Abstract
An outstanding feature of poultry production that provides animal protein yield for human feeding is its short production cycle. This characteristic has a linear relationship with waste production. Increasing the inclusion of this residue in diets in the near future is desirable in step with the growth of poultry production since it offers a better environmental and nutritional alternative to current methods. We evaluated the effects on the performance and carcass characteristics of broiler chickens produced by the inclusion of poultry offal meal (POM) in their feed. Treatments consisted of a control diet (corn, Zea mays and soybean, Glycine max) and four diets with inclusion of 30, 60, 90 and 120 g kg-1 of POM. The diets were formulated based on the level of digestible amino acid once categorized as isocalcic, isophosphoric, isosodic, isoenergetic and isonutritive for protein, methionine+cystine, lysine and threonine. The feed's electrolytes were corrected so that each diet had the same electrolytic balance. The variables analyzed were feed intake, weight gain, feed conversion ratio, body weight, carcass yield, chicken cut yield and abdominal fat. Feed intake was not affected by the quantities of POM added. The weight gain, feed conversion, carcass yield and noble cuts presented quadratic responses to the treatments. Abdominal fat increased linearly. The performance of the poultry, and carcass characteristics were maximized by the inclusion of 53 and 65 g kg-1, respectively, of POM in the diet, and the inclusion of 120 g kg-1 of POM provided greater disposition of abdominal fat.
alternative ingredient; byproduct of poultry; digestible amino acid
ANIMAL SCIENCE AND PASTURES
Poultry offal meal in broiler chicken feed
Edney Pereira da SilvaI; Carlos Bôa-Viagem RabelloII,* * Corresponding author < cbviagem@dz.ufrpe.br> ; Michele Bernardino de LimaI; Jorge Vitor LudkeIII; Emmanuele Maria Florêncio de ArrudaII; Luiz Fernando Teixeira AlbinoIV
ISão Paulo State University/FCAV, Via de Acesso Prof. Paulo Donato Castellane s/n - 14884-900 - Jaboticabal, SP - Brazil
IIFederal Rural University of Pernambuco - Dept. of Animal Science, R. Dom Manoel de Medeiros, s/n, Dois Irmãos - 52171-900 - Recife, PE - Brazil
IIIEmbrapa Swine and Poultry, C.P. 21 - 89700-000 - Concórdia, SC - Brazil
IVFederal University of Viçosa - Dept. of Animal Science, Av. Peter Henry Rolfs s/n, Campus Universitário - 36570-900 - Viçosa, MG - Brazil
ABSTRACT
An outstanding feature of poultry production that provides animal protein yield for human feeding is its short production cycle. This characteristic has a linear relationship with waste production. Increasing the inclusion of this residue in diets in the near future is desirable in step with the growth of poultry production since it offers a better environmental and nutritional alternative to current methods. We evaluated the effects on the performance and carcass characteristics of broiler chickens produced by the inclusion of poultry offal meal (POM) in their feed. Treatments consisted of a control diet (corn, Zea mays and soybean, Glycine max) and four diets with inclusion of 30, 60, 90 and 120 g kg1 of POM. The diets were formulated based on the level of digestible amino acid once categorized as isocalcic, isophosphoric, isosodic, isoenergetic and isonutritive for protein, methionine+cystine, lysine and threonine. The feed's electrolytes were corrected so that each diet had the same electrolytic balance. The variables analyzed were feed intake, weight gain, feed conversion ratio, body weight, carcass yield, chicken cut yield and abdominal fat. Feed intake was not affected by the quantities of POM added. The weight gain, feed conversion, carcass yield and noble cuts presented quadratic responses to the treatments. Abdominal fat increased linearly. The performance of the poultry, and carcass characteristics were maximized by the inclusion of 53 and 65 g kg1, respectively, of POM in the diet, and the inclusion of 120 g kg1 of POM provided greater disposition of abdominal fat.
Keywords: alternative ingredient, byproduct of poultry, digestible amino acid
Abbreviations: POM, poultry offal meal; FI, feed intake; WG, weight gain; FCR, feed conversion ratio; E, equation; FAT, deposition of fat in the abdominal and cloacal region.
Introduction
Among the branches of short-cycle animal protein yield for human intake, poultry production stands out thanks to its yield capacity. Concomitantly, this feature demonstrates a linear relationship with waste production. The poultry industry is able to absorb all the waste feed produced (Silva et al., 2011). Therefore, the increased inclusion of this residue in diets is desirable in the near future, with the growth of poultry production, because it provides a better environmental and nutritional alternative to current methods.
Currently, advances in the techniques applied in the nutrition of these animals and the accessibility of certain analysis to producers can even further increase the level of inclusion of that ingredient in poultry diets because POM (poultry offal meal) is an ingredient with specific characteristics, supplying amino acids, vitamins, minerals and energy, as well as being a resource generated inside the chain. Therefore, POM suffers less from market price speculation compared with soybean meal.
Even with these features, in the literature, little more than ten scientific papers are related to different recommendations regarding the optimal level of inclusion of this ingredient in diets. The lack of consensus has been attributed to variability in the composition of the flours and the different criteria used in the formulations. However, some researchers have announced positive results from the application of new concepts in the formulation of experimental diets, such as correction for metabolizable energy and digestible amino acids, the ratios of calcium and phosphorus of the diets, which have demonstrated improvements in the birds' performance (Bellaver et al., 2001; Caires et al., 2010). This study evaluated the effects on the performance and carcass characteristics of broiler chickens resulting from the inclusion of POM in their diet.
Materials and Methods
The present study was conducted in the municipality of Carpina, state of Pernambuco, Brazil (7º84' S; 35º25' W, 184 m a.s.l.).
Animals, experimental design, housing and management
700 one-day-old male chicks, Ross 308 breed, with initial mean weight of 45.5 g were selected and distributed in a completely randomized experimental design, with five treatments and seven repetitions composed of 20 animals per experimental unit (1.3 × 2.0 m). The environmental temperature and air humidity were recorded daily at 9h00, 15h00 and 21h00, including the maximum and minimum, using a digital thermo-hygrometer (Table 1). A lighting program of 23 h diary was adopted, except in the first week, when 24 h of light were provided. Starting from the 18th day of life of the chicks, four ventilators were installed, two in each row of boxes.
Experimental treatments and preparation of the poultry diet
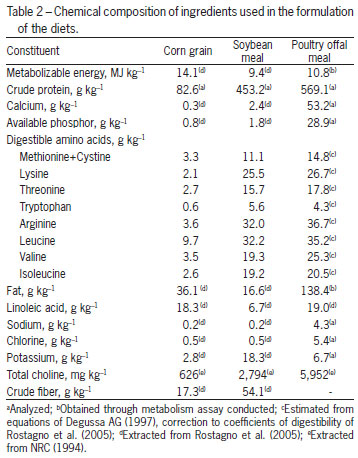
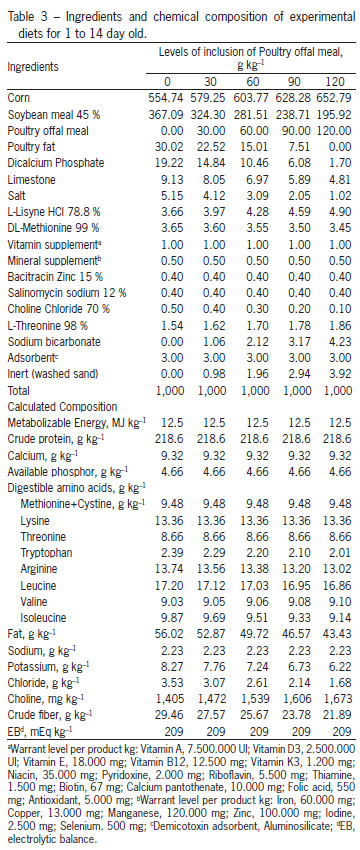
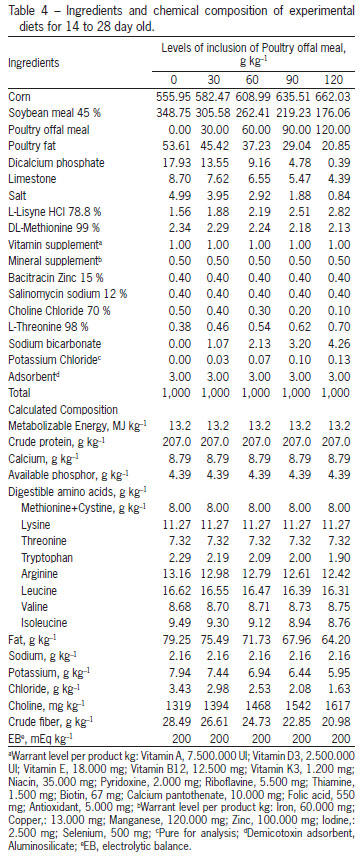
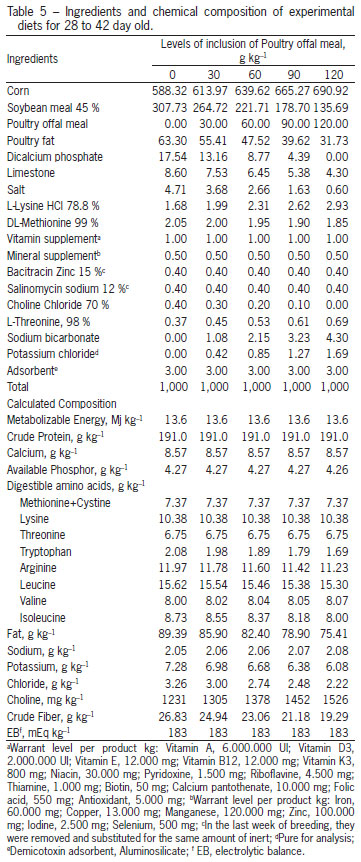
The treatments consisted of a control ration, based on corn (Zea mays) and soybean (Glycine max), T1, 0 g kg-1 of POM and four other treatments, composed by increasing the levels of inclusion of POM as follows: T2 - 30 g kg-1, T3 - 60 g kg-1, T4 - 90 g kg-1 and T5 - 120 g kg-1. To formulate the rations, a nutritional matrix was used for the ingredients corn, soybean meal and POM, according to Table 2. The food program applied was based on phases: initial (1 to 14), growth (14 to 28) and final (28 to 42 days old). The POM used throughout the experiment was part of the same lot. The rations are presented in Tables 3, 4 and 5. To allow the inclusion of 120 g kg-1 of POM in the experimental rations and maintain a constant calcium:phosphorus ratio, in the last phase (28 to 42 days), calcium and phosphorus were added at 6.4 and 6.2 %, respectively. To correct for the same electrolytic balance among the rations, the sodium and potassium values were fixed, with just the chloride values varying (Mongin, 1981).
Data Collection
The weight gain (WG) and feed intake were recorded weekly. To correct the feed intake (FI) and feed conversion ratio (FCR), the mortality occurring during the experimental period was computed. At the end of the performance experiment ( 42 days), two birds with the mean weight were selected and identified to represent the experimental unity in weight evaluation and in the carcass and parts yield. After being selected, the birds were submitted to 6 h of fasting. The birds were sacrificed using the cervical dislocation method. After this procedure the birds were exsanguinated, carcasses were scalded, plucked eviscerated and weighed without the head, neck, lungs and feet. The carcasses were immersed in water at a temperature close to 4 ºC for 10 min, and then, the carcasses were weighed to obtain the weight of the cold carcass. Next, the cuts were performed, separating the prime cuts (breast, drumstick and thigh) and the wings and back, all of them with skin. The abdominal fat value was obtained by adding up the fat from the abdominal region and the fat from the gizzard. The characteristics of the evaluated carcasses included the weight of the cold carcass, the weight of the cuts (breast, drumstick, thigh and wing), and the percentage of abdominal fat. The cold carcass yield was determined in relation to the weight at slaughter. The yield of the parts and the abdominal fat were obtained in relation to the cold carcass weight.
Statistical Analysis
The performance and carcass data from the broiler chickens were submitted initially to analysis to verify the normality of errors and homogeneity of variance using the Cramer-von Mises and Brown and Forsythe tests, respectively. Once the presuppositions were satisfied in relation to the data, the data were submitted to analysis of variance, considering a probability significance of p < 0.05. The mathematical models considered in the analysis of the data were linear, quadratic polynomial, broken line and segmented of two slopes (Robbins et al., 2006). The choice of regression model, the significance level and the coefficient of determination were considered as R2 = SQmodel/SQtreatment. The statistical procedures were conducted using PROC NLMIXED in SAS (Statistical Analysis System, version 9.2).
Results
The normality presuppositions of errors and homoscedasticity of variance for the performance and carcass results were tested and satisfied.
Performance of Broiler Chickens
The inclusion of POM in the diets favored the WG of the poultry (Table 6). Therefore, it is estimated that the best response to WG occurs with the inclusion of 67.12 g kg-1 of POM, producing a WG of 471.4 g per bird (E1, WG=471.4-0.338×(67.12-POM)-0.605×(POM-67.12). In the growing (14-28 days) and final phases (28-42 days), the equations for WG, in the respective phases, also suggest levels of 53.4 and 63 g kg-1, respectively for maximum performance. These events contributed to improve the FCR of the birds fed with a ration containing POM. According to the equations presented in Table 6, the inclusion of 62 g kg-1 POM in the diet enabled the best FCR throughout the entire period (1-42 days) of the experiment.
Carcass Yield of Broiler Chickens
As for the carcass constituents (Table 7), the majority of the absolute values (weight of cold carcass, breast, drumstick, thigh and wings) showed quadratic behavior, and the higher and lower estimated levels were found to be 57.1, 63.1 and 64.0 g kg-1 of POM for the breast, drumstick and thigh weight, respectively. The deposition of fat in the abdominal and cloacal region (FAT) in the birds increased by approximately 2.62 grams for each percentage point of POM included in the diet (E4, FAT =59.81+2.62×POM, R2 = 96.78 %).
The yield of the carcass parts in relation to the chilled carcass, shown in Table 7, was influenced by the treatments. The greatest improvement achieved in the yield of prime cuts occurred when an estimated mean of 67 g kg-1 POM was included in the diet (82, 60, 60 g kg-1 of POM for the best yield in the breast, drumstick and thigh, respectively), which may be attributed to the amino acid balance of the diets.
Discussion
Performance of Broiler Chickens
From 1 to 42 days, the treatments had no effect on FI. The first three limiting amino acids (methionine+cystine, lysine and threonine) were affected but this not the tryptophan. The amino acid levels are related to the regulation of intake by means of serotonin, ghrelin and gastric inhibitory peptide; such hormones involve tryptophan in their synthesis mechanism (Tsiolakis and Marks, 1984; Zhang et al., 2007). Variations of the plasmatic levels interfered with the standard consumption of swine and birds, as reported by Geelissen et al. (2006).
The magnitude of the effects of the tryptophan level on the feed may be inferred, although such mechanisms are still only partially explained in birds, because some formulations of POM tested with high levels and, without supplementation with synthetic amino acids, became deficient in this amino acid. Bellaver et al. (2001) used formulations based on corn and soybean meal with the inclusion of 140 g kg-1 of POM without tryptophan supplementation, and did not verify the effect of tryptophan on FI. Hassanabadi et al. (2008) also verified similar behavior with the inclusion of 150 g kg-1 of POM in diets based on corn (Zea mays), soybean (Glycine max), wheat (Triticum spp.) and meat meal.
The increased inclusion of POM and the supplementation of synthetic amino acids in the feed, with a subsequent decrease of soybean, demonstrated positive effects. That effect may be related to an improvement in the osmolarity of the birds' digestion by decreasing viscosity (Leske and Coon, 1999). Such a condition is essential to obtaining optimal activity of the hydrolytic enzymes present within the intestinal lumen.
Leske and Coon (1999) also reported that a decrease of soybean oligosaccharide (galactosides) enhanced the use of the essential amino acids (3.3; 4.4 and 5.6 % for lysine, threonine and methionine, respectively), and non-essential amino acids increased digestibility by 11.7 % (alanine). Thus, the decrease of such constituents by the removal of soybean meal from the diet may have improved hydrolysis in the lumen and in the intestinal mucosa.
Another hypothesis refers to the presence of protease inhibitors in the composition of soybean meal that, as reported by Feng et al. (2007), correspond to almost 3 mg g-1 of soybean dry matter. The use of processing to avoid such undesirable components is unavoidable. Parsons (1996) found a reduction of digestible lysine by 14.3 and 24.2 %, respectively, when the processing of soybean meal moved from mild to severe. The alteration in the diet's digestible lysine directly affects the relationships between amino acids that use lysine as a reference. This hypothesis is derived from the fact that inclusion of POM has improved the relation of lysine/amino acids in the diet. Bellaver et al. (2001) also verified improved performance for birds fed with POM. Superiority in WG was attributed to the improved balance of amino acids offered with the inclusion of 73.7 g kg-1 of POM in the diet, with supplementation of synthetic methionine and lysine.
Some authors found an equivalence between the protein, POM and soybean meal sources during this phase (Bellaver et al., 2001; Caires et al., 2010), which did not occur in the present study, in which the results obtained in the initial phase were maintained in the subsequent phases. Currently, data regarding the amino acid digestibility of ingredients are used to formulate feed for birds. The experimental feed was formulated in this way; however, the differences were maintained in the growth and final phases. This finding is attributed to a possible improvement in the availability of amino acids in the diet, above all lysine, with the removal of 280 g kg-1 of protein from the soybean meal, assuming that the processing of the feed may have caused greater incidence of lysine complex, which is reported to disappear in the gastrointestinal tract but is not used in protein anabolism (Plakas et al., 1988).
The POM and the soybean meal, similarly, use temperature as a main agent during processing, and among the factors that interfere in the quality of protein in food, the thermal treatment stands above all else. This step is necessary because both ingredients require denaturing and annulling of possible anti-nutritional and microbiological activities.
The occurrence of amino acid-carbohydrate combination complex is common and may be aggravated according to the degree of processing (Parsons, 1996). These include groupings, such as N 1-(1-deoxy-D-fructosyl)-Lysine, derived by the condensation of the -NH2 groups from residual lysine and the carbonyl groups of reducing sugars (glucose, fructose, lactose or maltose), by means of the Maillard reaction. Such an event may cause a decrease in available lysine content, which means that intestinal absorption still takes place (Plakas et al., 1985); however, at the cell level, the transcription by tRNAlis does not occur once this messenger is specific; consequently, the reading is not carried out, and codification to protein synthesis does not occur (Voet and Voet, 2010).
The greater efficiency of nutrients and energy in the diets with POM may be inferred to decrease the anti-nutritive compounds, such as trypsin and galactoside, present in soybean. Feng et al. (2007) quantified the enzymatic activity (in units of substrate composed per milligram of dry matter from digestion) in the duodenum, jejunum and ileum and verified an increase of 52 % in the proteolytic activity of trypsin in the duodenum and jejunum, with a decrease of this compound within the lumen. Furthermore, this activity increases the height of the villus and the depth of the crypt in the three segments of the large intestine, resulting in greater capacity of digestion and absorption. Such effects were verified by Mendonça Jr and Jensen (1989) using another methodology, in growth experiments, with the inclusion of 100 g kg-1 POM in diets based on corn, soybean and chicken fat. By means of partial collection of the excrement with 20 g kg-1 of chromic oxide, the authors observed that the inclusion of POM increased the capacity for metabolizing energy from the diet by up to 5.2 percentile units. This greater assimilation reflects a significant improvement in the birds' FCR.
Escalona and Pesti (1987) conducted two experiments. During the first, they verified in practical diets (with supplementation of methionine) based on corn and soybean meal that reaching 13.1 g kg-1 of total lysine did not overcome the FCR of birds fed with 50 g kg-1 POM, reaching 12.4 g kg-1 total lysine. The second experiment used just one different POM from another poultry rendering plant; the referred authors verified that the result obtained with inclusion of POM (5 to 10 %) in practical diets is not linear and depends on the POM used. The birds fed with inclusion of POM were less efficient, even with supplementation of lysine in the diet.
Carcass Yield of Broiler Chickens
A linear effect on abdominal fat with the replacement of POM in the diets of 0.115 g kg-1 (per unit inclusion) was observed. Mendonça Jr and Jensen (1989) and Hassanabadi et al. (2008) also verified that inclusion of POM in diets based on corn and soybean increased the deposition of fat by the birds. The greater disposition of FAT with inclusion of the POM is a proof that with a better usage of nutrients and energy, when an imbalance of amino acids occurs in the diet, the FAT tends to decrease (Yeh and Leveille, 1969; Cabel et al., 1988).
Aletor et al. (2000) verified that as the crude protein of the diet was reduced by using synthetic amino acid supplementation, the amino acid profile of the diet improved, which was later reflected in the lysine retention in the carcass and a greater deposition of FAT by the bird. This is because enzymes that are part of the synthesized complex in lipogenesis are sensitive to the levels of NAD; therefore, the equivalent of NADH reduction to NADP+ by the enzymes (NAD-malic dehydrogenase and malic enzyme) must be favored, so that biosynthesis of fatty acids may occur (Yeh and Leveille, 1969).
When an imbalance occurs, it causes an increase in the enzymatic activities of glutamic oxaloacetic transaminase and isocitrate dehydrogenase, which demand, via mitochondrial citrate, the intermediary alpha ketoglutaric acid that depends on NADPH for its activation (Rosebrough et al., 1990). The carbonic structure in the gluconeogenic process, converted into glucose, reduces the equivalent of the reduction (of NADPH levels) normally required by the addition of residues of acetyl during lipogenesis. In this way, through the decrease of the citrate and NADPH levels, the decrease of malic enzyme activity is verified. This is aggravated in birds, as there are reports that the carbonic structure cannot maintain the levels of NADPH by the pentose cycle (Yeh and Leveille, 1969).
In conclusion, the performance of poultry and improvement in the carcass and prime cuts were maximized with the inclusion of 53 to 67 g kg-1 POM, respectively, in the diet, and the inclusion of 120 g kg-1 POM (the maximum level included in this work) offered greater disposition of abdominal fat.
Acknowledgments
The authors thank CNPq (Brazilian National Council for Scientific and Technological Development) for providing the scholarship, Banco do Nordeste for financing the research, Agropecuária Serrote Redondo Company for donating the poultry offal meal, and the Pro-rectory of the Graduate School of the Federal Rural University of Pernambuco for financial aid to publish this article.
Received April 04, 2013
Accepted January 06, 2014
Edited by: Gerson Barreto Mourão
- Aletor, V.A.; Hamid, I.I.; Nieb, E.; Pfeffer, E. 2000. Low-protein amino acid-supplemented diets in broiler chickens, effects on performance, carcass characteristics, whole-body composition and efficiencies of nutrient utilization. Journal of the Science of Food and Agriculture 80:547-554.
- Bellaver, C.; Brum, P.A.R.; Lima, G.M.M.; Boff, J.; Kerber, J. 2001. Partial substitution of soybean meal by poultry offal meal in diets balanced according to protein and total or digestible amino acids for broilers. Brazilian Journal of Poultry Science 3: 233-240 (in Portuguese, with abstract in English).
- Cabel, M.C.; Goodwin, T.L.; Waldroup, P.W. 1988. Feather meal as nonspecific nitrogen source for fat reduction in broiler during the finishing period. Poultry Science 67:300-306.
- Caires, C.; Fernandes, E.; Fagundes, N.; Carvalho, A.; Maciel, M.; Oliveira, B. 2010. The use of animal byproducts in broiler feeds: use of animal co-products in broilers diets. Brazilian Journal of Poultry Science 12:41-46.
- Degussa AG. 1997. Amino Acid Recommendations for Poultry: Feed Formulation Guide. Degussa AG, Hanau, Germany.
- Escalona, R.R.; Pesti, G.M. 1987. Nutritive value of poultry by-product meal. 3. Incorporation into practical diets. Poultry Science 66:1067-1070.
- Feng, J.; Liu, X.; Xu, Z.R.; Lu, Y.P.; Liu, Y.Y. 2007. Effects of fermented soybean meal on intestinal morphology and digestive enzyme activities in weaned piglets. Digestive Diseases and Sciences 52:1845-1850.
- Geelissen, S.M.; Swennen, Q.; Geyten, S.V.; Kühn, E.R.; Kaiya, H.; Kangawa, K.; Decuypere, E.; Buyse, J.; Darras, V.M. 2006. Peripheral ghrelin reduces food intake and respiratory quotient in chicken. Domestic Animal Endocrinology 30:108-116.
- Hassanabadi, A.; Amanloo, H.; Zamanian. M. 2008. Effects of substitution of soybean meal with poultry by-product meal on broiler chickens performance. Journal of Animal and Veterinary Advances 7:303-307.
- Leske, K.L.; Coon, C.N. 1999. Hydrogen gas production of broiler chicks in response to soybean meal and α-galactoside free, ethanol-extracted soybean meal. Poultry Science 78:1313-1316.
- Mendonça Jr, C.X.; Jensen, L.S. 1989. Effect of formulating diets with different assigned energy data for poultry by-product meal on the performance and abdominal fat: content of finishing broilers. Poultry Science 68:1672-1677.
- Mongin, P. 1981. Recent advances in dietary anion-cation balance: applications in poultry. Proceedings of the Nutrition Society 40:285-294.
- National Research Council. 1994. Nutrient Requirements of Poultry. 9ed. National. Academies Press, Washington, DC, USA.
- Parsons, C.M. 1996. Digestible amino acids for poultry and swine. Animal Feed Science and Technology 59:147-153.
- Plakas, S.M.; Lee, T.C.; Wolke, R.E.; Meade, T.L. 1985. Effect of maillard browning reaction on protein utilization and plasma amino acid response by rainbow trout (Salmo gairdneri). Journal of Nutrition 115:1589-1599.
- Plakas, S.M.; Lee, T.C.; Wolke, R.E. 1988. Bioavailability of lysine in maillard browned protein as determined by plasma lysine response in Rainbow Trout (Salmo gairdneri). Journal of Nutrition 118:19-22.
- Robbins, K.R.; Saxton, A.M.; Southern, L.L. 2006. Estimation of nutrient requirements using broken-line regression analysis. Journal Animal Science 84 E155-165.
- Rosebrough, R.W.; Mitchell, A.D.; Von Vleck, M.F.; Steele, N.C. 1990. Protein and energy relations in the broiler chicken. British Journal of Nutrition 64:515-523.
- Rostagno, H.S.; Albino, L.F.T.; Donzele, J.L.; Gomes, P.C.; Oliveira, R.F.; Lopes, D.C.; Ferrreira, A.S; Barreto, S.L.T. 2005. Brazilian Tables for Poultry and Swine: Composition of Feedstuffs and Nutritional Requirements. UFV, Viçosa, MG, Brazil.
- Silva, E.P.; Lima, M.B.; Rabello, C.B.V.; Ludke, J.V.; Albino, L.F.T.; Sakomura, N.K.; 2011. Nutritional aspects of poultry offal meal and its use for broiler chicken feeding. Acta Veterinaria Brasilica 5:108-118 (in Portuguese, with abstract in English).
- Tsiolakis, D.; Marks, V. 1984. The differential effect of in tragastric and intravenous tryptophan on plasma glucose, insulin, glucagon, GLI and GIP in the fasted rat. Hormone and Metabolic Research 16:226-229.
- Voet, D.; Voet, J.G. 2010. Biochemistry. 4ed. John Wiley, Hoboken, NJ, USA.
- Yeh, Y.Y.; Leveille, G.A. 1969. Effect of dietary protein on hepatic lipogenesis in the growing chick. Journal of Nutrition 98:356-366.
- Zhang, H.; Yin, J.; Li, D.; Zhou, X.; Li, X. 2007. Tryptophan enhances ghrelin expression and secretion associated with increased food intake and weight gain in weanling pigs. Domestic Animal Endocrinology 33:47-61.
Publication Dates
-
Publication in this collection
15 May 2014 -
Date of issue
June 2014
History
-
Received
04 Apr 2013 -
Accepted
06 Jan 2014