Abstracts
Amyotrophic lateral sclerosis represents the most common neurodegenerative disease leading to upper and lower motor neuron compromise. Although the vast majority of cases are sporadic, substantial gain has been observed in the knowledge of the genetic forms of the disease, especially of familial forms. There is a direct correlation between the profile of the mutated genes in sporadic and familial forms, highlighting the main role ofC9orf72 gene in the clinical forms associated with frontotemporal dementia spectrum. The different genes related to familial and sporadic forms represent an important advance on the pathophysiology of the disease and genetic therapeutic perspectives, such as antisense therapy. The objective of this review is to signal and summarize clinical and genetic data related to familial forms of amyotrophic lateral sclerosis.
amyotrophic lateral sclerosis; motor neuron disease; neurogenetics; neurodegeneration; C9orf72 gene
A esclerose lateral amiotrófica representa a forma mais comum de doença neurodegenerativa com comprometimento do neurônio motor superior e inferior. Embora a maioria dos casos seja esporádica, ganho impressionante referente ao conhecimento das formas genética da doença foi observado, em especial das formas familiares. Há uma correlação direta entre o perfil de genes mutados nas formas familiares e esporádicas, destacando-se o papel principal do geneC9orf72 nas formas clínicas associadas com espectro da demência frontotemporal. Os diferentes genes relacionados às formas familiares e esporádicas representam um importante avanço na fisiopatologia da doença e perespectivas terapêuticas genéticas, como a terapia antisense. O objetivo desta revisão é apontar e resumir os principais dados clínicos e genéticos relacionados às formas familiares da esclerose lateral amiotrófica.
esclerose lateral amiotrófica; doença do neurônio motor; neurogenética; neurodegeneração; gene C9orf72
Amyotrophic Lateral Sclerosis (ALS) or Lou Gehrig’s disease is the main progressive adult-onset neurodegenerative motor neuron disease (MND), affecting primarily upper (UMN) and lower motor neurons (LMN) giving rise to its typical neurological manifestations. Overall prevalence of ALS is around two to seven cases per 100000 inhabitants11. Oliveira ASB, Pereira RDB. Amyotrophic lateral sclerosis (ALS): three letters that change the people’s life. For ever. Arq Neuropsiquiatr. 2009;67(3A):750-82. doi:10.1590/S0004-282X2009000400040,22. Leblond CS, Kaneb HM, Dion PA, Rouleau GA. Dissection of genetic factors associated with amyotrophic lateral sclerosis. Exp Neurol. 2014;262(Part B):91-101. doi:10.1016/j.expneurol.2014.04.013, and incidence around one to two new cases per 100000 inhabitants per year22. Leblond CS, Kaneb HM, Dion PA, Rouleau GA. Dissection of genetic factors associated with amyotrophic lateral sclerosis. Exp Neurol. 2014;262(Part B):91-101. doi:10.1016/j.expneurol.2014.04.013,33. He J, Mangelsdorf M, Fan D, Bartlett P, Brown MA. Amyotrophic Lateral Sclerosis genetic studies: from genome-wide association mapping to genome sequencing. Neuroscientist. 2014;pii:1073858414555404. Forthcoming. doi:10.1177/1073858414555404,44. Su XW, Broach JR, Connor JR, Gerhard GS, Simmons Z. Genetic heterogeneity of amyotrophic lateral sclerosis: implications for clinical practice and research. Muscle Nerve. 2014;49(6):786-803. doi:10.1002/mus.24198.
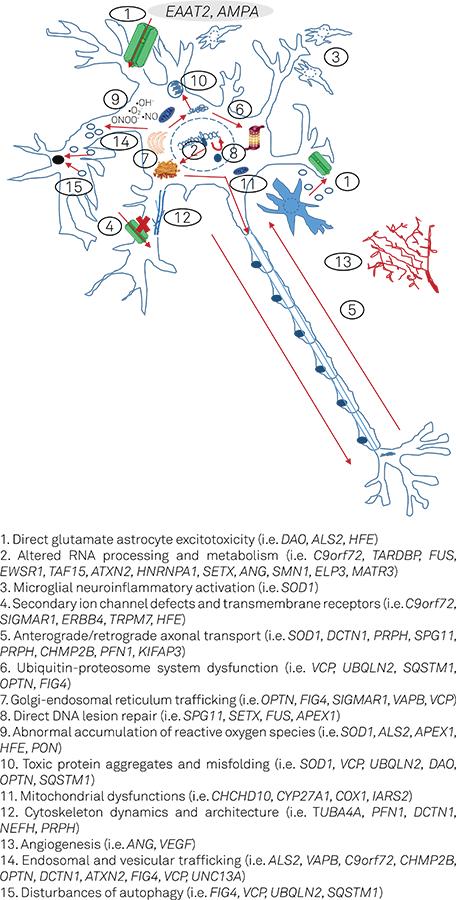
ALS arises as a consequence of multiple pathophysiological mechanisms and cellular dysfunctions (Figure 1), including protein misfolding and aggregation, altered RNA processing (mainly disturbed mRNA splicing and signaling), defects in axonal transport, abnormal metabolism and accumulation of reactive oxygen species, mitochondrial dysfunctions, microglial neuroinflammatory mechanisms, direct glutamate excitotoxicity by astrocytes, abnormal modulatory effects from other glial cells, disturbances of autophagy, ubiquitine-proteosome system abnormalities and primary and secondary ion channel defects55. Ferraiuolo L, Kirby J, Grierson AJ, Sendtner M, Shaw PJ. Molecular pathways of motor neuron injury in amyotrophic lateral sclerosis. Nat Rev Neurol. 2011;7(11):616-30. doi:10.1038/nrneurol.2011.152,66. Iguchi Y, Katsuno M, Ikenaka K, Ishigaki S, Sobue G. Amyotrophic lateral sclerosis: an update on recent genetic insights. J Neurol. 2013;260(11):2917-27. doi:10.1007/s00415-013-7112-y,77. Chen S, Sayana P, Zhang X, Le W. Genetics of amyotrophic lateral sclerosis: an update. Mol Neurodegener. 2013;8(1):28. doi:10.1186/1750-1326-8-28,88. Renton AE, Chiò A, Traynor BJ. State of play in amyotrophic lateral sclerosis genetics. Nat Neurosci. 2014;17(1):17-23. doi:10.1038/nn.3584. Environmental and toxic factors also represent major factors, including traumatic sports mechanisms and toxic causes (including the ALS-parkinsonism/dementia of Guam)99. Turner MR, Swash M. The expanding syndrome of amyotrophic lateral sclerosis: a clinical and molecular odyssey. J Neurol Neurosurg Psychiatry. 2015;86(6):667-73. doi:10.1136/jnnp-2014-308946.
Schematic representation of the main pathophysiological mechanisms involved in familial amyotrophic lateral sclerosis and the genetic changes involved in each case5,6,7,8.
Typical electrodiagnostic and clinical signs arise from a complex network of neuropathological changes including upper motor neuron degeneration in the frontal lobe (mainly Betz giant cells in the motor cortex), corticospinal and corticobulbar tracts (in the spinal cord, internal capsule and cerebral peduncles pathways), lower motor neurons in brainstem nuclei (motor nuclei of cranial nerves VII, X, XI and XII) and in anterior horn motoneurons of the spinal cord. Onufrowicz-Mannen’s nucleus and some cranial nerve motoneurons (motor nuclei of cranial nerves III, IV and VI) are generally spared, correlating with exceptionally rare compromises of facial and extrinsec ocular movements and sphincteral disturbances11. Oliveira ASB, Pereira RDB. Amyotrophic lateral sclerosis (ALS): three letters that change the people’s life. For ever. Arq Neuropsiquiatr. 2009;67(3A):750-82. doi:10.1590/S0004-282X2009000400040,22. Leblond CS, Kaneb HM, Dion PA, Rouleau GA. Dissection of genetic factors associated with amyotrophic lateral sclerosis. Exp Neurol. 2014;262(Part B):91-101. doi:10.1016/j.expneurol.2014.04.013. In microscopic evaluation, cytoplasmic Bunina bodies and Lewy body-like are motor neuron neuropathological hallmarks of ALS, although each genetic subtype commonly presents with their signatures. Neuropathological remarks include neurofilamentous swelling of proximal axons with reduced calibre of distal axons and axonal wallerian degeneration, accumulation of neurofilament and peripherin in axons and perikarium, reactive glyosis, Lewy body-like cytoplasmic neuronal inclusions, perikarial inclusions with phosphorylated neurofilament and ubiquitin immunoreactivity and positive immunoreactivity for other biochemical markers depending on genetic basis of the disease, mainly ubiquinated protein inclusions with positivity for TAR (Transactive response) DNA-binding protein 43 (TDP-43)22. Leblond CS, Kaneb HM, Dion PA, Rouleau GA. Dissection of genetic factors associated with amyotrophic lateral sclerosis. Exp Neurol. 2014;262(Part B):91-101. doi:10.1016/j.expneurol.2014.04.013. Furthermore, different authors believe there is a regional spreading of intracellular misfolded pathogenic proteins SOD1 and TDP-43 involved in ALS in a prion-like propagation in an intercellular contiguous spreading fashion22. Leblond CS, Kaneb HM, Dion PA, Rouleau GA. Dissection of genetic factors associated with amyotrophic lateral sclerosis. Exp Neurol. 2014;262(Part B):91-101. doi:10.1016/j.expneurol.2014.04.013,66. Iguchi Y, Katsuno M, Ikenaka K, Ishigaki S, Sobue G. Amyotrophic lateral sclerosis: an update on recent genetic insights. J Neurol. 2013;260(11):2917-27. doi:10.1007/s00415-013-7112-y,1010. Kanouchi T, Ohkubo T, Yokota T. Can regional spreading of amyotrophic lateral sclerosis motor symptoms be explained by prion-like propagation? J Neurol Neurosurg Psychiatry. 2012;83(7):739-45. doi:10.1136/jnnp-2011-301826.
Most cases of ALS present with asymmetric focal apendicular weakness progressing with bulbar dysfunction, quadriparesis and respiratory insufficiency leading to death in about two to three years after onset11. Oliveira ASB, Pereira RDB. Amyotrophic lateral sclerosis (ALS): three letters that change the people’s life. For ever. Arq Neuropsiquiatr. 2009;67(3A):750-82. doi:10.1590/S0004-282X2009000400040. The combination of upper motor neuron and lower motor neuron signs of compromise is classically described with variable degrees of each component during disease progression11. Oliveira ASB, Pereira RDB. Amyotrophic lateral sclerosis (ALS): three letters that change the people’s life. For ever. Arq Neuropsiquiatr. 2009;67(3A):750-82. doi:10.1590/S0004-282X2009000400040. Association with cognitive and behavioural disturbances is common and sometimes present with the classical phenotype of behavioral variant (bv) frontotemporal dementia (FTD). Although lability of affect commonly occurs, bvFTD, executive dysfunction and mood disorders occurs more frequently in some specific clinical and genetic conditions. A FTD clinical suspection can be properly analyzed applying specific criteria, including the Hodge’s and Neary criteria1111. Strong MJ, Grace GM, Freedman M, Lomen-Hoerth C, Woolley S, Goldstein LH et al. Consensus criteria for the diagnosis of frontotemporal cognitive and behavioural syndromes in amyotrophic lateral sclerosis. Amyotroph Lateral Scler 2009;10(3):131-46. doi:10.1080/17482960802654364. Other cognitive compromise patterns also occurs including primary progressive nonfluent aphasia, logopenic progressive aphasia and semantic dementia. Around 5% of sporadic and familial ALS cases fullfill properly diagnostic criteria for FTD1111. Strong MJ, Grace GM, Freedman M, Lomen-Hoerth C, Woolley S, Goldstein LH et al. Consensus criteria for the diagnosis of frontotemporal cognitive and behavioural syndromes in amyotrophic lateral sclerosis. Amyotroph Lateral Scler 2009;10(3):131-46. doi:10.1080/17482960802654364.
There are no specific clinical, neuroimaging and serum and cerebrospinal fluid biochemical markers to provide the definite diagnosis of ALS and clinicians must be aware about differential diagnosis and red-flags11. Oliveira ASB, Pereira RDB. Amyotrophic lateral sclerosis (ALS): three letters that change the people’s life. For ever. Arq Neuropsiquiatr. 2009;67(3A):750-82. doi:10.1590/S0004-282X2009000400040. Clinical and electrodiagnostic findings are essential to guide the diagnosis process using both the revised El Escorial criteria and the Awaji-shima electrodiagnostic criteria. Although causative genes and susceptibility loci are well-established in sporadic and familial ALS (Table 1), presymptomatic testing does not represent a reliable and available diagnostic method in most neurological centers22. Leblond CS, Kaneb HM, Dion PA, Rouleau GA. Dissection of genetic factors associated with amyotrophic lateral sclerosis. Exp Neurol. 2014;262(Part B):91-101. doi:10.1016/j.expneurol.2014.04.013,66. Iguchi Y, Katsuno M, Ikenaka K, Ishigaki S, Sobue G. Amyotrophic lateral sclerosis: an update on recent genetic insights. J Neurol. 2013;260(11):2917-27. doi:10.1007/s00415-013-7112-y.
Genetic causes of amyotrophic lateral sclerosis, year of description, pattern of inheritance and allelic conditions2,8,12,59.
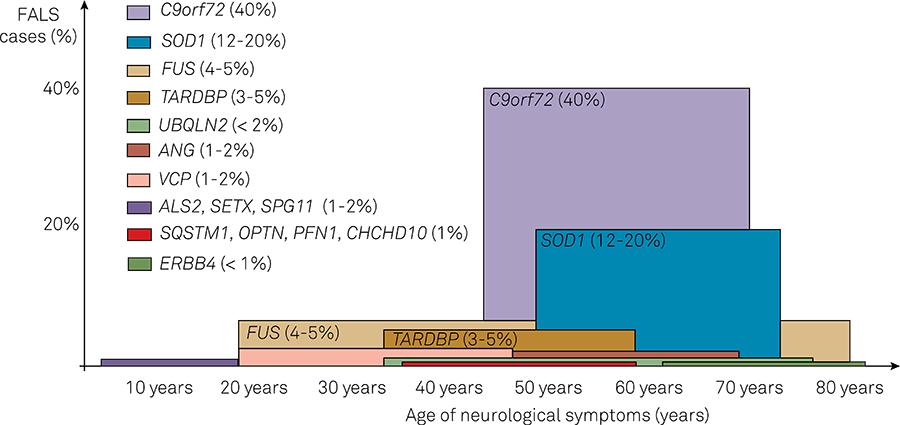
Most cases of ALS are sporadic (90 to 95% of ALS cases) and generally occurs in patients between the fifth and seventh decades of life. Familial ALS cases are defined when there is a context of more than one affected family member (relative) from first or second generation from the index or propositum case with the same disease presentation (despite the frequent occurence of intrafamilial clinical variability) and generally associates with: (i) earlier age at disease onset; (ii) more commonly symptom onset starts in the lower extremities; and (iii) longer or shorter disease duration, life expectancy and clinical progression, depending on particular genetic subtypes22. Leblond CS, Kaneb HM, Dion PA, Rouleau GA. Dissection of genetic factors associated with amyotrophic lateral sclerosis. Exp Neurol. 2014;262(Part B):91-101. doi:10.1016/j.expneurol.2014.04.013,1212. Al-Chalabi A, Jones A, Troakes C, King A, Al-Sarraj S, Berg LH. The genetics and neuropathology of amyotrophic lateral sclerosis. Acta Neuropathol. 2012;124(3):339-52. doi:10.1007/s00401-012-1022-4,11. Oliveira ASB, Pereira RDB. Amyotrophic lateral sclerosis (ALS): three letters that change the people’s life. For ever. Arq Neuropsiquiatr. 2009;67(3A):750-82. doi:10.1590/S0004-282X2009000400040 33. He J, Mangelsdorf M, Fan D, Bartlett P, Brown MA. Amyotrophic Lateral Sclerosis genetic studies: from genome-wide association mapping to genome sequencing. Neuroscientist. 2014;pii:1073858414555404. Forthcoming. doi:10.1177/1073858414555404. Familial cases can occur in an autosomal recessive or dominant or dominant X-linked inheritance patterns. Most adult-onset cases of familial ALS have autosomal dominant inheritance pattern, while juvenile-onset cases have autosomal recessive pattern, in a similar way to the observed with autosomal recessive cerebellar ataxias and autosomal dominant spinocerebellar ataxias. Overall penetrance is age and gene-dependent in familial ALS, as nearly half of patients with SOD1 and FUS genes mutations become symptomatic at their fifties and 90% at their seventies1212. Al-Chalabi A, Jones A, Troakes C, King A, Al-Sarraj S, Berg LH. The genetics and neuropathology of amyotrophic lateral sclerosis. Acta Neuropathol. 2012;124(3):339-52. doi:10.1007/s00401-012-1022-4. There is also a good correlation between genetic subtypes and general age at onset (Figure 2). Genetic anticipation is exceptionally seen. There is also a tendency to progress with more prominent bulbar symptoms in familial ALS than in sporadic cases with the same age. It is important to reiterate that no specific clinical, cerebrospinal and neuroimaging parameters can rightly and reliably differentiate familial from sporadic ALS in cases without a significant familial history of neurodegeneration22. Leblond CS, Kaneb HM, Dion PA, Rouleau GA. Dissection of genetic factors associated with amyotrophic lateral sclerosis. Exp Neurol. 2014;262(Part B):91-101. doi:10.1016/j.expneurol.2014.04.013,1212. Al-Chalabi A, Jones A, Troakes C, King A, Al-Sarraj S, Berg LH. The genetics and neuropathology of amyotrophic lateral sclerosis. Acta Neuropathol. 2012;124(3):339-52. doi:10.1007/s00401-012-1022-4,1313. Ticozzi N, Tiloca C, Morelli C, Colombrita C, Poletti B, Doretti A et al. Genetics of familial Amyotrophic lateral sclerosis. Arch Ital Biol. 2011;149(1):65-82. doi:10.4449/aib.v149i1.1262,1414. Conte A, Lattante S, Luigetti M, Del Grande A, Romano A, Marcaccio A et al. Classification of familial amyotrophic lateral sclerosis by family history: effects on frequency of genes mutation. J Neurol Neurosurg Psychiatry. 2012;83(12):1201-3. doi:10.1136/jnnp-2012-302897.
Distribution of the most important genetic causes of familial ALS according to the age of onset of neurological signs and symptoms. The proportion of each gene in relation to all familial cases is also represented4,8.
There are two other clinical situations which should be remembered in the context of familial and sporadic ALS: young-onset and juvenile ALS. Young-onset ALS starts before 45 years old, corresponds to 10% of all ALS cases and tends to present less commonly with bulbar-onset symptoms. Most cases arise in the context of sporadic ALS1515. Turner MR, Barnwell J, Al-Chalabi A, Eisen A. Young-onset amyotrophic lateral sclerosis: historical and other observations. Brain. 2012;135(9):2883-91. doi:10.1093/brain/aws144. Juvenile ALS represents cases starting very early before 25 years of age with slowly progressive ALS, serving as a guide and key-element for clinical suspicion of autosomal recessive familial cases66. Iguchi Y, Katsuno M, Ikenaka K, Ishigaki S, Sobue G. Amyotrophic lateral sclerosis: an update on recent genetic insights. J Neurol. 2013;260(11):2917-27. doi:10.1007/s00415-013-7112-y,1515. Turner MR, Barnwell J, Al-Chalabi A, Eisen A. Young-onset amyotrophic lateral sclerosis: historical and other observations. Brain. 2012;135(9):2883-91. doi:10.1093/brain/aws144. Juvenile forms are frequently described in ALS2, ALS4, ALS5, ALS6 (rarely), ALS15 and ALS1666. Iguchi Y, Katsuno M, Ikenaka K, Ishigaki S, Sobue G. Amyotrophic lateral sclerosis: an update on recent genetic insights. J Neurol. 2013;260(11):2917-27. doi:10.1007/s00415-013-7112-y.
CLINICAL AND GENETIC FORMS OF FAMILIAL ALS
From a historical perspective, Horton classified familial forms of motor neuron disease in three main clinico-pathological phenotypes: (i) the first with early-onset motor symptoms, before five years of age, with a rapidly progressive and lower motor neuron dominant ALS phenotype, with degeneration of corticospinal tracts and anterior horn motoneurons; (ii) the second form clinically similar, but also with posterior column and spinocerebellar tract degenerations; and (iii) a third form similar to the second, but with prolonged survival period of up to two decades1616. Horton WA, Eldridge R, Brody JA. Familial motor neuron disease: evidence for at least three different types. Neurology. 1976;26(5):460-5. doi:10.1212/WNL.26.5.460.
The genetic history of familial ALS can be summed up by the outstanding roles ofSOD1 and C9orf72 genes. By 2011, the year of the initial description of the hexanucleotide repeat expansion ofC9orf72 gene, only 30% of familial cases had established their genetic etiology55. Ferraiuolo L, Kirby J, Grierson AJ, Sendtner M, Shaw PJ. Molecular pathways of motor neuron injury in amyotrophic lateral sclerosis. Nat Rev Neurol. 2011;7(11):616-30. doi:10.1038/nrneurol.2011.152,88. Renton AE, Chiò A, Traynor BJ. State of play in amyotrophic lateral sclerosis genetics. Nat Neurosci. 2014;17(1):17-23. doi:10.1038/nn.3584,1717. Al-Chalabi A, Hardiman O. The epidemiology of ALS: a conspiracy of genes, environment and time. Nat Rev Neurol. 2013;9(11):617-8. doi:10.1038/nrneurol.2013.203,1818. Van Langenhove T, Zee J, Van Broeckhoven C. The molecular basis of the frontotemporal lobar degeneration-amyotrophic lateral sclerosis spectrum. Ann Med. 2012;44(8):817-28. doi:10.3109/07853890.2012.665471,1919. Blitterswijk M, v Es MA, Hennekam EA, Dooijes D, Rheenen W, Medic J et al. Evidence for an oligogenic basis of amyotrophic lateral sclerosis. Hum Mol Genet. 2012;21(17):3776-84. doi:10.1093/hmg/dds199. In the last decade, more than 20 different loci were related to familial ALS.
Despite the marked heterogeneity of familial ALS, it can be said that most cases relate to C9orf72, SOD1, FUS,TARDBP and UBQLN2 genes66. Iguchi Y, Katsuno M, Ikenaka K, Ishigaki S, Sobue G. Amyotrophic lateral sclerosis: an update on recent genetic insights. J Neurol. 2013;260(11):2917-27. doi:10.1007/s00415-013-7112-y,88. Renton AE, Chiò A, Traynor BJ. State of play in amyotrophic lateral sclerosis genetics. Nat Neurosci. 2014;17(1):17-23. doi:10.1038/nn.3584. Thus, it is also possible to set specific genetic test batteries for recessive and autosomal dominant and X-linked forms, according to inheritance patterns, clinical comorbitidities, natural history and clinical evolution, and population epidemiology. However, up to 32% of familial cases and up to 11% of sporadic cases still do not have a definite genetic diagnosis of ALS22. Leblond CS, Kaneb HM, Dion PA, Rouleau GA. Dissection of genetic factors associated with amyotrophic lateral sclerosis. Exp Neurol. 2014;262(Part B):91-101. doi:10.1016/j.expneurol.2014.04.013,77. Chen S, Sayana P, Zhang X, Le W. Genetics of amyotrophic lateral sclerosis: an update. Mol Neurodegener. 2013;8(1):28. doi:10.1186/1750-1326-8-28,88. Renton AE, Chiò A, Traynor BJ. State of play in amyotrophic lateral sclerosis genetics. Nat Neurosci. 2014;17(1):17-23. doi:10.1038/nn.3584,1919. Blitterswijk M, v Es MA, Hennekam EA, Dooijes D, Rheenen W, Medic J et al. Evidence for an oligogenic basis of amyotrophic lateral sclerosis. Hum Mol Genet. 2012;21(17):3776-84. doi:10.1093/hmg/dds199.
Establishing a definite diagnosis of familial ALS is complex. In an Italian study with 53 families, genetic screening for seven of the most important genes (SOD1, C9orf72, TARDBP, FUS, ANG, ATXN2, OPTN) disclosed only 25% of definite genetic diagnosis with 75% of them with two family members clinically affected and 17% with only one affected family member first and second degrees away1414. Conte A, Lattante S, Luigetti M, Del Grande A, Romano A, Marcaccio A et al. Classification of familial amyotrophic lateral sclerosis by family history: effects on frequency of genes mutation. J Neurol Neurosurg Psychiatry. 2012;83(12):1201-3. doi:10.1136/jnnp-2012-302897.
There is a lot of controversy regarding the role of most genes discovered nowadays in relation to ALS, as some of them do not have a unique causative mechanism (i.e.SOD1, C9ORF72, SETX, ANG, SPG11, FUS, TARDBP, VAPB, VCP, UBQLN2, OPTN, among others) but a disease-modifying function or susceptibility loci (i.e. PGRN, HFE, NEFH, UNC13A, VEGF, among others)44. Su XW, Broach JR, Connor JR, Gerhard GS, Simmons Z. Genetic heterogeneity of amyotrophic lateral sclerosis: implications for clinical practice and research. Muscle Nerve. 2014;49(6):786-803. doi:10.1002/mus.24198,1313. Ticozzi N, Tiloca C, Morelli C, Colombrita C, Poletti B, Doretti A et al. Genetics of familial Amyotrophic lateral sclerosis. Arch Ital Biol. 2011;149(1):65-82. doi:10.4449/aib.v149i1.1262. Further discussions regarding genetic and clinical basis of familial ALS types will be provided forth (Tables 1 and 2).
ALS1
ALS1 (MIM #105400) represents the second most common form of familial ALS, giving rise to up to 20% of cases with an autosomal dominant or recessive inheritance.SOD1 gene (superoxide dysmutase-1; 21q22.11) mutations give rise to abnormal function of copper/zinc superoxide dysmutase 1, responsible for converting superoxide free radical species from cytoplasm and inner intermembrane mitochondrial space into molecular oxygen and hydrogen peroxide. Secondary dysfunction of tyrosine phosphatases with phosphorilation inhibition by EGF (epidermal growth factor), IGF-1 (insulin-like growth factor 1) and FGF-2 (fibroblast growth factor-2) by MAPK (mitogen-activated protein kinase) pathway77. Chen S, Sayana P, Zhang X, Le W. Genetics of amyotrophic lateral sclerosis: an update. Mol Neurodegener. 2013;8(1):28. doi:10.1186/1750-1326-8-28,2020. Ince PG, Highley JR, Kirby J, Wharton SB, Takahashi H, Strong MJ et al. Molecular pathology and genetic advances in amyotrophic lateral sclerosis: an emerging molecular pathway and the significance of glial pathology. Acta Neuropathol. 2011;122(6):657-71. doi:10.1007/s00401-011-0913-0.
By 2011, SOD1-associated ALS (formerly the 21q-associated ALS) represented the most common form of familial and sporadic ALS. Most cases present with adult-onset ALS without cognitive compromise and variable clinical outcomes depending on the genetic background involved in each case44. Su XW, Broach JR, Connor JR, Gerhard GS, Simmons Z. Genetic heterogeneity of amyotrophic lateral sclerosis: implications for clinical practice and research. Muscle Nerve. 2014;49(6):786-803. doi:10.1002/mus.24198,77. Chen S, Sayana P, Zhang X, Le W. Genetics of amyotrophic lateral sclerosis: an update. Mol Neurodegener. 2013;8(1):28. doi:10.1186/1750-1326-8-28,2020. Ince PG, Highley JR, Kirby J, Wharton SB, Takahashi H, Strong MJ et al. Molecular pathology and genetic advances in amyotrophic lateral sclerosis: an emerging molecular pathway and the significance of glial pathology. Acta Neuropathol. 2011;122(6):657-71. doi:10.1007/s00401-011-0913-0. Rapidly progressive forms ofSOD1 familial ALS occur in USA with p.Ala4Val mutation. Slowly progressive cases correlate to p.Asp90Ala mutation in Scandinavia and p.His46Arg in Japan. A LMN dominant ALS variant linked to SOD1was described in cases of p.Ala4Val e p.Val148Gly mutations. A cerebellar ataxia variant with slow progression was described in Scandinavia in cases of p.Asp90Ala mutation77. Chen S, Sayana P, Zhang X, Le W. Genetics of amyotrophic lateral sclerosis: an update. Mol Neurodegener. 2013;8(1):28. doi:10.1186/1750-1326-8-28,2020. Ince PG, Highley JR, Kirby J, Wharton SB, Takahashi H, Strong MJ et al. Molecular pathology and genetic advances in amyotrophic lateral sclerosis: an emerging molecular pathway and the significance of glial pathology. Acta Neuropathol. 2011;122(6):657-71. doi:10.1007/s00401-011-0913-0.
ALS2
ALS2 (MIM #205100) represents a rare autosomal recessive slowly progressive UMN-dominant juvenile ALS as a consequence of homozigosity mutations inALS2 gene (2q33.1), coding alsin, involved in the membrane and endosomal intracellular trafficking as guanine nucleotide exchange factor for Rac1 and Rab5 GTPases, in neurite outgrowth in hippocampus mediated by Rac1 activation and in prevention of glutamartegic excitoxicity mediated by Glur2 subunit of AMPA (α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid) receptors1515. Turner MR, Barnwell J, Al-Chalabi A, Eisen A. Young-onset amyotrophic lateral sclerosis: historical and other observations. Brain. 2012;135(9):2883-91. doi:10.1093/brain/aws144,2121. Shirakawa K, Suzuki H, Ito M, Kono S, Uchiyama T, Ohashi T et al. Novel compound heterozygous ALS2 mutations cause juvenile amyotrophic lateral sclerosis in Japan. Neurology. 2009;73(24):2124-6. doi:10.1212/WNL.0b013e3181c67be0. It begins in preschool child until the young adult ages (up to third decade) and has been described in japanese, turkish, tunisian, kuwaitian, saudi arabian, cypriot and Amish population. ALS2 starts with lower limb and facial spasticity, moderate muscular atrophy, pseudobulbar signs, spastic dysathrophonia and bladder dysfunction evolving statically after two decades. Important clinical overlap with allelic forms of Juvenile primary lateral sclerosis and infantile ascending hereditary spastic paralysis occurs1313. Ticozzi N, Tiloca C, Morelli C, Colombrita C, Poletti B, Doretti A et al. Genetics of familial Amyotrophic lateral sclerosis. Arch Ital Biol. 2011;149(1):65-82. doi:10.4449/aib.v149i1.1262,1515. Turner MR, Barnwell J, Al-Chalabi A, Eisen A. Young-onset amyotrophic lateral sclerosis: historical and other observations. Brain. 2012;135(9):2883-91. doi:10.1093/brain/aws144,2121. Shirakawa K, Suzuki H, Ito M, Kono S, Uchiyama T, Ohashi T et al. Novel compound heterozygous ALS2 mutations cause juvenile amyotrophic lateral sclerosis in Japan. Neurology. 2009;73(24):2124-6. doi:10.1212/WNL.0b013e3181c67be0.
ALS3
ALS3 (MIM %606640) represents a rare autosomal dominant adult-onset familial ALS with lower limb onset associated with 18q21 locus. Classicaly evolves to respiratory failure and death five years after onset2222. Hand CK, Khoris J, Salachas F, Gros-Louis F, Lopes AA, Mayeux-Portas V et al. A novel locus for familial amyotrophic lateral sclerosis, on chromosome 18q. Am J Hum Genet. 2002;70(1):251-6. doi:10.1086/337945.
ALS4
ALS4 (MIM #602433) represents a rare autosomal dominant slowly progressive juvenile ALS associated with mutations in SETX gene (9q34.13), coding the protein senataxin, a DNA/RNA helicase domain involved in RNA processing and metabolism, in DNA repair mechanisms and in RNA polymerase II-dependent transcription. A toxic gain of function generally arises44. Su XW, Broach JR, Connor JR, Gerhard GS, Simmons Z. Genetic heterogeneity of amyotrophic lateral sclerosis: implications for clinical practice and research. Muscle Nerve. 2014;49(6):786-803. doi:10.1002/mus.24198,2323. Chen YZ, Bennett CL, Huynh HM, Blair IP, Puls I, Irobi J et al. DNA/RNA helicase gene mutations in a form of juvenile amyotrophic lateral sclerosis (ALS4). Am J Hum Genet. 2004;74(6):1128-35. doi:10.1086/421054. Very early-onset (generally before 6 years up to adolescence) with prominent distal muscular atrophy and eventually cerebellar ataxia are clues to diagnosis, mimicking spinal muscular atrophy with pyramidal signs and some forms of hereditary distal motor neuropathy. Corticospinal tract and dorsal column changes may be seen in neuroimaging. Ataxia with oculomotor apraxia type 2 is an allelic condition44. Su XW, Broach JR, Connor JR, Gerhard GS, Simmons Z. Genetic heterogeneity of amyotrophic lateral sclerosis: implications for clinical practice and research. Muscle Nerve. 2014;49(6):786-803. doi:10.1002/mus.24198,2323. Chen YZ, Bennett CL, Huynh HM, Blair IP, Puls I, Irobi J et al. DNA/RNA helicase gene mutations in a form of juvenile amyotrophic lateral sclerosis (ALS4). Am J Hum Genet. 2004;74(6):1128-35. doi:10.1086/421054.
ALS5
ALS5 (MIM %602099) represents a rare autosomal recessive slowly progressive juvenile ALS, arising from missense and frameshift mutations or deletions inSPG11 gene (15q21.1), coding spatacsin, involved in axonal outgrowth and intracellular trafficking. ALS5 represents solely the most important cause of autosomal recessive juvenile ALS44. Su XW, Broach JR, Connor JR, Gerhard GS, Simmons Z. Genetic heterogeneity of amyotrophic lateral sclerosis: implications for clinical practice and research. Muscle Nerve. 2014;49(6):786-803. doi:10.1002/mus.24198,2424. Hentati A, Ouahchi K, Pericak-Vance MA, Nijhawan D, Ahmad A, Yang Y et al. Linkage of a commoner form of recessive amyotrophic lateral sclerosis to chromosome 15q15-q22 markers. Neurogenetics 1998;2(1):55-60. doi:10.1007/s100480050052,2525. Daoud H, Zhou S, Noreau A, Sabbagh M, Belzil V, Dionne-Laporte A et al. Exome sequencing reveals SPG11 mutations causing juvenile ALS. Neurobiol Aging. 2012;33(4):839:e5-9. doi:10.1016/j.neurobiolaging.2011.11.012. Prominent tongue fasciculations and amyotrophy in the first two decades evolve in up to three decades with upper motor neuron signs and spastic dysarthrophonia. Faster clinical courses were described in late-onset cases2424. Hentati A, Ouahchi K, Pericak-Vance MA, Nijhawan D, Ahmad A, Yang Y et al. Linkage of a commoner form of recessive amyotrophic lateral sclerosis to chromosome 15q15-q22 markers. Neurogenetics 1998;2(1):55-60. doi:10.1007/s100480050052,2525. Daoud H, Zhou S, Noreau A, Sabbagh M, Belzil V, Dionne-Laporte A et al. Exome sequencing reveals SPG11 mutations causing juvenile ALS. Neurobiol Aging. 2012;33(4):839:e5-9. doi:10.1016/j.neurobiolaging.2011.11.012. Allelic condition to Hereditary Spastic Paraplegia type 112525. Daoud H, Zhou S, Noreau A, Sabbagh M, Belzil V, Dionne-Laporte A et al. Exome sequencing reveals SPG11 mutations causing juvenile ALS. Neurobiol Aging. 2012;33(4):839:e5-9. doi:10.1016/j.neurobiolaging.2011.11.012.
ALS6
ALS6 (MIM #608030) represents an autosomal recessive or dominant adult or late-onset LMN-dominant familial ALS associated with heterozygous mutations in the FUS/TLS gene (fused in sarcoma, translated in liposarcoma; 16p11.2), coding the FUS nucleoprotein, involved with DNA repair, transcription activation, and with RNA splicing and transport to the cytoplasm. FUS associates with TDP-43 protein during the formation of SMN complex in spliceosome maintenance66. Iguchi Y, Katsuno M, Ikenaka K, Ishigaki S, Sobue G. Amyotrophic lateral sclerosis: an update on recent genetic insights. J Neurol. 2013;260(11):2917-27. doi:10.1007/s00415-013-7112-y,2626. Hewitt C, Kirby J, Highley JR, Hartley JA, Hibberd R, Hollinger HC et al. Novel FUS/TLS mutations and pathology in familial and sporadic amyotrophic lateral sclerosis. Arch Neurol. 2010;67(4):455-61. doi:10.1001/archneurol.2010.52,2727. Bäumer D, Hilton D, Paine SM, Turner MR, Lowe J, Talbot K et al. Juvenile ALS with basophilic inclusions is a FUS proteinopathy with FUS mutations. Neurology. 2010;75(7):611-8. doi:10.1212/WNL.0b013e3181ed9cde. Up to 4% of familial and 1% of sporadic ALS arise from mutations in FUS gene, most commonly in USA, Cape Verdean and Europe (Germany, Italy, France, French-Canadian, United Kingdom). A rare juvenile variant with basophilic inclusions has also been described with a rapidly progressive motor phenotype starting during the second to third decade. Clinic and genetic correlations links p.Lys525Pro mutation with an aggressive rapidly progressive phenotype. Allelic disorders include hereditary essential tremor type 4 and FUS-related FTD66. Iguchi Y, Katsuno M, Ikenaka K, Ishigaki S, Sobue G. Amyotrophic lateral sclerosis: an update on recent genetic insights. J Neurol. 2013;260(11):2917-27. doi:10.1007/s00415-013-7112-y,2626. Hewitt C, Kirby J, Highley JR, Hartley JA, Hibberd R, Hollinger HC et al. Novel FUS/TLS mutations and pathology in familial and sporadic amyotrophic lateral sclerosis. Arch Neurol. 2010;67(4):455-61. doi:10.1001/archneurol.2010.52,2727. Bäumer D, Hilton D, Paine SM, Turner MR, Lowe J, Talbot K et al. Juvenile ALS with basophilic inclusions is a FUS proteinopathy with FUS mutations. Neurology. 2010;75(7):611-8. doi:10.1212/WNL.0b013e3181ed9cde.
ALS7
ALS7 (MIM %608031) represents a rare autosomal dominant adult-onset familial ALS, occurring in USA in association with ALS7 locus (20p13) without FTD or other systemic signs2828. Sapp PC, Hosler BA, McKenna-Yasek D, Chin W, Gann A, Genise H et al. Identification of two novel loci for dominantly inherited familial amyotrophic lateral sclerosis. Am J Hum Genet. 2003;73(2):397-403. doi:10.1086/377158.
ALS8
ALS8 (MIM #608627) represents a rare autosomal dominant slowly progressive LMN-dominant familial ALS, related to heterozygous mutation inVAPB gene (vesicle-associatedmembrane protein-associated protein B, synaptobrevin-associated membrane protein B; 20q13.32), coding the VAPB protein lnked to the response supression to unfolded protein accumulation in endoplasmic reticulum and microtubule-associated membrane transport, presynaptic neuronal terminal formation and vesicular trafficking2929. Nishimura AL, Mitne-Neto M, Silva HCA, Richieri-Costa A, Middleton S, Cascio D et al. A mutation in the vesicle-trafficking protein VAPB causes late-onset spinal muscular atrophy and amyotrophic lateral sclerosis. Am J Hum Genet. 2004;75(5):822-31. doi:10.1086/425287.
A complex spectrum of neurological emerges, including: (i) a rapidly progreressive adult-onset severe LMN-dominant ALS; (ii) atypical slowly progressive ALS with postural tremor; and (iii) late-onset spinal muscular atrophy type Finkel. Cases were described mainly in Brazilian families and portuguese and United Kingdom ascending patients2929. Nishimura AL, Mitne-Neto M, Silva HCA, Richieri-Costa A, Middleton S, Cascio D et al. A mutation in the vesicle-trafficking protein VAPB causes late-onset spinal muscular atrophy and amyotrophic lateral sclerosis. Am J Hum Genet. 2004;75(5):822-31. doi:10.1086/425287.
ALS9
ALS9 (MIM #611895) represents a rare bulbar-onset autosomal dominant familial ALS linked to heterozygous mutation in ANG gene (angiogenin; 14q11.2), coding angiogenin protein (pancreatic ribonuclease A, superfamily 5), involved with angiogenic activity in motoneurons acting as a neovascularization inducer, ribosomal RNA formation and inducing cellular proliferation by VEGF (vascular endothelial growth factor). Cases were reported in Ireland, Scotland, Italy, USA, northeastern Europe and France. ANG-related ALS represents near 2% of familial cases. Atypical parkinsonism with late-onset frontotemporal dementia, rapidly progressive variants and LMN-dominant ALS with parkinsonism and FTD have also been described44. Su XW, Broach JR, Connor JR, Gerhard GS, Simmons Z. Genetic heterogeneity of amyotrophic lateral sclerosis: implications for clinical practice and research. Muscle Nerve. 2014;49(6):786-803. doi:10.1002/mus.24198,3030. Millecamps S, Salachas F, Cazeneuve C, Gordon P, Bricka B, Camuzat A et al. SOD1, ANG, VAPB, TARDBP, and FUS mutations in familial amyotrophic lateral sclerosis: genotype-phenotype correlations. J Med Genet. 2010;47(8):554-60. doi:10.1136/jmg.2010.077180.
ALS10
ALS10 (MIM #612069) represents a rare early-onset autosomal LMN-dominant familial ALS, linked to heterozygous missense mutations in TARDBP gene (TDP-43, transactive response DNA binding protein 43 kDa; 1p36.22), coding the RNA-ligand TDP-43 (TAR DNA-binding protein 43-kD) ribonucleoprotein, involved in regulation of protein expression, transcription and translation, pre-mensager RNA alternative splicing and microRNA biogenesis66. Iguchi Y, Katsuno M, Ikenaka K, Ishigaki S, Sobue G. Amyotrophic lateral sclerosis: an update on recent genetic insights. J Neurol. 2013;260(11):2917-27. doi:10.1007/s00415-013-7112-y,2020. Ince PG, Highley JR, Kirby J, Wharton SB, Takahashi H, Strong MJ et al. Molecular pathology and genetic advances in amyotrophic lateral sclerosis: an emerging molecular pathway and the significance of glial pathology. Acta Neuropathol. 2011;122(6):657-71. doi:10.1007/s00401-011-0913-0,3131. Corcia P, Valdmanis P, Millecamps S, Lionnet C, Blasco H, Mouzat K et al. Phenotype and genotype analysis in amyotrophic lateral sclerosis with TARDBP gene mutations. Neurology. 2012;78(19):1519-26. doi:10.1212/WNL.0b013e3182553c88. ALS10 represent up to 5% of familial ALS cases and 2% of sporadic cases, being described in Italy (Sardinia), France, Germany, Japan, England, Australia and China. TARDBP-associated ALS also occur as long-standing symptoms in the upper limbs and with bulbar-onset in Asian patients. Clinical and genetical correlations are well-established: G298S mutations with rapidly progressive course, A315T and M337V mutations with longer survival periods66. Iguchi Y, Katsuno M, Ikenaka K, Ishigaki S, Sobue G. Amyotrophic lateral sclerosis: an update on recent genetic insights. J Neurol. 2013;260(11):2917-27. doi:10.1007/s00415-013-7112-y,2020. Ince PG, Highley JR, Kirby J, Wharton SB, Takahashi H, Strong MJ et al. Molecular pathology and genetic advances in amyotrophic lateral sclerosis: an emerging molecular pathway and the significance of glial pathology. Acta Neuropathol. 2011;122(6):657-71. doi:10.1007/s00401-011-0913-0,3131. Corcia P, Valdmanis P, Millecamps S, Lionnet C, Blasco H, Mouzat K et al. Phenotype and genotype analysis in amyotrophic lateral sclerosis with TARDBP gene mutations. Neurology. 2012;78(19):1519-26. doi:10.1212/WNL.0b013e3182553c88.
ALS11
ALS11 (MIM #612577) represents a rare autosomal dominant adult-onset ALS or PLS, related to heterozigous missense mutations in FIG4 gene (phosphatidylinositol 3,5-bisphophate 5-phosphatase, Sac domain-containing inositolphosphatase 3; 6q21), coding the FIG4 protein, a 5-phosphatase acting on phosphatidyl-inositol-3,5-biphosphate involved in dynamic changes in endosomal membranes during fission and fusion processes in intracellular transport from lysossomes and late endosomes to the trans-Golgi system, giving rise to authophagia dysfunction and motorneuron vacuolization in anterior horn. FIG4-related disorders include a broader spectrum including Yunis-Varon syndrome, Charcot-Marie-Tooth type 4J and bilateral temporooccipital polimicrogyria66. Iguchi Y, Katsuno M, Ikenaka K, Ishigaki S, Sobue G. Amyotrophic lateral sclerosis: an update on recent genetic insights. J Neurol. 2013;260(11):2917-27. doi:10.1007/s00415-013-7112-y,3232. Chow CY, Landers JE, Bergren SK, Sapp PC, Grant AE, Jones JM et al. Deleterious variants of FIG4, a phosphoinositide phosphatase, in patients with ALS. Am J Hum Genet. 2009;84(1):85-8. doi:10.1016/j.ajhg.2008.12.010.
ALS12
ALS12 (MIM #613435) represents a rare autosomal recessive or dominant slowly progressive familial ALS sith dominant UMN signs, resulting from homozygous or heterozygous mutations in OPTN gene (optineurin; 10p13), coding optineurin protein related to nuclear factor-kappa B (NF-κB) inhibition or activation by TNF-α pathways, Golgi complex manteinance, autophagy induction and interacting with huntingtin, Rab8 and transcription factor IIIA66. Iguchi Y, Katsuno M, Ikenaka K, Ishigaki S, Sobue G. Amyotrophic lateral sclerosis: an update on recent genetic insights. J Neurol. 2013;260(11):2917-27. doi:10.1007/s00415-013-7112-y,3333. Maruyama H, Morino H, Ito H, Izumi Y, Kato H, Watanabe Y et al. Mutations of optineurin in amyotrophic lateral sclerosis. Nature. 2010;465(7295):223-6. doi:10.1038/nature08971. Cases were described in japanese and italian families starting in the third or fifth decades with bulbar compromise at early and moderate disease stages. Allelic disorders include Paget disease of bone and primary open-angle glaucoma type 144. Su XW, Broach JR, Connor JR, Gerhard GS, Simmons Z. Genetic heterogeneity of amyotrophic lateral sclerosis: implications for clinical practice and research. Muscle Nerve. 2014;49(6):786-803. doi:10.1002/mus.24198,66. Iguchi Y, Katsuno M, Ikenaka K, Ishigaki S, Sobue G. Amyotrophic lateral sclerosis: an update on recent genetic insights. J Neurol. 2013;260(11):2917-27. doi:10.1007/s00415-013-7112-y,3333. Maruyama H, Morino H, Ito H, Izumi Y, Kato H, Watanabe Y et al. Mutations of optineurin in amyotrophic lateral sclerosis. Nature. 2010;465(7295):223-6. doi:10.1038/nature08971.
ALS13
ALS13 (MIM #183090) represents an autosomal dominant form of familial ALS in families with spinocerebellar ataxia phenotypes, related to intermediate repeat lengths of CAG trinucleotide (27-33 repeats, generally more than 31) in the 5-prime end of the coding region in the exon 1 of the ATXN2gene (ataxin 2; 12q24.12), coding ataxin-2, involved with EGF receptor trafficking as a negative regulator of endocytosis by interactions with endophilins A1 and A3, forms a RNA-dependent complex with TDP-43, and interacts with PABP protein (poly-A-binding-protein 1) in motoneurons66. Iguchi Y, Katsuno M, Ikenaka K, Ishigaki S, Sobue G. Amyotrophic lateral sclerosis: an update on recent genetic insights. J Neurol. 2013;260(11):2917-27. doi:10.1007/s00415-013-7112-y,3434. Van Damme P, Veldink JH, Blitterswijk M, Corveleyn A, Vught PW, Thijs V et al. Expanded ATXN2 CAG repeat size in ALS identifies genetic overlap between ALS and SCA2. Neurology. 2011;76(24):2066-72. doi:10.1212/WNL.0b013e31821f445b. Cases were described in Belgium, Netherlands, France and Canada. Intermediate repeat lengths also correlated with late-onset Parkinson’s disease and Progressive Supranuclear Palsy. More than 33 repeats are described in spinocerebellar ataxia type 2 (SCA2)66. Iguchi Y, Katsuno M, Ikenaka K, Ishigaki S, Sobue G. Amyotrophic lateral sclerosis: an update on recent genetic insights. J Neurol. 2013;260(11):2917-27. doi:10.1007/s00415-013-7112-y,3434. Van Damme P, Veldink JH, Blitterswijk M, Corveleyn A, Vught PW, Thijs V et al. Expanded ATXN2 CAG repeat size in ALS identifies genetic overlap between ALS and SCA2. Neurology. 2011;76(24):2066-72. doi:10.1212/WNL.0b013e31821f445b.
ALS14
ALS14 (MIM #613954) forms a rare autosomal dominant form of familial ALS resulting from heterozygous mutations in VCP gene (valosin-containing protein; 9p13.3), coding the AAA+-ATPase valosin-containing protein, involved in substrate extraction in ubiquitin-proteosome systems, in Golgi complex biogenesis, in chlatrin-mediated membrane trafficking in endocytosis and Golgi complex, regulates protein degradation at the outer mitochondrial membrane, peroxysomal assembly, autophagosome maturation, and regulates cell cycle44. Su XW, Broach JR, Connor JR, Gerhard GS, Simmons Z. Genetic heterogeneity of amyotrophic lateral sclerosis: implications for clinical practice and research. Muscle Nerve. 2014;49(6):786-803. doi:10.1002/mus.24198,3535. Johnson JO, Mandrioli J, Benatar M, Abramzon Y, Van Deerlin VM, Trojanowski JQ et al. Exome sequencing reveals VCP mutations as a cause of familial ALS. Neuron. 2010;68(5):857-64. doi:10.1016/j.neuron.2010.11.036. Up to 2% of familial ALS areVCP-related, occur in italian and north-american families and starts in the fourth to sixth decades with spine-onset ALS. Allelic disturbances include inclusion body myopathy, early-onset Paget disease, and frontotemporal dementia type 1, and distal myopathy44. Su XW, Broach JR, Connor JR, Gerhard GS, Simmons Z. Genetic heterogeneity of amyotrophic lateral sclerosis: implications for clinical practice and research. Muscle Nerve. 2014;49(6):786-803. doi:10.1002/mus.24198,3535. Johnson JO, Mandrioli J, Benatar M, Abramzon Y, Van Deerlin VM, Trojanowski JQ et al. Exome sequencing reveals VCP mutations as a cause of familial ALS. Neuron. 2010;68(5):857-64. doi:10.1016/j.neuron.2010.11.036.
ALS15
ALS15 (MIM #300857) represents the rare dominant X-linked form of familial ALS with incomplete penetrance related to mutations in UBQLN2 gene (ubiquilin 2; Xp11.21), coding ubiquilin-2, linked to ubiquilin regulator family of ubiquitin-proteosome system and autophagy66. Iguchi Y, Katsuno M, Ikenaka K, Ishigaki S, Sobue G. Amyotrophic lateral sclerosis: an update on recent genetic insights. J Neurol. 2013;260(11):2917-27. doi:10.1007/s00415-013-7112-y,3636. Deng HX, Chen W, Hong ST, Boycott KM, Gorrie GH, Siddique N et al. Mutations in UBQLN2 cause dominant X-linked juvenile and adult-onset ALS and ALS/dementia. Nature. 2011;477(7363):211-5. doi:10.1038/nature10353. Up to 2% of familial ALS cases have a X-linked pattern of inheritance, most commonly in the first third to fifth decades, and starts with bulbar-onset disease evolving with FTD, dystonia, athetosis and spastic tetraparesis. Neuroimaging unveils cortical and basal ganglia atrophy, rarely with brain iron accumulation in basal ganglia66. Iguchi Y, Katsuno M, Ikenaka K, Ishigaki S, Sobue G. Amyotrophic lateral sclerosis: an update on recent genetic insights. J Neurol. 2013;260(11):2917-27. doi:10.1007/s00415-013-7112-y,3636. Deng HX, Chen W, Hong ST, Boycott KM, Gorrie GH, Siddique N et al. Mutations in UBQLN2 cause dominant X-linked juvenile and adult-onset ALS and ALS/dementia. Nature. 2011;477(7363):211-5. doi:10.1038/nature10353.
ALS16
ALS16 (MIM #614373) represents an autosomal recessive UMN-dominant juvenile ALS (starting in preschool and early infancy), linked to homozygous E102Q mutations in the SIGMAR1 gene (sigma nonopioid intracellularreceptor 1; 9p13.3) in Saudi Arabia, coding the S1R receptor, an endoplasmic reticulum chaperone of the cholinergic post-synaptic membrane, which binds neurosteroids, psychostimulants and involved in potassium chanell regulation and calcium signaling by IP3 receptors in cortical and spinal cord motoneurons. Ubiquitin-proteosome system abnormalities and abnormal motoneuron apoptosis have also been described66. Iguchi Y, Katsuno M, Ikenaka K, Ishigaki S, Sobue G. Amyotrophic lateral sclerosis: an update on recent genetic insights. J Neurol. 2013;260(11):2917-27. doi:10.1007/s00415-013-7112-y,3737. Al-Saif A, Al-Mohanna F, Bohlega S. A mutation in sigma-1 receptor causes juvenile amyotrophic lateral sclerosis. Ann Neurol. 2011;70(6):913-9. doi:10.1002/ana.22534.
ALS17
ALS17 (MIM #614696) represents an autosomal dominant form of lower motor neuron dominant familial ALS (sometimes associated with FTD) related to missense heterozygous mutations in the CHMP2B gene (Chromatin-modifying protein 2B or charged multivesicular body protein 2B; 3p11.2), coding the VPS2B protein (vacuolar protein sorting 2), involved with the endosomal sorting complex ESCRT-III linked to degradation of surface receptors to the trans-Golgi and lysosomal networks, formation of multivesicular endocytic bodies, axonal transport, protein translation and MAPK intracellular pathways3838. Cox LE, Ferraiuolo L, Goodall EF, Heath PR, Higginbottom A, Mortiboys H et al. Mutations in CHMP2B in lower motor neuron predominant amyotrophic lateral sclerosis (ALS). PLoS One. 2010;5(3):e9872. doi:10.1371/journal.pone.0009872.
ALS18
ALS18 (MIM #614808) represents a rare autosomal dominant form of spinal-onset familial ALS linked to heterozygous missense mutations in thePFN1 gene (profilin 1; 17p13.2), coding the profilin-1 protein, which inhibits actin polymerization and regulates the outgrowth of the filamentous portion of (F)-actin by binding to monomeric (G)-actin. It represents up to 2% of familial cases starting in the fourth and fifth decades in caucasian and Sephardic Jewish populations without severe cognitive compromise22. Leblond CS, Kaneb HM, Dion PA, Rouleau GA. Dissection of genetic factors associated with amyotrophic lateral sclerosis. Exp Neurol. 2014;262(Part B):91-101. doi:10.1016/j.expneurol.2014.04.013,66. Iguchi Y, Katsuno M, Ikenaka K, Ishigaki S, Sobue G. Amyotrophic lateral sclerosis: an update on recent genetic insights. J Neurol. 2013;260(11):2917-27. doi:10.1007/s00415-013-7112-y,3939. Wu CH, Fallini C, Ticozzi N, Keagle PJ, Sapp PC, Piotrowska K et al. Mutations in the profilin 1 gene cause familial amyotrophic lateral sclerosis. Nature 2012;488(7412):499-503. doi:10.1038/nature11280.
ALS19
ALS19 (MIM #615515) represents a rare autosomal dominant form of late-onset slowly progressive familial ALS described in japanese and canadian families, resulting from heterozygous missense mutations in ERBB4 gene (V-ERB-B2 avian erythroblastic leucemia viral oncogene homolog 4; 2q34), coding the HER4 protein ligand for NDF/heregulin and neurregulin-ERBB4 pathways, involved in synaptic plasticity, cell proliferation and differentiation, glutamatergic hypofunction, and inhibition of NMDA currents and raise of AMPA currents4040. Takahashi Y, Fukuda Y, Yoshimura J, Toyoda A, Kurppa K, Moritoyo H et al. ERBB4 mutations that disrupt the neuregulin-ErbB4 pathway cause amyotrophic lateral sclerosis type 19. Am J Hum Genet. 2013;93(5):900-5. doi:10.1016/j.ajhg.2013.09.008.
ALS20
ALS20 (MIM #615426) represents an extremely rare autosomal dominant form of ALS with late-onset motor symptoms linked to heterozygous missense mutations in theHNRNPA1 gene (heterogeneous nuclear ribonucleoprotein A1; 12q13.13), coding the heterogeneous nuclear ribonucleoprotein A1, involved with splicing and processing of pre-messenger RNA and further metabolism and associates with core proteins of the protein moiety of the nuclear 40S ribonucleoprotein particle with RNA polymerase II transcripts. Allelic disorders are represented by inclusion body myopathy with early-onset Paget’s disease without frontotemporal dementia type 34141. Kim HJ, Kim NC, Wang YD, Scarborough EA, Moore J, Diaz Z et al. Mutations in prion-like domains in hnRNPA2B1 and hnRNPA1 cause multisystem proteinopathy and ALS. Nature. 2013;495(7442):467-73. doi:10.1038/nature11922.
ALS21
ALS21 (MIM #606070) represents a rare slowly-progressive autosomal dominant familial ALS linked to heterozygous missense mutations in the MATR3gene (matrin3; 5q31.2), coding the matrin-3 protein, an internal matrix nuclear protein which interacts with TDP-43 participating in the aberrant processing of RNA and stabilizing messenger RNA4242. Feit H, Silbergleit A, Schneider LB, Gutierrez JA, Fitoussi RP, Réyès C et al. Vocal cord and pharyngeal weakness with autosomal dominant distal myopathy: clinical description and gene localization to 5q31. Am J Hum Genet. 1998;63(6):1732-42. doi:10.1086/302166,4343. Johnson JO, Pioro EP, Boehringer A, Chia R, Feit H, Renton AE et al. Mutations in the Matrin 3 gene cause familial amyotrophic lateral sclerosis. Nat Neurosci. 2014;17(5):664-6. doi:10.1038/nn.3688.
ALS21 (formerly the vocal cord and pharyngeal dysfunction with distal myopathy type 2) presents with adult-onset dystal myopathy with inclusion body myopathy-like features and vocal cord and pharyngeal weakness, occasionally with lower limbs brisk reflexes and tongue fasciculations and death after 15 years of symptom-onset. Other European cases were described in assocation with dementia and MND. Other Indian patient presented with adult-onset ALS and hyper-CKemia4242. Feit H, Silbergleit A, Schneider LB, Gutierrez JA, Fitoussi RP, Réyès C et al. Vocal cord and pharyngeal weakness with autosomal dominant distal myopathy: clinical description and gene localization to 5q31. Am J Hum Genet. 1998;63(6):1732-42. doi:10.1086/302166,4343. Johnson JO, Pioro EP, Boehringer A, Chia R, Feit H, Renton AE et al. Mutations in the Matrin 3 gene cause familial amyotrophic lateral sclerosis. Nat Neurosci. 2014;17(5):664-6. doi:10.1038/nn.3688.
ALS22
ALS22 (MIM #616208), a recently described rare form of adult-onset familial ALS, correlates with TUBA4A gene (Tubulin, Alpha-4A; 2q35), coding the tubulin-alpha-4A protein, involved with microtubule network stabilization and reducing repolimerization dynamics. This form presents with late-onset familial spinal-dominant ALS with or without FTD4444. Smith BN, Ticozzi N, Fallini C, Gkazi AS, Topp S, Kenna KP et al. Exome-wide rare variant analysis identifies TUBA4A mutations associated with familial ALS. Neuron. 2014;84(2):324-31. doi:10.1016/j.neuron.2014.09.027.
FTD-ALS type 1
FTD-ALS type 1 (MIM #105550) represents the most common form of familial ALS. It occurs in an autosomal dominant inherited pattern in association with FTD and is associated with hexanucleotide repeat expansion (GGGGCC) in the non-conding intronic region of 5’ regulatory region of C9orf72 gene (chromosome 9 open reading frame 72; 9p21.2), coding the C9orf72 protein, involved in multiple intracellular mechanisms4545. Souza PV, Pinto WB, Oliveira AS. C9orf72-related disorders: expanding the clinical and genetic spectrum of neurodegenerative diseases. Arq Neuropsiquiatr. 2015;73(3):246-56. doi: 10.1590/0004-282X20140229,4646. Woollacott IO, Mead S. The C9ORF72 expansion mutation: gene structure, phenotypic and diagnostic issues. Acta Neuropathol. 2014;127(3):319-32. doi:10.1007/s00401-014-1253-7.
Loss of function by happloinsufficiency and toxic gain-of-function mechanisms are both present. C9orf72-related disorders result from abnormal membrane and endosomal trafficking (related to ubiquilin-2, HNRNPA1 and HNRNPA2B1 proteins), abnormal RNA processing and metabolism (with toxic RNA foci and gain-of-function), abnormal functions of ubiquitin-proteosome system, abnormal regulation of Rab guanine-nucleotide exchange factors involved with autophagia, and secondary axonal transport disturbances. Abnormal expansion repeats form RNA G-quadruplexes with distinct structures and promote formation of DNA/RNA hybrid R-loops with linkage of aborted transcripts to the expanded regions in ribonucleoproteins leading to nucleolar stress and neuronal apoptosis. Another mechanism involves the translation of expansion transcripts without the ATG start codon, the so-called RAN process (repeat-associated non-ATG translation), generating polipeptides with poli-glycine-arginine and poli-proline-arginine (also called dipeptide repeat proteins), which leads to neutoxicity by direct effects to DNA and binding to HNRNPA2 and changing RNA biogenesis88. Renton AE, Chiò A, Traynor BJ. State of play in amyotrophic lateral sclerosis genetics. Nat Neurosci. 2014;17(1):17-23. doi:10.1038/nn.3584,4646. Woollacott IO, Mead S. The C9ORF72 expansion mutation: gene structure, phenotypic and diagnostic issues. Acta Neuropathol. 2014;127(3):319-32. doi:10.1007/s00401-014-1253-7,4747. Cooper-Knock J, Shaw PJ, Kirby J. The widening spectrum of C9ORF72-related disease; genotype/phenotype correlations and potential modifiers of clinical phenotype. Acta Neuropathol. 2014;127(3):333-45. doi:10.1007/s00401-014-1251-9,4848. Renton AE, Majounie E, Waite A, Simón-Sánchez J, Rollinson S, Gibbs JR et al. A hexanucleotide repeat expansion in C9ORF72 is the cause of chromosome 9p21-linked ALS-FTD. Neuron. 2011;72(2):257-68. doi:10.1016/j.neuron.2011.09,4949. DeJesus-Hernandez M, Mackenzie IR, Boeve BF, Boxer AL, Baker M, Rutherford NJ et al. Expanded GGGGCC hexanucleotide repeat in noncoding region of C9ORF72 causes chromosome 9p-linked FTD and ALS. Neuron. 2011;72(2):245-56. doi:10.1016/j.neuron.2011.09.011.
Some neuropathological and molecular signatures fromC9orf72-related disorders differentiate them from other ALS and FTD genetic forms. Spinal motor neurons and glial cells may present with TDP-43 positive cytoplasmic inclusions in association with cortical and spinal cord intraneuronal cytoplasmic ubiquitin-positive, Tau-negative inclusions, FUS-negative and Ubiquilin 2 positive inclusions1212. Al-Chalabi A, Jones A, Troakes C, King A, Al-Sarraj S, Berg LH. The genetics and neuropathology of amyotrophic lateral sclerosis. Acta Neuropathol. 2012;124(3):339-52. doi:10.1007/s00401-012-1022-4. The same way, a nearly pathognomonic neuromolecular marker of C9orf72-related disorder is the presence of TDP-43 negative and p62-positive intraneuronal intracytoplasmic inclusions with dipeptide repeat proteins in extraspinal regions (dentate gyrus granule cells in the CA4 pyramidal cells of the hippocampus, frontal neocortex, and granule cells of the cerebellum). These dipeptide repeat proteins result from sense and antisense repeat associated non ATG-initiated translation of the expanded repeat noncoding region, previously described. Furthermore, the loss of dopaminergic neurons in substantia nigra has also been described inC9orf72-related ALS with parkinsonism, p62-positive inclusions and α-synuclein-negative Lewy bodies1212. Al-Chalabi A, Jones A, Troakes C, King A, Al-Sarraj S, Berg LH. The genetics and neuropathology of amyotrophic lateral sclerosis. Acta Neuropathol. 2012;124(3):339-52. doi:10.1007/s00401-012-1022-4.
C9orf72-associated ALS represents the main discovery in neuromuscular genetics since 2011 (former chromosome 9p-linked FTD with ALS)4848. Renton AE, Majounie E, Waite A, Simón-Sánchez J, Rollinson S, Gibbs JR et al. A hexanucleotide repeat expansion in C9ORF72 is the cause of chromosome 9p21-linked ALS-FTD. Neuron. 2011;72(2):257-68. doi:10.1016/j.neuron.2011.09,4949. DeJesus-Hernandez M, Mackenzie IR, Boeve BF, Boxer AL, Baker M, Rutherford NJ et al. Expanded GGGGCC hexanucleotide repeat in noncoding region of C9ORF72 causes chromosome 9p-linked FTD and ALS. Neuron. 2011;72(2):245-56. doi:10.1016/j.neuron.2011.09.011 and the major cause of ALS ranging from one third up to 46% of familial cases and from 6% up to 21% of sporadic cases2020. Ince PG, Highley JR, Kirby J, Wharton SB, Takahashi H, Strong MJ et al. Molecular pathology and genetic advances in amyotrophic lateral sclerosis: an emerging molecular pathway and the significance of glial pathology. Acta Neuropathol. 2011;122(6):657-71. doi:10.1007/s00401-011-0913-0. There is a clear tendency of low prevalence in cases from Asian countries and Pacific islands. On the other hand, high sporadic prevalences are detected in Finland (with a founder effect), Sweden, United Kingdom, Netherlands, Greece and USA, and high familial rates in Belgium, Sweden, Greece, Finland, Ireland, France and United Kingdom4646. Woollacott IO, Mead S. The C9ORF72 expansion mutation: gene structure, phenotypic and diagnostic issues. Acta Neuropathol. 2014;127(3):319-32. doi:10.1007/s00401-014-1253-7,4747. Cooper-Knock J, Shaw PJ, Kirby J. The widening spectrum of C9ORF72-related disease; genotype/phenotype correlations and potential modifiers of clinical phenotype. Acta Neuropathol. 2014;127(3):333-45. doi:10.1007/s00401-014-1251-9.
Neurological symptomatic cases arise with 250 up to 1600 expansion repeats, as healthy people most commonly present with 2 to 19 repeats4646. Woollacott IO, Mead S. The C9ORF72 expansion mutation: gene structure, phenotypic and diagnostic issues. Acta Neuropathol. 2014;127(3):319-32. doi:10.1007/s00401-014-1253-7,4747. Cooper-Knock J, Shaw PJ, Kirby J. The widening spectrum of C9ORF72-related disease; genotype/phenotype correlations and potential modifiers of clinical phenotype. Acta Neuropathol. 2014;127(3):333-45. doi:10.1007/s00401-014-1251-9. The clinical picture ofC9orf72-related disorders is expanding its phenotypical spectrum. Family history of FTD-ALS patients may unveil dementia (with FTD, dementia with diffuse Lewy bodies or Alzheimer’s disease), atypical parkinsonism (with rapidly progressive progressive supranuclear palsy and corticobasal degeneration), complex movement disorders (Huntington’s disease-like), and psychiatric disorders (mainly late-onset psychosis). Intense intrafamilial clinical variability makes difficult the recognition of this neurodegenerative complex4646. Woollacott IO, Mead S. The C9ORF72 expansion mutation: gene structure, phenotypic and diagnostic issues. Acta Neuropathol. 2014;127(3):319-32. doi:10.1007/s00401-014-1253-7,4747. Cooper-Knock J, Shaw PJ, Kirby J. The widening spectrum of C9ORF72-related disease; genotype/phenotype correlations and potential modifiers of clinical phenotype. Acta Neuropathol. 2014;127(3):333-45. doi:10.1007/s00401-014-1251-9.
Thus, C9orf72-related ALS must be investigated in cases of adult-onset familial ALS, mainly in non-Asian patient cases with FTD phenotype, or in cases with a complex familial network of neurodegenerative disorders, including adult and late-onset chorea, atypical parkinsonism or isolated psychiatric syndromes.
FTD-ALS type 2
FTD-ALS type 2 (MIM #615911) represents a rare autosomal dominant form of familial ALS with frontotemporal dementia, resulting from heterozygous mutations in the CHCHD10 gene (Coiled-coil-helix-coiled-coil-helixdomain-containing protein 10; 22q11.23), coding the CHCHD10 protein, involved in oxidative phosphorylation and maintenance of cristae morphology in the inner intermembranous mitochondrial space. In France and Spain patients around the fifth decade of life present with complex neurological phenotypes involving frontotemporal dementia, cerebellar ataxia, myopathy with ragged-red fibers and COX-negative fibers, motor neuron disease with progressive bulbar dysfunction, and rarely with akinetic-rigid parkinsonism and sensorineural deafness5050. Bannwarth S, Ait-El-Mkadem S, Chaussenot A, et al. A mitochondrial origin for frontotemporal dementia and amyotrophic lateral sclerosis through CHCHD10 involvement. Brain. 2014;137(8):2329-45. doi:10.1093/brain/awu138.
UNCLASSIFIED FORMS OF FAMILIAL ALS
Several genetic forms of familial ALS, well-recognized by specialists, are not characterized as a single familial ALS form in the Monogenic Muscle Gene Table classification (World Muscle Society)5151. Kaplan JC, Hamroun D. The 2015 version of the gene table of monogenic neuromuscular disorders (nuclear genome). Neuromuscul Disord. 2014;24(12):1123-53. doi:10.1016/j.nmd.2014.11.001. The most distinguished forms are discussed here.
Familial forms associated to NEFH gene (neurofilament, heavy polypepeptide; 22q12.2) are involved in monomelic ALS with dementia and severe dysphagia in Scandinavia. NEFH gene codes the heavy polypeptide neurofilament, the most important neuron-specific intermediate filament of cytoskeleton in myelinated axons, involved in maintenance of cytoskeleton and axonal architecture in proximal axonal region of spinal motoneurons5151. Kaplan JC, Hamroun D. The 2015 version of the gene table of monogenic neuromuscular disorders (nuclear genome). Neuromuscul Disord. 2014;24(12):1123-53. doi:10.1016/j.nmd.2014.11.001,5252. Al-Chalabi A, Andersen PM, Nilsson P, Chioza B, Andersson JL, Russ C et al. Deletions of the heavy neurofilament subunit tail in amyotrophic lateral sclerosis. Hum Molec Genet. 1999;8(2):157-64. doi:10.1093/hmg/8.2.157.
PRPH-associated familial ALS represents an adult-onset ALS with Lewy body-like inclusions and neuroaxonal spheroids in proximal axonal region of spinal motoneurons. It is associated with mutations in PRPH gene (peripherin; 12q13.12), coding peripherin, an intermediate filament type 3 neuronal cytoskeleton similar to neurofilament5151. Kaplan JC, Hamroun D. The 2015 version of the gene table of monogenic neuromuscular disorders (nuclear genome). Neuromuscul Disord. 2014;24(12):1123-53. doi:10.1016/j.nmd.2014.11.001,5353. Leung CL, He CZ, Kaufmann P, Chin SS, Naini A, Liem RK et al. A pathogenic peripherin gene mutation in a patient with amyotrophic lateral sclerosis. Brain Path. 2004;14(3):290-6. doi:10.1111/j.1750-3639.2004.tb00066.x.
DCTN1-related disorders are involved in a complex spectrum of neurodegenerative allelic disorders, including hereditary distal motor neuropathy type VIIB, FTD-like phenotypes and atypical parkinsonism, like PSP and Perry syndrome. A lower motor neuron dominant slowly progressive ALS with vocal cord and facial palsy arises as a consequence of G59S heterozygous mutation in DCTN1(dynactin 1; 2p13.1), coding dynactin-1, involved with dynactin complex connection with microtubules and cytoplasm dynein for axonal transport5454. LaMonte BH, Wallace KE, Holloway BA, Shelly SS, Ascaño J, Tokito M et al. Disruption of dynein/dynactin inhibits axonal transport in motor neurons causing late-onset progressive degeneration. Neuron. 2002;34(5):715-27. doi:10.1016/S0896-6273(02)00696-7.
DAO (D-aminoacid oxidase; 12q24.11) gene mutations are also involved with very rare adult-onset familial ALS. Dysfunction of D-aminoacid oxidase gives rise to abnormal downregulation of D-serine, a normal co-agonist of excitatory NMDA glutamate receptors, as a consequence of abnormal cerebellar and spinal oxidative deamination of D-aminoacids. There is association with schizophrenia in canadian patients66. Iguchi Y, Katsuno M, Ikenaka K, Ishigaki S, Sobue G. Amyotrophic lateral sclerosis: an update on recent genetic insights. J Neurol. 2013;260(11):2917-27. doi:10.1007/s00415-013-7112-y,5555. Millecamps S, Da Barroca S, Cazeneuve C, Salachas F, Pradat PF, Danel-Brunaud V et al. Questioning of the role of D amino acid oxidase in familial amyotrophic lateral sclerosis. Proc Natl Acad Sci U S A. 2010;107(26):E107. doi:10.1073/pnas.1006190107.
SQSTM1-associated ALS correlates with adult-onset autosomal dominant familial ALS associated with Paget disease of bone, representing up to 1% of familial cases. SQSTM1 gene (Sequestosome 1; 5q35.3) mutations are involved with abnormal coding of p62 protein, involved in ubiquitination and authophagy by activation of NF-κB pathway44. Su XW, Broach JR, Connor JR, Gerhard GS, Simmons Z. Genetic heterogeneity of amyotrophic lateral sclerosis: implications for clinical practice and research. Muscle Nerve. 2014;49(6):786-803. doi:10.1002/mus.24198,66. Iguchi Y, Katsuno M, Ikenaka K, Ishigaki S, Sobue G. Amyotrophic lateral sclerosis: an update on recent genetic insights. J Neurol. 2013;260(11):2917-27. doi:10.1007/s00415-013-7112-y.
TAF15 (TATA Box-binding protein-associated factor RNA polymerase II; 17q12) and EWSR1 (Ewing sarcoma breakpoint region 1; 22q12.2) genes are involved with extremely rare adult-onset familial ALS, both participating directly in transcription processes with other activators or repressors: the former coding RBP56 protein (RNA-binding protein 56), allelic to extraskeletal mixoid condrosarcoma; and the last coding EWS protein, allelic to neuroepithelioma and Ewing sarcoma44. Su XW, Broach JR, Connor JR, Gerhard GS, Simmons Z. Genetic heterogeneity of amyotrophic lateral sclerosis: implications for clinical practice and research. Muscle Nerve. 2014;49(6):786-803. doi:10.1002/mus.24198,66. Iguchi Y, Katsuno M, Ikenaka K, Ishigaki S, Sobue G. Amyotrophic lateral sclerosis: an update on recent genetic insights. J Neurol. 2013;260(11):2917-27. doi:10.1007/s00415-013-7112-y.
A rare autosomal recessive juvenile ALS form (MIM %205200) has been described in Dutch and Amish patients with early juvenile-onset dementia during the first decade with distal muscular atrophy and death after up to two decades of symptom-onset5656. Staal A, Went LN. Juvenile amyotrophic lateral sclerosis-dementia complex in a Dutch family. Neurology. 1968;18(8):800-6. doi:10.1212/WNL.18.8.800.
Another recently described familial ALS form has been described in Utah, USA, involving autosomal recessive inheritance and clinically manifest with upper motor neuron dominant juvenile ALS, with slowly progressive motor symptoms starting in the lower limbs and bulbar region in the first decade of life and evolving with partial eyelid ptosis, gynecomastia and mild distal hypopalesthesia22. Leblond CS, Kaneb HM, Dion PA, Rouleau GA. Dissection of genetic factors associated with amyotrophic lateral sclerosis. Exp Neurol. 2014;262(Part B):91-101. doi:10.1016/j.expneurol.2014.04.013,33. He J, Mangelsdorf M, Fan D, Bartlett P, Brown MA. Amyotrophic Lateral Sclerosis genetic studies: from genome-wide association mapping to genome sequencing. Neuroscientist. 2014;pii:1073858414555404. Forthcoming. doi:10.1177/1073858414555404.
Amyotrophic lateral sclerosis-parkinsonism/dementia complex type 1 results from mutations in the TRPM7 gene (Transient receptor potential cation channel, subfamily M, member 7; 15q21.2), coding the transient receptor potential cation channel subfamily M member 7, involved in regulation of oxidative stress metabolism and in the phosphorylation of different intracellular substrates. Susceptibility from direct exposure to neurotoxic effects β-methylamino-L-alanine found in local Guam species of flying fox (Pteropus tokudae) was described in patients from the Chamorro population from Guam Island in USA which posteriorly become called the Lytico-bodig disease5757. Morris HR, Al-Sarraj S, Schwab C, Gwinn-Hardy K, Perez-Tur J, Wood NW et al. A clinical and pathological study of motor neurone disease on Guam. Brain. 2001;124(11):2215-22. doi:10.1093/brain/124.11.2215. Most authors do not include Lytico-bodig disease as familial ALS. However, this apparently narrowed distribution of this ALS form has also been linked to another form of ALS described in the Kii Peninsula of Japan, also known as Muro disease with a complex neurological phenotype with dementia and movement disorders (including parkinsonism, dystonia and myoclonus), in which TRPM7, C9orf72 andSOD1-related mechanisms have been described5858. Shindo A, Ueda Y, Kuzuhara S, Kokubo Y. Neuropsychological study of amyotrophic lateral sclerosis and parkinsonism-dementia complex in Kii peninsula, Japan. BMC Neurol. 2014;14(1):151. doi:10.1186/1471-2377-14-151.
FAMILIAL AND SPORADIC FORMS ASSOCIATED WITH OTHER SUSCEPTIBILITY LOCI
In many of the so-called sporadic cases, there is an important role of genetic factors usually associated with incomplete penetrance and the complex stochastic predisposing events and the individual and environmental risk factors5959. Marangi G, Traynor BJ. Genetic causes of amyotrophic lateral sclerosis: new genetic analysis methodologies entailing new opportunities and challenges. Brain Res. 2014;1607:75-93. doi:10.1016/j.brainres.2014.10.009. Much data on susceptibility loci results from linkage analysis studies, candidate-gene association studies, genome-wide association studies and methylation analysis, copy number variants and chromosomal rearrangements studies, and whole-exome or genome-sequencing studies22. Leblond CS, Kaneb HM, Dion PA, Rouleau GA. Dissection of genetic factors associated with amyotrophic lateral sclerosis. Exp Neurol. 2014;262(Part B):91-101. doi:10.1016/j.expneurol.2014.04.013,33. He J, Mangelsdorf M, Fan D, Bartlett P, Brown MA. Amyotrophic Lateral Sclerosis genetic studies: from genome-wide association mapping to genome sequencing. Neuroscientist. 2014;pii:1073858414555404. Forthcoming. doi:10.1177/1073858414555404,5959. Marangi G, Traynor BJ. Genetic causes of amyotrophic lateral sclerosis: new genetic analysis methodologies entailing new opportunities and challenges. Brain Res. 2014;1607:75-93. doi:10.1016/j.brainres.2014.10.009. Most clinical descriptions are related to single nucleotide polymorphisms (SNPs), polyglutamine repeats, missense mutations, abnormal copy number of genes, gene promoter SNPs, and insertions and deletions77. Chen S, Sayana P, Zhang X, Le W. Genetics of amyotrophic lateral sclerosis: an update. Mol Neurodegener. 2013;8(1):28. doi:10.1186/1750-1326-8-28. More than 20 different gene loci have been described so far.
CYP27A1 gene (Cytochrome P450, Subfamily XXVIIA, Polypeptide 1; 2q35) mutations have been described in cerebrotendinous xanthomatosis (CTX) (MIM #213700) and as a susceptibility loci for sporadic ALS6060. Diekstra FP, Saris CG, van Rheenen W, Franke L, Jansen RC, Es MA et al. Mapping of gene expression reveals CYP27A1 as a susceptibility gene for sporadic ALS. PLoS One. 2012;7(4):e35333. doi:10.1371/journal.pone.0035333. In our experience, CTX may present with an early-onset lower motor dominant juvenile ALS phenotype in two Brazilian sisters with severe bulbar compromise, orofacial dyskinesia and generalized epilepsy. One of them presented also with typical systemic signs of CTX, including chronic diarrhea, juvenile bilateral cataracts and early-onset premenopausal osteopenia (unpublished data). Other gene loci are also involved with sporadic forms described as susceptibility loci (most as SNP associations or demonstrated by genome-wide association studies) for which there is some degree of suspicion related to familial cases (Table 3)22. Leblond CS, Kaneb HM, Dion PA, Rouleau GA. Dissection of genetic factors associated with amyotrophic lateral sclerosis. Exp Neurol. 2014;262(Part B):91-101. doi:10.1016/j.expneurol.2014.04.013,44. Su XW, Broach JR, Connor JR, Gerhard GS, Simmons Z. Genetic heterogeneity of amyotrophic lateral sclerosis: implications for clinical practice and research. Muscle Nerve. 2014;49(6):786-803. doi:10.1002/mus.24198,77. Chen S, Sayana P, Zhang X, Le W. Genetics of amyotrophic lateral sclerosis: an update. Mol Neurodegener. 2013;8(1):28. doi:10.1186/1750-1326-8-28,1212. Al-Chalabi A, Jones A, Troakes C, King A, Al-Sarraj S, Berg LH. The genetics and neuropathology of amyotrophic lateral sclerosis. Acta Neuropathol. 2012;124(3):339-52. doi:10.1007/s00401-012-1022-4,1313. Ticozzi N, Tiloca C, Morelli C, Colombrita C, Poletti B, Doretti A et al. Genetics of familial Amyotrophic lateral sclerosis. Arch Ital Biol. 2011;149(1):65-82. doi:10.4449/aib.v149i1.1262,5959. Marangi G, Traynor BJ. Genetic causes of amyotrophic lateral sclerosis: new genetic analysis methodologies entailing new opportunities and challenges. Brain Res. 2014;1607:75-93. doi:10.1016/j.brainres.2014.10.009.
CONCLUSION
Although most cases of ALS are sporadic, familial ALS cases represent important clinical, genetic and neuropathological keys to understand the natural history of different genetic forms, clinical and genetic correlations, and allow new genetic targeted therapies for ALS. Despite the current trend of ALS diagnoses are limited to syndromic clinical descriptions, in the near future it will be essential to establish the genetic types associated with the different clinical subtypes and presentations, especially in familial cases and in complex neurological phenotypes.
References
-
1Oliveira ASB, Pereira RDB. Amyotrophic lateral sclerosis (ALS): three letters that change the people’s life. For ever. Arq Neuropsiquiatr. 2009;67(3A):750-82. doi:10.1590/S0004-282X2009000400040
-
2Leblond CS, Kaneb HM, Dion PA, Rouleau GA. Dissection of genetic factors associated with amyotrophic lateral sclerosis. Exp Neurol. 2014;262(Part B):91-101. doi:10.1016/j.expneurol.2014.04.013
-
3He J, Mangelsdorf M, Fan D, Bartlett P, Brown MA. Amyotrophic Lateral Sclerosis genetic studies: from genome-wide association mapping to genome sequencing. Neuroscientist. 2014;pii:1073858414555404. Forthcoming. doi:10.1177/1073858414555404
-
4Su XW, Broach JR, Connor JR, Gerhard GS, Simmons Z. Genetic heterogeneity of amyotrophic lateral sclerosis: implications for clinical practice and research. Muscle Nerve. 2014;49(6):786-803. doi:10.1002/mus.24198
-
5Ferraiuolo L, Kirby J, Grierson AJ, Sendtner M, Shaw PJ. Molecular pathways of motor neuron injury in amyotrophic lateral sclerosis. Nat Rev Neurol. 2011;7(11):616-30. doi:10.1038/nrneurol.2011.152
-
6Iguchi Y, Katsuno M, Ikenaka K, Ishigaki S, Sobue G. Amyotrophic lateral sclerosis: an update on recent genetic insights. J Neurol. 2013;260(11):2917-27. doi:10.1007/s00415-013-7112-y
-
7Chen S, Sayana P, Zhang X, Le W. Genetics of amyotrophic lateral sclerosis: an update. Mol Neurodegener. 2013;8(1):28. doi:10.1186/1750-1326-8-28
-
8Renton AE, Chiò A, Traynor BJ. State of play in amyotrophic lateral sclerosis genetics. Nat Neurosci. 2014;17(1):17-23. doi:10.1038/nn.3584
-
9Turner MR, Swash M. The expanding syndrome of amyotrophic lateral sclerosis: a clinical and molecular odyssey. J Neurol Neurosurg Psychiatry. 2015;86(6):667-73. doi:10.1136/jnnp-2014-308946
-
10Kanouchi T, Ohkubo T, Yokota T. Can regional spreading of amyotrophic lateral sclerosis motor symptoms be explained by prion-like propagation? J Neurol Neurosurg Psychiatry. 2012;83(7):739-45. doi:10.1136/jnnp-2011-301826
-
11Strong MJ, Grace GM, Freedman M, Lomen-Hoerth C, Woolley S, Goldstein LH et al. Consensus criteria for the diagnosis of frontotemporal cognitive and behavioural syndromes in amyotrophic lateral sclerosis. Amyotroph Lateral Scler 2009;10(3):131-46. doi:10.1080/17482960802654364
-
12Al-Chalabi A, Jones A, Troakes C, King A, Al-Sarraj S, Berg LH. The genetics and neuropathology of amyotrophic lateral sclerosis. Acta Neuropathol. 2012;124(3):339-52. doi:10.1007/s00401-012-1022-4
-
13Ticozzi N, Tiloca C, Morelli C, Colombrita C, Poletti B, Doretti A et al. Genetics of familial Amyotrophic lateral sclerosis. Arch Ital Biol. 2011;149(1):65-82. doi:10.4449/aib.v149i1.1262
-
14Conte A, Lattante S, Luigetti M, Del Grande A, Romano A, Marcaccio A et al. Classification of familial amyotrophic lateral sclerosis by family history: effects on frequency of genes mutation. J Neurol Neurosurg Psychiatry. 2012;83(12):1201-3. doi:10.1136/jnnp-2012-302897
-
15Turner MR, Barnwell J, Al-Chalabi A, Eisen A. Young-onset amyotrophic lateral sclerosis: historical and other observations. Brain. 2012;135(9):2883-91. doi:10.1093/brain/aws144
-
16Horton WA, Eldridge R, Brody JA. Familial motor neuron disease: evidence for at least three different types. Neurology. 1976;26(5):460-5. doi:10.1212/WNL.26.5.460
-
17Al-Chalabi A, Hardiman O. The epidemiology of ALS: a conspiracy of genes, environment and time. Nat Rev Neurol. 2013;9(11):617-8. doi:10.1038/nrneurol.2013.203
-
18Van Langenhove T, Zee J, Van Broeckhoven C. The molecular basis of the frontotemporal lobar degeneration-amyotrophic lateral sclerosis spectrum. Ann Med. 2012;44(8):817-28. doi:10.3109/07853890.2012.665471
-
19Blitterswijk M, v Es MA, Hennekam EA, Dooijes D, Rheenen W, Medic J et al. Evidence for an oligogenic basis of amyotrophic lateral sclerosis. Hum Mol Genet. 2012;21(17):3776-84. doi:10.1093/hmg/dds199
-
20Ince PG, Highley JR, Kirby J, Wharton SB, Takahashi H, Strong MJ et al. Molecular pathology and genetic advances in amyotrophic lateral sclerosis: an emerging molecular pathway and the significance of glial pathology. Acta Neuropathol. 2011;122(6):657-71. doi:10.1007/s00401-011-0913-0
-
21Shirakawa K, Suzuki H, Ito M, Kono S, Uchiyama T, Ohashi T et al. Novel compound heterozygous ALS2 mutations cause juvenile amyotrophic lateral sclerosis in Japan. Neurology. 2009;73(24):2124-6. doi:10.1212/WNL.0b013e3181c67be0
-
22Hand CK, Khoris J, Salachas F, Gros-Louis F, Lopes AA, Mayeux-Portas V et al. A novel locus for familial amyotrophic lateral sclerosis, on chromosome 18q. Am J Hum Genet. 2002;70(1):251-6. doi:10.1086/337945
-
23Chen YZ, Bennett CL, Huynh HM, Blair IP, Puls I, Irobi J et al. DNA/RNA helicase gene mutations in a form of juvenile amyotrophic lateral sclerosis (ALS4). Am J Hum Genet. 2004;74(6):1128-35. doi:10.1086/421054
-
24Hentati A, Ouahchi K, Pericak-Vance MA, Nijhawan D, Ahmad A, Yang Y et al. Linkage of a commoner form of recessive amyotrophic lateral sclerosis to chromosome 15q15-q22 markers. Neurogenetics 1998;2(1):55-60. doi:10.1007/s100480050052
-
25Daoud H, Zhou S, Noreau A, Sabbagh M, Belzil V, Dionne-Laporte A et al. Exome sequencing reveals SPG11 mutations causing juvenile ALS. Neurobiol Aging. 2012;33(4):839:e5-9. doi:10.1016/j.neurobiolaging.2011.11.012
-
26Hewitt C, Kirby J, Highley JR, Hartley JA, Hibberd R, Hollinger HC et al. Novel FUS/TLS mutations and pathology in familial and sporadic amyotrophic lateral sclerosis. Arch Neurol. 2010;67(4):455-61. doi:10.1001/archneurol.2010.52
-
27Bäumer D, Hilton D, Paine SM, Turner MR, Lowe J, Talbot K et al. Juvenile ALS with basophilic inclusions is a FUS proteinopathy with FUS mutations. Neurology. 2010;75(7):611-8. doi:10.1212/WNL.0b013e3181ed9cde
-
28Sapp PC, Hosler BA, McKenna-Yasek D, Chin W, Gann A, Genise H et al. Identification of two novel loci for dominantly inherited familial amyotrophic lateral sclerosis. Am J Hum Genet. 2003;73(2):397-403. doi:10.1086/377158
-
29Nishimura AL, Mitne-Neto M, Silva HCA, Richieri-Costa A, Middleton S, Cascio D et al. A mutation in the vesicle-trafficking protein VAPB causes late-onset spinal muscular atrophy and amyotrophic lateral sclerosis. Am J Hum Genet. 2004;75(5):822-31. doi:10.1086/425287
-
30Millecamps S, Salachas F, Cazeneuve C, Gordon P, Bricka B, Camuzat A et al. SOD1, ANG, VAPB, TARDBP, and FUS mutations in familial amyotrophic lateral sclerosis: genotype-phenotype correlations. J Med Genet. 2010;47(8):554-60. doi:10.1136/jmg.2010.077180
-
31Corcia P, Valdmanis P, Millecamps S, Lionnet C, Blasco H, Mouzat K et al. Phenotype and genotype analysis in amyotrophic lateral sclerosis with TARDBP gene mutations. Neurology. 2012;78(19):1519-26. doi:10.1212/WNL.0b013e3182553c88
-
32Chow CY, Landers JE, Bergren SK, Sapp PC, Grant AE, Jones JM et al. Deleterious variants of FIG4, a phosphoinositide phosphatase, in patients with ALS. Am J Hum Genet. 2009;84(1):85-8. doi:10.1016/j.ajhg.2008.12.010
-
33Maruyama H, Morino H, Ito H, Izumi Y, Kato H, Watanabe Y et al. Mutations of optineurin in amyotrophic lateral sclerosis. Nature. 2010;465(7295):223-6. doi:10.1038/nature08971
-
34Van Damme P, Veldink JH, Blitterswijk M, Corveleyn A, Vught PW, Thijs V et al. Expanded ATXN2 CAG repeat size in ALS identifies genetic overlap between ALS and SCA2. Neurology. 2011;76(24):2066-72. doi:10.1212/WNL.0b013e31821f445b
-
35Johnson JO, Mandrioli J, Benatar M, Abramzon Y, Van Deerlin VM, Trojanowski JQ et al. Exome sequencing reveals VCP mutations as a cause of familial ALS. Neuron. 2010;68(5):857-64. doi:10.1016/j.neuron.2010.11.036
-
36Deng HX, Chen W, Hong ST, Boycott KM, Gorrie GH, Siddique N et al. Mutations in UBQLN2 cause dominant X-linked juvenile and adult-onset ALS and ALS/dementia. Nature. 2011;477(7363):211-5. doi:10.1038/nature10353
-
37Al-Saif A, Al-Mohanna F, Bohlega S. A mutation in sigma-1 receptor causes juvenile amyotrophic lateral sclerosis. Ann Neurol. 2011;70(6):913-9. doi:10.1002/ana.22534
-
38Cox LE, Ferraiuolo L, Goodall EF, Heath PR, Higginbottom A, Mortiboys H et al. Mutations in CHMP2B in lower motor neuron predominant amyotrophic lateral sclerosis (ALS). PLoS One. 2010;5(3):e9872. doi:10.1371/journal.pone.0009872
-
39Wu CH, Fallini C, Ticozzi N, Keagle PJ, Sapp PC, Piotrowska K et al. Mutations in the profilin 1 gene cause familial amyotrophic lateral sclerosis. Nature 2012;488(7412):499-503. doi:10.1038/nature11280
-
40Takahashi Y, Fukuda Y, Yoshimura J, Toyoda A, Kurppa K, Moritoyo H et al. ERBB4 mutations that disrupt the neuregulin-ErbB4 pathway cause amyotrophic lateral sclerosis type 19. Am J Hum Genet. 2013;93(5):900-5. doi:10.1016/j.ajhg.2013.09.008
-
41Kim HJ, Kim NC, Wang YD, Scarborough EA, Moore J, Diaz Z et al. Mutations in prion-like domains in hnRNPA2B1 and hnRNPA1 cause multisystem proteinopathy and ALS. Nature. 2013;495(7442):467-73. doi:10.1038/nature11922
-
42Feit H, Silbergleit A, Schneider LB, Gutierrez JA, Fitoussi RP, Réyès C et al. Vocal cord and pharyngeal weakness with autosomal dominant distal myopathy: clinical description and gene localization to 5q31. Am J Hum Genet. 1998;63(6):1732-42. doi:10.1086/302166
-
43Johnson JO, Pioro EP, Boehringer A, Chia R, Feit H, Renton AE et al. Mutations in the Matrin 3 gene cause familial amyotrophic lateral sclerosis. Nat Neurosci. 2014;17(5):664-6. doi:10.1038/nn.3688
-
44Smith BN, Ticozzi N, Fallini C, Gkazi AS, Topp S, Kenna KP et al. Exome-wide rare variant analysis identifies TUBA4A mutations associated with familial ALS. Neuron. 2014;84(2):324-31. doi:10.1016/j.neuron.2014.09.027
-
45Souza PV, Pinto WB, Oliveira AS. C9orf72-related disorders: expanding the clinical and genetic spectrum of neurodegenerative diseases. Arq Neuropsiquiatr. 2015;73(3):246-56. doi: 10.1590/0004-282X20140229
-
46Woollacott IO, Mead S. The C9ORF72 expansion mutation: gene structure, phenotypic and diagnostic issues. Acta Neuropathol. 2014;127(3):319-32. doi:10.1007/s00401-014-1253-7
-
47Cooper-Knock J, Shaw PJ, Kirby J. The widening spectrum of C9ORF72-related disease; genotype/phenotype correlations and potential modifiers of clinical phenotype. Acta Neuropathol. 2014;127(3):333-45. doi:10.1007/s00401-014-1251-9
-
48Renton AE, Majounie E, Waite A, Simón-Sánchez J, Rollinson S, Gibbs JR et al. A hexanucleotide repeat expansion in C9ORF72 is the cause of chromosome 9p21-linked ALS-FTD. Neuron. 2011;72(2):257-68. doi:10.1016/j.neuron.2011.09
-
49DeJesus-Hernandez M, Mackenzie IR, Boeve BF, Boxer AL, Baker M, Rutherford NJ et al. Expanded GGGGCC hexanucleotide repeat in noncoding region of C9ORF72 causes chromosome 9p-linked FTD and ALS. Neuron. 2011;72(2):245-56. doi:10.1016/j.neuron.2011.09.011
-
50Bannwarth S, Ait-El-Mkadem S, Chaussenot A, et al. A mitochondrial origin for frontotemporal dementia and amyotrophic lateral sclerosis through CHCHD10 involvement. Brain. 2014;137(8):2329-45. doi:10.1093/brain/awu138
-
51Kaplan JC, Hamroun D. The 2015 version of the gene table of monogenic neuromuscular disorders (nuclear genome). Neuromuscul Disord. 2014;24(12):1123-53. doi:10.1016/j.nmd.2014.11.001
-
52Al-Chalabi A, Andersen PM, Nilsson P, Chioza B, Andersson JL, Russ C et al. Deletions of the heavy neurofilament subunit tail in amyotrophic lateral sclerosis. Hum Molec Genet. 1999;8(2):157-64. doi:10.1093/hmg/8.2.157
-
53Leung CL, He CZ, Kaufmann P, Chin SS, Naini A, Liem RK et al. A pathogenic peripherin gene mutation in a patient with amyotrophic lateral sclerosis. Brain Path. 2004;14(3):290-6. doi:10.1111/j.1750-3639.2004.tb00066.x
-
54LaMonte BH, Wallace KE, Holloway BA, Shelly SS, Ascaño J, Tokito M et al. Disruption of dynein/dynactin inhibits axonal transport in motor neurons causing late-onset progressive degeneration. Neuron. 2002;34(5):715-27. doi:10.1016/S0896-6273(02)00696-7
-
55Millecamps S, Da Barroca S, Cazeneuve C, Salachas F, Pradat PF, Danel-Brunaud V et al. Questioning of the role of D amino acid oxidase in familial amyotrophic lateral sclerosis. Proc Natl Acad Sci U S A. 2010;107(26):E107. doi:10.1073/pnas.1006190107
-
56Staal A, Went LN. Juvenile amyotrophic lateral sclerosis-dementia complex in a Dutch family. Neurology. 1968;18(8):800-6. doi:10.1212/WNL.18.8.800
-
57Morris HR, Al-Sarraj S, Schwab C, Gwinn-Hardy K, Perez-Tur J, Wood NW et al. A clinical and pathological study of motor neurone disease on Guam. Brain. 2001;124(11):2215-22. doi:10.1093/brain/124.11.2215
-
58Shindo A, Ueda Y, Kuzuhara S, Kokubo Y. Neuropsychological study of amyotrophic lateral sclerosis and parkinsonism-dementia complex in Kii peninsula, Japan. BMC Neurol. 2014;14(1):151. doi:10.1186/1471-2377-14-151
-
59Marangi G, Traynor BJ. Genetic causes of amyotrophic lateral sclerosis: new genetic analysis methodologies entailing new opportunities and challenges. Brain Res. 2014;1607:75-93. doi:10.1016/j.brainres.2014.10.009
-
60Diekstra FP, Saris CG, van Rheenen W, Franke L, Jansen RC, Es MA et al. Mapping of gene expression reveals CYP27A1 as a susceptibility gene for sporadic ALS. PLoS One. 2012;7(4):e35333. doi:10.1371/journal.pone.0035333
Publication Dates
-
Publication in this collection
13 Oct 2015 -
Date of issue
Dec 2015
History
-
Received
08 Mar 2015 -
Reviewed
27 June 2015 -
Accepted
17 July 2015