Abstract
Toxic levels of Cd can cause protein denaturation and oxidative stress, which result in membrane damage, enzimatic activity changes and other metabolic damage. Some plants may show alteration in the activity of their antioxidant enzymes as a heavy metal tolerance mechanism. This study aims at evaluating the role of enzymes of the antioxidant system in adaptive responses of the accumulator P. glomerata species to levels of cadmium (Cd). Plants were cultivated in nutrient solutions containing concentrations of Cd in the form of CdSO4 (0, 45 and 90 µmol L-1), for 20 d. Cd concentrations and yields of root and shoot dry matter were determined at the end of the experiment. Malondialdehyde (MDA) production and the activities of antioxidant enzymes were determined after days 1, 12 and 20. Higher Cd concentrations in tissues of P. glomerata were found to reduce biomass production in both roots and shoots. The lipid peroxidation rates in leaves and roots were smaller at the start of the experiment for all Cd levels. Superoxide dismutase (SOD) activity increased in leaves on day 1 and in roots on day 20 as Cd levels increased. Cd stress induced an increase in the activity of APX in leaves, whereas in roots ascorbate peroxidase (APX) activity was reduced at high concentration of Cd. At the end of the experiment, catalase (CAT) activity in leaves was reduced as Cd concentration increased. Nevertheless, the glutathione reductase (GR) and guaiacol peroxidase (GPX) activities increased. In roots, GR activity was reduced on days 1 and 20.
antioxidant enzymes; oxidative stress; tolerance; cadmium toxicity
PLANT PHYSIOLOGY AND BIOCHEMISTRY
Antioxidant system of ginseng under stress by cadmium
Teresa Cristina Lara Lanza de Sá e Melo MarquesI; Angela Maria SoaresII,* * Corresponding author < amsoares@dbi.ufla.br>
IUFLA - Depto. de Ciência do Solo, C.P. 3037 - 35700-000 - Lavras, MG - Brasil
IIUFLA - Depto. de Biologia - Setor Fisiologia Vegetal, C.P. 3037 - 35700-000 - Lavras, MG - Brasil
ABSTRACT
Toxic levels of Cd can cause protein denaturation and oxidative stress, which result in membrane damage, enzimatic activity changes and other metabolic damage. Some plants may show alteration in the activity of their antioxidant enzymes as a heavy metal tolerance mechanism. This study aims at evaluating the role of enzymes of the antioxidant system in adaptive responses of the accumulator P. glomerata species to levels of cadmium (Cd). Plants were cultivated in nutrient solutions containing concentrations of Cd in the form of CdSO4 (0, 45 and 90 µmol L-1), for 20 d. Cd concentrations and yields of root and shoot dry matter were determined at the end of the experiment. Malondialdehyde (MDA) production and the activities of antioxidant enzymes were determined after days 1, 12 and 20. Higher Cd concentrations in tissues of P. glomerata were found to reduce biomass production in both roots and shoots. The lipid peroxidation rates in leaves and roots were smaller at the start of the experiment for all Cd levels. Superoxide dismutase (SOD) activity increased in leaves on day 1 and in roots on day 20 as Cd levels increased. Cd stress induced an increase in the activity of APX in leaves, whereas in roots ascorbate peroxidase (APX) activity was reduced at high concentration of Cd. At the end of the experiment, catalase (CAT) activity in leaves was reduced as Cd concentration increased. Nevertheless, the glutathione reductase (GR) and guaiacol peroxidase (GPX) activities increased. In roots, GR activity was reduced on days 1 and 20.
Keywords: antioxidant enzymes, oxidative stress, tolerance, cadmium toxicity
Introduction
Many heavy metals stimulate the formation of free radicals and reactive oxygen species (ROS), either by direct electron transfer involving metal cations or as a consequence of metal-mediated inhibition of metabolic reactions (Srivastava et al., 2005). Enhanced levels of ROS such as singlet oxygen may activate the pathways that set in train the cell death program (Foyer and Noctor, 2005). Plants that have a well developed defense system against ROS production, limiting their formation, are better equipped to tolerate adverse conditions in their habitat (Alscher et al., 2002). Antioxidants and antioxidant enzymes function to interrupt the cascades of uncontrolled oxidation. These defense systems can remove, neutralize or scavenge oxy-radicals and their intermediates (Foyer and Noctor, 2003).
Superoxide dismutase (SOD) acts as the first line defense against ROS, dismuting O2- to H2O2. Yet, the product of SOD activity is still toxic (H2O2) and should be eliminated in subsequent reactions, through the action of CAT, other peroxidases and by the ascorbate-glutathione cycle. The equilibrium of SOD, APX and CAT activities is essential in order to determine the steady-state level of O2- and H2O2 (Bowler et al., 1991). For instance, when CAT activity is reduced in plants, other ROS-scavenging enzymes such as APX and GPX are up-regulated (Willekens et al., 1997; Vandenabeele et al., 2004).
Pfaffia genus belongs to the family Amaranthaceae, and Pfaffia glomerata is of particular medical interest because its roots are popularly used for anti-tumor, anti-diabetes and aphrodisiac tonic properties (Montanari et al., 1999). Thus, studying its capability to tolerate heavy metals and how much is accumulated in the tissues is interesting since it is widely used as a medicinal plant. Besides that, the need to explore some of the mechanisms used by this species to tolerate metal concentrations has a major scientific interest. This study approached the capacity of P. glomerata to tolerate Cd by variations in the activities of antioxidant enzymes (SOD, CAT, APX, GPX and GR) in leaves and roots with different exposure time to the metal.
Material and Methods
Seeds of Pfaffia glomerata (Sprengel) Pedersen were obtained from at the Federal University of Mato Grosso do Sul (UFMS). Seeds of P.glomerata were placed directly on polystyrene cell trays containing vermiculite and were left to germinate, after which time they were transferred to a Clark nutrient solution (Clark, 1975). Plants remained in the solution for another three weeks for adaptive purposes and were then transferred to individual vases for treatment induction. Cd concentrations used in this work were chosen based on Carneiro et al. (2002). Treatments contained three concentrations of Cd (0, 45 and 90 µmol L-1) in the form of CdSO4, and the experiment was conducted for 20 days, with three assessments: 1, 12 and 20 days after treatment induction. Lipid peroxidation and the activities of antioxidant enzymes were determined on each of these dates. After 20 days, biomass production and Cd concentration in roots and shoots were also assessed.
Biomass analysis at the end of the 20-day-period, the plants were collected, separated into root and shoot fractions, placed in paper bags and taken to an air circulation oven at 70ºC till constant weight was reached for determination of dry matter. After determining dry matter, samples were ground in a Wiley mill equipped with a 0.38 mm mesh sieve and subsequent used for the analysis of Cd content.
For Cd determination, HNO3-HClO4 digestion of the dried and ground plant material was performed, and Cd determination was carried out on the acid extract by atomic absorption spectrometry (Silva, 1999).
The estimation of lipid peroxidation was based on the production of 2-thiobarbituric acid reactive metabolites, particularly malondialdehyde (MDA), following the methods of Heath and Packer (1968) and Buege and Aust (1978). Samples containing 200 mg of leaf and root tissue were macerated in 5 mL of 0.1% TCA (trichloroacetic acid). After complete homogenization, 1.4 mL of the homogenate was transferred to an eppendorf tube and centrifuged at 10,000 rpm for 5 minutes. An aliquot of 0.5 mL of the supernatant was added to 2 mL 0.5% (v/v) TBA (thiobarbituric acid ) in 20% TCA. The mixture was heated in a water bath at 95ºC for 30 minutes and then ice-cooled for 10 minutes. Readings were taken using a spectrophotometer at 535 and 600 nm. The MDA concentration was expressed as nmol g-1 of fresh tissue.
Extraction of the antioxidant enzymes (SOD, CAT, APX, GPX and GR) was carried from 200 mg of fresh tissue of macerated leaves and roots, with the addition of 800 µL extraction buffer containing 100 mmol L-1 potassium phosphate buffer (pH 7.8); 100 mmol L-1 EDTA and 1 mmol L-1 L-ascorbic acid. The protein content in all samples was determined using the Bradford (1976) method.
Superoxide dismutase assay (SOD; E.C. 1.15.1.1) - The activity of SOD was determined according to its capability to inhibit the photochemical reduction of nitroblue tetrazolium (NBT), as described by Beauchamp and Fridovich (1971). The reaction mixture containing 2 mL of final volume consisted of 50 mmol L-1 potassium phosphate buffer (pH 7.8), 14 mmol L-1 methionine, 0.1 µmol L-1 EDTA, 1 mmol L-1 NBT, 2 µmol L-1 riboflavin and 50 µL of protein extract, with riboflavin being added last. Sample test tubes and control test tubes (without samples) were placed under 20W fluorescent light tubes. The reaction was allowed to proceed with the light switched on for 10 min and then stopped by switching off the light. The reading of solution absorbance was carried out at 560 nm. SOD activity is the measure of NBT reduction in light without protein minus NBT reduction with protein. One unit of activity was defined as the amount of protein required to cause 50% inhibition of the initial NBT reduction under assay conditions.
Catalase assay (CAT; E.C. 1.11.1.6) - The activity of CAT was determined as described by Kraus et al. (1995), with minor alterations, as described by Azevedo et al. (1998). CAT was determined by spectrophotometry at 28ºC in a final reaction mixture of 2 mL containing 100 mmol L-1 potassium phosphate buffer (pH 7.0), 12.5 mmol L-1 hydrogen peroxide (H2O2) and water. The reaction was started by adding 30 µL of the extract and the activity determined following the decomposition of H2O2, for 1.5 minute at 10-second intervals, monitoring changes in absorbance at 240 nm, with a molar extinction coefficient of 0.0394 mmol L-1 cm-1. Results were expressed as µmol of H2O2 consumed per minute per mg protein.
Ascorbate peroxidase assay (APX; E.C.1.11.1.11) - The activity of APX was determined using the method of Nakano and Asada (1981), estimating the ascorbate oxidation rate (extinction coefficient of 2.8 mmol L-1 cm-1). The reaction mixture containing 100 mmol L-1 potassium phosphate buffer (pH 7.0), 0.1 mmol L-1 hydrogen peroxide (H2O2), 0.5 mmol L-1 L-ascorbic acid, distilled water and 30 µL of protein extract, in a total volume of 2 mL, was incubated at 28ºC. The ascorbate oxidation rate was monitored at 290 nm for 3 min at 15-s intervals and enzyme activity was expressed as µmol ascorbate oxidized per minute per mg of protein.
Guaiacol peroxidase assay (GPX; E.C. 1.11.1.7) - The activity of GPX was determined according to Souza and MacAdam (1998), observing the tetraguaiacol formation at 28ºC. The reaction mixture, with 2 mL of final volume, contained 50 mmol L-1 potassium phosphate buffer (pH 7.0), hydrogen peroxide (H2O2), 3.5 mmol L-1 guaiacol, distilled water and 10 µL of protein extract. The increase in absorbance due to guaiacol oxidation (extinction coefficient of 26.6 mmol L-1 cm-1) was measured at 470 nm for 1 min at 10-s intervals. The enzyme activity was expressed as µmol of guaiacol decomposed per minute per mg of protein.
Glutathione reductase assay (GR; E.C. 1.6.4.2)-GR activity was determined as described by Cakmak and Horst (1991), following the decrease in absorbance at 340 nm due to NADPH oxidation. The reaction mixture containing 50 mmol L-1 potassium phosphate buffer (pH 7.8), 1 mmol L-1 oxidized glutathione (GSSG), 0.075 mmol L-1 NADPH, distilled water and 30 µL of protein extract, was incubated at 28ºC. GR activity was calculated using an extinction coefficient of 6.2 mmol L-1 cm-1, measured at 340 nm for 3 min at 15-s intervals. The activity of the enzyme was expressed as µmol of NADPH per minute per mg of protein.
The experiment was conducted using a randomized block statistical design consisting of three Cd levels (0, 45 and 90 µmol L-1) and three sampling dates (1, 12 and 20 d). Data were submitted to analysis of variance using the F test and results were presented as mean ± standard error of mean (SEM) of three replicates, and to the Tukey test at 5%.
Results and Discussion
Increasing Cd content in the nutrient solution increased the metal content in roots and shoots of P. glomerata plants (Figure 1A). In the root part the highest values were observed in the treatment at 90 µmol Cd L-1 (1223 mg kg-1), while in the shoot part for the same treatment, Cd concentration was 238 mg kg-1. There were not significant differences in biomass production of the shoot between treatments, whereas the root biomass was higher in control treatment (Figure 1B). As a consequence of Cd accumulation in the tissues visual symptoms of toxicity were observed, including leaf wilting and yellowing as well as root darkening.
Overall, species that tolerate heavy metals should present them widely distributed in the roots which may result in morphological and metabolic changes, for instance reduced plant size and changes in antioxidant enzyme activity (Verbruggen et al., 2009). In this study, P. glomerata exhibited high concentrations of Cd in its tissues and yet it did not undergo major morphological changes, particularly in the treatment at 45 µmol L-1, thus demonstrating some tolerance to the heavy metal.
The impact that a heavy metal exert on a plant will depend on how sensitive the species is and the metals bioavailability present in the soil during the plant growing (Smical et al., 2008). A tolerant specie may be defined as that which produces a amount of roots in contaminated areas (Punz and Sieghardt, 1993), as was observed with P. glomerata. Many tolerant plants are endemic in contaminated places because they have developed a variety of avoidance mechanisms by which the excess of heavy metal can be rendered harmless. The hability to develop heavy metal resistance is due chiefly to morphophysiological changes (Raskin et al., 1994).
Reactive oxygen species are considered to be the indicators of cell damage because lipid peroxidation in vivo provides a steady supply of free radicals. The effect of Cd on cell membrane integrity was determined through MDA measurement (Figures 2A and B). An increase was observed in lipid peroxidation in the leaves of P. glomerata with the increase in Cd content and increase in the exposure periods. The maximum effect was noted at 20 d after induction (107%) on leaves, and 12 d after induction (154%) on roots, both at 90 µmol L-1 concentration A reduction was observed in MDA contents in the roots with increasing exposure to the metal up to concentration of 45 µmol Cd L-1 (58.7 (1 d) to 32.6 (20 d) nmol MDA g-1 FW), while at 90 µmol Cd L-1 concentration the MDA content increased on day 12 (45.5 (1 d) to 72.4 (12 d) nmol MDA g-1 FW).
Plant cell membranes are considered primary targets of metal-induced injuries and membrane destabilization is usually attributed to lipid peroxidation (Singh, 2006). Increased MDA formation in plants exposed to Cd has been verified in sunflowers (Gallego et al., 1996), in peas (Dixit et al., 2001), in Thlaspi caerulescens and Brassica juncea accumulator species and also Nicotiana tabacum, a non-accumulator species (Wang et al., 2008).
The enzymes of the antioxidant system exhibited variations in activity when exposed to different Cd concentrations and different exposure periods. The activity of SOD, which is responsible for the dismutation of superoxide radicals from the cells, increased in the leaves at the beginning with a maximum activity increase of 41% (at 45 µmol L-1 after 1 d). Increasing Cd contents did not change SOD activity in leaves on day 20. (Figure 3A). An increase was verified in the activity of SOD in the roots at 20 days (1.6 U mg-1 protein (0 µmol L-1) to 3.5 U mg-1 protein (90 µmol L-1) with increasing Cd contents in the nutrient solution. The maximum SOD activity increase observed in the roots was 203% (at 45 µmol L-1 after 20 d). (Figure 3B).
Treatments with Cd are reported to have reduced SOD activity in non-accumulator bean plants (Somashekaraiah et al., 1992), sunflowers (Gallego et al., 1996) and peas (Sandalio et al., 2001) and to have increased SOD activity in Alyssum lesbiacum, an accumulator species (Schickler and Caspi, 1999), and mustard (Mobin and Klan, 2007). In this study, high levels of SOD activity can protect P. glomerata against oxidative damage induced by Cd toxicity, when in combination with increased activity of other antioxidant enzymes, promoting the elimination of excess H2O2 which causes damage to the cells (Gossett et al., 1994).
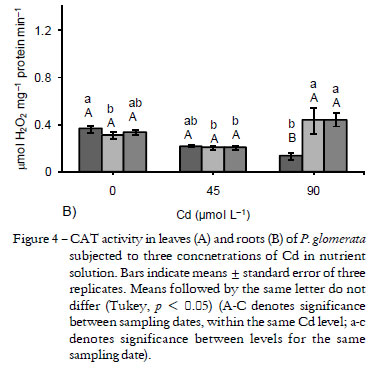
The excessive increase in H2O2 was minimized through the activity of CAT, APX and GPX. In the leaves, the maximum increase in CAT activity was of 54% in relation to the control (at 90 µmol Cd L-1 after 1 d). Yet, at this same concentration a maximum reduction was observed, down to 34%, after 20 d (Figure 4A). In the roots, CAT activity showed a maximum increase of 42% (at 90 µmol L-1 after 12 d) and it was similar to the control at 90 µmol Cd L-1 after 20 d (Figure 4B). Increased CAT activity is reported in Alyssum lesbiacum, a metal accumulator species (Schickler and Caspi, 1999), mustard (Mobin and Klan, 2007; Wang et al., 2008) and Thlaspi caerulesens (Wang et al., 2008). Considering that CAT is directly regulated by H2O2 levels (Polidoros and Scandalios, 1999), the increased activity in leaves with all levels of Cd at the initial stage suggests that this enzyme was more active when first exposed to metal, while in the roots this increased CAT activity was observed at 90 µmol L-1 after 12 (0.43 µmol H2O2 mg-1 protein min-1) and 20 d (0.44 µmol H2O2 mg-1 protein min-1). The maintenance of high CAT activity in Cd accumulator species is an aspect that may be part of the tolerance mechanism of this plant species to the metal.
At the start and end of the experiment, the levels of APX activity in the leaves were statistically similar to the control. Yet, the highest increase in APX activity in the leaves was observed after 12 d with maximum increases of 64% and 43% (at 45 and at 90 µmol L-1, respectively) (Figure 5A). APX activity in leaves appeared to be less affected than CAT activity when the plant was exposed to higher Cd contents for longer periods, since CAT was reduced at 90 µmol L-1 after 20 d, while APX activity remained similar to the control. In roots, the highest level of Cd (90 µmol L-1) after 20 d increased APX activity (4.9 µmol ascorbate mg-1 protein (12 d) to 9.4 µmol ascorbate mg-1 protein (20 d)), though after 12 d APX activity decreased in Cd presence (14 at control to 4.4 µmol ascorbate mg-1 protein min-1 at 90 µmol L-1) (Figure 5B). In the latter situation, the inadequate response of APX activity in the elimination of H2O2 was offset by the increase in CAT activity. Although higher amounts of Cd accumulated in root tissue, higher relative APX activities were observed in leaves of P. glomerata, when compared to the control, i.e. roots only presented a slight percentage increase in the enzyme activity. According to Vitória et al. (2001), evidence points to the existence of a sign of oxidative stress being transported from roots to leaves, and this signaling mechanism was reported by Karpinski et al. (1999), increasing APX transcription in leaves. Similar results to those in roots and leaves of P. glomerata, regarding APX activity, were found for Bacopa monnieri (Singh et al., 2006).
Guaiacol peroxidase activity increased in the leaves to a maximum of 183% and 95% (at 90 µmol L-1 after 12 and 20 d, respectively) (Figure 6A). After 12 and 20 days, there was an increase in the GPX activity following an increase in the metal concentration. In the roots, GPX activity changed only slightly in relation to the control to maximum rates of 21% and 17% (at 45 and at 90 µmol L-1, after 12 d, respectively). At the start of the experiment, after 1 d the stress induced by Cd reduced GPX activity by 48% in the treatment at 90 µmol L-1 (Figures 6B).
In this study there was an increase in GPX activity in leaves following exposure to Cd (0.8 at control to 1.6 µmol guaiacol mg-1 protein min-1 at 90 µmol L-1), suggesting that this enzyme acts as an intrinsic protection against oxidative damage in P. glomerata. In Bacopa monnieri, GPX activity in leaves is reported to have been stimulated by increase both in exposure time and in concentrations of Cd (Singh et al., 2006). However, these authors observed a reduction in the activity of the enzyme in roots with the increased exposure to Cd at the highest concentration (200 µmol. L-1). An increase in GPX activity was also observed in Lycopersicon esculentum when exposed to Cd, due to wide availability of H2O2 produced by stress, particularly toward the end of the experiment (Ammar et al., 2008).
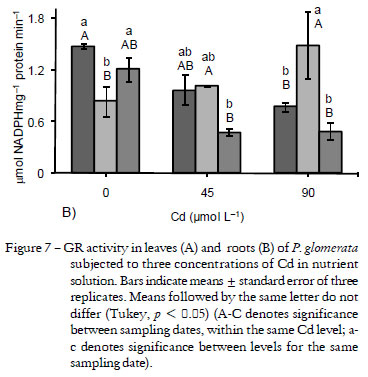
Glutathione redutase showed a increase in activity in the leaves after 12 and 20 d at both concentrations of Cd (45 and 90 µmol L-1) (Figure 7A). Both in leaves and in roots the highest increase in GR activity occurred after 12 d, with 63% in the leaves (at 45 µmol L-1) and 78% in the roots (at 90 µmol L-1). In the roots, the activity of the enzyme was reduced to levels below the control at all Cd concentrations tested after 1 d (1.4 at control to 0.8 µmol NADPH mg-1 protein min-1 at 90 µmol L-1) and 20 d (1.2 at control to 0.5 µmol NADPH mg-1 protein min-1 at 90 µmol L-1) of exposure (Figure 7B).
Glutathione reductase, which catalyses the NADPH-dependent reduction of oxidized GSSG to the reduced form GSH, is involved in defence against oxidative stress. Glutathione is one of the major redox buffers in most aerobic cells and plays an important role in physiological functions, including redox regulation, conjugation of metabolites, detoxification of xenobiotics and homeostasis and cellular signaling that trigger adaptive responses (Noctor et al., 2002). Moreover, accumulation of GSSG is often associated with death or quiescence (Foyer and Noctor, 2003). Small variations were observed in GR activity with increasing exposure time to Cd in pea leaves (Sandalio et al., 2001). As observed in this study, GR activity was higher in roots than in leaves of radish plants subjected to Cd (Vitória et al., 2001). In Alyssum argenteum hyperaccumulator species, low contents of Cd induced GR activity while higher contents reduced the activity (Schickler and Caspi, 1999). According to the authors, this reduction could be the result of direct binding of Cd to the sulfhydryl group of the enzyme, influencing the glutathione cycle. GR activity is important to maintain a high GSH/GSSG ratio, being critical to the functioning of the glutathione-ascorbate cycle and phytochelatin synthesis (Cobbett, 2000), which in turn are responsible for the tolerance of the species to contamination by heavy metals, especially Cd (Benavides et al., 2005).
The low CAT activity in leaves on 20 d may have favored a larger participation of the ascorbate-glutatione cycle as a tolerance mechanism. That is possible because there was an increase in the GR enzyme activity, whereas APX activity remained the same as the control. The ability to do so presumably reflects enhanced engagement of other pathways of H2O2 detoxification (Noctor et al., 2002), notably the ascorbate-glutathione cycle, as evidenced by the sustained oxidation of glutathione accompanied by increases in the total glutathione pool.
In this study, P. glomerata presented concentrations above 100 mg Cd kg-1 (Baker, 1981), meaning it is capable of tolerating high concentrations of Cd in its tissues. According to Baker (1981), it could be classified as a hyperaccumulator species, yet further studies are needed before characterizing it as a phytoremediator. Other results suggested that Cd-induced increase in the activity levels of antioxidant enzymes can constitute a secondary defense mechanism against oxidative stress, though not as direct a mechanism as phytochelatin production and compartmentalization in the vacuole. Here, the differentiated responses of antioxidant enzymes to Cd in roots and leaves can be attributed to variation in ROS formation levels due to differing functionality of the two plant organs, with the root being a non-photosynthetic tissue and thus resulting in reduced electron flow. As for the higher accumulation of Cd in the roots, the level of free Cd may be low given the fact that the bulk of the metal ions may be immobilized or compartmentalized in the vacuole or else forming a complex with phytochelatins. As for the leaves, the lower accumulation of Cd due to transport barriers, along with the activities of antioxidant enzymes, reduced oxidative damage, increasing the tolerance of the species to Cd.
P. glomerata showed some superior antioxidative defense to adapt to the oxidative stress induced by Cd toxicity. MDA content was similar to the control at the mean level of Cd. Accordingly, the activities of antioxidative enzymes increased or remained the same as the control until this concentration of the metal. Then, the coordinated increase of activities of antioxidative enzymes was effective in some protecting the plant from accumulation of ROS under Cd stress.
Acknowledgements
To Dr. Daniela Deitos Freis for the support on enzyme analyses and to the Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG) for providing financial support.
Received March 10, 2010
Accepted October 07, 2010
Edited by: Leonardo Oliveira Medici
- Alscher, R.G.; Erturk, N.; Heath, L.S. 2002. Role of superoxide dismutases (SODs) in controlling oxidative stress in plants. Journal of Experimental Botany 53: 1331-1341.
- Ammar, W.B.; Nouairi, I.; Zarrouk, M.; Ghorbel, M.H.; Jemal, F. 2008. Antioxidative response to cadmium in roots and leaves of tomato plants. Biologia Plantarum 52: 727-731.
- Azevedo, R.A..; Alas, R.M.; Smith, R.J.; Lea, P.J. 1998. Response of antioxidant enzymes to transfer from elevated carbon dioxide to air and ozone fumigation, in the leaves and roots of wild-type and a catalase-deficient mutant of barley. Physiologia Plantarum 104: 280-292.
- Baker, A.J.M. 1981. Accumulators and excluders: strategis in the response of plants to heavy metals. Journal of Plant Nutrition 3: 643-654.
- Beauchamp, C.; Fridovich, I. 1971. Superoxide dismutase improved assays and assay applicable to acrylamide gels. Analytical Biochemistry 44: 276-287.
- Benavides, M.P.; Gallego, S.M.; Tomaro, M.L. 2005. Cadmium toxicity in plants. Brazilian Journal of Plant Physiology 17: 21-34.
- Bradford, J.M. 1976. A rapid and sensitive method for qualification of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry 72: 248-254.
- Buege, J.A.; Aust, S.D. 1978. Microsomal lipid peroxidation. Methods Enzymology 52: 302-310.
- Cakmak, I.; Horst, W.J. 1991. Effect of aluminium on lipid peroxidation, superoxide dismutase, catalase and peroxidase activities in root tips of soybean (Glycine max). Physiologia Plantarum 83: 463-468.
- Carneiro, M.A.C.; Siqueira, J.O.; Moreira, F.M.S. 2002. Behavior of herbaceous species in soil mixes with different degree of contamination with heavy metal. Pesquisa Agropecuária Brasileira 37:1629-1638.
- Clark, R.B. 1975. Characterization of phosphatase of intact maize roots. Journal of Agricultural Food Chemistry 23: 458-460.
- Cobbett, C.S. 2000. Phytochelatins and their roles in heavy metal detoxification. Plant Physiology 123: 825-832.
- Dixit, V.; Pandey, V.; Shyam, R. 2001. Differential antioxidative responses to cadmium in roots and leaves of pea (Pisum sativum L. cv. Azad). Journal of Experimental Botany 52: 1101-1109.
- Foyer, C.H.; Noctor, G. 2003. Redox sensing and signaling associated with reactive oxygen in chloroplasts, peroxisomes and mitochondria. Plysiologia Plantarum 119: 355-364.
- Foyer, C.H.; Noctor, G. 2005. Redox homeostasis and antioxidant signaling: a metabolic interface between stress perception and physiological responses. Plant Cell 17: 1866-1875.
- Gallego, S.M.; Benavides, M.P.; Tomaro, M.L. 1996. Effect of heavy metal ion excess on sunflower leaves: evidence for involvement of oxidative stress. Plant Science 121: 151-159.
- Gossett, D.R.; Millhollon, E.P.; Lucas, M.C.; Bank, S.W.; Marney, M.M. 1994. The effects of NaCl on antioxidant enzyme activities in callus tissue of salt-tolerant and salt-sensitive cotton cultivars (Gossypium hirsutum L.). Plant Cell Report 13: 498-503.
- Heath, R.L.; Packer, L. 1968. Photoperoxidation in isolated chloroplasts. I. Kinetics and stoichiometry of fatty acid peroxidation. Archives of Biochemistry and Biophysics 125: 189-198.
- Karpinski, S.; Reynolds, H.; Karpinska, B.; Wingsle, G.; Creissen, G.; Mullineaux, P. 1999. Systemic signaling and acclimation in response to excess excitation energy in Arabidopsis Science 284: 654-657.
- Kraus, T.E.; Mckersie, B.D.; Fletcher, R.A. 1995. Paclobutrazol-induced tolerance of wheat leaves to paraquat may involve increased antioxidant enzyme activity. Journal of Plant Physiology 145: 570-576.
- Mobin, M.; Khan, N.A. 2007. Photosynthetic activity, pigment composition and antioxidative response of two mustard (Brassica juncea) cultivars differing in photosynthetic capacity subjected to cadmium stress. Journal of Plant Physiology 164: 601-610.
- Montanari, I.J.R.; Magalhães, P.M.; Queiroga, C.L. 1999. Influence of plantation density and cultivation cycle on root productivity and tonors of b-ecdsyone in Pfaffia glomerata (Spreng) Pedersen. Acta Horticulture 3: 125-128.
- Nakano, Y.; Asada, K. 1981. Hydrogen peroxide is scavenged by ascorbato-specific peroxidase in spinach chloroplasts. Plant Cell Physiology 22: 867-880.
- Noctor, G; Gomez, L.; Vanacker, H.; Foyer, C.H. 2002. Interactions between biosynthesis, compartimentation and transport in the control of glutathione homeostasis and signalling. Journal of Experimental Botany 53: 1283-1304.
- Polidoros, A.N.; Scandalios, J.G. 1999. Role of hydrogen peroxide and different classes of antioxidants in the regulation of catalase and glutathione S-transferase gene expression in maize (Zea mays L.). Physiologia Plantarum 106: 112-120.
- Punz, I.; Sieghardt, H. 1993. The response of roots of herbaceous plant species to heavy metal. Environmental and Experimental Botany 44: 85-98.
- Raskin, I.; Kumar, P.B.A.N.; Dushenkov, S.; Salt, D.E. 1994. Bioconcentration of heavy metals by plants. Current Opinion in Biotechnology 5: 285-290.
- Sandalio, L.M.; Dalurzo, H.C.; Gómez, M.; Romero-Puertas, M.C.; Rio, L.A. 2001. Cadmium-induced changes in the growth and oxidative metabolism of pea plants. Journal of Experimental Botany 52: 2115-2126.
- Schickler, H.; Caspi, H. 1999. Response of antioxidative enzymes to nickel and cadmium stress in hyperaccumulator plants of the genus Alyssum Physiologia Plantarum 105: 39-44.
- Silva, F.C. 1999. Manual of soil, plant, and fertilizer analyses. EMBRAPA, Brasília, DF, Brazil. (in Portuguese).
- Singh S.; Eapen, S.; D'Souza, S.F. 2006. Cadmium accumulation and its influence on lipid peroxidation and antioxidative system in an aquatic plant, Bacopa monnieri L. Chemosphere 62: 233-246.
- Smical, A.I.; Hotea, V.; Oros, V.; Juhaz, J.; Pop, E. 2008. Studies on transfer and bioaccumulation of heavy metals from soil into lettuce. Environmental Engineering and Management Journal 7:609-615.
- Somashekaraiah, B.V.; Padmajana, K.; Prasad, A.R.K. 1992. Phytotoxicity of cadmium ion on germinating seedlings of mung bean (Phaseolus vulgaris): involvement of lipid peroxides in chlorophyll degradation. Physiologia Plantarum 85: 85-89.
- Souza, I.R.P.; MacAdam, J.W. 1998. A transient increase in apoplastic peroxidase activity preceds decrease in enlogation rate of B73 maize (Zea mays L.) leaf blades. Physiologia Plantarum 104: 556-562.
- Srivastava, M.; Ma, L.Q.; Singh, N.; Singh S. 2005. Antioxidant responses of hyper-accumulator and sensitive fern species to arsenic. Journal of Experimental Botany 56: 1335-1342.
- Vandenabeele, S.; Vanderauwera, S.; Vuylsteke, M.; Rombauts, S.; Langebartels, C.; Seidlitz, H.K.; Zabeau, M.; Van Montagau, M.; Inzé, D.; Van Breusegem, F. 2004. Catalase deficiency drastically affects gene expression induced by high light in Arabidopsis thaliana The Plant Journal 39: 45-58.
- Verbruggen, N.; Hermans, C.; Schat, H. 2009. Mechanisms to cope with arsenic or cadmium excess in plants. Current Opinion in Plant Biology 12: 364-372.
- Vitória, A.P.; Lea, P.J.; Azevedo, R.A. 2001. Antioxidant enzymes responses to cadmium in radish tissues. Phytochemistry 57: 701-710.
- Wang, Z.; Zhang, Y.; Huang, Z.; Huang; L. 2008. Antioxidative response of metal-accumulator and non-accumulator plants under cadmium stress. Plant and Soil 310: 137-149.
- Willekens, H.; Chamnongpol, S.; Davey, M.; Schraudner, M.; Langebartels, C.; VanMontagu, M.; Inzé, D.; Van Camp, W. 1997. Calatase is a sink for H2O2 and is indispensable for stress defence in C3 plants. EMBO Journal 16: 4806-4816.
Publication Dates
-
Publication in this collection
22 Sept 2011 -
Date of issue
Aug 2011
History
-
Received
10 Mar 2010 -
Accepted
07 Oct 2010