ABSTRACT:
Tuberculosis is a chronic anthropozoonosis of worldwide occurrence, caused by the bacterium Mycobacterium tuberculosis and its variants. In Brazil, the National Program for the Control and Eradication of Brucellosis and Tuberculosis in cattle, is responsible for diagnosing and the correctly allocate positive animals, but there is still a lack of definitive diagnosis of the disease. This study described the use of five diagnostic tools that can be used, preferably together, for the confirmation of suspected cases. These tools included the clinical examination comparative cervical tuberculin test, macroscopic findings during the slaughtering and histopathology of the damaged tissues followed by histochemistry. We evaluated a total of 211 dairy cattle, where 15.1% (32/211) had classic clinical signs of bovine tuberculosis, 74 (35%) showed reactivity in the comparative cervical tuberculin test. Of the total number of animals, 141 (66.8%) were referred for sanitary slaughter due to legal and control issues in the outbreaks of the disease. In the follow-up of slaughtering and inspection of viscera and carcasses, 74 (52.5%) had macroscopic lesions compatible with bovine tuberculosis, while 67 (47.5%) showed no visible changes. During the inspection, fragments of lymph nodes and liver and lung parenchyma were collected from five cattle with macroscopic lesions and five with no lesions. The histopathological analysis showed numerous areas of caseous necrosis with or without central calcification and granulomatous inflammatory infiltrate. In the special staining of Ziehl-Neelsen, numerous acid-fast bacilli were evidenced in all cases.
INDEX TERMS:
Bovine; tuberculosis; diagnostic; dairy cattle; zoonosis; Mycobacterium spp.; cattle; bacterioses
RESUMO:
A tuberculose é uma antropozoonose crônica de ocorrência mundial, causada pela bactéria Mycobacterium tuberculosis e suas variantes. No Brasil existe o Programa Nacional de Controle e Erradicação da Brucelose e Tuberculose em bovinos que viabiliza o diagnóstico e a destinação correta dos animais positivos, porém ainda há carência quanto ao diagnóstico da doença. Assim, este trabalho descreve a utilização de cinco ferramentas diagnósticas para a confirmação de casos suspeitos de tuberculose. As ferramentas utilizadas compreenderam o exame clínico, teste tuberculínico cervical comparativo, os achados macroscópicos durante o abate sanitário e a histopatologia dos tecidos lesados seguido de histoquímica. O estudo avaliou um total de 211 bovinos leiteiros, dos quais 15,1% (32/211) apresentaram sinais clínicos clássicos de tuberculose bovina, 35,1% (74/211) apresentaram reatividade no teste tuberculínico cervical comparativo, e 143 animais (67,8%) foram encaminhados para abate sanitário devido a questões legais e de controle nos focos da doença. No acompanhamento do abate e inspeção sanitária de vísceras e carcaças verificou-se que 51,8% (74/143) dos bovinos abatidos apresentavam lesões macroscópicas compatíveis com tuberculose bovina, enquanto 48,2% (69/143) não apresentavam alterações visíveis. Durante a inspeção foram coletados fragmentos de linfonodos e parênquima de fígado e pulmão de cinco bovinos com lesões macroscópicas e de cinco sem lesões, que na análise histopatológica apresentaram numerosas áreas de necrose caseosa com ou sem calcificação central e infiltrado inflamatório granulomatoso. Na coloração de Ziehl-Neelsen foram evidenciados numerosos bacilos álcool-ácido resistentes em todos os casos. Assim, diante dos resultados obtidos verifica-se que as análises empregadas no presente estudo foram de extrema importância para o diagnóstico acurado de tuberculose em bovinos.
TERMOS DE INDEXAÇÃO:
Tuberculose; bovinos; diagnóstico; bovinos leiteiros; zoonose; Mycobacterium spp.; bacterioses
Introduction
Tuberculosis is a chronic anthropozoonosis of worldwide evolution (Acha & Szyfres 2005Acha P.N. & Szyfres B. 2005. Zoonoses and communicable diseases common to man and animals: bacterioses and mycoses. 3rd ed. Pan American Health Organization, Washington. 378p.). It is caused by the bacterium Mycobacterium tuberculosis and its variants, comprising M. tuberculosis var. tuberculosis, M. tuberculosis var. africanum, M. tuberculosis var. canetti, M. tuberculosis var. bovis, M. tuberculosis var. caprae, M. tuberculosis var. microti, M. tuberculosis var. pinnipedii, M. tuberculosis var. mungi, M. tuberculosis var. orygis and M. tuberculosis var. suricattae (Riojas et al. 2018Riojas M.A., McGough K.J., Rider-Riojas C.J., Rastogi N. & Hazbón M.H. 2018. Phylogenomic analysis of the species of the Mycobacterium tuberculosis complex demonstrates that Mycobacterium africanum, Mycobacterium bovis, Mycobacterium caprae, Mycobacterium microti and Mycobacterium pinnipedii are later heterotypic synonyms of Mycobacterium tuberculosis. Int. J. Syst. Evol. Microbiol. 68(1):324-332. <http://dx.doi.org/10.1099/ijsem.0.002507> <PMid:29205127>
https://doi.org/10.1099/ijsem.0.002507...
). According to the World Health Organization (WHO), tuberculosis is the infectious disease that causes the most deaths, even surpassing HIV. In 2016, about 10.4 million people fell ill from tuberculosis worldwide, and about 1.3 million died (WHO 2017WHO 2017. Bending the curve: ending TB. Annual report 2017, World Health Organization, Geneva. 72p. Available at <Available at http://apps.who.int/iris/handle/10665/254762
> Access on Aug. 1, 2018.
http://apps.who.int/iris/handle/10665/25...
). In Brazil, in 2016, 4,426 deaths were registered, and in 2017 69,569 new cases of tuberculosis were reported (Brasil 2018Brasil 2018. Implantação do Plano Nacional pelo fim da tuberculose como problema de saúde pública no Brasil: primeiros passos rumo ao alcance das metas. Boletim Epidemiológico 49(11):1-18.).
In cattle, the disease is caused by M. tuberculosis var. bovis and is one of the leading infectious diseases of the species (Acha & Szyfres 2005Acha P.N. & Szyfres B. 2005. Zoonoses and communicable diseases common to man and animals: bacterioses and mycoses. 3rd ed. Pan American Health Organization, Washington. 378p., Radostits et al. 2007Radostits O.M., Gay C.C., Hinchcliff K.W. & Constable P.D. 2007. Veterinary Medicine: a textbook of the diseases of cattle, horses, sheep, pigs, and goats. 10th ed. W.B. Saunders, Philadelphia. 2156p., Riojas et al. 2018Riojas M.A., McGough K.J., Rider-Riojas C.J., Rastogi N. & Hazbón M.H. 2018. Phylogenomic analysis of the species of the Mycobacterium tuberculosis complex demonstrates that Mycobacterium africanum, Mycobacterium bovis, Mycobacterium caprae, Mycobacterium microti and Mycobacterium pinnipedii are later heterotypic synonyms of Mycobacterium tuberculosis. Int. J. Syst. Evol. Microbiol. 68(1):324-332. <http://dx.doi.org/10.1099/ijsem.0.002507> <PMid:29205127>
https://doi.org/10.1099/ijsem.0.002507...
). The disease causes a reduction in meat and milk production and a high rate of slaughter carcass condemnation, as well as economic losses due to embargoes on the marketing of animals and animal products (Demelash et al. 2009Demelash B., Inangolet F., Oloya J., Asseged B., Badaso M., Yilkal A. & Skjerve E. 2009. Prevalence of bovine tuberculosis in Ethiopian slaughter cattle based on post-mortem examination. Trop. Anim. Health Prod. 41(1):755-765. <http://dx.doi.org/10.1007/s11250-008-9248-9> <PMid:19058024>
https://doi.org/10.1007/s11250-008-9248-...
, Asil et al. 2012Asil T.A., El Sanousi S.M., Gameel A., El Beir H., Fathelrahman M., Terab N.M., Muaz M.A. & Hamid M.E. 2012. Bovine tuberculosis in South Darfur State, Sudan: an abattoir study based on microscopy and molecular detection methods. Trop. Anim. Health Prod. 45(1):469-472. <PMid:22843216>, Paes & Franco 2016Paes A.C. & Franco M.M.J. 2016. Tuberculose em animais de produção, p.512-542. In: Paes A.C. & Franco M.M.J. (Eds), Doenças Infecciosas em Animais de Produção e Companhia. Roca, Rio de Janeiro.). Its transmission occurs mainly through direct contact with contaminated animals, consumption of infected water, and ingestion of raw milk and/or dairy products made from raw milk, such as clandestine cheese (Corrêa & Corrêa 1992Corrêa W.M. & Corrêa C.N.M. 1992. Tuberculose, p.317-337. In: Corrêa W.M. & Corrêa C.N.M. (Eds), Enfermidades Infecciosas dos Mamíferos Domésticos. 2ª ed. Medsi, Rio de Janeiro.). The national average of tuberculosis occurrence in cattle is 2.64%, with heterogeneity and fluctuation within and between the evaluated states, and in the southern region, highlighting milk production, Rio Grande do Sul, Santa Catarina and Paraná presented 2, 8%, 0.06% and 2.15%, respectively (Ferreira Neto et al. 2016Ferreira Neto J.S., Silveira G.B., Rosa B.M., Gonçalves V.S.P., Grisi-Filho J.H.H., Amaku M. & Lage A.P. 2016. Analysis of 15 years of the National Program for the Control and Eradication of Animal Brucellosis and Tuberculosis, Brazil. Semina, Ciênc. Agrárias 37(5):3385-3402.).
In Brazil, since 2001, the “Programa Nacional de Controle e Erradicação da Brucelose e Tuberculose” (PNCEBT, National Program for the Control and Eradication of Brucellosis and Tuberculosis) for cattle and buffaloes, created to establish norms and procedures for the diagnosis of these diseases (Brasil 2006Brasil 2006. Manual Técnico. Programa Nacional de Controle e Erradicação da Brucelose e da Tuberculose Animal (PNCEBT), Ministério da Agricultura, Pecuária e Abastecimento, Brasilia, DF.). According to PNCEBT, the basis for the diagnosis of in vivo tuberculosis is given by intradermal tuberculinization in cattle and buffaloes older than six months. Tuberculinization is immunologically characterized by late-type IV hypersensitivity reaction and can be performed using caudal, simple cervical or comparative cervical fold techniques (Brasil 2006Brasil 2006. Manual Técnico. Programa Nacional de Controle e Erradicação da Brucelose e da Tuberculose Animal (PNCEBT), Ministério da Agricultura, Pecuária e Abastecimento, Brasilia, DF., 2017aBrasil 2017a. Instrução Normativa Nº 10 de 03 de março de 2017. Regulamento Técnico do Programa Nacional de Controle e Erradicação da Brucelose e Tuberculose (PNCEBT), Ministério da Agricultura, Pecuária e Abastecimento, Brasília, DF.).
Brazilian legislation recommends sanitary slaughter of the positive tuberculin test animal, which must be performed in refrigerators under official inspection so that post mortem observations can be performed and, mainly, for the correct disposal of the carcass (Brasil 2017bBrasil 2017b. Decreto nº 9.013 de 29 de março de 2017, alterado pelo Decreto nº 9.069 de 31 de maio de 2017. Regulamento de Inspeção Industrial e Sanitária de Produtos de Origem Animal (RIISPOA), Ministério da Agricultura, Pecuária e do Abastecimento, Brasília, DF.). However, there are cases of nonspecific reaction where cattle show reaction to tuberculin without presenting the disease. There may also be cases in which the animal had contact with the agent, without tuberculosis lesions characteristic in the post mortem inspection, as well as negative animals in the test and presenting compatible lesions in the post mortem. These situations become questionablefor official agents who have to decide the fate of animals (Paes & Franco 2016Paes A.C. & Franco M.M.J. 2016. Tuberculose em animais de produção, p.512-542. In: Paes A.C. & Franco M.M.J. (Eds), Doenças Infecciosas em Animais de Produção e Companhia. Roca, Rio de Janeiro.). We used the comparative cervical tuberculin test, followed by thorough inspection of animals submitted to sanitary slaughter and tissue collection for histological and histochemical examination for diagnosis of bovine tuberculosis.
Materials and Methods
The application of tests for diagnosis of brucellosis and tuberculosis was part of the routine of the PNCEBT-qualified veterinarian and collaborator of the study, whose services were hired by the owners of the herds. The study evaluated 211 dairy cattle of different ages, from November 2017 to April 2018, coming from small farms of three municipalities of the north of Rio Grande do Sul. The importance of the diagnosis was explained to the owners, as well as clarified the legal measures in case there are positive animals in the herd. Thus, upon clarification, all farm animals were evaluated by a general clinical examination and subsequently submitted to tuberculin test.
The comparative cervical tuberculin test (CCTT) is considered a confirmatory test. We performed trichotomy in two areas: 1) cervical region, in front of the scapular spine and at 20cm from the withers, where the avian tuberculin was inoculated; 2) in the cervical region, behind the scapula spine, and 20cm from the withers, where bovine tuberculin was inoculated. Initially, a minimum distance from 15 to 20cm was respected between these two points, and the skinfold thickness of each point was measured with a cutimeter. Subsequently, we inoculated 0.1ml of avian tuberculin PPD (purified protein derivatitis) and 0.1ml of bovine PPD tuberculin. After 72 hours of inoculation, the skinfold thickness was checked again, and the differences were interpreted according to the criteria defined by the PNCEBT Technical Regulation (Brasil 2017aBrasil 2017a. Instrução Normativa Nº 10 de 03 de março de 2017. Regulamento Técnico do Programa Nacional de Controle e Erradicação da Brucelose e Tuberculose (PNCEBT), Ministério da Agricultura, Pecuária e Abastecimento, Brasília, DF.), in order to conclude which animals were reactive, nonreactive or inconclusive.
As recommended by the PNCEBT, all cattle considered reactive or inconclusive in the tuberculin test were marked on the right side of the face with a “P” in a circle of eight centimeters of diameter, and intended for slaughter in a slaughterhouse under official inspection. For sanitary and control reasons, cattle considered negative, but coming from properties where more than 50% of the lot was positive or inconclusive, were also destined for slaughter in a slaughterhouse under official inspection. The carcass and viscera inspection of all animals destined for sanitary slaughter was followed in order to locate and identify macroscopic lesions suggestive of bovine tuberculosis, especially caseous and/or calcified lesions. The analysis was performed by visual inspection, palpation and serial sectioning of the lymph nodes (mediastinal, bronchial, retropharyngeal, parotid, mandibular, mesenteric and hepatic), lung parenchyma and hepatic parenchyma and carcass.
Among the reactive CCTT cattle sent for slaughter, five animals with characteristic tuberculosis lesions and five without any caseous or calcified lesions were selected. From these, we collected lymph node fragments (retropharyngeal, mediastinal, bronchial, mesenteric and hepatic) and pulmonary and hepatic parenchyma fragments. The samples were placed in vials with 10% buffered formalin and sent for processing and laboratory analysis with histopathological examination (hematoxylin and eosin, HE) and histochemical analysis using the special Ziehl-Neelsen stain to identify acid-fast bacilli (AFB).
Results and Discussion
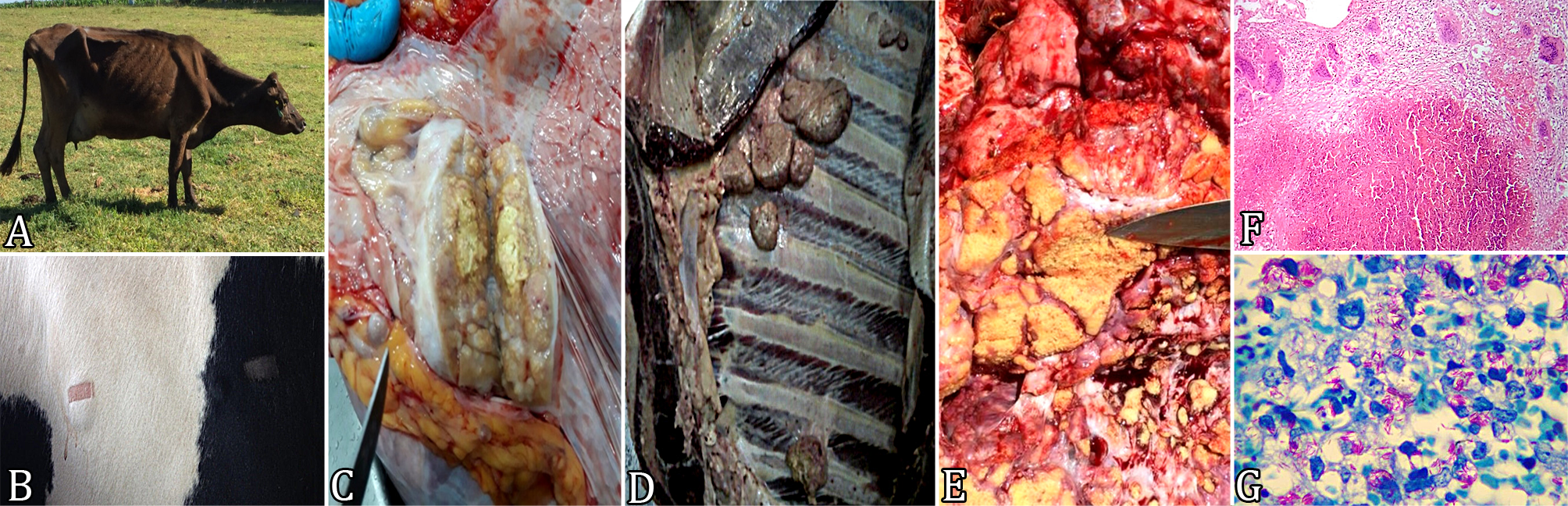
In the clinical evaluation, 15.1% (32/211) of dairy cattle presented classic clinical signs of bovine tuberculosis, such as decreased milk production, difficulty in conception and difficulty breathing, prostration and cachexia (Fig.1A). Subsequently, they were submitted to CCTT (Fig.1B), which showed that 35.1% (74/211) had reactivity, being 63.5% (47/74) positive and 36.5% (27/74) inconclusive. The test also showed that 64.9% (137/211) of the cattle were negative for bovine and avian tuberculin, although kept with test-reactive animals. Table 1 summarizes the results of the clinical evaluation, the CCTT, the frequency of animals destined for sanitary slaughter by the municipality and the number of macroscopic lesions found during post mortem inspection.
Diagnostic strategies for bovine tuberculosis. (A) Bovine female with difficulty breathing, prostration, and cachexia. (B) Cattle showing positive reaction on CCTT. (C) Caseous lesion in the mediastinal lymph node. (D) Caseous nodules adhering to the parietal pleura. (E) Extensive areas of caseous and mineralized lesions in the pulmonary parenchyma. (F) Lung with lesion with central area of caseous necrosis and granulomatous inflammatory infiltrate composed of epithelioid macrophages, multinucleated giant cells, and lymphocytes. HE, obj.10x. (G) Acid-resistant bacilli (AFB) in giant cell cytoplasm in bovine pre-scapular lymph node lesion with tuberculosis. Ziehl-Neelsen, obj.100x.
Clinical evaluation, cervical comparative tuberculin test (CCTT), animals destined for sanitary slaughter by the municipality, and number of animals with macroscopic lesions found in post mortem inspection during sanitary slaughter
Due to its chronic nature, the wide variability of manifestations makes the clinical diagnosis of tuberculosis a relative value, since the cattle may be locally infected and look healthy (Corrêa & Corrêa 1992Corrêa W.M. & Corrêa C.N.M. 1992. Tuberculose, p.317-337. In: Corrêa W.M. & Corrêa C.N.M. (Eds), Enfermidades Infecciosas dos Mamíferos Domésticos. 2ª ed. Medsi, Rio de Janeiro.). Animal studies inoculated with Mycobacterium tuberculosis var. bovis in contact with uninoculated animals showed no clinical signs compatible with the disease in either group (Cassidy et al. 1999Cassidy J.P., Bryson D.G., Pollock J.M., Evans R.T., Forster F. & Neill S.D. 1999. Lesions in cattle exposed to Mycobacterium bovis - inoculated calves. J. Comp. Pathol. 121(4):321-337. <http://dx.doi.org/10.1053/jcpa.1999.0330> <PMid:10542122>
https://doi.org/10.1053/jcpa.1999.0330...
). In cases of advanced tuberculosis, the clinical diagnosis assumes greater importance, since the animals have decreased milk production, conception difficulty, difficulty breathing, elimination of nasal discharge, prostration, and cachexia (Roxo 1997Roxo E. 1997. M. bovis como causa de zoonose. Revta Bras. Ciênc. Farmac. 18(1):101-108., Brasil 2006Brasil 2006. Manual Técnico. Programa Nacional de Controle e Erradicação da Brucelose e da Tuberculose Animal (PNCEBT), Ministério da Agricultura, Pecuária e Abastecimento, Brasilia, DF., Garbaccio et al. 2018Garbaccio S.G., Delgado F.O., Zumarraga M.J., Rodriguez L.R., Huertas P.S. & Garro C.J. 2018. Diagnóstico bacteriológico de tuberculosis bovina en bovinos reactores positivos a la prueba tuberculínica. Revta Investig. Agropec. 44(1):69-75.).
The sensitivity of the tuberculin test in cattle ranges from 32 to 99% and the specificity ranges from 75.5 to 99.9% (Vitale et al. 1998Vitale F., Capra G., Maxia L., Reale S., Vesco G. & Caracappa S. 1998. Detection of Mycobacterium tuberculosis complex in cattle by PCR using milk, lymph node aspirates and nasal swabs. J. Clin. Microbiol. 36(4):1050-1055. <http://dx.doi.org/10.1128/JCM.36.4.1050-1055.1998> <PMid:9542936>
https://doi.org/10.1128/JCM.36.4.1050-10...
). In the present study, the sensitivity was 35% (74/211) of reactive animals. The tuberculin test may yield false-positive results (Monaghan et al. 1994Monaghan M.L., Doherty M.L., Collins J.D., Kazda J.F. & Quinn P.J. 1994. The tuberculin test. Vet. Microbiol. 40(1):111-124. <http://dx.doi.org/10.1016/0378-1135(94)90050-7> <PMid:8073619>
https://doi.org/10.1016/0378-1135(94)900...
), as animals infected with M. avium, M. tuberculosis, M. avium subsp. paratuberculosis, Nocardia farcinius or other mycobacteria may be reactive to bovine PPD (Monaghan et al. 1994Monaghan M.L., Doherty M.L., Collins J.D., Kazda J.F. & Quinn P.J. 1994. The tuberculin test. Vet. Microbiol. 40(1):111-124. <http://dx.doi.org/10.1016/0378-1135(94)90050-7> <PMid:8073619>
https://doi.org/10.1016/0378-1135(94)900...
, Souza et al. 2016Souza M.A.D., Bombonato N.G., Soares P.M., Ramos G.B., Castro I.P., Medeiros A.A. & Lima A.M.C. 2016. Exames complementares no diagnóstico da tuberculose em bovinos reagentes à tuberculinização comparada. Arqs Inst. Biológico, São Paulo, 83(1):1-8., Garbaccio et al. 2018Garbaccio S.G., Delgado F.O., Zumarraga M.J., Rodriguez L.R., Huertas P.S. & Garro C.J. 2018. Diagnóstico bacteriológico de tuberculosis bovina en bovinos reactores positivos a la prueba tuberculínica. Revta Investig. Agropec. 44(1):69-75.). Thus, we recommend the CCTT since, compared to the simple tuberculin test, there is a reduction in the possibility of such cross-reactions (Collins et al. 1994Collins D.M., Radford A.J., de Lisle G.W. & Billman-Jacobe H. 1994. Diagnosis and epidemiology of bovine tuberculosis using molecular biological approaches. Vet. Microbiol. 40(1):83-94. <http://dx.doi.org/10.1016/0378-1135(94)90048-5> <PMid:7915446>
https://doi.org/10.1016/0378-1135(94)900...
). However, the occurrence of 12.8% (27/211) inconclusive animals demonstrates its limitation, as well as the need to use complementary methods for an accurate result (Monaghan et al. 1994Monaghan M.L., Doherty M.L., Collins J.D., Kazda J.F. & Quinn P.J. 1994. The tuberculin test. Vet. Microbiol. 40(1):111-124. <http://dx.doi.org/10.1016/0378-1135(94)90050-7> <PMid:8073619>
https://doi.org/10.1016/0378-1135(94)900...
, Romero et al. 1999Romero R.E., Garzón D.L., Mejía G.A., Monroy W., Patarroyo M.E. & Murillo L.A. 1999. Identification of Mycobacterium bovis in bovine clinical samples by PCR species-specific primers. Canadian J. Vet. Res. 63(2):101-106. <PMid:10369566>).
Some animals, although infected, do not respond to tuberculin tests (Brasil 2006Brasil 2006. Manual Técnico. Programa Nacional de Controle e Erradicação da Brucelose e da Tuberculose Animal (PNCEBT), Ministério da Agricultura, Pecuária e Abastecimento, Brasilia, DF.), which may have occurred in the present study. In municipalities 1 and 3, 100% of the tested animals were sent for sanitary slaughter control, of which 52% (27/52) and 47% (42/89) were negative in CCTT although they were together with animals positive, respectively. In animals from the same municipalities and sent for slaughter, 85% and 33.7% of macroscopic lesions suggestive of tuberculosis during post mortem inspection, respectively, were evidenced. Factors such as recent infection, late pregnancy, malnutrition, and advanced disease may cause false negatives in the tuberculin test. Also, such results may occur due to variations inherent in the test itself or due to variations in the reading and interpretation of the exam (Brasil 2006Brasil 2006. Manual Técnico. Programa Nacional de Controle e Erradicação da Brucelose e da Tuberculose Animal (PNCEBT), Ministério da Agricultura, Pecuária e Abastecimento, Brasilia, DF.). It is important to note that animals in advanced stages of infection may manifest the phenomenon called anergy, defined as absence of skin reactivity to tuberculin (Roxo 1996Roxo E. 1996. Tuberculose bovina: revisão [Bovine tuberculosis: review]. Arqs Inst. Biológico, São Paulo 63(2):91-97.).
Based on the CCTT results, the guidance of the PNCEBT (Brasil 2006Brasil 2006. Manual Técnico. Programa Nacional de Controle e Erradicação da Brucelose e da Tuberculose Animal (PNCEBT), Ministério da Agricultura, Pecuária e Abastecimento, Brasilia, DF.) and the Veterinary Inspectorate was that, in addition to the reactive cattle, CCTT negative animals coming from properties with more than 50% of reactive cattle for the bovine and avian tuberculin should also be slaughtered, accounting for 67.8% (143/211) of the animals examined. Slaughter was carried out in a slaughterhouse under official inspection, where all processes were suppervised and recorded, including the post mortem inspection of carcasses and viscera. The post mortem inspection showed that 51.8% (74/143) of the cattle presented purulent or caseous lesions, with or without mineralization. This occurred at one or more sites, mainly in the pre-scapular, retropharyngeal, retromammary, hepatic, mesenteric, and apical and/or mediastinal pulmonary lymph nodes (Fig.1C), in addition to the parietal (Fig.1D) and visceral pleurae and pulmonary (Fig.1E) and hepatic parenchyma. However, 48.2% (69/143) of the cattle showed no visible lesions.
Carcasses were considered positive when at least one organ had lesions suggestive of tuberculosis, as in the study by Furlanetto et al. (2012Furlanetto L.V., Figueiredo E.E.S., Conte Júnior C.A., Carvalho R.C.T., Silva F.G.S., Silva J.T., Lilenbaum W. & Paschoalin V.M.F. 2012. Uso de métodos complementares na inspeção post mortem de carcaças com suspeita de tuberculose bovina. Pesq. Vet. Bras. 32(11):1138-1144. <http://dx.doi.org/10.1590/S0100-736X2012001100011>
https://doi.org/10.1590/S0100-736X201200...
). In the present study, of the 143 cattle slaughtered, 51.8% (74/143) presented suggestive lesions, a result inferior to that reported by Fráguas et al. (2008)Fráguas S.A., Cunha-Abreu M.S., Ferreira A.M.R., Marassi C.D., Oelemann W., Fonseca L.S., Ferreira R. & Lilenbaum W. 2008. Estudo comparativo de métodos complementares para o diagnóstico da tuberculose bovina em animais reagentes à tuberculinização. Revta Bras. Ciênc. Vet. 15(3):117-121., who observed 72% of carcasses with lesions in a similar study. However, it was higher than that reported by Pinto et al. (2004)Pinto P.S.A., Viloria M.I.V. & Faria J.E. 2004. Avaliação do desempenho dos exames anatomopatológico e histopatológico na inspeção post mortem de bovinos suspeitos ou reagentes à prova de tuberculinização. Revta Bras. Ciênc. Vet. 11(1):27-31., who found 44% of bovine tuberculosis-reactive cattle also presenting tuberculous lesions. Corner et al. (1990)Corner L.A., Melville L., McCUBBIN K., Small K.J., McCORMICK B.S., Wood P.R. & Rothel J.S. 1990. Efficiency of inspection procedures for detection of tuberculous lesions in cattle. Aust. Vet. J. 67(11):389-392. <http://dx.doi.org/10.1111/j.1751-0813.1990.tb03020.x> <PMid:2085291>
https://doi.org/10.1111/j.1751-0813.1990...
reported that about 58% of tuberculosis animals have unique lesions, and detailed inspection of lymph nodes (head, thoracic, mesenteric and carcass) as well as liver, lung, spleen, kidneys, udder, and genitals increases the possibility visualization of these lesions (Corner 1994Corner L.A. 1994. Post mortem diagnosis of Mycobacterium bovis infection in cattle. Vet. Microbiol. 40(1):53-63. <http://dx.doi.org/10.1016/0378-1135(94)90046-9> <PMid:8073629>
https://doi.org/10.1016/0378-1135(94)900...
). Some aspects may be involved with the non-detection of suggestive lesions in CCTT-reactive animals, such as: early stage of the disease, contact with mycobacteria other than M. tuberculosis var. bovis, time and attention for insufficient post mortem inspection (Souza et al. 1999Souza A.V.D., Sousa C.F.A., Souza R.M.D., Ribeiro R.M.P. & Oliveira A.D.L. 1999. A importância da tuberculose bovina como zoonose. Higiene Alimentar 13(59):22-27., Corner 1994Corner L.A. 1994. Post mortem diagnosis of Mycobacterium bovis infection in cattle. Vet. Microbiol. 40(1):53-63. <http://dx.doi.org/10.1016/0378-1135(94)90046-9> <PMid:8073629>
https://doi.org/10.1016/0378-1135(94)900...
, Brasil 2006Brasil 2006. Manual Técnico. Programa Nacional de Controle e Erradicação da Brucelose e da Tuberculose Animal (PNCEBT), Ministério da Agricultura, Pecuária e Abastecimento, Brasilia, DF., Medeiros et al. 2012Medeiros L.S., Marassi C.D., Figueiredo E.E.S., Leite J., Ferreira A.M.R. & Lilenbaum W. 2012. Assessing the histopathology to depict the different stages of bovine tuberculosis infection in a naturally infected herd. Pesq. Vet. Bras. 32(2):135-139. <http://dx.doi.org/10.1590/S0100-736X2012000200008>
https://doi.org/10.1590/S0100-736X201200...
).
Of the ten cattle selected for collection and requested for histopathological and histochemical examination, five had yellowish lesions with purulent or caseous content surrounded by a fibrous capsule and sometimes calcified appearance at cut, as found by Souza et al. (2016)Souza M.A.D., Bombonato N.G., Soares P.M., Ramos G.B., Castro I.P., Medeiros A.A. & Lima A.M.C. 2016. Exames complementares no diagnóstico da tuberculose em bovinos reagentes à tuberculinização comparada. Arqs Inst. Biológico, São Paulo, 83(1):1-8.. Tissue fragments were also collected from five other reactive CCTT cattle, which did not present macroscopic lesions on inspection. As found by Furlanetto et al. (2012)Furlanetto L.V., Figueiredo E.E.S., Conte Júnior C.A., Carvalho R.C.T., Silva F.G.S., Silva J.T., Lilenbaum W. & Paschoalin V.M.F. 2012. Uso de métodos complementares na inspeção post mortem de carcaças com suspeita de tuberculose bovina. Pesq. Vet. Bras. 32(11):1138-1144. <http://dx.doi.org/10.1590/S0100-736X2012001100011>
https://doi.org/10.1590/S0100-736X201200...
, in the microscopic analysis the ten cattle presented lesions compatible with bovine tuberculosis. In the present study we identified severe or moderate lesions, delimited, with central area of caseous necrosis, sometimes mineralized, and presence of granulomatous inflammatory infiltrate composed of epithelioid macrophages, multinucleated giant cells, and lymphocytes (Fig.1F).
Granulomatous inflammatory processes with macroscopic tuberculosis-like characteristics may occur in cattle, but with different etiologies such as lymphosarcoma, nonspecific lymphadenitis, actinobacillosis and nocardiosis (Kantor et al. 1981Kantor I.N., De La Veja E. & Caballero P. 1981. Estudio de órganos bovinos decomisados por tuberculose, mataderos del gran Buenos Aires. Revta Med. Vet. 62(4):282-285., Reis et al. 1995Reis D.O., Almeida L. & Faria A.R. 1995. Estudo comparativo entre linfossarcoma, tuberculose e linfadenites inespecíficas ocorridas em bovinos abatidos e a confirmação histológica. Higiene Alimentar 9(35):28-30., Roxo 1997Roxo E. 1997. M. bovis como causa de zoonose. Revta Bras. Ciênc. Farmac. 18(1):101-108.). Macroscopic inspection can be subjective for carcass judgment (Reis et al. 1995Reis D.O., Almeida L. & Faria A.R. 1995. Estudo comparativo entre linfossarcoma, tuberculose e linfadenites inespecíficas ocorridas em bovinos abatidos e a confirmação histológica. Higiene Alimentar 9(35):28-30., Brasil 2017bBrasil 2017b. Decreto nº 9.013 de 29 de março de 2017, alterado pelo Decreto nº 9.069 de 31 de maio de 2017. Regulamento de Inspeção Industrial e Sanitária de Produtos de Origem Animal (RIISPOA), Ministério da Agricultura, Pecuária e do Abastecimento, Brasília, DF.), indicating that sanitary inspection of carcasses should be performed carefully by well-trained professionals, reducing the risk of suspicious food arriving consumers or carcasses to be unnecessarily condemned for non-tuberculous lesions (Fraguás et al. 2008Fráguas S.A., Cunha-Abreu M.S., Ferreira A.M.R., Marassi C.D., Oelemann W., Fonseca L.S., Ferreira R. & Lilenbaum W. 2008. Estudo comparativo de métodos complementares para o diagnóstico da tuberculose bovina em animais reagentes à tuberculinização. Revta Bras. Ciênc. Vet. 15(3):117-121., Brasil 2017bBrasil 2017b. Decreto nº 9.013 de 29 de março de 2017, alterado pelo Decreto nº 9.069 de 31 de maio de 2017. Regulamento de Inspeção Industrial e Sanitária de Produtos de Origem Animal (RIISPOA), Ministério da Agricultura, Pecuária e do Abastecimento, Brasília, DF.).
In the present study, Ziehl-Neelsen staining provided evidence of numerous acid-fast bacilli (AFB) in all tissue samples examined (Fig.1G), including those without macroscopic lesions. However, these findings differ from studies that consider low sensitivity Ziehl-Neelsen staining when applied to histological sections (Fráguas et al. 2008Fráguas S.A., Cunha-Abreu M.S., Ferreira A.M.R., Marassi C.D., Oelemann W., Fonseca L.S., Ferreira R. & Lilenbaum W. 2008. Estudo comparativo de métodos complementares para o diagnóstico da tuberculose bovina em animais reagentes à tuberculinização. Revta Bras. Ciênc. Vet. 15(3):117-121.). Andrade et al. (1991)Andrade G.B., Riet-Correa F., Mielke P.V., Méndez M.C. & Schild A.L. 1991. Estudo histológico e isolamento de micobactérias de lesões similares á tuberculose em bovinos no Rio Grande do Sul. Pesq. Vet. Bras. 11(3):81-86. observed AFB in only 9.1% of the histological samples examined in their study, while Salazar (2005)Salazar F.H.P. 2005. Ocorrência de tuberculose causada por Mycobacterium bovis em bovinos abatidos em frigoríficos no estado de Mato Grosso, Brasil. Master’s Thesis, Universidade Federal de Mato Grosso do Sul, Campo Grande. 73p. and Furlanetto et al. (2012)Furlanetto L.V., Figueiredo E.E.S., Conte Júnior C.A., Carvalho R.C.T., Silva F.G.S., Silva J.T., Lilenbaum W. & Paschoalin V.M.F. 2012. Uso de métodos complementares na inspeção post mortem de carcaças com suspeita de tuberculose bovina. Pesq. Vet. Bras. 32(11):1138-1144. <http://dx.doi.org/10.1590/S0100-736X2012001100011>
https://doi.org/10.1590/S0100-736X201200...
reported absence of AFB in tissue samples with macroscopic lesions suggestive of tuberculosis and confirmed on histopathological examination. According to Rodriguez et al. (2004)Rodriguez C.A.R., Zumárraga M.J., Oliveira E.M.D., Cataldi A.A., Romano M.I., Otto H.H., Bonafé V.L. & Ferreira Neto J.S. 2004. Caracterização molecular de isolados de Mycobacterium bovis do Estado de São Paulo Brasil, utilizando a técnica de Spoligotyping. Arqs Inst. Biológico, São Paulo 71(3):277-282., these divergences may occur because the test reveals the presence of AFB in concentrations higher than 104 mycobacteria in a histological section.
As in the study Silva et al. (2018)Silva D.A.V.D., Siconelli M.J.L., Bürger K.P. & Keid L.B. 2018. Comparison between tests for tuberculosis diagnosis in slaughtered bovines. Arqs Inst. Biológico, São Paulo 85(1):1-8., the results obtained in the present study allowed us to infer that the clinical examination, CCTT, macroscopic post mortem inspection, histopathological examination and identification of AFB are useful tools for the diagnosis of bovine tuberculosis. The present study highlights the histopathological and histochemical analysis in tissues with lesions suggestive of bovine tuberculosis, which allows characterizing the microscopic alterations of the lesions, as well as identifying the AFB. It is also noteworthy that the study was conducted in a dairy herd, demonstrating the circulation of the disease, which poses a threat to people who come into direct contact and/or consume meat and milk from these animals. Besides, positive cattle are also a source of infection for those working in the industry, as they will be exposed to the etiological agent when handling animals or meat products.
Conclusions
The present study demonstrated that the combination of low-cost alternatives increases the diagnostic accuracy for bovine tuberculosis, contributing to the good functioning of PNCEBT and prevention of this zoonosis in public health.
The clinical examination identified symptomatic animals, the CCTT identified reactive animals with suggestive lesions in the slaughterhouse, while the histopathology of the suggestive lesions showed caseous lesions, and Ziehl-Neelsen staining showed AFBs, even in CCTT reactive cattle and without macroscopic lesions.
References
- Acha P.N. & Szyfres B. 2005. Zoonoses and communicable diseases common to man and animals: bacterioses and mycoses. 3rd ed. Pan American Health Organization, Washington. 378p.
- Andrade G.B., Riet-Correa F., Mielke P.V., Méndez M.C. & Schild A.L. 1991. Estudo histológico e isolamento de micobactérias de lesões similares á tuberculose em bovinos no Rio Grande do Sul. Pesq. Vet. Bras. 11(3):81-86.
- Asil T.A., El Sanousi S.M., Gameel A., El Beir H., Fathelrahman M., Terab N.M., Muaz M.A. & Hamid M.E. 2012. Bovine tuberculosis in South Darfur State, Sudan: an abattoir study based on microscopy and molecular detection methods. Trop. Anim. Health Prod. 45(1):469-472. <PMid:22843216>
- Brasil 2006. Manual Técnico. Programa Nacional de Controle e Erradicação da Brucelose e da Tuberculose Animal (PNCEBT), Ministério da Agricultura, Pecuária e Abastecimento, Brasilia, DF.
- Brasil 2017a. Instrução Normativa Nº 10 de 03 de março de 2017. Regulamento Técnico do Programa Nacional de Controle e Erradicação da Brucelose e Tuberculose (PNCEBT), Ministério da Agricultura, Pecuária e Abastecimento, Brasília, DF.
- Brasil 2017b. Decreto nº 9.013 de 29 de março de 2017, alterado pelo Decreto nº 9.069 de 31 de maio de 2017. Regulamento de Inspeção Industrial e Sanitária de Produtos de Origem Animal (RIISPOA), Ministério da Agricultura, Pecuária e do Abastecimento, Brasília, DF.
- Brasil 2018. Implantação do Plano Nacional pelo fim da tuberculose como problema de saúde pública no Brasil: primeiros passos rumo ao alcance das metas. Boletim Epidemiológico 49(11):1-18.
- Cassidy J.P., Bryson D.G., Pollock J.M., Evans R.T., Forster F. & Neill S.D. 1999. Lesions in cattle exposed to Mycobacterium bovis - inoculated calves. J. Comp. Pathol. 121(4):321-337. <http://dx.doi.org/10.1053/jcpa.1999.0330> <PMid:10542122>
» https://doi.org/10.1053/jcpa.1999.0330 - Collins D.M., Radford A.J., de Lisle G.W. & Billman-Jacobe H. 1994. Diagnosis and epidemiology of bovine tuberculosis using molecular biological approaches. Vet. Microbiol. 40(1):83-94. <http://dx.doi.org/10.1016/0378-1135(94)90048-5> <PMid:7915446>
» https://doi.org/10.1016/0378-1135(94)90048-5 - Corner L.A. 1994. Post mortem diagnosis of Mycobacterium bovis infection in cattle. Vet. Microbiol. 40(1):53-63. <http://dx.doi.org/10.1016/0378-1135(94)90046-9> <PMid:8073629>
» https://doi.org/10.1016/0378-1135(94)90046-9 - Corner L.A., Melville L., McCUBBIN K., Small K.J., McCORMICK B.S., Wood P.R. & Rothel J.S. 1990. Efficiency of inspection procedures for detection of tuberculous lesions in cattle. Aust. Vet. J. 67(11):389-392. <http://dx.doi.org/10.1111/j.1751-0813.1990.tb03020.x> <PMid:2085291>
» https://doi.org/10.1111/j.1751-0813.1990.tb03020.x - Corrêa W.M. & Corrêa C.N.M. 1992. Tuberculose, p.317-337. In: Corrêa W.M. & Corrêa C.N.M. (Eds), Enfermidades Infecciosas dos Mamíferos Domésticos. 2ª ed. Medsi, Rio de Janeiro.
- Demelash B., Inangolet F., Oloya J., Asseged B., Badaso M., Yilkal A. & Skjerve E. 2009. Prevalence of bovine tuberculosis in Ethiopian slaughter cattle based on post-mortem examination. Trop. Anim. Health Prod. 41(1):755-765. <http://dx.doi.org/10.1007/s11250-008-9248-9> <PMid:19058024>
» https://doi.org/10.1007/s11250-008-9248-9 - Ferreira Neto J.S., Silveira G.B., Rosa B.M., Gonçalves V.S.P., Grisi-Filho J.H.H., Amaku M. & Lage A.P. 2016. Analysis of 15 years of the National Program for the Control and Eradication of Animal Brucellosis and Tuberculosis, Brazil. Semina, Ciênc. Agrárias 37(5):3385-3402.
- Fráguas S.A., Cunha-Abreu M.S., Ferreira A.M.R., Marassi C.D., Oelemann W., Fonseca L.S., Ferreira R. & Lilenbaum W. 2008. Estudo comparativo de métodos complementares para o diagnóstico da tuberculose bovina em animais reagentes à tuberculinização. Revta Bras. Ciênc. Vet. 15(3):117-121.
- Furlanetto L.V., Figueiredo E.E.S., Conte Júnior C.A., Carvalho R.C.T., Silva F.G.S., Silva J.T., Lilenbaum W. & Paschoalin V.M.F. 2012. Uso de métodos complementares na inspeção post mortem de carcaças com suspeita de tuberculose bovina. Pesq. Vet. Bras. 32(11):1138-1144. <http://dx.doi.org/10.1590/S0100-736X2012001100011>
» https://doi.org/10.1590/S0100-736X2012001100011 - Garbaccio S.G., Delgado F.O., Zumarraga M.J., Rodriguez L.R., Huertas P.S. & Garro C.J. 2018. Diagnóstico bacteriológico de tuberculosis bovina en bovinos reactores positivos a la prueba tuberculínica. Revta Investig. Agropec. 44(1):69-75.
- Kantor I.N., De La Veja E. & Caballero P. 1981. Estudio de órganos bovinos decomisados por tuberculose, mataderos del gran Buenos Aires. Revta Med. Vet. 62(4):282-285.
- Medeiros L.S., Marassi C.D., Figueiredo E.E.S., Leite J., Ferreira A.M.R. & Lilenbaum W. 2012. Assessing the histopathology to depict the different stages of bovine tuberculosis infection in a naturally infected herd. Pesq. Vet. Bras. 32(2):135-139. <http://dx.doi.org/10.1590/S0100-736X2012000200008>
» https://doi.org/10.1590/S0100-736X2012000200008 - Monaghan M.L., Doherty M.L., Collins J.D., Kazda J.F. & Quinn P.J. 1994. The tuberculin test. Vet. Microbiol. 40(1):111-124. <http://dx.doi.org/10.1016/0378-1135(94)90050-7> <PMid:8073619>
» https://doi.org/10.1016/0378-1135(94)90050-7 - Paes A.C. & Franco M.M.J. 2016. Tuberculose em animais de produção, p.512-542. In: Paes A.C. & Franco M.M.J. (Eds), Doenças Infecciosas em Animais de Produção e Companhia. Roca, Rio de Janeiro.
- Pinto P.S.A., Viloria M.I.V. & Faria J.E. 2004. Avaliação do desempenho dos exames anatomopatológico e histopatológico na inspeção post mortem de bovinos suspeitos ou reagentes à prova de tuberculinização. Revta Bras. Ciênc. Vet. 11(1):27-31.
- Radostits O.M., Gay C.C., Hinchcliff K.W. & Constable P.D. 2007. Veterinary Medicine: a textbook of the diseases of cattle, horses, sheep, pigs, and goats. 10th ed. W.B. Saunders, Philadelphia. 2156p.
- Reis D.O., Almeida L. & Faria A.R. 1995. Estudo comparativo entre linfossarcoma, tuberculose e linfadenites inespecíficas ocorridas em bovinos abatidos e a confirmação histológica. Higiene Alimentar 9(35):28-30.
- Riojas M.A., McGough K.J., Rider-Riojas C.J., Rastogi N. & Hazbón M.H. 2018. Phylogenomic analysis of the species of the Mycobacterium tuberculosis complex demonstrates that Mycobacterium africanum, Mycobacterium bovis, Mycobacterium caprae, Mycobacterium microti and Mycobacterium pinnipedii are later heterotypic synonyms of Mycobacterium tuberculosis Int. J. Syst. Evol. Microbiol. 68(1):324-332. <http://dx.doi.org/10.1099/ijsem.0.002507> <PMid:29205127>
» https://doi.org/10.1099/ijsem.0.002507 - Rodriguez C.A.R., Zumárraga M.J., Oliveira E.M.D., Cataldi A.A., Romano M.I., Otto H.H., Bonafé V.L. & Ferreira Neto J.S. 2004. Caracterização molecular de isolados de Mycobacterium bovis do Estado de São Paulo Brasil, utilizando a técnica de Spoligotyping. Arqs Inst. Biológico, São Paulo 71(3):277-282.
- Romero R.E., Garzón D.L., Mejía G.A., Monroy W., Patarroyo M.E. & Murillo L.A. 1999. Identification of Mycobacterium bovis in bovine clinical samples by PCR species-specific primers. Canadian J. Vet. Res. 63(2):101-106. <PMid:10369566>
- Roxo E. 1996. Tuberculose bovina: revisão [Bovine tuberculosis: review]. Arqs Inst. Biológico, São Paulo 63(2):91-97.
- Roxo E. 1997. M. bovis como causa de zoonose. Revta Bras. Ciênc. Farmac. 18(1):101-108.
- Salazar F.H.P. 2005. Ocorrência de tuberculose causada por Mycobacterium bovis em bovinos abatidos em frigoríficos no estado de Mato Grosso, Brasil. Master’s Thesis, Universidade Federal de Mato Grosso do Sul, Campo Grande. 73p.
- Silva D.A.V.D., Siconelli M.J.L., Bürger K.P. & Keid L.B. 2018. Comparison between tests for tuberculosis diagnosis in slaughtered bovines. Arqs Inst. Biológico, São Paulo 85(1):1-8.
- Souza A.V.D., Sousa C.F.A., Souza R.M.D., Ribeiro R.M.P. & Oliveira A.D.L. 1999. A importância da tuberculose bovina como zoonose. Higiene Alimentar 13(59):22-27.
- Souza M.A.D., Bombonato N.G., Soares P.M., Ramos G.B., Castro I.P., Medeiros A.A. & Lima A.M.C. 2016. Exames complementares no diagnóstico da tuberculose em bovinos reagentes à tuberculinização comparada. Arqs Inst. Biológico, São Paulo, 83(1):1-8.
- Vitale F., Capra G., Maxia L., Reale S., Vesco G. & Caracappa S. 1998. Detection of Mycobacterium tuberculosis complex in cattle by PCR using milk, lymph node aspirates and nasal swabs. J. Clin. Microbiol. 36(4):1050-1055. <http://dx.doi.org/10.1128/JCM.36.4.1050-1055.1998> <PMid:9542936>
» https://doi.org/10.1128/JCM.36.4.1050-1055.1998 - WHO 2017. Bending the curve: ending TB. Annual report 2017, World Health Organization, Geneva. 72p. Available at <Available at http://apps.who.int/iris/handle/10665/254762 > Access on Aug. 1, 2018.
» http://apps.who.int/iris/handle/10665/254762
Publication Dates
-
Publication in this collection
03 Apr 2020 -
Date of issue
Jan 2020
History
-
Received
28 June 2019 -
Accepted
10 July 2019