ABSTRACT:
Arachnoid cysts are accumulations of cerebrospinal fluid that occur within the arachnoid membrane due to the splitting or duplication of this structure. The aim of this retrospective study is to report the occurrence of this condition in 26 dogs, as well as to describe the findings of computed tomography imaging and magnetic resonance image scans and to correlate these with the clinical signs. A prevalence of the condition was observed in males, predominantly in the small breeds Miniature Pinscher and Shih Tzu, with age raging from 2 months to 9 years. The mainly clinical signs observed in these dogs were seizures and cerebellar ataxia. The study concluded that in 17 dogs the quadrigeminal arachnoid cyst was regarded as an incidental finding, in 6 dogs this condition was regarded as the determinant cause of the clinical signs, and in 3 dogs some clinical signs were associated with arachnoid cyst, but they also presented clinical signs involving others sites of intracranial lesion.
INDEX TERMS:
Quadrigeminal arachnoid cysts; Magnetic Resonance Imaging; Computed Tomography; dogs; seizures; cerebellar ataxia; hydrocephalus; brain diseases; patology
RESUMO:
Cistos aracnoide são acúmulos de líquido cefalorraquidiano que ocorre dentro da membrana aracnoide devido à divisão ou duplicação desta estrutura. O objetivo desse estudo retrospectivo é relatar a ocorrência dessa condição em 26 cães, assim como descrever os aspectos de imagem pela tomografia computadorizada e ressonância magnética e correlacionar com os sinais clínicos. No estudo observamos a prevalência em machos, em raças de pequeno porte, com predomínio de Pinscher miniatura e Shih Tzu, com idade variando de dois meses a nove anos. Os principais sinais neurológicos observados nestes animais foram crises epiléticas e ataxia cerebelar. Foi possível concluir que em 17 cães o cisto aracnoide quadrigeminal foi considerado um achado incidental, em seis cães essa condição foi considerada a causa determinante dos sinais clínicos, e em três cães alguns sinais estavam associados com o cisto aracnoide, porém esses animais apresentaram sinais clínicos relacionados a outros sítios de lesão intracraniana.
TERMOS DE INDEXAÇÃO:
Cistos aracnoides; cisterna quadrigeminal; ressonância magnética; tomografia computadorizada; caninos; convulsões; ataxia cerebelar; hidrocefalia; encefalopatias; patologia
Introduction
Arachnoid cysts are characterized by focal division of the leptomeninge which forms a bag or diverticulum within an arachnoid membrane (Rengachary & Watanabe 1981Rengachary S.S. & Watanabe I. 1981. Ultrastructure and pathogenesis of intracranial arachnoid cysts. J. Neuropathol. Exp. Neurol. 40(1):61-83. PMid:7205328. http://dx.doi.org/10.1097/00005072-198101000-00007.
https://doi.org/10.1097/00005072-1981010...
, Galassi et al. 1985Galassi E., Tognetti F., Frank F., Fagioli L., Nasi M.T. & Gaist G. 1985. Infratentorial arachnoid cysts. J. Neurosurg. 63(2):210-217. PMid:4020444. http://dx.doi.org/10.3171/jns.1985.63.2.0210.
https://doi.org/10.3171/jns.1985.63.2.02...
, Osborn 1994Osborn A.G. 1994. Miscellaneous tumors, cysts, and metastases, p.639-642. In: Ibid. (Ed.), Diagnostic Neuroradiology. Mosby, St Louis.). They can be classified as primary, which are congenital (Dei-Anang & Voth 1989Dei-Anang K. & Voth D. 1989. Cerebral arachnoid cyst: a lesion of the child’s brain. Neurosurg. Rev. 12(1):59-62. PMid:2747935. http://dx.doi.org/10.1007/BF01787131.
https://doi.org/10.1007/BF01787131....
, Matiasek et al. 2007Matiasek L.A., Platt S.R. , Shaw S. & Dennis R. 2007. Clinical and magnetic resonance imaging characteristics of quadrigeminal cysts in dogs. J Vet Intern Med 21(5):1021-1026. PMid:17939559.), that have active secretion of cerebrospinal fluid (CSF) through either the ectopic choroid plexus (Dei-Anang & Voth 1989Dei-Anang K. & Voth D. 1989. Cerebral arachnoid cyst: a lesion of the child’s brain. Neurosurg. Rev. 12(1):59-62. PMid:2747935. http://dx.doi.org/10.1007/BF01787131.
https://doi.org/10.1007/BF01787131....
, Osborn 1994Osborn A.G. 1994. Miscellaneous tumors, cysts, and metastases, p.639-642. In: Ibid. (Ed.), Diagnostic Neuroradiology. Mosby, St Louis.), or of the ependymal cells (Osborn 1994Osborn A.G. 1994. Miscellaneous tumors, cysts, and metastases, p.639-642. In: Ibid. (Ed.), Diagnostic Neuroradiology. Mosby, St Louis.); or as secondary, resulting from brain trauma (Rengachary & Watanabe 1981Rengachary S.S. & Watanabe I. 1981. Ultrastructure and pathogenesis of intracranial arachnoid cysts. J. Neuropathol. Exp. Neurol. 40(1):61-83. PMid:7205328. http://dx.doi.org/10.1097/00005072-198101000-00007.
https://doi.org/10.1097/00005072-1981010...
), intracranial infection and hemorrhage of the subarachnoid space (Di Rocco 1996Di Rocco C. 1996. Arachnoid cysts. p.967-994. In: Youmans J. (Ed.), Neurological Surgery. Vol.2. 4th ed. W.B. Saunders, Philadelphia.). They may also be defined as communicating or non-communicating depending on the relationship with the subarachnoid space (Rengachary & Watanabe 1981Rengachary S.S. & Watanabe I. 1981. Ultrastructure and pathogenesis of intracranial arachnoid cysts. J. Neuropathol. Exp. Neurol. 40(1):61-83. PMid:7205328. http://dx.doi.org/10.1097/00005072-198101000-00007.
https://doi.org/10.1097/00005072-1981010...
, Osborn 1994Osborn A.G. 1994. Miscellaneous tumors, cysts, and metastases, p.639-642. In: Ibid. (Ed.), Diagnostic Neuroradiology. Mosby, St Louis.).
A diagnosis of the condition can be obtained by computed tomography (CT) (Osborn 1994Osborn A.G. 1994. Miscellaneous tumors, cysts, and metastases, p.639-642. In: Ibid. (Ed.), Diagnostic Neuroradiology. Mosby, St Louis., Dewey et al. 2007Dewey C.W., Krotscheck U., Bailey K.S. & Marino D.J. 2007. Craniotomy with cystoperitoneal shunting for treatment of intracranial arachnoid cysts in dogs. Vet. Surg. 36(5):416-422. PMid:17614922. http://dx.doi.org/10.1111/j.1532-950X.2007.00287.x.
https://doi.org/10.1111/j.1532-950X.2007...
), magnetic resonance imaging (MRI) (Osborn 1994Osborn A.G. 1994. Miscellaneous tumors, cysts, and metastases, p.639-642. In: Ibid. (Ed.), Diagnostic Neuroradiology. Mosby, St Louis., Duque et al. 2005Duque C., Parent J., Brisson B., Costa R. & Poma R. 2005. Intracranial arachnoid cysts: are they clinically significant? J. Vet. Intern. Med. 19(5):772-774. PMid:16231727. http://dx.doi.org/10.1111/j.1939-1676.2005.tb02761.x.
https://doi.org/10.1111/j.1939-1676.2005...
, Dewey et al. 2007Dewey C.W., Krotscheck U., Bailey K.S. & Marino D.J. 2007. Craniotomy with cystoperitoneal shunting for treatment of intracranial arachnoid cysts in dogs. Vet. Surg. 36(5):416-422. PMid:17614922. http://dx.doi.org/10.1111/j.1532-950X.2007.00287.x.
https://doi.org/10.1111/j.1532-950X.2007...
, Matiasek et al. 2007Matiasek L.A., Platt S.R. , Shaw S. & Dennis R. 2007. Clinical and magnetic resonance imaging characteristics of quadrigeminal cysts in dogs. J Vet Intern Med 21(5):1021-1026. PMid:17939559.) and ultrasonography (Saito et al. 2001Saito M., Olby N.J. & Spaulding K. 2001. Identification of arachnoid cysts in the quadrigeminal cistern using ultrasonography. Vet. Radiol. Ultrasound 42(5):435-439. PMid:11678566. http://dx.doi.org/10.1111/j.1740-8261.2001.tb00966.x.
https://doi.org/10.1111/j.1740-8261.2001...
).
On MR images, the quadrigeminal arachnoid cysts present themselves as well-demarcated, extra-axial, cystic-appearing structures located between the occipital lobe of the cerebrum and rostral lobe of the cerebellum (Dewey et al. 2009Dewey C.W., Scrivani P.V., Krotscheck U., Cerda-Gonzalez S., Smith Bailey K. & Marino D.J. 2009. Intracranial arachnoid cysts in dogs. Compend. Contin. Educ. Vet. 31(4):160-167, quiz 168. PMid:19517408.) that displace or deform the adjacent structures (Osborn 1994Osborn A.G. 1994. Miscellaneous tumors, cysts, and metastases, p.639-642. In: Ibid. (Ed.), Diagnostic Neuroradiology. Mosby, St Louis.) and are isointense to CSF in all sequences (Osborn 1994Osborn A.G. 1994. Miscellaneous tumors, cysts, and metastases, p.639-642. In: Ibid. (Ed.), Diagnostic Neuroradiology. Mosby, St Louis., Matiasek et al. 2007Matiasek L.A., Platt S.R. , Shaw S. & Dennis R. 2007. Clinical and magnetic resonance imaging characteristics of quadrigeminal cysts in dogs. J Vet Intern Med 21(5):1021-1026. PMid:17939559.). These cysts do not enhance with intravenous gadolinium administration (Galassi et al. 1985Galassi E., Tognetti F., Frank F., Fagioli L., Nasi M.T. & Gaist G. 1985. Infratentorial arachnoid cysts. J. Neurosurg. 63(2):210-217. PMid:4020444. http://dx.doi.org/10.3171/jns.1985.63.2.0210.
https://doi.org/10.3171/jns.1985.63.2.02...
, Osborn 1994Osborn A.G. 1994. Miscellaneous tumors, cysts, and metastases, p.639-642. In: Ibid. (Ed.), Diagnostic Neuroradiology. Mosby, St Louis., Vernau et al. 2002Vernau K.M., LeCouteur R.A., Sturges B.K., Samii V., Higgins R.J., Koblik P.D. & Vernau W. 2002. Intracranial intra-arachnoid cyst with intracystic hemorrhage in two dogs. Vet. Radiol. Ultrasound 43(5):449-454. PMid:12375779. http://dx.doi.org/10.1111/j.1740-8261.2002.tb01032.x.
https://doi.org/10.1111/j.1740-8261.2002...
, Matiasek et al. 2007Matiasek L.A., Platt S.R. , Shaw S. & Dennis R. 2007. Clinical and magnetic resonance imaging characteristics of quadrigeminal cysts in dogs. J Vet Intern Med 21(5):1021-1026. PMid:17939559.), except if they have associated meningoencephalitis (Vernau et al. 2002Vernau K.M., LeCouteur R.A., Sturges B.K., Samii V., Higgins R.J., Koblik P.D. & Vernau W. 2002. Intracranial intra-arachnoid cyst with intracystic hemorrhage in two dogs. Vet. Radiol. Ultrasound 43(5):449-454. PMid:12375779. http://dx.doi.org/10.1111/j.1740-8261.2002.tb01032.x.
https://doi.org/10.1111/j.1740-8261.2002...
, Kitagawa et al. 2003Kitagawa M., Kanayama K. & Sakai T. 2003. Quadrigeminal cistern arachnoid cyst diagnosed by MRI in five dogs. Aust. Vet. J. 81(6):340-343. PMid:15080455. http://dx.doi.org/10.1111/j.1751-0813.2003.tb11509.x.
https://doi.org/10.1111/j.1751-0813.2003...
, Duque et al. 2005Duque C., Parent J., Brisson B., Costa R. & Poma R. 2005. Intracranial arachnoid cysts: are they clinically significant? J. Vet. Intern. Med. 19(5):772-774. PMid:16231727. http://dx.doi.org/10.1111/j.1939-1676.2005.tb02761.x.
https://doi.org/10.1111/j.1939-1676.2005...
). Occasionally, hemorrhage or high protein content may impair the appearance of an arachnoid cyst on MRI. On CT images the cysts also have features of a sharply defined, extra-axial, non-calcified and non-enhancing lesion. Unless that have present hemorrhage, most of the arachnoid cysts have similar attenuation to CSF (Osborn 1994Osborn A.G. 1994. Miscellaneous tumors, cysts, and metastases, p.639-642. In: Ibid. (Ed.), Diagnostic Neuroradiology. Mosby, St Louis.).
Matiasek et al. (2007)Matiasek L.A., Platt S.R. , Shaw S. & Dennis R. 2007. Clinical and magnetic resonance imaging characteristics of quadrigeminal cysts in dogs. J Vet Intern Med 21(5):1021-1026. PMid:17939559. observed three different phenotypes of quadrigeminal cysts in imaging examination: in association with the 3rd ventricle, in association with the 4th ventricle, and a loculated phenotype with relation to the 3rd and 4th ventricle.
The treatment can be performed with medical management with anticonvulsant drugs and corticosteroids (Vernau et al. 1997Vernau K.M., Kortz G.D., Koblik P.D., LeCouteur R.A., Bailey C.S. & Pedroia V. 1997. Magnetic resonance imaging and computed tomography characteristics of intracranial intra-arachnoid cysts in 6 dogs. Vet. Radiol. Ultrasound 38(3):171-176. PMid:9238786. http://dx.doi.org/10.1111/j.1740-8261.1997.tb00835.x.
https://doi.org/10.1111/j.1740-8261.1997...
, Duque et al. 2005Duque C., Parent J., Brisson B., Costa R. & Poma R. 2005. Intracranial arachnoid cysts: are they clinically significant? J. Vet. Intern. Med. 19(5):772-774. PMid:16231727. http://dx.doi.org/10.1111/j.1939-1676.2005.tb02761.x.
https://doi.org/10.1111/j.1939-1676.2005...
) or with surgical treatment such as fenestration (Vernau et al. 1997Vernau K.M., Kortz G.D., Koblik P.D., LeCouteur R.A., Bailey C.S. & Pedroia V. 1997. Magnetic resonance imaging and computed tomography characteristics of intracranial intra-arachnoid cysts in 6 dogs. Vet. Radiol. Ultrasound 38(3):171-176. PMid:9238786. http://dx.doi.org/10.1111/j.1740-8261.1997.tb00835.x.
https://doi.org/10.1111/j.1740-8261.1997...
) and cystoperitoneal shunting (Saito et al. 2001Saito M., Olby N.J. & Spaulding K. 2001. Identification of arachnoid cysts in the quadrigeminal cistern using ultrasonography. Vet. Radiol. Ultrasound 42(5):435-439. PMid:11678566. http://dx.doi.org/10.1111/j.1740-8261.2001.tb00966.x.
https://doi.org/10.1111/j.1740-8261.2001...
, Dewey et al. 2007Dewey C.W., Krotscheck U., Bailey K.S. & Marino D.J. 2007. Craniotomy with cystoperitoneal shunting for treatment of intracranial arachnoid cysts in dogs. Vet. Surg. 36(5):416-422. PMid:17614922. http://dx.doi.org/10.1111/j.1532-950X.2007.00287.x.
https://doi.org/10.1111/j.1532-950X.2007...
).
There are controversies about the clinical significance of arachnoid cyst in dogs and humans and they are considered in many cases to be incidental findings of imaging or post-mortem findings (Vernau et al. 1997Vernau K.M., Kortz G.D., Koblik P.D., LeCouteur R.A., Bailey C.S. & Pedroia V. 1997. Magnetic resonance imaging and computed tomography characteristics of intracranial intra-arachnoid cysts in 6 dogs. Vet. Radiol. Ultrasound 38(3):171-176. PMid:9238786. http://dx.doi.org/10.1111/j.1740-8261.1997.tb00835.x.
https://doi.org/10.1111/j.1740-8261.1997...
, Saito et al. 2001Saito M., Olby N.J. & Spaulding K. 2001. Identification of arachnoid cysts in the quadrigeminal cistern using ultrasonography. Vet. Radiol. Ultrasound 42(5):435-439. PMid:11678566. http://dx.doi.org/10.1111/j.1740-8261.2001.tb00966.x.
https://doi.org/10.1111/j.1740-8261.2001...
, Kitagawa et al. 2003Kitagawa M., Kanayama K. & Sakai T. 2003. Quadrigeminal cistern arachnoid cyst diagnosed by MRI in five dogs. Aust. Vet. J. 81(6):340-343. PMid:15080455. http://dx.doi.org/10.1111/j.1751-0813.2003.tb11509.x.
https://doi.org/10.1111/j.1751-0813.2003...
, Duque et al. 2005Duque C., Parent J., Brisson B., Costa R. & Poma R. 2005. Intracranial arachnoid cysts: are they clinically significant? J. Vet. Intern. Med. 19(5):772-774. PMid:16231727. http://dx.doi.org/10.1111/j.1939-1676.2005.tb02761.x.
https://doi.org/10.1111/j.1939-1676.2005...
, Matiasek et al. 2007Matiasek L.A., Platt S.R. , Shaw S. & Dennis R. 2007. Clinical and magnetic resonance imaging characteristics of quadrigeminal cysts in dogs. J Vet Intern Med 21(5):1021-1026. PMid:17939559.).
The aim of this retrospective study is to report the occurrence of the quadrigeminal arachnoid cysts in 26 dogs, as well to describe the imaging findings by computed tomography and magnetic resonance image scans and to correlate these with the clinical signs.
Materials and Methods
Records of head MRI and CT scans of dogs that were admitted to the Veterinary Neurology Service between February 2007 and July 2015 were reviewed by a radiologist resident and a professor. MRIs were performed with a 0.25 Tesla scanner (Vet-MR Grande, Esaote, Italy), including T1, T2, T2* and fluid-attenuation inversion recovery (FLAIR) sequences in the sagittal, transverse and dorsal planes. CTs were performed with helical equipment SCT-7800TC (Shimadzu; Kyoto, Japan) in the transverse plane, with most of the scans performed in sagittal and dorsal multiplanar reconstruction (MPR).
Images were evaluated using a medical digital imaging system (PACS, Synapse, Fuji Medical System; Tokyo, Japan). The degree of the cerebral and cerebellar compression was calculated from the mid-sagittal planes in 16/17 MRI and 5/9 CT scans as proposed in a previous study by Matiasek et al. (2007)Matiasek L.A., Platt S.R. , Shaw S. & Dennis R. 2007. Clinical and magnetic resonance imaging characteristics of quadrigeminal cysts in dogs. J Vet Intern Med 21(5):1021-1026. PMid:17939559..
The diagnosis of quadrigeminal arachnoid cyst was based on the following inclusion criteria: location of cystic-appearing structure between the occipital lobe and cerebellum, fluid-content intensity/attenuation similar to cerebrospinal fluid (CSF), and absence of enhancement after the intravenous contrast-medium administration (0.02ml/kg of gadoversetamide[Optimark®, Mallinckrodt Inc., Raleigh, NC, USA] to MRI; 0.2ml/kg of iopamidol [Iopamiron®300, Patheon Italia S.p.A., Ferentino, Frosinore, Italy] to CT).
Additional information was obtained from the medical records as well as signalment, presenting complaint, neurologic evaluation and blood profile (CBC and serum biochemistry). Cerebrospinal fluid (CSF) analysis was performed in seven dogs. The CSF protein was considered increased if greater than 25mg/dL (cerebellomedullary cistern). It was considered as mild (<50 mg/dL), moderate (<50-200 mg/dl) or severe (>200mg/dL) (Bohn et al., 2006Bohn A.A., Wills T.B., West C.L., Tucker R.L. & Bagley R.S. 2006. Cerebrospinal fluid analysis and magnetic resonance imaging in the diagnosis of neurologic disease in dogs: a retrospective study. Vet. Clin. Pathol. 35(3):315-320. PMid:16967416. http://dx.doi.org/10.1111/j.1939-165X.2006.tb00138.x.
https://doi.org/10.1111/j.1939-165X.2006...
). The total count of nucleated cells (CTCN) was characterized as pleocytosis when >5 nucleated cells per μL (Terlizzi & Platt, 2006Terlizzi R. & Platt S. 2006. The function, composition and analysis of cerebrospinal fluid in companion animals: Part I - Function and composition. Vet. J. 172(3):422-431. PMid:16154365. http://dx.doi.org/10.1016/j.tvjl.2005.07.021.
https://doi.org/10.1016/j.tvjl.2005.07.0...
). It was classified as primary, if the cell type comprised >85%, and mixed if none of the cell types comprised at least 85% of the nucleated cells. It was considered mild (<25 cells/μL), moderate (26-100 cells/μL), or marked (>100 cells/μL) and was further defined by the predominant cell type in the sample the neutrophilic, eosinophilic, mononuclear or mixed (Freeman & Raskin, 2001Freeman R.A. & Raskin R.E. 2001. Cytology of the central nervous system, p.967-994. In: Raskin R.E. & Meyer D.J. (Eds), Atlas of Canine and Feline Cytology. W.B. Saunders, Philadelphia.).
Results
Altogether, 650 records of canine and feline imaging brain scans were reviewed - 148 MRI images and 502 CT images. Twenty-six dogs were diagnosed with quadrigeminal arachnoid cyst, all of them small breeds weighing less than 10 kg (mean 4.5; SD = 2.83). Breed population consisted of 7 Miniature Pinschers, 7 Shih Tzus, 4 Lhasa Apsos, 2 Poodles, 2 mixed breed and 1 each of Yorkshire Terrier, Miniature Schnauzer, Boston Terrier, and Brazilian Terrier. Fifteen dogs were male and 11 dogs were female. The age at diagnosis ranged from 2 months to 9 years, with a mean of 3.1 years (SD = 2.43).
CBC and serum biochemistry did not reveal significant changes in any of the patients. CSF analysis of two dogs presented changes consistent with inflammatory brain disease showing lymphocytic pleocytosis (case 15) and neutrophilic pleocytosis (case 6). Five dogs presented with normal CSF analysis parameters (cases 10, 12, 14, 16). One CSF analysis could not be completed due to insufficient sample (case 1) that showed increased protein concentration with 81mg/dL (reference range, 0-30mg/dL) (Table 1).
The neurologic exams recorded revealed several clinical signs including generalized tonic-clonic seizures (13), focal seizures (1), cerebellar ataxia (8), hypermetria (3), wide-base stance (2), intention tremor (3), circling (7), compulsive walking (5), head pressing (2), deficit of menace response (8), behavioral changes (7), mental depression (2), conscious proprioceptive deficit (7), proprioceptive ataxia (5), paresis (3), head tilting (4), positional strabismus (1), pleurothotonus (1), parestesis (1), and neck pain (1) (Table 1).
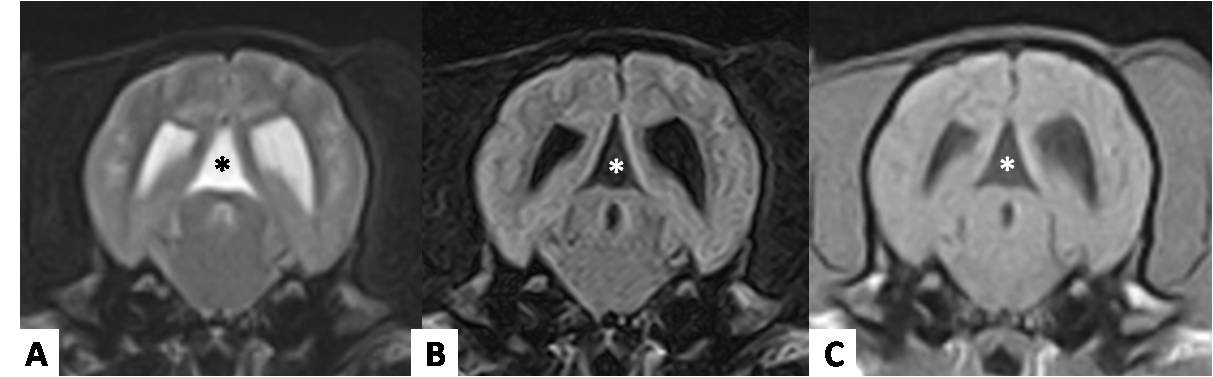
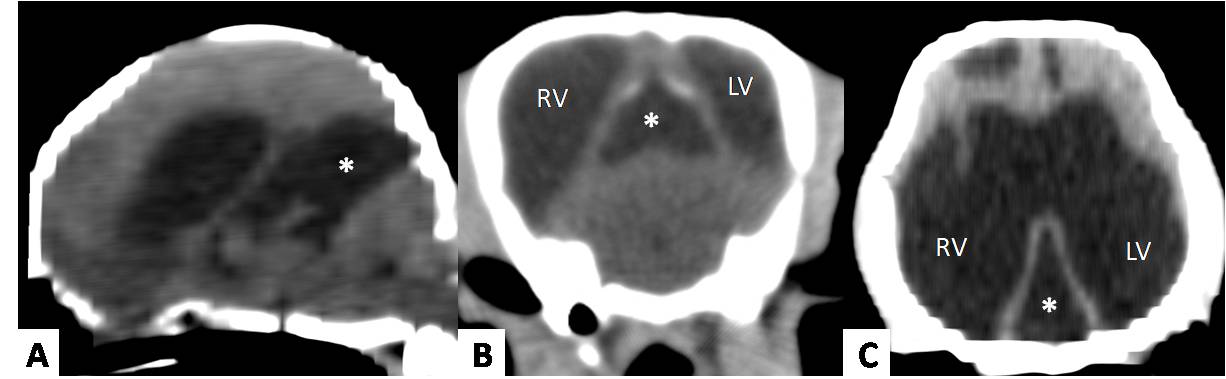
MRI showed a well-demarcated, extra-axial, cystic-appearing structure in various sizes located between the ventrocaudal border of the occipital lobes and the dorso-rostral border of the cerebellum, hypointense on T1, hyperintense on T2, and suppression on FLAIR images. The signal intensity of the cyst content was similar to CSF and non-contrast enhancing with gadolinium administration. These structures were observed on transverse planes in a triangular-like shape between the occipital lobes and the cerebellum (Fig.1). On CT images the structure showed fluid attenuation similar to CSF and no enhancement after contrast-medium administration (Fig.2).
(A) Transverse MRI of a 3-year-old female Boston Terrier with 9.6 kg presenting seizures weekly, agitation and behavior abnormal. Note the triangular-like shape of the structure (*) between occipital lobes showing the same signal intensity than CSF on T2-weighted, and (B) FLAIR with no contrast enhancement showed on T1-weighted (C).
(A) CT images of a 2-year-old male Pinscher Miniature with 1.4 kg presenting ataxia and circling to the right. Note the cyst-like structure (*) of fluid attenuation between occipital lobes and cerebellum extending from the third ventricle on the sagittal plane. (B) This cyst-like structure (*) can be seen between occipital lobes in a triangular-like shape on transverse, and (C) dorsal planes. This dog had severe hydrocephalus. LV = left ventricle; RV = right ventricle.
Other brain abnormalities were observed such as hydrocephaly, of which variations were observed ranging from discrete to severe in 20 dogs, with three obstructive hydrocephalus (cases 1, 5, 9) and two hydrocephalus ex vacuo (cases 6, 7). Other changes were consistent with inflammatory brain disease in five dogs, including meningoencephalitis (cases 2, 6), cortical (cases 8, 15) and interstitial edema (case 7), anomalous/congenital disease in 15 dogs, including hydrocephalus (cases 4, 5, 9-14, 17-22, 24, 26), syringohydromyelia (cases 5, 9), agenesis of the corpus callosum and lissencephaly (case 17), atlanto-occipital malformation and branchial cysts (case 4), degenerative disease, including cerebellar atrophy (case 25), and neoplasia in a dog with a tumor on lateral cerebellar pontine angle involving the fourth ventricle (case 1) (Table 2). Only three CT and MRI scans did not present abnormalities beyond the quadrigeminal cyst (cases 3, 16, 23).
MRI and CT findings, percentual of forebrain and cerebellar compression and phenotype of quadrigeminal arachnoid cyst in 26 dogs
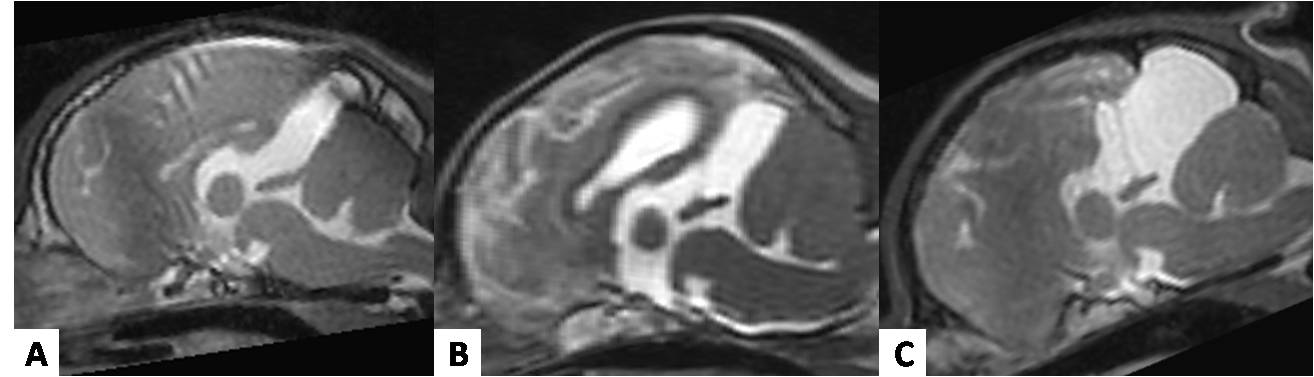
Three phenotypes of the quadrigeminal arachnoid cyst were easily distinguished on MRI and CT images of this study, including on CT transverse planes for which it was not possible to perform MPR mid-sagittal images (Fig.3). A predominance of phenotype was noted in association with the fourth ventricle (11 images) followed by association with the third ventricle (9 images) and loculated with the third and fourth ventricles (6 images). Four loculated phenotypes presented with equal relation to the third and forth ventricle, while the other two presented with phenotype loculated with a higher relation to the fourth ventricle than to the third ventricle.
Mid-sagittal T2-weighted MRI showing the 3 different phenotypes of quadrigeminal arachnoid cyst: (A) in association with the 3rd ventricle, (B) in association with the 4th ventricle, and (C) loculated phenotype to the 3rd and 4th ventricle.
The degree of cerebral and cerebellar compression was calculated from the mid-sagittal planes in 16/17 MRI scans (Fig.4 A and B), which obtained ranging from 0-24.2% (mean, 6.5%; SD = 9.2) of occipital lobe compression and from 0-53.3% (mean, 17.9%; SD = 17.8) of cerebellum compression. The same calculation was made in 5/9 CT scans (Fig.4C,D), as in another four CT scans it was not possible to perform the MPR in sagittal planes. The degree of compression of the occipital lobes ranged from 0-16.9% (mean, 9.4%; SD = 8.6) and the compression of the cerebellum ranged from 0-55.7% (mean, 26.8%; SD = 25.5%). Altogether, 21 CT and MRI scans were measured obtaining a mean of 7.2% (SD = 8.9) of occipital lobes compression and a mean of 20.0% (SD = 19.6) of cerebellum compression.
(A,B) Measurement of the compression degree of the cerebral and cerebellar parenchyma by mid-sagittal T2-weighted MRI, and (C,D) mid-sagittal CT images. The black line of (A) and (C) represents the expected diameter of the cerebellum and the measured length is set at 100%; the white line represents the actual diameter of the cerebellum. The black line of (B) and (D) is drawn for the expected longitudinal dimension of the forebrain and the measured length is set at 100%; the white line represents the actual longitudinal dimension of the forebrain.
Discussion
There are controversies regarding the quadrigeminal arachnoid cyst in literature about its clinical significance. There have been around 57 reported cases of intracranial arachnoid cyst in dogs (Vernau et al. 1997Vernau K.M., Kortz G.D., Koblik P.D., LeCouteur R.A., Bailey C.S. & Pedroia V. 1997. Magnetic resonance imaging and computed tomography characteristics of intracranial intra-arachnoid cysts in 6 dogs. Vet. Radiol. Ultrasound 38(3):171-176. PMid:9238786. http://dx.doi.org/10.1111/j.1740-8261.1997.tb00835.x.
https://doi.org/10.1111/j.1740-8261.1997...
, Koie et al. 2000Koie H., Kitagawa M., Kuwabara M. & Sakai T. 2000. Pineal arachnoid cyst demonstrated with magnetic resonance imaging. Canine Pract. 25:14-15., Saito et al. 2001Saito M., Olby N.J. & Spaulding K. 2001. Identification of arachnoid cysts in the quadrigeminal cistern using ultrasonography. Vet. Radiol. Ultrasound 42(5):435-439. PMid:11678566. http://dx.doi.org/10.1111/j.1740-8261.2001.tb00966.x.
https://doi.org/10.1111/j.1740-8261.2001...
, Vernau et al. 2002Vernau K.M., LeCouteur R.A., Sturges B.K., Samii V., Higgins R.J., Koblik P.D. & Vernau W. 2002. Intracranial intra-arachnoid cyst with intracystic hemorrhage in two dogs. Vet. Radiol. Ultrasound 43(5):449-454. PMid:12375779. http://dx.doi.org/10.1111/j.1740-8261.2002.tb01032.x.
https://doi.org/10.1111/j.1740-8261.2002...
, Kitagawa et al. 2003Kitagawa M., Kanayama K. & Sakai T. 2003. Quadrigeminal cistern arachnoid cyst diagnosed by MRI in five dogs. Aust. Vet. J. 81(6):340-343. PMid:15080455. http://dx.doi.org/10.1111/j.1751-0813.2003.tb11509.x.
https://doi.org/10.1111/j.1751-0813.2003...
, Duque et al. 2005Duque C., Parent J., Brisson B., Costa R. & Poma R. 2005. Intracranial arachnoid cysts: are they clinically significant? J. Vet. Intern. Med. 19(5):772-774. PMid:16231727. http://dx.doi.org/10.1111/j.1939-1676.2005.tb02761.x.
https://doi.org/10.1111/j.1939-1676.2005...
, Dewey et al. 2007Dewey C.W., Krotscheck U., Bailey K.S. & Marino D.J. 2007. Craniotomy with cystoperitoneal shunting for treatment of intracranial arachnoid cysts in dogs. Vet. Surg. 36(5):416-422. PMid:17614922. http://dx.doi.org/10.1111/j.1532-950X.2007.00287.x.
https://doi.org/10.1111/j.1532-950X.2007...
, Matiasek et al. 2007Matiasek L.A., Platt S.R. , Shaw S. & Dennis R. 2007. Clinical and magnetic resonance imaging characteristics of quadrigeminal cysts in dogs. J Vet Intern Med 21(5):1021-1026. PMid:17939559., Reed et al. 2009Reed S., Cho C.Y. & Paulsen D. 2009. Quadrigeminal arachnoid cysts in a kitten and a dog. J. Vet. Diagn. Invest. 21(5):707-710. PMid:19737770. http://dx.doi.org/10.1177/104063870902100519.
https://doi.org/10.1177/1040638709021005...
, Bernardino et al. 2015Bernardino F., Rentmeister K., Schmidt M.J., Bruehschwein A., Matiasek K., Matiasek L.A., Lauda A., Schoon H.A. & Fischer A. 2015. Inferior cerebellar hypoplasia resembling a Dandy-Walker-like malformation in purebred eurasier dogs with familial non-progressive ataxia: a retrospective and prospective clinical cohort study. PLoS One 10(2):e0117670. PMid:25668516. http://dx.doi.org/10.1371/journal.pone.0117670.
https://doi.org/10.1371/journal.pone.011...
) and more than half of these cases were deemed to be an incidental finding.
In humans, the incidence of intracranial arachnoid cysts is 1% of all non-traumatic mass lesions (Robertson et al. 1989Robertson S.J., Wolpert S.M. & Runge V.M. 1989. MR imaging of middle cranial fossa arachnoid cysts: temporal lobe agenesis syndrome revisited. AJNR Am. J. Neuroradiol. 10(5):1007-1010. PMid:2505512.), with 50-60% located in the middle fossa (Rengachary & Watanabe 1981Rengachary S.S. & Watanabe I. 1981. Ultrastructure and pathogenesis of intracranial arachnoid cysts. J. Neuropathol. Exp. Neurol. 40(1):61-83. PMid:7205328. http://dx.doi.org/10.1097/00005072-198101000-00007.
https://doi.org/10.1097/00005072-1981010...
, Robertson et al. 1989Robertson S.J., Wolpert S.M. & Runge V.M. 1989. MR imaging of middle cranial fossa arachnoid cysts: temporal lobe agenesis syndrome revisited. AJNR Am. J. Neuroradiol. 10(5):1007-1010. PMid:2505512., Yamakawa et al. 1991Yamakawa H., Ohkuma A., Hattori T., Niikawa S. & Kobayashi H. 1991. Primary intracranial arachnoid cyst in the elderly: a survey on 39 cases. Acta Neurochir. 113(1-2):42-47. PMid: 1799142. http://dx.doi.org/10.1007/BF01402113.
https://doi.org/10.1007/BF01402113....
, Wang et al. 1998Wang P.-J., Lin H.-C., Liu H.-M., Tseng C.-L. & Shen Y.-Z. 1998. Intracranial arachnoid cysts in children: related signs and associated anomalies. Pediatr. Neurol. 19(2):100-104. PMid:9744627. http://dx.doi.org/10.1016/S0887-8994(98)00020-4.
https://doi.org/10.1016/S0887-8994(98)00...
), mainly unilateral (Wang et al. 1998Wang P.-J., Lin H.-C., Liu H.-M., Tseng C.-L. & Shen Y.-Z. 1998. Intracranial arachnoid cysts in children: related signs and associated anomalies. Pediatr. Neurol. 19(2):100-104. PMid:9744627. http://dx.doi.org/10.1016/S0887-8994(98)00020-4.
https://doi.org/10.1016/S0887-8994(98)00...
), and 10% in the posterior fossa (Robertson et al. 1989Robertson S.J., Wolpert S.M. & Runge V.M. 1989. MR imaging of middle cranial fossa arachnoid cysts: temporal lobe agenesis syndrome revisited. AJNR Am. J. Neuroradiol. 10(5):1007-1010. PMid:2505512.).
The prevalence of this condition is low in dogs, representing about 0.7% of intracranial lesions (Matiasek et al. 2007Matiasek L.A., Platt S.R. , Shaw S. & Dennis R. 2007. Clinical and magnetic resonance imaging characteristics of quadrigeminal cysts in dogs. J Vet Intern Med 21(5):1021-1026. PMid:17939559.). Nevertheless, in the present study this percentage presented higher with 4% of total brain examinations evaluated. All arachnoid cysts presented on quadrigeminal cistern, as well as in most of the case reports in the literature (Vernau et al. 1997Vernau K.M., Kortz G.D., Koblik P.D., LeCouteur R.A., Bailey C.S. & Pedroia V. 1997. Magnetic resonance imaging and computed tomography characteristics of intracranial intra-arachnoid cysts in 6 dogs. Vet. Radiol. Ultrasound 38(3):171-176. PMid:9238786. http://dx.doi.org/10.1111/j.1740-8261.1997.tb00835.x.
https://doi.org/10.1111/j.1740-8261.1997...
, 2002Vernau K.M., LeCouteur R.A., Sturges B.K., Samii V., Higgins R.J., Koblik P.D. & Vernau W. 2002. Intracranial intra-arachnoid cyst with intracystic hemorrhage in two dogs. Vet. Radiol. Ultrasound 43(5):449-454. PMid:12375779. http://dx.doi.org/10.1111/j.1740-8261.2002.tb01032.x.
https://doi.org/10.1111/j.1740-8261.2002...
, Saito et al. 2001Saito M., Olby N.J. & Spaulding K. 2001. Identification of arachnoid cysts in the quadrigeminal cistern using ultrasonography. Vet. Radiol. Ultrasound 42(5):435-439. PMid:11678566. http://dx.doi.org/10.1111/j.1740-8261.2001.tb00966.x.
https://doi.org/10.1111/j.1740-8261.2001...
, Kitagawa et al. 2003Kitagawa M., Kanayama K. & Sakai T. 2003. Quadrigeminal cistern arachnoid cyst diagnosed by MRI in five dogs. Aust. Vet. J. 81(6):340-343. PMid:15080455. http://dx.doi.org/10.1111/j.1751-0813.2003.tb11509.x.
https://doi.org/10.1111/j.1751-0813.2003...
, Duque et al. 2005Duque C., Parent J., Brisson B., Costa R. & Poma R. 2005. Intracranial arachnoid cysts: are they clinically significant? J. Vet. Intern. Med. 19(5):772-774. PMid:16231727. http://dx.doi.org/10.1111/j.1939-1676.2005.tb02761.x.
https://doi.org/10.1111/j.1939-1676.2005...
, Dewey et al. 2007Dewey C.W., Krotscheck U., Bailey K.S. & Marino D.J. 2007. Craniotomy with cystoperitoneal shunting for treatment of intracranial arachnoid cysts in dogs. Vet. Surg. 36(5):416-422. PMid:17614922. http://dx.doi.org/10.1111/j.1532-950X.2007.00287.x.
https://doi.org/10.1111/j.1532-950X.2007...
, Matiasek et al. 2007Matiasek L.A., Platt S.R. , Shaw S. & Dennis R. 2007. Clinical and magnetic resonance imaging characteristics of quadrigeminal cysts in dogs. J Vet Intern Med 21(5):1021-1026. PMid:17939559.). Only two cases report the arachnoid cyst occurring at the cerebellomedullary angle (Vernau et al. 1997Vernau K.M., Kortz G.D., Koblik P.D., LeCouteur R.A., Bailey C.S. & Pedroia V. 1997. Magnetic resonance imaging and computed tomography characteristics of intracranial intra-arachnoid cysts in 6 dogs. Vet. Radiol. Ultrasound 38(3):171-176. PMid:9238786. http://dx.doi.org/10.1111/j.1740-8261.1997.tb00835.x.
https://doi.org/10.1111/j.1740-8261.1997...
) and in the pineal region (Koie et al. 2000Koie H., Kitagawa M., Kuwabara M. & Sakai T. 2000. Pineal arachnoid cyst demonstrated with magnetic resonance imaging. Canine Pract. 25:14-15.). There are only three reports of Persian cats presenting intracranial arachnoid cyst located in caudal fossa at level of the quadrigeminal cistern (Lowrie et al. 2009Lowrie M., Wessmann A., Gunn-Moore D. & Penderis J. 2009. Quadrigeminal cyst management by cystoperitoneal shunt in a 4-year-old Persian cat. J. Feline Med. Surg. 11(8):711-713. PMid:19261501. http://dx.doi.org/10.1016/j.jfms.2009.01.007.
https://doi.org/10.1016/j.jfms.2009.01.0...
, Reed et al. 2009Reed S., Cho C.Y. & Paulsen D. 2009. Quadrigeminal arachnoid cysts in a kitten and a dog. J. Vet. Diagn. Invest. 21(5):707-710. PMid:19737770. http://dx.doi.org/10.1177/104063870902100519.
https://doi.org/10.1177/1040638709021005...
) and cerebellar pontine (Milner et al. 1996Milner R.J., Engela J. & Kirberger R.M. 1996. Arachnoid cyst in cerebellar pontine area of a cat diagnosis by magnetic resonance imaging. Vet. Radiol. Ultrasound 37(1):34-36. http://dx.doi.org/10.1111/j.1740-8261.1996.tb00809.x.
https://doi.org/10.1111/j.1740-8261.1996...
), and a report of a European cat with supratentorial arachnoid cyst located mainly beneath the piriform lobe (Taroni et al. 2015Taroni M., Seurin M-J., Carozzo C. & Escriou C. 2015. Supratentorial arachnoid cyst management by cystoperitoneal shunt in a 1-year-old European cat. J. Feline Med. Surg.). However, in our study, neither record of feline imaging brain scans presented this condition. There also exists a report case of a chimpanzee with intracranial arachnoid cyst in the occipital convexity (Miyabe-Nishiwaki et al. 2014Miyabe-Nishiwaki T., Kaneko T., Sakai T., Kaneko A., Watanabe A., Watanabe S., Maeda N., Kumazaki K., Suzuki J., Fujiwara R., Makishima H., Nishimura T., Hayashi M., Tomonaga M., Matsuzawa T. & Mikami A. 2014. Intracranial arachnoid cysts in a chimpanzee (Pan troglodytes). Primates 55(1):7-12. PMid:24068629. http://dx.doi.org/10.1007/s10329-013-0384-0.
https://doi.org/10.1007/s10329-013-0384-...
).
Arachnoid cyst is present in small breed and brachycephalic dogs (Matiasek et al. 2007Matiasek L.A., Platt S.R. , Shaw S. & Dennis R. 2007. Clinical and magnetic resonance imaging characteristics of quadrigeminal cysts in dogs. J Vet Intern Med 21(5):1021-1026. PMid:17939559.), as seen in this study that the affected dogs weighed less than 10 kg. Among the dog breeds previously reported were Shih Tzu, Yorkshire Terrier, Pug, Lhasa Apso, Chihuahua, Bulldog, Staffordshire Bull Terrier, Pekingese, Jack Russel, Cavalier King Charles Spaniel, West Highland White Terrier, Cairn Terrier, Maltese, Pomeranian, Bichon Frisé, Beagle and Miniature Schnauzer (Vernau et al. 1997Vernau K.M., Kortz G.D., Koblik P.D., LeCouteur R.A., Bailey C.S. & Pedroia V. 1997. Magnetic resonance imaging and computed tomography characteristics of intracranial intra-arachnoid cysts in 6 dogs. Vet. Radiol. Ultrasound 38(3):171-176. PMid:9238786. http://dx.doi.org/10.1111/j.1740-8261.1997.tb00835.x.
https://doi.org/10.1111/j.1740-8261.1997...
, 2002Vernau K.M., LeCouteur R.A., Sturges B.K., Samii V., Higgins R.J., Koblik P.D. & Vernau W. 2002. Intracranial intra-arachnoid cyst with intracystic hemorrhage in two dogs. Vet. Radiol. Ultrasound 43(5):449-454. PMid:12375779. http://dx.doi.org/10.1111/j.1740-8261.2002.tb01032.x.
https://doi.org/10.1111/j.1740-8261.2002...
, Kitagawa et al. 2003Kitagawa M., Kanayama K. & Sakai T. 2003. Quadrigeminal cistern arachnoid cyst diagnosed by MRI in five dogs. Aust. Vet. J. 81(6):340-343. PMid:15080455. http://dx.doi.org/10.1111/j.1751-0813.2003.tb11509.x.
https://doi.org/10.1111/j.1751-0813.2003...
, Duque et al. 2005Duque C., Parent J., Brisson B., Costa R. & Poma R. 2005. Intracranial arachnoid cysts: are they clinically significant? J. Vet. Intern. Med. 19(5):772-774. PMid:16231727. http://dx.doi.org/10.1111/j.1939-1676.2005.tb02761.x.
https://doi.org/10.1111/j.1939-1676.2005...
, Dewey et al. 2007Dewey C.W., Krotscheck U., Bailey K.S. & Marino D.J. 2007. Craniotomy with cystoperitoneal shunting for treatment of intracranial arachnoid cysts in dogs. Vet. Surg. 36(5):416-422. PMid:17614922. http://dx.doi.org/10.1111/j.1532-950X.2007.00287.x.
https://doi.org/10.1111/j.1532-950X.2007...
, Matiasek et al. 2007Matiasek L.A., Platt S.R. , Shaw S. & Dennis R. 2007. Clinical and magnetic resonance imaging characteristics of quadrigeminal cysts in dogs. J Vet Intern Med 21(5):1021-1026. PMid:17939559.). However, the breeds most prevalent in this study were Miniature Pinscher and Shih Tzu (both representing 26.9%). Dogs with arachnoid cyst ranged in age from 2 months to 10 years and a greater incidence was found in males (Saito et al. 2001Saito M., Olby N.J. & Spaulding K. 2001. Identification of arachnoid cysts in the quadrigeminal cistern using ultrasonography. Vet. Radiol. Ultrasound 42(5):435-439. PMid:11678566. http://dx.doi.org/10.1111/j.1740-8261.2001.tb00966.x.
https://doi.org/10.1111/j.1740-8261.2001...
, Kitagawa et al. 2003Kitagawa M., Kanayama K. & Sakai T. 2003. Quadrigeminal cistern arachnoid cyst diagnosed by MRI in five dogs. Aust. Vet. J. 81(6):340-343. PMid:15080455. http://dx.doi.org/10.1111/j.1751-0813.2003.tb11509.x.
https://doi.org/10.1111/j.1751-0813.2003...
, Duque et al. 2005Duque C., Parent J., Brisson B., Costa R. & Poma R. 2005. Intracranial arachnoid cysts: are they clinically significant? J. Vet. Intern. Med. 19(5):772-774. PMid:16231727. http://dx.doi.org/10.1111/j.1939-1676.2005.tb02761.x.
https://doi.org/10.1111/j.1939-1676.2005...
, Matiasek et al. 2007Matiasek L.A., Platt S.R. , Shaw S. & Dennis R. 2007. Clinical and magnetic resonance imaging characteristics of quadrigeminal cysts in dogs. J Vet Intern Med 21(5):1021-1026. PMid:17939559., Reed et al. 2009Reed S., Cho C.Y. & Paulsen D. 2009. Quadrigeminal arachnoid cysts in a kitten and a dog. J. Vet. Diagn. Invest. 21(5):707-710. PMid:19737770. http://dx.doi.org/10.1177/104063870902100519.
https://doi.org/10.1177/1040638709021005...
). Similarly, more than half of the population of this retrospective study was male (57.7%), with an age range from 2 months to 9 years.
In humans, the posterior fossa clinical signs consist of headache, seizures, ataxia, vomiting, cerebellar disorders, mild cognitive impairment, and focal neurological signs (Wang et al. 1998Wang P.-J., Lin H.-C., Liu H.-M., Tseng C.-L. & Shen Y.-Z. 1998. Intracranial arachnoid cysts in children: related signs and associated anomalies. Pediatr. Neurol. 19(2):100-104. PMid:9744627. http://dx.doi.org/10.1016/S0887-8994(98)00020-4.
https://doi.org/10.1016/S0887-8994(98)00...
, Hanieh et al. 1988Hanieh A., Simpson D.A. & North J.B. 1988. Arachnoid cysts: a critical review of 41 cases. Childs Nerv. Syst. 4(2):92-96. PMid:3401876., Robertson et al. 1989Robertson S.J., Wolpert S.M. & Runge V.M. 1989. MR imaging of middle cranial fossa arachnoid cysts: temporal lobe agenesis syndrome revisited. AJNR Am. J. Neuroradiol. 10(5):1007-1010. PMid:2505512., Cincu et al. 2007Cincu R., Agrawal A. & Eiras J. 2007. Intracranial arachnoid cysts: current concepts and treatment alternatives. Clin. Neurol. Neurosurg. 109(10):837-843. PMid:17764831. http://dx.doi.org/10.1016/j.clineuro.2007.07.013.
https://doi.org/10.1016/j.clineuro.2007....
). Commonly, the clinical signs were associated with obstructive hydrocephalus (Hanieh et al. 1988Hanieh A., Simpson D.A. & North J.B. 1988. Arachnoid cysts: a critical review of 41 cases. Childs Nerv. Syst. 4(2):92-96. PMid:3401876., Cincu et al. 2007Cincu R., Agrawal A. & Eiras J. 2007. Intracranial arachnoid cysts: current concepts and treatment alternatives. Clin. Neurol. Neurosurg. 109(10):837-843. PMid:17764831. http://dx.doi.org/10.1016/j.clineuro.2007.07.013.
https://doi.org/10.1016/j.clineuro.2007....
). Macrocephaly and psychomotor retardation have been reported in young patients (Galassi et al. 1985Galassi E., Tognetti F., Frank F., Fagioli L., Nasi M.T. & Gaist G. 1985. Infratentorial arachnoid cysts. J. Neurosurg. 63(2):210-217. PMid:4020444. http://dx.doi.org/10.3171/jns.1985.63.2.0210.
https://doi.org/10.3171/jns.1985.63.2.02...
, Wang et al. 1998Wang P.-J., Lin H.-C., Liu H.-M., Tseng C.-L. & Shen Y.-Z. 1998. Intracranial arachnoid cysts in children: related signs and associated anomalies. Pediatr. Neurol. 19(2):100-104. PMid:9744627. http://dx.doi.org/10.1016/S0887-8994(98)00020-4.
https://doi.org/10.1016/S0887-8994(98)00...
). Dementia, ataxia, hemiparesis and gait disturbance have been reported as signs among the elderly (Yamakawa et al. 1991Yamakawa H., Ohkuma A., Hattori T., Niikawa S. & Kobayashi H. 1991. Primary intracranial arachnoid cyst in the elderly: a survey on 39 cases. Acta Neurochir. 113(1-2):42-47. PMid: 1799142. http://dx.doi.org/10.1007/BF01402113.
https://doi.org/10.1007/BF01402113....
).
Clinical signs more often described in animals were seizures, head tilt, ataxia, circling, paresis, dementia, and deficit of menace response (Vernau et al. 1997Vernau K.M., Kortz G.D., Koblik P.D., LeCouteur R.A., Bailey C.S. & Pedroia V. 1997. Magnetic resonance imaging and computed tomography characteristics of intracranial intra-arachnoid cysts in 6 dogs. Vet. Radiol. Ultrasound 38(3):171-176. PMid:9238786. http://dx.doi.org/10.1111/j.1740-8261.1997.tb00835.x.
https://doi.org/10.1111/j.1740-8261.1997...
, 2002Vernau K.M., LeCouteur R.A., Sturges B.K., Samii V., Higgins R.J., Koblik P.D. & Vernau W. 2002. Intracranial intra-arachnoid cyst with intracystic hemorrhage in two dogs. Vet. Radiol. Ultrasound 43(5):449-454. PMid:12375779. http://dx.doi.org/10.1111/j.1740-8261.2002.tb01032.x.
https://doi.org/10.1111/j.1740-8261.2002...
, Kitagawa et al. 2003Kitagawa M., Kanayama K. & Sakai T. 2003. Quadrigeminal cistern arachnoid cyst diagnosed by MRI in five dogs. Aust. Vet. J. 81(6):340-343. PMid:15080455. http://dx.doi.org/10.1111/j.1751-0813.2003.tb11509.x.
https://doi.org/10.1111/j.1751-0813.2003...
, Duque et al. 2005Duque C., Parent J., Brisson B., Costa R. & Poma R. 2005. Intracranial arachnoid cysts: are they clinically significant? J. Vet. Intern. Med. 19(5):772-774. PMid:16231727. http://dx.doi.org/10.1111/j.1939-1676.2005.tb02761.x.
https://doi.org/10.1111/j.1939-1676.2005...
, Saito et al. 2001Saito M., Olby N.J. & Spaulding K. 2001. Identification of arachnoid cysts in the quadrigeminal cistern using ultrasonography. Vet. Radiol. Ultrasound 42(5):435-439. PMid:11678566. http://dx.doi.org/10.1111/j.1740-8261.2001.tb00966.x.
https://doi.org/10.1111/j.1740-8261.2001...
, Reed et al. 2009Reed S., Cho C.Y. & Paulsen D. 2009. Quadrigeminal arachnoid cysts in a kitten and a dog. J. Vet. Diagn. Invest. 21(5):707-710. PMid:19737770. http://dx.doi.org/10.1177/104063870902100519.
https://doi.org/10.1177/1040638709021005...
). Interestingly, all these clinical signs were observed in the animals of this study with a predominance of seizures (54%), followed by cerebellar ataxia (31%) and deficit of menace response (31%).
Arachnoid cysts consist of a thin-walled, well-demarcated, fluid-filled cavity that has a triangular cross-section. On image evaluations the fluid is isodense to CSF in CT, the cysts have a very similar signal intensity to CSF in all sequences on MRI and the capsule does not frequently take up contrast (Lang & Seiler 2010Lang J. & Seiler G. 2010. Neuroradiology, p.140. In: Jaggy A. & Platt S.R. (Eds), Small Animal Neurology: an illustred text. Schlütersche Verlag, Hannover.). There were no other features on CT or MRI exams beyond those described in the literature.
Measurement of the degree of parenchymal compression of the occipital lobes and the cerebellum was proposed in a previous study using MRI on mid-sagittal T2- and T1-weighted images (Matiasek et al. 2007Matiasek L.A., Platt S.R. , Shaw S. & Dennis R. 2007. Clinical and magnetic resonance imaging characteristics of quadrigeminal cysts in dogs. J Vet Intern Med 21(5):1021-1026. PMid:17939559.). Likewise, the measurement was performed in both MRI and CT images. Although in only 1/17 MRI scan it was not possible to perform this measurement due the extensive dilatation of the ventricular system loosing the anatomical parameters (case 9) and also it was not possible to perform the measurement in 4/9 CT scans due to technical problems. Therefore, 21 MRI and CT scans had the degree of compression of the occipital lobe and the cerebellum measured.
The occipital lobe is responsible for processing visual information, so when there is damage in this region the main clinical sign observed is visual impairment or central blindness, represented by a deficit of menace response, with intact pupillary reflexes (Lorenz et al. 2010Lorenz M.D., Coates J.R. & Kent M. 2010. Neurologic history, neuroanatomy, and neurologic examination, p.5-14. In: Ibid. (Eds), Handbook of Veterinary Neurology. 5th ed. Elsevier, St Louis.). Seizures can occur when there is compression of the cerebral cortex including in the occipital lobe (Jaggy & Spiess 2010Jaggy A. & Spiess B. 2010. Neurological examination of small animals, p.1-37. In: Jaggy A. & Platt S.R. (Eds), Small Animal Neurology: an illustred text. Schlütersche Verlag, Hannover.).
In this study, these were the most common clinical signs with a total of 18 dogs, being 10 dogs presenting seizures, 4 presenting deficit of menace response, and 4 presenting both clinical signals (Table 1). The degree of occipital lobe compression in the dogs that presented clinical signs ranged from 0 to 24.2% (mean, 6.4%; SD = 9.2). Nevertheless, for 8 of these 18 dogs (cases 7, 8, 11, 13, 14, 15, 16, 18) the CT and MRI scans revealed 0% of occipital lobe compression, consisting of four dogs (cases 11, 13, 14, 18) with hydrocephalus, three (cases 7, 8, 15) with cortical inflammatory disease and only one (case 16) with no abnormality on MRI beyond the quadrigeminal cyst.
Six of the eighteen dogs that presented seizures and/or deficit of menace response had others brain abnormalities on CT and MRI, associated with some degree of the occipital lobe compression caused by quadrigeminal arachnoid cysts. Three dogs (cases 10, 12, 19) presented hydrocephalus, one dog (case 17) presented lissencephaly and agenesis of the callosum corpus, one (case 2) presented meningoencephalitis, and one (case 1) presented tumor on cerebellar pontine angle.
In four dogs that presented those clinical signs, the degree of occipital lobe compression could not be measured due to technical problems, with three dogs (cases 9, 24, 26) presenting hydrocephalus and one dog (case 23) having no abnormalities on CT images but with a cyst shown with mass-effect.
The cerebellum functions as a coordinator of movements that originate in the upper motor neuron system and serves to help maintain equilibrium, posture, body support against gravity, and eye movements (Lorenz et al. 2010Lorenz M.D., Coates J.R. & Kent M. 2010. Neurologic history, neuroanatomy, and neurologic examination, p.5-14. In: Ibid. (Eds), Handbook of Veterinary Neurology. 5th ed. Elsevier, St Louis.). In general, cerebellar disease causes severe ataxia, dysmetria (hypermetria), intention tremors, wide-legged stance (Jaggy & Spiess 2010Jaggy A. & Spiess B. 2010. Neurological examination of small animals, p.1-37. In: Jaggy A. & Platt S.R. (Eds), Small Animal Neurology: an illustred text. Schlütersche Verlag, Hannover.), and sometimes vestibular signs (paradoxical vestibulopathy) (Lorenz et al. 2010Lorenz M.D., Coates J.R. & Kent M. 2010. Neurologic history, neuroanatomy, and neurologic examination, p.5-14. In: Ibid. (Eds), Handbook of Veterinary Neurology. 5th ed. Elsevier, St Louis.). The same clinical signs were present in 9 dogs (cases 6, 12, 18-20, 22, 23, 25, 26) affected, of whom 6 dogs had a degree of cerebellum compression ranging from 0 to 55.7% (mean, 18.9%; SD = 24.4). Most of these cases presented CT and MRI scans with hydrocephalus (cases 12, 18-20, 22, 26) and one case presented cerebellar atrophy (case 25). Only one (case 23) presented no abnormalities beyond the quadrigeminal arachnoid cyst.
Among some clinical signs that were not correlated to local compression of the occipital lobe and the cerebellum were compulsive walking (frontal lobe, internal capsule, basal nuclei), paresis (frontal lobe, internal capsule, basal nuclei, brainstem, spinal cord, peripheral nerves), behavioral changes, as head pressing (limbic system, frontal or temporal cortex), head tilt and positional strabismus (vestibular system), mental depression (cerebrum, brainstem), circling and pleurothotonus (frontoparietal lobe, rostral thalamic) (Lorenz et al. 2010Lorenz M.D., Coates J.R. & Kent M. 2010. Neurologic history, neuroanatomy, and neurologic examination, p.5-14. In: Ibid. (Eds), Handbook of Veterinary Neurology. 5th ed. Elsevier, St Louis.), proprioceptive ataxia (parietal lobe, thalamus, brainstem, spinal cord and peripheral nerves) (Jaggy & Spiess 2010Jaggy A. & Spiess B. 2010. Neurological examination of small animals, p.1-37. In: Jaggy A. & Platt S.R. (Eds), Small Animal Neurology: an illustred text. Schlütersche Verlag, Hannover.).
Meanwhile, seizures occur when there is compression of the cerebrum. Nevertheless, some of these signs may be associated with hydrocephalus, as behavior changes, loss of vision and gait disturbance (Lorenz et al, 2010Lorenz M.D., Coates J.R. & Kent M. 2010. Neurologic history, neuroanatomy, and neurologic examination, p.5-14. In: Ibid. (Eds), Handbook of Veterinary Neurology. 5th ed. Elsevier, St Louis.). In our study, 20 dogs that were diagnosed with hydrocephalus ranging from discrete to severe, 15 dogs presented the clinical signs described above, but two dogs (cases 7, 15) had clinical signs consistent with multifocal disease observed on MRI as inflammatory brain disease. One dog (case 3) presented only clinical signs related with behavioral disturbance and its MRI showed no abnormalities beyond the quadrigeminal arachnoid cyst.
Even on CT scans that could not perform MPR mid-sagittal it was possible to identify the phenotype due its extension from third and/or mesencephalic aqueduct dorso-rostrally to the fourth ventricle. In 14 cases it was observed that the phenotype of the arachnoid cyst had correlation with the compression site. Five cases were observed that showed the phenotype of the arachnoid cyst was correlated with forebrain compression; 7 cases with phenotype in association to the fourth ventricle were correlated with cerebellar compression; and 2 cases with phenotype in association to the third and fourth were correlated with both forebrain and cerebellar compression. However, the clinical manifestation of these dogs indicated lesions in others sites of the brain not related with compression caused by quadrigeminal arachnoid cysts.
Clinical treatment based in seizure management with anticonvulsant and corticosteroids of those dogs improved the clinical signs, but the follow-up was in a short period or was not followed-up in some patients. Cystoperitoneal shunting may be an effective treatment method for dogs clinically affected with intracranial arachnoid cyst (Dewey et al. 2007Dewey C.W., Krotscheck U., Bailey K.S. & Marino D.J. 2007. Craniotomy with cystoperitoneal shunting for treatment of intracranial arachnoid cysts in dogs. Vet. Surg. 36(5):416-422. PMid:17614922. http://dx.doi.org/10.1111/j.1532-950X.2007.00287.x.
https://doi.org/10.1111/j.1532-950X.2007...
). A dog of this retrospective study was submitted to ventricle-peritoneal shunting which improved the clinical signs, such as seizures.
Based on the clinical signs, CSF analyses and other abnormalities on CT and/or MRI scans, 17 dogs (cases 1, 4-7, 9, 13, 15-25) had quadrigeminal arachnoid cyst regarded as an incidental finding. In 6 dogs (cases 2, 10-12, 14, 23) the findings were related with quadrigeminal arachnoid cyst. However, in the 3 remain dogs (cases 6, 8, 26), the findings were not associated exclusively with quadrigeminal arachnoid cyst, suggesting others sites of brain lesion.
According to Matiasek et al. (2007)Matiasek L.A., Platt S.R. , Shaw S. & Dennis R. 2007. Clinical and magnetic resonance imaging characteristics of quadrigeminal cysts in dogs. J Vet Intern Med 21(5):1021-1026. PMid:17939559., occipital lobe compression greater than 14% is uniformly associated with clinical signs. However in our study, in the six dogs that presented findings related to quadrigeminal arachnoid cyst, the mean of degree of compression of the occipital lobe was 9.1% (SD = 10). The cases in which the quadrigeminal arachnoid cyst was considered to be incidental had a considerable degree of cerebellum compression ranging from 15.7% to 55.7%.
In this retrospective study, it was concluded that the quadrigeminal arachnoid cyst is an incidental finding mainly when clinical signs presented by the patient do not associate with compression of the occipital lobe, cerebellum or with an increase of intracranial pressure. When there are clinical signs related to compression caused by the arachnoid cysts, ruling out other intracranial diseases, they can be regarded as the determinant cause, making therapeutic or surgical treatment necessary.
Acknowledgments
The authors are grateful to Heraldo André Catalan Rosa for MRI and CT technical support. The MRI equipment used in all cases of this retrospective study was acquired by means of researches under the procedure number 2009/54028-8 of the São Paulo Research Foundation (FAPESP) - R. Pio XI, 1500 Sao Paulo 05468-901, Brazil.
References
- Bernardino F., Rentmeister K., Schmidt M.J., Bruehschwein A., Matiasek K., Matiasek L.A., Lauda A., Schoon H.A. & Fischer A. 2015. Inferior cerebellar hypoplasia resembling a Dandy-Walker-like malformation in purebred eurasier dogs with familial non-progressive ataxia: a retrospective and prospective clinical cohort study. PLoS One 10(2):e0117670. PMid:25668516. http://dx.doi.org/10.1371/journal.pone.0117670.
» https://doi.org/10.1371/journal.pone.0117670 - Bohn A.A., Wills T.B., West C.L., Tucker R.L. & Bagley R.S. 2006. Cerebrospinal fluid analysis and magnetic resonance imaging in the diagnosis of neurologic disease in dogs: a retrospective study. Vet. Clin. Pathol. 35(3):315-320. PMid:16967416. http://dx.doi.org/10.1111/j.1939-165X.2006.tb00138.x.
» https://doi.org/10.1111/j.1939-165X.2006.tb00138.x. - Cincu R., Agrawal A. & Eiras J. 2007. Intracranial arachnoid cysts: current concepts and treatment alternatives. Clin. Neurol. Neurosurg. 109(10):837-843. PMid:17764831. http://dx.doi.org/10.1016/j.clineuro.2007.07.013.
» https://doi.org/10.1016/j.clineuro.2007.07.013. - Dei-Anang K. & Voth D. 1989. Cerebral arachnoid cyst: a lesion of the child’s brain. Neurosurg. Rev. 12(1):59-62. PMid:2747935. http://dx.doi.org/10.1007/BF01787131.
» https://doi.org/10.1007/BF01787131. - Dewey C.W., Krotscheck U., Bailey K.S. & Marino D.J. 2007. Craniotomy with cystoperitoneal shunting for treatment of intracranial arachnoid cysts in dogs. Vet. Surg. 36(5):416-422. PMid:17614922. http://dx.doi.org/10.1111/j.1532-950X.2007.00287.x.
» https://doi.org/10.1111/j.1532-950X.2007.00287.x. - Dewey C.W., Scrivani P.V., Krotscheck U., Cerda-Gonzalez S., Smith Bailey K. & Marino D.J. 2009. Intracranial arachnoid cysts in dogs. Compend. Contin. Educ. Vet. 31(4):160-167, quiz 168. PMid:19517408.
- Di Rocco C. 1996. Arachnoid cysts. p.967-994. In: Youmans J. (Ed.), Neurological Surgery. Vol.2. 4th ed. W.B. Saunders, Philadelphia.
- Duque C., Parent J., Brisson B., Costa R. & Poma R. 2005. Intracranial arachnoid cysts: are they clinically significant? J. Vet. Intern. Med. 19(5):772-774. PMid:16231727. http://dx.doi.org/10.1111/j.1939-1676.2005.tb02761.x.
» https://doi.org/10.1111/j.1939-1676.2005.tb02761.x. - Freeman R.A. & Raskin R.E. 2001. Cytology of the central nervous system, p.967-994. In: Raskin R.E. & Meyer D.J. (Eds), Atlas of Canine and Feline Cytology. W.B. Saunders, Philadelphia.
- Galassi E., Tognetti F., Frank F., Fagioli L., Nasi M.T. & Gaist G. 1985. Infratentorial arachnoid cysts. J. Neurosurg. 63(2):210-217. PMid:4020444. http://dx.doi.org/10.3171/jns.1985.63.2.0210.
» https://doi.org/10.3171/jns.1985.63.2.0210. - Hanieh A., Simpson D.A. & North J.B. 1988. Arachnoid cysts: a critical review of 41 cases. Childs Nerv. Syst. 4(2):92-96. PMid:3401876.
- Jaggy A. & Spiess B. 2010. Neurological examination of small animals, p.1-37. In: Jaggy A. & Platt S.R. (Eds), Small Animal Neurology: an illustred text. Schlütersche Verlag, Hannover.
- Kitagawa M., Kanayama K. & Sakai T. 2003. Quadrigeminal cistern arachnoid cyst diagnosed by MRI in five dogs. Aust. Vet. J. 81(6):340-343. PMid:15080455. http://dx.doi.org/10.1111/j.1751-0813.2003.tb11509.x.
» https://doi.org/10.1111/j.1751-0813.2003.tb11509.x. - Koie H., Kitagawa M., Kuwabara M. & Sakai T. 2000. Pineal arachnoid cyst demonstrated with magnetic resonance imaging. Canine Pract. 25:14-15.
- Lang J. & Seiler G. 2010. Neuroradiology, p.140. In: Jaggy A. & Platt S.R. (Eds), Small Animal Neurology: an illustred text. Schlütersche Verlag, Hannover.
- Lorenz M.D., Coates J.R. & Kent M. 2010. Neurologic history, neuroanatomy, and neurologic examination, p.5-14. In: Ibid. (Eds), Handbook of Veterinary Neurology. 5th ed. Elsevier, St Louis.
- Lowrie M., Wessmann A., Gunn-Moore D. & Penderis J. 2009. Quadrigeminal cyst management by cystoperitoneal shunt in a 4-year-old Persian cat. J. Feline Med. Surg. 11(8):711-713. PMid:19261501. http://dx.doi.org/10.1016/j.jfms.2009.01.007.
» https://doi.org/10.1016/j.jfms.2009.01.007. - Matiasek L.A., Platt S.R. , Shaw S. & Dennis R. 2007. Clinical and magnetic resonance imaging characteristics of quadrigeminal cysts in dogs. J Vet Intern Med 21(5):1021-1026. PMid:17939559.
- Milner R.J., Engela J. & Kirberger R.M. 1996. Arachnoid cyst in cerebellar pontine area of a cat diagnosis by magnetic resonance imaging. Vet. Radiol. Ultrasound 37(1):34-36. http://dx.doi.org/10.1111/j.1740-8261.1996.tb00809.x.
» https://doi.org/10.1111/j.1740-8261.1996.tb00809.x. - Miyabe-Nishiwaki T., Kaneko T., Sakai T., Kaneko A., Watanabe A., Watanabe S., Maeda N., Kumazaki K., Suzuki J., Fujiwara R., Makishima H., Nishimura T., Hayashi M., Tomonaga M., Matsuzawa T. & Mikami A. 2014. Intracranial arachnoid cysts in a chimpanzee (Pan troglodytes). Primates 55(1):7-12. PMid:24068629. http://dx.doi.org/10.1007/s10329-013-0384-0.
» https://doi.org/10.1007/s10329-013-0384-0. - Osborn A.G. 1994. Miscellaneous tumors, cysts, and metastases, p.639-642. In: Ibid. (Ed.), Diagnostic Neuroradiology. Mosby, St Louis.
- Reed S., Cho C.Y. & Paulsen D. 2009. Quadrigeminal arachnoid cysts in a kitten and a dog. J. Vet. Diagn. Invest. 21(5):707-710. PMid:19737770. http://dx.doi.org/10.1177/104063870902100519.
» https://doi.org/10.1177/104063870902100519. - Rengachary S.S. & Watanabe I. 1981. Ultrastructure and pathogenesis of intracranial arachnoid cysts. J. Neuropathol. Exp. Neurol. 40(1):61-83. PMid:7205328. http://dx.doi.org/10.1097/00005072-198101000-00007.
» https://doi.org/10.1097/00005072-198101000-00007. - Robertson S.J., Wolpert S.M. & Runge V.M. 1989. MR imaging of middle cranial fossa arachnoid cysts: temporal lobe agenesis syndrome revisited. AJNR Am. J. Neuroradiol. 10(5):1007-1010. PMid:2505512.
- Saito M., Olby N.J. & Spaulding K. 2001. Identification of arachnoid cysts in the quadrigeminal cistern using ultrasonography. Vet. Radiol. Ultrasound 42(5):435-439. PMid:11678566. http://dx.doi.org/10.1111/j.1740-8261.2001.tb00966.x.
» https://doi.org/10.1111/j.1740-8261.2001.tb00966.x. - Taroni M., Seurin M-J., Carozzo C. & Escriou C. 2015. Supratentorial arachnoid cyst management by cystoperitoneal shunt in a 1-year-old European cat. J. Feline Med. Surg.
- Terlizzi R. & Platt S. 2006. The function, composition and analysis of cerebrospinal fluid in companion animals: Part I - Function and composition. Vet. J. 172(3):422-431. PMid:16154365. http://dx.doi.org/10.1016/j.tvjl.2005.07.021.
» https://doi.org/10.1016/j.tvjl.2005.07.021. - Vernau K.M., Kortz G.D., Koblik P.D., LeCouteur R.A., Bailey C.S. & Pedroia V. 1997. Magnetic resonance imaging and computed tomography characteristics of intracranial intra-arachnoid cysts in 6 dogs. Vet. Radiol. Ultrasound 38(3):171-176. PMid:9238786. http://dx.doi.org/10.1111/j.1740-8261.1997.tb00835.x.
» https://doi.org/10.1111/j.1740-8261.1997.tb00835.x. - Vernau K.M., LeCouteur R.A., Sturges B.K., Samii V., Higgins R.J., Koblik P.D. & Vernau W. 2002. Intracranial intra-arachnoid cyst with intracystic hemorrhage in two dogs. Vet. Radiol. Ultrasound 43(5):449-454. PMid:12375779. http://dx.doi.org/10.1111/j.1740-8261.2002.tb01032.x.
» https://doi.org/10.1111/j.1740-8261.2002.tb01032.x. - Wang P.-J., Lin H.-C., Liu H.-M., Tseng C.-L. & Shen Y.-Z. 1998. Intracranial arachnoid cysts in children: related signs and associated anomalies. Pediatr. Neurol. 19(2):100-104. PMid:9744627. http://dx.doi.org/10.1016/S0887-8994(98)00020-4.
» https://doi.org/10.1016/S0887-8994(98)00020-4. - Yamakawa H., Ohkuma A., Hattori T., Niikawa S. & Kobayashi H. 1991. Primary intracranial arachnoid cyst in the elderly: a survey on 39 cases. Acta Neurochir. 113(1-2):42-47. PMid: 1799142. http://dx.doi.org/10.1007/BF01402113.
» https://doi.org/10.1007/BF01402113.
Publication Dates
-
Publication in this collection
Feb 2018
History
-
Received
16 Feb 2016 -
Accepted
04 Sept 2016