ABSTRACT
The excessive sun exposure, coupled with lack of sun protection represents one of the biggest risks to the occurrence of skin cancer and photoaging. Recent strategies for photoprotection have included the incorporation of natural antioxidant and anti-inflammatory compounds, into sunscreens, and the oral administration of natural antioxidant extracts. In this work, we use Brazilian red propolis extract because its antioxidant and anti-inflammatory activities. The aim of this work was to evaluate the sun protection factor and antioxidant activity of different hydroalcoholic extracts of red propolis (70% and 75%) prepared from lyophilized red propolis at room and high temperatures. The sun protection factor in vitro was determined by a spectrophotometric method developed by Mansur. The hydroalcoholic extracts of red propolis incorporated with Filter UVA-UVB 5% Gel Permulem TR-1 presented absorption in the UVB region. Also, the in vitro capacity of the hydroalcoholic extracts of red propolis to increase photoprotective activity of Filter UVA-UVB 5% Gel was evaluated. The hydroalcoholic extracts of red propolis incorporated presented higher values of sun protection factor and showed synergism in the photoprotective activity of Filter UVA-UVB 5% Gel Permulem TR-1. The antioxidant activity and sun protection factor are correlated with total phenolics content of the extracts and the hydroalcoholic extract of red propolis 75% at room temperature was choosen. The formulation developed with Filter UVA-UVB 5% Gel Permulem TR-1 with this hydroalcoholic extract of red propolis showed safe to be applied on the skin according HET-CAM test. Suggests indication of hydroalcoholic extract of red propolis (75% – room temperature) associated to photoprotective formulations for use in photoprotective products.
Keywords:
Sun protection factor; Red propolis; Filter UVA-UVB; Total phenolics; HET-CAM test; Antioxidant activity
Introduction
The search for sun protection has intensified in recent decades as the sun's harmful effects have become more known and publicized. It is a well-known fact that an over exposure of human skin by ultraviolet light included sunburn cells, accelerated skin aging and induction of skin cancer. Sunscreens are products that provide protection against the effects of the sun, but they can accumulate in humans and frequent exposure may induce adverse effects. Because of this, research has been done to find a natural protector that is expected to have fewer side effects. During our research for natural sunscreen agents, we have focused on propolis. Propolis or bee glue is a resinous sticky substance that Apis mellifera L. collects from various plants, mixed with wax and other secretions. It has been used since ancient times because of its biological properties as an antioxidant, anti-microbial, antibiotic, anti-inflammatory and sun photoprotective. These characteristics have attracted the researchers' attention. Propolis is one of the natural products arising from the Brazilian flora that has been used for the discovery and development of new therapeutic agents. Recently, the Brazilian red propolis type has been highlighted due to its features. The chemical composition and pharmacological activities of this specific red propolis, have been intensely explored since the 90s, which is evidenced by the publication of over 100 papers between scientific articles and patents (Moura et al., 2017). The main compounds found in propolis are distributed in several major classes: phenolic acids and their esters, flavonoids (flavones, flavonones, flavonols, dihydroflavonols, chalcones), terpenes, steroids, aromatic aldehydes, alcohols, sesquiterpenes, naphthalene, stilbene derivatives of benzopyran, benzophenone, caffeic acid, cinnamic acid derivatives, and benzoic acid (Castro et al., 2009Castro, L.M., Do Nascimento, A.M., Ikegaki, M., Costa-Neto, C.M., Alencar, S.M., Rosalen, P.L., 2009. Identification of a bioactive compound isolated from Brazilian propolis type 6. Bioorg. Med. Chem. 17, 5332-5335.). Flavonoids represent the most common and widely distributed group of phenolics in red propolis. These are among the most active compounds in this resin, which act in different physiological processes, and perform various functions, including sun protection.
The purpose of this study was to determine the photoprotective properties of pure lyophilized Brazilian red propolis and the lyophilized Brazilian red propolis incorporated into the formulation sunscreen (Coiffard et al., 2014). The lyophilized propolis was used to mask or reduce undesired smell of the hydroalcoholic extract of propolis. The photoprotective action of the propolis extract was measured by the Mansur method. The photoprotective action of a sunscreen is measured universally by the sun protection factor (SPF) which establishes the increase of the dose of sun exposure with the photoprotective product applied without the occurrence of erythema (Gonçalves et al., 2018Gonçalves, M.C., Rossoni Junior, J.V., Rabelo, A.C.S., Costa, D.C., Cazati, T., Taylor, J.G., Dos Santos, V.M.R., 2018. Estudos preliminares da citoxicidade e propriedades fotoprotetoras de derivados de benzofenonas e lactonas. Rev. Virtual Quim. 10, 600-608.).
Materials and methods
General considerations
Solvents were purchased from Neon and used without further purification. The in vitro Solar Protection Factor (SPF) and Photostability were determined by the Spectrophotometric method developed by Mansur. The UV–Vis Ultraviolet readings were performed on the Bel Engineering UV-M51 Spectrophotometer. Flavonoid concentrations were determined by ultraviolet–visible absorption spectroscopy (UV–Vis) at 420 nm in a Thermo Scientific – Genesys 840 spectrophotometer. Filter UVA-UVB 5% Gel Permulem TR-1 with 2-phenyl-benzimidazole-5-sulfonic acid and 2-hydroxy-4-methoxybenzophenone as sunscreen agents was obtained by the BioFarma. The lyophilized red propolis samples were bought in Pharma Néctar.
Red propolis samples
The lyophilized red propolis samples were bought in Pharma Néctar and the crude samples of red propolis were obtained in Marechal Deodoro, state of Alagoas, located in the Northeastern Region of Brazil (SL 094237 and WL355342).
Preparation of ethanolic extracts of red propolis (EEP-70% or 75%) heated
The lyophilized red propolis sample (20 g) was extracted using ethanol 70% or 75% (Neon), 15 ml of ethanol for each 2 g, in water bath at 70 °C for 30 min. After that, the sample was filtered on filter paper and 100 ml of ethanol (75% or 70%) were added to the residue, and another alcoholic extraction was performed. The solution obtained from the two extractions were dried and stored (Borges et al., 2014Borges, E.C., Da Silva, L.C., De Alencar, S.M., De Aguiar, C.L., 2014. Caracterização química de extratos etanólicos de própolis com atividade inibitória do crescimento de estafilococos isolados de mastite bovina. Rev. Bras. Tecnol. Agroindustr. 8, 1040-1153.).
Preparation of ethanolic extracts of red propolis (EEP-70% or 75% the room temperature)
The lyophilized red propolis sample (20 g) was extracted with 70% or 75% ethanol (15 ml of ethanol for each 2 g) for 48 h at room temperature and the resulting alcoholics extracts were filtered under vacuum on filter paper and 100 ml of ethanol (75% or 70%) were added to the residue, and another alcoholic extraction was made. The solution obtained from the two extractions were dried and stored (Nascimento et al., 2009Nascimento, C.S., Nunes, L.C.C., De Lima, A.A.N., Júnior, S.G., Neto, P.J., 2009. Incremento do SPF em formulação de protetor solar utilizando extratos de própolis verde e vermelha. Rev. Bras. Farm. 90, 334-339.).
Incorporation of red propolis extracts (EPP) in the filter UVA-UVB 5% Gel Permulem TR-1
Filter UVA-UVB 5% Gel Permulem TR-1 (1 g) and 1 ml of EEP solution (1 mg/ml) were added to a 100 ml beaker. The mixture was maintained under stirring for 30 min at room temperature. After that, the sample was stored (Nascimento et al., 2009Nascimento, C.S., Nunes, L.C.C., De Lima, A.A.N., Júnior, S.G., Neto, P.J., 2009. Incremento do SPF em formulação de protetor solar utilizando extratos de própolis verde e vermelha. Rev. Bras. Farm. 90, 334-339.).
Determination of maximum absorbance of EEP, filter UVA-UVB 5% Gel Permulem Tri and filter UVA-UVB 5% Gel Permulem TR-1 incorporated with EEP
The absorption readings of the ultraviolet UV–Vis by the samples were performed using the UV Spectrophotometer Femto800 Xi. For determination of absorbance in the ultraviolet regions UVA and UVB 10 mg of samples were diluted in 10 ml ethanol 70%, producing a concentration of 1 mg/ml that was diluted in ethanol 70% and producing diluted concentrations of 0.010, 0.020, 0.030, 0.050, 0.070 and 0.1 mg/ml. The scanning was performed for each concentration between the wavelengths of 200 and 600 nm in the UV spectrophotometer, using quartz bucket with an optical way of 1 cm. Ethanol 70% was used as white and the experiment was carried out in triplicate (Gonçalves et al., 2018Gonçalves, M.C., Rossoni Junior, J.V., Rabelo, A.C.S., Costa, D.C., Cazati, T., Taylor, J.G., Dos Santos, V.M.R., 2018. Estudos preliminares da citoxicidade e propriedades fotoprotetoras de derivados de benzofenonas e lactonas. Rev. Virtual Quim. 10, 600-608.).
In vitro determination of the Sun Protection Factor (SPF)
UVA-UVB 5% Gel Permulem TR-1 Filter (1 g) incorporated with EEP was weighed and the dilutions were performed in ethanol 70% (triplicate), until obtaining a concentration of 0.2 µl/ml. Also, separately, 1 mg/ml of each EEP and 1 mg/ml Filter UVA-UVB 5% Gel Permulem TR-1 were prepared and the dilutions were in ethanol 70% (triplicate), until obtaining concentrations of 0.010, 0.020, 0.030, 0.050, 0.070 and 0.1 mg/ml. The in vitro Solar Protection Factor was determined to each concentration by the spectrophotometric method developed by Mansur (1984) using Eq. (1).
where FC = 10 (constant), EE = erythemogenic effect, I = intensity of the sun and Abs = absorbance of the sample. Absorption readings were performed in the range of 290–320 nm with intervals of 5 nm and added in Eq. (1). The constants EE and I were pre-defined by Mansur (1984), according to Table 1.
Evaluation of photostability
The photostability test was performed using a light chamber with a UV lamp at 365 nm wavelength. Solutions of 0.1 mg/ml EPP, Filter UVA-UVB 5% Gel Permulem TR-1 and Filter UVA-UVB 5% Gel Permulem Tri incorporated with EEP were prepared in volumetric flasks, which were exposed to radiation for 2 h, evaluating the effective concentration every 30 min upon exposure to UV radiation.
Evaluation of antioxidant activity
The antioxidant activity was evaluated by photo-colorimetry in vitro methods using the free radicals 2,2-diphenyl-1-picrylhydrazyl (DPPH) (Sigma) and 2,2′-azinobis-3-ethylbenzotiazoline-6-sulfonic acid (ABTS) (Sigma).
For the DPPH method, the EEP and the quercetin (standard) were solubilized in ethanol to obtain stock solutions of 320.0 µg/ml. Different aliquots were pipetted to give final solutions from 0.5 to 64 µg/ml. The Filter UVA-UVB 5% Gel Permulem TR-1 and Filter UVA-UVB 5% Gel Permulem Tri incorporated with EEP were solubilized to give final solutions of × µl/ml. Then 100 µl of DPPH solution at 0.008% w/v in ethanol were added to each of these samples. The final volume was adjusted to 240 µl with ethanol. The negative control was obtained from 100 µl of the DPPH and 140 µl of the ethanol, which was used to calculate the inhibition percentage of free radical. Then, all the samples were incubated for 30 min at room temperature (25 ± 2 °C) protected from light and the absorbance (Abs) were read at a wavelength of 490 nm in a plate-reader. The test was performed in triplicate and the ability to scavenge free radicals was evaluated by the scavenging percentage of free radical (%I), calculated using the formula (Sousa et al., 2007):
The concentration required to obtain a 50% antioxidant effect (EC50) was calculated by linear regression for EEP and quercetin.
For the ABTS method, the working solution was obtained by mixing ABTS (7.4 mmol/l) with potassium persulfate (2.6 mmol/l) and kept for 16 h at room temperature protected from light. On the day of analysis, this solution was diluted with ethanol to an absorbance of 0.70 (±0.02) at 650 nm. The concentrations from EEP, quercetin and gels were obtained in the same manner as the previous test. Then 120 µl of ABTS were added to these samples. The final volume was adjusted to 150 µl with addition of ethanol. The negative control was obtained from 120 µl of the ABTS and 30 µl of the ethanol. Then, all the samples were incubated for 6 min at room temperature (25 ± 2 °C) protected from light (Li et al., 2009). The readings were performed at 650 nm and the % scavenging of the sample and EC50 was calculated like previously described.
Determination of total phenolic and flavonoid content
The measurement of total phenolic was made by the Folin-Ciocauteu method according Bonoli et al. (2004), with modifications. The extracts (80 µl) and fractions (5 mg/ml in ethanol 95%) were transferred to a 96-well plate and were added 60 µl of water and 10 µl of Folin-Ciocauteu (Cromoline). Following the plate was agitated for 1 min and were added 40 µl of sodium carbonate solution (15% w/v). The plate was agitated for 30 s, were added 10 µl of water and after incubation for 2 h the absorbance was read at 650 nm in microplate reader (Molecular Devices). The total phenolic were quantified by using a standard calibration curve of gallic acid (10–320 µg/ml; r2 = 0.9983; y = 0.0046x + 0.0886). The experiment was made in triplicate and the results were expressed as mg of gallic acid equivalents (GAE) per g of sample (mgGAE/g) and as percentage (w/w).
The measurement of total flavonoids was made by the aluminum chloride (AlCl3) colorimetric method according Chang et al. (2002), with modifications. The extracts (100 µl) and fractions (5 mg/ml in ethanol 95%) were transferred to a 96-well plate and were added 40 µl of ethanol 95%, 4 µl of AlCl3 (10% w/v), 4 µl of potassium acetate (1 mol/l) and 52 µl of water. The absorbance was read after incubation for 40 min in microplate reader (Molecular Devices) at 405 nm. The total flavonoids were quantified by using a standard calibration curve of quercetin (2–64 µg/ml; r2 = 0.9982; y = 0.0017x + 0.0418). The experiment was made in triplicate and the results were expressed as mg of quercetin equivalents (QE) per g of sample (mgQE/g) and as percentage (w/w).
HET-CAM test
HET-CAM test was performed according to a protocol suggested by Luepke, 1985Luepke, N.P., 1985. Hen's egg chorioallantoic membrane test for irritation potential. Chem. Toxic. 23, 287-291. for UVA-UVB 5% Gel Permulem TR-1 Filter incorporated or not with EEP 75% (room temperature). The assay bases the analysis of the appearance of irritative reactions in the chorioallantoic membrane of the fertilized chicken eggs in response to exposure of tested substances. For the procedure was obtained commercially fertile White Leghorn chicken eggs (Granja Tomolei, RJ, Brazil) without mycoplasms and used after the 9th day of incubation. After having been controlled for embryo viability are opened near the air cell using a pair of surgical scissors to reveal the highly vascularized chorioallantoic membrane (CAM). After the product application (0.1 mg), the CAM surface was observed over a period of 300 s and the time of the appearance of hemorrhage, lysis and coagulation occurring on the vascular system and the albumin was recorded. The positive irritant control was 0.1 N sodium hydroxide and the negative control was 0.9% sodium chloride. For each sample three eggs were used.
The irritant effects were classified by scores according to the time they were observed: less than 30 s (hyperemia: 5; hemorrhage: 7; clot formation/opacity: 9); between 30 and 120 s (hyperemia: 3; hemorrhage: 5; clot formation/opacity: 7); or between 120 and 300 s (hyperemia: 1; hemorrhage: 3; clotformation/opacity: 5). If an effect was not observed after 300 s, it was scored as zero. Each formulation was classified according to the scores mean value of three eggs: 0–4.99 corresponding to non-irritant/slightly irritant (NI/SI); 5.00–8.99 corresponding to moderately irritant (MI); and 9.00–21.00 corresponding to severely irritant (SVI) (Mansur et al., 2016Mansur, M.C.P.P.R., Leitão, S.G., Cerqueira-Coutinho, C., Vermelho, A.B., Silva, R.S., Presgrave, O.A.F., Leitão, A.A.C., Leitão, G.G., Ricci-Júnior, E., Santos, E.P., 2016. In vitro and in vivo evaluation of efficacy and safety of photoprotective formulations containing antioxidant extracts. Rev. Bras. Farmacogn. 26, 251-258.).
Statistical analysis
The results were statistically analyzed by the One-Way ANOVA test and multiple comparison by Tukey considering a significance level of 0.05 (p < 0.05). Pearson test was used to correlate phenolic and flavonoid content with SPF and antioxidant results. All statistical analysis were made using GraphPad Prism 5.0 software.
Results and discussion
Determination of absorbance maximum of EEP, UVA-UVB 5% Gel Permulem TR-1 Filter and UVA-UVB 5% Gel Permulem Tri Filter incorporated with EEP
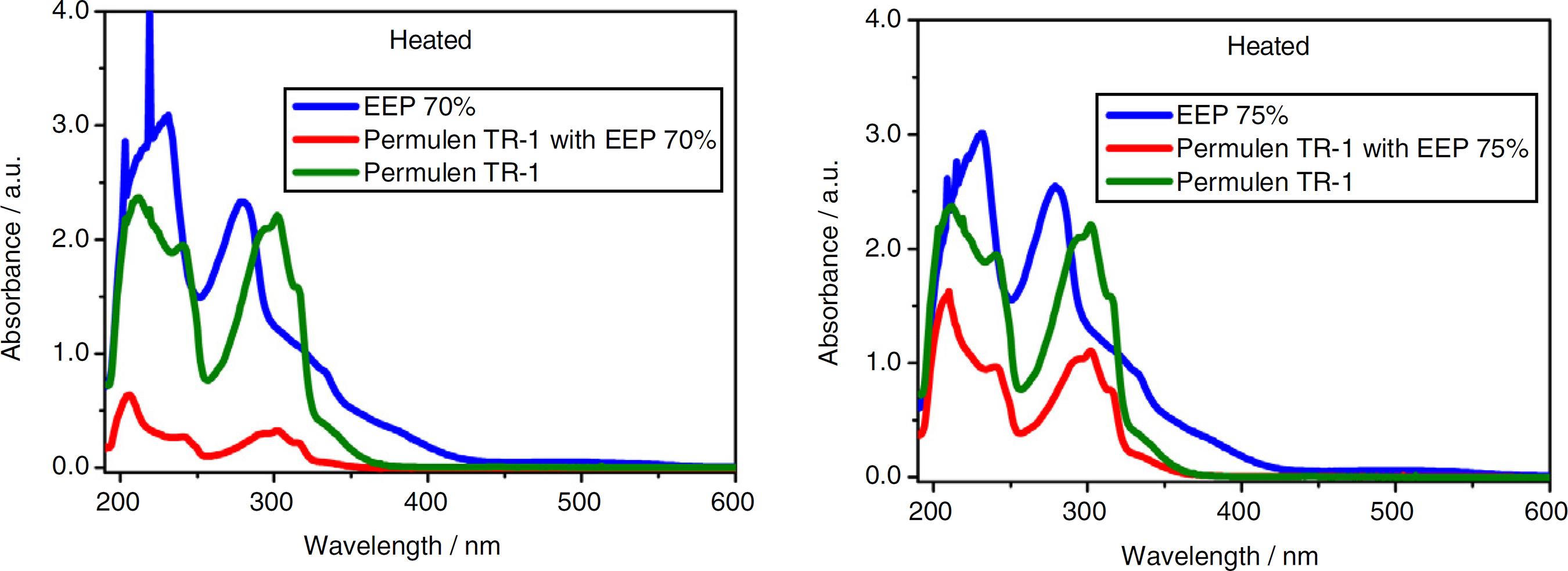
The EPP incorporated in the Filter UVA-UVB 5% Gel Permulem TR-1 caused a bathocromic displacement. The UV bands of the mixtures EPP (70% or 75% in the room temperature and heated) with Filter moved to a longer wavelenght, confirming the incorporation (Figs. 1 and 2).
UV/Vis absorption spectra of EEP (70% and 75%), EEP (70% and 75%) incorporated in Permulen TR-1 and Permulen TR-1 Filter, prepared at room temperature.
UV/Vis absorption spectra of heated EEP (70% and 75%), EEP (70% and 75%) incorporated in Permulen TR-1 and Permulen TR-1 Filter.
In vitro determination of the Sun Protection Factor (SPF)
The SPF was evaluated by the methodology developed by Mansur method. The analysis was carried out on an ultraviolet spectrophotometer, where the values of the obtained absorbances were placed in Eq. (1), providing the data of SPF values, presented in the Table 2. UVA-UVB 5% Gel Permulem TR-1 Filter has been used as positive control in previous studies. According to Table 2, the EEPs showed similar SPF than positive control, without statistical difference.
SPF values (mean ± SD) calculated from EEP and Positive Control (UVA-UVB 5% Gel Permulem Filter) in different concentrations.
The UVA-UVB 5% Gel Permulem TR-1 Filter at a concentration of 0.20 µl/ml presented an SPF of 10.22 (Table 3). The EEP 70% and 75% (room temperature) with Filter UVA-UVB 5% Gel Permulem TR-1 showed that there was a synergism between the formulations. Extracts of red propolis when incorporated with the filter showed more significant UV absorption values. The results obtained with the incorporation of EPP (room temperature) show that these mixtures cause a intensification in the SPF through the synergistic effect of the Filter (Table 3). In general, the incorporation of the EPP 70% and 75% (room temperature) on the filter show that these mixtures promote an intensification of the SPF through the synergistic effect of the extracts with the synthetic filter employed, ensuring greater sun protection.
Evaluation of photostability
EEP, UVA-UVB 5% Gel Permulem TR-1 Filter and EEP with UVA-UVB 5% Gel Permulem TR-1 Filter were analyzed separately in the photostability assay. The samples were dissolved in ethanol, forming solutions of concentration equal to 0.10 mg/ml. According Fig. 3, the graphs show how these solutions behave, in terms of photostability, after exposure to UV radiation at 365 nm wavelength. The samples were photostable in the UV radiation at 365 nm wavelength in the time of 2 h.
Graphs of photostability of the EEP (70% or 75%) and Permulem TR-1incorporated with EEP(70% or 75%), prepared at room temperature and heated. The inserts show the intensity of absorption versus exposure time to UV radiation.
Evaluation of antioxidant activity
Results of antioxidant activity showed that EPP had similar capacity of radicals scavenging (EC50%) (Table 4). Filter UVA-UVB 5% Gel Permulem TR-1 showed a weak antioxidant activity (low inhibition percentage %I) (Table 5). This result can be due to the lack of antioxidant activity of one of the both sunscreen agents present in this filter gel, the 2-phenyl-1H-benzimidazole-5-sulfonic acid (Bino et al., 2017Bino, A., Baldisserotto, A., Scalambra, E., Dissette, V., Vedaldi, D.E., Salvador, A., Durini, E., Manfredini, S., Vertuani, S., 2017. Design, synthesis and biological evaluation of novel hydroxy-phenyl-1H-benzimidazoles as radical scavengers and UV-protective agents. J. Enzyme Inhib. Med. Chem. 32, 527-537.). However, the results obtained with the incorporation of EPP with Filter UVA-UVB 5% Gel Permulem TR-1 showed that these extracts induced intensification in the antioxidant activity of this formulation (Table 5).
The addition of antioxidants to commercial sunscreens can prevent the damages caused by the oxidative stress induced by UVA radiation through reactive oxygen species (ROS) that leads to DNA lesions (Matsui et al., 2009Matsui, M.S., Hsia, A., Miller, J.D., Hanneman, K., Scull, H., Cooper, K.D., Baron, E., 2009. Non-sunscreen photoprotection: antioxidants add value to a sunscreen. J. Investig. Dermatol. Symp. Proc. 14, 56-59.). Extracts from medicinal plants due its antioxidant activity and SPF may provide new possibilities for the treatment and prevention of UV-mediated diseases (Ebrahimzadeh et al., 2014Ebrahimzadeh, M.A., Enayatifard, R., Khalili, M., Ghaffarloo, M., Saeedi, M., Yazdani, J.C., 2014. Correlation between sun protection factor and antioxidant activity, phenol and flavonoid contents of some medicinal plants. Iran. J. Pharm. Res. 13, 1041-1047.).
Determination of total phenolic and flavonoid content
In Brazilian red propolis samples, phenolic compounds have already been identified as tannins, catechins, flavonones and flavonols (Mendonça et al., 2015Mendonça, I.C., Porto, I.C., do Nascimento, T.G., de Souza, N.S., Oliveira, J.M., Arruda, R.E., Mousinho, K.C., dos Santos, A.F., Basílio-Júnior, I.D., Parolia, A., Barreto, F.S., 2015. Brazilian red propolis: phytochemical screening, antioxidant activity and effect against cancer cells. BMC Complement. Altern. Med. 15, http://dx.doi.org/10.1186/s12906-015-0888-9.
http://dx.doi.org/10.1186/s12906-015-088...
). Several studies demonstrated that phenolic acids and flavonoids derived from propolis, tea, grape, fern, and milk thistle provide photoprotection. These compounds can prevent penetration of radiation into the skin, resulting in the reduction of inflammation, oxidative stress, and DNA damaging effects (Nichols and Katiyar, 2010Nichols, J.A., Katiyar, S.K., 2010. Skin photoprotection by natural polyphenols: antiinflammatory, antioxidant and DNA repair mechanisms. Arch. Dermatol. Res. 302, 71-83.). Phenolic compounds also are important antioxidants and the elimination of free radicals is among the mechanisms related to this pharmacological action (Soobrattee et al., 2005Soobrattee, M.A., Neergheen, V.S., Luximon-Ramma, A., Aruoma, O.I., Bahorun, T., 2005. Phenolics as potential antioxidant therapeutic agents: mechanism and actions. Mutat. Res. 579, 200-213.). Because of this, it is reasonable to compare total phenolic and flavonoid content of the different extracts studied. Therefore, the content of both groups of phenolics was also determined in the extracts (Table 6).
EEP 75% hot presented a lower quantity of phenolic and flavonoids in relation to the others extracts. The minimum requirements according to the Brazilian regulation of identify and quality of propolis are 5% (w/w) for phenolic compounds and 0.5% (w/w) for flavonoids (Ministério da Agricultura, 2001Ministério da Agricultura, Pecuária e do Abastecimento, 2001. Instrução Normativa n. 3, de 19 de janeiro de 2001. Regulamentos técnicos de identidade e qualidade de apitoxina, cera de abelha, geléia real, geléia real liofilizada, pólen apícola, própolis e extrato de própolis. Diário Oficial da União, Brasília, 23 Janeiro, Seção I, pp. 18–23.). Therefore, between the extracts only EEP 75% is below that required by Brazilian regulation.
The total phenolic content have strong correlation (0.7 ≤ r2 ≤ 1.0) with the antioxidant results and moderate correlation (0.5 ≤ r2 < 0.7) with the SPF results (Table 7). In the other way, the total flavonoid content have weak correlation (0.0 ≤ r2 < 0.5) with the SPF results (Table 7). Thus, the activities are better related to total phenolic content that flavonoid.
HET-CAM test
The results of total phenolic content showed good correlation with antioxidant and SPF results, thus the toxicity test HET-CAM was performed to Filter UVA-UVB 5% Gel Permulem TR-1 incorporated or not with EEP 75% (room temperature), which presented the biggest total phenolic content among EEP (Table 6). HET-CAM results showed none alterations in the membrane when in contact with Filter UVA-UVB 5% Gel Permulem TR-1, but the Filter Gel incorporated with EEP 75% (room temperature) induced a pontual slight hyperemia (Fig. 4). According to the classification by scores, both formulations were classified as non-irritant/slightly irritant (NI/SI), suggesting that they are safe to be applied on the skin (Table 8). HET-CAM alterations is based upon vascular effects on the CAM, the non observation of these effects may suggest that formulation possess also a lower skin irritation potential, since skin irritation starts with vascular alterations and the results can be associated to cosmetic application on the face, near to the ocular mucous membrane (Mansur et al., 2016Mansur, M.C.P.P.R., Leitão, S.G., Cerqueira-Coutinho, C., Vermelho, A.B., Silva, R.S., Presgrave, O.A.F., Leitão, A.A.C., Leitão, G.G., Ricci-Júnior, E., Santos, E.P., 2016. In vitro and in vivo evaluation of efficacy and safety of photoprotective formulations containing antioxidant extracts. Rev. Bras. Farmacogn. 26, 251-258.).
Photographs of HET-CAM test results of Permulem TR-1incorporated or not with EEP 75%, prepared at room temperature. The arrows indicate the alterations.
Time for reaction (s) in HET-CAM test of Filter UVA-UVB 5% Gel Permulem Tri incorporated or not with EEP.
Conclusion
The present study demonstrated the importance and interest of using red propolis extracts in sunscreen preparations with incorporated UVA-UVB 5% Gel TR-1 Filter, since it was observed a relative increase of SPF and antioxidant activity, leading to greater skin protection. The results obtained by UV spectrophotometry allow a better evaluation of the efficacy of EPP 75% (room temperature) as sunscreens when associated with a photoprotective formulation. The antioxidant activity and SPF are correlated with total phenolics content of the extracts and the EEP 75% at room temperature was chosen. The formulation developed with Filter UVA-UVB 5% Gel Permulem with this EEP showed safe to be applied on the skin according HET-CAM test. It can be concluded in the experimental conditions of this study that EPP (75% - room temperature) associated to the photoprotective formulation showed an increase in the activity of the formulation, which suggests its indication for use in photoprotective products.
Acknowledgments
The authors thank Chemistry Department, Institute of Exact and Biological Sciences, Federal University of Ouro Preto. The authors also thanks the financial support from FAPEMIG and PROPP/UFOP (# 23109.003517/2018-85).
References
- Bino, A., Baldisserotto, A., Scalambra, E., Dissette, V., Vedaldi, D.E., Salvador, A., Durini, E., Manfredini, S., Vertuani, S., 2017. Design, synthesis and biological evaluation of novel hydroxy-phenyl-1H-benzimidazoles as radical scavengers and UV-protective agents. J. Enzyme Inhib. Med. Chem. 32, 527-537.
- Borges, E.C., Da Silva, L.C., De Alencar, S.M., De Aguiar, C.L., 2014. Caracterização química de extratos etanólicos de própolis com atividade inibitória do crescimento de estafilococos isolados de mastite bovina. Rev. Bras. Tecnol. Agroindustr. 8, 1040-1153.
- Castro, L.M., Do Nascimento, A.M., Ikegaki, M., Costa-Neto, C.M., Alencar, S.M., Rosalen, P.L., 2009. Identification of a bioactive compound isolated from Brazilian propolis type 6. Bioorg. Med. Chem. 17, 5332-5335.
- Ebrahimzadeh, M.A., Enayatifard, R., Khalili, M., Ghaffarloo, M., Saeedi, M., Yazdani, J.C., 2014. Correlation between sun protection factor and antioxidant activity, phenol and flavonoid contents of some medicinal plants. Iran. J. Pharm. Res. 13, 1041-1047.
- Gonçalves, M.C., Rossoni Junior, J.V., Rabelo, A.C.S., Costa, D.C., Cazati, T., Taylor, J.G., Dos Santos, V.M.R., 2018. Estudos preliminares da citoxicidade e propriedades fotoprotetoras de derivados de benzofenonas e lactonas. Rev. Virtual Quim. 10, 600-608.
- Luepke, N.P., 1985. Hen's egg chorioallantoic membrane test for irritation potential. Chem. Toxic. 23, 287-291.
- Mansur, M.C.P.P.R., Leitão, S.G., Cerqueira-Coutinho, C., Vermelho, A.B., Silva, R.S., Presgrave, O.A.F., Leitão, A.A.C., Leitão, G.G., Ricci-Júnior, E., Santos, E.P., 2016. In vitro and in vivo evaluation of efficacy and safety of photoprotective formulations containing antioxidant extracts. Rev. Bras. Farmacogn. 26, 251-258.
- Matsui, M.S., Hsia, A., Miller, J.D., Hanneman, K., Scull, H., Cooper, K.D., Baron, E., 2009. Non-sunscreen photoprotection: antioxidants add value to a sunscreen. J. Investig. Dermatol. Symp. Proc. 14, 56-59.
- Mendonça, I.C., Porto, I.C., do Nascimento, T.G., de Souza, N.S., Oliveira, J.M., Arruda, R.E., Mousinho, K.C., dos Santos, A.F., Basílio-Júnior, I.D., Parolia, A., Barreto, F.S., 2015. Brazilian red propolis: phytochemical screening, antioxidant activity and effect against cancer cells. BMC Complement. Altern. Med. 15, http://dx.doi.org/10.1186/s12906-015-0888-9
» http://dx.doi.org/10.1186/s12906-015-0888-9 - Ministério da Agricultura, Pecuária e do Abastecimento, 2001. Instrução Normativa n. 3, de 19 de janeiro de 2001. Regulamentos técnicos de identidade e qualidade de apitoxina, cera de abelha, geléia real, geléia real liofilizada, pólen apícola, própolis e extrato de própolis. Diário Oficial da União, Brasília, 23 Janeiro, Seção I, pp. 18–23.
- Nascimento, C.S., Nunes, L.C.C., De Lima, A.A.N., Júnior, S.G., Neto, P.J., 2009. Incremento do SPF em formulação de protetor solar utilizando extratos de própolis verde e vermelha. Rev. Bras. Farm. 90, 334-339.
- Nichols, J.A., Katiyar, S.K., 2010. Skin photoprotection by natural polyphenols: antiinflammatory, antioxidant and DNA repair mechanisms. Arch. Dermatol. Res. 302, 71-83.
- Soobrattee, M.A., Neergheen, V.S., Luximon-Ramma, A., Aruoma, O.I., Bahorun, T., 2005. Phenolics as potential antioxidant therapeutic agents: mechanism and actions. Mutat. Res. 579, 200-213.
Publication Dates
-
Publication in this collection
26 Aug 2019 -
Date of issue
Mar-Apr 2019
History
-
Received
18 Sept 2018 -
Accepted
7 Mar 2019 -
Published
20 Mar 2019