Abstracts
Objective:
To evaluate immediate and long-term results of cardiac transplantation at two different levels of urgency.
Methods:
From November 2003 to December 2012, 228 patients underwent cardiac transplantation. Children and patients in cardiogenic shock were excluded from the study. From the final group (n=212), 58 patients (27%) were hospitalized under inotropic support (Group A), while 154 (73%) were awaiting transplantation at home (Group B). Patients in Group A were younger (52.0±11.3 vs. 55.2±10.4 years, P=0.050) and had shorter waiting times (29.4±43.8 vs. 48.8±45.2 days; P=0.006). No difference was found for sex or other comorbidities. Haemoglobin was lower and creatinine higher in Group A. The characteristics of the donors were similar. Follow-up was 4.5±2.7 years.
Results:
No differences were found in time of ischemia (89.1±37.0 vs. 91.5±34.5 min, P=0.660) or inotropic support (13.8% vs. 11.0%, P=0.579), neither in the incidence of cellular or humoral rejection and of cardiac allograft vasculopathy. De novo diabetes de novo in the first year was slightly higher in Group A (15.5% vs. 11.7%, P=0.456), and these patients were at increased risk of serious infection (22.4% vs. 12.3%, P=0.068). Hospital mortality was similar (3.4% vs. 4.5%, P=0.724), as well as long-term survival (7.8±0.5 vs. 7.4±0.3 years).
Conclusions:
The results obtained in patients hospitalized under inotropic support were similar to those of patients awaiting transplantation at home. Allocation of donors to the first group does not seem to compromise the benefit of transplantation. These results may not be extensible to more critical patients.
Heart Transplantation; Thoracic Surgery; Postoperative Complications; Survival (Public Health)
Objetivo:
Avaliar os resultados imediatos e de longo prazo do transplante cardíaco em dois níveis diferentes de urgência.
Métodos:
De novembro de 2003 a dezembro de 2012, 228 pacientes foram submetidos a transplante cardíaco. Crianças e os pacientes em choque cardiogênico foram excluídos do estudo. Do grupo final (n=212), 58 pacientes (27%) estavam hospitalizados e em suporte inotrópico (Grupo A), enquanto 154 (73%) aguardavam transplante em casa (Grupo B). Os pacientes do Grupo A eram mais jovens (52,0±11,3 vs. 55,2±10,4 anos, P=0,050) e tinham menor tempo de espera (29,4±43,8 vs. 48,8±45,2 dias, P=0,006). Não foram encontradas diferenças entre os sexos ou outras comorbidades. Níveis de hemoglobina foram menores e de creatinina superiores no Grupo A. As características dos doadores foram semelhantes. O acompanhamento foi de 4,5±2,7 anos.
Resultados:
Não foram observadas diferenças no tempo de isquemia (89,1 ± 37,0 vs. 91,5 ± 34,5 min, P=0,660) ou no suporte inotrópico (13,8% vs. 11,0%, P=0,579), nem na incidência de rejeição celular ou humoral e de vasculopatia do enxerto. Incidência de diabetes de novo no início do primeiro ano foi um pouco maior no Grupo A (15,5% vs. 11,7%, P=0,456), e esses pacientes apresentaram maior risco de infecção grave (22,4% vs. 12,3%, P=0,068). A mortalidade hospitalar foi semelhante (3,4% vs. 4,5%, P=0,724), bem como a sobrevida a longo prazo (7,8±0,5 vs. 7,4±0,3 anos).
Conclusões:
Os resultados obtidos em pacientes hospitalizados em suporte inotrópico foram semelhantes aos de pacientes que aguardam o transplante em casa. Alocação de doadores para o primeiro grupo não parece comprometer o benefício do transplante. Esses resultados podem não ser estendidos aos pacientes mais críticos.
Transplante de Coração; Cirurgia Torácica; Complicações Pós-Operatórias; Sobrevida
INTRODUCTION
Despite advances in medical and surgical treatment of heart failure, heart
transplantation is still the elective treatment for patients refractory to other medical
therapies or conventional cardiac procedures. The criteria for selection of candidates
for transplantation have been gradually extended and the selection of donors and
recipients is essential to the success of a transplantation program[11 Stehlik J, Edwards LB, Kucheryavaya AY, Aurora P, Christie JD, Kirk R,
et al. The Registry of the International Society for Heart and Lung Transplantation:
twenty-seventh official adult heart transplant report-2010. J Heart Lung Transplant.
2010;29(10):1089-103.
2 Russo MJ, Chen JM, Hong KN, Stewart AS, Ascheim DD, Argenziano M, et al;
Columbia University Heart Transplant Outcomes Research Group. Survival after heart
transplantation is not diminished among recipients with uncomplicated diabetes
mellitus: an analysis of the United Network of Organ Sharing database. Circulation.
2006;114(21):2280-7.-33 McMurray JJ, Adamopoulos S, Anker SD, Auricchio A, Böhm M, Dickstein K,
et al; Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure
2012 of the European Society of Cardiology; ESC Committee for Practice Guidelines.
ESC guidelines for the diagnosis and treatment of acute and chronic heart failure
2012: The Task Force for the Diagnosis and Treatment of Acute and Chronic Heart
Failure 2012 of the European Society of Cardiology. Developed in collaboration with
the Heart Failure Association (HFA) of the ESC. Eur J Heart Fail.
2012;14(8):803-69.].
However, a significant reduction in supply has forced the spectrum of selection of donors to be broadned. Thus, the profile has changed from a young donor who died of cranio-cerebral trauma to that of an older donor whose death resulted from primary neurological disease. The latter is often accompanied by other types of diseases and cardiovascular risk factors, conditions which determine the choice of the recipient from a scarce and rarely ideal donor.
As a result, we have seen an increase in the number of patients in advanced stages of the disease at the time of transplantation, who are admitted for decompensated heart failure requiring intensive pharmacologic support and, at times, cardio-circulatory and mechanical respiratory support in those with worse clinical status, a situation that increases the urgency for transplantation, generally associated with greater risk of failure.
On a national level, the criteria/priority for heart transplantation of the Portuguese
Society of Transplantation (unpublished data; Table
1) include seven degrees in descending order of priority. As a result of this
and other types of stratification, the most critical patient passes ahead of others
whose clinical condition allows them to wait longer. However, these criteria do not
explain or quantify the risk profile. We know that the immediate results of cardiac
transplantation are very sensitive not only to the quality of the donor organ but also
to the clinical status of the candidate to transplantation[44 Fiorelli Al, Branco JN, Dinkhuysen JJ, Oliveira Junior JL, Pereira TV,
Dinardi LF, et al. Risk factor analysis of late survival after heart transplantation
according to donor profile: a multi-institutional retrospective study of 512
transplants. Transplant Proc. 2012;44(8):2469-72.
5 Forni A, Luciani GB, Chiominto B, Pizzuti M, Mazzucco A, Faggian G.
Results with expanded donor acceptance criteria in heart transplantation. Tranplant
Proc. 2011;43(4):953-9.-66 Holman WL, Kormos RL, Naftel DC, Miller MA, Pagani FD, Blume E, et al.
Predictors of death and transplant in patients with a mechanical circulatory support
device: a multi-institutional study. J Heart Lung Transplant.
2009;28(1):44-50.].
Strictly following the scale of priority or urgency for transplantation, we end up transplanting sicker hospitalized patients undergoing intensive treatment for decompensated heart failure, always accompanied by variable multiorgan function deterioration. This will result in a natural increase of morbidity and mortality after transplantation, triggering the question of who should be transplanted (?), which can raise probably insoluble questions of ethics in organ distribution.
In this study, we analyzed and compared the early and late results of transplantation following the criteria of priority in use by the Portuguese Society of Transplantation (SPT), in basic clinical aspects similar to the international scale INTERMACS (Interagency Registry for Mechanically Assisted Circulatory Support)[77 Starling RC, Naka Y, Boyle AJ, Gonzalez-Stawinski G, John R, Jorde U, et al. Results of the post-U.S. Food and Drug Administration-approval study with a continuous flow left ventricular assist device as a bridge to heart transplantation: a prospective study using the INTERMACS (Interagency Registry for Mechanically Assisted Circulatory Support). J Am Coll Cardiol. 2011;57(19):1890-8,88 Stevenson WL, Pagani FD, Young JB, Jessup M, Miller L, Kormos RL, et al. INTERMACS profiles of advanced heart failure: the current picture. J Heart Lung Transplant. 2009;28(6):535-41.].
METHODS
Origin, definition and collection of data
In the period from November 2003 to December 2012, 228 patients were consecutively submitted to heart transplantation at this Centre. Exclusion criteria for this study included patients previously transplanted with other organs (n=2), patients under 18 years of age (n=8), and patients with emergency priority (≤ grade IV, n=8). Thus, the study population included 212 patients, 58 of whom were in the ICU, under inotropic support without respiratory and/or mechanical ventricular assistance (SPT grade V; Group A), and 154 were at home on a waiting list with two or more hospitalization episodes in the ICU in the last 6 months (SPT grade VI; Group B).
The data were obtained from a national database, especially designed for the prospective registration (online platform) of data from the recipient, donor, surgery, immunosuppression protocol and follow-up of patients undergoing cardiac transplantation.
All surviving patients were followed via regular consultations at the Surgical Centre by a medical/surgical team, from one to 10 years, and no patient was lost to follow-up. The mean follow-up in this study was 4.5±2.7 years (total, 961.8 patient-years).
Recipients and Donors
Table 2 shows data from pre-operative variables of the recipients in groups A and B, as well as the results of the comparative analysis. When compared with the population of Group B, patients in Group A were younger (P=0.05), had lower body mass index (BMI; P=0.005), had significantly lower serum haemoglobin (P=0.001), higher total bilirubin (P=0.014) and creatinine (P=0.01), and shorter time on the waiting list (P=0.006).
Table 3 details the characteristics of the donors to patients of groups A and B, as well as the results of the comparative analysis, which showed no significant differences in any of the variables. However, male donors were the most common (82% vs. 73%). The cause of death was mostly traumatic (59.6% vs. 57.6%), but there was a growing trend in the number of donors dying from CVA, now approaching two thirds. In group A, there was a significant number of donors with prolonged inotropic or ventilator support (longer than one week, 12%).
Statistical analysis
Continuous variables are presented as mean ± standard deviation and evaluated using independent (comparison between groups) and paired (over time) Student's t-test. Categorical variables are reported as frequency and percentage, and compared using the chi-square test. Survival and event-free survival were calculated using the Kaplan -Meier method. Statistical significance was defined as P-value <0.05. Data were analyzed using the IBM SPSS Statistics for Windows program (Version 20.0. Armonk, NY: IBM Corp; released 2011).
RESULTS
Surgery
Table 4 shows the data for variables related to surgery in groups A and B, as well as the results of the comparative analysis. Patients in Group B had a significantly higher incidence of mechanical circulatory support (P=0.044). Of the ten patients who needed this kind of assistance, 2 required only intra-aortic balloon pump (IABP), 3 required the use of a left ventricular assist device, and 5 required total circulatory support with ECMO. No statistically significant differences were observed in relation to other variables, namely times of ischemia and CPB, mechanical ventilation and inotropic support >48 hours.
Cellular and humoral rejection and graft vascular disease
Table 5 shows the data on acute cellular rejection, humoral rejection and graft vascular disease (GVD) in groups A and B. Ninety patients (42.5%) had no episodes of acute cellular rejection (grade 0R of the ISHLT). The remaining patients (n=122, 57.5%) had at least one episode of rejection; however, in 83 of them it was only mild (1R), requiring no treatment. The results of the comparative analysis of the two groups showed no statistically significant difference in any of the cellular rejection grades. Thirty nine patients (18.4%) had at least one episode of cellular rejection grade ≥ 2R, the majority (33, 84.6%) occurring during the first year. No statistically significant difference (P=0.309) was observed in the comparative analysis of survival free from cellular rejection grade ≥2R between groups A and B (Figure 1A).
A - Survival free from cellular rejection grade ≥2R in groups A and B. 1B - Survival free from graft vascular disease in groups A and B.
Humoral rejection was diagnosed in six patients (2.8%), with no significant difference between the study groups (P=0.551).
Graft vascular disease (diagnosed by coronary angiography, as some degree of "new" irregularity or stenosis in any of the major coronary vessels or its main branches, even if less than 50%) was diagnosed in 20 patients (9.4%). The comparative analysis of this event showed no significant difference in the incidence between the study groups (P=0.781). Survival free from GVD in groups A and B was not statistically different (P=0.547, Figure 1B).
De novo diabetes and severe infections
The incidence of de novo diabetes in the first year after transplantation was 12.7%, with no difference between the two groups (15.5% vs. 11.7%, P=0.456).
The incidence of serious infections requiring hospitalization and intravenous antibiotics during the first year post-transplantation was higher in Group A (22.4% vs. 12.3%), but the difference did not reach statistical significance (P=0.068). Likewise, the incidence of pneumonia in the first 6 months was higher in Group A (17.2% vs. 9.7%), although the difference was not significant (P=0.131).
Behavior of renal function after transplantation
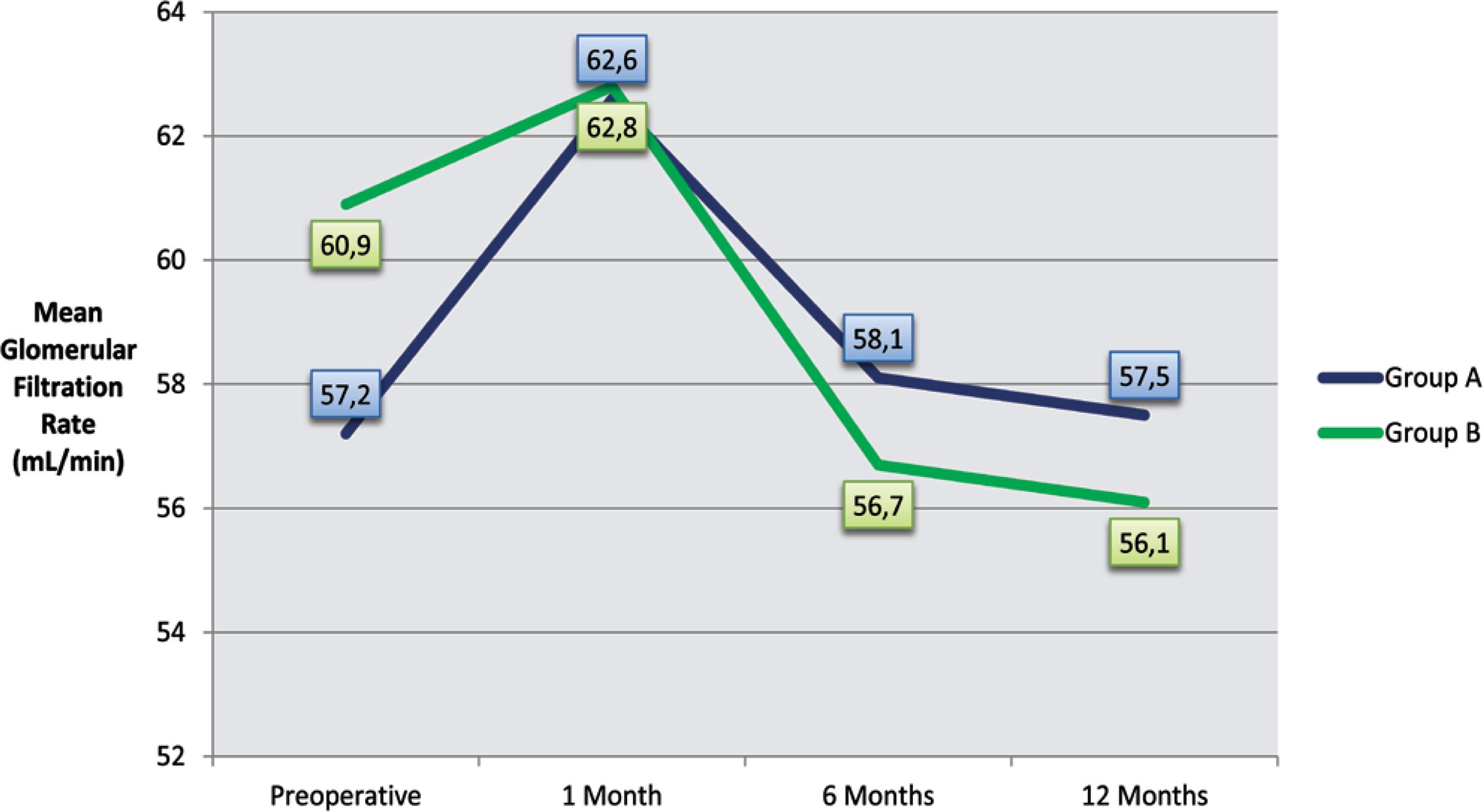
At the time of transplantation, patients in Group A showed a tendency to have lower rates of glomerular filtration (57.2±26.5 vs. 60.9±21.2 ml/min, P=0.301). However, early renal replacement therapy (≤ 1 month after transplantation) was necessary in only 4 patients, with no difference between groups.
In both groups, a tendency towards recovery of renal function was observed after one month of transplantation, although not statistically significant (P=0.343 and P=0.480 for groups A and B, respectively). However, this was followed by a progressive deterioration of the glomerular filtration rate, whose values dropped significantly in Groups A (P=0.050) and B (P<0.001) for the first sixth months post-transplantation, and continued to decline, though at a slower pace, up to twelve months after transplantation (Figure 2).
Evolution of the mean glomerular filtration rate during the first year after transplant in groups A and B.
Mortality and survival
After a mean follow-up of 4.5±2.7 years (1 to 10 years; 961.8 patient-years), the overall mortality did not show significant differences between the two groups of patients (19% vs. 23.4%, P=0.491) and the same goes for comparison of in-hospital mortality and death at 6 months, 1 year, and late (Table 6). The most frequent causes of death in both groups were infectious (5.2% vs. 6.5%, P=0.721) and vascular (5.2% vs. 5.8%, P=0.850), with no statistical significance in the comparison between the two groups.
The comparative analysis of the survival curves (Figure 3) also showed no significant difference between the two groups (P=0.659) and mean survival is currently 7.8±0.5 years in Group A and 7.4±0.3 years in Group B.
DISCUSSION
It is known that the pre-transplantation clinical situation of patients with the highest degree of urgency is different. Decompensated patients are refractory to ambulatory medical treatment for heart failure, which requires hospitalization in cardiac intensive care units where they are to undergo treatment with intravenous diuretics, inotropes (dobutamine and noradrenaline), and a large majority is treated with cardiotonics (levosimendan). On the other hand, patients having a lesser degree of urgency benefit from home and family environment as well as greater mobility, factors that contribute to improvement and even optimization of their psychological, nutritional, and muscular status, in addition to a drastic reduction of nosocomial infections. Undoubtedly, these are aspects that help decrease the potential for post-transplantation mortality and morbidity[99 Lietz K, Miller LW. Improved survival of patients with end-stage heart failure listed for heart transplantation: analysis of organ procurement and transplantation network/U.S. United Network of Organ Sharing data, 1990 to 2005. J Am Coll Cardiol. 2007;50(13):1282-90.].
Nevertheless, in this series we found a higher incidence of perioperative graft dysfunction in Group B. The most severe cases, in which it was necessary to use mechanical support (0% vs. 6.5%, P=0.047), were also detected in this group. But there was no significant difference in mortality between the two groups (3.4% and 4.5%, respectively, P=0.724).
These results point to a little adjusted (skewed) priority rank. The clinical situation of this less urgent group of patients is not homogeneous. The control of symptoms does not imply an improvement in the long-term prognosis. In fact, Lietz & Miller[99 Lietz K, Miller LW. Improved survival of patients with end-stage heart failure listed for heart transplantation: analysis of organ procurement and transplantation network/U.S. United Network of Organ Sharing data, 1990 to 2005. J Am Coll Cardiol. 2007;50(13):1282-90.] and Mokadam et al.[1010 Mokadam NA, Ewald GA, Damiano RJ Jr, Moazami N. Deterioration and mortality among patients with United Network for Organ Sharing status 2 heart disease: caution must be exercised in diverting organs. J Thorac Cardiovas Surg. 2006;131(4):925-6.] demonstrated deterioration in the clinical status in 32-48% of UNOS 2 patients (equivalent to our lower priority VI-VII) during the waiting time for transplantation. These patients come to transplantation in uncertain clinical condition (intermittent instability, recurrent and frequent decompensation, or chronic persistent symptoms), whose control is not always associated with a better prognosis. In addition, advances in the medical treatment of heart failure by cardiotonic drugs and artificial devices have distorted the actual phenotypic presentation of heart failure in late stage[1111 Ramani GV, Mehra MR. Risk assessment in critically ill patients awaiting transplantation: a step forward. Rev Esp Cardiol. 2011;64(3):175-6.,1212 Ramani GV, Uber PA, Mehra MR. Chronic heart failure: contemporary diagnosis and management. Mayo Clin Proc. 2010;85(2):180-95.].
The more favorable response in Group A suggests that patients recently undergoing
intensive anticongestive therapy reach transplantation better compensated from the
cardiorespiratory point of view, regardless of the fact that the pre-transplantation
analytical study showed a higher multi-organ involvement. In fact, bilirubin and
creatinine were higher in this group, both variables which are thought to have a strong
influence on early mortality after transplantation[1313 Weiss ES, Allen JG, Arnaoutakis GJ, George TJ, Russell SD, Shah AS, et
al. Creation of a quantitative recipient risk index for mortality prediction after
cardiac transplantation (IMPACT). Ann Thorac Surg.
2011;92(3):914-21.
14 Kilic A, Allen JG, Arnaoutakis GJ, George TJ, Cameron DE, Vricella LA,
et al. Adult-derived Index for Mortality Prediction After Cardiac Transplantation
(IMPACT) risk score predicts short-term mortality after pediatric heart
transplantation. Ann Thorac Surg. 2012;93(4):1228-34.
15 Vanhuyse F, Maureira P, Mattei MF, Laurent N, Folliguet T, Villemot JP.
Use of the model for end-stage liver disease score for guiding clinical
decision-making in the selection of patients for emergency cardiac transplantation.
Eur J Cardiothorac Surg. 2013;44(1):134-8.-1616 Kilic A, Allen JG, Weiss ES. Validation of the United States-derived
Index for Mortality Prediction after Cardiac Transplantation (IMPACT) using
international registry data. J Heart Lung Transplant.
2013;32(5):492-8.].
In addition, patients coming from intensive care are usually more delicate because they are in unfavorable physical, psychological and nutritional situation, exposed to a hospital environment, and they have been subject to different clinical monitoring devices, some quite invasive. Consequently, there was a greater number of serious infections in the first months post-transplantation in Group A, with values close to statistical significance (22.4% vs. 12.3%, P=0.068). The greatest risk of developing serious infections in this group of patients should alert us to the need for taking more forceful prevention measures, including adaptation of the immunosuppression protocol.
Also analyzed in this study was the evolution of renal function, measured by serum creatinine level and creatinine clearance during the first year, when one would expect greater divergence. There was an improvement in the values of serum creatinine immediately after transplantation in both groups, but a gradual deterioration over years and no significant difference between the groups. The incidence of de novo diabetes, often related to renal dysfunction, was comparable between the two groups, which is justified by similar immunosuppression protocols.
Global survival at 8 years and survival free from severe rejection (2R or 3R) were
similar in the two groups. These results also reinforce the idea that the high risk of
transplantation in these patients can be significantly reduced in units with high volume
of transplantations[1717 Arnaoutakis GJ, George TJ, Allen JG, Russell SD, Shah AS, Conte JV, et
al. Institutional volume and the effect of recipient risk on short-term mortality
after orthotopic heart transplant. J Thorac Cardiovasc Surg.
2012;143(1):157-67.
18 Weiss ES, Allen JG, Meguid RA, Patel ND, Merlo CA, Orens JB, et al. The
impact of center volume on survival in lung transplantation: an analysis of more than
10,000 cases. Ann Thorac Surg. 2009;88(4):1062-70.
19 Weiss ES, Meguid RA, Patel ND, Russell SD, Shah AS, Baumgartner WA, et
al. Increased mortality at low-volume orthotopic heart transplantation centers:
should current standards change?. Ann Thorac Surg.
2008;86(4):1250-9.-2020 Kilic A, Weiss ES, Arnaotakis GJ, George TJ, Conte JV, Shah AS, et al.
Identifying recipients at high risk for graft failure after heart retransplantation.
Ann Thorac Surg. 2012;93(3):712-6.]. The
advantage of having teams with greater experience to perform this procedure in high-risk
candidates is well known, as the results have been proven superior in these
centers[2121 Hosenpud JD, Breen TJ, Edwards EB, Daily OP, Hunsicker LG. The effect of
transplant center volume on cardiac transplant outcome. A report of the United
Network for Organ Sharing Scientific Registry. JAMA.
1994;271(23):1844-9.
22 Arnaoutakis GJ, George TJ, Allen JG, Russell SD, Shah AS, Conte JV, et
al. Institutional volume and the effect of recipient risk on short-term mortality
after orthotopic heart transplant. J Thorac Cardiovasc Surg.
2012;143(1):157-67.-2323 Pettit SJ, Jhund PS, Hawkins NM, Gardner RS, Haj-Yahia S, McMurray JJ,
et al. How small is too small? A systematic review of center volume and outcome after
cardiac transplantation. Cir Cardiovasc Qual Outcomes.
2012;5(6):783-90.].
The main message derived from this study is that, despite their higher risk profile and a further deteriorated clinical situation, urgent but not emergency patients showed similar outcomes to those of patients with lower priority. This may, therefore, raise the question of waste of a non-insignificant number of donors used in emergency cases with poor results in detriment of patients in better clinical conditions who die or deteriorate while on the waiting list, again conditioning the outcome of the transplantation[2424 Hong KN, Iribarne A, Worku B, Takayama H, Gelijns AC, Naka Y, et al. Who is the high-risk recipient? Predicting mortality after heart transplant using pretransplant donor and recipient risk factors. Ann Thorac Surg. 2011;92(2):520-7.]. Listing for transplantation of some of these patients should probably be revised or delayed to optimize their clinical state[2525 Young JB. Heart failure's near dead and dying: reconsidering our heart transplant wait list scheme. J Am Coll Cardiol. 2007;50(13):1291-3.]. Today, more efficient devices for temporary or permanent mechanical assistance (ECMO, ventricular assistance, artificial heart), can greatly improve the clinical situation and reverse multiorgan deterioration[2626 Wever-Pinzon O, Drakos SG, Kfoury AG, Nativi JN, Gilbert EM, Everitt M, et al. Morbidity and mortality in heart transplant candidates supported with mechanical circulatory support: is reappraisal of the current United network for organ sharing thoracic organ allocation policy justified? Circulation. 2013;127(4):452-62.].
In addition it seems important to call for other types of priority scales that include the characteristics of the recipient, other than the urgency of transplantation. Hong et al.[2424 Hong KN, Iribarne A, Worku B, Takayama H, Gelijns AC, Naka Y, et al. Who is the high-risk recipient? Predicting mortality after heart transplant using pretransplant donor and recipient risk factors. Ann Thorac Surg. 2011;92(2):520-7.] distinguished five groups of risk for transplantation, according to the presence of pre-transplant risk factors, where groups of higher risk have a first year survival of only 47-66% and a median survival of less than six years, results that, again, seriously question the use of donors that could have greater benefits in lower risk groups. These authors go as far as to say that donors should be allocated in priority for lower risk recipient groups and, only after that, for high-risk groups. They even warned about the need to consider whether we should not exclude high-risk patients from transplantation because the advantage of transplantation in the short and medium term is reduced in these patients, even when compared to medical treatment[2727 Barge-Caballero E, Paniagua-Martin MJ, Marzoa-Rivas R, Campo-Pérez R, Rodríguez-Fernández JÁ, Pérez-Pérez A, et al. Usefulness of the INTERMACS Scale for predicting outcomes after urgent heart transplantation. Rev Esp Cardiol. 2011;64(3):193-200.,2828 González A, Adsuar A, Hernández A, Borrego JM, Gutiérrez E, Lage E, et al. Is an emergency heart transplantation justified? Transplant Proc. 2006;38(8):2531-3.].
CONCLUSION
The number of patients awaiting transplantation has been increasing, unlike the supply of donors. This situation becomes more complex when one observes an increase in the number of non-priority patients whose clinical condition worsens in the waiting list, turning them into to higher priorities. This panorama requires strengthening of the borders of donation as well as considering responsible and pragmatic allocation of available organs.
In our experience, the results obtained from patients in urgent priority were similar to those of patients awaiting transplantation at home. The allocation of donors in this group does not seem to reduce the success of transplantation, a consideration that can probably not be extended to patients in more critical situations.
REFERENCES
-
1Stehlik J, Edwards LB, Kucheryavaya AY, Aurora P, Christie JD, Kirk R, et al. The Registry of the International Society for Heart and Lung Transplantation: twenty-seventh official adult heart transplant report-2010. J Heart Lung Transplant. 2010;29(10):1089-103.
-
2Russo MJ, Chen JM, Hong KN, Stewart AS, Ascheim DD, Argenziano M, et al; Columbia University Heart Transplant Outcomes Research Group. Survival after heart transplantation is not diminished among recipients with uncomplicated diabetes mellitus: an analysis of the United Network of Organ Sharing database. Circulation. 2006;114(21):2280-7.
-
3McMurray JJ, Adamopoulos S, Anker SD, Auricchio A, Böhm M, Dickstein K, et al; Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure 2012 of the European Society of Cardiology; ESC Committee for Practice Guidelines. ESC guidelines for the diagnosis and treatment of acute and chronic heart failure 2012: The Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure 2012 of the European Society of Cardiology. Developed in collaboration with the Heart Failure Association (HFA) of the ESC. Eur J Heart Fail. 2012;14(8):803-69.
-
4Fiorelli Al, Branco JN, Dinkhuysen JJ, Oliveira Junior JL, Pereira TV, Dinardi LF, et al. Risk factor analysis of late survival after heart transplantation according to donor profile: a multi-institutional retrospective study of 512 transplants. Transplant Proc. 2012;44(8):2469-72.
-
5Forni A, Luciani GB, Chiominto B, Pizzuti M, Mazzucco A, Faggian G. Results with expanded donor acceptance criteria in heart transplantation. Tranplant Proc. 2011;43(4):953-9.
-
6Holman WL, Kormos RL, Naftel DC, Miller MA, Pagani FD, Blume E, et al. Predictors of death and transplant in patients with a mechanical circulatory support device: a multi-institutional study. J Heart Lung Transplant. 2009;28(1):44-50.
-
7Starling RC, Naka Y, Boyle AJ, Gonzalez-Stawinski G, John R, Jorde U, et al. Results of the post-U.S. Food and Drug Administration-approval study with a continuous flow left ventricular assist device as a bridge to heart transplantation: a prospective study using the INTERMACS (Interagency Registry for Mechanically Assisted Circulatory Support). J Am Coll Cardiol. 2011;57(19):1890-8
-
8Stevenson WL, Pagani FD, Young JB, Jessup M, Miller L, Kormos RL, et al. INTERMACS profiles of advanced heart failure: the current picture. J Heart Lung Transplant. 2009;28(6):535-41.
-
9Lietz K, Miller LW. Improved survival of patients with end-stage heart failure listed for heart transplantation: analysis of organ procurement and transplantation network/U.S. United Network of Organ Sharing data, 1990 to 2005. J Am Coll Cardiol. 2007;50(13):1282-90.
-
10Mokadam NA, Ewald GA, Damiano RJ Jr, Moazami N. Deterioration and mortality among patients with United Network for Organ Sharing status 2 heart disease: caution must be exercised in diverting organs. J Thorac Cardiovas Surg. 2006;131(4):925-6.
-
11Ramani GV, Mehra MR. Risk assessment in critically ill patients awaiting transplantation: a step forward. Rev Esp Cardiol. 2011;64(3):175-6.
-
12Ramani GV, Uber PA, Mehra MR. Chronic heart failure: contemporary diagnosis and management. Mayo Clin Proc. 2010;85(2):180-95.
-
13Weiss ES, Allen JG, Arnaoutakis GJ, George TJ, Russell SD, Shah AS, et al. Creation of a quantitative recipient risk index for mortality prediction after cardiac transplantation (IMPACT). Ann Thorac Surg. 2011;92(3):914-21.
-
14Kilic A, Allen JG, Arnaoutakis GJ, George TJ, Cameron DE, Vricella LA, et al. Adult-derived Index for Mortality Prediction After Cardiac Transplantation (IMPACT) risk score predicts short-term mortality after pediatric heart transplantation. Ann Thorac Surg. 2012;93(4):1228-34.
-
15Vanhuyse F, Maureira P, Mattei MF, Laurent N, Folliguet T, Villemot JP. Use of the model for end-stage liver disease score for guiding clinical decision-making in the selection of patients for emergency cardiac transplantation. Eur J Cardiothorac Surg. 2013;44(1):134-8.
-
16Kilic A, Allen JG, Weiss ES. Validation of the United States-derived Index for Mortality Prediction after Cardiac Transplantation (IMPACT) using international registry data. J Heart Lung Transplant. 2013;32(5):492-8.
-
17Arnaoutakis GJ, George TJ, Allen JG, Russell SD, Shah AS, Conte JV, et al. Institutional volume and the effect of recipient risk on short-term mortality after orthotopic heart transplant. J Thorac Cardiovasc Surg. 2012;143(1):157-67.
-
18Weiss ES, Allen JG, Meguid RA, Patel ND, Merlo CA, Orens JB, et al. The impact of center volume on survival in lung transplantation: an analysis of more than 10,000 cases. Ann Thorac Surg. 2009;88(4):1062-70.
-
19Weiss ES, Meguid RA, Patel ND, Russell SD, Shah AS, Baumgartner WA, et al. Increased mortality at low-volume orthotopic heart transplantation centers: should current standards change?. Ann Thorac Surg. 2008;86(4):1250-9.
-
20Kilic A, Weiss ES, Arnaotakis GJ, George TJ, Conte JV, Shah AS, et al. Identifying recipients at high risk for graft failure after heart retransplantation. Ann Thorac Surg. 2012;93(3):712-6.
-
21Hosenpud JD, Breen TJ, Edwards EB, Daily OP, Hunsicker LG. The effect of transplant center volume on cardiac transplant outcome. A report of the United Network for Organ Sharing Scientific Registry. JAMA. 1994;271(23):1844-9.
-
22Arnaoutakis GJ, George TJ, Allen JG, Russell SD, Shah AS, Conte JV, et al. Institutional volume and the effect of recipient risk on short-term mortality after orthotopic heart transplant. J Thorac Cardiovasc Surg. 2012;143(1):157-67.
-
23Pettit SJ, Jhund PS, Hawkins NM, Gardner RS, Haj-Yahia S, McMurray JJ, et al. How small is too small? A systematic review of center volume and outcome after cardiac transplantation. Cir Cardiovasc Qual Outcomes. 2012;5(6):783-90.
-
24Hong KN, Iribarne A, Worku B, Takayama H, Gelijns AC, Naka Y, et al. Who is the high-risk recipient? Predicting mortality after heart transplant using pretransplant donor and recipient risk factors. Ann Thorac Surg. 2011;92(2):520-7.
-
25Young JB. Heart failure's near dead and dying: reconsidering our heart transplant wait list scheme. J Am Coll Cardiol. 2007;50(13):1291-3.
-
26Wever-Pinzon O, Drakos SG, Kfoury AG, Nativi JN, Gilbert EM, Everitt M, et al. Morbidity and mortality in heart transplant candidates supported with mechanical circulatory support: is reappraisal of the current United network for organ sharing thoracic organ allocation policy justified? Circulation. 2013;127(4):452-62.
-
27Barge-Caballero E, Paniagua-Martin MJ, Marzoa-Rivas R, Campo-Pérez R, Rodríguez-Fernández JÁ, Pérez-Pérez A, et al. Usefulness of the INTERMACS Scale for predicting outcomes after urgent heart transplantation. Rev Esp Cardiol. 2011;64(3):193-200.
-
28González A, Adsuar A, Hernández A, Borrego JM, Gutiérrez E, Lage E, et al. Is an emergency heart transplantation justified? Transplant Proc. 2006;38(8):2531-3.
-
No financial support
-
This study was carried out at Centre of Cardiothoracic Surgery and Transplantation of Thoracic Organs University Hospital and Faculty of Medicine of University of Coimbra, Portugal.
Publication Dates
-
Publication in this collection
Jul-Sep 2014
History
-
Received
10 May 2014 -
Accepted
25 May 2014