ABSTRACT:
We aimed to determine the concentration of MMP-2 and IL-1β in the aqueous humor of dogs with healthy eyes (n=8) and in those with mature (n=8) and hyper mature cataracts (n=8). Correlations between cytokines, cytokines, and intraocular pressure (IOP), as well as cytokines with ages of patients of each group, were also assessed. In patients with cataract, aqueous humor was collected at the end of the electroretinographic procedure. In healthy dogs, aqueous humor was collected before elective surgeries. Cytokine levels were determined using ELISA. IOP was assessed by applanation tonometry. IOP of patients with mature and hyper mature cataracts were lower than the ones measured in normal eyes (P=0.158). MMP-2 aqueous humor concentration was higher in patients with hyper mature cataracts, in comparisons with healthy patients (P=0.04). Average IL-1 β aqueous concentration was higher in patients with cataracts (P<0.0001). Significant higher values of IL-1 β were observed in patients with hyper mature, than in the ones with mature cataracts (P=0.0085). Correlations between MMP-2 and IL-1 β (r=-0.38, P=0.06), MMP-2 and IOP (r=-0.149, P=0.484), and IL-1 β and the ages of patients were not observed (P>0.05). IL-1 β and IOP correlated negatively (r=-0.42, P=0.04). MMP-2 and the ages of patients correlated only in dogs with mature cataracts (r=0.772, P=0.02). It can be concluded that the increment in the aqueous humor concentration of IL-1 β found in dogs with mature and hyper mature cataracts, in addition to the negative relationship of this cytokine with IOP, suggests that IL-1 β is involved in the pathogenesis of LIU. Despite dogs with hypermature cataracts presented significant higher concentrations of MMP-2 in the aqueous humor, such cytokine did not correlate with IL-1 β and IOP. In our study, a relationship between aqueous humor cytokines and the ages of patients was only confirmed between MMP-2 and the ages of dogs with mature cataracts.
INDEX TERMS:
Metalloproteinase-2; interleukin-1 β; dogs; cataractous; eyes; lens-induced uveitis; inflammatory cytokines; capsular fibrosis; lens
RESUMO:
Objetivou-se determinar as concentrações da metalloprotease-2 (MMP-2) e de interleucina-1 β (IL-1 β) em cães com olhos saudáveis (n=8) e naqueles com catarata madura (n=8) e hipermatura (n=8). Correlações entre ambas as citocinas, entre as citocinas e a pressão intraocular (PIO), assim como entre as citocinas e a idade dos pacientes dentro de cada grupo foram averiguadas. Nos pacientes com catarata, o humor aquoso foi colhido ao final da eletrorretinografia. Nos cães saudáveis, o humor aquoso foi colhido antes do início de cirurgias eletivas. Os níveis das citocinas foram determinados por ELISA e a PIO por tonometria de aplanação. A PIO dos pacientes com catarata madura e hipermadura foram mais baixas que aquelas dos pacientes controle (P=0.158). A concentração de MMP-2 no humor aquoso foi maior nos pacientes com catarata hipermtura, comparativamente aos pacientes saudáveis (P=0.04). A concentração de IL-1 β no humor aquoso foi mais elevada nos cães com catarata (P<0.0001). Nos pacientes com catarata hipermatura, os valores de IL-1 β foram significativamente mais altos que aqueles dosados nos pacientes com catarata madura (P=0.0085). Correlações entre MMP-2 e IL-1 β (r=-0.38, P=0.06), MMP-2 e PIO (r=-0.149, P=0.484) e IL-1 β e as idades dos pacientes não foram observadas (P>0.05). A IL-1 β se correlacionou negativamente com a PIO (r=-0.42, P=0.04). Correlação entre MMP-2 e a idades dos pacientes foi observada apenas nos cães com catarata madura (r=0.772, P=0.02). Conclui-se que o aumento na concentração de IL-1 β no humor aquoso de cães com catarata madura e hipermatura, associado à correlação negativa entre essa citocina e a PIO, sugerem que a mesma está envolvida na patogênese da uveíte induzida pela lente. Apesar dos cães com catarata hipermadura apresentarem concentrações significativamente maiores de MMP-2 no humor aquoso, essa citocina não se correlacionou com a IL-1 β e a PIO. Em nosso estudo, correlação entre as citocinas dosadas no humor aquoso e a idade dos pacientes foi confirmada apenas entre MMP-2 e a idade dos cães com catarata madura.
TERMOS DE INDEXAÇÃO:
Metaloprotease-2; interleucina-1 β; cães; olhos cataratoso; uveíte induzida pela lente; citocinas inflamatórias; fibrose capsular; lente
Introduction
Cataract is defined the opacity of the eye lens, and is considered to be the leading cause of blindness in dogs. The affection can be classified based on the age of onset (congenital, juvenile, or senile), position in the lens (capsular, cortical, nuclear, axial, equatorial, or total), degree and stage of opacification (incipient, immature, mature, or hypermature), and the possible cause (inherited, traumatic, metabolic, or toxic) (Davidson & Nelms 2013Davidson M.G. & Nelms S.R. 2013. Diseases of the lens and cataract formation, p.1199-1233. In: Gelatt K.N. & Gilger B.C. (Eds.), Veterinary Ophthalmology. 5th ed. Ames, Wiley-Blackwell., Renzo et al. 2014Renzo R., Ribeiro A.P., Silva M.L., Silva G.A., Ortencio K.P., Sobrinho A.A.F.B., Mineo T.W. & Laus J.L. 2014. Intraocular pressure, specular microscopy, and prostaglandin E2 concentration in dogs with mature and hypermature cataract. Vet. Ophthalmol. 17(4):280-285.).
As the pathologic alterations associated with cataract progresses, hydrolytic and proteolytic enzyme activity increases within the lens. This process results in rupture of cell membranes, loss of low molecular weight proteins, and an increase in water content. Further degradation of proteins into amino acids and polypeptides allows small products of proteolysis to diffuse from the lens (Davidson & Nelms 2013Davidson M.G. & Nelms S.R. 2013. Diseases of the lens and cataract formation, p.1199-1233. In: Gelatt K.N. & Gilger B.C. (Eds.), Veterinary Ophthalmology. 5th ed. Ames, Wiley-Blackwell.). The loss of water and nitrogenous materials may cause the lens to shrink as the cataract progresses to the hypermature stage (Davidson & Nelms 2013Davidson M.G. & Nelms S.R. 2013. Diseases of the lens and cataract formation, p.1199-1233. In: Gelatt K.N. & Gilger B.C. (Eds.), Veterinary Ophthalmology. 5th ed. Ames, Wiley-Blackwell.). Such diffusion of lens material into the aqueous humor is responsible for the inflammation of the iris and the ciliary body. An immune-mediated inflammatory response against lens antigens occurs after deviation of the normal low level of T-cell-mediated tolerance to lens proteins (Van der Woerdt 2000Van der Woerdt A. 2000. Lens-induced uveits. Vet. Ophthalmol. 3(4):227-234., Davidson & Nelms 2013Davidson M.G. & Nelms S.R. 2013. Diseases of the lens and cataract formation, p.1199-1233. In: Gelatt K.N. & Gilger B.C. (Eds.), Veterinary Ophthalmology. 5th ed. Ames, Wiley-Blackwell.). This process is called lens-induced uveitis (LIU), which is considered to be the most frequent type of anterior uveitis in dogs. LIU has been associated with all stages of cataract maturity in this species (Van der Woerdt 2000Van der Woerdt A. 2000. Lens-induced uveits. Vet. Ophthalmol. 3(4):227-234., Renzo et al. 2014Renzo R., Ribeiro A.P., Silva M.L., Silva G.A., Ortencio K.P., Sobrinho A.A.F.B., Mineo T.W. & Laus J.L. 2014. Intraocular pressure, specular microscopy, and prostaglandin E2 concentration in dogs with mature and hypermature cataract. Vet. Ophthalmol. 17(4):280-285.). Dogs with LIU, as those with uveitis due to other causes, present with lower intraocular pressure (IOP) because of the increased amounts of prostaglandins secreted by the inflammatory cells (Leasure et al. 2001Leasure J., Gelatt K.N. & Mackay E.O. 2001. The relationship of cataract maturity to intraocular pressure in dogs. Vet. Ophthalmol. 4(4):273-276., Renzo et al. 2014Renzo R., Ribeiro A.P., Silva M.L., Silva G.A., Ortencio K.P., Sobrinho A.A.F.B., Mineo T.W. & Laus J.L. 2014. Intraocular pressure, specular microscopy, and prostaglandin E2 concentration in dogs with mature and hypermature cataract. Vet. Ophthalmol. 17(4):280-285.).
Pro-inflammatory cytokines, such as interleukin-1 alpha (IL-1α) and interleukin-1 beta (IL-1 β), have been shown to actively recruit leukocytes to sites of inflammation. Both IL-1 α and β are predominantly secreted by activated macrophages, B lymphocytes, and endothelial cells (Chiou et al. 1994Chiou G.C., Yao Q.S. & Okawara T. 1994. Prevention of ocular inflammation induced by lens proteins, endotoxin, and interleukin-1 with synthetic interleukin-1 blockers. J. Ocul. Pharmacol. Ther. 10(3):577-586. , Medeiros et al. 2008Medeiros R., Rodrigues G.B., Figueiredo C.P., Rodrigues E.B., Grumman A. Jr., Menezes-de-Lima O. Jr., Passos G.F. & Calixto J.B. 2008. Molecular mechanisms of topical anti-inflammatory effects of lipoxin A(4) in endotoxin-induced uveitis. Mol. Pharmacol. 74(1):154-161., Ortencio et al. 2015Ortencio K.P., Renzo R., Sobrinho A.A.F.B., Kobashigawa K.K., Crivelaro R.M., Silva G.A., Aldrovani M., Ribeiro A.P., Mineo T.W. & Laus J.L. 2015. Effects of morphine on the expression of cytokines and inflammatory mediators in a rabbit model of endotoxin-induced experimental uveitis. Arq. Bras. Oftalmol. 78(6):371-375.). In animals and humans, IL-1 β seems to play a role in the pathogenesis of uveitis (Chiou et al. 1994Chiou G.C., Yao Q.S. & Okawara T. 1994. Prevention of ocular inflammation induced by lens proteins, endotoxin, and interleukin-1 with synthetic interleukin-1 blockers. J. Ocul. Pharmacol. Ther. 10(3):577-586. , Nishi et al. 1994Nishi O., Nishi K. & Ohmoto Y. 1994. Effect of interleukin 1 receptor antagonist on the blood-aqueous barrier after intraocular lens implantation. Brit. J. Ophthalmol. 78(12):917-920., El-Shabrawiet al. 2000El-Shabrawi Y.G., Christen W.G. & Foster S.C. 2000. Correlation of metalloproteinase-2 and -9 with proinflammatory cytokines interleukin-1b, interleukin-12 and the interleukin-1 receptor antagonist in patients with chronic uveitis. Curr. Eye Res. 20(3):211-214., Medeiros et al. 2008Medeiros R., Rodrigues G.B., Figueiredo C.P., Rodrigues E.B., Grumman A. Jr., Menezes-de-Lima O. Jr., Passos G.F. & Calixto J.B. 2008. Molecular mechanisms of topical anti-inflammatory effects of lipoxin A(4) in endotoxin-induced uveitis. Mol. Pharmacol. 74(1):154-161., Ortencio et al. 2015Ortencio K.P., Renzo R., Sobrinho A.A.F.B., Kobashigawa K.K., Crivelaro R.M., Silva G.A., Aldrovani M., Ribeiro A.P., Mineo T.W. & Laus J.L. 2015. Effects of morphine on the expression of cytokines and inflammatory mediators in a rabbit model of endotoxin-induced experimental uveitis. Arq. Bras. Oftalmol. 78(6):371-375., Wang et al. 2016Wang L., Zhang Z., Koch D.D., Jia Y., Cao W. & Zhang S. 2016. Anterior chamber interleukin 1β, interleukin 6 and prostaglandin E2 in patients undergoing femtosecond laser-assisted cataract surgery. Brit. J. Ophthalmol. 100(4):579-582., Aketa et al. 2017Aketa N., Yamaguchi T., Suzuki T., Higa K., Yagi-Yaguchi Y., Satake Y., Tsubota K. & Shimazaki J. 2017. Iris damage is associated with elevated cytokine levels in aqueous humor. Invest. Ophthalmol. Vis. Sci. 58(6):42-51.). One study demonstrated that the concentration of IL-1 β in the aqueous humor is correlated with the degree of iris damage in human patients who have undergone laser iridotomies and intraocular surgical procedures (Aketa et al. 2017Aketa N., Yamaguchi T., Suzuki T., Higa K., Yagi-Yaguchi Y., Satake Y., Tsubota K. & Shimazaki J. 2017. Iris damage is associated with elevated cytokine levels in aqueous humor. Invest. Ophthalmol. Vis. Sci. 58(6):42-51.). Another study reported that the concentration of IL-1 β in the aqueous humor was significantly increased in humans who have undergone femtosecond laser assisted cataract surgery, in comparison with the ones operated without laser assistance (Wang et al. 2016Wang L., Zhang Z., Koch D.D., Jia Y., Cao W. & Zhang S. 2016. Anterior chamber interleukin 1β, interleukin 6 and prostaglandin E2 in patients undergoing femtosecond laser-assisted cataract surgery. Brit. J. Ophthalmol. 100(4):579-582.). With regard to veterinary ophthalmology, no studies have focused on quantifying the level of this cytokine in the aqueous humor of dogs with uveitis.
Matrix metalloproteinases (MMPs) are proteolytic enzymes capable of degrading the extracellular matrix (ECM) that is present in almost every tissue in the body. Such enzymes are involved in many ocular physiological processes, including embryogenesis, angiogenesis, and wound healing (Di Girolamo et al. 1996Di Girolamo N., Verma M.J., McCluskey P.J., Lloyd A. & Wakefield D. 1996. Increased matrix metalloproteinases in the aqueous humor of patients and experimental animals with uveitis. Curr. Eye Res. 15(10):1060-1068., Hodgkinson et al. 2007Hodgkinson L.M., Duncan G., Wang L., Pennington C.J., Edwards D.R. & Wormstone I.M. 2007. MMP and TIMP expression in quiescent, dividing, and differentiating human lens cells. Invest. Ophthalmol. Vis. Sci. 48(9):4192-4199., Weinstein et al. 2007Weinstein W.L., Dietrich U.M., Sapienza J.S., Carmichael K.P., Moore P.A. & Krunkosky T.M. 2007. Identification of ocular matrix metalloproteinases present within the aqueous humor and iridocorneal drainage angle tissue of normal and glaucomatous canine eyes. Vet. Ophthalmol. 10(Suppl.1):108-116., Ribeiro et al. 2012Ribeiro A.P., Silva M.L., Araújo R.L., Ferrucci D.L., Mineo T., Thiesen R., Bandarra M.B. & Laus JL. 2012. Expression of matrix metalloproteinases, type IV collagen, and interleukin-10 in rabbits treated with morphine after lamellar keratectomy. Vet. Ophthalmol. 15(3):153-163., Jia et al. 2014Jia Y., Hu D.N., Zhu D., Zhang L., Gu P., Fan X. & Zhou J. 2014. MMP-2, MMP-3, TIMP-1, TIMP-2, and TIMP-3 protein levels in human aqueous humor: relationship with axial length. Invest. Ophthalmol. Vis. Sci. 55(6):3922-3928.). These enzymes are also up-regulated in several pathologic ocular disorders, including cataracts (Tamiya et al. 2000Tamiya S., Wormstone I.M., Marcantonio J.M., Gavrilovic J. & Duncan G. 2000. Induction of matrix metalloproteinases 2 and 9 following stress to the lens. Exp. Eye Res. 71(6):591-597., West-Mays 2007West-Mays J.A. 2007. Matrix metalloproteinases as mediators of primary and secondary cataracts. Expert Rev. Ophthalmol. 2(6):931-938., Alapure et al. 2012Alapure B.V., Praveen M.R., Gajjar D.U., Vasavada A.R., Parmar T.J. & Arora A.I. 2012. Matrix metalloproteinase-2 and -9 activities in the human lens epithelial cells and serum of steroid induced posterior subcapsular cataracts. Mol. Vis. 18:64-73., Eldred et al. 2012Eldred J.A., Hodgkinson L.M., Dawes L.J., Reddan J.R., Edwards D.R. & Wormstone I.M. 2012 MMP-2 activity is critical for TGF beta-2-induced matrix contraction: implications for fibrosis. Invest. Ophthalmol. Vis. Sci. 53(7):4085-4098.). MMPs are capable of denaturing most components of the lens capsule ECM; hence, they can facilitate lens fiber migration and differentiation into myofibroblasts, which are responsible for the contraction and opacification of the anterior and posterior capsule of the lens (West-Mays 2007West-Mays J.A. 2007. Matrix metalloproteinases as mediators of primary and secondary cataracts. Expert Rev. Ophthalmol. 2(6):931-938., Eldred et al. 2012Eldred J.A., Hodgkinson L.M., Dawes L.J., Reddan J.R., Edwards D.R. & Wormstone I.M. 2012 MMP-2 activity is critical for TGF beta-2-induced matrix contraction: implications for fibrosis. Invest. Ophthalmol. Vis. Sci. 53(7):4085-4098.). Higher activity of MMP-2 and MMP-9 was detected in both the lens epithelial cells and the serum of human patients with steroid-induced posterior capsular cataract (Alapure et al. 2012Alapure B.V., Praveen M.R., Gajjar D.U., Vasavada A.R., Parmar T.J. & Arora A.I. 2012. Matrix metalloproteinase-2 and -9 activities in the human lens epithelial cells and serum of steroid induced posterior subcapsular cataracts. Mol. Vis. 18:64-73.). In normal human lenses, gene expression (Hodgkinson et al. 2007Hodgkinson L.M., Duncan G., Wang L., Pennington C.J., Edwards D.R. & Wormstone I.M. 2007. MMP and TIMP expression in quiescent, dividing, and differentiating human lens cells. Invest. Ophthalmol. Vis. Sci. 48(9):4192-4199.) and concentration (Tamiya et al. 2000Tamiya S., Wormstone I.M., Marcantonio J.M., Gavrilovic J. & Duncan G. 2000. Induction of matrix metalloproteinases 2 and 9 following stress to the lens. Exp. Eye Res. 71(6):591-597., Sachdev et al. 2004Sachdev N.H., Di Girolamo N., Nolan T.M., McCluskey P.J., Wakefield D. & Coroneo M.T. 2004. Matrix metalloproteinases and tissue inhibitors of matrix metalloproteinases in the human lens: implications for cortical cataract formation. Invest. Ophthalmol. Vis. Sci. 45(11):4075-4078.) of MMP-2 and -9 are very low, whereas the expression of tissue inhibitor of MMPs (TIMPs) is high. Even so, MMPs can be detected in the aqueous humor of humans with and without cataracts (Di Girolamo et al. 1996Di Girolamo N., Verma M.J., McCluskey P.J., Lloyd A. & Wakefield D. 1996. Increased matrix metalloproteinases in the aqueous humor of patients and experimental animals with uveitis. Curr. Eye Res. 15(10):1060-1068.).
In humans with uveitis of different causes and in rabbits with experimentally induced anterior uveitis, concentrations of MMPs in the aqueous humor are elevated in comparison to controls (Di Girolamo et al. 1996Di Girolamo N., Verma M.J., McCluskey P.J., Lloyd A. & Wakefield D. 1996. Increased matrix metalloproteinases in the aqueous humor of patients and experimental animals with uveitis. Curr. Eye Res. 15(10):1060-1068., Määtää et al. 2006Määtää M., Tervahartiala T., Vesti E., Airaksinen J. & Sorsa T. 2006. Levels and activation of matrix metalloproteinases in aqueous humor are elevated in uveitis-related secondary glaucoma. J. Glaucoma 15(3):229-237., Bauer et al. 2018Bauer D., Kasper M., Walscheid K., Koch J.M., Müther P.S., Kirchhof B., Heiligenhaus A. & Heinz C. 2018. Multiplex cytokine analysis of aqueous humor in juvenile idiopathic arthritis-associated anterior uveitis with or without secondary glaucoma. Front. Immunol. 9(5):708.). Weinstein et al. (2007)Weinstein W.L., Dietrich U.M., Sapienza J.S., Carmichael K.P., Moore P.A. & Krunkosky T.M. 2007. Identification of ocular matrix metalloproteinases present within the aqueous humor and iridocorneal drainage angle tissue of normal and glaucomatous canine eyes. Vet. Ophthalmol. 10(Suppl.1):108-116. showed that only MMP-2 in its latent form was present in the aqueous humor of healthy dogs. One study showed there were no expressions of MMP-3 and -9 in the aqueous humor of dogs, before and after experimentally-induced uveitis (Pinard et al. 2011Pinard C.L., Gauvin D., Moreau M., Martel-Pelletier J., Pelletier J.P. & Troncy E. 2011. Measurements of canine aqueous humor inflammatory mediators and the effect of carprofen following anterior chamber paracentesis. Vet. Ophthalmol. 14(5):296-303.).
Identifying possible chemical mediators involved in the pathogenesis of anterior uveitis and LIU is important because could have direct therapeutic implications, and the knowledge can be used to minimize the potential vision-threatening sequelae that result from intraocular inflammation (Chiou et al. 1994Chiou G.C., Yao Q.S. & Okawara T. 1994. Prevention of ocular inflammation induced by lens proteins, endotoxin, and interleukin-1 with synthetic interleukin-1 blockers. J. Ocul. Pharmacol. Ther. 10(3):577-586. , Pinard et al. 2011Pinard C.L., Gauvin D., Moreau M., Martel-Pelletier J., Pelletier J.P. & Troncy E. 2011. Measurements of canine aqueous humor inflammatory mediators and the effect of carprofen following anterior chamber paracentesis. Vet. Ophthalmol. 14(5):296-303.). Aqueous humor concentrations of prostaglandin E2 (PGE2), tumor necrosis factor-α, cicloxigenase-2, nitric oxide synthase, nitrites, nitrates, MMP-3 and -9 have been studied in dogs subjected to experimental breakdown of the blood-aqueous barrier (Ribeiro et al. 2010Ribeiro A.P., Escobar A., Motheo T.F., Sakamoto C.A.M., Godoy G.S., Costa M.A.N. & Laus J.L 2010. Effects of carprofen administered by different routes to control experimental uveitis in dogs. Arq. Bras. Med. Vet. Zootec. 62(2):309-317., Pinard et al. 2011Pinard C.L., Gauvin D., Moreau M., Martel-Pelletier J., Pelletier J.P. & Troncy E. 2011. Measurements of canine aqueous humor inflammatory mediators and the effect of carprofen following anterior chamber paracentesis. Vet. Ophthalmol. 14(5):296-303., Gilmour et al. 2012Gilmour M.A. & Payton M.E. 2012. Comparison of the effects of IV administration of meloxicam, carprofen, and flunixin meglumine on prostaglandin E2 concentration in aqueous humor of dogs with aqueocentesis-induced anterior uveitis. Am. J. Vet. Res. 73(5):698-703.). Nonetheless, to date, the only cytokines dosed in the aqueous humor of dogs with cataracts were the vascular endothelial growth factor and PGE2 (Abrams et al. 2011Abrams K.L., Stabila P.F., Kauper K. & Elliott S. 2011. Vascular endothelial growth factor in diabetic and nondiabetic canine cataract patients. Vet. Ophthalmol. 14(2):93-99., Sandberg et al. 2012Sandberg C.A., Herring I.P., Huckle W.R., LeRoith T., Pickett J.P. & Rossmeisl J.H. 2012. Aqueous humor vascular endothelial growth factor in dogs: association with intraocular disease and the development of pre-iridal fibrovascular membrane. Vet. Ophthalmol. 15(Suppl.1):21-30., Renzo et al. 2014Renzo R., Ribeiro A.P., Silva M.L., Silva G.A., Ortencio K.P., Sobrinho A.A.F.B., Mineo T.W. & Laus J.L. 2014. Intraocular pressure, specular microscopy, and prostaglandin E2 concentration in dogs with mature and hypermature cataract. Vet. Ophthalmol. 17(4):280-285.). Therefore, we aimed to evaluate the concentrations of MMP-2 and IL-1 β in the aqueous humor of healthy dogs and those with cataracts at different stages. In addition, correlations between MMP-2 and IL-1 β, and between both cytokines and IOP, as well as both cytokines and the ages of patients were assessed.
Materials and Methods
Approval from the “Universidade Federal do Mato Grosso”, Ethics Committee for the Use of Animals in Research, as well as the consent from the patient owners were obtained prior to the experiment (23108.084384/2015-05). Male and female dogs of different breeds, with ages ranging from 6 to 13 years, which were referred to the Veterinary Ophthalmology Service of the University of Mato Grosso for cataract surgery, were enrolled in the study.
Patients underwent a full ophthalmological evaluation, which include Schirmer’s test (Schirmer’s Tear Test Strips, Ophthalmos, São Paulo, Brazil), biomicroscopy (XL-1 Slitlamp, Shin-Nippon, Japan), digital applanation tonometry (Tono Pen XL; Medtronic, Jacksonville/FL, USA), indirect binocular ophthalmoscopy (Oftalmoscopio binocular indireto FOH-5 - Eyetec S.A, São Carlos/SP, Brazil), fluorescein dye test (Fluorescein strips; Ophthalmos Ltda, São Paulo/SP, Brazil), ultrasound imaging (Ultra Scan, Alcon, Irvine/CA, USA), and flash electroretinography (Handheld Multi-species Electro- RetinoGraph; RetVetCorp, Columbia/MO, USA).
The criteria used to classify the stage of cataract maturity were described in the veterinary literature (Davidson & Nelms 2013Davidson M.G. & Nelms S.R. 2013. Diseases of the lens and cataract formation, p.1199-1233. In: Gelatt K.N. & Gilger B.C. (Eds.), Veterinary Ophthalmology. 5th ed. Ames, Wiley-Blackwell.). Only individuals with no ocular abnormalities other than mild LIU (no flare, mild to moderate conjunctival hyperemia and IOP ≤12mmHg), with mature (n=8) or hypermature (n=8) senile cataract, which were not under local or systemic steroidal or nonsteroidal anti-inflammatory therapy, were selected. Dogs with diabetic cataracts were not included in this study. A control group (n=8) composed of healthy dogs subjected to elective ovariohysterectomy (n=5) or orchiectomy (n=3), with ages ranging from 8 to 12 years, were also included. Dogs were included in the control group if no cataracts or other abnormalities were detected on ophthalmological evaluation. In addition, to be selected for the study, both healthy and cataractous dogs should have heart and respiratory rates, capillary refill time, temperature, skin turgor, bloodwork, and basic chemistry profile within the reference range for the species.
Anterior chamber paracentesis was performed under general anesthesia as previously described (Ribeiro et al. 2010Ribeiro A.P., Escobar A., Motheo T.F., Sakamoto C.A.M., Godoy G.S., Costa M.A.N. & Laus J.L 2010. Effects of carprofen administered by different routes to control experimental uveitis in dogs. Arq. Bras. Med. Vet. Zootec. 62(2):309-317., Gilmour et al. 2012Gilmour M.A. & Payton M.E. 2012. Comparison of the effects of IV administration of meloxicam, carprofen, and flunixin meglumine on prostaglandin E2 concentration in aqueous humor of dogs with aqueocentesis-induced anterior uveitis. Am. J. Vet. Res. 73(5):698-703.) and 0.2mL of aqueous humor was slowly aspirated. In patients with cataract, the aqueous humor was collect at the end of the eletroretinographic examination. The aqueous humor of dogs was not sampled in dogs that had abnormal ERG readings. In control healthy patients, aqueous humor was collected before the beginning of the aforementioned procedures. In all groups, propofol (10mg/kg) was injected intravenously to effect for anesthetic induction (Propovan®, Cristália, São Carlos/SP, Brazil). In control healthy patients, isoflurane (1.5V%) was used for maintenance of anesthesia (Isofluorane; BioChimico, Itatiaia, Rio de Janeiro/RJ, Brazil), and analgesic drugs were administered as deemed necessary by the anesthesiologist after the aqueous humor had been aspirated. After paracentesis, the eyes of patients of each group were treated with one drop of the following medications: 1% atropine (q 12h), 0.3% tobramycine (q 6h), and 1% prednisolone (q 6h), for 48 hours. The aqueous samples from all dogs were transferred to microtubes that were labeled (Tubos criogênicos, 2mL, Adamo, Brazil), and frozen at -80oC until cytokine quantitation was performed.
In all groups, the IOPs that were considered for statistical analyses were measured in the morning of the day the aqueous humor was collected. We believe that this strategy minimizes the influences of diurnal fluctuation of IOP and obtained measurements that closely reflect this parameter at the time of aqueous humor sampling.
Concentrations of MMP-2 and IL-1 β in the aqueous humor were measured using a sandwich ELISA by means of paired cytokine-specific monoclonal antibodies to canine total MMP-2 (Quantikine® ELISA form total MMP-2 Immunoassay, RnDSystems, USA) and IL-1β (Canine Interleukin-1 β ELISA Kit®, Sigma Aldrich/Merck, Germany) according to the manufacturer’s instructions. Known concentrations of recombinant MMP-2 and IL-1 β were added to each reaction, and the absorbance at 450 nanometers was read. The lower detection limits of the assays for MMP-2 and IL-1 β were 0.033ng/mL and 1pg/mL, respectively. Absorbance values read for MMP-2 and IL-1 β were converted to ng/mL and pg/mL, respectively; the estimated results were obtained from four-parameter logistic curves (http://www.myassays.com). None of the tested samples was thawed more than once.
Statistical analysis was performed using Prism version 7.04 (GraphPad Software, Inc., La Jolla, California, USA). Data were tested for normal distribution using the Shapiro-Wilk test. The concentrations of MMP-2 in the aqueous humor of healthy and cataractous eyes were compared using the Kruskal-Wallis test, followed by the Dunn test for multiple comparisons. Comparisons between healthy and cataractous eyes with respect to the concentration of IL-1 β in the aqueous humor were performed using the one way analysis of variance (ANOVA), followed by the Bonferroni test for multiple comparisons. ANOVA was used to assess differences in IOP between healthy and cataractous dogs; the same test was employed to assess differences between the ages. The Spearman´s test was employed to assess possible correlations between the concentrations of MMP-2 and IL-1 β, and between MMP-2 and IOP. The Person´s test was used to verify possible correlations between IL-1 β and IOP, and between cytokines and the ages of patients. On all occasions, a P value < 0.05 was considered statistically significant. Results are shown as mean ± standard deviation (SD) for parametric values, and as median and interquartile range for non-parametric values.
Results
The ages of healthy controls, and those with mature and hypermature cataracts, was 9.75±2.43, 7.65±1.76, and 9.37±2.50 years, respectively (P=0.158). Breeds of selected dogs with cataracts were Poodle (n=8), Labrador (n=1), mixed breed (n=2), Pincher (n=3), Lhasa Apso (n=1), and Maltese (n=1). The breeds of healthy controls were York Shire (n=2), mixed breed (n=3), Shih Tzu (n=2), and Pincher (n=1). The IOP of control healthy dogs was 13.50±3.25mmHg. The IOP of patients with mature (-1.00mmHg) and hypermature (-2.87mmHg) cataracts were lower than the ones measured in healthy controls (Table 1). However, significant differences were not observed among the groups (P=0.158).
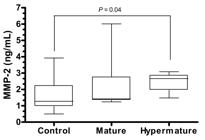
Median concentration of MMP-2 in the aqueous humor of healthy dogs was 1.67 (0.49-3.92) ng/mL. Median concentration of MMP-2 in the aqueous humor was 0.15 and 1.39ng/mL higher in patients with mature and hypermature cataracts, respectively, compared to healthy controls (Table 1, Fig.1). However, significant differences occurred only between healthy patients and those with hypermature cataracts (P=0.04) (Table 1, Fig.1).
Box-and-whisker plots of matrix metalloproteinase-2 (MMP-2) concentrations in the aqueous humor in studied dogs. Each box represents the first and third quartiles, the horizontal line in each box represents the median value, and the whiskers represent the smallest and largest sample value within 1.5 times the interquartile range.
The mean concentration of IL-1 β in the aqueous humor of healthy dogs was 5.95±0.02pg/mL. The concentration of IL-1β was significantly higher in patients with mature and hypermature cataracts than in healthy controls (P<0.0001) (Table 1, Fig.2). Significantly higher IL-1β concentrations were observed in patients with hypermature cataract, than in those with mature cataracts (P=0.0085) (Table 1, Fig.2).
Mean (line) and individual dispersion (circles) of interleukin 1 β (IL-1 β) concentrations in the aqueous humor in studied dogs.
The concentration of IL-1β did not correlate with the ages of dogs in any studied group (P>0.05). Regarding MMP-2, such cytokine also did not correlate with age in controls, and in dogs with hypermature cataracts (P>0.05). However, positive correlation was observed between MMP-2 and the ages of dogs with mature cataracts (r=0.772, P=0.02). Although there was a tendency toward a negative correlation between the concentrations of MMP-2 and IL-1 β (r=-0.38), statistical significance was not reached (P=0.06) (Fig.3). MMP-2 and IOP also did not correlate to each other (r=-0.149, P=0.484). However, IL-1 β and IOP correlated negatively (r=-0.42, P=0.04). (Fig.4).
Linear regression curve between matrix metalloproteinase-2 (MMP-2) and interleukin 1 β (IL-1 β) concentrations in the aqueous humor in the studied dogs. Sperman´s correlation test (r=-0.38, P=0.06)
Linear regression curves between both cytokines dosed in the aqueous humor and intraocular pressure in the studied dogs. (A) Sperman´s correlation test (r=-0.38, P=0.06). (B) Person´s correlation test (r=-0.42, P=0.04).
Discussion
Only two previous studies have focused on studying the relationship between cataract maturity and IOP (Leasure et al. 2001Leasure J., Gelatt K.N. & Mackay E.O. 2001. The relationship of cataract maturity to intraocular pressure in dogs. Vet. Ophthalmol. 4(4):273-276., Renzo et al. 2014Renzo R., Ribeiro A.P., Silva M.L., Silva G.A., Ortencio K.P., Sobrinho A.A.F.B., Mineo T.W. & Laus J.L. 2014. Intraocular pressure, specular microscopy, and prostaglandin E2 concentration in dogs with mature and hypermature cataract. Vet. Ophthalmol. 17(4):280-285.). Leasure et al. (2001)Leasure J., Gelatt K.N. & Mackay E.O. 2001. The relationship of cataract maturity to intraocular pressure in dogs. Vet. Ophthalmol. 4(4):273-276. reported that IOP tends to decrease as the cataracts progresses; however, significant results were only observed between dogs with immature and hypermature stages (Leasure et al. 2001Leasure J., Gelatt K.N. & Mackay E.O. 2001. The relationship of cataract maturity to intraocular pressure in dogs. Vet. Ophthalmol. 4(4):273-276.). Renzo et al. (2014)Renzo R., Ribeiro A.P., Silva M.L., Silva G.A., Ortencio K.P., Sobrinho A.A.F.B., Mineo T.W. & Laus J.L. 2014. Intraocular pressure, specular microscopy, and prostaglandin E2 concentration in dogs with mature and hypermature cataract. Vet. Ophthalmol. 17(4):280-285. also encountered lower IOP values in dogs with mature and hypermature cataracts in comparison to controls, with results that also did not reach statistical significance. Similarly, the dogs in our study showed the same tendency of lower IOPs in advanced stages of cataract maturity, but this tendency was not statistically significant.
This was the first study to evaluate the concentration of MMP-2 in the aqueous humor of dogs with cataracts. The decision to investigate MMP-2 was based on previous studies that reported that the latent form of such gelatinase was found to be the most relevant MMP in normal eyes of humans, rabbits, dogs, and chicken (Di Girolamo et al. 1996Di Girolamo N., Verma M.J., McCluskey P.J., Lloyd A. & Wakefield D. 1996. Increased matrix metalloproteinases in the aqueous humor of patients and experimental animals with uveitis. Curr. Eye Res. 15(10):1060-1068., Weinstein et al. 2007Weinstein W.L., Dietrich U.M., Sapienza J.S., Carmichael K.P., Moore P.A. & Krunkosky T.M. 2007. Identification of ocular matrix metalloproteinases present within the aqueous humor and iridocorneal drainage angle tissue of normal and glaucomatous canine eyes. Vet. Ophthalmol. 10(Suppl.1):108-116., Shimada et al. 2014Shimada A., Miyata Y. & Kosano H. 2014. Type I collagen accelerates the spreading of lens epithelial cells through the expression and activation of matrix metalloproteinases. Curr. Eye Res. 39(5):460-471.). Although slightly increased concentrations of MMP-2 were observed in dogs with mature cataracts than in controls, significantly higher values were only detected in patients with the hypermature stage of this affection. In the current study we quantified the concentration of MMP-2 in the aqueous humor using ELISA sensitive for latent and active MMP-2, as well as MMP-2 complexed with TIMP-2. Therefore, comparisons of our results with those reported in a previous study performed on the eyes of normal and glaucomatous dogs cannot be made because MMPs were quantified using zymography (Weinstein et al. 2007Weinstein W.L., Dietrich U.M., Sapienza J.S., Carmichael K.P., Moore P.A. & Krunkosky T.M. 2007. Identification of ocular matrix metalloproteinases present within the aqueous humor and iridocorneal drainage angle tissue of normal and glaucomatous canine eyes. Vet. Ophthalmol. 10(Suppl.1):108-116.). In the only study that compared healthy humans and those with uveitis, the researchers used zymography to quantify the concentration of MMPs in the aqueous humor (Di Girolamo et al. 1996Di Girolamo N., Verma M.J., McCluskey P.J., Lloyd A. & Wakefield D. 1996. Increased matrix metalloproteinases in the aqueous humor of patients and experimental animals with uveitis. Curr. Eye Res. 15(10):1060-1068.).
Määtää et al. (2006)Määtää M., Tervahartiala T., Vesti E., Airaksinen J. & Sorsa T. 2006. Levels and activation of matrix metalloproteinases in aqueous humor are elevated in uveitis-related secondary glaucoma. J. Glaucoma 15(3):229-237. and Kara et al. (2014)Kara S., Yildirim N., Ozer A., Colak O. & Sahin A. 2014. Matrix metalloproteinase-2, tissue inhibitor of matrix metalloproteinase-2, and transforming growth factor beta 1 in the aqueous humor and serum of patients with pseudoexfoliation syndrome. Clin. Ophthalmol. 28(8):305-309. <https://doi.org/10.214/OPTH.S55914>
https://doi.org/10.214/OPTH.S55914...
used a similar ELISA assay that we used in our study (total MMP-2); in patients with cataracts, these authors reported that the average concentration of MMP-2 in the aqueous humor was 25ng/mL (Määtää et al. 2006Määtää M., Tervahartiala T., Vesti E., Airaksinen J. & Sorsa T. 2006. Levels and activation of matrix metalloproteinases in aqueous humor are elevated in uveitis-related secondary glaucoma. J. Glaucoma 15(3):229-237., Kara et al. 2014Kara S., Yildirim N., Ozer A., Colak O. & Sahin A. 2014. Matrix metalloproteinase-2, tissue inhibitor of matrix metalloproteinase-2, and transforming growth factor beta 1 in the aqueous humor and serum of patients with pseudoexfoliation syndrome. Clin. Ophthalmol. 28(8):305-309. <https://doi.org/10.214/OPTH.S55914>
https://doi.org/10.214/OPTH.S55914...
). This was much higher than the concentrations observed in dogs with mature (1.42ng/mL) and hypermature (2.44ng/mL) cataracts in our study. Bauer et al. (2018)Bauer D., Kasper M., Walscheid K., Koch J.M., Müther P.S., Kirchhof B., Heiligenhaus A. & Heinz C. 2018. Multiplex cytokine analysis of aqueous humor in juvenile idiopathic arthritis-associated anterior uveitis with or without secondary glaucoma. Front. Immunol. 9(5):708. used multiplex bead immunoassay to quantify the concentration of MMP-2 in the aqueous humor of humans with cataracts and reported values that ranged from 0.25 to 0.75ng/mL; whereas in patients with juvenile idiopathic arthritis-associated anterior uveitis, median levels of such cytokine in the aqueous humor increased to 0.90ng/mL (0.30-1.60ng/mL). Reasonable explanations for the discrepancy between the results observed in humans studies with the ones found in our research can be related to interspecies variability, interassay variability, and for how long aqueous humor was stored. In addition, most of the studies conducted in humans focusing on the concentration of intraocular MMPs (El-Shabrawi et al. 2000El-Shabrawi Y.G., Christen W.G. & Foster S.C. 2000. Correlation of metalloproteinase-2 and -9 with proinflammatory cytokines interleukin-1b, interleukin-12 and the interleukin-1 receptor antagonist in patients with chronic uveitis. Curr. Eye Res. 20(3):211-214., Määtää et al. 2006Määtää M., Tervahartiala T., Vesti E., Airaksinen J. & Sorsa T. 2006. Levels and activation of matrix metalloproteinases in aqueous humor are elevated in uveitis-related secondary glaucoma. J. Glaucoma 15(3):229-237., Alapure et al. 2012Alapure B.V., Praveen M.R., Gajjar D.U., Vasavada A.R., Parmar T.J. & Arora A.I. 2012. Matrix metalloproteinase-2 and -9 activities in the human lens epithelial cells and serum of steroid induced posterior subcapsular cataracts. Mol. Vis. 18:64-73., Bauer et al. 2018Bauer D., Kasper M., Walscheid K., Koch J.M., Müther P.S., Kirchhof B., Heiligenhaus A. & Heinz C. 2018. Multiplex cytokine analysis of aqueous humor in juvenile idiopathic arthritis-associated anterior uveitis with or without secondary glaucoma. Front. Immunol. 9(5):708.) and IL-1 β (Takay et al. 2012Takay Y., Tanito M. & Ohira A. 2012. Multiplex cytokine analysis of aqueous humor in eyes with primary open-angle glaucoma, exfoliation glaucoma, and cataract. Invest. Ophthalmol. Vis. Sci. 53(1):241-247., Duvesh et al. 2017Duvesh R., Puthuran G., Srinivasan K., Rengaraj V., Krishnadas S.R., Rajendrababu S., Balakrishnan V., Ramulu P. & Sundaresan P. 2017. Multiplex cytokine analysis of aqueous humor from the patients with chronic primary angle closure glaucoma. Curr. Eye Res. 42(12):1608-1613., Zheng et al. 2018Zheng Y., Rao Y.Q., Li J.K., Huang Y., Zhao P. & Li J. 2018. Age-related pro-inflammatory and pro-angiogenic changes in human aqueous humor. Int. J. Ophthalmol. 11(2):196-200.) used individuals with senile cataracts as controls. In these studies, however, researches did not report the stages of the patients´ cataracts. Therefore, direct comparisons of our results with the ones obtained in human studies with regard to MMP-2 concentration and the stage of cataract maturity cannot be made.
MMPs present in the iridocorneal angle and in the aqueous humor play a role in controlling IOP by increasing the ECM turnover rate, which reduces the resistance to aqueous outflow through the meshwork (Bradley et al. 2001Bradley J.M., Kelley M.J., Zhu X.H., Anderssohn A.M., Alexander J.P. & Acott T.S. 2001. Effects of mechanical stretching on trabecular matrix metalloproteinases. Invest. Ophthalmol. Vis. Sci. 42(7):1505-1513., Weinstein et al. 2007Weinstein W.L., Dietrich U.M., Sapienza J.S., Carmichael K.P., Moore P.A. & Krunkosky T.M. 2007. Identification of ocular matrix metalloproteinases present within the aqueous humor and iridocorneal drainage angle tissue of normal and glaucomatous canine eyes. Vet. Ophthalmol. 10(Suppl.1):108-116.). Several studies have shown that the concentration of MMP-2 in the aqueous humor was significantly increased in human patients with uveitis of different causes (Di Girolamo et al. 1996Di Girolamo N., Verma M.J., McCluskey P.J., Lloyd A. & Wakefield D. 1996. Increased matrix metalloproteinases in the aqueous humor of patients and experimental animals with uveitis. Curr. Eye Res. 15(10):1060-1068., El-Shabrawi et al. 2000El-Shabrawi Y.G., Christen W.G. & Foster S.C. 2000. Correlation of metalloproteinase-2 and -9 with proinflammatory cytokines interleukin-1b, interleukin-12 and the interleukin-1 receptor antagonist in patients with chronic uveitis. Curr. Eye Res. 20(3):211-214., Määtää et al. 2006Määtää M., Tervahartiala T., Vesti E., Airaksinen J. & Sorsa T. 2006. Levels and activation of matrix metalloproteinases in aqueous humor are elevated in uveitis-related secondary glaucoma. J. Glaucoma 15(3):229-237., Alapure et al. 2012Alapure B.V., Praveen M.R., Gajjar D.U., Vasavada A.R., Parmar T.J. & Arora A.I. 2012. Matrix metalloproteinase-2 and -9 activities in the human lens epithelial cells and serum of steroid induced posterior subcapsular cataracts. Mol. Vis. 18:64-73., Bauer et al. 2018Bauer D., Kasper M., Walscheid K., Koch J.M., Müther P.S., Kirchhof B., Heiligenhaus A. & Heinz C. 2018. Multiplex cytokine analysis of aqueous humor in juvenile idiopathic arthritis-associated anterior uveitis with or without secondary glaucoma. Front. Immunol. 9(5):708.). However, researchers in all aforementioned studies did not assess the relationship between the concentration of MMP-2 and IOP (El-Shabrawi et al. 2000El-Shabrawi Y.G., Christen W.G. & Foster S.C. 2000. Correlation of metalloproteinase-2 and -9 with proinflammatory cytokines interleukin-1b, interleukin-12 and the interleukin-1 receptor antagonist in patients with chronic uveitis. Curr. Eye Res. 20(3):211-214., Määtää et al. 2006Määtää M., Tervahartiala T., Vesti E., Airaksinen J. & Sorsa T. 2006. Levels and activation of matrix metalloproteinases in aqueous humor are elevated in uveitis-related secondary glaucoma. J. Glaucoma 15(3):229-237., Alapure et al. 2012Alapure B.V., Praveen M.R., Gajjar D.U., Vasavada A.R., Parmar T.J. & Arora A.I. 2012. Matrix metalloproteinase-2 and -9 activities in the human lens epithelial cells and serum of steroid induced posterior subcapsular cataracts. Mol. Vis. 18:64-73.). Only Määtää et al. (2006)Määtää M., Tervahartiala T., Vesti E., Airaksinen J. & Sorsa T. 2006. Levels and activation of matrix metalloproteinases in aqueous humor are elevated in uveitis-related secondary glaucoma. J. Glaucoma 15(3):229-237. reported that the concentration of MMP-2 in the aqueous humor did not correlate with the IOP in patients with cataracts, primary open-angle glaucoma and secondary glaucomas associated with uveitis. Although there was a tendency toward a negative correlation between the concentration of MMP-2 and IOP in our study, such parameters did not correlate significantly.
It has been reported that the immunoreactivity for MMP-2 in normal human lenses is weak and localized within the lens epithelium and intracellularly, within a few early differentiating fibers at the lens cortex (Sachdev et al. 2004Sachdev N.H., Di Girolamo N., Nolan T.M., McCluskey P.J., Wakefield D. & Coroneo M.T. 2004. Matrix metalloproteinases and tissue inhibitors of matrix metalloproteinases in the human lens: implications for cortical cataract formation. Invest. Ophthalmol. Vis. Sci. 45(11):4075-4078.). In the same study, no staining for MMP-2 was observed in cataractous lenses, while zymograms and ELISA assays revealed increased levels of this cytokine (Sachdev et al. 2004Sachdev N.H., Di Girolamo N., Nolan T.M., McCluskey P.J., Wakefield D. & Coroneo M.T. 2004. Matrix metalloproteinases and tissue inhibitors of matrix metalloproteinases in the human lens: implications for cortical cataract formation. Invest. Ophthalmol. Vis. Sci. 45(11):4075-4078.). MMPs are capable of denaturing most components of the lens capsule ECM facilitating lens fiber migration and differentiation into miofibroblasts, which are responsible for the contraction and opacification of the anterior and posterior capsule of the lens (West-Mays 2007West-Mays J.A. 2007. Matrix metalloproteinases as mediators of primary and secondary cataracts. Expert Rev. Ophthalmol. 2(6):931-938., Eldred et al. 2012Eldred J.A., Hodgkinson L.M., Dawes L.J., Reddan J.R., Edwards D.R. & Wormstone I.M. 2012 MMP-2 activity is critical for TGF beta-2-induced matrix contraction: implications for fibrosis. Invest. Ophthalmol. Vis. Sci. 53(7):4085-4098.). These are the reasons because hypermature cataracts will have evidence of resorption, subcapsular plaques, lens-induced uveitis, and capsular wrinkling (Davidson & Nelms 2013Davidson M.G. & Nelms S.R. 2013. Diseases of the lens and cataract formation, p.1199-1233. In: Gelatt K.N. & Gilger B.C. (Eds.), Veterinary Ophthalmology. 5th ed. Ames, Wiley-Blackwell.). This can explain our findings with regard to higher levels of MMP-2 in the aqueous humor of dogs with hypermature cataracts. Although the degrees of anterior and posterior capsular opacification were not quantified in the dogs of our experiment, one can assume that the higher aqueous humor levels of MMP-2 detected in patients with hypermature cataracts can be associated with degradation of lens epithelium cells, as well anterior and posterior capsular fibrosis. Thus, further studies can be developed to evaluate the role of MMP-2 inhibitors in early stages of cataract maturity in order to prevent capsular fibrosis, which makes capsulorhexis harder to perform.
In our study, the concentration of IL-1 β in the aqueous humor varied very little among healthy controls and dogs with mature and hypermature cataracts. However, the concentration IL-1 β increased significantly in accordance with the degree of lens opacification. Contrary to our findings, it has been reported that the concentration of IL-1 β in the aqueous humor of humans with cataracts at stage II and III did not differ significantly; however, patients of that study received anti-inflammatory eye drops before phacoemulsification (Wang et al. 2016Wang L., Zhang Z., Koch D.D., Jia Y., Cao W. & Zhang S. 2016. Anterior chamber interleukin 1β, interleukin 6 and prostaglandin E2 in patients undergoing femtosecond laser-assisted cataract surgery. Brit. J. Ophthalmol. 100(4):579-582.). Chiou et al. (1994)Chiou G.C., Yao Q.S. & Okawara T. 1994. Prevention of ocular inflammation induced by lens proteins, endotoxin, and interleukin-1 with synthetic interleukin-1 blockers. J. Ocul. Pharmacol. Ther. 10(3):577-586. observed that the intraocular inflammation induced by lens proteins can be prevented with the use of interleukin-1 blockers; our results are consistent with these findings. There are no previous reports describing the concentration of IL-1 β in in the aqueous humor of dogs, cats, and horses. In rats, the concentration of IL-1 β in the aqueous humor quantified using ELISA was approximately 100pg/mL (Medeiros et al. 2008Medeiros R., Rodrigues G.B., Figueiredo C.P., Rodrigues E.B., Grumman A. Jr., Menezes-de-Lima O. Jr., Passos G.F. & Calixto J.B. 2008. Molecular mechanisms of topical anti-inflammatory effects of lipoxin A(4) in endotoxin-induced uveitis. Mol. Pharmacol. 74(1):154-161.). In humans with cataracts, Wang et al. (2016)Wang L., Zhang Z., Koch D.D., Jia Y., Cao W. & Zhang S. 2016. Anterior chamber interleukin 1β, interleukin 6 and prostaglandin E2 in patients undergoing femtosecond laser-assisted cataract surgery. Brit. J. Ophthalmol. 100(4):579-582. used ELISA to quantify the concentration IL-1β in the aqueous humor and reported concentrations that ranged from 14.9 to 19.4pg/mL; which were higher than the ones reported here for mature (6.00) and hypermature (6.03pg/mL) cataracts. In another studies that were also conducted in humans with cataracts in which multiplex bead immunoassay was used to quantify IL-1 β, much lower values of this cytokine were reported (0.002 to 1.0pg/mL) (Takay et al. 2012Takay Y., Tanito M. & Ohira A. 2012. Multiplex cytokine analysis of aqueous humor in eyes with primary open-angle glaucoma, exfoliation glaucoma, and cataract. Invest. Ophthalmol. Vis. Sci. 53(1):241-247., Duvesh et al. 2017Duvesh R., Puthuran G., Srinivasan K., Rengaraj V., Krishnadas S.R., Rajendrababu S., Balakrishnan V., Ramulu P. & Sundaresan P. 2017. Multiplex cytokine analysis of aqueous humor from the patients with chronic primary angle closure glaucoma. Curr. Eye Res. 42(12):1608-1613.). Quantification of IL-1 β in the aqueous humor of cataractous human eyes assessed by cytometric bead array revealed concentrations that ranged from 2.7±0.0 to 5.29±0.9pg/mL (Aketa et al. 2017Aketa N., Yamaguchi T., Suzuki T., Higa K., Yagi-Yaguchi Y., Satake Y., Tsubota K. & Shimazaki J. 2017. Iris damage is associated with elevated cytokine levels in aqueous humor. Invest. Ophthalmol. Vis. Sci. 58(6):42-51., Duvesh et al. 2017Duvesh R., Puthuran G., Srinivasan K., Rengaraj V., Krishnadas S.R., Rajendrababu S., Balakrishnan V., Ramulu P. & Sundaresan P. 2017. Multiplex cytokine analysis of aqueous humor from the patients with chronic primary angle closure glaucoma. Curr. Eye Res. 42(12):1608-1613.); which were closer to our results.
In dogs, LIU is most commonly associated with the presence of a hypermature cataract (Leasure et al. 2001Leasure J., Gelatt K.N. & Mackay E.O. 2001. The relationship of cataract maturity to intraocular pressure in dogs. Vet. Ophthalmol. 4(4):273-276., David & Nelms 2013Davidson M.G. & Nelms S.R. 2013. Diseases of the lens and cataract formation, p.1199-1233. In: Gelatt K.N. & Gilger B.C. (Eds.), Veterinary Ophthalmology. 5th ed. Ames, Wiley-Blackwell., Renzo et al. 2014Renzo R., Ribeiro A.P., Silva M.L., Silva G.A., Ortencio K.P., Sobrinho A.A.F.B., Mineo T.W. & Laus J.L. 2014. Intraocular pressure, specular microscopy, and prostaglandin E2 concentration in dogs with mature and hypermature cataract. Vet. Ophthalmol. 17(4):280-285.). Like in other causes of uveitis, dogs with LIU present lower IOPs because of the increased amounts of prostaglandins secreted by the inflammatory cells (David & Nelms 2013Davidson M.G. & Nelms S.R. 2013. Diseases of the lens and cataract formation, p.1199-1233. In: Gelatt K.N. & Gilger B.C. (Eds.), Veterinary Ophthalmology. 5th ed. Ames, Wiley-Blackwell., Renzo et al. 2014Renzo R., Ribeiro A.P., Silva M.L., Silva G.A., Ortencio K.P., Sobrinho A.A.F.B., Mineo T.W. & Laus J.L. 2014. Intraocular pressure, specular microscopy, and prostaglandin E2 concentration in dogs with mature and hypermature cataract. Vet. Ophthalmol. 17(4):280-285.). In one study, despite increased aqueous humor concentration of PGE2 has been found in dogs with mature and hypermature cataracts, the levels of such cytokine did not correlate with IOP values (Renzo et al. 2014Renzo R., Ribeiro A.P., Silva M.L., Silva G.A., Ortencio K.P., Sobrinho A.A.F.B., Mineo T.W. & Laus J.L. 2014. Intraocular pressure, specular microscopy, and prostaglandin E2 concentration in dogs with mature and hypermature cataract. Vet. Ophthalmol. 17(4):280-285.). In humans, the concentration of IL-1 β also did not correlate with IOP in subjects affected by cataracts, primary open-angle glaucoma and exfoliative syndrome (Takay et al. 2012Takay Y., Tanito M. & Ohira A. 2012. Multiplex cytokine analysis of aqueous humor in eyes with primary open-angle glaucoma, exfoliation glaucoma, and cataract. Invest. Ophthalmol. Vis. Sci. 53(1):241-247.). In dogs of our study, the concentration of IL-1 β correlated negatively with IOP, suggesting that this cytokine is involved in the pathogenesis of LIU. Therefore, further studies aiming to investigate the effects of anti-inflammatories to control of LIU should include IL-1 β as an inflammatory marker.
Aging is associated with the development of parainflammation, a term which describes a beneficial and controlled low level of inflammation in response to accumulating tissue stress such as oxidative and chemical stress (Zheng et al. 2018Zheng Y., Rao Y.Q., Li J.K., Huang Y., Zhao P. & Li J. 2018. Age-related pro-inflammatory and pro-angiogenic changes in human aqueous humor. Int. J. Ophthalmol. 11(2):196-200.). Aqueous humor is an important immunological milieu of the anterior segment (Zheng et al. 2018Zheng Y., Rao Y.Q., Li J.K., Huang Y., Zhao P. & Li J. 2018. Age-related pro-inflammatory and pro-angiogenic changes in human aqueous humor. Int. J. Ophthalmol. 11(2):196-200.). Therefore, we consider to asses possible correlations between the ages of our dogs with the cytokines dosed in the aqueous humor. It has been reported that the concentration of IL-1 β in the aqueous humor of children and adults affected by cataracts are similar, and that such cytokine did not correlate with the age of patients (Zheng et al. 2018Zheng Y., Rao Y.Q., Li J.K., Huang Y., Zhao P. & Li J. 2018. Age-related pro-inflammatory and pro-angiogenic changes in human aqueous humor. Int. J. Ophthalmol. 11(2):196-200.). Herein, we also did not observe any correlations between IL-1 β and the ages of dogs in any studied group. In one report, MMP-2 dosed in the aqueous humor did not correlate with the ages of humans patients with cataracts and uveitis (Bauer et al. 2018Bauer D., Kasper M., Walscheid K., Koch J.M., Müther P.S., Kirchhof B., Heiligenhaus A. & Heinz C. 2018. Multiplex cytokine analysis of aqueous humor in juvenile idiopathic arthritis-associated anterior uveitis with or without secondary glaucoma. Front. Immunol. 9(5):708.). Similarly, significant correlations were not found between the concentration of this cytokine and the ages of dogs with normal eyes, and those with hypermature cataracts. In the ones with mature cataracts, however, MMP-2 correlated positively with age.
Although our findings showed that the concentrations of MMP-2 and IL-1 β were increased in parallel with the cataract stage, a relationship between these two cytokines was not encountered. It has been reported that the levels of MMP-2, MMP-9, IL-1 β, and IL-12 are positively correlated with the severity of uveitis in humans (Di Girolamo et al. 1996Di Girolamo N., Verma M.J., McCluskey P.J., Lloyd A. & Wakefield D. 1996. Increased matrix metalloproteinases in the aqueous humor of patients and experimental animals with uveitis. Curr. Eye Res. 15(10):1060-1068., El-Shabrawi et al. 2000El-Shabrawi Y.G., Christen W.G. & Foster S.C. 2000. Correlation of metalloproteinase-2 and -9 with proinflammatory cytokines interleukin-1b, interleukin-12 and the interleukin-1 receptor antagonist in patients with chronic uveitis. Curr. Eye Res. 20(3):211-214., Hodgkinson et al. 2007Hodgkinson L.M., Duncan G., Wang L., Pennington C.J., Edwards D.R. & Wormstone I.M. 2007. MMP and TIMP expression in quiescent, dividing, and differentiating human lens cells. Invest. Ophthalmol. Vis. Sci. 48(9):4192-4199., Aketa et al. 2017Aketa N., Yamaguchi T., Suzuki T., Higa K., Yagi-Yaguchi Y., Satake Y., Tsubota K. & Shimazaki J. 2017. Iris damage is associated with elevated cytokine levels in aqueous humor. Invest. Ophthalmol. Vis. Sci. 58(6):42-51.). Notwithstanding, we did not investigate possible correlations between the uveitis score and the other parameters evaluated because all enrolled patients had mild uveitis.
Conclusions
The increment in the aqueous humor concentration of IL-1 β found in patients with mature and hypermature cataracts, in addition to the negative relationship of this cytokine with IOP, suggests that IL-1 β is involved in the pathogenesis of LIU in dogs.
Although dogs with hypermature cataracts presented higher concentrations of MMP-2 in the aqueous humor, such cytokine did not correlate with IL-1 β and IOP.
In our study, a relationship between aqueous humor cytokines and the ages of patients was only confirmed between MMP-2 and the ages of dogs with mature cataracts.
References
- Abrams K.L., Stabila P.F., Kauper K. & Elliott S. 2011. Vascular endothelial growth factor in diabetic and nondiabetic canine cataract patients. Vet. Ophthalmol. 14(2):93-99.
- Alapure B.V., Praveen M.R., Gajjar D.U., Vasavada A.R., Parmar T.J. & Arora A.I. 2012. Matrix metalloproteinase-2 and -9 activities in the human lens epithelial cells and serum of steroid induced posterior subcapsular cataracts. Mol. Vis. 18:64-73.
- Aketa N., Yamaguchi T., Suzuki T., Higa K., Yagi-Yaguchi Y., Satake Y., Tsubota K. & Shimazaki J. 2017. Iris damage is associated with elevated cytokine levels in aqueous humor. Invest. Ophthalmol. Vis. Sci. 58(6):42-51.
- Bauer D., Kasper M., Walscheid K., Koch J.M., Müther P.S., Kirchhof B., Heiligenhaus A. & Heinz C. 2018. Multiplex cytokine analysis of aqueous humor in juvenile idiopathic arthritis-associated anterior uveitis with or without secondary glaucoma. Front. Immunol. 9(5):708.
- Bradley J.M., Kelley M.J., Zhu X.H., Anderssohn A.M., Alexander J.P. & Acott T.S. 2001. Effects of mechanical stretching on trabecular matrix metalloproteinases. Invest. Ophthalmol. Vis. Sci. 42(7):1505-1513.
- Chiou G.C., Yao Q.S. & Okawara T. 1994. Prevention of ocular inflammation induced by lens proteins, endotoxin, and interleukin-1 with synthetic interleukin-1 blockers. J. Ocul. Pharmacol. Ther. 10(3):577-586.
- Davidson M.G. & Nelms S.R. 2013. Diseases of the lens and cataract formation, p.1199-1233. In: Gelatt K.N. & Gilger B.C. (Eds.), Veterinary Ophthalmology. 5th ed. Ames, Wiley-Blackwell.
- Di Girolamo N., Verma M.J., McCluskey P.J., Lloyd A. & Wakefield D. 1996. Increased matrix metalloproteinases in the aqueous humor of patients and experimental animals with uveitis. Curr. Eye Res. 15(10):1060-1068.
- Duvesh R., Puthuran G., Srinivasan K., Rengaraj V., Krishnadas S.R., Rajendrababu S., Balakrishnan V., Ramulu P. & Sundaresan P. 2017. Multiplex cytokine analysis of aqueous humor from the patients with chronic primary angle closure glaucoma. Curr. Eye Res. 42(12):1608-1613.
- Eldred J.A., Hodgkinson L.M., Dawes L.J., Reddan J.R., Edwards D.R. & Wormstone I.M. 2012 MMP-2 activity is critical for TGF beta-2-induced matrix contraction: implications for fibrosis. Invest. Ophthalmol. Vis. Sci. 53(7):4085-4098.
- El-Shabrawi Y.G., Christen W.G. & Foster S.C. 2000. Correlation of metalloproteinase-2 and -9 with proinflammatory cytokines interleukin-1b, interleukin-12 and the interleukin-1 receptor antagonist in patients with chronic uveitis. Curr. Eye Res. 20(3):211-214.
- Gilmour M.A. & Payton M.E. 2012. Comparison of the effects of IV administration of meloxicam, carprofen, and flunixin meglumine on prostaglandin E2 concentration in aqueous humor of dogs with aqueocentesis-induced anterior uveitis. Am. J. Vet. Res. 73(5):698-703.
- Hodgkinson L.M., Duncan G., Wang L., Pennington C.J., Edwards D.R. & Wormstone I.M. 2007. MMP and TIMP expression in quiescent, dividing, and differentiating human lens cells. Invest. Ophthalmol. Vis. Sci. 48(9):4192-4199.
- Jia Y., Hu D.N., Zhu D., Zhang L., Gu P., Fan X. & Zhou J. 2014. MMP-2, MMP-3, TIMP-1, TIMP-2, and TIMP-3 protein levels in human aqueous humor: relationship with axial length. Invest. Ophthalmol. Vis. Sci. 55(6):3922-3928.
- Kara S., Yildirim N., Ozer A., Colak O. & Sahin A. 2014. Matrix metalloproteinase-2, tissue inhibitor of matrix metalloproteinase-2, and transforming growth factor beta 1 in the aqueous humor and serum of patients with pseudoexfoliation syndrome. Clin. Ophthalmol. 28(8):305-309. <https://doi.org/10.214/OPTH.S55914>
» https://doi.org/10.214/OPTH.S55914 - Leasure J., Gelatt K.N. & Mackay E.O. 2001. The relationship of cataract maturity to intraocular pressure in dogs. Vet. Ophthalmol. 4(4):273-276.
- Määtää M., Tervahartiala T., Vesti E., Airaksinen J. & Sorsa T. 2006. Levels and activation of matrix metalloproteinases in aqueous humor are elevated in uveitis-related secondary glaucoma. J. Glaucoma 15(3):229-237.
- Medeiros R., Rodrigues G.B., Figueiredo C.P., Rodrigues E.B., Grumman A. Jr., Menezes-de-Lima O. Jr., Passos G.F. & Calixto J.B. 2008. Molecular mechanisms of topical anti-inflammatory effects of lipoxin A(4) in endotoxin-induced uveitis. Mol. Pharmacol. 74(1):154-161.
- Nishi O., Nishi K. & Ohmoto Y. 1994. Effect of interleukin 1 receptor antagonist on the blood-aqueous barrier after intraocular lens implantation. Brit. J. Ophthalmol. 78(12):917-920.
- Ortencio K.P., Renzo R., Sobrinho A.A.F.B., Kobashigawa K.K., Crivelaro R.M., Silva G.A., Aldrovani M., Ribeiro A.P., Mineo T.W. & Laus J.L. 2015. Effects of morphine on the expression of cytokines and inflammatory mediators in a rabbit model of endotoxin-induced experimental uveitis. Arq. Bras. Oftalmol. 78(6):371-375.
- Pinard C.L., Gauvin D., Moreau M., Martel-Pelletier J., Pelletier J.P. & Troncy E. 2011. Measurements of canine aqueous humor inflammatory mediators and the effect of carprofen following anterior chamber paracentesis. Vet. Ophthalmol. 14(5):296-303.
- Renzo R., Ribeiro A.P., Silva M.L., Silva G.A., Ortencio K.P., Sobrinho A.A.F.B., Mineo T.W. & Laus J.L. 2014. Intraocular pressure, specular microscopy, and prostaglandin E2 concentration in dogs with mature and hypermature cataract. Vet. Ophthalmol. 17(4):280-285.
- Ribeiro A.P., Escobar A., Motheo T.F., Sakamoto C.A.M., Godoy G.S., Costa M.A.N. & Laus J.L 2010. Effects of carprofen administered by different routes to control experimental uveitis in dogs. Arq. Bras. Med. Vet. Zootec. 62(2):309-317.
- Ribeiro A.P., Silva M.L., Araújo R.L., Ferrucci D.L., Mineo T., Thiesen R., Bandarra M.B. & Laus JL. 2012. Expression of matrix metalloproteinases, type IV collagen, and interleukin-10 in rabbits treated with morphine after lamellar keratectomy. Vet. Ophthalmol. 15(3):153-163.
- Sachdev N.H., Di Girolamo N., Nolan T.M., McCluskey P.J., Wakefield D. & Coroneo M.T. 2004. Matrix metalloproteinases and tissue inhibitors of matrix metalloproteinases in the human lens: implications for cortical cataract formation. Invest. Ophthalmol. Vis. Sci. 45(11):4075-4078.
- Sandberg C.A., Herring I.P., Huckle W.R., LeRoith T., Pickett J.P. & Rossmeisl J.H. 2012. Aqueous humor vascular endothelial growth factor in dogs: association with intraocular disease and the development of pre-iridal fibrovascular membrane. Vet. Ophthalmol. 15(Suppl.1):21-30.
- Shimada A., Miyata Y. & Kosano H. 2014. Type I collagen accelerates the spreading of lens epithelial cells through the expression and activation of matrix metalloproteinases. Curr. Eye Res. 39(5):460-471.
- Takay Y., Tanito M. & Ohira A. 2012. Multiplex cytokine analysis of aqueous humor in eyes with primary open-angle glaucoma, exfoliation glaucoma, and cataract. Invest. Ophthalmol. Vis. Sci. 53(1):241-247.
- Tamiya S., Wormstone I.M., Marcantonio J.M., Gavrilovic J. & Duncan G. 2000. Induction of matrix metalloproteinases 2 and 9 following stress to the lens. Exp. Eye Res. 71(6):591-597.
- Van der Woerdt A. 2000. Lens-induced uveits. Vet. Ophthalmol. 3(4):227-234.
- Wang L., Zhang Z., Koch D.D., Jia Y., Cao W. & Zhang S. 2016. Anterior chamber interleukin 1β, interleukin 6 and prostaglandin E2 in patients undergoing femtosecond laser-assisted cataract surgery. Brit. J. Ophthalmol. 100(4):579-582.
- Weinstein W.L., Dietrich U.M., Sapienza J.S., Carmichael K.P., Moore P.A. & Krunkosky T.M. 2007. Identification of ocular matrix metalloproteinases present within the aqueous humor and iridocorneal drainage angle tissue of normal and glaucomatous canine eyes. Vet. Ophthalmol. 10(Suppl.1):108-116.
- West-Mays J.A. 2007. Matrix metalloproteinases as mediators of primary and secondary cataracts. Expert Rev. Ophthalmol. 2(6):931-938.
- Zheng Y., Rao Y.Q., Li J.K., Huang Y., Zhao P. & Li J. 2018. Age-related pro-inflammatory and pro-angiogenic changes in human aqueous humor. Int. J. Ophthalmol. 11(2):196-200.
Publication Dates
-
Publication in this collection
29 May 2020 -
Date of issue
Mar 2020
History
-
Received
12 Apr 2019 -
Accepted
09 Aug 2019