ABSTRACT
Understanding the genetic structure and diversity of plants is fundamental to their conservation and permits their sustainable use by local communities. The genus Maytenus (Celastraceae) is composed of plants possessing pharmacological and antioxidant properties. However, the genetic and economic properties of the species M. dasyclada, a typical species of Araucaria forests in Brazil and Uruguay, have been little studied. In this work, the genetic structure and diversity of natural populations of M. dasyclada located in unprotected and preserved forest remnants were investigated using RAPD and isozymes markers. The results demonstrated that in areas of preservation, populations of M. dasyclada possess a relatively high degree of polymorphism and high values for Na, Ne, Shannon index, He and Ho, indicating high genetic variability. Moreover, these protected populations are very close to each other and potentially experience significant gene flow. The results presented here highlight the relevance of preservation areas for the conservation of M. dasyclada, and that populations inhabiting these areas could serve as a genetic source for the recovery of populations in regions where genetic diversity has been lost.
Keywords
conservation; forest reserves; genetic diversity; Maytenus dasyclada; molecular markers
Introduction
Many tropical countries, including Brazil, have employed extractive exploration with native species, sometimes resulting in their disappearance from natural ecosystems. Extinction of these species even at a local scale results in loss of important genetic information and, consequently, reduced capacity for ecosystem recuperation. Knowledge of the genetic structure and diversity of plants is fundamental for their conservation and permits their use by local communities (Nass et al. 2012Nass LL, Sigrist MS, Ribeiro CSC, Reifschneider FJB. 2012. Genetic resources: the basis for sustainable and competitive plant breeding. Crop Breeding and Applied Biotechnology 12: 75-86. ; Asma et al. 2016Asma M, Khaled M, Nadia BB. 2016. Genetic variability and phylogenetic relationships studies of Aegilops L. using some molecular markers. International Journal of Agronomy and Agricultural Research 8: 14-25.). In these sense, investigations of genetics in native plants with ecological and economical potential can inform both extractive activities and efficient forest management (Piotti et al. 2013Piotti A, Leonardi S, Heuertz M. et al. 2013. Within-population genetic structure in beech (Fagus sylvatica L.) stands characterized by different disturbance histories: does forest management simplify population substructure? PLoSone, 5,8, e73391. doi: 10.1371/journal.pone.0073391.
https://doi.org/10.1371/journal.pone.007...
).
Genetic variability is the basis for species conservation. Knowledge of the genetic structure of plants is essential for the establishment of effective conservation actions, particularly for species in which knowledge is lacking, such as tropical trees (Pautasso 2009Pautasso M. 2009. Geographical genetics and the conservation of forest trees. Perspectives in Plant Ecology, Evolution and Systematics 11: 157-189.). Molecular or biochemical genetic markers are widely used in studies of genetic variability both within and among species (Rutledge et al. 2000Rutledge J, Talbert RE, Sneller CH. 2000. RAPD analysis of genetic variation among propanil-resistant and -susceptible Echinochloa crus-galli populations in Arkansas. Weed Science 48: 669-674.; Amin et al. 2012Amin A, Dhillon TS, Mir H, Shah MA. 2012. Assessment of genetic diversity in carrot (Daucus carota L.) germplasms. SAARC Journal of Agriculture 10: 9-20.; Cruz et al. 2016Cruz ES, Dantas ACVL, Carmo CD, Bastos LP. 2016. Molecular characterization of jaboticaba tree genotypes located in the municipalities of recôncavo of Bahia. Revista Brasileira de Fruticultura 38: e-510. doi: 10.1590/0100-29452016510
https://doi.org/10.1590/0100-29452016510...
). RAPD (Random Amplified Polymorphic DNA) is a molecular marker that allows detection of polymorphism without previous knowledge of genetic sequences, and is commonly used in plant population studies (Ahmed et al. 2012Ahmed N, Mir JI, Mir RR, et al. 2012. SSR and RAPD analysis of genetic diversity in walnut (Juglans regia L.) genotypes from Jammu and Kashmir, India. Physiology and Molecular Biology of Plants 18: 149-160.; Zhang et al. 2013Zhang L, Zhang HG, Li XF. 2013. Analysis of genetic diversity in Larix gmelinii (Pinaceae) with RAPD and ISSR markers. Genetics and Molecular Research 12: 196-207.; Tiwari & Shrivastava 2016Tiwari P, Shrivastava A. 2016. Efficacy of RAPD markers for molecular diversity analysis of Withania somnifera (L) Dunal in central India. International Journal of Advanced Research in Biological Sciences 3: 126-130.). Isozymes are biochemical markers used in the investigation of genetic diversity in natural populations (Kaya & Bilben 2012Kaya N, Bilgen BB. 2012. Relationship between geographic proximity and genetic similarity among the natural populations of Pinus brutia Ten.: Its implication on genetic conservation. Pakistan Journal of Botany 44: 1047-1052. ; Saini & Yadav 2013Saini S, Yadav JP. 2013. Genetic variation in natural populations of Salvadora oleoides: An important medicinal plant that needs conservation. Pelagia Research Library 3: 20-27.), allowing analysis of genotype frequencies, coefficients of diversity genetic and heterozygosis, and estimation of possible derivations of Hardy-Weinberg´s equilibrium (Alfenas et al. 1998Alfenas CA, Brune W, Oliveira JR, Alonso SK, Scorticnhini M. 1998. In: Alfenas AC. (ed.) Eletroforese de isoenzimas e proteínas afins: fundamentos e aplicações em plantas e microrganismos. Viçosa, UFV. p. 85-114.). The combination of RAPD and isozymes is an effective wat to evaluate genetic diversity in plant populations for which there is limited knowledge of the genetic profile.
The genus Maytenus belongs to the family Celastraceae, which includes 55 genera and 850 species distributed among tropical and subtropical countries. There are 77 Maytenus species in the Brazilian flora (Carvalho-Okano 1992Carvalho-Okano RM. 1992. Estudos taxonômicos do gênero Maytenus Mol. emend. Mol. (Celastraceae) do Brasil extra-amazônico. PhD Thesis, Universidade Estadual de Campinas, Campinas.), some of which have pharmacological and antioxidant properties (Corsino et al. 2003Corsino J, Silva DHS, Zanoni MVB, et al. 2003. Antioxidant flavan-3-ols and flavonol glycosides from Mayteus aquifolium. Phytotherapy Research 17: 913-916.; Pereira et al. 2005Pereira AMS, Januário AH, Queiroz MEE, Biondo R, França SC. 2005. Evaluation of Maytenus aquifolia Mart. and Maytenus ilicifolia Mart. chemotypes for tannins, total phenols and triterpenes. Revista Brasileira de Plantas Medicinais 8: 13-17.; Tiberti et al. 2007Tiberti LA, Yariwake JH, Ndjoko K, Hostettmann K. 2007. Identification of flavonols in leaves of Maytenus ilicifolia and M. aquifolium (Celastraceae) by LC/UV/MS analysis. Journal of Chromatography B 846: 378-383.; Santos-Oliveira et al. 2009Santos-Oliveira R, Coulaud-Cunha S, Colaço W. 2009. Review of Maytenus ilicifolia Mart. ex Reissek, Celastraceae. Contribution to the studies of pharmacological properties. Brazilian Journal of Pharmacognosy 19: 650-659.; Cansian et al. 2015Cansian RL, Kubiak GB, Borsatti L, et al. 2015. Antioxidant and genotoxic properties of Maytenus dasyclada: a comparative study in relation to Maytenus reference species. Brazilian Journal of Biology 15: 471-476.; Santoyo et al. 2015Santoyo CZ, Espinoza LZ, Puente RZ, et al. 2015. Antipsychotic and behavior effect of the ethanolic extract from the bark of Maytenus macrocarpa (Ruiz & Pav.) Briq. in mice. Pharmacognosy Communications 5: 244-249.; Bevenutti et al. 2016Bevenutti DF, Della Monache F, Cechinel Filho V, Andrade SF, Niero R. 2016. Phytochemical analysis and gastroprotective activity of the root bark from Maytenus robusta. Natural Product Communications 11: 597-599.).
Among other Brazilian Maytenus species, Maytenus dasyclada, known as “coração-de-bugre”, is a conservation concern. This species is poorly studied, however it is known that populations are declining in the natural flora, especially because it is a bush species which is commonly removed to “clean” forest and urban areas. Although not currently on the list of endangered species, M. dasyclada is typical species of Araucaria forest and is in danger of extinction due to forest fragmentation (Higuchi et al. 2012Higuchi P, Silva AC, Ferreira TS, et al. 2012. Floristic composition and phytogeography of the tree component of Araucaria Forest fragments in southern Brazil. Brazilian Journal of Botany 35: 145-157.).
In this work, the genetic structure and diversity in natural populations of M. dasyclada were studied in unprotected and protected (institutionalized areas of preservation; Federal Reserve) forest remnants. Seven M. dasyclada populations were chosen in southern Brazil for comparison using RAPD and isozymes markers, with the goal of determining whether habitat fragmentation generated by anthropogenic activity has already caused perceptible effects at the population level (i.e., by reducing the index of genetic diversity). We hypothesized that populations located in protected forest remnants would be in a better state of genetic conservation than are populations located in unprotected areas.
Materials and methods
This study was conducted in Rio Grande do Sul and Santa Catarina states in southern Brazil (Fig. 1). Maytenus dasyclada Mart. were collected at seven sampling sites (N= 60 individuals). Three of the sampling sites lie in the Floresta Nacional de Passo Fundo (FLONA) forest reserve, with each site differing in landscape physiognomy and thus considered as a distinct population, as follows: native forest site (FLONA I); site with Pinus spp. (FLONA II); and site with Araucaria angustifolia (Bertol.) (FLONA III). Descriptive data of the seven sample sites are presented in Table 1. These data demonstrate very similar landscape features among the Erechim, Ronda Alta and Passo Fundo regions, and different landscape features in the Lages and Encruzilhada do Sul regions.
Characterization of the collection sites of Maytenus dasyclada and Matrix of geographic distances (Km) (A) and altitude differences (m) (B) in populations of Maytenus dasyclada sampling in Rio Grande do Sul and Santa Catarina (Brazil).
RAPD analyses
The freshly collected plants were kept in liquid nitrogen during the transport stored in a laboratory freezer at -85 °C until RAPD and isozymes analysis.
For RAPD, genomic DNA was extracted as described in Mossi et al. (2009Mossi AJ, Cansian RL, Leontiev-Orlov O, et al. 2009. Genetic diversity and conservation of native populations of Maytenus ilicifolia Mart. ex Reiss. Brazilian Journal of Biology 69: 447-453.) with minor modifications. The foliar tissues were macerated in liquid nitrogen, mixed with CTAB buffer (2 % CTAB, 1.4 M NaCl, 20 mM EDTA, 100 mM Tris-HCl pH 8.0, 0.2 % 2-mercaptoethanol) and incubated at 65 °C for 30 min. Proteins were eliminated with chloroform: isoamyl alcohol (24:1), and samples were washed once with ethanol absolute and once with 70 % ethanol. The DNA pellet was then re-suspended in TE buffer (10 mM TrisHCl pH 8.0, 1 mM EDTA). DNA quality was assessed using spectrophotometry (260 nm/ 280 nm) and visualization in agarose gel (0.8 %).
RAPD reactions were performed following Williams et al. (1990Williams JG, Kubelik AR, Livak KJ, Rafalski LA, Tingey SV. 1990. DNA polymorphism amplified by arbitrary primers are useful as genetic markers. Nucleic Acids Research 18: 6531-6535.). Briefly, PCR mixtures contained 40 ng of DNA, 1.5 U Taq DNA polymerase, specific primers (0.2 mM), MgCl2 (3 mM), Triton (0.25 mM), and Taq Buffer (Gibco BRL). The cycle program was as follows: initial denaturation (92 °C for 3 min) followed by 40 cycles of 92 °C for 1 min, 35 °C for 1 min, and 72 °C for 2 min, with a final extension step of 72 °C for 3 min. We then tested 30 primers using OPB, OPF, OPW, and OPY kits (Operon Technologies Inc.), and obtained the best amplification results with OPB 11, OPF 01, OPF 09, OPF 10, OPF 12, OPH 12, OPW 04, OPW 08, OPW 12, OPY 10, OPY 16, OPY 17 and OPY 18.
After amplification, DNA fragments were submitted to electrophoresis (240 min, 90V) in agarose gel (1.4 %) containing ethidium bromide (0.5 mg/mL) and photographed using a transilluminator digital system (GEL-PRO Imager, Media Cybernetics, Silver Spring, MD). The data were classified in relation to the presence or absence of specific bands, generating a matrix for analysis using the program PAST 2.0. The genograms of similarity (UPGMA) were constructed using the Jaccard coefficient. Principal component analyses (PCA) were carried out and Shannon index was calculated using PAST 2.0. The apparent number (Na) and effective number (Ne) of alleles, observed (Ho) and expected (He) heterozygosis, Shannon Index (Nei 1973Nei M. 1973. Analysis of gene diversity in subdivided populations. Proceedings of the National Academy of Sciences USA 3321-3323.), genetic distances (Nei 1972Nei M. 1972. Genetic distance between populations. American Naturalist 106: 283-292.), and analyses of molecular variance (AMOVA) were analyzed using GenAlEX 6.4 (Peakall & Smouse 2009Peakall R, Smouse PE. 2009. GENALEX 6.4: genetic analysis in Excel. Population genetic software for teaching and research. Molecular Ecology Notes 6: 288-295.).
Isozyme assays
Enzyme extractions were performed using approximately 1 g of leaf tissue per mL of extraction solution 1 (see Alfenas et al. 1998Alfenas CA, Brune W, Oliveira JR, Alonso SK, Scorticnhini M. 1998. In: Alfenas AC. (ed.) Eletroforese de isoenzimas e proteínas afins: fundamentos e aplicações em plantas e microrganismos. Viçosa, UFV. p. 85-114.), adding approximately 150 mg of PVP-40 during grinding to remove phenolic compounds and increase extract stability. The extract was then subjected to vertical electrophoresis (220 V, 40 mA) in a biphasic polyacrylamide gel formed by stacking phase (5 % polyacrylamide in Tris-HCl, pH 6.8) and running phase (12 % polyacrylamide in Tris-HCl, pH 8.8) (Alfenas et al. 1998).
Eleven enzymatic systems were tested, with six of them active in M. dasyclada and generating 15 loci that were able to be interpreted: ADH - alcohol dehydrogenase (E.C. 1.1.1.1; 3 loci); EST - esterase (E.C. 3.1.1.1; 1 locus); GDH - glucose dehydrogenase (E.C. 1.1.1.47; 3 loci); PO - peroxidase (E.C. 1.11.1.7; 2 loci); SDH - sorbitol dehydrogenase (E.C. 1.1.1.14; 3 loci); SOD - superoxide dismutase (E.C. 1.15.1.1; 3 loci). Results were evaluated based on the presence, distinctness, number, and location of bands for each enzymatic system.
Isozyme data were analyzed using GenAlEX 6.4, which generated allelic frequencies, genetic diversity index (percentage of polymorphic loci (P), apparent number of alleles (Na), effective number of alleles (Ne), observed heterozygosis (Ho), expected heterozygosis (He), and Shannon Index), Wright’s genetic divergence (Fst), gene flow (Nm), and Nei's genetic distances. Analyses of molecular variance (AMOVA), principal components analysis (PCA), and linear regressions were carried out using the same program.
Results
The RAPD assay resulted in 115 bands, with 88.70 % polymorphic and 11.30 % monomorphic. We obtained on average eight polymorphic bands per primer versus only 1 monomorphic. The M. dasyclada populations differed with respect to polymorphisms present (Tab. 2). Higher rates were observed in FLONA III (78.88 %) and Encruzilhada do Sul (74.41 %), followed by Lages (61.42 %). Ronda Alta and Erechim showed similar rates (41.79 % and 44.87 %, respectively).
Variation in the polymorphism degree between Maytenus dasyclada populations analyzed by RAPD.
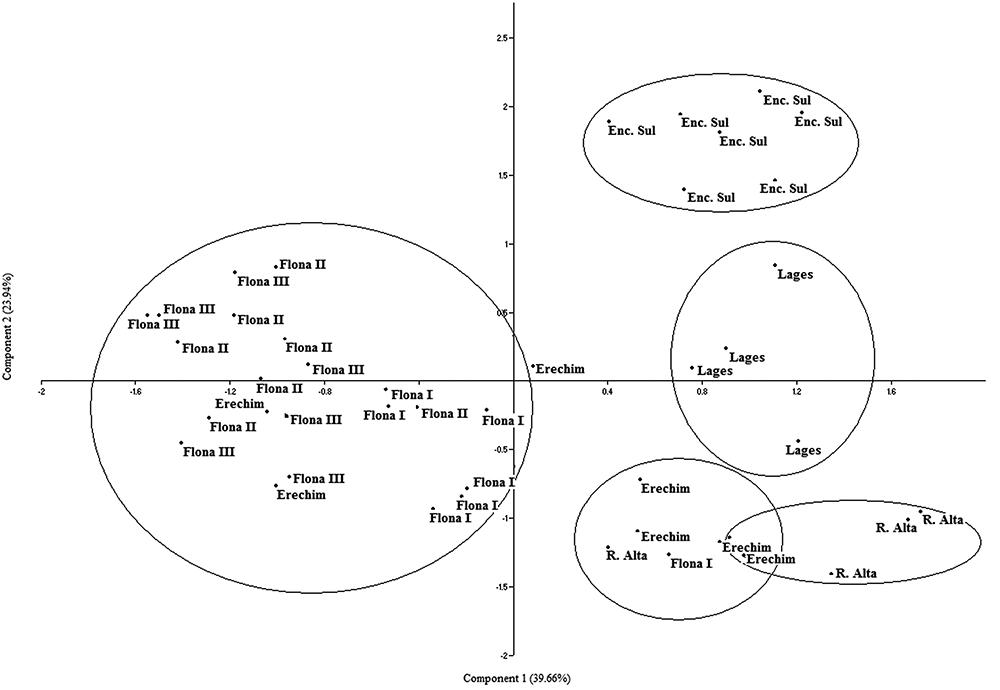
The genogram originated by RAPD (Fig. 2) showed clusters of plants belonging primarily to the same populations. However, different populations could be approximated by generating two major groups: i) FLONA I, FLONA II and Erechim; and ii) FLONA III and Ronda Alta. Similarity between all plants ranged from 0.47 to 0.90, pointing to high genetic diversity. Separation between the studied populations becomes more evident in the PCA (Fig. 3). FLONA I and FLONA III strongly overlap, while FLONA II is only partially superposed with these two populations. Although Lages and Encruzilhada do Sul are related (Fig. 2), they compose two separate populations (Fig. 3).
Genogram of seven Maytenus dasyclada populations collected in Rio Grande do Sul and Santa Catarina (Brazil) based on RAPD markers.
Principal component analyses (PCA) based on RAPD markers in Maytenus dasyclada populations collected in Rio Grande do Sul and Santa Catarina (Brazil).
Among the 15 isozymes that were active in M. dasyclada, about 58.76 % were polymorphic and 41.24 % monomorphic. Higher rates of polymorphic loci were observed in the FLONA populations (73.33 % and 66.67 % for FLONA II and FLONA I/FLONA III, respectively) and lower rates were observed in Erechim (40 %) and Lages (33.33 %). Regarding genetic equity, values for M. dasyclada populations were low with the exception of FLONA I (Tab. 3).
Frequency of polymorphic loci and genetic equity in populations of Maytenus dasyclada based in isozymes data.
The isoenzymatic markers identified loci with two to six alleles. The number of apparent alleles (Na) ranged from 1.667 (FLONA II) to 1.067 (Lages), while number of effective alleles (Ne) ranged from 1.202 to 1.475 (Tab. 4). For these two parameters (Na and Ne), FLONA populations presented the highest values. Shannon index values were also higher in populations from FLONA sites.
Number of apparent (Na) and effective (Ne) alleles, expected (He) and observed (Ho) heterozygosity and Shannon index in populations of Maytenus dasyclada, sampling in Rio Grande do Sul and Santa Catarina (Brazil) based in isozymes data.
Analysis of isozyme data resulted in observed heterozygosity (Ho) being higher than expected (He) in all study populations (Tab. 4). Plants collected in the three FLONA sites and Ronda Alta had higher Ho than other groups.
Analysis of genetic divergence using the Wright index (Fst) (Wright 1951Wright S. 1951. The genetical structure of populations. Annals of Eugenics 15: 395-420.) for isozyme data revealed that the Lages population is the most distant from all others M. dasyclada populations, while the FLONA I, FLONA II and FLONA III are very close. Ronda Alta and Erechim also have low genetic divergence (Tab. 5). Gene flow (Nm) is partially in accordance with divergence data, as it demonstrates high degree of similarity between the three FLONA populations, the Erechim and Ronda Alta populations, and the Encruzilhada do Sul and Ronda Alta populations (Tab. 5). According with Wright (Wright 1931Wright S. 1931. Evolution in Mendelian populations. Genetics 16: 97-159.), because Nm < 1 all the other populations compared in this study are in genetic isolation. Despite being a dominant marker, the RAPD data were not used for analyses of heterozygosity, divergence, and gene flow.
Genetic divergence (Fst) and flow gene (Nm) index in populations of Maytenus dasyclada, sampling in Rio Grande do Sul and Santa Catarina (Brazil) based in isozymes data.
The AMOVA results indicated that 34 % and 43 % of the genetic variability is inter-populational for RAPD and isoenzymatic data, respectively, while 66 % and 57 % of the variability was intra-populational. The PCA based on isozyme data showed a superposition between the FLONA populations, the group formed by Erechim/Ronda Alta, and the group formed by Encruzilhada do Sul/ Lages (Fig. 4).
Principal component analyses (PCA) based on isozyme markers in Maytenus dasyclada populations collected in Rio Grande do Sul and Santa Catarina (Brazil).
Nei´s genetic distances were calculated using RAPD and isozymes data (Tab. 6). We observed a positive correlation between the Nei's distances obtained by isozyme and RAPD data (p = 0.010, R2 = 0.33). The values of Nei's genetic distance with the RAPD data were more similar among populations than were values from isozyme data. The RAPD analysis indicates greater genetic distances in the populations of Ronda Alta, Lages and Encruzilhada do Sul, particularly in relation to FLONA II and FLONA III. The isozyme data indicates large genetic distances, primarily between Lages and Encruzilhada do Sul in relation to FLONA I, II, and III.
Values of Nei´s genetic distance in populations of Maytenus dasyclada based in RAPD and isozyme data.
The Lages population has the largest genetic distance in relation to all others except Encruzilhada do Sul, which exhibits short distance from Lages (0.134). The expected lower genetic distance among the three populations of FLONA was best demonstrated by Nei's genetic distance values from isozyme data.
Analysis of isoenzymatic data revealed positive correlations between Nei´s genetic distance and geographic distances of the M. dasyclada populations (p = 0.031, R2 = 0.3091), as well between Nei´s genetic distances and altitude differences (p = 0.008, R2 = 0.4192). No significant correlation between Nei´s distance and altitude or geographic distances was found using RAPD data.
Discussion
The genus Maytenus is a group of plants with pharmacological and antioxidant properties (Corsino et al. 2003Corsino J, Silva DHS, Zanoni MVB, et al. 2003. Antioxidant flavan-3-ols and flavonol glycosides from Mayteus aquifolium. Phytotherapy Research 17: 913-916.; Pereira et al. 2005Pereira AMS, Januário AH, Queiroz MEE, Biondo R, França SC. 2005. Evaluation of Maytenus aquifolia Mart. and Maytenus ilicifolia Mart. chemotypes for tannins, total phenols and triterpenes. Revista Brasileira de Plantas Medicinais 8: 13-17.; Tiberti et al. 2007Tiberti LA, Yariwake JH, Ndjoko K, Hostettmann K. 2007. Identification of flavonols in leaves of Maytenus ilicifolia and M. aquifolium (Celastraceae) by LC/UV/MS analysis. Journal of Chromatography B 846: 378-383.; Santos-Oliveira et al. 2009Santos-Oliveira R, Coulaud-Cunha S, Colaço W. 2009. Review of Maytenus ilicifolia Mart. ex Reissek, Celastraceae. Contribution to the studies of pharmacological properties. Brazilian Journal of Pharmacognosy 19: 650-659.; Cansian et al. 2015Cansian RL, Kubiak GB, Borsatti L, et al. 2015. Antioxidant and genotoxic properties of Maytenus dasyclada: a comparative study in relation to Maytenus reference species. Brazilian Journal of Biology 15: 471-476.; Santoyo et al. 2015Santoyo CZ, Espinoza LZ, Puente RZ, et al. 2015. Antipsychotic and behavior effect of the ethanolic extract from the bark of Maytenus macrocarpa (Ruiz & Pav.) Briq. in mice. Pharmacognosy Communications 5: 244-249.; Bevenutti et al. 2016Bevenutti DF, Della Monache F, Cechinel Filho V, Andrade SF, Niero R. 2016. Phytochemical analysis and gastroprotective activity of the root bark from Maytenus robusta. Natural Product Communications 11: 597-599.). M. dasyclada in particular is typical species of Araucaria forest that occurs only in Brazil and Uruguay (Carvalho-Okano & Leitão-Filho 2004Carvalho-Okano RM, Leitão Filho HFG. 2004. O gênero Maytenus Mol. emend. Mol. (Celastraceae) no Brasil extra-amazônico. In: Reis MS, Silva SR. (ed.) Conservação e uso sustentável de plantas medicinais e aromáticas: Maytenus spp., espinheira-santa. Brasília, Ibama. p. 11-51.), for which knowledge is lacking about its genetic properties and potential economic value. In this work, the structure and genetic diversity of M. dasyclada was investigated in seven natural populations of Rio Grande do Sul and Santa Catarina (Brazil), using RAPD and isozymes markers.
The two markers analyzed pointed to relatively high degree of polymorphism in FLONA populations, and moderate degree of polymorphism in Erechim and Ronda Alta. In the Lages and Encruzilhada do Sul populations, isozyme analysis revealed a very low degree of polymorphism compared to RAPD analysis (Tabs. 2, 3). The RAPD marker generated different groups of populations both in the genogram and the PCA (Figs. 2, 3), as well as for groups formed in the isozyme PCA (Fig. 4). PCAs from both RAPD and isozyme data confirmed the grouping tendency among the FLONA populations shown in the genogram. Differences in these two markers are well described in the literature. Royo & Itoiz (2004Royo JB, Itoiz R. 2004. Evaluation of the discriminance capacity of RAPD, isoenzymes and morphologic markers in apple (Malus x domestica Borkh.) and the congruence among classifications. Genetic Resources and Crop Evolution 51: 153-160.) and Aguiar et al. (2015Aguiar JLN, Albert ALM, Moreira JC, Leite PC. 2015. RAPD-PCR na identificação molecular de plantas medicinais regulamentadas pelo Sistema Único de Saúde do Brasil. Vigilância Sanitária em Debate 3: 34-40.) argue that RAPD has greater discrimination capacity between individuals of the same species compared to isozymes, although the two markers lead to similar taxonomic clusters.
Parameter values (Na, Ne, Shannon index, He and Ho) for M. dasyclada collected in FLONA (I, II and III) indicate higher genetic variability in these populations (Silva et al. 2016Silva EF, Araújo RL, Martins CSR, Martins LSS, Veasey EA. 2016. Diversity and genetic structure of natural populations of araçá (Psidium guineense Sw.) Revista Caatinga 29: 37-44.) (Tab. 4). Moreover, the FLONA populations are very close to each other and potentially experience significant gene flow (Tab. 5), and may even be considered as a single population due to geographical proximity (e.g., as was suggested by Mossi et al. (2009Mossi AJ, Cansian RL, Leontiev-Orlov O, et al. 2009. Genetic diversity and conservation of native populations of Maytenus ilicifolia Mart. ex Reiss. Brazilian Journal of Biology 69: 447-453.) for M. ilicifolia). In contrast, the Lages population overall seems to have less variability (Tab. 4). The Lages population was the also most distant in relation to the other groups, with which there is no gene flow (Tab. 5). We do, however, speculate that these lower values could have been caused by the low number of specimens sampled this population (n = 4).
Lower Nei´s distance values were observed between FLONA samples (Tab. 6). This result generally agrees with results from Sahyun (2007Sahyun SA. 2007. Variabilidade genética de populações de espinheira-santa (Maytenus aquifolium) por marcadores moleculares. MSc Thesis, Universidade Estadual de Londrina, Londrina.) in a study of three M. aquifolium populations, in which they encountered distance values ranging from 0.034 to 0.961. Bessega et al. (2000Bessega C, Saidman BO, Vilardi JC. 2000. Isozyme and RAPD studies in Prosopis glandulosa and P. velutina (Leguminsae, Mimosoideae). Genetics and Molecular Biology 23: 639-648.) also reported positive correlations between geographic distance, altitude, and genetic distances obtained with isoenzymatic data that were absent in the analysis of RAPD data. Greater geographic or altitudinal distances resulted in greater genetic distances in all cases except Lages and Encruzilhada do Sul. The lack of correlation in these populations may be partly explained by the low number of plants collected in the Lages population. Low sample size may also justify the result of genetic flow between Ronda Alta and Encruzilhada do Sul populations, which should be low, due to geographic distance. However, Bessega (1997) Bessega C. 1997. Estudios isoenzimáticos en especies Americanas del Género Prosopis (Leguminosae). MSc Thesis, Universidad de Buenos Aires, Buenos Aires.also observed high levels of genetic similarity among P. glandulosa and other South American species, despite geographical isolation.
FLONA is a forest reserve, and the elevated indices of variability and polymorphism in M. dasyclada populations is indicative of a good state of genetic conservation. The percentage of polymorphisms was also high in FLONA II, an area that has been reforested with Pinus spp. These results together demonstrate the importance of preserved lands for the maintenance of native plants. On the other hand, sites with lower indices of polymorphism, especially in Ronda Alta, Lages and Erechim, have experienced loss of genetic diversity. This loss of diversity is likely related to habitat fragmentation (Pautasso 2009Pautasso M. 2009. Geographical genetics and the conservation of forest trees. Perspectives in Plant Ecology, Evolution and Systematics 11: 157-189.). This is potentially reversible through introduction of genetically distinct individuals such as those from FLONA I, II or III populations, as they form a cluster of plants belonging primarily to the same populations (Fig. 2) and do not overlap with the other analyzed groups (Figs. 3, 4).
In forest trees, high genetic diversity is better maintained within populations than among them (Hamrick 2004Hamrick JL. 2004. Response of forest trees to global environmental changes. Forest Ecology and Management 197: 323-335.; Scotti et al. 2016Scotti I, González-Martínez SC, Budde KB, Lalagüe H. 2016. Fifty years of genetic studies: what to make of the large amounts of variation found within populations? Annals of Forest Science 73: 69-75.), and results from the current study M. dasyclada are in concordance with this notion. This characteristic could be due to having effective pollen dispersal, which facilities gene flow (Kremer et al. 2012Kremer A, Ronce O, Robledo-Arnuncio JJ, et al. 2012. Long-distance gene flow and adaptation of forest trees to rapid climate change. Ecology Letters 15: 378-392.). Plants in the genus Maytenus are highly sought after by pollinators such as bees, ants, and other small insects, and seeds undergo zoochoric dispersal by birds and small monkeys (Lorenzi 2002Lorenzi H. 2002. Árvores brasileiras: manual de identificação e cultivo de plantas arbóreas do Brasil. 4th. edn. Nova Odessa, Instituto Plantarum.; Catharino et al. 2005Catharino ELM, Bernacci LC, Franco GADC, Durigan G, Metzger JP. 2005. Aspectos da composição e diversidade do componente arbóreo das florestas da Reserva Florestal do Morro Grande, Cotia, SP. Biota Neotropica 6: 127-144.; Zipparro et al. 2005Zipparro VB, Guilherme FAG, Almeida-Scabria RJ, Morellato LPC. 2005. Levantamento Florístico de Floresta Atlântica no Sul do Estado de São Paulo, Parque Estadual Intervales, Base Saibadela. Biota Neotropica 5: 127-144.; Carvalho 2010Carvalho PER. 2010. Espécies arbóreas brasileiras. Brasília, Embrapa Informação Tecnológica; Colombo, Embrapa Florestas.). M. dasyclada in preserved areas may have increased rates of pollination and seed dispersal, as the animals that carry out these services benefit from the same habitat protection. In these sense, conditions in the FLONA Preservation Park may facilitate maintenance of greater genetic diversity in M. dasyclada.
Implications for conservation
The low genetic diversity of many M. dasyclada populations in remaining unprotected forest could lead to potential risk of extinction, primarily due to continued fragmentation of the Atlantic forest (with particular emphasis on Araucaria forest).
The results of current study point to the relevance of preservation areas for conservation of M. dasyclada. Studies around the word have reinforced the importance and needs of adequate management of protected areas for conserving plant genetic resources (Koskela et al. 2013Koskela J, Lefèvre F, Schueler S. et al. 2013. Translating conservation genetics into management: Pan-European minimum requeriments for dynamic conservation units of forest tree genetic diversity. Biological Conservation 157: 39-49.; Maxted 2003Maxted N. 2003. Conserving the genetic resources of crop wild relatives in European Protected Areas. Biological Conservation 113: 411-417.; Volis 2016Volis S. 2016. How to conserve threatened Chinese plant species with extremely small populations? Plant Diversity 38: 45-52.), particularly for tropical plants (Pautasso 2009Pautasso M. 2009. Geographical genetics and the conservation of forest trees. Perspectives in Plant Ecology, Evolution and Systematics 11: 157-189.; Howe 2014Howe HF. 2014. Diversity storage: Implications for tropical conservation and restoration. Global Ecology and Conservation 2: 349-358.). Knowledge of ecological and genetic characteristics of plant species may inform novel approaches for conservation planning, and may aid in identification of areas that best complement the established natural reserves (Prado et al. 2010Prado A, Hawkins JA, Yesson C, Bárcenas RT. 2010. Multiple diversity to identify complementary conservation areas for the Baja California peninsular cacti. Biological Conservation 143: 1510-1520.; Silva et al. 2016Silva EF, Araújo RL, Martins CSR, Martins LSS, Veasey EA. 2016. Diversity and genetic structure of natural populations of araçá (Psidium guineense Sw.) Revista Caatinga 29: 37-44.). This study showed that although the Ronda Alta population showed a low degree of polymorphism (Tabs. 3, 6), it did present indicators of general variability, with parameter values approaching those of FLONA populations (Tab. 4). In these sense, Ronda Alta could be a target to future policies that aim to enlarge the M. dasyclada preservation area. In general, we emphasize the high rates of observed heterozygosity, higher than expected population heterozygosity (including in non-protected areas), and high structuring of populations, as these results indicate strong genetic diversity which must be preserved for the conservation this species.
Acknowledgements
The authors thanks CNPq, CAPES, FAPERGS and SCIT-RS for financial support.
References
- Aguiar JLN, Albert ALM, Moreira JC, Leite PC. 2015. RAPD-PCR na identificação molecular de plantas medicinais regulamentadas pelo Sistema Único de Saúde do Brasil. Vigilância Sanitária em Debate 3: 34-40.
- Ahmed N, Mir JI, Mir RR, et al 2012. SSR and RAPD analysis of genetic diversity in walnut (Juglans regia L.) genotypes from Jammu and Kashmir, India. Physiology and Molecular Biology of Plants 18: 149-160.
- Alfenas CA, Brune W, Oliveira JR, Alonso SK, Scorticnhini M. 1998. In: Alfenas AC. (ed.) Eletroforese de isoenzimas e proteínas afins: fundamentos e aplicações em plantas e microrganismos. Viçosa, UFV. p. 85-114.
- Amin A, Dhillon TS, Mir H, Shah MA. 2012. Assessment of genetic diversity in carrot (Daucus carota L.) germplasms. SAARC Journal of Agriculture 10: 9-20.
- Asma M, Khaled M, Nadia BB. 2016. Genetic variability and phylogenetic relationships studies of Aegilops L. using some molecular markers. International Journal of Agronomy and Agricultural Research 8: 14-25.
- Bessega C. 1997. Estudios isoenzimáticos en especies Americanas del Género Prosopis (Leguminosae). MSc Thesis, Universidad de Buenos Aires, Buenos Aires.
- Bessega C, Saidman BO, Vilardi JC. 2000. Isozyme and RAPD studies in Prosopis glandulosa and P. velutina (Leguminsae, Mimosoideae). Genetics and Molecular Biology 23: 639-648.
- Bevenutti DF, Della Monache F, Cechinel Filho V, Andrade SF, Niero R. 2016. Phytochemical analysis and gastroprotective activity of the root bark from Maytenus robusta Natural Product Communications 11: 597-599.
- Cansian RL, Kubiak GB, Borsatti L, et al 2015. Antioxidant and genotoxic properties of Maytenus dasyclada: a comparative study in relation to Maytenus reference species. Brazilian Journal of Biology 15: 471-476.
- Carvalho PER. 2010. Espécies arbóreas brasileiras. Brasília, Embrapa Informação Tecnológica; Colombo, Embrapa Florestas.
- Carvalho-Okano RM. 1992. Estudos taxonômicos do gênero Maytenus Mol. emend. Mol. (Celastraceae) do Brasil extra-amazônico. PhD Thesis, Universidade Estadual de Campinas, Campinas.
- Carvalho-Okano RM, Leitão Filho HFG. 2004. O gênero Maytenus Mol. emend. Mol. (Celastraceae) no Brasil extra-amazônico. In: Reis MS, Silva SR. (ed.) Conservação e uso sustentável de plantas medicinais e aromáticas: Maytenus spp., espinheira-santa. Brasília, Ibama. p. 11-51.
- Catharino ELM, Bernacci LC, Franco GADC, Durigan G, Metzger JP. 2005. Aspectos da composição e diversidade do componente arbóreo das florestas da Reserva Florestal do Morro Grande, Cotia, SP. Biota Neotropica 6: 127-144.
- Corsino J, Silva DHS, Zanoni MVB, et al 2003. Antioxidant flavan-3-ols and flavonol glycosides from Mayteus aquifolium Phytotherapy Research 17: 913-916.
- Cruz ES, Dantas ACVL, Carmo CD, Bastos LP. 2016. Molecular characterization of jaboticaba tree genotypes located in the municipalities of recôncavo of Bahia. Revista Brasileira de Fruticultura 38: e-510. doi: 10.1590/0100-29452016510
» https://doi.org/10.1590/0100-29452016510 - Hamrick JL. 2004. Response of forest trees to global environmental changes. Forest Ecology and Management 197: 323-335.
- Higuchi P, Silva AC, Ferreira TS, et al 2012. Floristic composition and phytogeography of the tree component of Araucaria Forest fragments in southern Brazil. Brazilian Journal of Botany 35: 145-157.
- Howe HF. 2014. Diversity storage: Implications for tropical conservation and restoration. Global Ecology and Conservation 2: 349-358.
- Kaya N, Bilgen BB. 2012. Relationship between geographic proximity and genetic similarity among the natural populations of Pinus brutia Ten.: Its implication on genetic conservation. Pakistan Journal of Botany 44: 1047-1052.
- Koskela J, Lefèvre F, Schueler S. et al 2013. Translating conservation genetics into management: Pan-European minimum requeriments for dynamic conservation units of forest tree genetic diversity. Biological Conservation 157: 39-49.
- Kremer A, Ronce O, Robledo-Arnuncio JJ, et al 2012. Long-distance gene flow and adaptation of forest trees to rapid climate change. Ecology Letters 15: 378-392.
- Lorenzi H. 2002. Árvores brasileiras: manual de identificação e cultivo de plantas arbóreas do Brasil. 4th. edn. Nova Odessa, Instituto Plantarum.
- Maxted N. 2003. Conserving the genetic resources of crop wild relatives in European Protected Areas. Biological Conservation 113: 411-417.
- Mossi AJ, Cansian RL, Leontiev-Orlov O, et al 2009. Genetic diversity and conservation of native populations of Maytenus ilicifolia Mart. ex Reiss. Brazilian Journal of Biology 69: 447-453.
- Nass LL, Sigrist MS, Ribeiro CSC, Reifschneider FJB. 2012. Genetic resources: the basis for sustainable and competitive plant breeding. Crop Breeding and Applied Biotechnology 12: 75-86.
- Nei M. 1972. Genetic distance between populations. American Naturalist 106: 283-292.
- Nei M. 1973. Analysis of gene diversity in subdivided populations. Proceedings of the National Academy of Sciences USA 3321-3323.
- Pautasso M. 2009. Geographical genetics and the conservation of forest trees. Perspectives in Plant Ecology, Evolution and Systematics 11: 157-189.
- Peakall R, Smouse PE. 2009. GENALEX 6.4: genetic analysis in Excel. Population genetic software for teaching and research. Molecular Ecology Notes 6: 288-295.
- Pereira AMS, Januário AH, Queiroz MEE, Biondo R, França SC. 2005. Evaluation of Maytenus aquifolia Mart. and Maytenus ilicifolia Mart. chemotypes for tannins, total phenols and triterpenes. Revista Brasileira de Plantas Medicinais 8: 13-17.
- Piotti A, Leonardi S, Heuertz M. et al 2013. Within-population genetic structure in beech (Fagus sylvatica L.) stands characterized by different disturbance histories: does forest management simplify population substructure? PLoSone, 5,8, e73391. doi: 10.1371/journal.pone.0073391.
» https://doi.org/10.1371/journal.pone.0073391 - Prado A, Hawkins JA, Yesson C, Bárcenas RT. 2010. Multiple diversity to identify complementary conservation areas for the Baja California peninsular cacti. Biological Conservation 143: 1510-1520.
- Royo JB, Itoiz R. 2004. Evaluation of the discriminance capacity of RAPD, isoenzymes and morphologic markers in apple (Malus x domestica Borkh.) and the congruence among classifications. Genetic Resources and Crop Evolution 51: 153-160.
- Rutledge J, Talbert RE, Sneller CH. 2000. RAPD analysis of genetic variation among propanil-resistant and -susceptible Echinochloa crus-galli populations in Arkansas. Weed Science 48: 669-674.
- Sahyun SA. 2007. Variabilidade genética de populações de espinheira-santa (Maytenus aquifolium) por marcadores moleculares. MSc Thesis, Universidade Estadual de Londrina, Londrina.
- Saini S, Yadav JP. 2013. Genetic variation in natural populations of Salvadora oleoides: An important medicinal plant that needs conservation. Pelagia Research Library 3: 20-27.
- Santos-Oliveira R, Coulaud-Cunha S, Colaço W. 2009. Review of Maytenus ilicifolia Mart. ex Reissek, Celastraceae. Contribution to the studies of pharmacological properties. Brazilian Journal of Pharmacognosy 19: 650-659.
- Santoyo CZ, Espinoza LZ, Puente RZ, et al 2015. Antipsychotic and behavior effect of the ethanolic extract from the bark of Maytenus macrocarpa (Ruiz & Pav.) Briq. in mice. Pharmacognosy Communications 5: 244-249.
- Scotti I, González-Martínez SC, Budde KB, Lalagüe H. 2016. Fifty years of genetic studies: what to make of the large amounts of variation found within populations? Annals of Forest Science 73: 69-75.
- Silva EF, Araújo RL, Martins CSR, Martins LSS, Veasey EA. 2016. Diversity and genetic structure of natural populations of araçá (Psidium guineense Sw.) Revista Caatinga 29: 37-44.
- Tiberti LA, Yariwake JH, Ndjoko K, Hostettmann K. 2007. Identification of flavonols in leaves of Maytenus ilicifolia and M. aquifolium (Celastraceae) by LC/UV/MS analysis. Journal of Chromatography B 846: 378-383.
- Tiwari P, Shrivastava A. 2016. Efficacy of RAPD markers for molecular diversity analysis of Withania somnifera (L) Dunal in central India. International Journal of Advanced Research in Biological Sciences 3: 126-130.
- Volis S. 2016. How to conserve threatened Chinese plant species with extremely small populations? Plant Diversity 38: 45-52.
- Williams JG, Kubelik AR, Livak KJ, Rafalski LA, Tingey SV. 1990. DNA polymorphism amplified by arbitrary primers are useful as genetic markers. Nucleic Acids Research 18: 6531-6535.
- Wright S. 1931. Evolution in Mendelian populations. Genetics 16: 97-159.
- Wright S. 1951. The genetical structure of populations. Annals of Eugenics 15: 395-420.
- Zhang L, Zhang HG, Li XF. 2013. Analysis of genetic diversity in Larix gmelinii (Pinaceae) with RAPD and ISSR markers. Genetics and Molecular Research 12: 196-207.
- Zipparro VB, Guilherme FAG, Almeida-Scabria RJ, Morellato LPC. 2005. Levantamento Florístico de Floresta Atlântica no Sul do Estado de São Paulo, Parque Estadual Intervales, Base Saibadela. Biota Neotropica 5: 127-144.
Publication Dates
-
Publication in this collection
13 Mar 2017 -
Date of issue
Jan-Mar 2017
History
-
Received
05 Dec 2016 -
Accepted
29 Jan 2017