Abstract
Objective:
Functional neuroimaging techniques represent fundamental tools in the context of translational research integrating neurobiology, psychopathology, neuropsychology, and therapeutics. In addition, cognitive-behavioral therapy (CBT) has proven its efficacy in the treatment of anxiety disorders and may be useful in phobias. The literature has shown that feelings and behaviors are mediated by specific brain circuits, and changes in patterns of interaction should be associated with cerebral alterations. Based on these concepts, a systematic review was conducted aiming to evaluate the impact of CBT on phobic disorders measured by functional neuroimaging techniques.
Methods:
A systematic review of the literature was conducted including studies published between January 1980 and April 2012. Studies written in English, Spanish or Portuguese evaluating changes in the pattern of functional neuroimaging before and after CBT in patients with phobic disorders were included.
Results:
The initial search strategy retrieved 45 studies. Six of these studies met all inclusion criteria. Significant deactivations in the amygdala, insula, thalamus and hippocampus, as well as activation of the medial orbitofrontal cortex, were observed after CBT in phobic patients when compared with controls.
Conclusion:
In spite of their technical limitations, neuroimaging techniques provide neurobiological support for the efficacy of CBT in the treatment of phobic disorders. Further studies are needed to confirm this conclusion.
Phobia; cognitive therapy; functional magnetic resonance imaging; positron-emission tomography
Introduction
Functional neuroimaging techniques, such as magnetic resonance imaging (fMRI), single-photon emission-computed tomography (SPECT), spectroscopy, diffusion tensor imaging (DTI), and positron-emission tomography (PET), represent fundamental tools for translational research. These techniques allow the integration of neurobiology, psychopathology, neuropsychology, and therapeutics, contributing significantly to the understanding of the neurobiology of emotional regulation in healthy individuals and the neurocircuitry involved in the pathophysiology of mental disorders.11. Martínez-González AE, Piqueras-Rodríguez JA. [The effectiveness of cognitive-behavioural therapy in affective and anxiety disorders using functional neuroimaging]. Rev Neurol. 2010;50:167-78. Phobic disorders are characterized by marked and persistent fear prompted by a specific object or situation, and accompanied by the compelling effort to avoid such object or situation.22. Hamm AO. Specific phobias. Psychiatr Clin North Am. 2009;32:577-91. Social phobia, or social anxiety disorder (SAD), has a lifetime prevalence rate of over 10% and is characterized by a relevant fear of social or performance situations involving possible scrutiny by others.33. Kessler RC, Berglund P, Demler O, Jin R, Merikangas KR, Walters EE. Lifetime prevalence and age-of-onset distributions of DSM-IV disorders in the National Comorbidity Survey Replication. Arch Gen Psychiatry. 2005;62:593-602.,44. American Psychiatric Association. Diagnostic and statistical manual of mental disorders. 4th ed. Washington: American Psychiatric Association; 2000. Specific phobias (SP) are also frequent, with a lifetime prevalence of 7-11%, and are marked by excessive and unreasonable fear of specific objects or situations such as animals (e.g., spiders, snakes, dogs, and mice), flying, driving, enclosed places, heights, and blood/injury. Fear and avoidance cause significant distress and/or impairment in occupational, academic, or social functioning.44. American Psychiatric Association. Diagnostic and statistical manual of mental disorders. 4th ed. Washington: American Psychiatric Association; 2000.,55. Shin LM, Liberzon I. The neurocircuitry of fear, stress, and anxiety disorders. Neuropsychopharmacology. 2010;35:169-91. SP and SAD appear to alter different neural circuits,66. Linares IM, Trzesniak C, Chagas MH, Hallak JE, Nardi AE, Crippa JA. Neuroimaging in specific phobia disorder: a systematic review of the literature. Rev Bras Psiquiatr. 2012;34:101-11. and data from neuroimaging studies and clinical trials suggest that pharmacotherapy and cognitive-behavioral therapy (CBT) have positive effects on SAD. In SP, CBT may be one of the few useful interventions.77. Butler AC, Chapman JE, Forman EM, Beck AT. The empirical status of cognitive-behavioral therapy: a review of meta-analyses. Clin Psychol Rev. 2006;26:17-31.,88. Kumari V. Do psychotherapies produce neurobiological effects? Acta Neuropsychiatr. 2006;18:61-70. A combination of Beck's theoretical model and behavioral techniques, such as exposition and systematic desensitization, synthetizes CBT used in phobic disorders.11. Martínez-González AE, Piqueras-Rodríguez JA. [The effectiveness of cognitive-behavioural therapy in affective and anxiety disorders using functional neuroimaging]. Rev Neurol. 2010;50:167-78.
Studies involving fMRI in SP patients have demonstrated increased activation of the amygdala, anterior cingulate cortex (ACC), insular cortex, thalamus, and visual areas in patients exposed to or anticipating the presentation of phobic stimulation.9. Straube T, Glauer M, Dilger S, Mentzel HJ, Miltner WH. Effects of cognitive-behavioral therapy on brain activation in specific phobia. Neuroimage. 2006;29:125-35. 10. Etkin A, Wager TD. Functional neuroimaging of anxiety: a meta-analysis of emotional processing in PTSD, social anxiety disorder, and specific phobia. Am J Psychiatry. 2007;164:1476-88. 9-1111. Beauregard M. Mind does really matter: evidence from neuroimaging studies of emotional self-regulation, psychotherapy, and placebo effect. Prog Neurobiol. 2007;81:218-36. Such regions are the same as those involved in the pathophysiology of SAD observed in other functional neuroimaging studies.99. Straube T, Glauer M, Dilger S, Mentzel HJ, Miltner WH. Effects of cognitive-behavioral therapy on brain activation in specific phobia. Neuroimage. 2006;29:125-35. Moreover, a meta-analysis observed hyperactivation of the amygdala and insula in patients with SAD and SPs.1010. Etkin A, Wager TD. Functional neuroimaging of anxiety: a meta-analysis of emotional processing in PTSD, social anxiety disorder, and specific phobia. Am J Psychiatry. 2007;164:1476-88. Considering the correspondence between changes in brain and behavior, we aimed to systematically review the literature regarding clinical trials using functional neuroimaging techniques to assess the response to CBT in phobic disorders.
Methods
Three investigators (AGA, AB, CT) independently searched the databases of PubMed and ISI Web of Knowledge. Their search included clinical trials evaluating the changes in the pattern of functional neuroimaging methods before and after CBT for phobic disorders, published from January 1980 to April 2012. The keywords used in the search were functional magnetic resonance imaging or positron-emission tomography or single-photon emission-computed tomography or spectroscopy or diffusion tensor imaging and phobic disorders or phobia and cognitive therapy or behavior therapy. An initial searching protocol was prepared considering the following inclusion criteria: 1) studies written in English, Spanish or Portuguese; 2) clinical trials using any cognitive-behavioral technique for phobias; 3) diagnosis of phobia in patients over 18 years of age, and exclusion of any other psychiatric diagnosis based on a structured clinical diagnostic interview or according to the DSM-IV criteria44. American Psychiatric Association. Diagnostic and statistical manual of mental disorders. 4th ed. Washington: American Psychiatric Association; 2000.; 4) a functional neuroimaging method used before and after the psychotherapeutic intervention, with a symptom provocation paradigm; 5) presence of a control group.
Results
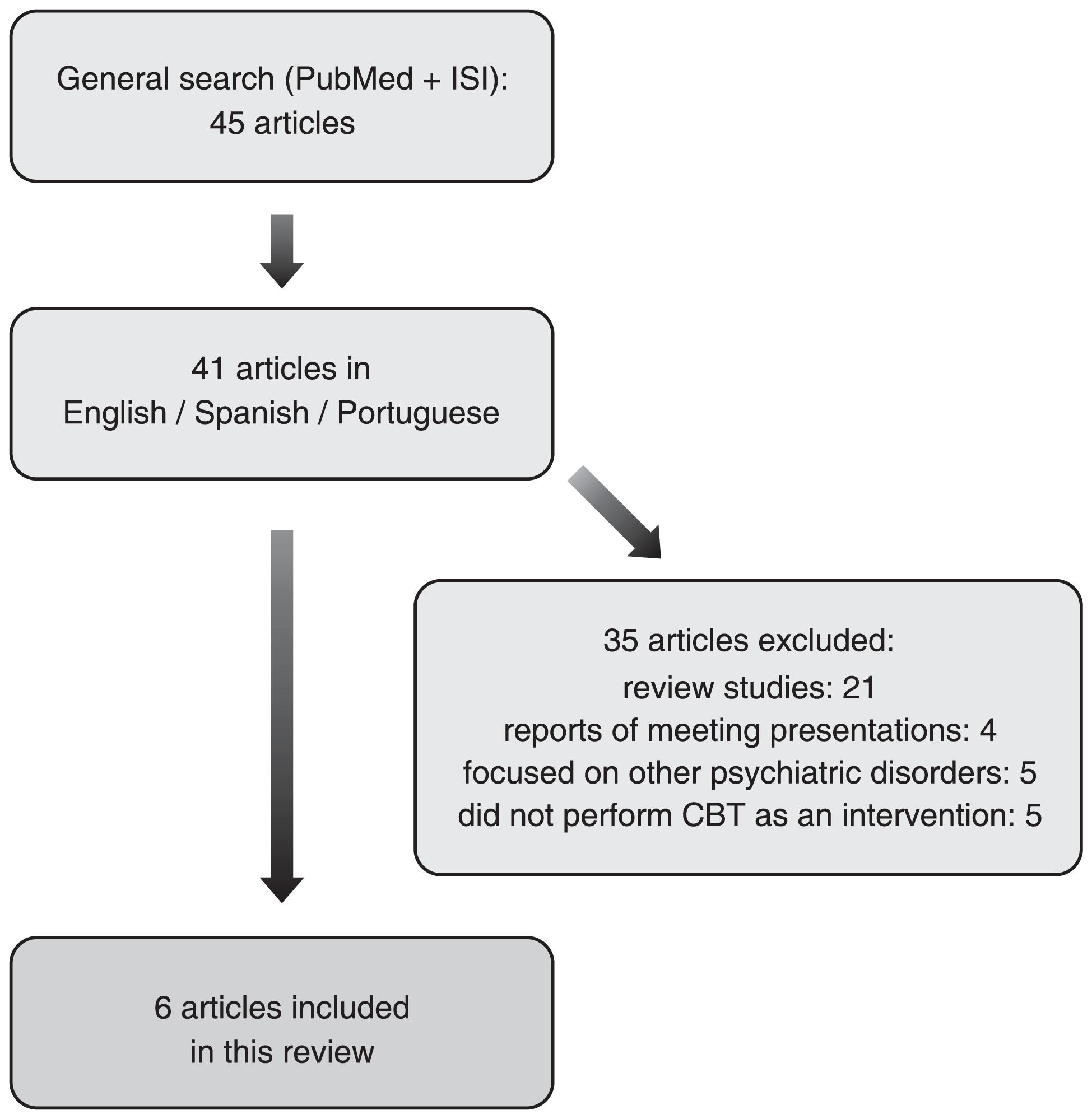
The initial search strategy yielded 45 studies. Forty-one of them (91.1%) were published in English, Spanish or Portuguese, which were screened for relevance related to the topic. Thirty-five studies (85.4%) were excluded for the following reasons: 21 were review studies; four consisted of reports on meeting presentations; five focused on other disorders (two on generalized anxiety disorder, one on panic disorder, one on posttraumatic stress disorder, and one on irritable bowel syndrome); finally, five studies did not perform CBT as an intervention. The systematic literature review search process and final results are shown in Figure 1. Six studies met all inclusion criteria. All of those studies were prospective and had a control group of non-phobic patients. Phobic patients were allocated in two groups: those submitted to CBT and a waiting list group. Patients and controls were submitted to a functional neuroimaging method before treatment, and all phobic patients (including those in the waiting lists) were submitted to a functional neuroimaging method after CBT. Comparisons were made between phobic patients and controls before treatment, and between patients who were submitted to CBT vs. waiting list patients. Blindness of imaging analysis was not declared in none of the studies. Overall, phobic patients presented higher activations of the amygdala, insula, thalamus and hippocampus, as well as lower activation of the medial orbitofrontal cortex (OFC) compared with a healthy control group. In addition, significant deactivations of the amygdala, insula, thalamus and hippocampus, as well as activation of the medial OFC, were observed in phobic patients after CBT when compared with controls (waiting list group). Those changes in brain activity were correlated with improvements of phobic symptoms. Table 1 summarizes the main characteristics and results of these studies included in the review.
Discussion
Previous studies have suggested that the mental functions and processes involved in CBT exert a significant influence on brain functioning and plasticity. Furthermore, functional neuroimaging studies have contributed to the understanding of the pathophysiological mechanisms underlying phobic disorders, thus providing an objective parameter to evaluate the impact of psychotherapeutic approaches and other non-pharmacological interventions.11. Martínez-González AE, Piqueras-Rodríguez JA. [The effectiveness of cognitive-behavioural therapy in affective and anxiety disorders using functional neuroimaging]. Rev Neurol. 2010;50:167-78.,11-1311. Beauregard M. Mind does really matter: evidence from neuroimaging studies of emotional self-regulation, psychotherapy, and placebo effect. Prog Neurobiol. 2007;81:218-36. Over the past decades, CBT has demonstrated good efficacy in treating most anxiety disorders, including phobias.77. Butler AC, Chapman JE, Forman EM, Beck AT. The empirical status of cognitive-behavioral therapy: a review of meta-analyses. Clin Psychol Rev. 2006;26:17-31.,88. Kumari V. Do psychotherapies produce neurobiological effects? Acta Neuropsychiatr. 2006;18:61-70. Individuals with phobias exposed to phobic stimuli showed increased amygdala, ACC, and insular cortex activation in functional neuroimaging studies when compared with healthy controls.55. Shin LM, Liberzon I. The neurocircuitry of fear, stress, and anxiety disorders. Neuropsychopharmacology. 2010;35:169-91. Reduced amygdala and insula and increased medial OFC activations after a successful CBT treatment are in agreement with expected cognitive and emotional changes. Amygdala activity modulates vigilance, which, in turn, facilitates an increased attentional interest in environmental factors in order to acquire information to resolve the ambiguity.1414. Goossens L, Sunaert S, Peeters R, Griez EJ, Schruers KR. Amygdala hyperfunction in phobic fear normalizes after exposure. Biol Psychiatry. 2007;62:1119-25.,1616. Schienle A, Schafer A, Stark R, Vaitl D. Long-term effects of cognitive behavior therapy on brain activation in spider phobia. Psychiatry Res. 2009;172:99-102.,1919. Fredrikson M, Furmark T. Amygdaloid regional cerebral blood flow and subjective fear during symptom provocation in anxiety disorders. Ann N Y Acad Sci. 2003;985:341-7.,2020. Ahs F, Pissiota A, Michelgard A, Frans O, Furmark T, Appel L, et al. Disentangling the web of fear: amygdala reactivity and functional connectivity in spider and snake phobia. Psychiatry Res. 2009;172:103-8. As the amygdala has been related to the pathophysiology of phobias, a reduction in the symptoms of autonomic arousal could be represented by reduced amygdala activity measured by functional neuroimaging methods.1414. Goossens L, Sunaert S, Peeters R, Griez EJ, Schruers KR. Amygdala hyperfunction in phobic fear normalizes after exposure. Biol Psychiatry. 2007;62:1119-25.,1919. Fredrikson M, Furmark T. Amygdaloid regional cerebral blood flow and subjective fear during symptom provocation in anxiety disorders. Ann N Y Acad Sci. 2003;985:341-7.,2020. Ahs F, Pissiota A, Michelgard A, Frans O, Furmark T, Appel L, et al. Disentangling the web of fear: amygdala reactivity and functional connectivity in spider and snake phobia. Psychiatry Res. 2009;172:103-8. Moreover, the insular cortex has been shown to be generally involved in the recognition and experience of aversive states, such as disgust, fear, and pain, and it is possible that successful CBT reduces the need to decode unpleasant emotional states usually related to exposure to phobic stimuli.1414. Goossens L, Sunaert S, Peeters R, Griez EJ, Schruers KR. Amygdala hyperfunction in phobic fear normalizes after exposure. Biol Psychiatry. 2007;62:1119-25. In addition, since the medial prefrontal cortex (MPFC) is involved in processes associated with extinction of conditioned fear, an increased activity in these areas could result in remission of emotion-related neural responses.2121. Freitas-Ferrari MC, Hallak JE, Trzesniak C, Filho AS, Machado-de-Sousa JP, Chagas MH, et al. Neuroimaging in social anxiety disorder: a systematic review of the literature. Prog Neuropsychopharmacol Biol Psychiatry. 2010;34:565-80.
We found that all clinical trials using functional neuroimaging techniques to assess the CBT response in phobic disorders found significant deactivation of the amygdala, insula, thalamus, and hippocampus, as well as activation of the medial OFC, after CBT in phobic patients when compared with controls. As these regions have been associated with the pathophysiology of either SP or SAD, deactivations of such areas after CBT would be a possible marker of improvement.1414. Goossens L, Sunaert S, Peeters R, Griez EJ, Schruers KR. Amygdala hyperfunction in phobic fear normalizes after exposure. Biol Psychiatry. 2007;62:1119-25.,1919. Fredrikson M, Furmark T. Amygdaloid regional cerebral blood flow and subjective fear during symptom provocation in anxiety disorders. Ann N Y Acad Sci. 2003;985:341-7.,2020. Ahs F, Pissiota A, Michelgard A, Frans O, Furmark T, Appel L, et al. Disentangling the web of fear: amygdala reactivity and functional connectivity in spider and snake phobia. Psychiatry Res. 2009;172:103-8. Such findings, which are similar to those observed in functional neuroimaging studies of phobic patients treated with antidepressants,2222. Kilts CD, Kelsey JE, Knight B, Ely TD, Bowman FD, Gross RE, et al. The neural correlates of social anxiety disorder and response to pharmacotherapy. Neuropsychopharmacology. 2006;31:2243-53.,2323. Carey PD, Warwick J, Niehaus DJ, van der Linden G, van Heerden BB, Harvey BH, et al. Single photon emission computed tomography (SPECT) of anxiety disorders before and after treatment with citalopram. BMC Psychiatry. 2004;4:30. could support the efficacy of CBT in the treatment of phobias.66. Linares IM, Trzesniak C, Chagas MH, Hallak JE, Nardi AE, Crippa JA. Neuroimaging in specific phobia disorder: a systematic review of the literature. Rev Bras Psiquiatr. 2012;34:101-11.,) Moreover, recent studies have observed that functional brain imaging may detect biomarkers that substantially improve predictions for the success of cognitive-behavioral interventions, suggesting that such biomarkers could offer personalized approaches for optimally selecting among treatment options for a patient.2424. Doehrmann O, Ghosh SS, Polli FE, Reynolds GO, Horn F, Keshavan A, et al. Predicting treatment response in social anxiety disorder from functional magnetic resonance imaging. JAMA Psychiatry. 2013;70:87-97.
Finally, a number of important limitations need to be considered, such as the reduced number of studies found in the literature and their small sample size. Moreover, differences across the studies precluded a direct comparison between them. Such heterogeneities were related to the phobic disorders examined, neuroimaging techniques used, data analysis methods, number of subjects, nature of volunteer controls, condition of data acquisition (resting state, activation task), and timing of the second imaging study during treatment. Although anxiety disorders are more prevalent in women, the studies had a gender limitation because all simple phobia patients were women, thus preventing generalization of results. In addition, the studies included in the present review did not allow determining whether the brain changes measured after psychotherapy were the cause or the effect of symptom reduction.
Nevertheless, despite such technical limitations, current literature data provide a possible neurobiological support for the efficacy of CBT in phobic disorders measured by functional neuroimaging studies.12. Furmark T, Tillfors M, Marteinsdottir I, Fischer H, Pissiota A, Langström B, et al. Common changes in cerebral blood flow in patients with social phobia treated with citalopram or cognitive-behavioral therapy. Arch Gen Psychiatry. 2002;59:425-33. 13. Paquette V, Lévesque J, Mensour B, Leroux JM, Beaudoin G, Bourgouin P, et al. “Change the mind and you change the brain”: effects of cognitive-behavioral therapy on the neural correlates of spider phobia. Neuroimage. 2003;18:401-9. 14. Goossens L, Sunaert S, Peeters R, Griez EJ, Schruers KR. Amygdala hyperfunction in phobic fear normalizes after exposure. Biol Psychiatry. 2007;62:1119-25. 15. Schienle A, Schafer A, Hermann A, Rohrmann S, Vaitl D. Symptom provocation and reduction in patients suffering from spider phobia: an fMRI study on exposure therapy. Eur Arch Psychiatry Clin Neurosci. 2007;257:486-93. 16. Schienle A, Schafer A, Stark R, Vaitl D. Long-term effects of cognitive behavior therapy on brain activation in spider phobia. Psychiatry Res. 2009;172:99-102. 17. Roffman JL, Marci CD, Glick DM, Dougherty DD, Rauch SL. Neuroimaging and the functional neuroanatomy of psychotherapy. Psychol Med. 2005;35:1385-98. 18. Linden DE. Brain imaging and psychotherapy: methodological considerations and practical implications. Eur Arch Psychiatry Clin Neurosci. 2008;258:71-5. 19. Fredrikson M, Furmark T. Amygdaloid regional cerebral blood flow and subjective fear during symptom provocation in anxiety disorders. Ann N Y Acad Sci. 2003;985:341-7. 20. Ahs F, Pissiota A, Michelgard A, Frans O, Furmark T, Appel L, et al. Disentangling the web of fear: amygdala reactivity and functional connectivity in spider and snake phobia. Psychiatry Res. 2009;172:103-8. 12-2121. Freitas-Ferrari MC, Hallak JE, Trzesniak C, Filho AS, Machado-de-Sousa JP, Chagas MH, et al. Neuroimaging in social anxiety disorder: a systematic review of the literature. Prog Neuropsychopharmacol Biol Psychiatry. 2010;34:565-80. Further complementary and longitudinal studies with larger and homogeneous samples, comparing different functional neuroimaging techniques in the same sample, are needed to better understand the underlying pathogenesis of phobic disorders and the efficacy of CBT in this context.66. Linares IM, Trzesniak C, Chagas MH, Hallak JE, Nardi AE, Crippa JA. Neuroimaging in specific phobia disorder: a systematic review of the literature. Rev Bras Psiquiatr. 2012;34:101-11.
Acknowledgement
Amanda Galvao-de Almeida, Fabiana Nery-Fernandes, Ângela Miranda-Scippa, and Irismar Reis de Oliveira receive research and other grants from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq). Andrea Parolin Jackowski receives research grants from CNPq and Fundação de Amparo è Pesquisa do Estado de São Paulo (FAPESP).
References
-
1Martínez-González AE, Piqueras-Rodríguez JA. [The effectiveness of cognitive-behavioural therapy in affective and anxiety disorders using functional neuroimaging]. Rev Neurol. 2010;50:167-78.
-
2Hamm AO. Specific phobias. Psychiatr Clin North Am. 2009;32:577-91.
-
3Kessler RC, Berglund P, Demler O, Jin R, Merikangas KR, Walters EE. Lifetime prevalence and age-of-onset distributions of DSM-IV disorders in the National Comorbidity Survey Replication. Arch Gen Psychiatry. 2005;62:593-602.
-
4American Psychiatric Association. Diagnostic and statistical manual of mental disorders. 4th ed. Washington: American Psychiatric Association; 2000.
-
5Shin LM, Liberzon I. The neurocircuitry of fear, stress, and anxiety disorders. Neuropsychopharmacology. 2010;35:169-91.
-
6Linares IM, Trzesniak C, Chagas MH, Hallak JE, Nardi AE, Crippa JA. Neuroimaging in specific phobia disorder: a systematic review of the literature. Rev Bras Psiquiatr. 2012;34:101-11.
-
7Butler AC, Chapman JE, Forman EM, Beck AT. The empirical status of cognitive-behavioral therapy: a review of meta-analyses. Clin Psychol Rev. 2006;26:17-31.
-
8Kumari V. Do psychotherapies produce neurobiological effects? Acta Neuropsychiatr. 2006;18:61-70.
-
9Straube T, Glauer M, Dilger S, Mentzel HJ, Miltner WH. Effects of cognitive-behavioral therapy on brain activation in specific phobia. Neuroimage. 2006;29:125-35.
-
10Etkin A, Wager TD. Functional neuroimaging of anxiety: a meta-analysis of emotional processing in PTSD, social anxiety disorder, and specific phobia. Am J Psychiatry. 2007;164:1476-88.
-
11Beauregard M. Mind does really matter: evidence from neuroimaging studies of emotional self-regulation, psychotherapy, and placebo effect. Prog Neurobiol. 2007;81:218-36.
-
12Furmark T, Tillfors M, Marteinsdottir I, Fischer H, Pissiota A, Langström B, et al. Common changes in cerebral blood flow in patients with social phobia treated with citalopram or cognitive-behavioral therapy. Arch Gen Psychiatry. 2002;59:425-33.
-
13Paquette V, Lévesque J, Mensour B, Leroux JM, Beaudoin G, Bourgouin P, et al. “Change the mind and you change the brain”: effects of cognitive-behavioral therapy on the neural correlates of spider phobia. Neuroimage. 2003;18:401-9.
-
14Goossens L, Sunaert S, Peeters R, Griez EJ, Schruers KR. Amygdala hyperfunction in phobic fear normalizes after exposure. Biol Psychiatry. 2007;62:1119-25.
-
15Schienle A, Schafer A, Hermann A, Rohrmann S, Vaitl D. Symptom provocation and reduction in patients suffering from spider phobia: an fMRI study on exposure therapy. Eur Arch Psychiatry Clin Neurosci. 2007;257:486-93.
-
16Schienle A, Schafer A, Stark R, Vaitl D. Long-term effects of cognitive behavior therapy on brain activation in spider phobia. Psychiatry Res. 2009;172:99-102.
-
17Roffman JL, Marci CD, Glick DM, Dougherty DD, Rauch SL. Neuroimaging and the functional neuroanatomy of psychotherapy. Psychol Med. 2005;35:1385-98.
-
18Linden DE. Brain imaging and psychotherapy: methodological considerations and practical implications. Eur Arch Psychiatry Clin Neurosci. 2008;258:71-5.
-
19Fredrikson M, Furmark T. Amygdaloid regional cerebral blood flow and subjective fear during symptom provocation in anxiety disorders. Ann N Y Acad Sci. 2003;985:341-7.
-
20Ahs F, Pissiota A, Michelgard A, Frans O, Furmark T, Appel L, et al. Disentangling the web of fear: amygdala reactivity and functional connectivity in spider and snake phobia. Psychiatry Res. 2009;172:103-8.
-
21Freitas-Ferrari MC, Hallak JE, Trzesniak C, Filho AS, Machado-de-Sousa JP, Chagas MH, et al. Neuroimaging in social anxiety disorder: a systematic review of the literature. Prog Neuropsychopharmacol Biol Psychiatry. 2010;34:565-80.
-
22Kilts CD, Kelsey JE, Knight B, Ely TD, Bowman FD, Gross RE, et al. The neural correlates of social anxiety disorder and response to pharmacotherapy. Neuropsychopharmacology. 2006;31:2243-53.
-
23Carey PD, Warwick J, Niehaus DJ, van der Linden G, van Heerden BB, Harvey BH, et al. Single photon emission computed tomography (SPECT) of anxiety disorders before and after treatment with citalopram. BMC Psychiatry. 2004;4:30.
-
24Doehrmann O, Ghosh SS, Polli FE, Reynolds GO, Horn F, Keshavan A, et al. Predicting treatment response in social anxiety disorder from functional magnetic resonance imaging. JAMA Psychiatry. 2013;70:87-97.
Publication Dates
-
Publication in this collection
Jul-Sep 2013
History
-
Received
2 July 2012 -
Accepted
12 Jan 2013