Abstract
The effects of roots are generally not considered in studies assessing crop responses to green manure. However, measuring such effects can contribute to a better understanding of crop rotation. In two experiments, we evaluated the content of legume-N in crop tissue and the fertilizer value of the roots and shoots of two legume species. Roots, shoots, or whole plants of the legumes sunhemp (Crotalaria juncea) and jack beans (Canavalia ensiformis) were cropped as green manure to supply nitrogen to cabbage crops (Brassica oleracea var. capitata). The principle of the A-value technique was applied to estimate the fertilizer value of each plant part. In a pot experiment, both the content of legume-N in cabbage and the fertilizer value of the whole plant was higher than the shoots, which was in turn higher than that of the roots. In field condition, roots had a decreasing effect on the N content of cabbage plants. Growing cabbage on legume root residue resulted in an increased absorption of 15N-urea, resulting in negative values for legume-N content: -13.59 g kg-1 and -3.51 g kg-1 for sunhemp and jack beans, respectively. Suggesting both low N supply by roots and N immobilization in soil organic matter or microbial biomass. Future research should focus on estimating the net N acquisition by plants from root residues under field conditions, where rooting patterns and biomass distribution differ from those in pot experiments, therefore giving a more realistic quantitative estimate.
Brassica oleracea var. capitata; Crotalaria juncea; Canavalia ensiformis; 15N, green manure
CROP SCIENCE
Nitrogen contributions of legume roots to cabbage nutrition
Thiago de Oliveira VargasI; Ellen Rúbia DinizII; Ricardo Henrique Silva SantosI,* * Corresponding author < rsantos@ufv.br> Edited by: Daniel Scherer de Moura ; Segundo UrquiagaIII; Paulo Roberto CeconIV
IUFV Depto de Fitotecnia, Av. P.H. Rolfs s/n Campus Universitário 36570-000 Viçosa, MG Brazil
IIInstituto Federal de Educação, Ciência e Tecnologia do Paraná, Rod. PR 466 - Secção C Parte 2, Gleba Pindaúva 86870-000 Ivaiporã, PR Brasil
IIIEmbrapa Agrobiologia, Rod. BR 465, km 7 23890-000 Seropédica, RJ Brasil
IVUFV Depto. de Informática
ABSTRACT
The effects of roots are generally not considered in studies assessing crop responses to green manure. However, measuring such effects can contribute to a better understanding of crop rotation. In two experiments, we evaluated the content of legume-N in crop tissue and the fertilizer value of the roots and shoots of two legume species. Roots, shoots, or whole plants of the legumes sunhemp (Crotalaria juncea) and jack beans (Canavalia ensiformis) were cropped as green manure to supply nitrogen to cabbage crops (Brassica oleracea var. capitata). The principle of the A-value technique was applied to estimate the fertilizer value of each plant part. In a pot experiment, both the content of legume-N in cabbage and the fertilizer value of the whole plant was higher than the shoots, which was in turn higher than that of the roots. In field condition, roots had a decreasing effect on the N content of cabbage plants. Growing cabbage on legume root residue resulted in an increased absorption of 15N-urea, resulting in negative values for legume-N content: -13.59 g kg1 and -3.51 g kg1 for sunhemp and jack beans, respectively. Suggesting both low N supply by roots and N immobilization in soil organic matter or microbial biomass. Future research should focus on estimating the net N acquisition by plants from root residues under field conditions, where rooting patterns and biomass distribution differ from those in pot experiments, therefore giving a more realistic quantitative estimate.
Keywords:Brassica oleracea var. capitata, Crotalaria juncea, Canavalia ensiformis, 15N, green manure
Introduction
The influence of legume material green manure on soil N availability and vegetable crop yields has been reported by Collins et al. (2007), Diniz et al. (2007), and Schellenberg et al. (2009). However, in most studies assessing the effects of green manure on crop performance, legume roots have not been considered.
Quantifying the N in legume root systems is fundamental to understanding its effects on soil mineral N fertility and on the supply of N to crops in legume-based rotations (Khan et al., 2002b). N derived from legume rhizodeposits contributed to an increase of 35-44 % in residual N content in soil and constituted 79-85 % of the below-ground N of plants at maturity (Mayer et al., 2003). Jensen (1996) and Sawatsky and Soper (1991) reported that 47 % of the total below-ground N derived from plants originated from root depositions. Russell and Fillery (1996a) verified that the deposition of lupine root accounted for 65 % of total residual N in the soil.
Direct methods to quantify root N, such as the physical recovery of roots, have been widely employed. However, their effectiveness is disputed (Khan et al., 2002a, b; McNeill et al., 1997; Russell and Fillery, 1996a, b), as they consider only the N present in the visible roots and ignore the N in unrecovered root fragments, such as that derived from exuded and mineralized roots during plant development (Wichern et al., 2007a, b; Wichern et al., 2008).
The estimated contribution of N derived from legumes can be determined indirectly using a fertilizer labeled with 15N as a reference, based on the principles of the A-value technique. The A-value technique is based on the assumption that the assimilation of N sources by plants is directly proportional to the N available from each source (Chalk, 1996; Fried and Dean, 1952). One of the main requirements of this technique is that the reference fertilizer in the soil be placed so as to minimize its interaction with the soil and the N sources under study.
The amounts of nitrogenous compounds released from legume roots increase immediately after cutting the shoots (Choi et al., 2008). However, little is known about contribution of below-ground parts of green manure legumes to the N nutrition of the succeeding crops.
For this reason, our main hypothesis was that legume roots would contribute positively to cabbage N supply. We evaluated the contributions of the roots, shoots, and whole plants of sunhemp (Crotalaria juncea) and jack beans (Canavalia ensiformis) to the N supply for cabbage (Brassica oleracea var. capitata) crop, to determine the content of legume-N in cabbage and the fertilizer value of the legume parts to the cabbage.
Materials and Methods
We conducted two experiments in which legume parts were applied to soils that were labeled with 15N and subsequently cultivated with cabbage. In this study the biological N fixation (BNF) of legume crops was estimated with the 15N natural abundance technique (Boddey et al., 2001). The content of legume-N in the cabbage was determined by applying the principle of the A-value technique (Boddey and Urquiaga, 1992), for which the reference fertilizer is located in the soil, looking up the lowest interaction with the soil and the green manures in the study.
Pot experiment
The experiment followed a (2 × 3) + 1 factorial design. The treatments were two legumes and three plant parts (roots, shoots, and whole plant) and one additional treatment (mineral fertilizer with 50 % of the recommended N dose). The experiment was set in completely randomized blocks with five replicates, totaling 35 plots of one plant per plot. The treatments were as follows: MF50 (50 % of the recommended N dose of Mineral Fertilizer), SRO MF50 (Sunhemp Root + MF50), SSH MF50 (Sunhemp Shoot + MF50), SWP MF50 (Sunhemp Whole Plant + MF50), JBRO MF50 (Jack Beans Root + MF50), JBSH MF50 (Jack Beans Shoot + MF50), and JBWP MF50 (Jack Beans Whole Plant + MF50).
Soil and legume production
Legumes were grown in 10-L pots filled with soil and sand in a 2:1 ratio. The final substrate had the following chemical characteristics after application of lime and single superphosphate: 5.8 g kg1 of organic matter (Walkley-Black), pH in water (1:2.5) 5.34, 56 mg dm3 available P (Mehlich I) and exchangeable cations: 86 mg dm3 K (Mehlich I) and 24.5 mmolc dm3 Ca, 6.3 mmolc dm3 Mg, and 0.0 mmolc dm3 Al (KCl 1M).
Legume seeds were inoculated with appropriate bacteria of the genus Bradyrhizobium produced by the following strains from the Embrapa Agrobiologia culture collection: Crotalaria juncea BR2001 and Canavalia ensiformis BR2003. Ten pots were planted with sunhemp and ten with jack beans, sowing nine sunhemp seeds and six jack beans seeds per pot. One week after the emergence of legumes, plants were thinned to three sunhemp and two jack bean plants in each pot. The legumes were cut at ground level 78 days after sowing.
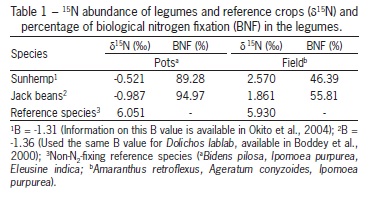
For the root treatment (RO), the shoots of the legumes were removed and the roots were kept in the soil. For the shoot treatment (SH), the removed shoots were applied to another pot. For the whole plant treatment (WP), the shoots were cut but the shoot biomass was not removed from the pot. The whole plant mass was determined by summing the root and shoot masses of legume plants. Additional pots were cultivated with non-N2-fixing reference species (Table 1) to determine the value of δ 15N of the soil and thus estimate the contribution of BNF in legumes via the 15N natural abundance technique (Boddey et al., 2001).
Cabbage cultivation
After the measuring legume shoot biomass, shoots were chopped using a paper guillotine and then redistributed on the soil surface for the SH or WP treatments, to reduce the effect of soil macrofauna absence in the pots. In the RO treatments the roots were not chopped and the soil was slightly loosened in order to receive the cabbage seedlings. 'Matsukaze' cabbage seedlings were then planted in the same pots, following the application of each treatment. The cabbage plants were grown in pots for 100 days and then harvested.
The N fertilizer was applied at a rate of 50 mg kg−1 N, corresponding to 0.6 g N per pot, equivalent to 50 % of the recommended N dose (Vargas et al., 2012). The N fertilizer, in the form of urea with 3 % 15N a.e. (atoms excess), was added immediately after transplant, in a small hole at a depth of 1 cm close to the seedling. The plants were irrigated daily to maintain soil moisture at approximately 80 % of water holding capacity. During the experiment, all leachate liquid after irrigation was collected in plastic plates and returned to the pots. Next, 70 mg kg−1 K and 1.0 g per pot of combined micronutrients (S 32 g kg1, B 18 g kg1, Cu 8 g kg1, Mn 20 g kg1, Mo 1 g kg1, Zn 90 g kg1, and Ca 18 g kg1) were applied. The entire supply of phosphorus (single superphosphate), calcium, and magnesium (calcined dolomitic limestone) was applied in the preparation of the sand and soil mixture before sowing the legumes.
Field experiment
Soil and legume production
Treatments and experimental design were the same as in the pot experiment. The cultivation was carried out in a Haplic Cambisol (FAO, 1998) in Viçosa, state of Minas Gerais, Brazil (20°45' S 42°51' W, 650 m a.s.l.), with the following chemical characteristics in the 0-20 cm layer: 26.60 g kg1 of organic matter (Walkley-Black), pH in water (1:2.5) 5.49, 30 mg dm3 available P (Mehlich I) and exchangeable cations: 176 mg dm3 K (Mehlich I), 30.4 mmolc dm3 Ca, 5.0 mmolc dm3 Mg, and 0.0 mmolc dm3 Al (KCl 1M). The region has a cold dry winter and a hot wet summer, with an average temperature of 19.4 °C (max. 26.4 °C and min. 14.8 °C) and mean annual precipitation of 1,221 mm.
Before sowing the legumes, the soil was ploughed, incorporating mainly jungle rice grass (Echinochloa colona), grated, and furrowed. A single superphosphate fertilizer was applied in the planting furrows at a dose of 400 kg ha−1 P2O5, in accordance with the usual recommendation for horticultural crops in Brazilian soils. Weed control was carried out manually. The legume seeds were inoculated with appropriate bacteria of the genus Bradyrhizobium as described earlier. Sunhemp was planted at a rate of 25 seeds per meter and jack beans at 7 seeds per meter, with 80 cm between rows.
Legumes were cut at ground level 118 days after planting. After cutting the legumes, parts were distributed as described in the pot experiment methods. Where legumes were not planted, non-N2-fixing reference species were grown to determine the δ 15N value of the soil and to estimate the legume BNF using the natural abundance of 15N.
Cabbage cultivation
On the day after the legumes were cut, 'Matsukaze' hybrid cabbage seedlings were transplanted into the rows where the legumes had been planted. Plants were spaced 0.8 m by 0.5 m, totaling 25 plants per plot. The experimental plot was comprised of nine central plants.
Nitrogen fertilization was applied to the cabbage at a dose of 75 kg ha−1 N, which corresponds to 50 % of the recommended dose of N; this fertilization was carried out at the time of transplanting so as not to affect legume mineralization and decomposition processes. Within each experimental plot, a central micro plot of four cabbage plants was established in which the N fertilizer was added immediately next to the seedling in the form of urea with 2 % 15N a.e. in a small, 1-cm-deep hole. The remaining plants received the same dose of N in the form of commercial urea.
Phosphorus was applied to all plots when the legumes were planted. Potassium was applied at a dose of 100 kg ha−1 K2O, with one-third applied at transplant followed by two equal applications at 37 and 57 days after transplant. FTE BR-12 was also applied at transplant in a dose of 62.5 kg ha−1. On average, the equivalent of 204, 518 and 721 kg N ha−1 from root, shoot, and whole plant treatments were added to the cabbage crop, respectively. For details about estimated legume biomass production, N content, C:N ratio, and nutrient accumulation, please see Vargas et al. (2011, 2012). The cabbage plants were grown in the field for 92 days and then harvested.
Sample collection and analysis in cabbage and legumes
After collecting the plants, representative fractions of the whole shoot (stem, leaf, and head) were removed, on the basis of the proportions of fresh matter. These samples were washed with deionized water, dried on paper towels, and then placed in paper bags and dried in a forced air ventilation oven at 65 °C until constant mass was reached. The material was then stored and ground for later determination of total N (Kjeldahl) and 15N content, allowing determination of the content of legume-N in the cabbage.
The BNF of sunhemp and jack beans was estimated using the 15N natural abundance technique (Shearer and Kohl, 1986; Boddey et al., 2001) with a mass spectrometer. The BNF of legumes grown in pots was 89 and 95 % for sunhemp and jack beans, respectively, and 46 and 56 % for those same species grown in the field (Table 1).
Content of legume-N in cabbage and determination of the fertilizer value of legume root, shoot, and whole plant mass
The fertilizer value of legume-N was estimated using the principle of the A-value technique (Fried and Dean, 1952; Boddey and Urquiaga, 1992). The content of N from legume roots, shoots, and whole plants in cabbage plants (% N-leg) and the legume A-value (Av) for cabbage crop was determined with the following equations.
Cabbage control (no legumes)
% fertilizer-N in cabbage control (% Nfert.) = (% 15N a.e. plant / % 15N a.e. fert.) 100 (1)
% soil-N in cabbage control (% Nsoil) = (100 % Nfert.) (2)
A-value Soil (As) = (% Nsoil / As) = (% Nfert. / Nfert. applic.) (3)
Cabbage (with legumes)
% fertilizer-N in cabbage with legumes (% Nfert.leg.) = (% 15N a.e. plant / % 15N a.e. fert.) 100 (4)
% soil-N in cabbage with legumes (% Nsoil leg.) = (% Nfert. leg. / Nfert. applic.) = (% Nsoil leg. / As) (5)
% legume-N in cabbage (% N-leg. cabbage): 100 (% Nfert. leg. + % Nsoil leg.) (6)
A-value Soil + legumes (A(s+leg.)) = 100 (% Nsoil leg. / A(s+leg.)) = (% Nfert. leg. / Nfert. applic.) (7)
Legumes A-value (Av): = (A(s+leg.) As) (8)
where % Nfert. = percentage of N derived from fertilizer in control cabbage plants; % 15N a.e. plant = percentage of 15N atoms in excess of those in the cabbage plants; % 15N a.e. fert. = percentage of 15N atoms in excess of those in the applied fertilizer (urea); % Nsoil = percentage of N derived from soil in control cabbage plants; As = amount of N available in soil in kg ha−1 equivalent to the urea form; Nfert. applic. = amount of N fertilizer applied in kg ha−1; % Nfert. leg. = percentage of N derived from fertilizer in cabbage plants grown with legume root, shoot, or whole plant treatments; % Nsoil leg. = percentage of N derived from soil in the cabbage plants grown with legume root, shoot, or whole plant treatments; % N-leg. Cabbage = percentage of N in cabbage plants derived from legume root, shoot or whole plant treatments; A(s+leg.) = amount of N available in the soil and from legume parts in kg ha−1; Av = amount of N available from legume parts in kg ha−1 expressed in the urea form.
The variance of the data was analyzed with the F-test, comparing the averages with Tukey's test (p < 0.05). Statistical analyses were performed with SAEG software (Funarbe, 2007).
Results
Pot experiment
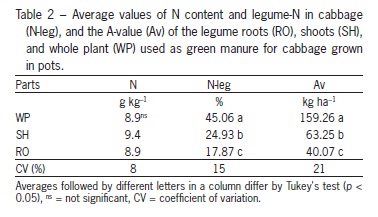
The application of legume parts did not affect the N content of the cabbage plants (Table 2). The legume-N content of cabbage grown on whole plant legume biomass was greater than that grown on shoot biomass, which was in turn greater than that grown on root biomass. As expected, the fertilizer value (A-value) of the whole legume plants as a source of N was higher than that of the other parts (Table 2), since the total N applied was significantly higher than that of the other parts.
Field Experiment
Although the variance analysis indicated an effect of the interaction between legume species and parts on legume-N taken up by cabbage, there was no difference (p ≥ 0.05) between species for each legume part (Table 3). Differences in the content of legume-N on cabbage were only found due to different legume parts. Applying the whole legume plant to cabbage resulted in higher N content than applying legume roots (Table 4). Cabbage plants grown on legume roots did not present any legume-N (Table 3). The fertilizer values of the legume whole plants and shoots were similar, and both were higher than the zero values found in plants grown on legume roots (Table 4). The root treatment resulted in a higher value of % 15N a.e. than the other plant part treatments (Table 5).
Discussion
Pot experiment
Applying the whole plant resulted in a higher fertilizer value (Av) in the urea form. These results were directly associated with the amounts of N applied to legume parts used as green manure. Legume-N content in cabbage was higher in the pot experiment than in the field experiment. Because of the specific nature of the pot experiment, with a relatively small soil volume per plant, root density will be higher and the shoot-to-root ratio will be larger than in field conditions (Mayer et al., 2003; Wichern et al., 2007b).
In the pot experiment, loss of N from urea and legume plants through leaching was avoided by returning leachate to the pots. Because of the possibility of volatilization by urea, care was taken to apply it deep in small holes in the soil. However, this procedure may have enhanced the process of N loss by volatilization from the legume shoots placed on the pot surface, since up to 14 % of N can be volatilized when not incorporated into the soil (Janzen and McGinn, 1991). This loss could have reduced the effect of legume species used as green manure in the study. In addition, nitrification and denitrification may also influence other types of N loss. Some studies have observed higher denitrification activity and N2O fluxes after retaining residues on the land (FAO, 2001). This effect may be related to the effect that residues on the soil surface have on moisture conditions of the topmost soil layer. The incorporation of residues also causes accelerated NO emissions.
Field experiment
Cabbage plants grown on legume root biomass presented zero values of legume-N. Roots had a negative effect on the availability of N to cabbage. We suggested that this finding may be attributed to the lower content of N from root fragments and because the N mineralized from root decomposition had been immobilized by the microbial biomass or incorporated into soil organic matter, as reported by Mayer et al. (2003). However, the competition of plant and microbial biomass for N uptake was not directly measured in our experiment. In this case, plant uptake of 15N-urea would increase, resulting in negative values of legume-N content. This theory is supported by the data on N content in the cabbage mass grown on legume roots, which is lower than that of the other treatments.
The high concentration of N derived from organic sources in the soil is often quoted in the literature, with values ranging between 44 and 89 % of N for green manures and between 12 and 53 % for mineral fertilizers (Azam et al., 1985; Harris and Hesterman, 1990; Harris et al., 1994; Scivittaro et al., 2003). This higher concentration is partially due to the greater absorption of N from mineral sources by plants in the short term (Ladd and Amato, 1986) and it may suggest the permanence of N derived mainly from the roots in soil organic matter.
The smaller availability of root-N would increase the value of 15N atomic excess (% 15N a.e.) in plants grown on legume roots. The consequent lack of N contributions from the roots to cabbage-N reflects the strong immobilizing effects of root biomass on the N available in soil, including 15N-urea. Nitrogen mineralization is highly influenced by the C:N ratio (Cabrera et al., 2005). The immobilization phenomenon can be linked to the high C:N ratio in sunhemp root biomass (42.70), as it has low N concentration when compared to jack bean (16.53).
The mineralization and immobilization potential of N from legumes was evaluated by Matos et al. (2008) during seven weeks. The authors report a higher mineralization in the first week, followed by negative mineralization, especially until the third or fifth week due to the predominance of the immobilization process, after which the mineral N concentration indicated positive mineralization. This finding suggests that, under field conditions and for the duration of the study, root biomass caused an N-immobilizing effect in the soil. In treatments consisting of roots of both legume species, there was a significant removal of soil nutrients when shoots were removed, thereby reducing the soil's ability to nourish the cultivated cabbage.
There are still unanswered questions related to the interaction of mineralized legume biomass N and its destination in the soil, compartmentalization/immobilization, remineralization, and availability processes. Rhizodeposition is a release process of organic and inorganic compounds derived from living roots (Wichern et al., (2008). It is possible that, in our study, low N contributions from legume roots were observed because the legume roots were dead and the next crop grown just as the roots started to decompose.
Greater net N mineralization from legume shoots than from root fractions has also been reported for other legume species. The lower N availability of legume roots compared with the shoots can be attributed to the resistance of certain organic N compounds to decomposition and to potentially higher immobilization of green manure N owing to associated additions of carbon (Wivstad, 1999).
Results show the low fertilizing value of legume roots. N immobilization by legume roots may have been the major factor behind the observed low contributions to cultivated cabbage plants, when compared with the problem of nutrient exportation by removing the shoot. This removal of the shoot must have depleted the soil in N and other nutrients. These possibilities are strengthened by the lack of significant differences in the A-values (Av) of the whole plant and shoot parts in this study. In this particular study to assess the local effect of the roots of legumes, cabbage plants were planted in the same environment without the presence of the shoot. Other techniques would require root production in another location and the incorporation of those roots in the experimental area.
These experiments suggest that the lower legume-N content in cabbage grown on root biomass was strongly influenced by the legume root material, emphasizing the importance of legume root characteristics, and by competition with the microbial biomass. This fact must be taken into account in studies of 15N mineralization from the legume root fraction. There is a need for further long-term research focusing on studies assessing nutrients from roots with the aim of achieving optimal synchronization between net N release and N uptake of succeeding crops.
In addition, future studies could be conducted by labeling of legumes with 15N in order to study the contribution of roots in the presence of shoots using the technique of cross-wise labeling, i.e. labeled roots with unlabelled shoots in one treatment and vice versa, in which labeled material (legumes) would have to be enriched in an adjacent area and brought into the environment under study.
Conclusions
Under pot conditions, the greatest fertilizer value of the legume was obtained when cabbage was grown over the whole plant biomass rather than only over legume shoot or root biomass. On the other hand, under field conditions, the legume shoots or whole plants resulted in higher values of legume-N on cabbage crops and higher fertilizer values. In addition, legume roots accounted for zero content of legume-N in cabbage, suggesting both low N supply by roots and N immobilization in soil organic matter or microbial biomass.
Acknowledgements
The authors thank the Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG) and the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for financial support.
Received August 23, 2012
Accepted June 28, 2013
- Azam, F.; Malik, K.A.; Sajjad, M.I. 1985. Transformations in soil and availability to plants of 15N Applied as inorganic fertilizer and legume residues. Plant and Soil 86: 3-13.
- Boddey, R.M.; Urquiaga, S. 1992. Calculations and assumptions involved in the use of the A-value and 15N isotope dilution techniques for the estimation of the contribution of plant-associated biological N2 fixation. Plant and Soil 145: 151-155.
- Boddey, R.M.; Peoples, M.B.; Palmer, B.; Dart, P. 2000. Use of the 15N natural abundance technique to quantify biological nitrogen fixation by woody perennials. Nutrient Cycling in Agroecosystems 57: 235-270.
- Boddey, R.M.; Polidoro, J.C.; Resende, A.S.; Alves, B.J.R.; Urquiaga, S. 2001. Use of delta 15N thecnique to estimation of biological nitrogen fixation in grasses and cereals. Australian Journal of Plant Physiology 28: 889-895.
- Cabrera, M.L.; Kissel, D.E.; Vigil, M.F. 2005. Nitrogen mineralization from organic residues: research opportunities. Journal of Environmental Quality 34: 75-79.
- Chalk, P. 1996. Estimation of N2 fixation by 15N isotope dilution: the A-value approach. Soil Biology and Biochemistry 28: 1123-1130.
- Choi, B.; Ohe, M.; Harada, J.; Daimon, H. 2008. Role of belowground parts of green manure legumes, Crotalaria spectabilis and Sesbania rostrata, in N uptake by the succeeding tendergreen mustard plant. Plant Production Science 11: 116-123.
- Collins, H.P.; Delgado, J.A.; Alva, A.K.; Follett, R.F. 2007. Use of nitrogen-15 isotopic techniques to estimate nitrogen cycling from a mustard cover crop to potatoes. Agronomy Journal 99: 27-35.
- Diniz, E.R.; Santos, R.H.S.; Urquiaga, S.S.; Peternelli, L.A.; Barrella, T.P.; Freitas, G.B. 2007. Green manure incorporation timing for organically grown broccoli. Pesquisa Agropecuária Brasileira 42: 199-206.
- Food and Agriculture Organization [FAO]. 1998. World Reference Base for Soil Resources. FAO, Rome, Italy. (World Soil Resources Reports, 84).
- Food and Agriculture Organization [FAO]. 2001. Global Estimates of Gaseous Emissions of NH3, NO and N2O from Agricultural Land. FAO, Rome, Italy.
- Fried, M.; Dean, L.A. 1952. A concept concerning the measurement of available soil nutrients. Soil Science 73: 263-271.
- Fundação Arthur Bernardes [Funarbe]. 2007. Statistics and Genetics Analysis System (SAEG), Version 9.1. = Sistema para Análises Estatísticas (SAEG), Version 9.1. UFV, Viçosa, MG, Brazil (in Portuguese).
- Harris, G.H.; Hesterman, O.B. 1990. Quantifying the nitrogen contribution from alfalfa to soil and two succeeding crops using nitrogen-15. Agronomy Journal 82: 129-134.
- Harris, G.H.; Hesterman, O.B.; Paul, E.A.; Peters, S.E.; Janke, R.R. 1994. Fate of legume and fertilizer nitrogen-15 in a long term cropping systems experiment. Agronomy Journal 86: 910-915.
- Janzen, H.H.; McGinn, S.M. 1991. Volatile loss of nitrogen during decomposition of legume green manure. Soil Biology and Biochemistry 23: 291-297.
- Jensen, E.S. 1996. Rhizodeposition of N by pea and barley and its effect on soil N dynamics. Soil Biology and Biochemistry 28: 65-71.
- Khan, D.F.; Peoples, M.B.; Chalk, P.M.; Herridge, D.F. 2002a. Quantifying below-ground nitrogen of legumes. 2. A comparison of N-15 and non isotopic methods. Plant and Soil 239: 277-289.
- Khan, D.F.; Peoples, M.B.; Herridge, D.F. 2002b. Quantifying belowground nitrogen of legumes. 1. Optimising procedures for N-15 shoot-labelling. Plant and Soil 245: 327-334.
- Ladd, J.N.; Amato, M. 1986. The fate of nitrogen from legume and fertilizer sources in soils successively cropped with wheat under field conditions. Soil Biology and Biochemistry 18: 417-425.
- Mayer, J; Buegger, F.; Jensen, E.S.; Schloter, M.; Hess, J. 2003. Estimating N rhizodeposition of grain legumes using a 15N in situ stem labelling method. Soil Biology and Biochemistry 35: 21-28.
- Matos, E.S; Mendonça, E.S.; Lima, P.C.; Coelho, M.S.; Mateus, R.F.; Cardoso, I.M. 2008. Green manure in coffee systems in the region of Zona da Mata, Minas Gerais: characteristics and kinetics of carbon and nitrogen mineralization. Revista Brasileira de Ciência do Solo 32: 2027-2035.
- McNeill, A.M.; Zhu, C.Y.; Fillery, I.R.P. 1997. Use of in situ N-15- labeling to estimate the total below-ground nitrogen of pasture legumes in intact soilplant systems. Australian Journal of Agricultural Research 48: 295-304.
- Okito, A.; Alves, B.J.R.; Urquiaga, S.; Boddey, R.M. 2004. Isotopic fractionation during N2 fixation by four tropical legumes. Soil Biology and Biochemistry 36: 1179-1190.
- Russell, C.A.; Fillery, I.R.P. 1996a. Estimates of lupin below-ground biomass nitrogen, dry matter, and nitrogen turnover to wheat. Australian Journal of Agricultural Research 47: 1047-1059.
- Russell, C.A.; Fillery, I.R.P. 1996b. In situ 15N labelling of lupin belowground biomass. Australian Journal of Agricultural Research 47: 1035-1046.
- Sawatsky, N.; Soper, R.J. 1991. A quantitative measurement of the nitrogen loss from the root system of field peas (Pisum arvense L.) grown in soil. Soil Biology and Biochemistry 23: 255-259.
- Schellenberg, D.L.; Morse, R.D.; Welbaum, G.E. 2009. Organic broccoli production on transition soils: Comparing cover crops, tillage and sidedress N. Renewable Agriculture and Food Systems 24: 85-91.
- Scivittaro, W.B.; Muraoka, T.; Boaretto, A.E.; Trivelin, P.C.O. 2003. Transformations of nitrogen from velvet bean and urea used as fertilizers in corn crop. Pesquisa Agropecuária Brasileira 38: 1427-1433 (in Portuguese, with abstract in English).
- Shearer, G.; Kohl, D.H. 1986. N2 fixation in field settings: estimations based on natural 15N abundance. Australian Journal of Plant Physiology 13: 699-756.
- Vargas, T.O.; Diniz, E.R.; Santos, R.H.S.; Lima, C.T.A.; Urquiaga, S.; Cecon, P.R. 2011. Influence of the biomass of legumes on cabbage yield held in two consecutive crops. Horticultura Brasileira 29: 562-568 (in Portuguese, with abstract in English).
- Vargas, T.O.; Diniz, E.R.; Santos, R.H.S.; Almeida, A.R.; Urquiaga, S.; Cecon, P.R. 2012. Production of cabbage grown in pots containing legumes' root and shoot. Revista Ceres 59: 689-694.
- Wichern, F.; Mayer, J.; Joergensen, R.G.; Müller, T. 2007a. Rhizodeposition of C and N in peas and oats after 13C15N double labelling under field conditions. Soil Biology and Biochemistry 39: 2527-2537.
- Wichern, F.; Mayer, J.; Joergensen, R.G.; Müller, T. 2007b. Release of C and N from roots of peas and oats and their availability to soil microorganisms. Soil Biology and Biochemistry 39: 2829-2839.
- Wichern, F.; Eberhardt, E.; Mayer, J.; Joergensen, R.G.; Müller, T. 2008. Nitrogen rhizodeposition in agricultural crops: methods, estimates and future prospects. Soil Biology and Biochemistry 40: 30-48.
- Wivstad, M. 1999. Nitrogen mineralization and crop uptake of N from decomposing 15N labeled red clover and yellow sweetclover plant fractions of different age. Plant and Soil 208: 21-31.
Publication Dates
-
Publication in this collection
03 Dec 2013 -
Date of issue
Dec 2013
History
-
Received
23 Aug 2012 -
Accepted
28 June 2013