Abstract
PURPOSE:
To compare the postoperative analgesic effects of lidocaine, morphine and lidocaine plus morphine administered by constant rate infusion (CRI) and analyzing their effects on opioid requirements after orthopedic surgery in dogs.
METHODS:
Twenty-four dogs underwent fracture repairs were premedicated with IM acepromazine (0.05 mg/kg) combined with morphine (0.3mg/kg). Anesthesia was induced with IV propofol (4 to 5 mg/ kg) and maintained with isoflurane. The dogs were randomly assigned to 3 groups and administered a CRI IV of lidocaine (T-L), morphine (T-M) or lidocaine plus morphine (T-LM) at the same doses. Postoperative analgesia was assessed for 24 hours using a Visual Analog Scale (VAS) and the Glasgow Composite Pain Scale (GCPS). Rescue analgesia was performed if the evaluation score exceeded 50% of the VAS and/or 33% of the GCPS.
RESULTS:
The pain score and postoperative opioid requirements did not differ among the treatments. Rescue analgesia was administered to 1/8 dogs in the T-M and T-LM, and to 3/8 dogs in the T-L.
CONCLUSION:
Lidocaine, morphine or lidocaine/morphine CRI may be efficacious techniques for pain management in the first 24 hours post-surgery. However, the two drugs administered together did not reduce the postoperative opioid requirement in dogs undergoing fracture repair. Key words: Anesthesia. Analgesics. Analgesics, Opioid. Lidocaine. Morphine. Dogs.
Anesthesia; Analgesics; Analgesics, Opioid; Lidocaine; Morphine; Dogs
Introduction
Orthopedic surgery in dogs is commonly associated with moderate to severe postoperative pain and inflammation11. Bergmann HM, Nolte I, Kramer S. Comparison of analgesic efficacy of preoperative or postoperative carprofen with or without preincisional mepivacaine epidural anesthesia in canine pelvic or femoral fracture repair. Vet Surg. 2007;36:623-32.
2. Kona-Boun JJ, Cuvelliez S, Troncy E. Evaluation of epidural administration of morphine or morphine and bupivacaine for postoperative analgesia after premedication with an opioid analgesic and orthopedic surgery in dogs. J Am Vet Med Assoc. 2006;229:1103-12.
3. Gruet P, Seewald W, King JN. Evaluation of subcutaneous and oral administration of robenacoxib and meloxicam for the treatment of acute pain and inflammation associated with orthopedic surgery in dogs. Am J Vet Res. 2011;72:184-93.
-
44. Moak P, Hosgood G, Rowe E, Lemke KA. Evaluation of intra-articular and subcutaneous administration of meloxicam for postoperative analgesia following stifle surgery in dogs. Vet Comp Orthop Traumatol. 2011;24:32-8.. Therefore, providing effective analgesia is essential during the immediate postoperative period and for several days or weeks thereafter33. Gruet P, Seewald W, King JN. Evaluation of subcutaneous and oral administration of robenacoxib and meloxicam for the treatment of acute pain and inflammation associated with orthopedic surgery in dogs. Am J Vet Res. 2011;72:184-93.
-
44. Moak P, Hosgood G, Rowe E, Lemke KA. Evaluation of intra-articular and subcutaneous administration of meloxicam for postoperative analgesia following stifle surgery in dogs. Vet Comp Orthop Traumatol. 2011;24:32-8.. Moreover, effective pain control may result in a more rapid recovery and return to normal function33. Gruet P, Seewald W, King JN. Evaluation of subcutaneous and oral administration of robenacoxib and meloxicam for the treatment of acute pain and inflammation associated with orthopedic surgery in dogs. Am J Vet Res. 2011;72:184-93..
Postoperative pain may be controlled by local anesthetic agents, opioids and non-steroidal anti-inflammatory drugs (NSAIDs) or a combination of these items55. Shih AC, Robertson S, Isaza N, Pablo L, Davies W. Comparison between analgesic effects of buprenorphine, carprofen, and buprenorphine with carprofen for canine ovariohysterectomy. Vet Anaesth Analg. 2008;35:69-79. - 66. Ortega M, Cruz I. Evaluation of a constant rate infusion of lidocaine for balanced anesthesia in dogs undergoing surgery. Can Vet J. 2011;52:856-60.. The administration of a single drug is often insufficient for postoperative analgesia. Conversely, multimodal or balanced analgesia combines different analgesic drugs to achieve beneficial additive or synergistic analgesic effects. Consequently, lower analgesic doses may be administered to potentially reduce the undesirable side effects77. Kehlet H, Dahl JB. The value of ''multimodal'' or ''balanced analgesia'' in postoperative pain treatment. Anaesth Analg. 1993;7:1048-563. - 88. Slingsby SL, Waterman-Pearson AE. Analgesic effects in dogs of carprofen and pethidine together compared with the effects of either drug alone. Vet Rec. 2001;148:441-4..
In humans, it is thought that systemic lidocaine infusion is an effective analgesic technique for some procedures, including major abdominal surgery99. Koppert W, Weigand M, Neumann F, Sittl R, Schuettler J, Schmelz M, Hering W. Perioperative intravenous lidocaine has preventive effects on postoperative pain and morphine consumption after major abdominal surgery. Anesth Analg. 2004;98:1050-5.
10. Kuo CP, Jao SW, Chen KM, Wong CS, Yeh CC, Sheen MJ, Wu CT. Comparison of the effects of thoracic epidural analgesia and i.v. infusion with lidocaine on cytokine response, postoperative pain and bowel function in patients undergoing colonic surgery. Br J Anaesth. 2006;97:640-6.
-
1111. Lauwick S, Kim J, Michelagnoli G, Mistraletti G, Feldman L, Fried G, Carli F. Intraoperative infusion of lidocaine reduces postoperative fentanyl requirements in patients undergoing laparoscopic cholecystectomy. Can J Anaesth. 2008;5:754-60., prostatectomy1212. Groudine SB, Fisher HA, Kaufman RP Jr, Patel MK, Wilkins LJ, Mehta SA, Lumb PD. Intravenous lidocaine speeds the return of bowel function, decreases postoperative pain, and shortens hospital stay in patients undergoing radical retropubic prostatectomy. Anesth Analg. 1998;86:235-9.
,
13 13. Lauwick S, Kim DJ, Mistraletti G, Carli F. Functional walking capacity as an outcome measure of laparoscopic prostatectomy: the effect of lidocaine infusion. Br J Anaesth. 2009;103:213-9.and some ambulatory surgeries1414. McKay A, Gottschalk A, Ploppa A, Durieux ME, Groves DS. Systemic lidocaine decreased the perioperative opioid analgesic requirements but failed to reduce discharge time after ambulatory surgery. Anesth Analg. 2009;109:1805-8.. Among the beneficial effects of lidocaine, studies have reported a morphine-sparing effect, an improved dynamic analgesia and a reduced hospital stay.
Additionally, several studies have shown that lidocaine infusion reduces the minimum alveolar concentration (MAC) of inhalation agents in horses1515. Doherty TJ, Frazier DL. Effect of intravenous lidocaine on halothane MAC in ponies. Equine Vet J. 1998;30:340-3., dogs1616. Himes RS, DiFazio CA, Burney RG. Effects of lidocaine on the anesthetic requirements for nitrous oxide and halothane. Anesthesiology. 1977;47:437-40.
17. Muir WW III, Wiese AJ, March PA. Effects of morphine, lidocaine, ketamine, and morphine lidocaine ketamine drug combination on minimum alveolar concentration in dogs anesthetized with isoflurane. Am J Vet Res. 2003;64:1155-60.
18. Valverde A, Doherty TJ, Hernandez J. Effect of intravenous lidocaine on isoflurane MAC in dogs. Vet Anaesth Analg. 2004;31:264-71.
-
1919. Matsubara LM, Oliva VN, Gabas DT, Oliveira GC, Cassetari ML. Effect of lidocaine on the minimum alveolar concentration of sevoflurane in dogs. Vet Anaesth Analg. 2009; 407-13., goats2020. Doherty TJ, Redua MA, Queiroz-Castro P, Egger C, Cox SK, Rohrbach BW. Effect of intravenous lidocaine and ketamine on the minimum alveolar concentration of isoflurane in goats. Vet Anaesth Analg. 2007;32:125-31., cats2121. Pypendop BH, Ilkiw JE. Effects of intravenous lidocaine administration on the minimum alveolar concentration of isoflurane in cats. Anesth Analg. 2005;100:97-101. and calves2222. Vesal N, Spadavecchia C, Steiner A, Kirscher F, Levionnois OL. Evaluation of the isoflurane-sparing effects of lidocaine infusion during umbilical surgery in calves. Vet Anaesth Analg. 2011;38:451-60.. Therefore, lidocaine infusion has been widely used as a supplement to general anesthesia77. Kehlet H, Dahl JB. The value of ''multimodal'' or ''balanced analgesia'' in postoperative pain treatment. Anaesth Analg. 1993;7:1048-563..
However, relatively few studies have focused on the postoperative analgesic effects of intravenous lidocaine infusion in dogs. Indeed, to our knowledge, the analgesic effect of a constant rate infusion (CRI) of lidocaine in dogs undergoing orthopedic surgery has not been reported.
The aim of the present study was to compare the postoperative analgesic effects of lidocaine, morphine or lidocaine plus morphine CRI in dogs undergoing fracture repair and analyzing their effects on opioid requirements after orthopedic surgery in dogs. We hypothesized that the combination of lidocaine with morphine would result in a superior analgesic effect compared with each drug administered alone.
Methods
This study was approved by the Institutional Animal Research Ethical Committee (protocol number 756), and permission for the participation of each dog was obtained from the owner.
Twenty-four dogs (19 males, five females), aged one to seven years (median, 2.6 years) and weighing between three and 19 kg (median, 12.3 kg), that underwent surgical repairs of a femoral, tibial, humeral or radial fracture were enrolled. The dogs were selected for this study using a physical examination and laboratory tests (i.e., a complete blood cell count, and measurements of the urea, creatinine, alanine aminotransferase and aspartate aminotransferase levels). The following exclusion criteria were applied: significant orthopedic trauma (multiple fractures), pregnancy, or severe concomitant disorders of the kidneys or liver.
After withdrawing food and water for 12 and three hours, respectively, all dogs were sedated with intramuscular (IM) acepromazine (0.05mg/kg; Acepran 0.2%, Univet, Sao Paulo-SP, Brazil) combined with morphine (0.3mg/kg; Dimorf, Cristalia, Campinas-SP, Brazil). Twenty min after sedation, an intravenous (IV) catheter (20 gauge, 32 mm; Insyte, Becton Dickinson, Juiz de Fora-MG, Brazil) was aseptically placed in a cephalic vein, and the CRI was started as follows: T-L (n=8) - bolus IV of lidocaine (1.0mg/kg), followed by lidocaine infusion (50µg/kg/min); T-M (n=8) - bolus IV of saline (0.5mL/kg) followed by morphine infusion (0.1mg/kg/h); T-LM (n=8) - bolus IV of lidocaine (1.0mg/kg), followed by a lidocaine infusion (50µg/kg/min) combined with a morphine infusion (0.1mg/kg/h). To preserve blinding, the initial bolus was made up to volumes equivalent to 0.5mL/kg, and then administrated for a period of 60 seconds. The drug infusion treatments were administered for 20 min before the anesthetic induction, and they were maintained using a peristaltic pump (ST1000; Samtronic, Sao Paulo, SP, Brazil) throughout the surgical procedure. The drugs were diluted in Ringer´s solution and administered at a rate of 5 to 10mL/kg/h.
Anesthesia was induced with IV propofol (4 to 5mg/kg; Propovan, Cristalia, Campinas-SP, Brazil). Orotracheal intubation was performed, and anesthesia was maintained with isoflurane in 100% oxygen in a small animal rebreathing circuit (Samurai; Takaoka, Sao Paulo-SP, Brazil). All dogs received initial doses of IV meloxicam (0.2mg/kg) 5 min before the surgical incisions were made, and then they received additional doses of meloxicam (0.1 mg/kg, SC, q 24 hour) beginning on day 1 and continuing for up to 10 days.
During the anesthetic procedure, the end-tidal carbon dioxide concentration (ETCO2), end-tidal isoflurane concentration (ETISO), oxygen saturation of hemoglobin (SpO2%) and heart rate (HR) were continuously measured using a capnograph, pulse oximeter and gas analyzer (VAMOS plus; Dräger, Sao Paulo-SP, Brazil). The end-tidal concentration of isoflurane was adjusted based on the arterial pressure and heart rate changes, and using the conventional signs of anesthesia. Systolic arterial blood pressure (SBP) was measured using a noninvasive method with a Doppler ultrasonic system (Doppler 841-A; Parks Medical Electronics, Las Vegas, EUA), with the width cuff approximately 40% of the antebrachium circumference. The respiratory rate (RR) was measured observing of the thoracic movements for one minute.
The administration of postoperative analgesic drugs was supervised by one anesthesiologist, and the pain assessment was performed by a blinded assessor. The pain was measured before the sedation (baseline scores), and at 30 min, and 1, 2, 4, 6, 8, 12, 20 and 24 hours after extubation, using the Glasgow Composite Pain Scale (GCPS)33. Gruet P, Seewald W, King JN. Evaluation of subcutaneous and oral administration of robenacoxib and meloxicam for the treatment of acute pain and inflammation associated with orthopedic surgery in dogs. Am J Vet Res. 2011;72:184-93.
,
2323. Holton L, Reid J, Scott EM, Pawson P, Nolan A. Development of a behaviour-based scale to measure acute pain in dogs. Vet Rec. 2001;148:525-31.. Next, pain was assessed using a Visual Analogue Scale (VAS) in which, 0=no pain and 100=worst possible pain, manifested by vocalization, aggression, and refusal to allow the examination)11. Bergmann HM, Nolte I, Kramer S. Comparison of analgesic efficacy of preoperative or postoperative carprofen with or without preincisional mepivacaine epidural anesthesia in canine pelvic or femoral fracture repair. Vet Surg. 2007;36:623-32.
2. Kona-Boun JJ, Cuvelliez S, Troncy E. Evaluation of epidural administration of morphine or morphine and bupivacaine for postoperative analgesia after premedication with an opioid analgesic and orthopedic surgery in dogs. J Am Vet Med Assoc. 2006;229:1103-12.
3. Gruet P, Seewald W, King JN. Evaluation of subcutaneous and oral administration of robenacoxib and meloxicam for the treatment of acute pain and inflammation associated with orthopedic surgery in dogs. Am J Vet Res. 2011;72:184-93.
4. Moak P, Hosgood G, Rowe E, Lemke KA. Evaluation of intra-articular and subcutaneous administration of meloxicam for postoperative analgesia following stifle surgery in dogs. Vet Comp Orthop Traumatol. 2011;24:32-8.
-
55. Shih AC, Robertson S, Isaza N, Pablo L, Davies W. Comparison between analgesic effects of buprenorphine, carprofen, and buprenorphine with carprofen for canine ovariohysterectomy. Vet Anaesth Analg. 2008;35:69-79..
When the GCPS score was >33% or/and the VAS pain score was >50%, IM morphine (0.5mg/kg) was administered as a rescue analgesic.
At each pain assessment, sedation was scored according to a scale that ranged from 1 to 5, as follows: normal behavior (1); mild sedation, some decrease in normal activity, some resistance to handling, responds to name (2); moderate sedation, easy to handle, still spontaneously active, responds to name, but more slowly (3); moderate-heavy sedation, less spontaneous activity, verbal encouragement needed to rise, minimal resistance to handling (4); sternal recumbency, physical stimulus required to rise, depressed, no resistance to handling (5)2424. MacDougall LM, Hethey JA, Livingston A, Clark C, Shmon CL, Duke-Novakovski T. Antinociceptive, cardiopulmonary, and sedative effects of five intravenous infusion rates of lidocaine in conscious dogs. Vet Anaesth Analg. 2009;36:512-22..
The data were recorded as mean±SD. A Kolmogorov-Smirnov-test was performed to assess normality of the variables. Values for HR, RR, SBP, SpO2, ETCO2 and ETISO concentrations were analyzed using a two-way analysis of variance (ANOVA) followed by a Tukey test to compare differences between treatment groups at each time point and differences in time for each treatment group. The pain and sedation scores were compared at different time-points by means of a nonparametric Friedman test for repeated measures. For comparisons at each time point a nonparametric Kruskal-Wallis test followed by post-hoc Dunn's test was employed. All analyses were performed using the computer software (GraphPad Instat5). Differences were considered significant when p<0.05.
Results
The three treatment groups were comparable in both demographic and clinical variables. There were no significant intergroup differences concerning the patient's ages, weights, surgical times, times to endotracheal extubation or recovery times (p>0.05) (Table 1).
The median pain scores did not significantly differ among the treatment groups at any time point (p>0.05) (Figures 1 and 2). The rescue analgesia was less frequently for T-M (1 of 8 dogs) and T-LM groups (1 of 8 dogs) than that in the T-L group (3 of 8 dogs). However, the number of dogs rescued in different treatment groups was not statistically different (p=0.29). One dog from T-M and TL-M groups and three dogs from T-L group required rescue analgesia twice (Table 2).
Mean±SD VAS score measured before surgery (ie, baseline) and at 30 min, and 1, 2, 4, 6, 8, 12, 20 and 24 hours after tracheal extubation in dogs undergoing elective orthopedic surgeries treated with intravenous lidocaine infusion (T-L), morphine infusion (T-M) or lidocaine plus morphine infusion (T-LM). The VAS ranged from 0 (lack of pain) to 10 (worst possible pain).
Mean ± SD GCPS score measured before surgery (ie, baseline) and at 30 min, and 1, 2, 4, 6, 8, 12, 20 and 24 hours after tracheal extubation in dogs undergoing elective orthopedic surgeries treated with intravenous lidocaine infusion (T-L), morphine infusion (T-M) or lidocaine plus morphine infusion (T-LM). The GCPS ranged from 0 (lack of pain) to 24 (worst possible pain).
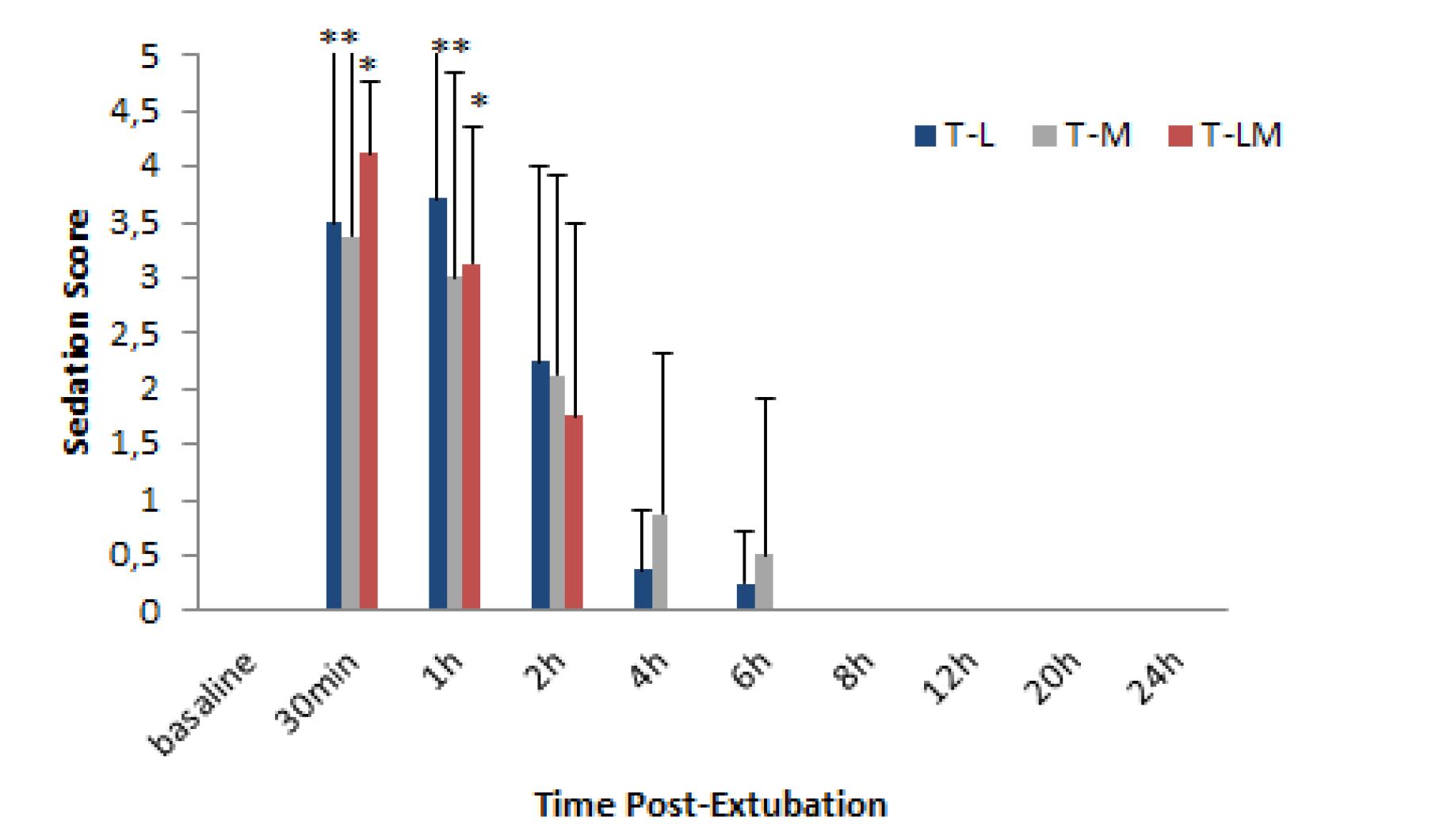
The degree of sedation was similar among the groups (p=0.30). The deepest degree of sedation was observed at 30 min (p<0.001) and 1 hour (p<0.001) after extubation for all treatment groups (Figure 3).
Mean ± SD sedation score measured before surgery (ie, baseline) and at 30 min, and 1, 2, 4, 6, 8, 12, 20 and 24 hours after tracheal extubation in dogs undergoing elective orthopedic surgeries treated with intravenous lidocaine infusion (T-L), morphine infusion (T-M) or lidocaine plus morphine infusion (T-LM). The sedation score ranged from 1 to 5 (Normal behavior =1; Mild sedation, some decrease in normal activity, some resistance to handling, responds to name =2; Moderate sedation, easy to handle, still spontaneously active, responds to name, but slower =3; Moderate-heavy sedation, less spontaneous activity, verbal encouragement needed to rise, minimal resistance to handling =4; Sternal recumbency, physical stimulus required to rise, depressed, no resistance to handling =5). *Within a treatment group, value differs significantly (p<0.05) from the baseline value.
Adverse effects, such as nausea, vomiting, excitement and shivering, were not observed in any of the dogs.
Discussion
The dosing regimens in the present study were selected based on recent trials involving lidocaine or morphine administered by CRI to dogs66. Ortega M, Cruz I. Evaluation of a constant rate infusion of lidocaine for balanced anesthesia in dogs undergoing surgery. Can Vet J. 2011;52:856-60.
,
2424. MacDougall LM, Hethey JA, Livingston A, Clark C, Shmon CL, Duke-Novakovski T. Antinociceptive, cardiopulmonary, and sedative effects of five intravenous infusion rates of lidocaine in conscious dogs. Vet Anaesth Analg. 2009;36:512-22.
25. Smith LJ, Bentley E, Shih A, Miller PE. Systemic lidocaine infusion as an analgesic for intraocular surgery in dogs: a pilot study. Vet Anaesth Analg. 2004;31:53-63.
-
2626. Guedes AG, Rudé EP, Rider MA. Evaluation of histamine release during constant rate infusion of morphine in dogs. Vet Anaesth Analg. 2006;33:28-35.. Moreover, previous studies have shown that the perioperative period seems to be particularly important for lidocaine-sensitive mechanisms of postoperative pain99. Koppert W, Weigand M, Neumann F, Sittl R, Schuettler J, Schmelz M, Hering W. Perioperative intravenous lidocaine has preventive effects on postoperative pain and morphine consumption after major abdominal surgery. Anesth Analg. 2004;98:1050-5.
,
1414. McKay A, Gottschalk A, Ploppa A, Durieux ME, Groves DS. Systemic lidocaine decreased the perioperative opioid analgesic requirements but failed to reduce discharge time after ambulatory surgery. Anesth Analg. 2009;109:1805-8.
,
2626. Guedes AG, Rudé EP, Rider MA. Evaluation of histamine release during constant rate infusion of morphine in dogs. Vet Anaesth Analg. 2006;33:28-35.. Thus, in our study, the infusions were started at least 20 min before surgery and were maintained throughout the surgical procedure.
Although the analgesic effects of systemic lidocaine have been proven for chronic pain, particularly for neuropathic pain states2727. Kastrup J, Petersen P, Dejgard A, Angelo HR, Hilsted J. Intravenous lidocaine infusion: a new treatment of chronic painful diabetic neuropathy?. Pain. 1987;28:69-75.
28. Galer BS, Miller KV, Rowbotham MC. Response to intravenous lidocaine infusion differs based on clinical diagnosis and site of nervous system injury. Neurology. 1993;43:1233-5.
-
2929. Baranowski AP, De Courcey J, Bonello E. A trial of intravenous lidocaine on the pain and allodynia of postherpetic neuralgia. J Pain Symptom Manage. 1999;17:429-33., conflicting results have been achieved for acute pain, such as postoperative pain1212. Groudine SB, Fisher HA, Kaufman RP Jr, Patel MK, Wilkins LJ, Mehta SA, Lumb PD. Intravenous lidocaine speeds the return of bowel function, decreases postoperative pain, and shortens hospital stay in patients undergoing radical retropubic prostatectomy. Anesth Analg. 1998;86:235-9.
,
2525. Smith LJ, Bentley E, Shih A, Miller PE. Systemic lidocaine infusion as an analgesic for intraocular surgery in dogs: a pilot study. Vet Anaesth Analg. 2004;31:53-63.
,
3030. Cassuto J, Wallin G, Hogstrom S, Faxén A, Rimbäck G. Inhibition of postoperative pain by continuous low-dose intravenous infusion of lidocaine. Anesth Analg. 1985;64:971-4..
In humans undergoing abdominal surgeries, systemic lidocaine reduced postoperative pain and morphine consumption when it was applied perioperatively, however, the effects were most prominent at 36 hours after surgery99. Koppert W, Weigand M, Neumann F, Sittl R, Schuettler J, Schmelz M, Hering W. Perioperative intravenous lidocaine has preventive effects on postoperative pain and morphine consumption after major abdominal surgery. Anesth Analg. 2004;98:1050-5.. Controversially, other studies have reported that systemic lidocaine neither reduced pain scores or induced any postoperative opioid-sparing effect3131. Cepeda MS, Delgado M, Ponce M, Cruz CA, Carr DB. Equivalent outcomes during postoperative patient-controlled intravenous analgesia with lidocaine plus morphine versus morphine alone. Anesth Analg. 1996;83:102-6.
32. Chia YY, Tan PH, Wang KY, Liu K. Lignocaine plus morphine in bolus patient-controlled intravenous analgesia lacks post-operative morphine-sparing effect. Eur J Anaesthesiol. 1998;15:664-8.
-
3333. Martin F, Cherif K, Gentili ME, Enel D, Abe E, Alvarez JC, Mazoit JX, Chauvin M, Bouhassira D, Fletcher D. Lack of impact of intravenous lidocaine on analgesia, functional recovery, and nociceptive pain threshold after total hip arthroplasty. Anesthesiology. 2008;109:118-23..
While opioids and local anesthetics have different sites and mechanisms of action, simultaneous administration of the two drugs have been suggested to enhance the analgesic effects and induce an opioid-sparing effect3434. Cohen SP, Mao J. Is the analgesic effect of systemic lidocaine mediated through opioid receptors? Acta Anaesthesiol Scand. 2003;47:910-1.. The mechanisms and site of action of systemic lidocaine remain unclear. The analgesic properties of lidocaine are mediated by various mechanisms, including sodium channel blockade3535. Hollmann MW, Durieux ME. Local anesthetics and the inflammatory response: a new therapeutic indication? Anesthesiology. 2000;93:858-75., inhibition of G protein-coupled receptors3636. Hollmann MW, Strumper D, Herroeder S, Durieux ME. Receptors, G proteins, and their interactions. Anesthesiology. 2005;103:1066-78.and NMDA receptors3737. Nagy I, Woolf CJ. Lignocaine selectively reduces C fibre-evoked neuronal activity in rat spinal cord in vitro by decreasing N-methyl-D-aspartate and neurokinin receptormediated post-synaptic depolarizations; implications for the development of novel centrally acting analgesics. Pain. 1996;64:59-70. , 3838. Sugimoto M, Uchida I, Mashimo T. Local anaesthetics have different mechanisms and sites of action at the recombinant N-methyl-D-aspartate (NMDA) receptors. Br J Pharmacol. 2003;138:876-82.. In addition, local anesthetics have been reported to interact with mu and kappa opioid receptors3434. Cohen SP, Mao J. Is the analgesic effect of systemic lidocaine mediated through opioid receptors? Acta Anaesthesiol Scand. 2003;47:910-1., potentiating the analgesic effect mediated by opioid drugs3939. Deneuche AJ, Dufayet C, Goby L, Fayolle P, Desbois C. Analgesic comparison of meloxicam or ketoprofen for orthopedic surgery in dogs. Vet Surg. 2004;33:650-60..
Therefore, we hypothesized that administering lidocaine plus morphine would provide better pain control for dogs in the first 24 hours after fracture repair compared to a single administration of each drug. However, based on the pain scores, the three treatments provided similar analgesia and adding lidocaine to morphine did not influence the postoperative opioid requirement. These results support the findings of Cepeda et al.3131. Cepeda MS, Delgado M, Ponce M, Cruz CA, Carr DB. Equivalent outcomes during postoperative patient-controlled intravenous analgesia with lidocaine plus morphine versus morphine alone. Anesth Analg. 1996;83:102-6. who reported no differences in analgesic effects and postoperative opioid requirements using morphine or morphine plus lidocaine infusions.
In dogs, a loading intravenous dose of 1.5mg/kg prior to anesthetic induction, followed by an infusion of lidocaine at a rate of 25µg/kg/h results in a similar analgesic effect to that provided by intraoperative IV morphine infusion (0.15mg/kg/h), and 50% of the dogs did not required any additional analgesics at any time after intraocular surgery2525. Smith LJ, Bentley E, Shih A, Miller PE. Systemic lidocaine infusion as an analgesic for intraocular surgery in dogs: a pilot study. Vet Anaesth Analg. 2004;31:53-63.. In the current study, the majority of dogs (7 of 8 in the T-M and T-LM groups and 5 of 8 in the T-L group) did not require any further analgesia following surgery. These dogs appeared calm and comfortable in the postoperative period.
A possible factor that may have contributed to the lack of a significant difference in this study for postoperative pain degree is the variability inherent in this type of clinical study. Although no significant differences were highlighted among the three patient groups regarding age, body weight, and length of surgery, the dogs were referred to the hospital with different medical and surgical conditions. A standardized surgical stimulus was not provided. The dogs included in this study underwent various types of fracture repair, involving different bones. As with most clinical studies, it is difficult to control certain factors, such as the degree of bone trauma, the surgical technique used, or the administration of pre-emptive analgesics. Dogs undergoing femoral and humeral fracture repairs are often presumed to have more significant pain than dogs undergoing tibial and radial fracture repairs. This variability may have confounded the pain scoring in the present study, particularly because different surgical procedures may result in different degrees of postoperative pain. Thus, variability in a clinical population is unavoidable.
The number of dogs was also limited, given the criteria and need for owner consent. The addition of a control group, receiving no analgesics would have provided a better measure of the effectiveness of the treatments administered in this study. However, for humanitarian and ethical reasons, we decided not to use an untreated control group.
Indeed, the provision of meloxicam before the surgical procedure may have masked the differences in the signs of pain among the groups. Meloxicam administered preoperatively is a safe and effective method of controlling postoperative pain for up to 24 hours in dogs undergoing orthopedic surgery33. Gruet P, Seewald W, King JN. Evaluation of subcutaneous and oral administration of robenacoxib and meloxicam for the treatment of acute pain and inflammation associated with orthopedic surgery in dogs. Am J Vet Res. 2011;72:184-93. , 44. Moak P, Hosgood G, Rowe E, Lemke KA. Evaluation of intra-articular and subcutaneous administration of meloxicam for postoperative analgesia following stifle surgery in dogs. Vet Comp Orthop Traumatol. 2011;24:32-8. , 3939. Deneuche AJ, Dufayet C, Goby L, Fayolle P, Desbois C. Analgesic comparison of meloxicam or ketoprofen for orthopedic surgery in dogs. Vet Surg. 2004;33:650-60..
Furthermore, pain recognition and assessment in animals are challenging because of their inability to communicate, the complexity of pain perception and variation in behavioral responses. Different pain scoring systems have been developed to assess the efficacy of analgesic techniques. Several investigators have reported the use of various scales (e.g., visual analog, numerical rating, simple descriptive and composite) developed and correlated specifically for dogs11. Bergmann HM, Nolte I, Kramer S. Comparison of analgesic efficacy of preoperative or postoperative carprofen with or without preincisional mepivacaine epidural anesthesia in canine pelvic or femoral fracture repair. Vet Surg. 2007;36:623-32. , 2323. Holton L, Reid J, Scott EM, Pawson P, Nolan A. Development of a behaviour-based scale to measure acute pain in dogs. Vet Rec. 2001;148:525-31. , 4040. Rialland P, Authier S, Guillot M, Del Castillo JR, Veilleux-Lemieux D, Frank D, Gauvin D, Troncy E. Validation of orthopedic postoperative pain assessment methods for dogs: a prospective, blinded, randomized, placebo-controlled study. PLoS One. 2012;7:1-10. , 4141. Hoelzler MG, Harvey RC, Lidbetter DA, Millis DL. Comparison of perioperative analgesic protocols for dogs undergoing tibial plateau leveling osteotomy. Vet Surg. 2005;34:337-44.. In the current study, two methods (i.e., the GCPS and VAS), were used to obtain a more accurate overall impression of pain. The VAS system is a simple way of scoring pain and is commonly used for both humans and animals. The GCPS system is internationally recognized and has been reported to be as reliable and reproducible for pain assessment in dogs as the VAS system4040. Rialland P, Authier S, Guillot M, Del Castillo JR, Veilleux-Lemieux D, Frank D, Gauvin D, Troncy E. Validation of orthopedic postoperative pain assessment methods for dogs: a prospective, blinded, randomized, placebo-controlled study. PLoS One. 2012;7:1-10.. In addition, only one observer scored all dogs to avoid interobserver variability in the subjective evaluation.
In addition to these factors, sedation may also influence the diagnosis of pain, whereas in animals with an intense degree of sedation, the discomfort can be masked by the lack of a response from the animal4242. Pibarot P, Dupuis J, Grisneaux E, Cuvelliez S, Planté J, Beauregard G, Bonneau NH, Bouffard J, Blais D. Comparison of ketoprofen, oxymorphone, and butorphanol in the treatment of postoperative pain in dogs. J Am Vet Med Assoc. 1997;211:438-44.. In the current study, based on the sedation score, mild to moderate sedation was observed in all treatment groups, with the maximal sedative effect observed from 30 min to 1 hour after extubation. Thus, the sedation appears to have not interfered with the pain evaluation.
Conclusions
Constant rate infusions of lidocaine, morphine or lidocaine plus morphine may be efficacious techniques for pain management for most of the dogs in the first 24 hours post-surgery. However, the two drugs administered together did not provide superior postoperative analgesia or reduce the requirement for rescue opioid compared to a single administration of each drug in dogs undergoing fracture repair.
References
-
1Bergmann HM, Nolte I, Kramer S. Comparison of analgesic efficacy of preoperative or postoperative carprofen with or without preincisional mepivacaine epidural anesthesia in canine pelvic or femoral fracture repair. Vet Surg. 2007;36:623-32.
-
2Kona-Boun JJ, Cuvelliez S, Troncy E. Evaluation of epidural administration of morphine or morphine and bupivacaine for postoperative analgesia after premedication with an opioid analgesic and orthopedic surgery in dogs. J Am Vet Med Assoc. 2006;229:1103-12.
-
3Gruet P, Seewald W, King JN. Evaluation of subcutaneous and oral administration of robenacoxib and meloxicam for the treatment of acute pain and inflammation associated with orthopedic surgery in dogs. Am J Vet Res. 2011;72:184-93.
-
4Moak P, Hosgood G, Rowe E, Lemke KA. Evaluation of intra-articular and subcutaneous administration of meloxicam for postoperative analgesia following stifle surgery in dogs. Vet Comp Orthop Traumatol. 2011;24:32-8.
-
5Shih AC, Robertson S, Isaza N, Pablo L, Davies W. Comparison between analgesic effects of buprenorphine, carprofen, and buprenorphine with carprofen for canine ovariohysterectomy. Vet Anaesth Analg. 2008;35:69-79.
-
6Ortega M, Cruz I. Evaluation of a constant rate infusion of lidocaine for balanced anesthesia in dogs undergoing surgery. Can Vet J. 2011;52:856-60.
-
7Kehlet H, Dahl JB. The value of ''multimodal'' or ''balanced analgesia'' in postoperative pain treatment. Anaesth Analg. 1993;7:1048-563.
-
8Slingsby SL, Waterman-Pearson AE. Analgesic effects in dogs of carprofen and pethidine together compared with the effects of either drug alone. Vet Rec. 2001;148:441-4.
-
9Koppert W, Weigand M, Neumann F, Sittl R, Schuettler J, Schmelz M, Hering W. Perioperative intravenous lidocaine has preventive effects on postoperative pain and morphine consumption after major abdominal surgery. Anesth Analg. 2004;98:1050-5.
-
10Kuo CP, Jao SW, Chen KM, Wong CS, Yeh CC, Sheen MJ, Wu CT. Comparison of the effects of thoracic epidural analgesia and i.v. infusion with lidocaine on cytokine response, postoperative pain and bowel function in patients undergoing colonic surgery. Br J Anaesth. 2006;97:640-6.
-
11Lauwick S, Kim J, Michelagnoli G, Mistraletti G, Feldman L, Fried G, Carli F. Intraoperative infusion of lidocaine reduces postoperative fentanyl requirements in patients undergoing laparoscopic cholecystectomy. Can J Anaesth. 2008;5:754-60.
-
12Groudine SB, Fisher HA, Kaufman RP Jr, Patel MK, Wilkins LJ, Mehta SA, Lumb PD. Intravenous lidocaine speeds the return of bowel function, decreases postoperative pain, and shortens hospital stay in patients undergoing radical retropubic prostatectomy. Anesth Analg. 1998;86:235-9.
-
13Lauwick S, Kim DJ, Mistraletti G, Carli F. Functional walking capacity as an outcome measure of laparoscopic prostatectomy: the effect of lidocaine infusion. Br J Anaesth. 2009;103:213-9.
-
14McKay A, Gottschalk A, Ploppa A, Durieux ME, Groves DS. Systemic lidocaine decreased the perioperative opioid analgesic requirements but failed to reduce discharge time after ambulatory surgery. Anesth Analg. 2009;109:1805-8.
-
15Doherty TJ, Frazier DL. Effect of intravenous lidocaine on halothane MAC in ponies. Equine Vet J. 1998;30:340-3.
-
16Himes RS, DiFazio CA, Burney RG. Effects of lidocaine on the anesthetic requirements for nitrous oxide and halothane. Anesthesiology. 1977;47:437-40.
-
17Muir WW III, Wiese AJ, March PA. Effects of morphine, lidocaine, ketamine, and morphine lidocaine ketamine drug combination on minimum alveolar concentration in dogs anesthetized with isoflurane. Am J Vet Res. 2003;64:1155-60.
-
18Valverde A, Doherty TJ, Hernandez J. Effect of intravenous lidocaine on isoflurane MAC in dogs. Vet Anaesth Analg. 2004;31:264-71.
-
19Matsubara LM, Oliva VN, Gabas DT, Oliveira GC, Cassetari ML. Effect of lidocaine on the minimum alveolar concentration of sevoflurane in dogs. Vet Anaesth Analg. 2009; 407-13.
-
20Doherty TJ, Redua MA, Queiroz-Castro P, Egger C, Cox SK, Rohrbach BW. Effect of intravenous lidocaine and ketamine on the minimum alveolar concentration of isoflurane in goats. Vet Anaesth Analg. 2007;32:125-31.
-
21Pypendop BH, Ilkiw JE. Effects of intravenous lidocaine administration on the minimum alveolar concentration of isoflurane in cats. Anesth Analg. 2005;100:97-101.
-
22Vesal N, Spadavecchia C, Steiner A, Kirscher F, Levionnois OL. Evaluation of the isoflurane-sparing effects of lidocaine infusion during umbilical surgery in calves. Vet Anaesth Analg. 2011;38:451-60.
-
23Holton L, Reid J, Scott EM, Pawson P, Nolan A. Development of a behaviour-based scale to measure acute pain in dogs. Vet Rec. 2001;148:525-31.
-
24MacDougall LM, Hethey JA, Livingston A, Clark C, Shmon CL, Duke-Novakovski T. Antinociceptive, cardiopulmonary, and sedative effects of five intravenous infusion rates of lidocaine in conscious dogs. Vet Anaesth Analg. 2009;36:512-22.
-
25Smith LJ, Bentley E, Shih A, Miller PE. Systemic lidocaine infusion as an analgesic for intraocular surgery in dogs: a pilot study. Vet Anaesth Analg. 2004;31:53-63.
-
26Guedes AG, Rudé EP, Rider MA. Evaluation of histamine release during constant rate infusion of morphine in dogs. Vet Anaesth Analg. 2006;33:28-35.
-
27Kastrup J, Petersen P, Dejgard A, Angelo HR, Hilsted J. Intravenous lidocaine infusion: a new treatment of chronic painful diabetic neuropathy?. Pain. 1987;28:69-75.
-
28Galer BS, Miller KV, Rowbotham MC. Response to intravenous lidocaine infusion differs based on clinical diagnosis and site of nervous system injury. Neurology. 1993;43:1233-5.
-
29Baranowski AP, De Courcey J, Bonello E. A trial of intravenous lidocaine on the pain and allodynia of postherpetic neuralgia. J Pain Symptom Manage. 1999;17:429-33.
-
30Cassuto J, Wallin G, Hogstrom S, Faxén A, Rimbäck G. Inhibition of postoperative pain by continuous low-dose intravenous infusion of lidocaine. Anesth Analg. 1985;64:971-4.
-
31Cepeda MS, Delgado M, Ponce M, Cruz CA, Carr DB. Equivalent outcomes during postoperative patient-controlled intravenous analgesia with lidocaine plus morphine versus morphine alone. Anesth Analg. 1996;83:102-6.
-
32Chia YY, Tan PH, Wang KY, Liu K. Lignocaine plus morphine in bolus patient-controlled intravenous analgesia lacks post-operative morphine-sparing effect. Eur J Anaesthesiol. 1998;15:664-8.
-
33Martin F, Cherif K, Gentili ME, Enel D, Abe E, Alvarez JC, Mazoit JX, Chauvin M, Bouhassira D, Fletcher D. Lack of impact of intravenous lidocaine on analgesia, functional recovery, and nociceptive pain threshold after total hip arthroplasty. Anesthesiology. 2008;109:118-23.
-
34Cohen SP, Mao J. Is the analgesic effect of systemic lidocaine mediated through opioid receptors? Acta Anaesthesiol Scand. 2003;47:910-1.
-
35Hollmann MW, Durieux ME. Local anesthetics and the inflammatory response: a new therapeutic indication? Anesthesiology. 2000;93:858-75.
-
36Hollmann MW, Strumper D, Herroeder S, Durieux ME. Receptors, G proteins, and their interactions. Anesthesiology. 2005;103:1066-78.
-
37Nagy I, Woolf CJ. Lignocaine selectively reduces C fibre-evoked neuronal activity in rat spinal cord in vitro by decreasing N-methyl-D-aspartate and neurokinin receptormediated post-synaptic depolarizations; implications for the development of novel centrally acting analgesics. Pain. 1996;64:59-70.
-
38Sugimoto M, Uchida I, Mashimo T. Local anaesthetics have different mechanisms and sites of action at the recombinant N-methyl-D-aspartate (NMDA) receptors. Br J Pharmacol. 2003;138:876-82.
-
39Deneuche AJ, Dufayet C, Goby L, Fayolle P, Desbois C. Analgesic comparison of meloxicam or ketoprofen for orthopedic surgery in dogs. Vet Surg. 2004;33:650-60.
-
40Rialland P, Authier S, Guillot M, Del Castillo JR, Veilleux-Lemieux D, Frank D, Gauvin D, Troncy E. Validation of orthopedic postoperative pain assessment methods for dogs: a prospective, blinded, randomized, placebo-controlled study. PLoS One. 2012;7:1-10.
-
41Hoelzler MG, Harvey RC, Lidbetter DA, Millis DL. Comparison of perioperative analgesic protocols for dogs undergoing tibial plateau leveling osteotomy. Vet Surg. 2005;34:337-44.
-
42Pibarot P, Dupuis J, Grisneaux E, Cuvelliez S, Planté J, Beauregard G, Bonneau NH, Bouffard J, Blais D. Comparison of ketoprofen, oxymorphone, and butorphanol in the treatment of postoperative pain in dogs. J Am Vet Med Assoc. 1997;211:438-44.
-
1
Research performed at Veterinary Hospital, Department of Veterinary Surgery and Anestesiology, Faculty of Veterinary Medicine, Oeste Paulista University (UNOESTE), Presidente Prudente-SP, Brazil.
-
Financial source: National Council of Technological and Scientific Development (CNPq)
Publication Dates
-
Publication in this collection
Apr 2014
History
-
Received
12 Dec 2013 -
Reviewed
14 Feb 2014 -
Accepted
11 Mar 2014