Abstract
Pseudomonas aeruginosa is the leading cause of nosocomial infections with high mortality rates owing to the limited therapeutic options for multidrug-resistant Pseudomonas aeruginosa (MDRPA) and metallo-beta-lactamase (MBL)-producing strains. Herein, we present a meta-analysis exploring the association between MDRPA and São Paulo MBL-1 (SPM-1)-producing strains vs. mortality. Online databases were screened to identify studies published between 2006 and 2016. A total of 15 studies, comprising 3,201 cases of P. aeruginosa infection, were included. Our results demonstrated a higher mortality rate among patients infected with MDRPA (44.6%, 363/813) than those with non-MDRPA infection (24.8%, 593/2,388) [odds ratio (OR) 2.39, 95% confidence interval (CI) 1.70-3.36, p <0.00001]. The risk of mortality in patients with non-SPM-1 strains was four times higher than that observed in the patients of the SPM-1 group; however, no statistically significant difference was observed (p = 0.43). In conclusion, the results of our study demonstrated that patients infected with MDRPA had a significantly higher mortality rate than that of patients infected with non-MDRPA strains, especially patients with bloodstream infection (BSI), immunosuppression, and inadequate antimicrobial therapy. The absence of studies on the molecular aspects of blaSPM-1 and its association with mortality limited the analysis; therefore, our results should be interpreted with caution. Our findings also highlight the need for more studies on the molecular aspects of resistance and the peculiarities of different nosocomial settings.
Keywords:
Pseudomonas aeruginosa; Mortality; Intensive care unit; Beta-lactamases; Nosocomial infection; Meta-analysis
INTRODUCTION
Pseudomonas aeruginosa is a versatile pathogen responsible for nosocomial infections with high mortality rates in critically ill patients, including those from general wards and outpatient clinics11. Hirsch EB, Cottreau JM, Chang KT, Caeiro JP, Johnson ML, Tam VH. A model to predict mortality following Pseudomonas aeruginosa bacteremia. Diagn Microbiol Infect Dis. 2012;72(1):97-102.
2. Gomes MZ, Machado CR, da Conceição MS, Ortega JA, Neves SM, Lourenço MC, et al. Outbreaks, persistence, and high mortality rates of multiresistant Pseudomonas aeruginosa infections in a hospital with AIDS-predominant admissions. Braz J Infect Dis. 2011;15(4):312-22.
3. Tumbarello M, Repetto E, Trecarichi EM, Bernardini C, De Pascale G, Parisini A, et al. Multidrug-resistant Pseudomonas aeruginosa bloodstream infections: risk factors and mortality. Epidemiol Infect. 2011;139(11):1740-9.-44. Suárez C, Peña C, Gavaldà L, Tubau F, Manzur A, Dominguez MA, et al. Influence of carbapenem resistance on mortality and the dynamics of mortality in Pseudomonas aeruginosa bloodstream infection. Int J Infect Dis. 2010;14(Suppl 3):e73-8.. In recent decades, publications worldwide have highlighted the increasing issue of carbapenem and antipseudomonal broad-spectrum cephalosporin resistance11. Hirsch EB, Cottreau JM, Chang KT, Caeiro JP, Johnson ML, Tam VH. A model to predict mortality following Pseudomonas aeruginosa bacteremia. Diagn Microbiol Infect Dis. 2012;72(1):97-102.,55. Hong DJ, Bae IK, Jang IH, Jeong SH, Kang HK, Lee K. Epidemiology and Characteristics of Metallo-β-Lactamase-Producing Pseudomonas aeruginosa. Infect Chemother. 2015;47(2):81-97.,66. Pollini S, Maradei S, Pecile P, Olivo G, Luzzaro F, Docquier JD, et al. FIM-1, a new acquired metallo-β-lactamase from a Pseudomonas aeruginosa clinical isolate from Italy. Antimicrob Agents Chemother. 2013;57(1):410-6.. The effect of bacterial resistance is directly related to infection severity and underlying disease, as well as the selected antibiotic therapy. Furthermore, the complexity of resistance mechanisms has contributed to a gradual increase in resistance rates, which are of particular concern in nosocomial settings33. Tumbarello M, Repetto E, Trecarichi EM, Bernardini C, De Pascale G, Parisini A, et al. Multidrug-resistant Pseudomonas aeruginosa bloodstream infections: risk factors and mortality. Epidemiol Infect. 2011;139(11):1740-9.,77. Peña C, Suarez C, Gozalo M, Murillas J, Almirante B, Pomar V, et al. Prospective multicenter study of the impact of carbapenem resistance on mortality in Pseudomonas aeruginosa bloodstream infections. Antimicrob Agents Chemother . 2012;56(3):1265-72.,88. Zavascki AP, Barth AL, Gonçalves AL, Moro AL, Fernandes JF, Martins AF, et al. The influence of metallo-beta-lactamase production on mortality in nosocomial Pseudomonas aeruginosa infections. J Antimicrob Chemother. 2006;58(2):387-92..
Among the multiple resistance mechanisms in P. aeruginosa, including activation and overexpression of efflux systems and alteration of outer membrane permeability, the production of metallo-beta-lactamases (MBL), which can hydrolyze almost all beta-lactam agents, is one of the most important ones as it severely limits therapeutic options. Moreover, it also has been associated with prolonged hospitalization, polymicrobial infections, previous antibiotic use, and inadequate therapy88. Zavascki AP, Barth AL, Gonçalves AL, Moro AL, Fernandes JF, Martins AF, et al. The influence of metallo-beta-lactamase production on mortality in nosocomial Pseudomonas aeruginosa infections. J Antimicrob Chemother. 2006;58(2):387-92.
9. Furtado GH, Bergamasco MD, Menezes FG, Marques D, Silva A, Perdiz LB, et al. Imipenem-resistant Pseudomonas aeruginosa infection at a medical-surgical intensive care unit: risk factors and mortality. J Crit Care. 2009;24(4):625.e9-14.-1010. Matos EC, Matos HJ, Conceição ML, Rodrigues YC, Carneiro IC, Lima KV. Clinical and microbiological features of infections caused by Pseudomonas aeruginosa in patients hospitalized in intensive care units. Rev Soc Bras Med Trop. 2016;49(3):305-11..
So far, 10 major clinically important MBL groups have been identified, including Imipenemase-type metallo-ß-lactamase (IMP), Verona integron-encoded metallo-ß-lactamases, (VIM), São Paulo metallo-ß-lactamase (SPM), Germany imipenemase (GIM), Seoul imipenemase (SIM), New Delhi metallo-ß-lactamase (NDM), Kyorin Health Science metallo-ß-lactamase (KHM), Tripoli metallo-ß-lactamase (TMB), Dutch imipenemase (DIM) and Florence imipenemase (FIM). These MBL variants quickly spread across Europe, then to North America, Latin America, Asia, and Oceania, reflecting a problem of global dimensions55. Hong DJ, Bae IK, Jang IH, Jeong SH, Kang HK, Lee K. Epidemiology and Characteristics of Metallo-β-Lactamase-Producing Pseudomonas aeruginosa. Infect Chemother. 2015;47(2):81-97.,66. Pollini S, Maradei S, Pecile P, Olivo G, Luzzaro F, Docquier JD, et al. FIM-1, a new acquired metallo-β-lactamase from a Pseudomonas aeruginosa clinical isolate from Italy. Antimicrob Agents Chemother. 2013;57(1):410-6.,1111. Khan AU, Maryam L, Zarrilli R. Structure, Genetics and Worldwide Spread of New Delhi Metallo-β-lactamase (NDM): a threat to public health. BMC Microbiol. 2017;17(1):101.,1212. Toleman MA, Bennett PM, Walsh TR. ISCR elements: novel gene-capturing systems of the 21st century? Microbiol Mol Biol Rev. 2006;70(2):296-316.. A relevant aspect regarding this fast dissemination of resistance traits among P. aeruginosa strains is related to the fact that the resistance genotype may be transferred through mobile genetic elements. The emergence of multidrug-resistant (MDR) strains demonstrates the need for changes in routine laboratory diagnostic procedures, such as the incorporation of the molecular detection of bla-variant genes, particularly blaSPM-1, which is the most prevalent variant in Brazil1313. Toleman MA, Simm AM, Murphy TA, Gales AC, Biedenbach DJ, Jones RN, et al. Molecular characterization of SPM-1, a novel metallo-beta-lactamase isolated in Latin America: report from the SENTRY antimicrobial surveillance programme. J Antimicrob Chemother . 2002;50(5):673-9.
14. Maciel WG, Silva KE, Bampi JV, Bet GM, Ramos AC, Gales AC, et al. Identification of São Paulo metallo-beta-lactamase-1-producing Pseudomonas aeruginosa in the Central-West region of Brazil: a case study. Rev Soc Bras Med Trop . 2017;50(1):135-7.-1515. Gales AC, Menezes LC, Silbert S, Sader HS. Dissemination in distinct Brazilian regions of an epidemic carbapenem-resistant Pseudomonas aeruginosa producing SPM metallo-beta-lactamase. J Antimicrob Chemother . 2003;52(4):699-702..
Several studies have reported conflicting results regarding mortality associated with multidrug-resistant Pseudomonas aeruginosa (MDRPA) infections, and few reports have highlighted this association with MBL-producing strains77. Peña C, Suarez C, Gozalo M, Murillas J, Almirante B, Pomar V, et al. Prospective multicenter study of the impact of carbapenem resistance on mortality in Pseudomonas aeruginosa bloodstream infections. Antimicrob Agents Chemother . 2012;56(3):1265-72.,88. Zavascki AP, Barth AL, Gonçalves AL, Moro AL, Fernandes JF, Martins AF, et al. The influence of metallo-beta-lactamase production on mortality in nosocomial Pseudomonas aeruginosa infections. J Antimicrob Chemother. 2006;58(2):387-92.,1010. Matos EC, Matos HJ, Conceição ML, Rodrigues YC, Carneiro IC, Lima KV. Clinical and microbiological features of infections caused by Pseudomonas aeruginosa in patients hospitalized in intensive care units. Rev Soc Bras Med Trop. 2016;49(3):305-11.,1616. Babu KV, Visweswaraiah DS, Kumar A. The influence of Imipenem resistant metallo-beta-lactamase positive and negative Pseudomonas aeruginosa nosocomial infections on mortality and morbidity. J Nat Sci Biol Med. 2014;5(2):345-51.,1717. Dantas RC, Ferreira ML, Gontijo-Filho PP, Ribas RM. Pseudomonas aeruginosa bacteraemia: independent risk factors for mortality and impact of resistance on outcome. J Med Microbiol. 2014;63(Pt 12):1679-87.. Therefore, the present meta-analysis evaluated the association of MDR, the presence of the blaSPM-1 gene, and the risk of mortality in patients with P. aeruginosa infection.
METHODS
Search strategy and inclusion criteria
The studies were analyzed based on the essential standards listed in the 2011 Cochrane Handbook for Systematic Review of Intervention, edited by Higgin and Green1818. Higgins JPT, Green S. Cochrane Handbook for Systematic Review of Intervention section, Version 5.1.0. The Cochrane Collaboration; 2011. Updated March 2011; cited 2017 Dec 27. Available from: Available from: http://handbook-5-1.cochrane.org/.
http://handbook-5-1.cochrane.org/. ...
. A search of the PubMed, MEDLINE, BIREME, and Embase databases was conducted using the following keywords and combinations thereof: Pseudomonas aeruginosa, P. aeruginosa, multidrug-resistance, MDR, non-multidrug-resistance, non-MDR, blaSPM-1 gene, and mortality. The articles were examined by two investigators and studies published in English, Portuguese, and/or Spanish between 2006 and 2016 were included. Studies including (I) humans, (II) at least one group of comparison to MDRPA, and (III) mortality evaluation and antimicrobial resistance were included in the analysis. Unpublished articles (abstracts) or articles that did not fit these criteria were excluded from analysis.
Data extraction and quality assessment
The data were extracted by two investigators. Information such as author, study period, study design, country, setting, definitions of mortality, mortality by MDRPA and non-MDRPA, and site of infection were extracted from each study, as well as whether the study authors had controlled for confounding clinical and demographic factors.
Definition of MDR
Multidrug-resistant was defined as resistance to at least three different classes of antimicrobials, including carbapenems (imipenem, meropenem), antipseudomonal cephalosporins (ceftazidime and cefepime), fluoroquinolones (ciprofloxacin), aminoglycosides (gentamicin and amikacin), and β-lactams with inhibitors (piperacillin-tazobactam).
Data analysis
Statistical analyses were performed by calculating the odds ratios (ORs) with the Mantel-Hansel test at 95% confidence interval (CI) and 5% significance level (p ≤ 0.05) to compare patients infected by MDRPA and non-MDRPA strains. Forest plots were produced using Review Manager 5.01919. Review Manager (RevMan). Computer program. Version 5.3. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014..
RESULTS
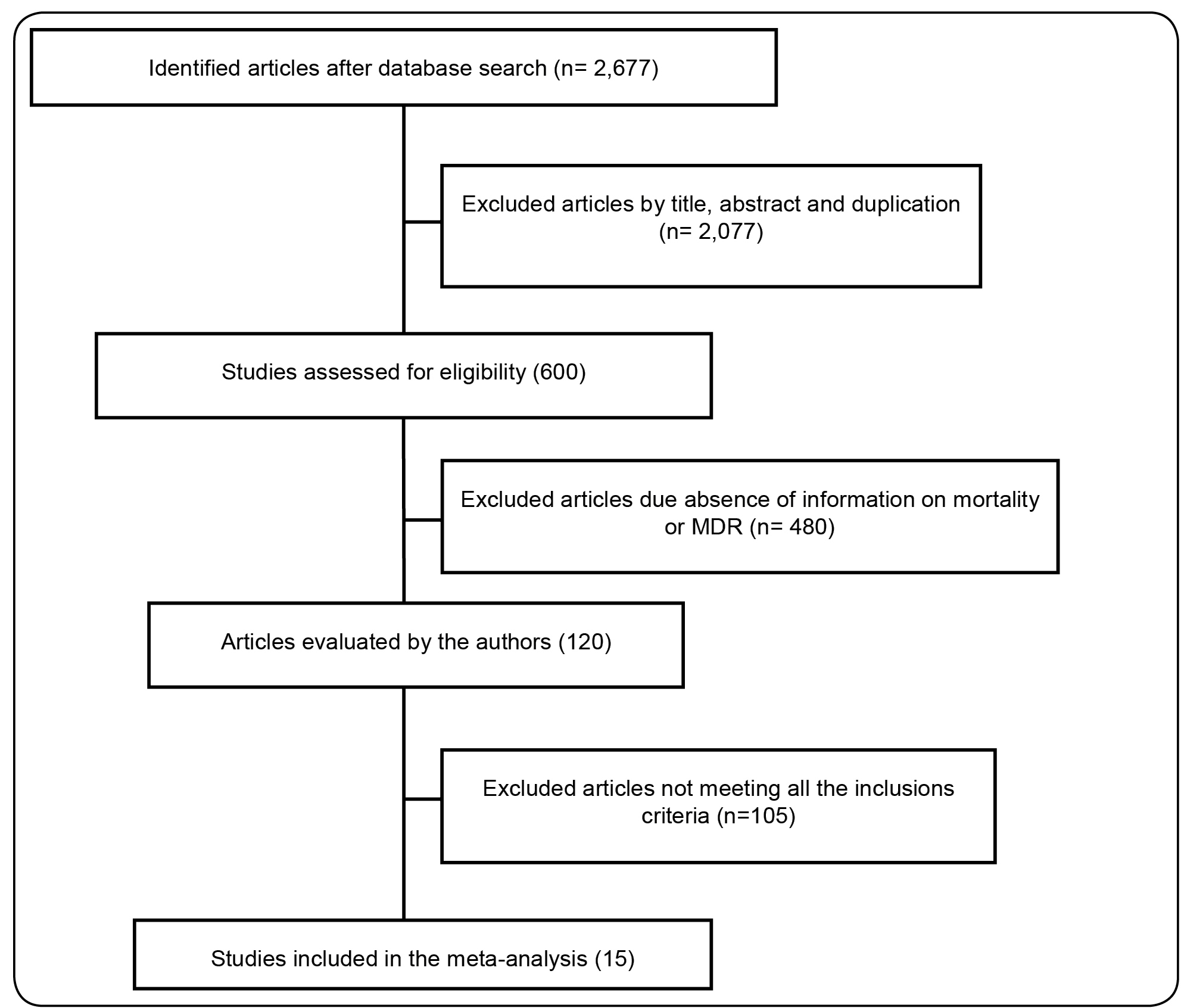
As shown in Figure 1, a total of 2,677 articles were retrieved, 2,077 of which were excluded after title and abstract evaluation, as well as because of duplication. In the second screening, 480 studies were excluded for their lack of information related to mortality and MDR and/or MBL detection; the remaining 120 articles were fully evaluated by the investigators. Finally, 15 studies matching the inclusion criteria were selected for analysis. The main characteristics of the 15 studies included in the meta-analysis are summarized in Table 1. The articles were published between 2006 and 2016, with a sample size comprising 3,201 P. aeruginosa isolates from five countries. Most of the studies were retrospective in design. The main sample source was bloodstream infection (BSI) and 30-day mortality was the outcome of interest.
The meta-analysis illustrated in Figure 2 revealed a higher proportion of deaths among patients with MDRPA (44.65%, 363/813) compared to that in patients with non-MDRPA infections (24.8%, 593/2,388) (OR 2.39, 95% CI 1.70-3.36, p <0.00001). Despite the substantial statistical heterogeneity (I2 = 66%), only the study by Peña2020. Peña C, Gómez-Zorrilla S, Oriol I, Tubau F, Dominguez MA, Pujol M, et al. Impact of multidrug resistance on Pseudomonas aeruginosa ventilator-associated pneumonia outcome: predictors of early and crude mortality. Eur J Clin Microbiol Infect Dis. 2013;32(3):413-20. presented results in favor of the non-MDRPA group; however, their results were not statistically significant (OR 0.82, 95% CI 0.35-1.97), which was possibly because they used the severity of the acute illness as the main predictor for mortality rather than the presence of an MDR isolate.
Forest plot of the association between MDR and the risk of mortality in patients with Pseudomonas aeruginosa infection. MDR: multidrug-resistant; non-MDR: non-multidrug-resistant; M-H: Mantel-Haenszel; random: random effect model; 95% CI: 95% confidence interval.
Forest plot of the association between MBL-producing and non-MBL-producing strains and the risk of mortality in patients with Pseudomonas aeruginosa infection. MDR: multidrug-resistant; non-MDR: non-multidrug-resistant; M-H: Mantel-Haenszel; random: random effect model; 95% CI: 95% confidence interval; MBL: metallo-beta-lactamase; non-MBL: non-metallo-beta-lactamase; SPM-1: São Paulo-metallo-ß-lactamase-1; non-SPM-1: non-São Paulo-metallo-ß-lactamase-1.
As presented in Figure 3, the risk of mortality in the non-SPM-1 group was four times greater than that observed in the SPM-1 group. However, the difference was not statistically significant (p = 0.43), and there was great heterogeneity between the studies (I2 = 82%) caused by Gomes22. Gomes MZ, Machado CR, da Conceição MS, Ortega JA, Neves SM, Lourenço MC, et al. Outbreaks, persistence, and high mortality rates of multiresistant Pseudomonas aeruginosa infections in a hospital with AIDS-predominant admissions. Braz J Infect Dis. 2011;15(4):312-22. as a result of clinical and methodological differences among the investigations.
DISCUSSION
This study selected several original articles for analysis, combining and performing a synthesis of the results based on variables associated with MDRPA and relating them to mortality. The meta-analysis of the data revealed that P. aeruginosa infections resulted in high mortality rates in cases of infection with MDRPA compared to those of non-MDRPA infections. P. aeruginosa infections are opportunistic and are considered to have a high mortality in a global context. This pathogen is closely related to nosocomial infections in critically ill patients, but it also affects immunocompetent individuals33. Tumbarello M, Repetto E, Trecarichi EM, Bernardini C, De Pascale G, Parisini A, et al. Multidrug-resistant Pseudomonas aeruginosa bloodstream infections: risk factors and mortality. Epidemiol Infect. 2011;139(11):1740-9.,2020. Peña C, Gómez-Zorrilla S, Oriol I, Tubau F, Dominguez MA, Pujol M, et al. Impact of multidrug resistance on Pseudomonas aeruginosa ventilator-associated pneumonia outcome: predictors of early and crude mortality. Eur J Clin Microbiol Infect Dis. 2013;32(3):413-20.
21. Araujo BF, Ferreira ML, Campos PA, Royer S, Batistão DW, Dantas RC, et al. Clinical and Molecular Epidemiology of Multidrug-Resistant P. aeruginosa Carrying aac(6')-Ib-cr, qnrS1 and blaSPM Genes in Brazil. PLoS One. 2016;11(5):e0155914.-2222. Tam VH, Rogers CA, Chang KT, Weston JS, Caeiro JP, Garey KW. Impact of multidrug-resistant Pseudomonas aeruginosa bacteremia on patient outcomes. Antimicrob Agents Chemother . 2010;54(9):3717-22..
Most of the selected studies focused on the bloodstream as the main site of infection. BSIs increase the costs to the hospital and the complexity of cases; they may prolong hospitalization and are often exacerbated by unfavorable outcomes. A study conducted at the teaching hospital in Pará State, Northern Brazil identified BSI as the most frequent route of infection among inpatients in intensive care units (ICU)1010. Matos EC, Matos HJ, Conceição ML, Rodrigues YC, Carneiro IC, Lima KV. Clinical and microbiological features of infections caused by Pseudomonas aeruginosa in patients hospitalized in intensive care units. Rev Soc Bras Med Trop. 2016;49(3):305-11.. A similar scenario was reported in India, wherein 48.2% of 593 blood cultures was determined to be positive for P. aeruginosa, 63.6% of which were MDR; this also highlights the increasing occurrence of MBL in the region, representing a major therapeutic problem2323. Sharma M, Yadav S, Chaudhary U. Metallo-beta-lactamase Producing Pseudomonas aeruginosa in Neonatal Septicemia. J Lab Physicians. 2010;2(1):146..
Among the selected works, none assessed neonatal inpatients exclusively. Considering the peculiarity of the neonatal ICU population, there were few reports in the literature regarding the behavior of these resistant microorganisms. Life support of these patients requires both direct and indirect special care, aside from presenting critical immunosuppression and possible prolonged hospitalization2424. Nangino GO, Oliveira CD, Correia PC, Machado NM, Dias ATB. Impacto financeiro das infecções nosocomiais em unidades de terapia intensiva em hospital filantrópico de Minas Gerais. Rev Bras Ter Intensiva. 2012;24(4);357-61.. However, a study conducted in 12 hospital centers of hematology and oncology in Italy described a retrospective series of nine years of BSI with P. aeruginosa in children diagnosed with cancer in pediatric oncology. The mortality rate reported in this study was 35.9% in cases of infection with MDRPA. This investigation emphasized that infection with this pathogen is a major concern in patients undergoing chemotherapy despite empirical antibiotic therapy containing at least one active drug, and that multidrug resistance is the greatest risk factor for a fatal outcome2525. Caselli D, Cesaro S, Ziino O, Zanazzo G, Manicone R, Livadiotti S, et al. Multidrug resistant Pseudomonas aeruginosa infection in children undergoing chemotherapy and hematopoietic stem cell transplantation. Haematologica. 2010;95(9):1612-5..
Although heterogeneous, the studied populations presented common aspects, including immunosuppression, underlying disease, use of immunosuppressive agents, antimicrobial therapy, inadequate empirical therapy, use of invasive procedures, and patient age range. Aside from infection with MDRPA, the main clinical features observed in the studies were HIV-positive patients with various complications, hematologic malignancy, cystic fibrosis, use of mechanical ventilation, and neonatal patients. The test of inconsistency showed a partial heterogeneity (I2 = 66%) between studies22. Gomes MZ, Machado CR, da Conceição MS, Ortega JA, Neves SM, Lourenço MC, et al. Outbreaks, persistence, and high mortality rates of multiresistant Pseudomonas aeruginosa infections in a hospital with AIDS-predominant admissions. Braz J Infect Dis. 2011;15(4):312-22.,33. Tumbarello M, Repetto E, Trecarichi EM, Bernardini C, De Pascale G, Parisini A, et al. Multidrug-resistant Pseudomonas aeruginosa bloodstream infections: risk factors and mortality. Epidemiol Infect. 2011;139(11):1740-9.,2626. Peña C, Suarez C, Gozalo M, Murillas J, Almirante B, Pomar V, et al. Prospective multicenter study of the impact of carbapenem resistance on mortality in Pseudomonas aeruginosa bloodstream infections. Antimicrob Agents Chemother . 2012;56(3):1265-72.
27. Joo EJ, Kang CI, Ha YE, Kang SJ, Park SY, Chung DR, et al. Risk factors for mortality in patients with Pseudomonas aeruginosa bacteremia: clinical impact of antimicrobial resistance on outcome. Microb Drug Resist. 2011;17(2):305-12.
28. Trecarichi EM, Tumbarello M, Caira M, Candoni A, Cattaneo C, Pastore D, et al. Multidrug resistant Pseudomonas aeruginosa bloodstream infection in adult patients with hematologic malignancies. Haematologica. 2011;96(1):e1-3.-2929. Morata L, Cobos-Trigueros N, Martínez JA, Soriano A, Almela M, Marco F, et al. Influence of multidrug resistance and appropriate empirical therapy on the 30-day mortality rate of Pseudomonas aeruginosa bacteremia. Antimicrob Agents Chemother . 2012;56(9):4833-7..
Zavascki et al.88. Zavascki AP, Barth AL, Gonçalves AL, Moro AL, Fernandes JF, Martins AF, et al. The influence of metallo-beta-lactamase production on mortality in nosocomial Pseudomonas aeruginosa infections. J Antimicrob Chemother. 2006;58(2):387-92. reported that patients with infections by MBL-producing P. aeruginosa have higher mortality rates than those infected with non-MBL-producing strains, emphasizing the importance of appropriate initial antimicrobial therapy to reduce mortality rates. Exposure to antimicrobial agents predisposes patients to infection by MDRPA strains, and this selective pressure has contributed to the increase and spread of this pathogen, as well as the expression of different resistance genes33. Tumbarello M, Repetto E, Trecarichi EM, Bernardini C, De Pascale G, Parisini A, et al. Multidrug-resistant Pseudomonas aeruginosa bloodstream infections: risk factors and mortality. Epidemiol Infect. 2011;139(11):1740-9.,55. Hong DJ, Bae IK, Jang IH, Jeong SH, Kang HK, Lee K. Epidemiology and Characteristics of Metallo-β-Lactamase-Producing Pseudomonas aeruginosa. Infect Chemother. 2015;47(2):81-97.,88. Zavascki AP, Barth AL, Gonçalves AL, Moro AL, Fernandes JF, Martins AF, et al. The influence of metallo-beta-lactamase production on mortality in nosocomial Pseudomonas aeruginosa infections. J Antimicrob Chemother. 2006;58(2):387-92.,1212. Toleman MA, Bennett PM, Walsh TR. ISCR elements: novel gene-capturing systems of the 21st century? Microbiol Mol Biol Rev. 2006;70(2):296-316.,1313. Toleman MA, Simm AM, Murphy TA, Gales AC, Biedenbach DJ, Jones RN, et al. Molecular characterization of SPM-1, a novel metallo-beta-lactamase isolated in Latin America: report from the SENTRY antimicrobial surveillance programme. J Antimicrob Chemother . 2002;50(5):673-9..
The data in Figure 3 show controversial results regarding the risk of death among patients infected with SPM-1-producing strains, as seen in the three studies that investigated this aspect22. Gomes MZ, Machado CR, da Conceição MS, Ortega JA, Neves SM, Lourenço MC, et al. Outbreaks, persistence, and high mortality rates of multiresistant Pseudomonas aeruginosa infections in a hospital with AIDS-predominant admissions. Braz J Infect Dis. 2011;15(4):312-22.,1010. Matos EC, Matos HJ, Conceição ML, Rodrigues YC, Carneiro IC, Lima KV. Clinical and microbiological features of infections caused by Pseudomonas aeruginosa in patients hospitalized in intensive care units. Rev Soc Bras Med Trop. 2016;49(3):305-11.,2121. Araujo BF, Ferreira ML, Campos PA, Royer S, Batistão DW, Dantas RC, et al. Clinical and Molecular Epidemiology of Multidrug-Resistant P. aeruginosa Carrying aac(6')-Ib-cr, qnrS1 and blaSPM Genes in Brazil. PLoS One. 2016;11(5):e0155914.. Our database search identified many studies that lacked information regarding the detection of MBL (blaSPM-1) and mortality, preventing a more concrete analysis; with the increasing prevalence of SPM-1-producing strains in different regions, particularly in Brazil, this demonstrates the need for investigations with representative sample sizes for analysis55. Hong DJ, Bae IK, Jang IH, Jeong SH, Kang HK, Lee K. Epidemiology and Characteristics of Metallo-β-Lactamase-Producing Pseudomonas aeruginosa. Infect Chemother. 2015;47(2):81-97.,88. Zavascki AP, Barth AL, Gonçalves AL, Moro AL, Fernandes JF, Martins AF, et al. The influence of metallo-beta-lactamase production on mortality in nosocomial Pseudomonas aeruginosa infections. J Antimicrob Chemother. 2006;58(2):387-92.,1010. Matos EC, Matos HJ, Conceição ML, Rodrigues YC, Carneiro IC, Lima KV. Clinical and microbiological features of infections caused by Pseudomonas aeruginosa in patients hospitalized in intensive care units. Rev Soc Bras Med Trop. 2016;49(3):305-11.,1414. Maciel WG, Silva KE, Bampi JV, Bet GM, Ramos AC, Gales AC, et al. Identification of São Paulo metallo-beta-lactamase-1-producing Pseudomonas aeruginosa in the Central-West region of Brazil: a case study. Rev Soc Bras Med Trop . 2017;50(1):135-7.,1515. Gales AC, Menezes LC, Silbert S, Sader HS. Dissemination in distinct Brazilian regions of an epidemic carbapenem-resistant Pseudomonas aeruginosa producing SPM metallo-beta-lactamase. J Antimicrob Chemother . 2003;52(4):699-702..
In conclusion, the results of our study indicate a significantly higher mortality among patients infected with MDRPA compared to those infected with non-MDRPA, especially patients with BSI, immunosuppression, and inadequate antimicrobial therapy. However, the lack of published studies on the molecular aspects of blaSPM-1 detection limited the analysis and its association with mortality; therefore, our results should be interpreted with caution. Heterogeneity of the populations from the studies in this meta-analysis was observed because of the particular characteristics of each setting, especially for the condition of immunosuppression caused by different co-morbidities. Our findings highlight the need for additional studies on the molecular aspects of resistance and the peculiarities of different nosocomial settings.
REFERENCES
-
1Hirsch EB, Cottreau JM, Chang KT, Caeiro JP, Johnson ML, Tam VH. A model to predict mortality following Pseudomonas aeruginosa bacteremia. Diagn Microbiol Infect Dis. 2012;72(1):97-102.
-
2Gomes MZ, Machado CR, da Conceição MS, Ortega JA, Neves SM, Lourenço MC, et al. Outbreaks, persistence, and high mortality rates of multiresistant Pseudomonas aeruginosa infections in a hospital with AIDS-predominant admissions. Braz J Infect Dis. 2011;15(4):312-22.
-
3Tumbarello M, Repetto E, Trecarichi EM, Bernardini C, De Pascale G, Parisini A, et al. Multidrug-resistant Pseudomonas aeruginosa bloodstream infections: risk factors and mortality. Epidemiol Infect. 2011;139(11):1740-9.
-
4Suárez C, Peña C, Gavaldà L, Tubau F, Manzur A, Dominguez MA, et al. Influence of carbapenem resistance on mortality and the dynamics of mortality in Pseudomonas aeruginosa bloodstream infection. Int J Infect Dis. 2010;14(Suppl 3):e73-8.
-
5Hong DJ, Bae IK, Jang IH, Jeong SH, Kang HK, Lee K. Epidemiology and Characteristics of Metallo-β-Lactamase-Producing Pseudomonas aeruginosa Infect Chemother. 2015;47(2):81-97.
-
6Pollini S, Maradei S, Pecile P, Olivo G, Luzzaro F, Docquier JD, et al. FIM-1, a new acquired metallo-β-lactamase from a Pseudomonas aeruginosa clinical isolate from Italy. Antimicrob Agents Chemother. 2013;57(1):410-6.
-
7Peña C, Suarez C, Gozalo M, Murillas J, Almirante B, Pomar V, et al. Prospective multicenter study of the impact of carbapenem resistance on mortality in Pseudomonas aeruginosa bloodstream infections. Antimicrob Agents Chemother . 2012;56(3):1265-72.
-
8Zavascki AP, Barth AL, Gonçalves AL, Moro AL, Fernandes JF, Martins AF, et al. The influence of metallo-beta-lactamase production on mortality in nosocomial Pseudomonas aeruginosa infections. J Antimicrob Chemother. 2006;58(2):387-92.
-
9Furtado GH, Bergamasco MD, Menezes FG, Marques D, Silva A, Perdiz LB, et al. Imipenem-resistant Pseudomonas aeruginosa infection at a medical-surgical intensive care unit: risk factors and mortality. J Crit Care. 2009;24(4):625.e9-14.
-
10Matos EC, Matos HJ, Conceição ML, Rodrigues YC, Carneiro IC, Lima KV. Clinical and microbiological features of infections caused by Pseudomonas aeruginosa in patients hospitalized in intensive care units. Rev Soc Bras Med Trop. 2016;49(3):305-11.
-
11Khan AU, Maryam L, Zarrilli R. Structure, Genetics and Worldwide Spread of New Delhi Metallo-β-lactamase (NDM): a threat to public health. BMC Microbiol. 2017;17(1):101.
-
12Toleman MA, Bennett PM, Walsh TR. ISCR elements: novel gene-capturing systems of the 21st century? Microbiol Mol Biol Rev. 2006;70(2):296-316.
-
13Toleman MA, Simm AM, Murphy TA, Gales AC, Biedenbach DJ, Jones RN, et al. Molecular characterization of SPM-1, a novel metallo-beta-lactamase isolated in Latin America: report from the SENTRY antimicrobial surveillance programme. J Antimicrob Chemother . 2002;50(5):673-9.
-
14Maciel WG, Silva KE, Bampi JV, Bet GM, Ramos AC, Gales AC, et al. Identification of São Paulo metallo-beta-lactamase-1-producing Pseudomonas aeruginosa in the Central-West region of Brazil: a case study. Rev Soc Bras Med Trop . 2017;50(1):135-7.
-
15Gales AC, Menezes LC, Silbert S, Sader HS. Dissemination in distinct Brazilian regions of an epidemic carbapenem-resistant Pseudomonas aeruginosa producing SPM metallo-beta-lactamase. J Antimicrob Chemother . 2003;52(4):699-702.
-
16Babu KV, Visweswaraiah DS, Kumar A. The influence of Imipenem resistant metallo-beta-lactamase positive and negative Pseudomonas aeruginosa nosocomial infections on mortality and morbidity. J Nat Sci Biol Med. 2014;5(2):345-51.
-
17Dantas RC, Ferreira ML, Gontijo-Filho PP, Ribas RM. Pseudomonas aeruginosa bacteraemia: independent risk factors for mortality and impact of resistance on outcome. J Med Microbiol. 2014;63(Pt 12):1679-87.
-
18Higgins JPT, Green S. Cochrane Handbook for Systematic Review of Intervention section, Version 5.1.0. The Cochrane Collaboration; 2011. Updated March 2011; cited 2017 Dec 27. Available from: Available from: http://handbook-5-1.cochrane.org/.
» http://handbook-5-1.cochrane.org/. -
19Review Manager (RevMan). Computer program. Version 5.3. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
-
20Peña C, Gómez-Zorrilla S, Oriol I, Tubau F, Dominguez MA, Pujol M, et al. Impact of multidrug resistance on Pseudomonas aeruginosa ventilator-associated pneumonia outcome: predictors of early and crude mortality. Eur J Clin Microbiol Infect Dis. 2013;32(3):413-20.
-
21Araujo BF, Ferreira ML, Campos PA, Royer S, Batistão DW, Dantas RC, et al. Clinical and Molecular Epidemiology of Multidrug-Resistant P. aeruginosa Carrying aac(6')-Ib-cr, qnrS1 and blaSPM Genes in Brazil. PLoS One. 2016;11(5):e0155914.
-
22Tam VH, Rogers CA, Chang KT, Weston JS, Caeiro JP, Garey KW. Impact of multidrug-resistant Pseudomonas aeruginosa bacteremia on patient outcomes. Antimicrob Agents Chemother . 2010;54(9):3717-22.
-
23Sharma M, Yadav S, Chaudhary U. Metallo-beta-lactamase Producing Pseudomonas aeruginosa in Neonatal Septicemia. J Lab Physicians. 2010;2(1):146.
-
24Nangino GO, Oliveira CD, Correia PC, Machado NM, Dias ATB. Impacto financeiro das infecções nosocomiais em unidades de terapia intensiva em hospital filantrópico de Minas Gerais. Rev Bras Ter Intensiva. 2012;24(4);357-61.
-
25Caselli D, Cesaro S, Ziino O, Zanazzo G, Manicone R, Livadiotti S, et al. Multidrug resistant Pseudomonas aeruginosa infection in children undergoing chemotherapy and hematopoietic stem cell transplantation. Haematologica. 2010;95(9):1612-5.
-
26Peña C, Suarez C, Gozalo M, Murillas J, Almirante B, Pomar V, et al. Prospective multicenter study of the impact of carbapenem resistance on mortality in Pseudomonas aeruginosa bloodstream infections. Antimicrob Agents Chemother . 2012;56(3):1265-72.
-
27Joo EJ, Kang CI, Ha YE, Kang SJ, Park SY, Chung DR, et al. Risk factors for mortality in patients with Pseudomonas aeruginosa bacteremia: clinical impact of antimicrobial resistance on outcome. Microb Drug Resist. 2011;17(2):305-12.
-
28Trecarichi EM, Tumbarello M, Caira M, Candoni A, Cattaneo C, Pastore D, et al. Multidrug resistant Pseudomonas aeruginosa bloodstream infection in adult patients with hematologic malignancies. Haematologica. 2011;96(1):e1-3.
-
29Morata L, Cobos-Trigueros N, Martínez JA, Soriano A, Almela M, Marco F, et al. Influence of multidrug resistance and appropriate empirical therapy on the 30-day mortality rate of Pseudomonas aeruginosa bacteremia. Antimicrob Agents Chemother . 2012;56(9):4833-7.
Publication Dates
-
Publication in this collection
Jul-Aug 2018
History
-
Received
01 Feb 2018 -
Accepted
06 July 2018