Abstract
The fish species Synbranchus marmoratus is widely distributed throughout the Neotropical region and exhibits a significant karyotype differentiation. However, data concerning the organization and location of the repetitive DNA sequences in the genomes of these karyomorphs are still lacking. In this study we made a physical mapping of the H3 and H4 histone multigene family and the transposable elements Rex1 and Rex3 in the genome of three known S. marmoratus karyomorphs. The results indicated that both histone sequences seem to be linked with one another and are scattered all over the chromosomes of the complement, with a little compartmentalization in one acrocentric pair, which is different from observations in other fish groups. Likewise, the transposable elements Rex1 and Rex3 were also dispersed throughout the genome as small clusters. The data also showed that the histone sites are organized in a differentiated manner in the genomes of S. marmoratus, while the transposable elements Rex1 and Rex3 do not seem to be compartmentalized in this group.
Synbranchidae; FISH; histone; retrotransposon
RESEARCH ARTICLE
Scattered organization of the histone multigene family and transposable elements in Synbranchus
Ricardo Utsunomia; José Carlos Pansonato-Alves; Priscilla Cardim Scacchetti; Claudio Oliveira; Fausto Foresti
Departamento de Morfologia, Instituto de Biociências, Universidade Estadual Paulista "Júlio de Mesquita Filho", Botucatu, SP, Brazil.
Send correspondence to Send correspondence to Ricardo Utsunomia. Departamento de Morfologia, Instituto de Biociências, Universidade Estadual Paulista"Júlio de Mesquita Filho", Distrito de Rubião Junior, s/n, 18618- 970 Botucatu, SP, Brazil. E-mail: utricardo@ibb.unesp.br
ABSTRACT
The fish species Synbranchus marmoratus is widely distributed throughout the Neotropical region and exhibits a significant karyotype differentiation. However, data concerning the organization and location of the repetitive DNA sequences in the genomes of these karyomorphs are still lacking. In this study we made a physical mapping of the H3 and H4 histone multigene family and the transposable elements Rex1 and Rex3 in the genome of three known S. marmoratus karyomorphs. The results indicated that both histone sequences seem to be linked with one another and are scattered all over the chromosomes of the complement, with a little compartmentalization in one acrocentric pair, which is different from observations in other fish groups. Likewise, the transposable elements Rex1 and Rex3 were also dispersed throughout the genome as small clusters. The data also showed that the histone sites are organized in a differentiated manner in the genomes of S. marmoratus, while the transposable elements Rex1 and Rex3 do not seem to be compartmentalized in this group.
Key words: Synbranchidae, FISH, histone, retrotransposon.
Introduction
The genomes of eukaryotic organisms are characterlittle information regarding other types of sequences, such as ized by a large number of repetitive DNA segments. These histone genes and transposable elements (TEs). Even though sequences are identifiable by a high variability in their nustudies on physical mapping of histone genes and TEs are cleotide composition, number of copies, function, distribufew, interesting features about those sequences have been retion and organization in the genome (Wagner et al., 1993; vealed, such as association with other repetitive families Charlesworth et al., 1994). Generally, these sequences may (Cioffi et al., 2010; Hashimoto et al., 2011, 2013; Limabe classified as coding sequences, represented by ribo-Filho et al., 2012), distinct modes of organization (Valente etsomal and histone multigene families, and noncoding seal., 2011; Ferreira et al., 2011a), and influences on karyotype quences, repeated in tandem or dispersed throughout the diversification (Pansonato-Alves et al., 2013a).
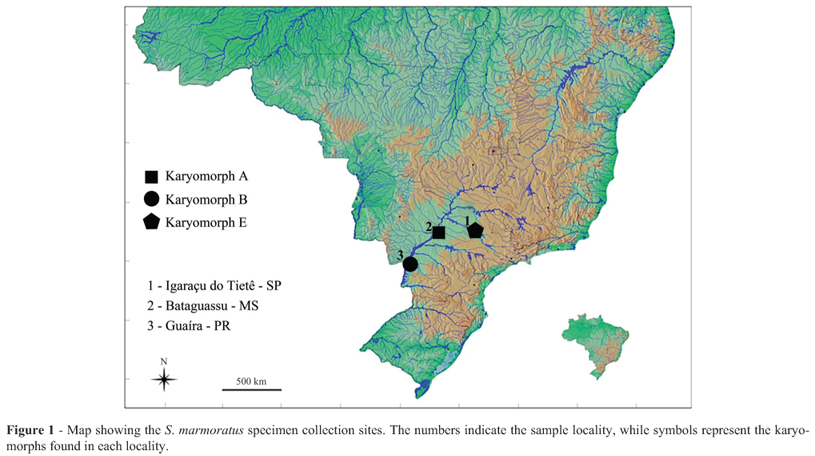
The repetitive nature of these sequences makes themideal for the development of probes for use in fluorescence in situ hybridization (FISH). Studies related to the organization and physical mapping of these types of sequences have enabled a better characterization of the biodiversity and karyoevolution of the ichthyofauna (Vicari et al., 2010). Furthermore, these data have also helped advancing in the knowledge of the organization, diversification, evolution and possible role of repetitive DNA sequences in the genome of vertebrates (Haff et al., 1993; Martins and Galetti Jr.1999; Gursel et al., 2003). However, the vast majority of mapping studies carried out on Neotropical fishes were focused on the location of ribosomal sites, and there is still very little information regarding other types of sequences, such as histone genes and transposable elements (TEs). Even though studies on physical mapping of histone genes and TEs are few, interesting features about those sequences have been revealed, such as association with other repetitive families (Cioffi et al., 2010; Hashimoto et al., 2011, 2013; Lim- Filho et al., 2012), distinct modes of organization (Valente et al., 2011; Ferreira et al., 2011a), and influences on karyotype diversification (Pansonato-Alves et al., 2013a). Although known as a single taxonomic entity, S. marmoratus (Synbranchiformes, Synbranchidae) displays considerable cytogenetic diversity. As a result, there are distinct karyotype variants (Foresti et al., 1992; Melilo et al., 1996; Sánchez and Fenocchio, 1996; Torres et al., 2005), resulting in five well-differentiated main karyomorphs (unpublished data). Individuals in karyomorph groups A and B have chromosome number 2n = 42. In addition, a pericentric inversion in a submetacentric chromosome of karyomorph A is related to the origin of karyomorph B. In contrast, analyses of chromosome rearrangements cannot definitively explain the origins of karyomorphs C (2n = 46), D and E (2n = 46), because the events responsible for their diploid chromosome numbers seem to originate from undependable and bidirectional events (unpublished data). This study describes the cytogenetic mapping of the histone sequences H3 and H4 and of the transposable elements Rex1 and Rex3 in samples from three S. marmoratus karyomorphs. The purpose of this study was to investigate the distribution patterns of each element in this group.
Material and Methods
Samples
Mitotic chromosomes were obtained from kidney tissue, as described by Foresti et al. (1981), from specimens of karyomorphs A, B and E, collected at different Brazilian locations, as specified in Table 1 and Figure 1. All samples were collected in accordance with the Brazilian Environmental Law (Collection permission MMA/IBAMA/SISBIO - Nr. 3245), and the procedures for fish collection, maintenance and analysis were performed in compliance with the Brazilian College of Animal Experimentation (COBEA) and approved (protocol Nr. 503) by the Bioscience Institute/UNESP Ethics Committee on Use of Animals (CEUA). After the analyses, the fishes were fixed in 10% formalin, conserved in 70% ethanol and deposited in the fish collection of the Fish Biology and Genetics Laboratory (Laboratório de Biologia e Genética de Peixes) - UNESP, Botucatu, São Paulo, Brazil. Voucher information is also presented in Table 1.
Isolation of repetitive DNA sequences and FISH
Genomic DNA of S. marmoratus (karyomorph A) was extracted using the Wizard Genomic DNA Purification Kit (Promega). Partial sequences of the histone genes H3 and H4 and the retrotransposable elements Rex1 and Rex3 were obtained by polymerase chain reaction (PCR) using previously described primers (White et al., 1990; Colgan et al., 1998; Volff et al., 1999, 2000; Pineau et al., 2005). During the secondary PCR assay, the H3 and H4 histone sequences were labeled with biotin-16-dUTP (Roche), and the Rex1 and Rex3 TEs and 18S rDNA with digoxigenin- 11-dUTP (Roche), by incorporating these modified nucleotides.
FISH was performed using the method described by Pinkel et al. (1986). Slides were incubated with RNase (50 µg/mL) for 1 h at 37 °C. Then, the chromosomal DNA was denatured in 70% formamide in 2x SSC for 5 min at 70 °C. For each slide, 30 µL of hybridization solution (containing 200 ng of each labeled probe, 50% formamide, 2x SSC and 10% dextran sulphate) were denatured for 10 min at 95 °C, dropped onto the slides and hybridized overnight at 37 °C in a 2x SSC moist chamber. After hybridization, the slides were washed in a 0.2x SSC solution with 15% formamide for 20 min at 42 °C, followed by a second wash in 0.1x SSC for 15 min at 60 °C, and a final wash in 4x SSC with 0.5% Tween for 10 min at room temperature. Probe detection was carried out with Avidin-FITC (Sigma) or anti-digoxigenin-rhodamine (Roche), the chromosomes were counterstained with DAPI-(4',6-diamidino-2-phenylindole, Vector Laboratories), and the FISH images were captured by an optical photomicroscope (Olympus, BX61) with the Image Pro Plus 6.0 software (Media Cybernetics).
Results
Cytogenetic analysis
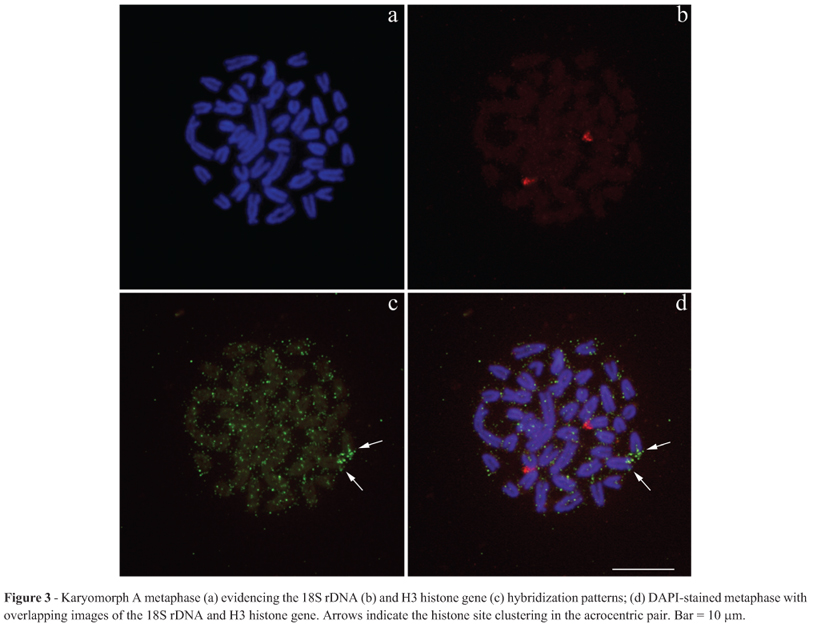
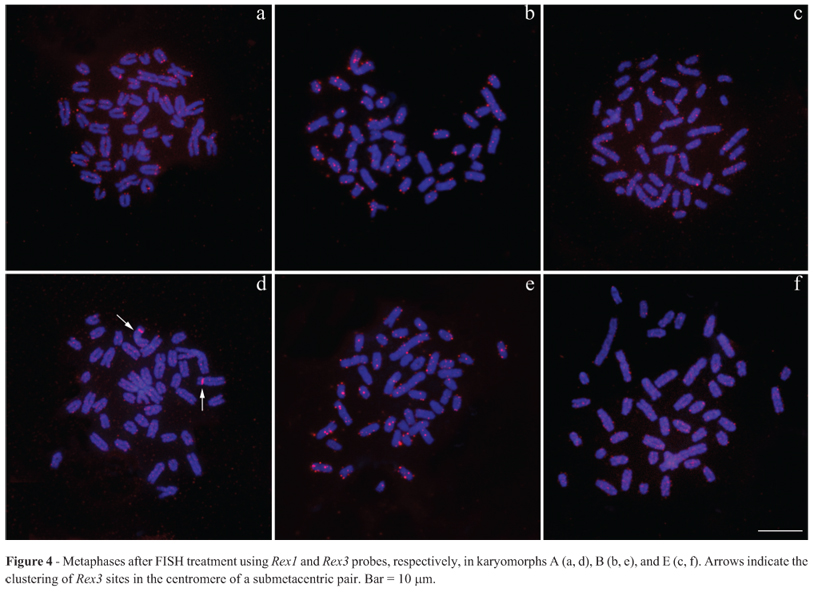
All repetitive probes used here were clearly visualized in the mitotic chromosomes. Both H3 and H4 histone sequences appeared to be clustered together and were distributed in a general pattern with dispersed signals on all chromosomes. Additionally, both sequences were accumulated in one acrocentric pair (Figure 2a-f). Since the histone sites mapped until now in fishes were found in conspicuous blocks, we used the double- FISH technique (18S rDNA + H3 histone sequences), in order to compare the hybridization patterns of both probes and check the veracity of the dispersed signal pattern of the histone sequences. Double-FISH confirmed that, as expected, in Synbranchus the histone sites are dispersed throughout the genome, while the 18S rDNA sites are only present in one big cluster (Figure 3a-d). Similarly, the Rex1 and Rex3 TEs are arranged in small clusters that are also dispersed throughout the genome (Figure 4a-f). However, in the individuals belonging to karyomorph A collected at Bataguassu, only the Rex3 elements demonstrated significant accumulation in chromosome pair 3 (Figure 4d). Discussion Histone genes constitute a complex multigene family and may show variations in copy number and organization within the genome (Kedes, 1979). Thus, some species may present up to a few thousand histone gene copies, usually organized into tandemly repeated copies, while in others these sequences may be dispersed throughout the genome in small groups of copies (reviewed in Rooney et al., 2002).
In fish species, data about chromosomal location of histone sequences are restricted to eight species mapped for H1 genes (Pendás et al., 1994; Hashimoto et al., 2011, 2013; Lima-Filho et al., 2012) and only five species with mapped H3 sites (Pansonato-Alves et al., 2013a, 2013b; Silva et al., 2013), all of them presenting histone sequences as conspicuous blocks in chromosomes. Despite the restricted sampling, these studies revealed some particular characteristics, such as differential dispersion of sites and association with 5S or 18S rDNA (Hashimoto et al., 2011, 2013; Lima-Filho et al., 2012; Pansonato-Alves et al., 2013a, 2013b). More recent studies also showed that H1, H3 and H4 histone genes are clustered at the same site in fishes of genus Astyanax (Pansonato-Alves et al., 2013b; Silva et al., 2013). Our present results show that H3 and H4 histone sequences appear to be linked to one another and that in Synbranchus their dispersion occurs in small clusters throughout the genome. Moreover, in some acrocentric chromosome pairs there may be a small accumulation of these sites, which may be considered the major histone clusters, characterizing a distinct mode of organization of these sequences in fish chromosomes.
To date, the mapped sites of histone sequences in fishes of distinct orders such as Characiformes (Hashimoto et al., 2011, Pansonato-Alves et al., 2013a), Siluriformes (Hashimoto et al., 2013; Pansonato-Alves et al., 2013a) and Perciformes (Lima-Filho et al., 2012) are shown as large chromosomal blocks. The distinct organization and distribution of the H3 and H4 histone sites in S. marmoratus lead to the conclusion that, in this group, these sites are organised in small and abundant repetitions throughout the genome. This extensive distribution may be attributed to one of the following factors: (i) extensive occurrence of orphon genes derived from in-tandem repetitive families in eukaryotes, as already demonstrated for histone and ribosomal genes (Childs et al., 1981; Eirín-López et al., 2004) and likely to be related to a birth-and-death evolutionary mechanism (Eirín-López et al., 2004); or (ii) associations between histone sequences and transposable elements (TEs) due to the similarity of their distribution patterns mapped in fishes (Ferreira et al., 2011a, 2011b).
Just as for histone sites, the physical mapping of TEs in representatives of the Neotropical ichthyofauna is restricted to a small number of species and to the non-LTR retrotransposons Rex1, Rex3 and Rex6 (Gross et al., 2009; Cioffi et al., 2010; Valente et al., 2011; Ferreira et al., 2011a,b; Pansonato-Alves et al., 2013a). The overlap of signals generated by FISH among TEs and other repetitive sequences raises questions about their role in the dispersion of repetitive DNA sequences (Mandrioli et al., 2001; Mandrioli and Manicardi, 2001; Cioffi et al., 2010). In S. marmoratus, the Rex1 and Rex3 elements are found in small clusters dispersed over all chromosomes. Notably, an accentuated accumulation of repetitions in the centromeric region of pair 3 was found in samples from Bataguassu (karyomorph A); however, this seems to be only a local amplification because the individuals of karyomorph B, which is believed to be recently derived from karyomorph A (unpublished data), did not present such blocks in this chromosome pair.
It is further worth noting that these elements may present variable modes of chromosomal distribution in different species, but tend to be distributed in a similar manner in close groups. The Rex1, Rex3 and Rex6 elements, for example, are primarily compartmentalized in the pericentromeric heterochromatic regions in Cichlid fishes (Teixeira et al., 2009; Valente et al., 2011) and dispersed throughout the genome in Loricariidae, Bathydraconidae and Artedidraconidae species (Ozouf-Costaz et al., 2004; Ferreira et al., 2011a; Pansonato-Alves et al., 2013a). However, in Characiform species, Rex3 elements may also be compartmentalized (Cioffi et al., 2010; Pansonato-Alves et al., 2013b; Silva et al., 2013), indicating that those elements are highly dynamic and their type of genomic organization does not reflect phylogenetic relationships among species.
Although S. marmoratus presents a remarkable variation in karyotype macrostructure, the physical mapping of histone sequences and transposable elements revealed that these sequences are all dispersed in different karyomorphs, and this seems to be a conserved feature. Thus, we stress the importance of further studies regarding the physical mapping of H1, H3 and H4 histone genes and other repetitive DNAs, which may be useful in determining the organization of these genes in eukaryote genomes. Similarly, the mapping of transposable elements can bring new perspectives on the genomic organization, dispersion of genes and speciation driven by those sequences.
Acknowledgments
This study was supported by grants from the Brazilian agencies Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP). Ricardo Utsunomia had a scholarship from FAPESP (2011/01370-0)
References
Charlesworth B, Snlegowski P and StephanW(1994) The evolutionary dynamics of repetitive DNA in eukaryotes. Nature 371:215-220.
Childs G, Maxson R, Cohn RH and Kedes L (1981) Orphons: Dispersed genetic elements derived from tandem repetitive genes of eukaryotes. Cell 23:651-663.
Cioffi MB, Martins C and Bertollo LAC (2010) Chromosome spreading of associated transposable elements and ribosomal DNA in the fish Erythrinus erythrinus. Implications for genome change and karyoevolution in fish. BMC Evol Biol 10:271-280.
Colgan DJ, McLauchlan A and Wilson GDF (1998) Histone H3 and U2 snRNA DNA sequences and arthropod molecular evolution. Aust J Zool 46:419-437.
Eirin-Lopez JM, González-Tizón AM, Martínez A and Méndez J (2004) Birth-and-death evolution with strong purifying selection in the Histone H1 multigene family and the origin of orphon H1 genes. Mol Biol Evol 21:1992-2003.
Ferreira DC, Oliveira C and Foresti F (2011a) Elements Rex1 and Rex3 in three fish species in the subfamily Hypoptopomatinae (Teleostei, Siluriformes, Loricariidae). Cytogenet Genome Res 132:64-70.
Ferreira DC, Oliveira C and Foresti F (2011b) A new dispersed element in the genome of the catfish Hisonotus leucofrenatus (Teleostei, Siluriformes, Hypoptopomatinae). Mob Genet Elements 1:103-106.
Foresti F, Almeida-Toledo LF and Toledo-Filho SA (1981) Polymorphic nature of nucleolus organizer regions on fishes. Cytogenet Cell Gen 31:137-144.
Foresti F, Oliveira C and Tien OS (1992) Cytogenetic studies of the genus Synbranchus (Pisces, Synbranchiformes, Synbranchidae). Naturalia 17:129-138.
Gross MC, Schneider CH, Valente GT, Porto JIR, Martins C and Feldberg E (2009) Comparative cytogenetic analysis of the genus Symphysodon (Discus fishes, Cichlidae): Chromosomal characteristics of retrotransposons and minor ribosomal DNA. Cytogenet Genome Res 127:43-53.
Gursel I, Gursel M, Yamada H, Ishii KJ, Takeshita F and Klinman DM (2003) Repetitive elements in mammalian telomeres suppress bacterial DNA-induced immune activation. J Immunol 171:1393-1400.
Haff T, Schmid M, Steinlein C, Galetti Jr PM, Willard H (1993) Organization and molecular cytogenetics of a satellite DNA family from Hoplias malabaricus (Pisces, Erythrinidae). Chromosome Res 1:77-86.
Hashimoto DT, Ferguson-Smith MA, Rens W, Foresti F and Porto-Foresti F (2011) Chromosome mapping of H1 histone and 5S rRNA genes clusters in three species of Astyanax (Teleostei, Characiformes). Cytogen Genome Res 134:64- 71.
Hashimoto DT, Ferguson-Smith MA, Rens W, Prado FD, Foresti F and Porto-Foresti F (2013) Cytogenetic mapping of H1 histone and ribosomal RNA genes in hybrids between catfish species Pseudoplatystoma corruscans and Pseudoplatystoma reticulatum. Cytogen Genome Res 139:102-106.
Kedes LH (1979) Histone genes and histone messengers. Annu Rev Biochem 48:837-870.
Lima-Filho PA, Cioffi MB, Bertollo LAC and Molina WF (2012) Chromosomal and morphological divergences in Atlantic populations of the frillfin Bathygobius soporator (Gobiidae, Perciformes). J Exp Mar Biol Ecol 434:63-70.
Mandrioli M, Manicardi GC, Machella N and Caputo V (2001) Molecular and cytogenetic analysis of the goby Gobius niger (Teleostei, Gobiidae). Genetica 110:73-78.
MandrioliMand Manicardi GC (2001) Cytogenetics and molecular analysis of the pufferfish Tetraodon fluviatilis (Osteichthyes). Genetica 111:433-438.
Martins C and Galetti Jr PM (1999) Chromosomal localization of 5S rDNA genes in Leporinus fish (Anostomidae, Characiformes). Chromosome Res 7:363-367.
Melilo IFM, Foresti F and Oliveira C (1996) Additional cytogenetic studies on local populations of Synbranchus marmoratus (Pisces, Synbranchiformes, Synbranchidae). Naturalia 21:201-208.
Nagoda N, Fukuda A, Nakashima Y and Matsuo Y (2005) Molecular characterization and evolution of the repeating units of histone genes in Drosophila americana: Coexistence of quartet and quintet units in a genome. Insect Mol Biol 14:713-717.
Ozouf-Costaz C, Brandt J, Korting C, Pisano E, Bonillo C, Coutanceau JP and Volff JN (2004) Genome dynamics and chromosomal localization of the non-LTR retrotransposons Rex1 and Rex3 in Antartic fish. Antartic Science 16:51-57.
Pansonato-Alves JC, Serrano EA, Utsunomia R, Scacchetti PC, Oliveira C and Foresti F (2013a) Mapping five repetitive DNA classes in sympatric species of Hypostomus (Teleostei, Siluriformes, Loricariidae): Analysis of chromosomal variability. Rev Fish Biol Fisher 23:477-489
Pansonato-Alves JC, Hilsdorf AWS, Utsunomia R, Silva DMZA, Oliveira C and Foresti F (2013b) Chromosomal mapping of repetitive DNA and cytochrome C oxidase I sequence analysis reveal differentiation among sympatric samples of Astyanax fasciatus (Characiformes, Characidae). Cytogenet Genome Res 141:133-142.
Pendás AM, Morán P and García-Vázquez E (1994) Organization and chromosomal location of the major histone cluster in brown trout, Atlantic salmon and rainbow trout. Chromosoma 103:147-152.
Pineau P, Henry M, Suspène R, Marchio A, Dettai A, Debruyne R, Petit T, Lécu A, Moisson P, Dejean A, et al. (2005) A universal primer set for PCR amplification of nuclear histone H4 genes from all animal species. Mol Biol Evol 22:582- 588.
Pinkel D, Straume T and Gray JW (1986) Cytogenetic analysis using quantitative, high-sensitivity, fluorescence hybridization. Proc Natl Acad Sci USA 83:2934-2938.
Sanchez S and Fenocchio AS (1996) Karyotypic analysis in three populations of the South-American eel like fish Synbranchus marmoratus. Caryologia 49:65-71.
Silva DMZA, Pansonato-Alves JC, Utsunomia R, Daniel SN, Hashimoto DT, Oliveira C, Porto-Foresti F and Foresti F (2013) Chromosomal organization of repetitive DNA sequences in Astyanax bockmanni (Teleostei, Characiformes): Dispersive location, association and co-localization in the genome. Genetica 141:329-336.
Sumner AT (2003) Chromosomes: Organization and Function. Blackwell Publishing Company, London, 287 pp.
Teixeira WG, Ferreira IA, Cabral-de-Melo DC, Mazzuchelli J, Valente GT, Pinhal D, Poletto AB, Venere PC and Martins C (2009) Organization of repeated DNA elements in the genome of the cichlid fish Cichla kelberi and its contributions to the knowledge of fish genomes. Cytogenet Genome Res 125:224-234.
Torres RA, Roper JJ, Foresti F and Oliveira C (2005) Surprising genomic diversity in the Neotropical Fish Synbranchus marmoratus (Teleostei, Synbranchidae): How many species? Neotrop Ichthyol 3:277-284.
Valente GT, Mazzuchelli J, Ferreira IA, Poletto AB, Fantinatti BEA and Martins C (2011) Cytogenetic Mapping of the retroelements Rex1, Rex3 and Rex6 among cichlid fish: New insights on the chromosomal distribution of transposable elements. Cytogenet Genome Res 133:34-42.
Vicari MR, Nogaroto V, Noleto RB, Cestari MM, Cioffi MB, Almeida MC, Moreira-Filho O and Artoni RF (2010) Satellite DNA and chromosomes in Neotropical fishes: Methods, applications and perspectives. J Fish Biol 76:1094-1116.
Volff JN, Körting C, Sweeney K and Schartl M (1999) The non-LTR retrotransposon Rex3 from the fish Xiphophorus is widespread among teleosts. Mol Biol Evol 16:1427-1438. Volff JN, Körting C and SchartlM(2000) Multiple lineages of the non-LTR retrotransposon Rex1 with varying success in invading fish genomes. Mol Biol Evol 17:1673-1684.
Wagner RP, Maguire MP and Stallings RL (1993) Chromosomes: A synthesis. Wiley-Liss Inc., New York pp. 523.
White TJ, Bruns T, Lee S and Taylor J (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ and White TJ (eds) PCR Protocols: A Guide to Methods and Applications. Academic Press Inc., New York, pp 315-322.
Received: August 1, 2013;
Accepted: October 3, 2013.
Associate Editor: Igor Schneider
All the content of the journal, except where otherwise noted, is licensed under a Creative Commons License CC BY-NC.
- Charlesworth B, Snlegowski P and StephanW(1994) The evolutionary dynamics of repetitive DNA in eukaryotes. Nature 371:215-220.
- Childs G, Maxson R, Cohn RH and Kedes L (1981) Orphons: Dispersed genetic elements derived from tandem repetitive genes of eukaryotes. Cell 23:651-663.
- Cioffi MB, Martins C and Bertollo LAC (2010) Chromosome spreading of associated transposable elements and ribosomal DNA in the fish Erythrinus erythrinus. Implications for genome change and karyoevolution in fish. BMC Evol Biol 10:271-280.
- Colgan DJ, McLauchlan A and Wilson GDF (1998) Histone H3 and U2 snRNA DNA sequences and arthropod molecular evolution. Aust J Zool 46:419-437.
- Eirin-Lopez JM, González-Tizón AM, Martínez A and Méndez J (2004) Birth-and-death evolution with strong purifying selection in the Histone H1 multigene family and the origin of orphon H1 genes. Mol Biol Evol 21:1992-2003.
- Ferreira DC, Oliveira C and Foresti F (2011a) Elements Rex1 and Rex3 in three fish species in the subfamily Hypoptopomatinae (Teleostei, Siluriformes, Loricariidae). Cytogenet Genome Res 132:64-70.
- Ferreira DC, Oliveira C and Foresti F (2011b) A new dispersed element in the genome of the catfish Hisonotus leucofrenatus (Teleostei, Siluriformes, Hypoptopomatinae). Mob Genet Elements 1:103-106.
- Foresti F, Almeida-Toledo LF and Toledo-Filho SA (1981) Polymorphic nature of nucleolus organizer regions on fishes. Cytogenet Cell Gen 31:137-144.
- Foresti F, Oliveira C and Tien OS (1992) Cytogenetic studies of the genus Synbranchus (Pisces, Synbranchiformes, Synbranchidae). Naturalia 17:129-138.
- Gross MC, Schneider CH, Valente GT, Porto JIR, Martins C and Feldberg E (2009) Comparative cytogenetic analysis of the genus Symphysodon (Discus fishes, Cichlidae): Chromosomal characteristics of retrotransposons and minor ribosomal DNA. Cytogenet Genome Res 127:43-53.
- Gursel I, Gursel M, Yamada H, Ishii KJ, Takeshita F and Klinman DM (2003) Repetitive elements in mammalian telomeres suppress bacterial DNA-induced immune activation. J Immunol 171:1393-1400.
- Haff T, Schmid M, Steinlein C, Galetti Jr PM, Willard H (1993) Organization and molecular cytogenetics of a satellite DNA family from Hoplias malabaricus (Pisces, Erythrinidae). Chromosome Res 1:77-86.
- Hashimoto DT, Ferguson-Smith MA, Rens W, Foresti F and Porto-Foresti F (2011) Chromosome mapping of H1 histone and 5S rRNA genes clusters in three species of Astyanax (Teleostei, Characiformes). Cytogen Genome Res 134:64- 71.
- Hashimoto DT, Ferguson-Smith MA, Rens W, Prado FD, Foresti F and Porto-Foresti F (2013) Cytogenetic mapping of H1 histone and ribosomal RNA genes in hybrids between catfish species Pseudoplatystoma corruscans and Pseudoplatystoma reticulatum. Cytogen Genome Res 139:102-106.
- Kedes LH (1979) Histone genes and histone messengers. Annu Rev Biochem 48:837-870.
- Lima-Filho PA, Cioffi MB, Bertollo LAC and Molina WF (2012) Chromosomal and morphological divergences in Atlantic populations of the frillfin Bathygobius soporator (Gobiidae, Perciformes). J Exp Mar Biol Ecol 434:63-70.
- Mandrioli M, Manicardi GC, Machella N and Caputo V (2001) Molecular and cytogenetic analysis of the goby Gobius niger (Teleostei, Gobiidae). Genetica 110:73-78.
- MandrioliMand Manicardi GC (2001) Cytogenetics and molecular analysis of the pufferfish Tetraodon fluviatilis (Osteichthyes). Genetica 111:433-438.
- Martins C and Galetti Jr PM (1999) Chromosomal localization of 5S rDNA genes in Leporinus fish (Anostomidae, Characiformes). Chromosome Res 7:363-367.
- Melilo IFM, Foresti F and Oliveira C (1996) Additional cytogenetic studies on local populations of Synbranchus marmoratus (Pisces, Synbranchiformes, Synbranchidae). Naturalia 21:201-208.
- Nagoda N, Fukuda A, Nakashima Y and Matsuo Y (2005) Molecular characterization and evolution of the repeating units of histone genes in Drosophila americana: Coexistence of quartet and quintet units in a genome. Insect Mol Biol 14:713-717.
- Ozouf-Costaz C, Brandt J, Korting C, Pisano E, Bonillo C, Coutanceau JP and Volff JN (2004) Genome dynamics and chromosomal localization of the non-LTR retrotransposons Rex1 and Rex3 in Antartic fish. Antartic Science 16:51-57.
- Pansonato-Alves JC, Serrano EA, Utsunomia R, Scacchetti PC, Oliveira C and Foresti F (2013a) Mapping five repetitive DNA classes in sympatric species of Hypostomus (Teleostei, Siluriformes, Loricariidae): Analysis of chromosomal variability. Rev Fish Biol Fisher 23:477-489
- Pansonato-Alves JC, Hilsdorf AWS, Utsunomia R, Silva DMZA, Oliveira C and Foresti F (2013b) Chromosomal mapping of repetitive DNA and cytochrome C oxidase I sequence analysis reveal differentiation among sympatric samples of Astyanax fasciatus (Characiformes, Characidae). Cytogenet Genome Res 141:133-142.
- Pendás AM, Morán P and García-Vázquez E (1994) Organization and chromosomal location of the major histone cluster in brown trout, Atlantic salmon and rainbow trout. Chromosoma 103:147-152.
- Pineau P, Henry M, Suspène R, Marchio A, Dettai A, Debruyne R, Petit T, Lécu A, Moisson P, Dejean A, et al. (2005) A universal primer set for PCR amplification of nuclear histone H4 genes from all animal species. Mol Biol Evol 22:582- 588.
- Pinkel D, Straume T and Gray JW (1986) Cytogenetic analysis using quantitative, high-sensitivity, fluorescence hybridization. Proc Natl Acad Sci USA 83:2934-2938.
- Sanchez S and Fenocchio AS (1996) Karyotypic analysis in three populations of the South-American eel like fish Synbranchus marmoratus. Caryologia 49:65-71.
- Silva DMZA, Pansonato-Alves JC, Utsunomia R, Daniel SN, Hashimoto DT, Oliveira C, Porto-Foresti F and Foresti F (2013) Chromosomal organization of repetitive DNA sequences in Astyanax bockmanni (Teleostei, Characiformes): Dispersive location, association and co-localization in the genome. Genetica 141:329-336.
- Sumner AT (2003) Chromosomes: Organization and Function. Blackwell Publishing Company, London, 287 pp.
- Teixeira WG, Ferreira IA, Cabral-de-Melo DC, Mazzuchelli J, Valente GT, Pinhal D, Poletto AB, Venere PC and Martins C (2009) Organization of repeated DNA elements in the genome of the cichlid fish Cichla kelberi and its contributions to the knowledge of fish genomes. Cytogenet Genome Res 125:224-234.
- Torres RA, Roper JJ, Foresti F and Oliveira C (2005) Surprising genomic diversity in the Neotropical Fish Synbranchus marmoratus (Teleostei, Synbranchidae): How many species? Neotrop Ichthyol 3:277-284.
- Valente GT, Mazzuchelli J, Ferreira IA, Poletto AB, Fantinatti BEA and Martins C (2011) Cytogenetic Mapping of the retroelements Rex1, Rex3 and Rex6 among cichlid fish: New insights on the chromosomal distribution of transposable elements. Cytogenet Genome Res 133:34-42.
- Vicari MR, Nogaroto V, Noleto RB, Cestari MM, Cioffi MB, Almeida MC, Moreira-Filho O and Artoni RF (2010) Satellite DNA and chromosomes in Neotropical fishes: Methods, applications and perspectives. J Fish Biol 76:1094-1116.
- Volff JN, Körting C, Sweeney K and Schartl M (1999) The non-LTR retrotransposon Rex3 from the fish Xiphophorus is widespread among teleosts. Mol Biol Evol 16:1427-1438.
- Volff JN, Körting C and SchartlM(2000) Multiple lineages of the non-LTR retrotransposon Rex1 with varying success in invading fish genomes. Mol Biol Evol 17:1673-1684.
- Wagner RP, Maguire MP and Stallings RL (1993) Chromosomes: A synthesis. Wiley-Liss Inc., New York pp. 523.
- White TJ, Bruns T, Lee S and Taylor J (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ and White TJ (eds) PCR Protocols: A Guide to Methods and Applications. Academic Press Inc., New York, pp 315-322.
Send correspondence to
Publication Dates
-
Publication in this collection
24 Mar 2014 -
Date of issue
2014
History
-
Received
01 Aug 2013 -
Accepted
03 Oct 2013