Abstract
Little information on the epidemiology and clinical characteristics of human parainfluenza virus (HPIV) infections, especially in children from tropical countries, has been published. The aim of this study was to determine the frequency of HPIV infections in children attended at a large hospital in Fortaleza in Northeast Brazil, and describe seasonal patterns, clinical and epidemiological characteristics of these infections. From January 2001 to December 2006, a total of 3070 nasopharyngeal aspirates collected from children were screened by indirect immunofluorescence for human parainfluenza viruses 1, 2, and 3 (HPIV-1, 2 and 3) and other respiratory viruses. Viral antigens were identified in 933 samples and HPIV in 117. The frequency of HPIV-3, HPIV-1 and HPIV-2 was of 83.76%, 11.96% and 4.27%, respectively. Only HPIV-3 showed a seasonal occurrence, with most cases observed from September to November, and with an inverse relationship to the rainy season. Most HPIV-3 infections seen in outpatients were diagnosed as upper respiratory tract infections.
Acute respiratory infections; parainfluenza virus; epidemiology; Brazil
ORIGINAL PAPERS
Parainfluenza virus infections in a tropical city: clinical and epidemiological aspects
Mariana Mota Moura FéI; André Jalles MonteiroII; Fernanda Edna Araújo MouraI
IPost-Graduation Program in Medical Microbiology, Virology Section, Department of Pathology and Forensic Medicine
IIDepartment of Statistics and Applied Mathematics; Federal University of Ceará; Fortaleza, CE, Brazil
Address for correspondence Address for correspondence: Dr. Fernanda Edna Araújo Moura E-mail: fernandaedna@terra.com.br Phone-fax: 55(85)33668303-55(85)33668316 Rua Alice, 58, C-07. Cidade dos Funcionários, 60822-610, Fortaleza- CE, Brazil. Zip code: 60822-610. Financial support: Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq- Grant: 620053/2004-6) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES).
ABSTRACT
Little information on the epidemiology and clinical characteristics of human parainfluenza virus (HPIV) infections, especially in children from tropical countries, has been published. The aim of this study was to determine the frequency of HPIV infections in children attended at a large hospital in Fortaleza in Northeast Brazil, and describe seasonal patterns, clinical and epidemiological characteristics of these infections. From January 2001 to December 2006, a total of 3070 nasopharyngeal aspirates collected from children were screened by indirect immunofluorescence for human parainfluenza viruses 1, 2, and 3 (HPIV-1, 2 and 3) and other respiratory viruses. Viral antigens were identified in 933 samples and HPIV in 117. The frequency of HPIV-3, HPIV-1 and HPIV-2 was of 83.76%, 11.96% and 4.27%, respectively. Only HPIV-3 showed a seasonal occurrence, with most cases observed from September to November, and with an inverse relationship to the rainy season. Most HPIV-3 infections seen in outpatients were diagnosed as upper respiratory tract infections.
Key-Words: Acute respiratory infections, parainfluenza virus, epidemiology, Brazil.
Human parainfluenza viruses (HPIV) are genetically and antigenically divided into types 1 to 4 [1]. HPIV-1 to HPIV-3 are major causes of acute respiratory infections (ARI), including lower respiratory tract infections, in infants and children. These three types of HPIV have been more extensively investigated than HPIV-4 [2-4]. All subtypes can cause a full spectrum of respiratory illness, including upper respiratory infections, croup, bronchiolitis and pneumonia [1,5]. Seasonal patterns of HPIV infections are distinct among the subtypes and geographic regions. In the USA, HPIV-2 can cause yearly outbreaks or biennial epidemics in co-circulation with HPIV-1 during the fall seasons, while HPIV-3 causes yearly spring and summer epidemics [5-10]. In England and Wales HPIV-3 causes yearly epidemics in the summer as shown [11]. In the northeast of England, a hospital-based study of the epidemiology of respiratory infections among pediatric inpatients over a six-year period showed that HPIV-1 and HPIV-2 epidemics occurred together, but only every 2 years [12]. In tropical countries, there have been fewer studies of HPIV and most of them do not demonstrate a seasonal pattern of HPIV infections [13,14].
The purposes of the study were: (1) To determine the rate of HPIV infections among children attended at the Albert Sabin Children's Hospital (HIAS) over six consecutive years; (2) To describe the clinical and epidemiological characteristics of these infections; (3) To show the seasonal pattern of HPIV infections and its relation with the rainy season and with periods of activity of other respiratory viruses; and (4) To compare the clinical characteristics of parainfluenza infections with infections caused by respiratory syncytial virus (RSV), influenza and adenovirus.
Material and Methods
Site of Study
This study was conducted at HIAS, a teaching hospital where care is provided to children from low income families living in Fortaleza and other cities in the state of Ceará. The city of Fortaleza is located at sea level, 4º south of the equator and has a tropical climate characterized by two distinct seasons: the rainy season, that occurs in the first semester of each year, usually from January to June, and the dry season, during the rest of the year. The humidity is high (>70%), and there is little temperature variation throughout the year. During this study, the minimum and maximum daily temperatures in Fortaleza were 21.1ºC and 33.9º C, respectively.
Population of Study
The patients included in this study were children and teenagers (0 to 16 years old) with acute respiratory symptoms. They were attended at the emergency department, outpatients' clinics and pediatric wards of HIAS from January 2000 to December 2006.
Inclusion Criteria
Patients were included in the study if they had one or more of the following symptoms with or without fever (>37.5ºC): cough, coryza, sore throat, earache, breathing difficulty, stridor and/or wheezing within seven days of onset.
Epidemiological and Clinical Data
The guardians and parents of each child were interviewed by our researchers to obtain basic information about symptoms, clinical presentation, risk factors and comorbidities. Description of physical signs, medical diagnosis, treatment and results of thoracic X-Rays were obtained from pediatricians at each setting of attendance.
Sample Collection
Nasopharyngeal aspirates were obtained from the patients by aspiration as described previously [15]. They were analyzed by indirect immunofluorescence assay (IFA) performed using the Respiratory Panel I Viral Screening and Identification kit (Chemicon International, Inc., Temucula, CA, USA), following the manufacturer's instructions, for detecting antigens of the following respiratory viruses: HPIV-1, 2 and 3, RSV, influenza A and B, and adenovirus. These samples were not cultured for the detection of bacteria.
Statistical Analysis
Descriptive statistics (mean, standard deviation, Student test) were used for univariate analysis. Comparisons were carried out using the Fisher exact test and Pearson's χ2 for categorical variables. All p values were considered significant if < 0.05. Spearman's correlation test was used to correlate the total number of ARI cases, the number of HPIV-3 cases and monthly rainfall.
Ethical Issues
The parents of the children included in the study were required to sign a formal written consent form, which was carefully explained to them. This study was approved by the Ethical Committee of HIAS.
Results
A total of 3,070 samples of nasopharyngeal aspirates were collected in the six-year study. The majority of the children with respiratory symptoms were attended mostly in the emergency room (58.66%), followed by outpatient's clinics (29.08%) and the pediatric wards (11.98%). Respiratory viruses were detected in 976 cases (31.8%), and there were 117 cases of HPIV (3.81%). HPIV-3 was the most frequently detected type among HPIV- positive cases, accounting for 98 cases (83.76%). HPIV-1 and HPIV-2 were detected in 14 (11.96%) and, 5 (4.27%) cases, respectively. The monthly distribution of infections caused by HPIV (1 to 3) during the period of study is shown in Figure 1. In summary, the viruses most frequently detected in the study were RSV (573), influenza A (200), HPIV-3 (98) and adenovirus (57). Mixed viral infections including HPIV were observed in six cases: HPIV-1 + RSV (two cases), HPIV1+RSV+ influenza A (1 case), HPIV-3 + influenza A + adenovirus (1 case), HPIV-1+ HPIV-3 (1 case) and, HPIV-2 + HPIV-3 (1 case).
As there were a high number of positive cases for HPIV-3 and low number of positive cases for HPIV-1 and HPIV-2 found in this study, a statistical analysis of epidemiological and clinical characteristics was able to be done by comparing infections caused by HPIV-3 with those caused by the other more common viruses detected in the study (RSV, influenza A and adenovirus).
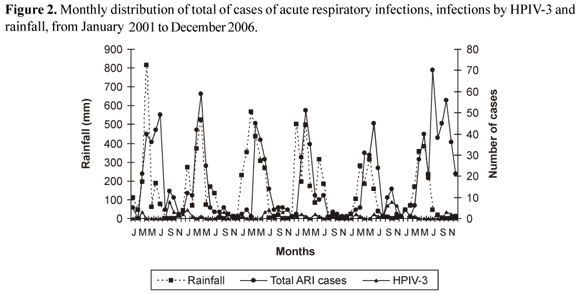
The results of this study show a statistically significant association in the occurrence of ARI with the rainy season (p<0.0001).The seasonal pattern of HPIV-3 in Fortaleza was characterized by the predominant occurrence from September to November (p<0.0001) and an inverse correlation of these infections with the rainy season (p=0.0067) (Figure 2). There was a contrasting relation of epidemic periods of RSV and influenza (associated to rainy periods) with HPIV-3 infections. There was not a correlation of occurrence of HPIV-3 infections and adenovirus infections (Figure 3).
Clinical and epidemiological characteristics of the study population and of children infected by HPIV are showed in the Table 1. The majority of children with ARI were outpatients (88%), as were the majority of HPIV patients (88%) and children infected by the most frequently detected viruses in the study. Hospitalized children represented 12% of the study population, and 12% of those were infected by HPIV. The number of hospitalized children due to RSV infection was higher than that caused by HPIV-3, and this was statistically significant (p=0.0223). The mean age of patients infected by HPIV-3 (20 months) was significantly higher than RSV cases (15 months) (p=0.0215) and lower than influenza A cases (34 months) (p<0.0001). No statistically significant differences were observed in relation to the age of children infected by HPIV- 3 or by other viruses analyzed in this study.
Upper airway infections were the most frequent clinical syndrome among HPIV-3 infected patients, diagnosed in 48% of the cases. These infections were more associated to HPIV-3 than RSV (p<0.0001) but less than influenza A (p=0.0285). The number of patients diagnosed with pneumonia due to HPIV-3 was statistically lower than caused by RSV (p=0.0007) and bronchiolitis in HPIV-3 patients was less frequent than in RSV patients (p=0.0068) but more frequent than in influenza A patients (p=0.0218).
Regarding clinical manifestations and chest X-ray findings more significant differences were observed between HPIV- 3 infected patients and RSV or influenza-infected patients. HPIV-3 patients significantly presented less dyspnea, cough, chest retractions, crackles and radiologic abnormalities than RSV patients. Consequently, HPIV-3 patients were prescribed less antibiotics, corticosteroids, oxygen, salbutamol and nebulization than RSV patients. HPIV-3 patients presented more conjunctivitis during episodes of ARI than RSV (p=0.0091) and influenza A (p=0.0420) infected patients and less than children infected by adenovirus (p=0.0960). The number of HPIV-3 infected patients with fever was slightly lower than those infected by influenza A (p=0.0927). Other clinical aspects (coryza, sneezing, vomiting, diarrhea, anorexia, otitis, cyanosis, rash) analyzed did not show statistically significant differences, when comparing infections by HPIV-3 and by other viruses.
Discussion
HPIVs have been underestimated as agents of pediatric ARI although they represent the second most common cause of lower respiratory tract infections in infants after RSV [1]. The rate of detection of HPIV in clinical samples of children included in the present study is within the range of minimum (0.31%) and maximum (13%) rates reported in other studies [16-23]. The highest rates of identification of HPIV (13%) have been achieved using isolation in cell culture and RT-PCR [22,23]. In the present study the rate of identification of HPÌV was comparable to other studies that also used direct and indirect IFA to detect the same agents [16-19]. The use of IFA as the single method to identify these infections may have contributed to the low frequency of HPIV infections in this study. Currently available IFA reagents for HPIV in pediatric specimens have shown variable sensitivity (0-100%) [1]. Factors such as specimen types, collection, processing, HPIV types and reagents help explain the reported variability in sensitivities. Although less sensitive than conventional cell culture, IFA has been uniformly specific, less expensive, and faster than conventional cell culture, allowing viral diagnosis in a few hours [19,24,25].
The predominance of HPIV-3 among the three subtypes of HPIV analyzed in this study was also demonstrated in several studies [6-8,18-20,26,27]. In an Italian study of ARI in hospitalized children, using nasal wash as the clinical sample and molecular techniques for thirteen respiratory viruses, HPIV-3 was the only HPIV found, representing 7.5% of identified viruses [21]. It is interesting to compare the results of the present study with those obtained during a community based study conducted in the city of Fortaleza over 29 consecutive months, when 62 infections by HPIV were identified and HPIV-2, HPIV-1 and HPIV-3 represented 54.8%, 21% and 13%, respectively of HPIV identified [28]. An HPIV-2 outbreak developed during the rainy season (February to June) in the second year of that study, when about 24 HPIV-2 viruses were detected. During this six-year study, the annual number of infections by HPIV-2 did not exceed two cases. Indeed, in the final three years no case of HPIV-2 occurred.
As observed for RSV and influenza, the seasonal pattern of HPIV infections has been described in more detail in studies from temperate areas [7,29,30]. Seasonal patterns of HPIV infections are distinct among the subtypes and geographic regions and have changed since the first descriptions of HPIV, when, in the 50's and 60's, infections by subtypes 1, 2, and 3 occurred throughout the year, in an endemic pattern. In the 70's, HPIV-1 switched to outbreaks in the odd-numbered years. HPIV-2 was more sporadically detected. The initial pattern of HPIV-3 was endemic, but since 1978 it has changed to epidemic with outbreaks in late winter, spring or early summer, in the United States and Australia [3,6,7,17,26,29,30]. A small number of studies have reported an epidemiology of HPIV-4. A Canadian study showed that HPIV-4 infections occur in autumn and winter of some years [31]. In some studies the seasonal distribution of HPIV infections has been described without identifying the serotypes circulating [17,22,32]. During a four-year study of the epidemiology of viral respiratory infections in Korean children, peaks of HPIV infections were identified from around April until June from 1994 to 1997. In this study a second peak of HPIV infections was observed from November 1994 to January 1995 [32]. During a 13 month-study in the district of Tlalpan, Mexico City, HPIV infections were detected throughout the year but were more frequent during summer and autumn [22].
The seasonal pattern of HPIV-3 in the city of Fortaleza during this study shows singular characteristics when compared to those described in the scarce publications relating to HPIV infections in tropical countries [13,14]. In Fortaleza, as in other tropical areas, the rainy season is associated with higher incidences of viral ARI, especially those caused by RSV and influenza [13,14,28,33]. In this study, a correlation between the total number of ARIs and the rainy season was observed, thus agreeing with most studies from tropical areas, where rainfall is the major climatic variable [13,14]. The majority of the cases of HPIV-3 infection in the city of Fortaleza occurred in the dry months, a seasonal pattern which had not previously been described in northeastern Brazil or in any other tropical countries. A previous study of viral ARI in children living in Fortaleza showed different findings for HPIV infections. First, the most prevalent subtype was HPIV-2, and second, the occurrence of HPIV-2 in one of the three years of the study was related to the rainy season [28]. Brazilian studies of viral ARI mention a possible interregional variation. Annual outbreaks of HPIV-3 infections in children living in a shanty town in Rio de Janeiro city were observed during the late winter or spring [34]. In a three-year-study conducted in Porto Alegre, a city located in the South of Brazil, only 10 cases of HPIV-3 were identified out of a total of 862 infections. The HPIV-3 cases were observed from September to November in 1991 and from July to August in 1992 [35].
Regarding the clinical aspects, the infections caused by HPIV-3 were milder than those caused by RSV, where there was a significantly higher involvement of lower respiratory tract, hospitalization, use of antibiotics, salbutamol, corticosteroids and need for oxygen. The occurrence of HPIV-3 infections in older children may have contributed to these characteristics. In several studies RSV is the leading viral pathogen associated with infants and young children being hospitalized due to serious ARI [36,37]. An interesting finding of this study was the lower occurrence of croup (laryngotracheobronchitis), the signal illness for HPIV. Six cases of croup were diagnosed, and only two were due to HPIV, both by HPIV-1. Of the etiologic agents of croup, HPIV -1 accounts for half of the cases and the other half is associated to HPIV-2, HPIV-3, RSV, influenza A, adenovirus and more rarely to Mycoplasma pneumoniae [38,39].
This study emphasizes the importance of HPIV, mainly HPIV-3, as an etiologic agent of ARI in children, and reinforces the need to diagnose viral respiratory infections quickly to provide epidemiological and virological data that may be useful in control efforts and vaccine trials, mostly in developing countries, where less information regarding respiratory viruses is available.
Received on 18 December 2007; revised 22 April 2008.
- 1. Henrickson K.J. Parainfluenza viruses. Clin Microbiol Rev 2003;16:242-64.
- 2. Lau S.K., To W.K., Tse P.W.T., et al. Human parainfluenza virus 4 outbreak and the role of diagnostic tests. J Clin Microbiol 2005;43:4515-21.
- 3. Chiu C.Y., Rouskin S., Koshy A., et al. Microarray detection of human parainfluenzavirus 4 infection associated with respiratory failure in an immunocompetent adult. Clin Infect Dis 2006;46:71-6.
- 4. Vanchon M.L., Dionne N., Leblanc E., et al. Human parainfluenza type 4 infections, Canada. Emerg Infect Dis 2006;12:1755-8.
- 5. Hall C.B. Respiratory syncytial virus and parainfluenza virus. N Engl J Med 2001;344:1917-28.
- 6. Reed G., Jewett P.H., Thompson J., et al. Epidemiology and clinical impact of parainfluenza infections in otherwise healthy infants and young children < 5 years old. J Infect Dis 1997;175:807-13.
- 7. Marx A., Török T.J., Holman R.C., et al. Pediatric hospitalizations for croup (laryngotracheobronchitis): Biennial increases associated with human parainfluenza virus 1 epidemics. J Infect Dis 1997;176:1423-7.
- 8. Weinberg G.A. Parainfluenza viruses an underappreciated cause of pediatric respiratory morbidity. Pediatr Infect Dis J 2006;25:447-8.
- 9. Knott A.M., Long C.E., Hall C.B. Parainfluenza viral infections in pediatric outpatients: seasonal patterns and clinical characteristics. Pediatr Infect Dis J 1994;13:269-73.
- 10. Fry A.M., Curns A.T., Harbour K., et al. Seasonal trends of human parainfluenza viral infections: United States, 1990-2004. Clin Infect Dis 2006;43:1016-22.
- 11. Easton A.J., Eglin R.P. Epidemiology of parainfluenza virus type 3 in England and Wales over a ten-year period. Epidemiol Infect 1989;102:531-3.
- 12. Martin A.J., Gardner P.S., McQuillin J. Epidemiology of respiratory viral infection among paediatric inpatients over a six-year period in north-east England. Lancet 1978;2:1035-8.
- 13. Chew F.T. Seasonal trends of viral respiratory tract infections in the tropics. Epidemiol Infect 1998;121:121-8.
- 14. Shek LP, Lee BW. Epidemiology and seasonality of respiratory tract virus infections in the tropics. Paediatr Respir Rev 2003;4:105-11.
- 15. Gardner P.S., McQuillin J. Rapid virus diagnosis: application of immunofluorescence. 2. ed. London: Butterworth, 1980
- 16. Irmen KE, Kelleher JJ. Use of monoclonal antibodies for rapid diagnosis of respiratory viruses in a community hospital. Clin Diagn Lab Immunol 2000;7:396-403.
- 17. Carballal G., Videla C.M., Espinosa M.A., et al. Multicentered study of viral acute lower respiratory infections in children from four cities in Argentina, 1993-1994. J Med Virol 2001;64:167-74.
- 18. Shetty AK., Treynor E., Hill D.W., et al. Comparison of conventional viral cultures with direct fluorescent antibody stain for diagnosis of community-acquired respiratory virus infections in hospitalized children. Pediatr Infect Dis J 2003;22:789-94.
- 19. Hazlett D.T.G., Bell T.M., Tukei P.M., et al. Viral etiology and epidemiology of acute respiratory infections in children in Nairobi, Kenya. Am J Trop Med Hyg 1988;39:632-40.
- 20. Lina B., Valette M., Foray S., et al. Surveillance of community-acquired viral infections due to respiratory viruses in Rhone-Alpes (France) during winter 1994 to 1995. J Clin Microbiol 1996;34:3007-11.
- 21. Pierangeli A., Gentile M., Di Marco P., et al. Detection and typing by molecular techniques of respiratory viruses in children hospitalized for acute respiratory infection in Rome, Italy. J Med Virol 2007;79:463-8.
- 22. Cabello C., Manjarrez M.E., Olvera R., et al. Frequency of viruses associated with acute respiratory infections in children younger than five years of age at a locality of México City. Mem Inst Oswaldo Cruz 2006;101:21-4.
- 23. Legg J.P., Warner J.A., Johnston S.L., et al. Frequency of detection of picornaviruses and seven other respiratory pathogens in infants. Pediatr Infect Dis J 2005;24:611-6.
- 24. Wong D.T., Welliver R.C., Riddlesberger K.R., et al. Rapid diagnosis of parainfluenza virus infection in children. J Clin Microbiol 1982;16:164-7.
- 25. Ray C.G., Minnich L. Efficiency of immunofluorescence for rapid detection of common respiratory viruses. J Clin Microbiol 1987;25:355-7.
- 26. De Silva L.M., Cloonan M.J. Parainfluenza virus type 3 infections: findings in Sydney and some observations on variations in seasonality worldwide. J Med Virol 1991;35:19-21.
- 27. Tsai H.P., Kuo P.H., Liu C.C., et al. Respiratory viral infections among pediatric inpatients and outpatients in Taiwan from 1997 to 1999. J Clin Microbiol 2001;39:111-8.
- 28. Arruda E., Hayden F.G., McAuliffe J.F., et al. Acute respiratory viral infections in ambulatory children of urban northeast Brazil. J Infect Dis 1991;164:252-8.
- 29. Glezen W.P. Parainfluenza virus type 3: seasonality and risk of infection and reinfection in young children. J Infect Dis 1984;150:851-7.
- 30. Monto A.S. Occurrence of respiratory virus: time, place and person. Pediatr Infect Dis J 2004;23:S58-S64.
- 31. Vachon M.L., Dionne N., Leblanc E., et al. Human parainfluenza type 4 infections in Canada. Emerg Infect Dis 2006;12:1755-8.
- 32. Kim M.R., Lee H.R., Lee G.M. Epidemiology of acute viral respiratory tract infections in Korean children. J Infect 2000;41:152-8.
- 33. Moura F.E.A., Nunes I.F.S., Silva Jr. G.B., et al. Respiratory syncytial virus infections in Northeastern Brazil: seasonal trends and general aspects. Am J Trop Med Hyg 2006;74:165-7.
- 34. Nascimento J.P., Siqueira M.M., Sutmoller F., et al. Longitudinal study of acute respiratory diseases in Rio de Janeiro: occurrence of respiratory viruses during four consecutive years. Rev Inst Med Trop São Paulo 1991;33:287-96.
- 35. Straliotto S.M., Siqueira M.M., Muller R.L., et al. Viral etiology of acute respiratory infections among children in Porto Alegre, RS, Brazil. Rev Soc Bras Med Trop 2002;35:283-91.
- 36. Craig P. Systematic review of the biology and medical management of respiratory virus infection. Respiratory Care 2003;48:209-33.
- 37. Iwane M.K., Edwards K.M., Szilagyi P.G., et al. Population-based surveillance for hospitalizations associated with respiratory syncytial virus, influenza virus, and parainfluenza viruses among young children. Pediatrics 2004;113:1758-64.
- 38. Denny F.W., Murphy T.F., Clyde W.A. Jr., et al. Croup: a 11-year study in pediatric pratice. Pediatrics 1983;71:871-6.
Publication Dates
-
Publication in this collection
25 Sept 2008 -
Date of issue
June 2008
History
-
Received
18 Dec 2007 -
Accepted
22 Apr 2008