Abstract
Dimorphandra wilsonii Rizzini is a rare species. Although cited as endemic to the Brazilian state of Minas Gerais, it has been recorded only for the municipalities of Paraopeba and Caetanópolis and therefore has not been extensively studied. This long-term, intensive survey, conducted from 2004 to 2012 in the central region of the state, was aimed at assessing its distribution, describing its habitat, and verifying its endemism, as well as assessing threats and determining its conservation status. Given the considerable size of the area to be studied and the difficulty of locating individuals of the species, we adopted popular participation as a complementary tool and we employed spatial distribution modeling. Communities were mobilized through the dissemination of print materials, and interviews were conducted. We visited 74 municipalities and addressed 900 people in search of this species. We found that D. wilsonii is endemic to the cerrado (savanna) and Atlantic Forest in the central region of Minas Gerais, occurring in 16 municipalities. Is not present in any fully protected conservation area, and its population (fewer than 250 individuals) is declining due to habitat destruction, caused mainly by agricultural/livestock and urban expansion, and its conservation status is "critically endangered".
Extinction; endemism; faveiro-de-wilson; inventory; popular participation
ARTICLES
Dimorphandra wilsonii Rizzini (Fabaceae): distribution, habitat and conservation status
Fernando Moreira FernandesI,II; Juliana Ordones RegoI
IJardim Botânico da Fundação Zoo-Botânica de Belo Horizonte, Belo Horizonte, Brasil
IIAuthor for correspondence: fernand@pbh.gov.br, fernandobrumas@yahoo.com.br
ABSTRACT
Dimorphandra wilsonii Rizzini is a rare species. Although cited as endemic to the Brazilian state of Minas Gerais, it has been recorded only for the municipalities of Paraopeba and Caetanópolis and therefore has not been extensively studied. This long-term, intensive survey, conducted from 2004 to 2012 in the central region of the state, was aimed at assessing its distribution, describing its habitat, and verifying its endemism, as well as assessing threats and determining its conservation status. Given the considerable size of the area to be studied and the difficulty of locating individuals of the species, we adopted popular participation as a complementary tool and we employed spatial distribution modeling. Communities were mobilized through the dissemination of print materials, and interviews were conducted. We visited 74 municipalities and addressed 900 people in search of this species. We found that D. wilsonii is endemic to the cerrado (savanna) and Atlantic Forest in the central region of Minas Gerais, occurring in 16 municipalities. Is not present in any fully protected conservation area, and its population (fewer than 250 individuals) is declining due to habitat destruction, caused mainly by agricultural/livestock and urban expansion, and its conservation status is "critically endangered".
Key words: Extinction, endemism, faveiro-de-wilson, inventory, popular participation
Introduction
Dimorphandra wilsonii Rizzini, a species that occurs in the state of Minas Gerais, Brazil, was discovered in the 1960s and described in 1969 (Rizzini 1969). Known locally as faveiro-de-wilson or faveira-da-mata, D. wilsonii is cited as endemic to the municipalities of Paraopeba and Caetanópolis, located in central region of the state (Rizzini & Mattos Filho 1986). Although it has been considered rare and endangered since 1986, it was not until 1997 that it was added to the International Union for Conservation of Nature (IUCN) Red List of Threatened Species, where it was listed in the "endangered" category (Walter & Gillett 1998); it was later withdrawn because of the lack of information about the species. In 2000, it was added to the Red List of Endangered Species in the Flora of Minas Gerais, although it was listed there as "vulnerable" rather than "endangered" (Mendonça & Lins 2000). In 2003, we made an expedition to the reported sites of occurrence. We found only 13 adults, all of them in pastures of Urochloa spp. on three farms within the municipalities cited, a situation similar to that reported by Rizzini & Mattos Filho (1986). At that time, we conducted a review of the literature and found only the records of occurrence mentioned above (for Paraopeba and Caetanópolis). Therefore, later that year, the Botanical Garden of the Belo Horizonte Zoological and Botanical Foundation (hereafter referred only as the Botanical Garden), in cooperation with other institutions, initiated a survey of the remaining population (Fernandes et al. 2007), in order to define the distribution of D. wilsonii, characterize its environment, verify its endemism, identify threats to the species, and determine its conservation status.
Material and methods
Study area
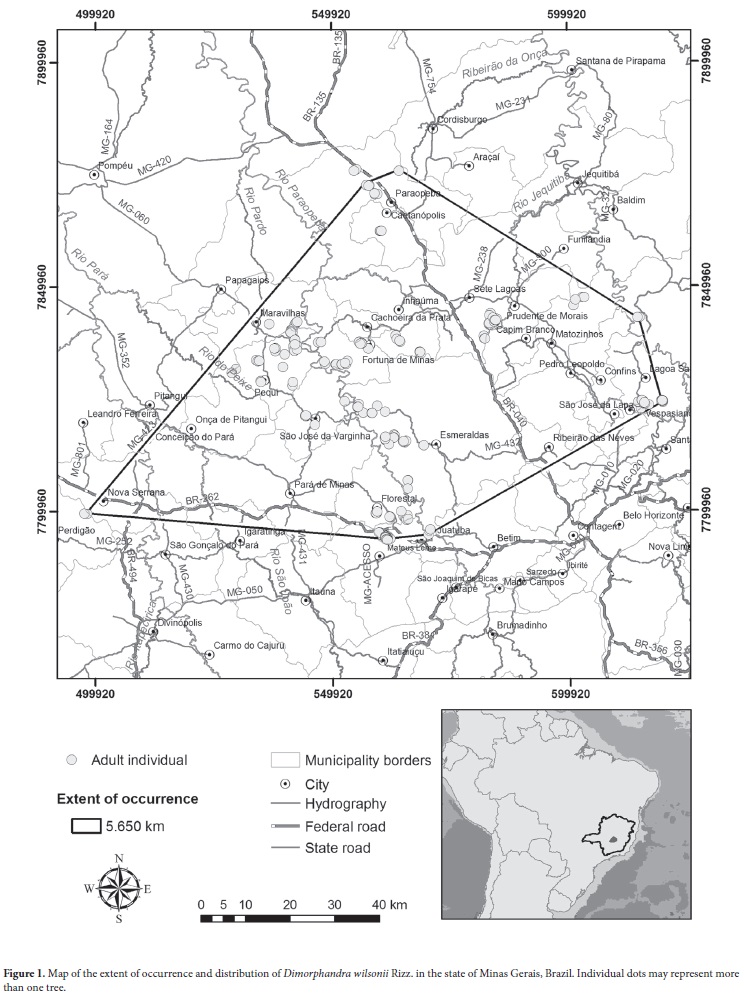
This study was conducted from January 2004 to October 2012. The study area comprises the central region of the state of Minas Gerais (Fig. 1). According to the Köppen-Geiger climate classification, the climate of the region is type Cwa (mesothermal), with dry winters and an average temperature in the warmest month > 22°C, and the annual rainfall varies between 1300 mm and 1500 mm, with 4-5 dry months (Silva et al. 2008).
Strategies for surveying the species
In an attempt to locate additional records of Dimorphandra wilsonii, we initially visited 13 herbaria within Brazil: PAMG; OUPR; VIC; HUFU; CESJ; ESAL; HXBH; GFJP; PMG; RB; R; SPF; and INPA. The INPA Herbarium was included because it contains the largest collection of Dimorphandra in the country, as would be expected, given that it is located within the Amazon Biome, where most species of the genus occur (Silva 1986). All acronyms are in accordance with the Index Herbariorum (Thiers [continuously updated]).
In addition to the inherent difficulties in assessing the abundance of a species, even one that is well-known (Kunin & Gaston 1997), conducting a survey of the Dimorphandra wilsonii population was complicated by the fact that it is such a rare, little known species and closely resembles several other Fabaceae species. In view of this situation and with the objective of obtaining the best possible results, we adopted an integrated strategy, adding community participation to our active search (the standard method). To mobilize the communities, we produced posters and leaflets-first in 2004 and subsequently in 2010-which were distributed in the interior regions of the state, in areas of cerrado (savanna) and areas of transition from cerrado to Atlantic Forest. We also produced an identification kit (a board with photographs of D. wilsonii and samples of its fruits, seeds, and leaves), which was shown to farmers, woodsman, herbalists, nurserymen, gardeners, researchers, government officials, and other residents of the region. We also interviewed people who had become aware of the search, either because they had seen the posters or leaflets or because they had seen a spontaneous media report, and had contacted us to report probable new occurrences. Their statements were verified by sending a sample (physically) or a photograph (by e-mail) to the Botanical Garden. In the active search, we observed altered landscapes (pasture, cropland, etc.) and fragments, in varying degrees of preservation, where 30 transects were surveyed, ranging from 100 to 600 m long and 20 to 40 m wide, established in several municipalities. Along the roads, we established observation points (with a field of view of 180° or 360°). At those points, we observed the landscape, impacted or not, with the naked eye and with 8×40 binoculars. With practice, it became possible to recognize the species at a distance of up to 600 m. At any sign of Dimorphandra, we would go to the site to inspect the individual. If confirmed as D. wilsonii, the tree was registered and the following data were collected: diameter at breast height (DBH), total height, tree health, and. We also photographed the individual and registered the point of occurrence, based on the geographic coordinates. Using the information collected, we assembled a database, in order to monitor the species and to support further studies. We also registered the observation points and established points of non-occurrence, defined as sites at which D. wilsonii would probably never occur, because the biotic and abiotic conditions are totally different from those of its natural habitat. From all of the individuals sampled, we collected leaf samples, which were sent to the Universidade Federal de Minas Gerais (UFMG, University of Minas Gerais) for genetic analysis. We also collected soil samples from some sites, as well as D. wilsonii fruits and seeds for study and for the production of seedlings. Some of the processed seeds were ceded to other studies conducted in cooperation with this one (Fonseca et al. 2010; Lopes & Matheus 2008). From several sites, we collected specimens of D. wilsonii and of related species, depositing the exsiccata in BHZB. The initial identification of Dimorphandra species was made from the material that was collected in the field, preserved and submitted to the Fabaceae specialist. Later, with the experience acquired, we were usually able to make the identification in the field, even in the absence of reproductive material. After the discovery of a each new occurrence of D. wilsonii, the owner of the land on which the individual occurred was informed of the discovery and instructed in the management of the species, as well as being encouraged to search for additional individuals.
Spatial distribution modeling
We began to use spatial distribution modeling (SDM) in August 2010, when 62% of the current population had already been found. We used 130 points of occurrence, corresponding to the individuals already known at that time, and 15 points of non-occurrence. We used seven bioclimatic variables extracted from the Worldclim database at a resolution of 1 km2 (Hijmans et al. 2005). The algorithms we used were maximum entropy (Phillips et al. 2006), environmental distance, and the genetic algorithm for rule-set prediction (Stockwell & Peters 1999), the last two available on the openModeller platform (Muñoz et al. 2009); to interpret the predictive maps, we applied a threshold rule to the training data and scheduled new field searches for validation.
Throughout the study period, because of new occurrences Dimorphandra wilsonii and at the suggestion of collaborators, as well as because of the introduction of SDM, the search area was extended to other mesoregions of the state, including the: northwestern, western, Triângulo Mineiro/Alto Paranaíba, southern, northern, and Jequitinhonha regions. In all of those regions, we made incursions and applied the local population approach. Consequently, during the study period, we visited 74 municipalities, established 180 observation points, and interviewed approximately 900 local residents. The extent of occurrence was calculated by the minimum convex polygon method (IUCN 2001). To calculate the area of occupancy, we applied grid plots of 1 ha and 10 ha. To determine the conservation status of D. wilsonii, we subjected the species distribution data, as well as the data regarding the biology, ecology, and current situation of the species, to the IUCN criteria (IUCN 2001).
Results and discussion
Collections and geographic distribution
Of the 13 herbaria consulted, only five (INPA, PAMG, PMG, RB, and VIC) had botanical material of Dimorphandra wilsonii in their collections, but all coming only from Paraopeba. However, at PAMG, there was one previously unidentified exsiccatum (M.B.Ferreira 5463 - PAMG 2541), collected in 1972 from an area near that municipality of Felixlândia (also within Minas Gerais), that we determined to be a new occurrence of D. wilsonii. We visited the municipality and noted that this specimen was dead and that the species no longer existed at the site. Considering the lack of material of this species at the herbaria consulted, as well as the need to make it more accessible to researchers in Brazil, duplicates of D. wilsonii collected during the inventory were sent to 11 other Brazilian herbaria in 2012 and 2013.
Our inventory revealed that Dimorphandra wilsonii, once recorded at only two municipalities in Minas Gerais, occurs in 16 municipalities (Tab. 1), all within the central region of the state. According to the geographical mapping of the area by the Brazilian Institute of Geography and Statistics (IBGE 2012), 14 of those municipalities are within the mesoregion of the greater metropolitan area of Belo Horizonte (in the microregions of Belo Horizonte, Pará de Minas and Sete Lagoas) and two are within the western mesoregion of the state (in the microregion of Divinópolis). All 16 municipalities are in river basins, either of the Paraopeba River, the Pará River or the Rio das Velhas River, all of which are tributaries of the São Francisco River (Fig. 1) and most of which are within the São Francisco-Velhas ecoregion (Arruda et al. 2008). The Espinhaço Mountain Range seems to be a natural geographic barrier, defining the eastern edge of the extent of D. wilsonii occurrence.
When only mature plants are considered (IUCN 2001), the total current population of Dimorphandra wilsonii comprises 219 individuals. We also recorded the deaths of 18 individuals (identified because part of the trunk still present or on the basis of the testimony of local residents). Although no individuals were found within any fully protected conservation area, three were identified within the Carste de Lagoa Santa Federal Environmentally Protected Area, albeit in pastureland. In addition to the mature individuals, we found 68 young non-fertile individuals, with heights of up to 6 m, within the area of occurrence.
Dimorphandra wilsonii has not been planted by local residents, because most are unaware of its existence or of its means of propagation. Although some farmers and ranchers are interested in cultivating D. wilsonii, we have not encouraged them to do so, because, if done randomly and without discretion, it would not necessarily contribute to the conservation of the species and, worse, could lead to increased inbreeding. Nevertheless, the Botanical Garden is reintroducing D. wilsonii on some farms in its natural area of occurrence, typically within legal reserves.
Although small, the population of Dimorphandra wilsonii is highly fragmented, and the total area occupied by the species is less than 9 km2. We obtained this area of occupancy by summing the occupied plot grids, adopting 10-ha grid cells, which most resembles the application of the smallest convex polygon method, tested in two subpopulations. The extent of occurrence is 5650 km2 (Fig. 1). Considering only the adult population of D. wilsonii, 40% of the individuals are located in municipalities within the Atlantic Forest Biome, as defined by Federal Law no. 11428/2006 and Decree no. 6660/2008 (Fundação SOS Mata Atlântica & Instituto Nacional de Pesquisas Espaciais 2011), more specifically in montane semideciduous forest (IBGE 2012). The remaining 60% are in the cerrado, usually in its transition to semideciduous forest (Fig. 2). However, it should be noted that even in the municipalities within the cerrado there are areas of semideciduous forest and cerradão (woodland savanna, the primary typology of the cerrado), and that D. wilsonii is usually found in or near such areas.
In the SDM of Dimorphandra wilsonii, the subsequent searches performed for validation (outside the central region of the state) revealed no new occurrences. The maximum entropy model performed best; the projections made by the other models were overly optimistic (Fig. 3). Although not allowing us to find new populations of D. wilsonii, the SDM served to direct the last searches toward regions where the species might be found in the future, thereby increasing the efficiency of fieldwork, as well as confirming the unlikelihood of its occurrence in other areas.
Habitat
Dimorphandra wilsonii occurs at elevations of 677-976 m, at different degrees of slope and in different soil types (predominantly red-yellow Spodosols and dark red Spodosols, usually well-drained and deep, with variable structure and fertility), suggesting that it is not very selective and is undemanding in terms of environment. Studies on the physiology of D. wilsonii (Fonseca et al. 2010) partially confirm this hypothesis, showing that the species has nutritional requirements compatible with the soil of its habitat and high adaptive capacity, mainly because of the association with nitrogen-fixing bacteria. Although it grows better in fertile soil, this adaptability allows it to develop well in areas of low fertility.
Mapping data for 2005, taken from Scolforo & Carvalho (2006), show that the municipalities where Dimorphandra wilsonii occurs then had collective totals of 260,300 ha of native forest remnants, cerrado accounting for 204,700 ha and Atlantic Forest accounting for 55,600 ha, respectively corresponding to 22.3% and 8.7% of the average original size of the vegetation formations in those municipalities. In addition, all of those remnants show some degree of disturbance, and habitat continues to be lost in the central region of Minas Gerais, at an annual rate of 0.2% (the equivalent of 1000 ha). We found that the habitat of D. wilsonii remains under severe anthropogenic pressure, mainly due to agricultural/livestock and urban expansion, as well as mining and fires. Another factor that negatively affects D. wilsonii is the cultivation of grasses of the genus Urochloa, especially U. decumbens (Stapf) R.D Webster, the most widely used grass in the region. Fonseca et al. (2013) found that U. decumbens competes with D. wilsonii for nutrients, inhibiting Bradyrhizobium and limiting the development of the juvenile plants. This might explain the rarity of D. wilsonii seedlings and juveniles settling near mature individuals in places where there is Urochloa grass, especially in dense pasture. The remaining trees of D. wilsonii individuals are situated in very different environments, many of which are also disturbed (Tab. 2).
In nine of the sixteen municipalities in which Dimorphandra wilsonii occurs, we also found individuals of Dimorphandra exaltata Schott, with new records for the species, and Dimorphandra mollis Benth. also occurs in 10 of those municipalities, not necessarily at the same sites. The latter is a species of wide distribution, common in most areas of cerrado, whereas D. exaltata is a little studied species of the Atlantic Forest Biome. The central region of Minas Gerais is just within the eastern border of the area of occurrence of D. mollis and the western/northern border of that of D. exaltata; the two areas extending out in opposite directions from exactly where D. wilsonii is found. In other words, there is a confluence of the three species in this region. Therefore, we suggest studies on the phylogeny and biogeography of these species, which could help explain the fact that D. wilsonii is restricted to such a small area in comparison with the other two species.
Dimorphandra wilsonii reaches maturity at around 15 years of age, when it presents a DBH of approximately 15 cm and a height of approximately 6 m. After its stem has been mown or cut, the plant sprouts again. Some individuals have multiple stems with up to 10 stalks due to cuts close to the base of the plant. The largest individual identified presented a DBH of 112 cm and a height of 17 m, its age having been estimated at over 100 years (E. Fernandes, Chico Mendes Institute for Biodiversity Conservation, personal communication). D. wilsonii has greater similarity to forest tree species than to savanna tree species; its rhytidome is predominantly reticulated (and thinner than that of savanna tree species), and its trunk is usually straight. When growing in the forest, it presents a tall leafless portion of trunk (up to 9 m) and the crown stands out in the forest canopy; when it grows in open areas, the leafless portion of its trunk is shorter, its rhytidome is rougher, and it presents a sparse, lower crown, although its trunk is always straight. On the basis of the division of ecological groups (Budowsky 1965; Ferreti 2002) and considering characteristics such as height, distribution, growth, longevity, wood, leafless stem, seed size, and seed viability, D. wilsonii can be framed as a late secondary successional species. The juveniles are always located close to the adults (usually within 500 m), which might indicate that the species has difficulty in dispersing over greater distances.
Many studies have shown that species classified as rare in terms of extent of occurrence usually have low dispersion ability (Kunin & Gaston 1997). Dimorphandra wilsonii seems to be one of these cases, because it presents a small proportion of young individuals, despite its regular, high production of fruit and seeds. There have been no studies of dispersion in D. wilsonii. However, Bizerril et al. (2005) noted that the tapir (Tapirus terrestris Lin.) is a seed spreader of D. mollis and suggested that in the past the extinct megafauna had this role in South America. The seeds of D. wilsonii and show great morphological similarity to those of D. mollis (Lopes & Matheus 2008); their fruits are very similar in terms of shape, color, and odor; and they have areas of occurrence in common. Therefore, they might also share seed dispersers. Because of the diversity of species that it consumes, the quantity it eats, and the distances that it travels, T. terrestris is considered an important seed disperser, although it is endangered (Drummond et al. 2008) and is no longer found in the region under study (IEF 2011). Ripe fruits of D. wilsonii fall in winter and spring, when pastures are dry, and are eagerly eaten by cattle, which seek them out. During the study period, we observed intact D. wilsonii seeds in cattle feces near a tree of the species, suggesting that cattle can be seed dispersers of D. wilsonii in disturbed areas.
Historical information, causes of extinction, and conservation status
According to reports (Fernandes, pers. comm.), Dimorphandra wilsonii was already scarce in Paraopeba and Caetanópolis in the 1960s; the established pastures were composed of Hyparrhenia rufa (Nees) Stapf and Melinis minutiflora P.Beauv, ("provisory" grass and molasses grass, respectively-both exotic species), as well as "capim-chato" (an unidentified native grass species). According to that same source, there was no ordinary use of D. wilsonii for specific purposes, although its wood, together with that of other local species, was used in the making of charcoal. In the 1940s and 1950s, there was extensive deforestation in Paraopeba and Caetanópolis, the wood being used in charcoal production for metallurgic/steel industries and as firewood for boilers. These areas were later converted to pastureland, without being allowed to regenerate. In the 1980s, the remaining D. wilsonii trees in these municipalities were saved from being cut down by the botanists Carlos Rizzini and Ezechias Heringer, who informed the farmers of the rarity of the species and the need to protect it. In the Brazilian Pharmacopoeia (Brazil 2010), there is no recorded medicinal use for D. wilsonii.
The information obtained in the first three years of the inventory conducted here, which already indicated the critical situation of Dimorphandra wilsonii, were submitted to the agencies and entities responsible for the red lists of endangered flora, along with a request for classification, inclusion and protection. As a result, in 2004, the government of Minas Gerais issued a decree declaring D. wilsonii exempt from cutting and exploitation in the state of Minas Gerais and empowering the State Forestry Institute to approve, encourage, and supervise scientific research on the species (GMG 2004). In 2006, the species was included in the IUCN Red List of Threatened Species, in the "critically endangered" category, where it appears until today (Fernandes 2006; IUCN 2013). In 2007, it was included in the Red List of Endangered Species in the Flora of Minas Gerais (Drummond et al. 2008). In 2008, was included in the Official List of Endangered Species in the Flora of Brazil (MMA 2008).
After the completion of the fieldwork in 2012, we applied the IUCN criteria, version 3.1 (IUCN 2001) to the information gathered during the inventory of the species. We found that Dimorphandra wilsonii continued to merit inclusion in the "critically endangered" category, because it presents a severely fragmented habitat, an area of occupancy < 10 km2, a reduction in the area, extent and/or quality of its habitat, a total population of fewer than 250 mature individuals, and no single subpopulation of more than 50 mature individuals, respectively meeting the criteria B2a, B2b (ii, iii, and v), and C2a (i). We also confirmed that the species is endemic to Minas Gerais.
The search for Dimorphandra wilsonii, even with extensive efforts made over the years, certainly was not exhaustive, and there is still a possibility that new individuals or populations will be found. since 2004, many collaborators have been sensitized and mobilized, and those collaborators sporadically send information. In 2013, we confirmed four new D. wilsonii trees on the basis of such information. However, the chance of finding a significant additional number of trees is admittedly small and is less likely outside the extent of occurrence outlined to date. The strategy of mobilizing communities with the methods first applied in 2004 (the use of "wanted" posters, leaflets, and interviews) was not only right but was essential to the success of the survey of the species. This campaign caught the attention of the media, which produced unsolicited television reports, such as those broadcast by TV Globo, the largest Brazilian television network, on the shows Jornal Nacional (the most widely watched prime-time news program in Brazil) and Globo Ecologia, as well as by Rede Minas (a statewide television network in Minas Gerais) on its show Planeta Minas. These reports helped mobilize the populace and surely increased the number of trees found. After its initial use in this work in 2004, this strategy was also used quite successfully in a similar study of another endangered species, the Brazilian Merganser (Mergus octosetaceus), in the Canastra Mountains (Lins et al. 2011).
The fact that few people knew or had heard of Dimorphandra wilsonii and that there were no records of any specific use of the plant, together with the fact that it was not discovered until 1968 (very recently compared with other arboreal species), suggest that the species has never been very abundant even when its habitat was more preserved. However, the extent of occurrence of Dimorphandra wilsonii (Fig. 1) is very large compared with its area of occupancy and population size. It is likely that the species once had a much larger population than it has today and that its subpopulations were connected, unlike the current situation, in which the population and its habitat are highly fragmented and severely affected by human activity. This hypothesis is somewhat supported by studies on the population genetics of D. wilsonii conducted by Souza & Lovato (2010), who showed that genetic variation was uniformly distributed between and within subpopulations, and that smaller variation among populations was smaller within D. wilsonii than within D. mollis, suggesting that the subpopulations of D. wilsonii were connected in the past, probable with gene flow, although its population size and density have never been very great. For the reasons set out above and due to the variety of environments in which the species is found (its adaptability), we can infer that the reduction to its current population was a consequence of habitat loss and that D. wilsonii is therefore another victim of the most common cause of species extinction in modern times. It is assumed that the reduction of the population has been caused directly and immediately by human occupation of its habitat, in this case, two global "hotspots", with deforestation, and indirectly and gradually by reducing or even locally extinguishing the populations of the seed disperser animals which would have prevented the species from recolonizing certain areas.
The fact that Dimorphandra wilsonii, categorized as a forest species, is now more often found in open areas that in forest fragments, raises certain questions. In the absence of a probable natural seed dispersers, mainly Tapirus terrestris, are the seeds of D. wilsonii being dispersed, even if only partially and poorly, by cattle, which not only reside in pastures and open areas but also occasionally venture into unfenced area that are somewhat more forested, where they feed on the fruits of D. wilsonii? In addition to having its population reduced by habitat destruction and the absence of major natural seed dispersers, might D. wilsonii be slowly and partially migrating from the forest to open areas? Answering those questions might help explain the discontinuous distribution of D. wilsonii and the existence of some rather isolated individuals and groups. As for its hypothetical migration to open areas, it is paradoxical that its chances of thriving in such areas are limited by the serious threats posed by factors such as competition from Urochloa, trampling/foraging by cattle, the periodic mowing of grassland, and the occasional grass fires.
The area of occurrence of Dimorphandra wilsonii is not unique in terms of geographic isolation or environmental conditions; nor does it harbor any other threatened species or species of restricted distribution that would justify the protection of natural areas as a strategy for conservation of the ecosystem or of a group of species. In addition, D. wilsonii is currently found only on private property. All of this makes it even more difficult and challenging to work toward the conservation of this species. We must therefore find sufficient justification to invest in studies that are more in-depth, in order to support interventions to preserve the species and its habitat.
The searches for Dimorphandra wilsonii and the push to gain a better understanding of its biology prompted the Botanical Garden to organize, in 2004, the Dimorphandra wilsonii Conservation and Management Project, which encouraged and promoted several studies developed jointly and cooperatively by a number of partners, moved by the desire to improve understanding of the species and contribute to its conservation. This shows the importance of multidisciplinary action and collective effort among institutions in the aid of an endangered species. The following types of studies were conducted: population genetics, by the UFMG (Souza & Lovato 2010); reproductive biology and phenology (Fernandes et al. 2007), by the Botanical Garden; physiology, by the Botanical Garden and the UFMG (Fonseca et al. 2010; 2013); seed conservation, by the Botanical Garden, UFMG and Federal University of Espírito Santo); and leaf mycobiota, by the Federal University of Viçosa (Silva 2012). However, it is necessary to expand the knowledge of the species and thus promote improved interventions for its management and conservation; conducting studies on plant-animal relationships, reproductive biology, reproductive structures, and dispersion, as well as studies of the pollination ecology of co-occurring congeneric species (D. exaltata and D. mollis) and the phylogeography of the Dimorphandra genus, to further elucidate obscure points about their biology. We also suggest a revision of the Dimorphandra genus, at least for the species not occurring in the Amazon, given that the last such revision was made almost three decades ago (Silva 1986) and the information about some species, including D. wilsonii, is therefore quite outdated. In that revision, D. wilsonii was analyzed only from the limited herbaria material available at the time (from Paraopeba only). Another major challenge is to increase the population of the species in nature through its reintroduction, which has already been initiated by the Dimorphandra wilsonii Conservation and Management Project, in a joint effort between the Botanical Garden and the UFMG, with some support from the private sector.
Acknowledgments
This study was sponsored by the Boticário Group Foundation for the Protection of Nature and the Liz Cement Company, as well as receiving logistical support from the Sociedade de Amigos da Fundação Zoo-Botânica de Belo Horizonte (Society of the Friends of the Belo Horizonte Zoological and Botanical Foundation), the Instituto Estadual de Florestas de Minas Gerais (IEF/MG, Minas Gerais State Forestry Institute), the Minas Gerais State Environmental Police, and the Brazilian Instituto Chico Mendes de Conservação da Biodiversidade (ICMBio, Chico Mendes Institute for Biodiversity Conservation)/Paraopeba National Forest. We offer our sincere thanks to Dr. Tania Sampaio Pereira, for the constant support and for reviewing the manuscript; to Professor Miele Tallon Matheus (in memoriam), for the encouragement and revisions; to Cássio Soares Martins, for providing the maps; to Dr. Luciana H. Y. Kamino, for collaborating on the SDM; to Enéas Fernandes (in memoriam), former woodsman of the ICMBio/Paraopeba National Forest, for the important historical reports; and to the interns Victor T. Giorni, Andreia Gomes, Pedro L. S. Miranda, and Ana Paula Otoni. We are also especially grateful to the farmers and residents of the local communities for the substantial assistance in the searches and in the protection of the species.
References
Arruda, M.B.; Proença, C.E.B; Rodrigues, S.C.; Campos, R.N.; Martins, R.C. & Martins, E.S. 2008. Ecorregiões, Unidades de Conservação e Representatividade Ecológica do Bioma Cerrado. Pp. 229-272. In Sano, S.M.; Almeida, S.P. & Ribeiro, J.F. (Eds). Cerrado: ecologia e flora, v.1. Brasília, Embrapa Cerrados.
Bizerril, M.X.A.; Rodrigues, F.H.G. & Hass, A. 2005. Fruit consumption and seed dispersal of Dimorphandra mollis Benth. (Leguminosae) by the lowland tapir in the Cerrado of Central Brazil. Brazilian Journal of Biology 65(3): 407-413.
Brasil, 2010. Agência Nacional de Vigilância Sanitária. Farmacopéia Brasileira. V. 2. Brasília, Anvisa.
Budowski, G. N. 1965. Distribution of tropical American rain forest species in the light of succession processes. Turrialba 15(1): 40-42.
Drummond, G.M.; Machado, A.B.M.; Martins, C.S.; Mendonça, M.P. & Stehmann, J.R. (Eds.) 2008. Listas vermelhas das espécies da fauna e da flora ameaçadas de extinção em Minas Gerais. 2ª ed. Belo Horizonte, Fundação Biodiversitas. Available from: http://www.biodiversitas.org.br/listas-mg/lista_floramg.asp. Cited 2013 Sep 11. .
Fernandes, F. M. 2006. Dimorphandra wilsonii. In: IUCN 2012. IUCN Red List of Threatened Species. Version 2012.1. Available from: http://www.iucnredlist.org/details/61926/0. Cited 2013 Aug 26.
Fernandes, F. M.; Fonseca, A. G.; Kaechele, K; Goulart, M. F.; Marinho, W.; souza, H A.V; Queiroz, A. R.; Giorni, V.T; Oliveira, G.; Rodrigues, M. J.; Bacelar, M. & Lovato, M. B. 2007. Tentando evitar mais uma extinção: o caso do "Faveiro de Wilson" (Dimorphandra wilsonii Rizzini). Pp. 87-98. In: Pereira, T. S.; Costa, M. L. M. N. & Jackson, P. W. (Orgs.). Recuperando o verde para as cidades: a experiência dos jardins botânicos brasileiros. Rio de Janeiro, Rede Brasileira de Jardins Botânicos, Instituto de Pesquisas Jardim Botânico do Rio de Janeiro, BGCI.
Ferreti, A.R. 2002. Fundamentos Ecológicos para o Planejamento da Restauração Florestal. Pp. 21-26. In: Galvão, A.P.M.; Medeiros, A.C.S.. Restauração da Mata Atlântica em Áreas de sua Primitiva Ocorrência Natural. Colombo, Embrapa.
Fonseca, M.B.; Franca, M.G.C.; Zonta, E. & Giorni, V.T. 2010. Crescimento inicial de Dimorphandra wilsonii (Fabaceae - Caesalpinioideae) em diferentes condições de fertilidade em solo de cerrado. Acta Botanica Brasilica 24(2): 322-327.
Fonseca, M.B.; Carolino, M.M.S.S. de L.; Dias, T.; Cruz, C. & França, M.G.C. 2013. Early growth of Brazilian tree Dimorphandra wilsonii is also threatened by African grass Urochloa decumbens, Journal of Plant Interactions 9(1):92-99.
Fundação SOS Mata Atlântica & Instituto Nacional de Pesquisas Espaciais. 2011. Atlas dos Remanescentes Florestais da Mata Atlântica Período 2008-2010. São Paulo, Fundação SOS Mata Atlântica e INPE.
GMG - Governo de Minas Gerais. 2004. Decreto 43904-2004. Diário Oficial do Estado de Minas Gerais. 27/10/2004. p. 5,col.1. Belo Horizonte, GMG.
Hijmans, R.J.; Cameron, S.E.; Parra, J.L.; Jones, P.G. & Jarvis, A. 2005. Very High Resolution Interpolated Climate Surfaces for Global Land Areas. International Journal of Climatology 25: 1965-1978.
IBGE - Instituto Brasileiro de Geografia e Estatística. 2012. Manual técnico da vegetação brasileira. Série Manuais Técnicos em Geociências. 2ª ed. Rio de Janeiro, IBGE.
IEF - Instituto Estadual de Florestas. 2011. Atlas da Fauna em Unidades de Conservação do Estado de Minas Gerais -Vol. 1 - Mastofauna. http://intranet.meioambiente.mg.gov.br/documentos/atlas-parte1.pdf Belo Horizonte, IEF.
IUCN 2001. IUCN Red List Categories and Criteria: Version 3.1. IUCN Species Survival Commission. Gland and Cambridge, IUCN.
IUCN 2013. IUCN Red List of Threatened Species. Version 2012.2. Available from: http://www.iucnredlist.org/search. Cited 2013 Oct 26.
Kunin, W.E. & Gaston, K.J. (Eds.) 1997. The biology of rarity. Causes and consequences of rare-common differences. London, Chapman & Hall.
Lins, L.V.; Ribeiro, F.; Andrade, R.D. & Rigueira, S.E. 2011. Educação ambiental para a conservação da região da Serra da Canastra, utilizando o pato- mergulhão (Mergus octosetaceus) como espécie bandeira. Belo Horizonte, Instituto Estadual de Florestas. Revista MG Biota 4(3): 34-55.
Lopes, J.C. & Matheus, M.T. 2008. Caracterização morfológica de sementes, plântulas e da germinação de Dimorphandra wilsonii Rizz.- Faveiro-de-Wilson (Fabaceae caesalpinioideae). Revista Brasileira de Sementes 30(1): 96-101.
Mendonça, M.P. & Lins, L.V. (orgs.). 2000. Lista vermelha das espécies ameaçadas de extinção da flora de Minas Gerais. Belo Horizonte, Fundação Biodiversitas e Fundação Zoo-Botânica de Belo Horizonte.
MMA - Ministério do Meio Ambiente. 2008. Instrução Normativa nº 6 de 23 de setembro de 2008. Diário Oficial da União de 24 de setembro de 2008. nº 185. Seção 1. p.75-83. Brasília, MMA.
Muñoz, M.E.S.; Giovanni, R.; Siqueira, M.F.; Sutton, T.; Brewer, P.; Pereira, R.S.; Canhos, D.A.L. & Canhos, V.P. 2009. OpenModeller: a generic approach to species' potential distribution modelling. GeoInformatica DOI: 10.1007/s10707-009-0090-7.
Phillips, S.J.; Anderson, R.P. & Schapire, R.E. 2006. Maximum entropy modeling of species geographic distributions. Ecological Modelling 190: 231-259.
Rizzini, C.T. 1969. Espécies novas de árvores do Planalto Central Brasileiro. Anais da Academia Brasileira de Ciências 41 (2): 239-244.
Rizzini, C.T. & Mattos Filho, A.1986. Espécies Vegetais em Extinção. Boletim da Fundação Brasileira para Conservação da Natureza 21: 99-103.
Scolforo, J.R. & Carvalho, L.M.T. 2006. Mapeamento e inventário da flora e dos remanescentes florestais de Minas Gerais. Lavras, UFLA.
Silva, M.F. 1986. Dimorphandra (Caesalpiniaceae). Flora Neotropica 44: 1-128.
Silva, F.A.M; Assad, E.D. & Evangelista, B.A. 2008. Caracterização Climática do Bioma Cerrado. Pp. 69-88. In Sano, S.M.; Almeida, S.P. & Ribeiro, J.F. (Eds). Cerrado: ecologia e flora, v.1. Brasília, Embrapa Cerrados.
Silva, M. 2012. Micobiota folícola de Dimorphandra wilsonii, espécie arbórea brasileira ameaçada de extinção. Dissertação de mestrado. Viçosa, Universidade Federal de Viçosa
Souza, H.A.V. & Lovato, M.B. 2010. Genetic diversity and structure of the critically endangered tree Dimorphandra wilsonii and of the widespread in the Brazilian Cerrado Dimorphandra mollis: Implications for conservation. Biochemical Systematics and Ecology 38 (2010) 49-56
Stockwell, D.R.B. & Peters, D. 1999. The GARP modelling system: Problems and solutions to automated spatial prediction. International Journal of Geographic Information Systems 13:143-158.
Thiers, B. [continuously updated]. Index Herbariorum: a global directory of public herbaria and associated staff. New York Botanical Garden's Virtual Herbarium. Website http://sweetgum.nybg.org/ih/. [accessed on October, 15, 2012].
Walter, K.S. & Gillett, H.J (eds.). 1998. 1997 IUCN Red List of Threatened Plants. Compiled by the World Conservation Monitoring Centre. Gland and Cambridge, The World Conservation Union, IUCN.
Received: 1 November, 2013. Accepted: 26 March, 2014
- Arruda, M.B.; Proença, C.E.B; Rodrigues, S.C.; Campos, R.N.; Martins, R.C. & Martins, E.S. 2008. Ecorregiões, Unidades de Conservação e Representatividade Ecológica do Bioma Cerrado. Pp. 229-272. In Sano, S.M.; Almeida, S.P. & Ribeiro, J.F. (Eds). Cerrado: ecologia e flora, v.1. Brasília, Embrapa Cerrados.
- Bizerril, M.X.A.; Rodrigues, F.H.G. & Hass, A. 2005. Fruit consumption and seed dispersal of Dimorphandra mollis Benth. (Leguminosae) by the lowland tapir in the Cerrado of Central Brazil. Brazilian Journal of Biology 65(3): 407-413.
- Brasil, 2010. Agência Nacional de Vigilância Sanitária. Farmacopéia Brasileira. V. 2. Brasília, Anvisa.
- Budowski, G. N. 1965. Distribution of tropical American rain forest species in the light of succession processes. Turrialba 15(1): 40-42.
- Drummond, G.M.; Machado, A.B.M.; Martins, C.S.; Mendonça, M.P. & Stehmann, J.R. (Eds.) 2008. Listas vermelhas das espécies da fauna e da flora ameaçadas de extinção em Minas Gerais 2ª ed. Belo Horizonte, Fundação Biodiversitas. Available from: http://www.biodiversitas.org.br/listas-mg/lista_floramg.asp Cited 2013 Sep 11.
- Fernandes, F. M. 2006. Dimorphandra wilsonii In: IUCN 2012. IUCN Red List of Threatened Species Version 2012.1. Available from: http://www.iucnredlist.org/details/61926/0 Cited 2013 Aug 26.
- Fernandes, F. M.; Fonseca, A. G.; Kaechele, K; Goulart, M. F.; Marinho, W.; souza, H A.V; Queiroz, A. R.; Giorni, V.T; Oliveira, G.; Rodrigues, M. J.; Bacelar, M. & Lovato, M. B. 2007. Tentando evitar mais uma extinção: o caso do "Faveiro de Wilson" (Dimorphandra wilsonii Rizzini). Pp. 87-98. In: Pereira, T. S.; Costa, M. L. M. N. & Jackson, P. W. (Orgs.). Recuperando o verde para as cidades: a experiência dos jardins botânicos brasileiros. Rio de Janeiro, Rede Brasileira de Jardins Botânicos, Instituto de Pesquisas Jardim Botânico do Rio de Janeiro, BGCI.
- Ferreti, A.R. 2002. Fundamentos Ecológicos para o Planejamento da Restauração Florestal. Pp. 21-26. In: Galvão, A.P.M.; Medeiros, A.C.S.. Restauração da Mata Atlântica em Áreas de sua Primitiva Ocorrência Natural Colombo, Embrapa.
- Fonseca, M.B.; Franca, M.G.C.; Zonta, E. & Giorni, V.T. 2010. Crescimento inicial de Dimorphandra wilsonii (Fabaceae - Caesalpinioideae) em diferentes condições de fertilidade em solo de cerrado. Acta Botanica Brasilica 24(2): 322-327.
- Fonseca, M.B.; Carolino, M.M.S.S. de L.; Dias, T.; Cruz, C. & França, M.G.C. 2013. Early growth of Brazilian tree Dimorphandra wilsonii is also threatened by African grass Urochloa decumbens, Journal of Plant Interactions 9(1):92-99.
- Fundação SOS Mata Atlântica & Instituto Nacional de Pesquisas Espaciais. 2011. Atlas dos Remanescentes Florestais da Mata Atlântica Período 2008-2010 São Paulo, Fundação SOS Mata Atlântica e INPE.
- GMG - Governo de Minas Gerais. 2004. Decreto 43904-2004. Diário Oficial do Estado de Minas Gerais 27/10/2004. p. 5,col.1. Belo Horizonte, GMG.
- Hijmans, R.J.; Cameron, S.E.; Parra, J.L.; Jones, P.G. & Jarvis, A. 2005. Very High Resolution Interpolated Climate Surfaces for Global Land Areas. International Journal of Climatology 25: 1965-1978.
- IBGE - Instituto Brasileiro de Geografia e Estatística. 2012. Manual técnico da vegetação brasileira Série Manuais Técnicos em Geociências. 2ª ed. Rio de Janeiro, IBGE.
- IEF - Instituto Estadual de Florestas. 2011. Atlas da Fauna em Unidades de Conservação do Estado de Minas Gerais -Vol. 1 - Mastofauna. http://intranet.meioambiente.mg.gov.br/documentos/atlas-parte1.pdf Belo Horizonte, IEF.
- IUCN 2001. IUCN Red List Categories and Criteria: Version 3.1 IUCN Species Survival Commission. Gland and Cambridge, IUCN.
- IUCN 2013. IUCN Red List of Threatened Species Version 2012.2. Available from: http://www.iucnredlist.org/search Cited 2013 Oct 26.
- Kunin, W.E. & Gaston, K.J. (Eds.) 1997. The biology of rarity Causes and consequences of rare-common differences. London, Chapman & Hall.
- Lins, L.V.; Ribeiro, F.; Andrade, R.D. & Rigueira, S.E. 2011. Educação ambiental para a conservação da região da Serra da Canastra, utilizando o pato- mergulhão (Mergus octosetaceus) como espécie bandeira. Belo Horizonte, Instituto Estadual de Florestas. Revista MG Biota 4(3): 34-55.
- Lopes, J.C. & Matheus, M.T. 2008. Caracterização morfológica de sementes, plântulas e da germinação de Dimorphandra wilsonii Rizz.- Faveiro-de-Wilson (Fabaceae caesalpinioideae). Revista Brasileira de Sementes 30(1): 96-101.
- Mendonça, M.P. & Lins, L.V. (orgs.). 2000. Lista vermelha das espécies ameaçadas de extinção da flora de Minas Gerais. Belo Horizonte, Fundação Biodiversitas e Fundação Zoo-Botânica de Belo Horizonte.
- MMA - Ministério do Meio Ambiente. 2008. Instrução Normativa nº 6 de 23 de setembro de 2008. Diário Oficial da União de 24 de setembro de 2008. nº 185. Seção 1. p.75-83. Brasília, MMA.
- Muñoz, M.E.S.; Giovanni, R.; Siqueira, M.F.; Sutton, T.; Brewer, P.; Pereira, R.S.; Canhos, D.A.L. & Canhos, V.P. 2009. OpenModeller: a generic approach to species' potential distribution modelling. GeoInformatica DOI: 10.1007/s10707-009-0090-7.
- Phillips, S.J.; Anderson, R.P. & Schapire, R.E. 2006. Maximum entropy modeling of species geographic distributions. Ecological Modelling 190: 231-259.
- Rizzini, C.T. 1969. Espécies novas de árvores do Planalto Central Brasileiro. Anais da Academia Brasileira de Ciências 41 (2): 239-244.
- Rizzini, C.T. & Mattos Filho, A.1986. Espécies Vegetais em Extinção. Boletim da Fundação Brasileira para Conservação da Natureza 21: 99-103.
- Scolforo, J.R. & Carvalho, L.M.T. 2006. Mapeamento e inventário da flora e dos remanescentes florestais de Minas Gerais. Lavras, UFLA.
- Silva, M.F. 1986. Dimorphandra (Caesalpiniaceae). Flora Neotropica 44: 1-128
- Silva, F.A.M; Assad, E.D. & Evangelista, B.A. 2008. Caracterização Climática do Bioma Cerrado. Pp. 69-88. In Sano, S.M.; Almeida, S.P. & Ribeiro, J.F. (Eds). Cerrado: ecologia e flora, v.1. Brasília, Embrapa Cerrados.
- Silva, M. 2012. Micobiota folícola de Dimorphandra wilsonii, espécie arbórea brasileira ameaçada de extinção. Dissertação de mestrado. Viçosa, Universidade Federal de Viçosa
- Souza, H.A.V. & Lovato, M.B. 2010. Genetic diversity and structure of the critically endangered tree Dimorphandra wilsonii and of the widespread in the Brazilian Cerrado Dimorphandra mollis: Implications for conservation. Biochemical Systematics and Ecology 38 (2010) 49-56
- Stockwell, D.R.B. & Peters, D. 1999. The GARP modelling system: Problems and solutions to automated spatial prediction. International Journal of Geographic Information Systems 13:143-158.
- Walter, K.S. & Gillett, H.J (eds.). 1998. 1997 IUCN Red List of Threatened Plants Compiled by the World Conservation Monitoring Centre. Gland and Cambridge, The World Conservation Union, IUCN.
Publication Dates
-
Publication in this collection
02 Oct 2014 -
Date of issue
Sept 2014
History
-
Received
01 Nov 2013 -
Accepted
26 Mar 2014