Abstracts
Because of their potential for N2 biological fixation, legumes are an alternative source of nitrogen to crops, and can even replace or supplement mineral fertilization. A greenhouse experiment was carried out to evaluate temporal patterns of velvet bean (Mucuna aterrima) green manure release of nitrogen to rice plants, and to study the fate of nitrogen from velvet bean in rice cultivation. The isotopic dilution methodology was used. Treatments consisted of a control and 10 incubation periods of soil fertilized with 15N-labeled velvet bean (0, 20, 40, 60, 90, 120, 150, 180, 210, and 240 days). The plant material was previously chopped, sifted (10 mm mesh sieve) and oven-dried (65ºC). Incubation of the plant material (2.2 g kg-1 soil) was initiated by the longest period, in order to synchronize the planting of the test crop, rice (Oryza sativa), at time zero for all treatments. Green manure incorporation promoted increases in rice dry matter yield and nitrogen uptake. These variables showed maximum values at incubation periods of 38 and 169 days, respectively. Green manure nitrogen utilization by rice plants was highest at an incubation period corresponding to 151 days. More than 60% of the green manure nitrogen remained in the soil after rice cultivation. The highest green manure nitrogen recovery from the soil-plant system occurred at an incubation period equivalent to 77 days.
Mucuna aterrima; effectiveness; recovery; losses
Em função de seu potencial de fixação de nitrogênio (N), as leguminosas representam uma alternativa ao suprimento do nutriente para as culturas, substituindo ou complementando a adubação mineral. Desenvolveu-se, sob condições semi-controladas, utilizando-se de metodologia de diluição isotópica, um experimento para determinar o padrão temporal de liberação de nitrogênio da mucuna-preta (Mucuna aterrima) e estudar a dinâmica do N contido nesse adubo verde no sistema solo-planta. O estudo compreendeu um tratamento testemunha e dez períodos de incubação do solo com mucuna-preta marcada com 15N (zero, 20, 40, 60, 90, 120, 150, 180, 210 e 240 dias). O material vegetal utilizado foi picado, peneirado (peneira com abertura de malha de 10 mm) e seco em estufa. A incubação do material vegetal, na dose de 2,2 g kg-1 de solo, foi iniciada pelo período mais longo, possibilitando coincidir o plantio da cultura teste, arroz (Oryza sativa), para todos os tratamentos, no tempo zero. A incorporação de mucuna-preta promoveu aumento da produção de matéria seca e da absorção de N pelas plantas de arroz. Os valores máximos para essas variáveis foram proporcionados pelos períodos de incubação de 38 e 169 dias, respectivamente. O máximo aproveitamento de N da mucuna-preta pelo arroz correspondeu ao período de incubação de 151 dias. Mais de 60% do N do adubo verde permaneceram no solo após a colheita do arroz. A recuperação máxima de N da mucuna-preta no sistema solo-planta correspondeu a um período de incubação de 77 dias.
Mucuna aterrima; eficiência; recuperação; perdas
SOILS AND PLANT NUTRITION
Fate of nitrogen (15N) from velvet bean in the soil-plant system
Dinâmica do nitrogênio (15N) da mucuna-preta no sistema solo-planta
Walkyria Bueno ScivittaroI, * * Corresponding author < wbscivit@cpact.embrapa.br> ; Takashi MuraokaII; Antonio Enedi BoarettoII; Paulo César Ocheuze TrivelinII
IEmbrapa Clima Temperado, C.P. 403 - 96001-970 - Pelotas, RS - Brasil
IIUSP/CENA - Lab. de Fertilidade do Solo, C.P. 96 - 13400-970 - Piracicaba, SP - Brasil
ABSTRACT
Because of their potential for N2 biological fixation, legumes are an alternative source of nitrogen to crops, and can even replace or supplement mineral fertilization. A greenhouse experiment was carried out to evaluate temporal patterns of velvet bean (Mucuna aterrima) green manure release of nitrogen to rice plants, and to study the fate of nitrogen from velvet bean in rice cultivation. The isotopic dilution methodology was used. Treatments consisted of a control and 10 incubation periods of soil fertilized with 15N-labeled velvet bean (0, 20, 40, 60, 90, 120, 150, 180, 210, and 240 days). The plant material was previously chopped, sifted (10 mm mesh sieve) and oven-dried (65ºC). Incubation of the plant material (2.2 g kg-1 soil) was initiated by the longest period, in order to synchronize the planting of the test crop, rice (Oryza sativa), at time zero for all treatments. Green manure incorporation promoted increases in rice dry matter yield and nitrogen uptake. These variables showed maximum values at incubation periods of 38 and 169 days, respectively. Green manure nitrogen utilization by rice plants was highest at an incubation period corresponding to 151 days. More than 60% of the green manure nitrogen remained in the soil after rice cultivation. The highest green manure nitrogen recovery from the soil-plant system occurred at an incubation period equivalent to 77 days.
Key words:Mucuna aterrima, effectiveness, recovery, losses
RESUMO
Em função de seu potencial de fixação de nitrogênio (N), as leguminosas representam uma alternativa ao suprimento do nutriente para as culturas, substituindo ou complementando a adubação mineral. Desenvolveu-se, sob condições semi-controladas, utilizando-se de metodologia de diluição isotópica, um experimento para determinar o padrão temporal de liberação de nitrogênio da mucuna-preta (Mucuna aterrima) e estudar a dinâmica do N contido nesse adubo verde no sistema solo-planta. O estudo compreendeu um tratamento testemunha e dez períodos de incubação do solo com mucuna-preta marcada com 15N (zero, 20, 40, 60, 90, 120, 150, 180, 210 e 240 dias). O material vegetal utilizado foi picado, peneirado (peneira com abertura de malha de 10 mm) e seco em estufa. A incubação do material vegetal, na dose de 2,2 g kg-1 de solo, foi iniciada pelo período mais longo, possibilitando coincidir o plantio da cultura teste, arroz (Oryza sativa), para todos os tratamentos, no tempo zero. A incorporação de mucuna-preta promoveu aumento da produção de matéria seca e da absorção de N pelas plantas de arroz. Os valores máximos para essas variáveis foram proporcionados pelos períodos de incubação de 38 e 169 dias, respectivamente. O máximo aproveitamento de N da mucuna-preta pelo arroz correspondeu ao período de incubação de 151 dias. Mais de 60% do N do adubo verde permaneceram no solo após a colheita do arroz. A recuperação máxima de N da mucuna-preta no sistema solo-planta correspondeu a um período de incubação de 77 dias.
Palavras-chave:Mucuna aterrima, eficiência, recuperação, perdas
INTRODUCTION
The inclusion of legumes as green manures into the management of cropping systems has drawn the attention of the scientific community because of their potential for the biological fixation of nitrogen. This practice represents a promising alternative to supplying N to crops, either as replacement or as supplementation for mineral fertilization. However, only adequate management practices elicit possible to derive maximum benefits from green manures, so as to supply the crops with their nitrogen requirements and thus sustain high yields. To that effect, the availability of information on the nitrogen supplying capacity and its temporal patterns of release by green manures, as well as on the fate of the N contained by them in the soil-plant system, is absolutely crucial.
The nitrogen applied to the soil in the form of green manures may take different pathways: part is absorbed by the plants, part is lost from the soil-plant system, and the rest remains in the soil, as non-decomposed residues, or is immobilized by microorganisms, and gradually converted into stable forms. The magnitude of these transformations varies depending on environmental conditions (Azam et al., 1985) and quality of the plant residues (Smith & Sharpley, 1993).
Under field conditions, most of the N that comes from plant residues remains in the soil, mainly in the organic form. In general, the recovery of N from green manures by the subsequent crop is low, ranging from 5% to 30% (Müller & Sundman, 1988; Harris & Hesterman, 1990; Bremer & Kessel, 1992; Harris et al., 1994). In potted experiments, the values obtained are higher, up to 55% (Azam et al., 1985; Janzen & Schaalje, 1992), which must be credited to the presence of more favorable conditions for residue decomposition, and to greater control over losses. The availability of residual N for a subsequent cultivation is rarely above 10% of the total amount applied (Müller & Sundman, 1988; Ta & Faris, 1990). There is considerable variation in the results of nitrogen from green manures that can be recovered or is lost from the soil-plant system, after one or more cultivations, ranging, respectively, from 66% to 102% and from 6% to 41% (Ladd et al., 1981; Azam et al., 1985; Harris & Hesterman, 1990).
The main routes through which N losses occur from the soil-plant system are leaching, denitrification, and ammonia volatilization (Ladd et al., 1981; Azam et al., 1985; Harris & Hesterman, 1990). In sandy-textured soils, nitrate leaching is the most important N loss mechanism of the system; on the other hand, great part of the N losses is attributed to denitrification in clayey-textured and poorly drained soils (Ladd et al., 1981; Harris & Hesterman, 1990). Furthermore, there have been reports of N losses by surface runoff and by gaseous exchanges in the above-ground part of plants (Janzen & McGinn, 1991; Bremer & Kessel, 1992). The potential for N losses in residues from decomposing plants on the soil surface is considerable (Bremer & Kessel, 1992), but becomes negligible, however, when the residues are incorporated (Janzen & McGinn, 1991).
Based on what has been considered above, an experiment was developed to determine the temporal pattern of N release from velvet bean and to study the fate of the nitrogen contained in this green manure crop in the soil-plant system.
MATERIAL AND METHODS
The experiment was carried out under greenhouse conditions in Piracicaba, SP, Brazil. It comprised a preliminary stage, during which velvet bean (Mucuna aterrima) was grown, marked with 15N, and then incorporated to soil samples, followed by a rice cultivation stage. The experiment used a surface sample (0.0-0.2 m) of a Rhodic Hapludox with the following chemical composition: 1.4 g kg-1 N (Parkinson & Allen, 1975); 21 g dm-3 organic matter; pH in CaCl2 0.01 mol L-1(1:2.5) 4.4; 52 mg dm-3 P; 4.3 mmolc dm-3 K; 20 mmolc dm3 Ca; 13 mmolc dm-3 Mg, and 59 mmolc dm-3 H+Al (Raij et al., 1987).
Velvet bean marked with 15N was produced in pots containing 5.0 kg of air-dried soil fertilized with 320 mg kg-1 P, in the form of triple superphosphate. Six seeds of velvet bean were used per pot, and three plants were left after thinning. Next, the legume was marked through the application of a mixture of urea and ammonium sulfate solutions containing 10 mg kg-1 N and enriched, respectively, to 10.2% and 5.4% in 15N atoms. Applications of 10 mg kg-1 N, in the form of 15N-urea solutions, were also made at 32, 44, 55, 60, and 65 days after the emergence of plants (DAE). On the first three applications, the urea showed an isotopic enrichment of 10.2% 15N atoms and on the other applications, enrichment was circa 30.2% 15N atoms.
The plants were harvested at 80 DAE; harvesting consisted of cutting the aerial part of plants and removing their roots, which were washed with both tap and distilled water. All plant material was air-dried, chopped, passed through a 10 mm mesh sieve, dried in a forced air circulation oven at 65ºC, and weighed for determination of dry matter yield.
Samples of the dried material were ground and analyzed to determine fiber and lignin contents by the fiber acid detergent method (Van Soest & Wine, 1968); polyphenols by the Folin-Denis method (Swain & Hillis, 1959); total C and nutrients contents (Bataglia et al., 1983), and isotopic abundance of 15N (Trivelin et al., 1973). Results obtained were: 42.8% fiber; 10.5% lignin; 55.2% polyphenols; 372 g kg1 C; 25.4 g kg-1 N; 2.8 g kg-1 P; 12.2 g kg-1 K; 19.8 g kg-1 Ca; 3.2 g kg-1 Mg; 2.7 g kg-1 S; 11 mg kg-1 Cu; 1128 mg kg-1 Fe; 996 mg kg-1 Mn, 46 mg kg-1 Zn, and isotopic enrichment of 2.383% in 15N atoms.
The treatments were then applied, consisting of ten incubation periods of soil containing 15N-marked velvet bean (zero, 20, 40, 60, 90, 120, 150, 180, 210, and 240 days) and one control without green manure, aiming to evaluate their potential in supplying N to the soil. The treatments were arranged in a completely randomized experimental design with four replicates. Pots containing 2 kg soil each represented the experimental plots. Incubation of the marked velvet bean, at the dose of 2.2 g dry matter per kg soil, started with the longest period, thus allowing the planting of the test crop to be synchronized, at time zero, for all treatments. With the exception of the control, all other treatments were moistened during the incubation period; soil moisture was kept near 70% of maximum water retention capacity.
Four rice plants, cultivar IAC-165, grown in washed sand for 44 days, were transplanted into each pot and then grown for 39 days. Phosphate and potassium fertilization was omitted, because of the high initial contents of P (52 mg dm-3) and K (4.3 mmolc dm-3) of the soil. Harvesting the rice involved cutting the aerial part of the plants and removing their roots, which were washed in both tap and distilled water, dried in a forced air circulation oven at 65ºC, and then weighed. The plant material was analyzed to determine N content (Bataglia et al., 1983) and 15N isotopic abundance, by the modified Dumas technique (Trivelin et al., 1973). Next, samples were taken from the soil in the pots to determine N content (Parkinson & Allen, 1975) and 15N isotopic abundance, by the Rittemberg method (Trivelin et al., 1973).
The treatments were evaluated based on their dry matter yield and N absorption by the rice plants, amount of N in the rice from the green manure, green manure nitrogen utilization by the rice, and percentage of nitrogen from the residual green manure in the soil. By associating the results relative to the rice plants with those relative to the soil, the percentages and amounts of N from the green manure recovered and lost from the soil-plant system were determined. The sum of the N amounts in the rice from the green manure and the residual N in the soil was considered as recovered N; the remainder, relative to the total added, was considered as lost from the soil-plant system. The ratios between those amounts and the amount of N incorporated into the soil by the green manure, expressed as percentages, were called the percentage of N from the green manure recovered, and percentage lost, respectively, from the soil-plant system.
The statistical analysis of the results consisted of analyses of variance and, after checking for significance through the F test, the Dunnett test at 5% was applied to discriminate the effects of treatments involving incorporation of green manure relative to the control. Polynomial regression analyses were also carried out to compare the different levels of the incubation period factor.
RESULTS AND DISCUSSION
Dry matter yield and nitrogen utilization by the rice
Velvet bean incorporation increased dry matter yield of rice plants relatively to the control, except for the 240-day incubation period (Table 1). This behavior confirms results obtained by Diekmann et al. (1996) and Ribeiro (1996), while studying the effect of green manure species on rice crop. An effect of incubation period on dry matter yield of the aerial part and whole rice plants (aerial part + roots) was observed. However, no influence on the dry matter yielded by the roots alone was observed (Figure 1). For both variables, the quadratic model provided the best fitting of the results. From the equation relative to the aerial part + roots, the maximum dry matter yield of rice plants was determined, corresponding to an incubation period of 38 days. Therefore, management for this green manure crop should be made for a short period of time just prior to planting the subsequent crop, to optimize yield.
The higher dry matter yield values obtained in the treatments with shorter incubation periods indicate that velvet beans undergo quick decomposition and release of nutrients. This behavior is associated with high contents of nitrogen accumulated in the dry matter from the green manure, connecting low C:N, lignin:N and polyphenol:N ratios to it, which favor mineralization of the organic material (Oliveira et al., 2002). The incorporation of plant residues into the soil, favoring microbial actin, must also have contributed toward this performance, similarly to what was reported by Wilson & Hargrove (1986). On the other hand, the equalization of the effect of the 240-day incubation period treatment and the effect observed for the control (Table 1) suggests that, as time passes, part of the nutrients in the green manure released to the system becomes unavailable, possibly because of microbial immobilization. Roots were responsible for approximately 30% of the dry matter yielded by the rice plants, demonstrating that it is important to consider root dry matter yield when evaluating treatments (Figure 1).
The velvet bean incorporation into the soil provided an increase in the amounts of nitrogen accumulated in the aerial part, and in the aerial part + roots of rice plants, as compared to the control, except for the day-zero incubation period treatment (Table 1). This is an indication of the nitrogen supplying potential of the green manure utilized.
The influence of the incubation period on the amounts of nitrogen accumulated in the aerial part, and in the aerial part + roots was described by quadratic models (Table 2). The maximum N accumulation in the rice plants was determined to occur at an incubation period of 169 days. However, satisfactory results were obtained starting at 90 days. The incubation period that provided maximum N accumulation by the rice plants was far superior to the 60 days reported by Ribeiro (1996), who worked with the same green manure crop on an Oxisol. These observed distinctions are possibly associated with differences in environmental conditions (Wilson & Hargrove, 1986) and between the soils that were utilized (Mary et al., 1996), thus conditioning distinct nitrogen mineralization rates.
Similarly to what was observed for dry matter yield, the previous incorporation of green manure into the soil was required for the release of N to the system to occur within the period of highest demand by the subsequent crop. In this case, however, the most favorable time period was longer. Because of, this difference, in addition to the release of N by the green manure, other factors influenced dry matter yield by rice plants.
More than 80% of the nitrogen absorbed by the plants was translocated to the aerial part, and small differences were observed between treatments with regard to the distribution of the absorbed N between the aerial part and the roots. Only results for the amount of green manure N in the aerial part of rice plants and in the aerial part + roots were influenced by the incubation period (Table 2). The behavior of these variables was similar to what was observed for nitrogen accumulated by the plants. The results were fitted by quadratic models, and a 147-day period was determined to provide the maximum amount of N from the green manure in rice plants.
The mean contribution of nitrogen from the green manure to rice plants was 25%, which can be considered a high value when compared to values obtained in field experiments. However, this value is appropriate for potted studies, where limited volumes of soil are used, which stimulates green manure decomposition and, as a consequence, also stimulates its contribution as a nutrient supply to plants. In addition, the chemical composition of the green manure and the use of rice seedlings produced during lengthy periods on a substrate practically devoid of nutrients, must also have contributed to high N absorption from the velvet bean. In a similar study, conducted under greenhouse conditions using the green manures velvet bean and tropical kudzu, the maximum N value from the green manure determined in rice plants was 11.6% (Ribeiro, 1996). That author did not consider, however, the N contained in the rice plant roots. On the other hand, Azam et al. (1985), growing maize in pots, determined N contributions from green manure higher than 40%. The differences in the results obtained for rice and maize plants can be explained in part by the differential ability between plant species in utilizing N from green manures, as proposed by Ta & Faris (1990).
No effect of the incubation period on N utilization by the roots was observed (Table 2). However, results relative to the aerial part of the plants and the aerial part + roots were influenced by the variations in incubation period length, and are described by quadratic models. For the aerial part + roots, the maximum utilization of N from the green manure occurred at an incubation period corresponding to 151 days. The incorporation of green manure into the soil for periods above 150 days reduced the efficiency of utilization of N from the velvet bean by rice, possibly because of microbial immobilization and/or to increasing nutrient losses to the atmosphere.
Even though the utilization of N from the green manure by the rice plants increased up to the 150-day period, for incubation periods of up to 90 days, the increments were higher, demonstrating a rapid release of N from the green manure. Therefore, very long incubation periods are not required to obtain high nitrogen utilization efficiency from the velvet bean.
Values found for utilization of N from the green manure by the rice plants, 20% on average, were comparable to those reported by other authors, when working under controlled conditions (Janzen & Schaalje, 1992; Rees et al., 1993); such values, however, were higher than those described by Ladd et al. (1981; 1983) for field experiments, where conditions are less favorable for the decomposition of green manures, and susceptibility to N losses is higher (Janzen & Schaalje, 1992; Rees et al., 1993).
Nitrogen from residual green manure in the soil
The effect of incubation period on the percentages of N from the residual green manure in the soil after the rice had been harvested fitted to a linear model, with values decreasing as the incubation period increased (Table 3). Despite the fact that the decomposition and release of nitrogen from the velvet bean residues was relatively easy, characterized by low C:N, lignin:N (Constantinides & Fownes, 1994), and polyphenol:N ratios (Oglesby & Fownes, 1992), these are long and gradual processes that extend beyond the subsequent cropping cycle.
The mean N percentage from the residual green manure in the soil was in excess of 60% of the amount of nutrient incorporated into the soil, in agreement with observations by Harris & Hesterman (1990), Harris et al. (1994), Ladd et al. (1981; 1983), and Müller & Sundman (1988), who reported that most of the N contained in green manures remains in the soil after an initial cropping cycle.
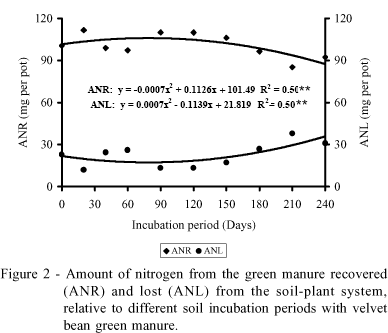
Nitrogen from the green manure recovered and lost from the soil-plant system
The amounts of nitrogen from the green manure recovered and lost from the soil-plant system were influenced by incubation period, and the results were fitted by quadratic models (Figure 2). The maximum N recovery from the velvet bean occurred at the 80-day incubation period, corresponding to 86% of the nitrogen contained in the green manure. Since recovery and loss are complementary, the behavior verified for losses of N from the green manure was the opposite of that described for the recovery results.
Nitrogen recovery from the green manure reflects variations observed for the variable utilization of N from the green manure by rice plants (Table 2), thus confirming the negative effect of long incubation periods on N recovery from the velvet bean. This behavior is possibly a consequence of greater susceptibility to N losses to the atmosphere in treatments involving longer incubation periods. The importance of synchronizing the periods of nitrogen release from the green manure with intake by the subsequent crop also becomes evident, in order to increase the efficiency of utilization of the nutrient from the green manure.
The values determined for N recovery from the green manure are intermediate, relative to the intervals described in the literature, which vary from 66% to 102% (Azam et al., 1985; Harris & Hesterman, 1990; Ladd et al., 1981). Considering the conditions under which the experiment was conducted, where the environment was partially controlled, the losses of nitrogen that occurred are attributed to denitrification and ammonia volatilization, since the leaching process was prevented. Additionally, it is more likely that a small amount of losses resulted from a less than complete removal of roots from the test crop, leading to an underestimation of the results of N recovery by the rice plants.
ACKNOWLEDGEMENTS
To Fundação de Amparo à Pesquisa do Estado de São Paulo and to IAEA - International Atomic Energy Agency, for providing financial support for this research.
Received March 13, 2003
Accepted February 02, 2004
- AZAM, F.; MALIK, K.A.; SAJJAD, M.I. Transformations in soil and availability to plants of 15N applied as inorganic fertilizer and legume residues. Plant and Soil, v.86, p.3-13, 1985.
- BATAGLIA, O.C.; FURLANI, A.M.C.; TEIXEIRA, J.P.F.; FURLANI, P.R.; GALLO, J.R. Métodos de análise química de plantas Campinas: IAC, 1983. 48p. (Boletim Técnico, 78).
- BREMER, E.; KESSEL, C. van. Plant-available nitrogen from lentil and wheat residues during a subsequent growing season. Soil Science Society of America Journal, v.56, p.1155-1160, 1992.
- DIEKMANN, K.H.; OTTOW, J.C.G.; DE DATTA, S.K. Yield and nitrogen response of lowland rice (Oryza sativa L.) to Sesbania rostrata and Aeschynomene afraspera green manure in different marginally productive soils in the Philippines. Biology and Fertility of Soils, v.21, p.103-108, 1996.
- HARRIS, G.H.; HESTERMAN, O.B. Quantifying the nitrogen contribution from alfafa to soil and two succeeding crops using nitrogen-15. Agronomy Journal, v.82, p.129-134, 1990.
- HARRIS, G.H.; HESTERMAN, O.B.; PAUL, E.A.; PETERS, S.E.; JANKE, R.R. Fate of legume and fertilizer nitrogen-15 in a long term cropping systems experiment. Agronomy Journal, v.86, p.910-915, 1994.
- JANZEN, H.H.; McGINN, S.M. Volatile loss of nitrogen during decomposition of legume green manure. Soil Biology and Biochemistry,v.23, p.291-297, 1991.
- JANZEN, H.H.; SCHAALJE, G.B. Barley response to nitrogen and non-nutritional benefits of legume green manure. Plant and Soil, v.142, p.19-30, 1992.
- LADD, J.N.; OADES, J.M.; AMATO, M. Distribution and recovery of nitrogen from legume residues decomposing in soils sown to wheat in field. Soil Biology and Biochemistry, v.13, p.251-256, 1981.
- LADD, J.N.; AMATO, M.; JACKSON, R.B.; BUTLER, J.H.A. Utilization by wheat crops of nitrogen from legume residues decomposing in soils in the field. Soil Biology and Biochemistry, v.15, p.231-238, 1983.
- MARY, B.; RECOUS, S.; DARWIS, D.; ROBIN, D. Interactions between decomposition of plant residues and nitrogen cycling in soil. Plant and Soil , v.181, p.71-82, 1996.
- MÜLLER, M.M.; SUNDMAN, V. The fate of nitrogen (15N) released from different plant materials during decomposition under field conditions. Plant and Soil, v.105, p.133-139, 1988.
- OGLESBY, K.A.; FOWNES, J.H. Effects of chemical composition on nitrogen mineralization from green manures of seven tropical leguminous trees. Plant and Soil, v.143, p.127-132, 1992.
- OLIVEIRA, F.H.T.; NOVAIS, R.F.; ALVAREZ V., V.H.; CANTARUTTI, R.B.; BARROS, N.F. Fertilidade do solo no sistema plantio direto. In: ALVAREZ V., V.H.; SCHAEFER, C.E.G.R.; BARROS, N.F. ; MELLO, J.W.V. de; COSTA, L.M. da (Ed.) Tópicos em ciência do solo Viçosa: SBCS, 2002. v.2, p.394-486.
- PARKINSON, J.A.; ALLEN, S.E. A wet oxidation procedure suitable for the determination of nitrogen and mineral nutrients in biological material. Communications in Soil Science and Plant Analysis, v.6, p.1-11, 1975.
- RAIJ, B. van; QUAGGIO, J.A.; CANTARELLA, H.; FERREIRA, M.E.; LOPES, A.S.; BATAGLIA, O.C. Análise química do solo para fins de fertilidade. Campinas: Fundação Cargill, 1987. 170p.
- REES, R.M.; YAN, L.; FERGUSON, M. The release and plant uptake of nitrogen from some plant and animal manures. Biology and Fertility of Soils, v.15, p.285-293, 1993.
- RIBEIRO, G. de. Efeito de períodos de incubação de adubos verdes (mucuna e puerária) na liberação de nitrogênio (15N) e enxofre (35S) para o arroz. Piracicaba: USP/CENA, 1996. 82p. (Dissertação - Mestrado).
- SMITH, S.J.; SHARPLEY, A.N. Nitrogen availability from surface-applied and soil-incorporated crop residues. Agronomy Journal, v.85, p.776-778, 1993.
- SWAIN, T.; HILLIS, W.E. The phenolic constituents of Prunus domestica I. The quantitative analysis if phenolic constituentes. Journal of the Science of Food and Agriculture, v.10, p.63-68, 1959.
- TA, T.C.; FARIS, M.A. Availability of N from 15N-labeled alfafa residues to three succeding barley crops under field conditions. Soil Biology and Biochemistry, v.23, p.835-838, 1990.
- TRIVELIN, P.C.O.; SALATI, E.; MATSUI, E. Preparo de amostras para análise de 15N por espectrometria de massas Piracicaba: CENA, 1973. 41p. (Boletim Técnico, 2).
- Van SOEST, P.J.; WINE, R.H. Determination of lignin and cellulose in acid-detergent fiber with permanganate. Journal of the AOAC, v.51, p.780-785, 1968.
- WILSON, D.O.; HARGROVE, W.L. Release of nitrogen from crimson clover residue under two tillage systems. Soil Science Society of America Journal, v.50, p.1251-1254, 1986.
Publication Dates
-
Publication in this collection
14 May 2004 -
Date of issue
Apr 2004
History
-
Accepted
02 Feb 2004 -
Received
13 Mar 2003