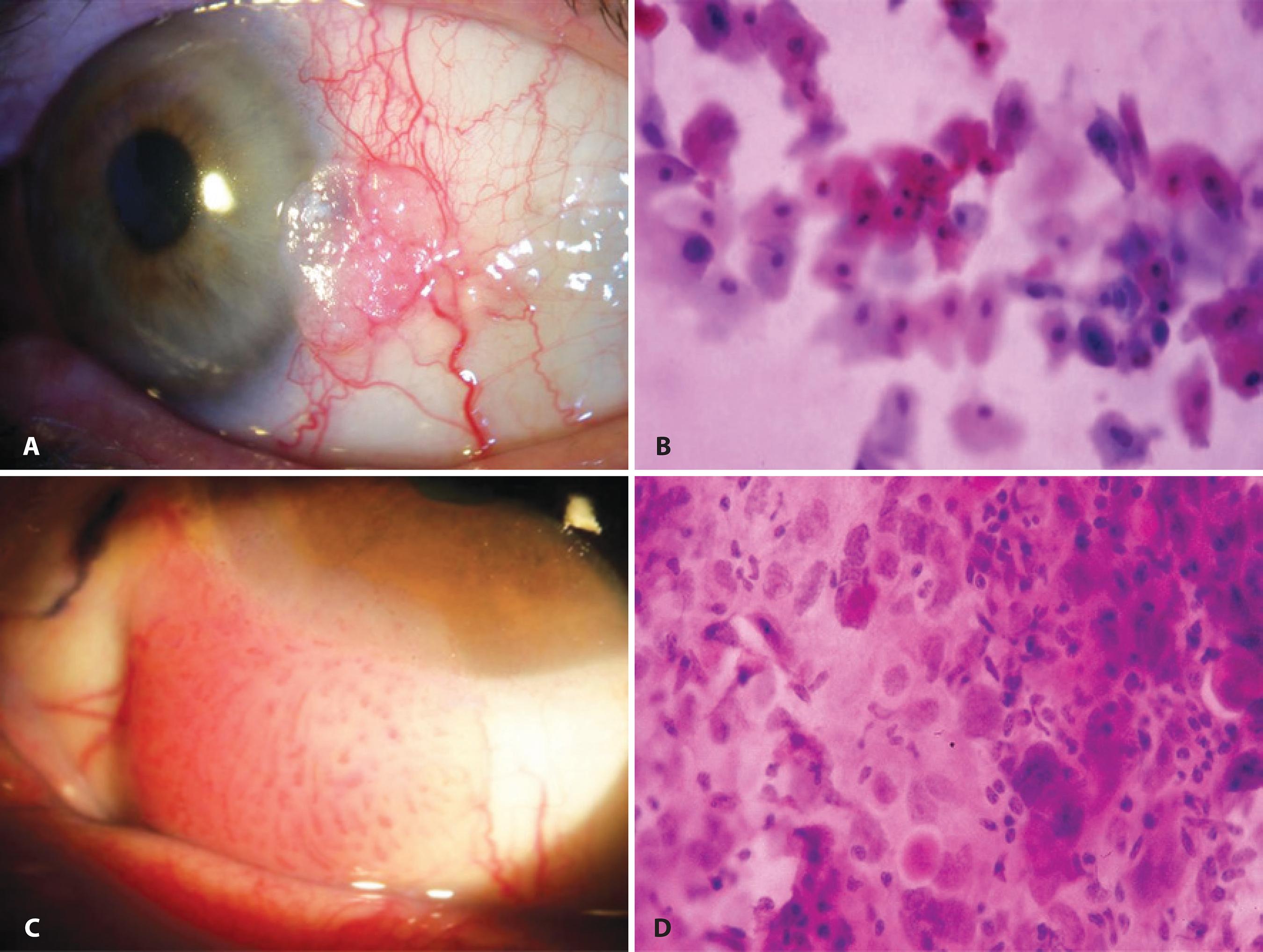
Example of IC in the evaluation of lesions of epithelial origin (ocular surface squamous neoplasia): A) Anterior segment slit-lamp photograph demonstrating conjunctival intraepithelial neoplasia. B) IC obtained from this lesion demonstrating atypical epithelial cells with mild nuclear enlargement, anisokaryosis, and remarkable hyperchromasia (Hematoxylin-Eosin staining; original magnification, 400×). C) Anterior segment slit-lamp photograph of invasive squamous cell carcinoma of the conjunctiva. D) IC demonstrating atypical epithelial cells showing nuclear enlargement, marked increase in the nuclear-to-cytoplasmic ratio, anisokaryosis, hyperchromasia, and a syncytial-like arrangement with absence of well-defined cytoplasmic borders (Hematoxylin-Eosin staining; original magnification, 400×).
Notably, the cytology of subclinical intraepithelial OSSN has already been described. The cytological pattern for OSSN with no clinically visible abnormality differed from that observed in the eyes with clinically detectable disease; there were often a few dysplastic cells lying within sheets of normal epithelium(1818 Nolan GR, Hirst, LW, Bancroft BJ. The citomorphology of ocular surface squamous neoplasia by using impression cytology. Cancer. 2001;93(1):60-7.).
In 2002, Chan et al. showed that IC obtained from surface cells overlying a pterygium was abnormal, typically exhibiting SM with increased goblet cell density. Altered cytology could also be demonstrated in the inferior bulbar conjunctiva and interpalpebral conjunctiva, without clinical evidence of pterygium. This suggested a graded series of changes occurring throughout the bulbar conjunctiva, with the most advanced occurring directly over the pterygium, confirming that it was indeed an ocular surface disorder(2020 Chan CM, Liu YP, Tan DT. Ocular surface changes in pterygium. Cornea. 2002;21(1):38-42.).
A case of conjunctiva-cornea intraepithelial neoplasia (CCIN) treated with topical mitomycin-C (MMC) and interferon alfa-2b in cycles was described in 2003. The patient was referred for LCSD and epithelial defect but IC specimens were suggestive of CCIN. After differentiation from LCSD by dye staining and IC, the patient was successfully treated(2121 Di Pascuale MA, Espana EM, Tseng SC. A case of conjunctiva-cornea intraepithelial neoplasia successfully treated with topical mitomycin C and interferon alfa-2b in cycles. Cornea. 2004;23(1):89-92.).
Another study found that IC had a positive and negative predictive value of 97.4% and 53.9%, respectively, when compared with histology(55 Larmande A, Timsit E. [Importance of cytodiganosis in ophthalmology: preliminar reporto f 8 cases of tumors of the sclero-corneal limbus]. Bull Soc Ophtalmol Fr. 1954;5:415-9. French.).
In 2009, Barros et al. described an index score modified from the Bethesda system for reporting cervical cytologic diagnoses to differentiate SCC from pre-invasive ocular surface lesions by IC (n=39). They revealed a predictive index score of ≥4.25 representing the best cut-off point for SCC with a sensitivity of 95%, specificity of 93%, positive predictive value of 95%, and negative predictive value of 93%(44 Barros JN, Lowen MS, Ballalai PL, Mascaro VL, Gomes JA, Martins MC. Predictive index to differentiate invasive squamous cell carcinoma from preinvasive ocular surface lesions by impression cytology. Br J Ophthalmol. 2009:93(2);209-14.). Four of seven parameters included in their regression model (nuclear enlargement > three-fold, syncytial-like groupings, increased NC ratio, and indistinct cytoplasm border) were visible using clinical confocal microscopy (CCM). One parameter (prominent nucleoli) is currently undetectable by CCM. The last two parameters (cellular hyperchromasia and eosinophilic cytoplasm) would require specific stains unavailable in vivo. The introduction of in vivo stains or biomarkers to better visualize these cellular details would be useful to improve image quality and to obtain more detailed information. A novel CCM specific index score to differentiate SCC from preinvasive ocular surface lesions is still necessary(2222 Parrozzani R, Lazzarini D, Dario A, Midena E. In vivo confocal microscopy of ocular surface squamous neoplasia. Eye (Lond). 2011;25(4):455-60.).
IC may be less sensitive for cases with keratotic lesions because an abundance of surface keratin can make sampling inaccurate(55 Larmande A, Timsit E. [Importance of cytodiganosis in ophthalmology: preliminar reporto f 8 cases of tumors of the sclero-corneal limbus]. Bull Soc Ophtalmol Fr. 1954;5:415-9. French.). To minimize this problem, authors have recommended collecting at least two samples over the same area from a suspicious lesion(44 Barros JN, Lowen MS, Ballalai PL, Mascaro VL, Gomes JA, Martins MC. Predictive index to differentiate invasive squamous cell carcinoma from preinvasive ocular surface lesions by impression cytology. Br J Ophthalmol. 2009:93(2);209-14.,2323 Kheirkhah A, Mahbod M, Farzbod F, Zavareh MK, Behrouz MJ, Hashemi H. Repeated applications of impression cytology to increase sensitivity for diagnosis of conjunctival intraepithelial neoplasia. Br J Ophthalmol. 2012;96(2):229-33.). For diagnosing OSSN, adding a second and a third evaluation of IC provided significantly more sensitivity than including only one(2323 Kheirkhah A, Mahbod M, Farzbod F, Zavareh MK, Behrouz MJ, Hashemi H. Repeated applications of impression cytology to increase sensitivity for diagnosis of conjunctival intraepithelial neoplasia. Br J Ophthalmol. 2012;96(2):229-33.). Nevertheless, it should considered that IC is very helpful, unless the result conflicts with the clinical scenario or when the actual clinical diagnosis is uncertain and the result is negative. In these cases, surgical biopsy needs to be performed for accurate diagnosis(55 Larmande A, Timsit E. [Importance of cytodiganosis in ophthalmology: preliminar reporto f 8 cases of tumors of the sclero-corneal limbus]. Bull Soc Ophtalmol Fr. 1954;5:415-9. French.,2323 Kheirkhah A, Mahbod M, Farzbod F, Zavareh MK, Behrouz MJ, Hashemi H. Repeated applications of impression cytology to increase sensitivity for diagnosis of conjunctival intraepithelial neoplasia. Br J Ophthalmol. 2012;96(2):229-33.).
In the study by Ballalai et al., 0.02% topical MMC was used to treat patients with OSSN. Before the treatment, cytology showed the presence of neoplastic cells in patients with primary tumors, avoiding surgical biopsy and treatment delay(2424 Ballalai PL, Erwenne CM, Martins MC, Lowen MS, Barros JN. Long-term results of topical mitomycin C 0.02% for primary and recurrent conjunctival-corneal intraepithelial neoplasia. Ophthal Plast Reconstr Surg. 2009;25(4):296-9.).
A great advantage of using IC is the preservation of limbal stem cells, responsible for renewal of corneal epithelium throughout life. In most OSSN cases, the lesions affect predominantly the limbus and have a tendency to recur. IC offers a safer tool for diagnosis than repeated biopsy(44 Barros JN, Lowen MS, Ballalai PL, Mascaro VL, Gomes JA, Martins MC. Predictive index to differentiate invasive squamous cell carcinoma from preinvasive ocular surface lesions by impression cytology. Br J Ophthalmol. 2009:93(2);209-14.). Moreover, IC can be used during post-surgery follow-up to identify any recurrence of the disease as well as the effects of topical treatment such as chemotherapy with antineoplastic drugs like MMC(2525 McKelvie P, Daniell M. Impression cytology following mitomycin C therapy for ocular surface squamous neoplasia. Br J Ophthalmol. 2001;85(9):1115-9.).
Application of IC following tumor treatment
Treatment for OSSN has historically been surgery but nonsurgical interventions have also been adopted. Adjunctive therapies allowed the treatment of subclinical disease at a site different from that of the clinically evident tumor. Nevertheless, topical chemotherapeutic drugs can be potentially toxic to the ocular surface(2626 Adler E, Turner JR, Stone DU. Ocular surface squamous neoplasia: a survey of changes in the standard of care from 2003 to 2012. Cornea. 2013;32(12):1558-61.). In 2001, IC was used to study the effects of topical MMC in the treatment of OSSN; 0.04% MMC induced cell death mainly by apoptosis or rarely by necrosis and changes induced in the ocular surface persisted for at least 8 months. MMC induced cytomegaly, cytoplasmic vacuolation, nucleomegaly with nuclear wrinkling, and binucleation or multinucleation. The N/C ratio in these enlarged cells was normal. These changes mimicked those observed following radiation therapy in uterine cervical cancer. Nuclear and cell size increased along with increasing N/C ratio in some dysplastic cells(2525 McKelvie P, Daniell M. Impression cytology following mitomycin C therapy for ocular surface squamous neoplasia. Br J Ophthalmol. 2001;85(9):1115-9.). Yamamoto et al. used IC during diagnosis and follow-up, resulting in successful treatment with 5-fluorouracil of an intraepithelial OSSN with LSCD that was refractive to topical MMC(2727 Yamamoto N, Ohmura T, Sizuki H, Shirasawa H. Successful treatment with 5-fluorouracil of conjunctival intraepithelial neoplasia refractive to mitomycin C. Ophthalmology. 2002;109(2):249-52. Comment in: Ophthalmology. 2003; 110(4):625-6; author reply 626; Ophthalmology. 2003; 110(7):1289; Ophthalmology. 2003;110(6):1262-3; author reply 1263.).
Dogru et al. evaluated the tear function and ocular surface alterations in patients with primary intraepithelial OSSN before and after treatment with 0.04% topical MMC. Initial IC specimens showed loss of goblet cells, higher grades of SM, and areas of isolated keratinized, binucleated, and actively mitotic disfigured epithelial cells in all patients. The mean goblet cell density and SM grade were observed to having significantly improved at the last visit of the patients. IC proved useful in attaining the diagnosis of OSSN, evaluating the effect of treatment and showing MMC-related long-term changes on the ocular surface(2828 Dogru M, Erturk H, Shimazaki J, Tsubota K, Gul M. Tear function and ocular surface changes with topical mitomycin C treatment for primary corneal intraepithelial neoplasia. Cornea. 2003;22(7):627-39.).
In 2005, Prabhasawat et al. reported complete tumor regression observed clinically and by IC, demonstrating the efficacy of 0.002% topical MMC as an adjunctive and alternative treatment in primary and recurrent OSSN; IC exhibited tumor-free specimens with cellular elongation as a result of chemotherapy(2929 Prabhasawat P, Tarinvorakup P, Tesavibul N, Uiprasertkul M, Kosrirukvongs P, Booranapong W, et al. Topical 0.002% mitomycin C for the treatment of conjunctival-corneal intraepithelial neoplasia and squamous cell carcinoma. Cornea. 2005; 24(4):443-8.).
Notably, cytological changes mimicking malignancy have been reported in conjunctiva up to 6 weeks following topical MMC therapy. Nevertheless, there are features which help to differentiate these changes: epithelial cells affected by the drug show a proportionate increase in both cytoplasm and nucleus, preserving a normal NC ratio (cytomegaly), unlike the case of increased NC ratio (cariomegaly) in OSSN. The distinction of MMC-related changes from OSSN cells in IC specimens can be performed when the cell border is clearly visible and the N/C ratio can be estimated. Differentiation becomes difficult in cells with large hyperchromatic nuclei where the cell outline is not clearly defined because of overlapping cells or attenuation of the vacuolated cytoplasm. Therefore, studying such cells for which cell size can be clearly assessed is crucial(2525 McKelvie P, Daniell M. Impression cytology following mitomycin C therapy for ocular surface squamous neoplasia. Br J Ophthalmol. 2001;85(9):1115-9.).
Westekemper et al. examined ocular surface integrity of ten patients with large and diffuse conjunctival melanoma who underwent proton beam radiation. The IC revealed conjunctival SM in nine cases, indicating a radiogenic, persisting disturbance in the differentiation of the conjunctival epithelial cells. The tear film instability correlated with goblet cell loss and meibomian gland dysfunction(3030 Westekemper H, Anastassiou G, Sauerwein W, Chauvel P, Bornfeld N, Steuhl KP, et al. [Analysis of ocular surface alterations following proton beam radiation in eyes with conjunctival malignant melanoma]. Ophthalmologe. 2006;103(7):588-95. German.).
The use of topical MMC has been described by some authors not only for OSSN but also for melanocytic lesions such as PAM with atypia. However, its prolonged use may be associated with a high incidence of complications like LSCD. IC diagnoses ocular surface lesions and also evaluates possible local side effects following treatment. Five cases of proven LSCD by IC resulting as a complication of topical treatment with MMC for PAM with atypia have been reported(3131 Lichtinger A, Pe'er J, Frucht-Pery J, Solomon A. Limbal stem cell deficiency after topical mitomycin C therapy for primary acquired melanosis with atipia. Ophthalmology. 2010;117(3):431-7.).
Rodríguez Feijoo et al. reported that making an accurate differential diagnosis between keratoacanthoma and SCC by histology as well as carrying out close monitoring after surgery due to the possibility of relapse and conversion to SCC is important. Therefore, they proposed the use of IC as a method for monitoring such patients. After the treatment, IC exhibited large altered epithelial cells with intracellular union changes and an NC ratio of 1:20. A second series of IC tests performed 3 months after the first series showed the same results(3232 Rodriguez Feijoo D, Romero Moreno I, Lopez Gutierrez C, Usabiaga Usandizaga M, Cisneros Carpio M, Amias Gorostiza A, et al. [Conjunctival keratoacanthoma: diagnosis, treatment and monitoring by conjunctival impression cytology]. Arch Soc Esp Oftalmol. 2012;87(3):82-5. Spanish.).
Recently, Faramarzi and Feizi evaluated the efficacy of perilesional/ subconjunctival injections of an antivascular endothelial growth factor, bevacizumab, for treatment of a group of 10 eyes with primary OSSN. Based on clinical presentation and IC results, they showed that the treatment was effective in terms of decreasing the size of conjunctival OSSN when the lesion was limited to the conjunctiva. However, this therapy had no effect on corneal extensions of the OSSN(3333 Faramarzi A, Feizi S. Subconjunctival bevacizumab injection for ocular surface squamous neoplasia. Cornea. 2013;32(7):998-1001.).
Application of IC in the evaluation of tumors of sebaceous origin
In 2003, Sawada et al. demonstrated that IC detected conjunctival intraepithelial invasion from sebaceous cell carcinoma of the eyelid in four patients with severe unilateral blepharoconjunctivitis. IC showed numerous inflammatory cells and abnormal tumor cells with atypia and characteristic cytoplasmic vacuoles, consistent with dissolved sebaceous contents(3434 Sawada Y, Fischer JL, Verm AM, Harrison AR, Yuan C, Huang AJ. Detection by impression cytologic analysis of conjunctival intraepithelial invasion from eyelid sebaceous cell carcinoma. Ophthalmology. 2003;110(10):2045-50.-3535 Santo RM, Bordon PB, Barros J de N, Schellini SA, Erwenne CM. Tumores da superficie ocular. In: Gomes JA, Alves MR, editores. Superficie ocular cornea limbo conjuntiva filme lacrimal. 2nd ed. Rio de Janeiro: Cultura Medica; 2011. p. 141-70.). They represented areas where lipid was contained before it was dissolved by alcohol; an example is shown in figure 3. The diagnosis was confirmed by histology from full-thickness wedge resection of the eyelids. When pagetoid spread in advanced cases of sebaceous cell carcinoma results in a superficial or full-thickness replacement of the normal conjunctival epithelium with tumor cells, the superficial abnormal cells can be detected by IC. However, areas on the conjunctiva with pagetoid spread may exist without full-thickness epithelial disease. In such cases, IC may sample only the superficial normal epithelial cells and may fail to detect the tumor cells concealed in the deeper layers. Because sebaceous carcinoma can masquerade as several benign conditions such as blepharitis, the investigations should include IC and biopsy in cases that are not responsive to medication. If cellular atypia is present, a full-thickness lid biopsy should be performed(3434 Sawada Y, Fischer JL, Verm AM, Harrison AR, Yuan C, Huang AJ. Detection by impression cytologic analysis of conjunctival intraepithelial invasion from eyelid sebaceous cell carcinoma. Ophthalmology. 2003;110(10):2045-50.).
A) Anterior segment slit-lamp photograph demonstrating conjunctival intraepithelial invasion from a sebaceous cell carcinoma of the eyelid. B) IC showing inflammatory cells and a tumor cell with atypia, abnormal prominent nucleoli, and characteristic cytoplasmic vacuoles (arrow) consistent with intracellular dissolved sebaceous contents. These cells were PAS-negative, suggestive of non-goblet cell origin. No evident goblet cells were observed in the epithelium (PAS and Hematoxylin-Eosin staining; original magnification, 400×).
Application of IC in the evaluation of ulcerative eyelid malignancy
Thirty-two histopathologically proven malignant eyelid lesions diagnosed over a 2-year period, including 13 basal cell carcinomas, 11 sebaceous carcinomas, four SCC, two malignant melanomas, and two poorly differentiated carcinomas, formed the study group described very recently. The results of IC were compared with those of obtained by histopathological analysis in the study group and with an age- and sex-matched group of benign cases as controls. The sensitivity of IC was 84% (27/32) for the diagnosis of malignancy and 28% (9/32) for categorization of the type of malignancy. Because of its low sensitivity in terms of cytological categorization of the type of malignancy, IC cannot be recommended in the primary diagnosis of eyelid malignancies. Nevertheless, with experience and improvement in the technique, it may prove to be a useful tool in deciding future management, particularly in recurrences of histopathologically confirmed eyelid malignancies, where biopsies may be avoided(3636 Sen S, Lyngdoh AD, Pushker N, Meel R, Bajaj MS, Chawla B. Impression cytology diagnosis of ulcerative eyelid malignancy. Cytopathology. Forthcoming 2014.).
IC , imaging, and histopathology
Clinical examination is subjective, is unable to assess cellular morphology, and may not detect subclinical microscopic diseases. A surgical biopsy to confirm the resolution of an OSSN could miss small residual lesions. Thus, an incisional biopsy may miss lesions that were not included in the excised tissue. The biopsy is based on clinically visible disease and may produce false-negative results. The false clinical impression of tumor resolution can result in premature termination of topical treatment and an increased risk of recurrence. These lesions can spread along the basal conjunctival layers far beyond the clinical lesion, and thus may be missed clinically. Excisional biopsy, despite being the most traditional and accurate means, may induce conjunctival scarring, LSCD, and visually disturbing corneal scarring. Due to the multifocal nature of OSSN, surgical excision results in extensive collateral damage to adjacent areas of normal epithelium(3737 Shousha MA, Karp CL, Canto AP, Hodson K, Oellers P, Kao AA, et al. Diagnosis of ocular surface lesions using ultra-high-resolution optical coherence tomography. Ophthalmology. 2013;120(5):883-91.).
In addition to IC, newer diagnostic techniques including CCM(2222 Parrozzani R, Lazzarini D, Dario A, Midena E. In vivo confocal microscopy of ocular surface squamous neoplasia. Eye (Lond). 2011;25(4):455-60.), toluidine blue(3838 Romero IL, Barros J de N, Martins MC, Ballalai PL. The use of 1% toluidine blue eye drops in the diagnosis of ocular surface squamous neoplasia. Cornea. 2013;32(1):36-9.), and ultra-high resolution anterior segment optical coherence tomography (UHR-OCT)(3939 Thomas BJ, Galor A, Nanji AA, El Sayyad F, Wang J, Dubovy SR, et al. Ultra high-resolution anterior segment optical coherence tomography in the diagnosis and management of ocular surface squamous neoplasia. Ocul Surf. 2014;12(1):46-58.) have been reported to aid in the diagnosis of OSSN. All these techniques have limitations and require skilled professionals to perform the tests and interpret the results. IC assesses only superficial layers of cells, which are not always representative of deeper layers, whereas CCM does not provide cross-sectional views hence not being useful for determining the vertical and horizontal extent of the lesion. Therefore, ensuring that the exact same area of the ocular surface is analyzed by CMM at follow-up examinations can be challenging. Regarding UHR-OCT, lesions, which are thickly pigmented lesions or show leukoplakia, tend to impede the penetration of light to deeper tissues, impairing the determination of the posterior limit of the lesion. Optical information at the time of the study was not sufficient to study signs of cellular atypia and was not able to rule out microinvasion(3737 Shousha MA, Karp CL, Canto AP, Hodson K, Oellers P, Kao AA, et al. Diagnosis of ocular surface lesions using ultra-high-resolution optical coherence tomography. Ophthalmology. 2013;120(5):883-91.). Similar to UHR-OCT, IC may not distinguish in situ from minimally invasive disease(66 Tananuvat N, Lertprasertsuk N, Mahanupap P, Noppanakeepong P. Role of impression cytology in diagnosis of ocular surface neoplasia. Cornea. 2008;27(3):269-74.).
Each imaging modality has the advantage of being noninvasive, and each has been is useful in the detection of OSSN. However, both UBM and some confocal microscopy devices require contact with the ocular surface, increasing both the length of time and technical expertise required for their performance. Furthermore, although confocal microscopy has the advantage of detailing individual cell morphology, which is currently outside of the capability of UHR-OCT, it targets a very limited area. OCT has the advantage of higher-resolution images, but shadowing may occur in thick lesions or those with leukoplakia. UBM has greater depth of penetration but lower resolution and cannot evaluate the epithelial versus subepithelial nature of a lesion. No data are available regarding inter- and intraobserver variability for the assessment of ocular surface pathology using the UHR-OCT(4040 Dart J. Impression cytology of the ocular surface - research tool or routine clinical investigation? Br J Ophthalmol. 1997;81(11):930.). Despite UHR-OCT having the advantage over IC of providing relatively deeper scans of the entire epithelium and the underlying tissue, it cannot reliably detect invasion(3737 Shousha MA, Karp CL, Canto AP, Hodson K, Oellers P, Kao AA, et al. Diagnosis of ocular surface lesions using ultra-high-resolution optical coherence tomography. Ophthalmology. 2013;120(5):883-91.). In addition, UHR-OCT machines are largely limited to academic institutions(3939 Thomas BJ, Galor A, Nanji AA, El Sayyad F, Wang J, Dubovy SR, et al. Ultra high-resolution anterior segment optical coherence tomography in the diagnosis and management of ocular surface squamous neoplasia. Ocul Surf. 2014;12(1):46-58.).
IC may be an inexpensive tool that can be used in the outpatient clinic setting to help provide an objective evaluation of suspicious lesions that enables patients to make better informed decisions regarding the treatment requirements. Results of IC may also help the ophthalmologist decide whether incisional or excisional biopsy should be performed and whether any other associated procedures, such as freeze-thaw cryotherapy of the sclera/limbus and/or ethanol application to the cornea, are required. IC provides a flat mount of an area as large as the size of the applied filter paper with well-preserved morphology. In comparison, conjunctival smears destroy much of the morphological information and conjunctival biopsies provide information from a relatively small sample of the surface epithelium, both because of the difficulty of preparing flat mounts and because of their small sizes. Therefore, IC is ideal for sampling the corneal epithelium(4040 Dart J. Impression cytology of the ocular surface - research tool or routine clinical investigation? Br J Ophthalmol. 1997;81(11):930.).
FINAL COMMENTS
OSSN masquerades as scar tissue or pannus; in addition, it can appear in association with pterygia(33 Lee GA, Hirst LW. Ocular surface squamous neoplasia. Surv Ophthalmol. 1995;39(6):429-50.). Thus, the question of using IC for the detection of OSSN in the setting of concomitant ocular surface disease requires further studies. Recently, Barros et al. reported that IC demonstrated high agreement with the results of the histopathological analysis for detecting atypical epithelial cells from unsuspected OSSN in cases of pterygia from Brazil, showing unsuspected and associated OSSN cells in 13 specimens (40%)(4141 Barros J de N, Lowen MS, Moraes-Filho MN, Martins MC. Impression cytology for detection of unsuspected ocular surface squamous neoplasia cells in pterygia. Arq Bras Oftalmol. 2014;77(5):305-9.).
IC presents great advantages: (1) it provides a source of intact and well-preserved epithelial cells from the ocular surface in any type of ocular surface pathology; (2) it is a nonsurgical, easy-to-perform, quick, and inexpensive technique that can always be performed on an outpatient basis; (3) only topical anesthesia is required, and no side effects or contraindications have ever been noted and thus it can be applied to children; (4) repeated IC sampling in the same patient over time is an excellent way to demonstrate changes due to a certain event, to monitor the progress of a disease, or to follow the effect of a therapeutic intervention; (5) IC maintains cell-to-cell contacts, preventing the problems of EC or brush cytology, which may destroy much of the cell morphology, cause overlapping of cells, and hamper clear visualization of the in vivo arrangement of the cells; (6) IC samples can be processed using any type of microscopy in addition to polymerase chain reaction (PCR), immunoblotting analyses, and/or flow cytometry. Based on all these advantages, IC has become the technique of choice for sampling ocular surface epithelium for being a very useful research tool in both basic and clinical aspects(4141 Barros J de N, Lowen MS, Moraes-Filho MN, Martins MC. Impression cytology for detection of unsuspected ocular surface squamous neoplasia cells in pterygia. Arq Bras Oftalmol. 2014;77(5):305-9.-4242 Calonge M, Diebold Y, Saez V, Enriquez de Salamanca A, Garcia-Vasquez C, Corrales RM, et al. Impression cytology of the ocular surface: a review. Exp Eye Res. 2004;78(3):457-72.). Although IC cannot replace histology, it has an important role in the diagnosis and management of patients with OSSN in a less invasive manner. A tool such as IC that aids the diagnosis of OSSN is of particular relevance to Brazilian patients, who live in a country closer to the equator line, with a climate and an ultraviolet-B light index that may contribute to the appearance and development of such tumors in its population. The correlation between sun exposure and OSSN has been well established(4343 Newton R, Ferlay J, Reeves G, Beral V, Parkin DM. Effect of ambient solar ultraviolet radiation on incidence of squamous-cell carcinoma of the eye. Lancet. 1996; 347(9013):1450-1.). The importance of IC lies in its capacity to detect both the presence and extent of OSSN when the clinical diagnosis is difficult, to detect subclinical disease and follow up on previously diagnosed disease(44 Barros JN, Lowen MS, Ballalai PL, Mascaro VL, Gomes JA, Martins MC. Predictive index to differentiate invasive squamous cell carcinoma from preinvasive ocular surface lesions by impression cytology. Br J Ophthalmol. 2009:93(2);209-14.,1818 Nolan GR, Hirst, LW, Bancroft BJ. The citomorphology of ocular surface squamous neoplasia by using impression cytology. Cancer. 2001;93(1):60-7.). Expertise in IC is acquired by continuing experience including close reviews, correlation with all possible subsequent histology specimens, and clinicopathological correlation. This enables the cytologist to gain familiarity and become aware of the eventual difficult areas such as keratinizing lesions(1717 Tole DM, McKelvie PA, Daniell M. Reliability of impression cytology for the diagnosis of ocular surface squamous neoplasia employing the biopore membrane. Br J Ophthalmol. 2001;85(2):154-8. Comment in: Br J Ophthalmol. 2001;85(7):888.).
Because IC has not presented sensitivity and specificity of 100%, the prospective use of the Barros score for predicting SCC needs to be further evaluated using a large number of patients(44 Barros JN, Lowen MS, Ballalai PL, Mascaro VL, Gomes JA, Martins MC. Predictive index to differentiate invasive squamous cell carcinoma from preinvasive ocular surface lesions by impression cytology. Br J Ophthalmol. 2009:93(2);209-14.). The development of a novel immunohistochemical analysis with a proliferative index such as that for Ki-67 could aid in IC specimens becoming a diagnostic marker for OSSN and in obtaining prognostic information regarding the risk of recurrence in a manner similar to the current use of histology(4444 Ohara M, Sotozono C, Tsuchihashi Y, Kinoshita S. Ki-67 labeling index as a marker of malignancy in ocular surface neoplasms. Jpn J Ophthalmol. 2004;48(6):524-9.). This combination of IC and immunocytochemistry was first described by Krenzer and Freddo in normal human conjunctiva in 1997, enabling the simultaneous evaluation of IC specimens for immunoreactivity to cytokeratin and morphological details(4545 Krenzer KL, Freddo TF. Cytokeratin expression in normal human bulbar conjunctiva obtained by impression cytology. Invest Ophthalmol Vis Sci. 1997;38(1):142-52.). Nevertheless, as indicated in this review, there was only a single case(1515 Tseng SH, Chen YT, Huang FC, Jin YT. Seborreic keratosis of conjunctiva simulating a malignant melanoma: an immunocytochemical study with impression cytology. Ophthalmology. 1999;106(8):1516-20.) using this combined technique in the evaluation of an ocular surface tumor. Thus, the sensitivity and reliability of IC combined with immunocytochemical staining in the differentiation of ocular surface tumors need further evaluation in large-scale studies.
-
Funding: No specific financial support was available for this study.
REFERENCES
-
1Varde MA, Biswas J. Ocular surface tumors. Oman J Ophthalmol. 2009;2(1):1-2.
-
2Rudkin AK, Dodd T, Muecke JS. The differential diagnosis of localised amelanotic limbal lesions: a review of 162 consecutive excisions. Br J Ophthalmol. 2011:95(3):350-4.
-
3Lee GA, Hirst LW. Ocular surface squamous neoplasia. Surv Ophthalmol. 1995;39(6):429-50.
-
4Barros JN, Lowen MS, Ballalai PL, Mascaro VL, Gomes JA, Martins MC. Predictive index to differentiate invasive squamous cell carcinoma from preinvasive ocular surface lesions by impression cytology. Br J Ophthalmol. 2009:93(2);209-14.
-
5Larmande A, Timsit E. [Importance of cytodiganosis in ophthalmology: preliminar reporto f 8 cases of tumors of the sclero-corneal limbus]. Bull Soc Ophtalmol Fr. 1954;5:415-9. French.
-
6Tananuvat N, Lertprasertsuk N, Mahanupap P, Noppanakeepong P. Role of impression cytology in diagnosis of ocular surface neoplasia. Cornea. 2008;27(3):269-74.
-
7Shields CL, Schields JA. Tumors of the conjunctiva and cornea. Surv Ophthalmol. 2004;49(1):3-24.
-
8Paridaens AD, McCartney AC, Curling OM, Lyons CJ, Hungerford JL. Impression cytology of conjunctival melanosis and melanoma. Br J Ophthalmol. 1992;76(4):198-201.
-
9Keijser S, Missotten GS, De Wolff-Rouendaal D, Verbeke SL, Van Luijk CM, Veselic- Charvat M, et al. Impression cytology of melanocytic conjunctival tumours using the Biopore membrane. Eur J Ophthalmol. 2007;17(4):501-6.
-
10Singh R, Joseph A, Umapathy T, Tint NL, Dua HS. Impression cytology of the ocular surface. Br J Ophthalmol. 2005;89(12):1655-9. Comment in: Br J Ophthalmol. 2008; 92(1):157-8.
-
11Oliveira CS, Barros J de N, Souza S de B, Cvintal T, Schellini SA. [Melanosis in eyelid margin with malignancy suspect: case report]. Arq Bras Oftalmol. 2009;72(5):706-9. Portuguese.
-
12Barros J de N, Lowen MS, Mascaro VLD, Andrade TP, Martins MC. Impression cytology features of conjunctival nevi referred as more noticeable. Arq Bras Oftalmol. 2009;72(2):205-10.
-
13Barros J de N, Motono M, Costa FD, Cunha MC, Chojniak MM. Amelanotic corneally displaced malignant conjunctival melanoma: a case report evaluated with impression cytology. Arq Bras Oftalmol. 2014;77(1):57-9.
-
14Lim L, Madigan MC, Conway RM. Conjunctival melanoma: a review of conceptual and treatment advances. Clin Ophthalmol. 2013;6:521-31.
-
15Tseng SH, Chen YT, Huang FC, Jin YT. Seborreic keratosis of conjunctiva simulating a malignant melanoma: an immunocytochemical study with impression cytology. Ophthalmology. 1999;106(8):1516-20.
-
16Nolan GR, Hirst, LW, Wright RG, Bancroft BJ. Application of impression cytology to the diagnosis of conjunctival neoplasms. Diagn Cytopathol. 1994;11(3):246-9.
-
17Tole DM, McKelvie PA, Daniell M. Reliability of impression cytology for the diagnosis of ocular surface squamous neoplasia employing the biopore membrane. Br J Ophthalmol. 2001;85(2):154-8. Comment in: Br J Ophthalmol. 2001;85(7):888.
-
18Nolan GR, Hirst, LW, Bancroft BJ. The citomorphology of ocular surface squamous neoplasia by using impression cytology. Cancer. 2001;93(1):60-7.
-
19McKelvie PA, Daniell M, McNab A, Loughnan M, Santamaria JB. Squamous cell carcinoma of the conjunctiva: a series of 26 cases. Br J Ophthalmol. 2002;86(2):168-73. Comment in: Br J Ophthalmol. 2002;86(12):1462.
-
20Chan CM, Liu YP, Tan DT. Ocular surface changes in pterygium. Cornea. 2002;21(1):38-42.
-
21Di Pascuale MA, Espana EM, Tseng SC. A case of conjunctiva-cornea intraepithelial neoplasia successfully treated with topical mitomycin C and interferon alfa-2b in cycles. Cornea. 2004;23(1):89-92.
-
22Parrozzani R, Lazzarini D, Dario A, Midena E. In vivo confocal microscopy of ocular surface squamous neoplasia. Eye (Lond). 2011;25(4):455-60.
-
23Kheirkhah A, Mahbod M, Farzbod F, Zavareh MK, Behrouz MJ, Hashemi H. Repeated applications of impression cytology to increase sensitivity for diagnosis of conjunctival intraepithelial neoplasia. Br J Ophthalmol. 2012;96(2):229-33.
-
24Ballalai PL, Erwenne CM, Martins MC, Lowen MS, Barros JN. Long-term results of topical mitomycin C 0.02% for primary and recurrent conjunctival-corneal intraepithelial neoplasia. Ophthal Plast Reconstr Surg. 2009;25(4):296-9.
-
25McKelvie P, Daniell M. Impression cytology following mitomycin C therapy for ocular surface squamous neoplasia. Br J Ophthalmol. 2001;85(9):1115-9.
-
26Adler E, Turner JR, Stone DU. Ocular surface squamous neoplasia: a survey of changes in the standard of care from 2003 to 2012. Cornea. 2013;32(12):1558-61.
-
27Yamamoto N, Ohmura T, Sizuki H, Shirasawa H. Successful treatment with 5-fluorouracil of conjunctival intraepithelial neoplasia refractive to mitomycin C. Ophthalmology. 2002;109(2):249-52. Comment in: Ophthalmology. 2003; 110(4):625-6; author reply 626; Ophthalmology. 2003; 110(7):1289; Ophthalmology. 2003;110(6):1262-3; author reply 1263.
-
28Dogru M, Erturk H, Shimazaki J, Tsubota K, Gul M. Tear function and ocular surface changes with topical mitomycin C treatment for primary corneal intraepithelial neoplasia. Cornea. 2003;22(7):627-39.
-
29Prabhasawat P, Tarinvorakup P, Tesavibul N, Uiprasertkul M, Kosrirukvongs P, Booranapong W, et al. Topical 0.002% mitomycin C for the treatment of conjunctival-corneal intraepithelial neoplasia and squamous cell carcinoma. Cornea. 2005; 24(4):443-8.
-
30Westekemper H, Anastassiou G, Sauerwein W, Chauvel P, Bornfeld N, Steuhl KP, et al. [Analysis of ocular surface alterations following proton beam radiation in eyes with conjunctival malignant melanoma]. Ophthalmologe. 2006;103(7):588-95. German.
-
31Lichtinger A, Pe'er J, Frucht-Pery J, Solomon A. Limbal stem cell deficiency after topical mitomycin C therapy for primary acquired melanosis with atipia. Ophthalmology. 2010;117(3):431-7.
-
32Rodriguez Feijoo D, Romero Moreno I, Lopez Gutierrez C, Usabiaga Usandizaga M, Cisneros Carpio M, Amias Gorostiza A, et al. [Conjunctival keratoacanthoma: diagnosis, treatment and monitoring by conjunctival impression cytology]. Arch Soc Esp Oftalmol. 2012;87(3):82-5. Spanish.
-
33Faramarzi A, Feizi S. Subconjunctival bevacizumab injection for ocular surface squamous neoplasia. Cornea. 2013;32(7):998-1001.
-
34Sawada Y, Fischer JL, Verm AM, Harrison AR, Yuan C, Huang AJ. Detection by impression cytologic analysis of conjunctival intraepithelial invasion from eyelid sebaceous cell carcinoma. Ophthalmology. 2003;110(10):2045-50.
-
35Santo RM, Bordon PB, Barros J de N, Schellini SA, Erwenne CM. Tumores da superficie ocular. In: Gomes JA, Alves MR, editores. Superficie ocular cornea limbo conjuntiva filme lacrimal. 2nd ed. Rio de Janeiro: Cultura Medica; 2011. p. 141-70.
-
36Sen S, Lyngdoh AD, Pushker N, Meel R, Bajaj MS, Chawla B. Impression cytology diagnosis of ulcerative eyelid malignancy. Cytopathology. Forthcoming 2014.
-
37Shousha MA, Karp CL, Canto AP, Hodson K, Oellers P, Kao AA, et al. Diagnosis of ocular surface lesions using ultra-high-resolution optical coherence tomography. Ophthalmology. 2013;120(5):883-91.
-
38Romero IL, Barros J de N, Martins MC, Ballalai PL. The use of 1% toluidine blue eye drops in the diagnosis of ocular surface squamous neoplasia. Cornea. 2013;32(1):36-9.
-
39Thomas BJ, Galor A, Nanji AA, El Sayyad F, Wang J, Dubovy SR, et al. Ultra high-resolution anterior segment optical coherence tomography in the diagnosis and management of ocular surface squamous neoplasia. Ocul Surf. 2014;12(1):46-58.
-
40Dart J. Impression cytology of the ocular surface - research tool or routine clinical investigation? Br J Ophthalmol. 1997;81(11):930.
-
41Barros J de N, Lowen MS, Moraes-Filho MN, Martins MC. Impression cytology for detection of unsuspected ocular surface squamous neoplasia cells in pterygia. Arq Bras Oftalmol. 2014;77(5):305-9.
-
42Calonge M, Diebold Y, Saez V, Enriquez de Salamanca A, Garcia-Vasquez C, Corrales RM, et al. Impression cytology of the ocular surface: a review. Exp Eye Res. 2004;78(3):457-72.
-
43Newton R, Ferlay J, Reeves G, Beral V, Parkin DM. Effect of ambient solar ultraviolet radiation on incidence of squamous-cell carcinoma of the eye. Lancet. 1996; 347(9013):1450-1.
-
44Ohara M, Sotozono C, Tsuchihashi Y, Kinoshita S. Ki-67 labeling index as a marker of malignancy in ocular surface neoplasms. Jpn J Ophthalmol. 2004;48(6):524-9.
-
45Krenzer KL, Freddo TF. Cytokeratin expression in normal human bulbar conjunctiva obtained by impression cytology. Invest Ophthalmol Vis Sci. 1997;38(1):142-52.
Publication Dates
-
Publication in this collection
Mar-Apr 2015
History
-
Received
17 Sept 2014 -
Accepted
04 Dec 2014