Abstracts
The essential oil obtained from the stem bark of Croton urucurana Baillon (Euphorbiaceae) was analysed by GC and GC-MS. 83 compounds were identified and borneol (14.7%), bornyl acetate (5.2%), 1-isopropyl-7-methyl-4-methylene-1,3,4,5,6,8-hexahydro-2H-naphthalen-4a-ol (14.7%), sesquicineole (10.5%) and gamma-gurjunene epoxide (5.4%) were the main components. The EC50 value of the crude essential oil in the DPPH free radical scavenging assay was 3.21 mg mL-1. The fraction of the crude essential oil that presented antioxidant activity was purified by prep-TLC on silica gel. GC and GC-MS analysis revealed that alpha-bisabolol (38.3%), alpha-eudesmol (9.3%) and guaiol (8.2%) were the main components of the antioxidant fraction. The EC50 value measured for the bioactive oil fraction in the DPPH assay was 1.05 mg mL-1. The antimicrobial activity of the crude essential oil was assayed against seven Gram-positive and Gram-negative bacteria and three yeasts. Measured MIC values ranged from 1.25 to 10.00 mg mL-1.
Croton urucurana; essential oil; antioxidant; DPPH assay; antimicrobial acticity
O óleo essencial obtido a partir das cascas do caule de Croton urucurana Baillon (Euphorbiaceae) foi analisado por CG e CG-EM. 83 compostos foram identificados, sendo borneol (14,7%), acetato de bornila (5,2%), 1-isopropil-7-metil-4-metileno-1,3,4,5,6,8-hexaidro -2H-naftalen-4a-ol (14,7%), sesquicineol (10,5%) e epóxido de gama-gurjuneno (5,4%) os principais componentes. Para o óleo essencial bruto, foi determinada em 3,21 mg mL-1 a EC50 no ensaio de atividade antioxidante frente ao radical livre DPPH. A fração do óleo essencial bruto que apresentou atividade antioxidante foi purificada por CCD sobre sílica gel. Análises de CG e CG-EM revelaram que alfa-bisabolol (38,3%), alfa-eudesmol (9,3%) e guaiol (8,2%) são os principais componentes da fração antioxidante. A EC50 medida no teste frente ao DPPH foi de 1,05 mg mL-1 para a fração bioativa. Foi determinada a atividade antimicrobiana do óleo essencial bruto frente a sete bactérias Gram-positivas e Gram-negativas e três fungos. Os valores de CIM medidos variaram de 1,25 a 10,00 mg mL-1.
ARTICLE
Chemical composition and evaluation of antibacterial and antioxidant activities of the essential oil of Croton urucurana Baillon (Euphorbiaceae) stem bark
Euclésio SimionattoI, * * e-mail: eusimionatto@yahoo.com.br ; Vanderléa F. L. BonaniI; Ademir Farias MorelIII; Nilva Ré PoppiII; Jorge Luiz Raposo JúniorII; Caroline Z. StukerIII; Gisele M. PeruzzoI; Marize T. L. P. PeresI; Sônia C. HessI
IDepartamento de Hidráulica e Transportes, Universidade Federal de Mato Grosso do Sul, 79070-900 Campo Grande-MS, Brazil
IIDepartamento de Química, Universidade Federal de Mato Grosso de Sul, 79070-900 Campo Grande-MS, Brazil
IIIDepartamento de Química, Universidade Federal de Santa Maria, 97105-900 Santa Maria-RS, Brazil
ABSTRACT
The essential oil obtained from the stem bark of Croton urucurana Baillon (Euphorbiaceae) was analysed by GC and GC-MS. 83 compounds were identified and borneol (14.7%), bornyl acetate (5.2%), 1-isopropyl-7-methyl-4-methylene-1,3,4,5,6,8-hexahydro-2H-naphthalen-4a-ol (14.7%), sesquicineole (10.5%) and g-gurjunene epoxide (5.4%) were the main components. The EC50 value of the crude essential oil in the DPPH free radical scavenging assay was 3.21 mg mL-1. The fraction of the crude essential oil that presented antioxidant activity was purified by prep-TLC on silica gel. GC and GC-MS analysis revealed that a-bisabolol (38.3%), a-eudesmol (9.3%) and guaiol (8.2%) were the main components of the antioxidant fraction. The EC50 value measured for the bioactive oil fraction in the DPPH assay was 1.05 mg mL-1. The antimicrobial activity of the crude essential oil was assayed against seven Gram-positive and Gram-negative bacteria and three yeasts. Measured MIC values ranged from 1.25 to 10.00 mg mL-1.
Keywords:Croton urucurana, essential oil, antioxidant, DPPH assay, antimicrobial acticity
RESUMO
O óleo essencial obtido a partir das cascas do caule de Croton urucurana Baillon (Euphorbiaceae) foi analisado por CG e CG-EM. 83 compostos foram identificados, sendo borneol (14,7%), acetato de bornila (5,2%), 1-isopropil-7-metil-4-metileno-1,3,4,5,6,8-hexaidro -2H-naftalen-4a-ol (14,7%), sesquicineol (10,5%) e epóxido de g-gurjuneno (5,4%) os principais componentes. Para o óleo essencial bruto, foi determinada em 3,21 mg mL-1 a EC50 no ensaio de atividade antioxidante frente ao radical livre DPPH. A fração do óleo essencial bruto que apresentou atividade antioxidante foi purificada por CCD sobre sílica gel. Análises de CG e CG-EM revelaram que a-bisabolol (38,3%), a-eudesmol (9,3%) e guaiol (8,2%) são os principais componentes da fração antioxidante. A EC50 medida no teste frente ao DPPH foi de 1,05 mg mL-1 para a fração bioativa. Foi determinada a atividade antimicrobiana do óleo essencial bruto frente a sete bactérias Gram-positivas e Gram-negativas e três fungos. Os valores de CIM medidos variaram de 1,25 a 10,00 mg mL-1.
Introduction
Croton (Euphorbiaceae) is one of the largest genera of flowering plants, with nearly 1,300 species of herbs, shrubs, and trees that are ecologically prominent and often important elements of secondary vegetation in the tropics and subtropics worldwide.1 Extracts (or infusions) of the different parts of the plant of several species of the genus Croton as C. lechleri, C. palanostigma, C. draconoides and C. urucurana are traditional remedies well known for their healing powers and are extensively used by indigenous cultures of the Amazon River for the treatment of infected wounds and to hasten wound healing.2-8 Its stem bark, when slashed, releases a blood-red sap and, for this reason, C. urucurana and other species of the genus Croton are known as Dragon's Blood (or Sangra d'água in Brazil).
Previous works reported that the essential oils from northeastern Brazilian Croton species, C. zenhtneri, C. nepetaefolius and C. argyrophylloides, exhibited good antioxidant activities.9Croton urucurana Baillon (Euphorbiaceae) is a tree commonly found in Paraguay, northern Argentina, southern Brazil, and Uruguay. Three different products from this species are used primarily in folk medicine: the red sap, the stem bark, and the gum exudate.10
Many pharmacological effects of Croton urucurana have been described in literature, which include wound and ulcer healing, antidiarrhoeic, anticancer, intestinal anti-inflammatory, antioxidant and antirheumatic properties.11-14,28 Peres et al.15 reported the antibacterial activity against Staphylococcus aureus and Salmonella typhimurium of the aqueous-EtOH extract, some fractions of the methanolic extract, catechin and acetyl aleuritolic acid obtained from C. urucurana.
Previous phytochemical analysis of Croton urucurana have identified the presence of acetyl aleuritolic acid, catechin, gallocatechin, sonderianin, b-sitosterol and its glucoside, stigmasterol, campesterol and two novel clerodane diterpenes.13,15,16 Fucoarabinogalactan, a polysaccharide, has been isolated from the gum exudate of C. urucurana.17 In this study, we report the chemical composition, antioxidant29 and antimicrobial activities of the essential oil obtained from the stem bark of Croton urucurana.
Experimental
Plant Material
The stem bark of Croton urucurana was collected in February and March 2006, from biological reserve of Federal University of Mato Grosso do Sul, at the town of Campo Grande, Mato Grosso do Sul state, Brazil. Voucher specimens (16859) have been deposited at the Herbarium of Federal University of Mato Grosso do Sul.
Essential oil isolation
The stem bark was subjected to hydrodistillation for 4 h using a modified Clevenger-type apparatus, followed by exhaustive extraction of the distillate with hexane. After removal of the solvent, the yield of the crude oil was 0.05%. The physical properties for oil were:  : 0.91;
: 0.91;  : 1.56;
: 1.56;  : 12.1 ( in CHCl3, c = 0.025) .
: 12.1 ( in CHCl3, c = 0.025) .
Gas Chromatography/Mass Spectrometry analysis
Analysis of the oil was performed using a Varian GC-MS-MS system comprising a CP-3900 gas chromatograph (Walnut Creek, CA, USA) with a 1077 injector, a CP-8410 autosampler and an ion-trap mass spectrometer (Varian Saturn 2100). Separations were carried out using a ZB-5 (5%-phenyl-95%-dimethylpolysiloxane) fused-silica capillary column (30 m × 0.25 mm i.d., 0.25 µm film thickness) from Phenomenex (Torrance, CA, USA). Oven temperature was programmed from 50 to 250 ºC at 3 ºC min-1. The temperatures of the manifold, GC-MS interface and the ion trap were 70, 240 and 200 ºC, respectively. Helium (99.999%) was used as carrier gas at a constant flow of 1.0 mL min-1 and an injection volume of 1 µL was employed (split ratio of 1:20). The MS scan parameters included electron impact ionization voltage of 70 eV, a mass range of 41-380 m/z and a scan interval of 0.5 s.
A C9-C21n-alkanes mixture diluted in n-hexane was prepared for determination of the temperature programmed retention indices. Samples diluted in n-hexane were analyzed. Internal standards (n-alkanes) were then added to each sample to aid in the standardization of retention times and the samples were analyzed again. Retention indices (RI) for all compounds were determined according to the Van den Dool and Kratz.27
Identification of essential oil constituents
The identification of the components was based on comparison of their mass spectra with those of NIST 2.0 and Saturn Libraries and those described by Adams,18 as well as by comparison of their retention indices with literature data.18
DPPH assay on TLC
Hydrogen atom- or electron-donation ability of the corresponding oils was measured from the bleaching of the purple-colored methanol solution of 2,2-diphenylpicrylhydrazyl (DPPH). Five microliters of a 1:10 dilution of the oils in hexane were applied to the TLC plates (aluminum sheets covered with silica gel 60 F254, Merck) and hexaneethyl acetate (9:1) mixture was used as eluent. The plate was sprayed with a 0.2% DPPH reagent in methanol and left at room temperature for 30 min. Yellow spots formed from bleaching of the purple colour of DPPH reagent were evaluated as positive antioxidant activity.19
Spectrophotometric DPPH assay
This spectrophotometric assay uses stable DPPH radical as a reagent.20,21 Fifty microliters of various concentrations of the oils in methanol were added to 5 mL of a 0.004% methanol solution of DPPH. After a 30 min incubation period at room temperature, the absorbance was read against a blank at 517 nm. Inhibition of DPPH free radical in percent (I%) was calculated in following way:
I % = (Ablank Asample) × 100
where Ablank is the absorbance of the control reaction (containing all reagents except the test compound), and Asample is the absorbance of the test sample. Oil concentration providing 50% inhibition (IC50) was calculated from the graph plotting inhibition percentage against oil concentration. Tests were carried out in triplicate. Commercial standard antioxidant butylated hydroxytoluene (BHT) was also tested against DPPH and used as a reference.
Isolation of antioxidant fraction
For the isolation of the antioxidant compounds, 100 mg of the crude oil were submitted to preparative TLC (silicagel 60 GF254, Merck; hexane-EtOAc, 90:10). The antioxidant fraction was detected by application of the DPPH solution (0.2% in ethanol) on the lateral of the plate. This fraction was withdrawn and washed with EtOAc to give the antioxidant fraction (6 mg).
Isolation of 1-isopropyl-7-methyl-4-methylene-1,3,4,5,6,8-hexahydro-2H-naphthalen-4a-ol (1)
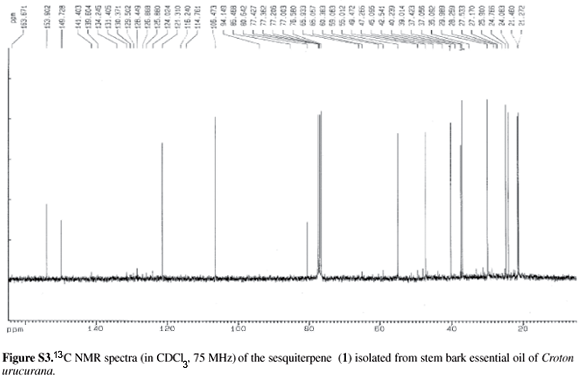
100 mg of crude essential oil of C. urucurana was further submitted to preparative TLC (SiO2; hexane-EtOAc, 85:15) afforded 1 (12 mg). Detection was achieved by UV light (254 nm) and by spraying with solutions of 10% H2SO4 and 2% vanillin in ethanol/H2SO4, followed by heating. The structure of (1) was deduced chiefly from GC-MS analyses and 1H /13C NMR experiments. The relative configuration was additionally confirmed by comparison of the spectral data and retention indices of isomers with those from leterature.25 (1):  : + 4.9 ( in CHCl3, c = 0.0075); 1H NMR (300 MHz, CDCl3): d 5.52 (brs,1H), 4.73 (s, 1H), 4.68 (s, 1H), 2.47-2.51 (m, 2H), 2.22-2.26 (m, 2H), 2.01-2.04 (m, 2H), 1.80-1.85 (m, 2H), 1.71-1.74 (m, 1H), 1.67 (m, 1H), 1.59 (m, 1H), 1.22 (s, 3H), 0.98 (d, J 2.7 Hz, 3H), 0.95 (d, J 2.7 Hz, 3H); 13C NMR (75 MHz, CDCl3) d 21.2, 21,4, 24.0, 24.7, 30.0, 37.0, 37.4, 40.2, 45.0, 55.0, 80.6, 106.4, 121.3, 149.7, 153.9; EI-MS: m/z = 220 [M]+, 202, 187, 159, 134, 119, 91.
: + 4.9 ( in CHCl3, c = 0.0075); 1H NMR (300 MHz, CDCl3): d 5.52 (brs,1H), 4.73 (s, 1H), 4.68 (s, 1H), 2.47-2.51 (m, 2H), 2.22-2.26 (m, 2H), 2.01-2.04 (m, 2H), 1.80-1.85 (m, 2H), 1.71-1.74 (m, 1H), 1.67 (m, 1H), 1.59 (m, 1H), 1.22 (s, 3H), 0.98 (d, J 2.7 Hz, 3H), 0.95 (d, J 2.7 Hz, 3H); 13C NMR (75 MHz, CDCl3) d 21.2, 21,4, 24.0, 24.7, 30.0, 37.0, 37.4, 40.2, 45.0, 55.0, 80.6, 106.4, 121.3, 149.7, 153.9; EI-MS: m/z = 220 [M]+, 202, 187, 159, 134, 119, 91.
Antimicrobial activity
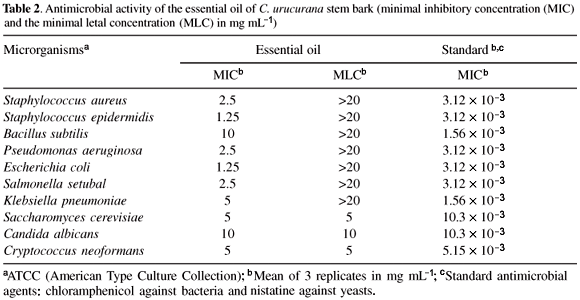
The antibacterial activity of the crude oil was assayed using the broth micro dilution method. A collection of ten microorganisms were used, including four Gram-positive bacteria: Staphylococcus aureus (ATCC 6538p), Staphylococcus epidermidis (ATCC 12228), Pseudomonas aeruginosa (ATCC27853) and Bacillus subtilis (ATCC 6633), three Gram-negative bacteria: Klebsiella pneumoniae (ATCC 10031), Escherichia coli (ATCC 11103) and Salmonella setubal (ATCC 19796), and three yeasts: Saccharomyces cerevisiae (ATCC 2601), Cryptococcus neoformans (ATCC 28952) and Candida albicans (ATCC 10231). Standard strains of microorganisms were obtained from American Type Culture Collection (ATCC), and standard antibiotics chloramphenicol and nistatine were used in order to control the sensitivity of the microbial test.22 The minimal inhibitory concentration (MIC) was determined on 96 well culture plates by a micro dilution method using a microorganism suspension at a density of 105 CFU mL-1 with Casein Soy Broth incubated for 24 h at 37 ºC for bacteria, and Sabouraud Broth incubated for 72 h at 25 ºC for yeasts. The cultures that did not present growth were used to inoculate plates of solid medium (Muller Hinton Agar and Sabouraud Agar) in order to determine the minimal letal concentration (MLC). Proper blanks were assayed simultaneously and samples were tested in triplicate. Technical data have been described previously.23,24
Results and Discussion
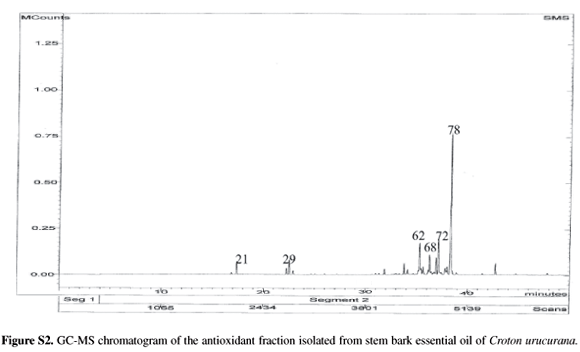
In every extractions were used 100 g of Croton urucurana stem bark and the crude oil yield was 0.05%. The GC-MS analysis led to the identification and quantification of a total of 83 components (Table 1), accounting for 94.6% of the total components present in the crude essential oil of C. urucurana.
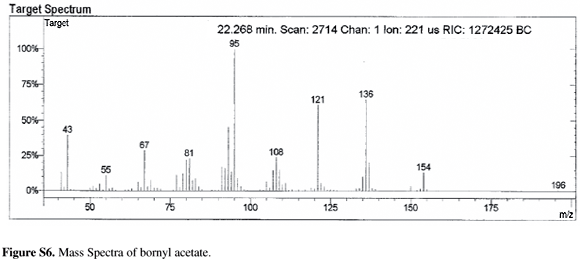
As shown in Table 1, borneol (14.7%), bornyl acetate (5.2%), o-cymene (3.2 %), terpineol (2.8%) and 1,8-cineole (1.8%) were the main monoterpenes identified in the C. urucurana stem bark essential oil. The analysis also showed that the essential oil is constituted by a high proportion of sesquiterpenes, dominated by sesquicineole (10.5%), 1-isopropyl-7-methyl-4-methylene-1,3,4,5,6,8-hexahydro-2H-naphthalen-4a-ol (14.7%), g-gurjunene epoxide (5.4%), a-bisabolol (1.8%), elemenone (1.8%) and a-eudesmol (1.5%).
The crude essential oil of C. urucurana stem bark (100 mg) was subjected to repeated preparative thin layer chromatography on silicagel to afford the derivative of cadinane-type sesquiterpene 1-isopropyl-7-methyl-4-methylene-1,3,4,5,6,8-hexahydro-2H-naphthalen-4a-ol (1, 12 mg), that is the main sesquiterpene component of the essential oil. Compound 1 was obtained as an colorless oil and its structure was established on the basis of the 1H and 13 C NMR spectra. This is the first report on the occurrence of 1 in C. urucurana and in the Euphorbiaceae family, but this compound has previously been isolated from the essential oil of Aglaia odorata Lour (Meliaceae) flowers.25
Next, DPPH free radical scavenging activity of crude essential oil was investigated. The model system of scavenging DPPH free radicals is a simple method to evaluate antioxidant activity. It is accepted that the DPPH free radical scavenging by antioxidants is due to their hydrogen-donating ability.26 The essential oil of C. urucurana stem bark exhibited the IC50 of 3.21 mg mL-1 in the DPPH free radical scavenging activity spectrophotometric assay. This IC50 value reveals that the tested essential oil is a weaker oxidant agent than the commercial standard antioxidant BHT, that presented IC50 0.18 mg mL-1 in the same test system. The isolated sesquiterpene 1-isopropyl-7-methyl-4-methylene-1,3,4,5,6,8-hexahydro-2H-naphthalen-4a-ol (1) showed no antioxidant activity.
Since the crude essential oil of C. urucurana stem bark exhibited antioxidant capacity in the DPPH test, it was submitted to prep-TLC, affording the fraction (6.0 mg) that is responsible for the antioxidant activity in the oil. After isolation, this fraction was analyzed by GC-MS for determination of the components. The GC-MS analysis of the antioxidant fraction of the essential oil of C. urucurana led to the identification and quantification of a total of the 21 major components (Table 1) accounting for 87.1% of the total components present and the main compounds are a-bisabolol (38.3%), a-eudesmol (9.3%) and guaiol (8.2%). These three compounds were present in the crude essential oil of C. urucurana stem bark in very lower yields.
The antioxidant fraction of the crude essential oil exhibited the IC50 of 1.05 mg mL-1 in the DPPH free radical scavenging assay. The measured IC50 indicates that this oil is also a weaker antioxidant agent than BHT.
The antimicrobial activity of the essential oils was examined by broth microdilution susceptibility assay against a panel of 10 microorganisms. The results, presented in Table 2, reveal that the crude essential oil of C. urucurana stem bark inhibited the growth of all microorganisms and that S. epidermidis and E. coli (MIC = 1.25 mg mL-1) were the most sensitive, while B. subtilis and C. albicans were the most resistant microorganisms (MIC =10 mg mL-1).
In comparison to the others essential oils from Croton species,9 the oil of the C. urucurana was more complex in composition. The main compounds found in the essential oil from C. urucurana, such as borneol, bornyl acetate, sesquicineole, g-gurjunene epoxide and the derivative of cadinane-type sesquiterpene (1) did not present in these others species.
Supplemenatry Information
Supplementary data are available free of charge at http://jbcs.sbq.org.br, as PDF file.
Acknowledgments
The authors are grateful to Fundação de Apoio ao Desenvolvimento de Ensino, Ciência e Tecnologia de Mato Grosso do Sul (FUNDECT/MS) and UFMS for the financial support. The authors also thank to CNPq for the IC, AT and DCR scholarships, to CAPES for the MS scholarship and Ubirazilda Resende for botanical support.
Received: September 29, 2006
Web Release Date: July 20, 2007
Supplementary information
- 1. Webster, G. L.; Taxon 1993, 42, 793.
- 2. Chen, Z. P.; Cai, Y.; Phillipson, J D.; Planta Med 1994, 60, 541.
- 3. Pieters, L.; De Bruyne, T.; Van Poel, B.; Vingerhoets Totte, J.; Vanden Berghe, D.; Vlietinck, A.; Phytomedicine1995, 2, 17.
- 4. Esmeraldino, L.E.; Souza, A.M.; Sampaio, S.V.; Phytomedicine 2005, 12, 570.
- 5. Gabriel, S. E.; Davenport, S. E.; Steagall, R. J.; Viaml, V.; Carlson, T.; Rozhon, R. J.; Am. J. Physiol. Gastrointest. Liver Physiol. 1999, 276, 58.
- 6. Holodniy, M.; Koch, J.; Mistal, M.; Schmidt, J. M.; Khandwala, A.; Pennington, J. E.; Porter, S. B.; Am. J. Gastroenterol. 1999, 94, 3267.
- 7. Miller, M.J.S.; MacNaughton, W. K.; Zhang, X. -J.; Thompson, J. H.; Charbonnet, R. M.; Bobrowski, P.; Lao, J.; Trentacosti, A.M.; Sandoval, M.; Am. J. Physiol. Gastrointest. Liver Physiol. 2000, 279, 192.
- 8. Jones, K.; J. Altern. Complement. Med 2003, 9, 877.
- 9. Morais, S. M. de; Catunda Júnior, F. E. A.; da Silva, A. R. A.; Neto, J. S. M.; Rondina, D.; Cardoso, J. H. L.; Quim. Nova 2006, 29, 907.
- 10. Gonzalez Torres, D. M.; Catalogo de plantas Medicinales (Alimenticias y Utiles) Usadas en Paraguay. Asuncion, Paraguay, 1970.
- 11. Gurgel, L. A.; Sidrim, J. J. C.; Martins, D. T.; Filho, V. C;. Rao, V. S.; J. Ethnopharmacol. 2005, 97, 409.
- 12. Gurgel, L. A.; Silva, R. M.; Santos, F. A.; Martins, D. T.; Matos, P. O.; Rao, V. S.; Phytother. Res. 2001, 15, 319.
- 13. Peres, M. T. L. P.; Delle Monache, F.; Pizzolatti, M. G.; Santos, A. R. S.; Beirith, A.; Calixto, J. B.; Yunes, R. A.; Phytother. Res. 1998, 12, 209.
- 14. Orlandi-Mattos, P. E.; Geremias, R.; Cordova, C. A. S.; Creczynski-Pasa, T. B., Rebello, J. M.; Wilhem Filho, D.; Martins, D. T. O.; Llesuy, S.; Pedrosa, R. C.; Free Radical Biol. Med. 2002, 33, 645.
- 15. Peres, M.T.L.P.; Delle Monache, F.; Cruz, A.B.; Pizzolatti, M.G.; Yunes, R.A.; J. Ethnopharmacol. 1997, 56, 223.
- 16. Peres, M.T.L.P.; Pizzolatti, M.G.; Yunes, R.A.; Delle Monache, F.; Phytochemistry 1998, 49, 171.
- 17. Milo, B.; Risco, E.; Vila, R., Iglesias, J.; Canigueral, S.; J. Nat. Prod. 2002, 65, 1143.
- 18. Adams, R. P.; Identification of Essential Oil Components by Gas Chromatography/Mass Spectroscopy, Allured Publishing: Illinois, 1995.
- 19. Mimica-Dukic, N.; Bozin, B.; Sokovic, M.; Mihajlovic, B.; Matavulj, B.; Planta Med. 2003, 69, 413.
- 20. Burits, M.; Bucar. F.; Phytother. Res. 2000, 14, 323.
- 21. Cuendet, M.; Hostettmann, K.; Potterat, O.; Helv. Chim. Acta 1997, 80, 1144.
- 22. FDA: Code of Federal Regulation 1991, 21, 300.
- 23. Hammer, K.A.; Carson, C.F.; Riley, T.V.; J. Appl. Microbiol. 1999, 86, 985.
-
24National Committee for Clinical Laboratory Standards: Reference Method of Broth Dilution Antifungal Susceptibility Testing of Yeasts Standard M77-T, NCCLS: Villanova,1995.
- 25. Weyerstahl, P.; Marschall, H.; Son, P. T.; Giang, P. M.; Flav. Fragr. J. 1999, 14, 219.
- 26. Chen, C. W.; Ho, C. T.; J. Food Lip. 1995, 2, 35.
- 27. Van den Dool, H.; Kratz, P. D.; J. Chromatogr 1963, 11, 463.
- 28. Salatino, A.; Salatino, M. L. F.; Negri, G.; J. Braz. Chem. Soc., 2007, 18, 11.
- 29. Núnez-Sélles, A. J.; J. Braz. Chem. Soc. 2005, 16, 699.
Publication Dates
-
Publication in this collection
25 Oct 2007 -
Date of issue
2007
History
-
Accepted
20 July 2007 -
Received
29 Sept 2006