Abstract
Background
Blood/injection phobia may have important consequences for health. These phobic individuals, in most cases, avoid contact with health systems, postpone or avoid medical procedures, avoid invasive treatments and do not participate in health promotion and early detection of disease initiatives such as vaccination, consultations, preventive exams or blood donation. Thus, specific and validated instruments are necessary to assess this variable. In addition, a lack of studies on this theme may be associated with the low availability of instruments. This study aimed to propose a Portuguese version of the Blood/Injection Fear Scale (BIFS-P) and assess its psychometric properties.
Methods
Translation and back-translation were performed. Content validity was assessed in two steps by a panel of 20 experts. The psychometric properties were assessed in a stratified and representative sample of primary health care service users of Ribeirão Preto, southeastern Brazil. Exploratory and confirmatory factor analyses were conducted using a polychoric correlation matrix.
Results
A total of 1054 primary health care users participated; 79.7% were female and the mean age was 40.6 (standard deviation = 15.16) years. According to the exploratory factor analysis, the items can be grouped into three or five factors with best fits being detected for the three- and five-factor models in confirmatory factor analysis.
Conclusion
Blood/Injection Fear Scale (Portuguese version) is easy to understand and apply in the general population, showed adequate psychometric properties, and represents an alternative in the assessment of blood/injection phobia for future studies.
Keywords:
Fear; Blood; Injections; Translating; Psychometrics
Introduction
Blood and injection phobias are characterized by an extreme aversion of see blood or receive injections or be subjected to invasive medical procedures.11 Hagopian LP, Crockett JL, Keeney KM. Multicomponent treatment for blood-injury-injection phobia in a young man with mental retardation. Res Dev Disabil. 2001;21(2):141-9.
According to Kose and Mandiracioglu,22 Kose S, Mandiracioglu A. Fear of blood/injection in healthy and unhealthy adults admitted to a teaching hospital. Int J Clin Pract. 2007;61(3):453-7. among the various types of specific phobias, there are two types of blood/injection phobia described in the scientific literature that are classified according to the presence or absence of a vasovagal reaction. The vasovagal reaction, characterized by a sudden initial increase in heart rate and blood pressure followed by a sudden drop, is a dysphasic cardiovascular response elicited in the face of the exposure of the individual to situations of fear. For this reason, the vasovagal reaction is accompanied by symptoms such as discomfort, nausea, pallor, expressions of disgust/loathing, and even fainting, all of which are very common among patients with blood and injection phobias.22 Kose S, Mandiracioglu A. Fear of blood/injection in healthy and unhealthy adults admitted to a teaching hospital. Int J Clin Pract. 2007;61(3):453-7.
3 Trijsburg RW, Jelicic M, Van den Broek WW, Plekker AEM, Verheij R, Passchier J. Exposure and participant modelling in a case of injection phobia. Psychother Psychosom. 1996;65(1):57-61.-44 Buodo G, Sarlo M, Poli S, Giada F, Madalosso M, Rossi C, et al. Emotional anticipation rather than processing is altered in patients with vasovagal syncope. Clin Neurophysiol. 2012;123(7):1319-27.
Unlike other types of specific phobias, blood and injection phobias may have important consequences for health. In most cases, these individuals avoid contact with health systems, postpone or avoid medical procedures, avoid invasive treatments and do not participate in health promotion and early detection of disease initiatives such as vaccination, consultations, preventive exams or blood donation.55 Armstrong T, Hemminger A, Olatunji BO. Attentional bias in injection phobia: overt components, time course, and relation to behavior. Behav Res Ther. 2013;51(6):266-73.,66 Miloyan B, Eaton WW. Blood-injection-injury phobia in older adults. Int Psychogeriatr. 2016;28(6):897-902.
In the face of these direct implications to the health, the development and validation of specific instruments to evaluate blood/injection phobia are necessary. Some instruments were proposed for this investigation in the general population, such as the Blood-Injection Symptom Scale (BISS)77 Page AC, Bennett KS, Carter O, Smith J, Woodmore K. The Blood-Injections Symptom Scale (BISS): assessing a structure of phobic symptoms elicited by blood and injections. Behav Res Ther. 1997;35(5):457-64. and the Blood/Injection Fear Scale (BIFS).22 Kose S, Mandiracioglu A. Fear of blood/injection in healthy and unhealthy adults admitted to a teaching hospital. Int J Clin Pract. 2007;61(3):453-7.
The BIFS was proposed by Kose and Mandiracioglu in 200722 Kose S, Mandiracioglu A. Fear of blood/injection in healthy and unhealthy adults admitted to a teaching hospital. Int J Clin Pract. 2007;61(3):453-7. and consists of 20 items divided into two factors, fear of blood and fear of injections and the symptoms involved. The original version of this scale was published in English and until now, it has not been translated to other languages.
Few studies have been conducted to investigate blood/injection phobia in the Brazilian population.88 D'El Rey GJ, Pacini CA. Prevalência da fobia de sangue-injeção-ferimentos em amostra da população de São Paulo-SP. Psicol Argum. 2005;23(43):53-9.,99 D'El Rey GJ, Peroni PC. Estudo de confiabilidade da versão em português da Escala de Sintomas de Sangue-Injeção (BISS). Psicologia. 2007;12(11):1-11. To the best of our knowledge, there is only one Brazilian study, published by D'El Rey and Pacini in 2005; it investigated the prevalence of blood and injection phobias in a representative sample of the general population of the city of São Paulo.88 D'El Rey GJ, Pacini CA. Prevalência da fobia de sangue-injeção-ferimentos em amostra da população de São Paulo-SP. Psicol Argum. 2005;23(43):53-9. Among the results, the combined prevalence of blood or injection phobias was 4.1%, with this fear being more common among women and among individuals with little schooling.
It is believed that the low availability of instruments that are appropriate and adapted for use in the general population is associated with the low number of epidemiological studies in Brazil on this type of phobia. The majority of studies in the area performed in Brazil refers to diagnostic and treatment techniques and are from the psychology and/or psychiatric fields.1010 Guimarães AM, Silva Neto AC, Vilar AT, Almeida BG, Albuquerque CM, Fermoseli AF. Transtornos de ansiedade: um estudo de prevalência sobre as fobias específicas e a importância da ajuda da psicologia. Cad Grad. 2015;3(1):115-28.
Objectives
In light of the foregoing, this study was conducted to present a Portuguese version of the BIFS and evaluate its psychometric properties in a representative sample of primary health care users in Ribeirão Preto, southeastern Brazil.
Methods
Instrument characteristics
BIFS is composed of 20 items divided into two factors according to the original proposal; the first is ‘fear of injections' (Items 1-12) and the second "fear of blood" (Items 13-20).22 Kose S, Mandiracioglu A. Fear of blood/injection in healthy and unhealthy adults admitted to a teaching hospital. Int J Clin Pract. 2007;61(3):453-7. Responses use a 5-point Likert scale (1 = Strongly agree; 2 = Agree; 3 = Neither agree nor disagree; 4 = Disagree; 5 = Strongly disagree).22 Kose S, Mandiracioglu A. Fear of blood/injection in healthy and unhealthy adults admitted to a teaching hospital. Int J Clin Pract. 2007;61(3):453-7.
Translation, back translation and cultural adaptation
Three bilingual translators, whose native language is Brazilian Portuguese, performed the translation of the BIFS, independently. The two researchers responsible for the study, who are also bilingual, compared the three translated versions. Thus, the Portuguese version of BIFS was defined.
The Portuguese version of the instrument was sent to a translator fluent in Brazilian Portuguese whose native language is English to perform the back translation. This translator was not aware of the content of the original scale and was not informed that it was a back-translation. The back translation was important to confirm that the translation of the instrument did not change the meaning of the items according to the original proposal.
After these steps, the Portuguese version of the BIFS (BIFS-P) was obtained and had its psychometric properties evaluated in this study.
Content validity
To evaluate the objectivity and relevance of the BIFS-P, the content of its items was assessed independently by a panel of eight judges, health professionals, with experience in hematology transfusion medicine and/or in professional services in primary health care. Judges were informed about the objectives of the study and were asked to classify each item of the instrument as adequate or not adequate for use in the target population. Suggestions of modifications and simplification of items were requested in these cases. The content validity index (CVI) was calculated for each item considering that IVC ≤ 0.70 is indicative of the need of a new translation (i.e., 30% or more of the judges classified the item as not adequate).1111 Polit DF, Beck CT. The Content Validity Index: are you sure you know what's being reported? Critique and recommendations. Res Nurs Health. 2006;29(5):489-97. According to the suggestions of the judges, some items were modified aiming to simplify and improve the understanding prior to the application of the instrument in the target population.
In a second phase of the content analysis, a panel of 20 judges with experience in hematology and transfusion medicine was invited to classify, also independently, each item of the instrument according to its essentiality (essential, useful but not essential or not necessary) to evaluate ‘fear of blood or injections'. The content validity ratio was calculated according to the proposal of Lawshe.1212 Lawshe CH. A quantitative approach to content validity. Pers Psychol. 1975;28(4):563-75. The significance of the decision was in accordance with the proposal of Wilson et al.,1313 Wilson FR, Pan W, Schumsky DA. Recalculation of the critical values for Lawshe's content validity ratio. Meas Eval Couns Dev. 2012;45(3):197-210. adopting a level of 5%.
Training of interviewers
Before starting data collection, the three interviewers responsible for this step participated in a training session on how to apply the instrument and all the strategies regarding the approach, explanation of the research objectives and instructions to patients were discussed.
Face validity
The pre-test of the BIFS-P in the target population was conducted in 30 primary health care users of Ribeirão Preto in a single healthcare clinic located in the central region, which caters for different medical specialties. The incomprehension index (proportion) of each item was calculated and, if necessary, the item was rewritten.
Data collection
The sample was composed by adult individuals (≥18 years), residents in Ribeirão Preto, southeastern Brazil, who are primary health care users in the municipality. Ribeirão Preto is a medium sized city of São Paulo State that has approximately 670,000 inhabitants. The municipality is divided into five health districts, North, West, Central, South and East. At the time of data collection, there were 41 primary government healthcare clinics in the municipality, which were classified according to the district in which they are located and to the Paulista Index of Social Vulnerability (PSVI) prevalent in their area. The PSVI is developed by the SEADE (State System of Data Analysis) from census data, with the geographic areas being classified into six distinct groups of social vulnerability.1414 Ferreira MP, Dini NP, Ferreira SP. Espaços e dimensões da pobreza nos Municípios do Estado de São Paulo: Índice Paulista de Vulnerabilidade Social - IPVS. São Paulo Perspect. 2006;20(1):5-17. Healthcare clinics were grouped into 12 strata according to the district where they are located and the predominant IPVS. The clinics where the PSVI was equal to 1 or 2 were grouped in the same stratum and the clinics with PSVI equal to or greater than 4 were grouped in another stratum.
It should be noted that this study is part of a wider project that aims to evaluate the association of some variables with blood donation. Thus, the sample size was obtained considering a stratified sampling plan,1515 Scheaffer RL, Mendenhall W, Ott RL, Gerow K. Elementary Survey Sampling. 7th ed. Boston: Cengage Learning; 2011. a 95% confidence level and an absolute precision of 3% for the estimative of the proportion of blood donors. This proportion, for maximization of variance, was considered equal to 50% in each stratum. Thus, a sample size of 1054 interviews was estimated, with the number of interviews to be conducted in each stratum being proportional to their estimated population size. The healthcare clinics where the interviews were conducted were selected randomly within each stratum. In addition to the BIFS-P, a questionnaire about sociodemographic characteristics and general health of the participants was used to characterize the sample.
Psychometric properties of BIFS-P
Factorial validity of the BIFS-P was assessed using exploratory factor analysis (EFA) and confirmatory factor analysis (CFA). EFA was performed using polychoric correlation methods implemented using the SAS software. The CFA was also performed using the polychoric correlations matrix implemented in the software Mplus, version 6.0. The estimation method used in CFA was the weighted least squares mean and variance adjusted (WLSMV). The ratio of Chi-square to its degrees of freedom (χ2/df), comparative fit index (CFI), Tucker-Lewis index (TLI) and root mean square error of approximation (RMSEA) were considered as goodness of fit indices. The fit of the models was considered adequate when χ2/df was ≤2.0, CFI and TLI was ≥0.90 and RMSEA was <0.10.1616 Maroco J. Análise de equações estruturais. 2nd ed. Lisboa: ReportNumber; 2014. Items with factor weights (λ) lower than 0.40 were considered underweight items for the corresponding factor. Modification indices, obtained from the Lagrange multipliers, were used to verify the existence of correlations between errors.1616 Maroco J. Análise de equações estruturais. 2nd ed. Lisboa: ReportNumber; 2014. Comparisons between the tested models were performed considering their respective fit in general, through the goodness of fit indices, with emphasis on the RMSEA index.1717 León DA. Análise Fatorial Confirmatória através dos Softwares R e Mplus. Porto Alegre, Brazil: Universidade Federal do Rio Grande do Sul; 2011. RMSEA estimates show how well the parameters of the model reproduce the population covariance and can be considered a parsimonious correction index. Thus, lower values of RMSEA are indicative of better models.1717 León DA. Análise Fatorial Confirmatória através dos Softwares R e Mplus. Porto Alegre, Brazil: Universidade Federal do Rio Grande do Sul; 2011.,1818 Kline RB. Principles and practice of structural equation modeling. New York: The Guilford Press; 1998, 354 pp.
Factorial invariance of the models was assessed using multigroup analysis considering the Chi-square difference (Δχ2). This analysis is used to evaluate the stability of the models in different samples. For this, the total sample was randomly divided into two equal parts the ‘test sample' and the ‘validation sample'. In this analysis the equivalence of factorial weights [metric invariance (λ)], the factorial weights and intercepts [scalar invariance (Int)], and the factorial weights, intercepts and variances/covariances [strict invariance (Cov)] were evaluated.1616 Maroco J. Análise de equações estruturais. 2nd ed. Lisboa: ReportNumber; 2014.,1919 Kaplan D. Structural equation modeling: foundations and extensions. Thousand Oaks, CA: Sage Publications; 2000.
Ethical aspects
This study was approved by the Ethics Committee on Human Research of the Hospital das Clínicas in Ribeirão Preto (CAAE: 38148814.2.0000.5440), with data collection being approved by the Department of Health of Ribeirão Preto. In addition, this study was conducted in accordance with the Helsinki Declaration as revised in 2008. Only adult individuals (≥18 years) who agreed and signed informed consent forms participated. The authors of the original scale authorized the translation and validation of the instrument.
Results
Content validity
The first step of the content validity process of the BIFS-P was performed by a panel of four male and four female judges with a mean age of 48.8 (SD 13.3) years who have been healthcare professionals for 20.6 years on average (SD 15.1). Of these, 37.5% work in hematology/transfusion medicine services and 62.5% are engaged in primary healthcare activities. According to the CVI values (CVI ≥ 0.75 for all items), there was no need for new translations of any item of the BIFS-P. However, some important suggestions were made and considered to simplify and adapt the instrument for the target population.
The second step of the content validity process was performed by 20 judges, who did not participate in the first step of this evaluation; 80% were female, the mean age was 36.1 (SD 9.6) years and the judges have worked as healthcare professionals for 10.6 (SD 8.2) years on average. Of these, 10% are engaged in activities of attending the population in primary healthcare services, 10% are nursing staff in transfusion medicine or blood donation services, and 80% are engaged in other types of activities in services of hematology/transfusion medicine or blood donation. The content validity ratio (CVR) was estimated and is presented in Table 1.
Original and Portuguese version of Blood/Injection Fear Scale and the results of content validity ratio (CVR) estimated for each item.
Only Items 1, 4, 8, 11, 15, and 18 were considered essentials by the judges.
Face validity
In the pre-test of the scale in the target population, 30 patients were interviewed (female: 73.3%; mean age: 36.97 ± 15.1 years) in a single healthcare clinic located in the central region of the municipality which attends different specialties. The incomprehension index was null for all items, indicating that there was no need for rewrites.
Psychometric properties
In the data collection step which aims to evaluate the psychometric properties of the BIFS-P, 1055 users of primary healthcare services in the municipality were interviewed in 12 healthcare clinics (female: 79.7%; mean age: 40.6 ± 15.2 years). Regarding the perception of health, 69.7% classified their health as ‘good', 26.5% as ‘regular', and 3.8% as ‘bad'. Arterial hypertension and diabetes mellitus were the most prevalent chronic diseases in the sample at 22.0% and 8.0%, respectively.
Figure 1 presents the polychoric correlations matrix for the 20 items of the BIFS-P. Higher correlation coefficients (>0.90) were detected between Items 11 and 12, 13 and 14, and 16 and 17. Items 2 and 19 were not strongly correlated with any other item of the BIFS-P, with correlation coefficients between 0.39 and 0.62.
Polychoric correlations matrix for the items of the Portuguese version of the Blood/Injection Fear Scale. Darker colors were attributed to higher correlation coefficients.
Table 2 shows the results of the EFA. According to the EFA, the items can be grouped in up to five factors, with Factors 4 and 5 being responsible for providing a more specific feature of the BIFS-P. These factors are related to fear and/or reactions on seeing their own blood (Factor 4) and to fear and/or reactions to seeing the blood of others (Factor 5).
Exploratory factor analysis (EFA) performed for the Portuguese version of Blood/Injection Fear Scale applied to a sample of primary health care users of Ribeirão Preto, southeastern Brazil.
Thus, three factorial structures of the BIFS-P were tested in the CFA: (1) original factorial structure with two first-order factors (fear of blood/fear of injections); (2) factorial structure with three main first-order factors resulting from the EFA; (3) factorial structure with 5 first-order factors resulting from EFA. These structures are called Model 1, Model 2 and Model 3, respectively.
Table 3 presents the results of the CFA for the factorial structures tested for the BIFS-P. It should be mentioned that it was necessary to exclude Item 19 due to its low factorial weight in all analyses and lower correlation coefficients with all the other items of the scale in order to have adequate fits of all models (Figure 1).
Factorial weights (λ), goodness of fit indices of the confirmatory factor analysis (χ 2/df, CFI, TLI, and RMSEA), modification indices, and correlation between errors adopted for the three factorial structures tested for the Portuguese version of Blood/Injection Fear Scale in a sample of primary health care users of Ribeirão Preto, southeastern Brazil.
Despite the appropriate factor weights of all items in each factor in the three models tested (λ > 0.60), the original structure with two factors (Model 1) does not present an adequate fit to the data considering the goodness of fit indices. Even after the exclusion of Item 19 and the model re-specification (seven correlations between errors considered), Model 1 presented results of RMSEA higher than adequate and high modification indices, which can be considered a poor adjustment (Table 3). On the other hand, Models 2 and 3 presented adequate fit to the data, with goodness of fit indices within acceptable limits (after re-specification of the models and the exclusion of Item 19). Despite high levels of χ2/df, this index is overestimated in larger samples, and should be evaluated together with the other indices to make decisions on the fit of the models. In Models 2 and 3, observing the RMSEA values and their confidence intervals, it is clear that no model has a better fit as both models are properly adjusted to the data and both structures can be considered in the assessment of fear of blood and injections in this sample.
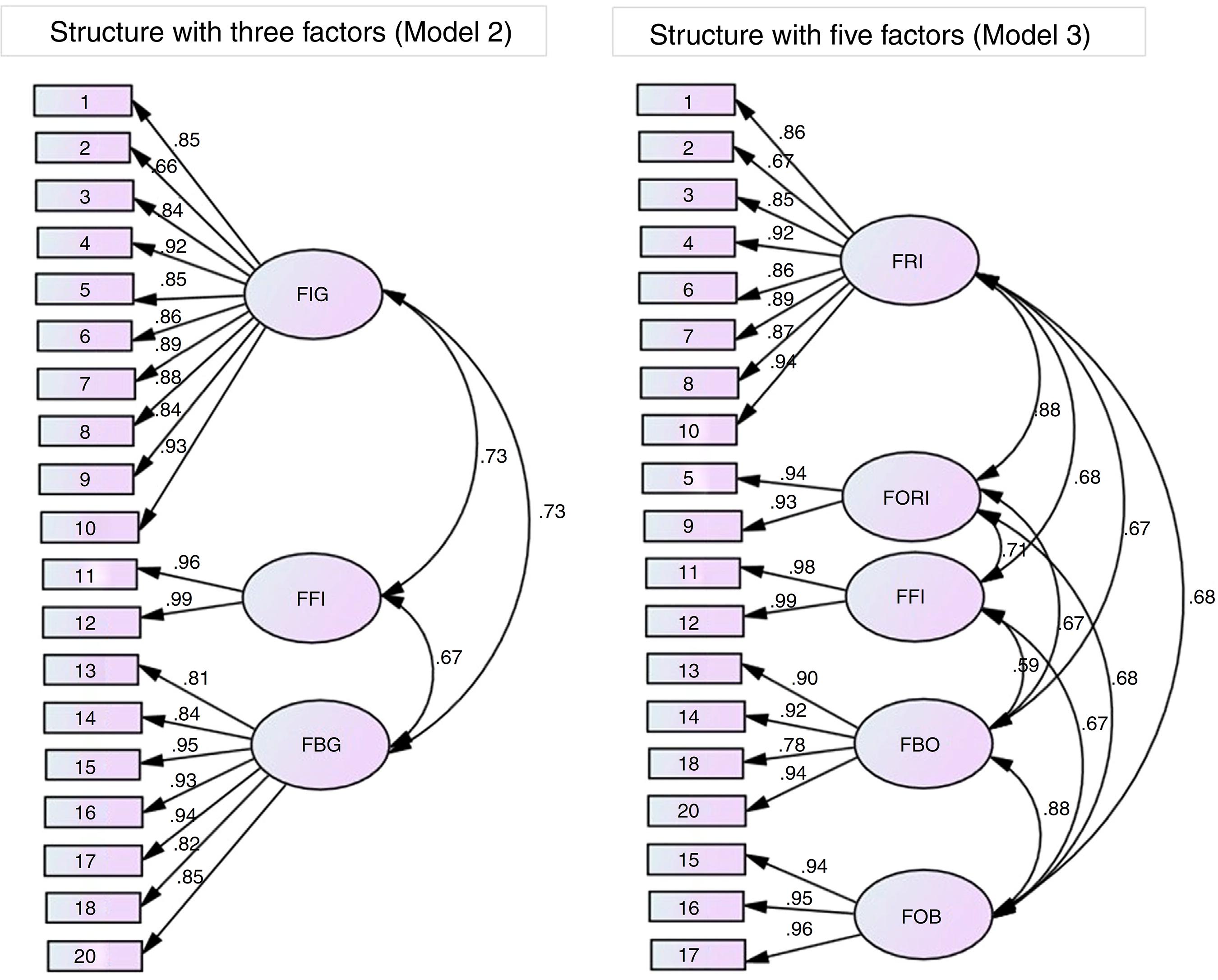
Distribution of the items in each factor considered in the confirmatory factor analysis of Models 2 and 3 and the factorial weights (λ) are presented in Figure 2. The names of the factors were adopted in accordance with the theoretical content of the items when grouped.
Distribution of items of the Portuguese version of Blood/Injection Fear Scale in each factor and the local fit (λ) of Models 2 and 3 of the data obtained in the confirmatory factor analysis. FIG: fear of injection in general; FFI: fainting due to fear of injection; FBG: fear of blood in general; FRI: fear of receiving injection; FORI: fear of seeing others receiving injection; FFI: fainting due to fear of injection; FBO: fear of seeing blood of others; FOB: fear of own blood.
All items presented adequate factorial weights for the corresponding factors (λ ≥ 0.66), which confirms an adequate distribution of items in the exploratory factor analysis.
Model 2 (structure with three factors) presented equivalence of factorial weights (metric invariance - λ: χ2 = 18.7; p-value = 0.29), of factorial weights and intercepts (scalar invariance - Int: χ2 = 85.2; p-value = 0.10), and of factorial weights, intercepts and variances/covariances (strict invariance - Cov: χ2 = 67.7; p-value = 0.09) considered very stable in different samples. Model 3 (structure with five factors) presented equivalence of factorial weights in different samples (metric invariance - λ: χ2 = 20.6; p-value = 0.11) with poor stability.
Discussion
There is a lack of studies and instruments to assess blood/injection phobia in the general Brazilian population, since most of the instruments available are specific to psychiatry and focus on diagnosis and treatment. In addition, little is known about the prevalence of this type of phobia and associated factors in the Brazilian population; this is an important gap in the scientific literature in the face of the important consequences that fear of blood and injection may have on the individual's health.
Regarding the CVR estimated for the items, only six items were considered essential by the judges (Table 1). Of these items, four are related to the fear of receiving injections and adverse reactions and two are related to the reactions such as discomfort/feeling ill on seeing their own blood and/or fainting at the sight of blood. As 90.0% of the judges are health professionals who perform some kind of activity in hematology/transfusion medicine/blood donation services, these results suggest that there may have been a trend by the professionals on give more importance to these items. However, content validity, analyzed using the CVR, should be considered as a complementary analysis and may not be used only as justification for the exclusion of items of an instrument.2020 Anastasi A. Psychological testing. 6th ed. New York: Macmillan Publishing Company; 1988.,2121 Pais-Ribeiro JL. Medida na avaliação psicológica. Psicol Saude Doe. 2013;14(1):245-63.
In the polychoric correlations matrix presented (Figure 1), most items of the BIFS-P were strongly correlated according to participants' responses. Despite the high correlation coefficient (>0.90) between three pairs of items (11 and 12, 13 and 14, and 16 and 17), these items showed high factor weights in all the models tested in the CFA (Figure 2) and for this reason they have been maintained in the instrument. However, this is not the case of Item 19, related to the smell of the injection application room. This item, besides the low correlation with all other items of the scale (Figure 1), presented low factor weight in all models tested, and hindered their fit. For this reason, we opted to exclude Item 19 in the analysis, and this exclusion was guided both by the low factorial weight detected and by the classification as a non-essential item in the content analysis.
The choice of the best factorial structure to be used to assess the fear of blood/injections in the general population is worth some consideration. Firstly, both models (with three and five factors) presented adequate fit to the data, suggesting that both are suitable to measure the construct. However, Model 2 provides a more comprehensive measure guided by a larger grouping of items. Model 3, in turn, provides a more specific measure, which can distinguish more precisely the situation of fear and reactions. Thus, the choice of the structure to be used will depend on the objectives of each study to be conducted. Furthermore, Model 2 was strongly stable in the analysis of factorial invariance in random subsamples, which allows us to suggest its use in studies of Brazilian users of healthcare services.
With respect to the overall score of the BIFS-P, the authors of the original proposal suggest the sum of all answers. However, there are strong arguments that contradict the use of the sum of responses when using psychometric scales mainly directed at the different factorial weights of items and distribution of factors in distinct populations.2020 Anastasi A. Psychological testing. 6th ed. New York: Macmillan Publishing Company; 1988.,2121 Pais-Ribeiro JL. Medida na avaliação psicológica. Psicol Saude Doe. 2013;14(1):245-63. Thus, we suggest using the arithmetic mean for the computation of an overall score or a score for each factor separately.
We conclude that the BIFS-P is an important instrument in the face of adequate psychometric properties found in the sample of primary health care users and its simplicity and ease of understanding within the public health context. We suggest that other studies should be conducted to assess the psychometric properties of the BIFS-P in different populations such as patients with chronic disease, blood donors, elderly people, high school students, students in healthcare courses, among others, aiming to expand the utility of this instrument and contribute to other areas of collective health.
-
Financial supportThis study was supported by the Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) under grant #2014/14020-6; and the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES).
Acknowledgements
This study was funded by the Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) 2014/14020-6 and the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES).
References
-
1Hagopian LP, Crockett JL, Keeney KM. Multicomponent treatment for blood-injury-injection phobia in a young man with mental retardation. Res Dev Disabil. 2001;21(2):141-9.
-
2Kose S, Mandiracioglu A. Fear of blood/injection in healthy and unhealthy adults admitted to a teaching hospital. Int J Clin Pract. 2007;61(3):453-7.
-
3Trijsburg RW, Jelicic M, Van den Broek WW, Plekker AEM, Verheij R, Passchier J. Exposure and participant modelling in a case of injection phobia. Psychother Psychosom. 1996;65(1):57-61.
-
4Buodo G, Sarlo M, Poli S, Giada F, Madalosso M, Rossi C, et al. Emotional anticipation rather than processing is altered in patients with vasovagal syncope. Clin Neurophysiol. 2012;123(7):1319-27.
-
5Armstrong T, Hemminger A, Olatunji BO. Attentional bias in injection phobia: overt components, time course, and relation to behavior. Behav Res Ther. 2013;51(6):266-73.
-
6Miloyan B, Eaton WW. Blood-injection-injury phobia in older adults. Int Psychogeriatr. 2016;28(6):897-902.
-
7Page AC, Bennett KS, Carter O, Smith J, Woodmore K. The Blood-Injections Symptom Scale (BISS): assessing a structure of phobic symptoms elicited by blood and injections. Behav Res Ther. 1997;35(5):457-64.
-
8D'El Rey GJ, Pacini CA. Prevalência da fobia de sangue-injeção-ferimentos em amostra da população de São Paulo-SP. Psicol Argum. 2005;23(43):53-9.
-
9D'El Rey GJ, Peroni PC. Estudo de confiabilidade da versão em português da Escala de Sintomas de Sangue-Injeção (BISS). Psicologia. 2007;12(11):1-11.
-
10Guimarães AM, Silva Neto AC, Vilar AT, Almeida BG, Albuquerque CM, Fermoseli AF. Transtornos de ansiedade: um estudo de prevalência sobre as fobias específicas e a importância da ajuda da psicologia. Cad Grad. 2015;3(1):115-28.
-
11Polit DF, Beck CT. The Content Validity Index: are you sure you know what's being reported? Critique and recommendations. Res Nurs Health. 2006;29(5):489-97.
-
12Lawshe CH. A quantitative approach to content validity. Pers Psychol. 1975;28(4):563-75.
-
13Wilson FR, Pan W, Schumsky DA. Recalculation of the critical values for Lawshe's content validity ratio. Meas Eval Couns Dev. 2012;45(3):197-210.
-
14Ferreira MP, Dini NP, Ferreira SP. Espaços e dimensões da pobreza nos Municípios do Estado de São Paulo: Índice Paulista de Vulnerabilidade Social - IPVS. São Paulo Perspect. 2006;20(1):5-17.
-
15Scheaffer RL, Mendenhall W, Ott RL, Gerow K. Elementary Survey Sampling. 7th ed. Boston: Cengage Learning; 2011.
-
16Maroco J. Análise de equações estruturais. 2nd ed. Lisboa: ReportNumber; 2014.
-
17León DA. Análise Fatorial Confirmatória através dos Softwares R e Mplus. Porto Alegre, Brazil: Universidade Federal do Rio Grande do Sul; 2011.
-
18Kline RB. Principles and practice of structural equation modeling. New York: The Guilford Press; 1998, 354 pp.
-
19Kaplan D. Structural equation modeling: foundations and extensions. Thousand Oaks, CA: Sage Publications; 2000.
-
20Anastasi A. Psychological testing. 6th ed. New York: Macmillan Publishing Company; 1988.
-
21Pais-Ribeiro JL. Medida na avaliação psicológica. Psicol Saude Doe. 2013;14(1):245-63.
Publication Dates
-
Publication in this collection
Jul-Sep 2017
History
-
Received
13 Feb 2017 -
Accepted
23 May 2017