Abstract
Fluid volume and hemodynamic management in hemodialysis patients is an essential component of dialysis adequacy. Restoring salt and water homeostasis in hemodialysis patients has been a permanent quest by nephrologists summarized by the ‘dry weight’ probing approach. Although this clinical approach has been associated with benefits on cardiovascular outcome, it is now challenged by recent studies showing that intensity or aggressiveness to remove fluid during intermittent dialysis is associated with cardiovascular stress and potential organ damage. A more precise approach is required to improve cardiovascular outcome in this high-risk population. Fluid status assessment and monitoring rely on four components: clinical assessment, non-invasive instrumental tools (e.g., US, bioimpedance, blood volume monitoring), cardiac biomarkers (e.g. natriuretic peptides), and algorithm and sodium modeling to estimate mass transfer. Optimal management of fluid and sodium imbalance in dialysis patients consist in adjusting salt and fluid removal by dialysis (ultrafiltration, dialysate sodium) and by restricting salt intake and fluid gain between dialysis sessions. Modern technology using biosensors and feedback control tools embarked on dialysis machine, with sophisticated analytics will provide direct handling of sodium and water in a more precise and personalized way. It is envisaged in the near future that these tools will support physician decision making with high potential of improving cardiovascular outcome.
Keywords:
Water-Electrolyte Balance; Hemodynamic Monitoring; Blood Pressure; Cardiovascular Deconditioning; Renal Dialysis; Treatment Outcome
Resumo
O volume de fluidos e o controle hemodinâmico em pacientes em hemodiálise é um componente essencial da adequação da diálise. A restauração da homeostase do sal e da água em pacientes em hemodiálise tem sido uma busca constante por parte dos nefrologistas, no que condiz à abordagem do “peso seco. Embora essa abordagem clínica tenha sido associada a benefícios no desfecho cardiovascular, recentemente tem sido questionada por estudos que mostram que a intensidade ou agressividade para remover fluidos durante a diálise intermitente está associada a estresse cardiovascular e dano potencial a órgãos.para remover fluidos durante a diálise intermitente está associada a estresse cardiovascular e dano potencial a órgãos. Uma abordagem mais precisa é necessária para melhorar o desfecho cardiovascular nessa população de alto risco. A avaliação e monitorização do estado hídrico baseiam-se em quatro componentes: avaliação clínica, ferramentas instrumentais não invasivas (por exemplo, US, bioimpedância, monitorização do volume sanguíneo), biomarcadores cardíacos (e.g. peptídeos natriuréticos), algoritmos e modelagem de sódio para estimar a transferência de massa. O manejo otimizado do desequilíbrio hídrico e de sódio em pacientes dialíticos consiste em ajustar a remoção de sal e líquido por diálise (ultrafiltração, dialisato de sódio), e restringir a ingestão de sal e o ganho de líquido entre as sessões de diálise. Tecnologia moderna que utiliza biosensores e ferramentas de controle de feedback, hoje parte da máquina de diálise, com análises sofisticadas, proporcionam o manejo direto sobre o sódio e a água de uma maneira mais precisa e personalizada. Prevê-se no futuro próximo que essas ferramentas poderão auxiliar na tomada de decisão do médico, com alto potencial para melhorar o resultado cardiovascular.
Palavras-chave:
Manejo Hídrico e de Sódio; Monitorização Hemodinâmica; Pressão Sanguínea; Descondicionamento Cardiovascular; Hemodiálise; Resultado do Tratamento
Fluid and hemodynamic management in hemodialysis patients: An identified modifiable cardiovascular risk factor
Optimal fluid volume management in dialysis patients is an essential component of dialysis adequacy but amplitude of volume fluctuation is still a quite challenging clinical condition11 Perl J, Dember LM, Bargman JM, Browne T, Charytan DM, Flythe JE, et al. The Use of a Multidimensional Measure of Dialysis Adequacy-Moving beyond Small Solute Kinetics. Clin J Am Soc Nephrol. 2017 May 8;12(5):839-847.. Restoring salt and water homeostasis in hemodialysis patients has been a permanent Holy Grail quest by nephrologists from the sixties. Salt and water management in dialysis patients is frequently summarized by the ‘dry weight’ approach33 Charra B. 'Dry weight' in dialysis: the history of a concept. Nephrol Dial Transplant. 1998 Jul;13(7):1882-5.,44 Chazot C, Charra B, Van CV, Jean G, Vanel T, Calemard E, et al. The Janus-faced aspect of 'dry weight'. Nephrol Dial Transplant. 1999 Jan;14(1):121-4.. Although this clinical approach has been associated with benefits on cardiovascular outcome, it is now challenged by recent studies showing that intensity or aggressiveness to remove fluid during conventional thrice-weekly dialysis might induce excessive hemodynamic stress and potential organ damage with potentially deleterious consequences on the long term55 McIntyre CW. Recurrent circulatory stress: the dark side of dialysis. Semin Dial. 2010 Sep/Oct;23(5):449-51.,66 London GM. Ultrafiltration intensification for achievement of dry weight and hypertension control is not always the therapeutic gold standard. J Nephrol. 2011 Jul/Aug;24(4):395-7.. In brief, ‘dry weight’ policy is necessary from a clinical perspective but it is not sufficient from a pathophysiologic perspective to ensure a fully cardioprotective effect in dialysis patients. A more balanced and precise approach is required to improve cardiovascular outcome in this high-risk population. To satisfy this unmet need, it is time to move to a broader approach embracing the whole hemodynamic management of dialysis patients rather than focusing only on their fluid management77 Canaud B, Kooman J, Selby NM, Taal M, Francis S, Kopperschmidt P, et al. Sodium and water handling during hemodialysis: new pathophysiologic insights and management approaches for improving outcomes in end-stage kidney disease. Kidney Int. 2019 Feb;95(2):296-309..
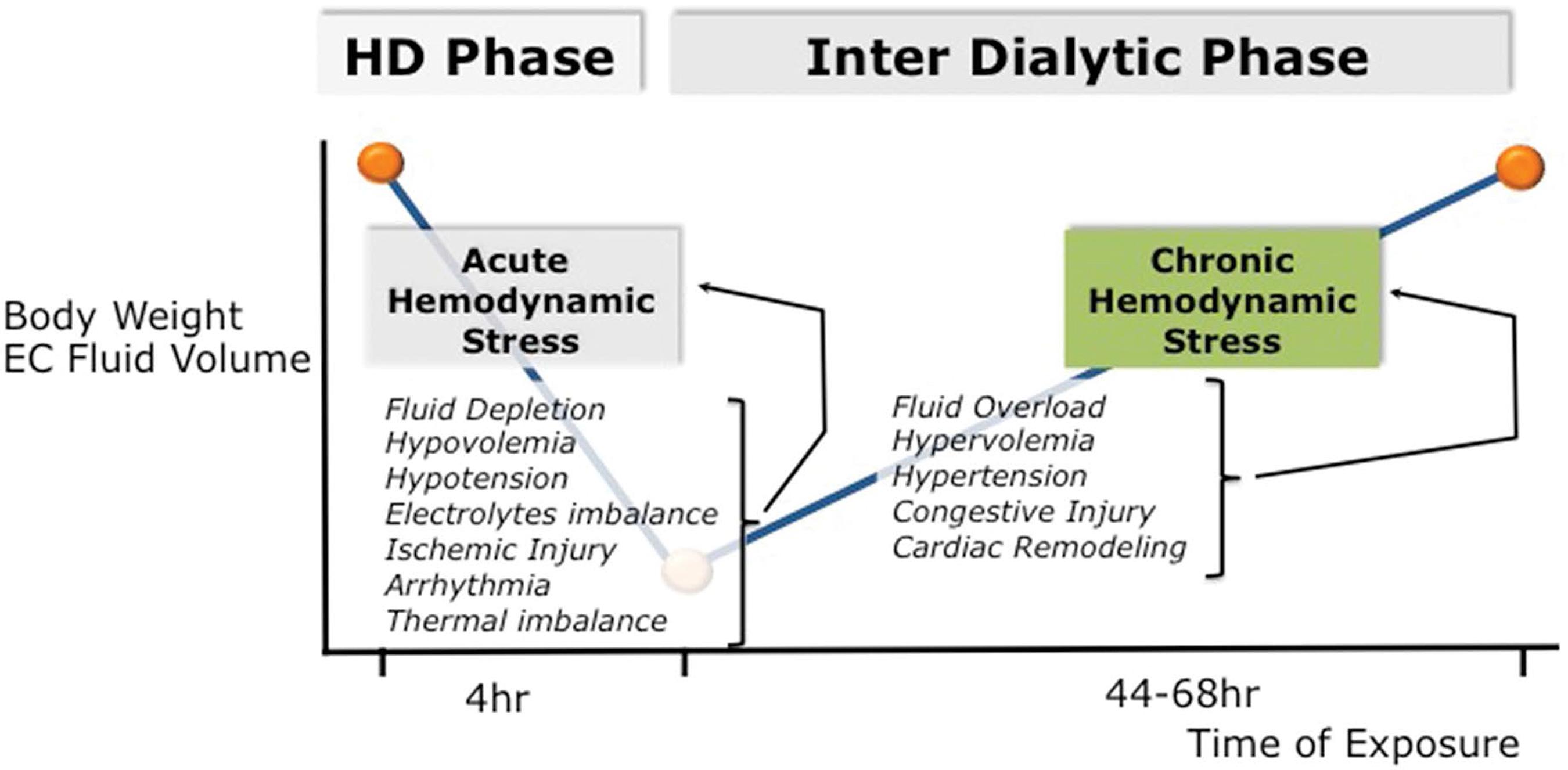
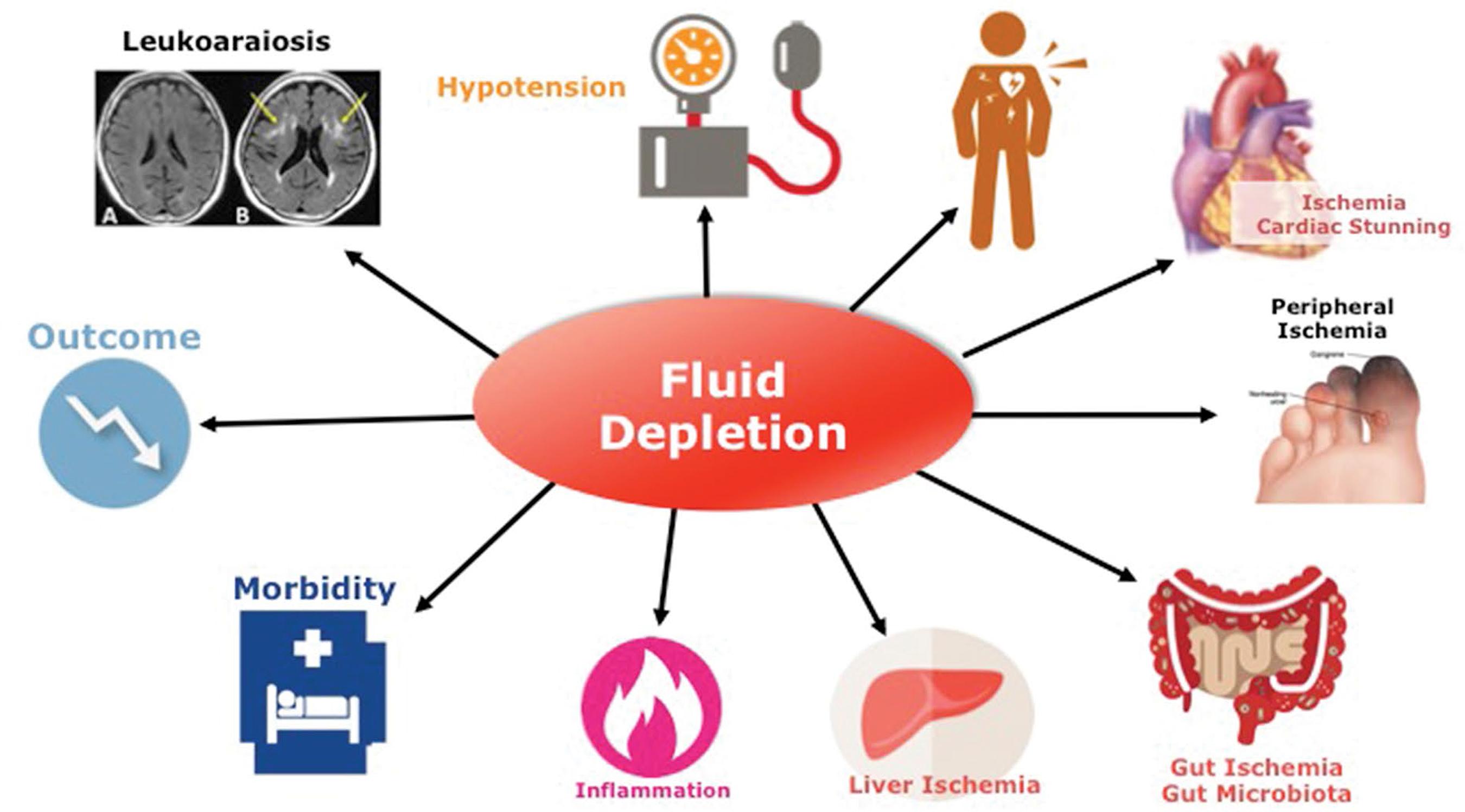
Intermittent renal replacement therapy exposes dialysis patients to continuous and repetitive hemodynamic stress conditions (Figure 1). By nature this is due to intermittency of treatment exposing patients to up (interdialytic period) and down (intradialytic period) fluid volume changes. This is best summarized by the “unphysiological profile” of short intermittent dialysis treatment88 Kjellstrand CM, Evans RL, Petersen RJ, Shideman JR, Von Hartitzsch B, Buselmeier TJ. The “unphysiology” of dialysis: a major cause of dialysis side effects?. Hemodial Int. 2004 Jan;8(1):24-9.,99 Kim GH. Dialysis unphysiology and sodium balance. Electrolyte Blood Press. 2009 Dec;7(2):31-7.. From a mechanistic approach, one can identify two different stress conditions: firstly, a chronic hemodynamic stress phase, which reflects extracellular fluid accumulation, often superimposed on a status of chronic fluid expansion during the interdialytic period; secondly, an acute hemodynamic stress phase, which reflects intravascular fluid depletion induced by dialysis session (ultrafiltration and sodium removal) marked by critical hypovolemia leading eventually to hypotensive episodes and impaired organ perfusion1010 McGuire S, Horton EJ, Renshaw D, Jimenez A, Krishnan N, McGregor G. Hemodynamic Instability during Dialysis: The Potential Role of Intradialytic Exercise. Biomed Res Int. 2018 Feb 27;2018:8276912..
Sodium and fluid accumulation that may occur in dialysis patients over time due to repetitive positive fluid imbalance is responsible for chronic extracellular fluid overload (Figure 2) with its adverse effects and cardiovascular consequences leading to poor outcomes1111 Heerspink HJL, Navis G, Ritz E. Salt intake in kidney disease: a missed therapeutic opportunity?. Nephrol Dial Transplant. 2012 Sep;27(9):3435-42.. Extracellular fluid overload and poor fluid management are the basic fundament of cardiovascular complications in hemodialysis patients1212 Kalantar-Zadeh K, Regidor DL, Kovesdy CP, Van Wyck D, Bunnapradist S, Horwich TB, et al. Fluid retention is associated with cardiovascular mortality in patients undergoing long-term hemodialysis. Circulation. 2009 Feb 10;119(5):671-9.,1313 Weiner DE, Brunelli SM, Hunt A, Schiller B, Glassock R, Maddux FW, et al. Improving clinical outcomes among hemodialysis patients: a proposal for a "volume first" approach from the chief medical officers of US dialysis providers. Am J Kidney Dis. 2014 Nov;64(5):685-95.. Hypertension as part of this constellation of disorders is widely recognized as a leading cause for left ventricular cardiomyopathy and accelerated atherosclerosis including coronary artery disease, peripheral artery disease, and cerebrovascular disease1414 Agarwal R, Martinez-Castelao A, Wiecek A, Massy Z, Suleymanlar G, Ortiz A, et al. The lingering dilemma of arterial pressure in CKD: what do we know, where do we go?. Kidney Int Suppl. 2011 Jun;1(1):17-20.,1515 Bansal N, McCulloch CE, Lin F, Alper A, Anderson AH, Cuevas M, et al. Blood Pressure and Risk of Cardiovascular Events in Patients on Chronic Hemodialysis: The CRIC Study (Chronic Renal Insufficiency Cohort). Hypertension. 2017 Aug;70(2):435-443.,1616 Georgianos PI, Agarwal R. Blood Pressure and Mortality in Long-Term Hemodialysis/ Time to Move Forward. Am J Hypertens. 2017 Mar 1;30(3):211-222.. Interestingly, as shown in a recent large cohort study, the presence of fluid overload per se has an independent and additive deleterious effect on blood pressure (either low or high blood pressure) in dialysis patient outcomes, which increases the global negative impact of blood pressure per se1717 Zoccali C, Moissl U, Chazot C, Mallamaci F, Tripepi G, Arkossy O, et al. Chronic Fluid Overload and Mortality in ESRD. J Am Soc Nephrol. 2017 Aug;28(8):2491-2497.. Hyponatremia, for reasons not entirely understood, is also associated with poor outcome in dialysis patients1818 Hecking M, Karaboyas A, Saran R, Sen A, Hörl WH, Pisoni RL, et al. Predialysis serum sodium level, dialysate sodium, and mortality in maintenance hemodialysis patients: the Dialysis Outcomes and Practice Patterns Study (DOPPS). Am J Kidney Dis. 2012 Feb;59(2):238-48.,1919 Kovesdy CP, Lott EH, Lu JL, Malakauskas SM, Ma JZ, Molnar MZ, et al. Hyponatremia, hypernatremia, and mortality in patients with chronic kidney disease with and without congestive heart failure. Circulation. 2012 Feb 7;125(5):677-84.,2020 Dekker MJ, Marcelli D, Canaud B, Konings CJ, Leunissen KM, Levin NW, et al. Unraveling the relationship between mortality, hyponatremia, inflammation and malnutrition in hemodialysis patients: results from the international MONDO initiative. Eur J Clin Nutr. 2016 Jul;70(7):779-84.. Management of sodium and fluid excess to restore fluid status homeostasis, (Figure 3) either by moderate or high ultrafiltration rate, or high plasma-to-dialysate sodium concentration gradient leading potentially to critical hypovolemia, is also associated with increased risk of mortality2121 Assa S, Hummel YM, Voors AA, Kuipers J, Westerhuis R, Jong PE, et al. Hemodialysis-induced regional left ventricular systolic dysfunction: prevalence, patient and dialysis treatment-related factors, and prognostic significance. Clin J Am Soc Nephrol. 2012 Oct;7(10):1615-23.,2222 Burton JO, Jefferies HJ, Selby NM, McIntyre CW. Hemodialysis-induced cardiac injury: determinants and associated outcomes. Clin J Am Soc Nephrol. 2009 May;4(5):914-20.,2323 Flythe JE, Kimmel SE, Brunelli SM. Rapid fluid removal during dialysis is associated with cardiovascular morbidity and mortality. Kidney Int. 2011 Jan;79(2):250-7.,2424 Flythe JE, Curhan GC, Brunelli SM. Disentangling the ultrafiltration rate-mortality association: the respective roles of session length and weight gain. Clin J Am Soc Nephrol. 2013 Jul;8(7):1151-61.,2525 Chazot C, Van CV, Lorriaux C, Deleaval P, Mayor B, Hurot JM, et al. Even a moderate fluid removal rate during individualised haemodialysis session times is associated with decreased patient survival. Blood Purif. 2017;44(2):89-97.,2626 Buchanan C, Mohammed A, Cox E, Köhler K, Canaud B, Taal MW, et al. Intradialytic Cardiac Magnetic Resonance Imaging to Assess Cardiovascular Responses in a Short-Term Trial of Hemodiafiltration and Hemodialysis. J Am Soc Nephrol. 2017 Apr;28(4):1269-1277.. Combination of these characteristics increases significantly the negative impact of each one on patient outcome2727 Chawla A, Sterns RH, Nigwekar SU, Cappuccio JD. Mortality and serum sodium: do patients die from or with hyponatremia?. Clin J Am Soc Nephrol. 2011 May;6(5):960-5.,2828 Shiba N, Shimokawa H. Chronic kidney disease and heart failure - Bidirectional close link and common therapeutic goal. J Cardiol. 2011 Jan;57(1):8-17.,2929 Khan YH, Sarriff A, Adnan AS, Khan AH, Mallhi TH. Chronic Kidney Disease, Fluid Overload and Diuretics: A Complicated Triangle. PLoS One. 2016 Jul 21;11(7):e0159335.,3030 Lim LM, Tsai NC, Lin MY, Hwang DY, Lin HY, Lee JJ, et al. Hyponatremia is Associated with Fluid Imbalance and Adverse Renal Outcome in Chronic Kidney Disease Patients Treated with Diuretics. Sci Rep. 2016 Nov 14;6:36817.. In this context, salt and fluid management of dialysis patients represents a major challenge for clinicians.
Fluid and hemodynamic management in hemodialysis patients: Challenges
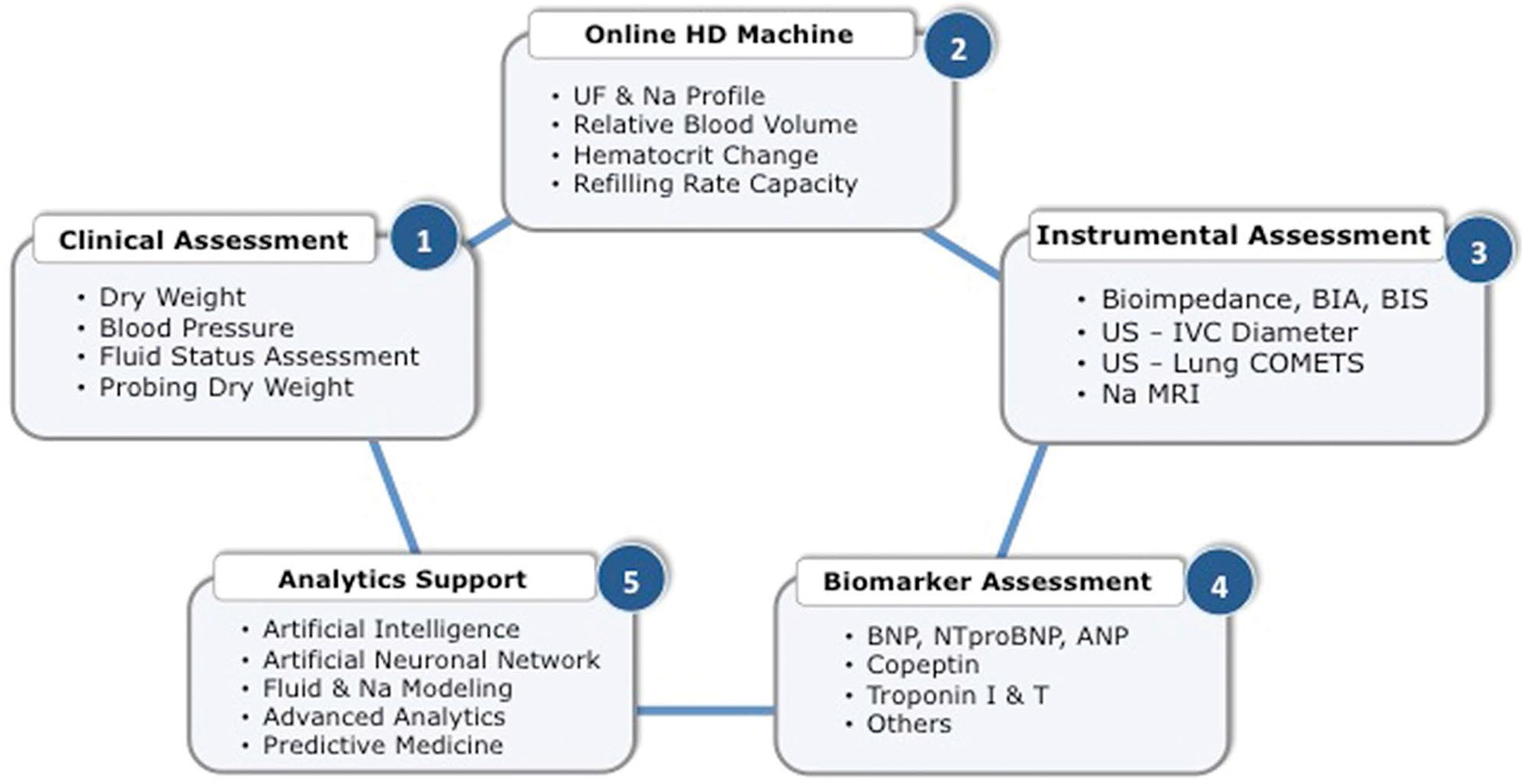
Assessing fluid status of dialysis patients is not an easy task from a clinical perspective. In that context, it is interesting to note that over time several tools have been proposed (Figure 4) to assess salt and water status in hemodialysis patients with a common objective of monitoring and guiding caregivers in their prescription3131 Ishibe S, Peixoto AJ. Methods of assessment of volume status and intercompartmental fluid shifts in hemodialysis patients: implications in clinical practice. Semin Dial. 2004 Jan/Feb;17(1):37-43.,3232 Raimann JG, Zhu F, Wang J, Thijssen S, Kuhlmann MK, Kotanko P, et al. Comparison of fluid volume estimates in chronic hemodialysis patients by bioimpedance, direct isotopic, and dilution methods. Kidney Int. 2014 Apr;85(4):898-908..
1. Clinical assessment focusing on fluid status, hemodynamic stability, and patient perception was the first attempt to address this issue in developing the concept of ‘dry weight’3333 Charra B, Jean G, Hurot JM, Terrat JC, Vanel T, Van CV, et al. Clinical Determination of Dry Body Weight. Hemodial Int. 2001 Jan;5(1):42-50.,3434 Charra B. Fluid balance, dry weight, and blood pressure in dialysis. Hemodial Int. 2007 Jan;11(1):21-31.. It relates in fact to the post-dialysis weight at which dialysis the patient has - in theory - no sign of fluid imbalance (neither excess nor depletion), blood pressure values in normal range for his or her clinical condition, and feels comfortable without functional limitations3535 Sinha AD, Agarwal R. Setting the dry weight and its cardiovascular implications. Semin Dial. 2017 Nov;30(6):481-488.. ‘Dry weight’ is probed over time by clinicians and reassessed periodically according to the patient conditions, changes, and/or intercurrent events in order to keep its fluid status balance over time3636 Canaud B, Lertdumrongluk P. Probing 'dry weight' in haemodialysis patients: 'back to the future'. Nephrol Dial Transplant. 2012 Jun;27(6):2140-3.. Further work has led investigators to refine assessment of the ‘dry weight’ concept3737 Agarwal R, Weir MR. Dry-weight: a concept revisited in an effort to avoid medication-directed approaches for blood pressure control in hemodialysis patients. Clin J Am Soc Nephrol. 2010 Jul;5(7):1255-60..
Subsequently, several tools have been proposed to help physicians in refining clinical acumen and defining more objectively ‘dry weight’ of dialysis patients3838 Leunissen KM, Kouw P, Kooman JP, Cheriex EC, DeVries PM, Donker AJ, et al. New techniques to determine fluid status in hemodialyzed patients. Kidney Int Suppl. 1993 Jun;41:S50-6.,3939 Leypoldt JK, Cheung AK. Evaluating volume status in hemodialysis patients. Adv Ren Replace Ther. 1998 Jan;5(1):64-74.. In brief, they utilize either instrumental tools or biomarkers4040 Ozkahya M, Ok E, Toz H, Asci G, Duman S, Basci A, et al. Long-term survival rates in haemodialysis patients treated with strict volume control. Nephrol Dial Transplant. 2006 Dec;21(12):3506-13.,4141 Ito K, Ookawara S, Ueda Y, Miyazawa H, Yamada H, Goto S, et al. A Higher Cardiothoracic Ratio Is Associated with 2-Year Mortality after Hemodialysis Initiation. Nephron Extra. 2015 Dec 19;5(3):100-10.,4242 Goldfarb-Rumyantzev AS, Chelamcharla M, Bray BE, Leypoldt JK, Lavasani I, Nelson N, et al. Volume indicators and left ventricular mass during aggressive volume management in patients on thrice-weekly hemodialysis. Nephron Clin Pract. 2009;113(4):c270-80..
2. Instrumental or technology-based tools use various non-invasive ways to assess volemia, fluid status, or hemodynamic surrogate indicators.
Inferior vena cava diameter (IVCD) and collapsibility has been proposed to monitor intravascular volume and right atrial pressure or central venous pressure in dialysis patients with interesting findings4343 Katzarski KS, Nisell J, Randmaa I, Danielsson A, Freyschuss U, Bergström J. A critical evaluation of ultrasound measurement of inferior vena cava diameter in assessing dry weight in normotensive and hypertensive hemodialysis patients. Am J Kidney Dis. 1997 Oct;30(4):459-65.,4444 Krause I, Birk E, Davidovits M, Cleper R, Blieden L, Pinhas L, et al. Inferior vena cava diameter: a useful method for estimation of fluid status in children on haemodialysis. Nephrol Dial Transplant. 2001 Jun;16(6):1203-6.,4545 Stawicki SP, Adkins EJ, Eiferman DS, Evans DC, Ali NA, Njoku C, et al. Prospective evaluation of intravascular volume status in critically ill patients: does inferior vena cava collapsibility correlate with central venous pressure?. J Trauma Acute Care Surg. 2014 Apr;76(4):956-63.. However, the practical difficulty in implementing these methods in a dialysis facility and the poor predictive value on blood pressure response in probing dry weight have precluded its generalizability4646 Agarwal R, Bouldin JM, Light RP, Garg A. Inferior vena cava diameter and left atrial diameter measure volume but not dry weight. Clin J Am Soc Nephrol. 2011 May;6(5):1066-72.. However, recent data in critically ill patients showed that IVCD collapsibility had reasonable value (c-statistic 0.72) in predicting tolerance to fluid removal4747 Kaptein MJ, Kaptein JS, Oo Z, Kaptein EM. Relationship of inferior vena cava collapsibility to ultrafiltration volume achieved in critically ill hemodialysis patients. Int J Nephrol Renovasc Dis. 2018 Jul;11:195-209..
Relative blood volume change (RBV) and refilling rate capacity during dialysis assessed by online blood volume sensor has been also proposed for fluid management. In expert hands, this tool provides useful information on individual patient volume status to facilitate hemodynamic guidance4949 Santoro A, Mancini E, Basile C, Amoroso L, Di Giulio S, Usberti M, et al. Blood volume controlled hemodialysis in hypotension-prone patients: a randomized, multicenter controlled trial. Kidney Int. 2002 Sep;62(3):1034-45.. Furthermore, blood volume monitoring has been shown helpful to set individual patient critical volemia beyond which occurrence of severe intradialytic hypotension is likely to occur5050 Barth C, Boer W, Garzoni D, Kuenzi T, Ries W, Schaefer R, et al. Characteristics of hypotension-prone haemodialysis patients: is there a critical relative blood volume?. Nephrol Dial Transplant. 2003 Jul;18(7):1353-60.. Despite the fact that most studies using blood volume monitoring (BVM) devices are reporting positive feedback on blood pressure control and hemodynamic stability5151 Covic A, Ciumanghel AI, Siriopol D, Kanbay M, Dumea R, Gavrilovici C, et al. Value of bioimpedance analysis estimated "dry weight" in maintenance dialysis patients: a systematic review and meta-analysis. Int Urol Nephrol. 2017;49(12):2231-2245., their clinical benefit is still matter of controversy5252 Keane DF, Baxter P, Lindley E, Rhodes L, Pavitt S. Time to reconsider the role of relative blood volume monitoring for fluid management in hemodialysis. ASAIO J. 2018 Nov/Dec;64(6):812-818.. Furthermore, in a randomized controlled trial (CLIMB Study) comparing BVM guided treatment versus standard of care had negative results that were associated with adverse outcomes5353 Reddan DN, Szczech LA, Hasselblad V, Lowrie EG, Lindsay RM, Himmelfarb J, et al. Intradialytic blood volume monitoring in ambulatory hemodialysis patients: a randomized trial. J Am Soc Nephrol. 2005 Jul;16(7):2162-9.. In a more recent study, BVM guided feedback did not result in an improvement in intradialytic hemodynamic stability although also no evidence of harm was found5454 Leung KCW, Quinn RR, Ravani P, Duff H, MacRae JM. Randomized Crossover Trial of Blood Volume Monitoring-Guided Ultrafiltration Biofeedback to Reduce Intradialytic Hypotensive Episodes with Hemodialysis. Clin J Am Soc Nephrol. 2017 Nov 7;12(11):1831-1840.. Absolute blood volume measurement, based on non-invasive measurement either by dilution or online calculation, has been proposed recently for a better assessment of this crucial parameter5555 Thijssen S, Kappel F, Kotanko P. Absolute blood volume in hemodialysis patients: why is it relevant, and how to measure it?. Blood Purif. 2013;35(1-3):63-71.,5656 Kron J, Schneditz D, Leimbach T, Aign S, Kron S. A simple and feasible method to determine absolute blood volume in hemodialysis patients in clinical practice. Blood Purif. 2014;38(3-4):180-7.,5757 Kron S, Schneditz D, Leimbach T, Czerny J, Aign S, Kron J. Determination of the critical absolute blood volume for intradialytic morbid events. Hemodial Int. 2016 Apr;20(2):321-6.. To date, no study has explored the clinical benefits of monitoring precisely this parameter.
Bioimpedance approach has been proposed over the last few years as a more objective way to assess fluid status in dialysis patients5858 Thomasset AL, Lenoir J, Roullet C, Jenin P, Béruard M, Bernard C, et al. The physiological surveillance of hemodialysis sessions by the continuous measurement of L.F. impedance of the circulating blood (Thomasset's method). Clin Exp Dial Apheresis. 1983;7(3):235-50.,5959 Chamney PW, Krämer M, Rode C, Kleinekofort W, Wizemann V. A new technique for establishing dry weight in hemodialysis patients via whole body bioimpedance. Kidney Int. 2002 Jun;61(6):2250-8.. Several approaches (segmental versus total body, single versus multifrequency) using various devices and algorithms have been developed with interesting results6060 Kotanko P, Levin NW, Zhu F. Current state of bioimpedance technologies in dialysis. Nephrol Dial Transplant. 2008 Mar;23(3):808-12.. In a systematic review, multifrequency bioimpedance spectroscopy (BIS) analysis [NICE, UK; CADTH, Canada] was recognized as the most precise and reliable tool in a clinical setting for guiding fluid management in dialysis patients at present available, although its use has not entered clinical guidelines yet6161 National Institute for Health and Care Excellence (NICE). NICE Guidance - Multiple frequency bioimpedance devices to guide fluid management in people with chronic disease having dialysis. London, UK: NICE; 2017 Jun; [cited 2019 Mar 23]. Available from: https://www.nice.org.uk/guidance/dg29/resources/multiple-frequency-bioimpedance-devices-to-guide-fluid-management-in-people-with-chronic-kidney-disease-having-dialysis-pdf-1053698654149
https://www.nice.org.uk/guidance/dg29/re...
,6262 CANADA ; [cited 2019 Mar 23]. Available from: https://www.cadth.ca/sites/default/files/pdf/htis/aug-2015/RC0695-Bioimpedance%20Final.pdf
https://www.cadth.ca/sites/default/files...
. In addition, extensive use of BIS in clinical studies has generated substantial evidences showing that BIS was able to detect subtle fluid volume variation6363 Keane D, Chamney P, Heinke S, Lindley E. Use of the Body Composition Monitor for Fluid Status Measurements in Subjects with High Body Mass Index. Nephron. 2016;133(3):163-8.,6464 Wabel P, Moissl U, Chamney P, Jirka T, Machek P, Ponce P, et al. Towards improved cardiovascular management: the necessity of combining blood pressure and fluid overload. Nephrol Dial Transplant. 2008 Sep;23(9):2965-71.,6565 Chazot C, Wabel P, Chamney P, Moissl U, Wieskotten S, Wizemann V. Importance of normohydration for the long-term survival of haemodialysis patients. Nephrol Dial Transplant. 2012 Jun;27(6):2404-10. and to support the notion that more precise fluid management might improve intermediate outcomes and dialysis patient endpoints6666 Antlanger M, Hecking M, Haidinger M, Werzowa J, Kovarik JJ, Paul G, et al. Fluid overload in hemodialysis patients: a cross-sectional study to determine its association with cardiac biomarkers and nutritional status. BMC Nephrol. 2013 Dec 2;14:266.,6767 Antlanger M, Josten P, Kammer M, Exner I, Lorenz-Turnheim K, Eigner M, et al. Blood volume-monitored regulation of ultrafiltration to decrease the dry weight in fluid-overloaded hemodialysis patients: a randomized controlled trial. BMC Nephrol. 2017 Jul 17;18(1):238.,6868 Canadian Agency for Drugs and Technologies in Health. Bioimpedance Devices for the Assessment of Body Fluid Volume for Patients Undergoing Dialysis: A Review of the Clinical Effectiveness, Cost-Effectiveness, and Guidelines [Internet]. CADTH Rapid Response Reports. 2014 Mar; [cited 2017 Sep 5]. Available from: https://www.cadth.ca/sites/default/files/pdf/htis/nov-2014/RC0534%20Bioimpedence%20Device%20Final.pdf
https://www.cadth.ca/sites/default/files...
. Few prospective clinical trials in advanced kidney disease or dialysis patients are ongoing to define more precisely the value of BIS in managing fluid status and its impact on preservation of residual kidney function and on cardiovascular outcomes6969 Stenberg J, Henriksson C, Lindberg M, Furuland H. Perspectives on clinical use of bioimpedance in hemodialysis: focus group interviews with renal care professionals. BMC Nephrol. 2018 May 23;19(1):121.,7070 Davies SJ, Caskey FJ, Coyle D, Lindley E, MacDonald J, Mitra S, et al. Rationale and design of BISTRO: a randomized controlled trial to determine whether bioimpedance spectroscopy-guided fluid management maintains residual kidney function in incident haemodialysis patients. BMC Nephrol. 2017 Apr 26;18(1):138..
More recently, it has also been proposed to extend the use of lung ultrasound in chronic hemodialysis patients for tracking silent fluid accumulation in the lung interstitium (extravascular edema). Interlobular septa thickening due to water accumulation reflects US beam and generates visible B line bundles (comet-like tail). A simple counting of these B lines provides an estimate of lung water excess and predictive value for patient outcomes7171 Enia G, Torino C, Panuccio V, Tripepi R, Postorino M, Aliotta R, et al. Asymptomatic pulmonary congestion and physical functioning in hemodialysis patients. Clin J Am Soc Nephrol. 2013 Aug;8(8):1343-8.,7272 Siriopol D, Onofriescu M, Voroneanu L, Apetrii M, Nistor I, Hogas S, et al. Dry weight assessment by combined ultrasound and bioimpedance monitoring in low cardiovascular risk hemodialysis patients: a randomized controlled trial. Int Urol Nephrol. 2017 Jan;49(1):143-153..
Sodium MRI has been introduced quite recently in the field of sodium and fluid assessment in chronic kidney disease patients in dialysis to assess tissue sodium accumulation7373 Francis S, Buchanan CE, Prestwich B, Taal MW. Sodium MRI: a new frontier in imaging in nephrology. Curr Opin Nephrol Hypertens. 2017 Nov;26(6):435-441.. Tissue sodium, namely ‘free-water sodium’ or ‘sodium bound to proteoglycans’, accumulates in chronic kidney disease and modulates lymphangiogenesis and blood pressure via proinflammatory resident cells7474 Titze J, Shakibaei M, Schafflhuber M, Schulze-Tanzil G, Porst M, Schwind KH, et al. Glycosaminoglycan polymerization may enable osmotically inactive Na+ storage in the skin. Am J Physiol Heart Circ Physiol. 2004 Jul;287(1):H203-8.,7575 Titze J, Rakova N, Kopp C, Dahlmann A, Jantsch J, Luft FC. Balancing wobbles in the body sodium. Nephrol Dial Transplant. 2016 Jul;31(7):1078-81.. Recent studies have shown that tissue sodium might contribute to systemic toxicity via local tissue and organ damage7676 Marvar PJ, Gordon FJ, Harrison DG. Blood pressure control: salt gets under your skin. Nat Med. 2009 May;15(5):487-8.,7777 Machnik A, Neuhofer W, Jantsch J, Dahlmann A, Tammela T, Machura K, et al. Macrophages regulate salt-dependent volume and blood pressure by a vascular endothelial growth factor-C-dependent buffering mechanism. Nat Med. 2009 May;15(5):545-52.. Left ventricular hypertrophy is positively associated with the amount of tissue sodium storage independent from blood pressure7878 Schneider MP, Raff U, Kopp C, Scheppach JB, Toncar S, Wanner C, et al. Skin Sodium Concentration Correlates with Left Ventricular Hypertrophy in CKD. J Am Soc Nephrol. 2017 Jun;28(6):1867-1876.. Vascular stiffness is also associated with sodium intake and sodium tissue storage independent from mechanical stress7979 Safar ME, Temmar M, Kakou A, Lacolley P, Thornton SN. Sodium intake and vascular stiffness in hypertension. Hypertension. 2009 Aug;54(2):203-9.,8080 Shirwany NA, Zou MH. Arterial stiffness: a brief review. Acta Pharmacol Sin. 2010 Oct;31(10):1267-76.. Furthermore, sodium tissue accumulation might contribute to metabolic and inflammatory disorders (e.g., insulin resistance, protein energy wasting) that increase cardiovascular risk. Due to its complex setting and limited number of scanning devices, sodium MRI remains an experimental tool with quite restricted access. However, it is envisioned that in the near future the dedicated extremity sodium MRI device, such as in rheumatologic field, could be used in a more systematic way to assess salt tissue content in dialysis patients8181 Jordan CD, McWalter EJ, Monu UD, Watkins RD, Chen W, Bargerter NK, et al. Variability of CubeQuant T1?, quantitative DESS T2, and cones sodium MRI in knee cartilage. Osteoarthritis Cartilage. 2014 Oct;22(10):1559-67..
3. Cardiac and vascular biomarkers have been used extensively in an attempt to disentangle fluid status and cardiac dysfunction in dialysis patients. Atrial natriuretic peptides (ANP, BNP, and NT-proBNP) are the most popular ones for assessing fluid overload8282 Wang AY, Lai KN. Use of cardiac biomarkers in end-stage renal disease. J Am Soc Nephrol. 2008 Sep;19(9):1643-52.,8383 Hickman PE. Biomarkers and cardiac disease in patients with end-stage renal disease on dialysis. Clin Biochem Rev. 2011 May;32(2):115-9. while, on the other hand, copeptin (a vasopressin precursor) is more reflective of fluid depletion8484 Kim JS, Yang JW, Chai MH, Lee JY, Park H, Kim Y, et al. Copeptin in Hemodialysis Patients with Left Ventricular Dysfunction. Yonsei Med J. 2015 Jul;56(4):976-80.. Cardiovascular biomarkers reflecting cardiac or endothelium injury are also of interest to set a more precise and personalized fluid management approach. Sensitive troponin family markers (troponin I and T) have been used to detect critical myocardial hypoperfusion. In this context, troponins (I and/or T) reflecting cardiac injury appear to be the most appealing ones being correlated with hemodynamic stress intensity, bearing a high predictive value for future cardiac events8585 McIntyre CW. Effects of hemodialysis on cardiac function. Kidney Int. 2009 Aug;76(4):371-5.,8686 Breidthardt T, Burton JO, Odudu A, Eldehni MT, Jefferies HJ, McIntyre CW. Troponin T for the detection of dialysis-induced myocardial stunning in hemodialysis patients. Clin J Am Soc Nephrol. 2012 Aug;7(8):1285-92.,8787 Drexler B, Heinisch C, Balmelli C, Lassus J, Siirilä-Waris K, Arenja N, et al. Quantifying cardiac hemodynamic stress and cardiomyocyte damage in ischemic and nonischemic acute heart failure. Circ Heart Fail. 2012;5(1):17-24.,8888 Vaduganathan M, Bhatt DL. Elevated Troponin Levels in Stable Patients Undergoing Hemodialysis: A Red Flag or a Red Herring?. Am J Nephrol. 2016;43(3):170-2.. Several other cardiac and endothelial biomarkers (e.g., ADMA, FG23, ROS, NO pathways) appear promising either isolated or combined in assessing cardiovascular risk but deserve further studies to define their exact place in fluid management strategy since they reflect tissue remodeling, uremic byproducts, inflammation, or oxidative stress mechanisms8989 Aucella F, Maas R, Vigilante M, Tripepi G, Schwedhelm E, Margaglione M, et al. Methylarginines and mortality in patients with end stage renal disease: a prospective cohort study. Atherosclerosis. 2009 Dec;207(2):541-5.,9090 Tripepi G, Raso FM, Sijbrands E, Seck MS, Maas R, Boger R, et al. Inflammation and asymmetric dimethylarginine for predictingdeath and cardiovascular events in ESRD patients. Clin J Am Soc Nephrol. 2011 Jul;6(7):1714-21.,9191 Niizuma S, Iwanaga Y, Yahata T, Miyazaki S. Renocardiovascular Biomarkers: from the Perspective of Managing Chronic Kidney Disease and Cardiovascular Disease. Front Cardiovasc Med. 2017 Mar 6;4:10.. Although these cardiac biomarkers have been shown to be quite useful for patient risk stratification, their predictive (specificity and sensitivity) and/or clinical value in term of fluid status management has remained limited to clinical cases management9292 Chazot C, Van CV, Zaoui E, Vanel T, Hurot JM, Lorriaux C, et al. Fluid overload correction and cardiac history influence brain natriuretic peptide evolution in incident haemodialysis patients. Nephrol Dial Transplant. 2011 Aug;26(8):2630-4.. Interpretation and clinical application of these cardiac biomarkers should be done with caution and be integrated in strategic care planning of dialysis patients since their circulating levels reflect not only fluid status but also kidney function deterioration and cardiac remodeling9393 D'Marco L, Bellasi A, Raggi P. Cardiovascular biomarkers in chronic kidney disease: state of current research and clinical applicability. Dis Markers. 2015;2015:586569.,9494 Savoj J, Becerra B, Kim JK, Fusaro M, Gallieni M, Lombardo D, et al. Utility of Cardiac Biomarkers in the Setting of Kidney Disease. Nephron. 2019;141(4):227-235.. Multi-markers approach and time trend analysis of these biomarkers have been proposed to better support physician decision in stratifying cardiovascular risk but raising a cost issue9595 Bargnoux AS, Morena M, Jaussent I, Maurice F, Chalabi L, Leray-Moragues H, et al. A combined index of cardiac biomarkers as a risk factor for early cardiovascular mortality in hemodialysis patients. Clin Chem Lab Med. 2013 Sep;51(9):1865-74.,9696 Ishii J, Takahashi H, Kitagawa F, Kuno A, Okuyama R, Kawai H, et al. Multimarker approach to risk stratification for long-term mortality in patients on chronic hemodialysis. Circ J. 2015;79(3):656-63..
4. In recent past years, several researchers have develop algorithms to quantify sodium and water mass transfer during hemodialysis sessions using either mass balance equations based on the law of conservation of mass within the dialysis/patient system9797 Santos SF, Peixoto AJ. Sodium balance in maintenance hemodialysis. Semin Dial. 2010 Nov/Dec;23(6):549-55.,9898 Lambie SH, Taal MW, Fluck RJ, McIntyre CW. Online conductivity monitoring: validation and usefulness in a clinical trial of reduced dialysate conductivity. ASAIO J. 2005 Jan/Feb;51(1):70-6. or by modeling sodium mass transfer using ionic dialysance with dialysate and plasma sodium concentrations9999 Odudu A, Lambie S, Taal MW, Fluck RJ, McIntyre CW. Use of online conductivity monitoring to study sodium mass balance in chronic haemodialysis patients: prospects for treatment individualisation. Kidney Blood Press Res. 2011;34(6):439-46.. Interestingly, on one hand, these studies have confirmed the validity of such an approach by direct dialysis quantification using partial or total dialysate collection. On the other hand, it has been clearly shown that sodium and water mass transfer and kinetics might be considered as a patient profile characteristic; in other words, intra-individual variability was relatively narrow while inter-individual variability was tremendously high. Several putative causal factors are associated with individual sodium and water profile (sodium or osmotic set-point) but most likely reflecting life style and diet observance101101 Keen ML, Gotch FA. The association of the sodium "setpoint" to interdialytic weight gain and blood pressure in hemodialysis patients. Int J Artif Organs. 2007 Nov;30(11):971-9.,102102 Song JH, Park GH, Lee SY, Lee SW, Lee SW, Kim MJ. Effect of sodium balance and the combination of ultrafiltration profile during sodium profiling hemodialysis on the maintenance of the quality of dialysis and sodium and fluid balances. J Am Soc Nephrol. 2005 Jan;16(1):237-46.,103103 Penne EL, Levin NW, Kotanko P. Improving volume status by comprehensive dietary and dialytic sodium management in chronic hemodialysis patients. Blood Purif. 2010;30(1):71-8.. In addition, it has been shown that acting on sodium and water mass transfer by individualizing dialysis sodium prescription it was possible to alter patient perception (reduce thirst, and sodium and water intake), reduce interdialytic weight gain, and modify outcomes (reduce dry weight and arterial blood pressure)104104 Raimann JG, Thijssen S, Usvyat LA, Levin NW, Kotanko P. Sodium alignment in clinical practice - implementation and implications. Semin Dial. 2011 Sep/Oct;24(5):587-92.,105105 Basile C, Lomonte C. A neglected issue in dialysis practice: haemodialysate. Clin Kidney J. 2015 Aug;8(4):393-9.,106106 Chevalier L, Tielemans C, Debelle F, Vandervelde D, Fumeron C, Mandart L, et al. Isonatric Dialysis Biofeedback in Hemodiafiltration with Online Regeneration of Ultrafiltrate in Hypertensive Hemodialysis Patients: A Randomized Controlled Study. Blood Purif. 2016;41(1-3):87-93.. However, this approach is hampered by the need for frequent lab sampling, which is cumbersome for clinical practice.
Fluid and hemodynamic management in hemodialysis patients: Opportunities
Optimal management of fluid and sodium imbalance in dialysis patients is achieved by adjusting salt and fluid removal through dialysis and salt intake restriction, and fluid gain between dialysis sessions. This is the conventional approach obtained by adjusting ‘dry weight’ according to clinical judgment and complementary tools including dialysate sodium prescription adaptation described earlier. However, this approach may be hampered by the discontinuous nature of the HD treatment and/or patient intolerance to fluid and sodium removal. An obvious solution would be to increase time and/or frequency of dialysis sessions in patients with high inter-dialytic weight gains and/or intolerance for fluid removal, as this has been shown to reduce intradialytic hemodynamic stress. However, this approach will not always be possible for financial or logistic reasons, or because of the wish of the patient.
Modern technology using biosensors and sophisticated analytics provide tools for handling directly sodium and water during hemodialysis session in a more precise and personalized way that have potential for improving patient outcome109109 Daugirdas JT, Tattersall JE. Automated monitoring of hemodialysis adequacy by dialysis machines: potential benefits to patients and cost savings. Kidney Int. 2010 Nov;78(9):833-5.. In this context, the use of calibrated conductivity meters or microsensors placed on dialysis fluid paths both inlet and outlet have been used to develop specific algorithms able to determine the precise contribution of sodium salt among the bulk of electrolytes110110 Locatelli F, Manzoni C, Pontoriero G, Cavalli A, Di Filippo S, Taher A. Ionic Dialysance and Conductivity Modeling. In: Azar AT, editor. Modeling and Control of Dialysis Systems. Switzerland: Springer-Verlag Berlin Heidelberg; 2013. p. 811-865.,111111 Mercadal L, Servais A, Venditto M, Renault N, Isnard-Bagnis C, Deray G, et al. Measuring plasma conductivity to detect sodium load in hemodialysis patients. Clin J Am Soc Nephrol. 2008 May;3(3):743-6.. Furthermore, the disposition of sensors on the dialysate path offers a means to ensure a precise mass balance due to a closed circuit. In addition, the combined use of advanced analytics embedded in the central processor unit provides a way to ensure direct handling of sodium and water according to the targeted prescription and patient baselines. Artificial intelligence has been recently proposed for clinical guidance and decision-making support in adapting dialysis prescription (e.g., ultrafiltration rate, dialysate sodium, treatment time) to ensure an optimal fluid status control and to minimize hemodynamic stress. The added value of these tools needs to be assessed in the future by clinical studies.
Complementary clinical studies on a large scale should help to better characterize dialysis patients in term of diet sodium intake over prolonged time period and explore effects of this precise sodium and fluid management approach on patients intermediary and clinical endpoint outcomes.
Take home message
Dialysis adequacy concept has evolved over time and based on patient outcomes. Due to more efficient hemodialyzers, more technically advanced hemodialysis machines, and wider use of ultrapure dialysis fluid, efficiency and biocompatibility of renal replacement therapy have improved tremendously114114 Himmelfarb J, Ikizler TA. Hemodialysis. N Engl J Med. 2010 Nov 4;363(19):1833-45.,115115 MacRae JM, Copland M, Kiaii M. Progress in dialysis practice: an introduction. Semin Dial. 2014 Mar;27(2):85-6.,116116 Hueso M, Navarro E, Sandoval D, Cruzado JM. Progress in the Development and Challenges for the Use of Artificial Kidneys and Wearable Dialysis Devices. Kidney Dis (Basel). 2019;5(1):3-10.. Cardioprotective hemodialysis requires further decisive actions in which sodium and fluid management are top ranking117117 Ronco C, Bowry S, Tetta C. Dialysis patients and cardiovascular problems: Can technology help solve the complex equation?. Blood Purif. 2006;24(1):39-45.. However, restoring homeostasis of extracellular volume, achieving adequately blood pressure control, and preserving hemodynamic equilibrium of dialysis patients still remains a matter of concern118118 Weiner DE, Brunelli SM, Hunt A, Schiller B, Glassock R, Maddux FW, et al. Improving clinical outcomes among hemodialysis patients: a proposal for a "volume first" approach from the chief medical officers of US dialysis providers. Am J Kidney Dis. 2014;64(5):685-95.,119119 Zoccali C, Mallamaci F. Mapping Progress in Reducing Cardiovascular Risk with Kidney Disease: Managing Volume Overload. Clin J Am Soc Nephrol. 2018 Sep 7;13(9):1432-1434.. Restoring sodium and fluid mass balance of dialysis patients is moving from an over simplistic ‘dry weight’ approach to a more precise fluid management approach with support of new diagnostic and monitoring tools and will enter in a new era with availability of smart hemodialysis machines with direct dialysis sodium and water handling options and with the support of advanced technology and analytics.
References
-
1Perl J, Dember LM, Bargman JM, Browne T, Charytan DM, Flythe JE, et al. The Use of a Multidimensional Measure of Dialysis Adequacy-Moving beyond Small Solute Kinetics. Clin J Am Soc Nephrol. 2017 May 8;12(5):839-847.
-
2Van Biesen W, Vanholder R, Lameire N. In search of the holy grail and the golden fleece. Perit Dial Int. 2004 Nov/Dec;24(6):501-3.
-
3Charra B. 'Dry weight' in dialysis: the history of a concept. Nephrol Dial Transplant. 1998 Jul;13(7):1882-5.
-
4Chazot C, Charra B, Van CV, Jean G, Vanel T, Calemard E, et al. The Janus-faced aspect of 'dry weight'. Nephrol Dial Transplant. 1999 Jan;14(1):121-4.
-
5McIntyre CW. Recurrent circulatory stress: the dark side of dialysis. Semin Dial. 2010 Sep/Oct;23(5):449-51.
-
6London GM. Ultrafiltration intensification for achievement of dry weight and hypertension control is not always the therapeutic gold standard. J Nephrol. 2011 Jul/Aug;24(4):395-7.
-
7Canaud B, Kooman J, Selby NM, Taal M, Francis S, Kopperschmidt P, et al. Sodium and water handling during hemodialysis: new pathophysiologic insights and management approaches for improving outcomes in end-stage kidney disease. Kidney Int. 2019 Feb;95(2):296-309.
-
8Kjellstrand CM, Evans RL, Petersen RJ, Shideman JR, Von Hartitzsch B, Buselmeier TJ. The “unphysiology” of dialysis: a major cause of dialysis side effects?. Hemodial Int. 2004 Jan;8(1):24-9.
-
9Kim GH. Dialysis unphysiology and sodium balance. Electrolyte Blood Press. 2009 Dec;7(2):31-7.
-
10McGuire S, Horton EJ, Renshaw D, Jimenez A, Krishnan N, McGregor G. Hemodynamic Instability during Dialysis: The Potential Role of Intradialytic Exercise. Biomed Res Int. 2018 Feb 27;2018:8276912.
-
11Heerspink HJL, Navis G, Ritz E. Salt intake in kidney disease: a missed therapeutic opportunity?. Nephrol Dial Transplant. 2012 Sep;27(9):3435-42.
-
12Kalantar-Zadeh K, Regidor DL, Kovesdy CP, Van Wyck D, Bunnapradist S, Horwich TB, et al. Fluid retention is associated with cardiovascular mortality in patients undergoing long-term hemodialysis. Circulation. 2009 Feb 10;119(5):671-9.
-
13Weiner DE, Brunelli SM, Hunt A, Schiller B, Glassock R, Maddux FW, et al. Improving clinical outcomes among hemodialysis patients: a proposal for a "volume first" approach from the chief medical officers of US dialysis providers. Am J Kidney Dis. 2014 Nov;64(5):685-95.
-
14Agarwal R, Martinez-Castelao A, Wiecek A, Massy Z, Suleymanlar G, Ortiz A, et al. The lingering dilemma of arterial pressure in CKD: what do we know, where do we go?. Kidney Int Suppl. 2011 Jun;1(1):17-20.
-
15Bansal N, McCulloch CE, Lin F, Alper A, Anderson AH, Cuevas M, et al. Blood Pressure and Risk of Cardiovascular Events in Patients on Chronic Hemodialysis: The CRIC Study (Chronic Renal Insufficiency Cohort). Hypertension. 2017 Aug;70(2):435-443.
-
16Georgianos PI, Agarwal R. Blood Pressure and Mortality in Long-Term Hemodialysis/ Time to Move Forward. Am J Hypertens. 2017 Mar 1;30(3):211-222.
-
17Zoccali C, Moissl U, Chazot C, Mallamaci F, Tripepi G, Arkossy O, et al. Chronic Fluid Overload and Mortality in ESRD. J Am Soc Nephrol. 2017 Aug;28(8):2491-2497.
-
18Hecking M, Karaboyas A, Saran R, Sen A, Hörl WH, Pisoni RL, et al. Predialysis serum sodium level, dialysate sodium, and mortality in maintenance hemodialysis patients: the Dialysis Outcomes and Practice Patterns Study (DOPPS). Am J Kidney Dis. 2012 Feb;59(2):238-48.
-
19Kovesdy CP, Lott EH, Lu JL, Malakauskas SM, Ma JZ, Molnar MZ, et al. Hyponatremia, hypernatremia, and mortality in patients with chronic kidney disease with and without congestive heart failure. Circulation. 2012 Feb 7;125(5):677-84.
-
20Dekker MJ, Marcelli D, Canaud B, Konings CJ, Leunissen KM, Levin NW, et al. Unraveling the relationship between mortality, hyponatremia, inflammation and malnutrition in hemodialysis patients: results from the international MONDO initiative. Eur J Clin Nutr. 2016 Jul;70(7):779-84.
-
21Assa S, Hummel YM, Voors AA, Kuipers J, Westerhuis R, Jong PE, et al. Hemodialysis-induced regional left ventricular systolic dysfunction: prevalence, patient and dialysis treatment-related factors, and prognostic significance. Clin J Am Soc Nephrol. 2012 Oct;7(10):1615-23.
-
22Burton JO, Jefferies HJ, Selby NM, McIntyre CW. Hemodialysis-induced cardiac injury: determinants and associated outcomes. Clin J Am Soc Nephrol. 2009 May;4(5):914-20.
-
23Flythe JE, Kimmel SE, Brunelli SM. Rapid fluid removal during dialysis is associated with cardiovascular morbidity and mortality. Kidney Int. 2011 Jan;79(2):250-7.
-
24Flythe JE, Curhan GC, Brunelli SM. Disentangling the ultrafiltration rate-mortality association: the respective roles of session length and weight gain. Clin J Am Soc Nephrol. 2013 Jul;8(7):1151-61.
-
25Chazot C, Van CV, Lorriaux C, Deleaval P, Mayor B, Hurot JM, et al. Even a moderate fluid removal rate during individualised haemodialysis session times is associated with decreased patient survival. Blood Purif. 2017;44(2):89-97.
-
26Buchanan C, Mohammed A, Cox E, Köhler K, Canaud B, Taal MW, et al. Intradialytic Cardiac Magnetic Resonance Imaging to Assess Cardiovascular Responses in a Short-Term Trial of Hemodiafiltration and Hemodialysis. J Am Soc Nephrol. 2017 Apr;28(4):1269-1277.
-
27Chawla A, Sterns RH, Nigwekar SU, Cappuccio JD. Mortality and serum sodium: do patients die from or with hyponatremia?. Clin J Am Soc Nephrol. 2011 May;6(5):960-5.
-
28Shiba N, Shimokawa H. Chronic kidney disease and heart failure - Bidirectional close link and common therapeutic goal. J Cardiol. 2011 Jan;57(1):8-17.
-
29Khan YH, Sarriff A, Adnan AS, Khan AH, Mallhi TH. Chronic Kidney Disease, Fluid Overload and Diuretics: A Complicated Triangle. PLoS One. 2016 Jul 21;11(7):e0159335.
-
30Lim LM, Tsai NC, Lin MY, Hwang DY, Lin HY, Lee JJ, et al. Hyponatremia is Associated with Fluid Imbalance and Adverse Renal Outcome in Chronic Kidney Disease Patients Treated with Diuretics. Sci Rep. 2016 Nov 14;6:36817.
-
31Ishibe S, Peixoto AJ. Methods of assessment of volume status and intercompartmental fluid shifts in hemodialysis patients: implications in clinical practice. Semin Dial. 2004 Jan/Feb;17(1):37-43.
-
32Raimann JG, Zhu F, Wang J, Thijssen S, Kuhlmann MK, Kotanko P, et al. Comparison of fluid volume estimates in chronic hemodialysis patients by bioimpedance, direct isotopic, and dilution methods. Kidney Int. 2014 Apr;85(4):898-908.
-
33Charra B, Jean G, Hurot JM, Terrat JC, Vanel T, Van CV, et al. Clinical Determination of Dry Body Weight. Hemodial Int. 2001 Jan;5(1):42-50.
-
34Charra B. Fluid balance, dry weight, and blood pressure in dialysis. Hemodial Int. 2007 Jan;11(1):21-31.
-
35Sinha AD, Agarwal R. Setting the dry weight and its cardiovascular implications. Semin Dial. 2017 Nov;30(6):481-488.
-
36Canaud B, Lertdumrongluk P. Probing 'dry weight' in haemodialysis patients: 'back to the future'. Nephrol Dial Transplant. 2012 Jun;27(6):2140-3.
-
37Agarwal R, Weir MR. Dry-weight: a concept revisited in an effort to avoid medication-directed approaches for blood pressure control in hemodialysis patients. Clin J Am Soc Nephrol. 2010 Jul;5(7):1255-60.
-
38Leunissen KM, Kouw P, Kooman JP, Cheriex EC, DeVries PM, Donker AJ, et al. New techniques to determine fluid status in hemodialyzed patients. Kidney Int Suppl. 1993 Jun;41:S50-6.
-
39Leypoldt JK, Cheung AK. Evaluating volume status in hemodialysis patients. Adv Ren Replace Ther. 1998 Jan;5(1):64-74.
-
40Ozkahya M, Ok E, Toz H, Asci G, Duman S, Basci A, et al. Long-term survival rates in haemodialysis patients treated with strict volume control. Nephrol Dial Transplant. 2006 Dec;21(12):3506-13.
-
41Ito K, Ookawara S, Ueda Y, Miyazawa H, Yamada H, Goto S, et al. A Higher Cardiothoracic Ratio Is Associated with 2-Year Mortality after Hemodialysis Initiation. Nephron Extra. 2015 Dec 19;5(3):100-10.
-
42Goldfarb-Rumyantzev AS, Chelamcharla M, Bray BE, Leypoldt JK, Lavasani I, Nelson N, et al. Volume indicators and left ventricular mass during aggressive volume management in patients on thrice-weekly hemodialysis. Nephron Clin Pract. 2009;113(4):c270-80.
-
43Katzarski KS, Nisell J, Randmaa I, Danielsson A, Freyschuss U, Bergström J. A critical evaluation of ultrasound measurement of inferior vena cava diameter in assessing dry weight in normotensive and hypertensive hemodialysis patients. Am J Kidney Dis. 1997 Oct;30(4):459-65.
-
44Krause I, Birk E, Davidovits M, Cleper R, Blieden L, Pinhas L, et al. Inferior vena cava diameter: a useful method for estimation of fluid status in children on haemodialysis. Nephrol Dial Transplant. 2001 Jun;16(6):1203-6.
-
45Stawicki SP, Adkins EJ, Eiferman DS, Evans DC, Ali NA, Njoku C, et al. Prospective evaluation of intravascular volume status in critically ill patients: does inferior vena cava collapsibility correlate with central venous pressure?. J Trauma Acute Care Surg. 2014 Apr;76(4):956-63.
-
46Agarwal R, Bouldin JM, Light RP, Garg A. Inferior vena cava diameter and left atrial diameter measure volume but not dry weight. Clin J Am Soc Nephrol. 2011 May;6(5):1066-72.
-
47Kaptein MJ, Kaptein JS, Oo Z, Kaptein EM. Relationship of inferior vena cava collapsibility to ultrafiltration volume achieved in critically ill hemodialysis patients. Int J Nephrol Renovasc Dis. 2018 Jul;11:195-209.
-
48Dasselaar JJ, Huisman RM, Jong PE, Franssen CF. Measurement of relative blood volume changes during haemodialysis: merits and limitations. Nephrol Dial Transplant. 2005 Oct;20(10):2043-9.
-
49Santoro A, Mancini E, Basile C, Amoroso L, Di Giulio S, Usberti M, et al. Blood volume controlled hemodialysis in hypotension-prone patients: a randomized, multicenter controlled trial. Kidney Int. 2002 Sep;62(3):1034-45.
-
50Barth C, Boer W, Garzoni D, Kuenzi T, Ries W, Schaefer R, et al. Characteristics of hypotension-prone haemodialysis patients: is there a critical relative blood volume?. Nephrol Dial Transplant. 2003 Jul;18(7):1353-60.
-
51Covic A, Ciumanghel AI, Siriopol D, Kanbay M, Dumea R, Gavrilovici C, et al. Value of bioimpedance analysis estimated "dry weight" in maintenance dialysis patients: a systematic review and meta-analysis. Int Urol Nephrol. 2017;49(12):2231-2245.
-
52Keane DF, Baxter P, Lindley E, Rhodes L, Pavitt S. Time to reconsider the role of relative blood volume monitoring for fluid management in hemodialysis. ASAIO J. 2018 Nov/Dec;64(6):812-818.
-
53Reddan DN, Szczech LA, Hasselblad V, Lowrie EG, Lindsay RM, Himmelfarb J, et al. Intradialytic blood volume monitoring in ambulatory hemodialysis patients: a randomized trial. J Am Soc Nephrol. 2005 Jul;16(7):2162-9.
-
54Leung KCW, Quinn RR, Ravani P, Duff H, MacRae JM. Randomized Crossover Trial of Blood Volume Monitoring-Guided Ultrafiltration Biofeedback to Reduce Intradialytic Hypotensive Episodes with Hemodialysis. Clin J Am Soc Nephrol. 2017 Nov 7;12(11):1831-1840.
-
55Thijssen S, Kappel F, Kotanko P. Absolute blood volume in hemodialysis patients: why is it relevant, and how to measure it?. Blood Purif. 2013;35(1-3):63-71.
-
56Kron J, Schneditz D, Leimbach T, Aign S, Kron S. A simple and feasible method to determine absolute blood volume in hemodialysis patients in clinical practice. Blood Purif. 2014;38(3-4):180-7.
-
57Kron S, Schneditz D, Leimbach T, Czerny J, Aign S, Kron J. Determination of the critical absolute blood volume for intradialytic morbid events. Hemodial Int. 2016 Apr;20(2):321-6.
-
58Thomasset AL, Lenoir J, Roullet C, Jenin P, Béruard M, Bernard C, et al. The physiological surveillance of hemodialysis sessions by the continuous measurement of L.F. impedance of the circulating blood (Thomasset's method). Clin Exp Dial Apheresis. 1983;7(3):235-50.
-
59Chamney PW, Krämer M, Rode C, Kleinekofort W, Wizemann V. A new technique for establishing dry weight in hemodialysis patients via whole body bioimpedance. Kidney Int. 2002 Jun;61(6):2250-8.
-
60Kotanko P, Levin NW, Zhu F. Current state of bioimpedance technologies in dialysis. Nephrol Dial Transplant. 2008 Mar;23(3):808-12.
-
61National Institute for Health and Care Excellence (NICE). NICE Guidance - Multiple frequency bioimpedance devices to guide fluid management in people with chronic disease having dialysis. London, UK: NICE; 2017 Jun; [cited 2019 Mar 23]. Available from: https://www.nice.org.uk/guidance/dg29/resources/multiple-frequency-bioimpedance-devices-to-guide-fluid-management-in-people-with-chronic-kidney-disease-having-dialysis-pdf-1053698654149
» https://www.nice.org.uk/guidance/dg29/resources/multiple-frequency-bioimpedance-devices-to-guide-fluid-management-in-people-with-chronic-kidney-disease-having-dialysis-pdf-1053698654149 -
62CANADA ; [cited 2019 Mar 23]. Available from: https://www.cadth.ca/sites/default/files/pdf/htis/aug-2015/RC0695-Bioimpedance%20Final.pdf
» https://www.cadth.ca/sites/default/files/pdf/htis/aug-2015/RC0695-Bioimpedance%20Final.pdf -
63Keane D, Chamney P, Heinke S, Lindley E. Use of the Body Composition Monitor for Fluid Status Measurements in Subjects with High Body Mass Index. Nephron. 2016;133(3):163-8.
-
64Wabel P, Moissl U, Chamney P, Jirka T, Machek P, Ponce P, et al. Towards improved cardiovascular management: the necessity of combining blood pressure and fluid overload. Nephrol Dial Transplant. 2008 Sep;23(9):2965-71.
-
65Chazot C, Wabel P, Chamney P, Moissl U, Wieskotten S, Wizemann V. Importance of normohydration for the long-term survival of haemodialysis patients. Nephrol Dial Transplant. 2012 Jun;27(6):2404-10.
-
66Antlanger M, Hecking M, Haidinger M, Werzowa J, Kovarik JJ, Paul G, et al. Fluid overload in hemodialysis patients: a cross-sectional study to determine its association with cardiac biomarkers and nutritional status. BMC Nephrol. 2013 Dec 2;14:266.
-
67Antlanger M, Josten P, Kammer M, Exner I, Lorenz-Turnheim K, Eigner M, et al. Blood volume-monitored regulation of ultrafiltration to decrease the dry weight in fluid-overloaded hemodialysis patients: a randomized controlled trial. BMC Nephrol. 2017 Jul 17;18(1):238.
-
68Canadian Agency for Drugs and Technologies in Health. Bioimpedance Devices for the Assessment of Body Fluid Volume for Patients Undergoing Dialysis: A Review of the Clinical Effectiveness, Cost-Effectiveness, and Guidelines [Internet]. CADTH Rapid Response Reports. 2014 Mar; [cited 2017 Sep 5]. Available from: https://www.cadth.ca/sites/default/files/pdf/htis/nov-2014/RC0534%20Bioimpedence%20Device%20Final.pdf
» https://www.cadth.ca/sites/default/files/pdf/htis/nov-2014/RC0534%20Bioimpedence%20Device%20Final.pdf -
69Stenberg J, Henriksson C, Lindberg M, Furuland H. Perspectives on clinical use of bioimpedance in hemodialysis: focus group interviews with renal care professionals. BMC Nephrol. 2018 May 23;19(1):121.
-
70Davies SJ, Caskey FJ, Coyle D, Lindley E, MacDonald J, Mitra S, et al. Rationale and design of BISTRO: a randomized controlled trial to determine whether bioimpedance spectroscopy-guided fluid management maintains residual kidney function in incident haemodialysis patients. BMC Nephrol. 2017 Apr 26;18(1):138.
-
71Enia G, Torino C, Panuccio V, Tripepi R, Postorino M, Aliotta R, et al. Asymptomatic pulmonary congestion and physical functioning in hemodialysis patients. Clin J Am Soc Nephrol. 2013 Aug;8(8):1343-8.
-
72Siriopol D, Onofriescu M, Voroneanu L, Apetrii M, Nistor I, Hogas S, et al. Dry weight assessment by combined ultrasound and bioimpedance monitoring in low cardiovascular risk hemodialysis patients: a randomized controlled trial. Int Urol Nephrol. 2017 Jan;49(1):143-153.
-
73Francis S, Buchanan CE, Prestwich B, Taal MW. Sodium MRI: a new frontier in imaging in nephrology. Curr Opin Nephrol Hypertens. 2017 Nov;26(6):435-441.
-
74Titze J, Shakibaei M, Schafflhuber M, Schulze-Tanzil G, Porst M, Schwind KH, et al. Glycosaminoglycan polymerization may enable osmotically inactive Na+ storage in the skin. Am J Physiol Heart Circ Physiol. 2004 Jul;287(1):H203-8.
-
75Titze J, Rakova N, Kopp C, Dahlmann A, Jantsch J, Luft FC. Balancing wobbles in the body sodium. Nephrol Dial Transplant. 2016 Jul;31(7):1078-81.
-
76Marvar PJ, Gordon FJ, Harrison DG. Blood pressure control: salt gets under your skin. Nat Med. 2009 May;15(5):487-8.
-
77Machnik A, Neuhofer W, Jantsch J, Dahlmann A, Tammela T, Machura K, et al. Macrophages regulate salt-dependent volume and blood pressure by a vascular endothelial growth factor-C-dependent buffering mechanism. Nat Med. 2009 May;15(5):545-52.
-
78Schneider MP, Raff U, Kopp C, Scheppach JB, Toncar S, Wanner C, et al. Skin Sodium Concentration Correlates with Left Ventricular Hypertrophy in CKD. J Am Soc Nephrol. 2017 Jun;28(6):1867-1876.
-
79Safar ME, Temmar M, Kakou A, Lacolley P, Thornton SN. Sodium intake and vascular stiffness in hypertension. Hypertension. 2009 Aug;54(2):203-9.
-
80Shirwany NA, Zou MH. Arterial stiffness: a brief review. Acta Pharmacol Sin. 2010 Oct;31(10):1267-76.
-
81Jordan CD, McWalter EJ, Monu UD, Watkins RD, Chen W, Bargerter NK, et al. Variability of CubeQuant T1?, quantitative DESS T2, and cones sodium MRI in knee cartilage. Osteoarthritis Cartilage. 2014 Oct;22(10):1559-67.
-
82Wang AY, Lai KN. Use of cardiac biomarkers in end-stage renal disease. J Am Soc Nephrol. 2008 Sep;19(9):1643-52.
-
83Hickman PE. Biomarkers and cardiac disease in patients with end-stage renal disease on dialysis. Clin Biochem Rev. 2011 May;32(2):115-9.
-
84Kim JS, Yang JW, Chai MH, Lee JY, Park H, Kim Y, et al. Copeptin in Hemodialysis Patients with Left Ventricular Dysfunction. Yonsei Med J. 2015 Jul;56(4):976-80.
-
85McIntyre CW. Effects of hemodialysis on cardiac function. Kidney Int. 2009 Aug;76(4):371-5.
-
86Breidthardt T, Burton JO, Odudu A, Eldehni MT, Jefferies HJ, McIntyre CW. Troponin T for the detection of dialysis-induced myocardial stunning in hemodialysis patients. Clin J Am Soc Nephrol. 2012 Aug;7(8):1285-92.
-
87Drexler B, Heinisch C, Balmelli C, Lassus J, Siirilä-Waris K, Arenja N, et al. Quantifying cardiac hemodynamic stress and cardiomyocyte damage in ischemic and nonischemic acute heart failure. Circ Heart Fail. 2012;5(1):17-24.
-
88Vaduganathan M, Bhatt DL. Elevated Troponin Levels in Stable Patients Undergoing Hemodialysis: A Red Flag or a Red Herring?. Am J Nephrol. 2016;43(3):170-2.
-
89Aucella F, Maas R, Vigilante M, Tripepi G, Schwedhelm E, Margaglione M, et al. Methylarginines and mortality in patients with end stage renal disease: a prospective cohort study. Atherosclerosis. 2009 Dec;207(2):541-5.
-
90Tripepi G, Raso FM, Sijbrands E, Seck MS, Maas R, Boger R, et al. Inflammation and asymmetric dimethylarginine for predictingdeath and cardiovascular events in ESRD patients. Clin J Am Soc Nephrol. 2011 Jul;6(7):1714-21.
-
91Niizuma S, Iwanaga Y, Yahata T, Miyazaki S. Renocardiovascular Biomarkers: from the Perspective of Managing Chronic Kidney Disease and Cardiovascular Disease. Front Cardiovasc Med. 2017 Mar 6;4:10.
-
92Chazot C, Van CV, Zaoui E, Vanel T, Hurot JM, Lorriaux C, et al. Fluid overload correction and cardiac history influence brain natriuretic peptide evolution in incident haemodialysis patients. Nephrol Dial Transplant. 2011 Aug;26(8):2630-4.
-
93D'Marco L, Bellasi A, Raggi P. Cardiovascular biomarkers in chronic kidney disease: state of current research and clinical applicability. Dis Markers. 2015;2015:586569.
-
94Savoj J, Becerra B, Kim JK, Fusaro M, Gallieni M, Lombardo D, et al. Utility of Cardiac Biomarkers in the Setting of Kidney Disease. Nephron. 2019;141(4):227-235.
-
95Bargnoux AS, Morena M, Jaussent I, Maurice F, Chalabi L, Leray-Moragues H, et al. A combined index of cardiac biomarkers as a risk factor for early cardiovascular mortality in hemodialysis patients. Clin Chem Lab Med. 2013 Sep;51(9):1865-74.
-
96Ishii J, Takahashi H, Kitagawa F, Kuno A, Okuyama R, Kawai H, et al. Multimarker approach to risk stratification for long-term mortality in patients on chronic hemodialysis. Circ J. 2015;79(3):656-63.
-
97Santos SF, Peixoto AJ. Sodium balance in maintenance hemodialysis. Semin Dial. 2010 Nov/Dec;23(6):549-55.
-
98Lambie SH, Taal MW, Fluck RJ, McIntyre CW. Online conductivity monitoring: validation and usefulness in a clinical trial of reduced dialysate conductivity. ASAIO J. 2005 Jan/Feb;51(1):70-6.
-
99Odudu A, Lambie S, Taal MW, Fluck RJ, McIntyre CW. Use of online conductivity monitoring to study sodium mass balance in chronic haemodialysis patients: prospects for treatment individualisation. Kidney Blood Press Res. 2011;34(6):439-46.
-
100Basile C, Libutti P, Lisi P, Vernaglione L, Casucci F, Losurdo N, et al. Sodium setpoint and gradient in bicarbonate hemodialysis. J Nephrol. 2013 Nov/Dec;26(6):1136-42.
-
101Keen ML, Gotch FA. The association of the sodium "setpoint" to interdialytic weight gain and blood pressure in hemodialysis patients. Int J Artif Organs. 2007 Nov;30(11):971-9.
-
102Song JH, Park GH, Lee SY, Lee SW, Lee SW, Kim MJ. Effect of sodium balance and the combination of ultrafiltration profile during sodium profiling hemodialysis on the maintenance of the quality of dialysis and sodium and fluid balances. J Am Soc Nephrol. 2005 Jan;16(1):237-46.
-
103Penne EL, Levin NW, Kotanko P. Improving volume status by comprehensive dietary and dialytic sodium management in chronic hemodialysis patients. Blood Purif. 2010;30(1):71-8.
-
104Raimann JG, Thijssen S, Usvyat LA, Levin NW, Kotanko P. Sodium alignment in clinical practice - implementation and implications. Semin Dial. 2011 Sep/Oct;24(5):587-92.
-
105Basile C, Lomonte C. A neglected issue in dialysis practice: haemodialysate. Clin Kidney J. 2015 Aug;8(4):393-9.
-
106Chevalier L, Tielemans C, Debelle F, Vandervelde D, Fumeron C, Mandart L, et al. Isonatric Dialysis Biofeedback in Hemodiafiltration with Online Regeneration of Ultrafiltrate in Hypertensive Hemodialysis Patients: A Randomized Controlled Study. Blood Purif. 2016;41(1-3):87-93.
-
107Tomson CR. Advising dialysis patients to restrict fluid intake without restricting sodium intake is not based on evidence and is a waste of time. Nephrol Dial Transplant. 2001 Aug;16(8):1538-42.
-
108Jefferies HJ, Virk B, Schiller B, Moran J, McIntyre CW. Frequent hemodialysis schedules are associated with reduced levels of dialysis-induced cardiac injury (myocardial stunning). Clin J Am Soc Nephrol. 2011;6(6):1326-32.
-
109Daugirdas JT, Tattersall JE. Automated monitoring of hemodialysis adequacy by dialysis machines: potential benefits to patients and cost savings. Kidney Int. 2010 Nov;78(9):833-5.
-
110Locatelli F, Manzoni C, Pontoriero G, Cavalli A, Di Filippo S, Taher A. Ionic Dialysance and Conductivity Modeling. In: Azar AT, editor. Modeling and Control of Dialysis Systems. Switzerland: Springer-Verlag Berlin Heidelberg; 2013. p. 811-865.
-
111Mercadal L, Servais A, Venditto M, Renault N, Isnard-Bagnis C, Deray G, et al. Measuring plasma conductivity to detect sodium load in hemodialysis patients. Clin J Am Soc Nephrol. 2008 May;3(3):743-6.
-
112Kuhlmann U, Maierhofer A, Canaud B, Hoyer J, Gross M. Zero Diffusive Sodium Balance in Hemodialysis Provided by an Algorithm-Based Electrolyte Balancing Controller: A Proof of Principle Clinical Study. Artif Organs. 2019 Feb;43(2):150-158.
-
113Barbieri C, Cattinelli I, Neri L, Mari F, Ramos R, Brancaccio D, et al. Development of an Artificial Intelligence Model to Guide the Management of Blood Pressure, Fluid Volume, and Dialysis Dose in End-Stage Kidney Disease Patients: Proof of Concept and First Clinical Assessment. Kidney Dis (Basel). 2019 Feb;5(1):28-33.
-
114Himmelfarb J, Ikizler TA. Hemodialysis. N Engl J Med. 2010 Nov 4;363(19):1833-45.
-
115MacRae JM, Copland M, Kiaii M. Progress in dialysis practice: an introduction. Semin Dial. 2014 Mar;27(2):85-6.
-
116Hueso M, Navarro E, Sandoval D, Cruzado JM. Progress in the Development and Challenges for the Use of Artificial Kidneys and Wearable Dialysis Devices. Kidney Dis (Basel). 2019;5(1):3-10.
-
117Ronco C, Bowry S, Tetta C. Dialysis patients and cardiovascular problems: Can technology help solve the complex equation?. Blood Purif. 2006;24(1):39-45.
-
118Weiner DE, Brunelli SM, Hunt A, Schiller B, Glassock R, Maddux FW, et al. Improving clinical outcomes among hemodialysis patients: a proposal for a "volume first" approach from the chief medical officers of US dialysis providers. Am J Kidney Dis. 2014;64(5):685-95.
-
119Zoccali C, Mallamaci F. Mapping Progress in Reducing Cardiovascular Risk with Kidney Disease: Managing Volume Overload. Clin J Am Soc Nephrol. 2018 Sep 7;13(9):1432-1434.
Publication Dates
-
Publication in this collection
24 Oct 2019 -
Date of issue
Oct-Dec 2019
History
-
Received
27 June 2019 -
Accepted
08 July 2019