Abstracts
Pectin lyase and polygalacturonase production by newly isolated Penicillium viridicatum strain Rfc3 was carried out by means of solid state fermentation using orange bagasse, corn tegument, wheat bran and mango and banana peels as carbon sources. The maximal activity value of polygalacturonase (Pg) (30U.g-1) was obtained using wheat bran as carbon source while maximal pectin lyase (Pl) (2000 U.g-1) activity value was obtained in medium composed of orange bagasse. Mixtures of banana or mango peels with sugar cane bagasse resulted in increased Pg and Pl production compared to fermentations in which this residue was not used. The mixture of orange bagasse and wheat bran (50%) increased the production of Pg and Pl to 55 U.g-1 and 3540 U.g -1 respectively. Fractions of Pg and Pl, isolated by gel filtration in Sephadex G50, presented optimum activity at pH 5.0 and 10.5 respectively. Maximal activity of Pg and Pl fractions was determined at 55ºC and 50ºC respectively. Pg was stable in neutral pH range and at 40ºC whereas Pl was stable in acidic pH and at 35ºC, for 1 h.
Penicillium viridicatum; polygalacturonase; pectin lyase; wastes, by-product
A produção de pectina liase (Pl) e poligalacturonase (Pg) por cepa de Penicillium viridicatum Rfc3, recentemente isolada, foi estudada por meio de fermentação em estado sólido usando bagaço de laranja, tegumento de milho, farelo de trigo e cascas de manga e banana como fontes de carbono. Quando os resíduos foram utilizados isoladamente, o valor máximo de atividade de Pg (30 U g-1) foi observado em meio de farelo de trigo, enquanto que o valor máximo para atividade de Pl (2000 U g-1) foi obtido em meio de bagaço de laranja. Misturas de cascas de banana ou de manga com bagaço de cana-de-açúcar (50% p/p), resultaram em aumento na produção tanto de Pl quanto de Pg, quando comparado com os experimentos nos quais esses materiais foram usados isoladamente. A mistura de bagaço de laranja e farelo de trigo (50%) elevou a produção de Pg e Pl para 55 U.g-1 e 3540 U.g-1, respectivamente. O fracionamento das enzimas presentes na solução enzimática bruta, através de filtração em gel Sephadex G50, resultou na obtenção de diferentes frações de Pl e de Pg. As frações de Pg e Pl, as quais foram caracterizadas, apresentaram atividade ótima em pH 5,0 e 10,5, respectivamente. A atividade máxima da fração de Pg foi obtida a 55ºC e, para Pl, a 50ºC. A Pg foi estável em valores de pH próximos à neutralidade e a 40ºC, enquanto que a Pl foi estável em pH ácido e a 35ºC, por uma hora.
Penicillium viridicatum; poligalacturonase; pectina liase; wastes, by-product
Pectinase production by Penicillium viridicatum RFC3 by solid state fermentation using agricultural wastes and agro-industrial by-products
Produção de pectinases por Penicillium viridicatum RFC3 através de fermentação em estado sólido, usando resíduos agrícolas e sub-produtos agroindustriais
Dênis Silva; Eduardo da Silva Martins; Roberto da Silva; Eleni Gomes
Laboratório de Bioquímica e Microbiologia, IBILCE, Universidade Estadual Paulista, São José do Rio Preto, SP, Brasil
Correspondence Correspondence to Universidade Estadual Paulista Departamento de Biologia Rua Cristóvão Colombo, 2265, Jardim Nazareth. 15054-000, São José do Rio Preto, SP, Brasil Tel.: (+5517) 221-2393. Fax: (+5517) 221-2390 E-mail: eleni@bio.ibilce.unesp.br.
ABSTRACT
Pectin lyase and polygalacturonase production by newly isolated Penicillium viridicatum strain Rfc3 was carried out by means of solid state fermentation using orange bagasse, corn tegument, wheat bran and mango and banana peels as carbon sources. The maximal activity value of polygalacturonase (Pg) (30U.g-1) was obtained using wheat bran as carbon source while maximal pectin lyase (Pl) (2000 U.g-1) activity value was obtained in medium composed of orange bagasse. Mixtures of banana or mango peels with sugar cane bagasse resulted in increased Pg and Pl production compared to fermentations in which this residue was not used. The mixture of orange bagasse and wheat bran (50%) increased the production of Pg and Pl to 55 U.g-1 and 3540 U.g 1 respectively. Fractions of Pg and Pl, isolated by gel filtration in Sephadex G50, presented optimum activity at pH 5.0 and 10.5 respectively. Maximal activity of Pg and Pl fractions was determined at 55ºC and 50ºC respectively. Pg was stable in neutral pH range and at 40ºC whereas Pl was stable in acidic pH and at 35ºC, for 1 h.
Key words:Penicillium viridicatum, polygalacturonase, pectin lyase, wastes, by-product
RESUMO
A produção de pectina liase (Pl) e poligalacturonase (Pg) por cepa de Penicillium viridicatum Rfc3, recentemente isolada, foi estudada por meio de fermentação em estado sólido usando bagaço de laranja, tegumento de milho, farelo de trigo e cascas de manga e banana como fontes de carbono. Quando os resíduos foram utilizados isoladamente, o valor máximo de atividade de Pg (30 U g-1) foi observado em meio de farelo de trigo, enquanto que o valor máximo para atividade de Pl (2000 U g-1) foi obtido em meio de bagaço de laranja. Misturas de cascas de banana ou de manga com bagaço de cana-de-açúcar (50% p/p), resultaram em aumento na produção tanto de Pl quanto de Pg, quando comparado com os experimentos nos quais esses materiais foram usados isoladamente. A mistura de bagaço de laranja e farelo de trigo (50%) elevou a produção de Pg e Pl para 55 U.g-1 e 3540 U.g-1, respectivamente. O fracionamento das enzimas presentes na solução enzimática bruta, através de filtração em gel Sephadex G50, resultou na obtenção de diferentes frações de Pl e de Pg. As frações de Pg e Pl, as quais foram caracterizadas, apresentaram atividade ótima em pH 5,0 e 10,5, respectivamente. A atividade máxima da fração de Pg foi obtida a 55ºC e, para Pl, a 50ºC. A Pg foi estável em valores de pH próximos à neutralidade e a 40ºC, enquanto que a Pl foi estável em pH ácido e a 35ºC, por uma hora.
Palavras-chave:Penicillium viridicatum, poligalacturonase, pectina liase, "wastes, by-product"
INTRODUCTION
A large number of microorganisms, isolated from different materials, have been screened for their ability to degrade polysaccharides present in biomass, producing protein or higher value products (7,29,39).
Among depolimerizing microbial enzymes, pectinolytic enzymes, which degrade pectin present in varying abundance in the middle lamella and primary cell walls, have great commercial importance for various industrial applications like improving juice yields and clarity (3,12). The use of liquefying enzymes for mash treatment results in improvement of juice flow, leading to a shorter press-time, without the necessity for pressing aids (35). At the same time, pectin is broken down to such an extent that the viscosity of mash is reduced (28). Other areas of application include the paper and pulp industry (36), waste management (13), animal feed (6) textile industry (4).
Pectinolytic enzymes are classified according to their mode of attack on the galacturonan part of the pectin molecule. They can be distinguished from pectin methylesterases (EC 3.1.11.1), that deesterify pectins to low methoxyl pectins or pectic acid, and form pectin depolymerases, that split the glycosidic linkages between galacturonosyl (methyl ester) residues. Polygalacturonases split glycosidic linkages next to free carboxyl groups by hydrolysis while pectate lyases split glycosidic linkages next to free carboxyl groups by b-elimination. Both endo types of Pgs and PAls (EC 3.2.1.15 and EC 4.2.2.2, respectively) are known to randomly split the pectin chain. Exo-Pgs (EC 3.2.1.67) release monomers or dimmers from the non-reducing end of the chain, whereas exo-PAls (EC 4.2.2.9) release unsaturated dimmers from the reducing end. Highly methylated pectins are degraded by endo-pectin lyases (Pl; EC 4.2.2.10) and also by a combination of pectin esterases with Pg or PAl (10,34).
Major impediments to the exploitation of commercial enzymes are their yield, stability, specificity and the cost of production. New enzymes for use in commercial applications with desirable biochemical and physico-chemical characteristics and a low cost of production have been the focus of much research. Application of agro-industrial wastes as carbon sources in enzyme production processes reduces the cost of production, and also helps in solving problems with their disposal (33).
In this paper we report the production of pectolytic enzymes by a newly isolated strain of Penicillium viridicatum Rfc3 by solid-state fermentation using agricultural wastes and agro-industrial by-products. Some physico-chemical characteristics of the enzymes fractions are presented.
MATERIALS AND METHODS
Microorganism
The Penicillium vericatum Rfc 3 strain used in this study was recently isolated from decaying vegetable collected in Bady Bassit-SP/Brazil. The strain was maintained on potato dextrose agar (PDA) slopes as a stock culture.
Media, cultivation of microorganism and enzyme production
The solid substrates were prepared as follows:
a) Orange bagasse. The pellet of orange bagasse (pressed mixture of pulp and peel) was provided by Citrovita Agro-industrial Ltda/Catanduva/SP/Brazil. Chemical analysis indicated that the dry material was composed of 11.8% fiber, 6.4% protein, 63.0% nitrogen, 6.7% ash, 19.0% total sugar (9.0% reducing sugar) and 0.1% pectin. The material was ground and particles sieved by a Bender USS 230 strainer and dried at 80ºC.
b) Sugar cane bagasse: The sugar cane bagasse was provided by Usina de Açúcar e Álcool São Domingos/Catanduva/SP/Brazil. Chemical analysis indicated that the dry material was composed of 75.0% fiber, 0.5% protein, 5.0% nitrogen, 3.4% ash, 10.4% total sugar (3.5% reducing sugar).The material was ground and particles sieved by a Bender USS 230 strainer and dried at 80ºC.
c) Wheat bran: This material was purchased from the local market, dried and used untreated. Chemical analysis indicated that the dry material was composed of 8.1% fiber, 15.7% protein, 65.0% nitrogen, 4.6% ash, 16.7% total sugar (5.2% reducing sugar).
d) Banana and mango were purchased from the local market. The peels were removed, dried and ground to about 2 mm in particle size. The total and reducing sugar contents in banana peel were 26.2% and 13.2%, respectively and in mango peel were 39.3% and 19.5%, respectively. No other chemical analyses were performed. The dried peels were not submitted to any chemical treatment.
e) Corn teguments were supplied by Laboratório de Cereais/Departamento de Engenharia de Alimentos/IBILCE/UNESP/São José do Rio Preto/SP/Brasil, as a residue from corn processing. The total and reducing sugar content were 10.4% and 3.5%, respectively. No other chemical analyses were performed.
Solid-state fermentation (SSF) was carried out using a 250 ml Erlenmeyer flask containing 5g of sterilized substrate (120ºC/40min) inoculated with 10 ml aliquots of conidia suspension (approx. 107 spores/g dry substrate) obtained from a 7-day agar slant culture suspended in sterile Tween 80 solution. After inoculation, 10 ml of nutrient solution, composed of 0.1% NH4NO3; 0.1% NH4H2PO4; 0.1% MgSO4 x 7H2O, was added to each flask. The final moisture content of the medium was approximately 67%. The substrates were mixed in proportions of 50%.
The cultivation was carried out at 30ºC for 14 days. At 48h intervals, the solid fermented material corresponding to one Erlenmeyer flask was mixed with 40 ml distilled water, stirred for 40 min, filtered under vacuum and centrifuged. The supernatant was used as crude enzyme solution.
Enzyme activity measurements
Polygalacturonase (Pg) activity was determined by measuring the release of reducing groups from citrus pectin using the 3,5-dinitrosalicylic acid (DNS) reagent assay (25). The reaction mixture containing 0.8 ml 1% citric pectin 67% methoxylated (Braspectina-Limeira/SP/Br) in 0.2M acetate buffer, pH 5.0 and 0.2 ml of crude enzyme solution, was incubated at 50ºC for 10 min. One unit of enzymatic activity (U) was defined as the amount of enzyme which releases one mmol of galacturonic acid per minute.
Pectin lyase (Pl) activity was determined by measuring the increase in absorbance at 235 nm of substrate solution (0.8 ml 1% citric pectin in 0.2M tris-HCl buffer, pH 8.5) hydrolyzed by 0.2 ml enzyme solution, at 50ºC. One unit of enzymatic activity (U) was defined as the amount of enzyme which releases 1 µmol of unsaturated uronide per minute, based on the molar extinction coefficient (e=5500) of the unsaturated products (2).
The enzyme production was expressed in units per gram of initial dry solid substrate (U.g-1).
Separation of the pectinases
The crude enzyme (300 ml), obtained after 2 and 14 cultivation days on orange bagasse and wheat bran mixture (50% w/w), was slowly dispersed in two volumes of iced ethanol and maintained at -20ºC for 2 hours. The resulting precipitate was collected by centrifugation (20,000xg for 20 min), dissolved in the smallest possible volume of tris-HCl buffer (50 mM, pH 7.4) and applied to a Sephadex G 50 column (3 x 100cm) equilibrated with 50 mM tris-HCl buffer (pH 7.4). The enzymes were eluted with the same buffer.
Enzyme characterization
The fractions of Pg and Pl, obtained from gel filtration, were used for the characterization assays.
Optimum pH and temperature for enzyme activity: The enzyme activity was determined at 50ºC, in different pH using sodium acetate (pH 3.0-5.0), citrate-phosphate (pH 5.0-7.0), tris-HCl (pH 7.0-8.5) and glycine-NaOH (pH 8.5-11.0) as buffers. The optimum temperature within the 30-70ºC range was determined by incubation of the reaction mixture at optimum pH.
pH and temperature stability: Enzyme solution was dispersed (1:1) in 0.2M buffer pH 3.0-5.0 (sodium acetate), pH 5.0-7.0 (citrate-phosphate), pH 7.0-8.5 (tris-HCl) and pH 8.5-11.0 (glycine-NaOH) and maintained at 25ºC for 24h. An aliquot was used to determine the remaining activity at the optimum pH and temperature for enzymes. For the temperature stability determination, the enzyme solution was incubated at different temperatures (30ºC - 70ºC) for 1h at pH 5.0. An aliquot was withdrawn and placed on ice before assaying for residual pectinase activity at optimum pH and temperature.
RESULTS AND DISCUSSION
Production of pectinases by SSF
The fungus Penicillium viridicatum Rfc3, when grown in a media containing wheat bran, orange bagasse, corn tegument, banana and mango peels or mixture of these materials with sugar cane bagasse (50% w/w) produced polygalacturonase and pectin lyase. Enzyme production by SSF was analyzed during 14 days and the experiments were carried out in five assays (Table 1). Polygalacturonase production peaked between the 4th and the 6th days of cultivation when the substrates contained no sugar cane bagasse. In media composed of orange bagasse, wheat bran and corn tegument, a first peak was obtained after the 4th of fermentation and a second and smaller peak, at the 12th day (data not shown). The maximal activity value of Pg (30U.g-1) was obtained using wheat bran as carbon source.
Pl production in media composed by only one type of waste was observed in the beginning of fermentation and peaks of production occurred between the 2th and the10th days. The maximal Pl activity value was obtained in media composed of orange bagasse (2000 U.g-1) (Table 1).
Mixtures of high sugar content wastes such as banana (13.2%) and mango (19.5%) peels with sugar cane bagasse resulted in increase in the Pg and Pl production compared to fermentations in which the bagasse was not used (Table 1). It is known that the synthesis of pectinases by fungi is subject to catabolic repression by high free sugar concentration affecting inducible and constitutive enzymes (1,19). On the other hand, the consistence and size of particles in media composed of banana and mango peels interfered in their packing during the fermentation, since the bed remained static (nonmixed system), affecting the gas and heat exchange within the system (27).The addition of fibrous material as sugar cane bagasse increased the interparticle spacing, possibly increasing the aeration and diffusion of nutrient and enzyme, as shown by Mitchell et al. (26). When solely sugar cane bagasse was used as carbon source, there was no growth of P. viridicatum (data not shown), indicating that the microorganism was not able to hydrolyse enough cellulose and hemicellulose fibers to support mycelium formation. These supposition was confirmed by the low level of cellulase and xilanase produced by this fungus in the experiments (data not shown). The sugar cane bagasse appeared to be only a solid inert support, being microorganism growth and enzyme production stimulated by wastes addition. The use of sugar cane bagasse as support is suitable for the growth of filamentous fungi and it allowed the utilization of high-concentration substrate solutions (30). This supposition is confirmed in solid-state culture of Aspergillus niger (31).
Our results indicated that, in addition to the effect of reducing sugar concentration, Pg and Pl production was influenced by media composition, reaching higher values in media containing high pectin and nutrient concentration (orange bagasse and wheat bran). The mixture of orange bagasse and wheat bran showed to be the best medium for pectinase production, with 55.2 U and 3540 U of Pg and Pl per gram of substrate, respectively. The effect of different carbon sources on pectinase synthesis by fungi in submerged and solid-state fermentation have already been studied, and it is generally agreed that the optimum medium for the enhanced production of extracellular pectinase is that containing pectic materials as an inducer (14,16,20,29,40).
The quantities of Pg and Pl obtained in our study were high compared to those reported for pectinolytic strains such as Aspergillus niger (25 U Pg and 350 U Pl per gram of substrate), P. italicum (6U Pl/g e 25U Pg/g), P. frequentans (3.4 U Pg /g) and A. foedidus (1000 U Pl/g) cultivated on solid substrates (8,9,18,21). However, the comparison of enzyme levels produced by different organisms is not straightforward, since distinct culture conditions and enzyme activity determinations have been used.
These results showed that SSF was suitable for pectinase production by P. viridicatum using agricultural and agro-industrial wastes and by-products.
Assays to evaluate the effect of temperature incubation (28ºC and 30ºC) and initial pH of the medium (4.5 and 5.0) on Pg and Pl production were carried out. The results indicated that maximal quantity of Pg was not significantly influenced by temperature or pH, but the production profile of this enzyme, during fermentation period, varied as a function of initial substrate pH (Fig. 1a; 1b). On the other hand, Pl production decreased at 30ºC, although the pH effect was similar to that observed for Pg production.
Enzymes separation by gel filtration
Gel filtration in Sephadex G50 of the crude enzyme solutions obtained after two days of fermentation resulted in two peaks of Pg and four peaks of Pl (Fig. 2a). Enzyme solution, obtained from medium after 14 days of fermentation, presented three peaks of Pg and two peaks of Pl (Fig. 2b). The position of the enzymes in the chromatogram suggests that two Pgs (I and II) and one Pl (IV) are produced during the whole fermentation period, while three Pl (I, II and III) are observed only in the initial stage of fermentation. After 14 days, both Pg (III) and Pl (V) are produced.
The comparison between Pg and Pl production during growth of P. viridicatum suggests a sequential induction of these enzymes. Sequential production of pectinases have been reported by various authors for different microorganisms (11,15,21,24,38) indicating that this is characteristic of fungi and bacteria. Other hemicellulolytic enzymes presented similar profiles (22).
Enzyme characterization
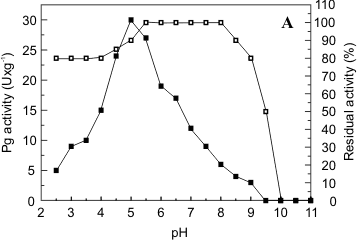
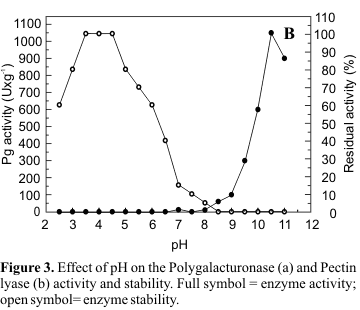
Characterization of the fractions of polygalacturonase (I) and pectin lyase (IV) obtained by gel filtration in Sephadex G50 was carried out. Pg exhibited maximal activity at pH 5.0 (Fig. 3a) and Pl, at pH 10.5 (Fig. 3b). Pg II was stable at pH 5.0-8.0 and maintained 80% of its activity at pH 9.0 (Fig. 3a). Pl was more sensitive to pH variation, presenting maximum stability at pH 3.5-4.5 which declined to 80% at pH 5.0 and to 60% at pH 6.0 (Fig. 3b).
The optimum temperature for Pg activity was 55ºC, presenting 90% of the maximum activity at 60ºC and 67% at 65ºC (Fig. 4a). As illustrated in Fig. 4b, Pl presented optimal activity at 50ºC and 67% of this activity at 55ºC. After incubation at 40ºC for 1h, the retained activities of Pg and Pl were 100% and 80% respectively. After 1h at 50ºC 55% of original Pg activity was maintained while the same conditions of incubation reduced original Pl activity by 60% (Fig. 4a, b).
Previous data indicated differences between physico-chemical properties of enzyme in crude solution (data not shown) and Pg and Pl fractions. Pg I fraction presented higher optimum temperature than crude enzyme solution, which maximal activity between 45 to 50ºC. The fraction studied was stable in neutral pH while activity of the crude enzyme was highest in acidic pH. Pl IV fraction showed to be more alkalophilic (optimum pH was 10.5) and more thermostable than the crude enzyme, which presented optimum pH of 8.5 and maintained only 30% of the original activity at 40ºC. These data indicate that pectinases with different properties were produced.
The optimum pH (5.0) found for Pg I from P. viridicatum Rfc3 is comparable to that from other fungal strains. On the other hand, optimum pH for Pl IV (10.5-11.0) is above the one found for fungal Pl (42), being similar to pectinases from bacteria (Bacillus sp) described by Kobayashi et al. (23).
In terms of thermal stability and optimum temperature for activity, pectinases from P. viridicatum Rfc3 were similar to those reported for several fungal species (36,37). Pectinases from Aspergillus strains have been described as susceptible to denaturation in temperature above 50ºC (5,17,41).
The present work shows that is feasible to use agro-industrial wastes and agro-industrial by-product for production of polygalacturonase and pectin lyase by Penicillium viridicatum strain Rfc3. This newly isolated fungus is able to produce extracellular polygalacturonase and pectin lyase during solid state fermentation, in which different pectinases are produced.
ACKNOWLEDGEMENTS
The authors wish to thank the Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) for financial support.
Submitted: September 13, 2002; Returned to authors for corrections: January 08, 2002; Approved: December 05, 2002.
- 1. Aguillar, G.; Huitron, C. Stimulation of production of extracellular pectinolytic activities of Aspergillus sp by galacturonic acid and glucose additions. Enz. Microbiol. Technol, 9: 690-696, 1987.
- 2. Albersheim, P. Pectin lyase from fungi. Methods Enzymol, 8: 628-631, 1966.
- 3. Alkorta, I.; Garbisu, C.; Llama J.M.; Serra, J.L. Industrial applications of pectic enzymes: a review. Process Biochem, 33: 21-28, 1998.
- 4. Bacarat, M.C.; Valentin, C.; Muchovej, J.J.; Silva, D.O. Selection of pectinolytic fungi for degumming of natural fibers. Biotechnol. Lett., 11: 899-902, 1989.
- 5. Bailey, M.J.; Pessa, E. Strain and process for production of polygalacturonase. Enzyme Microb. Technol, 12: 266-271, 1990.
- 6. Barreto de Menezes, T.J.; Salva, J.G.; Baldini, V.L.; Papini, R.S.; Sales, A.M. Protein enrichment of citrus wastes by solid substrate fermentation. Proc. Biochem., 23: 167-171, 1989.
- 7. Buswell, J.A.; Cai, Y.J.; Chang, S.T.; Peterdy, J.F.; Fu, S.Y.; Yu, H.S. Lignocellulolytic enzyme profiles of edible mushroom fungi. World J. Microb Biotech, 12: 537-542 1996.
- 8. Castilho, L.R.; Alves, T.L.M.; Medronho, R.A. Recovery of pectinolytic enzymes produced by solid state culture of Aspergillus niger Process Biochem, 34: 181-186, 1999.
- 9. Castilho, L.R.; Alves, T.L.M.; Medronho, R.A. Production and extraction of pectinases obtained by solid state fermentation of agro-industrial residues with Aspergillus niger. Biores. Technol., 71: 45-50, 2000.
- 10. Chahal, D.S. Production of Trichoderma reesei cellulase system with high hydrolytic potential by solid state fermentation. In: Leathem, G.F.; Himmel M.E.; (Eds): Enzymes in Biomass Conversion, ACS Symp. Series 460, American Chem. Soc, Whashington, 1991, p.247-269.
- 11. Crotti, L.B.; Jabopr, V.A.; Chellegatti, M.A.; Fonseca, M.J.; Said, S. Studies of pectic enzymes produced by Talaromyces flavus in submerged and solid substrate cultures. J. Basic Microbiol, 39: 27-35, 1999.
- 12. Da Silva, R.; Franco, C.M.L.; Gomes, E. Pectinases, hemicelulases e celulases: ação, produção e aplicação no processamento de alimentos: Revisão. Bol. Sociedade Bras. Ciênc. Tecnol. Aliment., 31: 49-260, 1997.
- 13. Duran, N.; Esposito, E.; Innocentini-Mei, L.H.; Canhos, V.P.; A new alternative process for Kraft E1 effluent treatment. A combination of photochemical and biological methods. Biodegradation., 5: 3-19, 1994.
- 14. Fonseca, M.J.V.; Said, S.; The pectinase produced by Tubercularia vulgaris in submerged culture using pectin or orange-pulp pellets as inducer. Appl. Microbiol. Biotechnol, 42: 32-35, 1994.
- 15. Fonseca, M.J.V.; Said, S. Sequential production of pectinases by Penicillium frequentans. World J. Microbiol. Biotechnol, 11: 174-177, 1995.
- 16. Galiotou-Panayotou, M.; Rodis, P.; Kapantai, M. Enhanced polygalacturonase production by Aspergillus niger NRRL-364 grown on supplemented citrus pectin. Lett. Apll. Microbiol, 17: 145-148, 1993.
- 17. Galiotou-Panayotou, M.; Kapantai, M.; Kalantzi, O. Growth conditions of Aspergillus sp ATHUM-3482 for polygalacturonase production. Appl. Microbiol. Biotechnol, 47: 425-429, 1997.
- 18. Garzón, C.G. and Hours, R.A. Citrus waste: an alternative substrate for pectinase production in solid-state culture. Biores. Technol, 39: 93-95, 1992.
- 19. Guevara, M.A.; Gonzalez-Jen, M.T.; Estevez, P. Multiple forms of pectic lyases and polygalacturonases from Fusarium oxysporum f. sp redicais lycopersici: Regulation of their synthesis by galacturonic acid. Canadian J. Microbiol., 43: 245-253, 1997.
- 20. Hang, Y.D.; Woodanms, E.E. Production of fungal polygalacturonase from apple pomace. Lebensm. Wiss U. Technol, 27: 194-196, 1994.
- 21. Hours, R.A.; Voget, C.E.; Ertola, R.J. Some factories affecting pectinase production from apple pomace in solid states cultures. Biological wastes., 24: 147-157, 1988.
- 22. Ismail, A.S. Utilization of orange peels for the production of multi-enzyme complexes by some fungal strains. Process Biochem, 1: 645-650, 1996.
- 23. Kobayashi, T.; Hatada, Y.; Higaki, N.; Lusterio, D.D.; Ozawa, T.; Koike, K.; Kawai, S.; Ito, S. Enzymatic properties and deduced amino acid sequence of a high-alkaline pectate lyase from na alkaliphilic Bacillus isolate. Bioch. Biophys. Acta., 1427: 145-154, 1999.
- 24. Leone, G.; Heuvel, J.; Van Den Heuvel, J. Regulation by carbohydrates of the sequential in vitro production of pectic enzymes by Botrytis cinerea Can. J. Bot., 65: 2133-2141, 1987.
- 25. Miller, G.L. Use of dinitrossalicylic acid reagent for determination of reducing sugars. Anal Chem., 31: 426-428, 1959.
- 26. Mitchell, D.A.; Do, D.D.; Greenfield, P.F.; Doelle, H.W. A semi-mechanistic mathematical model for growth of Rhizopus oligosporus in a model solid-state fermentation system. Biotechnol. Bioeng, 38: 353 62, 1991.
- 27. Mitchell, D.A.; Krieger, N.; Stuart, D.M.; Pandey, A. New developments in solid-state fermentation. II. Rotational approaches to the design, operation and scale-up of bioreactors. Process. Biochem., 35: 1211-1225, 2000.
- 28. Mutlu, M.; Srioglu, K.; Demir, N.; Ercan, M.T.; Acar, J. The use of commercial pectinase in fruit juice industry. Part I: viscosimetric determination of enzyme activity. J. Food Engen., 41: 147-150, 1999.
- 29. Naidu, G.G.N.; Panda, T. Production of pectolytic enzymes- a review. Bioprocess Eng, 19: 355-361, 1998.
- 30. Ooijkaas, L.; Weber, F.J.; Buitelaar, R.M.; Tramper, J.; Rinzema, A. Defined media and inert supports: their potential as solid-state fermentation production systems. Trends Biotechnol., 18: 356-360, 2000.
- 31. Oriol, E.; Schettino, B.; Viniegra-Gonzales, G.; Raimbaut, M. Solid-state culture of Aspergillus niger on support. J. Ferment. Technol, 66: 57-62, 1988.
- 32. Oudot, J.; Dupont, J.; Haloui, S.; Roquebert, M.F. Biodegradation potential of hydrocarbon-assimilating tropical fungi. Soil Biol. Biochem, 25: 1167-1173, 1993.
- 33. Pandey, A.; Soccol, C.R.; Nigam, P.; Soccol, V.T. Biotechnological potential of agro-industrial residues. I: sugar cane bagasse. Bioresource Technol, 74: 69-80, 2000.
- 34. Pilnik, W.; Voragen, A.G.J. Pectic enzymes in fruit and vegetable juice manufature. In: Nagodawithama, T.; Reed, G. (eds). Enzymes in Food Processing, Academic Press, New York 1993, p.363-399.
- 35. Reid, I.; Ricard, M. Pectinase in papermaking: solving retention problems in mechanical pulps bleached with hydrogen peroxide Enz. Microbiol. Technol., 26: 115-123, 2000.
- 36. Riou, C.; Freyssinet, G.; Feure, M. Purification and characterization of extracellular pectinolytic enzymes produced by Sclerotinia sclerotiorum. Appl. Env. Microbiol, 58: 578-583, 1992.
- 37. Ryazanova, L.P.; Mikhaleva, N.I.; Solov'eva, I.V.; Boev, A.V.; Okunev, O.N.; Kulaev, I.S. Pectolytic enzymes from Aspergillus heteromorphus Appl. Biochem. Microbiol, 32: 1-6, 1996.
- 38. Soares, M.M.C.N.; Da Silva, R.; Gomes, E. Screening of bacterial strains for pectinolytic activity characterization of the Pgase produced by Bacillus species Rev. Microbiol, 30: 229-303, 1999.
- 39. Soares, M.M.C.N.; Da Silva, R.; Carmona, E.C.; Gomes, E. Pectinolytic enzymes production by Bacillus species and their potential application on juice extraction. World J. Microbiol. Biotechnol., 17: 79-82, 2001.
- 40. Solis-Pereira, S.; Favela-Torres, E.; Viniegra-Gonzales,G.; Gutierrez-Rofas, M. Effects of different carbon source on the synthesis of pectinase by Aspergillus niger in submerged and solid state fermentations. Appl. Microbiol. Biothnol., 39: 36-41, 1993.
- 41. Ueda, S.; Yusaku, F.; Lim, J.Y. Production and some properties of pectic enzymes from Aspergillus oryzae A-3. J. Appl. Biochem, 4: 524-532, 1982.
- 42. Whitaker, J.R. Microbial Enzymes and Biotechnology, In: Fogarty, W.M.; Kelly, C.T. (eds). Elsevier, London, 1990, p.133-176.
Publication Dates
-
Publication in this collection
18 June 2003 -
Date of issue
Dec 2002
History
-
Accepted
05 Dec 2002 -
Reviewed
08 Jan 2002 -
Received
13 Sept 2002