Resumos
INTRODUÇÃO: O principal mecanismo de resistência entre isolados de Pseudomonas aeruginosa e Acinetobacter sp. é a produção de metalo-β-lactamases (MβLs). As MβLs são enzimas capazes de hidrolisar cefalosporinas, penicilinas e carbapenêmicos, mas não monobactâmicos (aztreonam) antibióticos que se encontram entre as principais opções terapêuticas para o tratamento de infecções causadas por bactérias não fermentadoras de glicose. MÉTODOS: Um estudo observacional, transversal, descritivo e retrospectivo foi desenvolvido para avaliar a frequência e o perfil de susceptibilidade cepas de P. aeruginosa e Acinetobacter sp. produtoras de MβLs isoladas no Hospital São Vicente de Paulo, Passo Fundo, Brasil. RESULTADOS: A produção de MβLs foi observada em 77,6% (n = 173/223) dos isolados de P. aeruginosa e em 22,4% (n = 50/223) dos isolados de Acinetobacter sp. Dentre as cepas produtoras de MβL, a maioria apresentou mais de 90% de resistência a seis antimicrobianos dos 12 testados, enfatizando a resistência a ceftazidima, gentamicina, aztreonam, piperaciclina/tazobactam, cefepime, ciprofloxacina, meropenem e tobramicina. CONCLUSÕES: Os índices de MβL encontrados confirmam a preocupação mundial com a disseminação desse mecanismo de resistência.
MβL; Resistência bacteriana; Pseudomonas aeruginosa; Acinetobacter
INTRODUCTION: The main mechanism of emerging resistance in Pseudomonas aeruginosa and Acinetobacter sp. isolates is the production of metallo-β-lactamases (MβLs). MβLs are enzymes capable of hydrolyzing cephalosporins, penicillins and carbapenems, but not monobactams (aztreonam), which are often used as antimicrobial therapy to treat nosocomial infections. METHODS: An observational descriptive and retrospective study was designed to assess the frequency of MβLs among strains of P. aeruginosa and Acinetobacter sp. obtained from a tertiary hospital in southern Brazil. RESULTS: MβL production was observed in 77.6% (n = 173/223) for P. aeruginosa isolates and 22.4% (n = 50/223) of Acinetobacter sp. isolates. The Acinetobacter sp. isolates showed 92.8% sensitivity to amikacin and P. aeruginosa isolates showed 58.9% sensitivity to aztreonam. CONCLUSIONS: The MβL indices determined confirm the global concern with this mechanism of resistance.
MβL; Bacterial resistance; Pseudomonas aeruginosa; Acinetobacter
Occurrence and the susceptibility to antimicrobial agents in Pseudomonas aeruginosa and Acinetobacter sp. at a tertiary hospital in southern Brazil
Ocorrência e perfil de sensibilidade a antimicrobianos em Pseudomonas aeruginosa e Acinetobacter sp. em um hospital terciário, no sul do Brasil
Gabriele Mariani MachadoI; Aldalise LagoII; Sérgio Roberto Riccardi FuentefriaII,III; Daiane Bopp FuentefriaII,III
IInstituto de Ciências Biológicas, Universidade de Passo Fundo, Passo Fundo, RS
IILaboratório de Análises Clínicas, Hospital São Vicente de Paulo, Passo Fundo, RS
IIIServiço de Análises Especializadas, Laboratório SANI, Passo Fundo, RS
Address to Address to: Drª Daiane Bopp Fuentefria Serv. Análises Especializadas/Laboratório SANI Av. Brasil Oeste 888, Centro 99010-001 Passo Fundo, RS, Brasil Phone/fax: 55 54 3312-2000 e-mail: dfuente@tpo.com.br
ABSTRACT
INTRODUCTION: The main mechanism of emerging resistance in Pseudomonas aeruginosa and Acinetobacter sp. isolates is the production of metallo-β-lactamases (MβLs). MβLs are enzymes capable of hydrolyzing cephalosporins, penicillins and carbapenems, but not monobactams (aztreonam), which are often used as antimicrobial therapy to treat nosocomial infections.
METHODS: An observational descriptive and retrospective study was designed to assess the frequency of MβLs among strains of P. aeruginosa and Acinetobacter sp. obtained from a tertiary hospital in southern Brazil.
RESULTS: MβL production was observed in 77.6% (n = 173/223) for P. aeruginosa isolates and 22.4% (n = 50/223) of Acinetobacter sp. isolates. The Acinetobacter sp. isolates showed 92.8% sensitivity to amikacin and P. aeruginosa isolates showed 58.9% sensitivity to aztreonam.
CONCLUSIONS: The MβL indices determined confirm the global concern with this mechanism of resistance.
Keywords: MβL. Bacterial resistance. Pseudomonas aeruginosa. Acinetobacter.
RESUMO
INTRODUÇÃO: O principal mecanismo de resistência entre isolados de Pseudomonas aeruginosa e Acinetobacter sp. é a produção de metalo-β-lactamases (MβLs). As MβLs são enzimas capazes de hidrolisar cefalosporinas, penicilinas e carbapenêmicos, mas não monobactâmicos (aztreonam) antibióticos que se encontram entre as principais opções terapêuticas para o tratamento de infecções causadas por bactérias não fermentadoras de glicose.
MÉTODOS: Um estudo observacional, transversal, descritivo e retrospectivo foi desenvolvido para avaliar a frequência e o perfil de susceptibilidade cepas de P. aeruginosa e Acinetobacter sp. produtoras de MβLs isoladas no Hospital São Vicente de Paulo, Passo Fundo, Brasil.
RESULTADOS: A produção de MβLs foi observada em 77,6% (n = 173/223) dos isolados de P. aeruginosa e em 22,4% (n = 50/223) dos isolados de Acinetobacter sp. Dentre as cepas produtoras de MβL, a maioria apresentou mais de 90% de resistência a seis antimicrobianos dos 12 testados, enfatizando a resistência a ceftazidima, gentamicina, aztreonam, piperaciclina/tazobactam, cefepime, ciprofloxacina, meropenem e tobramicina.
CONCLUSÕES: Os índices de MβL encontrados confirmam a preocupação mundial com a disseminação desse mecanismo de resistência.
Palavras-chaves: MβL. Resistência bacteriana. Pseudomonas aeruginosa. Acinetobacter.
INTRODUCTION
Hospital infections are a major concern for public health agencies and the internal committees of hospital infection control. Coco-bacilli and Gram-negative bacteria, such as Pseudomonas aeruginosa and Acinetobacter sp. respectively, are extremely important pathogens that cause serious infections in different parts of the human body and may lead to patient death1-3.
In the last decade, particularly in P. aeruginosa, enzymes were identified that promote hydrolysis of cephalosporins, penicillins and carbapenems but not monobactams, such as aztreonam, which are called arbapenamases or metallo-β-lactamases (MβLs) and confer resistance to these antimicrobial4,5.
The first reports of P. aeruginosa isolates producing MβLs in Japan occurred in 1991, since then Europe, Asia and America have also recorded such events6. In Brazil, the gene is prevalent bla SPM-1, originally isolated in São Paulo7. Currently, the increase in the number of isolates of P. aeruginosa with bla genes in intensive care units (ICUs) is a cause of serious concern8. Risk factors for nosocomial infections in this environment include exposure to highly invasive practices, such as mechanical ventilation, urinary catheters, intra-arterial and intravenous catheters and the use of antibiotics in large quantities9.
All metallo-β-lactamases (MβLs) hydrolyze imipenem, but this ability varies considerably and the rate of hydrolysis may or may not be correlated with the level of bacterial resistance to carbapenems. These enzymes are inhibited by EDTA (ethylenediaminetetraacetic acid) and other chelators of divalent cations and is thus correlated with its mechanism of action10,11.
This study was conducted in order to extend current knowledge regarding the incidence and susceptibility profiles of MβL-producing P. aeruginosa and Acinetobacter in a tertiary hospital in Passo Fundo, RS, Brazil.
METHODS
Place of the study
This study was conducted at the Laboratory of Clinical Analysis of São Vicente de Paulo Hospital (Hospital São Vicente de Paulo, HSVP) in Passo Fundo, State of Rio Grande do Sul, Brazil. The hospital has approximately 590 beds and 60 beds for support in a constructed area of 50,000m2, with about 28,000 admissions per year.
Study design and data collection
A retrospective cross-sectional observational study was conducted that included patients at the HSVP and bacterial cultures tests performed from June to December 2008. The data collected were complied on a spreadsheet where the following information was recorded: name, age, sex, hospital ward, number of laboratory records for the patient, type of biological material, underlying disease, culture results, susceptibility to antimicrobials and use of antibiotics before and after treatment. The data of patients with positive cultures for MβL-producing or nonproducing P. aeruginosa and Acinetobacter sp. were compared. For ethical reasons, the name of the patient was noted to only to prevent replication and was not disclosed.
Data collection was performed by the internal system of HSVP computers in the Laboratory of Clinical Analysis using Mylab for Windows; this system is used to disclose the results of patients to medical assistants and in the Program Requirements and Medical Patients, where the outcome of hospitalized patients is registered.
Definitions
The bacterial isolates were identified by classic morphological and biochemical tests, such as Gram stain, cytochrome oxidase, motility, TSI and pyoverdin production. The profile of antimicrobial susceptibility was determined by the disk-fusion method, in accordance with the standards of the Clinical Laboratory Standards Institute (CLSI), using the following agents: 30µg of ceftazidime (CAZ), 10mg of gentamicin (GEN), 110µg of piperacillin/tazobactam (PIT), 30µg of amikacin (AMI), 30µg of aztreonam (ATM), 30µg of cefepime (CPM), 5µg of ciprofloxacin (CIP), 10mg of meropenem (MER), 10mg of tobramycin (TOB), 20µg of ampicillin/sulbactam (SBA), 30µg of ceftriaxone (CRO) and 15µg of tigecycline (TGC). The phenotypic screening for the MβL production was performed by combined disk using imipenem (10ug) as the substrate and EDTA (750mcg) as an inhibitor12.
Statistical analysis
The percentage of strains producing MβL was calculated among cultures positive for pathogens. The data of the positive cultures and MβL-producing strains were compared with the type of biological sample, the profile of antimicrobial susceptibility and demographics, such as sex, hospital and age. Comparative analysis between the presence of MβLs and the different traits was performed by tthe Fisher exact test and Chi square test (χ2 test).
Qualitative categorical variables, such as the presence or absence of MβLs, type of biological sample, the hospital and patient sex were compared using univariate (Mantel-Haenszel common odds ratio estimate) and calculation of odds ratios (ORs) with 95% confidence interval (95%CI), using SPSS (version 11.0), where p < 0.05 was considered statistically significant.
RESULTS
A total of 7,849 cultures were analyzed during the study period. The percentage of positive cultures for pathogens was 2.8% (n = 223/7849) for P. aeruginosa and Acinetobacter sp. Among these, 77.6% (n = 173/223) were identified as P. aeruginosa and 22.4% (n = 50/223) were Acinetobacter sp. The screening test for MβLs was positive in 17.4% of isolates of P. aeruginosa and 6.3% of isolates of Acinetobacter sp.
The MβL-producing isolates were recovered mainly from samples of tracheal aspirate (n = 19/223, 8.5%), urine (n = 10/2,234, 48%), tracheal secretion (n = 7/223, 3.1%) and sputum (n = 6/223, 2.6%); however, the type of sample was not statistically significant for either of the pathogens studied.
Other types of biological samples, such as cultures of catheters, drains, bronchial washings, secretions and cerebrospinal fluid samples, occurred less frequently. Among the 53 MβL-producing isolates, 11 patients were female and 42 were male, although differences between sexes was only significant for isolates of P. aeruginosa (p = 0.034), with an odds ratio of 2.361 (95%CI, 0.987 to 5,649) for males.
A variation in rates of positive cultures for MβL-producing P. aeruginosa and Acinetobacter sp. was observed for different hospital sectors, with the highest rate of production of MβLs occurring in the central intensive care unit (ICU-C) with 21 isolates (n = 21/53, 39.6%), followed by the centre for critical care nursing (CCCN) with 17 infected patients (n = 17/53, 31.1%) and station 7 (S07) with seven cases (n = 7/53, 13.2%). A significant difference in the presence of MβL-producing Acinetobacter sp. (p = 0.13) was only observed for the ICU. For this sector, the odds ratio was 4.051 (95%CI, 1.343 to 13.213). The age group with the highest rate of MβL production was over 60 years-old (n = 28/53, 52.8%), followed by the range from 25 to 60 years-old (n = 15/53, 28.3%), from 12 to 25 years-old (n = 9/53, 16.9%) and up to 5 years-old (n = 1/53, 1.8%). Not one MβL producer was recovered from patients aged 5 to 12 years-old (Table 1), although age showed no significant difference (p = 0.564). The underlying diseases that manifested most often in relation to the presence of MβLs were the respiratory system, including pneumonia and bronchopneumonia, respiratory failure, chronic obstructive pulmonary disease and pulmonary embolism, and liver diseases, such as cirrhosis, renal and neurological complications.
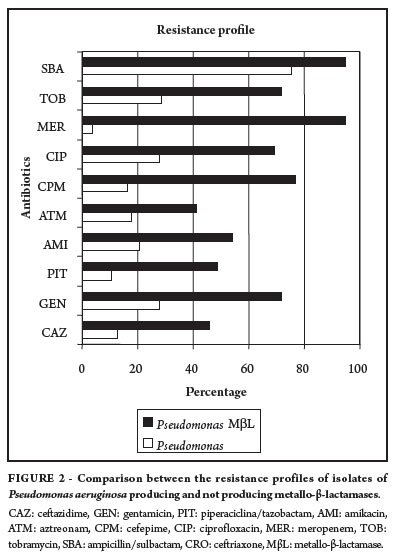
Comparing the isolates of MβL-producing Acinetobacter sp. with the nonproducing strains, the former present much greater resistance to the antimicrobial agents evaluated. Among the MβL-producing strains, most isolates showed over 90% of antimicrobial resistance to six of the 12 drugs tested, particularly to ceftazidime, gentamicin, aztreonam, piperacillin/tazobactam, cefepime, ciprofloxacin, meropenem and tobramycin (Figure 1). Only 7.1% of the MβL-producing isolates presented resistance to amikacin and tigecycline, while 28.5% of these isolates were resistant to ampicillin/sulbactam, providing the only therapeutic options for these cases. Meropenem, which has a high resistance rate and is used as standard treatment in cases of Acinetobacter sp. cannot be used in cases of MβL production, because resistance is superior to 90%, but in cases of MβL-nonproducing strains, it is the antibiotic of choice.
In MβL-nonproducing strains of Acinetobacter sp. the levels of antibiotic resistance were lower, with 34% of isolates resistant to ceftazidime, 42% to gentamicin, 26% to piperacillin/tazobactam, 24% to amikacin, 38% to aztreonam, 34% to cefepime, 42% to ciprofloxacin, 48% to tobramycin, 16% to ampicillin/sulbactam, 26% to ceftriaxone and only 2% to tigecycline and meropenem (Figure 1).
The MβL-producing strains of P. aeruginosa show greater resistance to the antibiotics tested than the nonproducing strains. However, MβL-producing strains present strength of more than 80% to only two antibiotics, ampicillin/sulbactam and meropenem, and resistance above 60% for six of the 10 antibiotics tested (Figure 2).
Among the MβL-nonproducing isolates of P. aeruginosa, resistance levels were much lower. The highest rates of resistance were observed against ampicillin/sulbactam (75.3%), followed by tobramycin (28.3%), gentamicin and ciprofloxacin (both 27.6%), amikacin (20.8%), aztreonam (17.9%), cefepime (16.4%), ceftazidime (12.6%), piperacillin/tazobactam (10.4%) and meropenem (3.7%).
DISCUSSION
The frequency of MβLs among isolates of P. aruginosa in this study was 17.4%. In a study conducted at the same institution, Graf et al9 detemined a rate of 10.8% of producers with phenotypes of resistance genes among isolates of P. aeruginosa in a period of three months. Sader et al5 verified a frequency of 19.7% for MβL-producing isolates at a hospital in São Paulo. Rates ranging from 70% of isolates resistant to carbapenems were reported by Gaspareto et al13 in Porto Alegre, in a two-year study. The high frequency of MβLs verified emphasizes the importance of phenotypically investigating the presence of MβLs in routine laboratory tests, since the enzymes are located in mobile genetic structures that can spread between bacterial strains and lead to epidemic outbreaks.
In this work, MβL-producing Acinetobacter sp. represented 6.3% of the samples identified. Similar rates are reported by the Mystic Program Brazil 2003, where 8.8% of bacterial isolates causing infections in ICUs were multidrug-resistant Acinetobacter sp.14.
Bacterial resistance to the antibiotics tested was consistent with several previous studies involving both pathogens. In MβL-producing P. aeruginosa, resistance exceeded 60% in six of the 11 antibiotics tested. The lowest resistance was observed against aztreonam, which can be used as a treatment option. For Acinetobacter sp. the lowest rates of resistance were observed against amikacin, tigecycline and sulbactam/ampicillin, making these the therapeutic options. The sensitivity profile of MβL-producing isolates of Acinetobacter sp. indicates the possible presence of other associated β-lactamase type oxacillinase (blaOXA23), similar to the epidemic clone disseminated in Brazil15.
A significant increase in resistance to antibiotics was observed for these pathogens compared to MβL-nonproducing isolates. For Acinetobacter sp. even aztreonam shows diminished treatment effectiveness in pathogens producing this enzyme, probably due to the presence of other resistance genes, including those capable of hydrolyzing monobactams, such as extended spectrum β-lactamases (ESBLs). Reis et al16 reported that in some Brazilian hospitals, rates of carbapenem resistance increased approximately 12% for isolates of Acinetobacter sp. The profile of the Acinetobacter sp. strains could be the type of enzymes associated with blaOXA-23. Dalla-Costa et al15 observed this strain in two hospitals in Curitiba, together with P. aeruginosa, and observed their similarity to MβL-producers due to carbapenem resistance and high sensitivity to aztreonam.
The high rate of resistance to most antibiotics tested indicates that the selection of resistant strains may be occurring and that the use of potent antibiotics may be contributing to this selection. Reis et al17 identified a strain of Acinetobacter sp. resistant even to polymyxin B, a worrying fact, since this is an extremely powerful drug and is often used as a last choice for the treatment of pan-resistant strains of Acinetobacter sp. According to Reis et al17, use of polymyxin B may have played a role in the selection of resistant strains, arguing against the theory of clonal spread. Furthermore, two isolates showed an identical genetic pattern, which raises the possibility of transmission of strains occurring between patients and the ability to create epidemics.
High rates of resistance observed in inpatient units, such as ICU-C, CCCN and Post 7, is probably related to the severity of cases among these patients, since these units are for hospitalized patients with more severe illnesses and greater susceptibility. MβL production occurred mainly in samples from the respiratory tract, stressing that both pathogens are important colonizers of artificial respiration and in patients with nosocomial pneumonia; another important risk factor is diabetes mellitus, which causes insulin resistance. Lautenbach et al18 reported that 44.6% of P. aeruginosa isolates were from respiratory tract samples. In nosocomial samples from Porto Alegre, Pulcinelli et al16 verified a rate of 70.7% in lower respiratory tract isolates of P. aeruginosa. In 2008, Carneiro et al19 reported that in a hospital in Londrina, Parana, the incidence of patients with ventilator-associated pneumonia was 25.2% for Acinetobacter sp.19.
According to the Manual of the Department of Infection Control, from 2009, of the HVSP, in-hospital precautions must be taken to prevent the spread of multidrug-resistant strains, which included contact precautions for patients colonized and/or infected, the use of aprons with long sleeves and appropriate use of procedure gloves and hand hygiene with antiseptic and water or alcohol gel before and after attending such patients, individual use of thermometers, stethoscopes and sphygmomanometers, individual, or preferably fourth cohort control within the hospital20.
MβL rates (17.4% of P. aeruginosa isolates and 6.3% of Acinetobacter sp. isolates) verified among the HVSP isolates were similar to those of other Brazilian institutions. Detection of MβL production in routine laboratory exams provides the foundation and assists in the selection of the appropriate antimicrobial therapy. With the spread of MβL-producing strains in hospitals around the world, knowledge regarding the prevalence of MβL production is essential to develop policies for empirical therapy in high risk units, where rates of infection by these microorganisms are high. Surveillance, combined with monitoring and daily review of cultures in the microbiology laboratory and the use of antimicrobials, are effective measures to identify outbreaks in time to carry out measures to optimize and intervene in the conduct adopted in the various hospital sectors21. Thus, the rates of MβLs verified confirm the concern of the global emergency of this resistance mechanism.
ACKNOWLEDGMENTS
The authors would like to thank the Clinical Laboratory and the Committee on Infection Control at the São Vicente de Paulo Hospital for allowing them to develop this study. The authors are also grateful to Dr. Gilberto Barbosa da Luz for his review of the manuscript.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
Received in 12/09/2010
Accepted in 08/11/2010
- 1. Gales AC, Menezes LC, Silbert S, Sader HS. Dissemination in distinct Brazilian regions of an epidemic carbapenem-resistant Pseudomonas aeruginosa producing SPM metallo-β-lactamase. J Antimicrob Chemother 2003; 52:699-702.
- 2. Gales AC, Tognim MC, Reis AO, Jones RN, Sader HS. Emergence of an IMP-like metallo-enzyme in an Acinetobacter baumannii clinical strain from a Brazilian teaching hospital. Diagn Microbiol Infect Dis 2003; 45:77-79.
- 3. Mendes RE, Castanheira M, Pignatari ACC, Gales AC. Metalo-β-lactamases. J Bras Patol Med Laboratorial2006; 42:103-113.
- 4. Castanheira M, Toleman MA, Jones, RN, Schimidt FJ, Walsh TR.Molecular characterization of a beta-lactamase gene, blaGIM-1, encoding a new subclass of metallo-β-lactamase. Antimicrob Agents Chemother2004; 48:4654-4661.
- 5. Sader HS, Reis AO, Silbert S, Gales AC. IMPs, VIMs and SPMs: the diversity of metallo-β-lactamases produced by carbapenem-resistant Pseudomonas aeruginosa in a Brazilian hospital. Clin Microbiol Infect 2004; 11:73-76.
- 6. Lauretti L, Riccio ML, Mazzariol A, Cornaglia G, Amicosante G, Fontana R, et al. Cloning and characterization of blaVIM, a new integron-borne Metallo-β-Lactamse gene from a Pseudomonas aeruginosa clinical isolate. Antimicrob Agents Chemother 1999; 43:1584-1590.
- 7. Lee K, Lim YS, Young D, Yum JH, Chong Y. Evaluation of the Hodge Test and the Imipenem-EDTA Double-Disk Synergy Test for Differentiating Metallo-β-Lactamase-Producing Isolates of Pseudomonas spp. and Acinetobacter spp. J Clin Microbiol 2003; 43:4623-4629.
- 8. Loureiro MM, Moraes BA, Mendonça VLF, Quadra MRR, Pinheiro GS, Asensi MD. Pseudomonas aeruginosa: Study of antibiotic resistance and molecular typing in hospital infection cases in a neonatal intensive care unit from Rio de Janeiro city, Brazil. Mem Inst Oswaldo Cruz 2002; 97:387-394.
- 9. Gräf T, Fuentefria DB, Corção G. Ocorrência de cepas de Pseudomonas aeruginosa multirresistentes produtoras de Metalo-β-Lactamase blaSPM-1 em amostras clínicas. Rev Soc Bras Med Trop 2008; 41:306-308.
- 10. Young D, Lee K, Yum JH, Shin HB, Rossolini GM, Chong Y. Imipenem-EDTA disk method for differentiation of Metallo-β-Lactamase-producing clinical isolates of Pseudomonas spp. and Acinetobacter spp. Clin Infect Dis 2002; 10: 3798-3801.
- 11. Walsh TR, Toleman MA, Poirel, L, Nordmann P. Metallo-β-Lactamases: the Quiet before the Storm? Clin Microbiol Rev 2005; 18:306-325.
- 12. Toleman MA, Simm AM, Murphy TA, Gales AC, Biedenbach DJ, Jones RN, et al. Molecular characterization of SPM-1, a novel metallo-beta-lactamase isolated in Latin America: report from the SENTRY antimicrobial surveillance programme. J Antimicrob Chemother 2002; 37:673-679.
- 13. Gaspareto PB, Martins AF, Zavascki AP, Barth AL. Occurrence of blaSPM-1 and blaIMP-1 genes of metalo-β-lactamases in clinical isolates of Pseudomonas aeruginosa from three universitary hospitals in the city of Porto Alegre, Brazil. Braz J Microbiol 2007; 38:108-109.
- 14. Kiffer C, Hsiung A, Oplustiul C, Sampaio J, Sakayami E, Turner P, et al. Mystic Brazil Group. Antimicrobial susceptibility of gram-negative bacteria in Brazilian hospitals: the MYSTIC program Brazil 2003. Braz J Infect Dis 2005; 9: 216-224.
- 15. Dalla-Costa LM, Coelho JM, Souza HAPHM, Castro MES, Stier CJN, Bragagnolo KL, et al. Outbreak of Carbapenem-Resistant Acinetobacter baumannii Producing the OXA-23 Enzyme in Curitiba, Brazil. J Clin Microbiol 2003; 41:3403-3406.
- 16. Pulcinelli RSR, Saucedo EM, Pereira R, Pilonetto M, Aquino ARC, Santos RCV. Detecção de metalo-β-lacmatases em amostras de Pseudomonas aeruginosa isoladas em Porto Alegre, Rio Grande do Sul. Rev Bras Anal Clin2009; 41: 197-199.
- 17. Reis AO, Luz DAM, Tognin MCB, Sader HS, Gales AC. Polymyxin-Resistant Acinetobacter spp. Isolates: What Is Next? Emerg Infect Dis2003; 9: 1025-1027.
- 18. Lautenbach E, Weiner MG, Nachamkin I, Bilker WB, Sheridan A, Fishman NO. Imipenem Resistance Among Pseudomonas aeruginosa Isolates: Risk Factors for Infection and Impact of Resistance on Clinical and Economic Outcomes. Infect Control Hosp Epidemiol 2006; 27:893-900.
- 19. Carneiro M, Saridakis HO. Pneumonia associada à ventilação mecânica por Acinetobacter baumannii resistente a carbapenem. Rev Panam Infectol2008; 10:28-33.
- 20. Barbosa GL, Schlichting D, Oliveira DR, De Paula NN. Manual do Serviço de Controle de Infecção Hospitalar. Hospital São Vicente de Paulo;2009.
- 21. Podnos YD, Cinat ME, Wilson SE, Cooke J, Gornick W, Thrupp LD. Eradication of multi-drug resistant Acinetobacter from an intensive care unit. Surg Infect 2001; 2:297-301.
Address to:
Datas de Publicação
-
Publicação nesta coleção
01 Abr 2011 -
Data do Fascículo
Abr 2011
Histórico
-
Recebido
12 Set 2010 -
Aceito
08 Nov 2010