ABSTRACT
A major problem in cauliflower crop is the fungus Alternaria brassicicola, which causes dark leaf spot on Brassicaceae family. The current use of copper salts in agriculture is questioned. In fact, these products present some disadvantages, connected mainly with their deposits in the soil and toxicity on plants. This work investigated the effects of arsenic treatments, in ultra high diluted form (UHD), prepared by a process of repeated dilution and succussion (shaking), through: 1) in vitro germination experiments, where spores of A. brassicicola were suspended in the treatments; 2) in planta experiments and 3) a field trial, where cauliflower plants infected by the fungus were sprayed with treatments. The results showed that ultra high dilutions of arsenic (where no more molecules of this substance are present) were effective in all the experiments, inhibiting spore germination by 60.0%, controlling fungal disease in in planta experiments (relative efficacy of 42.1%), and, in field trial, decreasing the mean infection level in cauliflower heads by 45.7% and 41.6% in artificially inoculated and naturally infected plants, respectively. This is the first study to demonstrate that ultra high dilutions effectively reduce in vitro spore germination and infection of A. brassicicola in cauliflower plants, both under controlled conditions and in the field. Our research is still very experimental, however, in light of the significant results obtained with ultra-diluted arsenic, and given that its extreme high dilution level precludes any toxicity or accumulation in the environment, the use of UHDs could be considered a potential and reliable approach for sustainable agriculture.
Keywords:
Brassica oleracea; agro-homeopathy; crop protection.
RESUMO
Um grande entrave no cultivo da couve-flor é a incidência do fungo Alternaria brassicicola, causador da mancha foliar escura em plantas da família Brassicaceae. A utilização de sais de cobre na agricultura é questionada. De fato, estes produtos apresentam desvantagens, principalmente relacionadas com os seus depósitos no solo e a sua fitotoxicidade. Neste trabalho investigou-se os efeitos do uso de arsenico na forma ultra diluída (UHD), preparado por um processo de diluição repetida e agitação, por meio de: 1) germinação in vitro de esporos em suspenção de A. brassicicola; 2) experimento in planta e 3) ensaio em campo onde plantas de couve-flor, infectadas pelo fungo, foram pulverizadas. Diluições ultra altas de arsenico (em que não há mais presença dessas moléculas) foram eficazes em todos os experimentos, inibindo a germinação de esporos em 60,0%, controlando a doença fúngica nos experimentos in planta (eficácia relativa de 42,1%) e, no teste de campo, diminuindo o nível de infecção médio em cabeças de couve flor em 45,7% e 41,6% respectivamente, em plantas inoculadas artificialmente e infectadas naturalmente. Este é o primeiro estudo para demonstrar que as soluções ultra diluídas de arsenico efetivamente reduzem a germinação de esporos in vitro e reduzem a infecção de A. brassicicola em plantas de couve-flor, tanto em condições controladas como em campo. Nossa pesquisa ainda é muito experimental; no entanto, à luz dos resultados significativos obtidos com arsênico ultra-diluído e, dado que o elevado nível de diluição impede qualquer toxicidade ou acumulo no ambiente, o uso de UHDs poderia ser considerado uma abordagem potencial e confiável para a agricultura sustentável.
Palavras-chave:
Brassica oleracea; agro-homeopatia; protecção de culturas.
Dark leaf spot of cauliflower (Brassica oleracea), caused by Alternaria brassicicola, is a very common disease that causes serious production losses worldwide. Symptoms appear on the leaves as small black dots and enlarge to form target-like dark spots: the tissue becomes dry, brittle and often falls out, resulting in a 'shot hole' appearance of the leaf. On cauliflower curds, spotting appears as dark brown irregular sunken areas, often accompanied by bacterial soft rot that liquefies the tissue. The conventional strategies for control of dark leaf spot consist in the application of synthetic fungicides. To complement these, and to minimize their possible adverse effects on the environment and human health, in recent decades more environmentally sound alternatives (organic and biodynamic agriculture) have been developed (Fließbach et al., 2007FLIEßBACH, A; OBERHOLZER, HR; GUNST, L; MÄDER, P. 2007. Soil organic matter and biological soil quality indicators after 21 years of organic and conventional farming. Agriculture, Ecosystems & Environment 118: 273-284.; Turinek et al., 2009TURINEK, M; GROBELNIK-MLAKAR, S; BAVEC, M; BAVEC, F. 2009. Biodynamic agriculture research progress and priorities. Renewable Agriculture and Food Systems 24: 146-154.). Among the natural compounds admitted in such systems, copper-based products are widely applied for the control of dark leaf spot, as well as of most fungal diseases. Unfortunately, copper use presents some disadvantages: it can be phytotoxic, and it can accumulate in the ground with negative consequences on soil microflora and microfauna (Naidu & Bolan, 2008NAIDU, R; BOLAN, NS. 2008. Contaminant chemistry in soils: key concepts and bioavailability. In: NAIDU, R (eds). Chemical bioavailability in terrestrial environment. Amsterdam: Elsevier. p. 9-37.). For these reasons, the European Community has set the maximum amount of copper metal that can be used per hectare in organic agriculture (Commission Regulation EC no. 473/2002 of 15th March, 2002). Therefore, there is a need for experimental research aimed at identifying alternative or integrative treatments to cupric products. From a sustainability perspective, the emerging discipline of "agro-homeopathy" - the application of homeopathy to agriculture - could offer potential benefits because homeopathic preparations, due to their high dilution, are relatively cheap and may have few or no ecological side-effects. This agricultural approach is currently being developed in Brazil, where it is legally provided by the organic production regulations. In particular, homeopathic ultra high dilutions (UHDs), being beyond the Avogadro limit and thus without molecules of the original substance, have the advantage of not provoking any toxicity or accumulation in the environment. The term UHD denotes treatments prepared by the process of dynamization, consisting of dilution and rhythmic shaking (called succussion), starting from a mother tincture. Dilutions are usually performed on a decimal (1:10) or centesimal (1:100) scale, designated with the letters x and c, respectively. The biological effectiveness of UHDs is still controversial, with the central issue of the dispute being that, at such high dilution levels, any biological activity is exceedingly unlikely, under the conventional scientific paradigm of the "molecule/receptor" model. Nevertheless, there is emerging evidence of UHD activity in vitro and in vivo (Witt et al., 2007WITT, CM; BLUTH, M; ALBRECHT, H; WEISSHUHN, TER; BAUMGARTNER, S; WILLICH, SN. 2007. The in vitro evidence for an effect of high homeopathic potencies - a systematic review of the literature. Complementary Therapies in Medicine 15: 128-38.; Endler et al., 2010ENDLER, PC; THIEVES, K; REICH, C; MATTHIESSEN, P; BONAMIN, L; SCHERR, C; BAUMGARTNER, S. 2010. Repetitions of fundamental research models for homeopathically prepared dilutions beyond 10-23: a bibliometric study. Homeopathy99: 25-36.).To explain this effectiveness, alternative models have been proposed that lie outside the conventional framework, but nevertheless possess strong theoretical cogency. The hypothesis is that, since the dynamization process introduces mechanical energy and turbulence into the system, the preparation procedure (dilution and succussion) can generate long-term alterations in UHDs, as evinced by numerous physical and chemical variables (Yinnon & Elia, 2013YINNON, TA; ELIA, V. 2013. Dynamics in perturbed very dilute aqueous solutions: theory and experimental evidence. International Journal of Modern Physics B27: 1350005 Available in: http://www.worldscientific.com/doi/abs/10.1142/S0217979213500057. Accessed in March 26, 2014.
http://www.worldscientific.com/doi/abs/1...
). Such alterations might act as an "external frequency", capable of communicating its oscillations, via a process of resonance, to biological fluids and complex "metastable" structures capable of oscillating (macromolecules, alpha-helixes, membranes, filamentous structures, receptors) (Bellavite et al., 2014BELLAVITE, P; MARZOTTO, M; OLIOSO, D; MORATTI, E; CONFORTI, A. 2014. High-dilution effect revisited. 1. Physicochemical aspects. Homeopathy103: 4-21.).The scientific literature on the effects of UHDs on plants includes several studies, recently reviewed (Majevsky et al., 2009MAJEWSKY, V; ARLT, S; SHAH, D; SCHERR, C; JÄGER, T; BETTI, L; TREBBI, G; BONAMIN, L; KLOCKE, P; BAUMGARTNER, S. 2009. Use of homeopathic preparations in experimental studies with healthy plants. Homeopathy98: 228-243.; Jäger et al., 2011aJÄGER, T; SCHERR, C; SHAH, D; MAJEWSKY, V; BETTI, L; TREBBI, G; BONAMIN, L; SIMÕES-WÜST, AP; WOLF, U; SIMON, M; HEUSSER, P; BAUMGARTNER, S. 2011a. Use of homeopathic preparations in experimental studies with abiotically stressed plants. Homeopathy 100: 275-287.), most of which deal with the classical test of in vitro germination and growth. Other experimental models are based on artificially diseased plants (phytopathological models), where the stress is a fungal, viral, bacterial or nematode pathogen. As far as plant/fungus interactions are concerned, the results of most studies are unreliable due to poor experimental methodology and inadequate statistics, as recently reviewed (Betti et al., 2009BETTI, L; TREBBI, G; MAJEWSKY, V; SCHERR, C; SHAH-ROSSI, D; JÄGER, T, BAUMGARTNER, S. 2009. Use of homeopathic preparations in phytopathological models and in field trials: a critical review. Homeopathy 98: 244-266.).With respect to the application of UHDs in field trials, only a few studies have been performed, on both healthy and infected crops, as reviewed by Betti et al. (2009)BETTI, L; TREBBI, G; MAJEWSKY, V; SCHERR, C; SHAH-ROSSI, D; JÄGER, T, BAUMGARTNER, S. 2009. Use of homeopathic preparations in phytopathological models and in field trials: a critical review. Homeopathy 98: 244-266.. One in particular (Sukul et al., 2007SUKUL, NC; CHATTOPADHYAY, S; DAS, C; SUKUL, A; GHOSH, S; SINHA BABU, SP. 2007. Enhancement of photosynthesis and plant growth by potentized drugs. Journal of Environment and Ecology 25S: 520-524.) reports a significant increase in certain physiological parameters observed in healthy plants following treatments with UHDs. Recently, a UHD of a plant growth retardant has been showed to promote growth and yield in rice varieties tested under natural field conditions (Mondal et al., 2013MONDAL, S; SUKUL, NEE CHUNARI, S; CHANDRA-SUKUL, N. 2013. A new homeopathic potency promotes growth and yield of rice: a field trial. International Journal of High Dilution Research 12: 115.). Moreover, the effects of high diluted preparations for pest management have been reported, showing significant (Modolon et al., 2012MODOLON, TA; BOFF, P; BOFF, MIC; MIQUELLUTI, DJ. 2012. Homeopathic and high dilution preparations for pest management to tomato crop under organic production system. Horticultura Brasileira 30: 51-57.) or not significant results (Rupp et al., 2012RUPP, LCD; BOFF, MIC; BOFF, P; DE, S; GONÇALVES, PA; BOTTON, M. 2012. High dilution of Staphysagria and fruit fly biotherapic preparations to manage South American fruit fly, Anastrepha fraterculus, in organic peach orchards. Biological Agriculture & Horticulture 28: 41-48.) in organic tomato crop and peach orchards, respectively.
The aim of the present investigation was to select some UHDs that might be proposed for practical use in a sustainable agriculture context. To this end, we conducted an initial screening in the laboratory to investigate whether certain test substances can have direct effects on A. brassicicola spores (in vitro germination experiments) or/and control fungal infections in cauliflower plants under controlled conditions (in planta experiments). After selecting the test substances which yielded the best pathogen control, the next step was to verify their efficacy in a field trial.
MATERIAL AND METHODs
Selection and preparation of treatments
The classes of treatments selected for testing were: 1) distilled water (C, negative control, p.A. Merck KGaA, Darmstadt, Germany); 2) D,L-3-ß-aminobutyric acid, 5 mM (BABA, positive control); 3) copper oxychloride 3, 1 and 0.3 g/L (Cu 3, as positive control, 1 and 0.3); 4) arsenic trioxide (As2O3) 35x and 45x (As 35x and 45x); 5) Cuprum metallicum 35x and 45x (Cupr 35x and 45x); 6) nosode 35x and 45x; 7) bentonite 10 g/L (Bent).
We selected the above treatments for initial screening based on a number of approaches. The first one was to follow the Hahnemannian "similia principle", according to which treatments are chosen on the basis of the symptoms: in our experimentation, we selected arsenic trioxide (As2O3, Sigma-Aldrich, St. Louis, MO, USA) because, at 1 mM, it induces necrotic spots on cauliflower leaves similar to those provoked by A. brassicicola infection. However, since As2O3 in mM concentrations can significantly contribute to soil contamination and its use in agricultural practice is not allowed, we opted to evaluate its efficacy in ultra high aqueous decimal dilutions: As2O3 35x and 45x, where no more molecules of arsenic are present, being the dilution levels beyond the Avogadro limit. These particular dynamization levels were chosen based on the results of a previous study which found that, in an in vitro wheat germination model, those same treatments had significant effects on seed germination rate (Brizzi et al., 2000BRIZZI, M; NANI, D; PERUZZI, M; BETTI, L. 2000. Statistical analysis of the effect of high dilutions of arsenic in a large data set from a wheat germination model. British Homeopathic Journal 89: 63-67.). Following on a second approach, since copper is known to have anti-fungal activity, we selected it for use in ultra high dilutions (35x and 45x). A third approach led to selection of a nosode (Shah-Rossi et al., 2009SHAH-ROSSI, D; HEUSSER, P; BAUMGARTNER, S. 2009. Homeopathic treatment of Arabidopsis thaliana plants infected with Pseudomonas syringaeThe Scientific World Journal 9: 320-330.) i.e., an extract of plant tissue infected by A. brassicicola, used in ultra highly diluted form (35x and 45x) to avoid the presence of mycelium particles and spores. Finally, we tested a natural treatment based on bentonite, a clay normally used in organic agriculture against fungal infections, which has an absorbent action that soaks up humidity, inhibiting fungal germination and growth (personal communication). To demonstrate the reactivity of the system, some substances well-known for their inhibiting effect on dark leaf spot infection were tested as positive controls: in particular, we chose D,L-3-ß-aminobutyric acid (BABA, Sigma-Aldrich, St. Louis, MO, USA) because it is a resistance inducer (Cohen, 2002COHEN, YR. 2002. ß-aminobutyric acid-induced resistance against plant pathogens. Plant Disease 86: 448-457.), and copper oxychloride because of its anti-fungal activity.
For what concerns the preparation method, the 35x and 45x treatments were obtained starting from As2O3 0.01 M and then performing serial dilutions in decimal scale: cleaned polyethylene bottles (250 mL) were filled to 72% of capacity (180 mL) with 162 mL of distilled water plus 18 mL of the previous dilution level.
For what concerns dynamization, at each dilution step the bottles were succussed using a mechanical apparatus (DYNA HV1 by Debofar N.V.S.A., Oostduinkerke, Belgium) which applied 100 vertical strokes at the frequency of 8.3 Hz in a single succussion process. The process of dynamization comprises several steps, each one having a dilution followed by 100 succussions. This procedure (serial dilution and succussion) was continued until the 35x and 45x dilution levels were reached. Cuprum metallicum 35x and 45x in aqueous solution were provided by Boiron srl (Milano, Italy), prepared according to the European Pharmacopoeia. The nosode (remedy prepared from a pathological specimen) was freshly prepared for each experiment as follows: 5 artificially inoculated leaves (see below) and 5 healthy leaves of cauliflower plants were mixed (Shah-Rossi et al., 2009SHAH-ROSSI, D; HEUSSER, P; BAUMGARTNER, S. 2009. Homeopathic treatment of Arabidopsis thaliana plants infected with Pseudomonas syringaeThe Scientific World Journal 9: 320-330.) and ground for 5 min in a mortar with 4 mL of distilled water. The extract, after filtration through a piece of cotton wool, was diluted and dynamized up to 35x and 45x, in the same manner described above. Bentonite (Cosmoonda snc, Sarsina, FC, Italy) was suspended in water at 10 g/L as indicated on the label. The concentration of copper oxychloride (SIAPA, Milan, Italy) at 3 g/L (indicated on the label) was chosen because it was within the 6 kg/ha/year limit permitted by Commission Regulation EC no. 473/2002 (in the field trial the plot area was 5.76 m2, the quantity of treatment/plot was 180 mL and thus the applied concentration was 5 kg/ha/year). Copper oxychloride at 0.3 and 1 g/L was tested to evaluate whether it retained some effectiveness also at lower concentrations.
All test samples were prepared the day before the experiment and stored at 4°C until use, in order to reduce microbial growth. Moreover, all treatments were letter-coded (blinded) by a person not involved in the experiments and the codes were kept by independent people until data analysis.
Plant material and fungal isolate
Plants of Brassica oleracea cultivar clx 33247 were used for both in planta and field experiments. Plants were grown in pots in a growth chamber with a photoperiod of 16:8 h light dark (1.87 W/m2) and 75-80% relative humidity (RH), at 25±1°C, up to vegetative stage of 5±1 leaves (about 4-5 weeks). An isolate of A. brassicicola (strain M 18), part of the fungi collection of Department of Agricultural Sciences (DipSA, Bologna University, Italy), was used. Conidia were harvested from 2-week old cultures grown in 4% potato-dextrose-agar (PDA, BD Difco, USA) plus 0.3 g/L streptomycin sulphate in 9-cm Petri dishes at 25°C in dark, by flooding the plates with sterile water and dislodging conidia by softly brushing the colonies with a sterile spatula.
In vitro germination experiments
We performed preliminary trials to determine which conidia concentration yielded the highest in vitro germination. Different concentrations, evaluated with a Thoma cell, were tested: 104, 105, 106. The concentration chosen for in vitro experiments was 105 spores/mL, which yielded a mean germination rate of about 30% and a sufficiently high number of spores in each frame. The stability of the experimental set-up was evaluated by performing a preliminary negative control experiment using distilled water as the only test substance in fungal suspensions, following the same protocol explained below.
Fungal suspensions were prepared in the following treatments: As 35x and 45x, Cupr 35x and 45x, nosode 35x and 45x and Cu 3, versus negative control. Two drops (30 μL each) of each spore suspension were placed on sterile slides, which were laid on 2 glass rods on filter paper moistened with 15 mL of distilled water in Petri dishes (150 by 20 mm) sealed with Parafilm. For each treatment, we used 10 Petri dishes, each containing 2 slides (control and treatment) with 2 drops/slide. After incubation at 25°C for 5 h, the conidia were observed using an optical microscope (Orthoplan Leitz, Wetzlar, Germany): we analysed 2 non-overlapping random frames for each drop (4 frames/slide), counting total and germinated spores to evaluate the germination rate. Germination was defined as the development of a hyphal tube of any size arising from a conidium.
In planta experiments
A negative control experiment was performed to evaluate the stability of the experimental set-up: three groups of twelve thirty-day-old cauliflower plants each were treated with distilled water as the only test substance, before and after fungal inoculation, following the same protocol applied in the growth chamber experiments (see below). The experiment was repeated 3 times and disease severity was evaluated blind.
In the experiments with test substances, the same treatments previously tested in vitro for spore germination and the BABA positive control were compared with respect to negative controls. Cauliflower plants were sprayed (12 plants/treatment, 20 mL/plant) both before and after fungal inoculation (pre- and post-inoculation treatments, respectively) in order to maximize any effects of the UHDs. Pre-inoculation treatments were sprayed on all the leaves of each plant on the 1st, 4th and 7th day from the start of the experiment. Plants were covered with clear polyethylene bags immediately before inoculation and for the ensuing 4 days to allow infection; inoculation was performed on the 11th day by spraying the whole plants with a conidial suspension at 106 conidia/mL (about 7 mL/plant), through a hole purposely created in the bag. Before each spraying, the fungal suspension was shaken to maintain a homogeneous concentration. Finally, post-inoculation treatments were sprayed on all the leaves of each plant through the same hole on the 12th, 13th and 14th days. On the 15th day the bags were taken away and 8 days after that (12 days after inoculation) disease development was assessed blind. For each plant, all the inoculated leaves were rated using a disease severity index scale ranging from 0 to 4, as modified from Doullah et al. (2006DOULLAH, MAU; MEAH, MB; OKAZAKI, K. 2006. Development of an effective screening method for partial resistance to Alternaria brassicicola (dark leaf spot) in Brassica rapaEuropean Journal of Plant Pathology 116: 33-43.), where 0= no visible symptoms; 1= a few pinpoint spots but no chlorosis; 2= some spots with a few lesions surrounded by light chlorosis; 3= increasing number and size of lesions and chlorosis on the leaf; 4= lesions with chlorosis on more than 75% of the leaf. In each experiment, two different treatments were tested against the negative control.
Field trial
The field trial was performed at the Bologna University experimental farm (Cadriano, Bologna, Italy) from September to December 2006. A field measuring 40 x 9.6 m (L x W) was prepared to accommodate 8 rows, each consisting of 66 seedlings; four-week old seedlings (3 or 4 fully expanded leaves) were transplanted on September 13th 2006. Seedlings were irrigated with an overhead sprinkler system immediately after transplanting and then watered for 1 h twice a week (approx. 6-8 mm rainfall). Rows were spaced 1.20 m apart, while plants on the same row were planted 0.6 m apart. Plants on the two external rows at either end of the test field area were boundary non-treated plants. The remaining internal rows were divided into plots (4.8×1.2 m, L x W), consisting of 6 plants per treatment, separated by 2 boundary plants to prevent interference between treatments. Each treatment was replicated four times in a randomized complete block design (4 plots/test substance). The following test substances were tested: As 35x, Bent, and Cu 3, 1 and 0.3 g/L, with respect to negative control. Three pre-inoculation (October 6th, 16th, 19th 2006) and 4 post-inoculation (November 10th, 17th, 28th and December 4th 2006) treatments were carried out by spraying the whole plant (30 mL/plant); contaminations of the ultra high dilutions and wind drift were avoided by using different spraying tools and a transparent plexiglass panel, respectively; inoculation with A. brassicicola at 107 conidia/mL was performed on October 26th 2006, by spraying 7 mL/plant. To evaluate the effectiveness of As 35x in the control of the natural infection induced by A. brassicicola, 8 plots (4 for C and 4 for As 35x) were not subjected to artificial inoculation, but otherwise treated on the same days as the other plots. Therefore the treatment classes were: i-H20 (control), i-As 35x, i-Bent, i-Cu 0.3, 1, 3, ni-H20, ni-As 35x, where i- and ni- denote inoculated and non-inoculated plants, respectively. Disease evaluation in each plot was performed 3 times on the leaves and 3 times on the head. In particular, as regards the leaves, assessments were conducted on inoculated leaves (November 13th and 28th 2006) and on new non-inoculated leaves (December 7th 2006) with the same infection scale used for the in planta experiments. As regards the cauliflower heads, disease assessments were carried out on December 14th, 20th and 28th 2006 by measuring the mean infection level on a scale from 0 (no infection) to 100 (total infection). Disease evaluation was always performed blind. After the last disease evaluation, the cauliflower heads were hand-harvested (December 28th 2006) and weighed individually; yields were expressed as kilograms of fresh weight/m2. To prevent damage by Epithrix hirtipennis and Pieris brassicae, we applied Rotena 43 (Serbios, Badia Polesine, RO, Italy) and Cifotrin 18 (CIFO, San Giorgio di Piano, BO, Italy) 3 times after planting. Daily rainfall data derived from real time measurements performed by automatic electronic equipment of the Hydrometeorological Service (ARPA, Emilia-Romagna) were obtained from the web site (www.arpa.emr.it).
Statistical analysis
In the in vitro germination tests, the experimental variable was the percentage of germinated conidia with respect to the total number (at least 230 conidia for each treatment group) for each frame. Treatment effects were evaluated by a comparison between two frequencies with normal approximation, suitable for binomial analysis of a large data set. Applying a normal approximation for large samples, we can use standard normal tables to check whether the value of z is significant or not. For each frequency we also indicate the corresponding standard error. Each treatment was compared with its individual control. For what concerns the in planta experiments, since the data were markedly skewed (very far from being normally distributed), we decided to apply the Kruskal-Wallis one-way analysis of variance, a nonparametric test based on ranks that involves ordering the observed data and replacing the values by the corresponding ranks, using the mean rank when there are repeated values. Such a test is used for comparing more than two samples that are independent, or unrelated, and is essentially based on a global ranking of sample data: if the ranks of data from one sample are considerably smaller or greater than those of the other sample, the null hypothesis is rejected. Whenever the Kruskal-Wallis test proved to be significant, we applied Dunn's post-hoc test to reduce the effect of multiple comparisons. Dunn's test compares the difference between the sum of ranks of two columns with the expected average difference, based on group number and size.
For what concerns the field trial, the data for the evaluation of infected leaves were subjected to a weighted mean, since we are here dealing with a frequency distribution, with the different observed disease intensities associated with their respective frequencies. On the other hand, for the evaluations of cauliflower head infection, which are count data expressed as proportions, we applied an arcsine transformation which results in a much better fit with the normal distribution. If f is expressed in decimal numbers, a useful transformation for normalizing the data distribution is W= arcsine (√ f ). Both weighted means for leaf infection and arcsine-transformed data for head infection were analysed by the repeated measures ANOVA test, followed by Dunnett's post hoc test (CoStat software, version 6.002, Cohort Software, Monterey, CA, USA). Moreover, to describe the difference from the control, the relative efficacy (of both in planta and field experiments) was calculated using Abbott's formula, and head weights were analysed by ANOVA.
RESULTS AND DISCUSSION
In vitro germination experiments
The evaluation of spore germination rates in standard conditions (negative control experiment) showed a mean germination rate of 30%, and the statistical evaluation of germination reproducibility did not evince any significant difference between controls (z= +0.029). This result shows that the experimental set-up was stable and did not generate false positive results. The inclusion of negative controls is considered good practice since it serves to demonstrate the non-existence of carry-over-effects (Stock-Schörer et al., 2009STOCK-SCHRÖER, B; ALBRECHT, H; BETTI, L; ENDLER, PC; LINDE, K; LÜDTKE, R; MUSIAL, F; VAN WIJK, R; WITT, C; BAUMGARTNER, S. 2009. Reporting experiments in homeopathic basic research (REHBaR) - A detailed guideline for authors. Homeopathy98: 287-298.).
The effects of UHDs on conidia vitality are reported in Figure 1, where controls are set equal to 100 to better display the differences between treatments. The results show that As 35x induced a highly significant inhibition (-60.0%) of spore germination rate compared to the control (p<0.001), whereas the 45th dilution/dynamization level of the same treatment did not have any significant effect. In our spore germination model, the inhibiting effect of As 35x and the null effect of As 45x suggest that the same treatment at different dilution/dynamization levels can have different effects, as has also been observed in other in vitro germination models (Majevsky et al., 2009MAJEWSKY, V; ARLT, S; SHAH, D; SCHERR, C; JÄGER, T; BETTI, L; TREBBI, G; BONAMIN, L; KLOCKE, P; BAUMGARTNER, S. 2009. Use of homeopathic preparations in experimental studies with healthy plants. Homeopathy98: 228-243.; Jäger et al., 2011aJÄGER, T; SCHERR, C; SHAH, D; MAJEWSKY, V; BETTI, L; TREBBI, G; BONAMIN, L; SIMÕES-WÜST, AP; WOLF, U; SIMON, M; HEUSSER, P; BAUMGARTNER, S. 2011a. Use of homeopathic preparations in experimental studies with abiotically stressed plants. Homeopathy 100: 275-287.), and particularly in the germination of wheat seeds, where a significant inhibition was obtained with As 35x whereas As 45x induced significant stimulation (Brizzi et al., 2000BRIZZI, M; NANI, D; PERUZZI, M; BETTI, L. 2000. Statistical analysis of the effect of high dilutions of arsenic in a large data set from a wheat germination model. British Homeopathic Journal 89: 63-67.).
Germination rate of Alternaria brassicicola spores suspended in different treatments at 35th or 45th decimal dynamization levels (35x and 45x, respectively) {taxas de germinação de esporos de Alternaria Brassicicola em suspensão em tratamentos diferentes aos níveis 35 e 45 de dinamização decimal (35x e 45x, respectivamente)}; C= control (testemunha); As= arsenic trioxide (trióxido de arsênio); Cupr= Cuprum metallicum; Nos= nosode; *** p<0.001; ns= not significant, Z test (não significativo, teste Z para uma frequência com aproximação normal). Italia, Bologna University, 2006.
For what concerns the Cupr 35x and 45x treatments, these yielded a highly significant reduction vs. control (p<0.001), with a reduction in germination rate of up to 70%. Other authors have also found that ultra high dilutions of Cuprum induce a significant inhibition of germination in A. solani spores (Rivas et al., 1996RIVAS, E; CECEÑA, C; GUAJARDO, G. 1996. Acción de 9 fármacos homeopáticos sobre la germinación de esporas de Alternaria solani y semillas de trigo y tomate. Boletin Mexicano de Homeopatia 29: 44-46.). As far as the nosode 35x and 45x is concerned, no significant effects were observed. These results are in line with those obtained by Carneiro et al. (2010CARNEIRO, SMTPG; ROMANO, EDB; PIGNONI, E; TEIXEIRA, MZ; VASCONCELOS, MEC; GOMES, JC. 2010. Effect of biotherapic of Alternaria solani on the early blight of tomato plant and the in vitro devolopment of the fungus. International Journal of High Dilution Research 9: 147-155.) with a nosode of A. solani. Finally, for what concerns the Cu 3 treatment, we can only hypothesize its inhibiting effects on germination, because it formed clusters around the conidia, preventing them from being clearly seen under the microscope. For this reason it was not included in Figure 1.
In planta experiments
The second aim of this work was to verify whether the UHDs previously tested on in vitro spore germination were able to control dark leaf spot disease in cauliflower seedlings inoculated with A. brassicicola. The stability of the test-system was verified by three negative control experiments (overall mean infection level = 1.28±0.03), the results of which found no significant difference whatsoever between controls (p= 0.969, p= 0.669, p= 0.366), thereby confirming that the experimental set-up was stable and did not generate false positive results.
The results of in planta experiments, reported in Figure 2, show that As 35x induced a significant decrease in the infection index (p<0.01) with a relative efficacy of 42.1% compared to the control. This finding might be ascribed to an increased plant resistance to fungal infection, but biochemical analyses are needed to confirm this hypothesis. In the literature, there is some evidence of the efficacy of high arsenic dilutions in the control of plant diseases in growth chamber (Betti et al., 2009BETTI, L; TREBBI, G; MAJEWSKY, V; SCHERR, C; SHAH-ROSSI, D; JÄGER, T, BAUMGARTNER, S. 2009. Use of homeopathic preparations in phytopathological models and in field trials: a critical review. Homeopathy 98: 244-266.); in particular, an increase in the resistance of tobacco plants to tobacco mosaic virus following treatments with As 45x has been reported (Betti et al., 2003BETTI, L; LAZZARATO, L; TREBBI, G; BRIZZI, M; CALZONI, GL; BORGHINI, F; NANI, D. 2003. Effects of homeopathic arsenic on tobacco plant resistance to TMV: theoretical suggestions about system variability, based on a large experimental data set. Homeopathy92: 195-202.). As far as Cuprum 35x and 45x are concerned, the results showed no significant differences vs. control. Likewise, nosode 35x and 45x did not induce any significant effect vs. control. The approach of using a diluted and dynamized extract of infected/stressed plant tissue (nosode) has also been adopted in other plant models, obtaining significant (Carneiro et al., 2010CARNEIRO, SMTPG; ROMANO, EDB; PIGNONI, E; TEIXEIRA, MZ; VASCONCELOS, MEC; GOMES, JC. 2010. Effect of biotherapic of Alternaria solani on the early blight of tomato plant and the in vitro devolopment of the fungus. International Journal of High Dilution Research 9: 147-155.; Jäger et al., 2010JÄGER, T; SCHERR, C; SIMON, M; HEUSSER, P; BAUMGARTNER, S. 2010. Effects of homeopathic Arsenicum album, nosode, and gibberellic acid preparations on the growth rate of arsenic-impaired duckweed (Lemna gibba L.). The Scientific World Journal 10: 2112-2129., 2011bJÄGER, T; SCHERR, C; SIMON, M; HEUSSER, P; BAUMGARTNER, S. 2011b. Development of a test system for homeopathic preparations using impaired duckweed (Lemna gibba L.). Journal of Alternative and Complementary Medicine 17: 315-323.), or not significant results (Shah-Rossi et al., 2009SHAH-ROSSI, D; HEUSSER, P; BAUMGARTNER, S. 2009. Homeopathic treatment of Arabidopsis thaliana plants infected with Pseudomonas syringaeThe Scientific World Journal 9: 320-330.). Finally, as we expected, the positive controls Cu 3 and BABA arrested the spread of disease (p<0.001, relative efficacy of 69.7% and 65.9%, respectively), confirming their antifungal activity and resistance inducing function, respectively. The results for BABA are in line with those obtained by Kamble & Bhargava (2007KAMBLE, A; BHARGAVA, S. 2007. ß-aminobutyric acid-induced resistance in Brassica juncea against the necrotrophic pathogen Alternaria brassicae. Journal of Phytopathology 155: 152-158.), who report a protective effect on Brassica plants against Alternaria brassicae following BABA treatment.
In planta experiments: infection index of cauliflower leaves inoculated with Alternaria brassicicola and treated with different test-substances. Error of means are represented by vertical bars (experimentos em planta: índice de infecção das folhas de couveflor inoculadas com A. brassicicola e tratadas com diferentes substâncias. Os erros das médias estão representados por barras verticais). C= control (testemunha); As= arsenic trioxide (trióxido de arsênio); Cupr= Cuprum metallicum; Nos= nosode; Cu 3= copper oxychloride 3 g/L (oxicloreto de cobre 3 g/L); BABA= aminobutyric acid, 5 mM (ácido aminobutírico, 5 mM); ***p<0.001; **p<0.01; ns= not significant (não significativo) (Kruskal-Wallis test, Dunn's post test) (teste de Kruskal-Wallis e pós-teste de Dunn).
Field trial
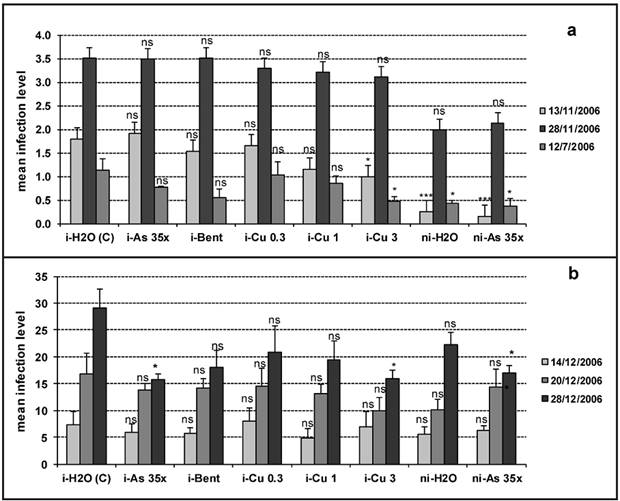
The UHDs that yielded significant effects in the in planta experiments were then tested in a field trial, along with Bent and Cu 0.3, 1 g/L, in comparison with Cu 3 g/L and H2O as positive and negative controls, respectively. Particular attention must be paid to the use of the preparation of arsenic. In fact, arsenic is a well known toxic chemical element used in the past (up to 50's and 60's) in the agricultural practices (Hughes et al., 2011HUGHES, MF; BECK, BD; CHEN, Y; LEWIS, AS; THOMAS, DJ. 2011. Arsenic exposure and toxicology: a historical perspective. Toxicological Sciences 123: 305-332.). In particular, the indiscriminate use of pesticides (herbicides and insecticides) based on arsenic has led to a high accumulation of this element in vast agricultural areas. In recent years, in more developed countries, most of these products containing arsenic have been banned from agriculture. However, in many developing countries these products are still widely used causing serious damage to the soil-water-plant, and consequently to the higher animals. Given the risks revealed by the use of arsenic in agriculture, in this experimentation our approach was to use ultra high dilutions of arsenic (beyond Avogadro number) in which no molecule of this element is left and, thus, totally non-toxic to the environment. The results of the infection level evaluated on leaves are reported in Figure 3a. In the first assessment (13/11/2006), only Cu 3 induced a significant decrease (p<0.05) in mean infection level in artificially inoculated plants, with a relative efficacy of 44.4% with respect to the inoculated control. Non-inoculated plants treated with H20 and As 35x showed a significantly lower infection level (p<0.001) with respect to the inoculated control, without any significant difference between the two treatments: the observed symptoms were probably induced by a natural infection caused by the spread of spores from inoculated plants carried by the wind. At the second evaluation on leaves (28/11/2006), all values were higher than on the first assessment, confirming that the disease has progressed, and none of the treatments showed significant effects vs. inoculated control. It is worth to noting that the decrease in infection induced by Cu 3 in the first evaluation was nullified in the second one. This unexpected result could be accounted for by the weather conditions during the week after the second post-inoculation treatment (average daily rainfall of 6.9 mm): the rain may have washed away the copper from the leaves, making the plant susceptible to fungal infection. On the third evaluation (7/12/2006), a significant reduction in symptoms (p<0.05) due to Cu 3 was obtained on new non-inoculated leaves: in fact, after the last two post-inoculation treatments (28/11/2006 and 4/12/2006) there had been no further rainfall. Disease assessments on cauliflower heads were performed on 3 successive occasions (Figure 3b): on the first (14/12/2006) and second (20/12/2006) evaluations, there were no test-substance that caused differences with respect to the inoculated control, whereas the last evaluation (28/12/2006) found a significant reduction. More specifically, in inoculated plants, As 35x and Cu 3 reduced disease symptoms (p<0.05) with a relative efficacy of 45.7% vs. inoculated control. For what concerns As 35x, significant control of disease was achieved. Since inoculation was performed on the leaves before flowering, we could hypothesize that As 35x induced an increase in the plants' natural resistance to fungal infection. This hypothesis is supported by the findings on non-inoculated plants: in fact, those treated with As 35x showed a significantly lower infection level than the inoculated controls (p<0.05) with a relative efficacy of 41.6%, whereas those treated with H2O reached an infection level similar to that of the inoculated control. This observed increase in natural resistance could be ascribed to an epigenetic effect of UHDs: regulation of the expression of certain specific and relevant genes has been demonstrated in both prokaryotes and in primitive forms of eukaryotes such as yeast (Das et al., 2011DAS, D; DE, A; DUTTA, S; BISWAS, R; BOUJEDAINI, N; KHUDA-BUKHSH, AR. 2011. Potentialyzed homeopathic drug Arsenicum Album 30C positively modulates protein biomarkers and gene expressions in Saccharomyces cerevisae exposed to arsenate. Journal of Chinese Integrative Medicine 9: 752-760.; De et al., 2012DE, A; DAS, D; DUTTA, S; CHAKRABORTY, D; BOUJEDAINI, N; KHUDA-BUKHSH, AR. 2012. Potentiated homeopathic drug Arsenicum album 30C inhibits intracellular reactive oxygen species generation and up-regulates expression of arsenic resistance gene in arsenite-exposed bacteria Escherichia coli. Journal of Chinese Integrative Medicine 10: 210-227.). The symptom reduction due to Cu 3 g/L confirms the well known inhibiting effect of Cu2+ ions on fungal spore germination (Borkow & Gabbay, 2005BORKOW, G; GABBAY, J. 2005. Copper as biocidal tool. Current Medicinal Chemistry 12: 2163-2175.). The other treatments (Cu 0.3, 1 g/L, and Bent), though they reduced the mean infection level, but not to statistically significant extent. Finally, the average yield of inoculated control was 0.92 kg/m2 and no significant differences were observed following the different treatments.
Field trial: mean infection level of leaves (a) and heads (b) in cauliflower plants artificially inoculated or not with A. brassicicola and treated with different test-substances {experimentos de campo: nível medio de infecção de folhas (a) e cabeças (b) de plantas de couve-flor inoculadas artificialmente (ou não) com A. brassicicola e tratadas com diferentes substâncias}; Error of means are represented by vertical bars (os erros das médias estão representados por barras verticais); i= inoculated (inoculada); ni= non-inoculated (não inoculada); C = control (testemunha); As= arsenic trioxide (trióxido de arsênio); Bent= bentonite (bentonita); Cu 0.3, 1, 3= copper oxychloride 0.3, 1, 3 g/L, respectively (oxicloreto de cobre 0.3, 1 ou 3 g/L, respectivamente); *** p<0.001; *p<0.05; ns= not significant (ANOVA) {não significativo (ANOVA)}.
In conclusion, this report demonstrates the ability of ultra high arsenic dilutions to inhibit in vitro spore germination of A. brassicicola and to control dark leaf spot both in planta and in field trials. Several bibliographic data have already been published about the UHD effects on in vitro and in planta trials (Betti et al., 2009BETTI, L; TREBBI, G; MAJEWSKY, V; SCHERR, C; SHAH-ROSSI, D; JÄGER, T, BAUMGARTNER, S. 2009. Use of homeopathic preparations in phytopathological models and in field trials: a critical review. Homeopathy 98: 244-266.; Majewsky et al., 2009MAJEWSKY, V; ARLT, S; SHAH, D; SCHERR, C; JÄGER, T; BETTI, L; TREBBI, G; BONAMIN, L; KLOCKE, P; BAUMGARTNER, S. 2009. Use of homeopathic preparations in experimental studies with healthy plants. Homeopathy98: 228-243.). On the contrary, to our knowledge few experiments in field have been carried out until now against fungal diseases, as reviewed in Betti et al. (2009)BETTI, L; TREBBI, G; MAJEWSKY, V; SCHERR, C; SHAH-ROSSI, D; JÄGER, T, BAUMGARTNER, S. 2009. Use of homeopathic preparations in phytopathological models and in field trials: a critical review. Homeopathy 98: 244-266.. In particular, the significant effects induced by ultra high diluted arsenic in the field represent a prove that UHDs are potential and reliable approaches for sustainable agriculture. Such approaches would be especially appropriate for organic farms that strive to be environmentally responsible, as well as economically viable. Nevertheless, our research is still experimental and is a long way from actually being available for use by farmers because i) controlled field experiments have to be repeated and on-farm pilot studies should follow; ii) the treatments we tested would be approved in certified organic farming systems and iii) farmers should learn how to use UHDs.
ACKNOWLEDGEMENTS
The authors wish to thank the Marche region, and in particular Dr. Leonardo Valenti, for the financial support and cooperation provided for this research. The authors are deeply grateful to Dr. Maria Grazia Fantino for scientific insights and fruitful discussions, and are also indebted to Laboratoires Boiron, in particular to Dr. Silvia Nencioni and Dr. Luigi Marrari, for their collaboration and technical assistance. The sponsors had no influence whatsoever upon the designing, conduction, evaluation and writing the manuscript of this investigation.
REFERENCES
- BELLAVITE, P; MARZOTTO, M; OLIOSO, D; MORATTI, E; CONFORTI, A. 2014. High-dilution effect revisited. 1. Physicochemical aspects. Homeopathy103: 4-21.
- BETTI, L; LAZZARATO, L; TREBBI, G; BRIZZI, M; CALZONI, GL; BORGHINI, F; NANI, D. 2003. Effects of homeopathic arsenic on tobacco plant resistance to TMV: theoretical suggestions about system variability, based on a large experimental data set. Homeopathy92: 195-202.
- BETTI, L; TREBBI, G; MAJEWSKY, V; SCHERR, C; SHAH-ROSSI, D; JÄGER, T, BAUMGARTNER, S. 2009. Use of homeopathic preparations in phytopathological models and in field trials: a critical review. Homeopathy 98: 244-266.
- BORKOW, G; GABBAY, J. 2005. Copper as biocidal tool. Current Medicinal Chemistry 12: 2163-2175.
- BRIZZI, M; NANI, D; PERUZZI, M; BETTI, L. 2000. Statistical analysis of the effect of high dilutions of arsenic in a large data set from a wheat germination model. British Homeopathic Journal 89: 63-67.
- CARNEIRO, SMTPG; ROMANO, EDB; PIGNONI, E; TEIXEIRA, MZ; VASCONCELOS, MEC; GOMES, JC. 2010. Effect of biotherapic of Alternaria solani on the early blight of tomato plant and the in vitro devolopment of the fungus. International Journal of High Dilution Research 9: 147-155.
- COHEN, YR. 2002. ß-aminobutyric acid-induced resistance against plant pathogens. Plant Disease 86: 448-457.
- DAS, D; DE, A; DUTTA, S; BISWAS, R; BOUJEDAINI, N; KHUDA-BUKHSH, AR. 2011. Potentialyzed homeopathic drug Arsenicum Album 30C positively modulates protein biomarkers and gene expressions in Saccharomyces cerevisae exposed to arsenate. Journal of Chinese Integrative Medicine 9: 752-760.
- DE, A; DAS, D; DUTTA, S; CHAKRABORTY, D; BOUJEDAINI, N; KHUDA-BUKHSH, AR. 2012. Potentiated homeopathic drug Arsenicum album 30C inhibits intracellular reactive oxygen species generation and up-regulates expression of arsenic resistance gene in arsenite-exposed bacteria Escherichia coli Journal of Chinese Integrative Medicine 10: 210-227.
- DOULLAH, MAU; MEAH, MB; OKAZAKI, K. 2006. Development of an effective screening method for partial resistance to Alternaria brassicicola (dark leaf spot) in Brassica rapaEuropean Journal of Plant Pathology 116: 33-43.
- ENDLER, PC; THIEVES, K; REICH, C; MATTHIESSEN, P; BONAMIN, L; SCHERR, C; BAUMGARTNER, S. 2010. Repetitions of fundamental research models for homeopathically prepared dilutions beyond 10-23: a bibliometric study. Homeopathy99: 25-36.
- FLIEßBACH, A; OBERHOLZER, HR; GUNST, L; MÄDER, P. 2007. Soil organic matter and biological soil quality indicators after 21 years of organic and conventional farming. Agriculture, Ecosystems & Environment 118: 273-284.
- HUGHES, MF; BECK, BD; CHEN, Y; LEWIS, AS; THOMAS, DJ. 2011. Arsenic exposure and toxicology: a historical perspective. Toxicological Sciences 123: 305-332.
- JÄGER, T; SCHERR, C; SHAH, D; MAJEWSKY, V; BETTI, L; TREBBI, G; BONAMIN, L; SIMÕES-WÜST, AP; WOLF, U; SIMON, M; HEUSSER, P; BAUMGARTNER, S. 2011a. Use of homeopathic preparations in experimental studies with abiotically stressed plants. Homeopathy 100: 275-287.
- JÄGER, T; SCHERR, C; SIMON, M; HEUSSER, P; BAUMGARTNER, S. 2011b. Development of a test system for homeopathic preparations using impaired duckweed (Lemna gibba L.). Journal of Alternative and Complementary Medicine 17: 315-323.
- JÄGER, T; SCHERR, C; SIMON, M; HEUSSER, P; BAUMGARTNER, S. 2010. Effects of homeopathic Arsenicum album, nosode, and gibberellic acid preparations on the growth rate of arsenic-impaired duckweed (Lemna gibba L.). The Scientific World Journal 10: 2112-2129.
- KAMBLE, A; BHARGAVA, S. 2007. ß-aminobutyric acid-induced resistance in Brassica juncea against the necrotrophic pathogen Alternaria brassicae Journal of Phytopathology 155: 152-158.
- MAJEWSKY, V; ARLT, S; SHAH, D; SCHERR, C; JÄGER, T; BETTI, L; TREBBI, G; BONAMIN, L; KLOCKE, P; BAUMGARTNER, S. 2009. Use of homeopathic preparations in experimental studies with healthy plants. Homeopathy98: 228-243.
- MODOLON, TA; BOFF, P; BOFF, MIC; MIQUELLUTI, DJ. 2012. Homeopathic and high dilution preparations for pest management to tomato crop under organic production system. Horticultura Brasileira 30: 51-57.
- MONDAL, S; SUKUL, NEE CHUNARI, S; CHANDRA-SUKUL, N. 2013. A new homeopathic potency promotes growth and yield of rice: a field trial. International Journal of High Dilution Research 12: 115.
- NAIDU, R; BOLAN, NS. 2008. Contaminant chemistry in soils: key concepts and bioavailability. In: NAIDU, R (eds). Chemical bioavailability in terrestrial environment. Amsterdam: Elsevier. p. 9-37.
- RIVAS, E; CECEÑA, C; GUAJARDO, G. 1996. Acción de 9 fármacos homeopáticos sobre la germinación de esporas de Alternaria solani y semillas de trigo y tomate. Boletin Mexicano de Homeopatia 29: 44-46.
- RUPP, LCD; BOFF, MIC; BOFF, P; DE, S; GONÇALVES, PA; BOTTON, M. 2012. High dilution of Staphysagria and fruit fly biotherapic preparations to manage South American fruit fly, Anastrepha fraterculus, in organic peach orchards. Biological Agriculture & Horticulture 28: 41-48.
- SHAH-ROSSI, D; HEUSSER, P; BAUMGARTNER, S. 2009. Homeopathic treatment of Arabidopsis thaliana plants infected with Pseudomonas syringaeThe Scientific World Journal 9: 320-330.
- STOCK-SCHRÖER, B; ALBRECHT, H; BETTI, L; ENDLER, PC; LINDE, K; LÜDTKE, R; MUSIAL, F; VAN WIJK, R; WITT, C; BAUMGARTNER, S. 2009. Reporting experiments in homeopathic basic research (REHBaR) - A detailed guideline for authors. Homeopathy98: 287-298.
- SUKUL, NC; CHATTOPADHYAY, S; DAS, C; SUKUL, A; GHOSH, S; SINHA BABU, SP. 2007. Enhancement of photosynthesis and plant growth by potentized drugs. Journal of Environment and Ecology 25S: 520-524.
- TURINEK, M; GROBELNIK-MLAKAR, S; BAVEC, M; BAVEC, F. 2009. Biodynamic agriculture research progress and priorities. Renewable Agriculture and Food Systems 24: 146-154.
- WITT, CM; BLUTH, M; ALBRECHT, H; WEISSHUHN, TER; BAUMGARTNER, S; WILLICH, SN. 2007. The in vitro evidence for an effect of high homeopathic potencies - a systematic review of the literature. Complementary Therapies in Medicine 15: 128-38.
- YINNON, TA; ELIA, V. 2013. Dynamics in perturbed very dilute aqueous solutions: theory and experimental evidence. International Journal of Modern Physics B27: 1350005 Available in: http://www.worldscientific.com/doi/abs/10.1142/S0217979213500057 Accessed in March 26, 2014.
» http://www.worldscientific.com/doi/abs/10.1142/S0217979213500057
Publication Dates
-
Publication in this collection
Jul-Sep 2016
History
-
Received
10 June 2015 -
Accepted
07 Mar 2016