ABSTRACT
This study aimed at identifying the best gender division pattern for swine at nursery stage, through the evaluation of behavioral and environmental parameters. Three treatments were established to achieve our objective: single-sex housing of 26 surgically castrated male pigs; mixed-sex housing of 13 castrated males and 13 females; and single-sex housing of 26 females. Environmental and behavioral data have been recorded. It was used an ethogram to make an inventory of behaviors and that was used in multivariate analysis. Bite addiction differed statistically among treatments, and the lowest occurrence was in mixed-sex housing. Results of the multivariate analysis suggested that regardless of the treatment chosen, relative humidity is positively associated with the production of gases and dry-bulb temperature with airspeed. Mixed-sex housing had multivariate relations suggesting less aggressive behaviors over food resources, being an example of social interaction that improves swine welfare.
KEYWORDS:
multivariate analysis; welfare; behavior; facilities
INTRODUCTION
Brazil is the world's fourth-largest pork producer and exporter. In the country prevails intensive pig farming to maximize economic gains and production (BAPTISTA et al., 2011BAPTISTA, R. I. A. A.; BERTANI, G. R.; BARBOSA, C. N. Indicadores do bem-estar em suínos. Ciência Rural, Santa Maria, v.41, n.10, p.1823-1830, out. 2011.). Reducing labor costs and simplifying logistics contribute to swine producers adopt wean-to-finish system. In this production system animals are kept in the same facility, from weaning to finish, optimizing thus the use of facilities and promoting animal welfare (MEDEIROS et al., 2014MEDEIROS, B.B.L.; MASSARI, J.M.; MOURA, D.J.; CURI, T.M.R.C.; MAIA, A.P.A. Uso da geoestatística na avaliação de variáveis ambientais em galpão de suínos criados em sistema “wean to finish” na fase de terminação. Engenharia Agrícola, Jaboticabal, v.34, n.5, p.800-811, set./out. 2014.).
Animal welfare can be measured by objective indicators (glucocorticoids) (RAULT, 2012RAULT, J.L. Friends with benefits: Social support and its relevance for farm animal welfare. Applied Animal Behaviour Science, Amsterdam, v.136, n.1, p.1-14, jan. 2012.) and subjective indicators (aggressive behavior, injuries, and with the analysis of images and vocalization) (PEREIRA et al., 2014PEREIRA, E.M.; NAAS, I.A.; GARCIA, R.G. Identification of acoustic parameters for broiler welfare estimate. Revista Engenharia Agrícola, Jaboticabal, v.34, n.3, p.413-421, maio/jun. 2014.). In a productive system, animal behavior may often be the first animal's response to a stressful environment (TEMPLE et al., 2011TEMPLE, D.; MANTECA, X.; VELARDE, A.; DALMAU, A. Assessment of animal welfare through behavioral parameters in Iberian pigs in intensive and extensive conditions. Applied Animal Behaviour Science, New York, v.131, n.1-2, p.29-39, abr. 2011.).
Regardless the swine production system adopted, there are no standards established for separating pigs by sex in pens, and single-sex housing is still the most common practice. In this regard, studies on separating swine by gender are concerned with production parameters of males, castrated males, and females (MORAES et al., 2010MORAES, E.; KIEFER, C.; SILVA, I.S. Ractopamina em dietas para suínos machos imunocastrados, castrados e fêmeas. Ciência Rural, Santa Maria, v.40, n.2, p.409-414, fev. 2010.). It is worth mentioning that studies on environmental quality, behavior, and housing (if animals are mixed or not) are relatively scarce, especially on wean-to-finish production system.
Multivariate analysis is a technique developed to analyze simultaneously correlated experimental variables (RIBEIRO et al., 2013RIBEIRO, F. A. D. L.; MANTOVANI, G. A.; POPPI, R. J.; ROSÁRIO, F. F. D.; BEZERRA, M. C. M.; BASTOS, A. L. M.; MELO, V. L. A. D. PCA: a tool for identification of chemical tracers for formation and injection waters associated with oil production. Química Nova, São Paulo, v.36, n.9, p.1281-1287, 2013.). Multivariate methods involve thus reduction processes, optimization, sorting, and classification of multidimensional data (GONÇALVES; FRITSCHE-NETO, 2012GONÇALVES, M. C.; FRITSCHE-NETO, R. Tópicos especiais de biometria no melhoramento de plantas: com exemplos numéricos e de programação no SAS®. Visconde do Rio Branco: Suprema, 2012.). Many authors have used multivariate analysis to evaluate livestock behavior (KROLOW et al., 2014KROLOW, R.H.; SILVA, M.A.D.; PAIM, N.R.; MEDEIROS, R.B.D.; VELHO, I.M.P.H. Comportamento ingestivo de vacas leiteiras em pastagem de azevém alimentadas com diferentes fontes proteicas. Arquivo Brasileiro de Medicina Veterinaria e Zootecnia, Belo Horizonte, v.66, n.3, p.845-852, jun. 2014.; VELOSO et al., 2015VELOSO, R.C.; PIRES, A.V.; TORRES FILHO, R.A.; FERREIRA, T.A.; SILVA, M. T. P.; BALLOTIN, L. M. V.; BONAFÉ, C. M. Divergência genética entre genótipos de frangos tipo caipira. Arquivo Brasileiro de Medicina Veterinária e Zootecnia, Belo Horizonte, v.67, n.5, p.1353-1360, out. 2015.). Studies have been developed on the importance of the pre-slaughter handling of swine for meat quality (MACHADO et al., 2014MACHADO, S.T.; SANTOS, R.C.; CALDARA, F.R.; GONÇALVES, M.C.; NÄÄS, I.A. Análise multivariada integrada para avaliação dos efeitos do manejo pré-abate na qualidade da carne suína. Engenharia Agrícola, Jaboticabal, v.34, n.3, p.435-444, maio/jun. 2014.) and swine carcass condemnation (BUENO et al., 2013BUENO, L.S.; CALDARA, F.R.; NAAS, I.A.; SALGADO, D.D.; GARCIA, R.G.; PAZ, I.C.L.A. Swine Carcass Condemnation in Commercial Slaughterhouses. Revista MVZ Córdoba, Montería, v.18, p.3836-3842, set./dez. 2013.).
According to the above mentioned, this study aimed at identifying the best gender division pattern for swine at nursery stage, based on behavioral and environmental parameters, correlating them by multivariate analysis (main components).
MATERIAL AND METHODS
This study was conducted in a commercial swine farm, located in Joaçaba, Santa Catarina, Brazil, between September 2012 and February 2013. The farm altitude is 522 meters, located at latitude 27°10′41″ and longitude 51°30′17″, with the predominance of Cfa climate – humid subtropical climate according to Köppen classification – with average temperature of 18.1 °C and average precipitation of 1,841 mm.
The curtain-barn had wooden and masonry walls, gable roof with ceramic tiles and 0.60 m eaves, 3 m of ceiling height, 1.3 meter masonry lateral walls, double lock curtains, and a 0.95 m wide corridor. The facility had no ridge vent and HVAC system, so the curtains were the only way to control the environment quality. The facility had 12 pens (6.4m × 3.85m) and 2 auxiliary pens (BA) (Fig. 1), each containing a semi-automatic feeder, a double nipple drinker (according to the animal's height) and two troughs drinkers, and 1 m hollow block walls separating the pens.
Since rations were constantly changed according to the cooperative and the farm management, following animals' nutritional requirements, weight, and age, the raising period was divided into phases. When compared to the conventional production system, this system has: first stage (nursery), second phase (growth), third stage (growth and finishing), and fourth stage (finishing). In the first three stages pigs are housed for 27 days and in the last stage for 34 days. In the present article we will emphasize only results from the first phase.
In total, 301 animals (Large White × Agroceres) were housed in a wean-to-finish barn, right after weaning (28 days old), with average initial weight of 7.5 kg and average slaughter weight of 125 kg. From this main group, 78 piglets at nursery stage were selected at random and distributed in only 3 of the 12 pens, arranged next to each other (Fig. 1). Once distributed in the pens, pigs were not relocated and the following treatments were carried out: Treatment 1 (T1), pen with 26 surgically castrated male pigs; Treatment 2 (T2), mixed sex pen with 13 surgically castrated male and 13 female pigs; and Treatment 3 (T3), pen with 26 female pigs. During all the experiment, water and ration were provided. The animals entered nursery stage with 46 days old and initial average weight of 13.4 kg, remaining at nursery until they were 73 days old with average weight of 37.9 kg.
Piglets' behavioral assessment was conducted to verify the influence of gender segregation on the proposed treatments. For this purpose simultaneous recordings were made with Sony Handycam DCR-SX40 cameras, installed on tripods inside the barn, positioned in a way that the observable visual field could include all animals.
Recordings started at 2:00 p.m. and the footage was analyzed by the same observer. Thus, eight repetitions (recordings) were carried out at nursery stage, two recordings per week in pre-established days. Each footage was twenty minutes long, being discarded the initial three minutes and the final two minutes, so that the animals could adapt to the environment without the presence of observers inside the barn, according to the methodology adapted from BIZERAY et al. (2002)BIZERAY, D.; ESTEVEZ, I.; LETERRIER, C.; FAURE, J.M. Effects of increasing environmental complexity on the physical activity of broiler chickens. Applied Animal Behaviour Science, Londres, v.79, n.1, p.27-41, set. 2002.. It was analyzed 15 minutes of continuous footage from each recording. An ethogram was adapted from the methodology proposed by MASSARI et al. (2015)MASSARI, J. M.; CURI, T. M. D. C.; MOURA, D. J.; MEDEIROS, B. B.; SALGADO, D. Behavioral characteristics of different gender division of growing and finishing swine in “wean to finish” system. Engenharia Agrícola, Jaboticabal, v.35, n.4, p.646-656, jul./ago. 2015..
The footage was paused every three minutes for analysis and the number of animals standing, lying, and at the feeder were counted, since these actions varied greatly during the recording, according to the focal animal sampling methodology proposed by ALTMANN (1974)ALTMANN, J. Observational study of behavior: sampling methods. Behaviour, Leiden, v.49, p.227-267, 1974.. Subsequently, each behavior was registered with its percentage. The other analyzed behaviors were registered during the whole recording, because they were more dynamic behaviors, e.g., agonistic behavior, social interaction, stereotypy, exploratory, and bite addiction according to methodology used by MASSARI et al. (2015)MASSARI, J. M.; CURI, T. M. D. C.; MOURA, D. J.; MEDEIROS, B. B.; SALGADO, D. Behavioral characteristics of different gender division of growing and finishing swine in “wean to finish” system. Engenharia Agrícola, Jaboticabal, v.35, n.4, p.646-656, jul./ago. 2015..
With data gathered, PCA graphs were generated using MINITAB 15.1 statistical software to study behavior of the swine at nursery stage, divided in pens by gender. Environmental data were measured before and immediately after the footage to verify any possible environmental variations that could interfere with the piglets' behavior. The ethogram used was adapted from MASSARI et al. (2015)MASSARI, J. M.; CURI, T. M. D. C.; MOURA, D. J.; MEDEIROS, B. B.; SALGADO, D. Behavioral characteristics of different gender division of growing and finishing swine in “wean to finish” system. Engenharia Agrícola, Jaboticabal, v.35, n.4, p.646-656, jul./ago. 2015. for assessment and is available on Table 1.
Established ethogram for behavioral analysis of swine divided by gender in wean-to-finish system.
Environmental variables were also considered to verify in what measure the environment influenced behaviors. The following variables were measured: dry bulb temperature (DBT, °C), relative humidity (RH, %), airspeed (AS, ms-1), and ammonia and carbon dioxide concentrations (NH3 and CO2, ppm). These data were collected at the beginning and at the end of each recording, with an interval of 20 minutes, in the geometric center of each pen, and at 1.5 m above the floor. Air temperature and speed were collected using Extech® thermal anemometer (model 407123), relative humidity using Instrutherm® digital multifunctional meter (model THDL-400), and NH3 and CO2 using BW® Technologies meter (GasAlert Micro5 IR model). With dry bulb temperature and relative humidity data, we calculated the temperature humidity index (THI) through [eq. (1)], proposed by THOM (1959)THOM, E.C. The discomfort index. Weatherwise, Washington, v.12, n.2, p.57-60, 1959., in which values between 64 and 74 are classified as “Comfort”; greater than 74 up to 78 as “Warning”, and values above 78 as “Danger”.
where,
-
DBT = dry bulb temperature (°C),
-
DPT = dew point temperature (°C).
We used Principal Component Analysis (PCA) to identify possible associations between the behaviors analyzed and the environmental variables considered. All environmental and behavioral parameters were considered for every PCA graph generated for each treatment. After conducting the test for normality of data, we concluded that data are nonparametric. Thus, we decided to use Kruskal-Wallis nonparametric test (p ≤ 0.05), using MINITAB® 15.1 software to compare the observed behaviors frequencies and environmental variables registered for each treatment.
RESULTS AND DISCUSSION
Table 2 presents summarized values from days of descriptive and confirmatory statistical data collection of environmental variables from the nursery where pigs were housed.
Descriptive and confirmatory statistics of environmental variables from wean-to-finish swine facility with animals at nursery stage.
No statistical differences were observed (p > 0.05) between environmental variables of each pen/treatment, because they are physically arranged next to each other, so it is pertinent that there were no great variations.
According to SAMPAIO et al. (2004) and GLOBALGAP (2012), all environmental variables are within the limit considered ideal for thermal and air comfort of animals, except average DBT value (19.83ºC) for T1, because comfort temperature for piglets at nursery stage should be between 20 and 24ºC (SAMPAIO et al., 2004). However, when analyzed the minimum values found, all treatments had values below 20ºC, so at some point of the day all animals were in an environment with temperature below the standard, causing thermal stress.
Average values of each behavior frequency (repetition) and the descriptive and confirmatory statistical analysis (Kruskall-Wallis test with 5% of significance) are on Table 3.
Descriptive and confirmatory statistics of behavioral variables in a wean-to-finish swine facility with animals at nursery stage.
Among the analyzed behaviors, only “Bite addiction” had statistical difference to Kruskall-Wallis test with 5% of significance, p value was 0.046. Thus, mixed sex housing was statistically different from groups of isolated males or females. Moreover, T1 and T3 were not statistically different when compared with one another. ZONDERLAND et al. (2010)ZONDERLAND, J.J.; BRACKE, M.B.M.; DEN HARTOG, L.A.D.; KEMP, B.; SPOOLDER, H.A.M. Gender effects on tail damage development in single- or mixed-sex groups of weaned piglets. Livestock Science, Amsterdam, v.129, p.151–158, 2010., at their final observations of piglets in same-sex and mixed-sex housing also found less frequently bite addiction in pigs in mixed-sex housing.
It is important to emphasize that bite addiction, most of the times, focus another piglet tail (tail biting), having a negative effect on animal welfare and also resulting in considerable economic losses by low carcass quality (TAYLOR et al., 2010TAYLOR, N. R.; MAIN, D. C. J.; MENDL, M.; EDWARDS, S.A. Tail-biting: A new perspective. The Veterinary Journal, London, v.186, p.137-147, out. 2010.). Therefore, it is possible to affirm that mixed-sex housing reduces this behavior occurrence, which is beneficial to the producer, to the animal, and even to the final consumer (meat quality).
There was no evidence to differentiate treatments regarding other behaviors occurrences, with individual tests. However, swine in mixed-sex pens (T2) had low frequency of most unwanted behaviors, such as “agonistic”, “stereotyped”, “mount”, and “suction addiction”. These behaviors are considered unwanted, because they affect animal health and welfare, causing losses to producers since quality of carcass becomes significantly lower (D'EATH et al., 2010D'EATH, R.B.; TURNER, S.P.; KURT, E.; EVANS, G.; THOLKING, L.; LOOFT, H.; WIMMERS, K.; MURANI, E.; KLONT, R.; FOURY, A.; ISON, S.H.; LAWRENCE, A.B.; MORMEDE, P. Pigs aggressive temperament affects pre-slaughter mixing aggression, stress and meat quality. Animal, Cambridge, v.4, 604-616, abr. 2010.). On the other hand, this treatment also showed the lowest average values for “ludic” and “interaction” behaviors.
For PCA analysis, vectors with the same direction and sense are positively and strongly associated. Situations with similar direction vectors but opposite senses imply strong negative associations and vectors forming angles near 90° are not correlated. Small magnitude vectors have been removed from the model, as they are not well explained by main components (VERCELLINO et al., 2013VERCELLINO, R.A.; MOURA, D.J.; NAAS, I.A.; MAIA, A.P.A.; MEDEIROS, B.B.L.; CARVALHO, T.M.R. The influence of side-curtain color in broiler chick behavior. Revista Brasileira de Ciência Avícola, Campinas, v.15, n.3, p.173-179, jul./set. 2013.; SALGADO, 2006SALGADO, D. D. Modelo estatístico para predição de bem-estar de reprodutoras de frango de corte baseado em dados de ambiente e análise do comportamento. 2006. 113f. Dissertação (Mestrado em Engenharia Agrícola) – Faculdade de Engenharia Agrícola, Universidade Estadual de Campinas, Campinas, 2006.).
The graph of main components for T1 is shown by Fig. 2. We used MINITAB® 15.1 software to calculate eigenvalues, which are equivalent to components variances. According to results obtained, the two components with higher eigenvalues have 80.7% of the sum of all eigenvalues. In other words, 80.7% of total variation in data has been explained by the two main components. For this graph (Fig. 2), only the vector for suction addiction variable was rejected for being little explained by components.
According to the graph generated (Fig. 2), “belly nosing” and “lying” behaviors are intensely and positively associated, indicating a tendency for animals to practice belly nosing when they are lying. Nevertheless, when piglets are lying they are not motivated to perform “standing”, “ludic”, “mount”, “biting”, and “stereotyped” behaviors. Likewise, when practicing belly nosing, they are not disposed to “interaction”, “agonistic”, and “exploratory” behaviors, because the abovementioned relations are intense and opposite.
“Standing” and “ludic” behaviors had a strong and positive relationship, because, according to the ethogram proposed for this study the ludic behavior was defined as runs with or without diversified purpose. HELD and SPINKA (2011)HELD, S.D.E.; SPINKA, M. Animal play and animal welfare. Animal Behaviour, New York, v.81, p.891–899, maio 2011. point out that the ludic behavior stimulates social interactions between animals, as the positive relationship between these two variables is observed.
“Agonistic” and “exploratory” behavioral variables are strongly and positively related, also associated with “interaction” in a lesser degree. It should consider the species natural tendency to explore the environment, forage, play with other piglets, and socialize (also aggressively). The majority of intensive pig farming systems do not usually offer appropriate substrates to encourage exploratory behavior, e.g., use of straw (ZWICKER et al., 2012ZWICKER, B.; GYGAX, L.; WECHSLER, B.; WEBER, R. Influence of the accessibility of straw in racks on exploratory behavior in finishing pigs. Livestock Science, Amsterdam, v.148, p.67–73, set. 2012.). Still, these two variables are positively related to “drinking”, denoting aggressive disputes over water sources.
“Stereotyped” behavior has a strong and positive relationship with “bite addiction” and “mount”, i.e., stereotyped behavior leads to other behaviors considered unwanted (mount, agonistic, bite and suction addiction), denoting low levels of animal welfare (MAIA et al., 2013MAIA, A.P.A.; SARUBBI, J.; MEDEIROS, B.B.L.; MOURA, D.J. Enriquecimento ambiental como medida para o bem-estar positivo de suínos (Revisão). Revista do Centro do Ciências Naturais e Exatas, Santa Maria, v.14, n.14, p.2862-2877, set. 2013.).
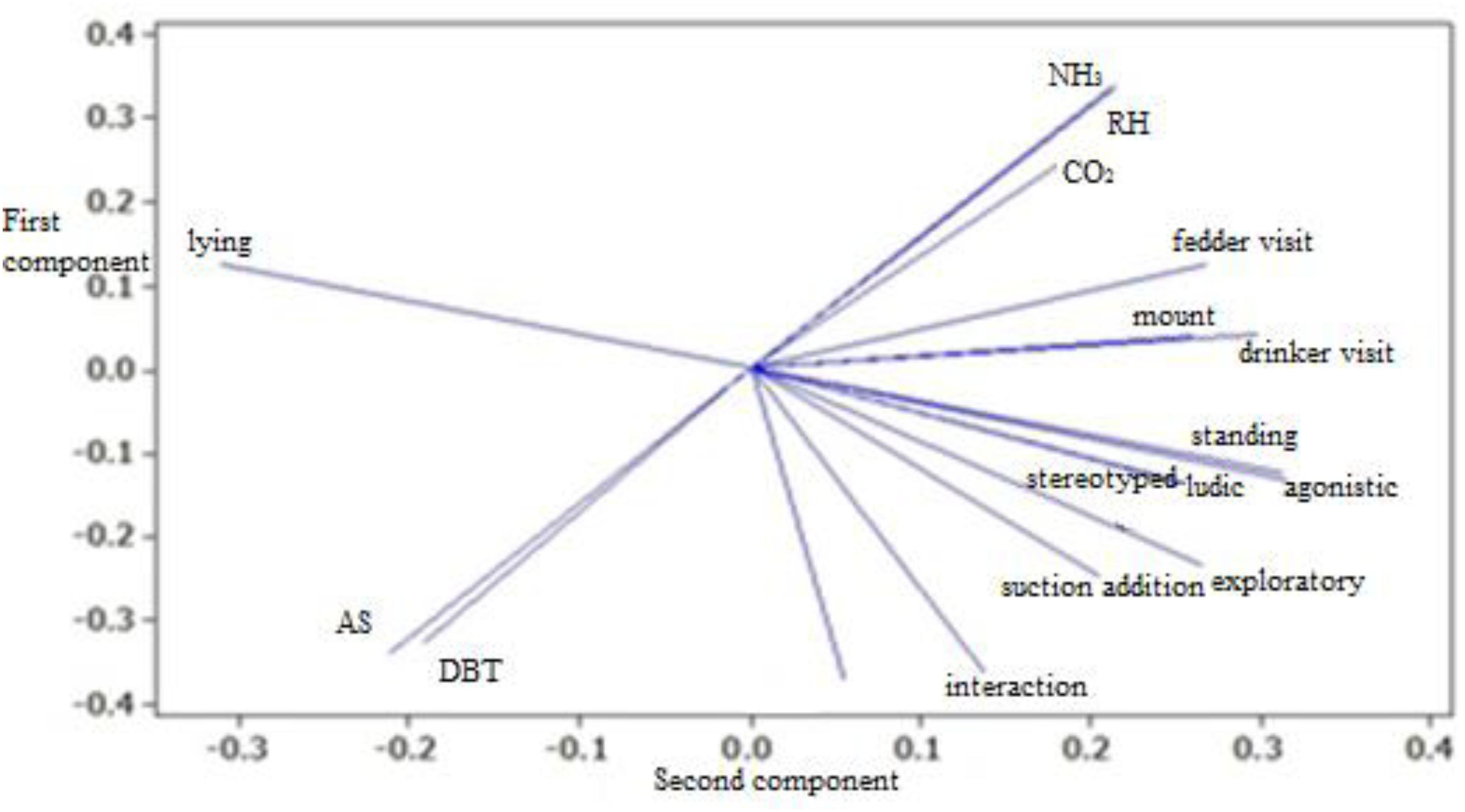
For T2, mixed sex housing, PCA results showed that 81.5% of total variation in data has been explained (Fig. 3). Vectors generated for “ludic” and “stereotyped” behaviors have been discarded for not being well explained by the components.
For this housing condition (Fig. 3), “lying” behavior does not have a strong positive association with other behavioral variable, but only moderately with “THI” and weak with “DBT” and “AS”. However, it has a strong negative association with “standing”, “mount”, and “agonistic”, which are positively and strongly related to each other. It is possible that mount behavior is a consequence of aggressive behavior (FORD, 1990FORD, J.J. Differentation of sexual behaviour in pigs. Journal of Reprodution Fertility, Cambridge, v.40, p.311-321, 1990.). “Belly nosing” has a strong positive relationship with “exploratory” behavior and in lesser extent with “interaction” and “eating”. Therefore, “belly nosing” may be related with hunger or feeding motivation (HÖTZEL et al. 2004HÖTZEL, M.J.; MACHADO FILHO, L.C.P.; WOLF, F.M.; COSTA, O.A.D. Behaviour of sows and piglets reared in intensive outdoor or indoor systems. Applied Animal Behaviour Science, New York, v.86, p.27–39, maio 2004.), but also may be preceded or followed by other behaviors, e.g., smell another piglet, being highly involved in social interactions (LI; GONYOU, 2002LI, Y.Z.; GONYOU, H.W. Analysis of belly nosing and associated behaviour among pigs weaned at 12–14 days-ofage. Applied Animal Behaviour Science, New York, v.77, p.285–294, jun. 2002.).
When swine are housed in mixed-sex pens, “eating” is positively and strongly related to “interaction”, “exploratory”, and “belly nosing”, but also positively and moderately to “agonistic” and “mount”, denoting a less aggressive hierarchy when compared to other treatments, what can be inferred from other generated graphs. Therefore, it is pertinent to affirm that mixed-sex housing provides a stable social hierarchy, essential for coexistence and an evolutionary advantage, for it prevents aggressions and injuries (MEESE; EWBANK, 1973MEESE, G.B.; EWBANK, R. The establishment and nature of the dominance hierarchy in the domesticated pig. Animal Behaviour, New York, v.21, p.326–334, maio 1973.). Swine in mixed-sex housing had the lowest frequencies of agonistic behavior, even without statistical difference (p>0.05).
Agonistic behavior (aggressive) is very complex and has aggression levels (from mild to critical) (OCZAK et al., 2013OCZAK, M.; ISMAYILOVA, G.; COSTA, A.; VIAZZI, S.; SONODA, L. T.; FELS, M.; VRANKEN, E. Analysis of aggressive behaviours of pigs by automatic video recordings. Computers and electronics in agriculture, New York, v.99, p.209-217, nov.2013.). We observed a positive and moderate tendency of concomitant occurrence of “suction addiction” and “bite addiction” – “bite addiction” is more related to “drinking”, denoting a dispute over the water source.
For T3, isolated females, PCA results also showed that 81.5% of total variation in data has been explained. To make this graph (Fig. 4) we disregarded “THI” and “belly nosing” vectors for not being totally explained by the components.
As has been found in Treatment 2, “lying” – for isolated females – does not positively associate with other behaviors, although it is strongly and negatively associated with other variables, especially “standing”, “agonistic”, “ludic”, and “stereotyped”.
“Eating” is directly related to “mount” and “drinking”, having a very strong positive connection with each other – this means that mount during feeding can be a dominant behavior used by top-ranking animals to reach the feeder and the drinker. It is thus probable that aggression and mount affect feeding behavior (FREDRIKSEN et al., 2004FREDRIKSEN, B.; NAFSTAD, O.; LIUM, B. M.; MARKA, C. H.; HEIER, B. T.; ANDRESEN, O. Entire male pig production in ‘born-to-finishing-pens'. Oslo: Norwegian Meat Research Centre, ago. 2004. (Project no. 6624)). Moreover, there was a moderate positive relationship between “agonistic” and “drinking”, which indicates that when sows are housed in same-sex pens, the highest aggression and dispute incidences are for reaching the drinker, and in a lesser degree the feeder. Nevertheless, for the other housing conditions (only males and mixed), agonistic interactions are related almost in the same proportion to “eating” and “drinking”, but in a lesser degree of intensity.
T3 had the highest averages of agonistic behavior and the same average of mount behavior of T1, both higher than T2, but without statistical difference. “Ludic” and “stereotyped” behaviors are extremely positively related to each other. Stereotyped behavior indicates special cases of abnormal behavior and welfare issues (MAIA et al., 2013MAIA, A.P.A.; SARUBBI, J.; MEDEIROS, B.B.L.; MOURA, D.J. Enriquecimento ambiental como medida para o bem-estar positivo de suínos (Revisão). Revista do Centro do Ciências Naturais e Exatas, Santa Maria, v.14, n.14, p.2862-2877, set. 2013.), on the other hand, ludic behavior means welfare and pleasure for young animals (HELD; SPINKA, 2011HELD, S.D.E.; SPINKA, M. Animal play and animal welfare. Animal Behaviour, New York, v.81, p.891–899, maio 2011.). It is thus pertinent to remember that associations established do not mean necessarily data dependence and may be related to other variables not included in the analysis (SALGADO, 2006SALGADO, D. D. Modelo estatístico para predição de bem-estar de reprodutoras de frango de corte baseado em dados de ambiente e análise do comportamento. 2006. 113f. Dissertação (Mestrado em Engenharia Agrícola) – Faculdade de Engenharia Agrícola, Universidade Estadual de Campinas, Campinas, 2006.).
From the PCA graphs generated (Fig. 2, 3, and 4); it was possible to identify similar occurrences in all treatments. Regarding the environmental variable “DBT”, in all treatments there is a strong and inverse association with the variable “eating”, especially T1, which had the strongest relationship, followed by T2 and T3. This tendency indicates that rising the temperature implies in reduction of eating frequency and difficulties for weight gain (BRETAS et al., 2011BRÊTAS, A.A.; FERREIRA, R.A.; AMARANTE JÚNIOR, V.S.; PEREIRA, W.E.; FONSECA, J.B.; CALDAS, F.R.L. Balanço eletrolítico para suínos machos castrados em crescimento mantidos em ambiente de alta temperatura. Ciência e Agrotecnologia, Lavras, v.35, n.1, p.186-194, jan./fev. 2011.).
Another similar tendency among treatments is found for environmental variables “NH3”, “CO2”, and “RH” that had vectors with the same direction and intensity, being positive and strongly related to each other. We emphasize that these environmental variables were positively associated to “eating” in T1 and T3 (more intensive for T1). For mixed-sex housing, however, it was associated with “drinking”.
Variable “AS” has a strong and negative relation with “CO2” and “NH3” – consistent data because ventilation is the main channel of air exchange and gas dispersion (PANDORFI et al., 2012PANDORFI, H.; ALMEIDA, G. L.P.; GUISELINI, C. Zootecnia de precisão: princípios básicos e atualidades na suinocultura. Revista Brasileira de Saúde e Produção Animal, Salvador, v.13, n.2, p. 558-568, abr./jun. 2012.). According to CURI et al. (2014)CURI, T.M.R.C., VERCELLINO, R.A.; MASSARI, J.M SOUZA, Z.M.; MOURA, D.J. Geoestatística para a avaliação do controle ambiental do sistema de ventilação em instalações comerciais para frangos de corte. Engenharia Agrícola, Jaboticabal, v.34, n.6, p.1062-1074, nov./dez. 2014. who considered the influence of temperature, relative humidity, air speed, CO2, and NH3 on poultry production, the concentration of ammonia is strongly related to relative humidity levels, confirming the data obtained in multivariate analysis.
CONCLUSIONS
According to what has been exposed, we may suggest mixed-sex housing as the best solution for swine at nursery stage in wean-to-finish systems, for although there is no statistical evidence of lower average frequency of agonistic behavior, it had the lowest average (p<0.05) of bite addiction and multivariate relations suggesting less aggressive disputes over food resources, revealing a social interaction that provides better conditions for the animals.
ACKNOWLEDGEMENTS
To Hamilton Abatti farm, the cooperative (Cooperio), CNPq, and FAPESP.
REFERENCES
- ALTMANN, J. Observational study of behavior: sampling methods. Behaviour, Leiden, v.49, p.227-267, 1974.
- BAPTISTA, R. I. A. A.; BERTANI, G. R.; BARBOSA, C. N. Indicadores do bem-estar em suínos. Ciência Rural, Santa Maria, v.41, n.10, p.1823-1830, out. 2011.
- BIZERAY, D.; ESTEVEZ, I.; LETERRIER, C.; FAURE, J.M. Effects of increasing environmental complexity on the physical activity of broiler chickens. Applied Animal Behaviour Science, Londres, v.79, n.1, p.27-41, set. 2002.
- BRÊTAS, A.A.; FERREIRA, R.A.; AMARANTE JÚNIOR, V.S.; PEREIRA, W.E.; FONSECA, J.B.; CALDAS, F.R.L. Balanço eletrolítico para suínos machos castrados em crescimento mantidos em ambiente de alta temperatura. Ciência e Agrotecnologia, Lavras, v.35, n.1, p.186-194, jan./fev. 2011.
- BUENO, L.S.; CALDARA, F.R.; NAAS, I.A.; SALGADO, D.D.; GARCIA, R.G.; PAZ, I.C.L.A. Swine Carcass Condemnation in Commercial Slaughterhouses. Revista MVZ Córdoba, Montería, v.18, p.3836-3842, set./dez. 2013.
- CURI, T.M.R.C., VERCELLINO, R.A.; MASSARI, J.M SOUZA, Z.M.; MOURA, D.J. Geoestatística para a avaliação do controle ambiental do sistema de ventilação em instalações comerciais para frangos de corte. Engenharia Agrícola, Jaboticabal, v.34, n.6, p.1062-1074, nov./dez. 2014.
- D'EATH, R.B.; TURNER, S.P.; KURT, E.; EVANS, G.; THOLKING, L.; LOOFT, H.; WIMMERS, K.; MURANI, E.; KLONT, R.; FOURY, A.; ISON, S.H.; LAWRENCE, A.B.; MORMEDE, P. Pigs aggressive temperament affects pre-slaughter mixing aggression, stress and meat quality. Animal, Cambridge, v.4, 604-616, abr. 2010.
- FREDRIKSEN, B.; NAFSTAD, O.; LIUM, B. M.; MARKA, C. H.; HEIER, B. T.; ANDRESEN, O. Entire male pig production in ‘born-to-finishing-pens'. Oslo: Norwegian Meat Research Centre, ago. 2004. (Project no. 6624)
- FORD, J.J. Differentation of sexual behaviour in pigs. Journal of Reprodution Fertility, Cambridge, v.40, p.311-321, 1990.
- GONÇALVES, M. C.; FRITSCHE-NETO, R. Tópicos especiais de biometria no melhoramento de plantas: com exemplos numéricos e de programação no SAS®. Visconde do Rio Branco: Suprema, 2012.
- HELD, S.D.E.; SPINKA, M. Animal play and animal welfare. Animal Behaviour, New York, v.81, p.891–899, maio 2011.
- HÖTZEL, M.J.; MACHADO FILHO, L.C.P.; WOLF, F.M.; COSTA, O.A.D. Behaviour of sows and piglets reared in intensive outdoor or indoor systems. Applied Animal Behaviour Science, New York, v.86, p.27–39, maio 2004.
- KROLOW, R.H.; SILVA, M.A.D.; PAIM, N.R.; MEDEIROS, R.B.D.; VELHO, I.M.P.H. Comportamento ingestivo de vacas leiteiras em pastagem de azevém alimentadas com diferentes fontes proteicas. Arquivo Brasileiro de Medicina Veterinaria e Zootecnia, Belo Horizonte, v.66, n.3, p.845-852, jun. 2014.
- LI, Y.Z.; GONYOU, H.W. Analysis of belly nosing and associated behaviour among pigs weaned at 12–14 days-ofage. Applied Animal Behaviour Science, New York, v.77, p.285–294, jun. 2002.
- MACHADO, S.T.; SANTOS, R.C.; CALDARA, F.R.; GONÇALVES, M.C.; NÄÄS, I.A. Análise multivariada integrada para avaliação dos efeitos do manejo pré-abate na qualidade da carne suína. Engenharia Agrícola, Jaboticabal, v.34, n.3, p.435-444, maio/jun. 2014.
- MAIA, A.P.A.; SARUBBI, J.; MEDEIROS, B.B.L.; MOURA, D.J. Enriquecimento ambiental como medida para o bem-estar positivo de suínos (Revisão). Revista do Centro do Ciências Naturais e Exatas, Santa Maria, v.14, n.14, p.2862-2877, set. 2013.
- MASSARI, J. M.; CURI, T. M. D. C.; MOURA, D. J.; MEDEIROS, B. B.; SALGADO, D. Behavioral characteristics of different gender division of growing and finishing swine in “wean to finish” system. Engenharia Agrícola, Jaboticabal, v.35, n.4, p.646-656, jul./ago. 2015.
- MEDEIROS, B.B.L.; MASSARI, J.M.; MOURA, D.J.; CURI, T.M.R.C.; MAIA, A.P.A. Uso da geoestatística na avaliação de variáveis ambientais em galpão de suínos criados em sistema “wean to finish” na fase de terminação. Engenharia Agrícola, Jaboticabal, v.34, n.5, p.800-811, set./out. 2014.
- MEESE, G.B.; EWBANK, R. The establishment and nature of the dominance hierarchy in the domesticated pig. Animal Behaviour, New York, v.21, p.326–334, maio 1973.
- MORAES, E.; KIEFER, C.; SILVA, I.S. Ractopamina em dietas para suínos machos imunocastrados, castrados e fêmeas. Ciência Rural, Santa Maria, v.40, n.2, p.409-414, fev. 2010.
- OCZAK, M.; ISMAYILOVA, G.; COSTA, A.; VIAZZI, S.; SONODA, L. T.; FELS, M.; VRANKEN, E. Analysis of aggressive behaviours of pigs by automatic video recordings. Computers and electronics in agriculture, New York, v.99, p.209-217, nov.2013.
- PANDORFI, H.; ALMEIDA, G. L.P.; GUISELINI, C. Zootecnia de precisão: princípios básicos e atualidades na suinocultura. Revista Brasileira de Saúde e Produção Animal, Salvador, v.13, n.2, p. 558-568, abr./jun. 2012.
- PEREIRA, E.M.; NAAS, I.A.; GARCIA, R.G. Identification of acoustic parameters for broiler welfare estimate. Revista Engenharia Agrícola, Jaboticabal, v.34, n.3, p.413-421, maio/jun. 2014.
- RAULT, J.L. Friends with benefits: Social support and its relevance for farm animal welfare. Applied Animal Behaviour Science, Amsterdam, v.136, n.1, p.1-14, jan. 2012.
- RIBEIRO, F. A. D. L.; MANTOVANI, G. A.; POPPI, R. J.; ROSÁRIO, F. F. D.; BEZERRA, M. C. M.; BASTOS, A. L. M.; MELO, V. L. A. D. PCA: a tool for identification of chemical tracers for formation and injection waters associated with oil production. Química Nova, São Paulo, v.36, n.9, p.1281-1287, 2013.
- SALGADO, D. D. Modelo estatístico para predição de bem-estar de reprodutoras de frango de corte baseado em dados de ambiente e análise do comportamento. 2006. 113f. Dissertação (Mestrado em Engenharia Agrícola) – Faculdade de Engenharia Agrícola, Universidade Estadual de Campinas, Campinas, 2006.
- TAYLOR, N. R.; MAIN, D. C. J.; MENDL, M.; EDWARDS, S.A. Tail-biting: A new perspective. The Veterinary Journal, London, v.186, p.137-147, out. 2010.
- TEMPLE, D.; MANTECA, X.; VELARDE, A.; DALMAU, A. Assessment of animal welfare through behavioral parameters in Iberian pigs in intensive and extensive conditions. Applied Animal Behaviour Science, New York, v.131, n.1-2, p.29-39, abr. 2011.
- THOM, E.C. The discomfort index. Weatherwise, Washington, v.12, n.2, p.57-60, 1959.
- VELOSO, R.C.; PIRES, A.V.; TORRES FILHO, R.A.; FERREIRA, T.A.; SILVA, M. T. P.; BALLOTIN, L. M. V.; BONAFÉ, C. M. Divergência genética entre genótipos de frangos tipo caipira. Arquivo Brasileiro de Medicina Veterinária e Zootecnia, Belo Horizonte, v.67, n.5, p.1353-1360, out. 2015.
- VERCELLINO, R.A.; MOURA, D.J.; NAAS, I.A.; MAIA, A.P.A.; MEDEIROS, B.B.L.; CARVALHO, T.M.R. The influence of side-curtain color in broiler chick behavior. Revista Brasileira de Ciência Avícola, Campinas, v.15, n.3, p.173-179, jul./set. 2013.
- ZONDERLAND, J.J.; BRACKE, M.B.M.; DEN HARTOG, L.A.D.; KEMP, B.; SPOOLDER, H.A.M. Gender effects on tail damage development in single- or mixed-sex groups of weaned piglets. Livestock Science, Amsterdam, v.129, p.151–158, 2010.
- ZWICKER, B.; GYGAX, L.; WECHSLER, B.; WEBER, R. Influence of the accessibility of straw in racks on exploratory behavior in finishing pigs. Livestock Science, Amsterdam, v.148, p.67–73, set. 2012.
Publication Dates
-
Publication in this collection
Jan-Feb 2017
History
-
Received
25 May 2016 -
Accepted
18 Aug 2016